Abstract
Aquatic animals are frequently exposed to environmental stressors such as elevated temperatures and various inorganic and organic contaminants. The present investigation examines the role of iron nanoparticles (Fe-NPs) in mitigating the combined effects of cadmium (Cd, 1/10th of LC50 0.69 mg L−1) toxicity and high-temperature stress (34 °C) in Pangasianodon hypophthalmus (striped catfish). Four treatments were designed in triplicate: a control group, a group exposed to Cd and high temperature (Cd + T), and two groups exposed to Cd + T but fed with Fe-NPs at 10 and 15 mg kg−1, respectively. Fe-NPs were biologically synthesized using fish waste (gill). Experimental diets were prepared by incorporating Fe-NPs at 0, 10, and 15 mg kg−1. Dietary supplementation with Fe-NPs significantly improved growth performance under Cd + T stress, including final weight gain %, specific growth rate, protein efficiency ratio, feed conversion ratio, relative feed intake, and daily growth index. Expression levels of growth genes GH and GHR1 in liver tissue were notably upregulated by Fe-NP-fed groups, while MYST expression was downregulated. Furthermore, stress and immune-related markers such as cortisol, HSP70, iNOS, CYP450, Caspase-3a, and TNF-α were significantly reduced in Fe-NP-treated groups compared to Cd + T and control groups, whereas Cd + T exposure alone markedly elevated these markers. Oxidative stress enzymes including catalase (CAT), superoxide dismutase (SOD), and glutathione-S-transferase (GST) in liver and kidney tissues were significantly elevated under Cd + T exposure, but their levels were reduced with dietary Fe-NP supplementation. Additionally, Fe-NPs at 10 and 15 mg kg−1 enhanced acetylcholine esterase activity in brain tissue compared to control and stressors group (Cd + T). Activities of alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), and malate dehydrogenase (MDH) in liver and gill tissues were significantly reduced in Fe-NP-fed groups under stress conditions. Importantly, dietary Fe-NPs also lowered Cd accumulation in various fish tissues. In conclusion, dietary supplementation with Fe-NPs at 10 and 15 mg kg−1 effectively mitigated the combined stress of Cd toxicity and elevated temperature in P. hypophthalmus by modulating gene expression, enhancing physiological performance, and alleviating cellular and metabolic stress.
Similar content being viewed by others
Introduction
Rising temperatures and pollution are becoming increasingly evident across all ecosystems, including aquatic environments1. In the current scenario, abrupt temperature fluctuations driven by climate change, pose significant global threats and challenges. According to IPCC2, the global mean temperature is projected to increase by 1.1 °C to 6.4 °C by the year 2100. At the same time, increasing anthropogenic activities are contributing to significant pollution in aquatic ecosystems. Among the various contaminants, heavy metals are of particular concern, with cadmium (Cd) recognized as one of the most toxic to aquatic organisms, especially fish. Even at low concentrations, cadmium exhibits high toxicity and a strong bioaccumulation potential because fish excrete it very slowly, leading to its buildup in multiple tissues3,4. Major sources of Cd contamination include industrial discharges from mining, metal plating, plastics, phosphate fertilizers, alloy production, nickel-cadmium batteries, dyes, electrical and electronic industries, as well as corrosion-resistant coatings used in aerospace and marine environments4,5. Aquatic organisms are particularly vulnerable to Cd toxicity, especially during sudden exposure events6. Cadmium has no known metabolic or physiological function in most organisms, except for its involvement in carbonic anhydrase activity in certain marine algae7,8. Moreover, Cd induces neurotoxicity by binding to sulfhydryl groups in enzymes, thereby inhibiting their biological activity and disrupting normal life functions in aquatic animals9. In the present study, fish were subjected to combined stress from cadmium and elevated temperature. As poikilothermic animals, fish are highly sensitive to temperature changes, which can drastically alter their physiological functions. Furthermore, the cadmium has a long biological half-life and can be transmitted through water, feed, and environmental exposure. The extent of Cd accumulation in fish tissues depends on several factors, including exposure duration and concentration, fish age and species, dietary mineral and antioxidant content, and overall water quality10.
