Abstract
Hyperuricemia is closely linked to metabolic diseases and cardiovascular conditions, while circadian syndrome (CircS) plays a pivotal role in metabolic syndrome (MetS) and its related disorders. This study examines the relationship between CircS, serum uric acid (SUA) levels, and the risk of hyperuricemia using data from the National Health and Nutrition Examination Survey (NHANES) from 2005 to 2018. Weighted multivariable linear and logistic regression analyses were employed to assess the relationships between CircS, its components, SUA levels, and the risk of hyperuricemia, supplemented by subgroup analyses and interaction tests to understand the influence of other risk factors. Restricted cubic spline (RCS) regression was utilized to investigate potential nonlinear relationships. The study included 16,785 adults aged 20 and older, of whom 5,833 exhibited CircS and 3,572 had hyperuricemia. The weighted linear regression revealed a significant positive association between CircS and SUA levels (β = 0.108, 95% CI 0.034, 0.182; P = 0.005), with triglyceride scores showing the strongest association (β = 0.168, 95% CI 0.105, 0.231; P < 0.001). Additionally, weighted multivariable logistic regression indicated a significant link between CircS and hyperuricemia occurrence (OR = 1.227; 95% CI: 1.073, 1.402; P = 0.003), with blood pressure scores having the most substantial contribution (OR = 1.538; 95% CI: 1.347, 1.756; P < 0.001). Subgroup analyses confirmed the robustness of these correlations across various populations. RCS results demonstrated significant nonlinear relationships between CircS, SUA levels, and hyperuricemia (P < 0.001). In conclusion, this study establishes a significant positive association between CircS and both SUA levels and the risk of hyperuricemia, highlighting a notable nonlinear relationship between the two.
Similar content being viewed by others
Introduction
Uric acid is the final product of purine metabolism in the human body, and under physiological conditions, its synthesis and excretion are balanced1. When this balance is disrupted, hyperuricemia occurs. With changes in lifestyle and dietary patterns, the prevalence of hyperuricemia has been rising, becoming the “fourth major” disease after hypertension, diabetes, and hyperlipidemia1,2. As of 2020, approximately 930 million people globally were affected by hyperuricemia and gout, with estimates suggesting this number could reach 1.18 billion by 20252. Hyperuricemia not only leads to gout but is also associated with cardiovascular events such as hypertension, atherosclerosis, and atrial fibrillation, as well as conditions like diabetes and chronic kidney disease3,4,5,6,7,8. However, the exact pathological mechanisms of hyperuricemia remain incompletely understood, and long-term treatment for hyperuricemia carries both costs and risks. Therefore, identifying high-risk populations for hyperuricemia and providing early prevention is crucial.
The circadian rhythm system is a major regulator of nearly all aspects of human health and metabolism9. It governs gene expression, the release of various hormones, body temperature, activity patterns, energy expenditure, and other vital bodily functions10. Circadian syndrome (CircS) is characterized by any of the following four conditions: dyslipidemia (elevated triglycerides or decreased high-density lipoprotein cholesterol), hypertension, central obesity, diabetes, depression, and sleep deprivation11. In recent years, lifestyle changes have led to an increased incidence of CircS, exacerbating the development of various chronic diseases, including metabolic disorders such as obesity, hypertension, and diabetes, as well as cancers12,13,14.
It is essential to identify risk factors within the causal chain to promote the prevention of hyperuricemia. However, most previous studies have only examined the relationship between hyperuricemia and individual independent risk factors. Circadian rhythm disturbances encompass multiple risk factors that contribute to hyperuricemia11,12. CircS represents a more coherent metabolic syndrome (MetS) clustering and its complications15. Yet, few studies to date have comprehensively examined the association between CircS and hyperuricemia. Therefore, this study aims to utilize the National Health and Nutrition Examination Survey (NHANES) data to explore the relationship between CircS and serum uric acid (SUA) levels as well as hyperuricemia.
Methods
Study design and population
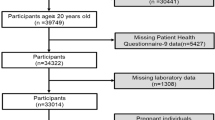
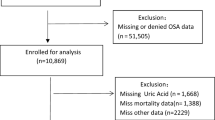
This cross-sectional study utilized data from the NHANES (https://www.cdc.gov/nchs/nhanes). NHANES is a national survey conducted by the Centers for Disease Control and Prevention (CDC) that employs a complex multistage sampling design to collect data representative of the non-institutionalized U.S. population. The NHANES program was approved by the National Center for Health Statistics (NCHS) Research Ethics Review Board, and all survey participants provided informed consent16. Data from NHANES cycles from 2005 to 2018 were analyzed. Exclusion criteria for this study included age < 20 years, and missing data on CircS and SUA levels, as detailed in Fig. 1.
