Abstract
Physical frailty may weaken structural brain integrity (SBI), which may lead to cognitive impairments. In this cross-sectional study, the number of final study participants was 2057 (700 men, 1357 women). The proportion of the frail men was 5.3% (n = 37), and the proportion of the frail women was 6.6% (n = 90). We used the Fried Frailty Phenotypes (FFP) to assess the physical frailty associated with SBI regarding cognitive impairments by sex. We performed multivariate linear and logistic regression to assess the associations between the FFP and SBI markers and probable mild cognitive impairment. The false discovery rate test using the Benjamini-Hochberg procedure was applied to fully adjusted models. Frail women had large white matter hyperintensities, low fractional anisotropy, and high mean diffusivity (β = 0.30, 95% Confidence Interval: 0.09 to 0.50; β = -0.28, 95% CI: -0.46 to -0.11; β = 0.31, 95% CI: 0.16 to 0.47). Frail men had a smaller thalamus and hippocampus (β = -0.36, 95% CI: -0.60 to -0.12; β = -0.47, 95% CI: -0.78 to -0.17). Compared with the opposite sex, white matter integrity was low in frail women, whereas gray matter integrity was low in frail men.
Similar content being viewed by others
Aging is accompanied by a decline in physical, social, and cognitive structures and functions, making the prevention of this decline vital for healthy aging. Physical frailty, a multidimensional decline due to aging, has attracted increasing attention as the aging population has rapidly expanded1. We used the Fried Frailty Phenotype (FFP) that assesses physical functions based on five simple binary phenotypes: unintentional weight loss, weakness, self-reported exhaustion, slowness, and low physical activity as proposed in 20012. The FFP has been recognized as a reliable indicator for predicting adverse health outcomes such as mortality, falls, and hospitalizations, particularly in community-dwelling middle-aged and aging general populations3,4.
Previous studies have reported that the frail participants had lower Mini-Mental State Examination (MMSE) scores and lower survival rates with cognitive impairment compared with robust participants5,6. As the FFP assesses functional decline, the relationship between the FFP and cognition has been linked to the brain structure using the magnetic resonance image (MRI) markers such as cortical thickness, gray matter volume (GMV), and white matter hyperintensities (WMH)7. Each of these structural brain markers has multiple clinical implications; however, a singular brain marker may provide insufficient clinical evidence in diagnosing cognitive impairment. Several studies have assessed multiple structural brain markers to reflect structural brain integrity relevant to cognitive impairment. GMV, WMV, and WMH, which are macrostructural markers, and fractional anisotropy (FA) and mean diffusivity (MD), which are microstructural markers, can be used to assess the association with FFP7,8,9. It has been reported that WMH volume indicates cerebral small vessel disease and cognitive decline10, and WMH increases in frail participants11. A meta-analysis reported that FA and MD in the white matter were low and high, respectively, in participants with mild cognitive impairment (MCI)12. Lower FA and higher MD were reported in frail participants4. Particularly, WMH, FA, and MD markers can assess white matter integrity that were known for significant indicators of cognitive impairments13,14,15. It was reported that white matter integrity markers were decreased in frail participants4,9. According to a study, WMV was likely to develop MCI but not as strongly as WMH16. A study reported that small WMV was related to high plasma glial fibrillary acidic protein, which indicates glial dysfunction in brain aging17. Consequently, WMV was included in white matter integrity in the current study to assess multiple patterns of the neuroimaging markers, which may provide insightful evidence in conducting further studies.
Although it is well established that physical frailty varies in prevalence and characteristics between sex18,19, there is an insufficient number of studies investigating the association between physical frailty and cognitive impairment through the use of MRI markers stratified by sex4. Rather than using the FFP index, most studies have evaluated specific phenotypes, such as handgrip strength and gait speed, associated with brain MRI markers8,20,21. For instance, Duchowny and colleagues reported that handgrip strength was associated with larger WMH, but smaller in women than in men20. However, they did not conduct the study stratified by sex. Several previous studies that defined FFP used participants who were ≥ 65 or 70 years old7,8,22 with fewer than a thousand participants11,23. Physical frailty has been established as a modifiable risk factor; thus, analyzing healthy participants who are 55–65 years old should be considered to assess a transitional stage of physical frailty.
We aimed to assess the cross-sectional association between FFP and multiple patterns of SBI (GMV, WMV, WMH, FA, and MD) in community-dwelling men and women aged 55–79 years in South Korea. With relatively large samples from five communities, we hypothesized that the FFP in the study population may be positively associated with SBI markers relevant to cognitive impairment, particularly with respect to sex.
Results
General characteristics of the study participants
The study included 2057 participants (700 men [34%] and 1357 women [66.0%]) (Table 1). Among the men, 336 were robust (48.0%), 327 were pre-frail (46.7%), and 37 were frail (5.3%). Among the women, 583 were robust (43.0%), 684 were pre-frail (50.4%), and 90 were frail (6.6%).
