Abstract
This study aimed to evaluate the potential of coffee parchment (CP), an underutilized agro-industrial by-product, as a functional feed additive for Nile tilapia (Oreochromis niloticus) reared in a biofloc system. Coffee parchment is rich in bioactive compounds and may offer sustainable benefits to aquaculture. To assess its effects, 300 fingerlings (average weight 18.60 ± 0.09 g) were randomly allocated to five groups and fed diets containing 0, 5, 10, 20, and 40 g kg−1 of CP for eight weeks. Growth performance, immune responses, the expression of growth-, immunity-, and antioxidant-related genes, and gut microbiota composition were examined. The results showed that CP supplementation significantly improved growth and immunological parameters, with the best overall effects observed in the CP5 group. CP5 led to significant upregulation of ghrelin, galanin, EF-1α, NPY-α, IL-1β, TNF-α, NFkB, MHC II-α, Hsp70, GPX, and Nrf2 genes. Microbiota analysis revealed 69 shared ASVs across treatments, with CP supplementation causing clear shifts in bacterial composition. CP5 reduced Proteobacteria abundance and microbial richness and evenness, suggesting selective modulation of gut communities. Polynomial regression analysis based on growth performance data estimated an optimal CP dosage range of 15.06–19.86 g kg−1. In conclusion, coffee parchment is a promising functional feed additive that enhances growth, immunity, gene expression, and gut microbiota modulation in Nile tilapia reared in biofloc systems. These findings support its application in sustainable aquaculture; however, further validation under commercial farming conditions is recommended.
Similar content being viewed by others
Introduction
In recent decades, intensive fish farming has expanded significantly to meet the growing global demand for animal protein1. Nile tilapia (Oreochromis niloticus) is a particularly important species in global aquaculture, ranking third in production with 4.4 million tons annually, accounting for approximately 9% of the world’s farmed fish output2. Its popularity stems from its high market demand, adaptability to a wide range of environmental conditions, and resilience to diseases3. However, the intensification of tilapia farming has led to increased disease incidence and environmental stress, posing serious threats to fish health and farm sustainability4. To manage disease outbreaks, conventional practices often rely on which contribute to the emergence of antimicrobial resistance, ecological degradation, and public health concerns due to residual contaminants in fish products5,6,7. As a result, there is growing interest in natural, environmentally friendly feed additives, particularly plant-based compounds for enhancing fish immunity and performance without side effects8,9. Among these, agriculture by-products have emerged as promising functional feed additives, contributing to sustainable aquaculture while aligning with circular economy principles10,11. Numerous studies highlight the benefits of such additives in enhancing fish health and performance in aquaculture systems12,13,14,15. Notably, coffee by-products stand out as a promising resource due to their abundance and rich bioactive compound profile16,17,18. They offer significant potential for improving fish health and performance, making them an attractive option for sustainable aquaculture.
Coffee is one of the most widely consumed beverages worldwide, with global production in 2023–2024 season estimated at 169.8 million 60-kg bags, projected to rise to 172.4 million bags in 2024–2025 19,20. Alongside this large-scaled production, the coffee industry generates approximately 6 million tons of waste annually, which poses significant environmental challenges if not properly managed21,22. This waste comprises about 50–60% of the coffee fruit’s mass and includes by-products such as skin (7–8%), pulp (29%), parchement (12%), and silverskin (1–2%)23,24,25. Coffee parchment, a fibrous endocarp layer that encases the green coffee bean, is particularly abundant and underutilized19,26. It is primarily composed of α-cellulose (40–49%), hemicelluloses (25–32%), lignin (33–35%), and minor fiber components (0.5–1%)27. These structural polymers are associated with valuable bioactive properties, including antioxidant and glucoregulatory activities23,28,29. However, despite its compositional richness, most coffee production facilities fail to repurpose this by-product. Large-scale industries often discard coffee parchment as waste rather than integrating it into value-added processes, thereby contributing to environmental degradation26,30,31. Utilizing coffee by-products offers an innovative and environmentally sustainable approach to enrich the coffee production value chain32. This strategy can support sustainable development while capitalizing on bioactive compounds with significant market potential23. However, little to no research has evaluated the potential of coffee parchment as a dietary supplement in aquafeeds, particularly in functional diets for Nile tilapia.
Biofloc technology (BFT) is an innovative and sustainable aquaculture approaches that reduces environmental impacts by recycling organic matter through microbial activity33,34. Bioflocs are aggregates of microorganisms, primary bacteria and microalgae, formed by manipulating the carbon/nitrogen ratio in culture water35. These microbial commmunities improve water quality, minimise harmful nitrogenous waste, and serve as a supplementary source of nutrients for farmed fish, thereby reducing reducing reliance on commercial feeds and fertilizers36. In Nile tilapia farming, BFT has been shown to enhance survival rates, promote nutrient uptake, and offer protection against pathogenic infections, positioning it as a cornerstone of sustainable aquaculture practices37. Despite the well-established benefits of BFT, the incorporation of novel plant-based feed additives into this system remains underexplored. Integrating such additives, particularly those derived from agro-industrial by-products may further enhance fish growth, immune function, and gut health by fostering beneficial interactions between dietary components and the biofloc microbial community. Therefore, this study aimed to address this research gap by evaluating the effects of dietary CP supplementation on growth performance, immune responses, gene expression, and gut microbiota composition in Nile tilapia reared under a biofloc system. The findings are expected to provide new insights into the functional use of agro-industrial waste in sustainable aquaculture production.
Materials and methods
Processing of coffee parchment (CP)
Arabica coffee parchment was obtained from the Lanna Thai Coffee Development Centre. The material was oven-dried until a stable moisture content of 10% was achieved, followed by grinding into a fine powder. The processed sample was then sent to the Division of Biotechnology, Faculty of Agro-Industry, Chiang Mai University, Chiang Mai 50,100, Thailand, for analysis of its proximate composition and bioactive compounds (Table 1).
Experimental fish
A total of 300 healthy Nile tilapia fingerlings (initial mean weight 18.60 ± 0.09 g) were sourced from a local hatchery in Chiang Mai, Thailand. The fish were acclimated to the experimental conditions by feeding them a commercial diet twice daily for two weeks prior to the introduction of the treatment diets. The fish were reared in 150-liter glass tanks. All experimental procedures adhered to the guidelines for animal care and use and were approved by the Chiang Mai University Ethics Committee (Approval No.: AG03007/2567).
Diet Preparation and experimental design
Dietary treatments containing varying levels of coffee parchment (CP) were formulated based on a standard diet, following the methodology outlined by Núñez, et al.38. The chemical composition of the diets was analyzed in accordance with AOAC guidelines AOAC39. The ingredients and their respective proportions are detailed in Table 2. Dry ingredients were thoroughly mixed, and the mixture was processed into 2-mm diameter pellets using a pelletizer after adding oil and water. The prepared feeds were stored at 4 °C until use. Following a two-week acclimation period, 300 Nile tilapia fingerlings (18.60 ± 0.09 g) were randomly distributed into five dietary treatment groups: CP0 (0 g kg−1), CP5 (5 g kg−1), CP10 (10 g kg−1), CP20 (20 g kg−1), and CP40 (40 g kg−1). Each treatment had three replicates, with 20 fish housed per 150-liter tank. The trial lasted eight weeks, with fish samples collected at week eight to assess growth performance and immunological parameters. Gene expression analysis for growth, immune, and antioxidant-related genes was conducted at the conclusion of the feeding trial. Skin mucus and blood samples were collected, and the fish were promptly returned to their tanks.
Biofloc and water management
Floc water in each experimental tank was prepared three weeks before the trial commenced. Each 150-liter tank was supplemented with 400 g of salt, 5 g of molasses, 5 g of dolomite, and 2 g of feed. To maintain a carbon-to-nitrogen (C: N) ratio of 15:1, molasses (containing 40% carbon) was added as the carbon source two hours after feeding40. The remaining nitrogen levels in the water and the carbon and nitrogen content of the feed were used to calculate the required carbon input41.
