Abstract
In angiosperms, microRNA156 (miR156) acts as an intrinsic, endogenous developmental timer for the age-dependent transition from the juvenile to the adult phase1,2,3. However, the mechanisms modulating the age-dependent expression pattern of miR156 are still poorly understood4. In this Article, we report that circular RNAs (ciMIR156Ds) derived from pri-miR156d negatively regulate miR156 levels in an aging-dependent manner in rice. The ciMIR156D levels increase as plants age, which is inversely correlated with the changes of pri-miR156d and miR156 abundance. Consistent with this observation, ciMIR156Ds deficiency caused by a spontaneous mutation increases pri-miR156d and miR156 levels, resulting in a delayed heading phenotype, whereas ciMIR156Ds overexpression has opposite effects, demonstrating that ciMIR156Ds are negative regulators of miR156. We further show that ciMIR156Ds form R-loops with MIR156D at the region where they derive in an aging-dependent manner, which reduces the occupancy of DNA-dependent RNA polymerase II at that location and hence impedes pri-miR156d elongation. These findings reveal a mechanism for regulating heading date by refining the aging-dependent expression of miR156.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Publicly available datasets used in this study include PlantcircBase v.7.0 (http://ibi.zju.edu.cn/plantcircbase/index.php) and Bedtools v.2.28.0 (https://bedtools.readthedocs.io/en/latest/index.html). DRIP–seq data used for R-loop identification in this study are available via ref. 31 (accession no. GSE111944). Pol II ChIP–seq data used in this study are available via ref. 34. RNA-seq datasets of NIL-sdh and Nip are available via the Sequence Read Archive of the National Center for Biotechnology Information (accession nos. PRJNA1171578 and PRJNA1171724). All other data supporting the findings of this study are available in the paper and its Supplementary Information and Supplementary Data 1–6. Source data are provided with this paper.
References
Wu, G. et al. The sequential action of miR156 and miR172 regulates developmental timing in Arabidopsis. Cell 138, 750–759 (2009).
Wang, J. W., Czech, B. & Weigel, D. miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138, 738–749 (2009).
Wang, J. W. Regulation of flowering time by the miR156-mediated age pathway. J. Exp. Bot. 65, 4723–4730 (2014).
Fouracre, J. P. & Poethig, R. S. The role of small RNAs in vegetative shoot development. Curr. Opin. Plant Biol. 29, 64–72 (2016).
Zeng, Q., Zhao, Z. H. & Zhao, S. Q. Signal pathways of flowering time regulation in plant. Yi Chuan 28, 1031–1036 (2006).
Doi, K. et al. Ehd1, a B-type response regulator in rice, confers short-day promotion of flowering and controls FT-like gene expression independently of Hd1. Genes Dev. 18, 926–936 (2004).
Xue, W. et al. Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nat. Genet. 40, 761–767 (2008).
Freytes, S. N., Canelo, M. & Cerdán, P. D. Regulation of flowering time: when and where? Curr. Opin. Plant Biol. 63, 102049 (2021).
Hyun, Y., Richter, R. & Coupland, G. Competence to flower: age-controlled sensitivity to environmental cues. Plant Physiol. 173, 36–46 (2017).
Guo, C. et al. Repression of miR156 by miR159 regulates the timing of the juvenile-to-adult transition in Arabidopsis. Plant Cell 29, 1293–1304 (2017).
Zhang, H. et al. Genome-wide mapping of the HY5-mediated gene networks in Arabidopsis that involve both transcriptional and post-transcriptional regulation. Plant J. 65, 346–358 (2011).
Xu, L. et al. Regulation of rice tillering by RNA-directed DNA methylation at miniature inverted-repeat transposable elements. Mol. Plant 13, 851–863 (2020).
Cui, Y. et al. The heterochronic gene Oryza sativa LIKE HETEROCHROMATIN PROTEIN 1 modulates miR156b/c/i/e levels. J. Integr. Plant Biol. 62, 1839–1852 (2020).
Kristensen, L. S. et al. The biogenesis, biology and characterization of circular RNAs. Nat. Rev. Genet. 20, 675–691 (2019).
Liu, R., Ma, Y., Guo, T. & Li, G. Identification, biogenesis, function, and mechanism of action of circular RNAs in plants. Plant Commun. 4, 100430 (2023).
Chu, Q. et al. Characteristics of plant circular RNAs. Brief. Bioinform. 21, 135–143 (2020).
Jiao, Y. et al. Regulation of OsSPL14 by OsmiR156 defines ideal plant architecture in rice. Nat. Genet. 42, 541–544 (2010).
Miura, K. et al. OsSPL14 promotes panicle branching and higher grain productivity in rice. Nat. Genet. 42, 545–549 (2010).
