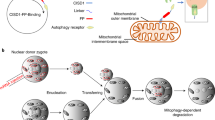
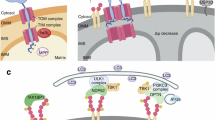
Abstract
The quality of mitochondria inherited from the oocyte determines embryonic viability, lifelong metabolic health of the progeny and lineage endurance. High levels of endogenous reactive oxygen species and exogenous toxicants pose threats to mitochondrial DNA (mtDNA) in fully developed oocytes. Deleterious mtDNA is commonly detected in mature oocytes, but is absent in embryos, suggesting the existence of a cryptic purifying selection mechanism. Here, we discover that in Caenorhabditis elegans, the onset of oocyte-to-zygote transition developmentally triggers a rapid mitophagy event. We show that mitophagy at oocyte-to-zygote transition (MOZT) requires mitochondrial fragmentation, the macroautophagy pathway and the mitophagy receptor FUNDC1, but not the prevalent mitophagy factors PINK1 and BNIP3. MOZT reduces the transmission of deleterious mtDNA and as a result, protects embryonic survival. Impaired MOZT drives the increased accumulation of mtDNA mutations across generations, leading to the extinction of descendant populations. Thus, MOZT represents a strategy that preserves mitochondrial health during the mother-to-offspring transmission and safeguards lineage continuity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data supporting the findings of this study are available in the main text, extended data figures and supplementary materials. Uncropped versions of confocal images supporting main figures are displayed in Supplementary Fig. 6. C. elegans strains used in this study are listed in Supplementary Table 1. Oligonucleotides used in the study are listed in Supplementary Table 2. All stable reagents generated in this study are available from the lead contact without restriction, upon request. Requests for further information and resources should be directed to and will be fulfilled by the lead contact, D.R.S. (david.sherwood@duke.edu). Source data are provided with this paper.
Code availability
No custom code was developed or used in this study.
References
Johnston, I. G. & Williams, B. P. Evolutionary inference across eukaryotes identifies specific pressures favoring mitochondrial gene retention. Cell Syst. 2, 101–111 (2016).
Thouas, G. A., Trounson, A. O., Wolvetang, E. J. & Jones, G. M. Mitochondrial dysfunction in mouse oocytes results in preimplantation embryo arrest in vitro. Biol. Reprod. 71, 1936–1942 (2004).
Taylor, R. W. & Turnbull, D. M. Mitochondrial DNA mutations in human disease. Nat. Rev. Genet. 6, 389–402 (2005).
Tesarik, J. & Mendoza-Tesarik, R. Mitochondria in human fertility and infertility. Int. J. Mol. Sci. 24, 8950 (2023).
Andreas, E., Reid, M., Zhang, W. & Moley, K. H. The effect of maternal high-fat/high-sugar diet on offspring oocytes and early embryo development. Mol. Hum. Reprod. 25, 717–728 (2019).
Malott, K. F. & Luderer, U. Toxicant effects on mammalian oocyte mitochondria. Biol. Reprod. 104, 784–793 (2021).
Otten, A. B. C. et al. Replication errors made during oogenesis lead to detectable de novo mtDNA mutations in zebrafish oocytes with a low mtDNA copy number. Genetics 204, 1423–1431 (2016).
Denver, D. R., Morris, K., Lynch, M., Vassilieva, L. L. & Thomas, W. K. High direct estimate of the mutation rate in the mitochondrial genome of Caenorhabditis elegans. Science 289, 2342–2344 (2000).
Fu, Y., Tigano, M. & Sfeir, A. Safeguarding mitochondrial genomes in higher eukaryotes. Nat. Struct. Mol. Biol. 27, 687–695 (2020).
Howe, D. K. & Denver, D. R. Muller’s Ratchet and compensatory mutation in Caenorhabditis briggsae mitochondrial genome evolution. BMC Evol. Biol. 8, 62 (2008).
Muller, H. J. The relation of recombination to mutational advance. Mutation Res. 1, 2–9 (1964).
Guo, Y. et al. Very low-level heteroplasmy mtDNA variations are inherited in humans. J. Genet. Genom. 40, 607–615 (2013).
Schwartz, A. Z. A., Tsyba, N., Abdu, Y., Patel, M. R. & Nance, J. Independent regulation of mitochondrial DNA quantity and quality in Caenorhabditis elegans primordial germ cells. eLife 11, e80396 (2022).
Lieber, T., Jeedigunta, S. P., Palozzi, J. M., Lehmann, R. & Hurd, T. R. Mitochondrial fragmentation drives selective removal of deleterious mtDNA in the germline. Nature 570, 380–384 (2019).
Palozzi, J. M. et al. Mitochondrial DNA quality control in the female germline requires a unique programmed mitophagy. Cell Metab. 34, 1809–1823.e1806 (2022).
Zhang, Y. et al. PINK1 inhibits local protein synthesis to limit transmission of deleterious mitochondrial DNA mutations. Mol. Cell 73, 1127–1137.e1125 (2019).
Hill, J. H., Chen, Z. & Xu, H. Selective propagation of functional mitochondrial DNA during oogenesis restricts the transmission of a deleterious mitochondrial variant. Nat. Genet. 46, 389–392 (2014).
Flowers, S. et al. Regulation of defective mitochondrial DNA accumulation and transmission in C. elegans by the programmed cell death and aging pathways. eLife 12, e79725 (2023).
Wai, T. et al. The role of mitochondrial DNA copy number in mammalian fertility. Biol. Reprod. 83, 52–62 (2010).
Charmpilas, N. & Tavernarakis, N. Mitochondrial maturation drives germline stem cell differentiation in Caenorhabditis elegans. Cell Death Diff. 27, 601–617 (2020).
Lee, S. K. et al. The association of mitochondrial potential and copy number with pig oocyte maturation and developmental potential. J. Reprod. Dev. 60, 128–135 (2014).
Ge, H. et al. The importance of mitochondrial metabolic activity and mitochondrial DNA replication during oocyte maturation in vitro on oocyte quality and subsequent embryo developmental competence. Mol. Reprod. Dev. 79, 392–401 (2012).
Braeckman, B. P., Smolders, A., Back, P. & De Henau, S. In vivo detection of reactive oxygen species and redox status in Caenorhabditis elegans. Antioxid. Redox Signal. 25, 577–592 (2016).