Floc volume in the tanks was measured using an Imhoff cone, following the procedure described by Avnimelech40. Briefly, water samples were gently collected from a depth of approximately 15 cm below the water surface to avoid surface debris and ensure representative sampling. One liter of water from each tank was transferred into a graduated Imhoff cone and allowed to settle undisturbed for 30 min at room temperature. After the settling period, the volume of the settled floc (biofloc solids) was recorded in milliliters (mL L−1) by reading the scale at the base of the cone. This value represents the floc volume, indicating the concentration of suspended biofloc particles in the culture water. Measurements were conducted in triplicate for each treatment to ensure consistency and accuracy.
Water quality parameters, including temperature, pH, dissolved oxygen (DO), and ammonium (NH4+), were monitored twice daily at 08:30 and 16:30 throughout the experimental period. Temperature, pH, and DO were measured using a portable multi-parameter meter (HI98196, Hanna Instruments, USA), while ammonium concentration was determined using a portable photometer (HI96733, Hanna Instruments, USA) based on the Nessler method, as outlined in the manufacturer’s protocol and standard water quality procedures42. During the feeding trial, water quality parameters remained stable: ammonium at 0.20 ± 0.07 mg L−1, temperature at 28.6 ± 0.75 °C, pH at 8.01 ± 0.64, and dissolved oxygen at 4.99 ± 0.04 mg L−1. To maintain optimal water conditions, a 5% water exchange was conducted weekly.
Growth performance analysis
Individual fish weights were recorded at the end of week eight post feeding. Growth performance indicators, including weight gain (WG), specific growth rate (SGR), feed conversion ratio (FCR), and survival rate (SR)were calculated according to the method described by Sherif, et al.43. The following formulas were used: Weight gain (WG, %) = [(FW–IW) × 100] /IW; Specific growth rate (SGR, % g/day] = 100 × [(LnFW – LnIW)/60 days; Feed conversion ratio (FCR) = FI/WG, and survival (%) = [final number/initial number] × 100. Where FW: final weight (g); IW: initial weight (g); WG: weight gain (g); FI: feed intake (g).
Immunity analysis
Collection of skin mucus and serum samples
Skin mucus samples were collected from three randomly selected fish per tank. The fish were briefly anesthetized with clove oil at a concentration of 5 mL L−1. After anesthesia, each fish was placed into a plastic bag containing 10 mL of 50 mM NaCl solution, and mucus was collected by gently rubbing the fish for one minute. The resulting solution was transferred into new tubes and stored at 4 °C for subsequent analysis.
For serum collection, blood samples were drawn from the caudal vein of three fish per replicate using a 1-mL syringe. The blood was transferred into sterile tubes without anticoagulant, allowed to clot at room temperature for one hour, and then incubated at 4 °C for four hours. The serum was separated and stored at -20 °C for further immunological assays.
Lysozyme activity
Lysozyme activity in undiluted serum and mucus was evaluated using a modified version of the Parry, et al.44 protocol, as adapted by Lubis, et al.45. In brief, 100 µL of skin mucus and 25 µL of undiluted serum from each fish were loaded in triplicate into 96-well microplates. Subsequently, 175 µL of Micrococcus lysodeikticus suspension (0.3 mg mL−1 prepared in 0.1 M citrate-phosphate buffer, pH 5.8) was added to each well. The mixture was immediately and thoroughly mixed, and the reduction in turbidity was measured at 540 nm every 30 s over a 5-minute period at 25 °C using a microplate reader. A standard curve was established using known concentrations (0–20 µg mL−1) of hen egg-white lysozyme (Sigma-Aldrich, USA). Lysozyme activity in the samples was then calculated and expressed as µg mL−1 equivalent based on this standard curve.
Peroxidase activity
Peroxidase activities in both serum and skin mucus were measured following a slightly adjusted method based on Quade and Roth46, with modifications outlined by Lubis, et al.45. Peroxidase activity was assessed using triplicate samples in flat-bottom 96-well microplates. A volume of 5 µL of either serum or skin mucus was dispensed into each well, followed by the addition of 45 µL of calcium- and magnesium-free Hank’s Balanced Salt Solution (HBSS). Subsequently, 100 µL of a chromogenic substrate solution, prepared by mixing 40 mL of distilled water, 10 mL of 30% hydrogen peroxide (H2O2, Sigma-Aldrich), and one tablet of 3,3′,5,5′-tetramethylbenzidine (TMB, Sigma-Aldrich) was added to initiate the reaction. After a brief incubation, the reaction was stopped by adding 50 µL of 2 M sulfuric acid (H2SO4), and absorbance was recorded at 450 nm using a microplate reader. Wells containing only the reaction mixture without biological samples served as blanks. One unit of peroxidase activity was defined as the amount of enzyme required to yield an absorbance increase of 1. Results were expressed as units (U) per mg of serum or mucus protein.
Alternative complement activity (ACH50)
The alternative complement activity (ACH50) was determined using the procedure described by Yanno47, with minor adjustments detailed by Lubis, et al.48. For the alternative complement activity (ACH50) assay, rabbit red blood cells (R-RBCs) were first washed twice with 0.01 M EGTA-magnesium-gelatin veronal buffer (EGTA-Mg-GVB), then centrifuged at 3000 rpm using phosphate-buffered saline (PBS). The washed cells were resuspended in 0.01 M EGTA-Mg-GVB to a final concentration of 2 × 108 cells mL−1. To prepare the 100% hemolysis control, 100 µL of R-RBC suspension was mixed with 3.4 mL of distilled water to induce complete lysis, and the absorbance of the resulting hemolysate was adjusted to approximately 0.740 at 414 nm using distilled water as the blank.
For the ACH50 test, 100 µL of fish serum was diluted with 400 µL of 0.01 M EGTA-Mg-GVB, followed by serial two-fold dilutions. To prevent premature complement activation, the diluted samples were kept on ice until use. Then, 100 µL of R-RBC suspension was added to each dilution tube, and the mixtures were incubated at 20 °C for 1.5 h with gentle, intermittent shaking.
To terminate the reaction, 3.15 mL of cold isotonic saline was added to each tube, followed by centrifugation at 1600 × g for 5 min. After centrifugation, 100 µL of the supernatant from each dilution was transferred to a 96-well plate, and absorbance was measured at 414 nm. The percentage hemolysis for each dilution was calculated by comparing the absorbance to the 100% lysis control. A log-log plot of serum volume versus percent hemolysis was used to determine the serum dilution causing 50% hemolysis. The ACH50 value (units mL−1) was calculated using the formula: ACH50 (units mL−1) = (1/K) × r × 1/2. Where K is the serum volume producing 50% hemolysis, r is the reciprocal of the dilution factor, and 1/2 is the correction factor. The assay was conducted at half the volume of the original protocol.
qPCR for immune gene expressions
Sample preparation
Gene expression levels related to immunity, antioxidation, and growth were analyzed using head kidney tissues collected from six fish per treatment group after the eight-week feeding trial. Tissue samples (25–50 mg) were placed in sterilized tubes containing 500 µL of Trizol reagent (Invitrogen, USA) and stored at − 80 °C until processing.
Total RNA was extracted using the PureLink™ RNA Mini Kit (Invitrogen, USA), following the manufacturer’s protocol. RNA concentration and purity were evaluated using a NanoDrop™ One spectrophotometer (Thermo Scientific, Wilmington, USA). Complementary DNA (cDNA) was synthesized from 1 µg of total RNA with the iScript™ cDNA Synthesis Kit (BIO-RAD, USA) according to the manufacturer’s instructions. The RT-qPCR primers utilized in this study are provided in Table 3.
Real-time PCR
Real-time PCR (qPCR) was conducted using a reaction mixture comprising 1 µL of cDNA (100 ng), 0.4 µL of each primer (10 µM), 10 µL of 2× Universal SYBR Green (BIO-RAD, USA), and DNase-free water to reach the final volume. The reactions were performed on the CFX Connect™ Real-Time PCR System (BIO-RAD, USA). The qPCR protocol adhered to the conditions described in Lubis, et al.48. Gene expression levels were calculated using the 2-ΔΔCt method Livak and Schmittgen49. The quantification cycle (Cq) values were used to determine the mRNA levels of target genes, with 18 S rRNA serving as the internal reference gene.