Regardless of sex, the frail participants were the oldest (men, mean age 70.9 [SD 5.0]; women, mean 70.4 [SD 5.8]). Across the five centers, Wonju had the highest proportion of frail men (n = 16, 43.2%), whereas Namwon had the highest proportion of frail women (n = 29, 32.2%). Yangpyeong and Kanghwa had the lowest proportion of frail men (Yangpyeong and Kanghwa: n = 3, 8.1%), whereas Yangpyeong had the lowest proportion of frail women (n = 7, 7.8%). Regarding cohabitation, there were two frail men (5.4%) and 23 frail women (25.6%) living alone. Regarding educational attainment, 175 men (25.0%) and 152 women (11.2%) had college degrees. Few frail men and women had college degrees (men: n = 7, 18.9%; women: n = 2, 2.2%, respectively).
Frail women had the lowest perceived social support scores (44.2 [SD 10.7]). Regarding smoking status (never, former, current), one frail woman was a former smoker, and two frail women were current smokers. Sleep duration was greater in frail men than in robust and pre-frail men (7.2 [SD 1.8]), whereas it was less in frail women than in the other groups (6.2 [SD 1.5]). The proportion of frail men and women was highest for those with cognitive scores < 24 (men: n = 6, 16.2%; women: n = 28, 31.1%).
The volume of the fried frailty phenotypes and structural brain integrity
The volume (mean [SD]) of the SBI markers was assessed in relation to the FFP (Supplementary Table S2). Among the macrostructural markers, the GMV and WMV were the smallest in frail men and women. WMH was the largest in both frail men and women. For microstructural markers, FA and MD were the lowest and highest, respectively, in frail participants.
Association between the components of fried frailty phenotypes and structural brain integrity
We assessed the association between five frailty phenotypes and SBI markers according to sex (Table 2). In men, weakness was the most significant phenotype among FFP except in MD (GMV: β = −0.46, 95% CI: −0.63 to −0.29; WMV: β= −0.40, 95% CI: −0.59 to −0.22; WMH: β = 0.30, 95% CI: −0.13 to 0.34; FA: β = −0.50, 95% CI: −0.69 to −0.31; MD: β = 0.43, 95% CI: 0.23 to 0.63). Exhaustion was the most significant phenotype in MD (β = 0.48, 95% CI: 0.23 to 0.73). In women, weakness was the most significant phenotype among FFP except in WMV and WMH (GMV: β = −0.35, 95% CI: −0.46 to −0.23; WMV: −0.20, 95% CI: −0.31 to −0.09; WMH: β = 0.33, 95% CI: 0.20 to 0.46; FA: β = −0.42, 95% CI: −0.55 to −0.29; MD: β = 0.49, 95% CI: 0.37 to 0.60). Slowness was the most significant phenotype in WMH (β = 0.50, 95% CI: 0.37 to 0.63).
Association between fried frailty phenotypes and structural brain integrity
We assessed the association between FFP and the SBI markers according to sex (Table 3). In fully adjusted model 3, among the three macrostructural markers, GMV in frail men was significantly smaller (GMV: β = −0.24, 95% CI: −0.42 to −0.06, P for trend = 0.010), whereas WMH in frail women was significantly larger (WMH: β = 0.30, 95% CI: 0.09 to 0.50, P for trend = 0.004). Two microstructural markers, FA and MD were significantly lower and higher in frail men and women, respectively (FA: β = −0.45, 95% CI: −0.72 to −0.17, P for trend = 0.001; β = −0.28, 95% CI: −0.46 to −0.11, P for trend = 0.001; MD: β = 0.53, 95% CI: 0.22 to 0.84, P for trend = 0.001; β = 0.31, 95% CI: 0.16 to 0.47, P for trend = < 0.001).
Association between fried frailty phenotypes and regions of interest
We assessed the association between the FFP and MRI markers of gray matter integrity by sex, including the subcortical GMV, thalamus, basal ganglia (caudate nucleus, putamen, globus pallidus, subthalamic nucleus), and hippocampus (Table 4). In the fully adjusted model 3, frail men had significantly small thalamus and hippocampus (thalamus: β = −0.36, 95% CI: −0.60 to −0.12, P for trend = 0.003; hippocampus: β = −0.47, 95% CI: −0.78 to −0.17, P for trend = 0.002).
We plotted the gray and white matter volume of the four lobes using a fully adjusted model stratified by sex (Figs. 2a and b and 3a and b). In the current study, the FFP and gray and white matter in the frontal, parietal, occipital, and temporal lobes exhibited distinct patterns by sex. Among the gray matter volumes of the four lobes in frail men, the frontal lobe was the lowest, and the occipital lobe was the highest. In frail women, the parietal lobe was the lowest, and the occipital lobe was the highest. Among the white matter volumes of the four lobes in frail men and women, the parietal lobe was the lowest, and the frontal lobe was the highest. None of these volumes was significant.
Association between fried frailty phenotypes and cognitive impairment
We assessed the association between the FFP and probable MCI according to sex (Table 5). In the fully adjusted model 3, the odds ratios (OR) for frail men and women and probable MCI were comparable (OR: 3.75, 95% CI: 1.09 to 12.9; OR: 3.94, 95% CI: 1.99 to 7.83), respectively. In model 3, the ORs for pre-frail women were higher than for pre-frail men (OR: 2.12, 95% CI: 1.29 to 3.49; OR: 1.24, 95% CI: 0.56 to 2.75).
