Abstract
The effects of organic acid (OA)-preserved grain and zinc oxide (ZnO) supplementation on post-weaning (PW) piglet performance and intestinal health were evaluated in a 2 × 2 factorial study. Ninety-six piglets (28 days old) were allocated to four diets: dried grain, OA-preserved grain, dried grain + ZnO, and OA-preserved grain + ZnO, for 35 days. Diets contained 600 g/kg grain (450 g/kg wheat, 150 g/kg barley). On day 10 PW, 28 piglets (n = 7/treatment) were euthanised for small intestinal morphology, gene expression, microbial, and volatile fatty acid (VFA) analyses. OA-preserved grain reduced dietary Ochratoxin A and Deoxynivalenol concentrations and increased average daily gain (P < 0.05), but provided no additional growth benefit when combined with ZnO. ZnO increased feed intake, body weight, colonic Lactobacillus abundance, and villus height-to-crypt depth ratio, while reducing faecal scores and colonic branched-chain fatty acids (P < 0.05). OA-preserved grain increased ileal Faecalibacterium and reduced Escherichia populations, and downregulated duodenal IL17 and ileal FOXP3 expression (P < 0.05). ZnO broadly suppressed pro-inflammatory cytokines and upregulated nutrient transporter genes (SLC15A1, SLC16A1). These findings indicate that OA-preserved grain improves growth and gut health but does not fully replace ZnO in mitigating PW diarrhoea.
Similar content being viewed by others
Introduction
In the commercial setting, piglets are typically weaned before their digestive and immune systems have fully matured1. The sudden transition from sow’s milk to a grain-based diet leads to reduced feed intake, intestinal inflammation, nutrient malabsorption, and microbial imbalances, which together impair growth performance2. This intestinal dysfunction fosters the growth of Escherichia coli, a primary cause of post-weaning diarrhoea (PWD)3. Zinc oxide (ZnO) supplementation has been shown to mitigate PWD and support growth4. However, the European Union banned the use of in-feed antimicrobials, including ZnO, in 2022 due to concerns around the environment and antimicrobial resistance5. Consequently, identifying nutritional strategies to enhance growth and intestinal health post-weaning is critical.
While antimicrobials inhibit microbial proliferation, organic acid (OA) supplementation encourages the growth of beneficial bacteria that outcompete pathogenic bacteria in the gut6. Supplementing piglets with OA modulates the intestinal microbiome, reduces inflammation and diarrhoea, and improves growth performance7. However, the effects of OA depend on factors such as the type of acid used, inclusion levels, and interactions with other dietary elements8. Additionally, OAs, particularly propionic acid, serve as effective grain preservatives9.
Cereal grains are essential components of pig diets10. In temperate, humid climates with significant rainfall, grain moisture content can reach 20–25% during harvest11, promoting fungal growth and mycotoxin production12. Grain can be mechanically dried13 to reduce moisture content, thereby limiting microbial load and preserving quality14. However, inconsistencies in mechanical drying can lead to uneven moisture content and elevated mycotoxin levels15. Moreover, mechanical drying is energy-intensive16 and generates substantial greenhouse gas emissions17.
Preserving grain with OA can reduce bacterial and fungal growth without affecting its chemical composition. Additionally, OA-preserving improves post-weaned piglet growth and gut health more effectively than direct OA supplementation18. Implementing post-harvest preservation techniques with OA to maintain grain quality may enhance piglet growth post-weaning through a linked mechanism: first, by reducing microbial contamination in the grain, thereby improving its quality and nutritional value; and second, by directly enhancing gastrointestinal function and health in piglets through lower levels of harmful pathogens and increased nutrient absorption. Previous research has reported that pigs offered OA-preserved grain exhibited increased growth, ileal and total tract nutrient digestibility, and beneficial gut microbial shifts at day 35 post-weaning compared to those fed dried grain19. Therefore, it is essential to compare the benefits and underlying mechanisms of OA-preserved grain with ZnO during the critical post-weaning period.
This study aimed to assess the intestinal health and growth of newly weaned piglets offered grain preserved with OA at harvest against those offered conventionally dried grain. Additionally, it sought to compare the mechanisms by which OA-preserved grain and ZnO improve piglet performance by assessing various intestinal health and growth parameters. The hypotheses were, firstly, that OA-preserved grain would enhance piglet intestinal health and growth compared to conventionally dried grain, and secondly, OA-preserved grain would have similar effects on piglet intestinal health and growth post-weaning as ZnO supplementation, presenting a viable alternative to in-feed antimicrobials.
Results
Grain quality
Prior to preservation, the initial quality assessment of wheat showed a moisture content of 180 g/kg, a hectolitre weight of 73 kg/hL, and a thousand grain weight (TGW) of 49.8 g. For barley, the corresponding values were 183 g/kg for moisture, 61 kg/hL for hectolitre weight, and a TGW of 48.2 g. Comprehensive chemical and microbial profiles of both grains at the point of diet formulation are detailed in Table 1. Grains preserved with OA exhibited lower DM content compared to those that were dried. Dried wheat and barley also had higher concentrations of deoxynivalenol (DON) than their OA-preserved counterparts. In barley, higher levels of HT-2 toxin and ochratoxin A (OTA) were observed in the dried samples. Across both preservation methods and grain types, levels of aflatoxins B1, B2, G1, and G2 (≤ 10 μg/kg), fumonisins B1 and B2 (< 1000 μg/kg), T-2 and HT-2 toxins (< 50 μg/kg), and zearalenone (ZEA; < 250 μg/kg) remained below the detectable threshold (Tables 2, 3 and 4).
Growth performance and faecal scores
Table 5 summarises the effects of the dietary treatments on growth metrics, body weight (BW), average daily gain (ADG), average daily feed intake (ADFI), gain-to-feed ratio (G:F), and faecal consistency scores (FS) over the post-weaning (PW) periods: days 0–21, 21–35, and the overall 0–35 days. One piglet per treatment group died during the first week PW due to causes unrelated to dietary intake.
During days 0–21, significant interactions between the grain preservation method and ZnO inclusion were observed for ADG, G:F, and BW (P < 0.05). Piglets fed OA-preserved grains had improved ADG and G:F compared to those receiving dried grains; however, these differences were not present when ZnO was added. In contrast, ZnO supplementation enhanced BW in piglets on dried grain diets but had no measurable effect in the OA-preserved grain group. ADFI was higher in ZnO-supplemented diets relative to those without ZnO (386 vs. 337 g/day, SEM 0.007; P < 0.001).During days 21–35, significant grain preservation × ZnO interactions were also found for ADFI, ADG, and BW (P < 0.05). ZnO inclusion increased ADFI in pigs fed dried grain diets, but not in those fed OA-preserved grains. ZnO reduced ADG in the OA-preserved grain group but showed no effect in the dried grain group. BW was increased by ZnO in the dried grain group only. Pigs fed OA-preserved grains had significantly higher G:F compared to those receiving dried grains (747 vs. 680, SEM 0.026; P < 0.05).
During the overall period (day 0–35), an interaction between grain preservation and ZnO supplementation influenced both ADFI and ADG (P < 0.05). ZnO enhanced ADFI and ADG in dried grain diets but had no such effect in OA-preserved diets. Overall, pigs fed OA-preserved grains achieved superior G:F ratios compared to dried grain-fed pigs (775 vs. 709, SEM 0.024; P < 0.05).
