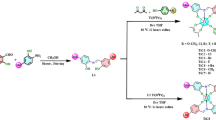
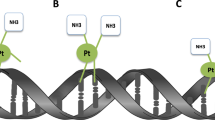
Abstract
Radiotherapy is used in more than half of cancer patients, yet most radiosensitizers increase reactive oxygen species (ROS) to enhance cytotoxicity in treated cells. This approach has limited use in hypoxic tumours and may cause oxidative injury to healthy tissues. We developed a platinum(II) azido complex (Complex 1) that releases platinonitrene upon X-ray exposure. Platinonitrene reacts with nucleophilic sites on DNA bases, forming covalent adducts that disrupt DNA integrity and cause double-strand breaks, leading to tumour cell death through a mechanism distinct from classical platinum coordination. Computational modelling elucidated this pathway and supported its role in radiosensitization. Complex 1 was synthesized by sequential ligand exchange of potassium tetrachloroplatinate with cyclohexanediamine, silver nitrate and sodium azide. In murine models, complex 1 showed negligible toxicity to major organs and normal immune cells while selectively reducing regulatory T-cell infiltration in tumours. Combined with low-dose radiotherapy and programmed cell death protein 1 blockade, it achieved complete regression of bilateral tumours in 40% of mice, demonstrating a strong abscopal effect. This work establishes metallonitrene-based, ROS-independent radiosensitization for precision radiotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All relevant data supporting the results in this study are available within the paper. The raw and analysed datasets generated during the study are available for academic purposes from the corresponding authors upon request. RNA sequencing data that support the findings of this study are deposited and made publicly available in the NCBI Gene Expression Omnibus repository under accession number GSE314315. Source data are provided with this paper.
References
Petroni, G., Cantley, L. C., Santambrogio, L., Formenti, S. C. & Galluzzi, L. Radiotherapy as a tool to elicit clinically actionable signalling pathways in cancer. Nat. Rev. Clin. Oncol. 19, 114–131 (2022).
Chabanon, R. M. et al. Targeting the DNA damage response in immuno-oncology: developments and opportunities. Nat. Rev. Cancer 21, 701–717 (2021).
Schaue, D. & McBride, W. H. Opportunities and challenges of radiotherapy for treating cancer. Nat. Rev. Clin. Oncol. 12, 527–540 (2015).
Weichselbaum, R. R., Liang, H., Deng, L. & Fu, Y.-X. Radiotherapy and immunotherapy: a beneficial liaison? Nat. Rev. Clin. Oncol. 14, 365–379 (2017).
Keener, A. B. Making radiation oncology more personal. Nature 585, S10 (2020).
Nishiga, Y. et al. Radiotherapy in combination with CD47 blockade elicits a macrophage-mediated abscopal effect. Nat. Cancer 3, 1351–1366 (2022).
Ngwa, W. et al. Using immunotherapy to boost the abscopal effect. Nat. Rev. Cancer 18, 313–322 (2018).
Grassberger, C., Ellsworth, S. G., Wilks, M. Q., Keane, F. K. & Loeffler, J. S. Assessing the interactions between radiotherapy and antitumour immunity. Nat. Rev. Clin. Oncol. 16, 729–745 (2019).
Hiniker, S. M. & C. D., Knox SJ. Abscopal effect in a patient with melanoma. N. Engl. J. Med. 366, 2035–2036 (2012).
De Ruysscher, D. et al. Radiotherapy toxicity. Nat. Rev. Dis. Primers 5, 13 (2019).
Price, J. M., Prabhakaran, A. & West, C. M. L. Predicting tumour radiosensitivity to deliver precision radiotherapy. Nat. Rev. Clin. Oncol. 20, 83–98 (2023).
Wang, H., Mu, X., He, H. & Zhang, X.-D. Cancer radiosensitizers. Trends Pharmacol. Sci. 39, 24–48 (2018).
Gill, M. R. & Vallis, K. A. Transition metal compounds as cancer radiosensitizers. Chem. Soc. Rev. 48, 540–557 (2019).
