Abstract
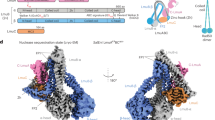
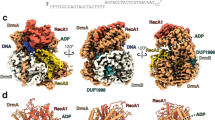
Bacteria have developed a variety of immune systems to combat phage infections. The Lamassu system is a prokaryotic immune system with a core conserved structural maintenance of chromosomes (SMC) superfamily protein LmuB and diverse effectors named LmuA, whose mechanism remains unclear. Here we present a series of cryo-electron microscopy structures of the type-I Lamassu complex from Bacillus cellulasensis and the type-II Lamassu complex from Vibrio cholerae, both in apo and dsDNA-bound states, revealing an unexpected stoichiometry and topological architecture distinct from canonical SMC complexes. Combined structural and biochemical analyses show how the nuclease effector LmuA is sequestered in an inactive monomeric form within the Lamassu complex and, upon sensing foreign DNA ends, dissociates and assembles into an active tetramer capable of DNA cleavage. Our findings elucidate the mechanism by which Lamassu systems detect viral replication and implement antiphage defense, highlighting the roles of SMC proteins in prokaryotic immunity.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Cryo-EM structures and atomic models generated in this study have been deposited in the wwPDB OneDep System under EMD accession codes EMD-64575, EMD-64576, EMD-64585, EMD-64586, EMD-64583 and EMD-64584 and PDB ID codes under accession codes 9UX7, 9UX8, 9UXK, 9UXL, 9UXH and 9UXI. Source data are provided with this paper.
References
Georjon, H. & Bernheim, A. The highly diverse antiphage defence systems of bacteria. Nat. Rev. Microbiol. 21, 686–700 (2023).
Deltcheva, E. et al. CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature 471, 602–607 (2011).
Barrangou, R. & Horvath, P. A decade of discovery: CRISPR functions and applications. Nat. Microbiol. 2, 17092 (2017).
Tock, M. R. & Dryden, D. T. F. The biology of restriction and anti-restriction. Curr. Opin. Microbiol. 8, 466–472 (2005).
Kuzmenko, A. et al. DNA targeting and interference by a bacterial Argonaute nuclease. Nature 587, 632–637 (2020).
Rousset, F. & Sorek, R. The evolutionary success of regulated cell death in bacterial immunity. Curr. Opin. Microbiol. 74, 102312 (2023).
Lopatina, A., Tal, N. & Sorek, R. Abortive infection: bacterial suicide as an antiviral immune strategy. Annu Rev. Virol. 7, 371–384 (2020).
Cohen, D. et al. Cyclic GMP–AMP signalling protects bacteria against viral infection. Nature 574, 691–695 (2019).
Ofir, G. et al. Antiviral activity of bacterial TIR domains via immune signalling molecules. Nature 600, 116–120 (2021).
Liu, X. et al. Target RNA activates the protease activity of Craspase to confer antiviral defense. Mol. Cell 82, 4503–4518 (2022).
Wang, X., Yao, J., Sun, Y.-C. & Wood, T. K. Type VII toxin/antitoxin classification system for antitoxins that enzymatically neutralize toxins. Trends Microbiol. 29, 388–393 (2021).
Stokar-Avihail, A. et al. Discovery of phage determinants that confer sensitivity to bacterial immune systems. Cell 186, 1863–1876 (2023).
Whiteley, A. T. et al. Bacterial cGAS-like enzymes synthesize diverse nucleotide signals. Nature 567, 194–199 (2019).
Duncan-Lowey, B. & Kranzusch, P. J. CBASS phage defense and evolution of antiviral nucleotide signaling. Curr. Opin. Immunol. 74, 156–163 (2022).
Doron, S. et al. Systematic discovery of antiphage defense systems in the microbial pangenome. Science 359, 6379 (2018).
Payne, L. J. et al. Identification and classification of antiviral defence systems in bacteria and archaea with PADLOC reveals new system types. Nucleic Acids Res. 49, 10868–10878 (2021).
