Abstract
Tuberculosis preventive treatment (TPT) is essential for pediatric tuberculosis control in children and adolescents. Evidence indicates most attrition occurs prior to TPT initiation rather than from post-initiation nonadherence. This study systematically evaluated TPT acceptance for latent tuberculosis infection (LTBI) among Chinese children and adolescents and identified key influencing factors through comprehensive database searches (PubMed, Web of Science, Embase, Cochrane, CNKI, Wanfang database, CBM, VIP) up to February 21, 2025. The heterogeneity was assessed using I2 statistics (95.770%, P < 0.001), with random-effects models employed for meta-analysis. Subgroup, meta-regression, and sensitivity analyses were performed to identify the sources of heterogeneity. Begg and Egger tests were used to detect any potential publication bias. Among 7,165 screened records, 31 eligible studies (n = 4,929) met inclusion criteria. The pooled TPT acceptance rate reached 64.0% (95%CI: 56.9%-70.9%), with significantly higher TPT acceptance rates observed among: central China residents (76.5%, 95% CI: 69.8%–82.5%), close household contacts (80.2%, 95% CI: 74.9% − 85.0%), IGRA positive (80.5%, 95% CI: 74.3% − 86.0%), recipients of isoniazid-rifapentine regimens (76.5%, 95% CI: 69.5%–82.9%), and twice-weekly dosing schedules (76.5%, 95% CI: 69.5%–82.9%). This study revealed the suboptimal uptake of TPT among children and adolescents with LTBI in China. These findings highlight the need to tailor interventions to regional epidemiological profiles, accurately identify the target population for TPT with reliable tools, and scale up optimized regimens under stringent monitoring, as these are critical strategies for enhancing TPT coverage and advancing the “End TB” initiative.
Similar content being viewed by others
Introduction
Latent tuberculosis infection (LTBI) is a state of persistent bacterial viability, immune control, and lack of evidence of clinically manifested active tuberculosis (TB), constituting a public health problem that requires effective interventions1,2. Approximately one-quarter of the global population is infected with Mycobacterium tuberculosis3. China is not only a high-TB burden nation but also ranks among the countries most severely affected by LTBI worldwide4,5. Approximately 250 million individuals in China are diagnosed with LTBI. The prevalence rate of LTBI was 18.08% in populations aged ≥ 5 years and 20.34% among those aged ≥ 15 years6,7. Latent tuberculosis infection constitutes a fundamental obstacle to TB epidemic containment and serves as a persistent disease reservoir that continuously generates new active cases8. Without intervention, approximately 5%–10% of individuals with LTBI will eventually experience symptoms and develop TB9. Tuberculosis can affect anyone regardless of age or sex. Moreover, compared to adults, children with LTBI are at a higher risk of progression to TB1,10. The 2-year cumulative incidence of LTBI in children aged < 5 years was up to 19%, and adolescents faced an increasing risk following childhood11. In 2023, there were approximately 1.3 million new cases of TB in children and adolescents aged 0–14 years and approximately 200,000 deaths12.
The implementation of tuberculosis preventive treatment (TPT) among children and adolescents plays a vital role in controlling pediatric TB13. Ayieko et al. demonstrated that isoniazid prophylaxis reduced the risk of TB development by 59% in children aged ≤ 15 years14. Moreover, Martinez et al. reported that, among children aged < 19 years who were in close contact of patients with TB and had tested positive for infection, the efficacy of preventive treatment reached 85%11. However, despite the proven high efficacy of preventive therapy for LTBI in averting TB onset15, the progress in providing such treatments to children and adolescents has been slow13. In 2022, only 37% of 1.56 million eligible household contacts < 5 years accessed TPT and coverage was even lower for household contacts above 5 years, with only 11% out of 11.4 million initiated on TPT16. In 2023, the global coverage of preventive therapy for close family contacts of patients with TB was only 21%12.
Reviews addressing the cascade of care for TB infection concluded that most attritions occurred before the initiation of TPT, rather than due to non-adherence to TPT after its initiation. However, at present, systematic reviews and meta-analyses regarding preventive therapy for LTBI in children and adolescents mainly focused on compliance, effectiveness, and safety of treatment regimens1,17,18. Although Rustage et al.19 and Sagili et al.20 evaluated the initiation and completion of LTBI treatment, their study populations were migrants and household contacts of patients with TB, respectively. No systematic review or meta-analysis has been conducted to assess the acceptance of preventive treatment in children and adolescents. We conducted this systematic review and meta-analysis to evaluate the acceptance of LTBI preventive therapy among Chinese children and adolescents and explore the factors influencing the acceptance of TPT, thereby providing evidence-based recommendations for strengthening TPT.
Methods
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standards21 (see Supplementary File 1), and the protocol was registered in PROSPERO (CRD420250655042).
