Abstract
High-fat diets (HFDs) are widely used in aquaculture but may adversely affect fish liver health by disturbing lipid metabolism and promoting oxidative stress. This study investigated the protective effects of Nannochloropsis oculata enriched with astaxanthin on zebrafish (Danio rerio) fed an HFD. All zebrafish (n = 135) were divided randomly into three groups: conventional diet (CD), high-fat diet (HFD), and HFD supplemented with 2% N. oculata (HFD + NO). After a four-week feeding trial, growth performance, hepatosomatic index, liver oxidative stress markers, and expression of immune cytokine genes were evaluated. HFD group exhibited significantly increased hepatic lipid accumulation, elevated serum triglycerides and total cholesterol, and increased liver marker enzymes. These changes were accompanied by reduced antioxidant enzyme and non-enzymatic antioxidant activity, heightened oxidative stress, and upregulated pro-inflammatory cytokine expression (il-1β and il-8). Histological analysis confirmed hepatic steatosis and structural deterioration. Conversely, N. oculata supplementation significantly alleviated these effects by reducing hepatic lipid deposition, restoring antioxidant defenses, and downregulating pro-inflammatory cytokines while upregulating anti-inflammatory responses (IL-10 and TGF-β). These protective effects are attributed to astaxanthin’s role in boosting mitochondrial β-oxidation, suppressing lipogenesis, and modulating inflammatory pathways. These findings suggest that N. oculata is a promising functional feed additive for mitigating diet-induced liver dysfunctions in aquaculture. Further research is needed to elucidate its molecular mechanisms and assess its long-term benefits across diverse aquatic environments.
Similar content being viewed by others
Introduction
Fish products constitute the half of global aquaculture production, highlighting their critical role in this industry1. The use of high-fat diets (HFDs) in aquaculture has increased significantly in recent years. HFDs are increasingly used in aquaculture to enhance energy density, reduce reliance on protein sources (which are often more expensive), and improve feed conversion efficiency. Lipids also play a vital role in membrane integrity, hormone synthesis, and overall fish growth and health2. Fats are widely regarded as a cost-effective energy source among macronutrients. They provide essential fatty acids, phospholipids, sterols, and fat-soluble vitamins necessary for the normal growth, development, and metabolic processes of fish3. Increasing dietary fat within an optimal range can exert a protein-sparing effect by reducing the use of protein as an energy source, thereby making it more available for growth4. As a result, high-fat diets are frequently used in contemporary aquaculture to preserve expensive and limited protein sources. Fish growth is encouraged, feed efficiency is increased, and feed costs are decreased with this method5. However, these diets are associated with numerous adverse effects on fish health6. Excessive dietary fat leads to abnormal fat accumulation, particularly in the liver, resulting in fatty liver disease7. This accumulation also elevates reactive oxygen species (ROS) levels, triggering oxidative stress and programmed cell death, including apoptosis and ferroptosis in liver cells8,9,10. Furthermore, chronic liver inflammation due to excessive fat intake has been documented across various fish species, compromising overall fish health11,12,13. These detrimental effects of HFDs pose significant challenges to aquaculture development, necessitating urgent solutions.
Astaxanthin, a red ketocarotenoid with potent antioxidant properties, has garnered significant interest in aquaculture for its dual role in enhancing pigmentation and providing robust protection against stress in aquatic animals14. Research demonstrates that astaxanthin improves growth performance, immune responses, stress tolerance, and disease resistance5,15. Furthermore, recent studies highlight its protective role in regulating lipid metabolism in fish fed with HFDs3,5. Studies indicate that astaxanthin derived from microalgae exhibits superior antioxidant and immunomodulatory effects compared to synthetic alternatives16,17. As a result, microalgal astaxanthin offers significant advantages and serves as an effective feed additive for aquatic animals. Nannochloropsis, a genus of unicellular microalgae with a polysaccharide-based cell wall and one chloroplast, is widely recognized for its applications in aquaculture18. Notably, Nannochloropsis oculata (Eustigmatophyceae), a marine unicellular microalga, is widely recognized for its high content of polyunsaturated fatty acids (PUFAs), particularly eicosapentaenoic acid (EPA), as well as astaxanthin, carotenoids, and antioxidant compounds. Due to these bioactive properties, it is increasingly studied as a functional feed additive in aquaculture to improve lipid metabolism, immunity, and oxidative status in fish19,20.
While previous studies have shown that astaxanthin supplementation in fish can mitigate inflammatory and immune responses3,5, the molecular mechanisms by which microalgal astaxanthin regulates lipid metabolism, liver lipid profiles, and liver inflammation remain unclear. This study aimed to assess the protective effects of N. oculata enriched with astaxanthin in zebrafish (Danio rerio) fed a high-fat diet. Specifically, it examined its impact on liver and serum lipid profiles, oxidative stress reduction, and the expression of cytokine genes associated with liver inflammation.