We assessed the exploratory analysis between the FFP and the Korean version of the Mini-Mental State Examination (K-MMSE) items by sex (Supplementary Table S3). Adjusting age, centers, and ICV, the score of the registration, recall, language, and drawing items were significantly low in frail men (registration: β = −0.28, 95% CI: −0.55 to −0.01; recall: β = −0.31, 95% CI: −0.59 to −0.04; language: β = −0.28, 95% CI: −0.54 to −0.02; drawing: β = −0.49, 95% CI: −0.77 to −0.21), whereas all scores except for orientation to place score (β = 0.03 95% CI: −0.18 to 0.25) were significantly low in frail women (registration: β = −0.66, 95% CI: −0.89 to −0.43; orientation to time: β = 0.63, 95% CI: −0.86 to −0.40; recall: β = −0.68, 95% CI: −0.90 to −0.45; attention & calculation: β = −0.40, 95% CI: −0.61 to −0.18; language: β = −0.59, 95% CI: −0.82 to −0.36; drawing: β = −0.42, 95% CI: −0.66 to −0.18).
Effect modification and interaction analysis
Age was a consistent effect modifier among the five SBI markers (Supplementary Table S4). Among the frail participants, age (≥ 65 years old) showed interactions with five SBI markers (Supplementary Table S5). The interactions between age and WMV and WMH among pre-frail participants were not significant.
Discussion
We assessed the association between the FFP and five SBI markers according to sex in community-dwelling late middle-aged and old adults from five centers in South Korea. For structural brain integrity, gray matter integrity was weak in frail men, whereas white matter integrity was weak in frail women. Among regions of interest (ROIs) of gray matter, the volume of the thalamus and hippocampus was significantly low in frail men, whereas none of the ROIs were significantly low in frail women. In frail participants, the gray matter of four lobes showed distinct but different patterns by sex, whereas the white matter of four lobes showed distinct but similar patterns regardless of sex. The ORs for probable MCI for frail men and women were comparable. The ORs for probable MCI for pre-frail women were higher than for pre-frail men.
Previous studies have reported that the FFP may be linked to SBI markers, represented by gray and/or white matter integrity markers, as these exhibit multiple patterns regarding cognitive impairments4,9. In line with our results, it was reported that frail participants had weakened white matter integrity characterized by larger WMH volume, lower FA, and higher MD than other participants, and this may progress to cognitive impairment4,7,10. As WMH indicates cerebral small vessel disease, and FA and MD indicate microvascular states, hypertension, smoking status, and high cholesterol may contribute to the relationship between the severity of physical frailty, cerebrovascular diseases, and brain atrophy. In the current study, frail women had more type 2 diabetes mellitus than frail men. A longitudinal study that used a South Korean cohort demonstrated that women with type 2 diabetes mellitus were vulnerable to larger WMH, which might consequently lead to cognitive impairments24. Vascular risk factors and type 2 diabetes mellitus might have an important role in the association between physical frailty and cognitive impairments exhibited by white matter integrity markers. One of the white matter integrity markers in the current study, WMV, was not a strong indicator of cognition aligned with our results16, and this should be investigated to elucidate what is associated with physical frailty in future studies. Components of the FFP were assessed with white matter integrity markers. Participants with weakness and slowness phenotypes together may be vulnerable to the onset of MCI21,25, and it was shown that weakness and slowness among WMV, WMH, FA, and MD were first or second significant phenotypes in frail women in the current study. However, these five phenotypes may vary depending on the characteristics of the study population; for instance, exhaustion and slowness were shown to be the most significant using UK Biobank9. Physical frailty was positively associated with increased WMH and smaller GMV9, which implies its relations with the SBI markers. Regarding the gray matter integrity, our results showed that frail men had a smaller thalamus and hippocampus compared with frail women. It is reported that the weakness and slowness were linked to changes in thalamic and hippocampal volume21,26. In the current study, among the gray matter of four lobes, low volume of the frontal lobe and parietal lobe was exhibited in frail men and women, respectively. A small GMV of the frontal lobe and parietal lobe might be linked to slowed gait speed and cognitive control task21,27,28, and mostly GMV of frontal and parietal lobe show significant change across lobes29. A study demonstrated that parietal WMH might be linked to executive function30. Nevertheless, previous studies have been reported asymmetry of ROIs as the magnitude of association could vary28,31. Taken together, these sex differences in physical frailty and multiple patterns of SBI should be further investigated. Since these results were not assessed on the association with FFP, the interpretation must not be directly linked to previous studies.
The association between FFP and SBI is likely to be affected by strong risk factors such as age4,32. As the age factor (≥ 65 years old) was an effect modifier for the current study, our results showed additive interactions between the age factor and the FFP. Thus, the results are partly consistent with those of previous studies. Note that, as GMV, WMV, and WMH are considered hallmarks of brain aging9,10,33, there can be brain regions that are not vulnerable to aging7. Aging is not preventable, and the severity of physical frailty increases with age. Wardlaw and colleagues reviewed that the blood-brain barrier permeability may increase with advancing age, with relatively healthy conditions, even in normal white matter10. They suggested that the MD marker may indicate the best of microstructural changes in normal appearing white matter structures10. As we used healthy participants without having major events such as CVD, heart diseases, and cancer, FA and MD as microstructural markers may imply there can be imperceptible changes using macrostructural markers. Consistently raised pathophysiological factor is a sex hormone. For instance, a study identified that WMH burden was higher and accelerated in postmenopausal women than premenopausal women of similar age34. Sex hormones such as estrone or follicle-stimulating hormone were positively associated with changes in frontal or temporal WMH that linked to cognition35. Further studies should address these sex hormonal issues regarding the relationship between physical frailty and SBI markers.