Research strategy
We searched PubMed, Web of Science, Embase, Cochrane Library, Chinese National Knowledge Infrastructure (CNKI), Wanfang Database, Chinese Biomedical Database, and VIP database for eligible studies published in English or Chinese as of February 21, 2025. This study aimed to collect articles on the prophylactic treatment of LTBI in Chinese children and adolescents. The search strategy combined MeSH and keywords for key concepts (e.g., China, students, tuberculosis, latent infection, preventive therapy). For instance, the Web of Science query was: (ALL= (China OR Chinese) AND TS= (schoo* OR kindergar* OR studen* OR infant OR infants OR newborn OR child OR children OR kid OR kids OR pediatri* OR adolescen* OR teenage* OR juvenil* OR freshman OR freshmen) AND TS= (tuberculo* OR TB OR MTB OR PTB) AND TS= (latent infectio* OR latent tuberculo* OR latent pulmonary tuberculo* OR latent TB OR LTBI OR tuberculosis infectio* OR TB infectio* OR latent Mycobacterium tuberculo* OR latent M. tuberculo* OR inactive tuberculo* OR inactive TB OR inactive mycobacterium tuberculo* OR Mycobacterium tuberculosis infectio* OR dorman* tuberculo* OR dorman* TB OR TBI) AND TS= (therap* OR treatmen* OR treat* OR prophyla* OR preven* OR precautio* OR pharmacotherap* OR immunotherapy OR chemotherapy)) OR (ALL= (China OR Chinese) AND TS= (schoo* OR kindergar* OR studen* OR infant OR infants OR newborn OR child OR children OR kid OR kids OR pediatri* OR adolescen* OR teenage* OR juvenil* OR freshman OR freshmen) AND TS= (tuberculo* OR TB OR MTB OR PTB) AND TS= (prevent* therap* OR prevent* treatmen* OR prophylactic therap* OR prophylactic treatmen* OR prevent* medication OR TPT OR chemoprevent* OR chemo-prevent* OR chemoprophyla* OR chemo-prophyla* OR chemical prophylaxis OR immunoprophylaxis OR immun* prevention)). Full strategies are in Supplementary File 2. The reference lists of the relevant articles or reviews were manually checked for other potentially relevant studies.
Inclusion and exclusion criteria
Studies relevant to children and adolescents aged < 18 years in China who had LTBI, irrespective of the definition of LTBI adopted by the authors, were included in this meta-analysis. The inclusion of studies was not restricted by study type; randomized controlled trials, case–control studies, cohort studies, and cross-sectional studies were all eligible. Studies that reported on preventive treatment regimens, the number of individuals eligible for TPT, and the number of individuals who initiated TPT were included. Studies that reported regimens including isoniazid monotherapy (INH, H), rifampicin monotherapy (RIF, R), combination therapy of isoniazid and rifampicin (INH-RIF, HR), combination therapy of isoniazid and rifapentine (INH-RFT, HP), and immunoprophylactic treatments were included. In case of multiple publications by the same researchers, only the earliest or the most complete dataset or the largest population study was adopted.
The exclusion criteria were as follows: (1) studies that focused on the contacts of patients with drug-resistant TB; (2) studies that implemented multiple preventive therapies but did not report the outcomes for each intervention separately or studies that failed to provide extractable data even after contacting the authors for clarification; (3) case reports, conference literature, reviews, editorials, letters, animal studies, and in vitro studies; and, (4) studies not published in English or Chinese and studies without full text.
Data extraction and quality assessment
Two reviewers (L.A.P and Y.Q.Z.) independently screened the studies retrieved from the literature search based on the inclusion and exclusion criteria and independently completed data extraction using a predefined data extraction form to ensure accuracy. Attempts were made to retrieve missing data by contacting the corresponding author. The following data were extracted from each included study: first author’s name, year of publication, title, study design, research setting, timing of data collection, study location, target population criteria for TPT, prophylactic treatment regimen, duration of treatment, number of individuals eligible for TPT, number of individuals who initiated TPT, and reasons for refusal of TPT.
Referring to previous meta-analyses22,23,24, two reviewers (L.A.P and Y.Q.Z.) independently used the Newcastle–Ottawa Scale (NOS) to assess the quality of cohort studies and a modified NOS to assess the quality of cross-sectional studies. The NOS22 has been widely employed to evaluate the methodological quality of observational studies. Official versions exist for cohort and case–control studies22, whereas modified versions exist for cross-sectional studies25,26. The maximum NOS score for cohort studies was nine points, and the maximum score for the modified NOS was 10 points.
Any discrepancies during data extraction and quality assessment were resolved by consulting a third reviewer (B.C.).
Data analysis
The extracted data were imported into Stata version 18.0 for the meta-analysis. The statistical heterogeneity among the included studies was assessed using Cochran Q test and the I2 statistic. A random-effects model was employed for the meta-analysis when there was significant heterogeneity among studies (P < 0.10 or I2 > 50%). Conversely, a fixed-effects model was used when no heterogeneity was detected27. The potential sources of heterogeneity were explored using subgroup, meta-regression, and sensitivity analyses. Subgroup analyses were conducted according to the region, research setting, regimen, duration of preventive therapy, dosage frequency, and study start time. Begg test and Egger linear regression test were used to assess publication bias28,29. Unless otherwise specified, P-values were two-tailed, and statistical significance was defined at P < 0.05.
In particular, China was categorized into four major regions (eastern, central, western, and northeastern) according to the latest economic zone classification standard issued by the National Bureau of Statistics30. This classification employs comprehensive socioeconomic criteria—covering geographical location, level of economic development, and technological advancement—to reflect regional disparities. Specifically, the eastern region includes 10 provinces/municipalities: Beijing, Tianjin, Hebei, Shanghai, Jiangsu, Zhejiang, Fujian, Shandong, Guangdong, and Hainan. The central region comprises six provinces: Shanxi, Anhui, Jiangxi, Henan, Hubei, and Hunan. The western region consists of 12 provinces/autonomous regions/municipalities: Inner Mongolia, Guangxi, Chongqing, Sichuan, Guizhou, Yunnan, Shaanxi, Gansu, Qinghai, Ningxia, Xinjiang, and Tibet. The northeastern region includes three provinces: Liaoning, Jilin, Heilongjiang.