Materials and methods
Cultivating microalgae
The microalga N. oculata was obtained from an algae cultivation center in Shahrood, Iran, and subsequently purified and stored at the Fisheries Laboratory, Razi Laboratory Complex, Science and Research Branch, Islamic Azad University. The cultivation was conducted in a modified BG-11 medium, composed of 36 mg CaCl₂·2 H₂O, 75 mg MgSO₄·7 H₂O, 20 mg Na₂CO₃, 40 mg K₂HPO₄, 3.15 mg FeCl₃·2 H₂O, 6.0 mg citric acid, 4.36 mg citric acid·2 H₂O, and 1 mL A5 mix per liter. The A5 mix, prepared per liter, contained 1.81 g MnCl₂·4 H₂O, 79 mg CuSO₄·5 H₂O, 2.86 g H₃BO₃, 222 mg ZnSO₄·7 H₂O, 0.39 g NaMoO₄·2 H₂O, 49.4 mg Co(NO₂O₃)₂, and 1 mL of 98% H₂SO₄3. Cultivation was carried out in 17 L plastic bottles containing 15 L of microalgal culture with continuous aeration. The culture was maintained at 24 ± 3 °C under fluorescent lighting, providing a constant intensity of 100 µmol/m²/sec. A fully automated, computer-controlled vertical photobioreactor with a 4000 L capacity was used to transfer the culture after 10 days. There, carbon dioxide was injected as a carbon source. The biomass increased until it reached 2–2.5 g/L. Algal biomass was harvested by centrifugation at 896 × g for 15 min using a basket centrifuge, washed twice with water, oven-dried at 50 °C, ground into a homogeneous powder, and stored in a refrigerator for further chemical and biological analysis. According to spectrophotometry21, the resultant microalgal powder had 1.92% astaxanthin, as well as 11.04% crude fat and 39.72% crude protein.
Animals and creating an experimental diet
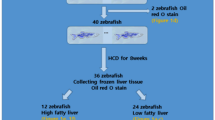
Wild-type, 4–5-month-old male short-finned zebrafish (Danio rerio; 0.40 ± 0.03 g) were obtained from a local supplier in Tehran, Iran, and housed in 40 L tanks at a density of up to 2 fish/L in non-chlorinated water. To prevent hormonal interference and aberrant fat accumulation, only male fish were used in the experiments22. Sex was determined based on morphological characteristics, such as body shape and coloration, and confirmed via gonadal examination after euthanasia with MS222. The fish were assigned an overdose of MS-222 (250 mg/L) to be euthanized23. In this regard, MS222 powder was diluted in 1 L of water, and the pH was maintained at 7.0-7.2 by adding sodium bicarbonate before introducing the fish. The fish were kept in the euthanasia solution until the opercular movement was stopped. The water temperature was maintained at 26 ± 3 °C, with a pH of 7.1–7.3. Fish were kept under a natural photoperiod (~ 12/12 h light/dark cycle) and fed twice daily with the basal diet (diet details in Table 1). Following a two-week acclimation period, the zebrafish were assigned randomly to 5.2 L tanks containing dechlorinated water (15 fish in each tank, 3 tanks per group) and grouped according to dietary regimen. Three experimental diets were used: a conventional diet (CD), a high-fat diet (HFD), and an HFD supplemented with 2% N. oculate (HFD + NO). The dosage of N. oculata was determined based on prior research3. Studies have demonstrated that a lipid content of 13% induces significant liver fat accumulation in zebrafish6,24. Accordingly, the lipid levels in the CD and HFD were set at 7% and 13.1%, respectively6,24 (Table 1). The pellet diet was prepared using the techniques previously described. Briefly, all dry ingredients (Table 1) were thoroughly mixed in a blender. Water was gradually added to achieve a dough-like consistency. It was then passed through a meat grinder to form strands, which were cut into small pellets (2–3 mm). The pellets were oven-dried at 50 °C for 24 h until a constant weight was achieved. After cooling to room temperature, the pellets were stored at -20 °C until use25. The fish in the experiment were fed for four weeks.
Sample collections
Following the feeding trial, fish from each tank were anesthetized with a clove oil bath (50 µL/L)26. Individual weights and the number of live fish were recorded. Blood was collected from the severed tails, and serum samples were obtained through centrifugation at 4 °C, 400 ×g, for 10 min. Twelve fish per treatment group were selected randomly; liver tissue from nine fish was immediately frozen in liquid nitrogen and stored at -80 °C, while tissue from the remaining three fish was preserved in Bowen’s solution.
Fish growth and hepato-somatic index
The growth pattern of the fish was evaluated morphometrically, including measurements of feed conversion ratio (FCR) and specific growth rate (SGR). The condition factors (K) were calculated using the formula:
FCR and SGR were determined using the following equations:
Feed conversion ratio (FCR) = Dry food fed (g)/Weight gained (g).