This is the first community-level study to assess the FFP and SBI markers according to sex in South Korea. Our study and the CAVAS-CA cohort have implications in public surveillance because five communities in this ongoing cohort are relatively small urban or rural areas in which healthcare access may not be sufficiently distributed compared to the mega city, Seoul. Our cross-sectional results will provide basic evidence in designing further studies and assessing health policy measures of healthcare blind spots for underprivileged areas. SBI markers have strengths for assessing multiple patterns that indicate any signal of cognitive level. Particularly, FA and MD have strengths in identifying any imperceptible changes, even when macrostructural MRI markers are normal, indicating a healthy participant. Our study has several limitations due to the study design, study hypothesis, and cohort status. We hypothesized that the physical frailty may lead to weakened SBI by sex, which was a unidirectional association. As previous studies have reported the bidirectional association between the physical frailty and brain structure markers and/or cognitive impairment4,9,36,37, the further studies should consider evaluation. Reverse causality cannot be excluded due to the nature of the cross-sectional design. We might not account for any potential residual confounding. Notably, educational attainment influenced the prevalence of physical frailty. However, for the current study, we adjusted for educational attainment employing multiple linear regressions stratified by sex and age, respectively. Educational attainment should be further considered as an effect modifier that influences brain volumetric in our future studies. Although we defined FFP, SBI, and other covariates considerably, response bias might have been present because of the sociocultural context in rural communities. For instance, older women in rural communities could hardly smoke. Apolipoprotein E and amyloid β variables were not available in the CAVAS-CA cohort. The current study does not provide which specific ROIs were located among the four lobes, and this issue must be solved in collecting follow-up datasets. Although we used a general population from five communities across South Korea, we cannot generalize our results. These limitations should be carefully solved in further studies.
As we assessed physical frailty using the FFP, there were differences in SBI and FFP according to sex. For SBI, frail men had weak gray matter integrity (GMV, thalamus, and hippocampus), and frail women had weak white matter integrity (WMV, WMH, FA, and MD).
Methods
Data sources and study participants
The Korean Genome and Epidemiology Study (KoGES) Cardiovascular Disease Association Study (CAVAS), funded by the Korea National Institute of Health, was originally constructed in 2005 to shed light on the burden of cardiovascular diseases concerning any health discrepancies between city and rural communities. The CAVAS has been described in detail elsewhere38. The CAVAS-Cognitive Aging (CA) is a prospective sub-cohort of the CAVAS that aims to investigate the relationship between healthy aging and cognition and to provide strong evidence for community-based prevention policies among community-dwelling adults in five centers across South Korea. To screen and assess the transition from late middle-aged to old population, the CAVAS-CA included adults who were ≥ 55 years old. The CAVAS-CA has its strength in collecting social and cognitive diagnostic tools and structural and functional brain MRI markers among five community settings. Baseline recruitment was conducted between 2020 and 2022. A follow-up survey is conducted every three years. The CAVAS-CA is currently collecting the first round of follow-up data. Data is collected using a standardized protocol following standardized education and training in the five regional centers.
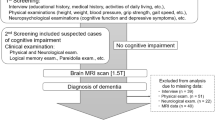
In the current study, reported artifacts in the acquisition of magnetic resonance images or examinations were removed, as these artifacts might distort any statistical analyses (n = 130). We excluded participants aged < 55 years and > 79 years (n = 4; n = 5). Non-respondents were excluded from the exposure and covariates (n = 47). We excluded participants with a history of cerebrovascular diseases (stroke, cerebral infarction), heart diseases (angina pectoris, myocardial infarction), and cancer regardless of their health status (n = 128; n = 246; n = 175, respectively). We excluded participants with missing frailty phenotypes, SBI markers, or covariates (n = 17). Finally, we included men (n = 700) and women (n = 1357) in the final analysis (n = 2057) who were 55–79 years of age at the time of baseline recruitment (2020–2022) of the CAVAS-CA (Fig. 1).
Flow diagram of the study participants. Abbreviations: KoGES CAVAS-CA, Korean Genome and Epidemiology Study (KoGES) Cardiovascular Disease Association Study (CAVAS)-Cognitive Aging (CA); MRI, magnetic resonance imaging; CVD, cerebrovascular diseases. Cerebrovascular diseases included stroke and cerebral infarction. Heart diseases included angina pectoris and myocardial infarction.
(a) Association between Fried Frailty Phenotypes and the gray matter volume of the four lobes in men. Fully-adjusted model is model 3, which adjusted for age, center, intracranial volume, cohabitation, educational attainment, perceived social support scores, sleep duration, smoking status, alcohol consumption, type 2 diabetes mellitus, hypertension, Korean version of mini-mental state examination, waist circumference, and total cholesterol. (b) Association between Fried Frailty Phenotypes and the gray matter volume of the four lobes in women. gray matter. Fully-adjusted model is model 3, which adjusted for age, center, intracranial volume, cohabitation, educational attainment, perceived social support scores, sleep duration, alcohol consumption, type 2 diabetes mellitus, hypertension, Korean version of mini-mental state examination, waist circumference, and total cholesterol. Smoking status was not adjusted in women due to influential points.