Sun, J. et al. A platinum(II) metallonitrene with a triplet ground state. Nat. Chem. 12, 1054–1059 (2020).
Domenianni, L. I. et al. Photoinduced metallonitrene formation by N2 elimination from azide diradical ligands. Angew. Chem. Int. Ed. 62, e202309618 (2023).
Wentrup, C. Carbenes and nitrenes: recent developments in fundamental chemistry. Angew. Chem. Int. Ed. 57, 11508–11521 (2018).
Vreeken, V. et al. C–H activation of benzene by a photoactivated NiII(azide): formation of a transient nickel nitrido complex. Angew. Chem. Int. Ed. 54, 7055–7059 (2015).
Henning, H., Hofbauer, K., Handke, K. & Stich, R. Unusual reaction pathways in the photolysis of diazido(phosphane)nickel(II) complexes: nitrenes as intermediates in the formation of nickel(0) complexes. Angew. Chem. Int. Ed. 36, 408–410 (1997).
Carsch, K. M. et al. Synthesis of a copper-supported triplet nitrene complex pertinent to copper-catalyzed amination. Science 365, 1138–1143 (2019).
Jung, H. et al. Mechanistic snapshots of rhodium-catalyzed acylnitrene transfer reactions. Science 381, 525–532 (2023).
Baek, Y. et al. C–H amination mediated by cobalt organoazide adducts and the corresponding cobalt nitrenoid intermediates. J. Am. Chem. Soc. 142, 11232–11243 (2020).
Wang, H. et al. Nitrene-mediated intermolecular N–N coupling for efficient synthesis of hydrazides. Nat. Chem. 13, 378–385 (2021).
Thornton, A. R., Martin, V. I. & Blakey, S. B. Pi-nucleophile traps for metallonitrene/alkyne cascade reactions: a versatile process for the synthesis of α-aminocyclopropanes and β-aminostyrenes. J. Am. Chem. Soc. 131, 2434–2435 (2009).
Geng, J. et al. Switching on prodrugs using radiotherapy. Nat. Chem. 13, 805–810 (2021).
Guillemard, L., Kaplaneris, N., Ackermann, L. & Johansson, M. J. Late-stage C–H functionalization offers new opportunities in drug discovery. Nat. Rev. Chem. 5, 522–545 (2021).
Cosio, M. N. & Powers, D. C. Prospects and challenges for nitrogen-atom transfer catalysis. Nat. Rev. Chem. 7, 424–438 (2023).
Herrera, F. G. et al. Low-dose radiotherapy reverses tumor immune desertification and resistance to immunotherapy. Cancer Discov. 12, 108–133 (2022).
Scott, J. G. et al. A genome-based model for adjusting radiotherapy dose (GARD): a retrospective, cohort-based study. Lancet Oncol. 18, 202–211 (2017).
Cheung, E. C. & Vousden, K. H. The role of ROS in tumour development and progression. Nat. Rev. Cancer 22, 280–297 (2022).
Kornepati, A. V. R., Rogers, C. M., Sung, P. & Curiel, T. J. The complementarity of DDR, nucleic acids and anti-tumour immunity. Nature 619, 475–486 (2023).
Vemparala, S., Domene, C. & Klein, M. L. Interaction of anesthetics with open and closed conformations of a potassium channel studied via molecular dynamics and normal mode analysis. Biophys. J. 94, 4260–4269 (2008).
Parikh, A. R. et al. Radiation therapy enhances immunotherapy response in microsatellite stable colorectal and pancreatic adenocarcinoma in a phase II trial. Nat. Cancer 2, 1124–1135 (2021).
Min, Y. et al. Antigen-capturing nanoparticles improve the abscopal effect and cancer immunotherapy. Nat. Nanotechnol. 12, 877–882 (2017).
Hoff, P. M. Oxaliplatin-related neurotoxicity: is chelation the solution? Nat. Clin. Pract. Oncol. 1, 78–79 (2004).
