Abstract
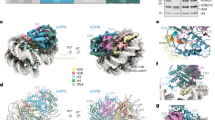
The DNA damage repair regulatory protein RNF168, a monomeric RING-type E3 ligase, has a crucial role in regulating cell fate and DNA repair by specific and efficient ubiquitination of the adjacent K13 and K15 (K13/15) sites at the H2A N-terminal tail. However, understanding how RNF168 coordinates with its cognate E2 enzyme UbcH5c to site-specifically ubiquitinate H2A K13/15 has long been hampered by the lack of high-resolution structures of RNF168 and UbcH5c~Ub (ubiquitin) in complex with nucleosomes. Here we developed chemical strategies and determined the cryo-electron microscopy structures of the RNF168–UbcH5c~Ub–nucleosome complex captured in transient H2A K13/15 monoubiquitination and adjacent dual monoubiquitination reactions, providing a ‘helix-anchoring’ mode for monomeric E3 ligase RNF168 on nucleosome in contrast to the ‘compass-binding’ mode of dimeric E3 ligases. Our work not only provides structural snapshots of H2A K13/15 site-specific monoubiquitination and adjacent dual monoubiquitination but also offers a near-atomic-resolution structural framework for understanding pathogenic amino acid substitutions and physiological modifications of RNF168.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The cryo-EM maps and atomic model of RNF168–UbcH5c in complex with nucleosomes were deposited to the EM Data Bank and PDB under accession codes EMD-38099 and PDB 8X7I (RNF1681–113–UbcH5c~Ub–nucleosome complex obtained using SICC strategy), EMD-38102 (RNF1681–113–UbcH5c~Ub–nucleosome complex obtained using CC strategy, 200-kV reconstruction), EMD-38100 and PDB 8X7J (RNF1681–113–UbcH5c~Ub–nucleosome complex, no Ub conformation, obtained using ACC strategy), EMD-38101 and PDB 8X7K (RNF1681–113–UbcH5c~Ub–H2A K13Ub–nucleosome complex obtained using ACC strategy), EMD-60066 (RNF1681–113–UbcH5c~Ub–H2A K13Ub–nucleosome complex, two-Ub conformation, obtained using ACC strategy) EMD-39800 (RNF1681–193–UbcH5c~Ub–nucleosome complex). The raw images of the RNF1681–113–UbcH5c~Ub–H2A K13Ub–nucleosome complex were deposited to the EM Public Image Archive under accession code EMPIAR-12143. The RNF168-related cancer missense mutations were derived from the ProteinPaint (https://proteinpaint.stjude.org/), which loads COSMIC data. Source data are provided with this paper.
References
Uckelmann, M. & Sixma, T. K. Histone ubiquitination in the DNA damage response. DNA Repair 56, 92–101 (2017).
Song, H. Y. et al. Histone post-translational modification and the DNA damage response. Genes Dis. 10, 1429–1444 (2023).
Stucki, M. et al. MDC1 directly binds phosphorylated histone H2AX to regulate cellular responses to DNA double-strand breaks. Cell 133, 549–549 (2008).
Gong, F. & Miller, K. M. Histone methylation and the DNA damage response. Mutat. Res. Rev. Mutat. Res. 780, 37–47 (2019).
Pinato, S. et al. RNF168, a new RING finger, MIU-containing protein that modifies chromatin by ubiquitination of histones H2A and H2AX. BMC Mol. Biol. 10, 55 (2009).
Mattiroli, F. et al. RNF168 ubiquitinates K13-15 on H2A/H2AX to drive DNA damage signaling. Cell 150, 1182–1195 (2012).
Gatti, M. et al. A novel ubiquitin mark at the N-terminal tail of histone H2As targeted by RNF168 ubiquitin ligase. Cell Cycle 11, 2538–2544 (2012).
Panier, S. & Boulton, S. J. Double-strand break repair: 53BP1 comes into focus. Nat. Rev. Mol. Cell Biol. 15, 7–18 (2014).
Shibata, A. & Jeggo, P. A. Roles for 53BP1 in the repair of radiation-induced DNA double strand breaks. DNA Repair 93, 102915 (2020).
Fradet-Turcotte, A. et al. 53BP1 is a reader of the DNA-damage-induced H2A Lys 15 ubiquitin mark. Nature 499, 50–54 (2013).
