Abstract
COVID-19 is associated with a wide spectrum of neurological alterations, ranging from headache and dizziness to severe encephalopathy and inflammatory neurological diseases (IND), and neuropathological findings suggest immune-mediated processes. Therefore, we sought to characterize profiles of cytokines, chemokines, growth factors, and markers of central nervous system (CNS) homeostasis in COVID-19 patients with neurological alterations to identify key factors and mechanisms underlying CNS disturbances in COVID-19. The study included a case series of 52 COVID-19 patients with neurological manifestations, which were categorized into three groups: isolated refractory headache (n = 14), encephalopathy (n = 24), and IND (n = 14). Individuals with non-inflammatory, non-infectious neurological conditions (n = 9) were included as negative controls. Paired CSF and serum samples were assessed for 56 biomarkers. Regardless of the neurological condition, COVID-19 patients exhibited elevated CSF levels of proinflammatory mediators, including IL-2, IL-3, IL-6, IL-15, IL-25, IFN-α2, CCL7, CCL11, and GM-CSF. Patients with encephalopathy and IND also showed increased IL-1β, IL-18, TNF-α, neopterin, IL-7, CXCL8, CXCL9, TGF-α, EGF, sTREM-2, and HMGB1, consistent with a CNS cytokine storm. In contrast, individuals with isolated refractory headache showed a modest inflammatory profile, compatible with the limited CNS involvement. COVID-19 patients showed elevated serum IL-13, IL-18, TNF-α, VILIP-1, TGF-α, and VEGF levels, indicating systemic inflammation and potential blood–brain barrier (BBB) disruption. β-NGF was increased in the CSF of patients with encephalopathy and IND, suggesting the activation of neuroprotective responses during patient recovery. Functional protein network analysis showed a significant enrichment of interactions between factors altered in the CSF of patients with encephalopathy and IND, many of them related to processes of neuroinflammation and microglial functions, and leukocyte chemotaxis, activation and proliferation. These findings support a model in which both systemic immune activation and localized neuroinflammation contribute to the diversity of neurological outcomes observed in COVID-19, and dysregulated cytokine production, glial activation, inflammasome activity and BBB disturbances represent key factors in neuro-COVID-19 pathogenesis.
Similar content being viewed by others
Introduction
At the onset of the Coronavirus Disease 2019 (COVID-19) pandemic, many groups reported the involvement of the central and peripheral nervous systems in SARS-CoV-2 infection. Patients displayed neurological manifestations as the initial presentation or after the onset of classic symptoms such as fever, cough, diarrhea, and fatigue1,2,3,4,5,6. Central nervous system (CNS) manifestations, including headache, dizziness, altered consciousness, and acute cerebrovascular disease, were more common in severe cases than in patients with mild to moderate symptoms as determined by their respiratory status5. In contrast, mass vaccination strategies against COVID-19 that were implemented in many countries since December 2020 promoted a dramatic reduction in the number of hospitalizations and deaths7,8.
Neurological symptoms are still among the most common manifestations in this new setting of widespread immunity against SARS-CoV-2. This includes fatigue, cognitive impairment or brain fog, dizziness, depression and anxiety, loss or alteration of smell/taste, among others9. However, mechanisms of neuroinvasion, cellular types involved in CNS infection by SARS-CoV-2, and the dynamics and duration of neuroinfection are still not fully understood10,11. Indeed, multiple features might contribute to the diversity of neurological outcomes in COVID-1912,13. Thus, despite the reduced incidence of neurological disorders such as encephalitis, demyelinating diseases, and encephalopathies in association with COVID-19 after widespread immunization by vaccine or prior infection, data on inflammation markers in patients with acute neurological outcomes, particularly those infected in the pre-Omicron era, are key to support a better understanding of events associated with general neurological involvement in COVID-19.
In this study, we aimed to identify possible mechanisms of neuropathogenesis in COVID-19. We assessed the concentration of cytokines, chemokines, growth factors, and factors associated with the activation of immune cells and CNS homeostasis in paired cerebrospinal fluid (CSF) and serum samples from patients with COVID-19 presenting with isolated refractory headache, encephalopathy, and inflammatory neurological diseases (IND). We observed that factors associated with vascular permeability were elevated in the serum of patients, regardless of their neurological status. Serum levels of interleukin (IL)-13, IL-18, TNF-α, TGF-α, VEGF, and visinin-like protein 1 (VILIP-1) were also increased. Patients with encephalopathy and IND had elevated CSF levels of neopterin and other markers of activation of immune cells, indicating the establishment of a CNS cytokine storm, and the activation of inflammasomes. In contrast, nerve growth factor beta (β-NGF) was increased in the CSF of these patients, suggesting the activation of neuroprotective responses.
Materials and methods
Study population and biological samples
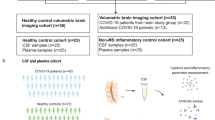
This study represents a case series of patients with COVID-19 and neurological manifestations (n = 52) admitted between April and June 2020 to reference hospitals in the cities of Rio de Janeiro (Hospital Federal dos Servidores do Estado and Instituto Nacional de Infectologia Evandro Chagas) and Niterói (Complexo Hospitalar de Niterói), Brazil. All patients enrolled in the study had COVID-19 confirmed by detection of SARS-CoV-2 RNA in nasopharyngeal swabs by quantitative reverse transcription PCR (RT-qPCR), as previously described6. CSF samples were collected and immediately processed, aliquoted, and stored at -80 °C. Cell counts, total protein, and glucose levels were determined in fresh CSF aliquots, and samples were also tested by PCR for exclusion of patients with concomitant infection by other neuropathogens6. Lumbar punctures were atraumatic, and CSF samples had adequate red blood cell (RBC) counts (4 RBC/mm3, interquartile range = 0–33 RBC/mm3), except for two patients with encephalopathy who had 2048 and 5547 RBC/mm3. Paired serum samples collected on the same day of the lumbar puncture were also available for 31 participants. Clinical data were obtained from medical records and data on the CSF cell counts, oligoclonal bands, and the concentration of neurofilament light chain (NfL) and total Tau protein are shown elsewhere6,14.
CSF (n = 9) and paired serum (n = 5) samples obtained from patients with non-inflammatory non-infectious neurological diseases (NI) (normal pressure hydrocephalus) who underwent CSF withdrawal before the emergence of COVID-19 in Brazil were used as uninfected controls. These individuals had normal intracranial opening pressure (≤ 200 mmH2O), and normal values for CSF cell counts (< 5 cells/mm3) and total proteins (≤ 45 mg/dL)14.
