Abstract
To evaluate the diagnostic utility of time-resolved fluoroimmunoassay (TRFIA) for detecting serum anti-Phospholipase A2 Receptor (PLA2R) antibodies, specifically the IgG (PLA2R-IgG) and IgG4 subclass (PLA2R-IgG4). A total of 175 patients with serum anti-PLA2R antibody range from 2 to 20 RU/ml, between January 2018 and December 2024, were retrospectively analyzed. 121 serum specimen were tested by TRFIA and ELISA. According to renal biopsy, 52% (91/175) patients were diagnosed with primary membranous nephropathy (PMN). Notably, TRFIA exhibited higher sensitivity and specificity in identifying patients with PMN. Based on the manufacturer’s recommended cutoff values, TRFIA demonstrated a higher sensitivity of 14.3% for PLA2R-IgG and 34.1% for PLA2R-IgG4 compared to ELISA (0%). Receiver operating characteristic analysis showed that TRFIA achieved an area under the curve (AUC) of 0.829 for PLA2R-IgG and 0.814 for PLA2R-IgG4.The optimal anti-PLA2R antibody cutoff values for diagnosing PMN were 0.44 RU/ml (PLA2R-IgG-TRFIA) and 25.07 ng/ml (PLA2R-IgG4-TRFIA) with a sensitivity/specificity of 83.3%/82.4% for PLA2R-IgG-TRFIA, and 83.3% /76.9% for PLA2R-IgG4-TRFIA, both of which were higher than those of ELISA (cutoff:2.84 RU/ml; sensitivity/specificity: 50%/88%). In conclusion, TRFIA demonstrates enhanced sensitivity and specificity in detecting anti-PLA2R antibodies, which may facilitate more accurate diagnosis of PMN and minimize the need for invasive renal biopsies.
Similar content being viewed by others
Introduction
Primary membranous nephropathy (PMN) is a prevalent form of nephrotic syndrome in adults, particularly associated with the autoantibodies against podocyte antigens, notably the M-type phospholipase A2 receptor (PLA2R)1,2.The clinical importance of PMN lies in its potential to progress to chronic kidney disease (CKD) and ultimately end-stage renal disease (ESRD) if not diagnosed and managed appropriately3,4. An early and accurate diagnosis is crucial for ensuring effective treatment and achieving favorable outcomes.
Traditionally, renal biopsy has been the gold standard for diagnosing PMN, which, while accurate, is an invasive procedure that carries risks such as bleeding and infection5,6. The discovery of serum anti-PLA2R antibody has revolutionized the diagnostic landscape for PMN. This non-invasive alternative enables early detection and offers insights into disease activity, prognosis, and treatment response, thereby reshaping clinical practice in nephrology7. Nowadays, 12 more antigens have been identified1,2,8,9,10,11, including Thrombospondin type-1 domain containing 7 (THSD7A), Protocadherin 7 (PCDH7), and Exostosis 1/Exostosis 2(EXT1/2), NELL1, NCAM1, SEMA3B, FAT1, CNTN1, HTRA1, NTNG1, PCSK6 and NDNF, overall accounting for 80∼90% of PMN cases.
The detection of anti-PLA2R antibody has become a cornerstone in the diagnosis of PMN. The 2021 KDIGO guidelines suggest that a renal biopsy may not be needed to confirm PMN in patients with stable nephrotic syndrome and positive anti-PLA2R antibodies 12. Initially, western blotting (WB)7 and indirect immunofluorescence (IIF)13 were the primary methods used to detect anti-PLA2R antibody. Due to the limitations of lengthy processing times of WB and imprecise quantification of IIF, the enzyme-linked immunosorbent assay (ELISA) is now widely used to accurately measure anti-PLA2R antibody in clinical practice. A positive result, particularly at a threshold of greater than 20 RU/ml, is strongly indicative of PMN. The specificity of anti-PLA2R antibody for PMN is reported to be as high as 100%, while sensitivity varies between 50% and 99.7%, depending on the population studied14,15,16,17,18,19,20,21,22. Diagnosing PMN becomes challenging when anti-PLA2R antibody levels are less than 20 RU/ml.