Hu, Q., Botuyan, M. V., Cui, G. F., Zhao, D. B. A. & Mer, G. Mechanisms of ubiquitin-nucleosome recognition and regulation of 53BP1 chromatin recruitment by RNF168/169 and RAD18. Mol. Cell 66, 473–487 (2017).
Becker, J. R. et al. BARD1 reads H2A lysine 15 ubiquitination to direct homologous recombination. Nature 596, 433–437 (2021).
Kitevski-LeBlanc, J. et al. The RNF168 paralog RNF169 defines a new class of ubiquitylated histone reader involved in the response to DNA damage. Elife 6, e23872 (2017).
Bartek, J., Bartkova, J. & Lukas, J. DNA damage signalling guards against activated oncogenes and tumour progression. Oncogene 26, 7773–7779 (2007).
Chroma, K. et al. Tumors overexpressing RNF168 show altered DNA repair and responses to genotoxic treatments, genomic instability and resistance to proteotoxic stress. Oncogene 36, 2405–2422 (2017).
Yu, N. et al. RNF168 facilitates proliferation and invasion of esophageal carcinoma, possibly via stabilizing STAT1. J. Cell. Mol. Med. 23, 1553–1561 (2019).
Stewart, G. S. et al. The RIDDLE syndrome protein mediates a ubiquitin-dependent signaling cascade at sites of DNA damage. Cell 136, 420–434 (2009).
Kelliher, J., Ghosal, G. & Leung, J. W. C. New answers to the old RIDDLE: RNF168 and the DNA damage response pathway. FEBS J. 289, 2467–2480 (2022).
Xie, T. Y. et al. Emerging roles of RNF168 in tumor progression. Molecules 28, 1417 (2023).
Mattiroli, F., Uckelmann, M., Sahtoe, D. D., van Dijk, W. J. & Sixma, T. K. The nucleosome acidic patch plays a critical role in RNF168-dependent ubiquitination of histone H2A. Nat. Commun. 5, 3291 (2014).
Horn, V. et al. Structural basis of specific H2A K13/K15 ubiquitination by RNF168. Nat. Commun. 10, 1751 (2019).
Kelliher, J. L., West, K. L., Gong, Q. & Leung, J. W. C. Histone H2A variants α1-extension helix directs RNF168-mediated ubiquitination. Nat. Commun. 11, 2462 (2020).
Dong, S. et al. Recent advances in chemical protein synthesis: method developments and biological applications. Sci. China Chem. 67, 1060–1096 (2024).
Ai, H. S., Pan, M. & Liu, L. Chemical synthesis of human proteoforms and application in biomedicine. ACS Cent. Sci. 10, 1442–1459 (2024).
Pan, M. et al. Structural insights into Ubr1-mediated N-degron polyubiquitination. Nature 600, 334–338 (2021).
Ai, H. S. et al. Synthetic E2-Ub–nucleosome conjugates for studying nucleosome ubiquitination. Chem 9, 1221–1240 (2023).
Deng, Z. H. et al. Mechanistic insights into nucleosomal H2B monoubiquitylation mediated by yeast Bre1–Rad6 and its human homolog RNF20/RNF40–hRAD6A. Mol. Cell 83, 3080–3094 (2023).
Baek, K. et al. NEDD8 nucleates a multivalent Cullin–RING–UBE2D ubiquitin ligation assembly. Nature 578, 461–466 (2020).
Horn-Ghetko, D. et al. Ubiquitin ligation to F-box protein targets by SCF–RBR E3–E3 super-assembly. Nature 590, 671–676 (2021).
Nakasone, M. A. et al. Structure of UBE2K-Ub/E3/polyUb reveals mechanisms of K48-linked Ub chain extension. Nat. Chem. Biol. 18, 422–431 (2022).
Cotton, T. R. et al. Structural basis of K63-ubiquitin chain formation by the Gordon–Holmes syndrome RBR E3 ubiquitin ligase RNF216. Mol. Cell 82, 598–615 (2022).
Streich, F. C. & Lima, C. D. Capturing a substrate in an activated RING E3/E2–SUMO complex. Nature 536, 304–308 (2016).