Small intestinal morphology
The effects of dietary treatments on small intestinal morphology are provided in Table 6. Neither grain preservation method, ZnO inclusion, nor their interaction significantly influenced villus height (VH), crypt depth (CD), or VH:CD ratio in the duodenum (P > 0.05).
In the jejunum, pigs receiving OA-preserved grain had shorter villi compared to those receiving dried grain (217.9 μm vs. 281.5 μm, SEM 15.03; P < 0.05). ZnO inclusion did not impact jejunal CD or VH:CD ratio (P > 0.05). In the ileum, a grain × ZnO interaction was evident for VH (P < 0.05); ZnO supplementation increased VH in the OA-preserved grain diet but not in the dried grain diet. ZnO also reduced CD (92.7 μm vs. 107.7 μm, SEM 4.71; P < 0.05) and improved VH:CD ratio (2.8 vs. 2.1, SEM 0.21; P < 0.05).
Gene expression in the gastrointestinal tract
Differential gene expression related to nutrient absorption, mucosal immunity, and barrier function is shown in Table 7.
In the duodenum, there was a significant interaction between grain preservation and ZnO on MUC2 expression (P < 0.05); ZnO reduced MUC2 expression in the dried grain group but had no effect in the OA-preserved group. IL17 expression was reduced in pigs fed OA-preserved grains (1.03 vs. 1.74, SEM 0.213; P < 0.05). ZnO supplementation significantly decreased expression of FABP2, IL6, and IL17 compared to non-supplemented diets (P < 0.05). In the jejunum, ZnO inclusion upregulated SLC15A1 (1.98 vs. 0.99, SEM 0.280) and OCCLN (1.55 vs. 0.95, SEM 0.207), while reducing IL1A, IL1B, CXCL8, IL17, IL22, and NOX1 (P < 0.05). Grain preservation had no effect (P > 0.05). In the ileum, a significant interaction was noted for TNF expression (P < 0.05); ZnO reduced TNF levels in OA-preserved diets only. The OA-preserved grain diets were associated with lower FABP2 and FOXP3 expression (P < 0.05). ZnO increased SLC15A1 expression and reduced pro-inflammatory markers (IL1A, IL1B, CXCL8, IL17, IL22, NOX1) (P < 0.05).
In the colon, ZnO × grain preservation interactions were observed for IL6 (P < 0.05); ZnO reduced IL6 in the dried grain group only. ZnO supplementation increased SLC16A1 and MUC2 expression while decreasing IL1A, IL1B, CXCL8, TLR4, IL17, and NOX1 (P < 0.05). Grain preservation had no significant effect on colonic gene expression.
Bacterial richness and diversity
Alpha diversity metrics are summarised in Table 8, with beta diversity visualised in Figs. 1 and 2. No significant effects of grain preservation, ZnO supplementation, or their interaction were observed on richness or diversity indices (Observed, Fisher, Shannon, Simpson) in the ileum (P > 0.05).
Bacterial beta diversity of ileal digesta based on Permanova analysis and through visualisation using the Bray Curtis distance matrix and multi-dimensional scaling.
Bacterial beta diversity of colonic digesta based on Permanova analysis and through visualisation using the Bray Curtis distance matrix and multi-dimensional scaling.
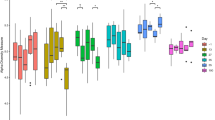
In the colon, OA-preserved grain diets led to a reduction in Shannon (3.31 vs. 3.55, SEM 0.060) and Simpson (0.94 vs. 0.96, SEM 0.004) diversity indices (P < 0.05). ZnO inclusion had no significant effect on colonic alpha diversity (P > 0.05). PERMANOVA analysis showed no significant differences in beta diversity for either intestinal region (P > 0.05).
Differential bacterial abundance
Phylum level
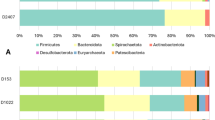
Table 9 presents the effects of grain preservation method and ZnO supplementation on the relative abundance of bacterial phyla. In the ileum, the predominant phyla were Firmicutes (~ 85.25%), Proteobacteria (~ 8.98%), Actinobacteria (~ 1.76%), and Bacteroidetes (~ 1.67%). Grain × ZnO interactions were detected for Proteobacteria (P < 0.05); OA-preserved grains reduced Proteobacteria abundance compared to dried grains, with no effect under ZnO supplementation. ZnO lowered Bacteroidetes abundance (1.03 vs. 2.24, SEM 0.421; P < 0.05). In the colon, Firmicutes (~ 91.60%) dominated, followed by Proteobacteria (~ 4.10%), and Bacteroidetes (~ 1.58%). Interactions were observed for Firmicutes and Proteobacteria (P < 0.05).
Family level
Table 10 presents the effects of grain preservation method and ZnO supplementation on the relative abundance of bacterial families. In the ileum, ZnO × preservation interactions affected Lactobacillaceae, Lachnospiraceae, and Enterobacteriaceae (P < 0.05). Ruminococcaceae were more abundant in OA-preserved diets, while ZnO increased Erysipelotrichaceae and decreased Oscillospiraceae (P < 0.05). In the colon, interactions were seen for Lachnospiraceae and Enterobacteriaceae. Lactobacillaceae abundance was reduced in OA-preserved grain diets but increased by ZnO supplementation (P < 0.01).
Genus level
Table 11 presents the effects of grain preservation method and ZnO supplementation on the relative abundance of bacterial genera. The OA-preserved grains increased Holdemanella and Faecalibacterium while reducing Escherichia in the ileum. ZnO also raised Faecalibacterium levels. In the colon, interactions affected Frisingicoccus, Gemmiger, and Faecalibacterium. Mediterraneibacter was higher and Lactobacillus lower in OA-preserved grain-fed pigs; ZnO reversed this trend.
Volatile fatty acids (VFAs)
Table 12 details the volatile fatty acid (VFA) profile. In the ileum, ZnO × grain preservation interactions influenced butyrate (P < 0.05); ZnO reduced butyrate in OA-preserved grain diets only. The OA-preserved grains raised acetate and lowered propionate proportions (P < 0.05). In the colon, OA-preserved diets increased propionate (0.267 vs. 0.237, SEM 0.0094; P < 0.05), while ZnO decreased branched-chain fatty acids (BCFA) (0.036 vs. 0.051, SEM 0.0046; P < 0.05). No significant interaction effects were found in the colon.
Discussion
The hypothesis of this study was that OA-preserved grain would improve piglet growth performance compared to conventionally dried grain by reducing the microorganism load in the grain, thereby increasing grain quality and directly influencing the gastrointestinal health and function of the pig. It was also hypothesised that OA-preserved grain would have similar effects on piglet growth and intestinal health compared to ZnO and could serve as an alternative to in-feed antimicrobials. The OA-preserved grain diet had the best BW, ADG, and G:F among all dietary treatments, while the beneficial effects of ZnO on growth performance were reduced after the initial 21 days PW. However, OA-preserved grain did not reduce FS to levels comparable with ZnO. These results indicate that OA-preserved grain could support piglet growth throughout the PW period. However, further dietary manipulation of OA-preserved grain diets is necessary to reduce FS to levels reported with ZnO.
