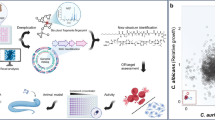
Abstract
The emerging fungal pathogen C. auris has recently been recognized as a global threat due to its ability to cause nosocomial and invasive candidemia with high mortality rates. C. auris has been widely reported for its multidrug resistance to antifungals, further complicating its clinical management. In this context, there is an urgent need for the research and development of more efficient alternatives for environmental control, disinfection, and treatment of C. auris. In this study, the antifungal properties of bio-oil aqueous phases (BOAP) derived from the pyrolysis of Cocos nucifera (coconut), Syagrus coronata (licuri), and Terminalia catappa (almond), with traditional antimicrobial efficiency, were evaluated against C. auris., All three BOAPs formulations exhibited remarkable antifungal activity, particularly the licuri BOAP I, with minimum inhibitory concentration (MIC) values ranging from 0.035 to 0.56 mg/mL, demonstrating higher efficacy. We have also evaluated the antifungal activity of volatile compounds in BOAPs, with licuri BOAP I showing the most significant inhibition halos in volatilization tests. Mass spectrometry analysis identified phenolic compounds, ketones, and aldehydes in the licuri BOAP I, with sequential organic solvent extractions further isolating active antifungal fractions. Chloroform-extractable compounds of licuri BOAP displayed the most potent antifungal activity, with MIC values of 0.14 mg/mL. In conclusion, our findings indicate the potential of bio-oils as natural antifungals, particularly the licuri-derived extracts, which could be further developed into effective treatments against resistant fungi. Furthermore, the in vivo efficacy of volatile and water-soluble active compounds could be assessed upon optimization of the extraction processes, thereby providing more sustainable and environment-friendly antifungal strategies.
Similar content being viewed by others
Introduction
Candida auris is an emerging fungus that has recently been classified as a critical threat to public health due to the increasing number of healthcare-associated infections worldwide, high mortality rates, and drug resistance1,2. This fungus was first documented in 2009, upon isolation from a patient’s ear canal in Japan, and subsequently reported as a novel Candida species2. However, the significant rise in C. auris infection rates, with several outbreaks recently reported across all continents, including recent ones in Latin American countries, is a deeply concerning scenario3, leading the World Health Organization (WHO) to include C. auris on the fungal pathogens priority list to guide public health actions4.
C. auris expresses various virulence factors and resistance traits, that collectively enable the fungus to survive in the e nvironment for extended periods, even after exposure to multiple disinfectants5. This striking environmental resilience is primarily attributed to its ability to produce biofilms on surfaces and materials, making it a significant risk to patients with comorbidities, immunosuppressed individuals, and those hospitalized in intensive care units, often due to contaminated medical devices6.
Recently, C. auris has also been recognized for its multidrug-resistance to various classes of antifungals used therapeutically worldwide, including fluconazole and other azoles, echinocandins, and amphotericin B, further complicating its clinical management7. Given this emergency and concerning scenario, it is imperative to research and develop more effective strategies for the environmental control and elimination of C. auris, in addition to novel therapeutic approaches for the clinical management of C. auris infections8.
In this context, the search for antifungal alternatives is crucial, with bioprospecting of natural products emerging as a promising alternative as biodiversity remains a rich source of a wide array of cost-effective and promptly available bioactive compounds9. Compounds obtained from plants through various extraction processes have already demonstrated efficacy in inhibiting the growth of C. albicans, and thus represent a potential new source of antifungals10.
Among the wide diversity of bioactive compounds found in plant extracts and essential oils, mixtures of volatile and odoriferous substances are well-documented for their antimicrobial activity11. Essential oils derived from plants are a valuable source of unique and complex biosynthetic structures, warranting further investigation as a potential source of new antifungals. Numerous studies in the literature corroborate the efficacy of selected plant extracts and essential oils against the growth of C. albicans12,13,14,15.
Notably, the oil derived from licuri (Syagrus coronata), a palm tree native to the caatinga biome present throughout the entire Brazilian eastern coast, stands out as an edible oilseed with significant oil production capacity and diverse clinical applications16. Aqueous fractions obtained through pyrolysis of licuri biomass have demonstrated insecticidal activity17. Another extract showing potential antifungal activity comes from the fruit of the coconut tree (Cocos nucifera). Bio-oil from the pyrolysis process of coconut biomass serves as a precursor in the medicinal field, exhibiting antimicrobial and antioxidant properties18,19. Additionally, it is associated with antifungal activity against C. albicans isolates in vitro20. A third noteworthy alternative comes from the almond Terminalia catappa fruit, which is composed of several chemical constituents. Extracts from its leaves exhibit pharmacological activity, including in vitro antifungal activity against C. albicans21.
The development and application of sustainable and environmentally friendly alternatives and solutions to address global challenges are a crucial points to be considered in modern scientific research. In this regard, pyrolysis involves the thermochemical decomposition of organic matter present in the feedstock by elevated temperatures in the absence of oxygen, which breaks down long carbon chains, resulting in a mixture of solid, liquid, and gaseous compounds22,23. Thus, it consists of an interesting method to convert plant-based biomass into new products, such as biochar, aqueous fractions of extracts and plant oils. Other advantages of the pyrolysis process include its simplicity and low cost to assemble/build the pyrolyzer devices, in addition to being a promising technology and sustainable approach for converting waste biomass into potentially valuable products22,24.
Historically, the pyrolysis process has been widely used for various applications. Practitioners of traditional Indian medicine use oily substances extracted through pyrolysis of plant stems to treat several illnesses25. Aboriginal people living away from the coast of Australia used pyrolysis of high salt and potassium-rich plants to produce ashes as a substitute for salt, while also accessing other essential dietary components and producing medicine to assist travelers on long journeys26,27. First Nation people employed pyrolysis of twigs and leaves to produce quicklime, calcium oxide ashes that could alkalinize solutions, facilitating the release of medicines and serving as excipients for chewing or transdermal medical applications, thereby enhancing the absorption into the bloodstream28. Charcoal, an important product of pyrolysis, can also be used in pigments and adhesives, contaminant immobilization, electric sensors and biosensors and drug delivery, with direct medical relevance29,30 and waste treatment and reuse, as a sustainable and protective approach to environmental protection31.
Upon the pyrolysis process, the first partition is usually performed to isolate the water-soluble phase (bio-oil aqueous phase, BOAP). These fractions often contain compounds with classical antimicrobial functions that can target key fungal metabolic pathways and cellular structures, offering innovative alternatives to address the rising issue of antimicrobial resistance23,32,33,34. Additionally, one of the main advantages of pyrolysis and bio-oil production is the feasibility of the process, the low production cost, and the use of waste mass as renewable resources, yielding encouraging potential applications as novel disinfectants and antifungal agents. Therefore, the present study offers promising new alternatives against C. auris, using sustainable approaches to generate potential bioproducts for environmental controlling/disinfection, or as a potential support to existing antifungals, which could reduce their effective concentration synergistically, thus minimizing both their in vivo toxicity and environmental impact.
Results
Growth and viability of C. auris strains in RPMI-MOPS
The selected C. auris strains (Table 1) were initially evaluated for their ability to grow in the standard RPMI-MOPS medium and conditions used for the antifungal susceptibility testing. Growth curves, determined by absorbance readings at 600 nm, demonstrated that the C. auris MMC1 strain exhibited similar growth to the C. albicans SC5314 reference strain (Fig. 1A). The growth of the other C. auris strains (Fig. 1A) was compared to that of the MMC1 strain. The C. auris MMC2 strain exhibited the lowest absorbance values within the first 24 h compared to the other strains; however, after 24 h, it achieved full growth, reaching similar final absorbance values (Fig. 1A and B). All other strains grew successfully in the RPMI-MOPS medium, allowing for subsequent susceptibility testing to be performed. By integrating the growth curve, the cumulative growth (area under curve) of each strain was compared to the C. albicans SC5314 reference strain (Fig. 1B). Overall, no substantial differences were observed when comparing the calculated average growth of the C. auris strains.