Specific growth rate (SGR, %/day) = log(Final body weight) − log(Initial body weight)/Time (days)×100.
Following dissection, liver moisture was removed using blotting paper, and liver weights were recorded. The hepato-somatic index (HSI) was calculated as the percentage of liver weight relative to body weight:
Hepato-somatic Index (HSI, %) = Liver weight (g)×100/Body weight (g).
Measurements of liver oxidative stress
To assess oxidative stress indices in liver tissues, samples were homogenized (Teflon-glass Potter-Elvehjem homogenizer (Sigma-Aldrich, USA)) in PBS (pH 7.4) at a 1:8 (w/w) ratio rather than the conventional 1:10 to ensure sufficient protein concentration (2.0–2.5 mg/mL) for robust measurement of antioxidant enzyme activities within the linear detection range of the ZellBio assay kits. Finally, the sample was centrifuged at 10,000 ×g for 10 min at 4 °C, and the resulting supernatant was used for subsequent assays. Protein concentration was determined using the Bradford method with bovine serum albumin as the standard27. All assays were conducted following standard protocols provided by ZellBio GmbH (Veltinerweg, Germany). Superoxide dismutase (ZB-SOD-96 A) and catalase (ZB-CAT-96 A) activities were measured using the method of28. Glutathione peroxidase (ZB-GPX-96 A) activity was quantified using a modified method with 5,5′-dithiobis-2-nitrobenzoic acid (DTNB)29. Glutathione S-transferase (ZB-GST-96 A) activity was assessed using 1-chloro-2,4-dinitrobenzene30. Glutathione (ZB-GSH-96 A) and oxidized glutathione (ZB-GSSG-96 A) levels were determined through the cyclic reaction of GSH with DTNB31,32, while glutathione reductase (GR) activity was evaluated by monitoring NADPH reduction33. Lipid peroxidation (LPO) was quantified by measuring malondialdehyde (MDA) levels using the thiobarbituric acid method34. Briefly, immediately after homogenization, a 200 µL aliquot of the crude liver homogenate was removed and mixed with TBA reagent. Samples were heated (95 °C for 30 min), cooled on ice, and then centrifuged (10,000 × g for 10 min) to pellet proteins. MDA–TBA complexes in the supernatant were quantified at 532 nm.
Lipid profiles in serum and liver enzymological indexes
The levels of total triglycerides (TG, 10120) and total cholesterol (TC, 10110) were measured following the guidelines provided by the ParsAzemun Co. assay kits (Tehran, Iran). Biochemical medical diagnostic kits (ParsAzemun Co., Tehran, Iran) were used to measure the levels of aspartate aminotransferase (AST, 12010), alanine aminotransferase (ALT, 12020), gamma-glutamyltransferase (GGT, 12130), alkaline phosphatase (ALP, 12100), and lactate dehydrogenase (LDH, 12300).
Hepatic lipid assay
Using the Folch method, lipids were extracted from the livers35. The sulfo-phospho-vanillin reaction was used to quantify total lipids36. Triglyceride, fatty acid, and total cholesterol levels were measured using an automatic biochemical analyzer (Tokyo Boeki Co., Japan, Prestige 24i) and commercial kits (Pars Azmoon Co., Tehran, Iran).
Liver histological analysis
The fixed liver tissue underwent standard histological preparation. After washing, the tissue was dehydrated through a graded ethanol series, cleared with xylene, and embedded in paraffin wax. Sections were cut into 5 μm slices using a microtome and mounted onto slides. The slides were stained with hematoxylin-eosin and examined under an optical microscope (E100, Nikon, Tokyo, Japan).
Expression of immune cytokine genes
RNA extraction and cDNA synthesis
Using the Sinaclon RNA Isolation Kit (EX6101, Sinaclon, Iran), liver tissue was treated to extract total RNA (20 mg per tissue sample) following the manufacturer’s instructions. A NanoDrop™ Lite plus spectrophotometer (NDLPLUSPRGL, Thermo Scientific™, USA) was used to measure the concentration and quality of the purified RNA at an absorbance ratio of 260/280 nm. After removing any remaining genomic DNA with DNase I, 1 µg of the RNA was reverse transcribed into cDNA using the First Strand cDNA Synthesis Kit (EX6101, Sinaclon, Iran). Parallel processing was done for negative controls that did not contain either RNA or enzyme. For subsequent analysis, the produced cDNA was stored at -80 °C after being diluted to a 1:10 ratio.