Krishnan, A., Burroughs, A. M., Iyer, L. M. & Aravind, L. Comprehensive classification of ABC ATPases and their functional radiation in nucleoprotein dynamics and biological conflict systems. Nucleic Acids Res. 48, 10045–10075 (2020).
Millman, A. et al. An expanded arsenal of immune systems that protect bacteria from phages. Cell Host Microbe 30, 1556–1569 (2022).
Aframian, N. & Eldar, A. Abortive infection antiphage defense systems: separating mechanism and phenotype. Trends Microbiol. 31, 1003–1012 (2023).
Jaskolska, M., Adams, D. W. & Blokesch, M. Two defence systems eliminate plasmids from seventh pandemic Vibrio cholerae. Nature 604, 323–329 (2022).
Robins, W. P., Meader, B. T., Toska, J. & Mekalanos, J. J. DdmABC-dependent death triggered by viral palindromic DNA sequences. Cell Rep. 43, 114450 (2024).
Burmann, F. & Lowe, J. Structural biology of SMC complexes across the tree of life. Curr. Opin. Struct. Biol. 80, 102598 (2023).
Yatskevich, S., Rhodes, J. & Nasmyth, K. Organization of chromosomal DNA by SMC complexes. Annu. Rev. Genet. 53, 445–482 (2019).
Lowey, B. et al. CBASS immunity uses CARF-related effectors to sense 3′–5′- and 2′–5′-linked cyclic oligonucleotide signals and protect bacteria from phage infection. Cell 182, 38–49 (2020).
Li, Y. et al. Structure and activation mechanism of a Lamassu phage and plasmid defense system. Nat Struct Mol Biol. https://doi.org/10.1038/s41594-025-01677-4 (2025).
Kashammer, L. et al. Mechanism of DNA end sensing and processing by the Mre11–Rad50 complex. Mol. Cell 76, 382–394 (2019).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Sophie, N. & David, S. The bacterial chromosome: architecture and action of bacterial SMC and SMC-like complexes. FEMS Microbiol. Rev. 38, 380–392 (2014).
Haudiquet, M. et al. Structural basis for Lamassu-based antiviral immunity and its evolution from DNA repair machinery. Proc. Natl Acad. Sci. USA 122, e2519643122 (2025).
Buckstein, M. H., He, J. & Rubin, H. Characterization of nucleotide pools as a function of physiological state in Escherichia coli. J. Bacteriol. 190, 718–726 (2008).
Rousset, F. et al. A conserved family of immune effectors cleaves cellular ATP upon viral infection. Cell 186, 3619–3631 (2023).
Tang, D., Chen, Y. & Yu, C. Y. Multiple enzymatic activities of a Sir2-HerA system cooperate for anti-phage defense. Mol. Cell 83, 4600–4613 (2023).
Li, J. et al. Structures and activation mechanism of the Gabija anti-phage system. Nature 629, 467–473 (2024).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Tang, G. et al. EMAN2: an extensible image processing suite for electron microscopy. J. Struct. Biol. 157, 38–46 (2007).
Pettersen, E. F. et al. UCSF ChimeraX: structure visualization for researchers, educators, and developers. Protein Sci. 30, 70–82 (2021).