Patients with COVID-19 and neurological alterations were grouped according to their primary neurological condition, as previously characterized6,14,15: (i) isolated refractory headache (n = 14), excluding patients with clinical or laboratory evidence for meningitis or meningoencephalitis, and inflammatory changes in CSF analysis; (ii) encephalopathy (n = 24), which was characterized by diffuse brain dysfunction, convulsions, alterations in cognition, consciousness, personality or behavior, and without signs of acute CNS inflammation; and (iii) inflammatory neurological diseases (IND) (n = 14), which included acute disseminated encephalomyelitis (ADEM) (n = 2), encephalitis (n = 2), meningitis (n = 2), meningoencephalitis (n = 4), acute myelitis (n = 3), and neuromyelitis optica (n = 1). Seizures and abnormalities on electroencephalogram (EEG) were considered for the diagnosis of both encephalopathy and encephalitis. To avoid overlapping between these groups, encephalopathy was defined as a syndrome of altered mental status (major criterion) without signs of acute brain inflammation, such as CSF pleocytosis and changes in brain magnetic resonance imaging (MRI). To define encephalitis, in addition to altered mental status, it was required at least two of the following minor criteria: fever, new‐onset seizures not attributable to a preexisting seizure disorder, focal neurologic signs, CSF pleocytosis, acute abnormalities on neuroimaging, or EEG abnormalities indicative of cerebral inflammation, such as generalized or focal slow‐wave activity, electrographic seizures16. In addition, patients with evidence of acute cerebrovascular disease in brain MRI or tomography were excluded from the study.
Quantification of factors associated with neuroinflammation
The CSF and serum levels of VILIP-1, brain-derived neurotrophic factor (BDNF), vascular endothelial growth factor (VEGF), soluble triggering receptor expressed on myeloid cells 1 (sTREM-1), sTREM-2, β-NGF, IL-18, transforming growth factor β1, (TGF- β1), and soluble receptor for advanced glycation end products (sRAGE) were determined using the cytometry bead-based multiplex assay LEGENDplex Human Neuroinflammation Panel 1 (Biolegend, Cat. No. 740795, USA), following the manufacturer’s instructions. The CSF was assessed undiluted, and serum samples were diluted at 1:2. Data was acquired in a FACSCanto II cytometer (BD Biosciences, USA), and the concentration of the analytes was calculated in 5-parameter logarithmic curves.
CSF and serum concentration of cytokines, chemokines and growth factors, including: IL-1α, IL-1β, IL-1RA, IL-2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-8/CXCL8, IL-9, IL-10, IL-12(p40), IL-12(p70), IL-13, IL-15, IL-17A, IL-17F, IL-22, IL-25, IL-27, IFN-α2, IFN-γ, soluble CD40 ligand (sCD40L), TGF-α, tumor necrosis factor α (TNF-α), TNF-β, monocyte chemoattractant protein-1 (MCP-1/CCL2), macrophage inflammatory protein 1α (MIP-1α/CCL3), MIP-1β/CCL4, monocyte chemotactic protein-3 (MCP3/CCL7), eotaxin/CCL11, macrophage-derived chemokine (MDC/CCL22), growth-related oncogene alpha (GROα/CXCL1), monokine induced by IFN-γ (MIG/CXCL9), IFN-γ-induced protein 10 (IP-10/CXCL10), fractalkine/CX3CL1, epidermal growth factor (EGF), fibroblast growth factor 2 (FGF2), granulocyte colony-stimulating factor (G-CSF), macrophage colony-stimulating factor (M-CSF), granulocyte/macrophage colony-stimulating factor (GM-CSF), platelet-derived growth factor (PDGF)-AA, PDGF-AB/BB, and Fms-like tyrosine kinase 3 ligand (FLT3L) were determined by Luminex with the MILLIPLEX MAP Human Cytokine/Chemokine/Growth Factor Panel A (Merck Millipore, Cat. No. HCYTA-60K-PX48) according to manufacturer’s instructions. Data was acquired in a MAGPIX system (Merck Millipore), and the concentration of the analytes was calculated in 5-parameter logarithmic curves. ELISA was used to determine the concentrations of neopterin (Neopterin ELISA, Cat #RE59325, IBL International, Germany) and high mobility group protein B1 (HMGB1) (FineTest, Cat. No. EH0884, China), following the manufacturer’s instructions. For the quantification of HMGB1, CSF and serum samples were diluted at 1:100 and 1:250, respectively.
Statistical analysis
Data analysis was performed using R software v.4.4.2. Data were log10-transformed and are shown as the median and interquartile range (IQR). Comparative analysis was carried out with Kruskal–Wallis and posthoc analysis with Dunn’s test with the Benjamini–Hochberg method (False Discovery Rate, FDR) for adjusting p-values in multiple comparisons. Summary data for all 56 factors evaluated in this study are presented in additional graphs and tables [see Suppl. Figs. 1–5 in the Additional file 1 and tables in the Additional file 2]. Factors were classified as proinflammatory and regulatory cytokines, chemokines, and growth factors as previously described elsewhere17. Circular boxplots were constructed for each neuro-COVID-19 condition and the control group using the proportion of individuals with concentration values above the median determined for each factor in the CSF and serum for the study population. Relative expression of factors significantly altered in the CSF and serum of COVID-19 patients in the comparison to uninfected controls was evaluated by clustering analysis using the pheatmap package for R. Spearman’s rank correlation coefficients were calculated to evaluate associations between factors that were significantly distinct in at least one study group and that had detectable levels in more than half of the samples. Patterns associated with each neurological outcome were determined by concomitant clustering analysis using the pheatmap package for R. Results were considered significant at p < 0.05. Data of CSF cell counts, total protein concentration, NfL and total Tau protein levels, neopterin (CSF/serum) ratio, oligoclonal bands, and intracranial opening pressure are shown in the supplementary material [see Additional file 3]. Gene coexpression analysis was performed with the STRING database tool (https://string-db.org/), in which coexpression scores are based on RNA expression patterns, and on protein co-regulation provided by ProteomeHD (www.proteomehd.net/predict) using the Gene Expression Omnibus (GEO) microarray expression data, corroborating evidence from GEO RNA-seq expression data (www.ncbi.nlm.nih.gov/geo/), and PRIDE proteomics data (www.ebi.ac.uk/pride/) [see Suppl. Fig. 6 in the Additional file 1]. Functional protein network analysis of factors significantly altered in the CSF and serum of patients according to the neurological outcomes associated with COVID-19 was performed with the STRINGdb package for R, version 12.0, accessing the human database (TaxID = 9606), using an interaction score set for high confidence (0.700), and the Kamada-Kawai method of force-directed layout [see Suppl. Figure 7 in the Additional file 1]. Enrichment analysis of factors according to biological functions and processes were retrieved from Gene Ontology database (GO-terms), Compartments subcellular localization database (GOCC identifiers), Reactome (HSA identifiers), and WikiPathways (WP identifiers).
