Abstract
Our study aimed to determine the association between baseline system inflammatory bioparameters and the characteristics of spectral domain optical coherence tomography (SD-OCT) and evaluate the efficiency of anti-vascular endothelial growth factor (VEGF) treatment in different stages of diabetic macular edema (DME). A total of 126 patients were enrolled in this prospective study, including 67 males and 59 females who were diagnosed with DME. The patients were initiatively treated with intravitreous anti-VEGF treatment at 1-month intervals for three consecutive months. With respect to the TCED-HFV grading system, ME was classified into early, advanced, severe, and atrophic stages. Best-corrected visual acuity (BCVA), central macular thickness (CMT), subretinal fluid (SRF), and quantities of hyperreflective foci (HRF) of the whole retina were assessed at baseline and three months after the first injection. System inflammatory bioparameters were calculated, including neutrophil-to-lymphocyte ratio (NLR), monocyte-to-lymphocyte ratio (MLR), platelet (PLT)-to-lymphocyte ratio (PLR), systemic immune-inflammation index (SII), and C-reactive protein (CRP). In addition, the association between HRF, SRF, and system inflammatory bioparameters were evaluated, and the efficiency of anti-VEGF treatment in different stages of DME was assessed. There were significant differences in systemic inflammatory bioparameters among the four stages, including CRP, NLR, PLR, MLR, and SII (all p < 0.001), which were significantly higher in the atrophic stage than another three stages. In contrast, the CRP, NLR, PLR, MLR, and SII levels were significantly lower. BCVA and CMT were significantly improved after treatment in the early, advanced, and severe stages except for the atrophic stage (all p < 0.05). The changes in the proportion of SRF and HRF ≥ 30 were most significant in the advanced stage after anti-VEGF therapy (p = 0.030, p = 0.041, respectively). In general, systemic inflammatory bioparameters are closely associated with the characteristics of different stages of DME based on the TCED-HFV grading system, which reflects the local inflammation of DME. The TCED-HFV grading system may be a precise indicator for predicting the efficiency of anti-VEGF treatment of DME and should be tested in further study.
Similar content being viewed by others
Introduction
Diabetic macular edema (DME) is a leading cause of vision loss and blindness in working-age adults worldwide1,2. It is well known that vascular endothelial growth factor (VEGF) and inflammation are the majority pathogenesis of DME. Romero-Aroca et al. suggested that angiogenesis and inflammation act interdependently during the development of DME3. Intravitreal anti-VEGF drugs have been considered as the first-line therapy for DME4. A large, multicenter study also demonstrated the intravitreal dexamethasone implant to be effective and safe in eyes with early and late stages of DME especially for the management of chronically persistent subjects who received regular intravitreal injection for at least four injections within 24 weeks5.
In recent studies, system inflammation was an emerging biomarker for the progression of DME, which might be accelerated by the destruction of the blood-retinal barrier (BRB) in the early episode. The inflammation-related factors contributing to BRB breakdown include but are not limited to VEGF, TNF-α, IL-1α, IL-1β, IL-6, IL-8, IL-10, ICAM-1, MCP-1, placental growth factor (PlGF), hepatocyte growth factor (HGF), insulin-like growth factor-1 (IGF-1), and histamine and complement factors, which were reported to be upregulated in the vitreous or aqueous humor of DME patients6,7. This indicated a significant contribution of inflammation-related factors to the pathogenesis of DME. In addition to inflammation-related factors, inflammatory cells, such as activated retinal microglia and infiltration of immune cells, including macrophages, lymphocytes, and neutrophils, also contribute to the pathogenesis of DME and DR8,9.
According to the morphological characteristics on spectral domain optical coherence tomography (SD-OCT), the European Institute of Advanced Ophthalmology (ESASO) proposed an innovative DME classification system, the TCED-HFV grading system (Table 1). The grading system includes foveal thickness (T), corresponding to either central subfoveal thickness or macular volume, intraretinal cysts (C), the ellipsoid zone (EZ) and/or external limiting membrane (ELM) status (E), the presence of disorganization of the inner retinal layers (DRIL), the number of hyperreflective foci (H), subfoveal fluid (F), and vitreoretinal relationship (V). Based on the first four variables, DME was defined as early stage, advanced stage, severe stage, and atrophic stage, respectively (Table 2)10.