Recent advancements in immunoassay technologies, including the time-resolved fluorescence immunoassay (TRFIA) developed by Huang in 201723, offer promising alternatives to traditional ELISA methods. TRFIA enhances detection sensitivity and specificity, enabling the differentiation of PMN from other renal pathologies without renal biopsies. In this study, we aimed to evaluate the diagnostic utility of TRFIA in detecting serum anti-PLA2R antibodies of IgG and IgG4 subclass in patients with anti-PLA2R antibody between 2 and 20 RU/ml. Additionally, we assessed the correlation of TRFIA results with clinical indicators, which may provide further insights into the relationship between antibody levels and disease severity.
Methods
Recruitment of patients
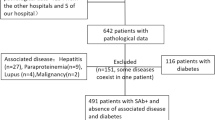
We screened all patients who underwent renal biopsy at Peking University Third Hospital from January 2018 to December 2024 (n = 1762). The inclusion criteria were: (1) age over 18 years, (2) availability of renal biopsy results, (3) a serum anti-PLA2R antibody test conducted prior to glucocorticoids or immunosuppressive therapy, and (4) serum anti-PLA2R antibody value between 2 and 20 RU/ml. Patients lacking information on anti-PLA2R antibody were excluded. The diagnosis of all kidney diseases depends on renal biopsy. The clinical data of all patients were collected and analyzed retrospectively. In this study, we finally included 175 patients, including primary membranous nephropathy (PMN, n = 91), IgA nephropathy (IgAN, n = 45), minimal change disease (MCD, n = 9), diabetic kidney disease (DKD, n = 9) and other kidney diseases (n = 21) (Fig. 1).
Flowchart of enrollment.
Clinical data collection
Clinical data were retrieved from the electronic medical records system. The clinical data comprised the following parameters: age, sex, clinical diagnosis, 24 h total urinary protein (24 h-UTP), serum anti-PLA2R antibody, albumin (ALB), uric acid (UA), blood urea (BUN), serum creatinine (Scr), total cholesterol, (TC), triglyceride (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), estimated glomerular filtration rate (eGFR),total bilirubin (TBIL), direct bilirubin (DBIL), blood routine results, glycated hemoglobin (HbA1c), C-reaction protein(CRP), erythrocyte sedimentation rate (ESR), D-dimer, fibrinogen, and immunological indicators such as complement components C3 and C4, immunoglobulins IgG, IgA, IgM, and IgE.
Anti-PLA2R antibody detection
At our hospital, serum anti-PLA2R antibody titers were assessed using a commercial ELISA kit (Euroimmune, Lubeck, Germany). According to the manufacturer’s recommendations, values ≥ 20 RU/ml are considered positive, value between 14 and 20 RU/mL are borderline, and values < 14 RU/ml are considered negative. In this study, values that are < 20 RU/ml are classified as negative.
In this study, we quantitatively detected serum anti-PLA2R IgG and the IgG4 subclass using TRFIA. PLA2R-IgG and PLA2R-IgG4 detection kits were purchased from Zhejiang Boshi Biotechnology Company (Hangzhou, Zhejiang, China), and fully automatic time-resolved fluorescence immunoassay analyzer TRF-1000 (Jiaxing Kaishi Biotechnology Co., Ltd. Jiaxing, Zhejiang, China) was used to detect anti-PLA2R antibodies. According to the specifications, PLA2R-IgG-TRFIA is positive for values ≥ 14 RU/mL, while PLA2R-IgG4-TRFIA is positive for values ≥ 200 ng/ml.
Statistical analysis
Statistical analyses were conducted using SPSS 22.0 and Prism 8.0. The measurement data were assessed to determine if they followed a normal distribution using the Shapiro–Wilk test. If the data followed a normal distribution, they were expressed as mean ± standard deviation, and differences between the two groups were compared using independent samples t-tests. If not, they were presented as the median along with the 25th and 75th percentiles, and differences between the two groups were analyzed using the Mann–Whitney U test. Categorical data were analyzed using the Chi-square test. The Spearman correlation coefficient was used to calculate correlations between the variables. Multivariate logistic regression models were employed to estimate the odds ratios (OR) and 95% confidence intervals (CIs) for different models. The difference in the areas under their receiver operating characteristic (ROC) curves (AUCs) was compared for statistical significance using DeLong’s test for two correlated curves. A two-tailed P value < 0.05 was considered significant statistically.
Ethics
This study followed the Declaration of Helsinki and received approval from the Ethics Review Committee of Peking University Third Hospital in China (M2018132). Informed consent was obtained from all individuals included in this study. We adhered to the declaration of Helsinki and national regulations during all study procedures.
