Abstract
Interleukin-6 (IL-6) is a major pro-inflammatory cytokine that demonstrates a robust correlation with age and body mass index (BMI) as part of the senescence-associated secretory phenotype. IL-6 cytokines also play a crucial role in metabolic homeostasis and regenerative processes primarily via the canonical STAT3 pathway. Thus, selective modulation of IL-6 signaling may offer a unique opportunity for therapeutic interventions. Our recent studies identified a novel non-canonical signaling pathway that involves prolonged activation of SRC family of kinases (SFKs) by IL-6/gp130, where genetic or pharmacological inhibition of this pathway was protective in several acute injury models. This study was designed to assess the effect of a small molecule (R159) that inhibits the non-canonical signaling in a mouse model of multimorbidity induced by chronic inflammation. Aged mice were fed a high-fat diet (HFD) to exacerbate chronic inflammation and inflammaging-related conditions, and R159 significantly decreased systemic inflammatory responses in adipose tissue and liver. R159 was protective against trabecular bone and articular cartilage loss and markedly prevented neurogenesis decline. Moreover, R159 reduced weight gain induced by HFD and increased physical activity levels. These findings suggest that selective pharmacological inhibition of SFK signaling downstream of IL6/gp130 offers a promising strategy to alleviate systemic chronic inflammation and relevant multimorbidity.
Similar content being viewed by others
Introduction
The mechanisms involved in systemic chronic inflammation, also known as “inflammaging”, is poorly understood and often associated with chronological aging, obesity, and chronic stress. This gap in knowledge limits the development of therapies used to manage inflammaging and associated multimorbidity, which is marked by the accumulation of senescent cells combined with increased systemic release and persistence of pro-inflammatory factors such as Interleukin 6 (IL-6)1,2. Inflammaging in older adults is usually marked in the clinic by stable elevated levels of C-reactive protein (CRP) and monocyte chemoattractant protein-1 (MCP-1)3,4 with no other symptoms of specific inflammatory disorders such as autoimmune conditions or chronic infection. Elevation of CRP (especially above 3 mg/L) and increased body mass index (BMI) (above 30 kg/m2) are associated with increased risk of multi-organ failure and mortality in multiple studies conducted in different ages and demographics5,6,7,8. Increased levels of MCP-1 in aging adults has been associated with decreases in memory, cardiovascular disease, and sarcopenia3,9. IL-6 is one of the key factors regulating the senescence-associated secreted phenotype produced by senescent cells in degenerating tissues and in immune cells activated by this progressive multiorgan degeneration and atrophy10,11,12. IL-6 is the principal inducer of CRP produced by the liver cells13 and a key activator of MCP-1 production in multiple tissues14,15.
All IL-6 family of cytokines, including IL-6, IL-11, and Oncostatin M (OSM), signal through the transmembrane receptor gp130, which mediates downstream signaling via its intracellular residues that impact distinct biological processes. In our recent study, we have identified a signaling tyrosine 814 (Y814) residue within the gp130 intracellular domain that acts as a “stress sensor” responsible for triggering degenerative and pathological changes in tissues via activation of SRC family of kinases (SFKs)16. Endogenous products arising from age-associated tissue senescence may also cause maladaptive hyperactivation of gp130-Y814, which is highly detrimental. Both genetic and drug-mediated inactivation of gp130/SFK signaling resulted in enhanced resolution of inflammation and accelerated regenerative outcomes in several injury models16.
However, the role of this novel gp130 signaling arm in chronic systemic inflammation is not yet defined. HFD leads to a significant elevation of IL-6, MCP-1, and CRP levels in systemic circulation and multiple studies have demonstrated that HFD can lead to the progressive dysfunction of multiple organs and tissues17,18,19. We hypothesized that non-canonical gp130/SFK signaling is a novel mechanism implicated in the pathogenesis of chronic systemic inflammation and associated multimorbidity. We predicted that selective targeting of this signaling can improve the healthspan of animals with systemic chronic inflammation induced by HFD. Our findings demonstrate that small molecule-mediated disruption of SFK activation via IL6/gp130 attenuated systemic inflammation and improved functional capacity of metabolic organs such as adipose tissue and liver, as well as musculoskeletal tissues including bone and cartilage, and promoted neurogenesis with increased physical activity in an accelerated aging model of mice fed with HFD. This intervention offers a promising strategy to alleviate systemic chronic inflammation and relevant multimorbidity, promoting the healthspan of aged or obese individuals.
Results
Pharmacological inhibition of gp130/SFK signaling reduces inflammatory responses in mice on HFD
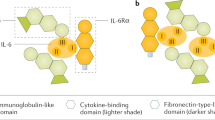
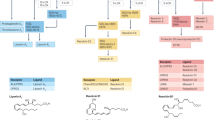
Our previous studies showed that pharmacological inhibition of SFK signaling downstream of IL6/gp130 leads to anti-inflammatory and anti-fibrotic effects in acute local injuries in small and large animal models16. Previously reported SFK recruitment inhibitor R805, developed by our group, had poor water solubility and was only suitable for localized applications16. R159, an analog of R805, was developed with an improved Absorbance Distribution Metabolism and Excretion (ADME) profile (Fig. 1a). OSM, a potent pro-inflammatory cytokine from the IL-6 family, induces phosphorylation of the gp130 receptor at residue Y814 (Fig. 1b). This phosphorylation event triggers downstream signaling pathways, including the activation of SFK. Treatment with the small molecule inhibitor R159 effectively blocks gp130-Y814 phosphorylation in response to IL-6 family cytokine stimulation, leading to a reduction in SFK activation (Fig. 1b and c).
R159 treatment inhibits gp130-Y814/SFK module in primary synovial cells and demonstrates improved ADME profile compared to previously developed analogs. (a) Pharmacokinetic characteristics of small molecule drug R805 and its analog, R159. TPSA (total polar surface area) range 60–140 Å2 is considered good solubility and permeability. cLogP and cLogD show predicted water solubility; cLogP less than 3 and cLogD less than 1 are considered favorable for water solubility. pKa indicates the ionization state of the compound at given pH. RLM (rat liver microsome)/HLM (human liver microsome) show stability in rat/human liver microsomes; %LBF shows the clearance attributed to liver blood flow. KS shows the solubility constant at a pH 7.4. MDCK (Madin-Darby canine kidney) WT and MDCK MDR1 cells are used to test drug transport and permeability. A higher Papp value in the A-B direction suggests better absorption potential and a higher Papp value in the B-A direction can indicate strong efflux activity. EER shows an efflux ratio; an EER equal to or close to 1 suggests balanced influx and efflux, indicating that efflux is not significantly hindering the compound’s permeability. (b) Western blots for phosphorylated gp130-Y814 in human synovial fibroblasts treated with or without OSM (10ng/mL) and R159 (10 µM) for 4 h, n = 4. (c) Western blots for phosphorylated SRC in human synovial fibroblasts treated with or without OSM (10ng/mL) and R159 (10 µM) for 4 h, n = 4. One-way ANOVA with multiple comparisons with Tukey test was used for statistical analysis of (b) and (c); p-values less than 0.05 were considered significant. Original blots for (b) and (c) are presented in Supplementary Fig. 1.