There are quantitative studies on the correlation between DME and system inflammatory biomarkers. Limited studies on the efficiency of anti-VEGF treatment of alternative severities of DME, was based on the TCED-HFV grading system were documented11. System inflammatory biomarkers have been thoroughly studied in the progression of tumors, cardiovascular abnormalities, and other multiple inflammatory diseases. Those biomarkers have dramatically changed in inflammation-related factors and the procoagulant capabilities of cells12,13. In this study, our goal was to identify the association between baseline system inflammatory bioparameters and the characteristics on SD-OCT and evaluate the efficiency of anti-VEGF treatment in different stages of DME.
Methods
The institutional Review Board of Hebei Eye Hospital approved this prospective study, which adhered to the Declaration of Helsinki. All participants gave written informed consent at the time of recruitment.
Patients
Patients were recruited from the Hebei Eye Hospital from October 2022 to November 2023. All patients were randomly received 0.05 mL of aflibercept or ranibizumab intravitreal injections for the first time, which were FDA-approved for DME treatment following a protocol of at least three monthly injections followed by pro re nata administration14,15. All patients underwent comprehensive ophthalmic examinations at baseline and one month after the third injection, including the best-corrected visual acuity (BCVA), slit-lamp biomicroscopy, color fundus photography, intraocular pressure (NIDEK NT-510), OCT (Spectralis, Heidelberg, Germany), and fundus fluorescein angiography (FFA). The inclusion criteria included (1) type 2 diabetic mellitus (DM) patients diagnosed with DME through OCT and FFA, (2) DME involving the fovea, which was defined as retinal thickening in the central macular subregion (diameter 1 mm) in the OCT.
The exclusion criteria were as follows: (1) other diseases that can result in macular edema, including retinal vein occlusion, age-related macular degeneration, polypoidal choroidal retinopathy, etc., (2) previous laser photocoagulation, intravitreal injection of anti-VEGF drugs or dexamethasone implant, surgery on the vitreous and (or) retina, cataract surgery within six months, (4) patients with comorbidities (acute or chronic inflammation or infection, severe cardiovascular and cerebrovascular disease, autoimmune diseases, kindney disorders and malignancy) or given long-term steroid hormone and immunosuppressive medications, and (5) patients with poor OCT imaging quality.
Hematologic laboratory indices and system inflammatory bioparameters
Blood samples were obtained from the antecubital vein before treatment at 6:00 am after an overnight fast of eight h. Serum was separated prior to intravitreal anti-VEGF treatment. Leukocyte subtypes (neutrophils, lymphocytes, and monocytes) and platelets were measured using an Automated Blood Coagulation Analyzer (Sysmex XN-550, Japan). Next, we analyzed lymphocytes, platelets (PLTs), monocytes, neutrophils, glycosylated hemoglobin (HbA1c), and creatinine. The neutrophil-to-lymphocyte ratio (NLR), PLT-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), and systemic immune-inflammation index (SII) were calculated for the following blood samples: neutrophils-to-lymphocytes, PLT-to-lymphocytes, monocytes-to-lymphocytes, and (neutrophils × PLT)-to-lymphocytes.
Assessment of OCT indices
OCT images were acquired using 6-mm vertical and horizontal scans centered on the fovea in high-resolution mode with spectral domain OCT (SD-OCT, Heidelberg Spectralis OCT, Heidelberg Engineering, Germany). Recorded quantitative variables included (1) central macular thickness (CMT) and (2) the total number of hyperreflective foci (HRF) across retinal layers. Qualitative parameters were assessed for (1) size of intraretinal cysts (IRC), (2) integrity of the ELM and EZ, (3) presence of disorganization of the inner retinal layers (DRIL), and (4) presence of subfoveal fluid (SRF). All the subjects underwent examinations twice. All OCT images were analysed by the blinded retina specialist(Li-na Lv) and averages were determined. According to the TCED-HFV grading system (Table 1), the DME was divided into early, advanced, severe, and atrophic stages10 (Table 2, represented in Figs. 1, 2, 3 and 4).