Argyriou, A. A., Bruna, J., Genazzani, A. A. & Cavaletti, G. Chemotherapy-induced peripheral neurotoxicity: management informed by pharmacogenetics. Nat. Rev. Neurol. 13, 492–504 (2017).
Kim, H. P. et al. The clinical spectrum and diagnosis of oxaliplatin liver injury in the era of nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 19, 2199–2201 (2021).
Wilson, W. R. & Hay, M. P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 11, 393–410 (2011).
Singleton, D. C., Macann, A. & Wilson, W. R. Therapeutic targeting of the hypoxic tumour microenvironment. Nat. Rev. Clin. Oncol. 18, 751–772 (2021).
Van Der Spoel, D. et al. GROMACS: fast, flexible, and free. J. Comput. Chem. 26, 1701–1718 (2005).
Humphrey, W., Dalke, A. & Schulten, K. V. M. D. Visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Salomon-Ferrer, R., Case, D. A. & Walker, R. C. An overview of the Amber biomolecular simulation package. WIREs Comput. Mol. Sci. 3, 198–210 (2013).
Pérez, A. et al. Refinement of the AMBER force field for nucleic acids: improving the description of α/γ conformers. Biophys. J. 92, 3817–3829 (2007).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Frisch, M. J. et al. Gaussian 09, Revision D.01 (Gaussian, Inc., 2009).
Bayly, C. I., Cieplak, P., Cornell, W. & Kollman, P. A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J. Phys. Chem. 97, 10269–10280 (1993).
Burger, S. K. et al. Automated parametrization of AMBER force field terms from vibrational analysis with a focus on functionalizing dinuclear zinc(II) scaffolds. J. Chem. Theory Comput. 8, 554–562 (2012).
Pereira, A. F., Prandi, I. G. & Ramalho, T. C. Parameterization and validation of a new force field for Pt(II) complexes of 2-(4′-amino-2′-hydroxyphenyl)benzothiazole. Int. J. Quantum Chem. 121, e26525 (2021).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Ding, Z. et al. Radiotherapy reduces N-oxides for prodrug activation in tumors. J. Am. Chem. Soc. 144, 9458–9464 (2022).
Chen, J. et al. Accurate and early metastases diagnosis in live animals with multimodal X-ray and optical imaging. Int. J. Radiat. Oncol. Biol. Phys. 115, 511–517 (2023).
Acknowledgements
This work was supported by: the National Natural Science Foundation of China (32271433, 32471437 and 31971299); 2025IHM01180, Start-Up grants KY2060000124 and KJ2060190030 from the University of Science and Technology of China and Fundamental Research Funds for the Central Universities (YD9100002031, WK2060190101) to Y.M. ; the National Natural Science Foundation of China (22422306) and National Key R&D Program of China (2024YFA1509203) to S.X.; the Natural Science Foundation of Anhui Province (2208085J48) and Hefei Comprehensive National Science Center Institute of Health and Medicine Project (DJK-LX-2022006, 2023IHM01044) to B.L.; the National Key R&D Program of China (2022YFA1303200) to K.Q, and the National Natural Science Foundation of China grants (T2125012 and 92574202) and Strategic Priority Research Program of Chinese Academy of Sciences (XDB0940301) to K.Q.; funding from the Global STEM Professorship, start-up fund (Grant No. 2023-Xing, HKPU), and Projects of RISA PolyU (UGC) (1-CDN9) to B.X. The work described in this paper was also partially supported by a grant from the NSFC/RGC Joint Research Scheme sponsored by the Research Grants Council of Hong Kong and the National Natural Science Foundation of China (Project No. N_PolyU521/25) to B.X.; and by the Fundamental Research Funds for the Central Universities (WK2060000099) to Y.X. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.M. conceived and designed the experiments with G.C. G.C. and X.L. performed the efficacy study. G.C. also performed the mechanistic study. Y. Huang and S.X. performed the calculations. C.Z., G.C., C.G. and H.Z. conducted radiotherapy for in vitro and in vivo experiments. G.C., H.F., Q. Zhang and B.L. designed and performed the flow cytometry analysis of Treg cells. G.C., K.L. and K.Q. performed the analysis of RNA sequencing. K.W., Qilong Zhu, R.D. and Y. He helped to perform the experiments at cellular level. Qiuhong Zhu and W.Z. helped to establish the glioblastoma model. G.C. and Y.M. wrote the manuscript. All authors analysed and discussed the data, revised the manuscript, and approved the submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characteristic NLMOs resulting from NBO analysis support the platinonitrene description of complex 2 and 3.
