Abstract
Industrialization and human urbanization have led to an increase in heavy metal (HM) pollution which often cause negative/toxic effect on agricultural crops. The soil-HMs cannot be degraded biologically however, microbe-mediated detoxification of toxic HMs into lesser toxic forms are reported. Considering the potentiality of HMs-tolerant soil microbes in metal detoxification, Pseudomonas fluorescence PGPR-7 and Trichoderma sp. T-4 were recovered from HM-affected areas. Under both normal and cadmium stress, the ability of both microorganisms to produce different plant hormones and biologically active enzymes was examined. Strains PGPR-7 and T-4 tolerated cadmium (Cd) an up-to 1800 and 2000 µg mL−1, respectively, and produced various plant growth regulating substances (IAA, siderophore, ACC deaminase ammonia and HCN) in Cd-stressed condition. The growth promoting and metal detoxifying ability of both strains were evaluated (either singly/combined) by applying them in chickpea (Cicer arietinum L.) plants endogenously contaminated with different Cd levels (0–400 µg kg−1 soils). The higher Cd concentration (400 µg kg−1 soils) negatively influenced the plant parameters which, however, improved following single/combined inoculation of P. fluorescence PGPR-7 and Trichoderma sp. T-4. Both microbial strains increased the growth of Cd-treated chickpeas however, their combined inoculation (PGPR-7 + T-4) caused the most positive effect. For instance, 25 µg Cd Kg−1 + PGPR-7 + T4 treatment caused maximum increase in germination percentage (10%), root dry biomass (71.4%) and vigour index (33%), chl-a (38%), chl-b (41%) and carotenoid content (52%). Furthermore, combined inoculation of P. fluorescence PGPR-7 and Trichoderma sp. T-4 maximally decreased the proline, MDA content, POD and CAT activities by 50%, 43% and 62%, respectively following their application in 25 µg Cd kg−1 soils-treated chickpea. Additionally, microbial strains lowered the plant uptake of Cd. For example, Cd-uptake in root tissues was decreased by 42 and 34% when 25 µg Cd Kg−1- treated chickpea plants were inoculated with P. fluorescence PGPR-7, Trichoderma sp. T-4 and co-inoculation (PGPR-7 + T4) of both strains, respectively. Therefore, from the current observation, it is suggested that dual inoculation of metal tolerant P. fluorescence and Trichoderma sp. may potentially be used in detoxification and reclamation of metal-contaminated soils.
Similar content being viewed by others
Introduction
In today’s agriculture, heavy metals (HMs) pollution is regarded as the most serious environmental issue1. Due to various human and natural activity, ecosystems have become poisoned by heavy metals2. The following activities largely contributed to the release of heavy metals into the environment: metal mining and smelting, burning of fossil fuels, fertilizers, sewage sludge, pesticides, municipal wastes, pigment production, wasted batteries, and vehicle exhaust3. In moderation, HMs can be utilized as plant nutrients (Cu, Zn, Fe, Mn, Ni, and Co are necessary components); nevertheless, higher HMs concentration in soil often cause severe toxic effects on edible crops4 and thus, reduced the agricultural productivity5.
These HMs are taken excessively by plant roots and transported to shoots and other plant organs. These translocations affected the metabolism and caused the stunted growth in the shoots6. Also, HM pose a negative effect on morphological7, physiological8 and biochemical9 properties as well as nutritional uptake10 and symbiotic features11 of leguminous crops including chickpeas. In addition, HMs caused a significant reduction in soil microbial activity, soil fertility and yields losses12.
HMs present in soil cannot degraded, but can only be converted to organic compounds through a biological process13. Therefore, it is necessary to choose plants, which interact with bacteria and plant roots, and investigate adequate metal concentrations in soil to carry out successful phytoremediation operations. Metal-resistant microorganisms can aid in the removal of metals from soil. In this regard, various metal-tolerant plant growth promoting bacteria14,15,16 and fungi17,18,19 isolated from different metal-contaminated areas are reported to detoxify the HMs toxicity. By producing/ synthesizing various growth regulating active biomolecules, these metal tolerant microbes increase the growth and yield of metal-polluted crops. For instance, growth of cadmium supplemented Sedum alfredii (L.) were enhanced by inoculating Cd-tolerant and endophytic bacterium Buttiauxella sp. SaSR13. The strain further decreased the concentration of cadmium in root and shoot tissues20. Furthermore, Bacillus sp. ZC3-2-1, a Zn and Cd tolerating PGPR strain improved the growth and ionic homeostasis in rice plants treated separately with heavy metals. Additionally, bio-inoculation of this bacterium enhanced the phytoextraction of Cd and metal immobilization14. Compared to un-inoculated treatment, the application of Cd-resistant strain decreased the bioavailability of Zn (32%) and Cd (93.3%) in rice seedlings.
It is widely reported that fungal strains have the ability to tolerate high HMs ions concentrations, play a crucial role in metal detoxification/reclamation21 and plant growth promotion22 and which constitutes a biological barrier against the transfer to the shoot tissues. Till date, a number of metals and other stress tolerant plant growth promoting fungi (PGPF) have been reported as stress reliever and growth promoter. For example, in a study, Cr and As-tolerating fungus Aspergillus welwitschiae strain Bk was isolated and recorded to synthesize phytohormone, solubilized insoluble P and produced stressor metabolites17. Following inoculation, strain Bk enhanced the root and shoot length of metal-affected Glycine max (L.). Also, Bk supported the antioxidative responses of metal-supplemented G. max and increased the CAT (1.58-fold increase), AAO (6.75–7.94-fold increase), POD (1.37-fold) and 1,1-diphenyl-2-picrylhydrazyl; DPPH (1.42-fold increase) activities of plant. Furthermore, strain Bk lowered the uptake of chromium (Cr) and arsenic (As) by 50% in plants and thus, lowered the metal phytotoxicity. In another study, multi-metal tolerant Aspergillus flvaus strain CR-500 had multifarious biochemical, physiological and growth regulating properties. The strain CR-500 potentially removed the metals such as As (99%), Pb (97.7%) and Ni (73.1%) by the mean of bioaccumulation and surface adsorption mechanisms. A. flavus CR-500 also exhibited excellent multi-metal (As, Ni, Pb and Cr) removal potential from simulated wastewater (SWW) and Tannery wastewater (TWW) with a high amount of biomass production rate23. The metal tolerant microbes hold a complex mechanism to adapt to metal stresses (a) binding of Cd by the elements of the cell wall, as well as produced siderophores and exopolysaccharides, to prevent it from entering the cell; (2) metalloproteases sequester Cd intracellularly; (3) Cd is eliminated from the cell through efflux pumps; (4) antioxidants reduce the harmful effects of Cd. However, only a few reports are available on dual/combined inoculation effect of metal tolerant microbes. So, considering these, the current project was undertaken with following specific objectives: (i) assessment of tolerance of different among bacterial and fungal isolates (ii) evaluation of PGP activities of P. fluorescence PGPR-7 and Trichoderma sp. T4 strains under Cd-stressed condition (iii) single/combined inoculation effect of metal tolerant microbes on chickpea raised in pot soils contaminated with different level of Cd (iv) Effect of Cd-tolerant PGPR-7 and T4 strains (single and dual) on growth, dry biomass and photosynthetic pigments of Cd-treated chickpea (v) assessing the dual inoculations of P. fluorescence PGPR-7 and Trichoderma sp. T4 strains on antioxidative response of Cd-treated chickpea (vi) evaluating the uptake of metal in Cd-treated and microbial strain inoculated chickpea.
Material and methods
Isolation of microbial isolates
By using the serial dilution procedure, the rhizosphere microbial isolates were recovered from rhizosphere soils of vegetables field irrigated with industrial/wastewater. The isolation of soil bacteria was accomplished using the nutrient agar medium/king’s B medium. Aseptically adding one mL of soil suspension from dilutions (10–5 and 10–6) to sterile Petri plates with ten mL of sterile media and incubating for 48 h at 28 °C. Following incubation, distinct individual colonies with yellow, green, and white pigments were identified. The isolated cultures were obtained by picking up the individual colonies with sterile loops, transferring them to Nutrient agar media slants, and then storing them in a refrigerator at 40 °C for later use. In a similar fashion, fungal isolates Trichoderma were recovered using serial dilutions (10–3 and 10–4) that were aseptically added to sterile Petri plates that contained 20 mL of Potato Dextrose agar (PDA) medium and incubated 37 °C for 3 days. A total of 20 bacterial and 15 fungal isolates were recovered and assessed for their cultural and biochemical properties.