The HRF was divided into two subgroups according to the number of HRF: HRF ≥ 30 and HRF < 30 for analysis. The identification for quantifying OCT parameters was as follows: CMT was the distance from the internal limiting membrane to the outer border of the retinal pigment epithelium (RPE) at the fovea. HRF were distinct punctate foci with reflection intensity equal to or greater than the nerve fiber layer, distributed across retinal layers. The HRF count was manual from the inner limiting membrane to Repute definition of IRC was the elliptical low-reflection areas within the neurosensory layer. IRCs were qualitatively classified as absent, mild, moderate, or severe, with total area measured. ELM and EZ integrity grading depended on the visibility and continuity of the outermost two reflective bands with the grades including recognizable integrity, disruption, and absence. DRIL was characterized by indistinct boundaries between the ganglion cell layer-inner plexiform layer (GCL-IPL) complex, inner nuclear layer (INL), and outer plexiform layer (OPL). DRIL grading was categorized as absent or present. SRF was shown to be subretinal hypo-reflective spaces and was classified as absent or present.
Early DME Multiple perifoveal cystoid spaces in the INL, the OPL, with mild thickening of the temporal area of foveal, absence of any visible adhesion or traction between vitreous cortex and retina, hyperreflective foci are less than 30 in number. The TCED-HFV grading is T = 1, C = 2, E = 0, D = 0, H = 0, F = 0, and V = 0.
Advanced DME Intermediate cystoid spaces in the fovea. The EZ is not gradable, but the ELM is disrupted subfoveally. The segmentation of the internal retinal layers is still visible. The presentation of vitreous macular traction is obvious. The TCED-HFV grading is T = 2, C = 2, E = 1, D = 1, H = 1, F = 1, and V = 3.
Severe DME Central macrocyst surrounded by large cystoid spaces involving the outer nuclear layer, the OPL, and the INL. Severe DRIL and some hyperreflective foci are shown. The external liming membrane and the EZ are disrupted under the fovea; nevertheless, the vitreoretinal relationship is normal. The TCED-HFV grading is T = 2, C = 3, E = 1, D = 1, H = 0, F = 1, and V = 0.
Atrophic DME Central retinal thinning with DRIL. The ELM and the EZ are still gradable subfoveally, and there are several hyperreflective foci in the outer nuclear layer and OPL. Thickness and volume are below normal values, but some moderate intraretinal cysts are present. The TCED-HFV grading is T = 0, C = 1, E = 0, D = 1, H = 0, F = 0, and V = 0.
Statistical analysis
All statistical analyses were conducted using the Statistical Program for the Social Sciences (SPSS) 23.0 (SPSS, Inc., Chicago, IL, USA). To avoid the impact of binocular homogeneity, data from the right eye were used for intergroup comparisons in patients with bilateral eyes influenced. The BCVA was converted to the logarithm of the minimum resolution angle (logMAR). Continuous variables in the measurement data were expressed as mean ± SD, and differences between groups were analyzed using independent-sample t-tests or one-way analysis of variance (ANOVA); paired t-tests were used for intragroup data before and after treatment. Non-normally distributed and non-homogeneity of variance data were expressed as medians, and the differences between groups were examined using the Mann-Whitney U or Kruskal-Wallis test; the Wilcoxon symbolic rank test was adopted for intragroup data before and after treatment. Categorical variables were described as percentages (%) and compared via χ2 test or Fisher exact probability test. A p-value of less than 0.05 was considered significant in all the analyses. Bonferroni correction was managed when the pairwise comparison intragroup was carried out, and a p-value less than 0.008 was considered significant.
Results
Baseline demographic data
There were 126 patients enrolled in this research, including 67 males and 59 females. The patients were divided into the following four stages: early (37 eyes), advanced (35 eyes), severe (36 eyes), and atrophic (18 eyes), according to the TCED-HFV grading system. Among the four stages of DME, no significance was detected in the distribution of gender, age, duration of DM, or diabetic retinopathy grading (Table 3).
Baseline system inflammatory bioparameters
In general, there were significant differences in C-reactive protein (CRP), NLR, MLR, PLR, and systemic immune-inflammation index (SII) among the four stages (p < 0.001); however, no significant differences in HbA1c were identified (p = 0.848).
C-reactive protein
There was a significant difference in CRP among the four stages (p < 0.001). Compared to early DME, the difference in CRP in the atrophic stage was significant (p < 0.001), but no significance was detected in the advanced and severe stages (p = 0.009, p = 0.038, respectively). Compared to advanced DME, the differences in CRP in the severe and atrophic stages were significant (p < 0.001, respectively). Compared to severe DME, the difference in CRP in the atrophic stage was significant (p < 0.001).