a, Structural representation of 1. b, N2 releases from 1 under X-ray irradiation, and produces metallonitrene compound 2 and 3. c, Scan of the Nα-Nβ bond length for singlet and triplet states of 1 to illustrate the interplay of singlet and triplet states during the loss of N2 from the molecule of 1. The triplet state of complex 2 is strongly stabilized by 12.7 kcal/mol compared with its singlet state. The calculations were performed at the level of PBE0-D/def2-TZVPP, and the step size during the scan is set to 0.01 Å. d-g, Characteristic NLMOs resulting from an NBO analysis. d-e, Pt-N σ-bond, singly occupied py, pz orbitals and s-type lone pair localized at N atom (d), and Pt-centred and doubly occupied d orbitals (e) of 2, which has only one nitrene group. f-g, Pt-N σ-bond, singly occupied py, pz orbitals and s-type lone pair localized at N atom (f), and Pt-centred and doubly occupied d orbitals (g) of 3, which has two nitrene groups. d to g show that the covalent Pt-N σ-bonds polarized towards nitrogen, a s-type lone pair and two singly occupied p-type NLMOs on the nitrene N atom as well as four essentially nonbonding Pt d orbitals which shared only insignificant N contributions (Pt-N Mayer bond order: 1.15 for 2 and 1.06 for 3).
Extended Data Fig. 2 Complex 1 sensitized RT effectively inhibits the growth and metastasis of 4T1 tumours.
a, Schemes of 4T1 tumour inoculation and therapies. b, Tumour growth curves of each mouse. c, Average tumour growth curves showed 1 sensitized RT effectively inhibited the growth of 4T1 tumours (n = 9). Tumour growth with the passage of time was compared by two-way ANOVA (Tukey’s test). d, Survival curves showed 1 sensitized RT prolonged the survival time of mice (n = 9). Differences in survival were determined for each group by the Kaplan-Meier method, and the overall P value was calculated by the log-rank test. e, Representative lung pathology sections of mice showed that 1 sensitized RT effectively inhibited lung metastasis of 4T1 tumours. Black arrow, pulmonary nodules. f, Lung metastasis count of mice (n = 3). One-way ANOVA multiple comparisons (Tukey’s multiple comparisons test) were used when more than two groups were compared. All the values in the present study are presented as the mean ± s.d., and all replicates are biological independent samples.
Extended Data Fig. 3 cis-Pt(NH3)2(N3)2 sensitized RT effectively inhibits the growth of established CT26 tumours.
a, Schemes of CT26 tumour inoculation and therapies. b, Tumour growth curves of each mouse. c, Average tumour growth curves showed cis-Pt(NH3)2(N3)2 sensitized RT effectively inhibited the growth of established CT26 tumours (n = 6). Tumour growth with the passage of time was compared by two-way ANOVA (Tukey’s test). d, Survival curves showed cis-Pt(NH3)2(N3)2 sensitized RT prolonged the survival time of mice (n = 6). Differences in survival were determined for each group by the Kaplan–Meier method, and the overall P value was calculated by the log-rank test. All the values in the present study are presented as the mean ± s.d., and all replicates are biological independent samples.