Nutritional approaches are among the most effective strategies to mitigate metal toxicity and high-temperature stress in fish11,12,13. These methods are practical, easily implementable, and highly effective for stress management in aquaculture systems14,15. In this study, iron nanoparticles (Fe-NPs) were applied as a nutritional intervention. Iron (Fe) is an essential micronutrient that plays a crucial role in metabolism and physiology, including oxygen transport and cellular respiration in fish16. Notably, Fe-NPs have been reported to enhance fish immunity through the NF-κB signaling pathway, involving immune-related genes such as TNFα and Ig17,18. Compared to conventional iron, Fe-NPs offer several advantages, including higher absorption and utilization efficiency due to their nanoscale size, elevated energy levels, increased surface activity, and enhanced ability to penetrate cell membranes19,20,21.
Oxidative stress biomarkers, including superoxide dismutase (SOD), catalase (CAT), and glutathione-S-transferase (GST), serve as critical indicators of oxidative damage arising from an imbalance between reactive oxygen species and antioxidant defenses under multiple stressors such as cadmium exposure and elevated temperature22. Stress-responsive genes HSP70, iNOS, CYP450, Ig, and TNFα are differentially expressed during adverse conditions and contribute to cellular tolerance. Neurotransmitter enzymes also act as sensitive biomarkers for pollution-induced neurotoxicity. Furthermore, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) are widely used markers of tissue injury in fish and typically increase under stress. Digestive enzyme activities provide additional insight into the metabolic and overall health status of aquatic organisms. Cadmium accumulation predominantly occurs in the kidney, gills, liver, muscle, and brain tissues of fish. Elevated temperatures further intensify Cd toxicity, as corroborated by the current study23. Factors such as pH, salinity, organic matter, and seasonal variation also modulate Cd bioaccumulation across different fish tissues24,25,26. In this study, Pangasianodon hypophthalmus was chosen as the experimental species because of its suitability for toxicity assessments, especially under multiple stressors. This species is economically and nutritionally important, with desirable taste qualities and strong potential for diversification in aquaculture. Therefore, the objective of this study was to investigate the role of iron nanoparticles (Fe-NPs) in mitigating the combined effects of cadmium toxicity and high-temperature stress in P. hypophthalmus.
Materials and methods
Experimental animal and design
Pangasianodon hypophthalmus (average weight: 45.50 ± 0.40 g; average length: 12.30 ± 0.25 cm) were used as experimental animals. Healthy fish were harvested from the NIASM farm pond. Prior to the experiment, the fish were acclimatized for two weeks in fiber-reinforced plastic (FRP) tanks with a capacity of 500 L. During the acclimatization period, the fish were fed a pelleted diet containing 30% crude protein. After acclimatization, a total of 144 fish were randomly distributed into 12 rectangular plastic tanks (each with a 150 L capacity), with 12 fish per tank. The experiment was arranged in a completely randomized design (CRD), consisting of four treatments with three replicates each. Details of the experimental treatments are provided in Table 1.
Fish were fed diets supplemented with Fe-NPs at 0, 10, and 15 mg kg−1 twice daily at 9:15 AM and 5:15 PM. Uneaten feed and fecal matter were removed daily by siphoning, and continuous aeration was provided using aerators throughout the experimental period. Water quality parameters were monitored periodically following standard methods described by APHA27. To induce cadmium toxicity, a stock solution of cadmium chloride (100 mg L−1) was prepared (Hi-Media, Mumbai, Maharashtra, India) as per the method described by Ganeshkar et al.28. In this study, the LC50 value for Cd under high-temperature stress (Cd + T) was determined to be 6.91 mg L−1, and 1/10th of this concentration (0.69 mg L−1) was used for experimental exposure. Water temperature was maintained at 34 °C using a thermostatic rod heater, as described by Kumar et al.29. The experimental diets were formulated as isonitrogenous (35% crude protein) and isocaloric (356.8 ± 1.21 kcal/100 g), as detailed in Table 2. The feed ingredients included fishmeal, groundnut meal, soybean meal, and wheat flour. After heat processing of the base ingredients, heat-sensitive nutrients such as cod liver oil, lecithin, iron nano-particles (Fe-NPs) and vitamin C were added. A iron free mineral mixture was used for feed formulation. For formulation of the pelleted diet, a manually prepared vitamin and mineral mixture was used. The dough was mixed properly and was pelleted, air dried and kept in a hot air oven at 60 °C until dry and was subsequently stored at 4 °C until required for feeding. The proximate composition of all the experimental diets and fish were analyzed as per the methods of AOAC30. Crude protein was measured based on nitrogen content, ether extract (EE) was determined via solvent extraction, and ash content was estimated using a muffle furnace at 550 °C. Carbohydrate content was calculated using the formula:
Gross energy content of the feed was calculated using the Halver method31. The feed formulation based on iron nanoparticles (Fe-NPs) were used in our previous study32. A detailed composition of the experimental diets is presented in Table 2.