NLR
There was significant difference in NLR among the four stages (p < 0.001). Compared to early DME, NLR was considerable in the atrophic stage (p < 0.001). However, no significance was manifested in the advanced and severe stages (p = 0.087, p = 0.870, respectively). Compared to advanced DME, the difference in NLR in the atrophic stage was significant (p < 0.001). Nevertheless, no significance was found in the severe stage (p = 0.045). Compared to severe DME, the difference in NLR in the atrophic stage was significant (p < 0.001).
MLR
There was a significant difference in MLR among the four stages (p < 0.001). Compared to early DME, MLR was considerably higher in the atrophic stage (p = 0.001); however, no significance was indicated in the advanced and severe stages (p = 0.213, p = 0.131, respectively). Compared to advanced DME, the difference in MLR in the atrophic stage was significant (p < 0.001); nevertheless, no significance was shown in the severe stage (p = 0.391). Compared to severe DME, the difference in MLR in the atrophic stage was significant (p < 0.001).
PLR
There was a significant difference in PLR among the four stages (p < 0.001). Compared to early Methera, no significant differences existed in the alternative stages (p = 0.033, p = 0.039, p = 0.124). Compared to advanced DME, the difference in PLR in the atrophic stage was significant (p < 0.001); however, no significance was detected in the severe stage (p = 0.844). Compared to severe DME, the difference in PLR in the atrophic stage was significant (p < 0.001).
Systemic immune-inflammation index
There was a significant difference in SII among the four stages (p < 0.001). Compared to early DME, the differences in SII in the advanced and atrophic stages were significant (p = 0.001, p < 0.001, respectively); however, no significance was shown in the severe stage (p = 0.174). Compared to advanced DME, the difference in SII in the atrophic stage was significant (p < 0.001); however, no significance was found in the severe stage (p = 0.080). Compared to severe DME, the difference in SII in the atrophic stage was significant (p < 0.001).
BCVA of subjects with different stages of DME
Baseline BCVA
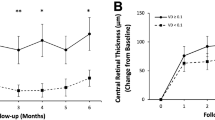
Significant differences in BCVA (logMAR) at the baseline in different stages of DME were detected (p < 0.001, Table 4; Fig. 5a). Compared to the early stage, the BCVA of the advanced, severe, and atrophic stages showed significant differences (p < 0.001). Compared to the advanced stage, the BCVA in the severe and atrophic stages were higher (p < 0.001). Compared to the severe stage, the BCVA in the atrophic stage was higher (p = 0.004).
BCVA after treatment
Significant differences in BCVA (logMAR) after treatment in different stages of DME were shown (p < 0.001, Table 4). Compared to the early stage, the BCVA of another three stages were much higher (p < 0.001). Compared to the advanced stage, the BCVA in the severe and atrophic stages were higher (p < 0.001). Compared to the severe stage, the BCVA in the atrophic stage was higher (p < 0.001).
BCVA changes
Significant differences in BCVA (logMAR) changes in different stages of DME were detected (p < 0.001, Table 4). Compared to the early stage, the differences in the changes in BCVA in the advanced and atrophic stages were significant (p < 0.001), but no significant difference was indicated in the severe stage (p = 0.013). Compared to the advanced stage, the differences in the changes in BCVA in the severe and atrophic stages were significant (p < 0.001, p = 0.005, respectively). Compared to the severe stage, the difference in the change in BCVA in the atrophic stage was significant (p < 0.001).
Intragroup BCVA comparison between the baseline and after treatment
Significant differences in BCVA between baseline and after treatment were manifested in the early, advanced, and severe stages (p < 0.001, p = 0.012, p < 0.001, respectively); nevertheless, no significance was shown in the atrophic stage (p = 0.407, Table 4).
CMT of subjects with different stages of DME
Baseline CMT
Significant differences in CMT at the baseline in different stages of DME were detected (p < 0.001, Table 5; Fig. 5b). Compared to the early stage, the CMT of the advanced, severe, and atrophic stages showed significant differences (p < 0.001). Compared to the advanced stage, the differences in CMT of the severe and atrophic stages were significant (p < 0.001). Compared to the severe stage, the CMT in the atrophic stage was much thinner (p < 0.001).
CMT after treatment
Significant differences in CMT after treatment in the different stages of DME were shown (p < 0.001, Table 5). Compared to the early stage, the CMT of the atrophic stage showed a significant difference (p < 0.001); however, no significances were detected in the advanced and severe stages (p = 0.221, p = 0.967, respectively). Compared to the advanced stage, the difference in CMT in the atrophic stage was significant (p < 0.001); nevertheless, the difference was apparent in the severe stage (p = 0.068). Compared to the severe stage, the CMT in the atrophic stage was much lower (p < 0.001).