De Fanti, S. et al. Intra-individual purifying selection on mitochondrial DNA variants during human oogenesis. Hum. Reprod. 32, 1100–1107 (2017).
Krakauer, D. C. & Mira, A. Mitochondria and germ-cell death. Nature 400, 125–126 (1999).
Perez, G. I., Trbovich, A. M., Gosden, R. G. & Tilly, J. L. Mitochondria and the death of oocytes. Nature 403, 500–501 (2000).
Chan, C. C. W. et al. Mitochondrial DNA content and 4977 bp deletion in unfertilized oocytes. Mol. Hum. Reprod. 11, 843–846 (2006).
Gibson, T. C., Kubisch, H. M. & Brenner, C. A. Mitochondrial DNA deletions in rhesus macaque oocytes and embryos. Mol. Hum. Reprod. 11, 785–789 (2005).
Ma, H. et al. Deleterious mtDNA mutations are common in mature oocytes. Biol. Reprod. 102, 607–619 (2019).
Brenner, C. A. et al. Mitochondrial DNA deletion in human oocytes and embryos. Mol. Hum. Reprod. 4, 887–892 (1998).
Monnot, S. et al. Mutation dependance of the mitochondrial DNA copy number in the first stages of human embryogenesis. Hum. Mol. Genet. 22, 1867–1872 (2013).
Barritt, J. A., Brenner, C. A., Cohen, J. & Matt, D. W. Mitochondrial DNA rearrangements in human oocytes and embryos. Mol. Hum. Reprod. 5, 927–933 (1999).
Kremer, L. S. et al. A role for BCL2L13 and autophagy in germline purifying selection of mtDNA. PLoS Genet. 19, e1010573 (2023).
Gitschlag, B. L., Tate, A. T. & Patel, M. R. Nutrient status shapes selfish mitochondrial genome dynamics across different levels of selection. eLife 9, e56686 (2020).
Liu, L. et al. Mitochondrial outer-membrane protein FUNDC1 mediates hypoxia-induced mitophagy in mammalian cells. Nat. Cell Biol. 14, 177–185 (2012).
McCarter, J., Bartlett, B., Dang, T. & Schedl, T. On the control of oocyte meiotic maturation and ovulation in Caenorhabditis elegans. Dev. Biol. 205, 111–128 (1999).
Stoeckius, M. et al. Global characterization of the oocyte-to-embryo transition in Caenorhabditis elegans uncovers a novel mRNA clearance mechanism. EMBO J. 33, 1751–1766 (2014).
Ahringer, J., Rosenquist, T. A., Lawson, D. N. & Kimble, J. The Caenorhabditis elegans sex determining gene fem-3 is regulated post-transcriptionally. EMBO J. 11, 2303–2310 (1992).
Bohnert, K. A. & Kenyon, C. A lysosomal switch triggers proteostasis renewal in the immortal C. elegans germ lineage. Nature 551, 629–633 (2017).
Yamamoto, H., Zhang, S. & Mizushima, N. Autophagy genes in biology and disease. Nat. Rev. Genet. 24, 382–400 (2023).
Tzur, Y. B. et al. Spatiotemporal gene expression analysis of the Caenorhabditis elegans germline uncovers a syncytial expression switch. Genetics 210, 587–605 (2018).
Shcherbo, D. et al. Far-red fluorescent tags for protein imaging in living tissues. Biochem. J. 418, 567–574 (2009).
Shang, A., Shao, S., Zhao, L. & Liu, B. Far-red fluorescent proteins: tools for advancing in vivo imaging. Biosensors 14, 359 (2024).
Burman, J. L. et al. Mitochondrial fission facilitates the selective mitophagy of protein aggregates. J. Cell Biol. 216, 3231–3247 (2017).
Mitra, K. & Lippincott-Schwartz, J. Analysis of mitochondrial dynamics and functions using imaging approaches. Curr. Protoc. Cell Biol. https://doi.org/10.1002/0471143030.cb0425s46 (2010).
Wang, S. et al. The mitophagy pathway and its implications in human diseases. Signal Transduct. Target. Ther. 8, 304 (2023).
Wu, W. et al. FUNDC1 regulates mitochondrial dynamics at the ER-mitochondrial contact site under hypoxic conditions. EMBO J. 35, 1368–1384 (2016).
Lim, Y. et al. Fndc-1 contributes to paternal mitochondria elimination in C. elegans. Dev. Biol. 454, 15–20 (2019).
Chatel-Chaix, L. et al. Dengue virus perturbs mitochondrial morphodynamics to dampen innate immune responses. Cell Host Microbe 20, 342–356 (2016).
Das, R. & Chakrabarti, O. Mitochondrial hyperfusion: a friend or a foe. Biochem. Soc. Trans. 48, 631–644 (2020).
Lim, Y. et al. FNDC-1-mediated mitophagy and ATFS-1 coordinate to protect against hypoxia-reoxygenation. Autophagy 17, 3389–3401 (2021).
Dilberger, B. et al. Mitochondrial oxidative stress impairs energy metabolism and reduces stress resistance and longevity of C. elegans. Oxidative Med. Cell. Longev. 2019, 6840540 (2019).
Terada, H. Uncouplers of oxidative phosphorylation. Environ. Health Perspect. 87, 213–218 (1990).
Hua, X., Liang, G., Chao, J. & Wang, D. Exposure to 6-PPD quinone causes damage on mitochondrial complex I/II associated with lifespan reduction in Caenorhabditis elegans. J. Hazard. Mater. 472, 134598 (2024).
Bess, A. S., Crocker, T. L., Ryde, I. T. & Meyer, J. N. Mitochondrial dynamics and autophagy aid in removal of persistent mitochondrial DNA damage in Caenorhabditis elegans. Nucleic Acids Res. 40, 7916–7931 (2012).
Zhang, W. & Wu, F. Effects of adverse fertility-related factors on mitochondrial DNA in the oocyte: a comprehensive review. Reprod. Biol. Endocrinol. 21, 27 (2023).
Leuthner, T. essC. et al. Resistance of mitochondrial DNA to cadmium and Aflatoxin B1 damage-induced germline mutation accumulation in C. elegans. Nucleic Acids Res. 50, 8626–8642 (2022).
Meshnik, L. et al. Mutant C. elegans mitofusin leads to selective removal of mtDNA heteroplasmic deletions across generations to maintain fitness. BMC Biol. 20, 40 (2022).