Results
Basic characteristics of enrolled patients and comparisons PMN and non-PMN
A total of 175 patients with serum anti-PLA2R antibody between 2 and 20 RU/ml were included (Fig. 1), of which 105 were men and 70 were women. The patient’s average age was 49 (range 19–77). Among the 175 patients, 52% (91/175) were PMN. Non-PMN patients included 25.7% (45/175) IgAN, 5.1% (9/175) MCD, 5.1% (9/175) DKD, and 12.1% (21/175) other kidney diseases (Fig. 2A). Serum anti-PLA2R antibody levels were significantly increased in patients with PMN (Fig. 2B). PMN patients account for a large proportion of kidney patients with different anti-PLA2R antibody levels (Fig. 2C), so further differential diagnosis was necessary. By comparison, we found significant differences in clinical baseline data between PMN and non-PMN patients (Table 1). Patients with PMN are significantly older and have higher levels of 24 h-UTP, HGB, TC, eGFR, LDL, and D-dimer (p < 0.001). They also exhibit lower levels of ALB, serum creatinine, BUN, IgG and IgA (p < 0.001).
Renal disease classification and anti-PLA2R antibody levels in 175 enrolled patients. (A) The pie chart illustrating disease distribution indicated that 52% of patients with anti-PLA2R antibody levels between 2 and 20 RU/ml were diagnosed with PMN. (B) Compared to IgAN, serum anti-PLA2R antibody levels were significantly higher in patients with PMN (p < 0.001). No statistically significant differences were observed between patients with PMN and those with MCD, DKD, or other conditions. (C)Patients with PMN constituted a substantial proportion of individuals across various anti-PLA2R antibody levels, as indicated by the numbers displayed in the column chart.
Continuous monitoring of anti-PLA2R antibodies in antibody-negative PMN patients
Of the 91 PMN patients, 52 patients had follow-up data on anti-PLA2R antibodies, and 15.4% (8 out of 52) tested positive for anti-PLA2R antibodies greater than 20 RU/ml during follow-up. For instance, Patient 1 and Patient 2 were tested positive for anti-PLA2R antibody (ELISA) 2 and 4 months after renal biopsy, respectively. Additionally, The antibody titers of Patient 2 gradually decreased following RTX treatment. (Supplementary data, Fig. S1). The time intervals between the detection of anti-PLA2R antibodies greater than 20 RU/ml and kidney biopsy were 1.5, 2, 4, 5, and 9 months for PMN patients without treatment, and 17, 36, and 42 months for those receiving corticosteroids.
Detecting PLA2R-IgG and IgG4 based on TRFIA improves the sensitivity of diagnosing PMN
In this study, we tested serum samples from 121 patients using the TRFIA method, including 91 PMN and 30 non-PMN. Additionally, 54 non-PMN patients without serum samples were excluded from the study. Based on the manufacturer’s cutoff value, the sensitivity were 14.3% (13/91) for PLA2R-IgG-TRFIA and 34.1% (31/91) for PLA2R-IgG4-TRFIA, respectively (Table 2). All of the 30 non-PMN patients were negative, predicting a specificity of 100% for both methods. We categorized PMN patients with anti-PLA2R antibodies detected by ELISA into three groups based on antibody levels: 2–7 RU/ml (n = 48), 7–14 RU/ml (n = 25), and 14–20 RU/ml (n = 18). The sensitivity of PLA2R-IgG4-TRFIA was 22.9% (11/48), 36% (9/25), and 66.7% (12/18), significantly surpassing the sensitivity of PLA2R-IgG-TRFIA, which was 4.2%, 16%, and 38.9% respectively. PLA2R-IgG4-TRFIA is more effective in diagnosing PMN patients with lower anti-PLA2R antibody levels ranging from 2 to 20 RU/ml (Fig. 3A and B, p < 0.05). Notably, among the eight patients who were antibody negative at baseline but later tested positive during follow-up, three showed baseline anti-PLA2R antibody positivity in the PLA2R-IgG4-TRFIA test, and one showed positivity in the PLA2R-IgG-TRFIA test.