Results
The study population was predominantly composed of patients with COVID-19 who displayed neurological manifestations within 14 days from symptom onset. Isolated refractory headache arose after a median interval of 2 days (IQR = 0–10 days), encephalopathy after 3 days (IQR = 2–7 days), and acute neuroinflammatory diseases after 4 days (IQR = 0–14 days) from the onset of common COVID‐19 symptoms. Fever and headache were the most frequent symptoms, observed in 69% and 60% of patients, respectively, followed by cough (51%), myalgia (37%), altered consciousness (34%), which included confusional state, delirium, and drowsiness; anosmia/ageusia (34%), respiratory alterations (26%)—including nasal congestion, dyspnea, and thorax computed tomography with ground glass and > 50% involvement, and fatigue (23%). Other less frequent symptoms included convulsion (11%), paresthesia (9%), diarrhea (9%), and aphasia (3%). Notably, three patients progressed to death, all classified with encephalopathy. Only three patients enrolled in the study developed neurological alterations after 14 days from COVID‐19 onset: one with encephalopathy, one with ADEM, and one with acute myelitis, starting at days 18, 19, and 29, respectively. At the time of participant inclusion in the study, FDA-approved antivirals for COVID-19 treatment, such as remdesivir and Paxlovid, were not yet available. Among 25 patients with data on pre-admission therapeutic interventions, nine received off-label treatments for COVID-19, including ivermectin, hydroxychloroquine, and azithromycin. In contrast, high-dose intravenous methylprednisolone and immunoglobulin therapy were initiated only after CSF was collected for laboratory analysis.
CSF and serum changes associated with acute neuro-COVID-19 conditions
Overall, patients with COVID-19 and neurological outcomes had increased CSF levels of IL-2, IL-3, IL-6, IL-15, IL-25, IFN-α2, MCP-3/CCL7, eotaxin/CCL11, and GM-CSF compared to a control group of non-inflammatory and non-infectious neurological diseases (NI) (Fig. 1). Patients with isolated headache and encephalopathy also had increased concentrations of IL-4 and fractalkine/CX3CL1 (Fig. 1). Both patients with encephalopathy and IND had higher CSF levels of inflammatory cytokines/factors associated with innate responses such as IL-18, TNF-α, and neopterin, of chemokines IL-8/CXCL8 and MIG/CXCL9, and growth factors, including IL-7, β-NGF, and TGF-α, in addition to elevated markers of cellular stress such as sTREM-2, and HMGB1, a damage-associated molecular pattern (DAMP) (Fig. 1). The CSF of patients with encephalopathy also displayed elevated levels of IL-1β, MIP-1α/CCL3, EGF, and FLT3L, while patients with IND had higher CSF levels of IL-5 and G-CSF (Fig. 1).
CSF analysis in COVID-19 patients with acute neurological disturbances. CSF samples from COVID-19 patients with isolated refractory headache (H) (n = 14), encephalopathy (E) (n = 24), and inflammatory neurological diseases (IND) (n = 14), and from a control group of individuals with non-inflammatory, non-infectious neurological diseases (NI) (n = 9) were assessed by Luminex (MILLIPLEX MAP Human Cytokine/Chemokine/Growth Factor Panel A, Merck Millipore), Cytometry bead array (LEGENDplex Human Neuroinflammation Panel 1, Biolegend), and ELISA. Concentration values in pg/mL were log10-transformed and are presented as median and interquartile range (IQR). Comparative analysis was performed with the Kruskal–Wallis test and posthoc analysis using Dunn’s test with Benjamini–Hochberg method for adjusting p-values in multiple comparisons. Results of proinflammatory factors (IL-1β, IL-6, TNF-α, IL-15, IL-25, IL-18, IFN-α2, and neopterin), regulatory cytokines (IL-4 and IL-5), chemokines (MIP-1α/CCL3, MCP-3/CCL7, eotaxin/CCL11, IL-8/CXCL8, MIG/CXCL9, and fractalkine/CX3CL1), growth factors (IL-2, IL-3, IL-7, FLT3L, EGF, GM-CSF, β-NGF, TGF-α, and G-CSF), and markers of cellular stress response (HMGB1 and sTREM-2) that presented significant changes (Kruskal–Wallis p < 0.05) are shown. Summary data for all factors evaluated are presented in additional graphs and tables [see Additional file 1 and Additional file 2]. *, adj. p < 0.05; **, adj. p < 0.01.
Serum analysis revealed that IL-13, IL-18, TNF-α, TGF-α, VEGF, and VILIP-1 were increased in all groups of patients with neuro-COVID-19 compared to controls (Fig. 2). Patients with isolated headache also had high serum levels of IL-10, while those with encephalopathy also showed increased levels of IL-6, IL-10, and IL-27. In turn, patients with encephalopathy had reduced serum levels of BDNF compared to individuals with headache and IND (Fig. 2).
Serum analysis in COVID-19 patients with acute neurological disturbances. Paired serum samples from COVID-19 patients with isolated refractory headache (H) (n = 7), encephalopathy (E) (n = 13), and inflammatory neurological diseases (IND) (n = 11), and from a control group of individuals with non-inflammatory and non-infectious neurological diseases (NI) (n = 5) were assessed by Luminex (MILLIPLEX MAP Human Cytokine/Chemokine/Growth Factor Panel A, Merck Millipore), Cytometry bead array (LEGENDplex Human Neuroinflammation Panel 1, Biolegend), and ELISA. Concentration values in pg/mL were log10-transformed and are presented as median and interquartile range (IQR). Comparative analysis was performed with the Kruskal–Wallis test and posthoc analysis using Dunn’s test with Benjamini–Hochberg method for adjusting p-values in multiple comparisons. Results of proinflammatory factors (IL-6, TNF-α, and IL-18), regulatory cytokines (IL-10, IL-13, and IL-27), growth factors (TGF-α, VEGF, and BDNF), and markers of cellular stress response (VILIP-1) that presented significant changes (Kruskal–Wallis p < 0.05) are shown. Summary data for all factors evaluated are presented in additional graphs and tables [see Additional file 1 and Additional file 2]. *, adj. p < 0.05; **, adj. p < 0.01.
COVID-19 patients showed distinct CSF profiles according to their neurological status, with upregulation of many factors. In encephalopathy and IND groups, a higher proportion of individuals exhibited elevated CSF levels of several proinflammatory and growth factors, chemokines, and markers of cellular stress responses. This pattern was less striking in the group with isolated refractory headache, suggesting a hierarchical pattern associated with neurological severity (Fig. 3a–d). In contrast, serum analysis showed that encephalopathy was associated with an increased proportion of patients with high serum levels of proinflammatory and immune regulatory factors, while IND and headache groups had a relatively higher frequency of patients with upregulated proinflammatory and growth factors compared to controls (Fig. 3e–h).
CSF and serum profiles of neuro-COVID-19 patients. Circular boxplots display data from CSF and serum analyses for proinflammatory and regulatory cytokines, chemokines, growth factors, and markers of cellular stress response in COVID-19 patients with isolated refractory headache (H), encephalopathy (E), and inflammatory neurological diseases (IND), and in a control group of individuals with non-inflammatory and non-infectious neurological diseases (NI). Each bar represents the proportion of individuals with concentrations above the median determined for each factor in the (a–d) CSF and (e–h) serum. Proportions are shown as gray (up to 50%), light gray (50–75%), and white (75–100%).