Quantitative real-time PCR (qPCR)
The LightCycler 480 system (Roche Diagnostics, Basel, Switzerland) was applied to measure the expression levels of the target genes il-1β, il-8, il-10, tgf-α, and tgf-β (Table 2). Each 10 µL amplification reaction consisted of 5 µL LightCycler 480 SYBR Green I Master (Roche Diagnostics), 2 µL PCR-grade water, 2 µL of 1:10 diluted cDNA, and 0.5 µL of each primer (500 nM final concentration). A variety of responses, including identical responses and a group without a specific template, were executed. The qPCR method consisted of an initial activation of enzymes at 95 °C for 5 min, followed by 40 cycles of heating at 95 °C for 10 s, annealing at the temperature specific to the primers for 10 s, and extension at 72 °C for 15 s. The second derivative maximum method was used to calculate the quantification cycle (Cq) values using LightCycler 480 software version 1.5. (Roche Diagnostics). Melting curve analysis and agarose gel electrophoresis were used to confirm the specificity of the PCR products. Pooled cDNA was serially diluted ten times to determine reaction efficiencies. Pooled cDNA was serially diluted ten times to determine reaction efficiencies37. The normalization factor was the geometric mean of the expression of β-act and gapdh. Three biological and three technical replicates were used in every RT-qPCR experiment.
Statistical analysis
The statistical analysis of the data was conducted using SPSS software, version 27 (SPSS Inc., Chicago, IL, USA). To identify significant differences between groups, a one-way analysis of variance (ANOVA) was performed, and post-hoc Tukey’s multiple range tests were performed. The significance level was set at P < 0.05.
Ethics approval
All experimental protocols were reported following ARRIVE guidelines and were approved by the Animal Ethics Committee of Islamic Azad University, Science and Research Branch, Tehran, Iran (Local ethical reference: IR.IAU.SRB.REC.1403.023). Also, all methods were carried out following the relevant guidelines and regulations.
Results
Fish growth and hepato-somatic index
The results indicated that the HFD + NO group exhibited the greatest increases in final weight, weight gain, specific growth rate (SGR), and hepatosomatic index (HSI) compared to the other test groups (P < 0.05; Table 3). However, no significant differences in FCR or HSI were observed between the CD and HFD + NO groups (P > 0.05; Table 3).
Liver oxidative stress indexes
A significant reduction in the activity of SOD, CAT, GPx, and GST enzymes was observed in the HFD group compared to the CD group (P < 0.05) (Table 4). However, fish in the HFD + NO group exhibited a marked increase in the activity of these enzymes compared to the HFD group (P < 0.05) (Table 4). A similar pattern was noted for the levels of GSH, GSSG, and GR across the experimental groups. In contrast, MDA levels were significantly higher in the HFD group compared to all other groups, while the HFD + NO group displayed significantly lower MDA levels than the HFD group (P < 0.05) (Table 4).
Serum and hepatic lipid and liver enzymological indexes
Comparison of liver enzyme indices and lipid profiles revealed that the HFD diet significantly elevated ALT, AST, and ALP levels, as well as CGT, LDH, TG, and TC levels in serum, and TL, TC, FC, and TG levels in the zebrafish liver, compared to the CD group (P < 0.05) (Table 5). However, these parameters were significantly reduced in the HFD + NO group compared to the HFD group (P < 0.05) (Table 5).
Cytokine gene expression
Figure 1 presents the results of cytokine gene expression analysis. The HFD diet significantly upregulated il-β, il-8, and tnf-α expression while downregulating il-10 and tgf-β expression in the zebrafish liver (P < 0.05). In the HFD + NO group, IL1β and IL-8 expression levels were significantly reduced compared to the HFD group (P < 0.05).
Cytokine gene expression in liver of zebrafish fed diets with a conventional diet (CD), a high-fat diet (HFD), and an HFD supplemented with 2% Nannochloropsis oculate (HFD + NO) for 4 weeks (means ± SD). The letters of the difference in each group indicate significance. P < 0.05 was considered as significant.
Liver histology
Figure 2 shows the results of hematoxylin-eosin staining of the liver. In CD-fed fish, liver cells were well preserved, with no evidence of lipid deposition. Conversely, in HFD-fed zebrafish, liver cells exhibited significant enlargement, with nuclei displaced to the cell periphery due to lipid infiltration. In the HFD + NO group, lipid infiltration was reduced compared to the HFD group, and the cellular structure appeared closer to normal.
Hematoxylin-eosin staining (n = 45) of liver tissues of zebrafish fed diets with a conventional diet (CD), a high-fat diet (HFD), and an HFD supplemented with 2% Nannochloropsis oculate (HFD + NO) for 4 weeks. LV: lipid vacuole.