In this study, OA-preserved grain had a lower DM and reduced levels of OTA and DON compared to dried grain, indicating improved grain quality. Post-weaned piglets are particularly vulnerable to mycotoxin contamination due to their physiological immaturity20. Mycotoxins impair immune function and increase pathogen susceptibility21, ultimately reducing voluntary feed intake and growth22. Specifically, OTA promotes oxidative stress and an inflammatory state23, while DON increases inflammation and digestive disorders24. Propionic acid and ammonium propionate are well-established grain preservatives26,27. Previous research suggests they provide greater benefits for gut health and growth when used as grain preservatives rather than as feed additives18. Therefore, improved grain quality could be partially responsible for the increased ADFI, growth performance, and improved gut microbiome observed in the OA-preserved grain diet. To confirm these protective effects, future studies could employ non-invasive mycotoxin biomarkers in piglet urine, faeces, or blood25.
Feed intake typically declines immediately PW28, leading to villous atrophy, crypt hyperplasia, elevated T-cell production29, and impaired intestinal development30. Reports on the effects of OA supplementation on piglet feed intake remain inconsistent, with studies showing no effect31,32,33,34, negative effects35, or positive effects36,37,38. Additionally, supplementation with glycerol polyethylene ricinoleate has been shown to increase ADG and improve feed efficiency in broiler chickens39‚40. In the present study, OA-preserved grain increased ADFI compared to dried grain, aligning with previous findings18,19. Therefore, the observed increase in ADFI may contribute to the gut microbial shifts and improved growth performance seen in piglets fed the OA-preserved grain diet.
Interestingly, ZnO supplementation improved BW and ADFI in dried grain but did not provide any additional benefit on BW and ADFI in OA-preserved grain, consistent with previous research19. Although research examining ZnO supplementation in OA-preserved grain is limited, ZnO supplementation in non-OA-preserved diets has consistently increased growth performance41,42,43. However, the beneficial effect of ZnO on growth performance in pigs fed dried grain was diminished during the second phase of the experiment (D21–35). This finding supports the notion that ZnO efficacy declines after the initial 21 days PW44. In contrast, OA-preserved grain maintained its positive effects on growth throughout the experimental period, likely due to its influence on feed intake and modulation of the gut microbiome. Although both OA-preserved grain and ZnO improved growth performance, their effects on FS differed. OA-preserved grain did not improve FS compared with dried grain, whereas ZnO supplementation led to higher FS values relative to non-ZnO diets, consistent with previous findings45,46. The growth and FS improvements associated with ZnO may stem from enhanced gut morphology, reduced immune activation, improved nutrient absorption, and alterations in intestinal microbial populations.
The intestinal microbiome is vital in maintaining the physiological, immunological, and nutritional state of the pig. Thus, manipulating the gut microbiota could reduce reliance on antimicrobials in pig production47. In this study, piglets offered OA-preserved grain had reduced colonic Shannon and Simpson diversity compared to those offered dried grain. Reduced microbial diversity has been associated with diarrhetic piglets PW48, however, other studies have reported no correlation between reduced diversity and piglet gut health49. Pigs offered the OA-preserved grain diet had the highest abundance of ileal and colonic Faecalibacterium among all treatments. Faecalibacterium, specifically Faecalibacterium prausnitzii are indicators of increased intestinal health due to their immunomodulatory50 and butyrate-producing abilities51. Supplementing Faecalibacterium prausnitzii to mice increased tight-junction protein expression, reduced pro-inflammatory cytokine expression, and decreased the abundance of faecal Staphylococcus and Klebsiella52. Additionally, the OA-preserved grain was associated with reduced abundance of ileal Escherichia compared to dried grain. Escherichia coli is one of the main causative agents of PWD53. Previous research has demonstrated OA supplementation can reduce E. coli54,55 and increase faecal Faecalibacterium in weaned piglets56. Despite the reduction in colonic bacterial diversity, the microbial shifts associated with OA-preserved grain could contribute to enhanced growth performance and intestinal health.
Supplementing ZnO can beneficially modulate the PW gut microbiota by promoting the growth of beneficial bacteria, enhancing short-chain fatty acid (SCFA) production, and inhibiting the proliferation of pathogens57, thereby supporting a more stable and mature gut ecosystem. ZnO supplementation has been reported to reduce the abundance of Enterobacteriaceae, Escherichia, and Lactobacillus spp. in the small intestine58. In contrast, the present study found that ZnO supplementation increased colonic Lactobacillus abundance compared with non-ZnO diets, differing from previous reports that observed no effect of ZnO on intestinal Lactobacillus populations4,45. Notably, Lactobacillus species have been associated with improved nutrient digestion and barrier integrity, reduced diarrhoea59, and decreased expression of pro-inflammatory cytokines60, which could partially explain the positive effects observed here. The beneficial influence of ZnO on the gut microbiota was further reflected in the VFA profile. ZnO supplementation reduced the molar proportions of colonic BCFAs compared with non-ZnO diets, indicating decreased protein fermentation61. Excessive protein fermentation generates harmful metabolites62, disrupts intestinal barrier function, promotes inflammation63, and facilitates the proliferation of pathogenic bacteria40. Together, the microbial and VFA shifts observed with ZnO supplementation may underlie the initial improvements in growth performance in pigs fed the dried grain diet with ZnO, as well as the reductions in FS and intestinal inflammatory markers.
Post-weaning inflammation reduces intestinal barrier function, alters the microbiome64 and affects intestinal morphology65. Supplementing OAs has been reported to reduce the expression of pro-inflammatory cytokines66 and increase the expression of genes related to intestinal barrier function67. In the current experiment, OA-preserved grain reduced the relative expression of duodenal IL17 and ileal FOXP3 compared to dried grain. The supplementation of ZnO decreased the relative expression of multiple pro-inflammatory cytokines, including IL6 and IL17 in the duodenum, IL1A, IL1B, CXCL8, IL17, and IL22 in the jejunum and ileum, and IL1A, IL1B, IL6, CXCL8, and IL17 in the colon on day 10 PW. Additionally, NOX1, an indicator of oxidative stress, was reduced in the jejunum, ileum, and colon of ZnO-supplemented pigs. The expression of genes involved in barrier integrity, such as OCCLN in the jejunum and MUC2 in the colon, were increased in ZnO-supplemented pigs. These results suggest that the reduction in pro-inflammatory cytokine expression and increased expression of genes associated with the integrity of the intestine in ZnO-supplemented diets could contribute to reduced FS. Moreover, physiological changes during an immune response create a homeorhetic state, redirecting nutrients from growth to the immune system68. Thus, the reduced expression of pro-inflammatory cytokines may increase the energy available for growth, partially explaining the improved growth performance of the dried grain diet with ZnO in the initial 21 days.
The relationship between intestinal morphology and growth performance is well established, with morphological disruptions associated with reduced growth performance69. Interestingly, although there was no negative effect on growth, OA-preserved grain reduced jejunal VH compared to dried grain. Furthermore, OA-preserved grain reduced the relative expression of ileal FABP2 compared to dried grain. The ability of OA-preserved grain to sustain superior growth performance, despite evidence of reduced absorptive capacity, may be attributed to its previously reported enhancement of nutrient digestibility in weaned pigs19,70, possibly mediated by increased digestive enzyme activity. Conversely, ZnO supplementation improved intestinal morphology by reducing ileal CD and increasing the ileal VH:CD ratio compared to non-ZnO supplemented diets. Supplementing ZnO also increased the relative expression of jejunal and ileal SLC15A1 and colonic SLC16A1 compared to non-ZnO supplemented diets, indicating increased protein and SCFA absorption. The increased digestive and absorptive capacity of ZnO-supplemented pigs may be linked to reduced FS and improved growth performance in the dried grain diet in the initial 21 days PW.