Clustering analysis of relative expression of factors differentially expressed in the CSF showed that patients with IND and encephalopathy had more similar neuroinflammatory profiles, whereas patients with isolated refractory headache and uninfected controls clustered closely (Fig. 4), corroborating the hierarchical pattern of neurological disturbance shown in Fig. 3a–d. Patients with IND and encephalopathy who clustered with patients with isolated headache and uninfected controls had normal CSF cell counts (8 of 9) and/or low NfL levels (6 of 9), indicating limited CNS involvement, neuroinflammatory activity, and neurological damage [see Additional file 3]. Patients with isolated headache that clustered with encephalopathy and IND patients had neopterin (CSF/serum) ratio > 1.0 (3 of 4), indicating active CNS production rather than peripheral diffusion. However, no other pattern of changes in CSF was observed, with each patient showing only one alteration, such as elevated total protein, or elevated NfL levels, or intrathecal IgG production (type 2 oligoclonal band), or high intracranial opening pressure (≥ 250 mmH2O) [see Additional file 3]. By assessing serum data, it was observed that uninfected controls and COVID-19 patients formed two distinct clusters, except for one patient with headache and two with IND. As observed in the CSF, the serum of patients with encephalopathy and IND also showed similar changes; however, no specific profile was clearly associated with neuro-COVID-19 conditions (Fig. 5).
Heatmap analysis of CSF factors differentially expressed in neuro-COVID-19 patients. Data from patients with isolated refractory headache (H), encephalopathy (E), and inflammatory neurological diseases (IND), and from a control group of individuals with non-inflammatory, non-infectious neurological diseases (NI) were log10-transformed and normalized for each factor (columns). Reduced expression is depicted in shades of blue, increased expression in shades of red, with the median represented by white. Heatmaps were generated with the R software v.4.4.2 (https://cran.r-project.org/).
Heatmap analysis of serum factors differentially expressed in neuro-COVID-19 patients. Data from patients with isolated refractory headache (H), encephalopathy (E), and inflammatory neurological diseases (IND), and from a control group of individuals with non-inflammatory, non-infectious neurological diseases (NI) were log10-transformed and normalized for each factor (columns). Reduced expression is depicted in shades of blue, increased expression in shades of red, with the median represented by white. Heatmaps were generated with the R software v.4.4.2 (https://cran.r-project.org/).
Correlation analysis of CSF factors significantly upregulated in COVID-19 patients revealed a common cluster across all three neurological conditions, including IL-1β, IL-2, IFN-α2, TNF-α, IL-25, MCP-3/CCL7, fractalkine/CX3CL1, eotaxin/CCL11, and TGF-α (Fig. 6). Individuals with isolated headache and IND also had FLT3L and EGF included in this cluster of factors. According to gene coexpression scores from the STRING database, these factors are not expected to strongly correlate, except for IL-1β and TNF-α (coexpression score = 0.616), and IL-6 and IL-8/CXCL8 (coexpression score = 0.445) [see Suppl. Fig. 6 in Additional file 1].
Correlation analysis of CSF factors differentially expressed in neuro-COVID-19 patients. Spearman’s rank correlation coefficients were calculated for factors that were significantly different in at least one study group compared to uninfected controls, and that had detectable levels in at least 50% of samples from patients with (a) isolated refractory headache, (b) encephalopathy, (c) inflammatory neurological diseases (IND), and (d) a control group of individuals with non-inflammatory, non-infectious neurological diseases. Clustering analysis delineated patterns associated with each neurological outcome. Reduced expression is depicted in shades of blue, increased expression in shades of red, with the median represented by white. Results were considered significant at p < 0.05. *, p < 0.05; **, p < 0.01; ***, p < 0.001.
Functional protein network analysis of CSF and serum changes to identify biological processes associated with acute neuro-COVID-19
Functional protein network analysis was performed with factors altered in the CSF and serum of neuro-COVID-19 patients. A significant enrichment of interactions was observed for all groups, indicating their association with intricate pathways (Fig. 7 and Suppl. Fig. 7 in the Additional file 1). Overall, the CSF of patients with neurological outcomes in COVID-19 showed an enrichment of factors associated with neuroinflammation (GO:0150077 and GO:0150078), Th2 immune responses (GO:2000551, Th2 cytokine production; GO:0002830, upregulation of Th2 response), STAT signaling (GO:0042531, upregulation of STAT protein; GO:0007259, signaling pathway via JAK-STAT), leukocyte differentiation (GO:0002521) and proliferation (GO:007665), lymphocyte-mediated immunity (GO:0002706), activation of myeloid leukocytes (GO:0002274), natural killer (NK) cells (GO:0030101), and microglia cells (GO:1903978), monocyte chemotaxis (GO:0002548), tissue remodeling (GO:0034105) and regulation of neurogenesis (GO:0050767), cytokine-mediated signaling (GO:0019221), particularly by IL-2 family (HSA-451927), IL-4 and IL-13 (HSA-9785807), and IL-10 (HSA-6783783) cytokines, the IL-12 (GOCC:0043514), IL-23 (GOCC:0070743), and TGF-β complex (GOCC:0099126), in addition to processes involving proinflammatory and profibrotic mediators (WP5095), COVID-19 adverse outcome (WP4891), and innate lymphoid cells (ILC) development and heterogeneity (WP3893), and SARS-CoV-2 signaling (WP5115) and immune evasion (WP5039). Encephalopathy was also related to microglia migration (GO:1904141), and NK cell chemotaxis (GO:2000503), EGF signaling (GO:0038134, ERBB2-EGFR signaling pathway), Th1 cytokine response (GO:2000556), upregulation of GM-CSF (GO:0032725), production of cell adhesion molecules (GO:0060353), pyroptosis (HSA-5620971), and the NLRP3 inflammasome complex (GOCC:0072559), including the signaling by the IL-1 family (HSA-446652), and IL-18 (HSA-9012546). Encephalopathy and IND shared the enrichment of factors associated with NF-κB (GOCC:0071159) and inflammasomes (GOCC:0061702), Th1 immune response (GO:0042088), leukocyte adhesion to endothelium (GO:1904996), and leukocyte chemotaxis (GO:0030595), myeloid dendritic cell activation (GO:0001773), proliferation of activated T cells (GO:0042104), IFN-γ production (GO:0032729), VEGF production (GO:0010573), glial cell proliferation (GO:0060252), and podosome assembly (GO:0071803), and processes of prostaglandin signaling (WP5088), and mitochondrial oxidative stress induced by SARS-CoV-2 (WP5183). In contrast, encephalopathy and isolated headache shared enrichment of factors related to neuron homeostasis (GO:0070050).
Functional protein network analysis of CSF factors differentially expressed in neuro-COVID-19 patients. Factors significantly altered in the cerebrospinal fluid (CSF) according to neurological outcomes—isolated refractory headache (H), encephalopathy (E), and inflammatory neurological diseases (IND)—were analyzed using the STRING database, with a high confidence interaction score (0.700). The analysis included Gene Ontology (GO) terms, subcellular localization data from the Compartments database (GOCC identifiers), Reactome pathways (HSA identifiers), and WikiPathways (WP identifiers). Biological functions and processes with significant enrichment (FDR test, p-value < 0.05) are shown. The protein ratio represents the proportion of proteins in the query relative to the total number associated with a given biological process.