1-year-old aged male C57BL/6J mice were placed on HFD and administered R159 or vehicle (Veh) twice a week via intraperitoneal injection for 12 weeks to evaluate the effects of gp130/SFK inactivation on systemic chronic inflammation (Fig. 2a). Older mice were used to closely model the progression of multimorbidity seen in middle-aged humans. HFD is a commonly used model to induce rapid weight gain, chronic systemic inflammation, and multimorbidity. Notably, mice treated with R159 gained significantly less weight compared to the Veh group (Fig. 2b). While serum IL-6 levels remained similar between Veh and R159 groups, R159 treatment significantly lowered both systemic MCP-1 and CRP levels (Fig. 2c). Concurrently, R159 attenuated expression of Ccl2 (MCP-1) and pro-inflammatory cytokines Tnf\(\:\alpha\:\) and Il1\(\:\beta\:\) in adipose tissue induced by HFD (Fig. 3a). Adipose tissue from R159-treated mice on HFD showed reduced macrophage presence compared to Veh-treated mice (Fig. 3b), and R159-treated livers also exhibited reduced macrophages and associated fibrosis due to liver steatosis compared to Veh counterparts (Fig. 3c, d). RNA-sequencing analysis of the liver showed an increase in inflammatory and fibrotic pathways in mice treated with Veh on HFD compared to the R159 group (Fig. 3e), where Veh treated mice had upregulated Ccl2, Ccr2, Ccr5, Cx3cr1, Spp1, Src, Tlr2, and Tlr5, suggesting increased macrophage presence and immune response, and upregulated Col1a1, Ctsk, Mmp2, and Pdgfc, suggesting more fibrosis activity compared to R159 counterparts (Fig. 3f). The RNA-seq results also suggested enrichment of atherosclerosis-associated genes (Apob, Lpl, Col1a1, Sele and Vcam1) in the Veh group compared to the R159 group. Serum lipid analysis revealed that R159 treatment also significantly reduced total cholesterol and LDL/VLDL, resulting in a higher HDL to LDL/VLDL ratio trends (Fig. 3g). Taken together, R159 treatment effectively mitigated systemic inflammatory responses of major metabolic organs and weight gain in mice on HFD and provided substantial protection to liver function. During R159 treatment, mice did not exhibit any notable adverse effects or signs of toxicity, and histological analysis of major organs showed no anomalies caused by the drug (Supplementary Fig. 2), demonstrating its potential as a therapeutic intervention for reducing inflammatory responses in metabolic organs and lowering systemic inflammation levels with minimal side effects.
R159 reduces weight gain and systemic inflammation in mice on HFD. (a) Experimental schematic of R159 and high-fat diet (HFD)-driven systemic chronic inflammation. 12-month-old mice were fed HFD for 5 weeks to induce systemic inflammation before R159 or vehicle (Veh) administration. R159 or Veh was injected twice a week intraperitoneally for 12 weeks with HFD continuation; mice were aged 16 months by the end of the experiment. (b) Weight change of Veh or R159-treated mice during HFD, n = 9 per group. Multiple unpaired t-tests with FDR = 5% were used to compare each time point, q-values less than 0.05 were considered significant. (c) Serum IL-6, MCP-1, and CRP levels of mice on HFD treated with Veh or R159 measured by ELISA. Two-tailed Student’s t-test, p-values less than 0.05 were considered significant.
R159 reduces HFD-induced inflammation in major metabolic organs and reduces systemic cholesterol levels in blood plasma. (a) Adipose tissue expression of Ccl2 (MCP-1), Tnf\(\:\alpha\:\) and Il-1\(\:\beta\:\) relative to housekeeping gene Rpl7 in mice on HFD treated by Veh or R159. Representative images of CD68 staining of adipose tissue (b) and liver (c) of mice on HFD treated by Veh or R159 and quantitative analysis. (d) Representative images of Picro Sirius Red staining marking fibrosis in the liver tissue of mice treated by Veh or R159 and quantitative analysis. (e) Enriched terms for genes upregulated in the liver of mice treated by Veh compared to R159 counterparts analyzed based on RNA-seq data (n = 3 per group). (f) Heat map showing differentially regulated genes (HFD-R159 vs. HFD-Veh) associated with inflammation and fibrosis in the liver from RNA-seq (n = 3 per group). (g) Serum levels of total cholesterol, High Density Lipoprotein (HDL), Low Density Lipoprotein (LDL)/ Very Low Density Lipoprotein (VLDL), and ratio of HDL levels to LDL/VLDL levels of HFD-fed mice treated by Veh or R159. Two-tailed Student’s t-test was used for statistical analysis, p-values less than 0.05 were considered significant.
R159 mitigates degenerative changes induced by HFD in musculoskeletal tissues
Previous studies have shown that chronic inflammation is one of the major drivers of bone and cartilage catabolism in various diseases20,21,22. Micro-computed tomography (µCT) was utilized to analyze bone mineral density of trabecular bone beneath the growth plate of proximal tibia (Fig. 4a), and we observed that bone loss induced by HFD was significantly mitigated by R159 (Fig. 4b). Furthermore, R159-treated mice showed reduced osteoclast activity marked by TRAP staining in trabecular bone sections as well as lower expression of RANKL (Tnfsf11), the gene required for osteoclast formation, in bone marrow cells compared to Veh counterparts (Fig. 4c,d). HFD is known to induce mild, slowly progressing osteoarthritis (OA) as a result of chronic systemic inflammation. We next tested if R159 treatment reduces the incidence and severity of OA in mice on HFD diet. The majority of Veh-treated mice exhibited more advanced OA compared to R159-treated mice but the protective effect of the drug was moderate (Fig. 4e).
HFD-induced musculoskeletal degeneration in mice on HFD treated by Veh or R159. (a) The selected area of interest for trabecular bone density beneath the growth plate of the proximal tibia measured by Micro-CT scan. (b) Representative 3D constructed images of trabecular bone density of Veh or R159 treated mice on HFD and quantitative analysis. (c) Representative images of TRAP staining (red) in trabecular bone (green) of Veh or R159 treated mice on HFD and quantitative analysis. (d) Expression of RANKL (Tnfsf11) relative to housekeeping gene Rpl7 in bone marrow cells of Veh or R159 treated mice on HFD. Two-tailed Student’s t-test was used for statistical analysis of (b-d) and p-values less than 0.05 were considered significant. (e) Representative images of Toluidine Blue staining on knee joint articular cartilage from mice on HFD treated with Veh or R159 and osteoarthritis (OA) evaluation by OARSI score (0–4: No OA; 5–9: Mild OA; 10–14: Moderate OA; 15 and over: Severe OA). Multiple Mann-Whitney test with FDR = 10% was used for statistical analysis, q-values less than 0.05 were considered significant.