The ability of OA-preserved grain to enhance piglet growth performance throughout the PW period highlights its potential as an alternative to ZnO following the EU’s restrictions on in-feed antimicrobials. However, the findings of this study may not be fully generalisable. The experiment used specific wheat (JB Diego) and barley (SY Errigal) varieties from Ireland, each with unique nutritional compositions that could influence how OA preservation affects grain quality, piglet growth, and intestinal health. Grains differing in variety, origin, or initial moisture content, as well as other cereals such as maize, oats, rye, triticale, or sorghum, may not respond identically to OA preservation. Variations in starch characteristics and fiber content can alter fermentation patterns and nutrient digestibility. Despite these potential differences, the core benefits of OA preservation, reducing microbial spoilage, stabilising nutrients, and supporting gut health, are likely to be broadly applicable across cereal types. Nonetheless, the magnitude and consistency of these effects are expected to depend on the specific properties of each grain and should be validated experimentally under different conditions. Finally, ZnO’s strong capacity to reduce FS and suppress pro-inflammatory cytokine expression suggests that future research should aim to further optimise OA-preserved grain diets to achieve comparable improvements in intestinal health. Approaches such as lowering dietary crude protein or incorporating complementary feed additives may help reduce FS and enhance gut integrity to levels similar to those observed with ZnO supplementation.
Material and methods
All of the experimental procedures detailed in this study were approved by the University College Dublin Animal Research Ethics Committee (AREC-20-21-ODoherty) and performed in accordance with Irish legislation (SI no.543/2012) and the EU directive 2010/63/EU for animal experimentation and in compliance with the ARRIVE guidelines. The authors confirm that the ethical policies of the journal, as noted on the journal’s author guidelines page, have been adhered to and the appropriate ethical review committee approval has been received. The authors confirm that they have followed the EU standards for the protection of animals used for scientific purposes and the ARRIVE guidelines.
Grain processing and quality evaluation
Winter wheat (cv. JB Diego) and spring barley (cv. SY Errigal) grains sourced from McAuley Feeds (Burtonstown, Co. Meath, Ireland) were utilised in this study, following appropriate management and preservation practises19. The wheat was sown in October 2019, following standard agronomic practices [3-spray fungicide program and a 3-split nitrogen (N) application rate of 180 kg N/ha] and harvested in August 2020 at 180.0 g/kg moisture content. The barley was sown in March 2020, and received a 2-spray fungicide program and a 2-split N application rate of 140 kg/ha, before being harvested in August 2020, at 182.0 g/kg moisture content.
Prior to storage, both the wheat and barley grains were separated into two groups and preserved19. Briefly, one group was dried using a continuous flow dryer (Cimbria, Thisted, Denmark) at 65 °C for 3 h before a 2 h cooling period, reducing the moisture content of the wheat and barley to 140 g/kg and 140.7 g/kg, respectively. The second group was preserved with an organic acid liquid surfactant mould inhibitor [MycoCURB ES Liquid; containing propionic acid (650 g/kg), ammonium propionate (70 g/kg), glycerol polyethyleneglycol ricinoleate (17.5 g/kg) and water]. It was applied to the grain by spray action at the inclusion rate of 4 g/kg. Even distribution of the acid throughout the grain was ensured by using a mixing auger. This product was manufactured by Kemin Industries (Des Moines, IA) and sourced from Adesco Nutricines, (Dungarvan, Co. Waterford, Ireland).
At the time of harvest, various grain quality parameters was evaluated including moisture content, density (hectolitre weight), and thousand-grain weight (TGW). Moisture content was measured with a DICKEY-john GAC 2500-UGMA electronic moisture meter (Illinois, USA), while density was assessed using a Pfeuffer Chondrometer along with a bulk density calibration chart. The TGW was determined by weighing 1000 grains and recording their weights using a Pfeuffer Contador seed counter (Kitzingen, Germany). Prior to diet manufacture, samples were collected from both the wheat and the barley using the grab sample technique. These samples were analysed for dry matter (DM), ash, gross energy (GE) crude protein, crude fibre, starch, fat and mycotoxin levels. Mycotoxins including Aflatoxin B1, B2, G1 and G2, and trichothecenes [T-2 toxin, HT-2 toxin and DON], Fumonisin B1 and B2, OTA and ZEA was detected using liquid chromatography-mass spectrometry71.
Experimental design and diet
Ninety-six newly piglets, which were weaned at 28 days (progeny of Meatline Hermitage boar (Sion Road, Kilkenny, Ireland) × (Large White × Landrace sow)) were selected from a commercial swine unit and assigned to 1 of 4 dietary treatments in a 2 × 2 factorial design across a 35-day experimental period (n = 8). The average weaning weight of the piglets was 7.00 ± 1.2 kg (SD), and they were blocked by litter of origin, sex and body weight. The dietary treatments were as follows: (T1) dried grain diet; (T2) OA-preserved grain diet; 3 dried grain diet with zinc oxide (ZnO) and4 OA-preserved grain diet with ZnO. The diets consisted of 600 g/kg of grain, with 450 g/kg being either dried or OA-preserved wheat and 150 g/kg being dried or OA-preserved barley. The remaining composition (400 g/kg) consisted of a concentrate obtained from Cargill (Naas, Co. Kildare, Ireland), as outlined in Table 2. The ZnO used in this study was obtained from Cargill (Naas, Co Kildare, Ireland) and contained 80% zinc. The ZnO was included at a concentration of 3.3 g ZnO/kg of feed, resulting in an inclusion level of 2.65 g Zn/kg of feed. After 15 days, the ZnO inclusion level was lowered to 1.5 g/ZnO/kg feed. The diets were formulated to have comparable levels of digestible energy (14.95 MJ/kg), net energy (10.95 MJ/kg) and standardised ileal digestible lysine (12.0 g/kg)72. The piglets’ amino acid requirements were met relative to lysine73. All diets were milled on the research facility. The ingredient composition of the dietary treatments is presented in Table 2, while the chemical and microbial composition of the dietary treatments are presented in Table 3.
Animal housing and management
Piglets were housed in fully slatted pens (1.7 × 1.2 m) with three animals per pen. Environmental conditions were controlled, with the room temperature set at 30 °C for the first week and reduced by 2 °C each subsequent week. Humidity was maintained at 65%. Feed in the form of mash and water were provided ad libitum through two-space feeders and nipple drinkers.
Body weight was recorded at baseline (day 0) and subsequently at 7-day intervals. These data were used to calculate ADG. Feed intake was recorded weekly to calculate ADFI and G:F. Faecal consistency was scored twice daily using a 1–5 scale: 1 = firm; 2 = slightly soft; 3 = soft, partially formed; 4 = loose, semi-liquid; 5 = watery, mucous-like46.
Sample collection
On day 10 PW, one piglet per pen (7 per treatment group) was selected for the collection of intestinal tissue and digesta samples, based on average body weight, as gut disturbances are greatest during the first 14 days PW69,74,75. Selected piglets were humanely euthanised. Euthanasia was performed by a trained technician in a separate area, using a lethal injection of pentobarbitone sodium (Euthanal, 200 mg/ml; Chanelle Pharma, Galway, Ireland) administered into the cranial vena cava at a dosage of 0.71 ml/kg body weight18.
Immediately following euthanasia, the gastrointestinal tract was excised. Tissue samples were taken from the duodenum (10 cm distal to the stomach), jejunum (60 cm distal), and ileum (15 cm proximal to the cecum), and fixed in 10% neutral buffered formalin. These samples were analysed via quantitative PCR (QPCR) to assess expression of nutrient transporters, mucins, tight junction proteins, and cytokines. Additionally, colonic tissue was harvested for analysis of SCFA transporters, mucin, and cytokine expression via QPCR. Digesta from the ileum and colon was collected into sterile containers (Sarstedt, Wexford, Ireland) and immediately frozen at − 20 °C for subsequent VFA profiling and 16S rRNA gene sequencing.
