Abstract
The systematics of lecanoroid lichens, particularly the genus Lecanora, are fraught with difficulties arising from hidden diversity and the morphological plasticity of these organisms. These challenges are further complicated in tropical regions, where despite the richness of the lichen species systematic studies exploring their diversity are still limited. Therefore, the recent molecular survey of Bolivian Lecanora s.l. revealed some taxonomic novelties including the discovery of the distinct evolutionary lineage representing a new genus, Flavonora described here. The genus Flavonora, is distinguished by the following features: brown-orange to reddish pigmentation of hypothecium and conidia if present filiform, in combination with lecanorine apothecia, large crystals always present in the amphithecium, brown-green epihymenium granulose or not, granules soluble in KOH, large spores up to 19.5 μm long, and the presence of perlatolic, 2’-O-methylperlatolic, confluentic acids, as well as usnic acid or xanthones in addition to atranorin. Comprehensive molecular data (mtSSU, nrITS, LSU, rpb1, rpb2, and mcm7) substantiates the establishment of the genus Flavonora as a new genus including the following species: Flavonora boliviana sp. nov., F. inversa comb. nov., F. fimbriatula comb. nov., and F. stramineoalbida comb. nov. (the generic type). Additionally, a focused analysis of Flavonora using mtSSU and nrITS loci was conducted to explore intrageneric relationships, revealing that F. inversa populations from Bolivia and Scotland are genetically distinct, suggesting potential cryptic speciation within this taxon.
Similar content being viewed by others
Introduction
The taxonomic classification of lecanoroid lichens exhibits a significant challenge due to their cryptic diversity, notable morphological plasticity, and their unresolved phylogenetic relationships. This implies especially to the tropical regions where contrary to the richness of lichen species systematic studies revealing the diversity are still insufficient. Lecanora (Lecanoraceae) is one of the genera comprising lichens with lecanorine apothecia and crustose rarely lobate thalli containing green-algal photobionts of the genus Trebouxia and is characterized by hyaline, non-septate ascospores, and Lecanora-type asci. It is one of the richest lichen genera found worldwide, and in the traditional circumscription it comprises more than 550 species1, with a type species L. allophana2. In the recent six-loci phylogenetic framework by Medeiros et al.3, such a broadly defined genus was proven to be paraphyletic, including Lecanora s. lat. and MPRPS clades (incorporating genera Myriolecis, Protoparmeliopsis, Rhizoplaca, Bryonora and groups ‘Lecanora’ polytropa and ‘L.’ saligna). Furthermore, the evolutionary lineage of Lecanora s. lat. remains polyphyletic3.
One of the distinct groups of Lecanora species with dark hypothecium was subjected to the comprehensive studies by Lumbsch et al. in 19964. This study provided detailed information and analysis of 17 tropical taxa, which were corticolous or saxicolous on siliceous rocks, and presented new taxonomic insights and revisions. The species with a pigmented hypothecium and filiform conidia were classified under Lecanora s. str. as L. coronulans-group4. Whereas all known taxa with baciliform conidia were treated in the work as not representing the genus Lecanora and listed among excluded species4. The genus Vainionora was validly and effectively published by Kalb in 1991 in the schedae to Lichenes neotropici Fasc. 12: p. 35. The author included five species in the new genus: V. pallidostraminea (Vain.) Kalb, designated as the type genus name, and four other species from South America (V. aemulans, V. flavovirens, V. stramineopallens and V. warmingii); three of these species originated from Brazil. In 2004, Kalb added V. americana as another newly described species to the genus6. The author established the genus based on the wide apothecia, well-developed and coloured hypothecium (brownish-red or yellowish), shorter bacilliform conidia compared to Lecanora s. str. and presence of xanthones5,6. Unfortunately, the genus Vainionora has not been characterized using molecular data since it was described. An attempt was made to establish a robust circumscription of taxa with coloured hypothecium assigned to the Lecanora s.l. as a part of a continuing project towards taxonomic treatment of Bolivian lecanoroid lichens. The species of our concern included: L. alboflavida, L. coronulans, Lecanora aff. fimbriatula, L. stramineoalbida and L. aff. stramineoalbida. The taxa were investigated in detail including morphological, chemical and molecular evidence, and the results of the study are presented here with a newly described genus.
Material and methods
Morphological, anatomical studies and secondary metabolites analyses
Morphological characters were examined using a Nikon Eclipse i80 light microscope and standard techniques. Cross-section preparations were mounted in either water or a solution containing c. 25% potassium hydroxide (K). Tissue and spore measurements were studied in water. The presence of granulation and crystals were observed under polarized light (pol), and their solubility was assessed using K and a 65% nitric acid (N) solution. The colour reaction of the epihymenium was tested using K, N, and 10% hydrochloric acid (HCl). The measurement results of 40 spores per specimen were presented as minimum–(mean ± standard deviation)–maximum for length (L) and for width (W), followed by spore’s ratio (L/W), and accompanied by the number of measurements (N). Thin-layer chromatography (TLC) was used to study the lichen substances, following the techniques described by Culberson and Kristinsson7 and Orange et al.8. Lichen substances were analysed using solvents A and C.
DNA extraction, PCR and sequencing
Apothecia excised from a solitary thallus were subjected to a thorough cleaning process using sterile, distilled water on a microscope slide. The QIAamp DNeasy™ Plant Mini Kit (Qiagen, Germany) was used to isolate the DNA, following the instructions provided by the manufacturer. To perform a phylogenetic analysis of Lecanoraceae, we applied the amplification technique to target specific genetic regions, which included the internal transcribed spacer (ITS), and mitochondrial small subunit (mtSSU). To amplify the ITS region, which encompasses the ITS1 spacer, 5.8S rRNA, and ITS2 spacer, the primers ITS1F9 and LR310 were utilized, whereas mtSSU was amplified using the primers MSU1 and MSU711. Polymerase chain reaction (PCR) was conducted using 1 μl of the DNA extract in a total volume of 25 μl, following the PCR conditions outlined by Medeiros3. Before sequencing PCR, the amplicon was assessed on a 1% agarose gel and purified using exonuclease I and shrimp alkaline phosphatase (ExoSap). The sequencing reactions were carried out utilizing the PCR primers. However, in one instance (F. stramineoalbida), primers mrSSU1 and mrSSU3R12 were substituted for MSU1 and MSU7 for sequencing of the mtSSU amplicons. Sanger sequencing was performed in Macrogen Europe B.V. (Amsterdam, the Netherlands). Supplementary materials 1 provides the GenBank numbers for all the sequences used, including the newly generated ones.