CMT changes
Significant differences in CMT changes in the different stages of DME were detected (p < 0.001, Table 5). Compared to the early stage, the difference in the change in CMT of the severe stage was significant (p < 0.001), but no significant differences were indicated in the advanced and atrophic stages (p = 0.011, p = 0.778, respectively). Compared to the advanced stage, the differences in the changes in CMT of the severe and atrophic stages were significant (p < 0.001). Compared to the severe stage, the difference in the change in CMT at the atrophic stage was significant (p < 0.001).
CMT intragroup comparison between baseline and after treatment
Significant differences in CMT between the baseline and after treatment were manifested in the early, advanced, severe, and atrophic stages (p = 0.038, p < 0.001, p < 0.001, and p = 0.031, respectively, Table 5).
SRF and HRF at different stages of DME
The differences in the distribution in SRF between the four stages at baseline were significant (p = 0.004), and the percentage of SRF in the advanced stage was maximum (40%) among all the stages at baseline. The differences in the distribution of SRF between the four stages after treatment were significant (p = 0.015), and the percentage of SRF in the severe stage was maximum (33.33%) among all the stages after treatment. The differences in the distribution of HRF ≥ 30 between the four stages at baseline were significant (p = 0.018), and the percentage of HRF ≥ 30 in the severe stage was maximum (47.22%) among all the stages at baseline. The differences in the distribution of HRF ≥ 30 between the four stages after treatment were significant (p = 0.033), and the percentage of HRF ≥ 30 in the severe stage was maximum (41.67%) among all stages after treatment.
The efficiency of anti-VEGF treatment was higher in the advanced stage, and the proportions of SRF and HRF ≥ 30 were significantly reduced from the baseline to after treatment from 40% to 14.29%, p = 0.030 and from 45.71% to 20%, p = 0.041, respectively (Table 6; Fig. 6).
Discussion
The relationship between inflammation and hyperreflective foci and subretinal fluid has been thoroughly studied in recent years. There was consensus that the local inflammation symbols were represented in the form of HRF and SRF shown on SD-OCT. HRF, which was initiated by the activation of microglial cells, was the lipoprotein extravasations prior to hard exudates or a degeneration of the photoreceptors or macrophages that engulf them16,17. It was interesting that SRF had a good reaction of intravitreal triamcinolone acetonide, which indirectly proved that inflammation played a key role in the development of SRF18.
Saijo et al.19 proved that Müller cells, astrocytes cells, and microglial cells were activated in the hyperglycemia condition, which initiated an inflammatory process in the retina. The microglial cells immigrated into the subretinal space and synthesized the cytokines, including interleukin 6 (IL-6) and interleukin 8 (IL-8) or C-C motif ligand 2 (CCL2), when the RPE was damaged. Low-grade inflammation was maintained by the cytokines. IL-6 altered the function of astrocytes, which gave structural support to the capillaries in the retina; thus, the BRB was broken20,21 and the subretinal fluid was finally accumulated.
The cytokines have been consistently elevated in DR patients with the progression of the destruction of hyperglycemia22. The early-and advanced-stage DME showed lower destruction of retinal layers, which might be attributed to the primary inflammatory circumstances. Moreover, the baseline system inflammatory bioparameters were smaller in the two stages. However, the CRP, NLR, MLR, PLR, and SII in the astrophic stage were more pronounced than the others. This phenomenon suggested that local inflammation was correlated with system inflammation. HbA1c of the atrophic stage was higher than another three stages, however, no significant differences in HbA1c were identified (p = 0.848) among the four stages.
Our research also suggested that system inflammatory bioparameters in early stage were higher than those in advanced stage which was consistent with Chen et al.11. Hyperglycemia caused oxidative stress reactions. As a result, macular edema was induced by increased systemic and ocular inflammatory levels23. There was a consensus that inflammatory activation had already occurred in the early stage, and then the body was in a compensated condition in the advanced stage and finally intensified with the progression of the DME from severe to atrophic stages11.