Feed analysis
During diet preparation, representative feed samples were collected and ground using a 1 mm screen (Christy and Norris Hammer Mill, Chelmsford, UK). GE was determined via an adiabatic bomb calorimeter (Parr Instruments, St. Moline, IL, USA). DM content was measured by drying at 55 °C for 72 h. Crude ash was determined by combusting the samples in a muffle furnace (Nabertherm) at 550 °C for 6 h.
Nitrogen concentration was analysed using a LECO FP 528 (Leco Instruments, Stockport, UK Ltd.). Amino acid profiles were determined using high-performance liquid chromatography (HPLC)76. Crude fibre was assessed following AOAC (1990; method 978.10), and neutral detergent fibre (NDF) was analysed using an Ankom 220 Fibre Analyzer (Ankom Technology, USA)77. Starch content was measured with the Megazyme total starch assay (Megazyme, Bray, Co. Wicklow, Ireland). Crude fat was quantified using Soxtec extraction with light petroleum ether (Tecator, Hillerod, Sweden). Table 3 summarises the complete chemical composition of the dietary treatments.
Gut morphological analysis
Histological processing of intestinal tissues (duodenum, jejunum, ileum) was performed following the paraffin-embedding method46. Tissue sections were cut to 5 μm long and stained with haematoxylin and eosin. Morphometric measurements were conducted using a light microscope fitted with an image analysis system (Image-Pro Plus; Media Cybernetics, Oxon, UK). For each tissue per animal, a minimum of 15 measurements of VH and CD were recorded. VH was measured from the villus tip to the crypt-villus junction, while CD was measured from the base of the crypt to the same junction.
Gene expression in the gastrointestinal tract
Tissue sample preparation and RNA Analysis
Tissue samples were collected from the mesenteric aspect of the duodenum, jejunum, ileum, and colon. Following collection, samples were rinsed with sterile phosphate-buffered saline (PBS; Oxoid), and the external smooth muscle layers were carefully removed. The mucosal tissues were then sectioned into smaller fragments using a sterile scalpel and placed in 15 mL of RNAlater™ (Applied Biosystems) for overnight preservation. Subsequently, the samples were stored at − 20 °C until RNA extraction.
Total RNA was isolated from the collected tissues using TRI Reagent (Sigma-Aldrich, St. Louis, MO, USA), adhering to the manufacturer’s instructions78. For reverse transcription, 2 µg of total RNA was converted to complementary DNA (cDNA) using the High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) with oligo (dT) primers, achieving a final reaction volume of 40 µL. This cDNA was then diluted with nuclease-free water to a final volume of 360 µL for downstream applications.
QPCR was carried out in 20 µL reaction volumes, comprising 10 µL GoTaq Master Mix (Promega, Madison, WI, USA), 1.2 µL of 5 µM forward and reverse primers, 3.8 µL nuclease-free water, and 5 µL of cDNA template. Each sample was analysed in duplicate using the 7500 ABI Prism Sequence Detection System (Applied Biosystems, Foster City, CA, USA). The thermocycling program included an initial denaturation step at 95 °C for 10 min, followed by 40 amplification cycles of 95 °C for 15 s and 60 °C for 1 min.
Primers were designed using Primer Express Software (Applied Biosystems) and synthesised by MWG Biotech UK Ltd. (Milton Keynes, UK). Melting curve analysis was performed to confirm amplicon specificity. PCR efficiency was assessed via standard curves generated from four-fold serial dilutions of cDNA, and only assays with efficiencies between 90–100% and single specific products were considered.
Normalised relative expression levels were calculated using qbase PLUS software (Biogazelle, Ghent, Belgium), based on stable reference genes. For duodenum, jejunum, and ileum tissues, HMBS, H3F3A, and YWHAZ were used, while ACTB and B2M were chosen for colonic tissues. These genes were selected based on GeNorm-calculated M values (< 1.5). Genes assessed included SLC15A1, FABP2, SLC2A1, SLC2A5, SLC2A7, SLC5A1, SLC16A1, CLDN1, TJP, OCCLN, IL1A, IL1B, IL10, IL6, IL17, IL22, INFG, TNF, CXCL8, MUC2, TLR2, TLR4, NOX1, GIP, SCT, HRH2, FOXP3, MMP1, and AQP10. Differentially expressed genes are presented in Table 4.
Volatile fatty acid (VFA) quantification
VFA levels in ileal and colonic digesta were determined using gas–liquid chromatography79. For each sample, 1 g of digesta was homogenised with distilled water (2.5 × the sample weight) and centrifuged at 1400 × g for 10 min using a Sorvall GLC-2 B centrifuge (DuPont). A 1 mL aliquot of the supernatant was mixed with 1 mL of an internal standard (0.05% 3-methyl-n-valeric acid in 0.15 M oxalic acid dihydrate) and 3 mL of distilled water. This mixture was then centrifuged at 500 × g for 10 min.
The resulting supernatant was filtered through a 0.45 µm polytetrafluoroethylene (PTFE) syringe filter and transferred to chromatographic vials. One microlitre of each sample was analysed using a Varian 3800 GC fitted with an EC™ 1000 Grace column (15 m × 0.53 mm I.D., 1.20 µm film thickness). The temperature program ramped from 75 to 90 °C at 3 °C/min, then from 95 to 200 °C at 20 °C/min, with a final hold of 0.5 min. Injector and detector temperatures were set at 240 °C and 280 °C, respectively. Each run lasted 12.42 min.
Microbial DNA extraction and illumina sequencing
Genomic DNA was extracted from the ileal and colonic digesta using the QIAamp PowerFecal Pro DNA Kit (Qiagen, West Sussex, UK), following the supplier’s protocol. DNA yield and purity were assessed using a Nanodrop ND-1000 spectrophotometer (Thermo Scientific, Wilmington, DE, USA).
Sequencing of the V3–V5 hypervariable regions of the 16S rRNA gene was performed on the Illumina MiSeq platform at Eurofins Genomics (Ebersberg, Germany). Amplification used universal primers with adapter overhangs compatible with Nextera XT indexing. Amplicons were purified using AMPure XP magnetic beads (Beckman Coulter, Indianapolis, IN), followed by indexing PCR. Indexed products were again purified and quantified using a fragment analyser (Agilent, Santa Clara, CA), pooled in equimolar ratios, and quantified using the Bioanalyser 7500 DNA kit (Agilent). Sequencing employed v3 chemistry for 2 × 300 bp paired-end reads.
Bioinformatic analysis
Sequence processing and analysis were carried out by Eurofins Genomics using QIIME (v1.9.1)80. Reads passing Illumina’s chastity filter and with Phred scores > 30 were demultiplexed. Primer sequences were trimmed, and reads with mismatches were excluded. Paired-end reads were merged using FLASH v2.2.0081, requiring a minimum 10 bp overlap. When merging was not possible, only forward reads were retained.
Reads were filtered to match the expected V3–V5 length and truncated to 300 bp to improve quality. Chimeric reads were identified using UCHIME in the VSEARCH tool82 and removed. Operational taxonomic units (OTU) were formed using Minimum Entropy Decomposition (MED)83. Representative OTU were classified taxonomically via DC-MEGABLAST against the NCBO nucleotide database, requiring ≥ 70% similarity over ≥ 80% of the sequence.