Comparison of (A) planktonic growth curves and (B) the calculated area under curve for Candida auris isolates (Table 1) and C. albicans (SC5314) reference strain used as a control. Planktonic cells were incubated, and absorbances were taken at 18, 24, 40, and 48 h to illustrate the distinct growth profiles of each species/isolate/strain. The integrated cumulative growth over the entire time period was assessed by calculating the area under the curve and compared with the MMC1 strain. Dashed lines represent the average growth for all the C. auris strains. Analyses were performed with biological triplicates with two technical replicates each.
Effect of bio-oil aqueous phase I from Cocos nucifera (coconut), Syagrus coronata (licuri) and Terminalia catappa (almond) on the growth of C. auris
The reference C. albicans SC5314 strain and the C. auris strains (Table 1) were initially tested against itraconazole as controls for antifungal susceptibility testing. The MIC value of the C. albicans SC5314 reference strain was 0.13 μg/mL, while for the C. auris strains and isolates, MIC values ranged from 0.063 to 1 μg/mL, with the MMC2 isolate displaying the highest susceptibility (Table 2 and Fig. 3A).
Evaluations with the C. albicans SC5314 reference strain demonstrated MIC values of 0.56 mg/mL for all three bio-oil aqueous phase I (BOAP I) fractions (Fig. 2A, B, and C, respectively, and Table 2). The fluconazole-resistant C. auris MMC1 strain displayed MIC values of 0.28 mg/mL for C. nucifera (coconut) BOAP I and 0.14 mg/mL for S. coronata (licuri) and T. catappa (almond) BOAPs I (Figs. 2D, E, and F and Table 2). Conversely, the fluconazole-sensitive C. auris MMC2 strain demonstrated MIC values of 0.14 mg/mL for coconut BOAP I and 0.07 mg/mL for licuri and almond BOAPs I, exactly half of the values of the C. auris MMC1 strain (Fig. 2G, H and I and Table 2).
Growth curves of (A-C) C. albicans (SC5314), (D-F) C. auris MMC1, and (G-I) C. auris MMC2 isolates in the presence of bio-oil aqueous phase I (BOAP I, from 4.5 to 0.009 mg/mL) of C. nucifera (coconut; left panel, A, D, and G), S. coronata (licuri; middle panel, B, E, and H) and T. catappa (almond; right panel, C, F, and I). Absorbances were measured at 18, 24, 40, and 48 h. Analyses were performed in three independent experiments, with three technical replicates each. The minimum inhibitory concentration (MIC) for each of the three extracts evaluated (coconut, licuri, and almond) was determined as the curve (concentration) where all the absorbances were below the absence of growth “cut off”, defined as three times the average of negative growth controls (dead fungal inoculum). Licuri showed the best performance; additionally, distinct batches of each licuri bio-oil were tested, with similar MIC values for the MMC1 and MMC2 strains.
The three BOAP I extracts were further evaluated against an additional 10 C. auris strains (Table 1), along with itraconazole as a control (Fig. 3A). MIC values for the C. nucifera (coconut) BOAP I ranged from 0.14 to 0.56 mg/mL, with 54% of the strains displaying MIC of 0.28 mg/mL (Fig. 3B and Table 2). MIC values for the S. coronata (licuri) BOAP I ranged from 0.035 to 0.56 mg/mL, with 38% of the strains displaying an MIC of 0.28 mg/mL (Fig. 3C and Table 2). MIC values for T. catappa (almond) BOAP I ranged from 0.070 to 0.56 mg/mL, with 46% of the strains displaying MIC values of 0.56 mg/mL (Fig. 3D and Table 2). Overall, MIC values for the BOAP I extracts were compared among the strains, and correlations were observed between the S. coronata (licuri) BOAP I and coconut BOAP I MIC values (Pearson r = 0.71, p = 0.007; Supplementary Figure 1), and the S. coronata (licuri) BOAP I and T. catappa (almond) BOAP I MIC values (Pearson r = 0.82, p = 0.001; Supplementary Figure 1). These correlation indices indicate lower MIC values and the best antifungal performance of the licuri BOAP I extract against C. auris.
Graphs illustrating the frequency of MIC values (0.035, 0.070, 0.14, 0.28, 0.56 mg/mL) obtained for each of the C. auris isolates (Table 1) and the C. albicans SC5314 reference strain facing the (A) itraconazole control, and (B-D) BOAP I from (B) C. nucifera (coconut), (C) S. coronata (licuri) and (D) T. catappa (almond). Licuri exhibited the highest frequency at the lowest MIC values, demonstrating the highest antifungal efficacy. Analyses were performed in three independent experiments, with three technical replicates each.
S. coronata (licuri) BOAP I impact on C. auris biofilms
We further evaluated the antifungal activity of the S. coronata (licuri) BOAP I on C. auris biofilms. For this, both MMC1 and MMC2 isolates of C. auris were seeded on plates under standard biofilm formation conditions, with different concentrations of amphotericin B and itraconazole (as controls) and the licuri BOAP I. Concentrations of licuri BOAP I as low as 0.28 mg/mL and 0.14 mg/mL impacted the formation and viability of biofilms of C. auris MMC1 and MMC2 isolates, respectively (Figs. 4A and B). In turn, for mature biofilms, concentrations of licuri BOAP I as low as 0.56 mg/mL were effective against both isolates (Figs. 4C and D).
Activity of S. coronata (licuri) BOAP I against biofilms of C. auris. (A-B) C. auris isolates (A) MMC1 and (B) MMC2 were seeded on microplates for biofilm formation (1 × 106 cells/mL) and incubated with different concentrations of the licuri bio-oil aqueous phase I (BOAP I, 4.5–0.009 mg/mL), amphotericin B and itraconazole as controls (16–0.0031 μg/mL). (C-D) C. auris mature biofilms of the (C) MMC1 and (D) MMC2 isolates were incubated with distinct concentrations of the licuri bio-oil aqueous phase I (BOAP I, 4.5–0.009 mg/mL) and amphotericin and itraconazole as controls (16–0.0031 μg/mL). The percentage of viability (%) was calculated using the Alamar blue metabolization (Thermo Scientific), dividing the absorbance of each treatment by the absorbance of the positive control for biofilm formation in the absence of drug/treatment (black bars, 100% viability). Results are the average of three independent experiments performed in triplicates. Asterisks denote the one-way ANOVA test using the Dunnet post-test (*p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001).
Antifungal properties of BOAP I volatile compounds
The antifungal properties of the volatile compounds in the BOAP I were detected in a disk volatilization test performed in Petri dishes due to their evaporation at 37 °C. The diameters of the growth inhibition zones were measured after 48 h of incubation and compared for both C. auris strains. For the MMC1, 10 mm disks embedded with either C. nucifera (coconut), S. coronata (licuri), or T. catappa (almond) BOAP I placed on the lid of the Petri dishes resulted in inhibition halos of 2.8 cm (p < 0.01), 3.1 cm and 2.2 cm in diameter (p < 0.05 for both), respectively (Fig. 5C), compared to no halo formation in the PBS diluent control. When counting the CFUs observed on the entire plate, despite a slight decrease, no significant difference was observed compared to the control (Fig. 5D, p > 0.05). For the MMC2 strain, coconut and licuri BOAPs produced similar halos (3.4 and 3.3 cm, respectively; p < 0.05), in comparison to 1.7 cm (p < 0.001) of the almond BOAP I and the absence of halo for the PBS control (Fig. 5E). Regarding the number of colonies observed on the entire plate, all three BOAPs decreased the number of colonies, with the best performance observed for the S. coronata (licuri) BOAP I (35% inhibition, p < 0.01; Fig. 5F).