Iron nanoparticles (Fe-NPs) synthesis using green approach
Preparation of fish tissue extract
Fe-NPs were synthesized using fish waste, specifically gill tissue. The gill tissues were first cut into small pieces and thoroughly washed with running water to remove blood. The cleaned tissues were then homogenized using a tissue homogenizer (Tissue Master Homogenizer, Kennesaw, GA), and the homogenate was centrifuged at 5,000–6,000 rpm to collect the supernatant. The resulting gill extract was subsequently filtered using Whatman filter paper with a pore size of 0.45 µm33.
Preparation and characterization of iron nanoparticles (Fe-NPs)
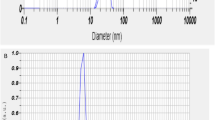
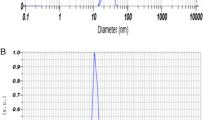
Ferrous sulfate (0.4 M) was mixed with the prepared gill extract and stirred at room temperature for 30 min. The mixture was then heated on a magnetic stirrer at 60 °C for 2.5 to 4 h, during which 0.8 M NaOH was added dropwise until a reddish-brown coloration developed, indicating the formation of iron nanoparticles (Fe-NPs). Once the reddish-brown color appeared, the solution was analyzed using a UV-Vis spectrophotometer in the range of 216–600 nm, which revealed a strong absorption peak at 250 nm, confirming the presence of Fe-NPs. The solution was then centrifuged, and the resulting pellet was washed three times with distilled water to remove any impurities. The purified pellet was dried using a concentrator, yielding Fe-NPs in the form of a reddish-brown powder. For characterization, the dried nanoparticles were re-dispersed in Milli-Q water, and their particle size and zeta potential were measured using a particle size analyzer (Litesizer 500, Anton Paar, Austria)21,32. The mean particle size and zeta potential were recorded as 130.17 nm and + 53.3 mV, respectively, as shown in Fig. 1A,B.
(A,B) Size of iron nanoparticles (Fe-NPs): 130 nm B. Zeta potential 53.7 mV.
Tissue homogenate preparation and blood collection
Under aseptic conditions, tissues including the gill, intestine, muscle, brain, liver, and kidney of P. hypophthalmus were collected from fish anesthetized with clove oil (50 µL L−1). The tissues were homogenized using an Omni Tissue Master Homogenizer (Kennesaw, GA, USA) in a cold 5% sucrose solution (w/v; 0.25 M) containing 1 mM EDTA for subsequent enzyme analysis. Muscle (MYST) and liver tissues were immediately snap-frozen in liquid nitrogen for gene expression analysis of HSP70, iNOS, TNFα, GHR1, Ig, GH, CYP450, and Cas 3a. The homogenates were centrifuged at 5,000 × g for 15 min at 4 °C using a refrigerated centrifuge (Eppendorf AG, 5430R, Hamburg, Germany), and the resulting supernatants were stored at − 20 °C for further biochemical analysis. Additionally, the blood was collected from the caudal vein for serum, from nine fish from each treatment without anticoagulant and allowed to clot for 2 h followed by collection of serum with a micropipette and stored at -22 °C until use.
Protein estimation
Protein content in different tissues (gill, intestine, muscle, brain, liver, and kidney) was determined following the method of Lowry et al.34, using freshly prepared 1 N Folin-Ciocalteu reagent and alkaline CuSO₄. For this, 0.1 mL of tissue homogenate was treated with 1 mL of 10% trichloroacetic acid (TCA) to precipitate proteins. The mixture was centrifuged at 5,000 rpm for 20 min, after which the supernatant was discarded. This residue was dissolved in 0.5 mL of 0.1 N NaOH, and 0.1 mL of the resulting solution was used for analysis. The absorbance was recorded at 660 nm.