In-vitro heavy metal testing of microbial isolates
In order to evaluate the heavy metal (HM) tolerant ability, recovered isolates were grown on Nutrient agar (NA; for bacterial isolates)24 and potato dextrose agar (PDA; for fungal isolates). Both of solid growth media were pre-supplemented with increasing doses (0–2400 µg mL−1) of salts of each chromium (Cr), cadmium (Cd). Lead (Pb), arsenic (As) and nickel (Ni)25. The freshly grown microbial (bacteria and fungi) isolates were spotted on metals-amended plates. Plates were incubated at 28 ± 2 °C for 48 h (for bacterial isolates) and 72 h (for fungal isolates) and observed for bacterial survivability. Bacterial and fungal isolates surviving the maximum level of metals were recorded as metal tolerant isolates. Of the total bacterial isolates (N = 20) and fungal isolates (N = 15), strain PGPR-7 and T-4 tolerated maximum concentrations of metals and therefore, selected for additional studies. All the isolates were morphologically and biochemically characterized.
Molecular identification of bacterial isolate
Isolate PGPR-7 showed the maximum tolerance towards the HMs. Therefore, it was decided to identify the isolate to genus level. For this, DNA was isolated using a PGPR-7 culture that had been cultivated overnight. A microcentrifuge tube containing 2.0 ml of the bacterial suspension was filled with the pellet and centrifuged for two minutes at 10,000 × g. To obtain an adequate number of cells, the aforementioned procedure was carried out twice with 2.0 ml of bacterial suspension. In order to release the genomic DNA, 0.2 ml of protease was added after the cells had been rinsed with 0.9% saline. This process digested and removed the protein and cellular components from the cells. The centrifuged tubes were inverted five to six times and heated to 55 °C in a water bath for one hour. One hour later, 0.1 ml of the DNA Salt solution was added, and it was then centrifuged at 5,000 rpm for five minutes. The centrifuge tube was then repeatedly inverted to thoroughly mix the components before 0.8 ml of the precipitated solution was progressively added. The cells were washed with 70% ethanol. Dried and suspended in TE buffer, the obtained DNA was kept at 4 °C. A 8% agarose gel was used to verify the extracted DNA's purity. Utilizing universal primers, the 16S rRNA gene fragments were amplified. Through the use of a DNA sequencing service, amplified 16S rRNA gene fragments were purified. The 16S rRNA gene sequences were collected and submitted to BLAST, a programme that searches local alignments for nucleotide sequences that match those in reference sequences.
Assessment of PGP traits of microbial strains under Cd stress
Evaluation of siderophore and indole-3-acetic acid production
For qualitative estimation of siderophore, FeCl3 test was performed by growing the microbial cultures in nutrient broth (NB) added with 0–400 µgCdmL−126. Additionally, the production of siderophores were assessed using the previously described method27, which involved spot-inoculating of freshly grown cultures of strain PGPR-7 and T-4 onto chrome azurol S (CAS) agar plates that had been amended with different concentrations of Cd. The plates were examined for the development of a yellow to orange zone (halo), which indicates the release of siderophores, around bacterial growth after incubation. Additionally, employing Modi media, a quantitative assessment of siderophores was carried out. For this, 100 µL of microbial cell suspension was added to Modi medium that had been supplemented with 0–400, µg mL−1 of Cd. The inoculated tubes underwent a 5-day incubation period at 28 ± 2 °C. The salicylate (SA) and 2, 3-dihydroxybenzoic acid (DHBA), catechol type phenolates, were then measured in the supernatant after centrifuging the cultures at 6000 rpm.
By using the modified protocol of Mayer28, the IAA secretion by the metal-tolerant bacterial strain PGPR-7 and T-4 was measured. In order to conduct the test, 100 mL of overnight-grown culture was added to 25 mL of Luria Bertani (LB) broth that had already been supplemented with 100 µg of tryptophan per mL. It was then exposed to 0, 25, 50, 100, 200, and 400 µg Cd mL−1. After inoculation, the cultures underwent a 48-h incubation period at 28 ± 2 °C and 125 rpm in a shaking incubator. After incubation, 4 mL of Salkowsky reagent and 2–3 drops of orthophosphoric acid were added to the supernatant from a centrifugation of 2 mL of each treatment's culture at 10,000 g for 15 min. For the purpose of colour development, samples were left in the dark at 28 ± 2 °C for one hour. Using pure IAA as a reference, the amount of IAA released in the supernatant was calculated at 530 nm by measuring the absorbance of the pink colour that emerged during the reaction.
ACC Deaminase production
By cultivating and spot inoculating microbial cultures on Dworkin and Foster (DF) salts minimum medium enriched with 3.0 mM ACC as the main N source, it was possible to determine the ACC deaminase generated by the metal-tolerant strains PGPR-7 and T-4 in a qualitative manner. The negative and positive control plates were made with DF agar plates that contained (NH4)2SO4 (0.2% w/v) but no ACC. The plates underwent a 72-h incubation period at 28 ± 2 °C, during which time bacterial growth was observed. The modified approach of Penrose and Glick29 and Honma and Shimomura30 was applied to quantitatively estimate the ACC deaminase. By measuring the absorbance of samples against a standard curve of pure α-ketobutyrate, the amount of α-ketobutyrate released as a result of ACC breakdown was determined. The ACC deaminase activity was expressed as μ mol α-ketobutyrate mg−1 protein h−1.
Ammonia and hydrogen cyanide (HCN) production
Ammonia production activity of metal tolerant microbial strains (PGPR-7 and T-4) was assessed under metal-stressed condition. For the assay, freshly grown microbial strains were inoculated in peptone water amended with 0–400 μg Cd mL−1 incubated for 4 days at 28 ± 2 °C31. The colour changed from yellow to reddish brown, indicating a favourable response. Further, cyanogenic compound released by PGPR-7 and T-4 was detected following the method of Bakker and Schipper32. In order to do this, King's B agar plates containing 4.4 g L−1 glycine and supplemented with 0, 25, 50, 100, 200 and 400 µgmL−1 of Cd were spread plated. Filter paper discs coated in 0.5% picric acid and made with 2% sodium carbonate were used to seal the Petri plate lids. To prevent the bacterial strain's volatile HCN from escaping, parafilm was used to seal each experimental plate. The plates were then incubated for 4 days at 28 ± 2 °C. Positive response was said to have occurred when the yellow filter paper turned orange.
Assessing the effect of metal tolerant microbial strains on chickpea raised in pot soils treated with Cd
Germination efficiency and growth parameters
For the purpose of evaluating how Cd treated chickpea plants react in the presence of single/combined inoculations of metal tolerant microbial strains (PGPR-7 and T-4). Uniform and healthy chickpea seeds were surface sterilised with 4% sodium hypochlorite (NaOCl) for 3 min. The seeds were then rinsed with 6–7 changes of sterile water. The seeds were sterilised before being immersed in metal-tolerant P. fluorescence PGPR-7 and Trichoderma asp. T-4 (single/dual inoculation) using 1% guar gum powder as an adhesive to deliver about 108 cells per seed. Additionally, the uncontaminated seedlings that were just steeped in sterile water served as a control. In 3.0 kg of non-sterilized alluvial sandy clay loam soil, the bio-primed and uninoculated seeds were planted in earthen pots. Two days before the seeds are sown, different concentrations (0, 25, 50, 100, 200 and 400 mg/kg soils) were added to soil. To replicate the quantities seen in the soil samples analysed, these Cd doses were used for the experimental trials. There were 23 treatments with three replications each and was as follows:
T1 = Control (un-inoculated), T2 = P. fluorescence PGPR-7, T3 = Trichoderma sp. T-4, T4 = 25 µg Cd kg−1 soil, T5 = 50 µg Cd kg−1 soil, T6 = 100 µg Cd kg−1 soil, T7 = 200 µg Cd kg−1 soil, T8 = 400 µg Cd kg−1 soil, T9 = 25 µg Cd kg−1 soil + P. fluorescence PGPR-7, T10 = 50 µg Cd kg−1 soil + P. fluorescence PGPR-7, T11 = 100 µg Cd kg−1 soil + P. fluorescence PGPR-7, T12 = 200 µg Cd kg−1 soil + P. fluorescence PGPR-7, T13 = 400 µg Cd kg−1 soil + P. fluorescence PGPR-7, T14 = 25 µg Cd kg−1 soil + Trichoderma sp. T-4, T15 = 50 µg Cd kg−1 soil + Trichoderma sp. T-4, T16 = 100 µg Cd kg−1 soil + Trichoderma sp. T-4, T17 = 200 µg Cd kg−1 soil + Trichoderma sp. T-4, T18 = 400 µg Cd kg−1 soil + Trichoderma sp. T-4, T19 = 25 µg Cd kg−1 soil + P. fluorescence PGPR-7 + Trichoderma sp. T-4, T20 = 50 µg Cd kg−1 soil + P. fluorescence PGPR-7 + Trichoderma sp. T-4, T21 = 100 µg Cd kg−1 soil + P. fluorescence PGPR-7 + Trichoderma sp. T-4, T22 = 200 µg Cd kg−1 soil + P. fluorescence PGPR-7 + Trichoderma sp. T-4, T23 = 400 µg Cd kg−1 soil + P. fluorescence PGPR-7 + Trichoderma sp. T-4.
A completely randomised block pattern was used to place the pots. Only two plants per container were kept alive until harvest fifteen days after the plants were thinned. The pots were maintained in open-air settings and periodically watered with tap water. To verify the accuracy and reproducibility of the results, the experiments were conducted repeatedly over the course of two years.
