Abstract
Insulin-like growth factor-binding protein 5 (IGFBP5) is associated with nonalcoholic fatty liver disease (NAFLD). IGFBP5 has a higher binding affinity to IGF1 and modulates its effects in serum. However, the effect of exercise on its expression remains unclear. Hence, this study investigated whether exercise regulated hepatic and circulating IGFBP5 levels to ameliorate NAFLD. The NAFLD mice were regularly trained for 12 weeks on a treadmill at a 0% grade. Serum levels of IGFBP5, total IGF1, and free IGF1 were detected using proteome analyses and ELISA. Critical cytokines, such as IRS1, Akt, and AMPKα, and their phosphorylation levels mediated by IGFBP5/IGF1 were confirmed via Western blotting. The results indicated that exercise remarkably alleviated HFD-induced NAFLD and insulin resistance. Although exercise lowered serum IGFBP5 levels and the IGFBP5/total IGF1 ratio in NAFLD mice, it had no effect on the hepatic expression of IGFBP5, IGF1, and IGF1R. Moreover, exercise increased serum levels of free IGF1 in NAFLD mice, which, when bound to IGF1R, accelerated the phosphorylation levels of hepatic IRS1, Akt, and AMPKα to ameliorate NAFLD. The present study confirmed that exercise activated the effects of IGF1 by reducing the serum IGFBP5/IGF1 ratio, consequently triggering the IRS1/Akt/AMPK pathway to ameliorate HFD-induced NAFLD.
Similar content being viewed by others
Introduction
Non-alcoholic fatty liver disease (NAFLD), the most common chronic liver disease, is an escalating public health problem1, with a prevalence of 25% globally2 and approximately 29.2% in China over the last decade3. The worldwide prevalence of NAFLD has continued to grow together with the global epidemic of obesity4. Symptoms associated with NAFLD can range from hepatic steatosis to fibrosis/cirrhosis, with some individuals even developing hepatocellular cancer. To date, no gold standard therapy exists for NAFLD, but exercise has been mostly recommended for the preventing and treating health in the United States, Europe, and China5,6,7. However, the exact mechanisms responsible for the protective and therapeutic effects of exercise against NAFLD have yet to be fully understood.
Insulin-like growth factor-binding proteins (IGFBPs) are a member of the insulin family of proteins. Studies have shown that IGFBP1–6 exhibit high affinity to insulin-like growth factors (IGFs)8 and that IGFBP4–6 play important roles as regulators of IGF action9. In most circumstances, IGFBPs have a higher affinity for binding with IGFs than with IGF receptors, with 99% of IGFBPs binding with IGFs in the blood9. IGFBP5, one of the conserved IGFBPs, it can translocate into the nucleus due to the existence of a nuclear localization sequence10. IGFBP5 binds to IGFs and modulates its effects on cell proliferation, differentiation, migration, invasion, senescence, apoptosis, and metabolism11,12,13,14. The binding affinity between IGFBP5 and IGF1 is higher than that between IGF1 and the IGF1 receptor (IGF1R)9. IGFBP5 has been shown to both inhibit and potentiate IGF1 action. Moreover, serum IGFBP5 levels have been correlated with NAFLD. In particular, NAFLD patients showed higher serum IGFBP5 levels than did healthy individuals, whereas patients with moderate-to-severe fibrosis displayed significantly higher IGFBP5/total IGF1 levels than did patients with no or mild fibrosis15. However, the mechanisms underlying these findings have remained unclear.
Exercise can regulate the expression of adipokines and various cytokines in the circulation and in several tissues16,17. Indeed, both acute and chronic exercises have been found to increase the level of IGF1, a member of the IGF family, in the achilles tendon and skeletal muscle18,19; however, other studies have shown that circulating total or free IGF1 either increased, decreased or remained unchanged, demonstrating controversial results20,21,22. Moreover, exercise showed similar effects on hepatic and circulating total IGF1 expression23,24, whereas both acute and chronic exercise altered circulating IGFBPs. Evidence also shows that IGFBP1–3 and IGFBP-6 all increased after exercise or training22,25,26. In fact, Nindl et al.. indicated that exercise increased IGFBP3 and IGFBP4 levels in muscle interstitial fluid20. Another study showed that plasma concentrations of IGF1 and IGFBP3 were significantly decreased after completing an exercise program, with a significant reduction in body weight and body fat in the diet/training group27. Exercise also upregulated the expression of IGFBP7 in muscle satellite cells28 and decreased the expression of IGFBP5 in muscle interstitial fluid20. Nevertheless, the effects of acute and chronic exercise on circulating and hepatic IGFBP5 remains unclear. The current study therefore aimed to investigate the ameliorating effects of regular exercise on NAFLD by determine its impact on circulating and hepatic IGFBP5 levels, as well as the IGFBP5/IGF1 binding ratio in NAFLD mice.