Lin, Y.-F. et al. Maintenance and propagation of a deleterious mitochondrial genome by the mitochondrial unfolded protein response. Nature 533, 416–419 (2016).
Liau, W. S., Gonzalez-Serricchio, A. S., Deshommes, C., Chin, K. & LaMunyon, C. W. A persistent mitochondrial deletion reduces fitness and sperm performance in heteroplasmic populations of C. elegans. BMC Genet. 8, 8 (2007).
Rodríguez-Nuevo, A. et al. Oocytes maintain ROS-free mitochondrial metabolism by suppressing complex I. Nature 607, 756–761 (2022).
Kim, K., Kenigsberg, S., Jurisicova, A. & Bentov, Y. The role of mitochondria in oocyte and early embryo health. OBM Genet. 03, 070 (2019).
Spikings, E. C., Alderson, J. & John, J. C. S. Regulated mitochondrial DNA replication during oocyte maturation is essential for successful porcine embryonic development. Biol. Reprod. 76, 327–335 (2007).
Han, L. et al. Increased mtDNA mutation frequency in oocytes causes epigenetic alterations and embryonic defects. Natl Sci. Rev. 9, nwac136 (2022).
Field, J. T. & Gordon, J. W. BNIP3 and Nix: atypical regulators of cell fate. Biochim. Biophys. Acta Mol. Cell Res. 1869, 119325 (2022).
Ren, J. et al. FUNDC1 interacts with FBXL2 to govern mitochondrial integrity and cardiac function through an IP3R3-dependent manner in obesity. Sci. Adv. 6, eabc8561 (2020).
Samuel, A. D. T., Murthy, V. N. & Hengartner, M. O. Calcium dynamics during fertilization in C. elegans. BMC Dev. Biol. 1, 8 (2001).
Kurihara, Y. et al. Mitophagy plays an essential role in reducing mitochondrial production of reactive oxygen species and mutation of mitochondrial DNA by maintaining mitochondrial quantity and quality in yeast. J. Biol. Chem. 287, 3265–3272 (2012).
Mathew, R. & White, E. Autophagy, stress, and cancer metabolism: what doesn’t kill you makes you stronger. Cold Spring Harb. Symp. Quant. Biol. 76, 389–396 (2011).
Cagnone, G. L. et al. Restoration of normal embryogenesis by mitochondrial supplementation in pig oocytes exhibiting mitochondrial DNA deficiency. Sci. Rep. 6, 23229 (2016).
Chiaratti, M. R. & Meirelles, F. V. Mitochondrial DNA copy number, a marker of viability for oocytes. Biol. Reprod. 83, 1–2 (2010).
Tsang, W. Y. & Lemire, B. D. Mitochondrial genome content is regulated during nematode development. Biochem. Biophys. Res. Commun. 291, 8–16 (2002).
Hsieh, R.-H. et al. Multiple rearrangements of mitochondrial DNA in unfertilized human oocytes. Fert. Steril. 77, 1012–1017 (2002).
Ru, Y. et al. Maternal age enhances purifying selection on pathogenic mutations in complex I genes of mammalian mtDNA. Nat. Aging 4, 1211–1230 (2024).
Fan, W. et al. A mouse model of mitochondrial disease reveals germline selection against severe mtDNA mutations. Science 319, 958–962 (2008).
Zhang, H. et al. Mitochondrial DNA heteroplasmy is modulated during oocyte development propagating mutation transmission. Sci. Adv. 7, eabi5657 (2021).
Ross, J. M. et al. Germline mitochondrial DNA mutations aggravate ageing and can impair brain development. Nature 501, 412–415 (2013).
Hahn, A. et al. Misregulation of mitochondrial 6mA promotes the propagation of mutant mtDNA and causes aging in C. elegans. Cell Metab. 36, 2528–2541.e2511 (2024).
Otten, A. B. C. et al. Differences in strength and timing of the mtDNA bottleneck between zebrafish germline and non-germline cells. Cell Rep. 16, 622–630 (2016).
Cao, L. et al. The mitochondrial bottleneck occurs without reduction of mtDNA content in female mouse germ cells. Nat. Genet. 39, 386–390 (2007).
Zhang, Y. et al. Polyamine metabolite spermidine rejuvenates oocyte quality by enhancing mitophagy during female reproductive aging. Nat. Aging 3, 1372–1386 (2023).
Xu, G. et al. Expression pattern and functional analysis of fundc1 in rare minnow (Gobiocypris rarus). Gene 626, 149–157 (2017).
Xu, G. et al. Fundc1 is necessary for proper body axis formation during embryogenesis in zebrafish. Sci. Rep. 9, 18910 (2019).
Kronja, I. et al. Quantitative proteomics reveals the dynamics of protein changes during Drosophila oocyte maturation and the oocyte-to-embryo transition. Proc. Natl Acad. Sci. USA 111, 16023–16028 (2014).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Dickinson, D. J., Ward, J. D., Reiner, D. J. & Goldstein, B. Engineering the Caenorhabditis elegans genome using Cas9-triggered homologous recombination. Nat. Methods 10, 1028–1034 (2013).
Shaner, N. C. et al. A bright monomeric green fluorescent protein derived from Branchiostoma lanceolatum. Nat. Methods 10, 407–409 (2013).
Fang, J. et al. Ribo-On and Ribo-Off tools using a self-cleaving ribozyme allow manipulation of endogenous gene expression in C. elegans. Commun. Biol. 6, 816 (2023).
Raiders, S. A., Eastwood, M. D., Bacher, M. & Priess, J. R. Binucleate germ cells in Caenorhabditis elegans are removed by physiological apoptosis. PLoS Genet. 14, e1007417 (2018).
Poteryaev, D., Squirrell, J. M., Campbell, J. M., White, J. G. & Spang, A. Involvement of the actin cytoskeleton and homotypic membrane fusion in ER dynamics in Caenorhabditis elegans. Mol. Biol. Cell 16, 2139–2153 (2005).
Stavoe, A. K. H. et al. KIF1A/UNC-104 transports ATG-9 to regulate neurodevelopment and autophagy at synapses. Dev. Cell 38, 171–185 (2016).
Sämann, J. et al. Caenorhabditits elegans LRK-1 and PINK-1 act antagonistically in stress response and neurite outgrowth. J. Biol. Chem. 284, 16482–16491 (2009).