Photosynthetic pigments estimation
Fresh leaf samples (0.5 g) from each replicated Cd-treated and microbial inoculated treatment were homogenised in 10 mL of 80% methanol. The samples were kept at 4 °C overnight after being centrifuged for 10 min. at 12,000 rpm. Utilizing a UV visible spectrophotometer, the absorbance of the extract was measured at 663, 645, and 480 nm for the amounts of chlorophyll a, b, and total carotenoid33,34. The following equations were used to determine the chlorophyll and carotenoid content of plants;
Estimation of proline and MDA content
The method of Bates et al.35 and previously done by Shahid et al.36 was followed to assess the proline contents in the fresh leaves of plants that had been either treated with increasing Cd concentrations and inoculated with metal tolerant microbial strains. In a nutshell, 5.0 mL of 3% (w/v) aqueous sulfosalicylic acid were used to homogenise 1.0 g of leaf samples. The homogenates were filtered through Whatman No. 2 filter paper and centrifuged at 8000 rpm for 20 min before being treated with acid ninhydrin (2.0 mL) and glacial acetic acid (2.0 mL) at 80 °C for one hour before being stopped in an ice bath. The colourful complex that formed was extracted with 4 mL of toluene, and absorbances at 520 nm were measured.
An evaluation of the MDA concentration was used to estimate the amount of lipid peroxidation. In this instance, 1.0 g of fresh tissue was homogenised in 10 mL of 10% (w/v) TCA and centrifuged for 10 min at 10,000 rpm. The supernatant was added to a 10% trichloroacetic acid (TCA) solution that included 0.5% of 2-thiobarbituric acid (TBA) and 1% polyvinylpyrrolidone (PVP) in an equal proportion. The sample was incubated for 30 min at 95 °C, immediately cooled in an ice bath, then centrifuged for 15 min at 10,000 rpm. In order to account for nonspecific absorbance, the absorbance was measured at 532 nm and corrected at 600 nm. Using an extinction value of 155 mM−1 cm−1 as M (MDA)g−1 FW, the concentration of MDA was determined.
Antioxidant enzymatic activities
1.0 g of fresh root and leaf samples were pulverised in liquid nitrogen with a mortar and pestle. The ground materials were homogenised on ice in 10 ml of a solution containing 50 mmol/l potassium phosphate buffer and 1% (w/v) PVP (pH 7.8), and the mixture was then maintained at 4 °C for 10 min. Filtering and centrifuging the homogenates at 4000 × g for 15 min. at 4 °C. After then, the antioxidant enzyme activity of the supernatant was evaluated. On a blank that didn't include any protein extract, each enzyme's activity was tested.
Extracts for the POX enzyme was made by combining 250 mg of crushed plant material with 1.2 mL of a 50 mM Tris–HCl buffer (pH 7.3) containing 0.5 M of CaCl2 and 5.0 mM of -mercaptoethanol, and then incubating the mixture at 4 °C for an hour. A crude extract of POX was made from the supernatant after the homogenate underwent centrifugation (14,000 × g 4 °C, 45 min). The homogenate was centrifuged (14,000 × g, 4 °C, 45 min) to produce a supernatant, and the supernatant was used to create a crude extract of POX37,38,39.
Following the breakdown of H2O2, the catalase (CAT) (EC 1.11.1.6) activity was measured as a reduction in absorbance at 240 nm for 1 min. The reaction mixture (total volume: 3 ml) contained the following ingredients: 20 µL of crude enzyme extract in 50 mM phosphate buffer (pH 7.0), 15 mM H2O2, and 25 °C. A blank solution of potassium phosphate buffer (50 mmol/L, pH 7.0) was used to measure the decrease in H2O2 absorbance during the 2 min experiment.
Assessment of inoculation effect of metal tolerant microbes on yield attributes and nutrient uptake in chickpea
At harvest, the grain attributes (seed number, seed weight and seed protein) were estimated in Cd-treated and microbial inoculated plants. The nutritional (nitrogen; N and phosphorous; P) content accumulated in metal treated and microbe-inoculated chickpea seeds were determined following the method of Jackson40 and Iswaran and Marwah41, respectively.
Inoculation effect of tolerant strains on Cd uptake
The uptake of Cd in dried organs (root and shoot) of bio-inoculated and cadmium-treated chickpea was analysed42. In order to do this, 100 mg of dried plant samples were digested separately in a mixture (ratio of 4:1) of HNO3 (nitric acid) and HClO4 (perchloric acid). After being digested, the residual suspensions were filtered through Whatman no. 2 filter paper and the volume was made up to 100 mL with double distilled water (ddH2O) and the content of Cd in each sample was determined by atomic absorption spectrophotometer (AAS) employing CRM standards (JT Baker, BAKER INSTRA-ANALYZED Reagent Grade for Trace Metal Analysis) Cd [1000 μg mL−1 (0.10% w/v)].
Statistical analysis
Each in-vitro and in-vivo experiments were designed/arranged and conducted in three replicates (n = 3) and arranged in a completely randomized design (CRD) factorial. The database for the measured parameters and findings was made using Excel sheets. To examine the gathered data, Statistics 8.1 was used to perform an analysis of variance. Results were statistically analysed using Minitab-17.1 software and One-way ANOVA. Additionally, Duncan's Multiple Range Test (DMRT) was performed on data at a 5% confidence level to compare the treatment means.
Results and discussion
Isolation of microbial culture, tolerance of heavy metals and molecular identification
Globally, heavy metal contamination is a significant obstacle to the sustainability of the food supply since it frequently affects plant growth, nutrient intake, photosynthesis, and other physiological functions. In order to re-solve this issue, we tried to isolate HMs tolerant microbes (bacteria and fungi) that may be utilised as a microbial resource to enhance the performance and yield of crops in metal-stressed conditions. Here, a total of 20 rhizosphere bacteria and 15 rhizosphere fungi were collected and assessed for their cultural and biochemical features. Additionally, the ability of each microbial isolate to produce/synthesize active biomolecules that promote plant development was evaluated. Both of the strains were found as strong siderophore producer and a considerable amount of phenolate siderophore was quantified (Fig. 1). Furthermore, all the recovered bacterial and fungal isolates exhibited a variable tolerance to each metal. Among the bacterial isolates, strain PGPR-7 tolerated a maximum concentration of Cd (800 µg mL−1), Cr (1000 µg mL−1), Pb (800 µg mL−1) and Ni (1200 µg mL−1). Moreover, Trichoderma T-4 isolate among fungi tolerated metals and survived even in the presence of 1000, 1200, 800 and 1400 µgmL−1 each of Cd, Cr, Pb and Ni, respectively (Table 1). The bacterial strain PGPR-7 was molecularly characterized using 16S rRNA sequencing and identified as Pseudomonas fluorescence. A phylogenetic tree was constructed based on the similar sequences obtained from NCBI data base (Fig. 2).
Pseudomonas PGPR-7 strain under UV light showing fluorescent (A). Ferric chloride (FeCl3) test indicating the production of siderophore (A1). Zone of halo formed by PGPR-7 strain on CAS gar plate (A2), and quantification of phenolate siderophore (A3). Trichoderma culture on PDA plate (B), FeCl3 test for siderophore production (B1). Panels B2 and B3 depicting the siderophore production ability of Trichoderma sp. T4.
Neighbor-joined phylogenetic tree of Pseudomonas fluorescence PGPR-7. The tree was constructed based on 16S rRNA partial gene sequence and closely related phylogenetic species (type cultures) derived using the NCBI BLAST search tool. Sequences were aligned using Clustal W sequence alignment tool in MEGA 7.0 software. The GenBank accession numbers of isolates and closely related species are presented in brackets.