Results
Exercise ameliorates HFD-induced NAFLD characteristics
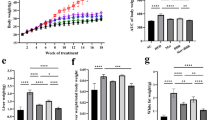
Hepatic steatosis, inflammation, and fibrosis are considered as the mainly characteristics of NAFLD29,30. HFD-induced NAFLD mice showed increased liver weight, which reduced significantly after regular exercise for 12 weeks (Fig. 1A). Moreover, exercise significantly reduced histological parameters reflecting hepatic steatosis and inflammation, such as the steatosis score, lobular inflammation score, ballooning score, and total NAS (Fig. 1B, C). Hepatic lipid accumulation, total cholesterol levels, triglyceride levels, and collagen accumulation were significantly lower in the HFD + EXE group than in the HFD group (Fig. 1D–G). Exercise downregulated serum alanine aminotransferase and aspartate transaminase levels (Fig. 1H). These results indicated that 12 weeks of regular exercise remarkably alleviated HFD-induced NAFLD.
Exercise reduced HFD-induced liver weight, hepatic steatosis, inflammation, and fibrosis. The liver weight (A) decreased after 12 weeks of treadmill training. Hepatic histological analysis of steatosis, inflammation, ballooning, and NAFLD activity score (NAS) (B). Liver sections stained with H&E (×200) (C). Hepatic TC levels and TG levels (D). Hepatic lipid accumulation as determined by Oil Red O staining (×200) (E). Collagen content determined by counting Sirius Red positive areas in six randomly selected fields using Image Pro Plus 6.0 software and hepatic mRNA levels of Collagen I (F). Liver sections stained with Sirius Red (×200) (G). Serum ALT and AST levels (H). Results are presented as the mean ± SEM, n = 6 per group. * P < 0.05, ** P < 0.01, and *** P < 0.001.
Exercise relieves HFD-induced insulin resistance
Fasting blood glucose and insulin levels distinctly increased in the HFD-fed mice but both sharply decreased after exercise training (Fig. 2A, B). The HOMA-IR index indicated that HFD-induced insulin resistance was ameliorated in the HFD + EXE mice (Fig. 2C). Exercise training improved tolerance to the glucose challenge (Fig. 2D). The HFD + EXE mice showed reduced blood glucose excursion during ITT compared to HFD mice (Fig. 2E). These results confirmed that exercise protects against insulin resistance.
Exercise relieves HFD-induced insulin resistance. Fasting blood glucose (A), serum insulin levels (B) and the HOMA-IR index (C). Glucose tolerance test (GTT) (D) and insulin tolerance test (ITT) (E). Results are presented as the mean ± SEM, n = 6 per group. *** P < 0.001 (HFD vs. HFD + EXE).
Exercise decreases serum IGFBP5 levels and the IGFBP5/IGF1 ratio in NAFLD mice
To investigate the mechanism by which exercise ameliorates HFD-induced NAFLD, the related protein profile was analyzed using proteomics. Label-free proteomics was used to detect serum protein profiles in the LFD, HFD, and HFD + EXE groups. The Reactome pathway database contains information on the relationship among molecules in biological pathways and processes, with the presented results being similar to enrichment analysis using Gene Ontology and Kyoto Encyclopedia of Genes and Genomes. Reactome pathway analysis showed that differentially expressed proteins between the three groups were significantly associated with the regulation of IGF transport (Fig. 3A). We then screened for the 10 most significantly differentially expressed proteins using a chord diagram to illustrate the differing protein expression profiles in serum samples. our results showed that the expression of IGFBP5 in the HFD group was obviously higher than that in the LFD and HFD + EXE groups (Fig. 3B). The expression of IGFBP5 in the HFD group was significantly greater than that in the LFD group using heat maps and enzyme-linked immunosorbent assay (Fig. 3C,D). However, exercise downregulated serum IGFBP5 levels in HFD-induced NAFLD mice (Fig. 3C,D). Although serum total IGF1 levels did not significantly differ between the three groups, free IGF1 levels were obviously higher in the HFD + EXE group than in the HFD group (Fig. 3D). Therefore, these results suggest that exercise decreases the serum IGFBP5/IGF1 ratio in HFD-induced NAFLD mice.