Results
Study selection and study characteristics
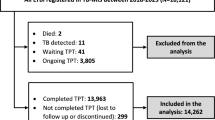
From our multi-database search, 7,165 potential articles were identified. After title, abstract, and full-text reviews, 31 articles with 4929 participants were included in this systematic review and meta-analysis, spanning 2004 to 202431,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61. Among these, 19 were cross-sectional studies and 12 were cohort studies. Seventeen studies31,32,35,38,39,41,42,45,50,51,52,53,54,56,58,60,61 were conducted in the eastern region of China; seven36,37,43,47,55,57,59, central; five33,34,40,46,48, western; one44, northeastern; and, one49, both central and western regions. Eighteen studies focused on school outbreak screening; two, household contact screening; and, 11, routine screening. Based on the criteria for defining the target population for TPT, the literature distribution was as follows: three studies used tuberculin skin test (TST) ≥ 10 mm or a strong reaction, 22 studies used TST ≥ 15 mm or a strong reaction, three studies used TST ≥ 20 mm or a strong reaction, one study used interferon-gamma release assay (IGRA) positivity, and two studies used TST ≥ 15 mm or a strong reaction or IGRA positivity. Prophylactic treatment regimens included isoniazid monotherapy (H), combination therapy with isoniazid and rifampicin (HR), and combination therapy with isoniazid and rifapentine (HP). The H and HR regimens were administered once daily, whereas the HP regimen was administered twice weekly. The studies were initiated between 2004 and 2023, with sample sizes ranging from 11 to 846. Figure 1 presents a flowchart detailing the study selection process. Table 1 provides detailed information about the selected studies.
PRISMA flow diagram for study selection.
Quality of the included studies
The quality scores of the 19 cross-sectional studies ranged from 5 to 9, whereas those of the 12 cohort studies ranged from 6 to 8 (see Supplementary File 3).
Acceptance rate of TPT among Chinese children and adolescents
We conducted a meta-analysis of the included cross-sectional and cohort studies. Considering that the data extraction for cohort studies was restricted to the baseline survey time point and given the homogeneity of their data structure with that of cross-sectional studies, we merged and analyzed these cohort studies with cross-sectional studies during the meta-analysis process. The pooled results showed that the acceptance rate of preventive therapy among Chinese children and adolescents with LTBI was 0.640 (95% CI: 0.569–0.709, P < 0.001), with high heterogeneity (I2random effects model = 95.770%, P < 0.001) (Fig. 2).
Acceptance rate of TPT among Chinese children and adolescents analyzed by the Forest plot.
The results of the subgroup analyses are presented in Table 2. The subgroup analysis revealed significant heterogeneity among the subgroups stratified by region (Q− between = 8.985, P = 0.029), research setting (Q− between = 19.766, P < 0.001), preventive therapy criteria (Q− between = 33.378, P < 0.001), regimen (Q− between = 9.755, P = 0.008), and dosage frequency (Q− between = 9.679, P = 0.002). In particular, among 17 studies conducted in the eastern region of China, the TPT acceptance rate among children and adolescents was 0.615 (95% CI: 0.509–0.717). The TPT acceptance rate in the western region was slightly lower than that in the eastern region, at 0.531 (95% CI: 0.284–0.771). The central region had a significantly higher TPT acceptance rate than those in the other areas, reaching 0.765 (95% CI: 0.698–0.825). The acceptance rate of TPT among Chinese children and adolescents with LTBI was highest in LTBI cases identified through household contact screening (0.802), followed by those detected through school outbreak screening (0.663), and lowest in cases identified via routine screening (0.522). Among different preventive therapy criteria, the TPT acceptance rate was highest at 0.805 when using IGRA positivity as the criterion, followed by 0.657 for the TST ≥ 20 mm or strong reaction group, 0.615 for the TST ≥ 15 mm or strong reaction group, and 0.559 for the TST ≥ 10 mm or strong reaction group. Regarding the different preventive treatment regimens, the acceptance rate of TPT for INH and RFT regimens was the highest at 0.765. In terms of medication frequency, the TPT acceptance rate for a twice-weekly regimen was 76.5%, compared to 58.9% for a daily regimen. We also performed a meta-regression analysis that included covariates for the following six subgroups: region (P = 0.238), research setting (P = 0.033), preventive therapy criteria (P = 0.224), regimen (P = 0.264), duration of preventive therapy (P = 0.840), dosage frequency (P = 0.086), and study start time (P = 0.218). Furthermore, the sensitivity analysis of the pooled acceptance rate of preventive therapy among Chinese children and adolescents with LTBI indicated that the results were stable (see Supplementary File 4).
Potential publication bias
We employed Begg test and Egger linear regression test to evaluate the potential publication bias for the pooled acceptance rate of preventive therapy among Chinese children and adolescents with LTBI (Fig. 3). The results of both Begg test (PB = 0.083) and Egger test (PE = 0.093) were consistent, suggesting no significant publication bias.
Combined assessment of publication bias using Begg test and Egger regression.
Discussion
Tuberculosis preventive treatment plays a pivotal role in controlling and eliminating TB. Achieving optimal TPT coverage is critical for protecting children and adolescents exposed to TB from developing TB17,62. In September 2023, at the second United Nations High-Level Meeting on the fight against TB, renewed commitments to address TB in children and adolescents have been defined63. By 2027, 90% of individuals at a high risk of developing TB will be provided with preventive treatment, which translates to providing TPT to 30 million household contacts of people with TB, including children. The political declaration also committed to urgently scaling up comprehensive efforts to close long-standing gaps in the prevention, diagnosis, treatment, and care of children with or at risk of TB62,63.