Disk volatilization test of the BOAP I from C. nucifera (coconut), S. coronata (licuri), and T. catappa (almond) extracted upon pyrolysis. (A) The volatilization test assembly, showing the fungal inoculum applied to the surface of Sabouraud agar plates (light grey lawn) and the disk soaked with the BOAP I (blue disk) placed on the center of the lid. (B) The Petri dishes are incubated with the lids facing down to promote the volatilization of compounds toward the seeded medium (arrows indicating volatilization), which inhibits fungal growth (as indicated by a dark grey lawn), resulting in a growth inhibition halo. (C-E) Fungal growth inhibition halo for C. auris strains (C) MMC1 and (E) MMC2 after disk volatilization tests with the coconut, licuri, and almond BOAP I on Sabouraud agar plates. (D-F) Number of colony-forming units (log CFU/mL) on the plates after the disk volatilization tests with the coconut, licuri, and almond tested for the C. auris strains (D) MMC1 and (F) MMC2. ”nd”: not detected. Results are the average of three independent experiments.
Mass spectrometry determination of the composition of the S. coronata (licuri) BOAP I
Given the inhibitory effect of S. coronata (licuri) BOAP I over planktonic and biofilm forms of C. auris MMC1 and MMC2 isolates, we performed mass spectrometry to determine its accurate composition. Despite the sample’s chemical complexity, 44 compounds displayed a peak area percentage above 0.05% (Table 3) and up to a retention time of 34.773 s. Among the 11 compounds that exhibit the highest peak area percentage, 5 were phenolic compounds (totalizing 51.51% area), including phenol and 2,6-dimethoxy-phenol, followed by ketones (7.51%) and aldehyde (2.86%,Supplementary Table 1). Additionally, their boiling point ranged from 88 to 280 °C, while their vapor pressure at 13–25 °C ranged from 0.1 to 56.8 mm Hg (Supplementary Table 1). All these compounds were water-soluble, with solubility values ranging from 6.13 to 1000 mg/mL (Supplementary Table 1).
Sequential extraction of the S. coronata (licuri) BOAP I with organic solvents
Since the S. coronata (licuri) bio-oil aqueous phase I (BOAP I) provided the best antifungal results against C. auris, we performed sequential organic solvent extraction following the methodology previously described by Ren et. al.34. Controls performed with blank tubes and evaporated solvents (considering the extraction volume used for each fractionation step) showed no impact on the growth of C. auris isolates. From the crude bio-oil and the initial water extraction used to generate BOAP I, subsequent extraction steps with organic solvents of increasing polarity were conducted (extraction yields are shown inSupplementary Table 2). The BOAPs and their respective organic phases were separated, dried, and tested against both C. auris MMC1 and MMC2 isolates (Fig. 6 and Table 4). For both isolates, BOAP I exhibited MIC values of 0.14 mg/mL for MMC1 and 0.07 mg/mL for MMC2, as previously mentioned. Following the first fractionation with hexane, BOAP II MIC values increased to 0.56 mg/mL for MMC1 and 0.14 mg/mL for MMC2. However, the subsequent extraction with chloroform produced a chloroform organic phase (COP), with comparable activity, showing MIC values of 0.14 mg/mL for both isolates. The resulting aqueous phase after chloroform partition, BOAP III, exhibited higher MIC values of 2.25 mg/mL for MMC1 and 0.28 mg/mL for MMC2, suggesting that residual active compounds were highly effective against the latter isolate. The final ethyl acetate extraction indicated that the remaining compounds with antifungal activity were extracted into the ethyl acetate organic phase (EAOP), with MIC values of 0.56 and 0.28 mg/mL, respectively, while the final aqueous phase (BOAP IV) displayed higher MIC values of 2.25 mg/mL for both isolates. Consequently, most antifungal compounds in the licuri bio-oil aqueous phase could be potentially extracted with chloroform.
Flowchart of the detailed fractionation process for S. coronata (licuri) bio-oil obtained through pyrolysis. BOOP: bio-oil organic phase; BOAP I: bio-oil aqueous phase I; BOAP II: bio-oil aqueous phase II; BOAP III: bio-oil aqueous phase III; BOAP IV: bio-oil aqueous phase IV; HOP: hexane organic phase; COP: chloroform organic phase; EAOP: ethyl acetate organic phase.
To assess the fungistatic/fungicidal activity of each fraction at their respective MICs, CFU counts were performed, and the growth inhibition percentages of the fractionated extracts were calculated (Fig. 7A and B, for MMC1 and MMC2 C. auris isolates, respectively, and Table 3). The EAOP exhibited fungicidal activity against MMC1 and MMC2, at their respective MICs of 0.56 mg/mL and 0.28 mg/mL. The BOAP IV was also fungicidal, but required higher MICs (2.25 mg/mL; Fig. 7A and B, respectively).
Colony forming units (log CFU/mL) at the minimum inhibitory concentration (MIC) for each BOAP I-IV and their respective organic phases: HOP: hexane organic phase; COP: chloroform organic phase and EAOP: ethyl acetate organic phase, obtained after S. coronata (licuri) pyrolysis, against C. auris (A) MMC1 and (B) MMC2 isolates. ”nd”: not detected. Bars represent the average of three independent experiments. Asterisks denote the one-way ANOVA test, using the Dunnet post-test (**p < 0.01 and ****p < 0.0001).
The minimal fungicidal concentration (MFC), which fully inhibits C. auris growth, was determined for all fractions. Lower MFC values were observed for BOAP I (0.56 mg/mL and 0.14 mg/mL, for MMC1 and MMC2 isolates, respectively) and COP (0.56 and 0.28 mg/mL, respectively). The MFC values for EAOP (0.56 and 0.28 mg/mL, respectively) were also consistent with COP, indicating that some fungicidal organic compounds remained in this phase (Table 3).
Comparison of the antifungal activity of volatile compounds in the S. coronata (licuri) BOAPs upon sequential organic extraction
Since the primary goal of fractionation was to identify the fractions with the highest antifungal activity in ascending order of polarity in solution, we also determined the activity of the volatile compounds in each S. coronata (licuri) BOAP fraction obtained. For the MMC1 isolate, BOAP I produced an inhibition growth halo of 3.2 cm, followed by 2.0 cm inhibition halo of for BOAP II and a 1.5 cm for BOAP III (Fig. 8A). Nevertheless, BOAP IV and COP extracts showed no growth inhibition halo. BOAP I, BOAP II, and BOAP III significantly reduced CFU counts in the remaining growth area of the plates (Fig. 8B). For the MMC2 isolate, BOAP I produced an inhibition halo of 3.3 cm, with halos of 3.6 and 2.1 cm for BOAP II and BOAP III, respectively (Fig. 8C). Moreover, BOAP I, BOAP II, and BOAP III statistically reduced the CFU numbers for this isolate (Fig. 8D).
Disk volatilization test of the fractions with the most pronounced antifungal activity of licuri (S. coronata) BOAPs and the chloroform organic phase, COP) against the MMC1 and MMC2 isolates of C. auris. (A) Inhibition halo and (B) colony-forming units on Petri dishes for the MMC1 isolate of C. auris. (C) Inhibition halo and (D) colony-forming units on Petri dishes for the MMC2 isolate of C. auris. Bars represent the average of three independent experiments. “nd”: not detected. Asterisks denote the one-way ANOVA test using the Dunnet post-test (*p < 0.05, **p < 0.01 and ****p < 0.0001).