Analysis of gut Microbiome
To investigate the gut microbiome, intestinal samples were collected from the control group (n = 6) and each coffee pulp treatment group (n = 6 per group) at the conclusion of the experiment. Genomic DNA was extracted using the TGuide S96 Magnetic Soil/Stool DNA Kit (Tiangen Biotech, Beijing, China). The V3 and V4 regions of the bacterial 16 S rRNA gene were amplified, and the resulting amplicons were sequenced on the Illumina Novaseq 6000 platform. The raw sequencing data have been submitted to the NCBI database with the accession number PRJNA1154970.
The raw sequencing reads were preprocessed by performing quality filtering with Trimmomatic v0.33 50 and removing primers using Cutadapt 1.9.151. Denoising was conducted with Dada2 implemented within the QIIME2 framework52, resulting in the generation of amplicon sequence variants (ASVs). Taxonomic classification of ASVs was performed using a Bayesian approach against the SILVA reference database53. Venn diagrams were created to depict shared and unique ASVs across the groups, highlighting common microbial features54.
Alpha and beta diversity analyses were performed on normalized datasets. Alpha diversity, measured through observed species richness and the Shannon index, was compared among groups using ANOVA. Beta diversity was evaluated using Bray-Curtis dissimilarity and visualized with Principal Coordinate Analysis (PCoA). Significant microbial features at the genus level were identified using the Linear Discriminant Analysis Effect Size (LEfSe) method, while STAMP software was utilized to determine genera with significant differences in relative abundance between treatment groups under default parameters55.
Statistical analysis
Statistical analyses were conducted using SAS software56. Group differences were analyzed with Analysis of Variance (ANOVA), and Duncan’s Multiple Range Test was used to identify significant differences among the means. Polynomial regression analysis was employed to determine the optimal CP level, following the approach described by57. A significance threshold of P ≤ 0.05 was used throughout the study. The Kolmogorov-Smirnov test was performed to evaluate the normality of the data distribution.
Results
Growth performance
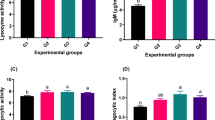
Table 4 summarizes the growth performance and feed utilization of Nile tilapia fed diets containing varying levels of coffee parchment (CP). Fish in the CP5 group exhibited significantly greater final weights, weight gain, and specific growth rates compared to the control group (CP0) and those receiving higher CP levels (CP10, CP20, and CP40). In contrast, no significant differences in growth parameters were observed between the control and CP40 groups. For feed conversion ratio (FCR), the CP5 group demonstrated significantly lower values compared to the control and CP40 groups, although no significant differences were detected among the CP5, CP10, and CP20 groups. Survival rates remained uniformly high across all treatments, with no significant variation observed (Table 4). The polynomial regression analysis (P < 0.05) shows that dietary coffee parchment (CP) significantly impacts Nile tilapia’s growth performance and feed efficiency (Fig. 1). Final body weight, weight gain, and specific growth rate peaked, while feed conversion ratio (FCR) improved at an optimal CP dosage of 15.06–19.86 g kg−1. Beyond this range, growth performance and feed efficiency declined, emphasizing the need for precise dietary optimization.
Polynomial regressions analysis (P < 0.05) between final body weight (a), weight gain (b), specific growth rate (c), and feed conversion ratio (d) of Nile tilapia and dietary levels of coffee parchment (CP) after 8 weeks post-feeding. Values expressed as means ± SE.
Immunological activities of lysozyme and peroxidase
The immunological responses of Nile tilapia, as measured by skin mucus lysozyme activity (SMLA) and peroxidase activity (SMPA), are shown in Table 5. Fish in the CP5 group exhibited the highest SMLA, significantly exceeding that of the control group (CP0). Similarly, SMPA was significantly higher in the CP5 group compared to the control and other supplemented groups. No significant differences in SMLA and SMPA were observed among the CP10, CP20, and CP40 groups. Additionally, SMLA did not differ significantly among the CP0, CP10, CP20, and CP40 groups.
Table 6 details the serum lysozyme (SL) and peroxidase (SP) activities of Nile tilapia fed diets with varying levels of coffee parchment. After eight weeks, the CP5 group demonstrated significantly greater SL and SP activities than the control and other treatment groups. Furthermore, the CP5 group showed the highest ACH50 value, significantly outperforming all other groups. No significant differences in SL were observed between the control, CP10, CP20, and CP40 groups. Similarly, SP levels did not differ significantly between the CP20, CP40, and control groups. For ACH50, no significant differences were found between the CP40 and control groups (Table 6).
mRNA expression of CP treated fish
Figure 2 presents the relative expression levels of growth-, immune-, and antioxidant-related genes in the kidney of Nile tilapia after eight weeks of feeding with graded levels of coffee parchment (CP): CP0 (control), CP5, CP10, CP20, and CP40 (g kg−1 diet). For growth-related genes (ghrelin, galanin, EF-α, and NPY-α), fish fed the CP5 diet showed the highest expression levels. All four genes were significantly upregulated in the CP5 group compared to the control and other treatments. Expression tended to decline at higher CP inclusion levels, with CP40 showing the lowest expression for most growth-related genes. Regarding immune-related genes (IL-1β, TNF-α, NF-κB, and MHC II-α), the CP5 group also exhibited the most pronounced upregulation. Expression of these genes peaked in CP5 and was significantly higher than in CP0. A gradual reduction in expression was observed with increasing CP levels, especially in CP40, where levels were similar to or lower than the control. For antioxidant-related genes (GPx, Hsp70, and Nrf2), CP5 again resulted in the highest gene expression, indicating enhanced antioxidant capacity in this group. Expression declined progressively in CP10, CP20, and CP40 groups.
Effect of coffee parchment on growth, immune and antioxidant related gene expressions in the kidney (n = 6) of Nile tilapia after feeding with experimental diets: CP0 (0 - control), CP5 (5 g kg−1 CP), CP10 (10 g kg−1 CP), CP20 (20 g kg−1 CP), and CP40 (40 g kg−1 CP). Significant differences between groups are denoted by different superscript letters (p < 0.05). Data presented are expressed as mean ± SD.
Gut microbiome
The Venn diagram (Fig. 3) displays the distribution of shared and unique amplicon sequence variants (ASVs) among treatment groups (CP0, CP5, CP10, CP20, CP40), with 69 core ASVs shared across all groups, suggesting a stable core microbiota. CP0 exhibited the highest number of unique ASVs (3,082), followed by CP10 (1,155) and CP20 (1,037), indicating microbial shifts driven by coffee parchment (CP) supplementation. Alpha diversity analysis (Fig. 4) showed a significant decrease in richness and evenness in CP5, while CP10 maintained greater microbial diversity, reflecting a more stable and diverse gut bacterial community. Beta diversity (Fig. 5) revealed distinct clustering among groups, with CP10 showing clear separation from CP0, CP20, and CP40, indicating that CP10 caused significant and structured changes in the bacterial community. Relative abundance at the phylum level (Fig. 6) identified Fusobacteria, Proteobacteria, Firmicutes, Bacteroidota, and Actinobacteria as the dominant phyla across all groups. CP10 showed a reduction in Proteobacteria, which is often linked to inflammation and gut dysbiosis, and a relative increase in Firmicutes, supporting improved gut health. The heatmap (Fig. 7) illustrated distinct bacterial composition at the genus level across treatments. CP0 showed higher levels of Cyanobacteria and Verrucomicrobiota, while CP10 and CP20 exhibited altered bacterial patterns, reflecting the influence of CP supplementation on microbial community structure. STAMP analysis (Fig. 8) showed that CP10 significantly increased the abundance of beneficial genera such as Cetobacterium compared to CP0, while CP20 increased Shinella. These shifts suggest that CP10 induced a more favorable bacterial profile linked to improved intestinal health.
Venn diagram illustrating the common and unique amplicon sequence variants (ASVs) across the different treatment groups.
Boxplots illustrating the alpha diversity indices across the treatment groups: (A) Observed richness and (B) Shannon index.
NMDS plot of beta diversity based on Bray-Curtis dissimilarity, showing separation of bacterial communities across treatment groups.