The capacity of this strain to withstand high metal concentrations is an intriguing feature since such microbes being may be utilised as an inoculant to increase crop productivity even when exposed to metal stress. Like the current observation, soil isolates recovered from metal polluted soil have been reported to displayed a dissimilar heavy metal tolerance profile24. Similarly, PGPR strain like Bacillus sp., Cupriavidus sp., Cupriavidus sp., Fulvimonas soli and Novosphingobium sp., isolated from rhizosphere soil tolerated heavy metal concentrations43. In yet another study, Fahsi and co-workers44 reported that Enterobacter hormaechei J-146 could withstand up to 1.5 mg L−1 of Cu/Cd, while B. halotolerans J-143 could tolerated up to 1.5 mg L−1 of Ni. Furthermore, P. frederiksbergensis J-158, was found to be the least metal tolerating microbe. Furthermore, numerous metal tolerant fungi like Alternaria sp., Bipolaris zeicola45, Trichoderma brevicompactum25 and Aspergillus welwitschiae17 have been reported for their crucial role in metal-clean up and crop enhancement. Global discussion and investigation surround the reason why microbial populations vary so greatly in their capacity to withstand such dangerous contaminants like heavy metals. According to some research, certain heavy metals cause different microorganisms to behave differently when it comes to metal tolerance. The reason behind could be (a) the variations in nutritional elements (macro- and micro) that are present in soils and growing medium that support bacterial development (b) dissimilarities in genetic constituents of microbial strains and (c) growth conditions/environmental variables affecting the growth of microorganisms46. Additionally, metal-tolerant microbes, which might be used as a bioremediation tool, have the ability to complex metals inside of cells, expel metal ions outside of cells, and enzymatically degrade/transform specific metal ions. These are some of the methods through which metal-tolerant bacteria can prevent the negative effects of metals on soil and plant health, hence reducing the risks to human health, whether they are used separately or in tandem. The metal tolerant microbial strains PGPR-7 and T-4 were subsequently characterized and molecularly identified to species level. Strain PGPR-7 was a Gram-positive, rod-shaped bacteria that fluoresced green on nutrient agar plates. The morphological and biochemical features of the plant were evaluated. According to its biochemical and cultural characteristics, PGPR-7 belonged to the genus Pseudomonas, and the 16S rRNA gene sequencing revealed that it was Pseudomonas at the species level. The 16S rRNA nucleotide sequence of strain PGPR-7 was deposited to GenBank. The bacterium PGPR-7 was identified as Pseudomonas fluorescence after a similarity search using the BLASTn programme showed that the two were closely related (99.64% sequence identical). Given that the study of the 16S rRNA gene sequence is a widely used technique for identifying bacterial strains, a phylogenetic tree was constructed using MEGA 7.0 software.
Growth regulators of PGPR-7 and T-4 strains under Cd stress
Siderophores and IAA
The metabolically important class of substances called siderophores is released by soil bacteria and fungi, and they chelate iron from the medium to prevent plant pathogens from absorbing it47,48. So, siderophores indirectly support plant development and survival in metal-polluted soils by reducing the number of pathogens49,50. The microbial strain PGPR-7 and T4 employed in this investigation was capable of producing an 19.0 mm and 25.0 mm zone on CAS agar medium, respectively and synthesising siderophores in the absence of metals (Table 2). When cultured in liquid medium as opposed to solid medium, both microbial strains continued to produce siderophores even when exposed to Cd stress. PGPR-7 produced 9.23 μg mL−1 SA and 5.51 μg mL−1 2, 3-DHBA when cultured in the presence of 25 μg Cd mL−1, which were 3% (Salicylate) and 2% (2,3-DHBA) reduced over untreated (0 μg Cd mL−1) control. Similarly, Cd at 200 μg mL−1 decreased the fungal siderophore production of by 14.4% (2,3-DHBA) and 211.6% (SA) while comparing control (Table 2). It's interesting to note that both metal tolerant microbial strains continued to produce siderophores at all concentrations of Cd, despite the fact that the levels were relatively insufficient at higher metal doses. Like our investigation, various metal-resistant and siderophore synthesizing soil bacteria such as Bacillus aerophilus51, Pseudomonas putida52, Enterobacter sp.53 and fungi like Piriformospora indica54, Trichoderma virens55, Aspergillus aculeatus56 are reported to augment the growth of different crops endogenously polluted with heavy metals.
Even when cultivated in soils with metal contamination, the IAA generated by soil microbiota has been shown to affect the plant growth by speeding up cell division and enlargement as well as causing root elongation and apical dominance in plants. So, considering this significance, the amount of IAA synthesized by P. fluorescence PGPR-7 and Trichoderma sp. T-4 under Cd-stressed condition was assessed. IAA was synthesized at its highest levels by strain PGPR-7 (123.1 µg mL−1) and T-4 (72.3 µg mL−1) when grown in media free of Cd among other plant growth promoters which, however, decreased by 17 and 22%, respectively when grown in LB broth was treated with 200 µg CdmL−1 (Table 2). Comparing the effects of different Cd concentrations on IAA generation by microbial strains, the highest measured concentration had the deadliest effect. Therefore, it was clear from the current results that both microbial strains maintained the plants' release of IAA into the outside environment even when growing at greater concentrations of Cd. In contrast to normal metal-free conditions, the amount of IAA that diffused externally was worse under metal stress. Similar to the tested bacterial and fungal strains, it has been discovered that numerous bacteria44,57,58 and fungi17,59 inhabiting rhizospheres produce indole acetic acid and have a favourable impact on a number of crucial physiological processes in plants. Therefore, this metal tolerant strain's capacity to promote IAA in addition to its ability to alleviate heavy metal stress could be a compelling choice for encouraging wheat plant growth even in metal-stressed conditions.
HCN and ammonia
A significant element that indirectly promotes plant development by stopping the formation of pathogens is hydrogen cyanide (HCN), a volatile secondary metabolite generated by numerous bacteria. Numerous soil bacterial and fungal species can produce HCN even when under metal pressure, which could greatly aid in boosting crop output in metal contaminated soil. Here, both microbial strains (PGPR-7 and T-4) could secrete HCN in metal free as well metal-stressed condition. Our findings are supported by the fact that various bacterial taxa, including Bacillus sp., and Pseudomonas sp. has a strong historical record of HCN production under stressed environment. Similarly, different doses of Cd had no discernible impact on ammonia produced by both of the microbial strains. However, the higher concentration of Cd lowered the production of ammonia, this could be seen from the way the colour of the resulting peptone water changed after being incubated. And, Cd at 200 μg mL−1 completely reduced the NH3 production activity of Trichoderma sp. T-4.
ACC deaminase
Numerous soil microbes including bacteria60,61,62 and fungi63,64, release the ACC deaminase enzyme, which is crucial in reducing ethylene levels in higher plants and promoting plant development even in stressful conditions. In light of this, the ability of both microbial strains to synthesise ACC deaminase was examined under metal stressed situations. The P. fluorescence PGPR-7 and Trichoderma sp. T-4 could produce 13.9 and 21.4 µmol α-ketobutyrate mg/g protein/h ACCD under metal-free environment. Even when different level of Cd was present, PGPR-7 and T-4 strains demonstrated ACC deaminase activity; however, this activity continually decreased as Cd concentrations increased. Among the Cd concentrations, 200 µg mL−1 induced a significant reduction (24%) fall in ACC deaminase activity of PGPR-7 strain in comparison to the untreated control (Table 2). It also showed the greatest reduction in α-ketobutyrate concentration. The concentration of α-ketobutyrate, in contrast, decreased to 17.2 mol mg/g protein/h in the presence of 200 µgCdmL−1 as opposed to very high production of ACCD (21.4 mol mg/g protein/h) by Trichoderma sp. T-4 (Table 2). It's interesting to note that the ability of both microbial strains to synthesise α-ketobutyrate in the presence of cadmium revealed that ACC deaminase coding genes of both soil microbes were expressed under metal-stressed condition. Other researchers have also noted that this specific property of microbial strains producing ACC deaminase enzyme even while under metal stress may be advantageous for promoting the development and subsequent production of agronomic crops even in metal-contaminated soils.
Single/dual inoculation effect of P. fluorescence PGPR-7 and Trichoderma sp. T-4 on chickpea raised in Cd-stressed soil
Germination, biometric characteristics and photosynthetic molecules
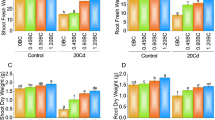
The germination and growth of chickpea plants inoculated with metal tolerant microbial strains and varying level of Cd was variable. Increasing cadmium doses decreased the chickpea germination. For instance, Cd at 400 µg kg−1 reduced the germination of chickpea seedlings by 33.3% over untreated control. In contrast, while using metal tolerant microbes as bio-inoculant, PGPR-7 and Trichoderma sp. T-4 strains increased the germination efficiency of 25 µg Cd kg−1 soil treated chickpea by 10% and 11% compared to un-inoculated but treated with identical Cd concertation (Table 3). The dual inoculation caused the maximum enhancement in germination rate (10% increase). As well, root and shoot length of metal-treated plant declined with increasing Cd concentration which, however, improved following metal-tolerant microbial inoculation in polluted soils. As an example, root length of 25 µg Cd kg−1 soil treated plant were increased by 25% and 21% after soil inoculation of PGPR-7, Trichoderma sp. T-4 and PGPR-7 + Trichoderma sp. T-4, respectively, over un-inoculated control (Table 3). The plant length of chickpea of PGPR-7, Trichoderma sp. T4 and PGPR-7 + Trichoderma sp. inoculated and 25 µg Cd kg−1 soil treated plants were enhanced by 26.5%, 23% and 34%, respectively (Table 3). Likewise, metal tolerating PGPR strain Bacillus anthracis PM21 increased the germination efficiency and growth parameters of Sesbania sesban (L.) grown in soil treated with increasing concentrations of heavy metal65.