Discussion
In recent years, there has been a significant increase in the number of systemic candidiasis cases attributed to the emerging fungus C. auris in healthcare-associated settings37. The clinical C. auris isolates frequently manifest resistance to one or more classes (multidrug-resistance) of antifungal agents, such as azoles, echinocandins, and amphotericin B, highlighting the urgent need for research and development of novel antifungal alternatives. In this regard, essential oils and bio-oils are natural compounds that constitute promising antifungal alternatives, due to their inherently lower toxicity compared to conventionally used antifungals38.
The literature provides solid evidence that supports their antimicrobial efficacy39, with many reports describing the products extracted from bio-oils as folk medicine antifungals, exemplifying their ethnopharmacological relevance23,33,40. For example, the simple pyrolysis of paper produced extracts containing furans, furanone, carboxylic acids, aldehydes and ketones which exhibited antifungal activity against Microsporum canis and Trichophyton longifusus with MICs of 0.28 and 0.36 mg/mL41; in addition to cellulose, pyrolysis of glucose, sucrose and starch also exhibited antifungal activity42.
The growth and viability of C. auris strains in RPMI-MOPS medium provide a foundational understanding for subsequent susceptibility testing. We tested a panel of C. auris isolates for their growth rates, including the first isolate described in 200943 and several others documented in the literature, most resistant to one or more classes of antifungals. MMC1 and MMC2 strains have been used in several studies due to their distinct resistance patterns to fluconazole, with one being highly resistant and the other sensitive2,35,36,44. The observation that the C. auris strains achieved full growth comparable to the C. albicans SC5314 reference strain confirms the adequacy of RPMI-MOPS for evaluating C. auris antifungal susceptibility.
From the pyrolysis process, the crude bio-oil obtained is usually a complex mixture that undergoes aqueous phase separation to obtain the bio-oil aqueous phase (BOAP I), a pale brown liquid containing about 91% (w/v) of water (data not shown). BOAP I demonstrated antifungal activity, with overall MICs ranging from 0.14–0.56 mg/mL for coconut, 0.035–0.56 mg/mL for licuri, and 0.070–0.56 mg/mL for almond. The observed correlations between the MIC values of different BOAP I extracts suggest consistent antifungal performance, with licuri BOAP I demonstrating superior efficacy against C. auris.The best activity observed for licuri BOAP I is supported by previous studies by Souza et. al45., which described that essential oils derived from S. coronata exhibited notable antimicrobial effectiveness against Staphylococcus aureus, including those with biofilm-forming abilities and multidrug-resistant phenotypes. This outcome can be attributed to the predominant presence of medium-chain fatty acids as the main components of the essential oils from S. coronata. These medium-chain fatty acids have been previously recognized as bioactive constituents and demonstrated antibacterial and antifungal properties46. Their lipophilic nature enables interaction with lipid structures in the cell membrane, resulting in membrane permeability and fluidity alterations. Consequently, the cell membranes undergo structural damage, ultimately culminating in cell death32,33,47,48.
A study conducted by Hughes et al.49 reported that the MIC of initial aqueous extracts from S. coronata against Bacillus cereus and S. aureus ranged from 0.19 to 0.78 mg/mL. These extracts exhibited robust antimicrobial activity, which was correlated to the presence of phenolic compounds within the extracted fractions. These compounds could form complexes with the microorganism’s secreted or surface proteins, leading to cell death49.
The literature indicates that most bio-oils aqueous phases from the pyrolytic process consist of organic volatile compounds, such as polyphenols. As indicated in Table 3, the primary identified compounds obtained after the pyrolysis process of S. coronata (licuri) were phenolic compounds, such as phenol and 2,6-dimethoxy-phenol, and ketones. These compounds often display relatively high vapor pressure values, allowing them to vaporize quickly at room temperature, in addition to displaying relatively low boiling points50. This suggests they are well-suited for aqueous extraction and potential volatilization, corroborating their applications to effectively inhibiting fungal growth.
For both C. auris strains evaluated in the volatilization tests, the BOAPs exhibited significantly larger halo zone diameters than the control group. The percentage of halo growth inhibition aligns with the expected results, as previous studies on using essential oils in filamentous fungi have shown their potent antifungal activity in the volatile phase51. Additionally, recent studies have demonstrated the formation of notable inhibition halos through volatilization tests conducted with bio-oils against C. auris strains52. A separate study investigated the activity of seven plant-derived polyphenols against C. auris and C. albicans. The results indicated that ellagic acid and caffeic acid phenethyl ester, both polyphenols, demonstrated more effective antifungal properties against both C. auris and C. albicans53.
Since the BOAPs obtained through pyrolysis hold significant potential for various applications, including the use as an effective antifungal agent, we performed a sequential organic solvent fractionation on the S. coronata (licuri) BOAP, to further understand the activity of their antifungal components and increase the effectiveness of the extraction process. Notably, the BOAP I and BOAP II aqueous fractions demonstrated the highest efficacy among the sequential aqueous phases in inhibiting fungal growth. Regarding the organic phases, the hexane fraction displayed MIC values of 1.13 mg/ml for both C. auris strains. On the contrary, the chloroform fraction showed MIC values as low as 0.14 mg/ml for both MMC1 and MMC2, indicating superior efficacy against both C. auris isolates. These results underscore the different antifungal activities of the sequential organic phase extract fractions and suggest that the most potent antifungal compounds in S. coronata (licuri) BOAP are chloroform-extractable, corroborating previously published studies that demonstrated the antifungal activity of chloroform extracts of Nigella sativa L. against C. pelliculosa54.
The pyrolysis of lignocellulosic biomass from Parthenium hysterophorus and the extraction of its aqueous phase produced numerous aromatic and organic compounds with the potential to inhibit the thioredoxin reductase (Trr1) from C. neoformans opening avenues for the development of novel antifungal agents24. Fractionation of the cashew (Anacardiaceae occidentale) nutshell pyrolysis products exhibited antifungal activity against C. albicans, with a growth inhibition halo of 1.83 cm observed in volatilization tests, which considered the inhibitory force as a strong category55. Ribeiro et. al. 2022 found that the utilization of Thymus spp. bio-oil demonstrated an inhibitory effect on the growth of C. auris, most likely due to high concentrations of phenol monoterpenes or aldehydes, which are known for their potent inhibitory effects on fungal cells, even in the vapor phase56.
Therefore, the pyrolysis process consists of an eco-friendly and efficient antimicrobial solution that features sustainable protocols. Additionally, converting waste biomass, specifically the S. coronata (licuri) endocarp, into biofuels or other valuable products through pyrolysis adds both economic and functional value to this resource. Licuri, a fruit from the palm S. coronata, which is native to the Sertão and Caatinga regions of the state of Bahia, Brazil, holds particular ethnobotanical significance and importance due to its natural medicinal applications, especially for the northeastern coastal populations of Brazil, including the use of its bio-oils as detergent and surfactants57,58. Apart from this, S. coronata (licuri) is also an important source of nutrients for humans and animals, as its fruit is extremely caloric. It could also be used as vitamin supplementation in rural areas and as an alternative to corn for poultry farming59. Compared to synthetic drugs, S. coronata (licuri) bio-oils often exhibit fewer side effects and offer a more personalized approach to healthcare, addressing specific community needs and cultural practices60.
As the main product, bio-oil fractions have demonstrated potent activity against resistant fungi such as C. auris, underscoring their potential as natural, versatile, cost-effective and renewable alternatives to synthetic antimicrobials. Nevertheless, the distinct antifungal susceptibility patterns displayed by the C. auris isolates to BOAPs suggest that additional extraction processes could be performed to enhance the potency of these extracts60,61. For example, the presence of potent antifungal compounds in S. coronata (licuri) BOAP I, particularly those extractable through feasible processes, such as the chloroform partition, opens avenues for developing novel processes and antifungal agents. Indeed, further comprehensive profiling of each phase would offer valuable comparative insights into improving extraction protocols, compound distribution and fractionation behavior, which could be expanded to target other fungal pathogens.