TFRIA has a stronger ability to diagnose PMN patients with lower PLA2R antibody levels. (A) Anti-PLA2R antibody was considered as positive when PLA2R-IgG-TRFIA ≥ 14RU/ml or PLA2R-IgG4-TRFIA ≥ 200 ng/ml. The number displayed above the column chart indicates the positive rate of the anti-PLA2R antibodies. PLA2R-IgG4-TRFIA demonstrated greater sensitivity across varying levels of anti-PLA2R antibodies (p < 0.05). (B) Scatter plot of anti-PLA2R antibody value by three methods. In the scatter plot, patients who tested positive for PLA2R-IgG-TRFIA are marked with red dots, those positive for PLA2R-IgG4-TRFIA are marked with green dots, and patients positive for both methods are indicated by yellow dots. PLA2R-IgG-TRFIA can diagnose patients with higher PLA2R-IgG-ELISA values, while PLA2R-IgG4-TRFIA had more sensitive in diagnosing patients with lower PLA2R-IgG-ELISA values. To account for the wide variation in PLA2R-IgG4-TRFIA values, we utilized log2 transformation for mapping purposes.
Comparison of the diagnostic ability of ELISA and TRFIA
The ELISA and TRFIA results showed moderate consistency in measuring serum anti-PLA2R antibodies in 121 patients. The Spearman correlation coefficient for PLA2R-IgG-TRFIA compared to PLA2R-IgG-ELISA was 0.50 (p < 0.001). In contrast, the coefficient for PLA2R-IgG4-TRFIA versus PLA2R-IgG-ELISA was 0.47 (p < 0.001). A strong correlation was observed between PLA2R-IgG-TRFIA and PLA2R-IgG4-TRFIA, with an rs value of 0.85 (p < 0.05). There were no significant correlations between anti-PLA2R antibody levels and clinical parameters, except for a negative correlation between PLA2R-IgG4-TRFIA and ALB at a low level (rs = -0.36, p < 0.001) (Fig. 4, Supplementary data, Table S1). PMN patients who tested positive for PLA2R-IgG4-TRFIA were older (57 vs 50, p = 0.044) and had lower levels of ALB (28.4 vs 32.4, p = 0.001), TBIL (8.6 vs 11.6, p = 0.011), DBIL (0.7 vs 0.9, p = 0.023), and C3 (0.9 vs 1.06, p = 0.003) (Supplementary data, Table S2).
Correlations between anti-PLA2R antibody value and clinical indicators. The Spearman correlation coefficient for PLA2R-IgG-TRFIA compared to PLA2R-IgG-ELISA is 0.50, while for PLA2R-IgG4-TRFIA, it is 0.47 (p < 0.001), whereas there was a strong correlation between PLA2R-IgG-TRFIA and PLA2R-IgG4-TRFIA (rs = 0.85, p < 0.05). No significant correlations were observed between anti-PLA2R antibody levels and clinical indicators, except for a weak negative correlation between PLA2R-IgG4-TRFIA and ALB (rs = −0.36, p < 0.001). The heatmap illustrates the correlations between anti-PLA2R antibodies and clinical indicators, with red ellipses indicating positive relationships and blue ellipses indicating negative relationships. *p < 0.05, **p < 0.01, ***p < 0.001.
Both ELISA and TRFIA indicated that anti-PLA2R antibody levels in PMN patients were significantly higher than those in non-PMN patients (ELISA p < 0.001, TRFIA p < 0.0001) (Fig. 5A, B, C). Furthermore, in the clinical performance study, TRFIA showed superior capability in distinguishing between PMN and non-PMN patients. The area under the curve (AUC) for PLA2R-IgG-TRFIA was 0.829 (95% CI: 0.752–0.905), while for PLA2R-IgG4-TRFIA, it was 0.814 (95% CI: 0.736–0.892). In contrast, the ELISA method achieved an AUC of 0.712 (95% CI: 0.594–0.830) (Fig. 5D, E, F).
Comparison of the identification ability of ELISA and TRFIA. (A–C) The anti-PLA2R antibody levels of PMN patients were significantly higher than that of non-PMN patients. The difference between PMN and non-PMN patients is more pronounced when using the TRFIA method. (D–F) The efficacy of ELISA and TRFIA in identifying PMN and non-PMN patients was analysis by ROC analysis, with AUC reaching 0.712 (ELISA), 0.829 (PLA2R-IgG-TRFIA) and 0.814 (PLA2R-IgG4-TEFIA), respectively. The p value for PLA2R-IgG-ELISA vs PLA2R-IgG-TFRIA was 0.078; the p value for PLA2R-IgG-ELISA vs PLA2R-IgG4-TRFIA was 0.128; the p value for PLA2R-IgG-TRFIA vs PLA2R-IgG4-TRFIA was 0.659. The adjusted cutoff values were as follows: 2.84 RU/ml for ELISA, 0.44 RU/ml for PLA2R-IgG-TRFIA, and 25.07 ng/ml for PLA2R-IgG4-TRFIA. The p-value for comparing PLA2R-IgG-TRFIA with PLA2R-IgG-ELISA was 0.078, while the p-value for comparing PLA2R-IgG4-TRFIA with PLA2R-IgG-ELISA was 0.12. AUC: area under the curve. ***p < 0.001, ****p < 0.0001.