Exercise reduced HFD-induced serum IGFBP5/IGF1 ratio. All identified proteins were classified according to the top 20 gene ontology ratios sorted by enrichment degree [− log10 (p-value)] (A). Chord diagram of top 10 differentially expressed proteins among the LFD, HFD and HFD + EXE groups (red represents up-regulation, blue represents down-regulation) (B). Heat map of differentially expressed proteins among the LFD, HFD and HFD + EXE groups (red represents up-regulation, blue represents down-regulation) (C). EILSA results of serum IGFBP5, total IGF1 and free IGF1 levels (D). Results are presented as the mean ± SEM, n = 3 or 6 per group. ns P > 0.05 and ***P < 0.001.
Exercise activated the IRS1/Akt/AMPK pathway by decreasing the serum IGFBP5/IGF1 ratio
We also determined the hepatic expression of IGFBP5, IGF1, and IGF1R in the three groups. Notably, no significant difference in hepatic protein expression levels of IGFBP5, IGF1, and IGF1R were observed between the HFD + EXE and HFD groups (Fig. 4A, B). However, the HFD + EXE group showed greater hepatic phosphorylation levels of IRS1 tyrosine residues, Akt, and AMPKα but lower phosphorylation levels of IRS1 serine residues than did the HFD group (Fig. 4A, B). These data indicated that exercise reduced the serum IGFBP5/total IGF1 ratio to promote the release IGF1, which activated the IRS1/Akt/AMPK signaling pathway.
Exercise activated IRS1/Akt/AMPK pathway due to reduce serum IGFBP5/total IGF1 ratio. (A). Protein levels of IGFBP5, IGF1, IGF1R, IRS1, p-IRS1 (Ser307), p-IRS1 (Tyr612), Akt, p-Akt (Ser473), AMPKα and p-AMPKα (Thr183/172) in mouse livers were assayed by western blot. (B). Protein expression results are presented as the mean ± SEM, n = 6 per group. ns P > 0.05, ** P < 0.01 and ***P < 0.001.
Discussion
NAFLD is a common liver disease that can become quite harmful when left uncontrolled. Reports have shown that IGFBP5 levels in the liver were correlated with NAFLD8. Moreover, many studies have shown that exercise regulates the expression of cytokines in the liver to alleviate NAFLD symptoms, but exercise whether affects the expression of hepatic IGFBP5 has not been reported. The current study found that although NAFLD mice showed decreased hepatic IGFBP5 levels, its expression did not significantly change after 12 weeks of regular exercise. Furthermore, our results showed no obvious change in hepatic IGF1 levels after exercise, which was consistent with the findings of a previous study23. These results suggested that exercise did not ameliorate NAFLD by increasing or decreasing IGFBP5 and IGF1 expression in the liver.
The circulating expression of IGFBP5 has also been associated with the occurrence and development of NAFLD. Indeed, Colak et al.. pointed out that serum IGFBP5 levels in NAFLD patients were higher than those in healthy people15. The same results were shown in HFD-induced NAFLD mice and in the low-fat diet-induced NAFLD mice. However, the same study showed that serum IGFBP5 levels in NAFLD mice decreased significantly after 12 weeks of regular exercise and that the serum total IGF1 levels did not differ between NAFLD mice with and without exercise. It indicated that exercise decreased the IGFBP5/total IGF1 by reduced IGFBP5 expression rather than increased IGF1 expression in serum. Although the previous study have indicated that IGFBP5/total IGF1 is associated with NAFLD, the mechanism remains unclear. How exercise reduced IGFBP5/total IGF1 to alleviate NAFLD? We carried out experimental research on the molecular mechanism to answer this question.