Screening conditions and process for the study population.
SUA levels and hyperuricemia diagnosis
Serum samples were collected from participants and stored at -30 °C before being sent to the CDC/NCEH/DLS for testing. SUA levels were measured using the Beckman Coulter UniCel® dx800 as part of the routine serum biochemical profile. Hyperuricemia was defined as serum uric acid levels ≥ 7 mg/dL in men and ≥ 6 mg/dL in women17.
Diagnosis of circadian syndrome
The diagnosis of CircS was based on seven components: reduced sleep duration (< 6 h), depressive symptoms (Patient Health Questionnaire-10 score ≥ 10), central obesity (waist circumference ≥ 85 cm for men and ≥ 80 cm for women), elevated triglycerides (≥ 150 mg/dL or use of lipid-lowering medications), low high-density lipoprotein cholesterol (HDL-C < 40 mg/dL in men and < 50 mg/dL in women or use of lipid-lowering medications), hypertension (systolic ≥ 130 and/or diastolic ≥ 85 mmHg or use of antihypertensive medications), and hyperglycemia (fasting blood glucose ≥ 100 mg/dL or use of glucose-lowering medications). Participants with at least four components were classified as having CircS12. Waist circumference and blood pressure measurements were taken by trained professionals at mobile examination centers. Participants had 1 to 4 blood pressure readings; those with no readings were excluded. If there was only one reading, it was recorded as the final value; if there were multiple readings, the first one was always excluded, and the average of the remaining readings was used18. Data on sleep duration, depressive symptoms, and medication history were collected through self-reported questionnaires. Triglycerides, HDL-C, and blood glucose concentrations were measured from fasting venous blood samples using enzymatic colorimetric methods.
Covariates
Covariates include demographic data (gender, age, education level, poverty income ratio), Body mass index (BMI), laboratory indicators (including albumin, alanine aminotransferase (ALT), and aspartate aminotransferase (AST)), as well as smoking, alcohol consumption history, and medical history (presence of coronary heart disease, stroke, or cancer). The poverty income ratio (PIR) was determined by calculating the ratio of monthly family income to the poverty levels defined by the Department of Health and Human Services, categorized as low income (≤ 1.30), middle income (1.31–3.50), and high income (> 3.50)19. BMI was calculated by dividing weight (kg) by height (m²). Smoking and alcohol consumption history, along with medical history, were collected through questionnaires. Participants’ creatinine clearance rates were assessed using the CKD-EPI Creatinine Eq. (2021) to evaluate kidney function20.
Statistical analyses
Statistical analyses were conducted following NHANES database analysis guidelines and recommended weights. The Kolmogorov-Smirnov test was used to assess data normality. Non-normally distributed continuous data were presented as medians and interquartile ranges, with group comparisons made using the Mann-Whitney U test. Categorical data were expressed as proportions, with group comparisons also made using the Mann-Whitney U test. Count data were presented as proportions, and group comparisons were conducted using the chi-square test. Weighted multivariable linear regression analyses were performed to explore the association between CircS and SUA levels. Weighted multivariable logistic regression analyses were used to examine the relationship between CircS and the risk of hyperuricemia. Three models were constructed: Model 1 was unadjusted, Model 2 was adjusted for gender, age, and race, and Model 3 was adjusted for gender, age, race, household poverty ratio, education level, BMI, eGFR, albumin, ALT, AST, alcohol, smoke and medical history of cancer, coronary heart disease, stroke. Restricted cubic spline (RCS) regression (with three knots) was employed to explore potential nonlinear relationships between circadian scores and SUA levels and hyperuricemia risk. All statistical analyses were performed using R software (version 4.0.0) and SPSS (version 25.0), with a significance level set at P < 0.05.
