Abstract
Habituation is a crucial sensory filtering mechanism whose dysregulation can lead to a continuously intense world in disorders with hypersensitivity. Although habituation is often termed the simplest form of learning, its circuit mechanisms remain elusive. Conventional peripheral explanations fail to fully account for long-term habituation in complex sensory environments, leading to theories proposing top-down regulation. Here we evaluated two competing top-down mechanistic explanations for habituation in mice (growth in predictive filtering and waning of novelty-driven amplification) and identified an unexpected role for the orbitofrontal cortex (OFC) in predictive filtering. After daily sound exposure, neural habituation in the primary auditory cortex (A1) was reversed by inactivating the OFC. Top-down projections from the OFC, but not other frontal areas, carried predictive signals that grew with daily sound experience and suppressed A1 via somatostatin-expressing inhibitory neurons. Thus, prediction signals from the OFC cancel out anticipated stimuli by generating their ‘negative images’ in sensory cortices.
Similar content being viewed by others
Main
Throughout our lives, we are constantly exposed to a flood of sensory information from the external world; however, only a small fraction of this information reaches our conscious perception and triggers behavioral responses. This remarkable filtering, which prioritizes inputs with expected meaningful outcomes while ignoring others, arises from our brain’s ability to build internal models of the world1,2. These models are continuously trained through our lifetime experiences and dictate predictive relationships between sensory objects in the world. The inability to build such predictive models, and thus to apply appropriate sensory filters, is a hallmark of mental disorders such as obsessive-compulsive disorder3, schizophrenia4, and most notably, autism spectrum disorders (ASDs)5,6.
One fundamental form of such sensory filtering is habituation, the brain’s ability to ignore familiar, inconsequential stimuli7. Habituation operates through multitudes of mechanisms, allowing animals to adapt to the statistical structures of their sensory environments over a wide range of timescales. While previous research primarily focused on mechanisms for short-term plasticity lasting from milliseconds to seconds, known as ‘stimulus-specific adaptation’8,9,10,11,12, habituation also extends to longer periods (days or even weeks)13,14,15 as evidenced by the gradual reduction in our sensitivity to perfumes or traffic noise. This long-term habituation is particularly relevant for understanding the pathology of filtering deficits, as a lack of accumulated habituation to daily sensory objects throughout one’s life could result in a persistently intense sensory world16,17.
Contrary to the widely held belief that habituation is the ‘simplest form of learning,’ accumulating evidence has challenged this perceived simplicity. Classical internal model-free mechanisms, such as sensory receptor fatigue, homosynaptic depression, and the short-term dynamics of inhibitory neuron firing, fail to fully account for several key characteristics of habituation. These characteristics include its persistence over weeks, capacity to process complex stimuli involving multiple sensory channels18, context-dependence19,20,21 and disruption by anesthesia15,22. These observations have prompted theories suggesting that habituation is associative and governed by top-down regulation informed by internal models of the external world16,22,23,24,25.
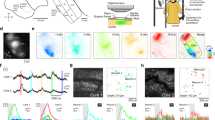
Two competing hypotheses have been proposed to explain the top-down circuit mechanisms of long-term sensory habituation. The predictive negative image hypothesis, aligned closely with the general ‘predictive processing’ framework24, posits that initially strong sensory responses are gradually canceled as the top-down predictive signal grows (Fig. 1a, left and Extended Data Fig. 1a,b). Supporting this hypothesis, somatostatin-expressing inhibitory neurons (SST cells) in sensory cortices increase their sensory responses over days of stimulus exposure26,27,28, providing neuronal substrates for the generation of a ‘negative image’ of the stimulus. An alternative is the novelty hypothesis, which assumes that bottom-up sensory inputs are weak but are initially amplified by a top-down novelty signal upon the animal’s first encounter with a stimulus (Fig. 1a, right and Extended Data Fig. 1b)29,30,31. This view proposes that the activation of vasoactive intestinal peptide-expressing inhibitory neurons (VIP cells) by the novelty signal disinhibits pyramidal neurons, and habituation occurs as the novelty signal wanes over time.
a, Schematics illustrating two theories explaining sensory habituation by top-down predictive mechanisms. b, Representative two-photon image of A1 L2/3 neurons expressing GCaMP6s. c, Schematic illustrating the 5-day auditory habituation paradigm. d, Heatmaps of sound-evoked responses in neurons imaged across 5 days in a representative mouse. Neurons are sorted by their responses on day 1. e, Average (solid line) and s.e.m. (shading) of the Change index across responsive neurons for excitatory (red) and inhibitory (blue) sound-evoked responses over days. n = 879 responsive neurons from 20 mice. f, Change index of excitatory responses in individual neurons on day 5 compared to day 1 across all mice. Black line on the right represents mean. n = 879 responsive neurons. ****P = 1.8 × 10−91 (two-sided Wilcoxon signed-rank test). g, Change in sound-evoked response traces from day 1 to day 5 averaged across all significantly responsive cells (left). Black bar, 7-s sound. The amplitudes of the difference trace at 1 and 7 s after sound onset (right). ****P = 1.1 × 10−67 (two-sided Wilcoxon signed-rank test). Box, 25th to 75th percentiles; whiskers, 1 × interquartile range (IQR) (95.7% of data); black line, median; red line, mean; outliers not shown. h, Histograms of changes in response magnitudes in all neurons. Orange and green bars show neurons with significant increase and decrease. ****P = 4.3 × 10−138 (two-sided Fisher’s exact test). i, Schematic illustrating vectors representing within-day habituation (purple, Modewithin) and across-day habituation (green, Modeacross) within a high-dimensional space. Each dimension corresponds to the response magnitudes of individual neurons. j, Projection of trial-to-trial sound-evoked A1 ensemble activity dynamics onto the Modewithin (left) and Modeacross (right) vectors. Data points represent individual trials (100 trials × 5 days). Ensemble activity patterns repeated fast daily plasticity along the Modewithin axis, whereas they show continuous slow plasticity along the Modeacross axis across 5 days. n = 20 mice, 2,398 cells imaged throughout the 5 days. k, Cosine similarity between Modewithin and Modeacross, indicating a nearly orthogonal relationship. l, Left, change in sound-evoked ensemble activity along Modewithin from trial 1 to trial 10, demonstrating prominent within-day, across-trial habituation around the tone onset. Right: Change in sound-evoked ensemble activity along Modeacross from day 1 to day 5. Across-day habituation slowly ramps up during the sustained tone. m, Summary data showing the Ramp-up Index for across-day plasticity (day 1 to day 5) and within-day plasticity (trial 1 to trial 10). n, Top left, schematic illustrating pupil monitoring during two-photon calcium imaging. Top right: Representative image from a pupil camera. Bottom right, representative pupil diameter dynamics during sound presentations. Black lines indicate tone timings. o, Change in normalized pupil diameter across days. n = 17 mice. p, Two alternative models illustrating the dependence of neuronal sound responses on normalized pupil diameter. Left, a model in which neuronal habituation depends on the decrease in arousal level. Right, a model in which neuronal habituation is independent of arousal level. Individual dots represent trials; black, day 1 trials; red, day 5 trials. Lines indicate regression lines. q, Experimental data showing neuronal response magnitudes binned by normalized pupil diameter separately for day 1 (black) and day 5 (red). Day 5 neuronal responses are smaller than day 1 responses regardless of normalized pupil diameter. n = 10 mice, 977 cells. Day 1 versus day 5, ****P = 2.0 × 10−30 (unbalanced two-way analysis of variance). Error bars and shading represent mean ± s.e.m.
Despite their fundamentally distinct mechanisms, activity measurements in sensory cortices alone have failed to differentiate between these hypotheses. Due to the reciprocal connections between SST and VIP cells, both models predict similar neuronal dynamics during habituation: an increase in SST cell activity and a decrease in VIP and pyramidal cell activity. To distinguish between these theories, we sought to identify the source of the top-down input to A1 during daily sensory habituation and to determine how sensory responses are affected when the top-down signal is removed.
Results
Distinct mechanisms underlie within-day and across-day habituation in A1
We induced sensory habituation in awake, head-fixed mice through repeated exposure to tones over 5 days (5–9 s pure tones, 7–16-s inter-trial interval, 70 dB sound pressure level (SPL), 200 trials per day) (Fig. 1b,c). To track the activity of the same A1 layer 2/3 (L2/3) neural ensembles across days, we conducted chronic two-photon calcium imaging. We first located A1 by intrinsic signal imaging32 and virally expressed GCaMP6s in the A1 of wild-type or VGAT-Cre × Ai9 mice. Three weeks after virus injection and optical window implantation, we recorded sound-evoked activities from 5,483 L2/3 neurons in 20 mice. We chose a fixed tone frequency for each mouse and imaged the corresponding tonotopic domain in A1, where we tracked the same neural ensembles throughout the experiment. Across days of repeated sound exposure, we observed clear habituation in tone responses, characterized by reduced excitation and increased suppression (Fig. 1d), consistent with previous studies26,27. We quantified the time course of the plasticity using a change index ((day X – day 1)/(day X + day 1); ranging from −1 to 1, with 0 indicating no change) separately for excitation and inhibition (Methods). This analysis revealed a gradual decrease in excitatory responses and a concurrent increase in inhibitory responses across days (Fig. 1e,f). The change in ΔF/F traces averaged across all excited neurons showed that the reduction in sound responses from day 1 to day 5 was more pronounced during the sustained portion of the sound presentation than at its onset (Fig. 1g). On day 5, 35.8% of neurons showed a significant reduction in sound responses while only 13.2% showed a significant increase (Fig. 1h and Extended Data Fig. 2a–d). There was a slight tendency for habituation effects to be stronger in superficial compared to deep L2/3 neurons; however, robust habituation was observed across all cortical depths (Extended Data Fig. 2e,f). Given comparable results between experiments imaging all L2/3 neurons and those imaging only VGAT− pyramidal neurons, data from both experiments were combined and treated as A1 pyramidal cells (Extended Data Fig. 2g–i).
We observed two timescales of habituation: within-day habituation, which repeated daily across trials, and across-day habituation, which built up over multiple days (Fig. 1i,j). To better understand the relationship between these plasticity timescales, we projected high-dimensional population dynamics onto the axes defined by within-day and across-day changes. Notably, these two habituation mechanisms represented nearly orthogonal axes in the high-dimensional space (78.2 degrees; Fig. 1k), indicating that across-day habituation is not simply the accumulated effect of within-day habituation. Within-day habituation markedly attenuated sound onset responses, whereas across-day habituation was greater during the sustained phase of tones (5 s after onset), implying two independent mechanisms for auditory habituation (Fig. 1g,l,m and Supplementary Discussion). This difference in kinetics supports the role of slow, top-down mechanisms in across-day habituation, while within-day habituation may be driven by faster, bottom-up mechanisms.
To assess the contribution of global neuromodulation to sensory habituation, we monitored pupil diameter during calcium imaging in a subset of experiments. Pupil diameter showed a gradual decrease across days, providing evidence of habituation in arousal responses (Fig. 1n,o); however, plots of trial-to-trial variability of neural activity against pupil diameter showed a distinct separation between day 1 and day 5 data regardless of pupil diameter, demonstrating that across-day habituation is independent of arousal levels (Fig. 1p,q). Additionally, we previously found that across-day habituation under these conditions is not inherited from subcortical pathways, as the plasticity was evident in L2/3 but not in the input layer, L4 (ref. 26). Therefore, we focused on across-day habituation for the rest of our analyses to determine its cortical circuit mechanisms.
Both the predictive negative image hypothesis and the novelty hypothesis involve the active recruitment of inhibitory neurons during habituation. To test whether these top-down mechanisms truly contribute to long-term habituation, it is essential to characterize inhibitory neural circuit dynamics before and after habituation. As VIP cell activity had not been previously monitored in A1 during across-day habituation, we performed cell type-selective calcium imaging of three inhibitory neuron subclasses (SST cells, VIP cells and parvalbumin-expressing neurons (PV cells)) during 5 days of repeated tone exposure. We targeted GCaMP6s to each cell type by injecting a conditional GCaMP6s-expressing virus in Cre transgenic mice. Throughout the 5-day sound exposure, L2/3 VIP and PV cells exhibited neural habituation similar to pyramidal cells (Extended Data Fig. 3). Notably, SST cells showed clear heterogeneity33; deep (>200 μm from the surface) SST neurons showed mild habituation, whereas superficial (<200 μm) neurons exhibited the opposite pattern26,27, suggesting the involvement of superficial SST cells in sensory habituation. The observed increase in superficial SST cell activity and reduced VIP cell activity align with inhibitory circuit dynamics proposed by both the predictive negative image and novelty hypotheses; however, these results do not conclusively support one hypothesis over the other, prompting further investigation into the modulation source for A1 inhibitory neurons.
OFC inactivation reverses sensory habituation in A1
We therefore sought to identify the source of top-down input regulating A1 neuronal activity (Fig. 2a). First, we performed anatomical tracing. After mapping auditory cortices with intrinsic signal imaging, we injected a retrograde tracer into A1 L2/3 (Fig. 2b). Consistent with previous studies, we found many presynaptic neurons in the OFC34,35,36, secondary motor cortex (M2)37 and anterior cingulate cortex (ACC)38 (Fig. 2c,d), with the ventrolateral and lateral OFC (hereafter, jointly called OFCvl) showing the densest inputs. The lack of overlap between the tracer and VGAT expression indicated that the top-down projections from OFCvl were glutamatergic (Extended Data Fig. 4a–d). Cell type-selective retrograde tracing with rabies virus further confirmed that both SST and VIP cells receive extensive top-down inputs from OFCvl (Fig. 2e–h and Extended Data Fig. 5; also Discussion). In contrast, tracer signals in adjacent areas, such as the frontal pole (FRP), prelimbic area (PL) and infralimbic area (IL), were weaker in both AAV and rabies tracing, indicating highly specific wiring of auditory circuits in the frontal cortex.