Chang, L. F., Zhang, Z. G., Yang, J., McLaughlin, S. H. & Barford, D. Atomic structure of the APC/C and its mechanism of protein ubiquitination. Nature 522, 450–454 (2015).
Brown, N. G. et al. RING E3 mechanism for ubiquitin ligation to a disordered substrate visualized for human anaphase-promoting complex. Proc. Natl Acad. Sci. USA 112, 5272–5279 (2015).
Scott, D. C. et al. Structure of a RING E3 trapped in action reveals ligation mechanism for the ubiquitin-like protein NEDD8. Cell 157, 1671–1684 (2014).
Gatti, M. et al. RNF168 promotes noncanonical K27 ubiquitination to signal DNA damage. Cell Rep. 10, 226–238 (2015).
Zheng, Q. Y. et al. A bifunctional molecule-assisted synthesis of mimics for use in probing the ubiquitination system. Nat. Protoc. 18, 530–554 (2023).
Zhang, X. et al. Structural basis for role of ring finger protein RNF168 RING domain. Cell Cycle 12, 312–321 (2013).
Wu, F. S., Zhu, J. S., Li, H. L. & Zhu, L. L. Structural analysis of recombinant human ubiquitin-conjugating enzyme UbcH5c. Acta Pharm. Sin. B 7, 390–394 (2017).
Vijaykumar, S., Bugg, C. E. & Cook, W. J. Structure of ubiquitin refined at 1.8 Å resolution. J. Mol. Biol. 194, 531–544 (1987).
Ai, H. S. et al. H2B Lys34 ubiquitination induces nucleosome distortion to stimulate Dot1L activity. Nat. Chem. Biol. 18, 972–980 (2022).
Cappadocia, L. & Lima, C. D. Ubiquitin-like protein conjugation: structures, chemistry, and mechanism. Chem. Rev. 118, 889–918 (2018).
Thorslund, T. et al. Histone H1 couples initiation and amplification of ubiquitin signalling after DNA damage. Nature 527, 389–393 (2015).
Mandemaker, I. K. et al. DNA damage-induced histone H1 ubiquitylation is mediated by HUWE1 and stimulates the RNF8–RNF168 pathway. Sci. Rep. 7, 15353 (2017).
Takahashi, T. S. et al. Structural insights into two distinct binding modules for Lys63-linked polyubiquitin chains in RNF168. Nat. Commun. 9, 170 (2018).
Panier, S. et al. Tandem protein interaction modules organize the ubiquitin-dependent response to DNA double-strand breaks. Mol. Cell 47, 383–395 (2012).
Plechanovová, A., Jaffray, E. G., Tatham, M. H., Naismith, J. H. & Hay, R. T. Structure of a RING E3 ligase and ubiquitin-loaded E2 primed for catalysis. Nature 489, 115–120 (2012).
Chakraborty, K. & Loverde, S. M. Asymmetric breathing motions of nucleosomal DNA and the role of histone tails. J. Chem. Phys. 147, 065101 (2017).
McGinty, R. K., Henrici, R. C. & Tan, S. Crystal structure of the PRC1 ubiquitylation module bound to the nucleosome. Nature 514, 591–596 (2014).
Bentley, M. L. et al. Recognition of UbcH5c and the nucleosome by the Bmi1/Ring1b ubiquitin ligase complex. EMBO J. 30, 3285–3297 (2011).
Hu, Q. et al. Mechanisms of BRCA1–BARD1 nucleosome recognition and ubiquitylation. Nature 596, 438–443 (2021).
Witus, S. R. et al. BRCA1/BARD1 site-specific ubiquitylation of nucleosomal H2A is directed by BARD1. Nat. Struct. Mol. Biol. 28, 268–277 (2021).
Zhao, F., Hicks, C. W. & Wolberger, C. Mechanism of histone H2B monoubiquitination by Bre1. Nat. Struct. Mol. Biol. 30, 1623–1627 (2023).
Onishi, S. et al. Structure of the human Bre1 complex bound to the nucleosome. Nat. Commun. 15, 2580 (2024).
Stewart, M. D., Ritterhoff, T., Klevit, R. E. & Brzovic, P. S. E2 enzymes: more than just middle men. Cell Res. 26, 423–440 (2016).