Overall, the antifungal efficacy demonstrated by volatile compounds in the licuri BOAPs and chloroform fractions further enhances the potential use of these bio-oil extracts for the decontamination of healthcare facilities and nosocomial environments62,63. Future studies should further focus on isolating and characterizing individual compounds that could demonstrate antifungal activity, and by optimizing the extraction processes and incorporating them into existing formulations, their efficacy could also be evaluated in vivo.
Methods
Biomass preparation of Cocos nucifera (coconut), Syagrus coronata (licuri) and Terminalia catappa (almond) shells
The green coconut husks (pericarp) and sea-almond fruits were obtained from wastes of local markets located in the city of Rio de Janeiro, Rio de Janeiro state (RJ), Brazil (22° 53′ 48.391"S and 43° 13′ 13.381"W), while Syagrus coronata (licuri) husks were obtained in the city of Caldeirão Grande, Brazil (11° 1´ 9.584″ S and 40° 18´ 11.465 W). In all instances, the obtained biomass was prepared by drying the material for 24 h at 105 °C to eliminate any signs of moisture. The dried material was then crushed and processed for the bio-oil production.
Bio-oil production and bio-oil aqueous phase separation
The pyrolytic liquid from the three biomasses was obtained through individual slow pyrolysis processes using a batch reactor located at the Synthesis, Chromatography, and Environment Laboratory, Chemistry Department, Fluminense Federal University, Niterói, RJ, Brazil. The fixed bed reactor of 140 cm × 10 cm made of borosilicate glass was inserted into a Heraeus R/O 100 oven coupled with a temperature controller, and the reactor was connected through ground glass joints to a condenser and a funnel for liquid collection. The non-condensable gases were directed to a series of gas scrubber flasks. Initially, 300 g of the dried shells were placed in the central region of the converter tube connected to the condensation system, subjected to a heating rate of 12 °C min-1, up to 400 °C, and held for 90 min. The system was purged with nitrogen flow at 500 mL/min, to ensure an inert sample atmosphere throughout the process. In the end, the resulting solid product (biochar) remained inside the reactor and was collected after the system cooled down. The condensable gases were collected as the pyrolytic liquid upon condensation. For the phase separation of pyrolysis liquids, the two phases were separated by density difference. For all feedstocks used bio-oil represented the heavier phase and pyroligneous acid, a water-rich phase, was the less dense liquid.
In this work, pyroligneous acid from C. nucifera (coconut), S. coronata (licuri) and T. catappa (almond) pyrolysis were named as crude bio-oil aqueous phase (BOAP). Prior to proceeding to biological tests, crude BOAP went through a simple pretreatment for clean-up, water was added to the crude oil obtained, and the solution was vigorously vortexed for homogenization. The crude BOAP solution was stored overnight at 4 °C, followed by centrifugation at 2000 xg for 30 min. Then, the supernatant was collected and separated from the bottom organic phase (bio-oil residue) by density difference64. The resulting bio-oil aqueous phase I (BOAP I) from the distinct shells were stored at −20 °C until further processing.
The BOAP I from distinct origins was dried out in a LABCONCO Centrivap benchtop concentrator (Kansas, MO, USA), and the material mass was weighed in an analytical balance (OHAUS, NJ, USA). In all cases, the BOAP I extracts were normalized to 90 mg/mL in phosphate-buffered saline, PBS, 137 mM NaCl, 2.7 mM KCl, 1.5 mM KH2PO4, 8.1 mM Na2HPO4, pH 7.4) and frozen at −20 °C to be used in all the subsequent experiments.
Cultivation of Candida spp
The C. albicans isolate SC5314 (ATCC MYA 2876) and the C. auris isolates MMC1 and MMC2—respectively resistant and sensitive to fluconazole and amphotericin B35,36 and the other isolates used in this study are shown in Table 1. All isolates were confirmed as C. auris by MALDI-TOF as described65. Candida spp. were grown on Sabouraud agar plates (SDA; Becton, Dickinson, BD, Franklin Lakes, NJ, USA) at 37 °C for 48 h. For all experiments, a single colony was picked and inoculated in 20 mL of Sabouraud liquid medium (Accumedia, Brazil) and cultivated for 24 h at 37 °C with 150 rpm shaking. Yeasts were centrifuged (1100 × g/10 min) and washed 3X (three times) with PBS, suspended in PBS, and enumerated in a hemocytometer (KASVI, Brazil).
Antifungigram evaluations (susceptibility testing) of BOAP I from C. nucifera (coconut), S. coronata (licuri) and T. catappa (almond)
The antifungal susceptibility testing was performed according to the standardized broth microdilution technique described by the Clinical and Laboratory Standards Institute (CLSI) for yeast-like cells (M27-A3)66. As experimental controls, antifungals such as itraconazole and amphotericin B (Sigma-Aldrich) were used, following the standardization protocols by the CLSI66. Upon cultivation in broth Sabouraud medium, yeasts were washed 3X with PBS and centrifuged (1100xg/10 min). Yeasts were then suspended in RPMI medium (Sigma Aldrich), pH 7.0, buffered with 0.165 mol/L N-morpholino propane sulfonic acid (MOPS, Sigma Aldrich) by vortexing. A small aliquot was withdrawn and counted in a hemocytometer. Yeasts were diluted to a final concentration of 5 × 103 cells/mL in RPMI-MOPS medium. In parallel, extracts were diluted in RPMI-MOPS medium to 9.0 mg/ml. Then serial dilutions (1:2) were carried out in a 96-well flat-bottomed translucent plate (Techno Plastic Products, Switzerland), with concentrations ranging from 9.0 to 0.018 mg/mL; controls of itraconazole and amphotericin B were included at concentrations ranging from 64 to 0.126 µg/mL67. Controls without extracts/drugs were also included. Subsequently, 100 µL of the yeast suspension was added to each well of the plates (for a final extract range concentration of 4.5–0.009 mg/mL), which were incubated at 37 °C for 48 h under agitation at 150 rpm (SL223 Shaker, SOLAB, Brazil). Plate absorbances were read at 560 and 600 nm in a Spectramax spectrophotometer (Molecular Devices) at 37 °C for 0, 18, 24, 42, and 48 h. The minimum inhibitory concentration (MIC) was considered the lowest concentration of the distinct extracts/standard antifungals responsible for limiting the growth of C. auris, leading to an absence of increase in absorbance. The MIC of itraconazole was defined as the lowest concentration of the drug capable of inhibiting 80% of the fungal growth compared to the growth control in the absence of the drug. In addition to the absorbance, the plate wells were visually evaluated at the aforementioned periods.
Effects of S. coronata (licuri) BOAP I on C. auris biofilms
Two distinct approaches were considered for evaluating the licuri BOAP I effect on biofilms: (i) antifungal effects during the formation of biofilms of C. auris MMC1 and MMC2 isolates and (II) antifungal effects on mature (formed) biofilms.
-
(i)
Antifungal effects during the formation of C. auris biofilms: The BOAP I was serially diluted in RPMI-MOPs as described in the antifungigram test using a final concentration ranging from 4.5 to 0.009 mg/mL, and 150 μL was added to the wells of a 96 well plate. Then, the same volume of either MMC1 or MMC2 C. auris inoculum (1 × 106 yeast/mL) was added and the plates were incubated for 6, 12, 24, and 48 h at 37 °C. Supernatants were discarded, and the wells were washed three times with PBS. Resazurin was added to the wells for the observation of fungal viability, and absorbances were measured at 570 nm according to the manufacturer’s instructions68.