R159 mitigates inflammation-induced decline in neurogenesis
Because exhaustion of stem cells and thus reduced regenerative potential is known to be a major hallmark of aging and systemic inflammation in various tissues23, we tested if R159 has protective effects in several stem cell niches in mice with HFD-induced accelerated aging. Although R159 treatment showed no effect on hematopoietic and skin stem cell pools (Fig. 5), R159 provided significant protective effects against inflammation-induced reduction of neurogenesis in the hippocampal area (Fig. 6). The dentate gyrus of the hippocampal region of brains from mice on HFD was assessed for neurogenesis. Cells with radial glial branch morphology that were positive for Nestin were counted as quiescent neural stem cells (NSCs). Among these cells, Minichromosome Maintenance Complex Component 2 (MCM2)-positive cells mark cells under mitosis, indicating proliferating NSCs. Doublecortin (DCX) marks a microtubule protein involved in cell migration of immature neurons, indicating neurogenesis. R159-treated mice on HFD showed significantly enhanced NSC maintenance and proliferation, and substantially increased neurogenesis compared to Veh-treated counterparts (Fig. 6).
R159 has no/ minimal effect on stem cells with high turnover potential. (a) Representative flow cytometry plots of hematopoietic stem cells and progenitor cell isolation. Lineage−/c-Kit+/Sca1+/CD34dim were considered hematopoietic stem cells and Lineage−/c-Kit+/Sca1+/CD34+ were considered hematopoietic progenitor cells. (b) Quantification of Lineage− cell ratio in total bone marrow cells. (c) Quantification of hematopoietic stem cells and progenitor cells in Lineage− cells. (d) Representative flow cytometry plots of hair follicle stem cell and progenitor cell isolation. ITGA6+/ITGB1+/Sca1−/CD34+ were considered hair follicle stem cells and ITGA6+/ITGB1+/Sca1+/CD34− were considered infundibulum/interfollicular progenitors. (e) Quantification of hair follicle stem cells or progenitors in ITGA6+/ITGB1+ cells. Two-tailed Student’s t-test was used statistical analysis, p-values less than 0.05 were considered significant.
Neuronal stem cells and neurogenesis of mice on HFD treated with Veh or R159. Representative images of Nestin (green) or MCM (red) cells (a) and Doublecortin (DCX, red) (b) in the dentate gyrus of hippocampus from mice on HFD treated with Veh or R159. (c) Quantification of total NSCs, proliferating NSCs, and new neurons; Two-tailed Student’s t-test, p-values less than 0.05 were considered significant.
R159 increased activity levels in mice on HFD
The observed tissue structural and molecular changes with R159 administration showed that the effects of chronic systemic inflammation can be mitigated in multiple tissues in aged mice on HFD. Lastly, we investigated whether these changes resulted in an overall functional improvement. Reduction in activity levels is associated with chronic systemic inflammation observed in aging and obesity in both animals and humans24,25,26, so we measured the voluntary activity levels of mice on HFD treated with either R159 or Veh via wheel running assay (Fig. 7a). R159-treated mice maintained their activity levels despite aging and weight gain in contrast to Veh-treated mice, which exhibited a significant decline in activity levels (Fig. 7b). Interestingly, motivation levels were not different between the groups, suggesting that R159-treated mice could endure longer and/or faster runs, suggesting less overall frailty (Fig. 7c). Hind leg muscle was examined for key inflammation and oxidative metabolism gene expression, but no significant differences were observed between the Veh and R159 groups (Fig. 7d).
Evaluation of activity levels of mice on HFD treated with Veh or R159. (a, b) Voluntary wheel running exercise activity and behavior were measured before and during the drug treatment to assess activity levels. Multiple unpaired t-tests with FDR = 5% were used for the statistical analysis, q-values less than 0.05 were considered significant. (c) Motivation for voluntary exercise behavior was evaluated by counting the number of attempts. Multiple Mann-Whitney tests with FDR = 5% were used for the statistical analysis, q-values less than 0.05 were considered significant. (d) Hind leg muscle gene expression of Il-6, Ccl2, Tnfα, Il-1β, and Nos2 relative to housekeeping gene Rpl7 in mice on HFD treated by Veh or R159. Two-tailed Student’s t-test was used for statistical analysis, p-values less than 0.05 were considered significant.
Discussion
Recent literature highlights gp130 and similar major receptors as complex multimodal sensors, not mere on/off switches, finely tuning diverse signaling pathways27,28,29. Selectively modulating a non-canonical gp130 signaling pathway holds unique therapeutic potential by preserving the beneficial canonical signaling while addressing systemic chronic inflammation associated with aging and obesity. Moreover, gp130 modulation offers a broader impact than blocking a specific IL-6 family cytokine, as all IL-6 family cytokines transduce their signaling through gp130.
In our previous study, we identified a novel non-canonical signaling pathway by the gp130-Y814 residue that is responsible for inflammation and fibrosis via SFK activation in an acute injury model16. The current study demonstrates that inactivation of gp130-Y814/SFK via systemic R159 administration in mice diminished the inflammatory response to HFD in adipose tissue and liver, the major metabolic organs. The disruption of gp130/SFK activation attenuated the upregulation of Ccl2 (MCP-1) in adipose tissue and liver, which resulted in reduced systemic MCP-1 levels. MCP-1 is a classical monocyte recruiter that attracts monocytes from the bone marrow into blood circulation, where they are then recruited to tissues and differentiate into macrophages30,31. The decreased MCP-1 led to a reduction in tissue macrophages, validated by reduced CD68 staining in the adipose tissue and liver sections from R159-treated mice. This result highlights the potential of gp130-Y814/SFK inactivation to curb systemic chronic inflammation and associated tissue damages such as liver fibrosis by mitigating the recruitment of monocytes and pro-inflammatory macrophages. The reduction in tissue macrophages is particularly significant as they are key players in perpetuating chronic inflammation32.
Histological examination and RNA-seq of the liver showed that Veh-treated mice had more severe liver fibrosis compared to R159-treated mice. Non-alcoholic fatty liver disease (NAFLD), precipitated by obesity from HFD, is characterized by excess fat accumulation, fibrosis, and increased immune cell presence33. NAFLD may progress to more severe stages like steatohepatitis, cirrhosis, and hepatocellular carcinoma if left untreated34, underscoring the clinical importance of effective interventions. Our findings align with existing literature which demonstrates that inhibition of SFKs can alleviate liver fibrosis caused by chemical injuries and metabolic stress35,36, suggesting gp130-Y814 as a promising potential therapeutic target for NAFLD. While exploring these implications for NAFLD falls beyond our current scope, our results encourage further investigation.