Copy number correction was applied using gene-specific linear copy numbers84. The resulting dataset, comprising the normalised OTU table, metadata, and phylogenetic tree, was imported into the R package phyloseq (v3.5.0) for differential abundance analysis at phylum, family, and genus levels.
Statistical analysis
All statistical analyses were conducted using SAS® software (v9.4, SAS Institute Inc.). The Shapiro–Wilk test (PROC UNIVARIATE) assessed data normality for growth performance, FS, gene expression, intestinal morphology, and VFA profiles. Where needed, data were transformed to meet normality assumptions. Growth metrics (ADFI, ADG, G:F, BW) were analysed via PROC GLM for days 0–21, days 22–35 and days 0–35. The model included fixed effects of grain preservation, ZnO supplementation, and their interactions. The pen was used as the experimental unit. Initial body weight was used as a covariate. The FS were averaged every three days and analysed via PROC MIX, using repeated measures. The model included fixed effects of grain preservation, ZnO supplementation, time, and their associated interactions. The pen was used as the experimental unit. PROC GLM was used to analyse gut morphology, gene expression, VFA concentrations, and alpha diversity. Bonferroni’s correction (P < 0.05) adjusted for multiple comparisons in gene expression. The Non-parametric microbial data were evaluated using PROC GLIMMIX, with P values corrected via the Benjamini–Hochberg method. Results are reported as least-square means ± standard error of the mean (SEM). Significance was set at P < 0.05, with 0.05 < P ≤ 0.10 considered a trend.
Alpha diversity (richness and evenness) was assessed using observed richness, Fisher, Shannon, and Simpson indices85,86. Beta diversity was calculated using the Bray–Curtis dissimilarity metric in phyloseq84,87, with data normalised prior to comparison.
Conclusion
In summary, OA-preserved grain improved piglet growth performance, feed intake, and beneficially altered the intestinal microbiota, offering a promising alternative to ZnO following regulatory restrictions. However, it did not match ZnO’s effectiveness in reducing FS or inflammatory responses. The growth-enhancing effects of OA-preserved grain may be linked to improved grain hygiene, increased feed intake, and favourable microbial profiles. Zinc oxide’s superior impact on FS and gut health markers highlights the need for future strategies, such as adjusting dietary protein or incorporating complementary additives, to optimise OA-preserved grain diets. These approaches could provide a sustainable alternative to in-feed antimicrobials, aligning with animal welfare and regulatory goals.
Data availability
All data generated and/or analysed during this study are available upon reasonable request from the corresponding author.
References
Eriksen, E. Ø. et al. Post-weaning diarrhea in pigs weaned without medicinal zinc: Risk factors, pathogen dynamics, and association to growth rate. Porc. Health Manag. 7(1), 54 (2021).
O’Doherty, J., Dowley, A., Conway, E. & Sweeney, T. Nutritional strategies to mitigate post-weaning challenges in pigs: A focus on glucans, vitamin D, and selenium. Animals 14(1), 13 (2023).
Rhouma, M., Fairbrother, J. M., Beaudry, F. & Letellier, A. Post weaning diarrhea in pigs: Risk factors and non-colistin-based control strategies. Acta Vet. Scand. 59(1), 31 (2017).
Heim, G. et al. Effect of seaweed-derived laminarin and fucoidan and zinc oxide on gut morphology, nutrient transporters, nutrient digestibility, growth performance and selected microbial populations in weaned pigs. Br. J. Nutr. 111(9), 1577–1585 (2014).
Bonetti, A., Tugnoli, B., Piva, A. & Grilli, E. Towards zero zinc oxide: Feeding strategies to manage post-weaning Diarrhea in piglets. Animals 11(3), 642 (2021).
Ravindran, V. & Kornegay, E. T. Acidification of weaner pig diets: A review. J. Sci. Food Agric. 62(4), 313–322 (1993).
Li, Z., Liu, S., Zhao, Y., Wang, J. & Ma, X. Compound organic acid could improve the growth performance, immunity and antioxidant properties, and intestinal health by altering the microbiota profile of weaned piglets. J. Anim. Sci. 101, skad196 (2023).
Connolly, K. R., Sweeney, T. & O’Doherty, J. V. Sustainable nutritional strategies for gut health in weaned pigs: The role of reduced dietary crude protein, organic acids and butyrate production. Animals 15(1), 66 (2024).
Khan, S. H. & Iqbal, J. Recent advances in the role of organic acids in poultry nutrition. J. Appl. Anim. Res. 44(1), 359–369 (2016).
Stein, H. H., Lagos, L. V. & Casas, G. A. Nutritional value of feed ingredients of plant origin fed to pigs. Anim. Feed Sci. Technol. 218, 33–69 (2016).
Burke, J., Spink, J., Hackett, R. Wheat in the Republic of Ireland. In: The World Wheat Book: A History of Wheat Breeding 107–18 (Lavoisier Publishing, 2011).
Hoffmans, Y., Schaarschmidt, S., Fauhl-Hassek, C. & Van Der Fels-Klerx, H. J. Factors during production of cereal-derived feed that influence mycotoxin contents. Toxins 14(5), 301 (2022).
Coradi, P. C., Fernandes, C. H. P., Helmich, J. C. & Goneli, A. L. D. Effects of drying air temperature and grain initial moisture content on soybean quality (Glycine Max (L.) Merrill). Eng. Agríc. 36(5), 866–876 (2016).
Alam, M. D. S., Sarker, M. D. S. H., Hasan, S. M. K., Ahmed, M. & Wazed, M. D. A. Comparison on drying characteristic, efficiency, unit drying cost and quality of maize dried by a novel multi-crop mobile dryer, existing industrial dryer and sun drying method. J. Agric. Food Res. 14, 100804 (2023).
Magan, N. & Aldred, D. Post-harvest control strategies: Minimizing mycotoxins in the food chain. Int. J. Food Microbiol. 119(1–2), 131–139 (2007).
Jimoh, K. A. et al. Recent advances in the drying process of grains. Food Eng. Rev. 15(3), 548–576 (2023).
Menon, A., Stojceska, V. & Tassou, S. A. A systematic review on the recent advances of the energy efficiency improvements in non-conventional food drying technologies. Trends Food Sci. Technol. 100, 67–76 (2020).
Connolly, K. R. et al. The role of propionic acid as a feed additive and grain preservative on weanling pig performance and digestive health. Anim. Feed Sci. Technol. 321, 116237 (2025).
Maher, S. et al. Organic acid preservation of cereal grains improves grain quality, growth performance, and intestinal health of post-weaned pigs. Anim. Feed Sci. Technol. 316, 116078 (2024).
Müller, L. K. F. et al. Post-weaning piglets fed with different levels of fungal mycotoxins and spray-dried porcine plasma have improved weight gain, feed intake and reduced diarrhea incidence. Microb. Pathog. 117, 259–264 (2018).
Kim, S. W., Holanda, D. M., Gao, X., Park, I. & Yiannikouris, A. Efficacy of a yeast cell wall extract to mitigate the effect of naturally co-occurring mycotoxins contaminating feed ingredients fed to young pigs: Impact on gut health, microbiome, and growth. Toxins 11(11), 633 (2019).
Popescu, R. G. et al. Natural antioxidant by-product mixture counteracts the effects of aflatoxin B1 and ochratoxin A exposure of piglets after weaning: A proteomic survey on liver mcrosomal fraction. Toxins 15(4), 299 (2023).