Mirza, Z., Walhout, A. J. M. & Ambros, V. A bacterial pathogen induces developmental slowing by high reactive oxygen species and mitochondrial dysfunction in Caenorhabditis elegans. Cell Rep. 42, 113189 (2023).
Tsang, W. Y. & Lemire, B. D. Stable heteroplasmy but differential inheritance of a large mitochondrial DNA deletion in nematodes. Biochem. Cell Biol. 80, 645–654 (2002).
Kelley, L. C. et al. Live-cell confocal microscopy and quantitative 4D image analysis of anchor-cell invasion through the basement membrane in Caenorhabditis elegans. Nat. Protoc. 12, 2081–2096 (2017).
Walston, T. & Hardin, J. An agar mount for observation of Caenorhabditis elegans embryos. Cold Spring Harb. Protoc. 2010, pdb.prot5540 (2010).
Wolke, U., Jezuit, E. A. & Priess, J. R. Actin-dependent cytoplasmic streaming in C. elegans oogenesis. Development 134, 2227–2236 (2007).
Burdet, V. et al. Ultrastructure Expansion Microscopy applied to C. elegans embryos. MicroPubl. Biol. https://doi.org/10.17912/micropub.biology.001033 (2024).
da Rosa, J. et al. Optimizing dsRNA engineering strategies and production in E. coli HT115 (DE3). J. Ind. Microbiol. Biotechnol. 51, kuae028 (2024).
Kamath, R. S. & Ahringer, J. Genome-wide RNAi screening in Caenorhabditis elegans. Methods 30, 313–321 (2003).
Rual, J. F. et al. Toward improving Caenorhabditis elegans phenome mapping with an ORFeome-based RNAi library. Genome Res. 14, 2162–2168 (2004).
Lowry, J. et al. High-throughput cloning of temperature-sensitive Caenorhabditis elegans mutants with adult syncytial germline membrane architecture defects. G3 5, 2241–2255 (2015).
Fu, H. et al. Wounding triggers MIRO-1 dependent mitochondrial fragmentation that accelerates epidermal wound closure through oxidative signaling. Nat. Commun. 11, 1050 (2020).
Kolli, S., Kline, C. J., Rad, K. M. & Wehman, A. M. Phagolysosomes break down the membrane of a non-apoptotic corpse independent of macroautophagy. PLoS ONE 19, e0306435 (2024).
Njume, F. N. et al. A lipid transfer protein ensures nematode cuticular impermeability. iScience 25, 105357 (2022).
Rodriguez, M. E. et al. Targeting of mitochondria by 10-N-alkyl acridine orange analogues: role of alkyl chain length in determining cellular uptake and localization. Mitochondrion 8, 237–246 (2008).
Ashley, N., Harris, D. & Poulton, J. Detection of mitochondrial DNA depletion in living human cells using PicoGreen staining. Exp. Cell. Res. 303, 432–446 (2005).
Hemel, I. M. G. M., Engelen, B. P. H., Luber, N. & Gerards, M. A hitchhiker’s guide to mitochondrial quantification. Mitochondrion 59, 216–224 (2021).
Soh, A. W. J. et al. On-demand delivery of fibulin-1 protects the basement membrane during cyclic stretching in C. elegans. Dev. Cell https://doi.org/10.1016/j.devcel.2025.07.015 (2025).
Klass, M. R. Aging in the nematode Caenorhabditis elegans: major biological and environmental factors influencing life span. Mech. Ageing Dev. 6, 413–429 (1977).
Hughes, S. E., Huang, C. & Kornfeld, K. Identification of mutations that delay somatic or reproductive aging of Caenorhabditis elegans. Genetics 189, 341–356 (2011).
Luo, S., Kleemann, G. A., Ashraf, J. M., Shaw, W. M. & Murphy, C. T. TGF-β and insulin signaling regulate reproductive aging via oocyte and germline quality maintenance. Cell 143, 299–312 (2010).
Montava-Garriga, L., Singh, F., Ball, G. & Ganley, I. G. Semi-automated quantitation of mitophagy in cells and tissues. Mech. Ageing Dev. 185, 111196 (2020).
Fan, X. et al. SapTrap assembly of Caenorhabditis elegans MosSCI transgene vectors. G3 10, 635–644 (2020).
Schärer, O. D. Nucleotide excision repair in eukaryotes. Cold Spring Harb. Perspect. Biol. 5, a012609 (2013).
Crowley, L. C., Christensen, M. E. & Waterhouse, N. J. Measuring mitochondrial transmembrane potential by TMRE staining. Cold Spring Harb. Protoc. https://doi.org/10.1101/pdb.prot087361 (2016).
Boyd, W. A., Smith, M. V. & Freedman, J. H. in Developmental Toxicology: Methods Protocols (eds. Harris, C. & Hansen, J. M.) 15–24 (Humana Press, 2012).
Taylor, S. K. B. et al. C. elegans electrotaxis behavior is modulated by heat shock response and unfolded protein response signaling pathways. Sci. Rep. 11, 3115 (2021).
Srivastava, V. & Gross, E. Detection of mitophagy in Caenorhabditis elegans and mammalian cells using organelle-specific dyes. J. Vis. Exp. https://doi.org/10.3791/65337 (2023).
Leuthner, T. C., Hartman, J. H., Ryde, I. T. & Meyer, J. N. in Mitochondrial Regulation: Methods and Protocols (eds. C. M. Palmeira & A. P. Rolo) 91–111 (Springer US, 2021).
Longhini, K. M. & Glotzer, M. Aurora A and cortical flows promote polarization and cytokinesis by inducing asymmetric ECT-2 accumulation. eLife 11, e83992 (2022).
d’Humières, J., Wang, L., Sherwood, D. R. & Plastino, J. The actin protrusion deforms the nucleus during invasion through basement membrane. Preprint at bioRxivhttps://doi.org/10.1101/2025.03.13.643012 (2025).
Norris, A. D., Kim, H. M., Colaiácovo, M. P. & Calarco, J. A. Efficient genome editing in Caenorhabditis elegans with a toolkit of dual-marker selection cassettes. Genetics 201, 449–458 (2015).
Rieckher, M. & Tavernarakis, N. Caenorhabditis elegans microinjection. Bio. Protoc. 7, e2565 (2017).
Kelly, W. G. & Fire, A. Chromatin silencing and the maintenance of a functional germline in Caenorhabditis elegans. Development 125, 2451–2456 (1998).