Hu, Q. et al. Mechanisms of RNF168 nucleosome recognition and ubiquitylation. Mol. Cell 84, 839–853.e12 (2024).
Buetow, L. et al. Activation of a primed RING E3–E2–ubiquitin complex by non-covalent ubiquitin. Mol. Cell 58, 297–310 (2015).
Patel, A., Sibbet, G. J. & Huang, D. T. Structural insights into non-covalent ubiquitin activation of the cIAP1–UbcH5B∼ubiquitin complex. J. Biol. Chem. 294, 1240–1249 (2019).
Buchwald, G. et al. Structure and E3-ligase activity of the RING–RING complex of polycomb proteins Bmi1 and Ring1b. EMBO J. 25, 2465–2474 (2006).
Kalb, R., Mallery, D. L., Larkin, C., Huang, J. T. J. & Hiom, K. BRCA1 is a histone-H2A-specific ubiquitin ligase. Cell Rep. 8, 999–1005 (2014).
Branigan, E., Carlos Penedo, J. & Hay, R. T. Ubiquitin transfer by a RING E3 ligase occurs from a closed E2–ubiquitin conformation. Nat. Commun. 11, 2846 (2020).
Xie, X. D. et al. The mTOR–S6K pathway links growth signalling to DNA damage response by targeting RNF168. Nat. Cell Biol. 20, 320–331 (2018).
Borad, M. J. et al. Integrated genomic characterization reveals novel, therapeutically relevant drug targets in FGFR and EGFR pathways in sporadic intrahepatic cholangiocarcinoma. PLoS Genet. 10, e1004135 (2014).
White, B. S. et al. A multiple myeloma-specific capture sequencing platform discovers novel translocations and frequent, risk-associated point mutations in IGLL5. Blood Cancer J. 8, 35 (2018).
Zhang, L. et al. Genomic analyses reveal mutational signatures and frequently altered genes in esophageal squamous cell carcinoma. Am. J. Hum. Genet. 107, 579–579 (2020).
Cheng, C. X. et al. Genomic analyses reveal FAM84B and the NOTCH pathway are associated with the progression of esophageal squamous cell carcinoma. Gigascience 5, 1 (2016).
Gingras, M. C. et al. Ampullary cancers harbor ELF3 tumor suppressor gene mutations and exhibit frequent WNT dysregulation. Cell Rep. 14, 907–919 (2016).
Pickering, C. R. et al. Mutational landscape of aggressive cutaneous squamous cell carcinoma. Clin. Cancer Res. 20, 6582–6592 (2014).
Bertotti, A. et al. The genomic landscape of response to EGFR blockade in colorectal cancer. Nature 526, 263–267 (2015).
Wang, H. B. et al. Role of histone H2A ubiquitination in polycomb silencing. Nature 431, 873–878 (2004).
Cao, R., Tsukada, Y. & Zhang, Y. Role of Bmi-1 and Ring1A in H2A ubiquitylation and Hox gene silencing. Mol. Cell 20, 845–854 (2005).
Nishiyama, A. et al. Uhrf1-dependent H3K23 ubiquitylation couples maintenance DNA methylation and replication. Nature 502, 249–253 (2013).
Ishiyama, S. et al. Structure of the Dnmt1 reader module complexed with a unique two-mono-ubiquitin mark on histone H3 reveals the basis for DNA methylation maintenance. Mol. Cell 68, 350–360 (2017).
Li, T. et al. Structural and mechanistic insights into UHRF1-mediated DNMT1 activation in the maintenance DNA methylation. Nucleic Acids Res. 46, 3218–3231 (2018).