Remarkably, R159-treated mice were protected against the loss of trabecular bone and degeneration of cartilage typically prompted by chronic inflammation in aging and obesity. Bone remodeling, a dynamic process involving osteoblasts constructing the bone matrix and osteoclasts degrading the matrix, can be disrupted by chronic inflammation, often leading to bone density loss driven by overactive osteoclasts21,37. IL-6 family cytokines influence this process by regulating RANKL, which is pivotal for osteoclast differentiation38, and the current study supports the link between IL-6 and RANKL by reporting that inhibition of gp130-Y814 significantly reduced RANKL transcription in obesity-driven systemic inflammatory conditions in vivo. Furthermore, the crucial role of SRC kinases in osteoclast function is demonstrated by osteopetrosis in SRC-deficient mice39, therefore, inhibiting gp130/SFK signaling provides a dual protective mechanism against bone density loss in a chronic inflammatory condition by reducing RANKL transcription and SFK activity. Additionally, SFK activity is reported to be critical to cartilage degeneration by promoting cartilage matrix degradation and synovial inflammation40,41.
Previous studies by our group demonstrated genetic or pharmacologic inhibition of gp130-Y814 signaling enhances cartilage preservation in post-traumatic osteoarthritis models in both mice and dogs16. While we observed a trend in attenuated cartilage degeneration in systemic R159-treated mice compared to the Veh group, the effect was not as substantial as the previous intra-articular injection model16. This discrepancy is likely attributed to the limitations of systemic administration affecting the drug’s bioavailability within the synovial capsule, resulting in less effective cartilage protection compared to direct intra-articular injection.
Systemic chronic inflammation suppresses neurogenesis in humans and mice42, and HFD is reported to exacerbate neurodegeneration in the hippocampus by disrupting the blood-brain barrier, especially in aged mice43. The hippocampus, a critical brain region for memory formation, harbors NSCs that continuously replenish neurons into adulthood and the NSC niche has a functional association with learning, memory, and mental health44. NSCs and progenitor cells diminish with age and obesity, likely contributing to the cognitive decline observed in aged, obese animals45,46. R159-treated mice showed substantially better NSC numbers, NSC proliferation, and neurogenesis compared to the Veh group. While reduced levels of systemic inflammation may be attributed to the protective effect of R159 against neurodegeneration induced by HFD, the striking differences in NSC niche preservation and neurogenesis suggest a potentially unknown role of IL-6/gp130-Y814 signaling in brain health, warranting further investigation.
IL-6 is a major cytokine of the senescence-associated secretory phenotype (SASP), a complex mix of factors secreted by senescent cells that can influence inflammation and tissue remodeling by affecting the surrounding cells and tissue environment47. Cell senescence and SASP not only increase with aging but also with increasing BMI, which is why obesity is considered a form of accelerated aging48. SASP-driven systemic chronic inflammation is associated with dementia, depression, atherosclerosis, cancers, diabetes, and mortality49. These morbidities often coexist in aging and obesity, making it hard to treat the root cause, and multimorbidity-driven systemic chronic inflammation perpetuates a pathological feed-forward loop of inflammaging23. Our findings demonstrate that selective disruption of IL-6/gp130-Y814 signaling significantly reduced the levels of systemic chronic inflammation, and while R159 did not eliminate SASP component IL-6, it may help mitigate the impact of SASP by reducing levels of subsequent systemic inflammation and degenerative changes across multiple tissues, such as liver, bone, and brain. Consequently, this could lead to an improved healthspan, as reflected in higher voluntary physical activity. Increased physical activity, in turn, promotes better bone density50 and improves the hippocampus function51, providing a clear example of breaking the pathological cycle of inflammaging. Interestingly, levels of inflammation and oxidative markers in muscle were not significantly different between the Veh and R159 groups, suggesting that the increased physical activity was not a direct consequence of inflammation or metabolism in the muscle.
R159-treated mice gained less weight compared to the Veh group, likely due to a combination of factors. First, R159-treated mice maintained higher levels of voluntary physical activity, which could increase energy expenditure and reduce weight gain. Additionally, RNA-seq analysis of liver tissue suggested that R159 improved lipid metabolism, potentially leading to more efficient fat utilization and storage. While we do not claim that R159 directly modulates lipid metabolism, the serum lipid panel results showed lower levels of total cholesterol and LDL/VLDL in the R159-treated group, which aligns with improved lipid handling. R159 treatment reduced macrophage infiltration and fibrosis in the liver, supporting the preservation of liver function—an organ where lipid metabolism is a major function. These combined effects on physical activity, liver health, and metabolic processes likely explain the reduced weight gain observed in R159-treated mice on HFD.
Studies have demonstrated the protective effects of blocking IL-6 family cytokines, including IL-6 and IL-11, on inflammation and fibrosis52,53,54,55. A recent publication showed that IL-11 inhibition improved metabolic decline and frailty of old mice and extended the lifespan of mice by almost 25%53. However, these interventions are limited to targeting a single cytokine and may lead to adverse effects due to the complex roles of IL-6 family cytokines in immune regulation, tissue regeneration, and metabolic homeostasis. Modulating gp130 offers a broader impact, as it transduces the signaling of all IL-6 family cytokines, and by selectively modulating gp130 signaling, we can preserve beneficial signaling pathways while effectively addressing inflammation, providing a better targeted therapeutic approach.
The current study has some limitations. All data presented is based on C57BL/6J male mice because female mice are reported to be less vulnerable to HFD-induced chronic inflammation, metabolic disorders, and degeneration including glucose sensitivity, bone density loss, and cognitive decline56,57,58,59. Additionally, although the ADME profile of R159 was performed, no detailed pharmacokinetic or pharmacodynamic studies of the drug were performed in vivo. Detailed pharmacological assessment is required to explore the bioavailability of the drug in the joints, central nervous system, and other locations notoriously difficult for small molecule targeting, which will be essential for optimization of the treatment regimen in preclinical studies.
Collectively, this study highlights the potential of selectively targeting SFK signaling downstream of IL-6/gp130 as an effective approach to reducing systemic chronic inflammation. While degenerative changes and tissue senescence are unavoidable consequences of obesity and aging, this study indicates that the related systemic responses and inflammation-driven multimorbidity can be therapeutically addressed.
Methods and materials
Ethics
All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Southern California (21242_TR01) and met or exceeded the requirements of the Public Health Service/National Institutes of Health and the Animal Welfare Act. All experiments were performed in accordance with Animal Research: Reporting of In Vivo Experiments (ARRIVE) Guidelines. Additionally, all methods were performed in accordance with the relevant guidelines and regulations.