Results
Baseline characteristics
Table 1 presents the baseline characteristics of the study population. A total of 16,785 adults aged 20 years and older were included in this study, of which 8,413 (50.12%) were male. Among the total study population, 3,572 participants were diagnosed with hyperuricemia. The median age of the study population was 50 years, with the hyperuricemia group having a median age of 58 years, which is older than the non-hyperuricemia group (median age 47 years). The median SUA level for the entire study population was 5.40 mg/dL, while the median level for the hyperuricemia group was 7.30 mg/dL. The proportion of males in the hyperuricemia group was higher (54.31% vs. 45.69%). Among the study population, 5,833 participants (34.75%) had CircS, with 1,877 (52.55%) of those in the hyperuricemia group meeting the criteria, compared to 29.94% in the non-hyperuricemia group. Additionally, of the seven components of circadian syndrome, all except for depressive status showed significantly higher positive rates in the hyperuricemia group (all P < 0.001).
Association between circs and SUA levels
Weighted linear regression analysis was performed to explore the strength of the association between CircS and its components with SUA levels. Three models were constructed: Model 1 was unadjusted, Model 2 adjusted for gender, age, and race, and Model 3 adjusted for all variables. As shown in Table 2, in the unadjusted model (Model 1), a positive association was observed between CircS and SUA levels (β = 0.625, 95% CI 0.55, 0.699; P < 0.001). After adjusting for all covariates, the positive association remained significant (β = 0.108, 95% CI 0.034, 0.182; P = 0.005). Furthermore, among the seven components of CircS, five components (excluding depressive status and reduced sleep) remained positively associated with SUA levels after adjusting for all covariates (all P < 0.05), with triglyceride scores showing the strongest effect (β = 0.168, 95% CI 0.105, 0.231; P < 0.001).
Association between circs and hyperuricemia
Weighted multivariable logistic regression analysis was conducted to investigate the relationship between CircS and the risk of hyperuricemia. As shown in Table 3, in the unadjusted model (Model 1), individuals with CircS had a 2.559 times higher risk of hyperuricemia compared to those without the syndrome (OR = 2.559; 95% CI: 2.3, 2.846; P < 0.001). After adjusting for age, gender, and race (Model 2), the association between CircS and hyperuricemia risk remained significant (OR = 2.314; 95% CI: 2.062, 2.596; P < 0.001). Even after adjusting for all covariates (Model 3), this positive association remained significant (OR = 1.227; 95% CI:1.073,1.402; P = 0.003). Among the seven components of CircS, blood pressure scores had the greatest contribution (OR = 1.538; 95% CI: 1.347, 1.756; P < 0.001).
Subgroup analysis
To further investigate the influence of other risk factors on the relationship between CircS and hyperuricemia risk, subgroup analyses and interaction tests were performed based on gender, age, race, and PIR. Overall results indicated a significant positive association between CircS and hyperuricemia (OR = 1.227, 95% CI: 1.073–1.402, P = 0.003). In the gender subgroup, the association was stronger in females (OR = 1.412, 95% CI:1.17, 1.704, P < 0.001), while no significant association was found in males (OR = 1.081, 95% CI:0.876, 1.334, P = 0.463), with a significant interaction between genders (P for interaction < 0.001). In age stratification, the association was significant in the younger population (< 65 years) (OR = 1.218, 95% CI:1.037, 1.432, P = 0.017), while the interaction by age was not significant (P for interaction = 0.940). Subgroup analyses by race and PIR also showed some differences, though interactions were not significant (Table 4).
Nonlinear regression analysis of circadian scores with SUA levels and hyperuricemia
Using RCS, we explored the nonlinear relationships between CircS and SUA levels as well as the risk of hyperuricemia. As shown in Fig. 2, there was a significant nonlinear relationship between CircS and both SUA levels and the occurrence of hyperuricemia after adjusting for all covariates (P < 0.001), with threshold effects appearing at a CircS score of 3. Specifically, the relationship between CircS score and SUA level did not follow a typical J-shaped or S-shaped pattern. Uric acid levels showed an approximately linear increase when the CircS score ranged from 0 to 3. Between scores of 3 and 4, the slope of the curve changed, and beyond a score of 4, the increase in uric acid levels gradually slowed, indicating a diminishing marginal effect (Fig. 2A). In contrast, the association between CircS score and hyperuricemia exhibited an overall S-shaped curve, characterized as an “acceleration–peak–plateau” pattern (Fig. 2B). When the CircS score was between 0 and 1, the risk increased slowly. From 1 to 2, the risk rose more rapidly, with a sharp increase observed between 2 and 3, which marked a critical risk escalation zone. From 3 to 4, the upward trend began to slow, and beyond a score of 4, the risk continued to increase slightly but approached a plateau.