Zheng, Q. Y. et al. An E1-catalyzed chemoenzymatic strategy to isopeptide-N-ethylated deubiquitylase-resistant ubiquitin probes. Angew. Chem. Int. Ed. 59, 13496–13501 (2020).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. Elife 7, e42166 (2018).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Acknowledgements
M.P. was supported by the National Key R&D Program of China (no. 2023YFA0915300), National Natural Science Foundation of China (no. 22277073), Shanghai Rising-Star Program (22QA1404900), Shanghai Pilot Program for Basic Research—Shanghai Jiao Tong University (21TQ1400224) and the Fundamental Research Funds for the Central University. L.Liu was supported by the National Key R&D Program of China (no. 2022YFC3401500), National Natural Science Foundation of China (nos. 22137005, 92253302, 22227810, and T2488301), XPLORER prize and New Cornerstone Investigator Program. C.T. was supported by the National Key R&D Program of China (no. 2022YFC3400502), National Natural Science Foundation of China (nos. 21825703 and 21927814) and Strategic Priority Research Program of Chinese Academy of Sciences (XDB37000000). H.A. thanks the National Facility for Translational Medicine (Shanghai) for funding. We acknowledge the Tsinghua University Branch of China National Center for Protein Sciences (Beijing) for cryo-EM data screening and collection.
Author information
Authors and Affiliations
Contributions
H.A., Z.T., Z.D., C.T., L.Liu and M.P. proposed the idea, designed the experiments and analyzed the results. Z.T. and L.Liu designed and optimized the SICC and ACC strategies. Z.T., Z.D., H.A., S.T. and Q.S. cloned the plasmids, expressed the proteins (RNF168, UbcH5c, histones and mutants) and reconstituted the nucleosomes. Z.T., H.A. and Z.D. synthesized the IntN-E2~Ub-conjugated H2A K15C, E2~Ub-conjugated H2A K15C, H2A K15CAT and H2A K13Ub/K15CAT nucleosome probes. H.A., Z.D., Z.T. and S.T. prepared the fluorescently labeled H2A histones. Q.S. and Z.D. synthesized the fluorescently labeled ubiquitinated histone H2A. Z.D. and J.L. synthesized the H2A K13Ubmut/K15 histone. H.A. and Z.D. performed the RNF168–H2A K13/15 selectivity assays and RNF168–UbcH5c mutant activity experiments on unmodified nucleosome substrates or acidic patch mutant nucleosomes. Z.T. performed the RNF168 ubiquitination experiments of Ub chain elongation and Ub mutants on the H2A K15Ub NCP. Z.D. performed the RNF8-related and SET8-related RNF168 ubiquitination experiments and the RNF168 mutant activity tests on ubiquitinated nucleosomes. Z.T. prepared the cryo-EM samples. H.A., Z.T. and Z.D. checked the samples and collected the cryo-EM data. H.A. processed the cryo-EM data, determined the cryo-EM structures and built the atomic models. Z.D. and G.S. conducted the immunofluorescence assay. Z.D., Z.T. and H.A. collated the experimental data and prepared the figure panels and tables. H.A. drafted the manuscript. H.A., Z.D., Z.T., C.T., L.Liu and M.P. revised the manuscript. All authors (H.A., Z.T., Z.D., Q.S., S.T., G.S., J.L., M.S., X.W., Q.Z., L.Liang, H.Y., J.-B.L., S.G., C.T., L.Liu and M.P.) read, discussed and analyzed the manuscript. M.P., L.Liu and C.T. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 RNF168 prefers to ubiquitinate H2AK13/15 without polyubiquitin chain linkage preference.
a, In vitro ubiquitination assay to compare the activity of RNF1681–113 and RNF168FL to generate monoubiquitination (H2AUb) and dual-monoubiquitination (H2AUb2) on H2A K13/K15 NCP. b, In vitro ubiquitination assay shows that RNF1681–113 preferentially ubiquitinates H2AK13/15 rather than H2BK120. c, In vitro ubiquitination assay shows that RNF1681–113 together with UbcH5c can generate short ubiquitin chains on H2A K13R/K15 NCP. Note that there is only one lysine (H2A K15) on H2A K13/K15 NCP that can be conjugated with ubiquitin and bands of H2AUb3 were observed, suggesting that at least three ubiquitin were conjugated to H2A K15. The PRC1RING E3 ligase that site-specifically catalyzes nucleosomal H2A K118/K119 monoubiquitination was used as a negative control and it failed to generate significant H2A ubiquitination on H2A K13R/K15 NCP. d. In vitro ubiquitination assay to test the ubiquitin chain elongation activity of RNF1681–113 or RNF168FL on H2A K13R/K15Ub NCP. Note that in this nucleosome substrate, all the lysines on H2A were substituted by arginines so that ubiquitin can only be conjugated to the Ub motif at the H2A K15 site. It was observed that RNF1681–113 or RNF168FL generated H2AUb3, suggesting that no more than two Ub were conjugated to the Ub motif of the H2A K13R/K15Ub NCP and the ubiquitin chain elongation activity of RNF1681–113 or RNF168FL is weak. e,f, In vitro ubiquitination assays to investigate the ubiquitin chain linkage preference of RNF1681–113 on H2A K13R/K15 NCP (e) and H2A K13R/K15Ub NCP (f). These results suggest that RNF1681–113 generates short and mixed ubiquitin chains at H2A K15 sites with no linkage preference in vitro. For all the biochemical results, three independent experiments were performed with similar results.