Kocbora et al.24 showed that the CRP and TNF-α levels increased in DME patients. Woo et al.25 found that the peripheral blood neutrophil was higher and correlated with the severity of DR. They concluded that system inflammation plays a key role in the pathogenesis of DR and DME. Similar findings were documented in our study. There were compensated mechanisms at the early stage of DME, and the immune system was in a relatively balanced condition. Nevertheless, in the late episode, the immune system was influenced at a severe level for lengthy hyperglycemia, which reached a decompensated stage. Furthermore, the retinal barrier was damaged more severely in the astrophic stage.
Multiple-center randomized controlled trials have identified intravitreal injection of ranibizumab and aflibercept as the first-line treatment for DME14,15 which were administered in our research. We also found that CMT declined significantly in every stage after anti-VEGF therapies, especially in advanced and severe DME. The BCVA was also improved to an acceptable level, particularly in the early and severe stages. The SRF and HRF showed more acceptable outcomes in the advanced stage, suggesting that inflammatory cells and cytokines in retinal layers were more sensitive to the anti-VEGF drugs despite the continued inflammation during constant hyperglycemia. In general, the structural and functional results of anti-VEGF therapy improved significantly, particularly in advanced DME.
SRF represented outer blood-retinal barrier disruption or retinal pigment epithelium (RPE) drainage dysfunction, which caused leakage via ELM or choroid capillary and the fluids accumulated under the neurosensory retina layer26. It was highly conceivable that HRF was induced by the activated microglial cell migration which were activated by inflammatory stimulation and migrated from the inner layer to the outer layer of the retina27.
Initially, the leakage and inflammation was relatively slight and the proportion of SRF and HRF was very low at the early stage. Consequently, the improvement of the SRF and HRF was not remarkablely. As mentioned above, the body was in a compensated condition at advanced stage and system inflammatory bioparameters were lower. The regression of SRF and HRF was more obvious and the improvement was significant after anti-VEGF treatment. The severe and astrophic stage were discompensated and intensified local inflammation and destruction of the blood-retinal barrier were more notable. As a result, the regression of SRF and HRF were not significant after anti-VEGF treatment.
Anti-VEGF therapy prevented BRB disruption and the release of proinflammatory cytokines, reducing the vascular permeability and inhibiting the inflammation progress; this resulted in reduced retinal thickness and elimination of cysts in the retinal layers28,29,30. This process might be managed at a balanced level in the early stage, consistent with the distribution of baseline CRP, NLR, MLR, PLR, and SII in our study. Moreover, the TCED-HFV grading based on SD-OCT provides an imaging illustration for prognosis and has a strong correlation with the results of the anti-VEGF treatment of DME. The grading system provided an alternative for assessing the outcomes of anti-VEGF treatment from both the local inflammation indices and the system inflammation bioparameters.
There are emerging meanings, including the intraocular fluid test, which can accurately evaluate inflammatory and angiogenesis factors before and after treatment. However, due to its invasive characteristics and high cost, this test was not applied in our research. As mentioned above, the TCED-HFV grading based on SD-OCT also implied reliable retinal inflammation in response to hyperglycemia. In addition, the indices on the SD-OCT were in compliance with system inflammatory bioparameters. Chen et al. drew a similar conclusion in their research11.
This study had several limitations and shortcomings. We did not set the placebo-treated group (DM patients without DR). Moreover, the DR grading was not comprehensively documented. The anti-VEGF drugs in our research were all FDA-approved. They are more expensive than bevacizumab, which is used off-label and shows promising results for DME therapy. The heavy economic burden might be considerable. In addition, the number of subjects was not high, and the quantitative and observation episodes were relatively short. Therefore, further exploration is needed to identify the significance of our research and its clinical application.
In summary, systemic inflamatory bioparameters are closely associated with the characteristics of different stages of DME based on the TCED-HFV grading system, which reflects the local inflammation of DME. The TCED-HFV grading system may be a precise indicator for predicting the efficiency of anti-VEGF treatment of DME but requires additional testing and further study.
(a) changes in BCVA at different stages of DME; (b) Changes in CMT at different stages of DME. ***p < 0.001, **p < 0.01, *p < 0.05, and ns versus baseline BCVA or baseline CMT.
(a,c) percentage of presence or absence of SRF before and after anti-VEGF treatment in different stages of DME. (b, d) Percentage of the number of HRF ≥ 30 or < 30 before and after anti-VEGF treatment at different stages of DME.
Data availability
The raw data is available from the corresponding author (wycocular@163.com) on reasonable request.