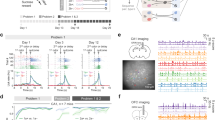
a, Schematics illustrating two theories explaining how to distinguish top-down predictive mechanisms from novelty mechanisms. b, Intrinsic signal imaging of pure tone responses superimposed on the cortical surface imaged through the skull (top). Yellow cross indicates the injection site. Schematic illustrating retrograde tracing from A1 L2/3 (bottom). c, tdTomato-expressing input neurons onto A1 L2/3. Coronal section of the frontal areas overlaid with area borders (left). Magnified images of OFC, ACC and M2 areas (right). d, Bar plots summarizing the distribution of input cells in ipsilateral frontal cortical areas. M1, primary motor cortex; CL, claustrum; DP, dorsal peduncular area. Inset, further classification of OFC into ventrolateral (OFCvl), lateral (OFCl) and medial (OFCm) subdivisions. n = 4 mice. e–g, Monosynaptic rabies retrograde tracing from A1 SST neurons. e, Coronal section of the injection site in A1 (left). Dotted lines show cortical borders. Magnified view of the area indicated by a square in the left image (right). f, Coronal section of the frontal areas overlaid with area borders. White dots show presynaptic neurons. g, Bar plots summarizing the distribution of input cells in ipsilateral frontal cortical areas. n = 4 mice. Inset shows further classification of OFC into OFCvl, OFCl and OFCm subdivisions. h, Same as g but for VIP-Cre mice. i, Schematic illustrating muscimol infusion (top). 4,6-diamidino-2-phenylindole (DAPI)-counterstained coronal section showing the cannula implantation site in OFC (bottom). j, Protocol for muscimol infusion after habituation. k, Trial-averaged sound-evoked response traces from three cells (top left). Scale, 100%, 1 s. Change index across days (bottom left). Pink shade shows muscimol infusion. n = 7 mice, 1,392 neurons. Change index of excitatory responses in individual neurons on day 6 compared to day 5 across all mice (right). n = 296 significantly excited neurons. ****P = 2.0 × 10−13 (two-sided Wilcoxon signed-rank test). l, Change in sound-evoked response traces from day 5 to day 6, averaged across all significantly excited cells (left). Bar, 7-s sound. The amplitudes of the difference trace at 1 and 7 s after sound onset (right). n = 1,288 neurons. ****P = 9.0 × 10−5 (two-sided Wilcoxon signed-rank test). Box shows 25th to 75th percentiles; whiskers show 1 × interquartile range (IQR); black line, median; red line, mean; outliers not shown. m, Histograms of changes in response magnitudes in all neurons. ****P = 5.7 × 10−25 (two-sided Fisher’s exact test). n–p, The same as k–m, but for control PBS infusion experiments. n = 3 mice, 482 total neurons, 49 significantly excited neurons. Scale, 100%. P = 0.76 (n); P = 0.089 (o); P = 0.19 (p). q, Schematic illustrating muscimol infusion in naive mice. r, Change index for excitatory responses across blocks of trials. Each data point represents a block of 25 trials. Red, muscimol (n = 4 mice); dark gray, PBS control (n = 6 mice). s, Change index of excitatory responses in individual neurons in blocks 6–8 compared to blocks 2–4 across all mice. n = 308 and 256 significantly excited neurons for PBS and muscimol. PBS, P = 0.15 (two-sided Wilcoxon signed-rank test); muscimol, P = 0.82; PBS versus muscimol, P = 0.49 (two-sided Wilcoxon rank-sum test). Error bars and shading represent mean ± s.e.m. NS, not significant.
We next asked whether the frontal top-down input causally contributes to A1 sensory habituation. Two hypotheses predict distinct outcomes with the removal of top-down input (Fig. 2a, red dotted arrows). According to the predictive negative image model, removing the predictive signal should increase sensory responses following habituation but have minimal impact on naive animals. Conversely, the novelty model suggests that eliminating the novelty signal would diminish sensory responses in naive animals but have little effect after habituation. To test these predictions, we targeted the OFCvl for pharmacological inactivation, given its strongest top-down connection to A1. After 5 days of tone habituation, we inactivated OFCvl on day 6 by infusing muscimol through a pre-implanted cannula (2.5 mg ml−1, 500 nl, 100 nl min−1) (Fig. 2i,j). Notably, OFCvl inactivation reversed the sensory habituation in A1 pyramidal cells by enhancing excitatory responses and reducing suppression responses, an effect not observed with the control phosphate-buffered saline (PBS) infusion (Fig. 2k–p and Extended Data Fig. 6) (change index from day 5 to day 6, muscimol: 0.31 ± 0.04, P = 2.0 × 10−13; control, 0.034 ± 0.094, P = 0.76). In a separate experiment, we evaluated the effects of OFCvl inactivation in naive animals by infusing muscimol on day 1 (Fig. 2q). Unlike the post-habituation inactivation, OFCvl inactivation before habituation did not alter tone responses (Fig. 2r,s) (change index before and after infusion, muscimol, −0.002 ± 0.017, P = 0.82; control, −0.037 ± 0.015, P = 0.15). The absence of any significant change in A1 responses upon muscimol infusion in naive animals rules out the role of OFCvl in transmitting novelty signals or modulating general sensory processing in A1. Instead, our findings demonstrate the causal role of the OFC specifically in the expression of long-term sensory habituation, aligning with the predictive negative image hypothesis.
Given the involvement of specific inhibitory neuron subtypes in both hypotheses, we further assessed whether inhibitory neurons follow the predictions of these models. We repeated the same 6-day OFCvl inactivation experiment while performing cell type-selective imaging of SST, VIP and PV cells (Fig. 3a). Upon OFCvl inactivation in habituated mice, sound responses in SST cells were significantly reduced (change index from day 5 to day 6, −0.50 ± 0.06, P = 1.1 × 10−11), as expected from the predictive negative image hypothesis (Fig. 3b–e). The reduction in sound responses was significantly greater in superficial SST cells compared to deeper SST cells, mirroring the depth dependence of enhancement observed during habituation (Extended Data Fig. 3m,n). In contrast, both VIP and PV cells showed a marked enhancement in their sound responses, consistent with the release from SST cell-mediated inhibition (PV, 0.18 ± 0.06, P = 0.036; VIP, 0.55 ± 0.10, P = 5.2 × 10−5) (Fig. 3f–m and Extended Data Fig. 6i). These findings further support that the predictive signals from the OFC activate SST cells to suppress other A1 cell types after habituation.
a, Schematic illustrating muscimol infusion after habituation (left). Circuit diagram of inhibitory neuron connectivity in A1 (right). b, Heatmaps of sound-evoked responses in SST neurons imaged across days 5 and 6 in a representative mouse. Neurons are sorted by their responses on day 5. c, Change index of excitatory responses in individual SST neurons on day 6 compared to day 5 across all mice. The line on the right represents the mean. n = 8 mice, 115 excited cells. ****P = 1.1 × 10−11 (two-sided Wilcoxon signed-rank test). d, Change in sound-evoked response traces from day 5 to day 6 averaged across all significantly excited SST cells (left). Solid line shows the average. Shading shows s.e.m. Bar, 7-s sound. The amplitudes of the difference trace at 1 and 7 s after sound onset (right). ****P = 1.4 × 10−11 (two-sided Wilcoxon signed-rank test). Box shows 25th to 75th percentiles; whiskers show 1 × IQR; black line shows median; red line shows mean; outliers not shown. e, Histograms of changes in response magnitudes in all SST neurons. Orange and green bars show neurons with significant increase and decrease. ****P = 3.5 × 10−8 (two-sided Fisher’s exact test). f–i, Same as b–e but for VIP neurons. n = 4 mice, 26 excited cells. ****P = 5.2 × 10−5 (g). ****P = 6.0 × 10−26 (h). ****P = 1.4 × 10−35 (i). j–m, Same as b–e but for PV neurons. n = 5 mice, 90 excited cells. *P = 0.036 (k). P = 0.17 (l). ****P = 1.6 × 10−12 (m). Error bars and shades represent the mean ± s.e.m.
Of note, while the effects of both sensory habituation (Fig. 1g,l,m) and OFCvl inactivation (Fig. 2l, Fig. 3d,h,l and Extended Data Fig. 6e, f) were evident at sound onset (0–1 s from the tone start), they were significantly more pronounced during the sustained sound stimulus. This slow kinetics of sensory habituation aligns with the recruitment of a long-distance top-down feedback loop and supports the role of prediction, as the sustained phase of the sound is more predictable than the onset.
OFC top-down projection conveys sound-specific predictive signals and suppresses A1
The OFC plays critical roles in various cognitive functions and has extensive connections with multiple brain regions, including the mediodorsal thalamus, striatum, amygdala, ACC, insular cortex and sensory cortical areas39. We next investigated whether the direct projection from OFCvl to A1 (Extended Data Fig. 4e–h) conveys predictive signals during sensory habituation (Fig. 4a).
a, Schematic illustrating direct and indirect pathways from OFC to A1. b, Schematic illustrating projection-specific calcium imaging of the OFC → A1 direct pathway (left). Representative two-photon image of a GCaMP6s-expressing OFC axon in A1 on day 1 and day 5 (right). c, Trial-averaged sound-evoked response traces from two axon boutons. Black, day 1; red, day 5. Scale bar, 50%. Dotted lines indicate sound onset and offset. d, Fraction of boutons with significant excitatory responses on day 1 and day 5 across mice. n = 12 mice, 595 boutons. *P = 0.035 (two-sided Wilcoxon signed-rank test). e, Averaged change index for excitatory responses across days. f, Change index of excitatory responses on day 5 compared to day 1. n = 32 excited boutons. ****P = 6.5 × 10−5 (two-sided Wilcoxon signed-rank test). g, Change in sound-evoked response traces from day 1 to day 5 averaged across boutons. All significantly excited boutons (top); all imaged boutons (bottom). Black bar, sound; solid line, average; shading, s.e.m. h, The amplitudes of the difference trace at 1 and 7 s after sound onset. *P = 0.029, **P = 0.0057 (two-sided Wilcoxon signed-rank test) (top). *P = 0.031, ***P = 2.9 × 10−4 (bottom). Box, 25th to 75th percentiles; whiskers, 1 × IQR; black line, median; red line, mean; outliers not shown. i, Histograms of changes in response magnitudes in all boutons. Orange and green bars show neurons with significant increase and decrease. **P = 0.0034 (two-sided Fisher’s exact test). j, Schematic illustrating single-unit recordings in M2 and OFC of naive and habituated mice (left). Coronal section showing the Neuropixels probe track (indicated by DiI) targeting M2 and OFC (center). The section was counterstained with DAPI and overlaid with area borders. Summary of all penetrations aligned to the Allen Common Coordinate Framework (right). Probe tracks within M2 and OFC are shown in green and red, respectively. n = 20 penetrations in 17 mice. k, Fraction of single units in OFCvl (left) and M2 (right) with significant excitatory responses in naive and habituated mice. OFCvl naive, n = 5 mice; habituated, n = 8 mice, *P = 0.045. M2 naive, n = 7 mice; habituated, n = 10 mice, P = 0.83 (two-sided Wilcoxon rank-sum test). l, Sound-evoked response traces averaged across all significantly excited single units in OFCvl (top) and M2 (bottom) of naive (black) and habituated (blue) mice (left). Bar, 7-s sound. Scale bar, 0.5 Hz. Cumulative plots of sound-evoked spike counts in all significantly excited single units for OFCvl (top right) and M2 (bottom right). OFCvl, naive, n = 38 units; habituated, n = 99 units, **P = 0.010 (two-sided Wilcoxon rank-sum test). M2, naive, n = 32 units; habituated, n = 66 units, P = 0.55. m, Schematic illustrating the intersectional strategy to express ChRmine selectively in A1-targeting OFC neurons (left). Coronal section showing the expression of ChRmine-oScarlet (right). n, Raster (top) and peristimulus time histogram (bottom) of a representative single unit in A1. o, modulation index of A1 L2/3 neural activity by transcranial photostimulation of OFC with a red laser. ChRmine: n = 6 mice, P = 0.012 (two-sided Wilcoxon signed-rank test). No opsin control, n = 3 mice, P = 0.67; ChRmine versus no opsin, P = 0.034 (two-sided Wilcoxon rank-sum test). Error bars and shades represent mean ± s.e.m.
We visualized sound-evoked activity in the OFCvl→A1 direct pathway by virally expressing axonGCaMP6s in OFCvl neurons. We performed two-photon calcium imaging of their axon terminals in L1 and superficial L2/3 of A1 to track the activity from the same axon boutons over a five-day habituation period (Fig. 4b). On day 1, significant tone-evoked excitation was observed in 11.0% of axon boutons, consistent with the sparse sound responses previously reported in OFC of naive mice34,40 (Fig. 4c,d). The sound responses are likely conveyed through the anatomical projections from the auditory cortex to the OFCvl (Extended Data Fig. 4i–o); however, following habituation, the proportion of excited boutons increased to 15.9%. In axon boutons identified across days, we observed a gradual increase in response magnitudes (Fig. 4c–f). This increase in OFCvl→A1 axonal activity after habituation was more pronounced during the sustained phase of tones (Fig. 4g, h), consistent with the slow kinetics of habituation in A1 neurons (Fig. 1g and Fig. 3d,h,l). On day 5, 17.7% of boutons showed a significant potentiation in sound responses while only 8.2% showed a significant reduction (P = 0.0034, Fisher’s exact test) (Fig. 4i). These results support the hypothesis that the OFC gradually forms an internal model of repeated stimuli, and its direct projection to A1 carries predictive signals that grow over days to recruit SST inhibitory neurons.