Discussion
Modern intensive aquaculture processes have made extensive use of high-fat diets because they are economical and effective at promoting growth and lowering dietary protein levels. Fish health is further hampered by excessive fat consumption, which also encourages fat deposition in tissues, which in turn triggers oxidative stress and inflammatory reactions12,38. This study demonstrated that an HFD suppressed growth and feed utilization in zebrafish after four weeks, consistent with previous findings in Trachinotus ovatu5, Monopterus albus39, and Cyprinus carpio40. High-fat diets may negatively impact digestion and nutrient assimilation over time, potentially inducing inflammation that disrupts digestive enzyme activity and the intestinal microbiota, ultimately reducing feed absorption and growth40,41,42. Notably, supplementing the HFD with astaxanthin-rich microalgae significantly improved growth performance and feed utilization. According to Amar et al. (2001), astaxanthin plays a key role in enhancing nutrient intake and promoting fish growth43,44. These findings align with those of Zhao et al. (2022a), who reported that dietary microalgae supplementation improved digestive function and fish growth25. By regulating lipid metabolism and supporting immune and digestive functions, microalgae may mitigate the adverse effects of an HFD and enhance fish growth45.
This study demonstrated that zebrafish fed a high-fat diet (HFD) experienced significant oxidative stress, as indicated by decreased activities of antioxidant enzymes (SOD, CAT, GPx, GST) and reduced levels of GSH, GSSG, and GR, alongside elevated MDA levels. These findings are consistent with previous research showing that excessive dietary fat increases reactive oxygen species (ROS) production, leading to oxidative stress and lipid peroxidation in liver tissues46. The elevated MDA levels further confirm increased lipid peroxidation, a hallmark of oxidative damage. Notably, supplementation with Nannochloropsis oculata enriched with astaxanthin (HFD + NO) significantly enhanced the antioxidant defense system. Compared to the HFD group, the HFD + NO group exhibited higher SOD, CAT, GPx, and GST activities, as well as increased GSH, GSSG, and GR levels, while MDA levels were markedly reduced. These results align with previous studies demonstrating astaxanthin’s potent antioxidant properties in mitigating oxidative stress by scavenging ROS and enhancing enzymatic antioxidant defenses47,48. The protective effects of astaxanthin in the liver may be mediated through the NRF2/ARE signaling pathway, which regulates antioxidant and detoxification gene expression49. The observed restoration of GSH levels and reduction in MDA concentrations in the supplemented group further support its antioxidant efficacy.
Excessive dietary fat intake is a well-established cause of lipid metabolism dysregulation. In this study, HFD-fed zebrafish exhibited significantly elevated serum triglycerides (TG) and total cholesterol (TC), along with increased hepatic lipid content (total lipids, TC, fatty acids, and TG), indicating disrupted lipid homeostasis and the development of hepatic steatosis characterized by excessive fat accumulation in liver cells. Histological analysis revealed hepatic steatosis and ballooning degeneration in the HFD group, marked by excessive lipid droplet accumulation and disrupted cellular architecture. These pathological changes align with previous findings linking high-fat diets to fatty liver disease in zebrafish through mechanisms involving ROS production, mitochondrial dysfunction, and endoplasmic reticulum stress50. Excess dietary lipids contribute to hepatic lipid accumulation through multiple pathways, including increased fatty acid uptake, enhanced de novo lipogenesis, and impaired β-oxidation51. In fish, prolonged exposure to high-fat diets suppresses peroxisome proliferator-activated receptor alpha (PPARα)-mediated fatty acid oxidation while upregulating lipogenic genes, exacerbating lipid deposition52. Additionally, chronic lipid overload induces endoplasmic reticulum stress and activates sterol regulatory element-binding protein 1c (SREBP-1c), further promoting lipid biosynthesis53.
This study demonstrated that an HFD significantly increased serum and hepatic triglycerides (TG) and total cholesterol (TC) levels, along with elevated liver enzyme activities (ALT, AST, GGT, ALP, and LDH), indicating hepatic lipid accumulation and liver damage. These findings align with previous research showing that excessive dietary fat promotes lipid deposition, contributing to non-alcoholic fatty liver disease (NAFLD) and impaired liver function in fish54. Elevated liver enzymes suggest hepatocellular damage resulting from oxidative stress and inflammation induced by lipid overload. Conversely, the HFD + NO group exhibited significantly reduced TG and TC levels in both serum and liver while normalizing liver enzyme activities. These results support prior findings in mammals that astaxanthin regulates lipid metabolism by inhibiting sterol regulatory element-binding proteins (SREBPs), which drive lipogenesis, and activating peroxisome proliferator-activated receptors (PPARs), which promote fatty acid oxidation55. Additionally, astaxanthin’s antioxidant properties likely mitigated oxidative damage to hepatocytes, reducing enzyme leakage into the bloodstream15,56,57.