The analysis of serum factors showed some redundancy with CSF findings (Fig. 8). All groups exhibited enrichment of factors related to neuroinflammation (GO:0150078) and neuron death (GO:1901214 and GO:1903203), upregulation of STAT protein (GO:0042531), IL-18 signaling (WP4754 and HSA-9012546), IL-4 and IL-13 signaling (HSA-6785807), regulation of vascular wound healing (GO:0061043), of endothelial cell apoptosis (GO:2000351), and of NIK/NF-κB signaling (GO:1901224), lung fibrosis (WP3624), proinflammatory and profibrotic mediators (WP5095), cytokines and inflammatory response (WP530), IL-12 (GOCC:0043514), IL-23 (GOCC:0070743), and TGF-β complex (GOCC:0099126), ILC development and heterogeneity (WP3893), SARS-CoV-2 signaling (WP5115), and mitochondrial oxidative stress induced by SARS-CoV-2 (WP5183). Patients with isolated headache and encephalopathy displayed enrichment in processes associated with chronic inflammation to antigen (GO:0002874 and GO:0002439), downregulation of nervous system process (GO:0031645), IFN-γ production (GO:0032649), regulation of lymphocyte proliferation (GO:0050670), extrafollicular and follicular B cell activation by SARS-CoV-2 (WP5218), upregulation of IL-18 (GO:0032661), NF-κB complex (GOCC:0071159), IL-10 signaling (HSA-6783783), and COVID-19 adverse outcome (WP4891). Moreover, encephalopathy was associated with the enrichment of processes involving the regulation of macrophage activation (GO:0043030), Th2 response (GO:0002830), leukocyte adhesion to endothelium (GO:1904996), and the upregulation of several cytokines, including VEGF (GO:0010573), IL-17 (GO:0032740), IL-10 (GO:0032733), IL-1β (GO:0032731), IL-8/CXCL8 (GO:0032757), and IL-6 (GO:0032755), and processes related to the IL-27 (GOCC:0070744) and the inflammasome complex (GOCC:0061702), prostaglandin signaling (WP5088), SARS-CoV-2 innate immunity evasion (WP5039), and CD163-mediated anti-inflammatory response (HSA-9662834). In contrast, IND patients did not exhibit any exclusive enrichment of relevant biological processes.
Functional protein network analysis of serum factors differentially expressed in neuro-COVID-19 patients. Serum factors significantly altered in patients according to neurological outcomes—isolated refractory headache (H), encephalopathy (E), and inflammatory neurological diseases (IND)—were analyzed using the STRING database, with a high confidence interaction score (0.700). The analysis included Gene Ontology (GO) terms, subcellular localization data from the Compartments database (GOCC identifiers), Reactome pathways (HSA identifiers), and WikiPathways (WP identifiers). Biological functions and processes with significant enrichment (FDR test, p-value < 0.05) are shown. The protein ratio represents the proportion of proteins in the query relative to the total number associated with a given biological process.
Discussion
Central and peripheral neurological manifestations have been extensively reported in patients with COVID-19 since early stages of the pandemic2,3,4,6 and even after established widespread immunity by mass vaccination and prior infections9,13. Neurological alterations in COVID-19 were initially attributed to neuroinfection by SARS-CoV-2. However, viral RNA is rarely detected in the CSF18, and post-mortem analysis has shown that SARS-CoV-2 is present at low viral load in the brain19,20. In contrast, CSF analysis, neuroimaging and neuropathologic findings, as well as patient’s responses to immunotherapies6,20,21, support the notion of immune-mediated neuropathogenesis triggered by viral infection or antigens and/or systemic inflammation. Data in our study and from previous observations further support this hypothesis.
It this study, several biomarkers of inflammation were assessed in the CSF and serum of patients with COVID-19 and neurological manifestations to identify possible mechanisms of neuropathogenesis. In general, these patients had increased serum levels of IL-13, IL-18, TNF-α, VILIP-1, TGF-α, and VEGF, which were associated with the systemic inflammation induced by SARS-CoV-2 infection. In turn, the CSF analysis revealed an association between the neurological involvement and the activation, regulation and function of immune and glial cells, CNS cytokine storm, and inflammasome activity, particularly in cases of encephalopathy and IND. In contrast, patients with isolated refractory headache showed limited laboratory changes in the CSF, suggesting that physiopathological mechanisms other than neurological or systemic inflammation may also play a role. Most individuals (85.4%) with isolated refractory headache showed an altered intracranial opening pressure on lumbar puncture (≥ 200 mmH2O) and half of them had intracranial hypertension (≥ 250 mmH2O)15. Thus, disturbances in CSF production and absorption may contribute to the manifestation of isolated refractory headache in COVID-19 in the absence of evidence of meningitis or cerebrovascular disease.
The CSF of COVID-19 patients, regardless of their neurological outcomes, showed increased levels of IL-2, IL-3, IL-6, IL-15, IL-25, IFN-α2, MCP-3/CCL7, eotaxin/CCL11, and GM-CSF as a common feature. Functional protein network analysis indicated that these factors are involved in JAK-STAT-controlled signaling pathways, and changes in their levels can affect several CNS functions, including neuronal survival and glial activation. Functional enrichment analysis showed that these factors are also associated with the differentiation, proliferation, and recruitment of blood-derived leukocytes, such as monocytes, lymphocytes and NK cells in neuroinflammatory processes, and with adverse outcomes in COVID-19.
Notably, the CSF and serum of neuro-COVID-19 patients presented unrelated profiles. Indeed, no serum profile was specifically associated with a neurological condition, reflecting both the small number of differentially expressed factors and their independence from neurological outcomes. The limited changes in serum likely resulted from the predominance of patients with mild to moderate respiratory symptoms. In contrast, the correlation analysis of CSF factors in neuro-COVID-19 revealed a cluster of upregulated mediators, including IL-1β, IL-2, IL-25, IFN-α2, CCL7, CCL11, CX3CL1, TGF-α, and TNF-α. This pattern corroborates the activation of neuroimmune responses, both innate and adaptive responses, which was supported by results in functional protein enrichment analysis, which indicated microglial activation, recruitment of peripheral immune cells, including T-cells, monocytes/macrophages, and NK cells, disruption of the BBB, and tissue repair. Upregulation of CX3CL1 and TGF-α in this CNS response axis also highlights neuron-glia interactions. Neurons represent a major source of CX3CL1 in the CNS, leading to microglial activation22, and also secrete TGF-α, which is involved in tissue remodeling and gliosis23, therefore potentially influencing the balance between neuronal regeneration and neurotoxicity. However, it is not possible to determine whether this resulted from a common event triggering CNS response or from intricate regulatory pathways activated by the neurological insult.