Restricted cubic spline regression explores the potential nonlinear relationships between circadian syndrome score and serum uric acid levels (A) and hyperuricemia (B). The β coefficient (solid lines) and 95% confidence levels (shaded areas) were adjusted for gender, age, race/ethnicity, household poverty ratio, household poverty ratio, education level, BMI, eGFR, albumin, ALT, AST, alcohol, smoking, and medical history of cancer, coronary heart disease, and stroke. OR: odds ratio; 95%CI, Confidence Interval.
Discussion
This study provides the first investigation into the association between CircS and both serum uric acid levels and the risk of hyperuricemia. The results revealed a significant positive association between CircS and serum uric acid levels/hyperuricemia, with a distinct nonlinear dose-response relationship (P-nonlinear < 0.001). Specifically, we observed a gradual increase in uric acid concentrations with rising CircS scores at lower levels. In contrast, the risk of hyperuricemia exhibited an abrupt surge when scores exceeded a critical threshold. This nonlinear pattern suggests the existence of a pivotal threshold effect in metabolic regulation. Subgroup analyses and interaction tests further confirmed the robustness of this positive association across diverse populations.
CircS is primarily influenced by circadian rhythm dysfunction, which refers to a set of biological variables generated by the biological clock over 24 hours11. The biological clock regulates not only daily physiological and molecular rhythms but also gene expression in a rhythmic manner21,22,23. Disruptions in circadian function and endocrine regulation are key factors contributing to circadian dysregulation. When circadian rhythms are altered, a variety of issues can arise, including inflammation, immune dysfunction, and metabolic disorders24,25. While extensive literature has established a close relationship between circadian disruption and metabolic diseases such as diabetes and obesity, as well as cardiovascular diseases, circadian disruption is a critical factor in metabolic diseases, which in turn are high-risk factors for hyperuricemia11,12,13,14,26. However, no previous studies have directly investigated the association between circadian syndrome and hyperuricemia. Additionally, most prior research has focused on the relationship between hyperuricemia and single independent risk factors, while circadian disruption encompasses multiple risk factors for hyperuricemia.
Circadian rhythm disruption may lead to elevated uric acid levels through several potential mechanisms. Firstly, circadian dysregulation can result in endocrine disorders and insulin resistance11,26both of which increase the production of uric acid while decreasing renal excretion, thereby raising SUA levels27. Secondly, disturbances in circadian rhythm may also trigger chronic inflammatory responses and oxidative stress, further exacerbating abnormalities in uric acid metabolism25. Inflammatory factors and oxidative stress products may directly influence the production and metabolism of uric acid28,29,30. Moreover, circadian disruption might impair renal excretion, interfering with uric acid clearance and leading to its accumulation in the body31,32. These mechanisms collectively suggest that circadian dysregulation could broadly affect uric acid metabolism through various metabolic pathways, increasing the risk of hyperuricemia.
Notably, among the various components of CircS, triglyceride levels demonstrated the most pronounced effect on serum uric acid concentrations. At the same time, blood pressure indicators showed the highest predictive value for the risk of hyperuricemia. These component-specific differences offer essential insights, suggesting that the coexistence of certain high-risk factors—such as hypertension combined with hypertriglyceridemia—may synergistically exacerbate kidney damage and promote oxidative stress through shared mechanisms33,34,35. This may accelerate the body’s progression beyond its metabolic compensation threshold. This phenomenon aligns with the “acceleration–inflection–plateau” pattern identified in the RCS analysis, in which a sharp increase in serum uric acid levels and hyperuricemia risk was observed when the CircS score reached 3 or higher. It also supports the previously proposed concept of metabolic inflammation following a “buffer–burst” dynamic pattern36. Therefore, early identification of high-risk individuals, risk stratification, and timely implementation of lifestyle modifications or pharmacological interventions before this pathological tipping point is reached may help delay disease progression and reduce the risk of associated complications.
This study has several strengths. First, it is the first to investigate the relationship between circadian syndrome and serum uric acid levels, as well as hyperuricemia. Second, a comprehensive analysis was conducted to examine the relationship between each component of circadian syndrome and serum uric acid levels, as well as hyperuricemia, thereby enhancing the specificity of the findings. Third, based on a large sample of American adult population data, the study ensures external validity and generalizability of the results. However, there are limitations. First, as a cross-sectional study, it cannot establish causality between circadian syndrome and serum uric acid levels or the risk of hyperuricemia; thus, future prospective studies are needed to clarify this relationship. Second, variables such as sleep duration, depressive symptoms, and medication history were self-reported, making them susceptible to recall bias. Additionally, although many confounding variables were accounted for, some potential unmeasured confounders may still influence the results.