Chen, S. & Phillips, C. M. Silencing of a NRDE-3 transgene in C. elegans germ cells and early embryos is mediated by the RNAi pathway. MicroPubl. Biol. https://doi.org/10.17912/micropub.biology.001308 (2024).
Luz, A. L., Smith, L. L., Rooney, J. P. & Meyer, J. N. Seahorse Xfe 24 extracellular flux analyzer-based analysis of cellular respiration in Caenorhabditis elegans. Curr. Protoc. Toxicol. 66, 25.27.21–25.27.15 (2015).
Acknowledgements
We thank the members of the laboratories of D.R.S. and J.M. for helpful discussions, A.W.J. Soh and L.P. Basta for reading and editing the manuscript and L. Yang for help with statistics and maintaining worm strains. FT2296, WEH722 and PHX5270 strains were provided as gifts by J. Nance (Univ. Wisconsin-Madison), A. M. Wehman (Univ. Denver) and P. Laurent (Univ. Libre de Bruxelles). We also thank L. Jameson for technical help with mitochondrial toxicants. This work was supported by R35GM118049 to D.R.S. and P42ES010356 and T32ES021432 to J.M. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
S.B.T. conceived the project, designed the study, performed most of the experiments, analysed the data, wrote the manuscript and prepared the figures. I.R. performed the qPCRs for measurement of uaDf5 ratios. S.B. helped with uaDf5 and UV exposure experiments under the supervision of J.M. K.M. performed measurements of mitochondrial respiration in MA lines and analysed the data under the supervision of J.M. Q.C. and I.K. helped construct molecular biology tools for the project. D.R.S. supervised the project, edited the manuscript and acquired funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Reduction in mtDNA nucleoids during the C. elegans oocyte-to-zygote transition.
a, Confocal fluorescence image of mitochondria during C. elegans OZT. Mitochondria are labelled using Nonyl acridine orange (NAO), a green fluorescent mitochondrial dye. Left: The blue box indicates the –2 oocyte and the pink box the zygote. Right: The –2 oocyte and zygote enclosed in boxed regions are magnified. Scale bars, 10 µm. b, Total fluorescence intensity of NAO in –2 oocytes and zygotes. The data represent the mean ± s.d. (n = 25 animals) from three biological replicates. P value using a two-tailed Mann–Whitney test. c, Top: Confocal fluorescence images of mitochondrial DNA nucleoids (HMG-5::GFP11) during C. elegans OZT. Germlines and zygotes were extruded prior to imaging to improve nucleoid visualization. Scale bars, 10 µm. Middle: Object map overlays depicting mtDNA nucleoid distribution shown in upper panels. Bottom: Boxed regions in upper panels are magnified. Scale bar, 5 µm. d, Total fluorescence intensity of HMG-5::GFP11 in –2 oocytes and zygotes. The data represent the mean ± s.d. (n = 17 animals) from three biological replicates. P value using a two-tailed paired Student’s t-tests. e, Quantification of mtDNA nucleoid number (HMG-5::GFP11) per µm3 compared between –2 oocytes and zygotes. Data represent mean ± s.d. (n = 25 extruded germlines and zygotes) from three biological replicates. P value using a two-tailed paired Student’s t-test. f, Top: Confocal fluorescence images of mitochondrial DNA nucleoids visualized by PicoGreen staining during C. elegans OZT. Scale bars, 10 µm. Middle: Object map depictions of mtDNA nucleoid distribution shown in upper panels. Bottom: Boxed regions in upper panels are magnified. Scale bar, 5 µm. g, Quantification of mtDNA nucleoid number (PicoGreen-positive foci) compared between –2 oocytes and zygotes. Data represent mean ± s.d. (n = 15 animals) from three biological replicates. P value using a two-tailed paired Student’s t-test.
Extended Data Fig. 2 Mitochondrial reduction during OZT is developmentally programmed and induced by sperm signalling.
a, Time-lapse images showing changes in mitochondrial abundance over 40 min in –1 oocytes of a hermaphrodite vs a feminized animal. Fluorescence images show mitochondrial reduction in the –1 oocyte during the transition to the zygote in the hermaphrodite (top), compared to unchanged mitochondrial abundance in an arrested –1 oocyte in a feminized animal (bottom). b, Quantification of mitochondrial reduction from the –2 to –1 oocyte in hermaphrodites versus feminized animals. Data represents the ratio of total NDUV-2::mNG fluorescence intensity in –1 oocytes to that in –2 oocytes, compared between hermaphrodites and feminized animals. The mean ± s.d. is displayed for n = 30 animals each condition from three biological replicates. P value using a two-tailed Student’s t-test. c-e, Changes to mitochondrial volume during the hermaphrodite –2 oocyte transitioning to the –1 oocyte (c), during hermaphrodite OZT (d), and in the –1 oocyte of feminized animals (e). Mitochondrial volume at each time point is normalized to mitochondrial volume at t = 0. The data represent six independent experiments for each condition. P values using a Kruskal–Wallis test followed by a Dunn’s multiple comparisons test. f, Fluorescence images of mitochondria (NDUV-2::mNG) in oocytes of a hermaphrodite and a feminized animal. Green boxes indicate -3 oocytes, blue boxes –2 oocytes, and purple boxes –1 oocytes. Oocytes enclosed in boxed regions are magnified. Yellow arrows indicate donut-shaped mitochondria. Scale bars, 10 µm. Right: Data represent mean ± s.d. proportions (n = 30 animals per condition) from four biological replicates. Refer to Supplementary Fig. 1 for detailed mitochondrial morphology analysis.
Extended Data Fig. 3 Upregulation of the autophagy-lysosome system during the C. elegans oocyte-to-zygote transition.
a, Endogenous ATG-9::GFP distribution in a –2 oocyte and a –1 oocyte. Scale bar, 10 µm. b, Confocal fluorescence images of autophagosomes visualized by germline-specific LC3 protein marker (mCherry::LGG-2) in a –2 oocyte and –1 oocyte. Scale bar, 10 µm. c,d, Confocal fluorescence images of lysosomes visualized using SCAV-3::GFP (c) and CTNS-1::wrmScarlet (d) in - 2 oocytes and –1 oocytes. Scale bars, 10 µm. e-h, Number of ATG-9 vesicles (e), autophagosomes (f), and lysosomes (g,h) in –2 oocytes and –1 oocytes. Data represent mean ± s.d. (n = 30 animals per strain) from three biological replicates. P values using two-tailed Wilcoxon tests (e,f) and two-tailed paired Student’s t-tests (g,h).