In the HFD group, the expression of pro-inflammatory cytokines (il-1β and il-8) was significantly upregulated, while anti-inflammatory cytokines (IL-10 and TGF-β) were downregulated compared to the control diet (CD) group. Chronic inflammation induced by excessive fat intake is well-documented in fish fed HFDs, as lipid accumulation activates nuclear factor-kappa B (NF-κB) signaling, driving the transcription of pro-inflammatory cytokines58. Supplementation with N. oculata reversed these effects by downregulating il-1β and il-8 while upregulating il-10 and tgf-β. This anti-inflammatory response is likely mediated by astaxanthin, which inhibits NF-κB activation and downregulating proinflammatory cytokines anti-inflammatory signaling through the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway59,60,61. The balance between pro- and anti-inflammatory cytokines is essential for hepatic homeostasis, suggesting that N. oculata effectively mitigates HFD-induced liver inflammation.
Conclusions
This study demonstrates that supplementing a high-fat diet with astaxanthin-rich N. oculata significantly alleviates liver dysfunction in zebrafish. N. oculata reduced hepatic lipid accumulation, improved antioxidant defenses, and modulated inflammatory cytokine expression, thereby protecting against diet-induced oxidative stress and inflammation. These beneficial effects are primarily attributed to astaxanthin’s ability to enhance lipid metabolism and regulate immune responses. Overall, N. oculata shows promise as a functional feed additive for improving liver health in aquaculture species. Further research is recommended to clarify its molecular mechanisms and evaluate long-term impacts in diverse aquatic environments.
Data availability
The data presented in this study are available in the article.
References
Tacon, A. G. Trends in global aquaculture and Aquafeed production: 2000–2017. Reviews Fisheries Sci. Aquaculture. 28 (1), 43–56 (2020).
Lim, C., Lee, C. S. & Webster, C. D. Alternative Protein Sources in Aquaculture Diets (CRC, 2023).
Zhao, W. & Huang, L. D. Astaxanthin-enriched oedocladium sp. improves liver health by enhancing fatty acid oxidation and suppressing inflammation in Trachinotus ovatus that was fed a high-fat diet. Anim. Feed Sci. Technol. 306, 115813 (2023).
Zhang, Y. et al. Calcium pyruvate attenuates fat deposition by augmenting fatty acid oxidation and inhibiting glucose oxidation in juvenile large yellow croaker (Larimichthys crocea) consuming a high-fat diet. Aquaculture 562, 738778 (2023).
Zhao, W. et al. Astaxanthin, bile acid and chlorogenic acid attenuated the negative effects of high-fat diet on the growth, lipid deposition, and liver health of Oncorhynchus Mykiss. Aquaculture 567, 739255 (2023).
Wang, C. et al. Glycerol monolaurate and triglycerol monolaurate alleviated high-fat diet induced lipid accumulation and damage of liver in zebrafish (Danio rerio). Aquaculture 561, 738616 (2022).
Kulkarni, A. et al. A novel 2-hit zebrafish model to study early pathogenesis of non-alcoholic fatty liver disease. Biomedicines 10 (2), 479 (2022).
He, L. et al. Hepatic oxidative stress and cell death influenced by dietary lipid levels in a fresh teleost. Antioxidants 13 (7), 808 (2024).
Park, K. H. et al. Palmitic acid-enriched diet induces hepatic steatosis and injury in adult zebrafish. Zebrafish 16 (6), 497–504 (2019).
Khazaei, M. & Ardeshir, R. A. Protective Effects of Sulfated Polysaccharides from Enteromorpha Intestinalis on Oxidative Stress, Liver Iron Overload and Ferroptosis in Zebra Fish Exposed To Ethanol181p. 117715 (Biomedicine & Pharmacotherapy, 2024).
Dai, Y. J. et al. Chronic Inflammation Is a Key To Inducing Liver Injury in Blunt Snout Bream (Megalobrama amblycephala) Fed with high-fat Diet97p. 28–37 (Developmental & Comparative Immunology, 2019).
Naiel, M. A. et al. The risk assessment of high-fat diet in farmed fish and its mitigation approaches: A review. J. Anim. Physiol. Anim. Nutr. 107 (3), 948–969 (2023).
Wu, D. et al. Resveratrol ameliorates oxidative stress, inflammatory response and lipid metabolism in common carp (Cyprinus carpio) fed with high-fat diet. Front. Immunol. 13, 965954 (2022).
Lim, K. C. et al. Astaxanthin as feed supplement in aquatic animals. Reviews Aquaculture. 10 (3), 738–773 (2018).
Li, M. Y. et al. Astaxanthin ameliorates high-carbohydrate diet-induced ER stress, immunosuppression and hepatic glucose metabolism through ampk/autophagy pathway in Channa Argus. Aquaculture 598, 742010 (2025).
Zhao, W. et al. Comparison of the retention rates of synthetic and natural Astaxanthin in feeds and their effects on pigmentation, growth, and health in rainbow trout (Oncorhynchus mykiss). Antioxidants 11 (12), 2473 (2022).
Capelli, B., Bagchi, D. & Cysewski, G. R. Synthetic Astaxanthin is significantly inferior to algal-based Astaxanthin as an antioxidant and May not be suitable as a human nutraceutical supplement. Nutrafoods 12, 145–152 (2013).