This first meta-analysis evaluating the acceptance rate of TPT among Chinese children and adolescents synthesized individual-level data from 31 regionally diverse studies. The pooled analysis revealed that only 64.0% of the eligible individuals received preventive therapy. This proportion was lower than that observed among immigrants (69.0%)19, highlighting a considerable shortfall compared to the global targets63. The acceptance rates for various preventive treatment regimens exhibited a wide range, from 9.7% to 96.8%, implying that enhancing the preventive treatment rate is indeed feasible.
The TB epidemic in China exhibited significant spatial heterogeneity, which stemmed from the interaction of multidimensional factors such as regional economic development gradients, population structure heterogeneity, spatial density of medical infrastructure, and local fiscal input capabilities64,65,66. Our study found significant heterogeneity in the acceptance rate of TPT across different regions of China. Unlike the situation in which regions with higher economic levels had lower TB epidemics, the Central region had the highest TPT acceptance rate, reaching 76.5%, whereas the acceptance rate in the Eastern region was only 61.5%. This phenomenon may be caused by multiple factors. First, owing to the high prevalence of TB, in addition to implementing the national TB strategy, central provinces implemented a series of non-fragmented local intervention combinations and achieved remarkable results67,68. For instance, in 2024, Hunan province achieved the largest decline in the TB incidence rate nationwide, with the reported incidence rate among students dropping to < 10/100,000 for the first time. The acceptance rate of preventive treatment among students increased by 65.7%69. Second, despite the relatively low prevalence of TB in the eastern region, awareness of the risk of TB may be inadequate. A survey of the public’s awareness of core TB knowledge in 2020 revealed that the overall awareness rate of five key TB prevention and control messages among the general public in Hunan Province was 88.3%70, compared with 81.5% in Beijing71. The acceptance of LTBI preventive treatment increases as the level of tuberculosis-related knowledge rises72,73. Moreover, residents in economically developed regions (eastern region) generally have a higher level of education, and the level of education is negatively correlated with the TPT acceptance rate74,75.
There were significant differences in the acceptance rates of targeted TPT for children and adolescents with LTBI across different research settings. In particular, the highest TPT acceptance rate was observed among household close contacts (80.2%), followed by those identified through school outbreak screening (66.3%). The lowest acceptance rate was observed among LTBI cases detected through general screening (52.2%). The disparities in the acceptance rate of TPT were primarily attributed to differences in awareness levels, intensity of medical management, social support, and policy safeguards76,77. In contrast to routine screening, the propensity to accept TPT is elevated among individuals with LTBI who are close contact with patients with TB73,78,79. Household close contacts generally had a higher level of awareness regarding the hazards of the disease and the necessity of TPT. Owing to their high exposure to and elevated risk of developing the disease, household close contacts were identified as the core target population for LTBI screening and intervention by the WHO and multiple countries12,80,81,82,83. Tn high-burden TB countries, the initiation rate of TPT for LTBI among household contacts of patients with TB exceeds 90%20. This rate was significantly higher than that reported in other populations. The Chinese government has attached great importance to TB control in schools. The “Guidelines for Tuberculosis Prevention and Control in Schools” explicitly required that once a case of active TB was confirmed within a school, a close contact investigation should be carried out, and individuals with a strongly positive result on the tuberculin skin test and normal findings on chest radiographs were advised to undergo preventive treatment84. Household and school contacts were typically incorporated into mandatory or semi-mandatory screening systems where they receive more intensive education and follow-up. These actions have improved the management of LTBI in close contacts of individuals with active TB4.
Subgroup analysis revealed a gradient in TPT acceptance rates across populations defined by different diagnostic criteria. The rate was highest in the IGRA-positive group (80.5%), attributable to IGRA’s superior specificity for LTBI. Unlike TST, IGRA was not affected by Bacille Calmette–Guérin (BCG) vaccination, most non-tuberculous mycobacterial infections, or subjective judgment of the operator, thus providing a more reliable diagnosis of true infection4,85. This diagnostic accuracy, coupled with meta-evidence showing a higher progression risk to active TB among IGRA-positive individuals86, enhanced both clinician recommendation confidence and participant acceptance. A dose-response relationship was observed with TST thresholds, where higher cut-offs (e.g., 10 mm, 15 mm, 20 mm) were associated with progressively increasing acceptance rates, consistent with clinical risk stratification. This trend aligned with evidence that larger TST induration reflects a greater risk of disease progression87, reinforcing risk perception among clinicians and participants and promoting willingness to initiate treatment. The lower TPT acceptance rate (57.2%) observed in the composite criteria group (TST ≥ 15 mm/strong reaction or IGRA positive) might be attributable to the limited number of included studies (only 2) and small sample size (n = 441), which could limit the precision and generalizability of the estimate.