Analysis of physiological indices
Growth performance
The growth performance of the fish was evaluated using our laboratory’s standardized methodologies, including weight gain percentage, specific growth rate (SGR), protein efficiency ratio (PER), feed conversion ratio (FCR), daily growth index (DGI), and relative feed intake (RFI). Weight measurements were recorded at 20-day intervals28,35.
Biochemical and gene expression analysis
Cortisol
Cortisol levels in the serum were measured using a commercially available ELISA kit (Cortisol EIA kit, catalogue no. 500360, Cayman Chemicals, USA). The assay was conducted following the instructions provided with the kit. An ELISA plate reader from Biotek India Pvt. Ltd. was used to perform the measurements, ensuring accurate detection of cortisol levels in the serum samples.
Antioxidant enzyme activities
Catalase assay (EC 1.11.1.6)
Catalase (CAT) activity in liver and kidney tissues was determined following the method of Takahara et al.36. The reaction mixture contained 1.0 mL of freshly prepared hydrogen peroxide solution (Cat. No. 1.93.007.0521, Merck Life Science, Vikhroli, Mumbai, India), 50 µL of tissue homogenate and 2.45 mL of 50 mM phosphate buffer (pH 7). After mixing, the decrease in absorbance at 240 nm was recorded for three minutes to measure catalase activity.
Superoxide dismutase assay (EC 1.15.1.1)
Superoxide dismutase (SOD) activity in liver and kidney tissues was analyzed following the procedure developed by Misra and Fridovich37. The reaction mixture was prepared by combining 0.5 mL of freshly prepared epinephrine (Cat. No. 1236970, Merck Life Science, Vikhroli, Mumbai, India), 1.5 mL of phosphate buffer, and 50 µL of tissue homogenate. The mixture was thoroughly mixed, and the absorbance was measured immediately at 480 nm for three minutes using a UV spectrophotometer (Shimadzu (Asia Pacific) Pte Ltd, Cintech IV, Science Park I, Singapore-118264).
Glutathione-S-transferase assay (EC 2.5.1.18)
Glutathione S-transferase (GST) activity in liver and kidney tissues was determined using a spectrophotometric method, with S-2,4-dinitrophenyl glutathione (CDNB) (Cat. No. S101915, Merck Life Science, Vikhroli, Mumbai, India) as the substrate, following the protocol established by Habing et al.38. The reaction involves the formation of an adduct between glutathione (GSH) and S-2,4-dinitrophenyl glutathione in the presence of CDNB. The increase in absorbance at 340 nm, relative to a blank, was used to quantify the production of this adduct, indicating GST activity.
Neurotransmitter enzyme activities
Acetylcholinesterase (EC 3.1.1.7) activity in brain tissue was assessed using a modified Hestrin method as described by Augustinsson39. In this procedure, 0.2 mL of the sample was mixed with 1 mL each of acetylcholine buffer and phosphate buffer, and the mixture was incubated at 37 °C for 30 min. Following incubation, alkaline hydroxylamine, HCl, and ferric chloride (Himedia, Cat. No. GRM 1379, Thane, Mumbai, India) were added. The enzymatic activity was determined by measuring the absorbance at 540 nm.
Aspartate aminotransferase (AST) and Alanine aminotransferase (ALT), lactate dehydrogenase (LDH), and malate dehydrogenase (MDH)
AST (E.C.2.6.1.1) and ALT (E.C.2.6.1.2) activities in liver and gill tissues were determined following the method described by Wootton40. The reaction mixture consisted of sodium hydroxide (1 N), potassium dihydrogen phosphate, dipotassium hydrogen phosphate, and 2,4-dinitrophenyl hydrazine. For the ALT assay, α-ketoglutarate and DL-alanine were used as substrates, while α-ketoglutarate and DL-aspartic acid were used in the AST assay. The tissue homogenate and substrate were mixed and incubated at 37 °C for 1 h, after which DNPH was added and the absorbance at 540 nm was measured. LDH activity in liver and gill tissues was evaluated using the Wroblewski and Ladue41 method. A reaction mixture was prepared by adding sodium dihydrogen phosphate (0.1 M) and disodium hydrogen phosphate to a phosphate buffer, followed by the addition of freshly prepared NADH and sodium pyruvate. After incubating the mixture with the sample/enzyme extract for 20 min, absorbance was measured at 320 nm. Similarly, MDH activity was assessed following Ochoa’s42 method, with the reaction mixture remaining the same but substituting sodium pyruvate with oxaloacetate as the substrate.