Marin, D. E., Pistol, G. C., Gras, M. A., Palade, M. L. & Taranu, I. Comparative effect of ochratoxin A on inflammation and oxidative stress parameters in gut and kidney of piglets. Regul. Toxicol. Pharmacol. 89, 224–231 (2017).
Van Le, T. B., Lessard, M., Chorfi, Y. & Guay, F. The efficacy of anti-mycotoxin feed additives in preventing the adverse effects of wheat naturally contaminated with Fusarium mycotoxins on performance, intestinal barrier function and nutrient digestibility and retention in weanling pigs. Can. J. Anim. Sci. 95(2), 197–209 (2015).
Tkaczyk, A. & Jedziniak, P. Mycotoxin biomarkers in pigs—Current state of knowledge and analytics. Toxins 13(8), 586 (2021).
Dijksterhuis, J., Kleinhoven, P., van Kuijk, S., Wolters, A. H. & Bruinenberg, P. G. Synergistic antifungal effects of the preservative ammonium propionate and medium chain fatty acids against dormant and germinating conidia, germ tubes and hyphae of Aspergillus chevalieri, a feed spoilage fungus. Int. J. Food Microbiol. 422, 110802 (2024).
Rutenberg, R., Bernstein, S., Fallik, E., Paster, N. & Poverenov, E. The improvement of propionic acid safety and use during the preservation of stored grains. Crop Prot. 110, 191–197 (2018).
Dong, G. Z. & Pluske, J. R. The low feed intake in newly-weaned pigs: Problems and possible solutions. Asian Aust. J. Anim. Sci. 20(3), 440–452 (2007).
McCracken, B. A., Spurlock, M. E., Roos, M. A., Zuckermann, F. A. & Gaskins, H. R. Weaning anorexia may contribute to local inflammation in the piglet small intestine. J. Nutr. 129(3), 613–619 (1999).
Lallès, J. P., Bosi, P., Smidt, H. & Stokes, C. R. Nutritional management of gut health in pigs around weaning. Proc. Nutr. Soc. 66(2), 260–268 (2007).
Lee, D. N. et al. Effects of diets supplemented with organic acids and nucleotides on growth, immune responses and digestive tract development in weaned pigs. Anim. Physiol. Nut. 91(11–12), 508–518 (2007).
Long, S. F. et al. Mixed organic acids as antibiotic substitutes improve performance, serum immunity, intestinal morphology and microbiota for weaned piglets. Anim. Feed Sci. Technol. 235, 23–32 (2018).
Namkung, H. et al. Impact of feeding blends of organic acids and herbal extracts on growth performance, gut microbiota and digestive function in newly weaned pigs. Can. J. Anim. Sci. 84(4), 697–704 (2004).
Wei, X. et al. Dietary organic acids modulate gut microbiota and improve growth performance of nursery pigs. Microorganisms 9(1), 110 (2021).
Wang, H. et al. Dietary acidifiers as an alternative to antibiotics for promoting pig growth performance: A systematic review and meta-analysis. Anim. Feed Sci. Technol. 289, 115320 (2022).
Kuang, Y. et al. Effects of dietary combinations of organic acids and medium chain fatty acids as a replacement of zinc oxide on growth, digestibility and immunity of weaned pigs. Anim. Feed Sci. Technol. 208, 145–157 (2015).
Tsiloyiannis, V. K., Kyriakis, S. C., Vlemmas, J. & Sarris, K. The effect of organic acids on the control of porcine post-weaning diarrhoea. Res. Vet. Sci. 70(3), 287–293 (2001).
Nowak, P., Zaworska-Zakrzewska, A., Frankiewicz, A. & Kasprowicz-Potocka, M. The effects and mechanisms of acids on the health of piglets and weaners—A review. Ann. Ani. Sci. 21(2), 433–455 (2021).
Yin, J., Yun, H. M. & Kim, I. H. Effects of supplemental glycerol polyethylene glycol ricinoleate in different energy density diets on the growth performance, blood profiles, nutrient utilization, and excreta gas emission of broilers: Focus on dietary glycerol polyethylene glycol ricinoleate in broilers. Korean J. Agric. Sci. 45(2), 219–228 (2018).
Rist, V. T. S., Weiss, E., Sauer, N., Mosenthin, R. & Eklund, M. Effect of dietary protein supply originating from soybean meal or casein on the intestinal microbiota of piglets. Anaerobe 25, 72–79 (2014).
Molist, F. et al. Effect and interaction between wheat bran and zinc oxide on productive performance and intestinal health in post-weaning piglets. Br. J. Nutr. 105(11), 1592–1600 (2011).
Sales, J. Effects of pharmacological concentrations of dietary zinc oxide on growth of post-weaning pigs: A meta-analysis. Biol. Trace Elem. Res. 152(3), 343–349 (2013).
Wang, Q. et al. Effects of dietary supplementation of humic acid sodium and zinc oxide on growth performance, immune status and antioxidant capacity of weaned piglets. Animals 10(11), 2104 (2020).
Conway, E. et al. Maternal and/or direct supplementation with a combination of a casein hydrolysate and yeast β-glucan on post-weaning performance and intestinal health in the pig. PLoS ONE 17(7), e0265051 (2022).
O’Shea, C. J., McAlpine, P., Sweeney, T., Varley, P. F. & O’Doherty, J. V. Effect of the interaction of seaweed extracts containing laminarin and fucoidan with zinc oxide on the growth performance, digestibility and faecal characteristics of growing piglets. Br. J. Nutr. 111(5), 798–807 (2014).
Rattigan, R., Sweeney, T., Vigors, S., Rajauria, G. & O’Doherty, J. V. Effects of reducing dietary crude protein concentration and supplementation with laminarin or zinc oxide on the faecal scores and colonic microbiota in newly weaned pigs. J. Anim. Physiol. Anim. Nutr. 104(5), 1471–1483 (2020).
Fouhse, J. M., Zijlstra, R. T. & Willing, B. P. The role of gut microbiota in the health and disease of pigs. Anim. Front. 6(3), 30–36 (2016).
Dou, S. et al. Characterisation of early-life fecal microbiota in susceptible and healthy pigs to post-weaning diarrhoea. PLoS ONE 12(1), e0169851 (2017).
Vigors, S., O’Doherty, J., Rattigan, R. & Sweeney, T. Effect of supplementing seaweed extracts to pigs until d35 post-weaning on performance and aspects of intestinal health. Mar. Drugs 19(4), 183 (2021).
Benevides, L. et al. New insights into the diversity of the genus Faecalibacterium. Front. Microbiol. 22(8), 1790 (2017).
Ferreira-Halder, C. V., de Sousa Faria, A. V. & Andrade, S. S. Action and function of Faecalibacterium prausnitzii in health and disease. Best Pract. Res. Clin. Gastroenterol. 31(6), 643–648 (2017).
Wang, X. et al. Faecalibacterium prausnitzii supplementation prevents intestinal barrier injury and gut microflora dysbiosis induced by sleep deprivation. Nutrients 16(8), 1100 (2024).
Renzhammer, R. et al. Prevalence of virulence genes and antimicrobial resistances in E. coli associated with neonatal diarrhea, postweaning diarrhea, and edema disease in pigs from Austria. Antibiotics 9(4), 208 (2020).
Gómez-García, M. et al. Antimicrobial activity of a selection of organic acids, their salts and essential oils against swine enteropathogenic bacteria. Porc. Health Manag. 5(1), 32 (2019).