Studies have reported that serum IGF1 levels were correlated with NAFLD and that its levels were decreased in NAFLD individuals31,32. Another study found that low serum IGF1 levels can be a predictor of advanced hepatic fibrosis33. IGFBP5 has been found to have a high affinity with IGF1 and can inhibit and potentiate IGF1 action in the circulation9. If the level of IGFBP5 is lower in the circulation, IGF1 combined with it will decrease, which useful for activating the signaling pathway mediated by IGF1. Therefore, a decline in the serum IGFBP5/IGF1 ratio can increase free IGF1 levels and promote its activation. Some studies indicated that IGFBPs increased or decreased after exercise in circulating, muscle interstitial fluid and muscle satellite cells20,23,24,26. It is reported that exercise downregulated the expression of IGFBP5 in muscle interstitial fluid18. However, the effect on hepatic IGFBPs by exercise has not been reported yet. We discovered that serum IGFBP5 decreased and hepatic IGFBP5 had no significant change. The current study also found that the IGFBP5/total IGF1 ratio was lower and the free IGF1 levels were higher after the exercise intervention. It is reported that long-term exercise induced a significant increase of plasma volume in healthy untrained individuals, but the effect of long-term exercise on plasma volume in trained individuals is no consensus concerning34. The serum volume of HFD mice and HFD + EXE mice was no significant different in this study. These results indicated that exercise reduced the serum IGFBP5/IGF1 ratio to activate the action of IGF1 rather than exercise regulated the hepatic IGFBP5 levels, resulting in the amelioration of NAFLD.
Insulin resistance is vital for the development of NAFLD35 and exercise can decline insulin resistance by reducing adipokines and proteins16,17,36. IRS1 and protein kinase B (Akt) are essential factors for the insulin signaling pathway37. One study showed that under NAFLD condition, tyrosine phosphorylation levels of IRS1 and phosphorylation levels of Akt in the livers were weakened, whereas serine phosphorylation levels of IRS1 were enhanced38. Our study showed that exercise promoted IGF1 binding to its receptor by reducing the serum IGFBP5/IGF1 ratio, which weakened IRS1 serine phosphorylation and enhanced the phosphorylation of IRS1 tyrosine residues and Akt. Surprisingly, exercise also increased AMPKα phosphorylation levels in the liver. Hepatic and adipose AMPK activation can increase insulin sensitivity39, whereas its phosphorylation can suppress de novo lipogenesis in NAFLD40,41.
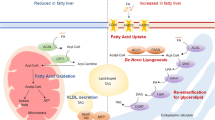
In Conclusion, although exercise did not affect IGFBP5 expression in the liver, it activated the effects of IGF1 by reducing the serum IGFBP5/IGF1 ratio in NAFLD mice (Fig. 5). The release of IGF1 then triggered IRS1/Akt/AMPK signaling pathway to ameliorate HFD-induced NAFLD (Fig. 5).
Exercise activated the effects of IGF1 by reducing the serum IGFBP5/IGF1 ratio, consequently triggering the IRS1/Akt/AMPK signaling pathway to ameliorate HFD-induced NAFLD.
Materials and methods
Animal model
Five-week-old male C57BL/6J mice were purchased from the Experimental Animal Center of Southern Medical University (Guangzhou, China) and acclimated for 1 week. The mice were housed on a 12-h light–dark cycle at 22℃–24℃ and were provided free access to food and water. All animal care and laboratory experimental procedures were conducted in accordance with the Chinese Guidelines for Animal Welfare and Experimental Protocols and were approved by the Animal Experiment Administration Committee of Guangzhou Sport University (2022DWLL-28). All mice were randomly divided into a low-fat diet control group (LFD, n = 8), a high-fat diet group (HFD, n = 8), and a HFD plus exercise group (HFD + EXE, n = 8). The LFD mice were fed a diet of 10% kcal from fat (D12450J, Research Diets Inc.) throughout the experimental period. The other two groups received a HFD containing 60% kcal from fat (D12492, Research Diets Inc.) for 24 weeks. After 12 weeks of HFD feeding, the HFD + EXE group mice were subjected to training on a treadmill at a 0% grade for 12 weeks. A day after the phase of final training, the serum and liver samples were collected from sacrificed mice under anesthesia (intraperitoneal-injected sodium pentobarbital 50 µg/g).