References
Flaxel, C. J. et al. Diabetic retinopathy preferred practice pattern®. Ophthalmology 127, 66–145. https://doi.org/10.1016/j.ophtha.2019.09.025 (2020).
Lee, R. et al. Epidemiology of diabetic retinopathy, diabetic macular edema and related vision loss. Eye Vis. 2, 17. https://doi.org/10.1186/s40662-015-0026-2 (2015).
Romero-Aroca, P. et al. Diabetic macular edema pathophysiology: vasogenic versus inflammatory. J. Diabetes Res. 2016, 2156273. https://doi.org/10.1155/2016/2156273 (2016).
Schmidt-Erfurth, U. et al. Guidelines for the management of diabetic macular edema by the European society of retina specialists (EURETINA). Ophthalmologica 237, 185–222. https://doi.org/10.1159/000458539 (2017).
Rosenblatt, A. et al. A collaborative retrospective study on the efficacy and safety of intravitreal dexamethasone implant (Ozurdex) in patients with diabetic macular edema: the European DME registry study. Ophthalmology 127, 377–393. https://doi.org/10.1016/j.ophtha.2019.10.005 (2020).
Rübsam, A. et al. Role of inflammation in diabetic retinopathy. Int. J. Mol. Sci. 19, 942. https://doi.org/10.3390/ijms19040942 (2018).
Adamis, A. P. & Berman, A. J. Immunological mechanisms in the pathogenesis of diabetic retinopathy. Semin. Immunopathol. 30, 65–84. https://doi.org/10.1007/s00281-008-0111-x (2008).
Lee, H. et al. Association between soluble CD14 in the aqueous humor and hyperreflective foci on optical coherence tomography in patients with diabetic macular edema. Investig. Ophthalmol. Vis. Sci. 59, 715–721. https://doi.org/10.1167/iovs.17-23042 (2018).
Xie, H. et al. Erythropoietin protects the inner blood-retinal barrier by inhibiting microglia phagocytosis via Src/Akt/cofilin signalling in experimental diabetic retinopathy. Diabetologia 64, 211–225. https://doi.org/10.1007/s00125-020-05299-x (2021).
Panozzo, G. et al. An optical coherence tomography-based grading of diabetic maculopathy proposed by an international expert panel: the European school for advanced studies in ophthalmology classification. Eur. J. Ophthalmol. 30, 8–18. https://doi.org/10.1177/1120672119880394 (2020).
Yanxia, C. et al. Optical coherence tomography-based grading of diabetic macular edema is associated with systemic inflammatory indices and imaging biomarkers. Ophthalmic Res. 67, 96–106. https://doi.org/10.1159/000535199 (2024).
Guthrie, G. J. K. et al. The systemic inflammation-based neutrophil-lymphocyte ratio: experience in patients with cancer. Crit. Rev. Oncol. Hematol. 88, 218–230. https://doi.org/10.1016/j.critrevonc.2013.03.010 (2013).
Kim, S. et al. Association of neutrophil-to-lymphocyte ratio with mortality and cardiovascular disease in the Jackson heart study and modification by the Duffy antigen variant. JAMA Cardiol. 3, 455–462. https://doi.org/10.1001/jamacardio.2018.1042 (2018).
Nguyen, Q. D. et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology 119, 789–801. https://doi.org/10.1016/j.ophtha.2011.12.039 (2012).
Brown, D. M. et al. Intravitreal Aflibercept for diabetic macular edema: 100-week results from the VISTA and VIVID studies. Ophthalmology 122, 2044–2052. https://doi.org/10.1016/j.ophtha.2015.06.017 (2015).
Zeng, H. et al. Microglial activation in human diabetic retinopathy. Arch. Ophthalmol. 126, 227–232. https://doi.org/10.1001/archophthalmol.2007.65 (2008).
Chen, X. et al. Early Spatiotemporal characterization of microglial activation in the retinas of rats with streptozotocin-induced diabetes. Graefe’s Archive Clin. Experimental Ophthalmol. 253, 519–525. https://doi.org/10.1007/s00417-014-2727-y (2015).
Shukla, D. et al. Serous macular detachment as a predictor of resolution of macular edema with intravitreal triamcinolone injection. Ophthalmic Surg. Lasers Imaging. 40, 115–119. https://doi.org/10.3928/15428877-20090301-20 (2009).
Graeber, M. B. et al. Role of microglia in CNS inflammation. FEBS Lett. 585, 3798–3805. https://doi.org/10.1016/j.febslet.2011.08.033 (2011).