Extended Data Fig. 4 Complex 1 sensitized RT enhances abscopal effect on syngeneic CT26 tumour model.
a, Schemes of primary and secondary tumour inoculation and therapies. b, Primary and secondary tumour growth curves of each mouse showed 1 in combination with RT and αPD-1 suppress growth of bilateral tumours. c, Average primary tumour growth curves of each group (n = 8). d, Average secondary tumour growth curves of each group (n = 8). Tumour growth with the passage of time was compared by two-way ANOVA (Tukey’s test). e, Survival curves showed 1 in combination with RT and αPD-1 prolonged survival time of mice in bilateral tumour model (n = 8). Differences in survival were determined for each group by the Kaplan–Meier method, and the overall P value was calculated by the log-rank test. f, Body weight change curve of mice (n = 8). All the values in the present study are presented as the mean ± s.d., and all replicates are biological independent samples.
Extended Data Fig. 5 Flow analysis of 1 sensitized RT enhancing immunotherapy and abscopal effect.
a, Quantification of DCs maturation in TDLNs by flow cytometry indicated 1 sensitized RT enhancing abscopal effect (Blank and RT + αPD-1, n = 4; 1 + RT + αPD-1, n = 3). b, Corresponding representative flow plots showed DCs in TDLNs. c, Image of representative primary and secondary tumours. d, Representative flow plots of primary and secondary tumour infiltrating CD45+CD3+, CD8+and Treg (CD4+Foxp3+) cells. One-way ANOVA multiple comparisons (Tukey’s multiple comparisons test) were used when more than two groups were compared. All the values in the present study are presented as the mean ± s.d., and all replicates are biological independent samples.
Extended Data Fig. 6 Platinonitrene-based radiosensitizer enhances abscopal effect that is superior to that of oxaliplatin and has reliable security.
a-b, Average primary and secondary tumour growth curves showed 1 in combination with low dose of RT and αPD-1 suppress growth of bilateral tumours (n = 10). Tumour growth with the passage of time was compared by two-way ANOVA (Tukey’s test). c, Representative TUNEL (TdT-mediated dUTP nick end labelling) and H&E (Haematoxylin and Eosin staining) section of kidneys showed 1 in combination with low dose of RT and αPD-1 would not cause the damage of kidney. d-h, Analysis of BUN, CRE, TBil (total bilirubin), ALT (alanine transaminase) and AST (aspartate aminotransferase) in serum showed 1 held a better biosecurity than oxaliplatin in combination with immunotherapy (n = 10). i, Representative TUNEL and H&E section of livers showed 1 in combination with low dose of RT and αPD-1 would not cause the damage of livers. One-way ANOVA was used for multiple comparisons (Tukey’s multiple comparisons test) when more than two groups were compared. All the values in the present study are presented as the mean ± s.d., and all replicates are biological independent samples.
Extended Data Fig. 7 Complex 1 sensitized RT effectively inhibits the growth of orthotopic glioblastoma.
a, Schemes of orthotopic glioblastoma inoculation and therapies. b, Bioluminescence imaging suggested complex 1 sensitized RT effectively inhibited the growth of orthotopic glioblastoma (n = 8). c, Changes of bioluminescence counts of brain tumour area of each mouse (n = 8). d, Body weight change curve of each mouse (n = 8). e, Average bioluminescence counts change curves showed 1 sensitized RT effectively inhibited the growth of orthotopic glioblastoma (n = 8). f, Average body weight change curve suggested mice in 1 + RT group had the slowest weight loss (n = 8). g, Survival curves showed 1 sensitized RT significantly prolonged survival time of mice in orthotopic glioblastoma model (n = 8). Average bioluminescence counts change with the passage of time was compared by two-way ANOVA (Tukey’s test). Differences in survival were determined for each group by the Kaplan–Meier method, and the overall P value was calculated by the log-rank test. f, Body weight change curve of mice (n = 8). All the values in the present study are presented as the mean ± s.d., and all replicates are biological independent samples.
Supplementary information
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, G., Li, X., Huang, Y. et al. X-ray activated platinum complex induces DNA damage and enhances cancer immunotherapy through abscopal effect. Nat. Biomed. Eng (2026). https://doi.org/10.1038/s41551-026-01612-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41551-026-01612-y