Conclusion
There is a significant positive association between CircS and SUA levels and the occurrence of hyperuricemia, along with a notable nonlinear relationship between the two. Further comprehensive and prospective cohort studies are necessary to explore the relationship between the two in greater depth.
Data availability
This study are supported by data from the National Health and Nutrition Examination Survey (NHANES). Access to the data is available on their official website at https://www.cdc.gov/nchs/nhanes.
Abbreviations
- CircS:
-
Circadian syndrome
- MetS:
-
Metabolic syndrome
- SUA:
-
Serum uric acid
- NHANES:
-
National Health and Nutrition Examination Survey
- RCS:
-
Restricted cubic spline
- CDC:
-
Centers for Disease Control and Prevention
- HDL-C:
-
High-density lipoprotein cholesterol
- BMI:
-
Body mass index
- NCHS:
-
National Center for Health Statistics
- PIR:
-
Poverty income ratio
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- OR:
-
Odds ratio
- CI:
-
Confidence Interval
References
Hao, Y. et al. Uricase and horseradish peroxidase hybrid cahpo₄ Nanoflower integrated with transcutaneous patches for treatment of hyperuricemia. J. Biomed. Nanotechnol. 15, 951–965 (2019).
Singh, G., Lingala, B. & Mithal, A. Gout and hyperuricaemia in the USA: prevalence and trends. Rheumatol. (Oxford). 58, 2177–2180 (2019).
Kuwabara, M. et al. Metabolically healthy’ obesity and hyperuricemia increase risk for hypertension and diabetes: 5-year Japanese cohort study. Obes. (Silver Spring). 25, 1997–2008 (2017).
Kuwabara, M. et al. Asymptomatic hyperuricemia without comorbidities predicts cardiometabolic diseases: Five-Year Japanese cohort study. Hypertension 69, 1036–1044 (2017).
Kuwabara, M. et al. Hyperuricemia is an independent competing risk factor for atrial fibrillation. Int. J. Cardiol. 231, 137–142 (2017).
Kuwabara, M. et al. Elevated serum uric acid increases risks for developing high LDL cholesterol and hypertriglyceridemia: A five-year cohort study in Japan. Int. J. Cardiol. 261, 183–188 (2018).
Kuwabara, M. et al. Uric acid is a strong risk marker for developing hypertension from prehypertension: A 5-Year Japanese cohort study. Hypertension 71, 78–86 (2018).
Maruhashi, T., Hisatome, I., Kihara, Y. & Higashi, Y. Hyperuricemia and endothelial function: from molecular background to clinical perspectives. Atherosclerosis 278, 226–231 (2018).
Sj, K., Lm, C. & Jf, D. Introduction to Chronobiology. Cold Spring Harbor perspectives in biology 10, (2018).
Kalsbeek, A. et al. Circadian disruption and SCN control of energy metabolism. FEBS Lett. 585, 1412–1426 (2011).
Zimmet, P. et al. The circadian syndrome: is the metabolic syndrome and much more! J. Intern. Med. 286, 181–191 (2019).
Shi, Z. et al. The circadian syndrome predicts cardiovascular disease better than metabolic syndrome in Chinese adults. J. Intern. Med. 289, 851–860 (2021).
Xiao, Y. et al. Association between circadian syndrome and the prevalence of kidney stones in overweight adults: a cross-sectional analysis of NHANES 2007–2018. BMC Public. Health. 23, 960 (2023).
Wang, Y., Yang, L., Zhang, Y. & Liu, J. Relationship between circadian syndrome and stroke: A cross-sectional study of the National health and nutrition examination survey. Front. Neurol. 13, 946172 (2022).
Hong, F. et al. Melatonin orchestrates lipid homeostasis through the hepatointestinal circadian clock and microbiota during constant light exposure. Cells 9, 489 (2020).
Skrivankova, V. W. et al. Strengthening the reporting of observational studies in epidemiology using Mendelian randomization: the STROBE-MR statement. JAMA 326, 1614–1621 (2021).
Zhang, T. et al. Edible mushroom consumption and incident hyperuricemia: results from the TCLSIH cohort study. Food Funct. 12, 9178–9187 (2021).