-
(ii)
Antifungal effects on mature biofilms. Biofilms of either C. auris MMC1 or MMC2 were produced in 96-well plates by adding 300 μL per well of a 1 × 106 yeast/mL suspension and plates were incubated for 48 h at 37 °C. Upon washes, licuri’s BOAP I (ranging from 4.5 to 0.009 mg/mL) was added over the mature biofilms from each strain. Plates were incubated additionally for 48 h, and upon washes, Resazurin was added, and absorbances were measured at 570 nm68.
Disk volatilization tests of the BOAP I
The antifungal properties of the bio-oil volatile components were evaluated by the disk volatilization tests in Petri dishes. The test was conducted using 10 mm diameter filter paper disks soaked with 100 µL of the extracts evaluated in this study (at 90 mg/mL, 9.0 mg per disk) or their 1:10 dilutions (in PBS, 0.9 mg per disk). One hundred µL of yeast suspensions of the MMC1 and MMC2 C. auris strains at 3 × 104 cells/mL were plated on Sabouraud agar 90 mm plates using the Copacabana method69 to guarantee a uniform dispersion of colonies on a plate upon fungal growth. Then, the disks were placed on the center of the lids of 90 mm Petri dishes (Fig. 5A) and plates incubated at 37 °C with the lids facing down (Fig. 5B). After 48 h, the growth inhibition halo was measured with a pachymeter, and diameters were compared among groups. In addition, colony-forming units (CFU) were counted on the entire surface of the medium and compared.
Gas chromatography-mass spectrometry (GC–MS) analysis
GC–MS was selected because the pyrolysis-derived BOAP I fractions primarily contain volatile and semi-volatile low molecular weight compounds, including phenolics and ketones, which are better suited to GC separation. Many of these components are thermally stable and amenable to electron ionization detection. While LC–MS could indeed complement this approach by detecting less volatile or thermolabile compounds, GC–MS is the standard method used in the characterization of pyrolysis liquids17,23,34. The BOAP I of S. coronata (licuri) was suspended in methanol and filtered using PTFE 0.45 µm syringe filters before analysis. Analysis was carried out by a GC–MS model GCMS—QP2010 (Shimadzu, Kyoto, Japan) with a quadrupole analyzer equipped with a SUPELCOWAX® 10 column (30 m in length × 0.25 mm of diameter × 0.25 μm of film thickness). The carrier gas used was pure helium (99.999%). The oven temperature program was isotherm at 35 °C for 8 min, raised to 230 °C at 10 °C/min, and held for 18 min, with a total analysis time of 45.5 min. An injection volume of 1 μL was performed using a ratio of 1:20 and the injector temperature at 230 °C. The mass spectrometer detector was operated under an ion source temperature of 240 °C, electron ionization energy of 70 eV, and an m/z scan range of 35–600 Da. The chromatograms obtained were processed by the LabSolutions software. For tentative peak assignment, the National Institute of Standards and Technology (NIST) library-NIST 05 (as of June 2023) was used and similarly indexes higher than 85%. GC–MS RAW-files are deposited at www.figshare.com (https://doi.org/10.6084/m9.figshare.29621078). The substance’s distribution was done by normalizing the area of each assigned peak in the chromatogram by the total area of the identified compounds. Information of boiling point, vapor pressure, and solubility in water of the eleven most abundant identified compounds were retrieved from publicly available databases, including in order or preference: (I) PubChem (https://pubchem.ncbi.nlm.nih.gov), (II) ChemSpider (https://www.chemspider.com), (III) CAS common chemistry https://commonchemistry.cas.org), (IV) Merck Index (https://merckindex.rsc.org) and the (V) ChemSrc database (https://www.chemsrc.com/en/), and added to the Supplementary Table 1. Specifically, values of solubility in water and vapor pressure were confirmed with available handbooks70,71.
Sequential organic solvent extraction for the fractionation of chemical groups from S. coronata (licuri) BOAP I
With slight modifications, the BOAP I from licuri was used for the fractionation experiments as described elsewhere17. Three organic solvents of increasing polarity (hexane, chloroform, and ethyl acetate) were chosen for sequential extraction of chemicals. Initially, BOAP I (at 90 mg/mL) was mixed in a chemically resistant glass tube with hexane in a 1:1 (v/v) solvent-to-feed ratio. The mixture was vigorously vortexed for 30 min and incubated for 2 h at room temperature. After centrifugation at 1,100 xg for 10 min, the bio-oil aqueous phase II (BOAP II) was collected. An aliquot of the BOAP II was reserved, and the remaining liquid was further extracted with chloroform in a 1:1 (v/v) solvent-to-feed ratio and extracted as described above. After separation, the bio-oil aqueous phase III (BOAP III) was collected. An aliquot of the BOAP III was reserved. The remaining liquid was mixed with ethyl acetate to a 1:1 (v/v) solvent-to-feed ratio and extracted. The BOAP IV was collected after separation from the ethyl acetate organic phase (EAOP). The sequential bio-oils and organic phases were recovered by evaporating the organic solvents using a LABCONCO Centrivap benchtop concentrator at RT, to preserve the chemical integrity of the compounds throughout the process. After this process, the components were weighed in an OHAUS analytical balance and suspended in RPMI-MOPS for normalization. As negative controls, blank tubes were added with the same volume of solvent used for the extraction of each bio-oil organic phase, dried out and the negligible content was resuspended in RPMI-MOPs.
Antifungigram evaluations of sequentially extracted BOAPs and organic fractions from S. coronata (licuri)
For the comparison of antifungal activity of all the fractions obtained upon sequential extraction of licuri bio-oil (BOAP I, 90 mg/mL), all the extracts obtained were diluted 1:10 in RPMI-MOPS and sequential 1:2 dilutions were performed. Yeasts were diluted to a final concentration of 5 × 103 cells/mL in RPMI-MOPS medium, and 100 µL was added to each well of the diluted extracts (to a final concentration range from 4.5 to 0.009 mg/mL). Controls of itraconazole and amphotericin B (to a final concentration range from 16 to 0.031 µg/mL) were included, as well as fungal growth in the absence of extracts/drugs. Plates were incubated at 37 °C for 48 h under agitation at 150 rpm, and absorbances were read at 600 nm at 18, 24, 40, and 48 h. Upon cultivation, the fungal viability was checked in the maximum inhibiting dilution by determining the colony-forming units (CFUs) in each case by plating on Sabouraud agar, and the percentage of inhibition of fungal growth was determined. The sequentially extracted BOAPs and organic fractions from S. coronata (licuri) were also evaluated in the disk volatilization test as described previously (10 mm disks, 9 mg/disk of each fraction).
Statistical analysis
The statistical analysis was performed using the GraphPad Prism 8 software (GraphPad Software, Boston, MA, USA). The groups were compared using the One-way analysis of variance (One-way ANOVA), with a statistical significance level of 95% (p ≤ 0.05). The Tukey post-test was used to compare every mean with every other mean, whereas the Dunnett test was used to compare every mean to a control mean. Each experiment included at least two biological replicates, and for each biological replicate, three technical replicates were performed.
Data availability
All the data supporting the findings of this study are fully contained in the paper. The datasets used and analyzed during the current study are available from the corresponding author, Allan Guimarães, upon reasonable request.
References
Garcia-Bustos, V. et al. What do we know about Candida auris? state of the art, knowledge gaps, and future directions. Microorganisms 9, 2177 (2021).
Zamith-Miranda, D. et al. Transcriptional and translational landscape of Candida auris in response to caspofungin. Comput. Struct. Biotechnol. J. 19, 5264–5277 (2021).
Chowdhary, A., Sharma, C. & Meis, J. F. Candida auris: A rapidly emerging cause of hospital-acquired multidrug-resistant fungal infections globally. PLoS Pathog. 13, e1006290 (2017).
World Health Organization. WHO fungal priority pathogens list to guide research, development and public health action. (2022).