Additionally, dry biomass of Cd-treated plant was reduced in a concentration-dependent way. when chickpea plants were grown in the presence of 400 µg Cd kg−1 as opposed to the uninoculated control, the dry matter accumulation in the roots and shoots were reduced by 83.3% and 69%, respectively (Table 3). An earlier study conducted by Imtiaz et al. (2018) supported our findings noted a comparable drop in a number of biological indices in chickpea plants cultivated in the presence of different concentrations of vanadium (Vd). Metal-tolerant microbes detoxify the metal toxicity and enhanced the root and shoot dry biomass of plant. It's interesting to note that adding metal tolerant PGPR-7 to the experimental soils treated with 25 µg Cd kg−1 increased the root and shoot dry biomass by 52% and 37%, respectively, over non-inoculated control (Table 3). Like the current observation, a study found that Zea mays (L.) plants cultivated in the presence of increasing Pb concentrations displayed a concentration-dependent drop in germination rate, length of plant organs, fresh and dry biomass, and total protein content66. Additionally, a notable improvement in the development indices, such as the rate of germination, and growth parameters was recorded when dual inoculation were subjected to chickpeas grown in Cd stressed soil. These results imply that the addition of metal tolerant microbes to the culture reduced/eliminated the toxicity of heavy metals because of its capacity to detoxify metals, and as a result, enhanced a number of parameters of wheat plants grown in metal-treated soils.
Furthermore, in the presence of 400 µg Cd kg−1 soil, the Chl a, chl b and carotenoid content accumulated in fresh foliage of chickpea plants were significantly (p ≤ 0.05) reduced by 48, 56 and 67%, respectively. However, the leaves of inoculated plants showed more chlorophyll than the uninoculated one. Previous studies have found that under heavy metal stress circumstances, plant chlorophyll concentrations are lowered, photosynthetic pigment concentrations fluctuate, and gas exchange factors are inhibited. This drop in plant chlorophyll content may be caused by ROS interference, chlorophyll activation, modifications to important thylakoid membrane components, their manifestation mechanism, and modifications to the ultrastructure of proteins and DNA.
Contrarily, a 34, 29 and 41% increase in chl a, chl b and carotenoid content was recorded when strain PGPR-7 was applied to chickpea plants raised in soil treated with 25 µg Cd kg−1 soil, respectively (Fig. 3). In addition, Trichoderma sp. T-4 inoculation enhanced the chlorophyll a, chl b and carotenoid content by 28, 32 and 38% even at 100 µg Cd kg−1 soil and successfully bioremediated experimental soil that had been exposed to metal. Similar research showing the disruption in ability of plant to photosynthesize when Davidia involucrata plants were exposed to increasing doses of Cd and Pb stress has been reported67. In contrast, research of plant inoculated with metal-tolerant PGPR strain and cultivated under Cd stress discovered a significant increase in leaf photosynthetic pigments and other important growth indices of Glycine max and Lolium multiflorum68.
Single inoculation effect of PGPR (A–C), Trichoderma spp. (D–F) and combined inoculation of PGPR + Trichoderma sp. (G–I) on chl a, chl b and carotenoid content of fresh foliage detached from chickpea plants raised in soil affected with 0, 25, 50, 100, 200 and 400 μgCdmL−1. Each bar diagram represents the mean of three replicates (n = 3). Corresponding error bars represent standard deviation (S.D.) of replicates. Different letters represent that mean values are significantly different according to DMRT test.
Response of metal tolerant strains to antioxidant enzymes of Cd-treated chickpea
When inoculated/uninoculated chickpea plants were grown in the presence of 0–400 µg/kg doses of Cd, a dose-dependent increase in the antioxidant enzyme activity of the foliage was observed. While comparing the metal concentrations, 400 µgkg−1 of Cd had the maximum accumulation of CAT and POD in chickpea foliage. The build-up of antioxidant enzymes in the metal-treated plants was noticeably reduced when metal tolerant PGP strain PGPR-7 and T-4 strain was applied singly/combined to Cd-treated chickpea. For instance, when compared to uninoculated, plants inoculated with P. fluorescence PGPR-7 and treated with 25 µg Cdkg−1. Likewise, CAT activity was maximally reduced (55% reduction) when Trichoderma sp. T-4 was inoculated with 25 µg/kg of cadmium. Furthermore, dual inoculation of PGPR-7 and T-4 strains caused the maximum reduction of 29% and 32% in CAT and POD activity in 25 µg Cd kg−1 soil treated chickpea plants (Fig. 4). In conclusion, it is quite likely that metal tolerant microbial inoculants (P. fluorescence and Trichoderma sp.) lowered the amount of antioxidant enzymes, which would have otherwise increased in the presence of Cd stress. Even in soils with metal contamination, the overall growth of chickpea was significantly increased as a result of the clear decrease in antioxidants brought on by single/dual inoculation with the metal-tolerant microbes. Similar to our findings, it has been discovered that CAT activity was significantly increased in a concentration dependent manner in the leaf and root tissue of different legume seedlings69. Furthermore, Park et al.70 also noticed a decrease in oxidative stress (CAT and POD) of soybean plants following the inoculation PGPR Bacillus aryabhattai strain. Similarly, IAA producing and Cd tolerant Burkholderia gladioli and P. aeruginosa strains declined the level of antioxidant enzymes by lowering the Cd-uptake in tomato71. Likewise, phytohormone producing endophytic fungus Penicillium roqueforti decreased the metal induced oxidative stress in wheat and improved the growth72.
Antioxidant enzymes; catalase (A–C) and peroxidase (D–F) activities in cadmium supplemented and inoculated (either single/combined) with PGPR-7, Trichoderma sp. T4 strain. Each bar diagram represents the mean of three replicates (n = 3). Corresponding error bars represent standard deviation (S. D.) of replicates. Different letters represent that mean values are significantly different according to DMRT test.
Proline and MDA content in inoculated and Cd-treated Chickpea
Proline is thought to act as an osmo-regulator, protecting plants from the harmful effects of ROS produced by plants when they are exposed to environmental challenges such heavy metals, salt and drought etc. Proline also helps plants maintaining homeostasis and osmotic equilibrium. In addition to this, it functions as a ROS scavenger and is essential for maintaining the subcellular structures of plants to help them survive a variety of environmental challenges. Taking this into account, the proline content of chickpea cultivated with or without Cd was measured. In general, when Cd level increases, the proline content in chickpea leaves gradually increased. It's interesting to note that when plants were cultivated in the presence of 400 µg Cd kg−1, compared to the uninoculated control, a 67% upsurge in proline concentration was observed. Conversely, following inoculation of metal tolerant microbial strains PGPR-7 and T-4, the accumulation of proline in chickpea foliage were significantly declined by 31% and 42%, respectively when plants were raised in soil polluted with 400 µg Cd kg−1 soil over uninoculated but treated with identical Cd doses (Fig. 5). The maximum decline (48%) in proline concentration was recorded when dual cultures were applied to chickpea plants grown in soil treated with 25 µg Cd kg−1 soil. A comparable decrease in proline content in the plant organs of Cd-treated Hordeum vulgare (L.) inoculated with mycorrhizal fungi73 and cultivated in stressful settings was observed, correlating with our findings.
Proline content estimation in Cd treated chickpeas and inoculated with Trichoderma sp. T4 (A), PGPR-7 strain (B) and Trichoderma sp. T4 + PGPR-7 strain (C). Panels D, E and F represents the MDA content extracted from Cd-treated and inoculated with Trichoderma sp. T4 (D), PGPR-7 strain (E) and Trichoderma sp. T4 + PGPR-7 strain (F) chickpeas. Here, line diagram represents the mean of three replicates (n = 3). Corresponding error bars represent standard deviation (S. D.) of replicates. Different letters represent that mean values are significantly different according to DMRT test.
Since, single/combined inoculation of metal tolerant microbial bacterial strain lessened the effects of Cd stress and shielded the plants from oxidative damage, the metal tolerant PGPR utilised in this study allowed chickpea plants to survive and flourish even under metal stress. These findings support the idea that metal tolerant microbial strains might function as a potential organism in bioremediation procedures, eventually improving the functionality of chickpea grown under metal stress.
Many plants, including legumes, are affected by heavy metal stress that results in the production of ROS, which affects lipid peroxidation (LPO) in cellular membrane. Malondialdehyde (MDA) is a by-product of LPO in plant tissues. MDA thus serves as a marker of the intensity of stress for plants. In light of this, production of MDA was measured in chickpea leaves affected with varying dosages of Cd. With larger metal doses, leaves generally contained more MDA. For instance, compared to the un-inoculated control, 400 µg Cd kg−1 soil markedly raised the MDA concentration by 54%. However, even with 200 µg Cd kg−1 soil, P. fluorescence PGPR-7 and Trichoderma sp. T-4 were still effective in reducing metal stress and, consequently reduced the level of MDA by 43% and 49%, respectively (Fig. 6). Moreover, combined inoculation of bacteria and fungi had maximum detoxifying effect and lowered the MDA concentration maximally in chickpea plants contaminated with 25 µg Cd kg−1 soil. Saif and Khan74, investigated that HM stress-induced seedlings of chickpea had a significantly higher MDA and proline contents than control seedlings which however, decreased following the inoculation of Ni and Cr tolerant Pseudomonas aeruginosa.