The serum of patients with neuro-COVID-19 exhibited an overall increase in IL-13, IL-18, TNF-α, TGF-α, VEGF, and VILIP-1 levels. VILIP-1 is a calcium-sensor protein primarily expressed in the brain that is involved in regulating neuronal growth, survival, and synaptic plasticity; its release into the extracellular space due to disturbed calcium homeostasis has led to its use as a biomarker of neuronal injury24,25. However, VILIP-1 expression is also expressed in the heart, liver, kidney, and lung26, and CSF and serum VILIP-1 levels correlate weakly25. VILIP-1 was significantly increased in the serum of COVID-19 patients without corresponding changes in CSF, suggesting that elevated serum VILIP-1 was linked to systemic inflammation rather than CNS injury. In contrast, BDNF, another factor regulating neuronal homeostasis, was reduced in the serum of patients with encephalopathy compared to patients with isolated refractory headache and IND. However, BDNF and its receptor are also expressed by lung cells, and it is abundant in peripheral blood and can be secreted by lymphocytes and monocytes27. In severe/moderate COVID-19, patients present a transient reduction in serum BDNF levels compared to individuals with mild disease, which are restored during recovery28. Although consecutive serum samples were not available in our study to assess BDNF kinetics, lower serum BDNF levels in patients with encephalopathy may reflect differences in respiratory involvement rather than neurological manifestations.
Neuro-COVID-19 patients displayed elevated blood levels of TGF-α and VEGF, and high CSF levels of GM-CSF. Although VEGF has angiogenic and protective effects on endothelial cells, it can promote BBB leakage by transiently affecting cell–cell adhesion29,30. GM-CSF also influences the BBB disruption by stimulating the expression of proinflammatory cytokines, such as IL-6 and TNF-α, that downregulate adhesion molecules in cerebral microvasculature31. Thus, both CSF and blood factors may contribute to neurological alterations in COVID-19 by inducing endothelial activation, increased vascular permeability, and BBB disruption.
Patients with IND and encephalopathy had elevated CSF neopterin, in agreement with the literature32,33, and half of these patients had a neopterin (CSF/serum) ratio > 1.0 [see the Additional file 3], indicating active CNS production rather than peripheral diffusion. Neopterin is secreted by activated monocytes/macrophages in response to IFN-γ34 and is a biomarker of immune activation, and associated with CNS disturbances triggered by infections, autoimmune disorders, and primary CNS lymphoma35,36. High CSF neopterin in IND patients corroborated the mononuclear pleocytosis, whereas patients with encephalopathy did not exhibit CNS infiltration by immune cells. Resident CNS cells, such as microglia and astrocytes, can also secrete neopterin in response to IFN-γ37,38. Edén et al.32 reported that CSF neopterin and IFN-γ levels correlated with SARS-CoV-2 nucleocapsid antigen concentrations, which was detected even in the absence of viral RNA. In our study population, encephalopathy and IND were associated with neuronal injury and elevated levels of proinflammatory factors that can disturb the BBB6,14. Therefore, systemic inflammation in COVID-19 and/or the presence of SARS-CoV-2 antigens in CNS likely influenced the development of encephalopathy and IND, given that active viral replication in the CNS was not detected. However, we were not able to assess SARS-CoV-2 nucleocapsid protein levels in CSF and serum samples to confirm this.
Patients with encephalopathy and IND had elevated CSF levels of HMGB1 and IL-18, in addition to IL-1β in cases of encephalopathy. This supports the involvement of inflammasomes in COVID-19-associated neuroinflammation. HMGB1 can be passively released after cell death or actively secreted by stimulated cells upon triggering of NLRP3 inflammasomes, which finally results in IL-1β and IL-18 secretion39,40. Although the exact source of elevated HMGB1 in the CSF of these patients cannot be specified, it is likely that neuronal damage has contributed to its increase. Patients with IND had elevated CSF NfL—a marker of injury to large myelinated axons—especially in cases with meningeal involvement6. Moreover, HMGB1 mediates acute and chronic inflammation by recruiting cells to sites of injury and infection and stimulating macrophages and endothelial cells to produce cytokines, particularly IL-1β, IL-6, TNF-α, and type I IFNs40,41, which were elevated in the CSF of patients with encephalopathy and IND. Patients with IND also had higher levels of IL-2, IL-6, TNF-α, CXCL8, and CXCL10 in the CSF than in serum, indicating that neuroinflammation was driven by CNS-intrinsic events rather than a secondary response to systemic inflammation. In an experimental coronavirus neuroinfection model, microglia were identified as the main source of the CNS cytokine storm. Additionally, type I astrocytes can also contribute to neuroinflammation by upregulating IL-1α/β, IL-2, IL-7, IL-13, IL-15, IL-17, IL-18, and IFNs42,43. Collectively, these findings indicate that CNS resident cells such as microglia and astrocytes play a major role in the development of acute neurological syndromes in COVID‑19, while infiltration by peripheral mononuclear immune cells exacerbates neuroinflammation. Results from functional protein network analysis also support this by indicating the enrichment of processes associated with microglial functions and chemotaxis, proliferation, activation, and cytokine production in various peripheral blood mononuclear cell types. Similar to findings in IND, individuals with encephalopathy exhibited a marked upregulation of multiple proinflammatory mediators, indicative of a cytokine storm and neuroinflammation. However, the lack of signs of acute brain inflammation, such as CSF pleocytosis and changes in brain magnetic resonance imaging (MRI), suggests a temporal sequence in which neurological manifestations arise from CNS response to preceding systemic inflammation, rather than from primary CNS involvement, which defined the IND.
Many factors upregulated in the CSF in encephalopathy and IND cases were associated with Th1 responses, which is typically induced by viral infections. However, the enrichment of factors associated with Th2 responses was also seen. In isolated headache and encephalopathy, IL-25, an anti-inflammatory cytokine of the IL-17 family, was elevated. In the CNS, IL-25 helps to prevent inflammatory cell infiltration and modulates BBB repair, and regulates Th2 cytokine production, as shown in murine models44,45. This corroborates the self-limited neuroinflammation observed in patients in our study, who experienced spontaneous recovery or symptom resolution after corticotherapy or intravenous immunoglobulin6, as also reported elsewhere2,3,32. Additionally, the CSF of patients with encephalopathy and IND had increased β-NGF, a growth factor promoting neuronal survival and plasticity46, and encephalopathy patients also had elevated EGF, a factor supporting differentiation, maturation, and survival of multiple cell types, including neurons47, further indicating ongoing neural recovery responses.
However, our findings may differ in the context of current SARS-CoV-2 variants or vaccinated populations, since study participants were enrolled in 2020, before the emergence of Omicron and COVID-19 vaccination. Omicron variant has been shown to be more transmissible and infectious than previous SARS-CoV-2 variants but causing milder unspecific and respiratory symptoms48. In contrast, Omicron can also trigger new‐onset neurological conditions or worsen pre‐existing neurological diseases, and para-infectious cases of neuroimmune complications, including Guillain-Barré syndrome, spinal meningitis/myelitis, neuromyelitis optica, ADEM, autoimmune encephalitis, and encephalopathy have been described yet49,50,51. Unfortunately, changes in the prevalence of acute neurological complications associated with Omicron and pre-Omicron variants are still unknown. However, the prevalence of post-infectious conditions such as long COVID showed a reduction after infection with Omicron lineages13. In addition, full vaccination has been also related to a reduced risk for cerebrovascular diseases52. Indeed, the humoral response (IgG and IgA) induced by a second booster dose of COVID-19 vaccines showed a protective effect on neurological symptoms, such as brain fog, sleep quality, impaired coordination, and physical pain53, and at least one vaccine dose had a protective effect against long COVID54. Protection against neurological manifestations conferred by immunization has been associated with short-term and long-term suppression of cytokine storm responses upon SARS-CoV-2 infection, with lower levels of inflammatory factors such as IL-2RA, IL-7, IL-8, IL-15, IL-29/IFN-λ, IP-10/CXCL10, CCL2/MCP-1, and TNF-α compared to unvaccinated counterparts55. This highlights the importance of ongoing immunization programs in reducing the frequency and severity of neurological manifestations in COVID-19.