Bar chart illustrating the relative abundance of bacterial phyla across the different coffee parchment treatment groups.
Heatmap illustrating the abundance of various bacterial genera across the coffee parchment treatment groups.
STAMP analysis results showing significant differences in the mean proportions of bacterial genera between the CP0 group and other treatment groups. (A) CP5. (B) CP20.
Discussion
The efficiency of aquaculture is continuously advancing, as highlighted by recent research58. With growing concerns about regulatory measures and environmental pollution, there is an increased emphasis on enhancing aquaculture productivity34,59. The indiscriminate use of chemotherapeutic agents in aquaculture poses significant risks, such as reduced fish resistance, the development of multi-resistant pathogens, the onset of other diseases, and economic setbacks60. As a result, the industry is actively searching for effective, affordable, and sustainable alternatives, including immunostimulatory and chemotherapeutic agents61,62. One promising area of interest is plant-based immunostimulants, known for their rich content of secondary metabolites and bioactive compounds63. These natural agents have the potential to drive the adoption of more sustainable aquaculture practices on a global scale.
Our study revealed that incorporating coffee parchment (CP) into the diet of Nile tilapia for eight weeks significantly improved growth and feed efficiency compared to the control diet, with the most notable results observed at a supplementation level of 5 g kg−1 CP. To date, there is no published research on the use of CP in fish diets or its effects on the growth of any aquaculture species. However, studies investigating other plant seeds and by-products have demonstrated similar positive effects growth performance in various fish species, including European sea bass (Dicentrarchus labrax) fed okra leaves64, black rockfish (Sebastes schlegelii) fed by-products from producing yacon65, largemouth bass (Micropterus salmoides) fed fermented tea residues66, common carp (Cyprinus carpio) fed pomegranate peel67, Nile tilapia (Oreochromis niloticus) fed cumin stem, makiang seed, and dragon fruit peel68,69,70. Additionally, bagrid catfish (Mystus nemurus) fed sweet orange peel waste showed notable improvements71. The observed growth enhancement in our study may be attributed to the bioactive constituents of CP, which include high levels of fiber, phenolic compounds23,72, and chlorogenic acids73, all of which are known to positively influence fish metabolism and growth performance74,75,76. Polyphenols-rich plant materials have been shown to stimulate digestive secretions and enzyme activity, enhancing nutrient absorption and utilization, which ultimately supports animal growth77,78. Furthermore, phenolic compounds can induce metabolic adaptations that improve nutrient use efficiency and energy metabolism79. However, higher levels of CP supplementation (≥ 20 g kg−1) led to a reduction in growth performance, which may be attributed to its high fiber content (72.13%, Table 1). Excess dietary fiber can act as a prebiotic and affect gut fermentation dynamics80. While low to moderate levels of dietary fiber can be beneficial, excess fiber may reduce food intake, slower weight gain, and increase the secretion of satiety-related hormones, such as GLP-1 and PYY81,82. These hormones are stimulated by colonic fermentation products, such as short-chain fatty acids like butyrate, acetate, and propionate, which bind to free fatty acid receptors that enhance satiety signaling83. In addition to these physiological effects, CP has demonstrated promising in vitro hypoglycemic activity, such as glucose adsorption, inhibition of glucose diffusion, and α-amylase activity reduction, which may lower starch digestibility. Furthermore, CP exhibits hypolipidemic effects through the inhibition of pancreatic lipase and cholesterol binding72.
In terms of immune function, fish mucus and serum play essential roles as the first line of defense against pathogens84,85. Our study showed that the diet with CP supplementation significantly elevated lysozyme and peroxidase activities in both skin mucus and serum after 8 weeks. To our knowledge, this is the first report demonstrating the effects of CP on these innate immune markers in Nile tilapia. Nevertheless, similar benefits have been reported in other species fed fruit and plant by-products, such as Nile tilapia (Oreochromis niloticus)69,86 and gilthead seabream (Sparus aurata L.)87. Additionally, gene expression analysis revealed significant upregulation of growth-related genes (ghrelin, galanin, EF-α, and NPY-α), immunity (il-1β, MHC II-α, NFkB, and TNF-α), and antioxidant defense (hsp70, GPX, and nrf2) in CP-fed fish. Similar findings have been reported in other studies, such as largemouth bass (Micropterus salmoides) fed fermented tea residues66 and Nile tilapia (Oreochromis niloticus) fed makiang seed, dragon fruit peel, and banana flower powder68,69,88. Additional positive effects were observed in Rohu carp (Labeo rohita) fed seaweed polysaccharides89, and rainbow trout (Oncorhynchus mykiss) fed phytogenic diet90. The mechanisms underlying these responses may involve the epigenetic effects of dietary polyphenols, which can modulate gene expression via DNA methylation, histone modifications, and miRNA interactions91. Certain polyphenols also activate immune signaling pathways through interactions with cell surface receptors on immune cells, influencing intracellular cascades that regulate immunity92. Coffee parchment, a notable source of pectic polysaccharides and xylooligosaccharides80,93, may contribute further to immune modulation by stimulating dendritic cell (DC) maturation via PI3K/AKT, MAPK, NF-κB, and Dectin-1/Syk pathways, ultimately enhancing cytokine production, antigen presentation, and T-cell activation94.
Notably, elucidating the connection between physiological functions and specific gut microbial groups is essential for identifying the roles of bacteria that may benefit fish health and well-being95,96,97. Numerous studies have emphasized the significant involvement of intestinal microbiota in various physiological functions of fish, including growth, nutrient metabolism, intestinal development, and immune regulation98,99,100. Moreover, research has indicated that the richness and composition of gut microbiota can serve as a reliable indicator of fish health, with elevated levels of Fusobacteria, particularly Cetobacterium, being associated with favorable health status101,102. This study demonstrated that dietary supplementation with coffee parchment (CP) modulated the gut microbiota of Nile tilapia reared in a biofloc system in a dose-dependent manner, with the most favorable outcomes observed in the CP10 group. Key findings included higher microbial diversity in CP10, reduced abundance of potentially pathogenic Proteobacteria, and a significant increase in the beneficial genus Cetobacterium, which together indicate improved gut microbial balance. These findings align with previous studies reporting the central role of gut microbiota in supporting various physiological processes in fish, including growth, metabolism, nutrient absorption, intestinal development, and immune regulation95,103,104. Gomes, et al.105 and Lenin, et al.106 emphasized that analyzing specific bacterial clades and their relationship with host physiology is crucial for understanding their functional contributions to fish health and welfare. In this regard, our results provide further evidence that CP supplementation can positively influence host-microbiota interactions in aquaculture systems. A particularly important observation was the significant increase in Cetobacterium abundance in the CP10 group. Cetobacterium is a known commensal bacterium in freshwater fish, associated with vitamin B12 production and improved nutrient metabolism107. Prior research has suggested that a higher relative abundance of Fusobacteria/Cetobacterium, along with a lower abundance of Proteobacteria, is indicative of gut health and better performance108. This pattern was clearly observed in our study, supporting the hypothesis that CP10 helped maintain gut microbiota homeostasis. Furthermore, the observed shifts in microbiota composition are consistent with previous studies involving prebiotics, paraprobiotics, and postbiotics. For example, Chen, et al.95 showed that supplementation with compounds derived from autochthonous microorganisms improved the gut microbiota of common carp. Similarly, Ballantyne, et al.109 reported that heat-killed microbial supplements enhanced the intestinal health of white shrimp (Penaeus vannamei). Likewise, Mohammady, et al.110 and Shija, et al.111 found that a diet enriched with Bacillus spp. improved the intestinal microbiota of Nile tilapia. These studies, like ours, commonly report an increase in beneficial bacteria and a reduction in harmful taxa such as Proteobacteria, contributing to better physiological outcomes. The mechanism by which CP exerts these effects is likely due to its composition. CP contains bioactive compounds such as polyphenols, chlorogenic acids, pectic polysaccharides, and xylooligosaccharides, which act as fermentable substrates that selectively promote the growth of beneficial bacteria23,93. These prebiotic-like effects may suppress opportunistic pathogens and support microbial species that contribute to gut health and immune regulation. The reduction of Proteobacteria in the CP10 group, often linked to inflammation and gut barrier dysfunction, suggests that CP may also help maintain intestinal integrity.