Taxon sampling for phylogenetic analyses
To resolve the phylogenetic relationship of the taxa being the subject of the study, i.e., L. alboflavida, L. coronulans, Lecanora aff. fimbriatula, L. stramineoalbida and L. aff. stramineoalbida the dataset was created including downloaded GenBank’s sequences of the latter taxa and sequences representing major evolutionary lineages within the genus Lecanora s. lat., as well as representatives of selected genera segregated from Lecanora in its traditional sense. The selection of sequences was made sourcing the following publications dedicated to lecanoroid lichens systematics:3,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32. During this study, eight new sequences were generated for nrITS and five for mtSSU. The sequences were deposited in GenBank. Letharia columbiana, Protoparmelia badia, and Protoparmelia picea were used as outgroups3,14. Sequences retrieved from GenBank and newly generated sequences utilized in phylogenetic studies are listed in Supplementary materials 1, Supplementary materials 2.
Phylogenetic analyses
The trimming of the ends of newly generated sequences was performed using the Geneious Prime 2024.0 software. The NCBI nucleotide database was used in conjunction with BLAST to analyse the sequences and detect any indications of contamination or misidentification of specimens. The phylogenetic framework was based on six regions: mtSSU, nrITS (including ITS1, 5.8S and ITS2), nrLSU, rpb1, rpb2, and mcm7 (8 loci) proved informative for lecanoroid lichens3,14. Six distinct alignments were prepared to represent each region. The sequences were aligned using the MAFFT online server, specifically utilizing the G-INS-1 option as described by Katoh33. Subsequently, all instances of uncertain positions were eliminated by employing Gblocks v. 0.91b34,35 through the website http://phylogeny.lirmm.fr/phylo_cgi/one_task.cgi?task_type=gblocks. Each alignment for a single locus was then utilized as input for a maximum likelihood phylogenetic analysis conducted in IQ-TREE 1.6.1236,37, executed on the CIVIB server38 to assess incongruence. The concatenated alignment, encompassing all loci and representing the fungal symbiotic partner, was used as input for phylogenetic analysis in IQ-TREE, including UFBoot2 bootstrap analysis, SH-aLRT, and approximate Bayes tests. The input partitioning scheme divided the concatenated alignment based on the individual loci. To evaluate support for each single-locus tree, phylogenetic tree reconstructions were performed with 5000 ultrafast bootstrap pseudo-replicates, following the approach outlined by Hoang39. Nodes that attained bootstrap values of ≥ 80% of the SH-aLRT test or > 0.95 of accurate approximation of branch supports (aBayes) or ≥ 95% of UFBoot2 support were considered to be strongly supported. The resulting tree was visualized using Figtree v.1.4.4.
Results and discussion
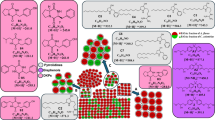
Phylogenetic reconstruction (Fig. 1) was based on six regions: mtSSU, nrITS, nrLSU, rpb1, rpb2, mcm7. In total, 305 sequences including 68 taxa were used. The exact numbers of sequences for a particular locus are 54, 59, 59, 55, 45, and 33 respectively. Detailed alignment statistics are presented in Table 1.
Phylogeny of Flavonora and related genera were inferred from a concatenated six-locus dataset (mtSSU + nrITS + nrLSU + rpb1 + rpb2 + mcm7). The concatenated alignment was input for SH-aLRT and accurate approximation of branch supports (aBayes) tests and UFBoot2 bootstrap analysis in IQ-TREE. Branches in bold received bootstrap ≥ 80% of SH-aLRT test or > 0.95 of aBayes branch supports or ≥ 95% of UFBoot2 support.
Substitution models were as follows 1—HKY + F + I + G4; 2—K2P + G4; 3—TIM2e + I + G4; 4—K2P + I + G4; 5—TIM + F + I + G4.
Most of the genetic lineages are well supported in our phylogeny. The phylogenetic analysis reveals that the samples named Lecanora alboflavida, L. aff. fimbriatula, L. stramineoalbida, and L. aff. stramineoalbida form a distinct, statistically supported clade. Notably, this lineage includes not only specimens from Bolivia but also reference material from Scotland. We recognize this clade as an undescribed genus and named it Flavonora and provide a taxonomic treatment for all representatives of the new genus below. Lecanora coronulans was found to be phylogenetically distant and will not be discussed further in this study.
The tree topology is consistent with the previous studies on Lecanoraceae3,14, except the position of Lecidella, which represents a sister lineage to the most recently described genus Nimisora16 in our study. Both these genera form a statistically supported clade, sister to the clade comprising representatives of the genera Glaucomaria, Lecanora, Myriolecis, Protoparmeliopsis, Pulvinora and Rhizoplaca.
As one of the specimens of Flavonora inversa was recovered in a clade with F. fimbriatula, an additional analysis exclusively with mtSSU and nrITS regions was carried out for selected voucher specimens (Supplementary materials 2). Phylogeny was done for 21 mtSSU sequences and 18 nrITS. The second tree (Fig. 2) clearly shows that F. fimbriatula is genetically distinct from F. inversa. Furthermore, Bolivian specimens of F. inversa (in green) are separated from those from Scotland (in blue) on the tree which might indicate the case of cryptic speciation. For this analysis we additionally used more specimens of F. inversa and F. stramineoalbida in contrast to the six loci phylogeny, where we selected specimens with a larger number of available DNA regions, including protein-coding ones.