Next, we examined whether the increase in the representation of habituated sounds is specific to the OFCvl. Previous research found predictive filtering of A1 responses to self-generated sounds by M2 → A1 top-down input37. Thus, predictive filtering of sensory responses may be a general function shared equally across frontal cortical areas. Alternatively, distinct frontal regions might apply unique filters on A1 activity, tailored to different types of predictions: OFC for predictions based on a history of experiences and M2 for predictions related to motor corollary discharges.
To investigate these possibilities, we directly compared tone-evoked activities between OFCvl and M2 neurons within the same animals. We performed single-unit recordings using a Neuropixels 1.0 probe that penetrated both regions (Fig. 4j). When we compared tone responses between naive and habituated mice, we found a 1.7-fold larger fraction of significantly excited OFCvl single units in habituated mice (habituated, 8.2%; naive, 4.7%, P = 0.045), whereas M2 single units showed no difference (habituated, 6.1%; naive, 5.8%, P = 0.83) (Fig. 4k). Sound-evoked response magnitudes averaged across all excited single units showed a significant increase after habituation in OFCvl (104% increase; P = 0.010; Fig. 4l). In contrast, M2 units showed a tendency for reduced activity, likely reflecting a weaker input from the habituated auditory cortex (47% reduction; P = 0.55). Recordings in other frontal regions also showed no signs of increased tone responses (Extended Data Fig. 7a–f). This OFCvl-specific plasticity is consistent with the OFCvl→A1 axonal imaging data and supports a division of labor between OFC and M2 in the predictive filtering of A1 activity across different contexts.
To determine whether activation of the OFCvl→A1 pathway is sufficient to suppress A1 neuronal activity, we next performed pathway-selective optogenetic activation. We injected AAVretro-syn-Cre in the A1 identified by intrinsic signal imaging, and AAV8-nEF-Con/Foff-ChRmine-oScarlet in the OFCvl. This intersectional viral approach restricted the expression of red-shifted opsin ChRmine specifically to A1-projecting OFCvl neurons (Fig. 4m). Transcranial illumination of the frontal cortex with a red laser successfully evoked action potentials in ChRmine-expressing OFCvl neurons (Extended Data Fig. 7g,h)41. A1 recording during activation of OFCvl→A1 pathway revealed significant suppression of A1 activity, an effect absent in control mice without opsin expression (modulation index, ChRmine, −0.23 ± 0.06, P = 0.012; control, 0.05 ± 0.07, P = 0.668; ChRmine versus control: P = 0.034) (Fig. 4n,o and Supplementary Discussion). Therefore, the increased activity in the OFCvl→A1 top-down pathway observed following repeated tone exposure is sufficient to cause neuronal habituation in A1.
We next asked whether the OFCvl→A1 top-down pathway conveys signals specifically predictive of familiar stimuli, or instead sends generalized signals that broadly suppress auditory cortical responses (Fig. 5a). To differentiate between these possibilities, we imaged axonal boutons of the OFCvl→A1 pathway and assessed their responses to two sound frequencies (tone A and tone B, selected to flank the population best frequency of the imaged area). Tone A was presented daily to induce habituation, whereas tone B was presented only on the first and last days, serving as a control (Fig. 5b). To minimize habituation to tone B, presentations before (‘day 0’) and after habituation (‘day 6’) were limited to ten trials per sound. On day 6, OFCvl axonal boutons showed enhanced excitatory responses specifically to tone A (Fig. 5c–g). In contrast, tone B responses on day 6 remained similar to pre-exposure levels, demonstrating the specificity of top-down signals conveyed through this pathway (change index: tone A, 0.55 ± 0.14, P = 0.0042; tone B, 0.06 ± 0.17, P = 0.71; two-sided Wilcoxon signed-rank test).
a, Schematic illustrating two hypotheses for how prediction circuits respond to a novel sound (tone B) following habituation to tone A. b, Protocol for assessing sound specificity of habituation. c, Trial-averaged response traces from two axon boutons to tone A and tone B, before (blue) and after (red) tone A habituation. Scale, 20%, 1 s. d, Averaged tone-evoked response traces from all imaged boutons before and after tone A habituation (top). Averaged changes in response traces across all boutons (n = 11 mice, 161 boutons) (bottom). Due to variability in tone durations and the limited number of trials, only the first 5 s common to all trials are shown. Solid line shows mean; shading shows s.e.m. e, Amplitudes of the difference traces at 1 and 5 s after sound onset for tone A (left) and tone B (right) responses. Two-sided Wilcoxon signed-rank test. Box, 25th to 75th percentiles; whiskers, 1 × IQR; black line, median; red line, mean; outliers not shown. n = 161 boutons. f, Change index of excitatory responses for individual boutons on day 6 compared to day 0. Black lines on the right represent mean. n = 22 (tone A) and 26 (tone B) significantly excited boutons. **P = 0.0042 (two-sided Wilcoxon signed-rank test). g, Histograms showing changes in response magnitudes across all boutons. Orange and green bars show boutons with significant potentiation and suppression for tone A (top) and tone B (bottom). n = 161 boutons, **P = 0.0011 (two-sided Fisher’s exact test). h–l, Same as c–g but for A1 neurons. n = 4 mice, 551 neurons (i). ****P = 1.5 × 10−12; ***P = 5.3 × 10−4 (j). Tone A, n = 160 excited cells, ****P = 8.8 × 10−22; tone B, n = 196 (k). n = 551 neurons, ****P = 1.6 × 10−21 (l). Error bars and shades represent mean ± s.e.m.
This observed sound-specific increase in OFCvl→A1 activity, combined with its causal role in suppressing A1 neuronal activity (Fig. 4m–o), suggests that A1 L2/3 neurons should also show sound-specific habituation. To test this hypothesis, we repeated the two-tone frequency experiment while imaging A1 L2/3 neurons. On day 6, A1 neurons showed significantly reduced excitatory responses to tone A, whereas responses to tone B remained unchanged (Fig. 5h–k) (change index (CI) tone A, −0.66 ± 0.04, P = 8.8 × 10−22; tone B, 0.06 ± 0.05, P = 0.17; two-sided Wilcoxon signed-rank test). The fraction of neurons with suppressed compared to potentiated responses after tone A exposure was significantly greater in tone A but not tone B (Fig. 5l) (tone A, suppressed, 36.0%; potentiated, 12.9%, P = 1.6 × 10−21; tone B, suppressed, 17.1%; potentiated, 20.2%, P = 0.19; Fisher’s exact test). These data together indicate that repeated sound exposure induces sound-specific plasticity in both OFCvl→A1 axons and A1 neurons.
Finally, to examine the causal role of OFCvl’s predictive signaling in driving sound-specific habituation, we performed optogenetic experiments comparing the effects of OFCvl silencing on habituated (tone A) versus novel (tone B) stimuli. We virally expressed ChRmine in GABAergic neurons in the OFCvl of VGAT-Cre mice, enabling transcranial optogenetic silencing of the OFCvl (Extended Data Fig. 8a,b)41. Again, we imaged A1 L2/3 neurons during alternating presentations of tone A and tone B on day 0 and day 6, with repeated tone A presentations from day 1 to day 5 (Extended Data Fig. 8c). On days 0 and 6, we silenced the OFCvl during half of the trials ((A LED−) – (B LED−) – (A LED+) – (B LED+)…), synchronously with tone presentations. This design allowed comparisons of A1 neuronal responses between LED− and LED+ trials and evaluation of OFCvl silencing effects before and after habituation within the same neuronal population. On day 0, OFCvl silencing did not alter A1 responses to either tones, consistent with pharmacological inactivation results before habituation (Extended Data Fig. 8d and Fig. 2q–s). In contrast, on day 6, OFCvl silencing slightly increased responses to tone B but had an over tenfold greater effect on responses to tone A (Extended Data Fig. 8e–i) (Δ(ΔF/F), LED-no LED, tone A, day 0, 1.7 ± 1.0%; day 6, 9.4 ± 0.9%, day 0 versus day 6, P = 2.2 × 10−8; tone B, day 0, −0.09 ± 1.0%, day 6, 0.61 ± 0.92%, day 0 versus day 6, P = 9.5 × 10−5). LED illumination alone on day 6 without sound did not affect A1 activity (Extended Data Fig. 8g). Collectively, these findings support the idea that the OFCvl stores the information of repeated sensory stimuli and directly transmits predictive signals to A1, triggering sound-specific neural habituation.
Plasticity in SST cells amplifies sensory habituation
Our results have demonstrated neural plasticity in the OFC, leading to the predictive filtering of A1 sensory responses. However, the OFC is considered to support flexible learning not only through its own plasticity42,43,44 but also by driving plasticity in other brain regions45,46,47. A previous study found that pairing OFC → A1 axon terminal activation with tone presentations results in plastic changes in A1 that suppress its responses to the paired frequency34. Therefore, we wondered whether the enhanced OFCvl → A1 input during habituation triggers synaptic plasticity in A1 to further amplify sensory habituation.
We first explored the involvement of overall A1 synaptic plasticity in sensory habituation through the area-selective knockout of N-methyl-D-aspartate (NMDA) receptors. We co-injected AAV9- GCaMP6s and AAV8-mCherry-Cre into the A1 of floxed GluN1 mice (Grin1flox/flox) (Fig. 6a). Previous studies using the same mouse showed that Cre-dependent knockout of NMDA receptors became evident within 2 weeks post-transfection and grew over subsequent weeks48,49. Five weeks post-injection, tone response magnitudes of A1 pyramidal cells were smaller in GluN1 knockout mice compared to control mice (Extended Data Fig. 9a–d), consistent with the loss of NMDA receptors. Comparing 5-day sensory habituation of GluN1 knockout mice to controls revealed a significant reduction in across-day sensory habituation (Fig. 6b). It is unlikely that this diminished habituation can be explained by lower baseline sensory responses in GluN1 knockout mice, as reducing baseline auditory responses by lowering tone intensity to 60 dB SPL did not alter habituation (Extended Data Fig. 10). Thus, our results suggest that local NMDA receptor-dependent synaptic plasticity in A1 amplifies long-term sensory habituation.
a, Schematic illustrating the viral strategy to knock out NMDA receptors in A1 neurons (top). Representative two-photon image of A1 L2/3 neurons from a Grin1flox/flox mouse expressing GCaMP6s (green) and Cre (red) (bottom). b, Change index for excitatory (solid line) and inhibitory (dotted line) sound-evoked responses across days in Grin1flox/flox (red) and control (black) mice. Grin1flox/flox, n = 4 mice, 905 cells; control, n = 5 mice, 1,708 cells. c,d, Same as a,b but for knockout of NMDA receptors selectively in SST neurons. d, SST-Cre × Grin1flox/flox, n = 5 mice, 1,364 cells; control, n = 6 mice, 1,740 cells. e,f, Same as a,b but for knockout of NMDA receptors selectively in VIP neurons. f, VIP-Cre × Grin1flox/flox, n = 4 mice, 996 cells; control, n = 5 mice, 1,494 cells. g, Summary scatter-plots showing the change index of excitatory responses in individual neurons on day 5 compared to day 1. Grin1flox/flox, Cre− control (Ctrl), n = 229 excited cells. Grin1flox/flox + AAV-Cre (KOA1), n = 220. SST-Cre × Grin1flox/flox (KOSST), n = 262. VIP-Cre × Grin1flox/flox (KOVIP), n = 255. Ctrl versus KOA1, ****P = 3.6 × 10−5. Ctrl versus KOSST, ****P = 1.8 × 10−5. Ctrl versus KOVIP, P = 1.0. KOA1 versus KOSST, P = 1.0. KOA1 versus KOVIP, **P = 0.0019. KOSST versus KOVIP, **P = 0.0020 (two-sided Wilcoxon rank-sum test with Bonferroni correction). h, Summary diagrams of sensory habituation pathways. Error bars and shades represent mean ± s.e.m.
Finally, we focused on synaptic plasticity specifically in SST cells. Given that GluN1 is also expressed in SST cells and regulates their plasticity50,51, we generated Grin1flox/flox mice carrying SST-Cre to delete GluN1 selectively in SST cells. A1 pyramidal cells in these mice showed normal tone response properties in a naive condition (Extended Data Fig. 9h–k); however, quantification of tone responses across days revealed a significantly diminished habituation in SST cell GluN1 knockout mice compared to the control group (Fig. 6c,d). In contrast, selective deletion of GluN1 in VIP cells did not affect sensory habituation (Fig. 6e–g). Thus, while GluN1 deletion in SST cells does not alter basal sound processing in A1, it attenuates sensory habituation through decreased synaptic plasticity. Collectively, our experiments demonstrate that experience-dependent plasticity in both OFCvl circuits and A1 SST cells leads to sensory habituation via the recruitment of SST inhibitory circuits by top-down predictive signals.
Discussion
Theories based on human psychophysics have suggested the role of internal predictive models in sensory habituation (Extended Data Fig. 1). Our findings identify the neural substrates for these predictive models and the negative images of sensory stimuli they generate. In naive animals, bottom-up sensory signals evoke strong responses in the A1; however, with repeated exposure to the same sound over days, a predictive model forms in the OFC. The OFC then sends top-down predictive signals to SST cells in the A1, which in turn suppress the activity of pyramidal, VIP and PV cells. This finding may seem at odds with the focus of previous studies on the top-down regulation of VIP cells and the recruitment of the VIP → SST disinhibitory circuit33,52,53; however, quantification of cell type-specific retrograde tracing from sensory cortical areas has revealed denser top-down connectivity from frontal cortical areas to SST than VIP cells36 (Extended Data Fig. 5). Furthermore, SST and VIP cells are known to form a mutual inhibitory loop54,55, with SST → VIP connections exhibiting one of the highest connection probabilities, largest inhibitory post-synaptic potential amplitudes, and strongest facilitation within cortical circuits56. Therefore, it is not unexpected that top-down input from OFCvl, which connects to both SST and VIP cells, could engage the often overlooked SST → VIP inhibitory connection to suppress the A1 circuit. Synaptic plasticity in SST neurons may further shift the balance of SST–VIP mutual inhibition toward SST dominance to facilitate habituation (Fig. 6h). The balance in this mutually inhibitory SST–VIP circuit likely determines the direction of sensory response modulation, while the broad inhibition exerted by SST neurons onto both pyramidal and PV neurons could enable this modulation while simultaneously preserving excitation-inhibition balance within cortical circuits (Extended Data Fig. 3o). Our model successfully explains recent studies in V1 that identified a VIP-dominant disinhibitory circuit during animals’ first encounter with novel stimuli29,30. We do not exclude the roles of novelty or top-down prediction error signals in other contexts (Supplementary Discussion); however, our data indicate that daily sensory habituation is mediated by an increase in predictive signals, rather than a decrease in novelty signals. Thus, plasticity in the OFC, through the accumulation of experiences over extended periods42,43,44, creates and updates the internal predictive model of the sensory environment, which is then used to filter neural activity in sensory cortices.
