Abstract
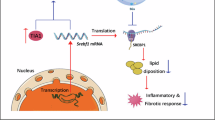
The prevalence of metabolic-dysfunction-associated steatohepatitis (MASH) is rising globally, yet effective treatments remain limited1. Here we found that systemic or hepatocyte-specific ablation of the gene encoding glycoprotein non-metastatic melanoma protein B (Gpnmb)—a top upregulated gene in MASH—protected mice from diet-induced MASH. Notably, MASH progression was driven specifically by the secreted GPNMB ectodomain (G-ECD), rather than full-length GPNMB. Serum G-ECD levels showed a strong positive correlation with MASH severity in human patients. Using an unbiased screen of a cell-surface-displayed transmembrane protein library, we identified related to receptor tyrosine kinase (RYK) as a functional receptor for G-ECD. Hepatocyte-specific Ryk ablation protected mice against MASH and abolished the pathogenic effects of G-ECD. Mechanistically, G-ECD binding to RYK activated ERK1/2 signaling, resulting in transcriptional activation of PPARγ-CD36 and SREBP1C pathways that promote hepatic lipid uptake and lipogenesis. Multiple therapeutic strategies targeting the GPNMB–RYK axis—including vaccination, short hairpin RNA, neutralizing antibody and N-acetylgalactosamine small interfering RNA—effectively prevented and treated MASH in preclinical models. Our findings identify the GPNMB–RYK axis as a new pathogenic ligand–receptor pathway and a promising therapeutic target for MASH.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Raw images for Western blots are provided in Supplementary Fig. 1. The RNA-seq data generated in this study are available through GEO accession no. GSE260956. Source data are provided with this paper.
References
Do, A., Zahrawi, F. & Mehal, W. Z. Therapeutic landscape of metabolic dysfunction-associated steatohepatitis (MASH). Nat. Rev. Drug Discov. 24, 171–189 (2025).
Harrison, S. A., Allen, A. M., Dubourg, J., Noureddin, M. & Alkhouri, N. Challenges and opportunities in NASH drug development. Nat. Med. 29, 562–573 (2023).
Francque, S. et al. Nonalcoholic steatohepatitis: the role of peroxisome proliferator-activated receptors. Nat. Rev. Gastroenterol. Hepatol. 18, 24–39 (2021).
Rinella, M. E. et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology 78, 1966–1986 (2023).
Loomba, R., Friedman, S. L. & Shulman, G. I. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 184, 2537–2564 (2021).
Tsou, P. S. & Sawalha, A. H. Glycoprotein nonmetastatic melanoma protein B: a key mediator and an emerging therapeutic target in autoimmune diseases. FASEB J. 34, 8810–8823 (2020).
Hoashi, T. et al. Glycoprotein nonmetastatic melanoma protein B, a melanocytic cell marker, is a melanosome-specific and proteolytically released protein. FASEB J. 24, 1616–1629 (2010).
Tucher, J. et al. LC-MS based cleavage site profiling of the proteases ADAM10 and ADAM17 using proteome-derived peptide libraries. J. Proteome Res. 13, 2205–2214 (2014).
Govaere, O. et al. A proteo-transcriptomic map of non-alcoholic fatty liver disease signatures. Nat. Metab. 5, 572–578 (2023).
Yang, L. et al. GFRAL is the receptor for GDF15 and is required for the anti-obesity effects of the ligand. Nat. Med. 23, 1158–1166 (2017).
Halford, M. M. & Stacker, S. A. Revelations of the RYK receptor. Bioessays 23, 34–45 (2001).
Yoshikawa, S., McKinnon, R. D., Kokel, M. & Thomas, J. B. Wnt-mediated axon guidance via the Drosophila derailed receptor. Nature 422, 583–588 (2003).
Kim, H. T. et al. WNT/RYK signaling restricts goblet cell differentiation during lung development and repair. Proc. Natl Acad. Sci. USA 116, 25697–25706 (2019).