Exercise protocol
The exercise mice were trained on a treadmill at 0% grade 5 days per week for 12 weeks. The first week is an adaptation period, a 5 min warm-up period at 6 m/min, a 20 min of the main exercise at 10 m/min, and a 5 min of cool down at 6 m/min were performed. The mice performed 5 min warm up session at 6 m/min, a 50 min of the main training at 12 m/min, and a 5 min of cool down at 6 m/min from the 2nd to the final week.
Blood analysis
The serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels were determined using commercial kits (Jiancheng Bioengineering Institute, Nanjing, China).
Glucose tolerance test (GTT) and insulin tolerance test (ITT)
A GTT was carried out after 11 weeks of diet and exercise intervention. The levels of blood glucose were detected after overnight fasting using a glucometer (Accu-Chek, Roche). Thereafter, mice received an intraperitoneal injection of glucose at a dose of 1 mg/g body weight, after which blood collection was from the tail vein at 0, 30, 60, 90, 120 min after injection, respectively.
An ITT was performed in the previous week of final diet and exercise intervention. The mice were performed an intraperitoneal injection of insulin (Sigma) at a dose of 0.75 IU/kg body weight after a 3-h fast. Blood glucose levels were determined from tail vein at 0, 30, 60, 90, and 120 min after injection using a glucometer (Accu-Chek, Roche).
Homeostatic model assessment for insulin resistance (HOMA-IR) calculation
HOMA-IR was calculated to evaluate the insulin resistance. The formula is as follows: HOMA-IR = fasting glucose (mmol/L) × fasting insulin (mIU/L)/22.542.
Hepatic triglyceride and total cholesterol analysis
Hepatic TG and TC levels were measured using commercial kits (Jiancheng Bioengineering Institute, Nanjing, China), according to the manufacturer’s instructions.
Histological analysis of the liver
Fresh liver tissues were fixed with 4% paraformaldehyde solution for 24 h, embedded in paraffin, and sliced into 4-mm sections for hematoxylin–eosin (H&E) and Sirius Red staining. The NAFLD activity score (NAS) was then calculated according to the guidance supplied by the Pathology Committee of the NASH Clinical Research Network43: steatosis (< 5% = 0; 5–33% = 1; 33–66% = 2; >66% = 3), lobular inflammation (none = 0; <2 foci = 1; 2–4 foci = 2; >4 foci = 3), and hepatocellular ballooning (none = 0; few = 1; prominent = 2). All features of per sample were scored in a blinded manner based on six fields of view. Individual scores for each field of view were summed to estimate the NAS for each mouse.
Serum proteomic analyses
Label-free mass spectrometry technology, coupled with liquid chromatography with tandem mass spectrometry, was used to quantitatively analyze proteins and identify those differentially expressed. All such analyses were performed by Metabo-Profile Biotechnology (Shanghai) Co., Ltd., China. Results were analyzed using FragPipe software, whereas protein data were screened from the UniProt database.
ELISA assays
The serum levels of total IGF1 (Mediagnost, Reutlingen, Germany), free IGF1 (AnshLabs, Webster, TX, USA) and IGFBP5 (Raybiotech, Norcross, Georgia, USA) were measured following the manufacturers’ instructions. Absorbance was read by spectrophotometry (BioTek, Winooski, USA).
Quantitative real-time PCR
Total RNA was extracted from frozen liver using RNeasy Plus Mini Kit (Qiagen) following the manufacturer’s instructions. The primer sequences are detailed in Table 1. Expression levels were normalized to those of the housekeeping gene GAPDH.