Bourgeois, B. et al. Associations between height and blood pressure in the united States population. Med. (Baltim). 96, e9233 (2017).
Alaimo, K., Briefel, R. R., Frongillo, E. A. & Olson, C. M. Food insufficiency exists in the united states: results from the third National health and nutrition examination survey (NHANES III). Am. J. Public. Health. 88, 419–426 (1998).
Inker, L. A. et al. New Creatinine- and Cystatin C-Based equations to estimate GFR without race. N Engl. J. Med. 385, 1737–1749 (2021).
Kolarski, D. et al. Reversible modulation of circadian time with chronophotopharmacology. Nat. Commun. 12, 3164 (2021).
Camandola, S. & Mattson, M. P. Brain metabolism in health, aging, and neurodegeneration. EMBO J. 36, 1474–1492 (2017).
Geltink, R. I. K., Kyle, R. L. & Pearce, E. L. Unraveling the complex interplay between T cell metabolism and function. Annu. Rev. Immunol. 36, 461–488 (2018).
Timmons, G. A., O’Siorain, J. R., Kennedy, O. D., Curtis, A. M. & Early, J. O. Innate rhythms: clocks at the center of monocyte and macrophage function. Front. Immunol. 11, 1743 (2020).
Reale, M., Conti, L. & Velluto, D. Immune and Inflammatory-Mediated Disorders: From Bench to Bedside. J Immunol Res 7197931 (2018). (2018).
Mulè, A. et al. Sex differences in Rest-Activity circadian rhythm in patients with metabolic syndrome. Front. Physiol. 12, 641461 (2021).
Miao, Z. et al. Insulin resistance acts as an independent risk factor exacerbating high-purine diet induced renal injury and knee joint gouty lesions. Inflamm. Res. 58, 659–668 (2009).
Yu, W. & Cheng, J. D. Uric acid and cardiovascular disease: an update from molecular mechanism to clinical perspective. Front. Pharmacol. 11, 582680 (2020).
Xiao, J. et al. Soluble uric acid increases NALP3 inflammasome and interleukin-1β expression in human primary renal proximal tubule epithelial cells through the Toll-like receptor 4-mediated pathway. Int. J. Mol. Med. 35, 1347–1354 (2015).
Johnson, R. J. et al. Hyperuricemia, acute and chronic kidney disease, hypertension, and cardiovascular disease: report of a scientific workshop organized by the National kidney foundation. Am. J. Kidney Dis. 71, 851–865 (2018).
Carriazo, S. et al. Chronodisruption: A poorly recognized feature of CKD. Toxins (Basel). 12, 151 (2020).
Xiong, Y., Zhong, Q., Zhang, Y., Liu, Z. & Wang, X. The association between circadian syndrome and chronic kidney disease in an aging population: a 4-year follow-up study. Front. Endocrinol. 15, 1338110 (2024).
Lertsakulbunlue, S. et al. Associations of triglyceride-glucose index with hyperuricemia among Royal Thai army personnel. BMC Endocr. Disorders. 24, 17 (2024).
Mazidi, M., Kengne, A. P., Katsiki, N., Mikhailidis, D. P. & Banach, M. Lipid accumulation product and triglycerides/glucose index are useful predictors of insulin resistance. J. Diabetes Complications. 32, 266–270 (2018).
Choi, H. K., Atkinson, K., Karlson, E. W. & Curhan, G. Obesity, weight change, hypertension, diuretic use, and risk of gout in men: the health professionals follow-up study. Arch. Intern. Med. 165, 742–748 (2005).
Hotamisligil, G. S. Foundations of immunometabolism and implications for metabolic health and disease. Immunity 47, 406–420 (2017).
Author information
Authors and Affiliations
Contributions
MHY: conceived the study, designed the study, wrote the manuscript, statistical calculation, and data interpretation. HSS and LT: statistical calculation, and data interpretation. DY: participated in the data interpretation and manuscript writing. YZX: review of revised manuscripts.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The National Center for Health Statistics (NCHS) Research Ethics Review Board (ERB) approved the NHANES program, and all participants provided informed consent. No additional informed consent was required for this study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mao, H., Huang, S., Lin, T. et al. Exploring the relationship between circadian syndrome, serum uric acid levels, and hyperuricemia: evidence from NHANES 2005–2018. Sci Rep 15, 28984 (2025). https://doi.org/10.1038/s41598-025-14676-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14676-8