Seo, S. H. et al. Metabolic improvement and liver regeneration by inhibiting CXXC5 function for non-alcoholic steatohepatitis treatment. Exp. Mol. Med. 54, 1511–1523 (2022).
Wang, S. et al. Nonalcoholic fatty liver disease induced by noncanonical Wnt and its rescue by Wnt3a. FASEB J. 29, 3436–3445 (2015).
Cheng, Z. et al. Luciferase reporter assay system for deciphering GPCR pathways. Curr. Chem. Genomics 4, 84–91 (2010).
Talebi, A. et al. Sustained SREBP-1-dependent lipogenesis as a key mediator of resistance to BRAF-targeted therapy. Nat. Commun. 9, 2500 (2018).
Boonsong, T. et al. Effect of exercise and insulin on SREBP-1c expression in human skeletal muscle: potential roles for the ERK1/2 and Akt signalling pathways. Biochem. Soc. Trans. 35, 1310–1311 (2007).
Prusty, D., Park, B. H., Davis, K. E. & Farmer, S. R. Activation of MEK/ERK signaling promotes adipogenesis by enhancing peroxisome proliferator-activated receptor gamma (PPARgamma) and C/EBPalpha gene expression during the differentiation of 3T3-L1 preadipocytes. J. Biol. Chem. 277, 46226–46232 (2002).
Suda, M. et al. Senolytic vaccination improves normal and pathological age-related phenotypes and increases lifespan in progeroid mice. Nat. Aging 1, 1117–1126 (2021).
Xiong, X. et al. Landscape of intercellular crosstalk in healthy and NASH liver revealed by single-cell secretome gene analysis. Mol. Cell 75, 644–660 e645 (2019).
Gosis, B. S. et al. Inhibition of nonalcoholic fatty liver disease in mice by selective inhibition of mTORC1. Science 376, eabf8271 (2022).
Gong, X. M. et al. Gpnmb secreted from liver promotes lipogenesis in white adipose tissue and aggravates obesity and insulin resistance. Nat. Metab. 1, 570–583 (2019).
Springer, A. D. & Dowdy, S. F. GalNAc-siRNA conjugates: leading the way for delivery of RNAi therapeutics. Nucleic Acid Ther. 28, 109–118 (2018).
Debacker, A. J., Voutila, J., Catley, M., Blakey, D. & Habib, N. Delivery of oligonucleotides to the liver with GalNAc: from research to registered therapeutic drug. Mol. Ther. 28, 1759–1771 (2020).
Shah, V. N. & Pyle, L. Lumasiran, an RNAi therapeutic for primary hyperoxaluria type 1. N. Engl. J. Med. 385, e69 (2021).
Ray, K. K. et al. Two phase 3 trials of inclisiran in patients with elevated LDL cholesterol. N. Engl. J. Med. 382, 1507–1519 (2020).
Patthy, L. The WIF module. Trends Biochem. Sci 25, 12–13 (2000).
Mendrola, J. M., Shi, F., Park, J. H. & Lemmon, M. A. Receptor tyrosine kinases with intracellular pseudokinase domains. Biochem. Soc. Trans. 41, 1029–1036 (2013).
Halford, M. M., Oates, A. C., Hibbs, M. L., Wilks, A. F. & Stacker, S. A. Genomic structure and expression of the mouse growth factor receptor related to tyrosine kinases (Ryk). J. Biol. Chem. 274, 7379–7390 (1999).
Liu, H. et al. Roles of activin A and Gpnmb in metabolic dysfunction-associated steatotic liver disease (MASLD). Diabetes 73, 260–279 (2024).
Katayama, A. et al. Beneficial impact of Gpnmb and its significance as a biomarker in nonalcoholic steatohepatitis. Sci. Rep. 5, 16920 (2015).
Harrison, S. A. et al. A phase 3, randomized, controlled trial of resmetirom in NASH with liver fibrosis. N. Engl. J. Med. 390, 497–509 (2024).