(a) Association between Fried Frailty Phenotypes and the white matter volume of the four lobes in men. Fully-adjusted model is model 3, which adjusted for age, center, intracranial volume, cohabitation, educational attainment, perceived social support scores, sleep duration, smoking status, alcohol consumption, type 2 diabetes mellitus, hypertension, Korean version of mini-mental state examination, waist circumference, and total cholesterol. (b) Association between Fried Frailty Phenotypes and the white matter volume of the four lobes in women. Fully-adjusted model is model 3, which adjusted for age, center, intracranial volume, cohabitation, educational attainment, perceived social support scores, sleep duration, smoking status, alcohol consumption, type 2 diabetes mellitus, hypertension, Korean version of mini-mental state examination, waist circumference, and total cholesterol. Smoking status was not adjusted in women due to influential points.
Assessment of fried frailty phenotypes
The FFP has been validated in East Asian countries and is a reliable indicator of physical frailty39,40,41. We defined the FFP according to the Fried method2,39. We defined each of the five components as follows: unintentional weight loss, weakness, self-reported exhaustion, slowness, and low activity. For unintentional weight loss, we used participants who lost more than 5% of their weight (kg). For weakness, we used participants whose handgrip strength (kg) was the lowest 20th percentile stratified by sex and BMI. For self-reported exhaustion, we used two questions from the Korean version of the Center for Epidemiologic Studies Depression Scale battery test; namely, responses were dichotomized using these two questions: “I felt that everything I did was an effort” or “I could not get going.” For slowness, we used participants with the highest 20th percentile of a gait speed (meters per second) stratified by sex and height. Lastly, for low activity, the lowest 20th percentile of total energy consumption stratified by sex was used. We categorized the FFP into robust, pre-frail, and frail statuses according to the component scores. Frail was defined as scoring three or more components. Pre-frail was defined as scoring one or two components. Robust was defined as scoring none of these components. Detailed definitions and criteria were described in the supplement (Supplementary Table S1).
MRI acquisition and data processing
The participants were scanned on 3.0T MRIs (Philips Ingenia CX 5.4/Achieva dStream at the Yangpyeong center; Philips Ingenia CX 5.3 at the Namwon, Goryeong, and Kanghwa centers; Siemens Skyra at the Wonju-Pyeongchang center). Cortical and subcortical GMV and WMV were measured from T1-weighted images using the CIVET (Constrained Iterative Version of Topology) v2.1 pipeline. WMH were extracted from FLAIR images using a U-Net (U-shaped Network)-based white matter segmentation model. Lastly, the Computational Neuroimage Analysis DTI processing pipeline was used to measure FA and MD as the results of corrections, co-registration, and DTI fitting. All MRI data were standardized by the processing protocol of the harmonization developed by Hanyang CAVAS-MRI Quality Control Center. Gray matter ROIs were segmented using an atlas, Automatic Nonlinear Image Matching, and Anatomical Labeling42. FA and MD were calculated using 48 ROIs atlas developed by Johns Hopkins University43. In the current study, all SBI markers, including ROIs, were converted to z-scores to fit the normal distribution and compare across outcomes coefficients.
Assessment for probable mild cognitive impairment
The Korean version of the Mini-Mental State Examination (K-MMSE) was adopted to evaluate the cognitive status using scores 0 to 30. K-MMSE may not reflect all the clinical characteristics of mild cognitive impairment; therefore, we used cutoffs (< 24) to define probable mild cognitive impairment44. Seven items (registration, orientation to time, orientation to place, recall, attention & calculation, language, and drawing) of K-MMSE were further explored as a secondary analysis.
Covariates
We used age, center (Yangpyeong, Namwon, Goryeong, Wonju-Pyeongchang, and Kangwha), cohabitation (alone, spouse or family), intracranial volume (ICV), educational attainment (middle school, middle and high school, college), perceived social support score, alcohol consumption (0–10 g, 10–70 g, 70 g+), smoking status (never, former, current), sleep duration (hours), hypertension (no, yes), type 2 diabetes mellitus (no, yes), K-MMSE (< 24, ≥ 24), waist circumference (cm), and total cholesterol (mg/dL) as covariates. We assessed the frequency of cohabitation, educational attainment, perceived social support scores, sleep duration, alcohol consumption, and smoking status using self-reported questionnaires. We assessed alcohol consumption (g/times) by calculating approximately 10 g of alcohol per soju (a distilled liquor that is popular in Korea) cup. We considered participants to be heavy drinkers when they drank 70 + g/time regardless of sex45. Hypertension was defined using a history of hypertension or systolic blood pressure or diastolic blood pressure ≥ 140/90 mmHg or the use of antihypertensive medications. Type 2 diabetes mellitus was defined using a history of type 2 diabetes mellitus or fasting glucose level ≥ 126 mg/dL or HbA1c ≥ 6.5% or the use of antidiabetic medications. Waist circumference was measured to the nearest 0.1 cm, and the range of waist circumference was considered as the half between the lowest ribcage and the iliac crest during expiration. Total cholesterol level was measured using an ADVIA 1650 automated analyzer (Siemens, New York, NY, USA).