Animals and treatments
All animals used in this study were C57BL/6J males purchased from Jackson Laboratory. 11 months old mice were purchased, acclimated, and fed a normal diet for 1 month until they reached 12-months-old. Mice were fed a 60% high-fat diet (HFD) (Envigo, TD.06414) from 12-months-old for 5 weeks to induce obesity before R159 or vehicle treatment. Mice were 13-months-old when R159 or vehicle treatment began and were continuously given a high-fat diet during the 12-week R159/vehicle treatment period, and experimental mice were sacrificed at 16-months-old.
Drug R159 was dissolved in a 1% w/v Captisol solution in 0.9% saline at a concentration of 3 mg/mL and administered by intraperitoneal injection at 10 µg per gram body weight. The vehicle group was injected with a 1% w/v Captisol solution in 0.9% saline alone by intraperitoneal injection; the injection volume was 200 µL.
Serum ELISA
Mouse blood was collected from the submandibular vein and left to clot for 2 h at room temperature before centrifuging for 20 min at 2000 x g. Collected serum was flash-frozen in liquid nitrogen and kept in -80 °C until the assay. ELISA kits were purchased from R&D Systems and experiment were performed following the manufacturer’s instructions; Mouse CRP (MCRP00), Mouse IL-6 (M6000B), Mouse CCL2/MCP-1 (MJE00B).
Quantitative real-time PCR
Total RNA was extracted using the RNeasy Mini Kit (Qiagen) following the manufacturer’s protocol. For real-time PCR, cDNA was generated using the Maxima First Strand cDNA Synthesis Kit (Thermo Scientific). Power SYBR Green (Applied Biosystems) was used for RT-PCR amplification and detection was performed using ViiA7 (Life Technologies) or Step One Plus Real-Time PCR system (Applied Biolsystems). The comparative Ct method for relative quantification (1/2^(ΔCt)) was used to quantitate gene expression, where results were normalized to Rpl7 (ribosomal protein L7) expression levels. Primer sequences used were: housekeeping gene Rpl7: forward 5’ ACCGCACTGAGATTCGGATG 3’; reverse 5’ GAACCTTACGAACCTTTGGGC 3’, Ccl2: forward 5’ TTAAAAACCTGGATCGGAACCAA 3’; reverse 5’ GCATTAGCTTCAGATTTACGGGT 3’, Tnfα: forward 5’ GACGTGGAACTGGCAGAAGAG 3’; reverse 5’ TTGGTGGTT TGTGAGTGTGAG 3’, Tnfsf11: forward 5’ CAGCATCGCTCTGTTCCTGTA 3’; reverse 5’ CTGCGTTTTCATGGAGTCTCA 3’, Il1β: forward 5’ TTCAGGCAGGCAGTATCACTC 3’; reverse 5’ GAAGGTCCACGGGAAAGACAC 3’; Il6: forward 5’ CTGCAAGAGACTTCCATCCAG 3’; reverse 5’ AGTGGTATAGACAGGTCTGTTGG 3’, Nos2: forward 5’ CTATGTCCTATCTCCATTCT 3’; reverse 5’ CATGACCTTTCGCATTAG 3’.
RNA sequencing library preparation and sequencing
Total RNA was isolated using QIAGEN RNeasy Mini kit (Qiagen) and quantified using Qubit fluorometer (ThermoFisher Scientific). Quality of the isolated RNA was checked using Agilent Bioanalyzer 2100. Universal Plus mRNA-Seq Library with NuQuant (TECAN) was used to generate stranded RNA-seq libraries. Briefly, poly(A) RNA was selected followed by RNA fragmentation. Double stranded cDNA was generated thereafter using a mixture of random and oilgo(dT) priming. The library was then constructed by end repairing the cDNA to generate blunt ends, ligation of Unique Dual Index (UDI) adaptors, strand selection and PCR amplification. Different adapters were used for multiplexing samples in one lane. The quality of the purified libraries was further assessed using Qubit fluorometer and Agilent Tapestation. Libraries were pooled and sequencing was performed at UCLA Technology Center for Genomics & Bioinformatics (TCGB) core on NovaSeq SP/ X Plus with paired end 50 bp reads. Data quality check was done on Illumina SAV. Demultiplexing was performed with Illumina CASAVA 1.8.2.
RNA sequencing data analysis
Quality of the raw fastq files were determined using FastQC (v0.11.9) (https://www.bioinformatics.babraham.ac.uk/projects/fastqc/). Adapters were trimmed using Cutadapt60. Trimmed and filtered reads were aligned to mouse reference genome (mm39- GENCODE Release M30) using STAR aligner (version 2.7.8a)61. Transcript levels were quantified to the reference using a modified expectation-maximization (EM) algorithm. Normalization was done using median ratio method. Differential expression analysis was performed using DESeq2 (v3.5)62. Genes were considered to be differentially expressed based on fold change > 1.5 and p-value < 0.05. Functional enrichment analysis for the differentially expressed genes was performed using Ingenuity Pathway analysis (IPA, Qiagen). Heatmaps were generated in R (v4.2.3) using pheatmap package (v1.0.12).
Picro sirius red staining
Mouse livers were dissected and fixed in 4% PFA overnight. Tissues were embedded in paraffin and cut at a thickness of 5 μm. Deparaffinized and rehydrated sections were stained with Picro-Sirius Red Stain Kit (Abcam, ab15068) according to the manufacturer’s instructions. Stained sections were imaged using a Zeiss Axio Imager.A2 Microscope Axiocam 105 color camera at three random locations. Positive staining was quantified using ImageJ and an average of three images per sample was used for analysis.
Immunohistochemistry (IHC)
Mouse tissues were dissected and fixed in 4% PFA for 24 h at 4 °C. Mouse legs were decalcified with 14% EDTA, pH 7.4, for 14–21 days at 4 °C before paraffin embedding. Tissues were embedded in paraffin and cut at a thickness of 5 μm. Antigen was retrieved by Tris-HCL pH 10 (Sigma) at 95 °C. Sections were blocked with 2.5% normal horse serum for 1 h at room temperature and incubated with primary antibody (CD68, Invitrogen, PA5-178996) in 1% BSA at 4 °C overnight. Slides were washed and incubated at room temperature for an hour in secondary antibody-HRP (Vector Laboratories, MP-7401). Antibodies were then visualized by peroxidase substrate kit DAB (Vector Laboratories, SK-4100). Slides were imaged using a Zeiss Axio Imager.A2 Microscope Axiocam 105 color camera with Zen 2 program at three random locations. Positive stain was quantified using ImageJ and an average of three images per sample was used for analysis.