Extended Data Fig. 2 Cryo-EM data processing for RNF1681–113/UbcH5c–Ub/NCP complex achieved by SICC strategy.
a, A representative micrograph. The micrographs collected in this dataset show similar particle density and dispersion to the representative micrograph. b, CTF estimation of the micrograph shown in a. c, Representative 2D classifications. d, Data processing flowcharts. e, Euler angle distributions of the final particles for cryo-EM reconstruction. f, Local resolution colored cryo-EM density map. g, Gold standard Fourier shell correlation (FSC) curves showing the overall resolution of 3.27 Å and 3.62 Å for the final density map of RNF168/UbcH5c–Ub/NCP, and RNF168/UbcH5c, respectively.
Extended Data Fig. 3 Cryo-EM data processing for RNF1681–113/UbcH5c–Ub/NCP complex achieved by CC strategy.
a, Schematic presentation of nucleosomal H2A ubiquitination intermediate showing the tetrahedron transition state and two different mimicries. b, Data processing flowcharts. c, Euler angle distributions of the final particles for cryo-EM reconstruction. d, Gold standard Fourier shell correlation (FSC) curves showing the overall resolution of 7.13 Å.
Extended Data Fig. 4 Cryo-EM data processing for RNF1681–193/UbcH5c–Ub/NCP complex achieved by CC strategy.
a, A representative micrograph. The micrographs collected in this dataset show similar particle density and dispersion to the representative micrograph. b, CTF estimation of the micrograph shown in a. c, Representative 2D classifications. d, Data processing flowcharts. e, Euler angle distributions of the final particles for cryo-EM reconstruction of RNF1681–193/UbcH5c/NCP conformation. f, Euler angle distributions of the final particles for cryo-EM reconstruction of RNF1681–193/UbcH5c-Ub/NCP conformation. g, Gold standard Fourier shell correlation (FSC) curves showing the overall resolution of 3.23 Å and 3.59 Å for the final density map of RNF1681–193/UbcH5c/NCP and RNF1681–193/UbcH5c-Ub/NCP conformation, respectively.
Extended Data Fig. 5 Comparison of CC strategy, SICC strategy, and ACC strategies.
a. Cartoon illustration of the CC strategy. E2-Ub is covalently attached to the nucleosome by chemical synthesis, followed by the addition of E3 ligase to form the complex. b. Cartoon illustration of the SICC strategy. Given the basis of the E2-Ub-nucleosome conjugation strategy, the split intein enables protein trans-splicing reaction resulting in the formation of a covalent bond between E3 and E2. A covalent complex of E3-E2-Ub-nucleosome is finally acquired. c. Cartoon illustration of the ACC strategy. Firstly, E3 ligase and E2-Ub are covalently linked by a trans-splicing reaction, and a covalent bond is formed between E2-Ub and nucleosome based on the E3 ligase activity.
Extended Data Fig. 6 Cryo-EM data processing for RNF1681–113/UbcH5c–Ub/H2AK13Ub NCP complex achieved by ACC strategy.
a, A representative micrograph. The micrographs collected in this dataset show similar particle density and dispersion to the representative micrograph. b, Representative 2D classifications. c, Data processing flowcharts. d, Euler angle distributions of the final particles for cryo-EM reconstruction of RNF168/UbcH5c/two Ub conformation. e, Euler angle distributions of the final particles for cryo-EM reconstruction of RNF168/UbcH5c/NCP conformation. f, Gold standard Fourier shell correlation (FSC) curves showing the overall resolution of 3.52 Å and 3.20 Å for the final density map of RNF1681–113/UbcH5c/two Ub conformation and RNF1681–113/UbcH5c/NCP conformation, respectively.