Statistical analyses
Descriptive analyses were performed using one-way analysis of variance and chi-square tests. WMH was naturally log-transformed to alleviate skewness.
We performed linear regression analyses to assess the association between the FFP and SBI markers according to sex. We modeled multiple linear regressions adjusting for sociodemographic, behavioral, anthropometrics, and blood specimens: (1) for model 1, we did not adjust for confounders; (2) for model 2, we adjusted for age, center, and ICV; (3) for model 3, we further adjusted for cohabitation, educational attainment, perceived social support, sleep duration, smoking status, alcohol consumption, comorbidities (type 2 diabetes mellitus, hypertension), WC, and total cholesterol. ICV was adjusted for models 2 and 3 because the adjustment approach may be flexible, accounting for multiple factors that affect structural brain MRI markers46. We solely adjusted for smoking status in men because smokers in women were extremely scarce in the CAVAS-CA. We modeled for multiple logistic regressions, adjusting for the same covariates except for ICV and K-MMSE. We tested linear trends to assess the dose-response relationship. In the multivariate linear regression analysis, we added age groups (< 65 years old, ≥ 65 years old) as an interaction term for all the models. We plotted fully adjusted models to assess patterns of four lobes (frontal, parietal, temporal, occipital) of gray matter and white matter associated with FFP. To assess SBI, we performed multivariate linear regressions on subcortical gray matter structures, including the thalamus, caudate nucleus, putamen, globus pallidus, hippocampus, and subthalamic nucleus. The false discovery rate tests using the Benjamini-Hochberg procedure were applied to twenty-one fully-adjusted models, and these significances were shown in bold text. Any other details of the methodology, as well as additional tables and figures, can be found in the supplementary material.
All statistical analyses were performed using STATA SE 18.0 (StataCorp LLC, College Station, TX, USA).
Data availability
The current dataset, CAVAS-CA, is not publicly available; however, it will be available in the next few years. All inquiries should be sent to the following email: [kimyumi@hanyang.ac.kr](mailto: kimyumi@hanyang.ac.kr) **.** Otherwise, all data analyzed during the current study are included in the manuscript (and its Supplementary Information file).
References
Dent, E. et al. Management of frailty: opportunities, challenges, and future directions. Lancet 394, 1376–1386. https://doi.org/10.1016/S0140-6736(19)31785-4 (2019).
Fried, L. P. et al. Frailty in older adults: evidence for a phenotype. J. Gerontol. Biol. Sci. Med. Sci. 56, M146–156. https://doi.org/10.1093/gerona/56.3.m146 (2001).
Cesari, M., Gambassi, G., van Kan, G. A. & Vellas, B. The frailty phenotype and the frailty index: different instruments for different purposes. Age Ageing. 43, 10–12. https://doi.org/10.1093/ageing/aft160 (2014).
Ducca, E. L. et al. Physical frailty and brain white matter abnormalities: the atherosclerosis risk in communities study. J. Gerontol. Biol. Sci. Med. Sci. 78, 357–364. https://doi.org/10.1093/gerona/glac111 (2023).
Lee, Y. et al. The effects of frailty and cognitive impairment on 3-year mortality in older adults. Maturitas 107, 50–55. https://doi.org/10.1016/j.maturitas.2017.10.006 (2018).
Vahedi, A. et al. Cognitive impairment in people with physical frailty using the phenotype model: A systematic review and meta analysis. Int. J. Geriatr. Psychiatry. 37 https://doi.org/10.1002/gps.5822 (2022).
Cipolli, G. C. et al. Frailty and brain changes in older adults without cognitive impairment: A scoping review. Arch. Gerontol. Geriatr. 123, 105395. https://doi.org/10.1016/j.archger.2024.105395 (2024).
Lopez-Sanz, D. et al. Scoping review of neuroimaging studies investigating frailty and frailty components. Front. Med. (Lausanne). 5, 284. https://doi.org/10.3389/fmed.2018.00284 (2018).
Jiang, R. et al. Associations of physical frailty with health outcomes and brain structure in 483 033 middle-aged and older adults: a population-based study from the UK biobank. Lancet Digit. Health. 5, e350–e359. https://doi.org/10.1016/S2589-7500(23)00043-2 (2023).
Wardlaw, J. M., Hernandez, V., Munoz-Maniega, S. & M. C. & What are white matter hyperintensities made of? Relevance to vascular cognitive impairment. J. Am. Heart Assoc. 4, 001140. https://doi.org/10.1161/JAHA.114.001140 (2015).
Avila-Funes, J. A. et al. Vascular cerebral damage in frail older adults: the AMImage study. J. Gerontol. Biol. Sci. Med. Sci. 72, 971–977. https://doi.org/10.1093/gerona/glw347 (2017).