Phylogeny of Flavonora and Lecanora allophana was inferred from a concatenated two-locus dataset (mtSSU + nrITS). The concatenated alignment was input for SH-aLRT and accurate approximation of branch supports (aBayes) tests and UFBoot2 bootstrap analysis in IQ-TREE. Branches in bold received bootstrap ≥ 80% of SH-aLRT test or > 0.95 of aBayes branch supports or ≥ 95% of UFBoot2 support.
The phylogenetic topologies depicted in Figs. 1 and 2 demonstrate incongruence in the placement of certain Flavonora specimens. Figure 1 offers a robust and statistically supported topology for elucidating higher taxonomic ranks, while Fig. 2 focuses on resolving species-level relationships within the genus Flavonora. In Fig. 2, F. inversa 4 is nested within the clade corresponding to this species, consistent with its morphological, anatomical, and chemical characteristics concerning other F. inversa specimens. Notably, F. fimbriatula does not cluster with F. inversa 4 in this analysis. Considering the match between phenotypic and chemical data with the species-level phylogeny in Fig. 2, it has been determined to retain the current taxonomic designation for F. inversa 4 rather than reclassifying it as F. fimbriatula.
According to our morphological observations, the representatives of the genus Flavonora share numerous features, and those are: lecanorine apothecia, large crystals always present in the amphithecium, green–brown epihymenium granulose or not, granules soluble in KOH, orange-brown to reddish pigmentation of hypothecium, large spores up to 19.5 μm long, and filiform conidia if present. Chemical profile of Flavonora species is characterized by the presence of depsidones (perlatolic, 2’-O-methylperlatolic, confluentic acids) and usnic acid or xanthones in addition to atranorin.
The newly described genus, although separate genetically, has numerous features in common with Lecanora s. str. that is extremely diverse concerning morpho-anatomical and chemical evidence. Dark hypothecium limits this similarity to the L. coronulans group within Lecanora s. str., from which Flavonora is phylogenetically distant. In many respects, Flavonora is very similar to the genus Vainionora that is also characterized by the pigmented hypothecium. However, the presence of filiform instead of bacilliform conidia sets the former genus apart. Flavonora also appears to be less closely aligned with tropical areas as suggested by the broad ecological preferences indicated in this genus. Considering all this, the classification of Flavonora as a distinct genus within Lecanoraceae is justified most of all on its phylogenetic distinctiveness.
Taxonomy
Flavonora Mazur, Malíček & Śliwa, gen. nov.
MycoBank: 853319.
Diagnosis
The combination of the following characteristics sets the genus apart from other genera within Lecanoraceae: the lecanorine apothecium margin, large crystals always present in the amphithecium, green–brown epihymenium granulose or not, granules soluble in KOH, orange-brown pigmentation of hypothecium, presence of perlatolic, 2’-O-methylperlatolic, confluentic acids, as well as usnic acid or xanthones in addition to atranorin, large spores up to 19.5 μm long, and filiform conidia if present.
Generic type
Flavonora stramineoalbida (Vain.) Mazur, Malíček & Śliwa, comb. nov.
Figure 3E–F
Morphological variations of Flavonora species: A–F. boliviana, Bolivia, A.Flakus 28846 (KRAM); B–F. inversa, Bolivia, A.Flakus 29124 (KRAM); C–F. fimbriatula, Bolivia, A.Flakus 29926 (KRAM); D–F. fimbriatula, India, J.Watt (BM!); E–F. stramineoalbida, Bolivia, A.Flakus 19939 (KRAM); F–F. stramineoalbida, Saint Vincent and the Grenadines, W.R.Elliott (BM!).
Etymology
The epithet refers to the slightly yellowish tinge of the thallus caused by the presence of usnic acid or xanthones in addition to atranorin (flavo = yellow, nora as sharing several features with Lecanora including Lecanora-type asci and filiform conidia).
The species
Flavonora boliviana Mazur & Śliwa, sp. nov.
MB: 856419
Figure 3A
Diagnosis
The species is characteristic due to its conspicuous, thick, rimose to areolate thallus, creamy to olive or grey discs of apothecia, greenish brown and granulose epihymienium, orange-red hypothecium, and large and broadly ellipsoid ascospores: 16.0–19.5 × 7.1–10.0 μm (L/W 2.1). Secondary metabolites produced: atranorin, confluentic acid, perlatolic acid, usnic acid, and a complex of unknown substances.
Etymology
The epithet refers to the country of origin of the type collection.
Type
BOLIVIA. Dept. La Paz, prov. Nor Yungas, Coroico village, 1870 m alt., 16°11′56′′S 67°43′26′′W, 10 Jan. 2020, P.Rodriguez 4004 & A.Flakus (holotype: KRAM L-74769; isotype: LPB).
Description
Thallus conspicuous, grey, green or white, thick, rimose to areolate, verrucose, pruinose or epruinose, irregular in outline; areoles undulate and irregular. Prothallus not visible or black, dark-green filmlike, or white filamentous. Apothecia scarce to abundant, scattered, adnate to sessile, 0.3–1.9 mm in diam.; margin lecanorine, circular, undulate or flexuose, crenate, prominent, at the same level with the disc or below; discs concave, flat to convex, epruinose, creamy, sandy, olive or grey, without blackness and discolouration.
Amphithecium, measured in the middle of the thalline margin, 120 μm thick, with crystals (pol +) insoluble in K and insoluble in N; algal cells present but scarce; amphithecial cortex gelatinous, 30 μm thick; parathecium not visible. Hymenium hyaline, 70 μm high. Epihymenium greenish brown, HCl‒, K + (discoloured), N‒, with granules (pol +) soluble in K and insoluble in N. Hypothecium orange-red, incl. subhymenium 110 μm high; subhymenium olive. Paraphyses simple, slender, 3.4 μm thick, not thickened in the apical part. Asci clavate, 8-spored. Ascospores simple, hyaline, ellipsoid, 16.0–(17.9 ± 1.3)–19.5 μm × 7.1– (8.4 ± 0.8)–10.0 μm, L/W ratio 2.1, N = 40. Pycnidia and conidia unknown.