Chakrabarti, A. & Sood, P. On the emergence, spread and resistance of Candida auris: host, pathogen and environmental tipping points. J. Med. Microbiol. 70(3), 001318 (2021).
Ahmad, S. & Alfouzan, W. Candida auris: Epidemiology, diagnosis, pathogenesis, antifungal susceptibility, and infection control measures to combat the spread of infections in healthcare facilities. Microorganisms 9, 807 (2021).
Rodrigues, M. L. & Nosanchuk, J. D. Fungal diseases as neglected pathogens: A wake-up call to public health officials. PLoS Negl. Trop Dis. 14, e0007964 (2020).
Fisher, M. C. et al. Tackling the emerging threat of antifungal resistance to human health. Nat. Rev. Microbiol. 20, 557–571 (2022).
Barros, D. B. et al. Antifungal activity of terpenes isolated from the Brazilian Caatinga: A review. Braz. J. Biol. 83, e270966 (2023).
Estruzani, T. & Kozusny-Andreani, D. I. Eficácia de extratos plantas medicinais no controle in vitro de Candida albicans. RSD 11, e53311326779 (2022).
Islam, Md. T. et al. Therapeutic potential of essential oils focusing on diterpenes: Diterpenes, the family members of essential oils. Phytother. Res. 30, 1420–1444 (2016).
Kumar, P. The influence of Azadirachta indica, Melaleuca alternifolia, and Cocos nucifera on Candida albicans strain in tissue conditioner at varying time intervals. J. Indian Prosthodont. Soc. 20, 171 (2020).
Swari, D. A. M. A., Santika, I. W. M. & Aman, I. G. M. Antifungal activities of ethanol extract of rosemary leaf (Rosemarinus officinalis L.) Against Candida albicans. JPSA 2, 28–35 (2020).
Kume, J. E. P., Andreani Junior, R., Di-Tanno, M. F. P. & Kozusny-Andreani, D. I. Uso de óleos essenciais in natura e ozonizados no controle in vitro de Trichophyton mentagrophytes. RSD 10, e4710111233 (2021).
Proškovcová, M., Čonková, E., Váczi, P., Marcinčáková, D. & Harčárová, M. Efficacy of Lamiaceae essential oils with selected azoles against Candida albicans clinical isolates. Pol. J. Vet. Sci. 25, 279–285 (2022).
Cibele, M. A. D. S. B. et al. Syagrus coronata seed oils have antimicrobial action against multidrug-resistant Staphylococcus aureus. J. Med. Plants Res. 10, 310–317 (2016).
Mattos, C. et al. Fumigant activity of pyrolysis fractions derived from waste licuri’s pericarp (Syagrus coronata - Arecaceae) in the control the red flour beetle Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Environ. Chall. 5, 100294 (2021).
DebMandal, M. & Mandal, S. Coconut (Cocos nucifera L.: Arecaceae): In health promotion and disease prevention. Asian Pac. J. Trop. Med. 4, 241–247 (2011).
Sivakumar, M. K., Moideen, M. M., Varghese, R., Sheik, B. & Dhanapal, C. Preliminary phytochemical screening and anti-bacterial activity of cocos nucifera linn root. Res. J. Pharm., Biol. Chem. Sci. 2, 468 (2011).
Shino, B. et al. Comparison of antimicrobial activity of chlorhexidine, coconut oil, probiotics, and ketoconazole on Candida albicans isolated in children with early childhood caries: An in vitro study. Scientifica 2016, 1–5 (2016).
Terças, A. L. G. et al. Antifungal drug susceptibility of Candida species isolated from HIV-Positive patients recruited at a public hospital in São Luís, Maranhão, Brazil. Front. Microbiol. 8, 298 (2017).
Imran, A., Bramer, E. A., Seshan, K. & Brem, G. An overview of catalysts in biomass pyrolysis for production of biofuels. Biofuel Res. J. 5, 872–885 (2018).
Mattos, C., Veloso, M. C. C., Romeiro, G. A. & Folly, E. Biocidal applications trends of bio-oils from pyrolysis: Characterization of several conditions and biomass, a review. J. Anal. Appl. Pyrol. 139, 1–12 (2019).
Krishnan, S. et al. Waste to drugs: Identification of pyrolysis by-products as antifungal agents against Cryptococcus neoformans. J. Biomol. Struct. Dyn. 41, 15386–15399 (2023).
Shanmugavasan, A., Vaitheeswaran, K. S. R. & Ramachandran, T. Design and development of pyrolyser to extract medicinal oil from the stem of Ziziphus jujuba. J. Anal. Appl. Pyrol. 92, 176–183 (2011).
Australian National University. National Reconciliation Week: Highlights. 2021.
Australian Curriculum, Assessment and Reporting Authority (ACARA). Australian Curriculum. 2021.
Pirintsos, S. et al. From traditional ethnopharmacology to modern natural drug discovery: A methodology discussion and specific examples. Molecules 27, 4060 (2022).
Hasibuan, R. et al. Effect of pyrolysis temperature and adhesive type on the quality of biobriquette from coffee stem biomass. E3S Web. Conf. 519, 02001 (2024).
Zhuo, Q. et al. Applications of biochar in medical and related environmental fields: Current status and future perspectives. Carbon Res. 2, 32 (2023).
Deng, Q., Li, A., Wu, Y. & Sun, L. Life Cycle Assessment of Biochar Preparation of Chinese Traditional Medicine Residue by Low-temperature Pyrolysis. E3S Web. Conf. 350, 03005 (2022).
Lourençon, T. V., Mattos, B. D., Cademartori, P. H. G. & Magalhães, W. L. E. Bio-oil from a fast pyrolysis pilot plant as antifungal and hydrophobic agent for wood preservation. J. Anal. Appl. Pyrol. 122, 1–6 (2016).
Mohan, D. et al. Fungicidal values of bio-oils and their lignin-rich fractions obtained from wood/bark fast pyrolysis. Chemosphere 71, 456–465 (2008).
Ren, S., Ye, X. P. & Borole, A. P. Separation of chemical groups from bio-oil water-extract via sequential organic solvent extraction. J. Anal. Appl. Pyrol. 123, 30–39 (2017).
Zamith-Miranda, D. et al. Multi-omics Signature of Candida auris, an Emerging and Multidrug-Resistant Pathogen. mSystems. 4, e00257-e319 (2019).
Kim, S. H. et al. Genetic analysis of Candida auris implicates Hsp90 in Morphogenesis and Azole Tolerance and Cdr1 in Azole resistance. MBio 10, e02529-e2618 (2019).
Cristina, M. L. et al. An Overview on Candida auris in Healthcare Settings. J. Fungi (Basel) 9, 913 (2023).
Singh, S., Kumar Chaurasia, P., Lata Bharati, S. & Golla, U. A mini-review on the safety profile of essential oils. MOJBM 7, 33–36 (2022).
Boukhatem, M. N. & Setzer, W. N. Aromatic herbs, medicinal plant-derived essential oils, and phytochemical extracts as potential therapies for coronaviruses: Future perspectives. Plants 9, 800 (2020).
Ramos, M. L. N. S. et al. Potential of cattle manure pyrolysis liquid as an alternative environmentally friendly source of agricultural fungicides. J. Anal. Appl. Pyrol. 149, 104862 (2020).
Perveen, S., Hussain, Z. & Khan, K. M. Antifungal activity of the liquid obtained from pyrolysis of paper. J. Chem. Soc. Pakistan 30, 876–878 (2008).
Hussain, Z., KHAN, K. M., PERVEEN, S. & BUKHARI, I. H. Antifungal Activity of the Pyrolyzate of Glucose, Sucrose and Starch in Comparison to Paper Pyrolyzate. 33, 694–697 (2011).