The OFC is considered to support flexible behaviors by encoding the values of predicted outcomes57,58,59, assigning prediction error credits to corresponding actions60, and tracking associative relationships between stimuli1,2,46; however, the extent to which the OFC’s top-down projections influence sensory processing remains debated34,45,61. Our finding of the OFC’s role in the predictive filtering of sensory inputs aligns with the concept that the OFC constructs and uses internal associative models, or ‘cognitive maps,’ which dictate predictive relationships between the sensory task space and possible outcomes1,2. Furthermore, our results suggest a more generalized function of the OFC beyond merely predicting explicitly rewarding or punishing action outcomes, extending to the attenuation of salience in neutral stimuli during passive experiences. This broader role is reminiscent of the OFC’s involvement in ‘devaluation,’ where initially favorable stimuli lose their value with satiety or new associations with unfavorable outcomes62,63,64. It is plausible that sensory inputs inherently carry behavioral values, as they may signal the approach of rewards or threats crucial for an animal’s survival. By forming internal associative models, the OFC ‘devalues’ not only explicitly rewarding stimuli but also neutral sensory inputs without meaningful outcomes, thereby inducing habituation.
Our findings align with the broader predictive processing framework, in which the brain encodes prediction errors, the differences between top-down predictions and actual bottom-up sensory inputs, rather than faithfully relaying the sensory input24. Predictive processing has been documented across various domains, including reward association learning65, motor learning66, visuomotor coupling67,68 and cross-modal association18. In most of these studies, predictions are driven by external or internal cues (for example, reward cues, self-generated movements, or inputs from another sensory modality) rather than by the sensory stimuli themselves. We propose that sensory habituation fits within this predictive processing framework, but with the sensory stimulus itself acting as the predictive cue. Here, the onset of a familiar stimulus acts as a cue predicting its continuation. In naive animals, no internal model exists for a novel stimulus, resulting in robust prediction errors in A1 (Extended Data Fig. 1b, left). With repeated exposure, these error signals train an internal model in the OFC, which subsequently suppresses A1 error responses via top-down predictions. This self-predicting feedback mechanism explains the slow ramp up of habituation effects observed within each sound presentation (Fig. 1g,l) and highlights its potential relevance for habituation to prolonged environmental noise exposures.
The OFC’s long-range top-down predictive inputs onto A1 SST neurons parallel inhibitory circuit motifs described in other predictive processing contexts. In audiomotor coupling, top-down projections from M2 onto A1 PV neurons have been shown to suppress error signals, scaling down responses to self-generated sounds37. On the other hand, in a visuomotor mismatch paradigm, SST neurons were shown to suppress error signals in the primary visual cortex, consistent with our observations67. This latter study also reported the correlation between visual inputs and SST neuron activity, which we interpret as SST neurons receiving habituation-related top-down predictions in addition to bottom-up sensory inputs, as their experiments were conducted after multiple days of stimulus exposure, similar to day 5 in our study. These studies together suggest that multiple top-down prediction mechanisms (movement-related predictions from M2 and familiarity-based predictions from OFC) likely operate simultaneously, recruiting local inhibitory neurons to filter predictable sensory inputs. Beyond predictive coding, previous studies also suggested attentional filtering of A1 activity by the prefrontal cortex69,70,71. Understanding how individual frontal areas are recruited in specific contexts to apply these appropriate filters, and how these multiple predictive processing mechanisms function in concert, would be an important area for future research.
Impaired habituation to repeated stimuli and resultant sensory hypersensitivity are commonly observed in mental disorders such as obsessive-compulsive disorders3 and ASDs5,6. Although theories have proposed that a lack of predictive filtering may be central to the symptoms associated with ASDs16,17, the neural substrates linking altered neural wiring with reduced sensory filtering have remained unclear. Our discovery that top-down projections from the OFC to A1 are essential for auditory habituation provides crucial insights into the circuit mechanisms underlying reduced habituation in individuals with ASD. It is believed that the neocortical network in ASD displays long-range hypoconnectivity, as indicated by less cohesive activity across brain regions and reduced white matter volume72. This reduced long-range communication between frontal areas and sensory cortices could therefore be a culprit for the insufficient habituation in these disorders. This general framework of top-down predictive filtering highlights the importance of targeting long-range frontal projections in developing interventions to alleviate sensory pathophysiology.
Methods
Animals
Mice were at least 6 weeks old at the time of experiments. The strains used include: C57BL/6J (JAX 000664), B6N-Cdh23tm2.1Kjn/Kjn (JAX 018399), Slc32a1tm2(cre)Lowl/J (VGAT-Cre; JAX 028862), Pvalbtm1(cre)Arbr/J (PV-Cre; JAX 017320), Ssttm2.1(cre)Zjh/J (Sst-Cre; JAX 013044), Viptm1(cre)Zjh/J (VIP-Cre; JAX 010908), Gt(ROSA)26Sortm9(CAG-tdTomato)Hze/J (Ai9; JAX 007909) and B6.129S4-Grin1tm2Stl/J (Grin1flox/flox; JAX 005246). Both female and male mice were used. The animals were housed at 21 °C and 40% humidity, with ad libitum access to food pellets and water. They were kept under a reverse light cycle (12–12 h), and all experiments were conducted during their dark cycle. All experimental procedures were approved and conducted in accordance with the Institutional Animal Care and Use Committee at the University of North Carolina at Chapel Hill and the guidelines of the National Institutes of Health.
Auditory stimuli
Auditory stimuli were calculated in MATLAB (MathWorks) at a sample rate of 192 kHz and delivered via a free-field electrostatic speaker (ES1; Tucker-Davis Technologies). The speakers were calibrated across a range of 2–64 kHz to give a flat response (±1 dB). Stimuli were delivered to the ear contralateral to the imaging or recording site. The delivery of auditory stimuli was controlled by Bpod (Sanworks), running on MATLAB.
For areal mapping with intrinsic signal imaging, 3, 10 and 30 kHz pure tones (75 dB SPL, 1-s duration) were presented at 30-s intervals for 5–20 trials. The tonal receptive fields of individual neurons were determined by presenting 1-s pure tone pips covering 17 frequencies (4–64 kHz, log-spaced) at 30, 50 and 70 dB SPL. Tone pips were presented in a randomized order at 5-s intervals, with each stimulus repeated across five trials. For auditory habituation experiments, a single frequency was chosen for each mouse (see ‘Two-photon calcium imaging’ section). Prolonged pure tones (70 dB SPL unless otherwise specified, 5, 7 or 9-s duration) were presented at 7–16-s intervals, 100 or 200 trials per day. For linear probe recordings in A1, the cortical layers were identified through current source density analysis using click sounds, which were generated as 0.1-ms monopolar rectangular pulses and presented across 200 trials at 0.5-s intervals. All pure tone stimuli had a 5-ms linear rise and fall at their onset and offset.
Intrinsic signal imaging
Intrinsic signal imaging was conducted to locate the auditory cortical areas, following a previously described protocol32. Signals were acquired using a custom tandem lens macroscope comprising Nikkor 35 mm 1:1.4 and 135 mm 1:2.8 lenses, attached to a 12-bit CCD camera (DS-1A-01M30, Dalsa). Mice were anesthetized with isoflurane (0.8–2%) vaporized in oxygen (1 l min−1) and maintained on a heating pad at 36 °C. The muscle overlying the right auditory cortex was removed, and a custom stainless-steel head bar was secured to the skull with dental cement. For initial mapping before craniotomies, the brain surface was imaged through a skull kept transparent with PBS. For re-mapping before two-photon calcium imaging, the brain surface was imaged through an implanted glass window. Mice received a subcutaneous injection of chlorprothixene (1.5 mg kg−1) before imaging. Images of surface vasculature were captured under green LED illumination (530 nm), and intrinsic signals were recorded at 16 Hz using red illumination (625 nm). Images of reflectance were acquired at a resolution of 717 × 717 pixels (2.3 × 2.3 mm). Images collected during the response period (0.5–2 s from sound onset) were averaged across trials and normalized to the average image during the baseline period. The images were Gaussian filtered and thresholded for visualization. Individual auditory areas, including A1, AAF, VAF and A2, were identified based on their characteristic tonotopic patterns.
Two-photon calcium imaging
Following initial transcranial mapping of auditory cortical areas using intrinsic signal imaging, a craniotomy (2 × 3 mm) was performed over the right auditory cortex, leaving the dura intact. Except for axon terminal imaging, viruses were injected using sharpened glass pipettes (15–20 μm outer diameter) at 7–10 locations around A1 (180–250 μm deep from the pial surface, 30–40 nl per site at 10 nl min−1). For pyramidal cell imaging, AAV9-syn-GCaMP6s was injected in C57BL/6J or VGAT-Cre×Ai9 mice. For interneuron subtype-selective imaging, AAV9-syn-Flex-GCaMP6s was injected in either PV-Cre, SST-Cre, or VIP-Cre mice. For imaging with A1-selective NMDA receptor knockout, AAV9-syn-GCaMP6s and AAV8-mCherry-Cre were co-injected in Grin1flox/flox mice. For cell type-selective knockout of NMDA receptors, AAV9-syn-GCaMP6s was injected in SST-Cre×Grin1flox/flox or VIP-Cre×Grin1flox/flox mice. For imaging OFCvl axon terminals in A1, AAV1-syn-axonGCaMP6s was stereotaxically targeted to the right OFCvl at two coordinates (anterior, lateral, ventral) = (2.4, 0.85, 2.5–2.7) and (2.7, 0.85, 2.5–2.65) from bregma in mm (300 nl per site at 20 nl min−1). A glass window was then placed over the craniotomy and secured with dental cement. Dexamethasone (2 mg kg−1) was injected before the craniotomy. Enrofloxacin (10 mg kg−1) and meloxicam (5 mg kg−1) were administered before the mice were returned to their home cage.
Two-photon calcium imaging was conducted 2–7 weeks after chronic window implantation to ensure an appropriate level of GCaMP6s expression. A second intrinsic signal imaging through the chronic window was conducted 1–3 days before two-photon calcium imaging to confirm the integrity of the tonotopic map. During calcium imaging, awake mice were head fixed under a two-photon microscope in a custom-built sound-attenuation chamber. One to two fields of view (FOVs) in A1 were selected based on intrinsic signal maps. Before starting habituation experiments, tonal receptive fields of individual neurons were determined by measuring responses to pure tone pips. After constructing the best frequency maps, one FOV was selected in the low-to-middle-frequency domain of A1 to avoid the influence of high-frequency hearing loss. Sound frequencies for passive exposure (4–22 kHz) were chosen in each mouse as the frequency that evoked excitation in the largest fraction of cells in the imaged FOV. For sound-specific habituation experiments, two frequencies (tone A and tone B) with a one-octave separation flanking the best frequency of the FOV were selected to ensure comparable response magnitudes in naive conditions. During habituation, mice underwent either a single day or five consecutive days of imaging, with a pure tone presented 100 or 200 trials per day, except for one mouse imaged for 3 days (SST cells) and one for 4 days (axonal imaging). For sound-specific habituation experiments, tone A was presented for 200 trials per day across 5 days (days 1–5), whereas on days 0 and 6, tone A and tone B were alternately presented, limited to ten trials per sound to minimize habituation. In experiments involving sound-specific habituation combined with optogenetic OFCvl inactivation, photostimulation was applied during half of the trials on days 0 and 6, following a sequence of alternating trials ((A LED−) – (B LED−) – (A LED+) – (B LED+)…) repeated ten trials per day. GCaMP6s and tdTomato were excited at 925 nm and mCherry was excited at 1,070 nm (InSight DS+, Newport). Images (512 × 512 pixels covering 620 × 620 μm for imaging soma and 124 × 124 μm for imaging axon terminals) were acquired at 30 Hz using a commercial microscope (MOM scope, Sutter) equipped with a ×16 objective (Nikon) and running Scanimage software (Vidrio). Imaging was conducted in L2/3 (120–280 μm below the surface) for soma and in L1 and shallow L2 (60–130 μm below the surface) for axon terminals.
Muscimol infusion
For the pharmacological inactivation of the OFC, a cannula was implanted during the same surgery as the craniotomy. A guide cannula (outer diameter 0.41 mm, inner diameter 0.25 mm; RWD) was stereotaxically inserted at coordinates (A, L, V) = (2.7, 0.85, 2.7) in the right hemisphere. The guide cannula was secured with dental cement and sealed with a dummy cannula. On the day of muscimol infusion, the dummy cannula was replaced with an internal cannula (outer diameter 0.21 mm, inner diameter 0.11 mm; RWD) containing either a muscimol solution (2.5–5 mg ml−1 in PBS) or PBS (for control). A total volume of 500 nl of the solution was infused at 100 nl min−1 using a micropump (UMP3 and Micro4, WPI). Infusions were carried out before starting day 6 of sound exposure (habituated mice) or after 100 trials of sound exposure on day 1 (naive mice).