Chemistry
Atranorin, confluentic acid, perlatolic acid, usnic acid, and a complex of unknown substances detected in TLC. Characteristic of the unknown substances: UN1, UN2 and UN3 (in solvent A UN1—RF 0,5 daylight before sulfuric acid, light red, after light brown; UN2 –RF 0,3 daylight before sulfuric acid, light red, after light brown; UN3—RF 0,2 daylight before sulfuric acid, not visible, after light brown; in solvent C – UN1 is not visible, UN2—RF 0,2 daylight before and after sulfuric acid, grey; UN3 —RF 0,1 daylight before and after sulfuric acid, grey).
Distribution and ecology
This corticolous species from Bolivia has been repetitively recorded in some areas of the country (dept. La Paz, e.g. prov. Nor Yungas). It might be the species neotropical distribution range since it was discovered also from eastern Caribbean during the revision study of herbarium materials.
Specimens examined
BOLIVIA. Dept. La Paz, prov. Franz Tamayo, between Apollo and Vaqueri, 1565 m alt., 14°39′39′′S, 68°27′46′′W, 18 Nov. 2016, A.Flakus 28312 (KRAM L-74759, LPB); dept. La Paz, prov. Nor Yungas, Coroico village, Uchumachi footpath, 1897 m alt., 16°11′36′′S, 67°43′21′′W, 23 Nov. 2016, A.Flakus 28846 (KRAM L-74761, LPB); ibid., A.Flakus 28854 (KRAM L-74755, LPB); dept. La Paz, prov. Nor Yungas, Coroico village, 1550 m alt., 16°11′10′′S, 67°43′16′′W, Dec. 2019, P.Rodriguez 3995 (KRAM L-74760, LPB); ibid., P.Rodriguez 4001 (KRAM L-74756, LPB); dept. La Paz, prov. Nor Yungas, Coroico village, 1870 m alt., 16°11′56′′S 67°43′26′′W, 10 Jan. 2020, P.Rodriguez 3996 (KRAM L-74758, LPB); ibid., P.Rodriguez 4003 (KRAM L-74757, LPB).
Notes
The taxon was preliminary circumscribed as Lecanora aff. stramineoalbida in the phylogenetic framework by Medeiros et al.3 because its morphology and anatomy are almost the same as in L. stramineoalbida. However, the species differ morphologically, in the composition of secondary metabolites and genetically (Table 2, Fig. 2). Two other known species resemble the newly described one, namely Lecanora hypocrocea Vain. and L. dispersogranulata Szatala. The former species is different due to its apothecial discs, which are orange-brown and presence of xanthones in the thallus. The latter species can be distinguished by its paler hypothecium − yellowish instead of the yellow–red one. Flavonora boliviana morphologically resembles Lecanora achroa Nyl., L. helva Stizenb., and L. leprosa Fée. However, all three species differ by having a hyaline hypothecium.
Flavonora fimbriatula (Stirt.) Mazur, Malíček & Śliwa, comb. nov.
MB: 853322.
Figures 3C–D.
Basionym
Lecanora fimbriatula Stirt., Proceedings of the Philosophical Society of Glasgow 11: 311 (1879).
Type
‘India. Neilgherri, [D.G.] Watt’ (holotype: BM!).
Description
Thallus grey-white, conspicuous, thick, rimose, irregular in outline, without pruinosity and vegetative propagules. Prothallus present, film-like dark green and filamentous white. Apothecia scarce and scattered, sessile, 0.3‒2 mm in diam.; margin lecanorine, circular or undulate, continuous, thin, below the disc level to elevated; discs concave, flat or convex, epruinose, black brown.
Amphithecium, measured in the middle of the thalline margin, 160 μm thick; with large crystals insoluble in K and insoluble in N; algal cells present but scarce; amphithecial cortex of gelatinous structure, 40 μm thick; parathecium absent. Hymenium hyaline, ca. 75 μm high. Epihymenium greenish black, HCl‒, K + green, N + red, without granules. Hypothecium orange, incl. subhymenium 130 μm high; subhymenium yellow. Paraphyses simple, slender, ca. 1.7 μm thick, in the apical part up to 3.4 μm. Asci clavate, 8-spored. Ascospores simple, hyaline, broadly ellipsoid, 17.9–(18.2 ± 0.4)–19.0 μm 9.9– (10.8 ± 0.4)–11.2 μm, L/W ratio 1.7, N = 40. Pycnidia and conidia not seen; filiform according to Lumbsch et al.4.
Chemistry
Atranorin, confluentic acid, and xanthones detected in TLC. Characteristic of the xanthones: X1, X2 and X3 (in solvent A, X1—RF 0,63 daylight before and after sulfuric acid, orange, in UV yellow; X2—RF 0,57 daylight before sulfuric acid, orange, after light orange, in UV white; X3—RF 0,52 daylight before sulfuric acid, orange, after light blue, in UV blue; in solvent C, X1 – RF 0,58 daylight before sulfuric acid, yellow, after brown, in UV light yellow; X2—RF 0,52 daylight before sulfuric acid, orange, after blue, in UV white; X3—RF 0,45 daylight before sulfuric acid, orange, after brown, in UV light blue).
Distribution and ecology
This is the first species recorded from the South America. It was found in Bolivia at an elevation above 2000 m a.s.l. It was also seen from India. According to Lumbsch et al.4 the species occurs on the bark with Lecanora spp. and Pyxine subcinerea Stirt. in areas of high humidity, e.g. a cloud forest with Alnus acuminata near the river, as well as at high elevations in Central Himalayas.
Specimens examined
BOLIVIA. Dept. Cochabamba, prov. Carrasco, protected area ‒ Parque Nacional Carrasco, Sehuencas, 2217 m alt., 17°29′57′′S, 65°16′30′′W, 5 Nov. 2016, A.Flakus 27926 (KRAM L-74754, LPB).