Lu, T. et al. Transcriptome-wide investigation of circular RNAs in rice. RNA 21, 2076–2087 (2015).
Ye, C. Y., Chen, L., Liu, C., Zhu, Q. H. & Fan, L. Widespread noncoding circular RNAs in plants. New Phytol. 208, 88–95 (2015).
Zhang, P. et al. PlantCircNet: a database for plant circRNA-miRNA-mRNA regulatory networks. Database 2017, bax089 (2017).
Fan, J. et al. circRNAs are involved in the rice–Magnaporthe oryzae interaction. Plant Physiol. 182, 272–286 (2020).
Xu, X. et al. PlantcircBase 7.0: full-length transcripts and conservation of plant circRNAs. Plant Commun. 3, 100343 (2022).
Tanaka, N. et al. The COP1 ortholog PPS regulates the juvenile-adult and vegetative-reproductive phase changes in rice. Plant Cell 23, 2143–2154 (2011).
Xie, K. et al. Gradual increase of miR156 regulates temporal expression changes of numerous genes during leaf development in rice. Plant Physiol. 158, 1382–1394 (2012).
Yoine, M. et al. The lba1 mutation of UPF1 RNA helicase involved in nonsense-mediated mRNA decay causes pleiotropic phenotypic changes and altered sugar signalling in Arabidopsis. Plant J. 47, 49–62 (2006).
Kurihara, Y. et al. Genome-wide suppression of aberrant mRNA-like noncoding RNAs by NMD in Arabidopsis. Proc. Natl Acad. Sci. USA 106, 2453–2458 (2009).
Chen, L. L. The biogenesis and emerging roles of circular RNAs. Nat. Rev. Mol. Cell Biol. 17, 205–211 (2016).
Zhang, Y. et al. Circular intronic long noncoding RNAs. Mol. Cell 51, 792–806 (2013).
Conn, V. M. et al. A circRNA from SEPALLATA3 regulates splicing of its cognate mRNA through R-loop formation. Nat. Plants 3, 17053 (2017).
Fang, Y. et al. Characterization of functional relationships of R-loops with gene transcription and epigenetic modifications in rice. Genome Res. 29, 1287–1297 (2019).
Xu, W. et al. The R-loop is a common chromatin feature of the Arabidopsis genome. Nat. Plants 3, 704–714 (2017).
Shivji, M. K. K., Renaudin, X., Williams, Ç. H. & Venkitaraman, A. R. BRCA2 regulates transcription elongation by RNA polymerase II to prevent R-loop accumulation. Cell Rep. 22, 1031–1039 (2018).
Xie, L. et al. RiceENCODE: a comprehensive epigenomic database as a rice encyclopedia of DNA elements. Mol. Plant 14, 1604–1606 (2021).
Gonzalo, L. et al. R-loops at microRNA encoding loci promote co-transcriptional processing of pri-miRNAs in plants. Nat. Plants 8, 402–418 (2022).
Liu, Y. et al. Back-spliced RNA from retrotransposon binds to centromere and regulates centromeric chromatin loops in maize. PLoS Biol. 18, e3000582 (2020).
Li, X. et al. Linking circular intronic RNA degradation and function in transcription by RNase H1. Sci. China Life Sci. 64, 1795–1809 (2021).
Huang, R. et al. BLISTER promotes seed maturation and fatty acid biosynthesis by interacting with WRINKLED1 to regulate chromatin dynamics in Arabidopsis. Plant Cell 34, 2242–2265 (2022).
Quinlan, A. R. BEDTools: the Swiss-Army tool for genome feature analysis. Curr. Protoc. Bioinformatics https://doi.org/10.1002/0471250953.bi1112s47 (2014).
Acknowledgements
We thank W. Zhang of Nanjing Agricultural University for providing R-loop results and Q. Song of Nanjing Agricultural University for analysing BSA results. This work was supported by BaGui Scholars Foundation of Guangxi (to Y.L.); National Natural Science Foundation of China (31970603 to Y.S.); National Institute of Health (GM127414 to B.Y.); and National Science Foundation (MCB-1818082 to B.Y.).
Author information
Authors and Affiliations
Contributions
Y.S., Y.Y., S.G., Z. Wang, X.L. and Z. Wei performed the experiments and analysed the data. Y.S., S.G. and H.W. modelled and wrote the paper. C.Z., S.G. and Q.X. carried out data analysis. Y.Q. helped create rice mutants. Y.L. and B.Y. jointly supervised the research and wrote the paper. All authors provided feedback on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
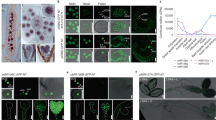
Extended Data Fig. 1 The effect of the sdh allele on heading date.
a, c, The heading date of indicated genotypes under long-days (LDs) and short-days (SDs). The days towards heading of each plant were recorded as the number of days beginning from transplanting seedlings to soil to the appearance of the first panicle. b, d, The tiller numbers of indicated genotypes under LDs. Values in a–d are given as means ± SD. *, P < 0.05; **, P < 0.01 compared with the wild type by single-sided Student’s t-test. e, Phenotypes of 98-day-old Nipponbare (Nip) and NIL-sdh plants grown under LD conditions in paddy fields of Nanning (China) during maturation period.