Considering that anti-PLA2R antibody detected by TRFIA shows stronger capability than ELISA in diagnosing PMN, we further employed multivariate logistic regression to clarify the diagnostic ability of PLA2R-IgG-TFRIA and PLA2R-IgG4-TFRIA for PMN in 121 kidney patients. After fully adjusting for potential confounders, the positive association between PLA2R-IgG-TRFIA and the risk of PMN remained robust. This suggested that PLA2R-IgG-TRFIA is an independent diagnostic factor for PMN in kidney patients with low anti-PLA2R antibody levels. The detailed results of the multivariate logistic regression, including adjusted odds ratios and confidence intervals, are presented in Table 3.
Exploring and determining optimal cutoff values for ELISA and TRFIA
TRFIA enhanced the sensitivity of PMN diagnosis based on the manufacturer’s recommended cutoff values (Fig. 3 and Table 2). We analyzed the data to find the optimal cutoff values for ELISA and TRFIA, using the following lower borderline values: ELISA at 14 RU/ml, IgG-TRFIA at 7 RU/ml, and IgG4-TRFIA at 100 ng/ml. The results indicated that ELISA had a sensitivity of 19.8% and a specificity of 90%. In contrast, IgG-TRFIA showed a sensitivity of 26.4% and a specificity of 100%, while IgG4-TRFIA demonstrated a sensitivity of 48.4% and a specificity of 93.3%. ROC curve analysis determined that the optimal cutoff value for ELISA was 2.84 RU/ml, yielding a sensitivity of 50% and a specificity of 88%. The optimal cutoff value for IgG-TRFIA was 0.44 RU/ml with a sensitivity of 83.3% and a specificity of 82.4%. For IgG4-TRFIA, the optimal cutoff was 25.07 ng/ml, resulting in a sensitivity of 83.3% and a specificity of 76.9% (Table 2).
Discussion
This research explored the potential of TRFIA as a more sensitive diagnostic tool than traditional ELISA. Our findings showed that TRFIA was more sensitive and specific in identifying PMN patients. When using the cutoff value recommended by the manufacturer, PLA2R-IgG4-TRFIA demonstrated higher sensitivity and specificity. TRFIA achieved an AUC of 0.829 for PLA2R-IgG and 0.814 for PLA2R-IgG4, compared to 0.712 for ELISA, indicating its robust diagnostic performance. By establishing optimal cutoff values for accurate diagnosis, this research emphasizes the need for better diagnostic methods in clinical settings.
Anti-PLA2R antibody serve as a biomarker that can accurately diagnose PMN. Several detection methods for anti-PLA2R antibodies have been developed, such as western blotting7, immunofuorescence assay (IFA)13 and enzyme-linked immunosorbent assay (ELISA)24. The Euroimmun ELISA is widely used in clinical laboratories to quantitatively measure anti-PLA2R antibody in serum. The manufacturer’s cut-off value is recommended at 20 RU/ml, and values ≥ 20 RU/ml are considered positive and those < 14 RU/ml are considered negative. In previous clinical studies, this threshold of 20 RU/ml for PLA2R-IgG-ELISA demonstrates a specificity of approximately 94–98%, indicating that a positive result is highly indicative of PMN. However, the test sensitivity varies, typically ranging from 50 to 99.7% 5,14,15,20,22,25,26,27,28. Bobart et al. found that patients with ELISA results ranging from 2 to 20 RU/ml require further confirmation with IFA method29. However, 57.5% of patients with negative values via IFA were confirmed as MN via biopsy5,14,15,20,22,25,26,27,28. Therefore, adopting more sensitive methods is crucial for the differential diagnosis of patients with ELISA results between 2 and 20 RU/ml.