Sexton, C. E., Kalu, U. G., Filippini, N., Mackay, C. E. & Ebmeier, K. P. A meta-analysis of diffusion tensor imaging in mild cognitive impairment and alzheimer’s disease. Neurobiol. Aging. 32, 2322e2325–2322e2318. https://doi.org/10.1016/j.neurobiolaging.2010.05.019 (2011).
Kantarci, K. et al. White matter integrity determined with diffusion tensor imaging in older adults without dementia: influence of amyloid load and neurodegeneration. JAMA Neurol. 71, 1547–1554. https://doi.org/10.1001/jamaneurol.2014.1482 (2014).
Snir, J. A., Bartha, R. & Montero-Odasso, M. White matter integrity is associated with gait impairment and falls in mild cognitive impairment. Results from the gait and brain study. Neuroimage Clin. 24, 101975. https://doi.org/10.1016/j.nicl.2019.101975 (2019).
Zhang, W., Zhou, C. & Chen, A. A systematic review and meta-analysis of the effects of physical exercise on white matter integrity and cognitive function in older adults. Geroscience 46, 2641–2651. https://doi.org/10.1007/s11357-023-01033-8 (2024).
Zhang, S. et al. Value of white matter hyperintensity volume and total white matter volume for evaluating cognitive impairment in patients with cerebral small-vessel disease. Front. Aging Neurosci. 15, 1096808. https://doi.org/10.3389/fnagi.2023.1096808 (2023).
Asken, B. M. et al. Lower white matter volume and worse executive functioning reflected in higher levels of plasma GFAP among older adults with and without cognitive impairment. J. Int. Neuropsychol. Soc. 28, 588–599. https://doi.org/10.1017/S1355617721000813 (2022).
O’Caoimh, R. et al. Prevalence of frailty in 62 countries across the world: a systematic review and meta-analysis of population-level studies. Age Ageing. 50, 96–104. https://doi.org/10.1093/ageing/afaa219 (2021).
Ofori-Asenso, R. et al. Global incidence of frailty and prefrailty among Community-Dwelling older adults: A systematic review and Meta-analysis. JAMA Netw. Open. 2, e198398. https://doi.org/10.1001/jamanetworkopen.2019.8398 (2019).
Duchowny, K. A. et al. Associations between handgrip strength and dementia Risk, Cognition, and neuroimaging outcomes in the UK biobank cohort study. JAMA Netw. Open. 5, e2218314. https://doi.org/10.1001/jamanetworkopen.2022.18314 (2022).
Tajimi, T. et al. Association of gait speed with regional brain volumes and risk of dementia in older japanese: the Hisayama study. Arch. Gerontol. Geriatr. 106, 104883. https://doi.org/10.1016/j.archger.2022.104883 (2023).
Kim, G. S. et al. Cross-Lagged Panel Analysis between Physical Frailty, Cognitive Function, and Falls by Sex. J Am Med Dir Assoc 24, 1541–1548 e1541 (2023). https://doi.org/10.1016/j.jamda.2023.07.004
Del Brutto, O. H., Recalde, B. Y., Rumbea, D. A. & Mera, R. M. Pre-Existing frailty is unrelated to progression of diffuse subcortical damage of vascular origin: A longitudinal prospective study in Community-Dwelling older adults. J. Prim. Care Community Health. 13, 21501319221092245. https://doi.org/10.1177/21501319221092245 (2022).
Schweitzer, N. et al. Sex-specific risk factors and clinical dementia outcomes for white matter hyperintensities in a large South Korean cohort. Alzheimers Res. Ther. 16, 243. https://doi.org/10.1186/s13195-024-01598-2 (2024).
Kiiti Borges, M. et al. The relationship between physical frailty and mild cognitive impairment in the elderly: A systematic review. J. Frailty Aging. 8, 192–197. https://doi.org/10.14283/jfa.2019.29 (2019).
Meysami, S. et al. Handgrip strength is related to hippocampal and Lobar brain volumes in a cohort of cognitively impaired older adults with confirmed amyloid burden. J. Alzheimers Dis. 91, 999–1006. https://doi.org/10.3233/JAD-220886 (2023).
Ramanoel, S. et al. Gray matter volume and cognitive performance during normal Aging. A Voxel-Based morphometry study. Front. Aging Neurosci. 10, 235. https://doi.org/10.3389/fnagi.2018.00235 (2018).
Rosso, A. L. et al. Slowing gait and risk for cognitive impairment: the hippocampus as a shared neural substrate. Neurology 89, 336–342. https://doi.org/10.1212/WNL.0000000000004153 (2017).
Fjell, A. M. et al. What is normal in normal aging? Effects of aging, amyloid and alzheimer’s disease on the cerebral cortex and the hippocampus. Prog Neurobiol. 117, 20–40. https://doi.org/10.1016/j.pneurobio.2014.02.004 (2014).
Garnier-Crussard, A. et al. White matter hyperintensities across the adult lifespan: relation to age, Abeta load, and cognition. Alzheimers Res. Ther. 12, 127. https://doi.org/10.1186/s13195-020-00669-4 (2020).
Allali, G. et al. Brain structure covariance associated with gait control in aging. J. Gerontol. Biol. Sci. Med. Sci. 74, 705–713. https://doi.org/10.1093/gerona/gly123 (2019).