Micro-computed tomography (µCT) data collection and analysis
Mouse legs were dissected and fixed in 4% PFA for 24 h and scanned with XT H 225 ST (Nikon) in 20 μm resolution, at 80 kV energy, 120 µA current, and 9.6 W power at USC Molecular Imaging Center. Trabecular bone under the growth plate near the proximal tibia was reconstructed and analyzed using Amira (Thermo Fisher Scientific). 1 mm length of tibia bone mineral density was measured by bone volume over total volume (BV/TV).
Flow cytometry
Flow cytometry was performed on a BD FACS Aria II cell sorter. For hematopoietic stem cells, bone marrow cells were stained using BD Stemflow™ Mouse Hematopoietic Stem and Progenitor Cell Isolation Kit (BD, 560492) following the manufacturer’s instructions. For skin stem cells, populations of interest were based on negative DAPI (Fisher Scientific, 5748) expression and surface marker expressions ITGA6 (Invitrogen, 12-0495-82), ITGB1(BioLegend, 102215), CD34 (Invitrogen, 11-0341-82), Sca1 (BioLegend, 122513) were used63. Flow cytometry data was analyzed using FlowJo software.
TRAP staining and quantification
Mouse legs were dissected and fixed in 4% PFA for 24 h, then decalcified with 14% EDTA (pH7.4) for 21 days at 4 °C. Tissues were embedded in paraffin and cut at a thickness of 5 μm. Deparaffinized and rehydrated sections were incubated in 50 mL TRAP staining medium (110 mM Sodium Acetate (Sigma, S-2889), 50 mM Tartaric Acid (Sigma, T-6521), 4.5 mM Naphthol AS-BI Phosphate (Sigma, N-2125) in distilled water) for 1 h at 37 °C. After incubation, 1 mL of Pararosaniline Dye in Sodium Nitrite solution was mixed into the medium and incubated for 30 min at 37 °C. Sections were counter stained with 0.08% Fast Green for 90 s. To quantify TRAP staining, images were taken from three different locations per sample. Slides were imaged using a Zeiss Axio Imager.A2 Microscope Axiocam 105 color camera with Zen 2 program at three random locations. Positive stains were quantified using ImageJ and an average of three images per sample was used for analysis.
OARSI scoring
Mouse legs were dissected and fixed in 4% PFA for 24 h, then decalcified with 14% EDTA (pH7.4) for 21 days at 4 °C. Knee joint tissues were embedded in the sagittal plane in paraffin and cut at a thickness of 5 μm. For osteoarthritis grade evaluation, deparaffinized and rehydrated sections were stained in 0.4% Toluidine Blue solution for 10 min at room temperature and counterstained in 0.02% Fast Green for 3 min. Research Society International (OARSI) scoring performed as described previously64. Observers performing the analysis were blinded from the sample IDs.
Western blot
Cells were lysed in RIPA Lysis and Extraction Buffer (Thermo Scientific) containing Pierce Halt Protease inhibitors (Thermo Scientific) followed by sonication with a 15-second pulse at a power output of 2 using the VirSonic 100 (SP Industries Company). Protein concentrations were determined by BCA protein assay (Thermo Scientific). Proteins were resolved with SDS-PAGE utilizing 4–15% Mini-PROTEAN TGX Precast Gels (Biorad) and transferred to Trans-Blot Turbo Transfer Packs (Biorad) with a 0.2-µm pore-size nitrocellulose membrane (Biorad). Nitrocellulose membranes were blocked in 5% nonfat milk in 0.05% (v/v) Tween 20 (PBST) (Corning). Membranes were then incubated with primary antibodies p-gp130 Y814 (Evseenko lab, AbClonal), SRC (Cell Signaling, 2123), p-SRC (Cell Signaling, 6943), or Histone H3 (Cell Signaling, 9515) overnight at 4 °C. After washing, membranes were incubated with rabbit IgG-HRP (Thermo Scientific, 31460) for 1 h at room temperature. After washing, membranes were developed with the Clarity Western ECL Blotting Substrate (Biorad). Quantification was performed using ImageJ.
Voluntary activity assay
Mice were placed in a cage individually with access to a wheel (Panlab/Harvard apparatus, LE904) for 24 h. The wheel movement was tracked continuously and recorded automatically in 30-minute increments by a multicounter (Panlab/Harvard apparatus, LE3806). The total number of wheel turns was used to assess activity levels, and the number of attempts was used to assess motivation levels.
Brain tissue processing and staining
Mice were deeply anesthetized with isoflurane and then transcardially perfused with 10 units of heparin-PBS. The brains were isolated and fixed overnight in 4% PFA, followed by incubation in 30% sucrose solution for a subsequent 48 h prior to sectioning. Frozen coronal sections in 45-µm thickness through the entire dentate gyrus were performed using a sliding microtome (SM2010R, Leica, Wetzlar, Germany). The sections were then transferred to a cryoprotectant solution (27.3% sucrose, 45.5% glycerol, 27.3% ethylene glycol) and stored at − 20 °C until processing for immunohistochemistry staining.
Immunohistology was performed with antibodies as previously described65. Briefly, coronal sections of the hemisphere were stereologically sampled throughout the entire hippocampus (5 - 6 sections per dentate gyrus), mounted on SuperFrost Plus slides (Thermo Scientific), dried overnight, rinsed in PBS, incubated in 0.01 mol/L citric buffer (pH6.5) for 40 min at 95 - 98 °C, and rinsed again in PBS. Subsequently, the sections were incubated 2 overnights at 4 °C with primary antibodies against the following antigens: Goat anti-Nestin (R&D, AF2736), mouse anti-MCM2 (BD Laboratories, 610701), rabbit anti-DCX (Cell Signaling Technology, 4604 S). Sections were then washed with PBS and incubated in appropriate secondary antibodies conjugated to fluorophores (Jackson Immunoresearch) with DAPI counterstaining (Roche, 10236276001). Stained sections were washed, air dried, and coverslipped with PVA/DABCO.
Quantification of NSCs and neurogenesis
Images of the stained sections were acquired as a tiled z-stack across the area and the depth containing the dentate gyrus region using a confocal microscope system (Axio.A1 Observer with LSM700 Scanhead, Carl Zeiss, Germany) at 40X. Morphological and co-labeling analysis was done using ZEN 2012 SP1 (black edition, Carl Zeiss, Germany). Nestin+ only cells with radial glial branch are quiescent neural stem cells, same morphological cells that are Nestin+/Mcm2+ indicate active neural stem cells, and DCX+ only cells repregent neurogenesis. The neural stem cell activation rate percentages were calculated through dividing the number of active neural stem cells by the number of total neural stem cells (both Nestin+/Mcm2- and Nestin+/Mcm2+ cells). The total neural stem cell numbers and neurogenesis data were quantified by cells/mm3.