The study aimed to evaluate the diagnostic utility of TRFIA for detecting serum PLA2R antibody IgG and its IgG4 subclass in a cohort of biopsy-confirmed patients whose ELISA results ranging from 2 to 20 RU/ml. Our findings indicate that TRFIA significantly improves the sensitivity of detecting both total anti-PLA2R IgG and its IgG4 subclass compared to ELISA. Our study demonstrated that TRFIA exhibited a sensitivity of 34.1% for PLA2R-IgG4 and 13.1% for PLA2R-IgG when differentiating PMN from other nephropathies with low PLA2R-IgG levels. The method also had a high specificity, as confirmed by Huang et al.30. The high sensitivity of the TRFIA method resulted in a positive rate of 89.7% among patients confirmed to have PMN, compared to a 66.7% positive rate with ELISA23. Beyond its diagnostic advantages for PMN, PLA2R-IgG4-TRFIA has also been validated for its role in disease risk stratification31 and prognosis assessment32. Importantly, the combined detection of serum and urine PLA2R-IgG4-TRFIA improves the efficiency of PMN risk stratification, and can provide a better assessment of PMN monitoring33.This represents a significant advancement, as prior studies indicate that patients with nephrotic syndrome and low antibody titers may still require a kidney biopsy for diagnosis. TRFIA can improve clinical decision-making and patient outcomes while minimizing procedural risks.
It is necessary to adjust the optimal cutoff value to improve sensitivity and specificity. This study analyzed the sensitivity and specificity of TRFIA and ELISA methods in 121 patients with anti-PLA2R antibodies between 2 and 20 RU/ml. Besides the manufacturer’s recommended cutoff value, we found that when the cutoff for PLA2R-IgG4-TRFIA is set at 150 ng/ml, the sensitivity is 39.6% and specificity is 100%. At a cutoff value of 100 ng/ml, the sensitivity improves to 48.4%, while specificity decreases to 93.3%. The cutoff value for PLA2R-IgG-TRFIA is 7 RU/ml, yielding a sensitivity of 26.4% and a specificity of 100%. The cutoff value of PLA2R-IgG-ELISA is 14 RU/ml, the sensitivity/specificity are 19.8%/90%. According to ROC curve analysis, the adjusted cutoff values are 25.07 ng/ml for PLA2R-IgG4-TRFIA, 0.44 RU/ml for PLA2R-IgG-TRFIA, and 2.8 RU/ml for PLA2R-IgG-ELISA. The sensitivity of the three methods was further improved at the expense of specificity. Whether the optimal cutoff value is applicable to the patients with anti-PLA2R antibody ≥ 2 to 20 RU/ml is uncertain and requires further study.
Can serum anti-PLA2R antibody serve as a viable alternative to renal biopsy in diagnosing PMN? When deciding whether to perform a renal biopsy, it is essential to consider both the additional benefits and risks. Although patients with anti-PLA2R antibody ≥ 20 RU/ml can obtain additional pathological information through renal biopsy, such as combined diagnosis of IgAN, DKD, renal tubular injury, interstitial fibrosis, etc., but the prevalence of such cases is low33, and it does not significantly impact diagnosis and treatment. For patients with low anti-PLA2R antibody titer (< 20 RU/ml), it is advisable to perform a renal biopsy and stain the sample for the PLA2R antigen. Our study found that PLA2R-IgG4-TRFIA can re-diagnose 34.1% of PMN patients with high specificity when their anti-PLA2R antibody ranging from 2 to 20 RU/ml, enabling these patients to avoid renal biopsy and thus lowering both risk and economic burden. In addition, continuous monitoring of anti-PLA2R antibody can identify additional patients who test positive for the antibody, thereby avoiding the need for a renal biopsy. In our study, 15.4% (8/52) patients monitored had anti-PLA2R antibodies > 20 RU/ml during the follow-up process, aligning with previous studies34,35. These results highlight the necessity for a more sensitive testing method and ongoing monitoring of anti-PLA2R antibodies for suspected PMN patients who may be hesitant to undergo a renal biopsy. In this study, the manufacturer’s recommended cut-off value (PLA2R-IgG-ELISA ≥ 20 RU/ml; PLA2R-IgG-TRFIA ≥ 14 RU/ml; PLA2R-IgG4-TRFIA ≥ 200 ng/ml) showed very high specificity, and patients with positive antibodies may be considered for not undergoing renal biopsy.
Despite these promising findings, our study does have limitations that warrant consideration. The retrospective nature of our analysis and the small sample size may limit the applicability of our results. Furthermore, the lack of multicenter validation and long-term follow-up may weaken the reliability of our conclusions about the prognostic value of anti-PLA2R antibodies. To build on our findings, future research should focus on conducting larger, prospective studies for confirmation. Addressing these gaps will help us refine our understanding of PMN and improve diagnostic strategies.