Omri, S. et al. Microglia/macrophages migrate through retinal epithelium barrier by a transcellular route in diabetic retinopathy: role of PKCζ in the Goto Kakizaki rat model. Am. J. Pathol. 179, 942–953. https://doi.org/10.1016/j.ajpath.2011.04.018 (2011).
Ambrosini, E. & Aloisi, F. Chemokines and glial cells: a complex network in the central nervous system. Neurochem. Res. 29, 1017–1038. https://doi.org/10.1023/b:nere.0000021246.96864.89 (2004).
Vujosevic, S. et al. Proteome analysis of retinal glia cells-related inflammatory cytokines in the aqueous humour of diabetic patients. Acta Ophthalmol. 94, 56–64. https://doi.org/10.1111/aos.12812 (2016).
Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications (EDIC) Research Group. Effect of intensive diabetes therapy on the progression of diabetic retinopathy in patients with type 1 diabetes: 18 years of follow-up in the DCCT/EDIC. Diabetes 64, 631–642. https://doi.org/10.2337/db14-0930 (2015).
Kocabora, M. S. et al. Serum and aqueous concentrations of inflammatory markers in diabetic macular edema. Ocul. Immunol. Inflamm. 24, 549–554. https://doi.org/10.3109/09273948.2015.1034804 (2016).
Woo, S. J. et al. Elevated systemic neutrophil count in diabetic retinopathy and diabetes: a hospital-based cross-sectional study of 30,793 Korean subjects. Investig. Ophthalmol. Vis. Sci. 52, 7697–7703. https://doi.org/10.1167/iovs.11-7784 (2011).
Kim, J. T. et al. Changes in choroidal thickness in relation to the severity of retinopathy and macular edema in type 2 diabetic patients. Investig. Ophthalmol. Vis. Sci. 54, 3378–3384. https://doi.org/10.1167/iovs.12-11503 (2013).
Lee, H. et al. Correlation between optical coherence tomographic hyperreflective foci and visual outcomes after anti-vegf treatment in neovascular age-related macular degeneration and polypoidal choroidal vasculopathy. Retina 36, 465–475. https://doi.org/10.1097/IAE.0000000000000645 (2016).
Khandelwal, P. J. et al. Inflammation in the early stages of neurodegenerative pathology. J. Neuroimmunol. 238, 1–11. https://doi.org/10.1016/j.jneuroim.2011.07.002 (2011).
Campochiaro, P. A. Ocular neovascularization. J. Mol. Med. (Berlin Germany) 238, 91. https://doi.org/10.1007/s00109-013-0993-5 (2013).
Stewart, M. W. The expanding role of vascular endothelial growth factor inhibitors in ophthalmology. Mayo Clin. Proc. 87, 77–88. https://doi.org/10.1016/j.mayocp.2011.10.001 (2012).
Acknowledgements
Huan Li and Yun-chang Wang conducted the experiments. Rong-rong Li, Li-na Lv and performed data analysis, visualization, and wrote the manuscript. Jie Wang revised and edited the manuscript. Ya-cong Wang, Jun-kun Gao, Chuan-qi Zhou and Wei Wei provided technical assistance. All authors read and approved the final manuscript.
Funding
This work was supported by the Municipal Science and Technology Plan Project of Xingtai City, Hebei Province, No. 2022ZC232.
Author information
Authors and Affiliations
Contributions
Huan Li and Yun-chang Wang conducted the experiments. Rong-rong Li, Li-na Lv and performed data analysis, visualization, and wrote the manuscript. Jie Wang revised and edited the manuscript. Ya-cong Wang, Jun-kun Gao, Chuan-qi Zhou and Wei Wei provided technical assistance. All authors read and approved the fnal manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was approved by the Ethics Committee of the Hebei Eye Hospital, Hebei Provincial Key Laboratory of Ophthalmology, Hebei Provincial Clinical Research Center for Eye Diseases ([2022KY19]).
Informed consent
This study involves human participants. All research procedures adhered to the tenets of the Declaration of Helsinki. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Rr., Lv, Ln., Li, H. et al. Shifting of the local inflammatory characteristics of diabetic macular edema manifested in SD-OCT after anti-VEGF treatment and associations with system inflammatory bioparameters. Sci Rep 15, 39437 (2025). https://doi.org/10.1038/s41598-025-23067-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23067-y