Optogenetic OFC inactivation
For optogenetic inactivation of the OFCvl, VGAT-Cre mice underwent injections of AAV8-nEF-Con/Foff-ChRmine-oScarlet (300 nl per site at 20 nl min−1) into the right OFCvl during the same surgery as chronic window implantation in A1. Injections were stereotaxically targeted at two coordinates: (A, L, V) = (2.4, 0.75, 2.7) and (2.8, 0.75, 2.65). Head bar and head cap implantation procedures were identical to those used in standard two-photon calcium imaging experiments, except that head bars with a large hole were used to allow optical access to the frontal cortex. Craniotomies were sealed with Duragel (Cambridge Neurotech), covered with a thin layer of transparent cement, and protected with an additional layer of silicone. The mice were allowed to recover in home cages for 2–3 weeks to achieve the appropriate level of ChRmine expression. On experimental days 0 and 6, the silicone was removed, and a fiber-coupled 625 nm LED (Thorlabs) was positioned 1–2 mm above the transparent cement covering the frontal cortex. ChRmine-expressing GABAergic neurons were activated by transcranial illumination (50 Hz, 10-ms pulses; maximum intensity of 60 mW at the fiber tip outside the skull) simultaneously with tone presentations in alternating trials. Effective OFCvl inactivation was confirmed by independent electrophysiological recording experiments under the same experimental conditions. To compare tone response magnitudes between LED and No LED conditions within the same cells, significantly tone-responsive neurons were identified independently for LED and No LED trials, and neurons significantly responsive in at least one condition were included in the analysis (Extended Data Fig. 8d,e).
Pupillometry
The eye contralateral to the craniotomy was imaged using an infrared camera (Genie Nano M640-NIR, Teledyne Dalsa, or Firefly FFY-U3-16S2M-S, FLIR). The two-photon excitation laser transmitted through the eye was used to visualize the pupil. A violet LED (395 nm) provided low-intensity illumination, maintaining the pupil at approximately mid-range in diameter. The intensity and position of the LED were kept consistent across chronic imaging sessions to allow for cross-day comparisons of pupil diameters. Pupil videos were captured and synchronized with sound presentation trials using Bonsai software (https://bonsai-rx.org/). Images of 360 × 480 pixels were collected at 30 Hz. For each acquired video, an intensity threshold was set, and the images were binarized to extract the pupil pixels. The pupil diameter was then determined as the maximum Feret diameter of the segmented pupil pixels for each frame. Frames with undetectable pupils due to blinking were excluded using a deviant detection program. Pupil sizes were normalized against the minimum and maximum diameters observed throughout the entire multi-day recordings for each mouse.
Analysis of two-photon calcium imaging data
Lateral motion was corrected using cross-correlation-based image alignment. Regions of interest (ROIs) corresponding to individual cell bodies or axon boutons were detected automatically using Suite2P software73 and supplemented by manual drawing. All ROIs were individually inspected and edited for appropriate shapes using a graphical user interface in MATLAB. Pixels within each ROI were averaged to derive a fluorescence time series, Fcell-measured(t). To correct for background contamination, ring-shaped background ROIs (starting at 2 pixels and ending at 8 pixels from the border of the ROI) were created around each cell ROI. From this background ROI, pixels that contained cell bodies or processes from neighboring cells (detected as pixels showing large increases in ΔF/F uncorrelated to that of the cell ROI during the entire imaging session) were excluded. The remaining pixels were averaged to generate a background fluorescence time series Fbackground(t). The fluorescence signal for each cell was estimated as F(t) = Fcell_measured(t) – 0.9 × Fbackground(t). To ensure robust neuropil subtraction, only cell ROIs that were at least 3% brighter than their corresponding background ROIs were included. To compensate for slow drifts in baseline fluorescence intensity, a normalized trace, Fnorm(t), was calculated as F(t)/F0(t), where F0(t) represents a time-varying drifting trace estimated by smoothing the inactive portions of F(t) using an iterative procedure.
The total receptive fields of individual cells were measured using their responses to 1-s pure tones. Cells were judged as significantly excited if they fulfilled two criteria: (1) Fnorm(t) had to exceed a fixed threshold value consecutively for at least 0.5 s in more than half of the trials, and (2) Fnorm(t) averaged across trials had to exceed the same fixed threshold value consecutively for at least 0.5 s. The threshold for excitation (2.5 × s.d. during the baseline period) was determined using receiver operating characteristic analysis to yield a 90% true positive rate in tone responses, whereas the threshold for inhibition (−1.25 × s.d.) was set at half of that for excitation. Response amplitudes were quantified using a 1.2-s window after the tone onset. Best frequencies were identified as the frequency evoking the strongest excitatory response, independent of tone intensity. Bandwidth (BW70) was calculated as the average of the range of frequencies that evoked significant responses and the range of frequencies with a Gaussian fit exceeding a threshold at 70 dB SPL.
Cellular activity for multi-day experiments was assessed for significant excitation or inhibition based on two criteria: (1) the area above baseline for individual trials was significantly larger (or smaller for inhibition) than zero, using a two-sided Wilcoxon signed-rank test, and (2) the peak amplitude of the response exceeded 1 × s.d. for excitation (or –0.5 × s.d. for inhibition). For axon bouton imaging, a two-sided Wilcoxon signed-rank test with a significance level of 0.1 was used for excitation, and inhibition was not assessed due to the low baseline fluorescence values. Given variations in tone durations (5, 7 and 9 s), statistical analyses focused on the first 5 s of tone presentation. Trial-averaged sound response traces for each cell were compiled from responses across the three tone durations. The area above (or below for inhibition) the baseline of the average trace during the first 5 s was used to calculate the CI as CI = (Areaafter – Areabefore)/(Areaafter + Areabefore). CIs were calculated separately for excitatory and inhibitory responses, and only responsive cells were included in the analysis. For example, if the CI was calculated from day 1 to day X, only cells that were responsive on day 1, day X, or both were included, and CI was calculated as CI = (day X – day 1)/(day X + day 1). In muscimol infusion experiments, responses on day 6 were compared to those on day 5. In single-day experiments, averaged traces were calculated for blocks of 25 trials, and responses were compared to those in block 1.
High-dimensional analysis of calcium imaging data
To compare within-day and across-day changes in the response patterns of A1 neuronal ensembles, we analyzed A1 population activity in a high-dimensional space, with each dimension corresponding to the response magnitude of individual neurons. We included only ROIs that were consistently visible across all imaging sessions and showed significant excitatory or inhibitory responses to tones in at least one session. Responses in each ROI were normalized to their minimum and maximum values across all sessions, resulting in normalized values that ranged from 0 to 1. We concatenated time series data from all trials across sessions to create an ensemble response matrix structured as ROIs × frames × trials. Although tone durations varied between 5, 7 and 9 s, only the first 5 s of tone presentations, shared across all trials, were included in the analysis. Direction vectors, representing plasticity in ensemble response patterns, were calculated for within-day (Modewithin) and across-day (Modeacross) changes. To ensure balanced contributions from onset-locked and sustained activities, we calculated a neuron’s response magnitude in a trial by summing its normalized activity from 0.5–1.5 s and 4–5 s after sound onset. Modewithin was obtained by computing the vector from the mean of trials 1–10 to the mean of trials 91–100 for each day and averaging these vectors over days 1–5. Modeacross was derived by calculating trial-averaged responses for days 1 and 5 and computing their differences. The entire ensemble response matrix was projected onto Modewithin and Modeacross to visualize the within-day and across-day habituation of population response patterns. The kinetics of within-day plasticity were visualized by computing the difference between the ensemble activity in trial 10 and that in trial 1, and projecting this difference onto the Modewithin axis. Similarly, the kinetics of across-day plasticity were visualized by computing the difference between the trial-averaged ensemble activity on day 5 and day 1, and projecting this difference onto the Modeacross axis. These kinetics were quantified using the Ramp-up Index, which is defined as the ratio of the plasticity trace at 1 s and that at 5 s after tone onset. For the 6-day muscimol experiments, data from day 6 (post-muscimol infusion) were projected onto Modewithin and Modeacross, which were calculated based on data from days 1 to 5.
NMDA receptor knockout experiments
To selectively ablate NMDA receptors from the A1 region, AAV8-syn-mCherry-Cre was co-injected with AAV9-syn-GCaMP6s into the A1 of Grin1flox/flox mice during the window implantation surgery. Following the injections and window implantation, mice were allowed to recover in their home cages for at least 5 weeks to ensure a sufficient reduction in NMDA receptor expression before habituation experiments. During two-photon calcium imaging, neurons expressing Cre were identified by their mCherry fluorescence. Control groups included both Grin1flox/flox mice without Cre virus injections and Grin1+/+ mice with Cre virus injections. For targeted NMDA receptor knockout in specific inhibitory neuron subtypes, SST-Cre or VIP-Cre mice were crossed with Grin1flox/flox mice. The procedures for virus injections and surgeries were identical to those used for two-photon imaging in C57BL/6J mice. Mice were at least 8 weeks old at the start of habituation experiments to ensure sufficient reduction in NMDA receptor expression. Data from mice carrying both homozygous and heterozygous alleles of floxed Grin1 were pooled, as no significant differences were found between these genotypes. Control mice included Grin1flox/flox mice without Cre transgenes, SST-Cre mice without floxed Grin1 transgenes, and VIP-Cre mice without floxed Grin1 transgenes.
Electrophysiology
For single-unit recordings in the frontal cortex, mice were first implanted with a custom stainless-steel head bar and a cement cap. For recordings in habituated mice, the animals were head fixed and exposed to prolonged 10 kHz pure tones (70 dB SPL, 5, 7 or 9-s durations, 7–16-s intervals) for 200 trials per day over 5 days before the recording. On the recording day, following a small craniotomy (<0.3 mm) and durotomy in the right frontal cortex, mice were head fixed in an awake state, and a 384-channel probe (Neuropixels 1.0, IMEC) was slowly inserted (approximately 2 μm s−1) perpendicular to the brain surface. To simultaneously record from OFCvl and M2, the probe was stereotaxically targeted to the coordinates (A, L, V) = (2.6, 0.75, 2.8). Additional probe insertion sites sampled a range of stereotaxic locations to record from surrounding frontal regions, including ACC, FRP, PL, IL and OFCm. A reference electrode was placed at the dura above the frontal cortex of the contralateral hemisphere or the somatosensory cortex of the ipsilateral hemisphere. The probe was allowed to settle for at least 1 h before collecting data. Unit activity was amplified, digitized, and acquired at 30 kHz using the OpenEphys system (https://open-ephys.org). During recordings, the mice sat quietly in a loosely fitted plastic tube in a sound-attenuating enclosure (Gretch-Ken). The tube was lined with fleece fabric for comfort and attenuation of noise from scratching. In two of the habituated mice, recordings were repeated over 2 days to sample multiple brain regions.
An intersectional viral approach was used to selectively activate OFCvl neurons projecting to A1 L2/3. Following the initial transcranial mapping of auditory cortical areas using intrinsic signal imaging in C57BL/6J mice, two small craniotomies (<300 μm) were performed over the low- and high-frequency domains of the right A1, where AAVretro-syn-Cre was injected (200–250 μm deep, 40 nl per site at 10 nl min−1). In the same surgery, the right OFCvl was stereotaxically targeted for the injection of AAV8-nEF-Con/Foff-ChRmine-oScarlet (300 nl per site at 20 nl min−1) at two coordinates: (A, L, V) = (2.4, 0.75, 2.7) and (2.8, 0.75, 2.65). A stainless-steel head bar was cemented to the skull, and the skull’s top surface was covered with black cement, leaving the areas around the craniotomies. The craniotomies were sealed with Duragel (Cambridge Neurotech), covered with a thin layer of transparent cement, and protected by an additional layer of silicone. The mice were allowed to recover in their home cages for 2–3 weeks to achieve the appropriate level of ChRmine expression. On the day of recording, following a small craniotomy (<0.3 mm) and durotomy in the 10-kHz-responding domain of the A1, mice were head fixed in an awake state, and a 384-channel probe was slowly inserted (approximately 1 μm s−1) perpendicular to the brain surface. Spikes were monitored during probe insertion, and the probe was advanced until it reached the white matter, where no spikes were detected. The reference electrode was placed at the dura above either the visual or somatosensory cortex. A fiber-coupled 638 nm laser (RLM638TA-200FC; Shanghai Laser) was positioned 1–2 mm above the transparent cement covering the frontal cortex. ChRmine-expressing neurons were activated by transcranial illumination (1-s duration, continuous pulse, 50 Hz, 10-ms ramped pulses or 20-Hz sinusoidal pulses; maximum intensity of 80 mW at the fiber tip outside the skull) at a 10-s or 20-s interval. Control experiments were conducted with mice that did not express ChRmine. Successful transcranial activation of OFCvl units was confirmed by simultaneous recording from OFCvl during optogenetic activation.
Analysis of unit recording data
Single- and multi-units were isolated using Kilosort v.2.5 software74 and the spike-sorting graphical user interface Phy75. Single-unit isolation was confirmed based on the consistency of the spike waveform and the inter-spike interval histogram. Specifically, after correction for the overall spike frequency, less than 5% of the spikes should fall within the refractory period, which was 2 ms and 1.5 ms for regular-spiking and fast-spiking units, respectively. The initial automated classification was conducted using the Bombcell spike classification toolbox76. Following this automated step, spike clusters were individually inspected and edited to eliminate artifacts and duplications. Multi-unit spikes were calculated by aggregating all spikes within each channel or layer bin.