Trabuco, L. G., Villa, E., Mitra, K., Frank, J. & Schulten, K. Flexible fitting of atomic structures into electron microscopy maps using molecular dynamics. Structure 16, 673–683 (2008).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Acknowledgements
We thank the Cryo-EM Center at the University of Science and Technology of China (USTC) for their assistance with the cryo-EM experiments. This work is supported by National Key Research and Development Program of China (grant nos. 2024YFA0916903 to Y.Z., 2022YFC3401500 to Y.F., 2022YFA1302700 to K.Z., 2022YFC2303700 to K.Z. and 2022YFC2104800 to Y.F.), the National Natural Science Foundation of China (grant nos. 32371329 to Y.Z., 32171274 to Y.F., 32371345 to K.Z., 32301044 and 32471301 to S.L.), Scientific Research Innovation Capability Support Project for Young Faculty (grant no. ZYGXQNJSKYCXNLZCXM-B1) to Y.F., the Fundamental Research Funds for the Central Universities (grant no. QNTD2023-01) to Y.F., Anhui Provincial Natural Science Foundation (grant no. 2308085QC80) to S.L., the Strategic Priority Research Program of the Chinese Academy of Sciences (grant no. XDB0490000) to K.Z., the Center for Advanced Interdisciplinary Science and Biomedicine of IHM (grant no. QYPY20220019) to K.Z. and the USTC Research Funds of the Double First-Class Initiative (grant nos. YD9100002048 to K.Z. and YD9100002044 to S.L.).
Author information
Authors and Affiliations
Contributions
Y.Z., K.Z. and Y.F. conceived and supervised the project and designed experiments. M.L., Xingyu Z., D.L., W.X., Z.G., L.H. and L.A. purified the proteins and performed in vitro activity analysis and in vivo assays. M.L., Xiaolong Z., Y.G. and K.Z. collected the cryo-EM data and solved the cryo-EM structures. S.L. built and refined the models. Y.Z. wrote the original manuscript. Y.F., K.Z. and Y.Z. revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Type I and type II Lamassu systems both form heterooligomers.
a–c, Purification profile of BcLmuA (a), BcLmuB (b) and BcLmuAB complex (c) on Superdex 200 column. d, SEC-MALS (Size Exclusion Chromatography with Multi-Angle Light Scattering) analysis of BcLmuB, BcLmuAB and VcLmuACB complex. Respective calculated molecular weight is marked on top of the peak. e, Statistics of the SEC-MALS results and theoretical molecular weight based on predicted complex ratios. f, g, Purification profile of VcLmuA (f) and VcLmuC (g) on Superdex-200 column. h, Purification test of His-SUMO-TEV-VcLmuB and SDS-PAGE gel of the samples. 1-bacteria pre-induced. 2-bacteria post-induced. 3-supernatant. 4-pellet. 5-flowthrough after Ni-column. 6-beads after wash. 7-Elution. 8-beads after elution. i, Purification profile of VcLmuACB complex on Superdex 200 column.
Extended Data Fig. 2 DNA binding, ATPase and nuclease activity of the Lamassu system.
a, EMSA to test the binding of BcLmuAK59AB and VcLmuAK57GCB to dsDNA. BcLmuAK59AB and VcLmuAK57GCB is included at a concentration of 0.25, 0.5, 1, 2, 4, 8 μM, respectively. b, pUC19 digestion by VcLmuACB in the presence or absence of ATP or ATPγS, respectively. c, Measurement of ATP or ATPγS hydrolysis activity for VcLmuACB and BcLmuAB at 0.5 μM final with or without 0.5 μM 59 bp DNA duplex, 0.25 mM ATP or ATPγS. Each group with three replicate measurements. Means and standard deviations are shown.
Extended Data Fig. 3 Single-particle cryo-EM analysis of the BcLmuAB complex.
a, Representative cryo-EM micrograph (upper) and reference-free 2D class averages (lower) of BcLmuAB. b, Workflow of the cryo-EM data processing. The final map resolution is color coded for different regions. c, Gold standard FSC plots for the 3D reconstructions of the whole map, calculated in cryoSPARC. d, Euler angle distribution of the particle images.
Extended Data Fig. 4 Single-particle cryo-EM data processing of VcLmuACB.
a, Representative cryo-EM micrograph of VcLmuACB. b, Representative cryoEM 2D averages of VcLmuACB. c, Cryo-EM data processing workflow for VcLmuACB. d, Gold standard FSC plot for the final 3D reconstruction of the whole map, calculated in cryoSPARC. e, Euler angle distribution of the particle images. f, The final map resolution is colored for different regions.