Extended Data Fig. 4 Mitochondria are acidified during OZT; DRP-1 and FUNDC1 are necessary for mitochondrial acidification.
a,b, Top: Confocal fluorescence images of mitochondria endogenously tagged with both eGFP and mKate2 in the oocyte and zygote. Scale bar, 10 µm. Bottom: Magnifications of boxed regions. Scale bar, 1 µm. Blue arrows indicate non-acidified mitochondria with robust green and red fluorescence. Yellow arrows indicate acidified mitochondria, where red fluorescence dominates. Mitochondria marked by arrowheads are magnified in (b). Scale bar, 1 µm. Green-to-red (G/R) ratio of the overall –2 oocyte mitochondria was normalized to be 1, and the relative G/R values of marked regions in (b) are denoted. c, Quantification of overall mitochondrial eGFP/mKate2 ratio in –2 oocytes and zygotes, revealing mitochondrial acidification in zygotes. Average eGFP/mKate2 ratio of –2 oocytes was normalized to be 1. Data represent mean ± s.d. (n = 25 animals) from three biological replicates. P value using a two-tailed Student’s t-test. d, Quantification of eGFP/mKate2 ratio in whole oocyte mitochondrial network and in the eGFP-diminished mitochondrial regions noted in zygotes. Average G/R ratio of the overall –2 oocyte mitochondrial network was normalized to be 1. Relatively decreased G/R ratio of these zygotic mitochondria suggests they are acidified. Data represent mean ± s.d. (n = 24 animals) from three biological replicates. P values using a two-tailed Wilcoxon test. e, Semi-automated quantification of the number of acidified mitochondria (mitolysosomes) in –2 oocytes and zygotes quantified using the ‘mitoQC counter’ plugin. Data represent mean ± s.d. (n = 26 animals per strain) from three biological replicates. P values using a two-tailed Wilcoxon test. f–i, Mitochondrial acidification visualized and quantified in drp-1 RNAi-treated animals, similar to a-e. Scale bars, 10 µm (top) and 2 µm (bottom) (f), 2 µm (g). Data represent mean ± s.d. from three biological replicates. P values using a two-tailed Student’s t-test (n = 22 animals) (h) and a two-tailed Mann–Whitney test (n = 16 animals) (i). j–m, Mitochondrial acidification visualized and quantified in fndc-1 mutant animals. Scale bars, 5 µm (top) and 1 µm (bottom) (j), 2 µm (k). Data represent mean ± s.d. from three biological replicates. P values using a two-tailed Student’s t-test (n = 25 animals) (l) and a two-tailed Mann–Whitney test (n = 16 animals) (m). In all panels, blue arrows indicate non-acidified mitochondria, and yellow arrows indicate acidified mitochondria. ‘G/R ratio’ denotes the eGFP/mKate2 fluorescence ratio.
Extended Data Fig. 5 Rapid mitochondrial fragmentation during the oocyte-to-zygote transition.
a, A schematic of the experimental setup to assess changes to mitochondrial interconnectedness during OZT— photoconversion of a specific mitochondrial region and tracking changes to its morphology. Mitochondrial fusion would result in the intermixing of the photoconverted region with the greater network, while fragmentation would be reflected as breaks in the photoconverted region. b, Top: Examples of mitochondrial photoconversion experiments performed on the –1 oocyte mitochondria (TOMM-20::Dendra2). Boxed region is photoconverted (t = 0 s), and mitochondrial structure is assessed at t = 20. Scale bar, 10 µm. Bottom: Boxed region is magnified. Scale bar, 5 µm. White arrows indicate fragmentation of the photoconverted mitochondrial region. The second example (animal 2) shows rapid fragmentation occurring between t = 20 s and t = 22 s, as indicated by white arrows. c-e, Network parameters of the photoconverted mitochondrial region compared immediately upon photoconversion (t = 0 s) and after 20 s. Data represent mean ± s.d. (n = 10 animals) from three biological replicates. P values using two-tailed paired Student’s t-tests.
Extended Data Fig. 6 Mitochondrial fragmentation is not sufficient to regulate the timing and extent of mitochondrial reduction at OZT.
a, Top: Confocal fluorescence images showing the change in mitochondrial morphology (visualized with NDUV-2::mNG) between the –2 oocyte and zygote in an empty vector control compared to a fzo-1 RNAi-treated animal. Scale bar, 10 µm. Boxed regions are magnified under original images. Bottom: Skeletonized images of the mitochondria shown in middle panels. Scale bar, 2 µm. b, Quantification of mitochondrial network parameters (NDUV-2::mNG) compared between –2 oocytes and zygotes in control and fzo-1 RNAi-treated animals. Data represent mean ± s.d. (n = 30 animals per condition) from three biological replicates. P values using two-tailed Student’s t-tests and two-tailed Mann–Whitney tests, depending on normality of distribution. c, Quantification of MOZT in control compared to fzo-1 RNAi-treated animals. Data represent ratio of total NDUV-2::mNG fluorescence intensity in the zygote to that in the –2 oocyte for each animal. The data represent the mean ± s.d. (n = 30 animals each condition) from three biological replicates. P value using a two-tailed Student’s t-test. d, Quantification of mean volume (µm3) of individual fragmented mitochondrion, autophagosome, and lysosome in the –1 oocyte. Data represent mean ± s.d. (n = 30 animals per strain) from three biological replicates. P values using a Kruskal–Wallis test followed by a Dunn’s multiple comparisons test.