RNA isolation and quantification and cDNA synthesis and quantitative PCR
Total RNA was extracted from the liver tissue of P. hypophthalmus using the TRIzol method (Catalogue no. 15596018; Invitrogen™ Life Technologies, Carlsbad, CA, USA). Liver samples were homogenized in liquid nitrogen, followed by phase separation with chloroform. For each treatment, nine fish were used; samples from three fish within a replicate were pooled, yielding three biological replicates for RNA extraction. RNA was separated by centrifugation, dissolved in nuclease-free water, and stored at − 80 °C. Integrity was verified on a 1% agarose gel, and concentration was determined using a NanoDrop spectrophotometer (Thermo Scientific, Wilmington, DE, USA). For cDNA synthesis, residual DNA was removed with DNase I, and first-strand cDNA was generated using the RevertAid First Strand cDNA Synthesis Kit (Catalog no. K1622, Thermo Fisher Scientific Baltics UAB, Lithuania). Each reaction contained 100 ng RNA in 12 µl with 15 pmol oligo(dT) primers, incubated at 65 °C for 5 min and chilled on ice. Subsequently, 1 µl RiboLock RNase inhibitor (20 U/µl), 2 µl 10 mM dNTP mix, and 1 µl reverse transcriptase were added. The mixture was centrifuged briefly, incubated at 60 °C for 42 min, then at 70 °C for 5 min, and stored at − 20 °C. β-actin served as the reference gene to confirm cDNA synthesis. Quantitative real-time PCR was performed on a QuantStudio 5 system (Applied Biosystems, Thermo Fisher Scientific) using SYBR Green (Catalog no. A25742, Bio-Rad, Lithuania) and gene-specific primers. Cycling conditions included initial denaturation at 95 °C for 10 min, followed by 39 cycles of 95 °C for 15 s and 60 °C for 1 min, following Pfaffl43. Primer sequences are provided in Table 3.
Cadmium analysis in fish tissues and experimental water
Cadmium concentrations in water and fish tissues (liver, muscle, gill, brain, and kidney) were analyzed across all treatment groups. Water samples were filtered through 0.45 μm membranes and acidified with 100 µl of concentrated HNO₃ (69%, Himedia Laboratory Pvt. Ltd., Mumbai, India). Tissue samples were digested in a microwave reaction system (Multiwave PRO, Anton Paar GmbH, Austria) using a 5:1 mixture of HNO₃ and H2O2. After digestion, the solutions were cooled, filtered through 0.45 μm Whatman paper, and diluted to a final volume of 50 ml. Cadmium levels were then quantified using Inductively Coupled Plasma Mass Spectrometry (ICP-MS, Agilent 7700 series, Agilent Technologies, USA) following the protocol of Kumar et al.44,45.
Statistics
Data analysis was performed using the Statistical Package for the Social Sciences (SPSS) version 16 software. To assess the normality of the data, the Shapiro-Wilk test was employed, while the Levene test was used to examine the homogeneity of variances. A One-Way Analysis of Variance (ANOVA) was conducted, followed by Duncan’s multiple range test for post-hoc comparisons. The significance threshold for all tests was set at p < 0.05.
Results and discussion
Growth performance
In the present investigation, the growth performance of fish was evaluated based on final weight gain percentage, feed conversion ratio (FCR), specific growth rate (SGR), protein efficiency ratio (PER), daily growth index (DGI), and relative feed intake (RFI). The fish were exposed to concurrently with cadmium toxicity and high-temperature stress for 40 days. Fish were fed either a control diet or Fe-NPs-supplemented diet to reduce the impact of Cd + T stress. The results indicated that supplementation of Fe-NPs at 10 and 15 mg kg− 1 in the diet with the stressors (Cd + T) significantly (p = 0.0024) enhanced weight gain percentage, SGR (p = 0.001), PER (p = 0.0015), DGI (p = 0.001), and RFI (p = 0.012) compared to the control group and the group treated with Cd + T stress alone. Moreover, the group exposed to both Cd + T stress and fed the control diet exhibited the lowest growth performance. Additionally, the FCR was significantly (p = 0.0014) reduced with Fe-NPs supplementation at 10 and 15 mg kg− 1 in the diet under stress conditions (Table 4).