Western blot analysis
After extracting the protein from experimental animal livers, total protein concentrations were measured using a BCA protein assay kit (Thermo Fisher Scientific). Equal amounts of total protein were separated using sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. These membranes were then blocked and incubated with primary antibodies against IGFBP5 (Abcam, ab254324, Cambridge, MA, USA), IGF1(Abcam, ab223567, Cambridge, MA, USA), IGF1R (Abcam, ab182408, Cambridge, MA, USA), IRS1 (Proteintech, 17509, China), phospho-IRS1 (Tyr612) (Thermo Fisher, 44816G, Waltham, MA, USA), phospho-IRS1 (Ser307) (Bioss, bs3200R, China), Akt (Servicebio, GB15689, China), phospho-Akt (Ser473) (ImmunoWay, YP0006, Plano, TX, USA), AMPKα (ImmunoWay, YT0216, Plano, TX, USA), phospho-AMPKα (Thr183/172) (ImmunoWay, YP0575, Plano, TX, USA), and β-actin (Servicebio, GB15003, China). The membranes were incubated for 1 h with the following secondary antibody: goat anti-rabbit IgG-HRP (Servicebio, GB23303, China). The immunoblot signals were quantified using Quantity One software after signal detection.
Statistical analysis
All data were presented expressed as mean ± standard error of the mean (mean ± SEM). One-way analysis of variance followed by Tukey’s test for three groups comparisons. A t-test was used for two groups comparisons in the analysis of protein profile. All analyses were performed using GraphPad Prism (version 8.0.2), with a P value less than 0.05 indicating statistical significance.
Data availability
The datasets used in the analyses described in this study are available from the corresponding author on reasonable request.
References
Yang, W. et al. Exercise suppresses NLRP3 inflammasome activation in mice with diet-induced NASH: a plausible role of adropin. Laboratory investigation; a journal of technical methods and pathology (2020).
Chalasani, N. et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American association for the study of liver diseases. Hepatology 67 (1), 328–357 (2018).
Zhou, F. et al. Unexpected rapid increase in the burden of NAFLD in China from 2008 to 2018: A systematic review and Meta-Analysis. Hepatology 70 (4), 1119–1133 (2019).
Luci, C., Bourinet, M., Leclère, P. S., Anty, R. & Gual, P. Chronic inflammation in Non-Alcoholic steatohepatitis: molecular mechanisms and therapeutic strategies. Front. Endocrinol. 11, 597648 (2020).
Chalasani, N. et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American gastroenterological association, American association for the study of liver diseases, and American college of gastroenterology. Gastroenterology 142 (7), 1592–1609 (2012).
European Association for the Study of the. European association for the study of, D.; European association for the study of, O., EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. Diabetologia 59 (6), 1121–1140 (2016).
Fan, J. G. & Farrell, G. C. Epidemiology of non-alcoholic fatty liver disease in China. J. Hepatol. 50 (1), 204–210 (2009).
Xiao, Z., Chu, Y. & Qin, W. IGFBP5 modulates lipid metabolism and insulin sensitivity through activating AMPK pathway in non-alcoholic fatty liver disease. Life Sci. 256, 117997 (2020).
Bach, L. A. Insulin-like growth factor binding proteins 4–6. Best Pract. Res. Clin. Endocrinol. Metab. 29 (5), 713–722 (2015).
Yamaguchi, Y., Yasuoka, H., Stolz, D. B. & Feghali-Bostwick, C. A. Decreased caveolin-1 levels contribute to fibrosis and deposition of extracellular IGFBP-5. J. Cell. Mol. Med. 15 (4), 957–969 (2011).
Chen, Z. et al. Down-regulation of insulin-like growth factor binding protein 5 is involved in intervertebral disc degeneration via the ERK signalling pathway. J. Cell. Mol. Med. 23 (9), 6368–6377 (2019).
Gleason, C. E. et al. Role of insulin-like growth factor-binding protein 5 (IGFBP5) in organismal and pancreatic beta-cell growth. Mol. Endocrinol. 24 (1), 178–192 (2010).
Han, N. et al. Local application of IGFBP5 protein enhanced periodontal tissue regeneration via increasing the migration, cell proliferation and osteo/dentinogenic differentiation of mesenchymal stem cells in an inflammatory niche. Stem Cell. Res. Ther. 8 (1), 210 (2017).