Notes
Characteristic features of F. fimbriatula are dark apothecial discs, epihymenium that reacts K + greenish, large spore’s size reaching 19 μm length and 11 μm wide, and the presence of 2’‒O‒methylperlatolic acid4. The latter secondary metabolite was not detected in the Bolivian specimen of F. fimbriatula, however, it is treated as a chemotype of this species. See Table 2 for comparison with other Flavonora species.
Flavonora inversa (Nyl.) Mazur, Malíček & Śliwa, comb. nov.
MB: 853321.
Nomenclatural type of Flavonora inversa collected by Taylor (BM; the lectotype), scale = 1 mm.
Basionym
Lecanora inversa Nyl., Flora (Regensburg) 62: 361 (1879) ≡ Ochrolechia inversa (Nyl.) J. R. Laundon, Lichenologist 2: 130 (1963)40.
Type
‘Ireland, on furze Ulex europaeus, Finnihy River, Co. Kerry, Taylor’ [Flora (Regensburg) 62: 361 (1879)], (lectotype: BM!—BM000975547, designated here).
Description
Thallus pale grey-yellowish or greenish white, conspicuous, thick, rimose, verrucose, irregular in outline, with or without numerous vegetative propagules. Prothallus not visible or black, filmlike or white filamentous. Soralia usually present, 0.4‒1.3 mm in diam., white to greenish, rounded, regular to erumpent, or part of the thallus eroding into soredia. Apothecia absent or scarce and scattered, adnate to sessile, 0.3–2.3 mm in diam.; margin lecanorine, circular or flexuose, crenate, verruculose, prominent and below the disc level; discs concave or convex, epruinose, olive grey, an unusual umbo-like structure concolourous with the thallus is visible in older apothecia.
Amphithecium, measured in the middle of thalline margin, 100‒160 μm thick; containing giant crystals, 40‒55 μm in diam., (pol +) insoluble in K and insoluble in N; algal cells scarce; amphithecial cortex, if visible, 100 μm thick; parathecium not visible. Hymenium hyaline, ca. 70 μm high. Epihymenium brown or orange-brown, HCl‒, K + (discoloured), N‒; containing granules (pol +) soluble in K and insoluble in N. Hypothecium orange-brown to dark orange-brown (in K very dark orange-brown), incl. subhymenium 185‒190 μm high; subhymenium hyaline. Paraphyses simple, slender, 2‒3.4 μm thick, not thickened in the apical part. Asci clavate, 8-spored. Ascospores simple, hyaline, broadly ellipsoid, 13.6–(13.6 ± 0.2)–14.5 μm × 8.5–(10.2 ± 0.3)–10.8 μm, L/W ratio 1.3–1.5, N = 40. Pycnidia and conidia unknown.
Chemistry
Atranorin, arthothelin, thiophanic acid, and two or three unidentified xanthones detected in TLC. Characteristic of the xanthones: X4, X5 and X6 (in solvent A, X4—RF 0,8 daylight before sulfuric acid, not visible, after light yellow, in UV yellow; X5—RF 0,7 daylight before sulfuric acid, not visible, after yellow, in UV yellow; X6—RF 0,52 daylight before sulfuric acid, orange, after orange-violet, in UV orange; in solvent C, X4—RF 0,56 daylight before sulfuric acid, not visible, after orange, in UV white; X5—RF 0,5 daylight before sulfuric acid, not visible, after orange, in UV violet; X6—RF 0,65 daylight before sulfuric acid, light orange, after grey, in UV light orange).
Distribution and ecology
According to Cannon et al.41 the species grows on the acidic bark of old trees in moist forests, such as temperate oceanic and tropical rainforests. Its occurrence in South America and Europe was supported here by molecular evidence. The saxicolous records of the taxon refer to Lecanora alboflavida.
Specimens examined
BOLIVIA. Dept. La Paz, prov. Franz Tamayo, between Apollo and Mapiri, 1521 m alt., 14°38′51′′S, 68°24′44′′W, 18 Nov. 2016, A.Flakus 28262 (KRAM L-74750); dept. La Paz, prov. Nor Yungas, Coroico village, 1550 m alt., 16°11′10′′S, 67°43′16′′W, 18 Nov. 2016, P.Rodriguez 3997 (KRAM L-74751); dept. Santa Cruz, prov. Comarapa, protected area ‒ Parque Nacional y Área Natural de Manejo Integrado Amboró, Remate, 2550 m alt., 17°52′11′′S, 64°20′53′′W, 15 May 2017, A.Flakus 29124 (KRAM L-74770); ibid., A.Flakus 29143 (KRAM L-74752); ibid., A.Flakus 29196 (KRAM L-74753). SCOTLAND. Kirkenbrightshire, Glen Trool, Caldon’s wood, on Betula in woodland, 80 m alt., 55°04′39′′N, 4°30′40′′W, 30 Mar. 2016, B. Coppins (herb. J.Malíček 9718).
Notes
Flavonora inversa [= L. inversa Nyl. (1879)], recently reported as Lecanora alboflavida Taylor (e.g.11,21,44,45), is a rare and little-known species. It has been reported in Norway, France, Macaronesia and Great Britain42, and Bolivia3. The taxonomic classification of this species has been a subject of confusion and debate among researchers43. Taylor’s original description of L. alboflavida in 1836 identified the specimen being saxicolous. However, Nylander described a species named L. inversa based on Taylor’s epiphytic material in 187944. Hue45 concluded that L. inversa Nyl. being identical to L. alboflavida Tayl. Later, Laundon40 examined the material of L. alboflavida from the Taylor’s collection at the Farlow Herbarium, as well as the type specimen of L. inversa deposited in BM; according to Laundon, the specimen in FH labelled as L. alboflavida matches both the morphology and habitat described for L. alboflavida by Taylor and should be considered as the holotype of this name. However, Laundon concluded that L. alboflavida is identical to L. epanora (Ach.) Ach., and therefore, L. alboflavida is simply a synonym of that species. On the hand, Laundon transferred L. inversa to the genus Ochrolechia based on the morphology of paraphyses and with no signs of asci. Despite this conflicting information, Cannon et al.41 consider L. alboflavida to be the correct name for Ochrolechia inversa.