Post hoc identification of probe trajectories in the frontal cortex was achieved by examining DiI or DiO fluorescence in brain sections counterstained with DAPI. Brain sections were aligned to the Allen Common Coordinate Framework using MATLAB codes modified from an existing repository (https://github.com/petersaj/AP_histology). Channels in the ventrolateral and lateral regions of OFC were collectively referred to as OFCvl. For A1 recordings, the positions of the cortical surface, layer borders and white matter were identified using current source density analysis, the distribution of multi-unit spikes, and the correlation coefficient across channels77.
Single units were considered significantly excited by tones if their spike rates during tone presentations were significantly higher than those during baseline period (5 s before the tone onset), as determined across trials using a two-sided Wilcoxon signed-rank test. Out of the 200 trials, the first 10 trials were excluded from analysis to remove the effect of rapid within-day habituation. In each mouse, the fraction of excited single units was calculated only for areas containing at least 30 isolated single units.
Peristimulus time histograms (PSTHs) were constructed using a 500-ms bin width. For visualization in Fig. 4l and Extended Data Figs. 7 and 8, PSTHs were smoothed using a Gaussian kernel. Tone response magnitudes in individual units were quantified by calculating the total spikes during tone presentations and subtracting the baseline firing rate obtained from the averaged PSTH. The effects of optogenetic manipulation were quantified using the modulation index, which was calculated as (L − B)/(L + B), where L represents the activity during laser stimulation, and B represents the baseline activity. The modulation index values range from −1 to 1, where −1 indicates a complete loss of activity, 1 indicates the emergence of activity from nothing, and 0 indicates no change.
Anatomical tracing
Cell type-nonspecific retrograde tracing from A1 was conducted using two approaches: injecting AAVretro-syn-Cre in Ai9 mice or AAVretro-CAG-EGFP in VGAT-Cre × Ai9 mice. Following transcranial mapping with intrinsic signal imaging, the viruses were injected into the low-to-mid-frequency domain of A1 using a sharpened glass pipette (250 μm or 750 μm deep, 20 nl at 10 nl min−1). Data from injections at both depths were combined, as no clear difference was observed in the distribution of presynaptic neurons.
For trans-synaptic retrograde tracing from specific neuron subtypes in A1, we used rabies virus-mediated monosynaptic tracing78. Following transcranial mapping, a mixture of AAV5-CAGGS-Flex-Rev-mKate-T2A-CVS-N2cG and AAV5-CAG-Flex-Rev-mKate-T2A-TVA (for Emx1-Cre, SST-Cre and PV-Cre-transgenic mice), or AAV9-CAGGS-Flex-Rev-mKate-T2A-CVS-N2cG and AAV9-CAG-Flex-Rev-mKate-T2A-TVA (for VIP-Cre mice) was injected into the mid-frequency domain of A1 (250 μm deep, 40 nl, 10 nl min−1). Two weeks later, EnvA-pRbv-CVS-N2c(ΔG)-H2B-EGFP was injected at the same location (400 nl, 20 nl min−1), resulting in the visualization of mKate+/EGFP+ starter cells around the injection site and EGFP+ presynaptic cells in the frontal cortical areas.
Cell type-nonspecific tracing from the OFCvl was conducted by injecting AAV5-syn-EYFP in C57BL/6J mice for anterograde tracing and by injecting AAVretro-syn-mCherry in C57BL/6J mice or AAVretro-syn-Cre in Ai9 mice for retrograde tracing. The viruses were stereotaxically injected at the coordinates (A, L, V) = (2.7, 0.85, 2.7) (60–80 nl at 10 nl min−1).
Two weeks (AAV) or 1 week (rabies) after virus injection, cardiac perfusion was performed using PBS, followed by 4% paraformaldehyde in PBS under isoflurane anesthesia. The brains were then extracted, fixed overnight with 4% paraformaldehyde, and immersed in 30% sucrose in PBS overnight for cryoprotection. The brains were coronally sectioned at 40-μm thickness using a freezing microtome (SM2010R, Leica). After the sections were mounted on slide glasses and counterstained with DAPI, images were acquired using a fluorescence microscope (Axio Observer 7, ZEISS). Brain section images were aligned to the Allen Common Coordinate Framework using ABBA software (https://abba-documentation.readthedocs.io/en/latest/) to delineate anatomical area borders. The somas of retrogradely labeled presynaptic neurons were automatically detected and sorted into brain regions using QuPath software (https://qupath.github.io/). Histology figure panels were created by overlaying signals from multiple colors using Fiji (https://imagej.net/Fiji) or ZEN Microscopy (ZEISS) software.
Immunohistochemistry
For NMDA receptor immunostaining, brain slices were incubated in PBS containing 0.1% Triton X-100 and 10% normal donkey serum on a shaker for 1 h at room temperature. The slices were rinsed three times with PBS for 5 min each and incubated in PBS with rabbit anti-NR1 primary antibodies (1:100 dilution, ab52177, lot 1030057-4, Abcam) on a shaker overnight at 4 °C. The slices were then rinsed three times in PBS and incubated in PBS with Alexa 488-conjugated donkey anti-rabbit secondary antibodies (1:200 dilution, 711-545-152, lot 134352, Jackson ImmunoResearch) for 2 h at room temperature. After rinsing three times in PBS, the slices were mounted on slide glasses.
Statistics and reproducibility
Sample sizes were not predetermined by statistical methods but were selected based on standards commonly used in the field. All key results were replicated across multiple mice. Mice were randomly assigned to experimental groups. While the experiments were not performed blind, the experimenters were blind to the types and timings of trials during the ROI segmentation in two-photon imaging and spike sorting in electrophysiology. Data are presented as individual data points or mean ± s.e.m., as specified in the figure legends. Statistical differences between conditions were evaluated using standard two-sided nonparametric tests, specifically the Wilcoxon rank-sum test for unpaired data and the Wilcoxon signed-rank test for paired data. A Bonferroni correction was applied for multiple comparisons, and corrected P values were reported. Fractions were compared using Fisher’s exact test. Correlation coefficients were calculated as Spearman’s correlation using MATLAB’s corr function. A two-way analysis of variance was used to evaluate the effects of two independent variables.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
ΔF/F0 trace data for all imaged cells are available at Zenodo (https://doi.org/10.5281/zenodo.18613596)79. Source data are provided with this paper.
Code availability
This project did not develop broadly applicable software packages; however, code is available from the corresponding author upon request.
References
Wilson, R., Takahashi, Y., Schoenbaum, G. & Niv, Y. Orbitofrontal cortex as a cognitive map of task space. Neuron 81, 267–279 (2014).
Costa, K. M. et al. The role of the lateral orbitofrontal cortex in creating cognitive maps. Nat. Neurosci. 26, 107–115 (2023).
Hallett, M. Tourette syndrome: update. Brain Dev. 37, 651–655 (2015).
Titone, D., Ditman, T., Holzman, P. S., Eichenbaum, H. & Levy, D. L. Transitive inference in schizophrenia: impairments in relational memory organization. Schizophr. Res. 68, 235–247 (2004).
Guiraud, J. A. et al. Differential habituation to repeated sounds in infants at high risk for autism. Neuroreport 22, 845–849 (2011).
Green, S. A. et al. Neurobiology of sensory overresponsivity in youth with autism spectrum disorders. JAMA Psychiatry 72, 778–786 (2015).
Thompson, R. F. Habituation: a history. Neurobiol. Learn. Mem. 92, 127–134 (2009).
Wehr, M. & Zador, A. Synaptic mechanisms of forward suppression in rat auditory cortex. Neuron 47, 437–445 (2005).
Natan, R. G. et al. Complementary control of sensory adaptation by two types of cortical interneurons. Elife 4, e09868 (2015).
Castellucci, V., Pinsker, H., Kupfermann, I. & Kandel, E. R. Neuronal mechanisms of habituation and dishabituation of the Gill-withdrawal reflex in Aplysia. Science. 167, 1745–1748 (1970).
Malmierca, M. S., Cristaudo, S., Pérez-González, D. & Covey, E. Stimulus-specific adaptation in the inferior colliculus of the anesthetized rat. J. Neurosci. 29, 5483–5493 (2009).
Ulanovsky, N., Las, L., Farkas, D. & Nelken, I. Multiple time scales of adaptation in auditory cortex neurons. J. Neurosci. 24, 10440–10453 (2004).
Westenberg, I. S. & Weinberger, N. M. Evoked potential decrements in auditory cortex. II. Critical test for habituation. Electroencephalogr. Clin. Neurophysiol. 40, 356–369 (1976).
Cooke, S. F., Komorowski, R. W., Kaplan, E. S., Gavornik, J. P. & Bear, M. F. Visual recognition memory, manifested as long-term habituation, requires synaptic plasticity in V1. Nat. Neurosci. 18, 262–271 (2015).
Kato, H. K., Chu, M. W., Isaacson, J. S. & Komiyama, T. Dynamic sensory representations in the olfactory bulb: modulation by wakefulness and experience. Neuron 76, 962–975 (2012).
Ramaswami, M. Network plasticity in adaptive filtering and behavioral habituation. Neuron 82, 1216–1229 (2014).
Sinha, P. et al. Autism as a disorder of prediction. Proc. Natl Acad. Sci. USA 111, 15220–15225 (2014).
Garner, A. R. & Keller, G. B. A cortical circuit for audio-visual predictions. Nat. Neurosci. 25, 98–105 (2022).
Jordan, W. P., Strasser, H. C. & McHale, L. Contextual control of long-term habituation in rats. J. Exp. Psychol. Anim. Behav. Process. 26, 323–339 (2000).
Dissegna, A., Turatto, M. & Chiandetti, C. Context-specific habituation: a review. Animals 11, 1767 (2021).
Fiser, A. et al. Experience-dependent spatial expectations in mouse visual cortex. Nat. Neurosci. 19, 1658–1664 (2016).
Sokolov, E. N. Higher nervous functions; the orienting reflex. Annu. Rev. Physiol. 25, 545–580 (1963).
Wagner, A. R. Habituation and memory. in Mechanisms of Learning and Motivation (eds Dickinson, A. & Boakes, R. A.) Ch. 3 (Psychology Press, 1980).
Keller, G. B. & Mrsic-Flogel, T. D. Predictive processing: a canonical cortical computation. Neuron 100, 424–435 (2018).
Poort, J. et al. Learning enhances sensory and multiple non-sensory representations in primary visual cortex. Neuron 86, 1478–1490 (2015).
Kato, H. K., Gillet, S. N. & Isaacson, J. S. Flexible sensory representations in auditory cortex driven by behavioral relevance. Neuron 88, 1027–1039 (2015).
Gillet, S. N., Kato, H. K., Justen, M. A., Lai, M. & Isaacson, J. S. Fear learning regulates cortical sensory representations by suppressing habituation. Front. Neural Circuits 11, 112 (2018).
Makino, H. & Komiyama, T. Learning enhances the relative impact of top-down processing in the visual cortex. Nat. Neurosci. 18, 1116–1122 (2015).
Garrett, M. et al. Experience shapes activity dynamics and stimulus coding of VIP inhibitory cells. Elife 9, e50340 (2020).
Aitken, K., Campagnola, L., Garrett, M. E., Olsen, S. R. & Mihalas, S. Simple synaptic modulations implement diverse novelty computations. Cell Rep. 43, 114188 (2024).
Homann, J., Koay, S. A., Chen, K. S., Tank, D. W. & Berry, M. J. Novel stimuli evoke excess activity in the mouse primary visual cortex. Proc. Natl Acad. Sci. USA 119, e2108882119 (2022).
Narayanan, D. P., Tsukano, H., Kline, A. M., Onodera, K. & Kato, H. K. Biological constraints on stereotaxic targeting of functionally-defined cortical areas. Cereb. Cortex 33, 3293–3310 (2023).
Furutachi, S., Franklin, A. D., Aldea, A. M., Mrsic-Flogel, T. D. & Hofer, S. B. Cooperative thalamocortical circuit mechanism for sensory prediction errors. Nature 633, 398–406 (2024).
Winkowski, D. E. et al. Orbitofrontal Cortex Neurons Respond to Sound and Activate Primary Auditory Cortex Neurons. Cereb. Cortex 28, 868–879 (2018).
Ying, R. et al. Organization of orbitofrontal-auditory pathways in the Mongolian gerbil. J. Comp. Neurol. 531, 1459–1481 (2023).
Tasaka, G. I., Maggi, C., Taha, E. & Mizrahi, A. The local and long-range input landscape of inhibitory neurons in mouse auditory cortex. J. Comp. Neurol. 531, 502–514 (2023).
Schneider, D. M., Nelson, A. & Mooney, R. A synaptic and circuit basis for corollary discharge in the auditory cortex. Nature 513, 189–194 (2014).
Sun, W. et al. The anterior cingulate cortex directly enhances auditory cortical responses in air-puffing-facilitated flight behavior. Cell Rep. 38, 110506 (2022).
Cavada, C., Compañy, T., Tejedor, J., Cruz-Rizzolo, R. J. & Reinoso-Suárez, F. The anatomical connections of the macaque monkey orbitofrontal cortex. A review. Cereb. Cortex 10, 220–242 (2000).
Srivastava, H. K. & Bandyopadhyay, S. Parallel lemniscal and non-lemniscal sources control auditory responses in the orbitofrontal cortex (OFC). eNeuro 7, 1–19 (2020).
Chen, R. et al. Deep brain optogenetics without intracranial surgery. Nat. Biotechnol. 39, 161–164 (2021).
Hattori, R. et al. Meta-reinforcement learning via orbitofrontal cortex. Nat. Neurosci. 26, 2182–2191 (2023).
Namboodiri, V. M. K. et al. Single-cell activity tracking reveals that orbitofrontal neurons acquire and maintain a long-term memory to guide behavioral adaptation. Nat. Neurosci. 22, 1110–1121 (2019).
Johnson, C. M., Peckler, H., Tai, L. H. & Wilbrecht, L. Rule learning enhances structural plasticity of long-range axons in frontal cortex. Nat. Commun. 7, 1–14 (2016).