In the present investigation, Fe-NPs were incorporated into the diet at 10 and 15 mg kg−1 to evaluate their effectiveness in reducing the adverse effects of low-dose cadmium exposure combined with high-temperature stress (34 °C). The results demonstrated substantial improvements in weight gain, FCR, PER, SGR, RFI, and DGI in P. hypophthalmus subjected to Cd + T stress for 40 days following Fe-NP supplementation. The growth-promoting effects of Fe-NPs are likely attributed to their role in maintaining iron homeostasis within the organism, thereby enhancing growth performance under combined Cd + T stress21,46. Furthermore, the smaller particle size of Fe-NPs may contribute to their efficient utilization when incorporated into the diet47,48,49,50. The higher bioavailability and improved absorption of Fe-NPs further support their positive influence on growth in fish, both under control and stressful conditions21,32. In contrast, concurrent exposure to Cd + T resulted in reduced growth and feed utilization. However, Fe-NPs supply the essential nutrients through the bloodstream, providing energy and acting as enzyme cofactors that enhance nutrient breakdown and absorption51. The supplementation of Fe-NPs at 10 and 15 mg kg− 1 also improved the daily growth index and relative feed intake, underscoring their role in supporting growth attributes.
Effect of Fe-NPs on growth performing genes (GH, GHR1 and MYST)
The expression of growth-promoting genes, such as GH and GHR1 in liver tissue and MYST in muscle tissue of P. hypophthalmus reared under concurrent exposure to cadmium and high-temperature stress for 40 days, is presented in Fig. 2A,B. The results showed that GH (p = 0.0016) and GHR1 (p = 0.0021) were significantly upregulated by dietary Fe-NPs at 10 mg kg− 1 in P. hypophthalmus under Cd + T stress, compared to the control and stressor groups. In contrast, MYST was substantially (p = 0.0017) downregulated by dietary Fe-NPs at both 10 and 15 mg kg− 1 in fish exposed to Cd + T stress, relative to the control and stressor groups. Additionally, the group exposed to Cd and high temperature and fed with the control diet exhibited significantly downregulated GH and GHR1 genes, while the MYST gene was upregulated in P. hypophthalmus throughout 40-day experimental period.
(A,B) Potential role of Fe-NPs diets on gene expression of GH, GHR1 and MYST in liver tissue in fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
The regulation of GH, GHR1, and MYST genes was significantly downregulated in fish exposed to Cd + T stress and fed with the control diet, indicating that the combined exposure to cadmium and high temperature negatively impacts the gene regulatory mechanisms associated with growth enhancement. However, supplementation with Fe-NPs at 10 and 15 mg kg− 1 diet resulted in the upregulation of GH and GHR1 gene expression, while MYST expression was downregulated. This effect could be attributed to the role of Fe-NPs in reducing myoblast activity, which promotes terminal differentiation and muscle fiber enlargement52. Additionally, Fe-NPs may facilitate the binding of GH to its receptor in liver cells, stimulating the release and synthesis of IGF, further enhancing growth. Fe-NPs also play a crucial role in the secretion and regulation of IGF, which directly influences growth performance via the central nervous system of fish, whether reared under normal or stressed conditions53,54. Ghrelin, which regulates GH release from the pituitary gland, is also essential in this process. It stimulates the growth hormone-releasing hormone and somatostatin axis, leading to increased IGF production in the liver55.
Effect of Fe-NPs on cortisol, HSP 70 and iNOS
Cortisol is the primary stress hormone, and its levels increase during stress conditions. In this study, cortisol levels were significantly elevated (p = 0.0015) in the group exposed to concurrent Cd + T stress and fed with the control diet. However, dietary supplementation with Fe-NPs at 15 mg kg− 1 resulted in a significant reduction in cortisol levels, followed by a reduction in the group fed with Fe-NPs at 10 mg kg− 1, compared to the control and Cd + T groups (Fig. 3A). Additionally, the gene expression of HSP 70 and iNOS in liver tissue of P. hypophthalmus reared under control and combined Cd + T stress for 40 days is presented in Fig. 3B. Gene expression of HSP 70 (p = 0.0013) and iNOS (p = 0.001) was significantly downregulated by dietary Fe-NPs at 10 and 15 mg kg-1 in the presence of stressors, compared to the control and Cd + T groups. In contrast, upregulation of HSP 70 and iNOS was observed in the group exposed to Cd + T stress and fed the control diet, compared to both the control and Fe-NPs supplemented groups.