Extended Data Fig. 2 Positioning cloning of SDH.
a, Identification of genomic regions possibly harboring causative mutations using MutMap. The X-axis represents the chromosome positions. The Y-axis represents SNP index for the mutant. The red arrow indicates initial mutation locus interval. b, The diagram of predicted genes between markers RM12584 and RM12590. The boxes represent exons, while the slim lines and bold lines represent introns and regions between genes, respectively. c, PCR analysis of sdh. Primer positions were indicated in Fig. 1d. WT: Gui649; sdh: SDH/sdh obtained from Gui649 x sdh crossing. d, Association analysis between phenotype segregation and DNA deletion in MIR156D in F2 progenies generated from Gui649 and sdh crossing. The analyzed samples were collected based on WT or delayed heading phenotypes and subject to PCR reactions using primer shown in (Fig. 1d).
Extended Data Fig. 3 Functional verification of SDH.
a, The relative expression levels of SPL genes in the flag leaves of Nip and NIL-sdh detected by RT-qPCR. b, The relative expression levels of pri-miR156d in the flag leaves of various genotypes detected by RT-qPCR. c, Heading dates of various genotypes under LDs. The days to heading of each plant were recorded as the number of days beginning from transplanting seedlings to soil to the appearance of the first panicle. d, The tiller numbers of various genotypic plants under LDs. Data in (c, d) are shown as means ± SD, *, P < 0.05 **, P < 0.01 by single-sided Student’s t-test. e, The diagram of structure of STTM, including the promoter of MIR156D, spacer linker between STTMs, and designed STTM156 in which a weak imperfect ‘stem-loop’ was included (marked by red). f, The relative expression levels of pri-miR156d in the flag leaves of various genotypes detected by RT-qPCR. Expression of Ubiquitin was used as an internal control, and the expression level of each gene in Nip was set as 1 in (a, b, f). Data are shown as means ± SD (n = 3), *, P < 0.05; **, P < 0.01 by single-sided Student’s t-test.
Extended Data Fig. 4 Identification and over-expression of ciMIR156Ds.
a, Diagram showing the positions of ciMIR156Ds at the MIR156D locus and their validation with divergent primers followed by Sanger sequencing. Dashed lines indicate in electropherogram represent reads matching the back-splicing junction. The typical locations of divergent primers used for circRNA-specific amplification from exon 3 by RT–PCR are shown as grey arrows. b, Schematic diagram of constructs used to over-express ciMIR156D (OE-miR156dcirc) harboring a region from the 1st to the 4th exon of MIR156D. The red lines indicate that miR156 and miR156* were mutated in the construct. Mutated miR156d and miR156* sequences were shown below. c, ciMIR156D transcript levels in 11 independent OE-miR156dcirc transgenic plants detected by RT-PCR. Nip was used as a positive control. Line #1, #4 and #6 were selected for subsequent experiments as OE-miR156dcirc rice.
Extended Data Fig. 5 The effects of circRNAs on expressions of miR156 and its target genes.
a, The relative expression levels of pri-miR156d in the flag leaves of various genotypes detected by RT-qPCR using primers located at 5th exon. b, miR156 and miR172 were detected in flag leaves of various genotypes using Northern blot. U6 was used as the loading control. c, The relative expression levels of SPL genes in the flag leaves of various genotypes detected by RT-qPCR. Ubiquitin was used as an internal control, and the expression levels of each gene in Nip were set as 1 in (a, c). Data are shown as means ± SD (n = 3), *, P < 0.05; **, P < 0.01; ns, not significant by single-sided Student’s t-test.
Extended Data Fig. 6 The effects of linear RNAs of OE-156dw/t3rd,intron on miR156.