The subgroup analysis also revealed that, compared with different preventive treatment regimens, the acceptance rate of INH and RFT administered twice weekly was higher than that of daily INH or daily INH and RIF. Combination drug therapy was more effective than single drug therapy for TPT88. The combination of two bactericidal drugs with different mechanisms of action can enhance the therapeutic effects18. Although the INH regimen was the earliest, most widely used, and most mature scheme with a relatively high protection rate, its acceptance was relatively low owing to issues such as side effects and the long duration of medication89,90,91. In contrast, the intermittent combination regimen of INH and RFT was more readily accepted and had higher compliance owing to its better tolerability, shorter duration, and long-acting intermittent dosing characteristics of RFT77,91. The combination regimen of INH and RIF is characterized by a short treatment duration and comparable protective efficacy. However, its acceptance rate was relatively low, which may be partly attributed to the higher dosing frequency compared to the INH and RFT regimen, as well as the more pronounced hepatotoxicity92. Santos et al. conducted a systematic review and network meta-analysis to retrospectively compare the effectiveness and safety of different treatment regimens for LTBI in patients aged < 15 years. They found that the combination of INH and RFT was likely the most effective option compared to other active drugs and placebos for preventing TB in the overall pediatric population18. Considering the individual genetic background and the fact that the frequency of the rapid acetylator genotype of N-acetyltransferase 2 is higher among Asians (approximately 50%) than among Caucasians (approximately 5%), China has widely adopted a 3-month regimen of INH and RFT (3H2P2) administered twice weekly for the treatment of LTBI, rather than the WHO-recommended 3HP regimen (a 3-month regimen of weekly INH and RFT). This regimen has also been shown to have a high protection rate and low incidence of liver dysfunction4.
Additionally, our study found that the primary barriers to preventive treatment were concerns about drug side effects and inadequate awareness of the disease. The concerns of some individuals with LTBI and their families regarding the costs, resources, and time required for TB infection treatment also influence decision-making. These factors have been identified in previous studies74,77,79,93,94. The existing evidence demonstrates that both the TPT regimen using INH alone and the combination of INH with RFT/RIF have good safety profiles. However, there was a deviation between the public’s perception of risk and the actual risk. Future efforts should focus on bridging this gap by enhancing health education, optimizing services, and reinforcing policy safeguards. Moreover, continuous endeavors are needed to increase the population’s acceptance of TPT.
This study has a few limitations. First, the included studies failed to provide detailed reports on TB awareness levels and economic status of individuals with LTBI (or their guardians), which are potentially important factors that could affect the true attribution analysis of the acceptance rate. Second, the eligible studies were mainly concentrated in the eastern provinces of China; however, the number of articles was still inadequate in some less developed or relatively sparsely populated regions. Third, compared to the vast population of individuals with LTBI, the sample size included in this study was relatively limited. Despite these limitations, this study systematically analyzed all available research data and, for the first time, revealed the acceptance rate of preventive treatment for LTBI among children and adolescents in China, providing important insights for advancing TPT in this population.
Conclusions
This study revealed the suboptimal uptake of TPT among children and adolescents with LTBI in China. Tailoring interventions to regional epidemiological profiles, accurately identifying the target population for TPT with reliable tools, and scaling up optimized regimens under stringent monitoring are critical strategies for enhancing TPT coverage and advancing the “End TB” initiative. Given the limitations of these studies, future research is recommended to collect sociodemographic variables related to individuals with LTBI and explore their impact on acceptance rates. Alternatively, mixed-methods approaches combining quantitative data with qualitative interviews can be employed to uncover the drivers of decision making.
Data availability
All original data generated or analyzed during this research are included in the article and its supplementary information. Additional requests may be directed to the corresponding authors.
References
Sabella-Jiménez, V. et al. Effectiveness and safety of available preventive tuberculosis treatment regimens for children and adolescents: protocol for a systematic review and network meta-analysis. BMJ Paediatr. Open. 6, e001551 (2022).
Getahun, H., Matteelli, A., Chaisson, R. E. & Raviglione, M. Latent Mycobacterium tuberculosis infection. N Engl. J. Med. 372, 2127–2135 (2015).
Cohen, A., Mathiasen, V. D., Schön, T. & Wejse, C. The global prevalence of latent tuberculosis: a systematic review and meta-analysis. Eur. Respir J. 54, 1900655 (2019).
Cui, X., Gao, L. & Cao, B. Management of latent tuberculosis infection in china: exploring solutions suitable for high-burden countries. Int. J. Infect. Dis. 92, S37–S40 (2020).
Houben, R. M. G. J. & Dodd, P. J. The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLoS Med. 13, e1002152 (2016).
Gao, L., Zhang, H. & Hu, M. Expert consensus on the estimation of the national burden on latent tuberculosis infection. Chin. J. Antituberc. 44, 4–8 (2022).
Gao, L. et al. Estimation of the national burden on latent tuberculosis infection based a multi-center epidemiological survey and the space statistics model. Chin. J. Antituberc. 44, 54–59 (2022).
Wang, X. et al. Current status of tuberculosis burden in China. Chin. J. Epidemiol. 45, 857–864 (2024).
World Health Organization. Fact sheets: tuberculosis. (2025). https://www.who.int/news-room/fact-sheets/detail/tuberculosis
Sterling, T. R. et al. Guidelines for the treatment of latent tuberculosis infection: recommendations from the National Tuberculosis Controllers Association and CDC, 2020. Mmwr Recomm Rep. 69, 1–11 (2020).
Martinez, L., Cords, O., Horsburgh, C. R. & Andrews, J. R. The risk of tuberculosis in children after close exposure: a systematic review and individual-participant meta-analysis. Lancet 395, 973–984 (2020).
World Health Organization. Global Tuberculosis Report 2024 (World Health Organization, 2024).
Wang, Q., Wang, J., Li, Y., Liu, E. & Zhou, L. Attach importance to childhood tuberculosis and care for children’s health. Chin. J. Antituberc. 45, 1–5 (2023).
Ayieko, J. et al. Efficacy of Isoniazid prophylactic therapy in prevention of tuberculosis in children: a meta-analysis. BMC Infect. Dis. 14, 91 (2014).