This study had limitations, including a small sample size, the lack of consecutive CSF and serum samples, and limited follow-up after discharge. Our study was also not sufficiently powered to formally adjust for confounding variables such as hypertension, diabetes, and cardiovascular disorders. These pre-existing conditions contribute to systemic inflammation, endothelial dysfunction, and the compromise of BBB, predisposing individuals to severe neurological manifestations in COVID-19 by exacerbating the cytokine storm and immune-mediated damage within the CNS2,56. The exploratory design also limited the evaluation of biomarkers with potential prognostic value. However, strengths include a well-characterized cohort, comprehensive investigation of multiple factors related to inflammation, neuronal damage, and CNS homeostasis in paired CSF and serum samples, evaluation of a diverse range of neurological outcomes, and inclusion of non-hospitalized patients with mild COVID-19 as well as uninfected controls. Consequently, our data allowed us to identify mechanisms likely associated with distinct neurological manifestations in acute COVID-19.
Conclusions
In summary, changes in the CSF and blood during the acute phase of COVID-19 (within 14 days from symptom onset) in patients with neurological manifestations were diverse. However, analysis of multiple factors and functional networks analysis identified common mechanisms, at least in patients with encephalopathy and IND, primarily involving the activation, regulation, and function of glial cells, CNS cytokine storm, immune cell responses, inflammasome activity, and probable disturbances of BBB integrity.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Azim, D., Nasim, S., Kumar, S., Hussain, A. & Patel, S. Neurological consequences of 2019-nCoV infection: A comprehensive literature review. Cureus 12, e8790 (2020).
Ellul, M. A. et al. Neurological associations of COVID-19. Lancet Neurol. 19, 767–783 (2020).
Garg, R. K., Paliwal, V. K. & Gupta, A. Encephalopathy in patients with COVID-19: A review. J. Med. Virol. https://doi.org/10.1002/jmv.26207 (2020).
Helms, J. et al. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 382, 2268–2270 (2020).
Mao, L. et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. https://doi.org/10.1001/jamaneurol.2020.1127 (2020).
Espíndola, O. M. et al. Cerebrospinal fluid findings in neurological diseases associated with COVID-19 and insights into mechanisms of disease development. Int. J. Infect. Dis. 102, 155–162 (2021).
Sobczak, M. & Pawliczak, R. COVID-19 vaccination efficacy in numbers including SARS-CoV-2 variants and age comparison: A meta-analysis of randomized clinical trials. Ann. Clin. Microbiol. Antimicrob. 21, 32 (2022).
Steele, M. K. et al. Estimated Number of COVID-19 infections, hospitalizations, and deaths prevented among vaccinated persons in the US, December 2020 to September 2021. JAMA Netw. Open 5, e2220385 (2022).
Thaweethai, T. et al. Development of a definition of postacute sequelae of SARS-CoV-2 infection. JAMA 329, 1934–1946 (2023).
Vargas, G. et al. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and glial cells: Insights and perspectives. Brain, Behav. Immun. Health 7, 100127 (2020).
Sha, A. & Chen, H. Infection routes, invasion mechanisms, and drug inhibition pathways of human coronaviruses on the nervous system. Front. Neurosci. 17, 1169740 (2023).
Pezzini, A. & Padovani, A. Lifting the mask on neurological manifestations of COVID-19. Nat. Rev. Neurol. 16, 636–644 (2020).
Fernández-de-Las-Peñas, C. et al. Long-COVID symptoms in individuals infected with different SARS-CoV-2 variants of concern: A systematic review of the literature. Viruses 14, 2629 (2022).
Espíndola, O. M. et al. Inflammatory cytokine patterns Associated with neurological diseases in coronavirus disease 2019. Ann. Neurol. 89, 1041–1045 (2021).
Silva, M. T. T. et al. Isolated intracranial hypertension associated with COVID-19. Cephalalgia 40, 1452–1458 (2020).
Venkatesan, A. et al. Case definitions, diagnostic algorithms, and priorities in encephalitis: Consensus statement of the international encephalitis consortium. Clin. Infect. Dis. 57, 1114–1128 (2013).
Jardim-Santos, G. P. et al. Unbalanced networks and disturbed kinetics of serum soluble mediators associated with distinct disease outcomes in severe COVID-19 patients. Front Immunol 13, 1004023 (2022).
de Espíndola, O. M. et al. Patients with COVID-19 and neurological manifestations show undetectable SARS-CoV-2 RNA levels in the cerebrospinal fluid. Int. J. Infect. Dis. 96, 567–569 (2020).
Puelles, V. G. et al. Multiorgan and renal tropism of SARS-CoV-2. N. Engl. J. Med. 383, 590–592 (2020).
Solomon, I. H. et al. Neuropathological features of Covid-19. N. Engl. J. Med. 383, 989–992 (2020).
Pilotto, A. et al. Steroid-responsive encephalitis in coronavirus disease 2019. Ann. Neurol. https://doi.org/10.1002/ana.25783 (2020).
Luo, P., Chu, S.-F., Zhang, Z., Xia, C.-Y. & Chen, N.-H. Fractalkine/CX3CR1 is involved in the cross-talk between neuron and glia in neurological diseases. Brain Res. Bull. 146, 12–21 (2019).
Rabchevsky, A. G. et al. A role for transforming growth factor alpha as an inducer of astrogliosis. J. Neurosci. 18, 10541–10552 (1998).
Schnurra, I., Bernstein, H. G., Riederer, P. & Braunewell, K. H. The neuronal calcium sensor protein VILIP-1 is associated with amyloid plaques and extracellular tangles in Alzheimer’s disease and promotes cell death and tau phosphorylation in vitro: A link between calcium sensors and Alzheimer’s disease?. Neurobiol. Dis. 8, 900–909 (2001).
Halbgebauer, S. et al. Visinin-like protein 1 levels in blood and CSF as emerging markers for Alzheimer’s and other neurodegenerative diseases. Alzheimers Res. Ther. 14, 175 (2022).
Gierke, P. et al. Expression analysis of members of the neuronal calcium sensor protein family: Combining bioinformatics and Western blot analysis. Biochem. Biophys. Res. Commun. 323, 38–43 (2004).
Azoulay, D., Urshansky, N. & Karni, A. Low and dysregulated BDNF secretion from immune cells of MS patients is related to reduced neuroprotection. J. Neuroimmunol. 195, 186–193 (2008).
Azoulay, D. et al. Recovery from SARS-CoV-2 infection is associated with serum BDNF restoration. J. Infect. 81, e79–e81 (2020).