Moonen, J. E. F. et al. Race, sex, and mid-life changes in brain health: cardia MRI substudy. Alzheimers Dement. 18, 2428–2437. https://doi.org/10.1002/alz.12560 (2022).
Salat, D. H. et al. Regional white matter volume differences in nondemented aging and alzheimer’s disease. Neuroimage 44, 1247–1258. https://doi.org/10.1016/j.neuroimage.2008.10.030 (2009).
Lohner, V. et al. Relation between Sex, Menopause, and white matter hyperintensities: the Rhineland study. Neurology 99, e935–e943. https://doi.org/10.1212/WNL.0000000000200782 (2022).
Thurston, R. C. et al. Reproductive hormones in relation to white matter hyperintensity volumes among midlife women. Alzheimers Dement. 20, 6161–6169. https://doi.org/10.1002/alz.14093 (2024).
Sharma, B., Wang, M., McCreary, C. R., Camicioli, R. & Smith, E. E. Gait and falls in cerebral small vessel disease: a systematic review and meta-analysis. Age Ageing. 52 https://doi.org/10.1093/ageing/afad011 (2023).
Ogama, N. et al. Impact of regional white matter hyperintensities on specific gait function in alzheimer’s disease and mild cognitive impairment. J. Cachexia Sarcopenia Muscle. 12, 2045–2055. https://doi.org/10.1002/jcsm.12807 (2021).
Kim, Y., Han, B. G. & Ko, G. E. S. g. Cohort profile: the Korean genome and epidemiology study (KoGES) consortium. Int. J. Epidemiol. 46, e20. https://doi.org/10.1093/ije/dyv316 (2017).
Won, C. W. et al. Korean frailty and aging cohort study (KFACS): cohort profile. BMJ Open. 10, e035573. https://doi.org/10.1136/bmjopen-2019-035573 (2020).
Seo, Y., Kim, M., Shim, H. & Won, C. W. Differences in the association of neighborhood environment with physical frailty between urban and rural older adults: the Korean frailty and aging cohort study (KFACS). J. Am. Med. Dir. Assoc. 22, 590–597e591. https://doi.org/10.1016/j.jamda.2020.09.044 (2021).
Lee, Y., Kim, E., Yun, J. & Chuck, K. W. The influence of multiple frailty profiles on institutionalization and all-cause mortality in community-living older adults. J. Cachexia Sarcopenia Muscle. 13, 2322–2330. https://doi.org/10.1002/jcsm.13033 (2022).
Collins, D. L., Evans, A. C. & Animal Validation and applications of nonlinear Registration-Based segmentation. Int. J. Pattern recognit. Artif. Intell. 11, 1271–1294. https://doi.org/10.1142/s0218001497000597 (1997).
Tsuchida, A. et al. Age-Related variations in regional white matter volumetry and microstructure during the Post-adolescence period: A Cross-Sectional study of a cohort of 1,713 university students. Front. Syst. Neurosci. 15, 692152. https://doi.org/10.3389/fnsys.2021.692152 (2021).
Lee, H. J. et al. Association between change in lifestyle and cognitive functions among elderly koreans: findings from the Korean longitudinal study of aging (2006–2016). BMC Geriatr. 20, 317. https://doi.org/10.1186/s12877-020-01693-7 (2020).
Lee, S. J. et al. Moderate alcohol intake reduces risk of ischemic stroke in Korea. Neurology 85, 1950–1956. https://doi.org/10.1212/WNL.0000000000002165 (2015).
Wang, J. et al. Comparison of approaches to control for intracranial volume in research on the association of brain volumes with cognitive outcomes. Hum. Brain Mapp. 45, e26633. https://doi.org/10.1002/hbm.26633 (2024).
Acknowledgements
We appreciate to all participants and staff of the Korean Genome and Epidemiology Study Cardiovascular Disease Association Study-Cognitive Aging, supported by the Korea National Institute of Health research project (Project No. 2020-E7104-00, 2021-E0607-00, 2022-E0601-00), for their contributions.
Funding
This research was supported by the Korea National Institute of Health research project (project No. 2020-E7104-00, 2021-E0607-00, 2022-E0601-00).
Author information
Authors and Affiliations
Contributions
DYL: Conceptualization, Data curation, Formal analysis, Software, Methodology, Visualization, Writing – Original draft, Review & Editing; YSK: Review & Editing, Supervision; MKK: Data curation, Review editing; MHS: Review & Editing; SBK: Review & Editing; HCK: Review & Editing; IC: Review & Editing; JML: Review & Editing; YK: Review & Editing; YMK: Conceptualization, Data curation, Review & Editing, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study protocol was approved by the Institutional Review Boards of Hanyang University (HYU-2019-11-002-1, HYUIRB-202011-012), Chonnam University (06–062), Keimyung University (2020-01-058), Yonsei Wonju College of Medicine (CR320120), and Yonsei University (4-2019-1206, 4-2020-0817). The study was conducted in accordance with the ethical standards of the Declaration of Helsinki. All participants provided written informed consent prior to this study. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, D.Y., Kim, Y.S., Kim, M.K. et al. Physical frailty and MRI markers of structural brain integrity in the community-dwelling late middle-aged and old adults. Sci Rep 15, 40464 (2025). https://doi.org/10.1038/s41598-025-24208-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24208-z