Martinez, L. et al. Effectiveness of preventive treatment among different age groups and Mycobacterium tuberculosis infection status: a systematic review and individual-participant data meta-analysis of contact tracing studies. Lancet Respir Med. 12, 633–641 (2024).
World Health Organization. Global Tuberculosis Report 2023 (World Health Organization, 2023).
Pangprasertkul, S., Buawangpong, N., Pinyopornpanish, K., Jiraporncharoen, W. & Angkurawaranon, C. Treatment completion and safety profile of once-weekly 3HP regimen for tuberculosis preventive treatment in children and adolescents: a systematic review. BMC Infect. Dis. 25, 436 (2025).
Santos, J. M. et al. Systematic review with network meta-analysis on the treatments for latent tuberculosis infection in children and adolescents. J. Infect. Chemother. 28, 1645–1653 (2022).
Rustage, K. et al. Initiation and completion of treatment for latent tuberculosis infection in migrants globally: a systematic review and meta-analysis. Lancet Infect. Dis. 21, 1701–1712 (2021).
Sagili, K. D. et al. Strategies to detect and manage latent tuberculosis infection among household contacts of pulmonary TB patients in high TB burden countries - a systematic review and meta-analysis. Trop. Med. Int. Health. 27, 842–863 (2022).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Wells, G. A. et al. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Hospital Research Institute. (2021). https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp
Moskalewicz, A. & Oremus, M. No clear choice between Newcastle–Ottawa scale and appraisal tool for cross-sectional studies to assess methodological quality in cross-sectional studies of health-related quality of life and breast cancer. J. Clin. Epidemiol. 120, 94–103 (2020).
Zhou, Y. et al. Association of variant vitamin statuses and tuberculosis development: a systematic review and meta-analysis. Ann. Med. 56, 2396566 (2024).
Modesti, P. A. et al. Panethnic differences in blood pressure in europe: a systematic review and meta-analysis. PLoS One. 11, e0147601 (2016).
Yoo, H., Kim, J. Y., Lee, Y. M. & Kang, M. Y. Occupational risk factors associated with lower urinary tract symptoms among female workers: a systematic review. Occup. Environ. Med. 80, 288–296 (2023).
Hedges, L. V. & Vevea, J. L. Fixed- and random-effects models in meta-analysis. Psychol. Methods. 3, 486–504 (1998).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Begg, C. B. & Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 50, 1088–1101 (1994).
National Bureau of Statistics. Part IV: Statistical Systems and Classification Standards (16 Items). (2024). https://www.stats.gov.cn/hd/cjwtjd/202302/t20230207_1902279.html
Dong, S., Wang, L., Liu, X. & He, B. Analysis of tuberculin skin test results among 14,501 primary school students. Chin. J. Sch. Doctor. 20, 299 (2006).
Guo, X. & Liu, Q. Screening of tuberculosis among freshmen in senior high schools in Rugao City from 2018 to 2021. Jiangsu J. Prev. Med. 35, 182–184 (2024).
Wu, B. Public health publicity on controlling infection and prophylactic medication of close contacts in the families of tuberculosis patients. Chin. Health Ind. 16, 156–157 (2019).
Shi, S. Investigation on the current situation of latent tuberculosis infection among new students in compulsory education schools in Longchang City. Chin. Health Care Nutr. 28, 279–280 (2018).
Zhou, Y., Liu, C. & Liu, S. Investigation and analysis of tuberculosis patients found in the college entrance examination physical examinations of four middle schools in Yangjiang City, Guangdong Province. Chin. J. Antituberc. 35, 1029–1030 (2013).
Yu, G. & Ma, X. Analysis of tuberculin skin test results of new entrants to primary and secondary schools in Wancheng District, Nanyang City in 2010. Chin. Pract. J. Rural Doctor. 19, 21–22 (2012).
Hu, Y., Chen, G. & Yu, G. Analysis of tuberculin skin test results of new students in primary and secondary schools in Wancheng District, Nanyang City in 2017. Henan Med. Res. 27, 2945–2946 (2018).
Guan, L. & Yan, B. Investigation on the infection status of Mycobacterium tuberculosis among primary and secondary school students in Qufu City. Occup. Health. 21, 1191 (2005).
Jiang, K. & Zhao, C. Investigation on PPD test and TB control knowledge among primary and secondary school students in Sishui County. Occup. Health. 26, 917–918 (2010).
Pan, D., Cui, Z., Tan, H. & Lan, R. Dynamic changes of tuberculosis infection among high school of local and non-local students after one year of enrollment. Chin. Trop. Med. 21, 939–942 (2021).
Jia, X. et al. Investigation of pulmonary tuberculosis cluster in a boarding school in Beijing. J. Tuberc Lung Dis. 4, 13–19 (2023).
Weng, L. & Wang, Y. Investigation of tuberculosis cases occurring during the preventive medication process for a school tuberculosis epidemic. Chin. J. Infect. Dis. 35, 699–700 (2017).
Li, Y., Jin, X., Wei, X., Duan, H. & Jin, H. Epidemiological investigation and management of a tuberculosis cluster epidemic in a school. J. Tuberc Lung Dis. 1, 89–92 (2020).
Zheng, J. Investigation and its control measures on outbreak of the general level of tuberculosis. Chin. Trop. Med. 16, 184–185 (2016).
Gong, S., Qiao, B. & Ding, Y. Investigation of a tuberculosis epidemic in a primary school in Yiwu City. Shanghai J. Prev. Med. 29, 957–959 (2017).