Wang, W., Dentler, W. L. & Borchardt, R. T. VEGF increases BMEC monolayer permeability by affecting occludin expression and tight junction assembly. Am. J. Physiol. Heart Circ. Physiol. 280, H434-440 (2001).
Gavard, J. & Gutkind, J. S. VEGF controls endothelial-cell permeability by promoting the beta-arrestin-dependent endocytosis of VE-cadherin. Nat. Cell Biol. 8, 1223–1234 (2006).
Lotfi, N. et al. Roles of GM-CSF in the pathogenesis of autoimmune diseases: An update. Front. Immunol. 10, 1265 (2019).
Edén, A. et al. Viral antigen and inflammatory biomarkers in cerebrospinal fluid in patients With COVID-19 infection and neurologic symptoms compared with control participants without infection or neurologic symptoms. JAMA Netw. Open 5, e2213253 (2022).
Chaumont, H. et al. Cerebrospinal fluid biomarkers in SARS-CoV-2 patients with acute neurological syndromes. Rev. Neurol. (Paris) 179, 208–217 (2023).
Hoffmann, G., Wirleitner, B. & Fuchs, D. Potential role of immune system activation-associated production of neopterin derivatives in humans. Inflamm. Res. 52, 313–321 (2003).
Eisenhut, M. Neopterin in diagnosis and monitoring of infectious diseases. J. Biomark. 2013, 1–10 (2013).
Viaccoz, A. et al. CSF neopterin level as a diagnostic marker in primary central nervous system lymphoma. Neuro Oncol. 17, 1497–1503 (2015).
Ghisoni, K., de Martins, R. P., Barbeito, L. & Latini, A. Neopterin as a potential cytoprotective brain molecule. J. Psychiatr. Res. 71, 134–139 (2015).
de Paula Martins, R. et al. Neopterin preconditioning prevents inflammasome activation in mammalian astrocytes. Free Radic. Biol. Med. 115, 371–382 (2018).
Vande Walle, L., Kanneganti, T.-D. & Lamkanfi, M. HMGB1 release by inflammasomes. Virulence 2, 162–165 (2011).
Yang, H., Wang, H. & Andersson, U. Targeting inflammation driven by HMGB1. Front. Immunol. 11, 484 (2020).
Ren, W., Zhao, L., Sun, Y., Wang, X. & Shi, X. HMGB1 and Toll-like receptors: Potential therapeutic targets in autoimmune diseases. Mol. Med. 29, 117 (2023).
Savarin, C. & Bergmann, C. C. Fine tuning the cytokine storm by IFN and IL-10 following neurotropic coronavirus encephalomyelitis. Front. Immunol. 9, 3022 (2018).
Lavi, E. & Cong, L. Type I astrocytes and microglia induce a cytokine response in an encephalitic murine coronavirus infection. Exp. Mol. Pathol. 115, 104474 (2020).
Kleinschek, M. A. et al. IL-25 regulates Th17 function in autoimmune inflammation. J. Exp. Med. 204, 161–170 (2007).
Sonobe, Y. et al. Interleukin-25 expressed by brain capillary endothelial cells maintains blood-brain barrier function in a protein kinase Cepsilon-dependent manner. J. Biol. Chem. 284, 31834–31842 (2009).
Aloe, L., Rocco, M. L., Bianchi, P. & Manni, L. Nerve growth factor: From the early discoveries to the potential clinical use. J. Transl. Med. 10, 239 (2012).
Wong, R. W. C. & Guillaud, L. The role of epidermal growth factor and its receptors in mammalian CNS. Cytokine Growth Fact. Rev. 15, 147–156 (2004).
Fan, Y. et al. SARS-CoV-2 Omicron variant: Recent progress and future perspectives. Signal Transduct. Target Ther. 7, 141 (2022).
Chang, T. et al. Neurological disorders associated with COVID-19 in Sri Lanka. BMC Neurol. 23, 351 (2023).
Gong, S. et al. Clinical and immunological features in patients with neuroimmune complications of COVID-19 during Omicron wave in China: A case series. Front. Immunol. 15, 1499082 (2024).
Lu, L. et al. Neurological complications during the omicron COVID-19 wave in China: A cohort study. Eur. J. Neurol. 31, e16096 (2024).
Chen, S.-Y. et al. Prior COVID-19 vaccination and reduced risk of cerebrovascular diseases among COVID-19 survivors. J. Med. Virol. 96, e29648 (2024).
Liu, M. et al. Immune-boosting effect of the COVID-19 vaccine: Real-world bidirectional cohort study. JMIR Public Health Surveill. 9, e47272 (2023).
Ceban, F. et al. COVID-19 vaccination for the prevention and treatment of long COVID: A systematic review and meta-analysis. Brain Behav. Immun. 111, 211–229 (2023).
Zhu, X. et al. Dynamics of inflammatory responses after SARS-CoV-2 infection by vaccination status in the USA: A prospective cohort study. Lancet Microbe 4, e692–e703 (2023).
Gupta, A. et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 26, 1017–1032 (2020).
Acknowledgements
We thank the technical team of the Laboratório Neurolife for performing CSF and paired serum laboratory analyses, and Dr. Eric Henrique Roma (Laboratório de Imunologia e Imunogenética em Doenças Infecciosas of the Instituto Nacional de Infectologia Evandro Chagas—FIOCRUZ) for the assistance with the MAGPIX equipment.
Funding
This study was supported by the INOVA FIOCRUZ program of the Fundação Oswaldo Cruz (Grant VPPCB-005-FIO-20–2-65 to M.T.T.S.), by the Carlos Chagas Foundation for the Advancement of Science of the State of Rio de Janeiro (FAPERJ) (Grant E-26/200.157/2023 to O.M.E.), by scholarships granted to N.F.L., Y.C.P.G, and J.V.C.D by the Coordination of Superior Level Staff Improvement (CAPES) of the Brazilian Ministry of Education (Finance code 001), and by scholarship granted to K.S. by the Brazilian National Council for Scientific and Technological Development (CNPq).
Author information
Authors and Affiliations
Contributions
N.L.F. performed cytometry bead-based multiplex assays with assistance from R.C.T., conducted ELISA, data analysis, and drafted the manuscript; J.V.C.D. carried out functional network analysis; K.S., performed Luminex assays; Y.C.P.G. performed data analysis; C.O.B. performed CSF and paired serum laboratory analysis; C.N.S. and M.T.T.S. acquired clinical data and conducted neurological assessments; N.F.L., J.V.C.D., and O.M.E assembled the figures; O.M.E. and M.T.T.S contributed to the study concept and design; O.M.E performed data analysis, supervised all phases of the project, and drafted the manuscript. All authors contributed to interpretation of the results, read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study protocol was approved by the Brazilian National Committee of Ethics in Research on April 22, 2020 (Protocol number CAAE 30611720.6.0000.5262), and written informed consent was obtained from all participants. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Freitas, N.L., Deus, J.V.C., Sampaio, K. et al. Central nervous system and systemic inflammatory networks associated with acute neurological outcomes in COVID-19. Sci Rep 15, 24154 (2025). https://doi.org/10.1038/s41598-025-08632-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-08632-9