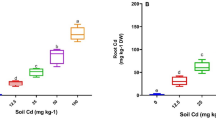
Uptake of Cd in chickpea plants treated with different concentrations (0, 25, 50, 100, 200 and 400 μg Cd kg−1) of Cd and inoculated with PGPR-7 (A), Trichoderma sp. T-4 (B) and PGPR-7 + Trichoderma sp. T-4 (C). Each bar and line diagram represents the mean of three replicates (n = 3). Corresponding error bars represent standard deviation (S. D.) of replicates. Different letters represent that mean values are significantly different according to DMRT test.
Yield attributes and nutritional content in Cd-treated and bio-inoculated chickpeas
At harvest (120 DAS), the seed yield (SY) and seed protein (SP) showed a steady decline with successively increasing doses of Cd. However, the inoculation of metal tolerant microbial strain caused an increase in these parameters. The maximum number of seeds/plants (25 seed/plants) were recorded when chickpeas were detached from soil inoculated with microbial consortia (PGPR-7 strain and T4). Furthermore, chickpea plants inoculated with PGPR-7, Trichoderma sp. T-4 and PGPR-7 + T-4 and treated with 25 µg Cd kg−1 soil increased the seed yield by 26%, 28.5% and 32%, respectively over uninoculated but treated with similar Cd dose (Table 4). The seed protein content was also were significantly declined with increasing Cd concentration. In contrast, the Cd-tolerant Pseudomonas PGPR-7, Trichoderma sp. T4 and Pseudomonas PGPR-7 + Trichoderma sp. T4 on the other hand, enhanced the SP of chickpea by 20%, 27% and 38%, respectively, at 25 µg Cd kg−1 soil, when compared to uninoculated chickpea cultivated in soil amended with identical doses of cadmium. In a similar study, Oves et al.75 demonstrated that Pseudomonas aeruginosa improved the yield attributes of chickpea plants raised in chromium (Cr) contaminated soil. Furthermore, research by Tripathi et al.76 demonstrated that growth characteristics and yield of chickpea were improved when the plant was grown with a consortium of Trichoderma while being exposed to the arsenic (As). The statistical study significantly demonstrated how Cd concentrations and microbial inoculations both had an interaction effect on the examined parameter.
Understanding the toxic effect of heavy metals on nutritional values of legumes and the efficiency of Cd-tolerant microbial consortia in toxicity removal and enhanced uptake of nutrient was evaluated. Here, the increasing Cd doses declined the nitrogen and phosphorous content of chickpea. However, inoculation of Pseudomonas PGPR-7, Trichoderma T-4 and Pseudomonas PGPR-7 + Trichoderma T-4 significantly increased the N (21, 13 and 39%, respectively) and P (14, 6 and 20%, respectively) content at 25 µg Cd kg−1 soil (Table 4). Two elements for the computed components, an ANOVA revealed that the single and dual effect of microbial inoculants and their interaction (inoculation + cadmium) were significant (p < 0.05). In almost treatment, the dual inoculation of microbial consortia potentially mitigated the hazard of Cd and maximally increased/improved the biometric parameters.
Cd uptake in inoculated chickpeas
Although there were significant differences in the amount of Cd uptake in roots and shoot tissues of chickpea, the concentration of metals consistently rose with higher Cd dosages. In terms of plant organs, shoots (1.89 µg g−1) and roots (2.78 µg g−1) exhibited the highest Cd concentrations when detached from soil contaminated with 400 µg Cd kg−1. Intriguingly, the plant organs showed the greatest decrease in metal concentration after inoculation of Cd-tolerant P. fluorescence PGPR-7 and Trichoderma sp. T4. For instance, the Cd uptake by roots that were removed from plants growing at 400 mgCdkg−1 was reduced by 43% after inoculation of P. fluorescence PGPR-7 strain (Fig. 6). Similar to this, it was found that Trichoderma sp. T4 inoculated plant roots had a 37% decrease in metal deposition in the presence of 400 µg Cd kg−1. The reduction/decrease in Cd uptake following metal tolerant microbes was observed followed the order: P. fluorescence PGPR-7 + Trichoderma sp. T4 (dual inoculation) > Trichoderma sp. T4 > P. fluorescence PGPR-7. Although cadmium (Cd) is a highly hazardous element, Cd ions are easily transferred from the soil to different plant components. It's possible that this is caused by the production of protons, root exudates, and other metabolites, which facilitate the simple dissolution of Cd ions, most likely by way of the formation of metal chelating complexes. Therefore, plants inoculated with metal tolerant microbial species could showed the decreased metal deposition in each organ, probably as a result of the metal-tolerant strain accumulating the majority of the metal component present in the soil. Because of this, the plants could only absorb a lesser amount of metal. Thus, when metal pressure was released, the inoculated chickpea plants had a better chance of surviving even in soil that had been treated with Cd. In a similar manner, Cd-tolerant strain Bradyrhizobium lowered the uptake of cadmium in Glycine max (L.) and Lolium multiflorum (L.) by reducing the toxicity77. Furthermore, metal tolerant T. harzianum increased the growth of Hordeum vulgare (L.) by decreasing the uptake of Cd78.
Conclusions
The present investigation was primarily concerned with the detrimental effects of Cd on biological and chemical properties of chickpea. It was observed that Cd-induced severe oxidative damage in chickpeas led to membrane LPO and a powerful antioxidant defence response. Either singly/combined inoculation of metal-resistant P. fluorescence PGPR-7 and Trichoderma sp. T-4 prevented Cd toxicity in chickpeas. Even under Cd pressure, microbial strains were remarkably capable of producing a variety of growth regulating active biomolecules. These elements may have contributed significantly to improving the functioning of chickpeas, either alone or concurrently, and they have shed light on why chickpea plants kept growing and functioning even when exposed to Cd stress. Consequently, it was discovered that both microbial strains were effective/potential candidates that could be used to remediate the metal contaminated soils. As a result, the performance of Cd-treated chickpeas as a whole was improved when combined inoculum of both strains was used. The Cd-resistant Pseudomonas fluorescence and Trichoderma sp. could, therefore, serve as a potent microbial inoculant that could be used in a straightforward, affordable, and environmentally friendly method to remediate the heavy metal contaminated soils because of the commendable metal tolerance ability and other distinctive growth promoting features. Furthermore, from the study, it is recommended that irrigation of edible crops with industrially discharged effluents/wastewater should be minimized. In a nutshell, we can say that the applied HM-tolerant microbes could play an essential in agricultural sectors as an army to clean-up/decontaminate the heavy metal hazard. Furthermore, efficacy of HMs tolerant microbes can be increased by transferring the genes from theses HM-resistant microorganisms to another laboratory cultivated genetically engineered microbes.
Data availability
Sequence data of identified bacterial strain used in this study have been submitted to the NCBI archives (https://ncbi.nlm.nih.gov). The name of the repository and accession number can be found here; https://www.ncbi.nlm.nih.gov/genbank/, OP925885.
References
Sheikh, L. et al. Evaluating the effects of cadmium under saline conditions on leafy vegetables by using acidified biochar. Pak. J. Bot. 55, 1537. https://doi.org/10.30848/PJB2023-SI(4) (2023).
Shahzad, K. et al. Exogenous application of proline and glycine betaine mitigates nickel toxicity in mung bean plants by up-regulating growth, physiological and yield attributes. Pak. J. Bot. 55, 1536. https://doi.org/10.30848/PJB2023-SI(3) (2023).
Khatun, J., Intekhab, A., Dhak, D. Effect of uncontrolled fertilization and heavy metal toxicity associated with arsenic(As), lead(Pb) and cadmium (Cd), and possible remediation. Toxicology. 477, 153274 (2022).
Sandeep, G., Vijayalatha, K. R. & Anitha, T. Heavy metals and its impact in vegetable crops. Int. J. Chem. Stud. 7, 1612–1621 (2019).
Tighe-Neira, R., Gonzalez-Villagra, J., Nunes-Nesi, A. & Inostroza-Blancheteau, C. Impact of nanoparticles and their ionic counterparts derived from heavy metals on the physiology of food crops. Plant Physiol. Biochem. 172, 14–23 (2022).
Sarathchandra, S. S., Rengel, Z. & Solaiman, Z. M. Remediation of heavy metal-contaminated iron ore tailings by applying compost and growing perennial ryegrass (Lolium perenne L.). Chemosphere 288, 132573 (2022).
Sofo, A., Khan, N. A., D’Ippolito, I. & Reyes, F. Subtoxic levels of some heavy metals cause differential root-shoot structure, morphology and auxins levels in Arabidopsis thaliana. Plant Physiol. Biochem. 173, 68–75 (2022).
Rahman, S. U. et al. State-of-the-art OMICS strategies against toxic effects of heavy metals in plants: A review. Ecotoxicol. Environ. Saf. 242, 113952 (2022).
Ma, J. et al. Individual and combinatorial effects of SNP and NaHS on morpho-physio-biochemical attributes and phytoextraction of chromium through Cr-stressed spinach (Spinacia oleracea L.). Front. Plant Sci. 13, 973740 (2022).