Hou, Y., Cai, L., Qin, F. & Huang, X. Intervention effect of health education among close contacts of tuberculosis in schools on TST strongly positive individuals. Health Prot. Promot. 24, 1786–1789 (2024).
Wang, N. et al. Feasibility of preventive therapy for latent Mycobacterium tuberculosis infection in middle school students. Dis. Surveill. 32, 43–47 (2017).
Fan, J. et al. Investigation and analysis of a school pulmonary tuberculosis aggregated epidemic in Chongqing. Chin. J. Antituberc. 44, 792–796 (2022).
Chen, H. et al. Adherence to preventive treatment for latent tuberculosis infection in close contacts of pulmonary tuberculosis patients: a cluster-randomized controlled trial in China. Int. J. Infect. Dis. 147, 107196 (2024).
Li, H. & Wang, Q. Analysis report on the tuberculosis screening of primary and secondary school students in the urban area of Fengxian in 2004. Harbin Med. J. 28, 9–10 (2008).
Li, C. et al. Analysis on epidemic and management of tuberculosis among senior middle school students from Qinghai Tibetans in a school of Daxing district of Beijing from 2010 to 2019. Chin. J. Antituberc. 42, 1318–1322 (2020).
Fang, Y., Ma, Y., Lu, Q., Sun, J. & Pei, Y. An outbreak of pulmonary tuberculosis and a follow-up investigation of latent tuberculosis in a high school in an Eastern City in China, 2016–2019. PLoS One. 16, e0247564 (2021).
Fang, Y. et al. Outbreak of pulmonary tuberculosis in a Chinese high school, 2009–2010. J. Epidemiol. 23, 307–312 (2013).
Huang, H. Study on Risk Factors of Tuberculosis Outbreak among Adolescent Students in Guangdong Area and Intervention Measures (Southern Medical University, 2018).
Zou, X. et al. Investigation and analysis of tuberculosis epidemic situation in a school in Wugang, hu’nan. J. Tuberc Lung Dis. 2, 366–369 (2021).
Hu, J. et al. Analysis of tuberculosis infection and preventive treatment among close contacts in different types of high schools in Qingpu district of Shanghai. Chin. Prim. Health Care. 37, 78–81 (2023).
Zhou, J. et al. Baseline screening and follow-up cohort study of close contacts of school tuberculosis epidemic. J. Tuberc Lung Dis. 4, 120–127 (2023).
Weng, L. & Wang, Y. Screening close contacts of smear-positive tuberculosis patients and the effectiveness of preventive medication in close contacts in school. Int. J. Epidemiol. Infect. Dis. 41, 258–260 (2014).
Tang, H., Su, H. & Sun, Q. Investigation, treatment and analysis of a cluster epidemic of tuberculosis in a school. Anhui J. Prev. Med. 28, 73–76 (2022).
Zhang, H., Zhang, D., Zhou, J. & Xu, Z. Investigation and analysis on tuberculosis outbreak aggregated in a vocational middle school of Yixing City. J. Environ. Hyg. 7, 43–46 (2017).
Xu, Z. et al. Survey of a school clustering epidemic of pulmonary tuberculosis. J. Med. Pest Control. 37, 275–279 (2021).
World Health Organization. Roadmap Towards Ending TB in Children and Adolescents 3rd edn (World Health Organization, 2023).
United Nations General Assembly. Political declaration of the high-level meeting on the fight against tuberculosis. A/RES/78/5. UN General Assembly, (2023).
He, W. et al. Spatial inequality, characteristics of internal migration, and pulmonary tuberculosis in China, 2011–2017: a Spatial analysis. Infect. Dis. Poverty. 9, 159 (2020).
Wang, L. et al. Spatio-temporal variation in tuberculosis incidence and risk factors for the disease in a region of unbalanced socio-economic development. BMC Public. Health. 21, 1817 (2021).
Hu, M. et al. Unbalanced risk of pulmonary tuberculosis in China at the subnational scale: Spatiotemporal analysis. JMIR Public. Health Surveill. 8, e36242 (2022).
Huang, G. et al. Spatiotemporal analysis of tuberculosis in the Hunan Province, China, 2014–2022. Front. Public. Health. 12, 1426503 (2024).
Jiang, H. et al. Assessment of strategies and epidemiological characteristics of tuberculosis in Henan Province, china: observational study. JMIR Public. Health Surveill. 7, e24830 (2021).
Wang, L., Xu, Z. & Yang, Z. Incidence of tuberculosis in Hunan ranks first in the nation in terms of decline. Hunan today (2025). https://m.voc.com.cn/xhn/news/202503/28199092.html (accessed 4 April 2025).
Huang, J., Ming, H., Wang, Q., Gong, D. & Tang, Y. Awareness rate of core knowledge about tuberculosis prevention and treatment in Hunan Province, 2020. Pract. Prev. Med. 29, 1455–1458 (2022).
Xu, Y. et al. A network survey on knowledge of tuberculosis prevention and control in Beijing. Cap J. Public. Health. 16, 101–104 (2022).
Bar-Meir, M., Pariente, G., Romem, A. & Wiener-Well, Y. Identifying factors affecting latent tuberculosis treatment acceptance among healthcare workers: a retrospective analysis in a tertiary care centre. BMJ Open. 11, e047444 (2021).
Li, Y. et al. Acceptance of chemo-prophylaxis for latent tuberculosis infection among high school/college student contacts of tuberculosis patients in Shanghai, China. Biomed. Environ. Sci. 31, 317–321 (2018).