Yang, Y., Lee, K. Y. & Kim, I. H. Effects of dietary protected organic acids on growth performance, nutrient digestibility, fecal microflora, diarrhea score, and fecal gas emission in weanling pigs. Can. J. Anim. Sci. 99(3), 514–520 (2019).
Han, Y. S. et al. Effects of dietary supplementation with combinations of organic and medium chain fatty acids as replacements for chlortetracycline on growth performance, serum immunity, and fecal microbiota of weaned piglets. Livest. Sci. 216, 210–218 (2018).
Tang, X., Xiong, K., Zeng, Y. & Fang, R. The mechanism of zinc oxide in alleviating diarrhea in piglets after weaning: A review from the perspective of intestinal barrier function. IJMS 25(18), 10040 (2024).
Starke, I. C., Pieper, R., Neumann, K., Zentek, J. & Vahjen, W. The impact of high dietary zinc oxide on the development of the intestinal microbiota in weaned piglets. FEMS Microbiol. Ecol. 87(2), 416–427 (2014).
Zhu, C. et al. A meta-analysis of Lactobacillus-based probiotics for growth performance and intestinal morphology in piglets. Front. Vet. Sci. 9, 1045965 (2022).
Sun, Z., Li, H., Li, Y. & Qiao, J. Lactobacillus salivarius, a potential probiotic to improve the health of LPS-challenged piglet intestine by alleviating inflammation as well as oxidative stress in a dose-dependent manner during weaning transition. Front. Vet. Sci. 16(7), 547425 (2020).
García, K. E. et al. Microbial fermentation patterns, diarrhea incidence, and performance in weaned piglets fed a low protein diet supplemented with probiotics. FNS 05(18), 1776–1786 (2014).
Heo, J. M. et al. Gastrointestinal health and function in weaned pigs: A review of feeding strategies to control post-weaning diarrhoea without using in-feed antimicrobial compounds: Feeding strategies without using in-feed antibiotics. J. Anim. Physiol. Anim. Nutr. 97(2), 207–237 (2013).
Xia, J. et al. Research progress on diarrhoea and its mechanism in weaned piglets fed a high-protein diet. Anim. Physiol. Nut. 106(6), 1277–1287 (2022).
Pluske, J. R., Turpin, D. L. & Kim, J. C. Gastrointestinal tract (gut) health in the young pig. Anim. Nut. 4(2), 187–196 (2018).
Bomba, L. et al. Gut response induced by weaning in piglet features marked changes in immune and inflammatory response. Funct. Integr. Genom. 14(4), 657–671 (2014).
Grilli, E. et al. Impact of dietary organic acids and botanicals on intestinal integrity and inflammation in weaned pigs. BMC Vet. Res. 11(1), 96 (2015).
Ma, J. et al. Mixed organic acids as an alternative to antibiotics improve serum biochemical parameters and intestinal health of weaned piglets. Anim. Nut. 7(3), 737–749 (2021).
Rauw, W. M. Immune response from a resource allocation perspective. Front. Genet. 3, 267. https://doi.org/10.3389/fgene.2012.00267/abstract (2012).
Pluske, J. R., Hampson, D. J. & Williams, I. H. Factors influencing the structure and function of the small intestine in the weaned pig: A review. Livest. Prod. Sci. 51(1–3), 215–236 (1997).
Connolly, K. R., Sweeney, T., Ryan, M. T., Vigors, S. & O’Doherty, J. V. Impact of reduced dietary crude protein and propionic acid preservation on intestinal health and growth performance in post-weaned pigs. Animals 15(5), 702 (2025).
Soleimany, F., Jinap, S. & Abas, F. Determination of mycotoxins in cereals by liquid chromatography tandem mass spectrometry. Food Chem. 130(4), 1055–1060 (2012).
Sauvant, D., Perez, J. M., Tran, G., (eds) Tables of composition and nutritional value of feed materials: Pigs, poultry, cattle, sheep, goats, rabbits, horses and fish [Internet]. Brill | Wageningen Academic; 2004 [cited 2024 Mar 19]. 77–289. Available from: https://brill.com/view/title/68487
NRC. Nutritional Requirements of Swine (National Academcic Press, Washington DC, USA, 2012).
Campbell, J. M., Crenshaw, J. D. & Polo, J. The biological stress of early weaned piglets. J. Anim. Sci. Biotechnol. 4(1), 19 (2013).
Lallès, J. P., Bosi, P., Smidt, H. & Stokes, C. R. Weaning—A challenge to gut physiologists. Livest. Sci. 108(1–3), 82–93 (2007).
Iwaki, K. et al. Amino acid analysis by reversed-phase high-performance liquid chromatography. J. Chromatogr. A 407, 273–279 (1987).
Van Soest, P. J., Robertson, J. B. & Lewis, B. A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 74(10), 3583–3597 (1991).
Kiernan, D. P., O’Doherty, J. V., Connolly, K. R., Ryan, M. & Sweeney, T. Exploring the differential expression of a set of key genes involved in the regulation and functioning of the stomach in the post-weaned pig. Vet. Sci. 10(7), 473 (2023).
Clarke, L. C. et al. Effect of β-glucanase and β-xylanase enzyme supplemented barley diets on nutrient digestibility, growth performance and expression of intestinal nutrient transporter genes in finisher pigs. Anim. Feed Sci. Technol. 238, 98–110 (2018).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5), 335–336 (2010).
Magoč, T. & Salzberg, S. L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21), 2957–2963 (2011).
Rognes, T., Flouri, T., Nichols, B., Quince, C. & Mahé, F. VSEARCH: A versatile open source tool for metagenomics. PeerJ 18(4), e2584 (2016).
Eren, A. M. et al. Oligotyping: Differentiating between closely related microbial taxa using 16S rRNA gene data. Methods Ecol. Evol. 4(12), 1111–1119 (2013).
Angly, F. E. et al. CopyRighter: A rapid tool for improving the accuracy of microbial community profiles through lineage-specific gene copy number correction. Microbiome 2(1), 11 (2014).
Kim, B. R. et al. Deciphering diversity indices for a better understanding of microbial communities. J. Microbiol. Biotechnol. 27(12), 2089–2093 (2017).
Wagner, B. D. et al. On the use of diversity measures in longitudinal sequencing studies of microbial communities. Front. Microbiol. 22(9), 1037 (2018).
McMurdie, P. J. & Holmes, S. Phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8(4), e61217 (2013).
Acknowledgements
The authors express their gratitude to Ms Denise Cunnigham, Mr John Ryan, Dr. Maria Markiewicz-Keszycka, and Mr Kevin Thornton for their assistance with the analysis of feed, digesta and tissue samples.
Funding
This research was supported by funding from the Department of Agriculture, Food and the Marine (DAFM), grant number 2019R518.
Author information
Authors and Affiliations
Contributions
Conceptualisation and Methodology, K.R.C. and J.V.O.; Investigation, K.R.C.; Resources, J.V.O. and T.S.; Data curation, K.R.C., S.M., M.R. and V.G.; Formal analysis, K.R.C., J.V.O. and S.V.; Writing—original draft, K.R.C.; Writing—review and editing, K.R.C., J.V.O., T.S., and S.V.; Funding acquisition and Supervision, J.V.O. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Connolly, K.R., Sweeney, T., Ryan, M.T. et al. Organic acid-preserved grain improves growth and gut health in weanling pigs fed zinc oxide free diets. Sci Rep 16, 1034 (2026). https://doi.org/10.1038/s41598-025-30702-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30702-1