The biofloc system used in this study may have played a synergistic role in enhancing the effects of CP. Biofloc technology is widely recognized for its ability to improve water quality, recycle nutrients, and promote the growth of beneficial microbes through heterotrophic bacterial activity112. The addition of carbohydrates like CP to biofloc systems enhances microbial protein production, supporting nitrogen removal and generating additional feed biomass33,113. This dual benefit of CP as both a dietary supplement and a floc-enhancing carbon source likely explains the reduced feed conversion ratio (FCR) observed in our experiment. Furthermore, the improved immune responses may stem from enhanced microbial diversity in the floc environment and reduced pathogen load, as biofloc systems are known to stimulate fish immunity and suppress opportunistic pathogens. Taken together, these findings highlight the potential of CP as a sustainable and functional feed additive in tilapia aquaculture under biofloc systems. The study demonstrates that CP can improve multiple performance and health parameters, particularly at optimized inclusion levels, while supporting the principles of waste valorization and circular economy. While the results of this study are promising, several limitations should be acknowledged. The experiment was conducted under controlled laboratory conditions, which may not fully reflect outcomes in commercial aquaculture settings. Additionally, although we observed beneficial changes in gene expression and gut microbial communities, the long-term physiological impacts and specific mechanisms of action remain unclear. Importantly, the study did not assess the effect of coffee parchment (CP) on disease resistance through pathogen challenge trials, which are essential for confirming functional immune benefits. Future research should investigate dose–response relationships beyond 40 g kg−1 to determine CP’s upper tolerance limit, conduct extended feeding trials under practical farming conditions, and include pathogen challenge tests to validate immunoprotective effects. Moreover, metabolomic and functional microbiome analyses are recommended to gain deeper insights into the interactions between CP supplementation, host physiology, and microbial ecology.
Conclusion
This study demonstrates that dietary inclusion of coffee parchment (CP) in the feed of Nile tilapia reared under a biofloc system significantly enhances growth performance, innate immune responses, and the expression of growth-, immune-, and antioxidant-related genes. Among the tested levels, the 5 g kg−1 CP supplementation yielded the most pronounced biological effects, particularly in gene expression. Polynomial regression analysis, based on growth performance data only, estimated an optimal CP inclusion range of 15.06 to 19.86 g kg−1 for maximizing growth. These findings highlight coffee parchment as a promising functional feed additive with sustainable benefits for tilapia aquaculture. Nonetheless, further research under field-scale conditions and across longer production cycles is recommended to validate its efficacy and elucidate the underlying physiological mechanisms.
Data availability
The raw sequencing data generated during this study have been deposited in the NCBI Sequence Read Archive (SRA) under the accession number PRJNA1154970.
References
Oftebro, T. L. et al. What’s next for Norwegian salmon farming? Stakeholder perceptions on what influences industry development. Aquaculture 599, 742130. https://doi.org/10.1016/j.aquaculture.2025.742130 (2025).
FAO. (ed Food and Agriculture Organization of the United Nations). FAO, Rome, Italy, (2024).
Yang, Z., Fu, G., Lee, M., Yeo, S. & Yue, G. H. Genes for editing to improve economic traits in aquaculture fish species. Aquaculture Fisheries. 10, 1–18. https://doi.org/10.1016/j.aaf.2024.05.005 (2025).
Clyde, C. W., Tan, J. P., Yeap, S. K. & Yong, C. Y. Current updates on viral infections affecting tilapia. Aquaculture Fisheries. https://doi.org/10.1016/j.aaf.2024.05.002 (2024).
Hoseinifar, S. H. et al. Reducing the use of antibiotics in European aquaculture with vaccines, functional feed additives and optimization of the gut microbiota. J. Mar. Sci. Eng. 12, 204 (2024).
Kaur, K., Singh, S. & Kaur, R. Impact of antibiotic usage in food-producing animals on food safety and possible antibiotic alternatives. Microbe 4, 100097. https://doi.org/10.1016/j.microb.2024.100097 (2024).
Salma, U. et al. Occurrence, risks, and mitigation of antibiotic pollution in Bangladeshi aquaculture systems. Environ. Chem. Ecotoxicol. 7, 351–363. https://doi.org/10.1016/j.enceco.2025.01.007 (2025).
Chakroborty, K., Lima, R. A., Hossain, F., Rafiquzzaman, S. M. & M. & Biobased functional feed additives in Asian aquaculture: trends, impacts, and future directions. Anim. Feed Sci. Technol. 320, 116222. https://doi.org/10.1016/j.anifeedsci.2025.116222 (2025).
Kuebutornye, F. K. A., Roy, K., Folorunso, E. A. & Mraz, J. Plant-based feed additives in aquaculture. Reviews Aquaculture. 16, 309–336. https://doi.org/10.1111/raq.12840 (2024).
Sabaghi, M. & Seyedalmoosavi, M. M. Applications of sustainable proteins in food and feed, and perspectives on health and circular bioeconomy. Int. J. Biol. Macromol. 309, 143193. https://doi.org/10.1016/j.ijbiomac.2025.143193 (2025).
Quagliardi, M. et al. Use of grape by-products in aquaculture: new frontiers for a circular economy application. Heliyon 10, e27443. https://doi.org/10.1016/j.heliyon.2024.e27443 (2024).
Bhattacharya, S. et al. Dietary pineapple Peel ethanolic extract alleviates crowding stress in Labeo rohita fingerlings with enhancement of growth performance and IGF-1 expression, physio-biochemical profile, antioxidant status and immunological responses. Anim. Feed Sci. Technol. 320, 116200. https://doi.org/10.1016/j.anifeedsci.2024.116200 (2025).
Velázquez-Carriles, C. A., Angulo, C., Macías-Rodríguez, M. E. & Reyes-Becerril, M. Phytochemical properties of Cyrtocarpa Edulis Peel exert antimicrobial activity and enhance Immunobiological parameters in Almaco Jack Seriola rivoliana cells. Fish Shellfish Immunol. 156, 110044. https://doi.org/10.1016/j.fsi.2024.110044 (2025).
Chakroborty, K., Lima, R. A., Hossain, F., Rafiquzzaman, S. M. & M. & Biobased functional feed additives in Asian aquaculture: trends, impacts, and future directions. Anim. Feed Sci. Technol. 116222 https://doi.org/10.1016/j.anifeedsci.2025.116222 (2025).
Wannavijit, S. et al. Evaluation of Longan (Dimocarpus Longan) Peel powder as fruit by-product additive in nile tilapia (Oreochromis niloticus) feed: effects on growth, immunity, and immune-antioxidant gene expressions. Heliyon e41609 https://doi.org/10.1016/j.heliyon.2024.e41609 (2025).
Maiyah, N. et al. Recovering bioactive compounds and antioxidant capacity of medium roasted spent coffee grounds through varied hydrothermal brewing cycles. J. Agric. Food Res. 20, 101789. https://doi.org/10.1016/j.jafr.2025.101789 (2025).
Bojórquez-Quintal, E. et al. Bioactive compounds and valorization of coffee By-Products from the origin: A circular economy model from local practices in Zongolica, Mexico. Plants 13, 2741 (2024).
Chomphoosee, T. et al. A novel beverage of coffee Cherry (cascara) water Kefir rich in antioxidants, bioactive compounds, and exhibiting promising antibacterial and sensory qualities. LWT 219, 117539. https://doi.org/10.1016/j.lwt.2025.117539 (2025).
dos Muchangos, L. S., Mejia, C., Gupta, R., Sadreghazi, S. & Kajikawa, Y. A systematic review of life cycle assessment and environmental footprint for the global coffee value chain. Environ. Impact Assess. Rev. 111, 107740. https://doi.org/10.1016/j.eiar.2024.107740 (2025).
Temaj, J. B. a. K. (ed World Bank) (2025).
Thamaraiselvi, C. et al. Effective reduction of carbon-containing pollutants in coffee Cherry pulping wastewater using natural polysaccharide from Tamarindus indica L. seeds. Environ. Res. 259, 119527. https://doi.org/10.1016/j.envres.2024.119527 (2024).