Banerjee, A. et al. Value-guided remapping of sensory cortex by lateral orbitofrontal cortex. Nature 585, 245–250 (2020).
Gardner, M. P. H. & Schoenbaum, G. The orbitofrontal cartographer. Behav. Neurosci. 135, 267–276 (2021).
Miller, K. J., Botvinick, M. M. & Brody, C. D. Value representations in the rodent orbitofrontal cortex drive learning, not choice. Elife 11, e64575 (2022).
Larsen, R. S. et al. Synapse-specific control of experience-dependent plasticity by presynaptic NMDA receptors. Neuron 83, 879–893 (2014).
Iwasato, T. et al. Cortex-restricted disruption of NMDAR1 impairs neuronal patterns in the barrel cortex. Nature 406, 726–731 (2000).
Perez-Rando, M. et al. NMDA receptors regulate the structural plasticity of spines and axonal boutons in hippocampal interneurons. Front. Cell. Neurosci. 11, 271022 (2017).
Nyíri, G., Stephenson, F. A., Freund, T. F. & Somogyi, P. Large variability in synaptic N-methyl-D-aspartate receptor density on interneurons and a comparison with pyramidal-cell spines in the rat hippocampus. Neuroscience 119, 347–363 (2003).
Fu, Y. et al. A cortical circuit for gain control by behavioral state. Cell 156, 1139–1152 (2014).
Zhang, S. et al. Long-range and local circuits for top-down modulation of visual cortex processing. Science 345, 660–665 (2014).
Pfeffer, C. K., Xue, M., He, M., Huang, Z. J. & Scanziani, M. Inhibition of inhibition in visual cortex: the logic of connections between molecularly distinct interneurons. Nat. Neurosci. 16, 1068–1076 (2013).
Karnani, M. et al. Cooperative Subnetworks of Molecularly Similar Interneurons in Mouse Neocortex. Neuron 90, 86–100 (2016).
Campagnola, L. et al. Local connectivity and synaptic dynamics in mouse and human neocortex. Science 375, eabj5861 (2022).
Padoa-Schioppa, C. & Assad, J. A. Neurons in the orbitofrontal cortex encode economic value. Nature 441, 223–226 (2006).
Hirokawa, J., Vaughan, A., Masset, P., Ott, T. & Kepecs, A. Frontal cortex neuron types categorically encode single decision variables. Nature 576, 446–451 (2019).
Schoenbaum, G., Chiba, A. A. & Gallagher, M. Orbitofrontal cortex and basolateral amygdala encode expected outcomes during learning. Nat. Neurosci. 1, 155–159 (1998).
Walton, M. E., Behrens, T. E. J., Buckley, M. J., Rudebeck, P. H. & Rushworth, M. F. S. Separable learning systems in the macaque brain and the role of orbitofrontal cortex in contingent learning. Neuron 65, 927–939 (2010).
Liu, D. et al. Orbitofrontal control of visual cortex gain promotes visual associative learning. Nat. Commun. 11, 1–14 (2020).
Howard, J. D. & Kahnt, T. Identity-specific reward representations in orbitofrontal cortex are modulated by selective devaluation. J. Neurosci. 37, 2627–2638 (2017).
Gardner, M. P. H., Conroy, J. S., Shaham, M. H., Styer, C. V. & Schoenbaum, G. Lateral orbitofrontal inactivation dissociates devaluation-sensitive behavior and economic choice. Neuron 96, 1192–1203.e4 (2017).
Gottfried, J. A., O'Doherty, J. & Dolan, R. J. Encoding predictive reward value in human amygdala and orbitofrontal cortex. Science 301, 1104–1107 (2003).
Schultz, W., Dayan, P. & Montague, P. R. A neural substrate of prediction and reward. Science 275, 1593–1599 (1997).
Wolpert, D. M., Ghahramani, Z. & Jordan, M. I. An internal model for sensorimotor integration. Science 269, 1880–1882 (1995).
Attinger, A., Wang, B. & Keller, G. B. Visuomotor coupling shapes the functional development of mouse visual cortex. Cell 169, 1291–1302.e14 (2017).
Keller, G., Bonhoeffer, T. & Hübener, M. Sensorimotor mismatch signals in primary visual cortex of the behaving mouse. Neuron 74, 809–815 (2012).
Nakajima, M., Schmitt, L. I. & Halassa, M. M. Prefrontal cortex regulates sensory filtering through a basal ganglia-to-thalamus pathway. Neuron 103, 445–458.e10 (2019).
Fritz, J., Shamma, S., Elhilali, M. & Klein, D. Rapid task-related plasticity of spectrotemporal receptive fields in primary auditory cortex. Nat. Neurosci. 6, 1216–1223 (2003).
Fritz, J. B., Elhilali, M. & Shamma, S. A. Differential dynamic plasticity of A1 receptive fields during multiple spectral tasks. J. Neurosci. 25, 7623–7635 (2005).
Courchesne, E. & Pierce, K. Why the frontal cortex in autism might be talking only to itself: local over-connectivity but long-distance disconnection. Curr. Opin. Neurobiol. 15, 225–230 (2005).
Pachitariu, M. et al. Suite2p: beyond 10,000 neurons with standard two-photon microscopy. Preprint at bioRxiv https://doi.org/10.1101/061507
Pachitariu, M., Steinmetz, N., Kadir, S., Carandini, M. & Harris, K. D. Kilosort: realtime spike-sorting for extracellular electrophysiology with hundreds of channels. preprint at bioRxiv https://doi.org/10.1101/061481
Rossant, C. et al. Spike sorting for large, dense electrode arrays. Nat. Neurosci. 19, 634–641 (2016).
Fabre, J. M. J., van Beest, E. H., Peters, A. J., Carandini, M. & Harris, K. D. Bombcell: automated curation and cell classification of spike-sorted electrophysiology data. Zenodo https://doi.org/10.5281/ZENODO.8172822 (2023).
Senzai, Y., Fernandez-Ruiz, A. & Buzsáki, G. Layer-specific physiological features and interlaminar interactions in the primary visual cortex of the mouse. Neuron 101, 500–513.e5 (2019).
Wickersham, I. et al. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 53, 639–647 (2007).
Tsukano, H., Garcia, M. M., Dandu, P. R. & Kato, H. K. Dataset for Tsukano et al. 2026 "Orbitofrontal cortex drives predictive filtering of sensory responses". Zenodo https://doi.org/10.5281/zenodo.18613596 (2026).
Acknowledgements
We thank P. Manis, J. Isaacson, D. Polley and the members of the Kato Laboratory for their comments on the manuscript. We thank G. Lopes for his support in setting up Bonsai for pupil monitoring. This work was supported by National Institute on Deafness and Other Communication Disorders (R01DC017516; H.K.K.), National Institute of Neurological Disorders and Stroke and BRAIN Initiative (R01NS128873; H.K.K.), Simons Foundation (SFARI 979212; H.K.K.), Eagles Autism Foundation (24-1395; H.K.K.) and Foundation of Hope (H.K.K. and H.T.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
H.T. and H.K.K. designed the project, performed data analysis, and wrote the manuscript. H.T. performed calcium imaging experiments. H.K.K. performed electrophysiology experiments. H.T., M.M.G. and P.R.D. performed histological experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks Abhishek Banerjee, Yang Dan, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Predictive negative image hypothesis for sensory habituation.
a, Schematic illustrating the steps of sensory habituation mechanisms proposed by the predictive negative image hypothesis. The exact brain loci where the internal predictive model is stored and how this model filters sensory inputs remain unknown. b, Detailed schematics comparing how two theories explain changes in neural circuit dynamics before and after repeated sensory exposure. c, Summary diagrams of neuronal substrates for the negative image hypothesis proposed in this paper.
Extended Data Fig. 2 Additional data characterizing A1 sensory habituation.
a, Trial-averaged sound-evoked response traces from three cells tracked over five days. Red dots: significantly excited. Blue dots: significantly suppressed. Scale bars: 100% ΔF/F. b, Fraction of significantly excited (top) and suppressed (bottom) cells over five days. n = 20 mice. ****P = 8.9 × 10−5, **P = 2.5 × 10−4 (two-sided Wilcoxon signed-rank test). Solid line: mean. c, Change index of excitatory responses from Day 1 to Day 5 in individual neurons plotted against the difference between their best frequency (BF) and the experienced frequency. Red line indicates a linear regression line. Sensory habituation occurs uniformly regardless of a cell’s frequency tuning. n = 317 significantly responsive cells. R = − 0.074, P = 0.19 (Spearman correlation). d, Sound-evoked response amplitudes (% ΔF/F, averaged over five seconds) on Day 1 and Day 5 for individual neurons significantly excited on Day 1, Day 5, or both (corresponding to Fig. 1f). n = 879 neurons. ****P = 8.2 × 10−74 (two-sided Wilcoxon signed-rank test). Black lines on the right represent mean. e, Laminar profile of the Change index for excitatory (left) and inhibitory (right) sound-evoked responses in individual neurons on Day 5 compared to Day 1. Dots, individual cells. Solid lines represent mean for data binned at 10 μm intervals. f, Change index of excitatory responses from Day 1 to Day 5 in individual neurons, plotted separately for shallow and deep layers. Excitatory: shallow, ****P = 3.0 × 10−46; deep, ****P = 1.7 × 10−16 (two-sided Wilcoxon signed-rank test); shallow vs. deep, P = 0.16 (two-sided Wilcoxon rank-sum test). Inhibitory: shallow, ****P = 1.1 × 10−39; deep, ****P = 3.1 × 10−8; shallow vs. deep, P = 0.20. g, Representative two-photon image of A1 L2/3 neurons expressing GCaMP6s in all neurons and tdTomato in inhibitory neurons. h, Change index of excitatory (red) and inhibitory (blue) sound-evoked responses across days, plotted separately for VGAT− excitatory neurons (left), VGAT+ inhibitory neurons (middle), and neurons from mice without tdTomato (right). 1,882 VGAT− neurons, 896 VGAT+ neurons from 10 VGAT-Cre::Ai9 mice, 2,771 neurons from 10 mice without tdTomato. i, Change index of excitatory (left) and inhibitory responses (right) from Day 1 to Day 5 in individual neurons, plotted separately for VGAT−, VGAT+ neurons, and neurons from mice without tdTomato. Excitatory: VGAT−, ****P = 2.7 × 10−64 (two-sided Wilcoxon signed-rank test with Bonferroni correction), VGAT+, ****P = 7.8 × 10−40; No tdTomato, ****P = 1.6 × 10−44. VGAT− vs. VGAT+, P = 0.51; VGAT− vs. No tdTomato, P = 0.47; VGAT+ vs. No tdTomato, P = 0.98 (two-sided Wilcoxon rank-sum test with Bonferroni correction). Inhibitory: VGAT−, ****P = 4.7 × 10−17; VGAT+, ****P = 8.5 × 10−30; No tdTomato, ****P = 1.4 × 10−26. VGAT− vs. VGAT+, P = 0.27; VGAT− vs. No tdTomato, P = 0.25; VGAT+ vs. No tdTomato, **P = 0.007. Error bars and shades represent mean ± s.e.m.
Extended Data Fig. 3 Across-day habituation in three inhibitory neuron subtypes.
a, Representative two-photon image of A1 L2/3 PV neurons expressing GCaMP6s. b, Averaged Change index for excitatory (red) and inhibitory (blue) sound-evoked responses across days. n = 7 mice, 385 cells. c, Change index of excitatory (left) and inhibitory (right) responses in individual neurons on Day 5 compared to Day 1 across all mice. Lines on the right represent mean. n = 71 significantly excited neurons and 265 significantly inhibited neurons. Excitatory, ****P = 2.2 × 10−10 (two-sided Wilcoxon signed-rank test). Inhibitory, ****P = 2.0 × 10−5. d-f, Same as a-c but for VIP neurons. e, n = 6 mice, 336 cells. f, n = 70 significantly excited neurons and 268 significantly inhibited neurons. Excitatory: ****P = 1.1 × 10−23. Inhibitory: ****P = 9.7 × 10−26. g-i, Same as a-c but for superficial ( < 200 μm from the surface) SST neurons. h, n = 5 mice, 104 cells. i, n = 60 significantly excited neurons and 34 significantly inhibited neurons. Excitatory, ***P = 6.3 × 10−4. Inhibitory, **P = 0.0022. j-l, Same as a-c but for deep ( > 200 μm from the surface) SST neurons. k, n = 7 mice, 253 cells. l, n = 126 significantly excited neurons and 76 significantly inhibited neurons. Excitatory, ****P = 3.8 × 10−15. Inhibitory, ****P = 1.5 × 10−6. m, Laminar profile of the Change index for excitatory (left) and inhibitory (right) sound-evoked responses in individual SST neurons on Day 5 compared to Day 1. Dots, individual cells. Solid lines represent mean for data binned at 10 μm intervals. Excitatory: R = 0.38, P = 7.7 × 10−9. Inhibitory: R = 0.41, P = 9.5 × 10−7 (Spearman correlation; two-sided). n = 12 mice. n, The same as m, but for the comparison between Day 6 (Muscimol) and Day 5. Excitatory: R = 0.57, P = 1.3 × 10−10. Inhibitory: R = 0.26, P = 0.07. n = 8 mice. o, Schematics illustrating the inhibitory neuron circuit dynamics before and after repeated sensory exposure. Error bars and shades represent mean ± s.e.m.