Hibberd, D. J. Notes on the taxonomy and nomenclature of the algal classes eustigmatophyceae and tribophyceae (synonym Xanthophyceae). Bot. J. Linn. Soc. 82 (2), 93–119 (1981).
Abdelghany, M. F. et al. Effects of dietary Nannochloropsis oculata on growth performance, serum biochemical parameters, immune responses, and resistance against Aeromonas veronii challenge in Nile tilapia (Oreochromis niloticus). Fish & shellfish immunology, 107: pp. 277–288. (2020).
Navalho, S. et al. Nannochloropsis lipids and polyunsaturated fatty acids: Potential applications and strain improvement. Mar. Drugs. 23 (3), 128 (2025).
Wang, F. et al. A novel strategy for the hyper-production of Astaxanthin from the newly isolated microalga haematococcus pluvialis JNU35. Algal Res. 39, 101466 (2019).
Landgraf, K. et al. Short-term overfeeding of zebrafish with normal or high-fat diet as a model for the development of metabolically healthy versus unhealthy obesity. BMC Physiol. 17, 1–10 (2017).
Parichy, D. M. et al. Normal table of postembryonic zebrafish development: Staging by externally visible anatomy of the living fish. Dev. Dyn. 238 (12), 2975–3015 (2009).
Sun, S. et al. Fat-1 Transgenic zebrafish are protected from abnormal lipid deposition induced by high-vegetable oil feeding. Appl. Microbiol. Biotechnol. 104, 7355–7365 (2020).
Zhao, W. et al. Beneficial changes in growth performance, antioxidant capacity, immune response, hepatic health, and flesh quality of Trachinotus ovatus fed with oedocladium carolinianum. Front. Immunol. 13, 940929 (2022).
Ahmadifar, E. et al. Can dietary ginger (Zingiber officinale) alter biochemical and immunological parameters and gene expression related to growth, immunity and antioxidant system in zebrafish (Danio rerio)? Aquaculture 507, 341–348 (2019).
Bradford, N. A rapid and sensitive method for the quantitation microgram quantities of a protein isolated from red cell membranes. Anal. Biochem. 72 (248), e254 (1976).
Marklund, S. & Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 47 (3), 469–474 (1974).
Hafeman, D., Sunde, R. & Hoekstra, W. Effect of dietary selenium on erythrocyte and liver glutathione peroxidase in the rat. J. Nutr. 104 (5), 580–587 (1974).
Habig, W. H., Pabst, M. J. & Jakoby, W. B. Glutathione S-transferases: The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249 (22), 7130–7139 (1974).
Baker, M. A., Cerniglia, G. J. & Zaman, A. Microtiter plate assay for the measurement of glutathione and glutathione disulfide in large numbers of biological samples. Anal. Biochem. 190 (2), 360–365 (1990).
Tietze, F. Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: Applications to mammalian blood and other tissues. Anal. Biochem. 27 (3), 502–522 (1969).
Cribb, A. E., Leeder, J. S. & Spielberg, S. P. Use of a microplate reader in an assay of glutathione reductase using 5, 5′-dithiobis (2-nitrobenzoic acid). Analytical biochemistry, 183(1): pp. 195–196. (1989).
Ohkawa, H., Ohishi, N. & Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 95 (2), 351–358 (1979).
Folch, J., Lees, M. & Stanley, G. S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 226 (1), 497–509 (1957).
Frings, C. S. & Dunn, R. T. A colorimetric method for determination of total serum lipids based on the sulfo-phospho-vanillin reaction. Am. J. Clin. Pathol. 53 (1), 89–91 (1970).
Muller, P. Y. et al. Short technical report processing of gene expression data generated by quantitative real-time RT-PCR. Biotechniques 32 (6), 1372–1379 (2002).
Zhou, W. H. et al. Dietary sodium acetate improves high-fat diet utilization through promoting differential nutrients metabolism between liver and muscle in nile tilapia (Oreochromis niloticus). Aquaculture 565, 739142 (2023).
Shi, Y. et al. Taurine supplements in high-fat diets improve survival of juvenile monopterus albus by reducing lipid deposition and intestinal damage. Aquaculture 547, 737431 (2022).
Abasubong, K. P. et al. Effects of dietary xylooligosaccharide prebiotic supplementation on growth, antioxidant and intestinal immune-related genes expression in common carp Cyprinus Carpio fed a high‐fat diet. J. Anim. Physiol. Anim. Nutr. 106 (2), 403–418 (2022).
Zhang, W. et al. Effects of dietary lipid levels on growth, digestive enzyme activities, antioxidant capacity, and lipid metabolism in turbot (Scophthalmus Maximus L.) at three different stages. Aquacult. Nutr. 2022 (1), 1042263 (2022).