Aswathi, K. N. & Murthy, P. S. Pulped natural/honey coffee process: an innovative approach. Food Humanity. 2, 100287. https://doi.org/10.1016/j.foohum.2024.100287 (2024).
Barcellos Silva, I. G. C. et al. Method optimization for the extraction of chlorogenic acids from coffee parchment: an ecofriendly alternative. Food Chem. 458, 139842. https://doi.org/10.1016/j.foodchem.2024.139842 (2024).
Bondam, A. F., da Silveira, D. D., dos Santos, J. P. & Hoffmann, J. F. Phenolic compounds from coffee by-products: extraction and application in the food and pharmaceutical industries. Trends Food Sci. Technol. 123, 172–186 (2022).
Hu, S. et al. Valorization of coffee pulp as bioactive food ingredient by sustainable extraction methodologies. Curr. Res. Food Sci. 6, 100475 (2023).
Tsigkou, K. et al. Coffee processing waste: unlocking opportunities for sustainable development. Renew. Sustain. Energy Rev. 210, 115263. https://doi.org/10.1016/j.rser.2024.115263 (2025).
Gondim, F. F., Rodrigues, J. G. P., Aguiar, V. O., de Fátima Vieira Marques, M. & Monteiro, S. N. Biocomposites of cellulose isolated from coffee processing By-Products and incorporation in Poly(Butylene Adipate-Co-Terephthalate) (PBAT) matrix: an overview. Polymers 16, 314 (2024).
Makiso, M. U., Tola, Y. B., Ogah, O. & Endale, F. L. Bioactive compounds in coffee and their role in Lowering the risk of major public health consequences: A review. Food Sci. Nutr. 12, 734–764. https://doi.org/10.1002/fsn3.3848 (2024).
Khat-udomkiri, N. & Myo, H. in In Coffee in Health and Disease Prevention (Second Edition). 867–880 (eds Victor, R., Preedy, Vinood, B. & Patel) (Academic, 2025).
Dari, D. N. et al. Spent coffee grounds: insights and future prospects for bioenergy and circular economy applications. Green. Technol. Sustain. 100213 https://doi.org/10.1016/j.grets.2025.100213 (2025).
Chéron-Bessou, C. et al. Unravelling life cycle impacts of coffee: why do results differ so much among studies? Sustainable Prod. Consum. 47, 251–266. https://doi.org/10.1016/j.spc.2024.04.005 (2024).
De Felice, F., Rehman, M., Petrillo, A., Ortiz Barrios, M. A. & Baffo, I. Integrating IoT and circular economy in textile supply chains: A closed-loop model for sustainable production using recycled PET and spent coffee grounds. J. Clean. Prod. 501, 145277. https://doi.org/10.1016/j.jclepro.2025.145277 (2025).
Ghosh, A. K., Hasanuzzaman, A. F. M., Sarower, M. G., Islam, M. R. & Huq, K. A. Unveiling the Biofloc culture potential: Harnessing immune functions for resilience of shrimp and resistance against AHPND -causing Vibrio parahaemolyticus infection. Fish Shellfish Immunol. 109710. https://doi.org/10.1016/j.fsi.2024.109710 (2024).
Yang, H. et al. Advancements in freshwater aquaculture wastewater management: A comprehensive review. Aquaculture 594, 741346. https://doi.org/10.1016/j.aquaculture.2024.741346 (2025).
Wan Mahari, W. A. et al. Emerging paradigms in sustainable shellfish aquaculture: microalgae and Biofloc technologies for wastewater treatment. Aquaculture 587, 740835. https://doi.org/10.1016/j.aquaculture.2024.740835 (2024).
Yu, Y. B. et al. The use, application and efficacy of Biofloc technology (BFT) in shrimp aquaculture industry: A review. Environ. Technol. Innov. 33, 103345. https://doi.org/10.1016/j.eti.2023.103345 (2024).
Helal, A. M. et al. Biofloc supplementation improves growth performances, nutrient utilization, and histological status of nile tilapia (Oreochromis niloticus) while enhancing zooplankton diversity, community, and abundance. Aquaculture 585, 740711. https://doi.org/10.1016/j.aquaculture.2024.740711 (2024).
Núñez, A. C. et al. The use of coffee pulp as a potential alternative supplement in ruminant diets. J. Agric. Sci. Technol. A. 5, 214–217 (2015).
AOAC. Association of Official Analytical Chemists, Arlington, (1998).
Avnimelech, Y. Biofloc Technology: a Practical Guide Book (World Aquaculture Society, 2015).
Tarigan, N. B., Amal, M., Ekasari, J., Keesman, K. J. & Verdegem, M. Nitrogen, phosphorus, and carbon dynamics in Biofloc system of nile tilapia fed with high non-starch polysaccharides diet. Aquaculture 596, 741714. https://doi.org/10.1016/j.aquaculture.2024.741714 (2025).
Federation, W. E. & Association, A. P. H. Standard methods for the examination of water and wastewater. American Public Health Association (APHA): Washington, DC, USA (2005).
Sherif, E. M. et al. Growth performance, antioxidative status, and immune response of nile tilapia (Oreochromis niloticus) fed dietary fermented Spirulina platensis. Aquaculture Rep. 39, 102324. https://doi.org/10.1016/j.aqrep.2024.102324 (2024).
Parry, R. M. Jr., Chandan, R. C. & Shahani, K. M. A rapid and sensitive assay of muramidase. Proc. Soc. Exp. Biol. Med. 119, 384–386 (1965).
Lubis, A. R. et al. Effects of passion fruit Peel (Passiflora edulis) pectin and red yeast (Sporodiobolus pararoseus) cells on growth, immunity, intestinal morphology, gene expression, and gut microbiota in nile tilapia (Oreochromis niloticus). Sci. Rep. 14 https://doi.org/10.1038/s41598-024-73194-1 (2024).
Quade, M. J. & Roth, J. A. A rapid, direct assay to measure degranulation of bovine neutrophil primary granules. Vet. Immunol. Immunopathol. 58, 239–248 (1997).
Yanno, T. Techniques in Fish Immunology (eds J.Set al.) 131–141 (SOS, 1992). Stolen.
Lubis, A. R. et al. Impact of Mango Peel pectin and Bacillus iranensis on growth, immunity, and gene expression in nile tilapia (Oreochromis niloticus). Aquaculture Rep. 41, 102661. https://doi.org/10.1016/j.aqrep.2025.102661 (2025).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using Real-Time quantitative PCR and the 2 – ∆∆CT method. Methods 25, 402–408. https://doi.org/10.1006/meth.2001.1262 (2001).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Callahan, B. J. et al. DADA2: High-resolution sample inference from illumina amplicon data. Nat. Methods. 13, 581–583. https://doi.org/10.1038/nmeth.3869 (2016).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41 (596), D590. https://doi.org/10.1093/nar/gks1219 (2013).
Chen, H. & Boutros, P. C. VennDiagram: a package for the generation of highly-customizable Venn and Euler diagrams in R. BMC Bioinform. 12 https://doi.org/10.1186/1471-2105-12-35 (2011).
Parks, D. H., Tyson, G. W., Hugenholtz, P. & Beiko, R. G. STAMP: statistical analysis of taxonomic and functional profiles. Bioinformatics 30, 3123–3124 (2014).
SAS Institute Inc. SAS Campus Drive, Cary, NC USA 27513 – 2414. (2003).
Yossa, R. & Verdegem, M. Misuse of multiple comparison tests and underuse of contrast procedures in aquaculture publications. Aquaculture 437, 344–350 (2015).
Tao, L. T. et al. The application and potential of postbiotics as sustainable feed additives in aquaculture. Aquaculture 592, 741237. https://doi.org/10.1016/j.aquaculture.2024.741237 (2024).
Samat, N., Goh, K. H. & See, K. F. Review of the application of cost–benefit analysis to the development of production systems in aquaculture. Aquaculture 587, 740816. https://doi.org/10.1016/j.aquaculture.2024.740816 (2024).