Extended Data Fig. 7 FUNDC1 puncta associate with ER and mitochondria, and exhibit two distinct behaviours on oocyte mitochondria.
a-d, Left: mRb3::FNDC-1 puncta visualized together with the endoplasmic reticulum (SP12::GFP) (a), mitochondria (NDUV-2::mNG) (b), ATG-9 puncta (ATG-9::GFP) (c) and lysosomes (SCAV-3::GFP) (d) in the –2 oocyte. Scale bar, 10 µm. Right: Boxed regions are magnified to the right. e, Quantification of Pearson’s correlation coefficient (r) to measure colocalization between mRb3::FNDC-1 puncta and three organelles in the –2 oocyte—the ER, mitochondria, and ATG-9 puncta. Data represent mean ± s.d. (n = 14, 29, 23 animals, respectively) from three biological replicates. P values using one-way ANOVA followed by a Tukey’s multiple comparison test. f, Object-based colocalization analysis measuring proportion of mRb3::FNDC-1 puncta overlapping with ATG-9::GFP positive puncta (n = 15 animals) and lysosomes (SCAV-3::GFP) (n = 19 animals). Data represent mean ± s.d. from three biological replicates. g, Top: Endogenous mRb3::FNDC-1 distribution in the oocyte of a control animal compared to a drp-1 RNAi-treated animal. Yellow and cyan arrows indicate MERC and OMM-localized mRb3::FNDC-1 signal, respectively. Region within dashed lines is segmented below. Bottom: Spectral fluorescence-intensity map of the segmented region, which displays the minimum (L, low) and maximum (H, high) pixel value of the original image. Scale bars, 10 µm. h-j, Number (h), mean fluorescence intensity of mRb3::FNDC-1 puncta (i), and mean fluorescence intensity of OMM-localized mRb3::FNDC-1 signal (j), in oocytes of controls compared to drp-1 RNAi-treated animals. Data represent mean ± s.d. (n = 33 animals per condition) from three biological replicates. P values using two-tailed Mann–Whitney tests. k, Time-lapse images showing dynamic associations of FUNDC1 punctae with mitochondria in the –1 oocyte. Scale bars, 2 µm. Boxes indicate short-dwelling (yellow) and mitochondria-stabilized (blue) FUNDC1 puncta. Note that FUNDC1 puncta observed are not colocalized on mitochondria at t = 0 min, but both gradually localize onto mitochondria. FUNDC1 foci indicated by yellow box starts localizing on mitochondrial surface (NDUV-2::mNG) around t = 2:27 min, fully co-localizes on mitochondria 10 s later, stays associated with mitochondria for 45 s, after which it moves away from mitochondria. FUNDC1 foci indicated by blue box start localizing on mitochondrial surface around t = 1:00 min and stays fully associated with mitochondria for the rest of the time-lapsing experiment (2:37 min). This data represents the two distinct behaviours of FUNDC1 puncta on the –1 oocyte mitochondria shown in Fig. 5e. White arrows indicate the start of FUNDC1 colocalization with mitochondria. Yellow arrow indicates the end of FUNDC1 colocalization with mitochondria. Orange arrow indicates intact mitochondria after short-dwelling FUNDC1 puncta is no longer mitochondrial localized. Supplementary Video 2 shows the time-lapse data from which (k) is derived. l, Pearson’s correlation coefficient (r) plotted over time measuring colocalization of FUNDC1 and mitochondria for both FUNDC1 puncta shown in time-lapse (k). Arrows in representative images from (k) correspond to arrows shown on the line graphs. Data represent FUNDC1 puncta dynamics observed in –1 oocytes of n = 35 animals from 8 independent experiments. m, Proposed model for FUNDC1 localization and function during C. elegans OZT. Panels 1 – 4 show the sequence of molecular events involved in MOZT.
Extended Data Fig. 8 Oocytes upregulate FUNDC1 levels upon mitochondrial stress, but OZT mitochondrial reduction and fragmentation are not altered.
a, Endogenous mRb3::FNDC-1 distribution in oocytes of a control animal compared to Paraquat, FCCP, and 6PPD-Q treated animals. Scale bar, 10 µm. Refer to Supplementary Fig. 3d for the same representative images with mitochondrial (NDUV-2::mNG) signal. b,c, Mean fluorescence intensity of OMM-localized (b) and MERC localized (c) mRb3::FNDC-1 in oocytes of control animals compared to animals treated with Paraquat, FCCP, and 6PPD-Q. Data represent mean ± s.d. for control (n = 10 animals), Paraquat (n = 10 animals), FCCP (n = 11 animals), and 6PPD-Q (n = 16 animals), from two independent experiments. P values using a one-way ANOVA followed by a Tukey’s multiple comparison test. d, Confocal fluorescence images showing the change in mitochondrial abundance and morphology (visualized with NDUV-2::mNG) between the –2 oocyte and zygote in a control animal compared to a UV-treated animal. Scale bar, 2 µm. e, Quantification of mitochondrial network parameters compared between –2 oocytes and zygotes in control and UV-treated animals. Data represent mean ± s.d. (n = 30 animals per condition) from three biological replicates. P values using two-tailed Student’s t-tests (mitochondrial count) and a two-tailed Mann–Whitney test (mitochondrial volume). f, Quantification of mitochondrial reduction in control vs UV-treated animals. Data represent the ratio of total NDUV-2::mNG fluorescence intensity in the zygote to that in the –2 oocyte for each animal. The data represent the mean ± s.d. (n = 30 animals each condition) from three biological replicates. P value using a two-tailed Student’s t-test. g, Distribution of endogenous ATG-9::GFP in –1 oocytes of a control and UV-treated animal. Scale bar, 10 µm. The same control oocyte is displayed in Extended Data Fig. 3a to show autophagy activation. h, Quantification of number and size (surface area, µm²) of ATG-9 vesicles in –1 oocytes of control vs UV-treated animals. Data represent mean ± s.d. (n = 30 animals) from three biological replicates. P values were calculated from a two-tailed Mann–Whitney test (number) and a two-tailed Student’s t-tests (size).
Extended Data Fig. 9 fndc-1 embryos are vulnerable to maternally inherited UV-induced damage.