Statistical analysis
The numbers of biological replicates for each experiment are indicated in the figure legends. Unless otherwise indicated, statistical analysis was performed using one-way ANOVA followed by the Tukey test to compare more than two groups or two-tailed Student’s t test to compare two groups. P values less than 0.05 were considered significant.
Data availability
All data is deposited in GEO data base and is available under the accession number GSE273795.
References
Ferrucci, L. & Fabbri, E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 15, 505–522 (2018).
Rodrigues, L. P. et al. Hallmarks of aging and immunosenescence: Connecting the dots. Cytokine Growth Fact. Rev. 59, 9–21 (2021).
Bettcher, B. M. et al. Increases in a pro-inflammatory chemokine, MCP-1, are related to decreases in memory over time. Front. Aging Neurosci. https://doi.org/10.3389/fnagi.2019.00025 (2019).
Alberro, A. et al. Inflammaging markers characteristic of advanced age show similar levels with frailty and dependency. Sci. Rep. 11, 4358 (2021).
Visser, M., Bouter, L. M., McQuillan, G. M., Wener, M. H. & Harris, T. B. Elevated C-reactive protein levels in overweight and obese adults. JAMA 282, 2131–2135 (1999).
O’Doherty, M. G. et al. Repeated measures of body mass index and C-reactive protein in relation to all-cause mortality and cardiovascular disease: Results from the consortium on health and ageing network of cohorts in Europe and the United States (CHANCES). Eur. J. Epidemiol. 29, 887–897 (2014).
Chen, C. et al. Combined associations of hs-CRP and cognitive function with all-cause mortality among oldest-old adults in Chinese longevity areas: A prospective cohort study. Immun. Ageing I A 16, 30 (2019).
Maluf, C. B. et al. Association between C reactive protein and all-cause mortality in the ELSA-Brasil cohort. J. Epidemiol. Commun. Health 74, 421 (2020).
Yousefzadeh, M. J. et al. Circulating levels of monocyte chemoattractant protein-1 as a potential measure of biological age in mice and frailty in humans. Aging Cell 17, e12706 (2018).
Yue, Z. et al. Senescence-associated secretory phenotype and its impact on oral immune homeostasis. Front. Immunol. 13, 1019313 (2022).
Coppé, J.-P., Desprez, P.-Y., Krtolica, A. & Campisi, J. The senescence-associated secretory phenotype: The dark side of tumor suppression. Annu. Rev. Pathol. 5, 99–118 (2010).
Kumari, R. & Jat, P. Mechanisms of cellular senescence: Cell cycle arrest and senescence associated secretory phenotype. Front. Cell Dev. Biol. 9, 645593 (2021).
Ridker, P. M. C-reactive protein: Eighty years from discovery to emergence as a major risk marker for cardiovascular disease. Clin. Chem. 55, 209–215 (2009).
Hosaka, K. et al. Monocyte chemotactic protein-1–interleukin-6–osteopontin pathway of intra-aneurysmal tissue healing. Stroke 48, 1052–1060 (2017).
Biswas, P. et al. Interleukin-6 induces monocyte chemotactic protein-1 in peripheral blood mononuclear cells and in the U937 cell line. Blood 91, 258–265 (1998).
Shkhyan, R. et al. Inhibition of a signaling modality within the gp130 receptor enhances tissue regeneration and mitigates osteoarthritis. Sci. Transl. Med. 15, eabq2395 (2023).
Duan, Y., Zeng, L. & Xu, K. Inflammatory links between high fat diets and diseases. Front. Immunol. 9, (2018).
Kanda, H. et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Invest. 116, 1494–1505 (2006).
King, D. E., Egan, B. M. & Geesey, M. E. Relation of dietary fat and fiber to elevation of C-reactive protein. Am. J. Cardiol. 92, 1335–1339 (2003).
Bjarnason, I. et al. Reduced bone density in patients with inflammatory bowel disease. Gut 40, 228–233 (1997).
Abdelmagid, S. M., Barbe, M. F. & Safadi, F. F. Role of inflammation in the aging bones. Life Sci. 123, 25–34 (2015).
Greene, M. A. & Loeser, R. F. Aging-related inflammation in osteoarthritis. Osteoarthr. Cartil. 23, 1966–1971 (2015).
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Inflammaging: A new immune–metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Wärnberg, J., Cunningham, K., Romeo, J. & Marcos, A. Physical activity, exercise and low-grade systemic inflammation. Proc. Nutr. Soc. 69, 400–406 (2010).
Johannsen, D. L. et al. Physical activity in aging: Comparison among young, aged, and nonagenarian individuals. J. Appl. Physiol. 105, 495–501 (2008).
Hamrick, M. W. et al. Age-related loss of muscle mass and bone strength in mice is associated with a decline in physical activity and serum leptin. Bone 39, 845–853 (2006).
Garbers, C., Aparicio-Siegmund, S. & Rose-John, S. The IL-6/gp130/STAT3 signaling axis: Recent advances towards specific inhibition. Curr. Opin. Immunol. 34, 75–82 (2015).
Murakami, M., Kamimura, D. & Hirano, T. Pleiotropy and specificity: Insights from the interleukin 6 family of cytokines. Immunity 50, 812–831 (2019).
Nusse, R. & Clevers, H. Wnt/β-catenin signaling, disease, and emerging therapeutic modalities. Cell 169, 985–999 (2017).
Ingersoll, M. A., Platt, A. M., Potteaux, S. & Randolph, G. J. Monocyte trafficking in acute and chronic inflammation. Trends Immunol. 32, 470–477 (2011).
Serbina, N. V. & Pamer, E. G. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat. Immunol. 7, 311–317 (2006).
Oishi, Y. & Manabe, I. Macrophages in age-related chronic inflammatory diseases. npj Aging Mech. Dis. 2, 1–8 (2016).
Godoy-Matos, A. F., Silva Júnior, W. S. & Valerio, C. M. NAFLD as a continuum: From obesity to metabolic syndrome and diabetes. Diabetol. Metab. Syndr. 12, 1–20 (2020).
Kim, H. et al. Metabolic spectrum of liver failure in type 2 diabetes and obesity: From NAFLD to NASH to HCC. Int. J. Mol. Sci. 22, 4495 (2021).
Seo, H.-Y. et al. Src inhibition attenuates liver fibrosis by preventing hepatic stellate cell activation and decreasing connective tissue growth factor. Cells 9, 558 (2020).
Du, G. et al. Targeting Src family kinase member Fyn by Saracatinib attenuated liver fibrosis in vitro and in vivo. Cell Death Dis. 11, 1–12 (2020).
Mundy, G. R. Osteoporosis and inflammation. Nutr. Rev. 65, S147–S151 (2007).