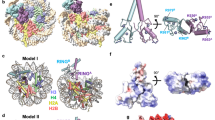
Extended Data Fig. 7 Analysis of the E3-E2 interfaces and canonical closed E2~Ub conformation.
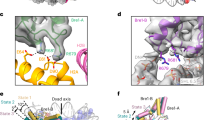
a, Structural alignment of RNF1681–113/UbcH5c–Ub/NCP complex achieved by SICC strategy, and the structure of RNF1681–113/UbcH5c–Ub/H2A K13Ub NCP complex captured by ACC strategy, highlighting the congruence in nucleosome-binding mode and E3 /E2 arrangement. b,c, Close-up views of RNF168/UbcH5c on the nucleosomal acidic patch in the two structures including the RNF1681–113/UbcH5c–Ub/NCP complex by SICC strategy (PDB:8X7I) (b), and the RNF1681–113/UbcH5c–Ub/H2A K13Ub NCP complex by ACC strategy (PDB:8X7K) (c). The Cαs of UbcH5c active center C85 and H2A K15 are depicted as spheres and the distances between them are indicated. d. Overview of interactions of RNF1681–113 and UbcH5c. Rectangle regions indicate closed-up interfaces shown in e and f. e, Close-up view highlighting the RNF168–UbcH5c SPA motif interface. f, Close-up view highlighting the interface centered on the RNF1681–113 linchpin R55 and UbcH5c N92. g. Alignment of four E3/E2–Ub complexes (PDB: 4AUQ, 4AP4, 6TTU, and 8GRM) that contain UbcH5 family E2s with Ub at the close conformation and the current RNF168/UbcH5c structure. The Cαs of UbcH5 E2 residues that play important roles in mediating closed E2~Ub conformation (I88, R90, L97, L104, D116, and D117) and non-covalent ubiquitin binding stimulation (S22) are colored red and blue, respectively. h. In vitro ubiquitination assay using WT-H2A NCP to investigate the effects of UbcH5c mutants on overall nucleosomal H2A ubiquitination pattern. Data show the mean ± SD (bars) from n = 3 independent biological replicates. i. Alignment of our RNF168/UbcH5c in our complex structure with four nucleosomal E3/E2 complexes (PDB: 4R8P, 7JZV, 8IEG, and 8IEJ), highlighting the conserved E3/E2 spatial arrangement and the E3–E2 interface.
Extended Data Fig. 8 Cryo-EM data processing for RNF1681–113/UbcH5c–Ub/NCP complex achieved by ACC strategy.
a, A representative micrograph. The micrographs collected in this dataset show similar particle density and dispersion to the representative micrograph. b, Representative 2D classifications. c, Data processing flowchart. d, Cryo-EM density map at an overall resolution of 6.87 Å representing the closed E2~Ub conformation. e, Local resolution analysis of the 3.39 Å cryo-EM density map. f, Gold standard Fourier shell correlation (FSC) curves showing the overall resolution of 3.39 Å and 3.27 Å for the final density map of RNF1681–113/UbcH5c–Ub/NCP and RNF1681–113/UbcH5c/NCP conformation, respectively.
Extended Data Fig. 9 Sample densities for the cryo-EM reconstructions of RNF168/UbcH5c-mediated nucleosomal H2A ubiquitination.
a–f, Main chain traces of the whole complex (a), DNA (b), histone octamer (c), RNF168 (d), UbcH5c (e), and Ub (f) fit into the cryo-EM density map of the RNF1681–113/UbcH5c–Ub/NCP complex achieved by SICC strategy. g,h, Two views of the representative region of the cryo-EM density map of the RNF1681–113/UbcH5c–Ub/NCP complex achieved by SICC strategy for the RNF168–acidic patch interface with side chains of RNF168 R57, S60, and R63, and some H2A/H2B residues shown. i–n, Main chain traces of the whole complex (i), DNA (j), histone octamer (k), RNF168 (l), UbcH5c (m), and Ub (n) fit into the cryo-EM density map of the RNF1681–113/UbcH5c–Ub/NCP complex achieved by ACC strategy. o and p, Two views of the representative region of the cryo-EM density map of the RNF1681–113/UbcH5c–Ub/NCP complex achieved by ACC strategy for the RNF168–acidic patch interface with side chains of RNF168 S60 and R63, and some H2A/H2B residues shown. q–u, Main chain traces of the whole complex (q), DNA (r), histone octamer (s), RNF168 (t), and UbcH5c (u) fit into the cryo-EM density map of the RNF1681–113/UbcH5c–Ub/H2AK13Ub NCP complex achieved by ACC strategy. v,w, Two views of the representative region of the cryo-EM density map of the RNF1681–113/UbcH5c–Ub/H2AK13Ub NCP complex achieved by ACC strategy for the RNF168–acidic patch interface with side chains of RNF168 R57, S60, and R63, and some H2A/H2B residues were shown.