a, Endogenous mRb3::FNDC-1 distribution in the –2 oocyte of a control animal compared to a UV-treated animal. Blue and yellow arrows indicate OMM and MERC-associated mRb3::FNDC-1 signal, respectively. Scale bar, 10 µm. b, Mean fluorescence intensity of OMM-localized mRb3::FNDC-1 signal (left); number (middle) and mean fluorescence intensity of mRb3::FNDC-1 puncta (right) in –2 oocytes of controls compared to those of UV-treated animals. Data represent mean ± s.d. (n = 30 animals per condition) from three biological replicates. P values using a two-tailed Student’s t-test (left graph) and two-tailed Mann–Whitney tests (middle and right graphs). c, Schematic of experimental design to generate mtDNA lesions in oocytes. Inset: Red dots in mitochondria and nucleus denote DNA lesions. d, DIC images of representative F1 progeny of wild-type and fndc-1 mutant parents (P0) treated with UV radiation at L4 stage. Scale bars, 10 µm. Mean embryonic lethality percentage ± s.d. among F1 progeny is indicated. Data represents 10 progeny observed per strain across three replicates. e, Embryonic lethality (mean ± s.d.) in F1 progeny of untreated controls and UV-treated parents (P0), in wild-type and fndc-1 mutant backgrounds. n = 10 animals were used as P0 parents per strain per condition in three independent experiments. P values using a two-tailed Wilcoxon test (wild-type) and a two-tailed Student’s t-test (fndc-1). f, Quantification of the proportion of fndc-1 embryos arrested at specific stages during embryonic development. Embryos assessed are arrested F1 progeny of UV-treated fndc-1 mothers. g, Merged confocal fluorescence images (top) of mitochondria in F1 embryos from control fndc-1 mutant parents (P0) compared to arrested F1 embryos from UV-treated fndc-1 mutant parents (P0). Green, mitochondrial signal; grey, DIC signal. Scale bars, 10 µm. Boxed regions of mitochondrial signal and their skeletonized representations are magnified below. Scale bar, 2 µm. h-j, Mitochondrial network parameters (NDUV-2::mNG) compared between F1 embryos from untreated (control) and UV-treated fndc-1 mutant parents. Data represent mean ± s.d. (n = 11, 12 embryos per condition) from three biological replicates. P values using a two-tailed Mann–Whitney test (h) and two-tailed Student’s t-test (i,j). k, Top to bottom: Confocal fluorescence images of mitochondria (NDUV-2::mNG), TMRE signal, and merged, in a representative F1 embryo from an untreated fndc-1 mutant parent (P0) compared to two arrested F1 embryos from UV-treated fndc-1 mutant parents (P0). Scale bars, 10 µm. l,m, Mitochondrial membrane potential (TMRE signal per mitochondrial signal) compared between mitochondria of F1 embryos from untreated (control) and UV-treated fndc-1 mutant parents. Data represent mean ± s.d. (n = 18 embryos per condition) from three biological replicates. P values using a two-tailed Welch’s test (l) and a two-tailed Mann–Whitney test (m). n, A Schematic of population bottlenecking experimental design to accelerate mtDNA damage accumulation. Green animals denote untreated control worms and magenta animals denote UV-treated worms. o, Relative brood size as an indicator of reproductive fitness of MA lines across generations. Total brood size was calculated at each generation (G0 to G10) in UV-bottlenecked lines and normalized against that of the respective control bottlenecked lines. Green and magenta lines denote the trajectory of reproductive fitness in wild-type (UV) and fndc-1 (UV) bottlenecked lines, respectively. Graph indicates a decline in reproductive fitness of fndc-1 lines relative to controls across generational time upon UV treatment repeated at each generation. Data points represent mean ± s.e.m (n = 11 animals per strain per treatment) from three independent experiments. P values using Kruskal–Wallis test followed by Dunn’s multiple comparisons test. p, Extinction rates of wild-type (UV) and fndc-1 (UV) bottlenecked lines. Y-axis denotes the percentage of bottlenecked lines present at each generation out of 11 wild-type UV lines (green line) and 11 fndc-1 UV lines (magenta line).
Extended Data Fig. 10 Reproductive decline and embryonic lethality in fndc-1 (UV) lines are strictly maternal effects.
a, A schematic of the experimental design to rescue sperm health in fndc-1 UV lines. Green animals denote untreated worms, and magenta animals denote worms with mtDNA damage. b, Quantification of reproductive fitness (brood size) of self-fertilizing hermaphrodites from fndc-1 UV-bottlenecked lines (fourth generation, G4) compared to those crossed with untreated wild-type males. Data represent mean ± s.d. (n = 11 animals [selfed] and 10 animals [crossed]) from three biological replicates. P values using a two-tailed Student’s t-test. c, Verification of successful crossing between wild-type males and hermaphrodites from fndc-1 UV-bottlenecked lines (G4). Merged confocal images showing F1 progeny with paternally inherited plasma membrane signal (mCherry::PH, magenta; DIC, grey). Magenta signal in progeny confirms successful cross-fertilization by wild-type males. Scale bar, 10 µm. d, Schematic of experimental design to rescue embryonic expression of FUNDC1 in fndc-1 UV-bottlenecked lines (G4). Green denotes the untreated condition, and magenta denotes UV-treated animal. e, Embryonic lethality among F1 progeny of parents from the fndc-1 UV-bottlenecked lines (G4) compared to F1 progeny of parents (from the same lines) injected with FNDC-1 rescuing embryo expression plasmid [pie-1p::mRb3::fndc-1::tbb-2 3′UTR]. Data represent mean ± s.d. (n = 10 animals per condition) from three biological replicates. P values using a two-tailed Mann–Whitney test. f, Merged confocal fluorescence images of mRb3::FNDC-1 (FNDC-1, magenta; DIC, grey) in oocytes of a fndc-1 UV-bottlenecked (G4) parent injected with embryo rescue plasmid and the resulting F1 embryo. Magenta signal in the F1 embryo, but not the parental oocytes confirm embryo-specific rescue of mRb3::FNDC-1 expression. Scale bar, 10 µm.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–7 and Tables 1 and 2.
Supplementary Video 1 (download MP4 )
Mitochondrial fragmentation events during C. elegans OZT; FUNDC1 puncta mark sites of mitochondrial fragmentation.
Supplementary Video 2 (download MP4 )
Dynamic interactions of FUNDC1 puncta with –1 oocyte mitochondria.
Source data
Source Data Fig. 1 (download XLSX )
Statistical Source Data.
Source Data Fig. 2 (download XLSX )
Statistical Source Data.
Source Data Fig. 3 (download XLSX )
Statistical Source Data.
Source Data Fig. 4 (download XLSX )
Statistical Source Data.
Source Data Fig. 5 (download XLSX )
Statistical Source Data.
Source Data Fig. 6 (download XLSX )
Statistical Source Data.
Source Data Fig. 7 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical Source Data.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical Source Data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thendral, S.B., Bacot, S., Ryde, I.T. et al. Programmed mitophagy at the oocyte-to-zygote transition promotes lineage endurance. Nat Cell Biol 28, 268–284 (2026). https://doi.org/10.1038/s41556-025-01854-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41556-025-01854-z