Kannt, A. et al. Activation of thyroid hormone receptor-β improved disease activity and metabolism independent of body weight in a mouse model of non-alcoholic steatohepatitis and fibrosis. Br. J. Pharmacol. 178, 2412–2423 (2021).
Newsome, P. N. & Ambery, P. Incretins (GLP-1 receptor agonists and dual/triple agonists) and the liver. J. Hepatol. 79, 1557–1565 (2023).
Levien, T. L. & Baker, D. E. Resmetirom. Hosp. Pharm. 60, 12–20 (2025).
Wharton, S. et al. Gastrointestinal tolerability of once-weekly semaglutide 2.4 mg in adults with overweight or obesity, and the relationship between gastrointestinal adverse events and weight loss. Diabetes Obes. Metab. 24, 94–105 (2022).
Wang, J. Q. et al. Inhibition of ASGR1 decreases lipid levels by promoting cholesterol excretion. Nature 608, 413–420 (2022).
Lu, X. Y. et al. Feeding induces cholesterol biosynthesis via the mTORC1–USP20–HMGCR axis. Nature 588, 479–484 (2020).
Xi, Y. et al. The anti-fibrotic drug pirfenidone inhibits liver fibrosis by targeting the small oxidoreductase glutaredoxin-1. Sci. Adv. 7, eabg9241 (2021).
Kleiner, D. E. et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41, 1313–1321 (2005).
Acknowledgements
We thank Y. Wang (Wuhan University) for providing AAV cloning vector pAAV-TBG-3×Flag, and Y. Liu (Wuhan University) for providing AAV shRNA vector pAAV-ZsGreen-shRNA. This work was supported by Excellence Research Group Program of NSFC (grant no. 32588301), National Natural Science Foundation of China (grant nos. 3230110204, 22327901 and 32301066), Ministry of Science and Technology (grant nos. 2023YFA0913404 and 2024YFA1306102) and Postdoctoral Science Foundation of China (grant no. 2023M742708). Y.X. was supported in part by the fellowship of China National Postdoctoral Program for Innovative Talents (grant no. BX20230272).
Author information
Authors and Affiliations
Contributions
B.-L.S. conceived and mentored this research. Y.X., W.Q., and B.-L.S. designed the study. Y.X., W.Z., J. L., J.S. and Z.H. performed the experiments and analysed the data. J.Z. and L.W. from Novo Nordisk Research Centre China only contributed to the screening of GPNMB binders using the cell-surface display library. W.L. and S.Z. provided technical support and conceptual advice. Y.X., B.-L.S. and W.Q. wrote the manuscript. All authors edited the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Shuang Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer review reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
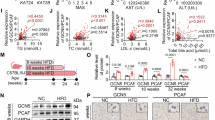
Extended Data Fig. 1 GPNMB is a top upregulated gene in MASH and Gpnmb ablation ameliorates steatosis.
(a and b) RNA-seq analysis of differentially expressed hepatic genes from WT mice fed a normal chow diet (ND), a high-fat/high-cholesterol (HFHC) diet, or an AMLN diet (GSE119340) for 4 months. (c) Hepatic GPNMB expression (log2) in healthy, MASLD, and MASH patients (GSE162694, health n = 31, MASLD n = 65, MASH n = 47; GSE135251, health n = 10, MASLD n = 51, MASH n = 155; GSE89632, health n = 24, MASLD n = 20, MASH n = 19; GSE274114, health n = 9, MASH n = 10; GSE240729, MASLD n = 26, MASH n = 41). The box plots show the interquartile range (IQR; Q1-Q3) (box limits), the median (center line), and the minimum (Q1-1.5×IQR) and maximum (Q3 + 1.5×IQR) values (whiskers). P values were calculated using unpaired two-tailed Student’s t-tests. (d to j) Eight-week-old male WT and Gpnmb−/− mice were fed a HFD for 4 months. BW (d), cumulative food intake (e), H&E and Oil Red O staining, α-SMA immunostaining, and Sirius Red staining, scale bar, 200 μm; representative gross appearance of livers and eWAT, scale bar, 5000 μm (f), histological evaluation of MASLD activity and fibrosis (g), liver TG (h), serum AST and ALT (i), and hepatic expression of inflammatory and fibrogenic genes (j) (WT n = 9, Gpnmb−/− n = 8 mice). Data are presented as mean ± SEM (d, g-j). Unpaired two-tailed Student’s t-test (d, g-j).