(A,B) Potential role of Fe-NPs diets on cortisol and gene expressions of HSP 70 and iNOS in liver tissue in fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
The overexpression of HSP70 and iNOS gene expression under Cd + T stress may be attributed to the activation of heat shock promoters by heat shock factors (HSF), triggered by high temperature and cadmium toxicity56. Moreover, both stressors can reduce the methylation levels at the HSF binding motif in the HSP70 promoter, resulting in its hypomethylation and subsequent overexpression57. The iNOS gene, on the other hand, is an early inflammatory marker and plays a central role in initiating cellular inflammatory responses58. Its expression helps regulate cellular metabolism during concurrent exposure to Cd + T. Elevated cortisol levels under Cd + T stress conditions may be due to disruptions in the interrenal cells of the kidney within the pituitary-adrenal axis, impairing corticosteroidogenesis and leading to excessive cortisol production59. However, dietary supplementation with Fe-NPs at 10 and 15 mg kg−1 significantly downregulated HSP70 expression, likely by protecting against proteasomal degradation, preventing protein aggregation, aiding the degradation of denatured proteins, and supporting the correct folding of misfolded proteins60,61. Furthermore, Fe-NPs may influence HSP transcription factor activity by reducing heat shock element binding, thereby decreasing HSP70 expression62,63. A strong interrelationship exists between iron metabolism and iNOS gene expression. Iron nanoparticles have been shown to reduce oxidative stress by preventing the accumulation of reactive oxygen species via a NOS-dependent mechanism64. Additionally, iron can promote cellular hypertrophy by upregulating anti-apoptotic proteins such as Bcl-2 and survivin, as well as hypertrophic agents like endothelin-1 (ET-1) and leukemia inhibitory factor (LIF), all of which contribute to reduced H2O2-induced necrosis65,66.
Effect of Fe-NPs on CYP 450, Cas 3a, Ig and TNFα
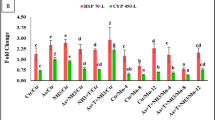
The expression patterns of CYP450, Caspase 3a (Cas 3a), Immunoglobulin (Ig), and Tumor Necrosis Factor-alpha (TNFα) genes in the liver of Pangasianodon hypophthalmus reared under control conditions and concurrent exposure to cadmium and high temperature (Cd + T) stress for 40 days are presented in Fig. 4A,B. The gene expression of CYP450 was significantly downregulated (p = 0.0011) with dietary supplementation of Fe-NPs at 10 mg kg−1, followed by 15 mg kg−1, under stress conditions when compared to the control and Cd + T groups. Similarly, Cas 3a expression was also markedly reduced (p = 0.0025) in the groups supplemented with Fe-NPs at 10 and 15 mg kg−1 compared to the control and stress groups (Fig. 4A). Furthermore, Ig gene expression was significantly upregulated (p = 0.001) by Fe-NPs at 10 mg kg−1 diet when compared to other treatment groups. Meanwhile, TNFα expression was strongly downregulated (p = 0.00016) in fish fed Fe-NPs at 10 and 15 mg kg−1 diet under Cd + T stress compared to the control and stress-exposed groups. These findings clearly indicate that concurrent exposure to cadmium and elevated temperature significantly alters the regulation of genes associated with xenobiotic metabolism, apoptosis, immune response, and inflammation in P. hypophthalmus, while dietary Fe-NPs mitigate these effects in a dose-dependent manner.