Based on the literature review and our results, we suggest Lecanora inversa is the proper name for the epiphytic material incorrectly reported as L. alboflavida in the past. The name L. alboflavida is considered synonymous with L. epanora as suggested by Laundon40. After studying the original historical collection of L. inversa by Taylor housed at the BM herbarium, we also designate the lectotype for the name. See Table 2 for comparisons with other Flavonora species.
Flavonora stramineoalbida (Vain.) Mazur, Malíček & Śliwa, comb. nov.
MB: 853323.
Figure 4E‒F.
Basionym
Lecanora stramineoalbida Vain., Journal of Botany, British and Foreign 34: 35 (1896).
Type
‘Saint Vincent and the Grenadines. Richmond Valley, W.R.Elliot’ (isotype?: BM!).
Thallus grey, conspicuous, thick, rimose, verrucose, epruinose, irregular in outline. Prothallus not visible or dark-green, filmlike, or white filamentous. Apothecia scarce and scattered, adnate, 0.3–1.1 mm in diam.; margin lecanorine, circular or undulate, crenate, prominent; discs concave, epruinose to slightly grey pruinose, creamy, becoming orange–brown.
Amphithecium, measured in the middle of the thalline margin, 147 μm thick; with large crystals (pol +) insoluble in K and insoluble in N; algal cells abundant; amphithecial cortex amorphous, ca. 62 μm thick; parathecium not visible. Hymenium hyaline, 70 μm high. Epihymenium brown HCl‒, K + (discoloured), N‒, with granules (pol +) soluble in K and insoluble in N. Hypothecium orange-brown, incl. subhymenium 170 μm high; subhymenium olive. Paraphyses simple, slender, 3.4 μm thick, not thickened in the apical part. Asci clavate, 8-spored. Ascospores simple, hyaline, broadly ellipsoid, 12.1–(13.4 ± 1.0)–15.2 μm × 7.1–(7.7 ± 0.6)–9.0 μm, L/W ratio 1.7, N = 40. Pycnidia black. Conidia filiform, straight 21–(23.8)–27 μm × 1–1.7 μm, N = 10.
Chemistry
Atranorin, usnic acid and 2’‒O‒methylperlatolic detected in TLC.
Distribution and ecology
The species (reported under the name L. stramineoalbida) occurs in Central America4, North America (Georgia), Western China and South America (Bolivia) as presented by Śliwa et al.46. In Bolivia it is known from various habitats including sub-Andean Amazon forest, the Yungas cloud forest and Tucumano-Boliviano Forest as well as savannah, between 400 and 2544 m a.s.l. elevations. It is known to be corticolous but was also noted on lignum and once on a rock. The Bolivian record quoted below is confirmed by molecular data.
Specimen examined
BOLIVIA, dept. Tarija, prov. Aniceto Arce, Papachacra, Tucumano-Boliviano montane forest with Alnus acuminata, 2195 m alt., 21°41′36′′S 64°29′33′′W, 26 Nov. 2010, A.Flakus 19939 & J.Quisbert (KRAM L-74984).
Notes
Morphologically F. stramineoalbida is distinguished by its large apothecia up to 1.3 mm in diameter with yellowish to orange-brown discs (that may also become dark grey) and a prominent, entire, flexuose to verrucose margin. The major lichen substances produced by the species are atranorin and 2’-O-methylperlatolic acid, and usnic acid4,46. The species may be confused with Lecanora helva Stizenb. due to the pale colour of its apothecial discs and its chemistry, however, the latter species differs by having a hyaline hypothecium. For comparison with other Flavonora species see Table 2.
Conclusions
The recent circumscription of the genus Flavonora within the family Lecanoraceae offers a new insight into the ongoing discourse concerning the taxonomic boundaries within this group, particularly concerning the species exhibiting a dark hypothecium. Representatives of the genus Lecanora s. lat. having a dark hypothecium are found in tropical regions3,4,24,47,48. However, it is noteworthy that species displaying this feature are also present in temperate climates.
The study conducted by Malíček et al.19 on the phylogeny of corticolous sorediate Lecanora species, including L. alboflavida (currently Flavonora inversa), identified two closely related species Lecanora barkmaniana and L. variolascens. However, both species are characterized by the absence of the dark hypothecium. Moreover, these species were incorporated into the prepared draft of our phylogeny, however they did not cluster within the clade containing the representatives of the newly established genus Flavonora.
Papong et al.24 demonstrated that a dark hypothecium similarly to the presence of usnic acid evolved independently in different lineages of Lecanora using molecular analyses, suggesting that these features are the result of convergent evolution. This has greatly complicated the genus-level taxonomy within Lecanoraceae, as many of the traditionally used morphological and chemical characters do not correspond to monophyletic groups. The presence of dark hypothecium in both Lecanora s. str. and Vainionora species supports this idea of multiple evolutionary origins for these traits. The delimitation of Flavonora additionally confirms the concept that species with dark hypothecia do not form a monophyletic group3,24,48. Furthermore, the findings suggest that dark hypothecia may not be reliable indicators of phylogenetic relationships at the generic level.
The separation of Flavonora highlights the need for continued taxonomic revision within the family Lecanoraceae. While morphological and chemical traits have traditionally been used to differentiate lichen genera, molecular phylogenetic analyses increasingly reveal the limitations of these characters in taxonomic studies and highlights the importance of integrating molecular data into taxonomic studies. Further research should focus on expanding phylogenetic analyses to include additional loci, as well as incorporating a wider range of species from tropical and subtropical regions, in where the evolutionary pressures are likely to be strongest in driving the development of these traits.
Data availability
The data supporting the findings of this study are accessible in GenBank (https://www.ncbi.nlm.nih.gov/genbank/), as detailed in Supplementary materials 1 and 2 and within the accompanying text.
References
Lücking, R., Hodkinson, B. P. & Leavitt, S. D. The 2016 classification of lichenized fungi in the Ascomycota and Basidiomycota—Approaching one thousand genera. Bryologist 119, 361–416. https://doi.org/10.1639/0007-2745-119.4.361 (2017).