Extended Data Fig. 2 Systemic or hepatocyte-specific Gpnmb ablation protects against AMLN diet-induced MASH.
(a to h) Eight-week-old male WT and Gpnmb−/− mice were fed a ND or an AMLN diet for 6 months. BW (a), hepatic Gpnmb expression (b), serum G-ECD (c), H&E and Oil Red O staining, α-SMA immunostaining, and Sirius Red staining of liver sections, scale bar, 200 μm; representative gross appearance of livers, scale bar, 5000 μm (d), histological evaluation of MASLD activity and liver fibrosis (e), liver TG and TC (f), and serum AST and ALT (g) (n = 8 mice per group). Hepatic gene expression (ND n = 6; AMLN WT n = 7; AMLN Gpnmb−/− n = 7 mice) (h). (i) Gpnmb expression in primary hepatocytes (HCs) isolated from WT mice fed a ND or an AMLN diet for 3 months (n = 3 biologically independent experiments). (j to q) Eight-week-old male Gpnmbfl/fl and GpnmbΔHC mice were fed the AMLN diet for 6 months. BW (j), hepatic Gpnmb expression (k), serum G-ECD (l), H&E and Oil Red O staining, α-SMA immunostaining, and Sirius Red staining of liver sections, scale bar, 200 μm; representative gross appearance of livers, scale bar, 5000 μm (m), histological evaluation of MASLD activity and liver fibrosis (n), liver TG (o), and serum AST and ALT (p) (Gpnmbfl/fl n = 10; GpnmbΔHC n = 11 mice). Hepatic gene expression (n = 7 mice per group) (q). Data are presented as mean ± SEM (a-c, e-l, n-q). Unpaired two-tailed Student’s t-test (h-l, n-q) and one-way ANOVA with Tukey’s multiple-comparison test (a-c, e-g).
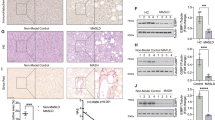
Extended Data Fig. 3 Systemic or hepatocyte-specific Gpnmb ablation ameliorates CDHFD-induced MASH.
(a to j) Eight-week-old male WT and Gpnmb−/− mice were fed a CDHFD for 4 months. BW (a), cumulative food intake (b), hepatic Gpnmb expression (c), serum G-ECD (d), H&E staining, Oil Red O staining, α-SMA immunostaining, and Sirius Red staining, scale bar, 200 μm; representative gross appearance of livers, scale bar, 5000 μm (e), histological evaluation of MASLD activity and fibrosis (f), liver TG (g), and serum AST and ALT (h) (n = 8 mice per group). Hepatic gene expression (n = 7 mice per group) (i). Protein expression of F4/80 and α-SMA (n = 3 mice per group) (j). (k to r) Eight-week-old male Gpnmbfl/fl and GpnmbΔHC mice were fed a CDHFD for 4 months. BW (k), cumulative food intake (l), serum G-ECD (m), H&E staining, Oil Red O staining, α-SMA immunostaining, and Sirius Red staining, scale bar, 200 μm; representative gross appearance of livers, scale bar, 5000 μm (n), histological evaluation of MASLD activity and fibrosis (o), liver TG (p), and serum AST and ALT (q) (n = 7 mice per group). Hepatic gene expression (Gpnmbfl/fl n = 6; GpnmbΔHC n = 7 mice) (r). Data are presented as mean ± SEM (a, c-d, f-i, k, m, o-r). Unpaired two-tailed Student’s t-test (a, c-d, f-i, k, m, o-r).