Extended Data Fig. 10 Biochemical investigations of the role of the structurally observed Ubback in the RNF168-mediated H2A ubiquitination.
a, Models showing the spatial proximity of SHL DNA 3.5 and Ubback and the potential interface of UbcH5c and Ubback. The models are from the RNF168/UbcH5c–Ub/NCP complex achieved by intein-based E2–Ub–nucleosome conjugation strategy. Nucleosomal DNA is present as cryo-EM density surface and Ubback, UbcH5c, and H2A are shown as ribbon models. RNF168, H2B, H3, and H4 are hidden for clarity. The Cαs of Ubback positive residues near the SHL DNA 3.5 (K6, R42, K48, and R72) and Ubback residues near UbcH5c (E34, G35, D39, and R74) are colored red, light blue, and medium blue, respectively. b, Schematic representation of biochemical assays to investigate the effects of Ubback mutations in the second H2A ubiquitination step. Note the Ubback mutations are introduced to the H2A K13Ub/K15 NCP which is the substrate in the 2nd H2A ubiquitination step. c, In vitro ubiquitination assays described in b using H2A K13UbMut/K15 NCP as substrates. Coomassie Brilliant Blue-stained bands of H2AUb (substrate) and H2AUb2 (product) are indicated, and the band of H4 in each lane was used as a control to calculate the H2AUb2 generation efficiency in d. d, Quantified H2AUb2 generation efficiency in c. Combination mutations of Ubback positive residues near the SHL DNA 3.5 (K6D, R42E, K48D, and R72E) and Ubback residues near UbcH5c (D39K and R74E) slightly reduced the second Ub conjugation efficiency at the H2A K15 site on the H2A K13UbMut/K15 NCPs. Data show the mean ± SD (bars) from n = 3 independent biological replicates.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–3 and Figs. 1–12.
Supplementary Data (download PDF )
Uncropped gels.
Source data
Source Data Fig. 1 (download PDF )
Uncropped gels.
Source Data Fig. 2 (download PDF )
Uncropped gels.
Source Data Fig. 3 (download PDF )
Uncropped gels.
Source Data Fig. 4 (download PDF )
Uncropped gels.
Source Data Fig. 6 (download PDF )
Uncropped gels.
Source Data Extended Data Fig. 1 (download PDF )
Uncropped gels and blots.
Source Data Extended Data Fig. 10 (download PDF )
Uncropped gels.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ai, H., Tong, Z., Deng, Z. et al. Mechanism of nucleosomal H2A K13/15 monoubiquitination and adjacent dual monoubiquitination by RNF168. Nat Chem Biol 21, 668–680 (2025). https://doi.org/10.1038/s41589-024-01750-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41589-024-01750-x
This article is cited by
-
Structural basis for E4 enzyme Ufd2-catalyzed K48/K29 branched ubiquitin chains
Nature Chemical Biology (2026)
-
Unique gluing effect of ASXL1 K351 monoubiquitination stimulates the PR-DUB activity
Nature Chemical Biology (2026)
-
Allosteric activation of RNF20/RNF40–RAD6A-mediated H2BK120 monoubiquitylation by H2BS112 GlcNAcylation
Nature Chemical Biology (2026)
-
Structural visualization of HECT-type E3 ligase Ufd4 accepting and transferring ubiquitin to form K29/K48-branched polyubiquitination
Nature Communications (2025)
-
Advances in the chemical synthesis of human proteoforms
Science China Life Sciences (2025)