Brodo, I. & Vitikainen, O. The typification of Lecanora subfasca (L.) Ach., its varieties, and some of its related taxa published before 1850. Mycotaxon 21, 281–298 (1984).
Medeiros, I. D. et al. Turnover of lecanoroid mycobionts and their Trebouxia photobionts along an elevation gradient in Bolivia highlights the role of environment in structuring the lichen symbiosis. Front. microbiol. 12, 774839. https://doi.org/10.3389/fmicb.2021.774839 (2021).
Lumbsch, H. T., Guderley, R. & Elix, J. A revision of some species in Lecanora sensu stricto with a dark hypothecium (Lecanorales, Ascomycotina). Bryologist 99, 269–291 (1996).
Kalb, K. Lichenes Neotropici ausgegeben von Klaus Kalb. Fascikel XII (No. 476–525). vol. Publ. by author, Neumarkt/OPf (1991).
Kalb, K. New or otherwise interesting lichens II. Biblioth. Lichenol. 88, 301–329 (2004).
Culberson, C. F. & Kristinsson, H. A standardized method for the identification of lichen products. J. Chromatogr. A. 46, 85–93 (1970).
Orange, A., James, P. W. & White, F. J. Microchemical methods for the identification of lichens (British Lichen Society, 2001).
Gardes, M. & Bruns, T. D. ITS primers with enhanced specificity for basidiomycetes—Application to the identification of mycorrhizae and rusts. Mol. Ecol. 2, 113–118 (1993).
Vilgalys, R. & Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 172, 4238–4246 (1990).
Zhou, S. & Stanosz, G. R. Primers for amplification of mt SSU rDNA, and a phylogenetic study of Botryosphaeria and associated anamorphic fungi. Mycol. Res. 105, 1033–1044 (2001).
Zoller, S., Scheidegger, C. & Sperisen, C. PCR primers for the amplification of mitochondrial small subunit ribosomal DNA of lichen-forming ascomycetes. Lichenologist 31, 511–516 (1999).
Arup, U. & Grube, M. Is Rhizoplaca (Lecanorales, lichenized Ascomycota) a monophyletic genus?. Can. J. Bot. 78, 318–327 (2000).
Zhao, X. et al. Towards a revised generic classification of lecanoroid lichens (Lecanoraceae, Ascomycota) based on molecular, morphological and chemical evidence. Fungal Divers. 78(1), 293–304. https://doi.org/10.1007/s13225-015-0354-5 (2016).
Davydov, E. et al. The new genus Pulvinora (Lecanoraceae) for species of the ‘Lecanora pringlei’ group, including the new species Pulvinora stereothallina. Bryologist 124, 242–256. https://doi.org/10.1639/0007-2745-124.2.242 (2021).
Pérez-Ortega, S., Turégano, Y., Svensson, M. & Zamora, J. C. Nimisora (Lecanoraceae, Ascomycota), a new genus for a common lecideoid epiphytic species from the central Iberian Peninsula. Lichenologist 55(5), 335–345. https://doi.org/10.1017/S0024282923000427 (2023).
Mamut, R., Li, P., Abbas, A. & Fu, C. Morphology, chemistry and molecular phylogeny revealed a new species and a new combination of Myriolecis (Lecanoraceae, Ascomycota) from China. Bryologist 122(3), 375–383. https://doi.org/10.1639/0007-2745-122.3.375 (2019).
Kistenich, S., Timdal, E., Bendiksby, M. & Ekman, S. Molecular systematics and character evolution in the lichen family Ramalinaceae (Ascomycota: Lecanorales). Taxon 67, 871–904. https://doi.org/10.12705/675.1 (2018).
Malíček, J., Berger, F., Palice, Z. & Vondrák, J. Corticolous sorediate Lecanora species (Lecanoraceae, Ascomycota) containing atranorin in Europe. Lichenologist 49(5), 431–455. https://doi.org/10.1017/S002428291700038X (2017).
Leavitt, S. D. et al. Cryptic diversity and symbiont interactions in rock-posy lichens. Mol. Phylogenet. Evol. 99, 261–274. https://doi.org/10.1016/j.ympev.2016.03.030 (2016).
Miadlikowska, J. et al. A multigene phylogenetic synthesis for the class Lecanoromycetes (Ascomycota): 1307 fungi representing 1139 infrageneric taxa, 317 genera and 66 families. Mol. Phylogenet. Evol. 79, 132–168. https://doi.org/10.1080/15572536.2006.11832636 (2014).
Singh, G. et al. The sister-group relationships of the largest family of lichenized fungi, Parmeliaceae (Lecanorales, Ascomycota). Fungal Biol. 117(10), 715–721. https://doi.org/10.1016/j.funbio.2013.08.001 (2013).
Kirika, P., Parnmen, S. & Lumbsch, H. T. Two new species of Lecanora sensu stricto (Lecanoraceae, Ascomycota) from East Africa. MycoKeys 3, 37–47. https://doi.org/10.3897/mycokeys.3.3201 (2012).
Papong, K., Boonpragob, K., Parnmen, S. & Lumbsch, H. T. Molecular phylogenetic studies on tropical species of Lecanora sensu stricto (Lecanoraceae, Ascomycota). Nova Hedwig. 96, 1–13. https://doi.org/10.1127/0029-5035/2012/0072 (2012).
Schoch, C. L. et al. Fungal barcoding consortium; fungal barcoding consortium author list nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for fungi. Proc. Natl. Acad. Sci. U. S. A. 109, 6241–6246. https://doi.org/10.1073/pnas.111701810 (2012).
Śliwa, L., Miadlikowska, J., Redelings, B. D., Molnar, K. & Lutzoni, F. Are widespread morphospecies from the Lecanora dispersa group (lichen-forming Ascomycota) monophyletic?. Bryologist 115, 265–277. https://doi.org/10.1639/0007-2745-115.2.265 (2012).