a, Schematic diagram of constructs used to over-express truncated MIR156D harboring a region from the 1st to the 4th exon of MIR156D without 3rd intron (OE-156dw/t 3rd intron). The gene structure is represented by exons (boxes) and introns (lines), and the green box in the 1st exon indicates the pre-miR156d coding region. The red lines in 1st exon indicate that miR156 and miR156* were mutated in the construct, and mutated miR156d and miR156* sequences were shown in Supplementary Fig. 4b. The red dashed line indicates the deleted 3rd intron in the construct. b, The relative transgene transcript levels of OE-156dw/t 3rd intron and OE-156dcirn in the flag leaves of various genotypes detected by RT-qPCR using primers located at the 3rd exon. The transcript levels of pri-mR156d in Nip were set as 1. The line #10 was used for the subsequent data. c, The relative expression levels of ciMIR156Ds in the flag leaves of Nip and OE-156dw/t 3rd intron #10 detected by RT-qPCR. d, The relative expression levels of pri-miR156d in the flag leaves detected by RT-qPCR using primers located at 5th exon. e, The relative expression levels of miR156 in the flag leaves among NIP and OE-156dw/t 3rd intron #10 detected by stem loop RT-qPCR. f,g, The heading date of indicated genotypes under long days (LDs). Ubiquitin (b-d) or U6 (e) was used as an internal control. Data are shown as means ± SD (n = 3), *, P < 0.05; **, P < 0.01; ns, not significant by single-sided Student’s t-test.
Extended Data Fig. 7 The pri-miR156d of Nip and NIL-sdh were not affected by NMD.
a, The representative diagram of Os07g0495900 gene structure. Exons and introns are indicated by boxes and lines respectively. The os07g0495900 mutant with a 4 bp deletion in the 5th exon was generated by CRISPR-Cas9 system. b, c, The relative levels of pri-miR156d (b) and miR156 (c) in Nip, os07g0495900, NIL-sdh (sdh) and os07g0495900 sdh were detected by RT-PCR and stem-loop RT-qPCR, respectively. Ubiquitin (UBQ) and U6 were used as the control in b and c, respectively. Data are shown as means ± SD (n = 3), *, P < 0.05; ns, not significant by single-sided Student’s t-test.
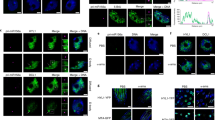
Extended Data Fig. 8 The cellular localization of circRNAs and epigenetic modifications of MIR156D region.
a, RT-qPCR analysis of circRNAs in the nuclear and cytosolic fractions in flag leaves of Nip. The nuclear abundance is set as 1. Data is shown as means ± SD (n = 3). b,c, The levels of H3K27ac (b) and H3K27me3 (c) at the MIR156D locus in wild-type and NIL-sdh. Four pairs of primers (used in Fig. 3a) around the deletion region were used to check the enrichment of H3K27ac or H3K27me3 modifications. The flag leaves were collected for ChIP-qPCR assays. The levels of H3K27ac and H3K27me3 of Actin gene in wild-type was set to 1.0. Three biological replicates were performed. Data is shown as means ± SD (n = 3). A single-sided Student’s t-test was used to generate the P values, *, P < 0.05; **, P < 0.01; ns, not significant.
Extended Data Fig. 9 Transcript levels of individual miR156 members in Nip and NIL-sdh.
The transcript levels of individual miR156 members in RNA-seq assay from 4th leaves of Nip and NIL-sdh were shown as TPM (Transcripts Per Kilobase of exon model per Million mapped reads) in heatmap, with three biological replicates for each sample.
Extended Data Fig. 10 The circRNA-R-loop may exist in other MIR156 loci of Arabidopsis and rice.
a,b, Diagram showing the circRNAs derived from MIR156F locus of Arabidopsis (a) and MIR156J locus of rice (b) overlap with the R-loop.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1.
Supplementary Data 1 (download XLSX )
Summary of identified circRNAs in OsMIR156D locus.
Supplementary Data 2 (download XLSX )
The circRNAs derived from MIR156 locus of Arabidopsis and rice overlap with the R-loop.
Supplementary Data 3 (download XLSX )
The identified circRNAs located within a 2-kb range upstream and downstream of miRNAs in rice.
Supplementary Data 4 (download XLSX )
The identified circRNAs originated from miRNA loci of rice overlaps with R-loop.
Supplementary Data 5 (download XLSX )
The sequences of primers used in this study.
Supplementary Data 6 (download XLSX )
Source data for Supplementary Table 1.
Source data
Source Data Fig. 1 (download XLSX )
Unprocessed gels.
Source Data Fig. 2 (download XLSX )
Unprocessed gels and statistical source data.
Source Data Fig. 3 (download XLSX )
Unprocessed dot blots and statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Unprocessed gels.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Unprocessed gels.
Source Data Extended Data Fig. 5 (download XLSX )
Unprocessed gels and statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Unprocessed gels and statistical source data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Su, Y., Yi, Y., Ge, S. et al. Circular RNAs derived from MIR156D promote rice heading by repressing transcription elongation of pri-miR156d through R-loop formation. Nat. Plants 11, 709–716 (2025). https://doi.org/10.1038/s41477-025-01961-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41477-025-01961-7
This article is cited by
-
Transcriptome and small RNA sequencings reveal the response of tobacco to aphid infestation
BMC Genomics (2025)
-
Dual roles of DEAD-box RNA helicase Brr2 in genome stability regulation
Nature Communications (2025)