Fleming, J. M., Brandimarto, J. A. & Cohick, W. S. The mitogen-activated protein kinase pathway tonically inhibits both basal and IGF-I-stimulated IGF-binding protein-5 production in mammary epithelial cells. J. Endocrinol. 194 (2), 349–359 (2007).
Colak, Y. et al. Serum concentrations of human insulin-like growth factor-1 and levels of insulin-like growth factor-binding protein-5 in patients with nonalcoholic fatty liver disease: association with liver histology. Eur. J. Gastroenterol. Hepatol. 24 (3), 255–261 (2012).
Tayebi, S. M., Ghanbari-Niaki, A., Saeidi, A. & Hackney, A. C. Exercise training, neuregulin 4 and obesity. Annals Appl. Sport Sci. 5 (2), 1–2 (2017).
Saeidi, A. et al. Independent and combined effects of antioxidant supplementation and circuit resistance training on selected adipokines in postmenopausal women. Front. Physiol. 10, 484 (2019).
Tsuchiya, Y., Takakura, H., Osawa, S. & Izawa, T. High-intensity interval training enhances mRNA expression of IGF1Ea in rat Achilles tendon. Mol. Biol. Rep. 51 (1), 374 (2024).
Matheny, R. W., Merritt, E., Zannikos, S. V., Farrar, R. P. & Adamo, M. L. Serum IGF-I-deficiency does not prevent compensatory skeletal muscle hypertrophy in resistance exercise. Exp. Biol. Med. (Maywood). 234 (2), 164–170 (2009).
Nindl, B. C. et al. Microdialysis-Assessed exercised muscle reveals localized and differential IGFBP responses to unilateral stretch shortening cycle exercise. Front. Endocrinol. 11, 315 (2020).
Stokes, K., Nevill, M., Frystyk, J., Lakomy, H. & Hall, G. Human growth hormone responses to repeated bouts of sprint exercise with different recovery periods between bouts. J. Appl. Physiol. (1985). 99 (4), 1254–1261 (2005).
Dall, R. et al. No evidence of insulin-like growth factor-binding protein 3 proteolysis during a maximal exercise test in elite athletes. J. Clin. Endocrinol. Metab. 86 (2), 669–674 (2001).
Cetinkaya, C. et al. Positive effects of aerobic exercise on learning and memory functioning, which correlate with hippocampal IGF-1 increase in adolescent rats. Neurosci. Lett. 549, 177–181 (2013).
Leme, J. A. et al. Long-term physical training increases liver IGF-I in diabetic rats. Growth Horm. IGF Res. 19 (3), 262–266 (2009).
Copeland, J. L., Heggie, L. & IGF-I IGFBP-3 during continuous and interval exercise. Int. J. Sports Med. 29 (3), 182–187 (2008).
Nindl, B. C. et al. Overnight responses of the Circulating IGF-I system after acute, heavy-resistance exercise. J. Appl. Physiol. (1985). 90 (4), 1319–1326 (2001).
Ben Ounis, O. et al. Effect of individualized exercise training combined with diet restriction on inflammatory markers and IGF-1/IGFBP-3 in obese children. Ann. Nutr. Metab. 56 (4), 260–266 (2010).
Chen, Z. et al. Exercise protects proliferative muscle satellite cells against exhaustion via the Igfbp7-Akt-mTOR axis. Theranostics 10 (14), 6448–6466 (2020).
Tiniakos, D. G., Vos, M. B. & Brunt, E. M. Nonalcoholic fatty liver disease: pathology and pathogenesis. Annu. Rev. Pathol. 5, 145–171 (2010).
Liu, W., Baker, R. D., Bhatia, T., Zhu, L. & Baker, S. S. Pathogenesis of nonalcoholic steatohepatitis. Cell. Mol. Life Sci. 73 (10), 1969–1987 (2016).
Nguyen, A. et al. Liver fat content in people with pituitary diseases: influence of serum IGF1 levels. Horm. Metab. Res. 50 (4), 303–307 (2018).
Eroglu, I. et al. Nonalcoholic fatty liver disease, liver fibrosis, and utility of noninvasive scores in patients with acromegaly. J. Clin. Endocrinol. Metab. 109 (1), e119–e129 (2023).