Ma, J., et al. Impact of foliar application of syringic acid on tomato (Solanum lycopersicum L.) under heavy metal stress-insights into nutrient uptake, redox homeostasis, oxidative stress, and antioxidant defense. Front. Plant Sci. 13. (2022).
Stambulska, U.Y. & Bayliak, M. M. Legume-rhizobium symbiosis: secondary metabolites, free radical processes, and effects of heavy metals. Co-Evol. Second. Metab. 291–322 (2020).
Naz, M. et al. The soil pH and heavy metals revealed their impact on soil microbial community. J. Environ. Manage. 321, 115770 (2022).
Meena, M., Sonigra, P. & Yadav, G. Biological-based methods for the removal of volatile organic compounds (VOCs) and heavy metals. Environ. Sci. Pol. Res. 28, 2485–2508 (2020).
Liu, A. et al. Improvement of the Cd and Zn phytoremediation efficiency of rice (Oryza sativa) through the inoculation of a metal-resistant PGPR strain. Chemosphere 302, 134900 (2022).
Ke, T. et al. Improvement of the Cu and Cd phytostabilization efficiency of perennial ryegrass through the inoculation of three metal-resistant PGPR strains. Environ. Pol. 271, 116314 (2021).
Tirry, N., Kouchou, A., El Omari, B., Ferioun, M. & El Ghachtouli, N. Improved chromium tolerance of Medicago sativa by plant growth-promoting rhizobacteria (PGPR). J. Genet. Eng. Biotechnol. 19, 1–14 (2021).
Hussain, A., Shah, M., Hamayun, M., Qadir, M. & Iqbal, A. Heavy metal tolerant endophytic fungi Aspergillus welwitschiae improves growth, ceasing metal uptake and strengthening antioxidant system in Glycine max L.. Environ. Sci. Pol. Res. 29, 15501–15515 (2022).
Anam, G. B., Reddy, M. S. & Ahn, Y. H. Characterization of Trichoderma asperellum RM-28 for its sodic/saline-alkali tolerance and plant growth promoting activities to alleviate toxicity of red mud. Sci. Total Environ. 662, 462–469 (2019).
Fuentes-Quiroz, A. et al. Rhizosphere fungi regulate the expression of metal tolerance genes in Solanum lycopersicum L. (Solanaceae) growing in a metal (loid)-contaminated soil. Rhizosphere 24, 100599 (2022).
Wu, K. et al. Endophytic bacterium Buttiauxella sp. SaSR13 improves plant growth and cadmium accumulation of hyperaccumulator Sedum alfredii. Environ. Sci. Pol. Res. 25, 21844–21854 (2018).
Mehmood, H., Arif Ali, M. & Hussain, S. Growth and yield of rice under variable application methods of zinc with and without arbuscular mycorrhizae in normal and saline soils. Pak. J. Bot. 55, 1534. https://doi.org/10.30848/PJB2023-SI(1) (2023).
Banaay, C. G. B. & Cuevas, V. C. Alleviating the yield constraints posed by copper contamination in lowland rice and vegetable crop areas using Trichoderma technologies. Sci. Eng. J. 15(1), 17–24 (2022).
Kumar, V. & Dwivedi, S. K. Multi-metal tolerant fungus Aspergillus flavus CR500 with remarkable stress response, simultaneous multiple metal/loid removal ability and bioremediation potential of wastewater. Environ. Technol. Innov. 20, 101075 (2020).
Syed, A. et al. Heavy metals induced modulations in growth, physiology, cellular viability, and biofilm formation of an identified bacterial isolate. ACS Omega 6, 25076–25088 (2021).
Zhang, D., Yin, C., Abbas, N., Mao, Z. & Zhang, Y. Multiple heavy metal tolerance and removal by an earthworm gut fungus Trichoderma brevicompactum QYCD-6. Sci. Rep. 10, 1–9 (2020).
Shahid, M. & Khan, M. S. Pesticide-induced alteration in proteins of characterized soil microbiota revealed by sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE). J. Proteins Proteom. 11, 1–9 (2020).
Shahid, M. et al. Stress-tolerant endophytic isolate Priestia aryabhattai BPR-9 modulates physio-biochemical mechanisms in wheat (Triticum aestivum L.) for enhanced salt tolerance. Int. J. Environ. Res. Public Health 19(17), 10883 (2022).
Mayer, A. M. Determination of indole acetic acid by the Salkowsky reaction. Nature 182(4650), 1670–1671 (1958).
Penrose, D. M. & Glick, B. R. Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol. Planta. 118, 10–15 (2003).
Honma, M. & Shimomura, T. Metabolism of 1-aminocyclopropane-1-carboxylic acid. Agric. Biol. Chem. 42, 1825–1831 (1978).
Oves, M., Khan, M. S. & Qari, H. A. Ensifer adhaerens for heavy metal bioaccumulation, biosorption, and phosphate solubilization under metal stress condition. J. Taiwan Inst. Chem. Eng. 80, 540–552 (2017).
Bakker, A. W. & Schippers, B. O. B. Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas spp-mediated plant growth-stimulation. Soil Biol. Biochem. 19, 451–457 (1987).
Arnon, D. I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 24(1), 1 (1949).
Kirk, J. T. O. & Allen, R. L. Dependence of chloroplast pigment synthesis on protein synthesis: Effect of actidione. Biochem. Biophys. Res. Commun. 21, 523–530 (1965).
Bates, L. S., Waldren, R. P. & Teare, I. D. Rapid determination of free proline for water-stress studies. Plant Soil 39(1), 205–207 (1973).
Shahid, M., Ahmed, B., Zaidi, A. & Khan, M. S. Toxicity of fungicides to Pisum sativum: A study of oxidative damage, growth suppression, cellular death and morpho-anatomical changes. RSC Adv. 8, 38483–38498 (2018).
Khan, S. et al. Fungicide-tolerant plant growth-promoting rhizobacteria mitigate physiological disruption of white radish caused by fungicides used in the field cultivation. Int. J. Environ. Res. Public Health 17(19), 7251 (2020).
Shahid, M. & Khan, M. S. Fungicide tolerant Bradyrhizobium japonicum mitigate toxicity and enhance greengram production under hexaconazole stress. J. Environ. Sci. 78, 92–108 (2019).
Shahid, M., Khan, M. S. & Kumar, M. Kitazin-pea interaction: Understanding the fungicide induced nodule alteration, cytotoxicity, oxidative damage and toxicity alleviation by Rhizobium leguminosarum. RSC Adv. 9, 16929–16947 (2019).
Jackson, M. L. Soil Chemical Analysis (Prentice Hall, 1976).
Iswaran, V. & Marwah, T. S. A modifed rapid Kjeldahl method for determination of total nitrogen in agricultural and biological materials. Geobios 7, 281–282 (1980).
Intawongse, M. & Dean, J. R. Uptake of heavy metals by vegetable plants grown on contaminated soil and their bioavailability in the human gastrointestinal tract. Food Addit. Contam. 23, 36–48 (2006).
Lee, S. Y., Lee, Y. Y. & Cho, K. S. Characterization of heavy metal tolerant and plant growth-promoting rhizobacteria isolated from soil contaminated with heavy metal and diesel. Microbiol. Biotechnol. Lett. 49, 413–424 (2021).
Fahsi, N., Mahdi, I., Mesfioui, A., Biskri, L. & Allaoui, A. Plant Growth-Promoting Rhizobacteria isolated from the Jujube (Ziziphus lotus) plant enhance wheat growth, Zn uptake, and heavy metal tolerance. Agriculture 11(4), 316 (2021).
Shadmani, L., Jamali, S. & Fatemi, A. Isolation, identification, and characterization of cadmium-tolerant endophytic fungi isolated from barley (Hordeum vulgare L.) roots and their role in enhancing phytoremediation. Braz. J. Microbiol. 52, 1097–1106 (2021).
Fakhar, A. et al. Heavy metal remediation and resistance mechanism of Aeromonas, Bacillus, and Pseudomonas: A review. Crit. Rev. Environ. Sci. Tech. 52, 1868–1914 (2022).
Ali, S. S. & Vidhale, N. N. Bacterial siderophore and their application: A review. Int. J. Curr. Microbiol. Appl. Sci. 2, 303–312 (2013).
Das, A. et al. Fungal siderophores: Structure, functions and regulation. In Microbial Siderophores (eds Varma, A. & Chincholkar, S. B.) 1–42 (Springer, 2007).
Tamariz-Angeles, C., Huamán, G. D., Palacios-Robles, E., Olivera-Gonzales, P. & Castañeda-Barreto, A. Characterization of siderophore-producing microorganisms associated to plants from high-Andean heavy metal polluted soil from Callejón de Huaylas (Ancash, Perú). Microbiol. Res. 250, 126811 (2021).
Patel, M. et al. Cadmium-tolerant plant growth-promoting bacteria Curtobacterium oceanosedimentum improves growth attributes and strengthens antioxidant system in chili (Capsicum frutescens). Sustainability 14(7), 4335 (2022).