Suyamud, B. et al. Antimicrobial resistance in aquaculture: occurrence and strategies in Southeast Asia. Sci. Total Environ. 907, 167942. https://doi.org/10.1016/j.scitotenv.2023.167942 (2024).
Elgendy, M. Y. et al. Alternative therapies recently applied in controlling farmed fish diseases: mechanisms, challenges, and prospects. Aquacult. Int. https://doi.org/10.1007/s10499-024-01603-3 (2024).
Xia, J., Ge, C. & Yao, H. Antimicrobial peptides: an alternative to antibiotic for mitigating the risks of antibiotic resistance in aquaculture. Environ. Res. 251, 118619. https://doi.org/10.1016/j.envres.2024.118619 (2024).
Ivanova, S. et al. Medicinal plants: A source of phytobiotics for the feed additives. J. Agric. Food Res. 16, 101172. https://doi.org/10.1016/j.jafr.2024.101172 (2024).
Guebebia, S. et al. Effects of Okra (Abelmoschus esculentus L.) leaves, fruits and seeds extracts on European sea bass (Dicentrarchus labrax) leukocytes, and their cytotoxic, bactericidal and antioxidant properties. Fish Shellfish Immunol. 138, 108799. https://doi.org/10.1016/j.fsi.2023.108799 (2023).
Oh, H. Y. et al. Effects of by-products from producing Yacon (Smallanthus sonchifolius) juice as feed additive on growth performance, digestive enzyme activity, antioxidant status, related gene expression, and disease resistance against Streptococcus iniae in juvenile black rockfish (Sebastes schlegelii). Aquaculture 569, 739383 (2023).
Hu, M. et al. Use of fermented tea residues as a feed additive and effects on growth performance, body composition, intestinal enzyme activities, and inflammatory biomarkers in juvenile largemouth bass (Micropterus salmoides). Aquaculture Rep. 31, 101671. https://doi.org/10.1016/j.aqrep.2023.101671 (2023).
Yousefi, M. et al. Effects of dietary pomegranate Peel supplementation on growth performance and biochemical responses of common carp, Cyprinus Carpio, to chronic crowding stress. Aquaculture Rep. 30, 101532 (2023).
Xuan, C. L. et al. Modulation of growth, immunity, and immune-antioxidant gene expression in nile tilapia, Oreochromis niloticus, culture under Biofloc system by Dragon fruit, Hylocereus Undatus, Peel powder. J. World Aquac. Soc.. https://doi.org/10.1111/jwas.13088 (2024).
Le Xuan, C. et al. Influences of Makiang (Syzygium nervosum) seed powder on growth performance, immunological response, antioxidant and immune related gene expression in juvenile nile tilapia (Oreochromis niloticus). Aquaculture 588, 740943. https://doi.org/10.1016/j.aquaculture.2024.740943 (2024).
Deng, H. et al. Elucidating the effects of Cumin (Cuminum cyminum) fruit and stem as feed additives on growth, antioxidant capacity, liver and intestinal health, and gut Microbiome of nile tilapia (Oreochromis niloticus). Aquaculture Rep. 31, 101687. https://doi.org/10.1016/j.aqrep.2023.101687 (2023).
See, M. S., Musa, N., Liew, H. J., Harun, N. O. & Rahmah, S. Sweet orange Peel waste as a feed additive in growth promoting and protective effect against Aeromonas hydrophila of juvenile Bagrid catfish Mystus Nemurus. J. Environ. Manage. 351, 119677. https://doi.org/10.1016/j.jenvman.2023.119677 (2024).
Benitez, V. et al. Coffee parchment as a new dietary fiber ingredient: functional and physiological characterization. Food Res. Int. 122, 105–113 (2019).
Machado, M. et al. Bioactive potential and chemical composition of coffee By-Products: from pulp to Silverskin. Foods 12, 103390foods12122354 (2023).
Zhou, H. et al. Effects of dietary pectin on the growth performance, intestinal barrier, and antioxidant status of juvenile rainbow trout (Oncorhynchus mykiss). Aquaculture Rep. 36, 102055. https://doi.org/10.1016/j.aqrep.2024.102055 (2024).
Waqas, M., Salman, M. & Sharif, M. Application of polyphenolic compounds in animal nutrition and their promising effects. J. Anim. Feed Sci. 32, 233–256 (2023).
Santos, É. M. et al. Coffee by-products in topical formulations: A review. Trends Food Sci. Technol. 111, 280–291. https://doi.org/10.1016/j.tifs.2021.02.064 (2021).
Muntaha, S. T. et al. Polyphenol-protein particles: A nutraceutical breakthrough in nutrition and food science. J. Agric. Food Res. 19, 101641. https://doi.org/10.1016/j.jafr.2025.101641 (2025).
Manzoor, Z. et al. Polyphenols as antiviral agents: assessing their potential usage and benefits in aquaculture. Aquacult. Int. 33 https://doi.org/10.1007/s10499-024-01778-9 (2025).
Zhang, R. et al. Ameliorative effect of phenolic compound-pterostilbene on corticosterone-induced hepatic lipid metabolic disorder in broilers. J. Nutr. Biochem. 137, 109822. https://doi.org/10.1016/j.jnutbio.2024.109822 (2025).
Machado, M., Ferreira, H., Oliveira, M. B. P. P. & Alves, R. C. Coffee by-products: an underexplored source of prebiotic ingredients. Crit. Rev. Food Sci. Nutr. 64, 7181–7200. https://doi.org/10.1080/10408398.2023.2181761 (2024).
Wu, S. et al. A new dietary Fiber can enhance satiety and reduce postprandial blood glucose in healthy adults: A randomized Cross-Over trial. Nutrients 15 https://doi.org/10.3390/nu15214569 (2023).
Azemi, A. K. et al. Obesity-driven hunger: from pathophysiology to intervention. Obes. Med. 54, 100588. https://doi.org/10.1016/j.obmed.2025.100588 (2025).
Miyamoto, J. et al. Nutritional signaling via free fatty acid receptors. Int. J. Mol. Sci. 17, 450 (2016).
Esteban, M. Á. A review of soluble factors and receptors involved in fish skin immunity: the tip of the iceberg. Fish Shellfish Immunol. 145, 109311. https://doi.org/10.1016/j.fsi.2023.109311 (2024).
Dong, M. et al. Proteomic characterization of the interactions between fish serum proteins and waterborne bacteria reveals the suppression of anti-oxidative defense as a serum-mediated antimicrobial mechanism. Fish Shellfish Immunol. 62, 96–106. https://doi.org/10.1016/j.fsi.2017.01.013 (2017).
Salem, M. E., Almisherfi, H. M., El-Sayed, A. F. M., Makled, S. O. & Abdel-Ghany, H. M. Modulatory effects of dietary prickly Pear (Opuntia ficus-indica) Peel on high salinity tolerance, growth rate, immunity and antioxidant capacity of nile tilapia (Oreochromis niloticus). Fish Physiol. Biochem. 1–14 (2024).
Bahi, A., Bhaskaracharya, R., Esteban, M. A. & Guardiola, F. A. Dietary administration impact of Olive pulp on growth performance, metabolic profile, immune status, and antioxidant potential of Gilthead seabream (Sparus aurata L). Front. Sustain. Food Syst. 8 https://doi.org/10.3389/fsufs.2024.1395436 (2024).
Phinyo, M., Sangarun, P., Wangkahart, E. & Sujipuri, K. Effects of banana flower powder (Musa sp.) supplementation on growth performance, whole body composition, antioxidant and immune responses, gene expression and liver histology in nile tilapia (Oreochromis niloticus). Anim. Feed Sci. Technol. 308, 115882 (2024).
Abdel-Tawwab, M., Harikrishnan, R., Devi, G., Bhat, E. A. & Paray, B. A. Stimulatory effects of seaweed Laminaria digitata polysaccharides additives on growth, immune-antioxidant potency and related genes induction in Rohu carp (Labeo rohita) during Flavobacterium columnare infection. Aquaculture 579, 740253. https://doi.org/10.1016/j.aquaculture.2023.740253 (2024).