Chen, W. et al. Dietary sodium butyrate supplementation improves fish growth, intestinal microbiota composition, and liver health in largemouth bass (Micropterus salmoides) fed high-fat diets. Aquaculture 564, 739040 (2023).
Amar, E. et al. Influence of various dietary synthetic carotenoids on bio-defence mechanisms in rainbow trout, Oncorhynchus Mykiss (Walbaum). Aquac. Res. 32, 162–173 (2001).
Li, M. Y. et al. Exploring cr (VI)-induced blood-brain barrier injury and neurotoxicity in zebrafish and Snakehead fish, and inhibiting toxic effects of Astaxanthin. Environ. Pollut. 355, 124280 (2024).
Song, X. et al. Dietary Astaxanthin improved the body pigmentation and antioxidant function, but not the growth of discus fish (Symphysodon spp). Aquac. Res. 48 (4), 1359–1367 (2017).
Yu, D. et al. High fat diet-induced oxidative stress blocks hepatocyte nuclear factor 4α and leads to hepatic steatosis in mice. J. Cell. Physiol. 233 (6), 4770–4782 (2018).
Kalinowski, C. T. et al. Influence of dietary Astaxanthin on the hepatic oxidative stress response caused by episodic hyperoxia in rainbow trout. Antioxidants 8 (12), 626 (2019).
Elia, A. C. et al. Effects of Astaxanthin and canthaxanthin on oxidative stress biomarkers in rainbow trout. J. Toxicol. Environ. Health Part. A. 82 (13), 760–768 (2019).
Allameh, A. et al. Oxidative stress in liver pathophysiology and disease. Antioxidants 12 (9), 1653 (2023).
Schlegel, A. Studying non-alcoholic fatty liver disease with zebrafish: A confluence of optics, genetics, and physiology. Cell. Mol. Life Sci. 69, 3953–3961 (2012).
Musso, G., Gambino, R. & Cassader, M. Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (NAFLD). Prog. Lipid Res. 48 (1), 1–26 (2009).
Shen, Y. et al. Differential regulatory effects of optimal or excessive dietary lipid levels on growth, lipid metabolism and physiological response in black seabream (Acanthopagrus schlegelii). Aquaculture 560, 738532 (2022).
Fu, S., Watkins, S. M. & Hotamisligil, G. S. The role of Endoplasmic reticulum in hepatic lipid homeostasis and stress signaling. Cell Metabol. 15 (5), 623–634 (2012).
Jia, R. et al. Effects of high-fat diet on steatosis, Endoplasmic reticulum stress and autophagy in liver of tilapia (Oreochromis niloticus). Front. Mar. Sci. 7, 363 (2020).
Xu, J. et al. A combination of flaxseed oil and Astaxanthin improves hepatic lipid accumulation and reduces oxidative stress in high fat-diet fed rats. Nutrients 9 (3), 271 (2017).
Abou-Zeid, S. M. et al. Astaxanthin mitigates thiacloprid-induced liver injury and immunotoxicity in male rats. Mar. Drugs. 19 (9), 525 (2021).
Lv, Y. et al. Hepatocytes and mitochondria dual-targeted Astaxanthin WPI-SCP nanoparticles for the alleviation of alcoholic liver injury. Int. J. Biol. Macromol. 285, 137992 (2025).
Rives, C. et al. Oxidative stress in NAFLD: Role of nutrients and food contaminants. Biomolecules 10 (12), 1702 (2020).
Ashrafizadeh, M. et al. Astaxanthin and Nrf2 signaling pathway: A novel target for new therapeutic approaches. Mini Rev. Med. Chem. 22 (2), 312–321 (2022).
Niu, X. T. et al. The major role of glucocorticoid receptor (GR) in Astaxanthin alleviates immune stress in Channa Argus lymphocyte. Aquaculture 584, 740637 (2024).
Zhao, L. et al. Astaxanthin Ameliorates Cr (VI) Accumulation, Antioxidant, Digestive, Apoptosis and Inflammatory Responsep. 104240 (Regional Studies in Marine Science, 2025).
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the Conceptualization and design. Writing-Original draft preparation and conducting experiments were performed by Sh.J. Material preparation and resources were supplied by M.A-B and S.P.H.S. Statistical and data Analyses were done by S.M.M., and Methodology, Reviewing, editing, and final version of the manuscript were done by L.E. and K.L. All listed authors have approved the manuscript before submission, including the names and order of authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no conflict interests.
Ethical approval
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jafari, S., Akhavan-Bahabadi, M., Shekarabi, S.P.H. et al. Astaxanthin-rich Nannochloropsis oculata mitigates high-fat diet-induced liver dysfunction in zebrafish by modulating lipid metabolism and oxidative stress. Sci Rep 15, 28862 (2025). https://doi.org/10.1038/s41598-025-13642-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13642-8