Ren, J. et al. Barriers to the acceptance of tuberculosis preventive treatment: a multicenter cross-sectional study in China. Biomed. Environ. Sci. 37, 1303–1309 (2024).
Spruijt, I. et al. Implementation of latent tuberculosis infection screening and treatment among newly arriving immigrants in the netherlands: a mixed methods pilot evaluation. PLoS One. 14, e0219252 (2019).
Szkwarko, D. et al. Child contact management in high tuberculosis burden countries: a mixed-methods systematic review. PLoS One. 12, e0182185 (2017).
Campbell, J. I., Sandora, T. J. & Haberer, J. E. A scoping review of paediatric latent tuberculosis infection care cascades: initial steps are lacking. BMJ Glob Health 6 (2021).
Wang, Z. et al. The current situation of willingness to receive prophylactic treatment among students with latent tuberculosis infection and its influencing factors in Jiangsu Province. Chin. Trop. Med. 24, 244–251 (2024).
Yuan, Y. et al. Factors associated with refusal of preventive therapy after initial willingness to accept treatment among college students with latent tuberculosis infection in Shandong, China. BMC Infect. Dis. 23, 38 (2023).
Velen, K., Shingde, R. V., Ho, J. & Fox, G. J. The effectiveness of contact investigation among contacts of tuberculosis patients: a systematic review and meta-analysis. Eur. Respir. J. 58 (2021).
World Health Organization. WHO Consolidated Guidelines on Tuberculosis: Module 1: Prevention: Tuberculosis Preventive Treatment 2nd edn (World Health Organization, 2024).
Zhao, Y. & Chen, M. Technical Guide on Tuberculosis Prevention and Control in China (People’s Medical Publishing House, 2021).
Du, Y. et al. Declining incidence rate of tuberculosis among close contacts in five years post-exposure: a systematic review and meta-analysis. BMC Infect. Dis. 23, 373 (2023).
Chen, H. et al. Tuberculosis in schools and requirements for prevention and control in China. China CDC Wkly. 3, 58–60 (2021).
Lamberti, M. et al. Tuberculin skin test and quantiferon test agreement and influencing factors in tuberculosis screening of healthcare workers: a systematic review and meta-analysis. J. Occup. Med. Toxicol. 10, 2 (2015).
Zhou, G. et al. Management of latent tuberculosis infection in china: exploring solutions suitable for high-burden countries. Int. J. Infect. Dis. 20, 1457–1469 (2020).
Cao, D. et al. The association between tuberculin skin test result and active tuberculosis risk of college students in Beijing, china: a retrospective cohort study. BMC Infect. Dis. 19, 619 (2019).
Shah, R., Khakhkhar, T. & Modi, B. Efficacy and safety of different drug regimens for tuberculosis preventive treatment: a systematic review and meta-analysis. Cureus 15, e38182 (2023).
World Health Organization. Report of a Lessons Learnt Workshop on the Six ProTEST Pilot Projects in Malawi, South Africa and Zambia (World Health Organization, 2004).
Alsdurf, H., Hill, P. C., Matteelli, A., Getahun, H. & Menzies, D. The cascade of care in diagnosis and treatment of latent tuberculosis infection: a systematic review and meta-analysis. Lancet Infect. Dis. 16, 1269–1278 (2016).
Chee, C. B. E., Reves, R., Zhang, Y. & Belknap, R. Latent tuberculosis infection: ppportunities and challenges. Respirology 23, 893–900 (2018).
Yoopetch, P. et al. Efficacy of anti-tuberculosis drugs for the treatment of latent tuberculosis infection: a systematic review and network meta-analysis. Sci. Rep. 13, 16240 (2023).
Wang, N. et al. Influence of tuberculosis knowledge on acceptance of preventive treatment and the moderating role of tuberculosis stigma among china’s general population: cross-sectional analysis. BMC Public. Health. 24, 2300 (2024).
Campbell, J. I. & Menzies, D. Testing and scaling interventions to improve the tuberculosis infection care cascade. J. Pediatr. Infect. Dis. Soc. 11, S94–S100 (2022).
Funding
The study was funded by the “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2025C01134), the Natural Science Foundation of Shenzhen Municipality (JCYJ20240815110459001), the Guangdong Medical Research Fund (A2023156), the Bao’an District’s Medical and Health Research Project (2023JD152), the Science and Technology Program on Disease Prevention and Control of Zhejiang Province (2025JK181) and the Shenzhen Medical Research Fund (B2303003).
Author information
Authors and Affiliations
Contributions
B.C. had the idea for the study and L.A.P., Y.Q.Z., M.W. and Z.P.Z. were responsible for the study inception and protocol design. L.A.P., Y.Q.Z. and B.C. searched literature, selected the studies, and completed the extraction of data. L.A.P. and Y.Q.Z. were involved in analysis the data. Interpretation of the results and drafting of the manuscript was done by L.A.P., M.W., Z.P.Z., Q.W., X.Y.C., Y.Z., and B.X. contributed to revising it critically for intellectual content. B.C., Y.X.W., and L.A.P. were involved in the final approval of the version to be published. All authors reviewed and approved the final manuscript, confirmed that they met the established authorship criteria, and agreed to be accountable for all aspects of the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Peng, L., Zhou, Y., Wang, M. et al. Acceptance of preventive therapy for latent tuberculosis infection in Chinese children and adolescents: a systematic review and meta-analysis. Sci Rep 16, 1471 (2026). https://doi.org/10.1038/s41598-025-31419-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31419-x