In conclusion, our findings highlight that time-resolved fluoroimmunoassay (TRFIA) is a more sensitive diagnostic tool for PMN than ELISA. The ability of TRFIA to detect both PLA2R-IgG and PLA2R-IgG4 may significantly reduce the need for renal biopsies, thereby alleviating patient discomfort and risk. By enhancing the sensitivity of detecting PLA2R antibodies, especially in patients with low antibody titers, we can offer a more effective approach to identifying and managing PMN.
Data availability
The data that support the findings of this study are not publicly available due to privacy reasons, but are available from the corresponding author upon reasonable request.
References
Sethi, S. & Fervenza, F. C. Membranous nephropathy-diagnosis and identification of target antigens. Nephrol. Dial. Transpl. 39(4), 600–606 (2024).
Hoxha, E., Reinhard, L. & Stahl, R. Membranous nephropathy: New pathogenic mechanisms and their clinical implications. Nat. Rev. Nephrol. 18(7), 466–478 (2022).
Radhakrishnan, Y., Zand, L., Sethi, S. & Fervenza, F. C. Membranous nephropathy treatment standard. Nephrol. Dial. Transpl. 39(3), 403–413 (2024).
Ronco, P. et al. Membranous nephropathy. Nat. Rev. Dis. Primers 7(1), 69 (2021).
Ragy, O. et al. Can use of the serum anti-PLA2R antibody negate the need for a renal biopsy in primary membranous nephropathy?. PLoS ONE 18(2), e0281726 (2023).
Roccatello, D., Fenoglio, R. & Sciascia, S. The role of kidney biopsy in the diagnosis of membranous nephropathy. Clin. Kidney J. 17(10), sfae292 (2024).
Beck, L. H. Jr. et al. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N. Engl. J. Med. 361(1), 11–21 (2009).
Avasare, R., Andeen, N. & Beck, L. Novel antigens and clinical updates in membranous nephropathy. Annu. Rev. Med. 75, 219–332 (2024).
Avasare, R. S. et al. Characteristics and outcomes of NELL1 membranous nephropathy in lipoic acid users and nonusers. Kidney Int. Rep. 9(5), 1379–1386 (2024).
Fu, N., Yuan, S., Yang, G., Li, H. & Wang, T. Concurrent glomerular PCDH7 deposits in PLA2R-associated membranous nephropathy. CEN Case Rep. 13(4), 297–301 (2024).
Sethi, S. New ‘Antigens’ in membranous nephropathy. J. Am. Soc. Nephrol. 32(2), 268–278 (2021).
KDIGO. Clinical practice guideline for the management of glomerular diseases. Kidney Int. 100(4S), S1–S276 (2021).
Hoxha, E. et al. An immunofluorescence test for phospholipase-A(2)-receptor antibodies and its clinical usefulness in patients with membranous glomerulonephritis. Nephrol. Dial. Transpl. 26(8), 2526–2532 (2011).
Chen, J. et al. Optimal value for serum anti-PLA2R antibody in primary membranous nephropathy: A multicenter observational study. Am. J. Nephrol. 53(2–3), 129–138 (2022).
Wu, X., Liu, L., Guo, Y. & Yang, L. Clinical value of a serum anti-PLA2R antibody in the diagnosis and monitoring of primary membranous nephropathy in adults. Int. J. Nephrol. Renovasc. Dis. 11, 241–247 (2018).
Mukherjee, S. et al. Utility of serum anti-phospholipase A2 receptor antibody as a biomarker in membranous nephropathy: An experience from a tertiary care center of Eastern India. Indian J. Pathol. Microbiol. 66(2), 269–277 (2023).
Porcelli, B. et al. Diagnostic accuracy of anti-phospholipase A2 receptor (PLA2R) antibodies in idiopathic membranous nephropathy: An Italian experience. J. Nephrol. 34(2), 573–579 (2021).
Liu, Y. et al. Serum anti-PLA2R antibody as a diagnostic biomarker of idiopathic membranous nephropathy: The optimal cut-off value for Chinese patients. Clin. Chim. Acta 476, 9–14 (2018).
Tampoia, M. et al. Definition of a new cut-off for the anti-phospholipase A2 receptor (PLA2R) autoantibody immunoassay in patients affected by idiopathic membranous nephropathy. J. Nephrol. 31(6), 899–905 (2018).