Extended Data Fig. 4 Characterization of GPNMB variants.
(a) Schematic illustration of GPNMB structure. (b) Illustration of full-length (FL) GPNMB and deletion mutants. (c) Secreted G-ECD in culture medium of Huh7 cells transfected with plasmids expressing GPNMB FL or deletion mutants for 48 h (n = 3 biologically independent experiments). (d) Illustration of membrane-adjacent segment mutations to glycine. (e) Medium G-ECD released from Huh7 cells transfected with plasmids expressing individual GPNMB mutants (n = 3 biologically independent experiments). (f) Medium G-ECD levels of Huh7 cells co-transfected with ADAM10 and GPNMB mutant expression plasmids (n = 3 biologically independent experiments). (g) Illustration of shedding-resistant GPNMB mutants. (h and i) G-ECD/GPNMB in culture medium and cell lysate of Huh7 cells transfected with plasmids expressing GPNMB mutants (h, n = 3 biologically independent experiments; i, repeated twice independently with similar results). (j) Representative images of two independent experimental repeats demonstrating membrane localization of GPNMB variants in Huh7 cells. (k) Histological evaluation of steatosis, ballooning, and inflammation in GpnmbΔHC mice reconstituted with GPNMB variants (n = 8 mice per group). Data are presented as mean ± SEM (c, e-f, h, k). Two-way (f) and one-way (c, e, h, k) ANOVA with Tukey’s multiple-comparison test.
Extended Data Fig. 5 Hepatic GPNMB and circulating G-ECD are positively associated with MASH severity.
(a to c) WT mice were fed a ND for 6 months or an AMLN diet for 3 or 6 months. Hepatic mRNA and protein expression of GPNMB and serum levels of G-ECD (n = 8 mice per group). (d) Spearman’s rank correlation coefficient analysis of hepatic GPNMB expression (log2) with MAS score and Pearson correlation coefficient analysis with serum ALT and AST (GSE89632). (e) Circulating proteomics of G-ECD in healthy individuals and patients with advanced MASLD without T2DM (Female: health n = 25, MASLD n = 3; male: health n = 36, MASLD n = 11). Data are presented as mean ± SEM (a, c, e). Unpaired two-tailed Student’s t-test (e) and one-way ANOVA with Tukey’s multiple-comparison test (a, c).
Extended Data Fig. 6 RYK is required for G-ECD-driven MASH, and G-ECD activates ERK1/2 signaling.
(a to f) Eight-week-old male Alb-G-ECD mice and their WT littermates were subjected to AAV8-shRNA transduction and the AMLN diet-induced MASH model. BW gain (a), serum G-ECD (b), H&E and Oil Red O staining, α-SMA immunostaining, and Sirius Red staining, scale bar, 200 μm; representative gross appearance of livers, scale bar, 5000 μm (c), histological evaluation of MASLD activity and fibrosis (d), liver TG (e), and serum AST and ALT (f) (n = 8 mice per group). (g and h) Gene expression in Huh7 cells treated with the WNT inhibitor ICG-001 (5 μM) and G-ECD (100 ng/ml) for 24 h (n = 3 biologically independent experiments). (i) Luciferase reporter activity in Huh7 cells transfected with individual luciferase reporter genes and treated with G-ECD (100 ng/ml) for 24 h (n = 4 biologically independent experiments). Data are presented as mean ± SEM (a, b, d-i). Unpaired two-tailed Student’s t-test (g, i) and one-way ANOVA followed by Tukey’s multiple-comparison test (a, b, d-f, h).
Extended Data Fig. 7 G-ECD vac prevents MASH development and systemic metabolic abnormalities.