Boyle, W. J., Simonet, W. S. & Lacey, D. L. Osteoclast differentiation and activation. Nature 423, 337–342 (2003).
Soriano, P., Montgomery, C., Geske, R. & Bradley, A. Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 64, 693–702 (1991).
Novikov, F. N. et al. Inhibition of SYK and cSrc kinases can protect bone and cartilage in preclinical models of osteoarthritis and rheumatoid arthritis. Sci. Rep. 11, 23120 (2021).
Goldring, M. B. Integrin-dependent recruitment of Src to ROS-producing endosomes in osteoarthritic cartilage. Sci. Signal. 16, e9760 (2023).
Kempuraj, D. et al. Neuroinflammation induces neurodegeneration. J. Neurol. Neurosurg. Spine 1, 1003 (2016).
Tucsek, Z. et al. Obesity in aging exacerbates blood-brain barrier disruption, neuroinflammation, and oxidative stress in the mouse hippocampus: Effects on expression of genes involved in beta-amyloid generation and Alzheimer’s disease. J. Gerontol. Ser. A 69, 1212–1226 (2014).
Peng, L. & Bonaguidi, M. A. Function and dysfunction of adult hippocampal neurogenesis in regeneration and disease. Am. J. Pathol. 188, 23–28 (2018).
Gontier, G. et al. Tet2 rescues age-related regenerative decline and enhances cognitive function in the adult mouse brain. Cell Rep. 22, 1974–1981 (2018).
Park, H. R. et al. A high-fat diet impairs neurogenesis: involvement of lipid peroxidation and brain-derived neurotrophic factor. Neurosci. Lett. 482, 235–239 (2010).
Freund, A., Orjalo, A. V., Desprez, P.-Y. & Campisi, J. Inflammatory networks during cellular senescence: Causes and consequences. Trends Mol. Med. 16, 238–246 (2010).
Tchkonia, T. et al. Fat tissue, aging, and cellular senescence. Aging Cell 9, 667–684 (2010).
Tchkonia, T., Zhu, Y., van Deursen, J., Campisi, J. & Kirkland, J. L. Cellular senescence and the senescent secretory phenotype: therapeutic opportunities. J. Clin. Invest. 123, 966–972 (2013).
Hoshi, A., Watanabe, H., Chiba, M. & Inaba, Y. Effects of exercise at different ages on bone density and mechanical properties of femoral bone of aged mice. Tohoku J. Exp. Med. 185, 15–24 (1998).
Greenwood, B. N., Strong, P. V., Foley, T. E. & Fleshner, M. A behavioral analysis of the impact of voluntary physical activity on hippocampus-dependent contextual conditioning. Hippocampus 19, 988–1001 (2009).
Sarkar, D. & Fisher, P. B. Molecular mechanisms of aging-associated inflammation. Cancer Lett. 236, 13–23 (2006).
Widjaja, A. A. et al. Inhibition of IL-11 signalling extends mammalian healthspan and lifespan. Nature https://doi.org/10.1038/s41586-024-07701-9 (2024).
Rose-John, S. Interleukin-6 signalling in health and disease. F1000Research 9, 1013 (2020).
Villa, P. et al. The interleukin-8 (IL-8/CXCL8) receptor inhibitor reparixin improves neurological deficits and reduces long-term inflammation in permanent and transient cerebral ischemia in rats. Mol. Med. 13, 125–133 (2007).
Pettersson, U. S., Waldén, T. B., Carlsson, P.-O., Jansson, L. & Phillipson, M. Female mice are protected against high-fat diet induced metabolic syndrome and increase the regulatory T cell population in adipose tissue. PLOS ONE 7, e46057 (2012).
Huang, K.-P. et al. Sex differences in response to short-term high fat diet in mice. Physiol. Behav. 221, 112894 (2020).
Hwang, L.-L. et al. Sex differences in high-fat diet-induced obesity, metabolic alterations and learning, and synaptic plasticity deficits in mice. Obesity 18, 463–469 (2010).
Gautam, J. et al. Micro-architectural changes in cancellous bone differ in female and male C57BL/6 mice with high-fat diet-induced low bone mineral density. Br. J. Nutr. 111, 1811–1821 (2014).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Dobin, A. et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Kostic, L., Sedov, E., Soteriou, D., Yosefzon, Y. & Fuchs, Y. Isolation of stem cells and progenitors from mouse epidermis. Curr. Protoc. Stem Cell Biol. 41, 1C.20.1-1C.20.11 (2017).
Glasson, S. S., Chambers, M. G., Van Den Berg, W. B. & Little, C. B. The OARSI histopathology initiative—Recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthr. Cartil. 18, S17–S23 (2010).
Bonaguidi, M. A. et al. In vivo clonal analysis reveals self-renewing and multipotent adult neural stem cell characteristics. Cell 145, 1142–1155 (2011).
Acknowledgements
We thank Dr. Thomas Lozito and Dr. Benjamin Van Handel for their helpful input on the manuscript, Tautis Skorka in the USC Molecular Imaging Center for μCT scanning, and Seth Ruffins in the USC Optical Imaging Facility for providing bone density analysis and methodology. We also thank Dr. Lei Peng for mouse perfusion and brain dissection training and the USC Department of Animal Resources for animal care. Schematics were created with BioRender.com.
Funding
Research in this study was supported by the Department of Defense grant W81XWH-13-1-0465 (DE), National Institute on Aging grants R01AG058624 (DE) and R01AG076956 (MB), National Institute of Arthritis and Musculoskeletal and Skin Diseases grant R01AR071734 (DE), National Institute of Dental and Craniofacial Research grant T90DE021982 (JT), and National Institutes of Health grants F31AG084302 (JNL) and T32HD060549 (JNL). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
DE and YL conceived the project. DE, YL, and MB designed experiments. YL conducted vehicle/drug treatment. YL, JT, SL, ACD, JM, US, JL, and DG conducted mouse tissue collection and process. YL, JNL, ACD, JM, HT, TM, and FB conducted tissue staining, imaging, and quantification. YL and SL conducted ELISA assay. YL, FN, and JY conducted RNA extraction and qPCR. AS conducted bulk RNA sequencing and analysis. YL conducted bone density analysis. NL and DG evaluated OARSI scores. JT conducted flow cytometry and analysis. YL and HT conducted voluntary activity assay. YL and TM conducted cholesterol assay. JL and RS conducted western blot. YL, JT and DE wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Denis Evseenko is a co-founder and major stockholder of Carthronix Inc. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, Y., Tassey, J., Sarkar, A. et al. Pharmacological inactivation of a non-canonical gp130 signaling arm attenuates chronic systemic inflammation and multimorbidity induced by a high-fat diet. Sci Rep 14, 31151 (2024). https://doi.org/10.1038/s41598-024-82414-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82414-7