Leavitt, S. D. et al. Local representation of global diversity in a cosmopolitan lichen-forming fungal species-complex (Rhizoplaca, Ascomycota). J. Biogeogr. 40, 1792–1806. https://doi.org/10.1111/jbi.12118 (2013).
Leavitt, S. D. et al. Complex patterns of speciation in cosmopolitan “rock posy” lichens—disentangling the Rhizoplaca melanophthalma species complex. Mol. Phylogenet. Evol. 61, 45–63. https://doi.org/10.1016/j.ympev.2011.03.020 (2011).
Schmull, M. et al. Phylogenetic affiliations of members of the heterogeneous lichen-forming fungi of the genus Lecidea sensu Zahlbruckner (Lecanoromycetes, Ascomycota). Mycologia 103, 983–1003. https://doi.org/10.3852/10-234 (2011).
Kalb, K., Staiger, B., Elix, J. A., Lange, U. & Lumbsch, H. T. A new circumscription of the genus Ramboldia (Lecanoraceae, Ascomycota) based on morphological and molecular evidence. Nova Hedwigia 86, 23–42 (2008).
Arup, U., Ekman, S., Grube, M., Mattsson, J. E. & Wedin, M. The sister group relation of Parmeliaceae (Lecanorales, Ascomycota). Mycologia 99, 42–49 (2007).
Crespo, A. et al. Testing morphology-based hypotheses of phylogenetic relationships in Parmeliaceae (Ascomycota) using three ribosomal markers and the nuclear RPB1 gene. Mol. Phylogenet. Evol. 44, 812–824 (2007).
Katoh, K., Rozewicki, J. & Yamada, K. D. MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 20, 1160–1166. https://doi.org/10.1093/bib/bbx108 (2019).
Castresana, J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 17, 540–552 (2000).
Dereeper, A. et al. Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 36, 465–469 (2008).
Chernomor, O., von Haeseler, A. & Minh, B. Q. Terrace aware data structure for phylogenomic inference from supermatrices. Syst. Biol. 65, 997–1008. https://doi.org/10.1093/sysbio/syw037 (2016).
Nguyen, L. T., Schmidt, H. A., von Haeseler, A. & Minh, B. Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32(1), 268–274. https://doi.org/10.1093/molbev/msu300 (2015).
Trifinopoulos, J., Nguyen, L. T., von Haeseler, A. & Minh, B. Q. W-IQ-TREE: A fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res. 44, 232–235. https://doi.org/10.1093/nar/gkw256 (2016).
Hoang, D. T., Chernomor, O., von Haeseler, A., Minh, B. Q. & Vinh, L. S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 35, 518–522. https://doi.org/10.1093/molbev/msx281 (2018).
Laundon, J. R. The tamy of sterile crustaceous lichens in the British Isles 2 Corticolous and lignicolous species. Lichenologist 2, 101–151 (1963).
Cannon, P. et al. Lecanorales: Lecanoraceae, including the genera Ameliella, Bryonora, Carbonea, Claurouxia, Clauzadeana, Glaucomaria, Japewia, Japewiella, Lecanora, Lecidella, Miriquidica, Myriolecis, Palicella, Protoparmeliopsis, Pyrrhospora and Traponora. Revis. Br. Irish Lichens 25, 1–83 (2022).
Edwards, B., Aptroot, A., Hawksworth, D. L. & James, P. W. Lecanora Ach. in Luyken (1809). In The Lichens of Great Britain and Ireland (eds Smith, C. W. et al.) 465–502 (British Lichen Society, 2009).
Purvis, O., Coppins, B., Hawksworth, D., James, P. W. & Moore, D. M. The Lichenflora of Great Britain and Ireland (Natural Museum Publication in association with The British Lichen Society, 1992).
Nylander, W. Addenda nova ad Lichenographiam Europaeam Cont. XXXII. Flora 62, 353–362 (1879).
Hue, A. Addenda Nova ad Lichenographiam Europaeam. p. 93 (1886)
Śliwa, L., Rodriguez, F. P., Wilk, K. & Flakus, A. New records of Lecanora for Bolivia II. Pol. Bot. J. 59(1), 97–103 (2014).
Vänskä, H. Lecanora vainioi and L. ahtii, two new lichen species from Brazil, compared with five allied species. Annal. Bot. Fenn. 23, 121–141 (1986).
Dos Santos, L. A. et al. Lecanora s.lat. (Ascomycota, Lecanoraceae) in Brazil: DNA barcoding coupled with phenotype characters reveals numerous novel species. J. Fungi 9, 415. https://doi.org/10.3390/jof9040415 (2023).
Acknowledgements
The authors express their gratitude to the personnel at the Herbario Nacional de Bolivia, Instituto de Ecología, Universidad Mayor de San Andrés, La Paz, for their valuable and enduring collaboration with the lichenologists at the W. Szafer Institute of Botany, Polish Academy of Sciences. Special thanks are due to the collectors, A. Flakus and P. Rodriguez-Flakus, for providing their specimens for this research. M. Kukwa (University of Gdańsk) is greatly thanked for the valuable advice and support with TLC processing and secondary metabolites identification.
Funding
This work was supported by SYNTHESYS + for visiting The Natural History Museum (GB-TAF-TA4-035) and Visegrad Fellowship no. 62320151 awarded to EM, by the long-term research development project RVO 67985939 awarded to JM and by statutory funds from the W. Szafer Institute of Botany, Polish Academy of Sciences, and the National Science Centre, Poland, project 2016/21/B/NZ8/02463 granted to LŚ.
Author information
Authors and Affiliations
Contributions
E.M.: conceptualization, investigation, analyses, visualization, writing original draft and editing; J.M.: conceptualization, revision and editing manuscript; G.W. revision and editing manuscript; L.Ś. investigation, writing, critical revision, editing manuscript. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mazur, E., Malíček, J., Weerakoon, G. et al. Flavonora: a new genus of lecanoroid lichens (Ascomycota, lichenized fungi). Sci Rep 15, 43429 (2025). https://doi.org/10.1038/s41598-025-26012-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26012-1