Ichikawa, T. et al. Role of growth hormone, insulin-like growth factor 1 and insulin-like growth factor-binding protein 3 in development of non-alcoholic fatty liver disease. Hepatol. Int. 1 (2), 287–294 (2007).
Zouhal, H. et al. The effects of exercise training on plasma volume variations: A systematic review. Int. J. Sports Med. (2023).
Akhtar, D. H., Iqbal, U., Vazquez-Montesino, L. M., Dennis, B. B. & Ahmed, A. Pathogenesis of insulin resistance and atherogenic dyslipidemia in nonalcoholic fatty liver disease. J. Clin. Transl Hepatol. 7 (4), 362–370 (2019).
Saeidi, A. et al. Astaxanthin Supplemented with High-Intensity Functional Training Decreases Adipokines Levels and Cardiovascular Risk Factors in Men with Obesity. Nutrients 15, (2) (2023).
Saeidi, A. et al. Effects of exercise training on type 2-diabetes: the role of Meteorin-like protein. Health Promotion Perspect. 9 (2), 89–91 (2019).
Sakurai, Y., Kubota, N., Yamauchi, T. & Kadowaki, T. Role of Insulin Resistance in MAFLD. Int J Mol Sci 22 (8). (2021).
Jung, T. W., Park, H. S., Choi, G. H., Kim, D. & Lee, T. beta-aminoisobutyric acid attenuates LPS-induced inflammation and insulin resistance in adipocytes through AMPK-mediated pathway. J. Biomed. Sci. 25 (1), 27 (2018).
Li, Y. et al. AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell. Metab. 13 (4), 376–388 (2011).
Kamikubo, R., Kai, K., Tsuji-Naito, K. & Akagawa, M. beta-Caryophyllene attenuates palmitate-induced lipid accumulation through AMPK signaling by activating CB2 receptor in human HepG2 hepatocytes. Mol. Nutr. Food Res. 60 (10), 2228–2242 (2016).
Yang, W. et al. Exercise ameliorates the FGF21-adiponectin axis impairment in diet-induced obese mice. Endocr. Connections. 8 (5), 596–604 (2019).
Kleiner, D. E. et al. Nonalcoholic steatohepatitis clinical research, N., design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41 (6), 1313–1321 (2005).
Acknowledgements
The authors thank all the laboratory members for their help (Haohui Lai, Jianhua Wang, Wenbi He, Huan Wang, Gaoyuan Yang and Yu Wang).
Funding
This work was supported by the Department of Education of Guangdong Province, China. (2023ZDZX2034) (to Y.Y.), and the Science and Technology Plan Project of Liwan District of Guangzhou, China (to L.Y.).
Author information
Authors and Affiliations
Contributions
Yang Yu: Writing – review & editing, Visualization, Supervision, Formal analysis, Data curation, Funding acquisition, Conceptualization. Guanxiong Zhu: Writing – original draft, Visualization, Investigation, Validation, Formal analysis, Data curation. Zeyu Zhang: Writing – original draft, Visualization, Investigation, Validation, Formal analysis, Data curation. Huiguo Wang: Writing – review & editing, Supervision, Methodology, Investigation. Liting Zeng: Writing – review & editing, , Validation, Formal analysis, Data curation. Jingbo Xia: Writing – review & editing, Supervision, Methodology. Xiaoguang Liu: Writing – review & editing, Supervision, Methodology. Chunlu Fang: Writing – review & editing, Validation, Methodology. Shujing Liu: Writing – review & editing, Validation, Methodology. Yunjie Yang: Writing – review & editing, Validation, Methodology. Lina Yu: Writing – review & editing, Supervision, Project administration, Funding acquisition, Conceptualization. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
All animal experiments in this study were performed in accordance with the ARRIVE guidelines 2.0, and the Institutional Animal Care and Use Committee of Guangzhou Sport University approved all procedures performed on animals.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, Y., Zhu, G., Zhang, Z. et al. Exercise ameliorates nonalcoholic fatty liver disease by reducing the IGFBP5 to IGF1 ratio to activate AMPK pathway. Sci Rep 15, 23083 (2025). https://doi.org/10.1038/s41598-025-07857-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07857-y