Extended Data Fig. 4 Additional tracing data.
a, Schematic illustrating two-color labeling of VGAT+ inhibitory neurons (red) and retrogradely labeled neurons from A1 (green). b, Coronal section around the injection site in the A1. c, Magnified image of the OFCvl in a coronal section. There is no overlap between GFP+ and tdTomato+ neurons. d, Summary bar plot quantifying the fraction of VGAT+ and VGAT− cells out of retrogradely labeled neurons in the ipsilateral OFCvl. n = 2 mice. e, Schematic illustrating anterograde tracing from OFCvl. f, Coronal section around the injection site in the OFCvl. g, Left: Coronal section showing the laminar distribution of OFCvl axons in the auditory cortex (ACx). Right: Magnified image of L2/3. h, Distribution of EGFP signal intensity across the normalized cortical depth. Solid lines and shadings show mean and s.e.m. n = 4 sections from 2 mice. i, Schematic illustrating retrograde tracing from OFCvl. j, Coronal section around the injection site in the OFCvl. k, Coronal section showing the OFCvl-projecting neurons in the auditory cortex. l, Distribution of mCherry signal intensity across the normalized cortical depth. n = 8 sections from 4 mice. m, Coronal section showing no OFCvl-projecting neurons in the medial geniculate body (MGB). n, Coronal section showing no OFCvl-projecting neurons in the inferior colliculus (IC). o, Count of OFCvl-projecting neurons in the sections of the ipsilateral auditory cortex, MGB, and IC. n = 8 sections for ACx, 8 sections for MGB, 4 sections for IC, from 4 mice. APT, anterior pretectal nucleus; Cb, cerebellum; CIC/DCIC/ECIC, central nucleus/dorsal cortex/external cortex of IC; Hp, hippocampus; MGd/MGm/MGv, dorsal/medial/ventral subdivisions of MGB; PB, parabrachial nucleus; PIL, posterior intralaminar thalamic nucleus; PoT, posterior triangular thalamic nucleus; PP, peripeduncular nucleus; SG, suprageniculate thalamic nucleus. Error bars and shades represent mean ± SEM.
Extended Data Fig. 5 Monosynaptic innervation of A1 SST cells by frontal cortical areas.
a, Schematic illustrating the rabies viral strategies for monosynaptic retrograde tracing from Cre-expressing neurons in A1 L2/3. Red circles: mKate-expressing AAV-infected neurons. Green circles: EGFP-expressing rabies-infected neurons. Yellow circles: starter cells co-expressing mKate and EGFP. b-e, Retrograde tracing from A1 L2/3 pyramidal neurons using Emx1-Cre mice. b, Left: Coronal section of the injection site in A1. Dotted lines: cortical borders. Right: Magnified view of the area indicated by a square in the left image. c, Coronal section of the frontal areas overlaid with area borders. White dots: presynaptic neurons. d, Bar plots summarizing the distribution of input cells in ipsilateral frontal cortical areas. n = 4 mice. e, Further classification of OFC into ventrolateral (OFCvl), lateral (OFCl), and medial (OFCm) subdivisions. f-i, Same as b-e but from A1 SST neurons using SST-Cre mice (n = 4). j-m, Same as b-e but from A1 VIP neurons using VIP-Cre mice (n = 3). n-q, Same as b-e but from A1 PV neurons using PV-Cre mice (n = 3). White dotted lines in the frontal cortical images indicate the boundary of the combined OFCvl and OFCl.
Extended Data Fig. 6 Additional data characterizing the effect of OFC inactivation.
a-b, Additional data from A1 L2/3 pyramidal cell imaging during muscimol infusion into the OFCvl. a, Fraction of significantly excited (left) and suppressed (right) cells over six days. n = 7 mice, 1,177 responsive cells. Excited: *P = 0.031. Suppressed: *P = 0.016. Two-sided Wilcoxon signed-rank test. b, Same as a but for PBS infusion into the OFCvl. n = 3 mice, 439 responsive cells. Excited: P = 0.25. Suppressed: P = 1.0. c, Projection of sound-evoked A1 ensemble trial-to-trial activity dynamics onto Modewithin (left) and Modeacross (right) defined using data from Days 1–5. Data points represent individual trials (100 trials × 6 days) before and after muscimol infusion. The ensemble activity pattern shows partial recovery along Modeacross but not Modewithin following muscimol infusion. n = 7 mice, 734 cells imaged throughout the six days. d, Cosine similarity between Modewithin and Modeacross, indicating a nearly orthogonal relationship. e, Left: Change in sound-evoked ensemble activity along Modewithin between Trial 1 and Trial 10, illustrating prominent within-day, across-trial habituation around tone onset. Right: Change in sound-evoked ensemble activity along Modeacross from Day 1 to Day 5 (green) and from Day 5 to Day 6 (red). Both across-day habituation and its recovery by muscimol slowly ramp up during the sustained tone. f, Summary data showing the Ramp-up Index for across-day plasticity (day 1 to day 5), muscimol-induced recovery (Day 5 to Day 6), and within-day plasticity (Trial 1 to Trial 10). g, Change index for inhibitory responses across blocks of trials during muscimol infusion on Day 1 in naïve mice. Each data point represents a block of 25 trials. Red: muscimol (n = 4 mice). Dark gray: PBS control (n = 6 mice). h, Change index for inhibitory responses in individual neurons in blocks 6–8 compared to blocks 2–4 across all mice. n = 894 and 492 significantly suppressed neurons for PBS and muscimol. P = 0.55 (two-sided Wilcoxon rank-sum test). i, Change index for inhibitory responses in SST, VIP, and PV neurons comparing Day 5 and Day 6. SST, n = 51 responses from 8 mice, P = 0.30; VIP, n = 197 responses from 5 mice, ****P = 3.9 × 10−28; PV, n = 150 responses from 5 mice, ****P = 5.2 × 10−12 (two-sided Wilcoxon signed-rank test). Error bars and shades represent mean ± s.e.m.
Extended Data Fig. 7 Additional data characterizing sound-evoked responses in frontal areas.
a, Summary of all probe penetrations in naïve mice aligned to the Allen Common Coordinate Framework. Probe tracks within M2 and OFC are shown in green and red, respectively. n = 9 penetrations from 9 mice. View angles from top right, right, and front are shown. b, Fraction of single units with significant excitatory responses in the frontal areas of naïve mice. Individual dots represent mice, and bar plot represents mean. c-d, Same as a-b but for recordings in habituated mice. n = 11 penetrations from 8 mice. e, Left: Sound-evoked response traces averaged across significantly excited single units in naïve (black) and habituated (blue) mice. Black bar represents the sound timing. Scale bar: 0.5 Hz. Right: Cumulative plots of sound-evoked spike counts in significantly excited single units. Top: OFCvl (n = 38 and 99 units for naïve and habituated; **P = 0.010). Middle: M2 (n = 32 and 66 for naïve and habituated; P = 0.55). Bottom: other frontal areas, including ACC, FRP, PL, IL, and OFCm (n = 56 and 29 for naïve and habituated; P = 9.8 × 10−4). Two-sided Wilcoxon rank-sum test. OFCvl and M2 data are the same as Fig. 4l. f, Same as e but for all single units, including non-responsive ones. Top: OFCvl (n = 785 and 1203 units for naïve and habituated. *P = 0.027. Middle: M2 (n = 632 and 1,059 for naïve and habituated; P = 0.49). Bottom: other frontal areas (n = 731 and 549 for naïve and habituated; P = 0.054). Scale bar: 0.1 Hz. g, Raster (top) and peristimulus time histogram (bottom) of a representative single unit in OFCvl activated by a laser. h, The fraction of OFCvl single units significantly excited by a laser (n = 5 mice). Error shades represent mean ± s.e.m.
Extended Data Fig. 8 Optogenetic inactivation of OFCvl selectively enhances A1 responses to habituated sounds.
a, Left, Schematics illustrating optogenetic inactivation of OFCvl during two-photon calcium imaging of A1. Right, Expression of ChRmine-oScarlet in the OFCvl GABAergic neurons. b, Electrophysiological confirmation of optogenetic silencing of OFCvl (n = 2 mice). Left, Normalized peristimulus time histograms of tone-responsive multi-units in the OFCvl. Right, Summary plot showing response magnitudes during tone presentations. c, Protocol for OFCvl inactivation before and after habituation. d, Top: Averaged tone-evoked response traces from all significantly responsive cells with (red) and without (black) LED for Tone A (left) and Tone B (right). Bottom: Averaged changes in sound-evoked response traces between LED and No LED trials across all responsive neurons. Insets: The amplitudes of the difference trace at 1 and 5 s after sound onset (n = 7 mice, 1,195 and 1,153 responsive neurons for Tone A and B; ****P = 1.7 × 10−7, two-sided Wilcoxon signed-rank test). Box: 25th to 75th percentiles. Whiskers: 1 × IQR. Black line: median. Red line: mean. Outliers not shown. e, The same as d but for Day 6 (n = 1,327 and 1,340 responsive neurons for Tones A and B; ****P = 7.1 × 10−9). Note that the smaller onset responses to Tone A on Day 6 compared to Day 0 traces are due to the predominance of suppressed cells among responsive cells on Day 6. f, LED-induced change in sound responses, compared between Day 0 and Day 6 conditions. Tone A: P = 2.1 × 10−8, Tone B: P = 9.5 × 10−5 (two-sided Wilcoxon rank-sum test). g, Averaged changes in traces between LED and No LED trials without tone presentation. h, Change index of Tone A excitatory (left) and inhibitory (right) responses in individual neurons in LED compared to No LED trials across all mice (n = 258 excited cells, *P = 0.013; n = 544 inhibited cells, *P = 0.011; two-sided Wilcoxon signed-rank test). i, The same as h but for Tone B (n = 242 excited cells, P = 0.95; n = 536 inhibited cells, P = 0.57). Error bars and shades represent mean ± s.e.m.
Extended Data Fig. 9 Additional data characterizing NMDA receptor knockout experiments.
a-d, Characterization of tonal receptive fields using one-second tone pips in mice with NMDA receptor knockout from A1. a, Normalized response traces to the best frequency tone pips, averaged across all responsive neurons from Grin1flox/flox + AAV-Cre (KO; blue) and control (black) experiments. Bar, tone. b, Fraction of cells with significant excitatory (Exc), inhibitory (Inh), and both excitatory and inhibitory (Exc/Inh) responses across mice. No resp: nonresponding cells. Control: n = 6 mice, 1,101 cells from 6 FOVs; KO: n = 4 mice, 1,275 cells from 5 FOVs. c, Scatter plot showing the response magnitudes of neurons at their best frequencies. n = 236 and 143 responsive cells for control and KO mice. ****P = 1.7 × 10−6. Two-sided Wilcoxon rank-sum test. d, Scatter plot showing the bandwidth of neurons at 70 dB SPL (BW70). P = 0.84. e, Fraction of Cre-positive and Cre-negative neurons in the imaged fields of view, determined by their mCherry expression. n = 905 imaged neurons. f, Grin1flox/flox + AAV-Cre data from Fig. 6b, categorized by the presence (red) or absence (orange) of Cre expression in imaged neurons. Reduced habituation is also observed in Cre-negative neurons, suggesting a network-level effect of habituation. n = 226, 154, and 42 for Control, Cre-positive, and Cre-negative. Control vs. Cre-positive: ***P = 8.4 × 10−4. Control vs. Cre-negative: ****P = 3.0 × 10−5. Cre-positive vs. Cre-negative: P = 0.091. Two-sided Wilcoxon rank-sum test with Bonferroni correction. g, Top: Coronal section around the AAV injection site, showing NMDA receptor immunostaining. Bottom: Fluorescence intensity quantified along the dotted rectangle in the top image. h-k, Same as a-d but for mice with NMDA receptor knockout from SST neurons. i, Control: n = 6 mice, 1,095 cells from 6 FOVs. KO: n = 5 mice, 1,157 cells from 5 FOVs. j, n = 243 and 106 responsive cells for control and KO mice. P = 0.72. k, P = 0.13. l-o, Same as a-d but for mice with NMDA receptor knockout from VIP neurons. m, Control: n = 5 mice, 921 cells from 5 FOVs. KO: n = 4 mice, 1,256 cells from 6 FOVs. n, n = 191 and 154 responsive cells for control and KO mice. P = 0.14. o, P = 0.76. Error shades represent mean ± s.e.m.
Extended Data Fig. 10 Tones at 60 dB SPL induce habituation comparable to that at 70 dB SPL.
a, Distributions of excitatory response magnitudes across conditions: 70 dB SPL in WT mice (n = 20 mice, 1,241 neurons), 70 dB SPL in GluN1 KO mice (n = 4 mice, 267 neurons), and 60 dB SPL in WT mice (n = 3 mice, 153 neurons). 70 dB WT vs. 70 dB GluN1 KO: P = 4.7 × 10−8; 70 dB WT vs. 60 dB WT: P = 0.0093; 70 dB GluN1 KO vs. 60 dB WT: P = 0.46; two-sided Wilcoxon rank-sum test with Bonferroni correction. b, Change index of excitatory (left) and inhibitory (right) responses in individual neurons after five days of tone exposure at 70 dB SPL and 60 dB SPL. (Excitation: 70 dB, P = 2.9 × 10−88; 60 dB, P = 5.1 × 10−13, two-sided Wilcoxon signed-rank test; 70 dB vs. 60 dB, P = 0.92, two-sided Wilcoxon rank-sum test; Inhibition: 70 dB, P = 5.7 × 10−60; 60 dB, P = 2.9 × 10−18; 70 dB vs. 60 dB, P = 0.0036). c, Histograms of changes in response magnitudes for all neurons in the 60 dB SPL habituation experiments. Orange and green bars show neurons with significant potentiation and suppression. ****P = 5.0 × 10−21, two-sided Fisher’s exact test. Error bars represent mean ± s.e.m.
Supplementary information
Supplementary Information (download PDF )
Supplementary Discussion.
Source data
Source Data (download XLSX )
Statistical source data for all figures and extended data figures.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tsukano, H., Garcia, M.M., Dandu, P.R. et al. Orbitofrontal cortex drives predictive filtering of sensory responses. Nat Neurosci 29, 888–900 (2026). https://doi.org/10.1038/s41593-026-02217-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41593-026-02217-z