Extended Data Fig. 5 Single-particle cryo-EM data processing of VcLmuACB-pUC19.
a, Representative cryo-EM micrograph of VcLmuACB-pUC19. b, Reference-free 2D class averages of VcLmuACB-pUC19 c, Cryo-EM data processing workflow for VcLmuACB-pUC19. d, Gold standard FSC plot for the final 3D reconstruction. e, Euler angle distribution of the particle images. f, Resolution distribution (ResMap).
Extended Data Fig. 6 Structural alignment between different states of BcLmuAB and VcLmuACB complexes.
a, Structural alignment between the two apo state structures of VcLmuACB with different protein lengths. b–d, Structural alignment between the apo and DNA-bound states of BcLmuAB (b), between the apo (partial structure) and DNA-bound states of VcLmuACB (c), and between the apo (full length) and DNA-bound states of VcLmuACB (d).
Extended Data Fig. 7 Inter-subunit interactions in the apo state of VcLmuACB (full length).
a–e, Atomic model of the VcLmuACB complex is shown in a, in which detailed interactions between dsDNA and BcLmuAB are shown in b–e.
Extended Data Fig. 8 Single-particle cryo-EM analysis of the BcLmuAB-DNA complex.
a, Representative cryo-EM micrograph (left) and reference-free 2D class averages (right) of BcLmuAB-DNA. b, Workflow of the cryo-EM data processing. The final map resolution is color coded for different regions. c, Gold standard FSC plots for the 3D reconstructions of the whole map, calculated in cryoSPARC (left) and Euler angle distribution of the particle image(right).
Extended Data Fig. 9 Single-particle cryo-EM data processing of VcLmuACB-59bp DNA complex and VcLmuA-tetramer complex.
a, Representative cryo-EM micrograph of VcLmuACB-59bp DNA. b, Workflow of cryo-EM data processing of VcLmuACB-59bp DNA. c, Reference-free 2D class average of VcLmuACB-DNA. d, Reference-free 2D class average of VcLmuA-tetramer. e, Gold standard FSC plots for the 3D reconstructions of the whole map of VcLmuACB-DNA, calculated in cryoSPARC. f, Gold standard FSC plots for the 3D reconstructions of the whole map of VcLmuA-tetramer, calculated in cryoSPARC. g, Euler angle distribution of the particle image of VcLmuACB-DNA. h, Euler angle distribution of the particle image of VcLmuA-tetramer. i, The final map resolution is color coded for different regions of VcLmuACB-DNA. j, The final map resolution is color coded for different regions of VcLmuA-tetramer.
Extended Data Fig. 10 Enzymatic cleavage activity towards five types of DNA substrates.
a, Schematic representation and sequences of the five DNA structures. b–f, Time-course cleavage of double-stranded (b), single-stranded (c), palindromic DNA (d), circular DNA (pUC19) (e) and Linear phage DNA (extracted from phage T1) (f) by two Lamassu systems. The reaction contains 80 nM VcLmuACB or 20 nM BcLmuAB and 1 μM DNA substrates (b–d), 22 nM pUC19 or 1.2 nM T1 phage genomic DNA.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1-2 and Figs. 1–3.
Supplementary Data 1 (download XLSX )
DNA and primer sequences used in this study.
Source data
Source Data Fig. 1 (download PDF )
Unprocessed gels and photos.
Source Data Fig. 3 (download PDF )
Unprocessed gels and photos.
Source Data Fig. 4 (download PDF )
Unprocessed gels and photos.
Source Data Fig. 5 (download PDF )
Unprocessed gels and photos.
Source Data Extended Data Fig. 1 (download PDF )
Unprocessed gels and photos.
Source Data Extended Data Fig. 2 (download PDF )
Unprocessed gels and photos.
Source Data Extended Data Fig. 10 (download PDF )
Unprocessed gels and photos.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, M., Zhao, X., Zhao, X. et al. Structural insights into type-I and type-II Lamassu antiphage systems. Nat Chem Biol (2026). https://doi.org/10.1038/s41589-025-02102-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41589-025-02102-z