Satoh, K. et al. Candida auris sp. nov., a novel ascomycetous yeast isolated from the external ear canal of an inpatient in a Japanese hospital. Microbiol. Immunol. 53, 41–44 (2009).
Lockhart, S. R. et al. simultaneous emergence of multidrug-resistant candida auris on 3 continents confirmed by whole-genome sequencing and epidemiological analyses. CLINID 64, 134–140 (2017).
Souza Dos Santos, B. et al. Anti-staphylococcal activity of Syagrus coronata essential oil: Biofilm eradication and in vivo action on Galleria mellonela infection model. Microb. Pathog. 131, 150–157 (2019).
Sternkopf Lillebæk, E. M., Lambert Nielsen, S., Scheel Thomasen, R., Færgeman, N. J. & Kallipolitis, B. H. Antimicrobial medium- and long-chain free fatty acids prevent PrfA-dependent activation of virulence genes in Listeria monocytogenes. Res. Microbiol. 168, 547–557 (2017).
Kim, K. H. et al. Evaluation of the antifungal effects of bio-oil prepared with lignocellulosic biomass using fast pyrolysis technology. Chemosphere 89, 688–693 (2012).
Ferreira, E. D. S. et al. The use of essential oils and their isolated compounds for the treatment of oral candidiasis: A literature review. Evidence-Based Comp. Altern. Med. 2021, 1–16 (2021).
Hughes, A. F. D. S., Lima, F. G. D., Lucchese, A. M., Góes Neto, A. & Uetanabaro, A. P. T. Antimicrobial activity of Syagrus coronata (Martius) Beccari. Braz. arch. biol. technol. 56, 269–274 (2013).
Fujitani, Y. et al. Characteristics of different volatility classes of organic compounds emitted by a municipal solid waste incineration plant. Atmos. Environ.: X 17, 100197 (2023).
Mandras, N. et al. Liquid and vapour-phase antifungal activities of essential oils against Candida albicans and non-albicans Candida. BMC Comp. Altern. Med. 16, 330 (2016).
Santos Júnior, C. J. D., Rodrigues, D. K. D. B., Dantas, R. D. S. & Souza, A. K. P. Atividade antifúngica de isolados clínicos de Candida não-albicans aos óleos essenciais de Syzygium aromaticum (cravo-da-índia) e Eucalyptus globulus (eucalipto-comum). Medicina (Ribeirão Preto). 54, e168684 (2021).
Possamai Rossatto, F. C. et al. Antifungal Activity of the Phenolic Compounds Ellagic Acid (EA) and Caffeic Acid Phenethyl Ester (CAPE) against Drug-Resistant Candida auris. JoF 7, 763 (2021).
Tiji, S. et al. Chemical composition related to antimicrobial activity of Moroccan Nigella sativa L. extracts and isolated fractions. Evidence-Based Comp. Altern. Med. 2021, 1–14 (2021).
Ustamuni, U., Nurliana, L. & Musta, R. Aplikasi dan Tinjauan Kinetika Hasil Fraksinasi Produk Pirolisis Cangkang Biji Jambu Mete Uji sebagai Antijamur Candida albicans. Ind. J. Chem. Anal 2, 47–53 (2019).
Ribeiro, R. et al. Comparing the effect of Thymus spp. essential oils on Candida auris. Ind. Crops Prod. 178, 114667 (2022).
Alves, A. Q. et al. The fatty acid composition of vegetable oils and their potential use in wound care. Adv. Skin Wound Care 32, 1–6 (2019).
Rufino, M. U. D. L., Costa, J. T. D. M., Silva, V. A. D. & Andrade, L. D. H. C. Conhecimento e uso do ouricuri (Syagrus coronata) e do babaçu (Orbignya phalerata) em Buíque, PE. Brasil. Acta Bot. Bras. 22, 1141–1149 (2008).
Crepaldi, I. C., Almeida-Muradian, L. B. D., Rios, M. D. G., Penteado, M. D. V. C. & Salatino, A. Composição nutricional do fruto de licuri (Syagrus coronata (Martius) Beccari). Rev. bras. Bot. 24, 155–159 (2001).
Drugkar, K. et al. Advanced separation strategies for up-gradation of bio-oil into value-added chemicals: A comprehensive review. Sep. Purif. Technol. 283, 120149 (2022).
Chan, Y. H. et al. Fractionation and extraction of bio-oil for production of greener fuel and value-added chemicals: Recent advances and future prospects. Chem. Eng. J. 397, 125406 (2020).
Bukłaha, A. et al. New trends in application of the fumigation method in medical and non-medical fields. Ann. Agric. Environ. Med. 29, 185–189 (2022).
Byrns, G. & Fuller, T. P. The risks and benefits of chemical fumigation in the health care environment. J. Occup. Environ. Hyg. 8, 104–112 (2011).
Folly, E. et al. Processo Para Obtenção de uma Composição Bioinseticida, Composição Bioinseticida e uso da mesma. (2021).
Abdolrasouli, A. & Fraser, M. A. Candida auris Identification and Profiling by MALDI–ToF Mass Spectrometry. in Candida auris (ed. Lorenz, A.) vol. 2517 21–32 (Springer US, New York, NY, 2022).
Wayne, P. A. CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. (Clinical and Laboratory Standard Institute, 2008).
Höfling, J. F. et al. Antimicrobial potential of some plant extracts against Candida species. Braz. J. Biol. 70, 1065–1068 (2010).
Cordero, R. J. B. et al. Enhanced virulence of Histoplasma capsulatum through transfer and surface incorporation of glycans from Cryptococcus neoformans during co-infection. Sci. Rep. 6, 21765 (2016).
Worthington, M. T., Luo, R. Q. & Pelo, J. Copacabana method for spreading E. coli and yeast colonies. Biotechniques 30, 738–742 (2001).
Guillory, J. K. Handbook of Aqueous Solubility Data By Samuel H. Yalkowsky and Yan He. CRC Press, Boca Raton, FL. 2003. xii + 1496 pp. 18 × 26 cm. ISBN 0–89493–1532–8. $299.95. J. Med. Chem. 46, 4213–4213 (2003).
Stull, D. R. Vapor pressure of pure substances. organic and inorganic compounds. Ind. Eng. Chem. 39, 517–550 (1947).
Acknowledgements
AJ.G. was supported by the Brazilian agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grants 311470/2018–1 and 309736/2021–8) and Fundação Carlos Chagas de Amparo à Pesquisa no Estado do Rio de Janeiro (FAPERJ E-26/200.997/2022). J.N. and D.Z.M. are supported by R21AI156104 and R01AI171093. The current manuscript is part of a thesis to be submitted by Deborah Santos Cintra in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate Program of Applied Microbiology and Parasitology of the Fluminense Federal University (PPGMPA, UFF).
Funding
National Institutes of Health, R21AI156104; R01AI171093, R21AI156104; R01AI171093, Conselho Nacional de Desenvolvimento Científico e Tecnológico, 311470/2018–1; 309736/2021–8, Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro, FAPERJ E-26/200.997/2022.
Author information
Authors and Affiliations
Contributions
DSC, LMA, KAN, MRV, PCOO, BSP, MCCV, GAR and AJG designed the study; DSC, LMA, KAN, MRV, PCOO, BSP, MCCV, and AJG conducted the study; DSC, LMA, KAN and AJG prepared the figures; DSC and AJG wrote the manuscript text; DSC, PCOO, BSP, DZM, JDN, MRP and AJG reviewed the manuscript; All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cintra, D.S., Alcantara, L.M., Nardy, K.A. et al. Antifungal efficacy of pyrolysis-derived bio-oil aqueous phases from Tropical plant biomass against Candida auris. Sci Rep 15, 33514 (2025). https://doi.org/10.1038/s41598-025-17626-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17626-6