Yilmaz, S. et al. Effects of a phytogenic diet on growth, haemato-immunological parameters, expression of immune- and stress-related genes, and resistance of Oncorhynchus mykiss to Lactococcus garvieae infection. Aquaculture 587, 740845. https://doi.org/10.1016/j.aquaculture.2024.740845 (2024).
Ding, S., Jiang, H. & Fang, J. Regulation of immune function by polyphenols. J. Immunol. Res. 2018, 1264074–1264074. https://doi.org/10.1155/2018/1264074 (2018).
Kolanchi, P. et al. Phytohormonal signaling network and immune priming pertinence in plants to defend against insect herbivory. Plant. Stress. 16, 100850. https://doi.org/10.1016/j.stress.2025.100850 (2025).
Machado, M., Ferreira, H., Oliveira, M. & Alves, R. C. Coffee by-products: an underexplored source of prebiotic ingredients. Crit. Rev. Food Sci. Nutr. 64, 7181–7200. https://doi.org/10.1080/10408398.2023.2181761 (2024).
Xiong, H., Han, X., Cai, L. & Zheng, H. Natural polysaccharides exert anti-tumor effects as dendritic cell immune enhancers. Front. Oncol. 13 https://doi.org/10.3389/fonc.2023.1274048 (2023).
Chen, X. et al. Effect of Bacillus velezensis T23 solid-state fermentation product on growth, gut and liver health, and gut microbiota of common carp (Cyprinus carpio). Aquaculture 596, 741733. https://doi.org/10.1016/j.aquaculture.2024.741733 (2025).
Zhou, G. et al. Exploring the gut microbiota and metabolome of Lateolabrax Japonicus: A multi-omics approach. Comp. Biochem. Physiol. D: Genomics Proteomics. 54, 101408. https://doi.org/10.1016/j.cbd.2024.101408 (2025).
Tolas, I. et al. A fishy gut feeling – current knowledge on gut microbiota in teleosts. Front. Mar. Sci. 11–2024 https://doi.org/10.3389/fmars.2024.1495373 (2025).
Qu, P. et al. Yellow mealworm (Tenebrio molitor) meal replacing dietary fishmeal alters the intestinal microbiota, anti-oxidation and immunity of large yellow croaker (Larimichthys crocea). Fish Shellfish Immunol. 161, 110272. https://doi.org/10.1016/j.fsi.2025.110272 (2025).
Hu, R. G. et al. Improvement of non-specific immunity, intestinal health and microbiota of crucian carp (Carassius auratus) juvenile with dietary supplementation of Bacillus coagulans BC1. Aquaculture 580, 740327, (2024). https://doi.org/10.1016/j.aquaculture.2023.740327
Zhu, L. et al. Comparative effects of different dietary pectin types on growth performance and intestinal health in Pearl Gentian grouper (Epinephelus fuscoguttatus♀ × E. lanceolatus♂). Aquaculture Rep. 40, 102547. https://doi.org/10.1016/j.aqrep.2024.102547 (2025).
Kanika, N. H. et al. Fish gut Microbiome and its application in aquaculture and biological conservation. Front. Microbiol. 15, 1521048. https://doi.org/10.3389/fmicb.2024.1521048 (2024).
Amthungphong, P. et al. Comprehensive profiling of gut bacterial communities in hybrid red tilapia with and without streptococcosis. Aquaculture Rep. 40, 102556. https://doi.org/10.1016/j.aqrep.2024.102556 (2025).
Zhang, Z. et al. Effects of supplemental feeding of feeds with different protein levels on the metabolism of the gut microbiota of Bighead carp (Hypophthalmichthys nobilis). Aquaculture Rep. 40, 102576. https://doi.org/10.1016/j.aqrep.2024.102576 (2025).
Hao, Q. et al. Dietary Solid-state-fermentation product of Bacillus velezensis T23 alleviate hepatic steatosis, oxidative stress, gut barrier damage, and microbiota dysbiosis in juvenile genetically improved farmed tilapia (GIFT, Oreochromis niloticus). Aquaculture Rep. 40, 102523. https://doi.org/10.1016/j.aqrep.2024.102523 (2025).
Gomes, N. C. M. et al. Shifts in turbot (Scophthalmus maximus) larvae bacterial communities from neutrality to non-neutrality: A window of opportunity for Microbiome interventions. Aquaculture 599, 742074. https://doi.org/10.1016/j.aquaculture.2024.742074 (2025).
Lenin, K. L. D., Maharajan, T., Ceasar, S. A. & Antony, S. P. Transcriptomic and bioinformatic analysis of histone H2A-genes in Lates calcarifer and Oreochromis niloticus upon Vibrio vulnificus challenge. Comp. Immunol. Rep. 7, 200180. https://doi.org/10.1016/j.cirep.2024.200180 (2024).
Qi, X. et al. Vitamin B(12) produced by Cetobacterium somerae improves host resistance against pathogen infection through strengthening the interactions within gut microbiota. Microbiome 11, 135. https://doi.org/10.1186/s40168-023-01574-2 (2023).
Luan, Y. et al. The fish microbiota: research progress and potential applications. Engineering 29, 137–146. https://doi.org/10.1016/j.eng.2022.12.011 (2023).
Ballantyne, R. et al. Dietary administration of a postbiotic, heat-killed Pediococcus pentosaceus PP4012 enhances growth performance, immune response and modulates intestinal microbiota of white shrimp, Penaeus vannamei. Fish Shellfish Immunol. 139, 108882. https://doi.org/10.1016/j.fsi.2023.108882 (2023).
Mohammady, E. Y., Khafagy, S. S., Shawer, E. E., Soaudy, M. R. & Hassaan, M. S. Effects of selenium-enriched Bacillus pumilus on the growth performance, blood biochemical parameters, innate immune response, and oxidative activity nile tilapia (Oreochromis niloticus) fed fishmeal-free diets. Anim. Feed Sci. Technol. 320, 116202. https://doi.org/10.1016/j.anifeedsci.2024.116202 (2025).
Shija, V. M. et al. Effects of dietary supplementation with fish-derived Bacillus amyloliquefaciens AV5 on growth status, immune response, microbiota, and intestinal health of nile tilapia (Oreochromis niloticus). Aquaculture Rep. 41, 102658. https://doi.org/10.1016/j.aqrep.2025.102658 (2025).
Bhadra, S., Krishnani, K. K., Sharma, A., Sahoo, U. & Majeedkutty, B. R. A. Curcuma longa and Allium sativum as health promoters in genetically improved farmed Tilapia (GIFT) - A green drug approach in hi-tech aquaculture using Biofloc. Aquaculture 582, 740516. https://doi.org/10.1016/j.aquaculture.2023.740516 (2024).
Khanjani, M. H., Sharifinia, M. & Hajirezaee, S. Biofloc: A sustainable alternative for improving the production of farmed cyprinid species. Aquaculture Rep. 33, 101748. https://doi.org/10.1016/j.aqrep.2023.101748 (2023).
Acknowledgements
This research work was partially support by Chiang Mai University.
Author information
Authors and Affiliations
Contributions
CRediT authorship contribution statementHien Van Doan: Conceptualization, Investigation, Methodology, Conceptual Writing – Original draft, Formal analysis, Data curation, Software analysis, Writing, Review, Editing, Formal analysis, and Data curation. Toungporn Uttarotai: Formal Analysis. Supreya Wannavijit: Investigation and Methodology. Khambou Tayyamath: Investigation and Methodology. Punika Ninyamasiri: Investigation and Methodology. Tran Thi Diem Quynh: Formal analysis. Orranee Srinual: Formal analysis. Thanongsak Chaiyaso: Methodology. Pimporn Khamtavee: Data curation. Wanaporn Tapingkae: Conceptualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All animal experiments comply with AAALAC guide-lines approval by Chiang Mai University Committee (Approval No.: AG03007/2567). The study is reported in accordance with ARRIVE guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Van Doan, H., Uttarotai, T., Wannavijit, S. et al. Impacts of coffee parchment on growth, immunity, and gut microbiota of Nile tilapia (Oreochromis niloticus) in biofloc system. Sci Rep 15, 25057 (2025). https://doi.org/10.1038/s41598-025-03109-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03109-1