(a to j) Mice are the same as described in Fig. 5a–g. BW (a), food intake (b), histological evaluation of MASLD activity (c), serum AST and ALT (d), fat mass/BW and lean mass/BW ratios (e), weights of eWAT, ingWAT, and BAT, and their BW ratios (f), serum TG and TC (g), and blood glucose (h) (ND n = 10; Con vac n = 8; G-ECD vac n = 8 mice). GTT and ITT (Con vac n = 4; G-ECD vac n = 7 mice) (i). Energy expenditure (n = 7 mice per group) (j). Data are presented as mean ± SEM (a, c-j). Unpaired two-tailed Student’s t-test (a, i, j) and one-way ANOVA with Tukey’s multiple-comparison test (c-h).
Extended Data Fig. 8 AAV8-shGpnmb alleviates established MASH without affecting systemic metabolism.
(a to q) Eight-week-old male WT mice were fed the AMLN diet for 4 months to induce MASH. At this point, a subset was euthanized (4 months). The remaining mice were injected with AAV8-shNC or -shGpnmb and fed the AMLN diet for 2 additional months (a). BW (b), food intake (c), hepatic GPNMB (d), serum G-ECD (e), H&E and Oil Red O staining, α-SMA immunostaining, and Sirius Red staining, scale bar, 200 μm; representative gross appearance of livers, scale bar, 5000 μm (f), histological evaluation (g), liver TG (h), serum AST and ALT (i) (4 months n = 8; shNC n = 7; shGpnmb n = 7 mice). Hepatic gene expression (n = 7 mice per group) (j and k). Fat mass/BW and lean mass/BW ratios (l), weights of eWAT, ingWAT, and BAT, and their BW ratios (m), serum TG and TC (n), and blood glucose (o) (4 months n = 8; shNC n = 7; shGpnmb n = 7 mice). GTT and ITT (shNC n = 5; shGpnmb n = 4 mice) (p), and energy expenditure (n = 6 mice per group) (q). Data are presented as mean ± SEM (b, e, g-q). Unpaired two-tailed Student’s t-test (b, j-l, p, q) and one-way ANOVA followed by Tukey’s multiple-comparison test (e, g-i, m-o).
Extended Data Fig. 9 G-ECD neutralizing antibody reverses established MASH progression and improves systemic metabolism.
(a to j) Mice are the same as described in Fig. 5h–n. BW (a), food intake (b), histological evaluation (c), serum AST and ALT (d), fat mass/BW and lean mass/BW ratios (e), weights of eWAT, ingWAT, and BAT, and their BW ratios (f), serum TG and TC (g), and blood glucose (h) (ND n = 10; 4 months n = 8; IgG n = 8; G-ECD Ab n = 8 mice). GTT and ITT (n = 4 mice per group) (i) and energy expenditure (n = 7 mice per group) (j). Data are presented as mean ± SEM (a, c-j). Unpaired two-tailed Student’s t-test (a, i, j) and one-way ANOVA with Tukey’s multiple-comparison test (c-h).
Extended Data Fig. 10 GalNAc-siGpnmb ameliorates established MASH and improves insulin resistance.
(a to j) Mice are the same as described in Fig. 5o–u. BW (a), food intake (b), histological evaluation of steatosis, ballooning, and inflammation (c), serum AST and ALT (d), fat mass/BW and lean mass/BW ratios (e), weights of eWAT, ingWAT, and BAT, and their BW ratios (f), serum TG and TC (g), and blood glucose (h) (ND n = 10; 4 months n = 7; siNC n = 9; siGpnmb n = 9 mice). GTT and ITT (siNC n = 7; siGpnmb n = 9 mice) (i) and energy expenditure (siNC n = 9; siGpnmb n = 9 mice) (j). Data are presented as mean ± SEM (a, c-j). Unpaired two-tailed Student’s t-test (a, i, j) and one-way ANOVA with Tukey’s multiple-comparison test (c-h).
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1 and 2 and Supplementary Table 1.
Source data
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xi, Y., Zeng, W., Luo, J. et al. RYK is a GPNMB receptor that drives MASH. Nature (2026). https://doi.org/10.1038/s41586-026-10160-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41586-026-10160-z