Kumar, A., Maleva, M., Bruno, L. B. & Rajkumar, M. Synergistic effect of ACC deaminase producing Pseudomonas sp. TR15a and siderophore producing Bacillus aerophilus TR15c for enhanced growth and copper accumulation in Helianthus annuus L.. Chemosphere 276, 130038 (2021).
Tripathi, M., Munot, H. P., Shouche, Y., Meyer, J. M. & Goel, R. Isolation and functional characterization of siderophore-producing lead-and cadmium-resistant Pseudomonas putida KNP9. Curr. Microbiol. 50, 233–237 (2005).
Chen, Y. et al. Metal-tolerant Enterobacter sp. strain EG16 enhanced phytoremediation using Hibiscus cannabinus via siderophore-mediated plant growth promotion under metal contamination. Plant Soil 413, 203–216 (2017).
Sepehri, M. & Khatabi, B. Combination of siderophore-producing bacteria and Piriformospora indica provides an efficient approach to improve cadmium tolerance in alfalfa. Microbial. Ecol. 81, 717–730 (2021).
Babu, A. G., Shim, J., Bang, K. S., Shea, P. J. & Oh, B. T. Trichoderma virens PDR-28: a heavy metal-tolerant and plant growth-promoting fungus for remediation and bioenergy crop production on mine tailing soil. J. Environ. Manag. 132, 129–134 (2014).
Xie, Y. et al. Characterization of the Cd-resistant fungus Aspergillus aculeatus and its potential for increasing the antioxidant activity and photosynthetic efficiency of rice. Ecotoxicol. Environ. Safe. 171, 373–381 (2019).
Oubohssaine, M., Sbabou, L. & Aurag, J. Native heavy metal-tolerant plant growth promoting rhizobacteria improves Sulla spinosissima (L.) growth in post-mining contaminated soils. Microorganisms 10(5), 838 (2022).
Pramanik, K., Mitra, S., Sarkar, A. & Maiti, T. K. Alleviation of phytotoxic effects of cadmium on rice seedlings by cadmium resistant PGPR strain Enterobacter aerogenes MCC 3092. J. Hazard. Mater. 351, 317–329 (2018).
Xie, Y. et al. Identification of Cd-resistant microorganisms from heavy metal-contaminated soil and its potential in promoting the growth and Cd accumulation of bermudagrass. Environ. Res. 200, 111730 (2021).
Murali, M. et al. Bio-prospecting of ACC deaminase producing Rhizobacteria towards sustainable agriculture: A special emphasis on abiotic stress in plants. Appl. Soil Ecol. 168, 104142 (2021).
Ali, S. & Kim, W. C. Plant growth promotion under water: Decrease of waterlogging-induced ACC and ethylene levels by ACC deaminase-producing bacteria. Front. Microbiol. 9, 1096 (2018).
Glick, B. R., Cheng, Z., Czarny, J. & Duan, J. Promotion of plant growth by ACC deaminase-producing soil bacteria. in New Perspectives and Approaches in Plant Growth-Promoting Rhizobacteria Research 1st edn (eds Bakker, P. A. H. M. et al.) 329–339 (Springer, Dordrecht, 2007).
Rauf, M. et al. Molecular mechanisms of the 1-aminocyclopropane-1-carboxylic acid (ACC) deaminase producing Trichoderma asperellum MAP1 in enhancing wheat tolerance to waterlogging stress. Front. Plant Sci. 11, 614971 (2021).
Zhang, S., Gan, Y. & Xu, B. Mechanisms of the IAA and ACC-deaminase producing strain of Trichoderma longibrachiatum T6 in enhancing wheat seedling tolerance to NaCl stress. BMC Plant Biol. 19, 1–18 (2019).
Ali, J. et al. Mechanistic elucidation of germination potential and growth of Sesbania sesban seedlings with Bacillus anthracis PM21 under heavy metals stress: An in vitro study. Ecotoxicol. Environ. Safe. 208, 111769 (2021).
Hussain, A. et al. Effects of diverse doses of Lead (Pb) on different growth attributes of Zea-mays L. Agric. Sci. 4, 262–265 (2013).
Yang, Y. et al. Response of photosynthesis to different concentrations of heavy metals in Davidia involucrata. PLoS ONE 15(3), e0228563 (2020).
Guo, J. & Chi, J. Effect of Cd-tolerant plant growth-promoting rhizobium on plant growth and Cd uptake by Lolium multiflorum Lam. and Glycine max (L.) Merr. in Cd-contaminated soil. Plant Soil. 375, 205–214 (2014).
Chehelpar, N., Tohidi-Moghadam, H. R. & Ghoushchi, F. Hexaconazole foliar application alleviates water deficit effects in common bean. Pesquisa Agropecuária Trop. 46, 301–310 (2016).
Park, Y. G. et al. Bacillus aryabhattai SRB02 tolerates oxidative and nitrosative stress and promotes the growth of soybean by modulating the production of phytohormones. PLoS ONE 12(3), e0173203 (2017).
Khanna, K., Jamwal, V. L., Gandhi, S. G., Ohri, P. & Bhardwaj, R. Metal resistant PGPR lowered Cd uptake and expression of metal transporter genes with improved growth and photosynthetic pigments in Lycopersicon esculentum under metal toxicity. Sci. Rep. 9, 1–14 (2019).
Ikram, M. et al. IAA producing fungal endophyte Penicillium roqueforti Thom., enhances stress tolerance and nutrients uptake in wheat plants grown on heavy metal contaminated soils. PLoS One 13(11), e0208150 (2018).
Keshavarz, H., Hosseini, S. J., Sedibe, M. M. & Achilonu, M. C. Arbuscular mycorrhizal fungi used to support iranian barley cultivated on cadmium contaminated soils (Hordeum vulgare L.). Appl. Ecol. Environ. Res. 20(1), 43–53 (2022).
Saif, S. & Khan, M. S. Assessment of toxic impact of metals on proline, antioxidant enzymes, and biological characteristics of Pseudomonas aeruginosa inoculated Cicer arietinum grown in chromium and nickel-stressed sandy clay loam soils. Environ. Monit. Assess. 190, 1–18 (2018).
Oves, M., Khan, M. S. & Zaidi, A. Chromium reducing and plant growth promoting novel strain Pseudomonas aeruginosa OSG41 enhance chickpea growth in chromium amended soils. Eur. J. Soil Biol. 56, 72–83 (2013).
Tripathi, P., Singh, P. C., Mishra, A., Tripathi, R. D. & Nautiyal, C. S. Trichoderma inoculation augments grain amino acids and mineral nutrients by modulating arsenic speciation and accumulation in chickpea (Cicer arietinum L.). Ecotoxicol. Environ. Safe. 117, 72–80 (2015).
Guo, J. & Chi, J. Effect of Cd-tolerant plant growth-promoting rhizobium on plant growth and Cd uptake by Lolium multiflorum Lam. and Glycine max (L.) Merr. in Cd-contaminated soil. Plant Soil 375, 205–214 (2014).
Taghavi Ghasemkheili, F. et al. Ameliorative effects of Trichoderma harzianum and Rhizosphere soil microbes on Cadmium biosorption of Barley (Hordeum vulgare L.) in Cd-polluted soil. J. Soil Sci. Plant Nut. 22, 527–539 (2022).
Acknowledgements
The authors extend their appreciation to The Researchers Supporting Project number (RSP2023R56), King Saud University, Riyadh, Saudi Arabia.
Funding
The authors extend their appreciation to The Researchers Supporting Project number (RSP2023R56) King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conceptualization; A.S.; A.M.E.; A.H.B.; Conducted experiment; A.S.; A.M.E.; A.H.B.; Formal analysis; A.S.; A.M.E.; A.H.B.; Methodology; A.S.; A.M.E.; A.H.B.; R.K.I. Writing—original draft; A.S.; S.D.; Writing—review & editing; A.S.; S.D.; A.M.E..; R.E., R.K.I.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Syed, A., Elgorban, A.M., Bahkali, A.H. et al. Metal-tolerant and siderophore producing Pseudomonas fluorescence and Trichoderma spp. improved the growth, biochemical features and yield attributes of chickpea by lowering Cd uptake. Sci Rep 13, 4471 (2023). https://doi.org/10.1038/s41598-023-31330-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-023-31330-3
This article is cited by
-
PGPR-mediated alleviation of arsenic and cadmium toxicity in rice (Oryza sativa L.) through regulation of rhizosphere Microbiome and oxidative stress biomarkers
Plant Growth Regulation (2026)
-
Plant nickel-exclusion versus hyperaccumulation: a microbial perspective
Microbiome (2025)
-
Plant growth-promoting native rhizobia isolated from red clover (Trifolium pratense L.) nodules: potential for metal-phytostabilization
BMC Microbiology (2025)
-
Minimization of cadmium toxicity and improvement in growth and biochemical attributes of spinach by using acidified biochar
Scientific Reports (2025)
-
Effect of Chitosan and Trichoderma on Pisum sativum Germinating Seeds under Induced cadmium toxicity
Journal of Plant Growth Regulation (2025)