Ragy, O. et al. PLA2R autoantibodies, a multifaceted biomarker in nephrotic syndrome and membranous nephropathy. Nephrol. Dial. Transpl. 16, gfaf012 (2025).
Dahnrich, C. et al. Development of a standardized chemiluminescence immunoassay for the detection of autoantibodies against human M-Type phospholipase A2 receptor in primary membranous nephropathy. Kidney Int. Rep. 5(2), 182–188 (2020).
Guo, H. et al. The cutoff value and prognosis of anti-PLA2R antibody for idiopathic membranous nephropathy: A single-center retrospective study in China. Ren. Fail. 45(2), 2253922 (2023).
Huang, B. et al. A novel time-resolved fluoroimmunoassay for the quantitative detection of antibodies against the phospholipase A2 receptor. Sci. Rep. 7, 46096 (2017).
Dähnrich, C. et al. Development of a standardized ELISA for the determination of autoantibodies against human M-type phospholipase A2 receptor in primary membranous nephropathy. Clin. Chim. Acta 421, 213–218 (2013).
Zhou, K. et al. Predictive value of the domain specific PLA2R antibodies for clinical remission in patients with primary membranous nephropathy: A retrospective study. PLoS ONE 19(5), e0302100 (2024).
Kukuy, O. L. et al. The prognostic value of anti-PLA2R antibodies levels in primary membranous nephropathy. Int. J. Mol. Sci. 24(10), 9051 (2023).
Bajcsi, D. et al. The value of PLA2R antigen and IgG subclass staining relative to anti-PLA2R seropositivity in the differential diagnosis of membranous nephropathy. BMC Nephrol. 24(1), 230 (2023).
Lu, S. et al. Diagnostic value of renal biopsy in anti-phospholipase A2 receptor antibody-positive patients with proteinuria in China. Sci. Rep. 14(1), 2907 (2024).
Bobart, S. A. et al. Noninvasive diagnosis of primary membranous nephropathy using phospholipase A2 receptor antibodies. Kidney Int. 95(2), 429–438 (2019).
Huang, B. et al. Phospholipase A2 receptor antibody IgG4 subclass improves sensitivity and specificity in the diagnosis of idiopathic membranous nephropathy. Kidney Blood Press. Res. 44(4), 848–857 (2019).
Liu, X. et al. A PLA2R-IgG4 antibody-based predictive model for assessing risk stratification of idiopathic membranous nephropathy. J. Healthc. Eng. 2021, 1521013 (2021).
Huang, Y. et al. PLA2R-IgG4 antibody as a predictive biomarker of treatment effectiveness and prognostic evaluation in patients with idiopathic membranous nephropathy: A retrospective study. PeerJ 10, e14193 (2022).
Zheng, T. et al. Urine PLA2R antibody detection in hazard stratification of PLA2R-associated membranous nephropathy. Arch. Pathol. Lab. Med. https://doi.org/10.5858/arpa.2024-0161-OA (2024).
Van de Logt, A. E., Hofstra, J. M. & Wetzels, J. F. Serum anti-PLA2R antibodies can be initially absent in idiopathic membranous nephropathy: Seroconversion after prolonged follow-up. Kidney Int. 87(6), 1263–1264 (2015).
Ramachandran, R., Kumar, V., Nada, R. & Jha, V. Serial monitoring of anti-PLA2R in initial PLA2R-negative patients with primary membranous nephropathy. Kidney Int. 88(5), 1198–1199 (2015).
Acknowledgements
Thank you to Zhe Wang for his great help in the production of figures.
Funding
This study was supported by the Clinical Cohort Construction Program of Peking University Third Hospital (BYSYDL2021017) and the National Natural Science Foundation of China (82070736).
Author information
Authors and Affiliations
Contributions
Z.L.D., Y.W. and D.X.Z. designed the study, confirm the authenticity of all the raw data, revised the manuscript and finalized the article. Z.L.D. collected serum samples and clinical data of patients, and wrote the manuscript. Y.L.Q and Y.M.G contributed to data analysis. Y.T.L. and T.Y.Z. performed the experiments. B.H. reviewed drafts of the article. All authors read and approved the final draft.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Deng, Z., Qu, Y., Liu, Y. et al. Time-resolved fluoroimmunoassay improves sensitivity in the diagnosis of primary membranous nephropathy with low phospholipase A2 receptor antibody titer. Sci Rep 15, 34403 (2025). https://doi.org/10.1038/s41598-025-17354-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17354-x