(A,B) Potential role of Fe-NPs diets on gene expressions of CYP 450, CAS 3a, Ig and TNFα in liver tissue in fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
In this study, concurrent exposure to cadmium (Cd) and elevated temperature (34 °C) stress led to a marked upregulation of CYP450 gene expression, likely due to the activation of oxidative stress-related kinases and the consequent generation of reactive oxygen species (ROS), which in turn triggered apoptosis and elevated inflammatory cytokine levels67. The increased transcription of CYP450 under Cd + T stress also suggests its involvement in Cd detoxification processes, during which excessive ROS are produced, intensifying oxidative stress68. However, dietary supplementation with Fe-NPs significantly downregulated CYP450 expression, indicating their role in modulating oxidative damage induced by Cd + T stress69. Cd exposure also disrupts cellular homeostasis by promoting ROS accumulation, which affects intracellular Ca²⁺ mobilization, leads to mitochondrial membrane depolarization, and causes the release of cytochrome c into the cytoplasm, ultimately activating caspase-3 and initiating apoptosis70. Notably, Fe-NPs appear to mitigate these effects through the involvement of protein disulfide isomerase (PDI), a multifunctional enzyme capable of regulating apoptosis via caspase pathways71, and by binding transition metals such as copper and zinc at active thiol sites72. Cd-induced stress also activates death receptor pathways, particularly via TNFα, enhancing its expression and contributing to liver inflammation and programmed cell death in fish73,74. Conversely, dietary Fe-NPs suppressed TNFα expression, potentially through their regulatory influence on ferritin levels within macrophages75, thereby exerting anti-inflammatory effects. Additionally, Cd + T stress significantly reduced total immunoglobulin (Ig) gene expression, whereas Fe-NP supplementation was able to restore Ig expression levels, suggesting an immunoprotective role under combined metal and thermal stress.
Effect of Fe-NPs on catalase, SOD, GST and AChE
The activities of catalase (CAT), superoxide dismutase (SOD), and glutathione S-transferase (GST) in liver and kidney tissues, along with acetylcholinesterase (AChE) activity in brain tissue of Pangasianodon hypophthalmus reared under control conditions and concurrent exposure to cadmium and high temperature (Cd + T) for 40 days, are presented in Figs. 5 and 6A,B. CAT activity in both liver (p = 0.001) and kidney (p = 0.001) tissues was significantly elevated in the Cd + T group fed the control diet, compared to both the control and Fe-NP-supplemented groups. Interestingly, dietary supplementation with Fe-NPs at 10 mg kg−1 diet, followed by 15 mg kg−1, markedly reduced CAT activity in these tissues. Similarly, SOD activity in the liver and kidney was substantially lowered in groups supplemented with Fe-NPs at both 10 and 15 mg kg−1 diets when compared to the control and Cd + T groups (Fig. 5A,B). Furthermore, GST activity in the liver (p = 0.0011) and kidney (p = 0.012) was significantly reduced in the group supplemented with 10 mg kg−1 Fe-NPs relative to other groups. In contrast, concurrent exposure to Cd + T stress in fish fed the control diet significantly elevated the activities of CAT, SOD, and GST in both liver and kidney tissues compared to the Fe-NP-supplemented and control groups. Regarding neurological effects, AChE activity in brain tissue was significantly inhibited (p = 0.00015) by combined Cd and high-temperature stress. However, supplementation with Fe-NPs at 10 mg kg−1, followed by 15 mg kg−1, restored AChE activity in stressed fish, showing higher levels compared to both the control and Cd + T only groups (Fig. 6A,B).
(A,B) Potential role of Fe-NPs diets on CAT, and SOD in liver and kidney tissues of fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
(A,B) Potential role of Fe-NPs diets on GST in liver and kidney tissues and AChE in brain of fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
Concurrent exposure to cadmium and elevated temperature (Cd + T) triggered oxidative stress in P. hypophthalmus, as evidenced by elevated activities of antioxidant enzymes such as CAT, SOD, and GST. This response indicates an overproduction of reactive oxygen species (ROS), which can lead to cellular damage76,77. Under such stress, the fish initiate protective mechanisms to neutralize superoxide (O2−) radicals; however, these mechanisms can be overwhelmed. Supplementation with Fe-NPs significantly modulated this response by reducing the activities of CAT, SOD, and GST, suggesting a protective role in oxidative stress mitigation21,32. Fe-NPs exhibit multiple biological functions, including serving as components of the antioxidant defense system. They are likely to enhance the activity of enzymes like SOD and GPx, thereby preventing excessive free radical generation and lipid peroxidation78,79. Catalase, a heme-containing enzyme, works synergistically with SOD to neutralize cellular ROS and improve the antioxidant capacity of fish cells80. Additionally, AChE activity, which was markedly inhibited by Cd + T exposure, reflects oxidative damage to critical cellular structures such as the endoplasmic reticulum, plasma membrane, and Golgi apparatus81. Interestingly, Fe-NPs were able to restore AChE activity under combined stress conditions, likely by preserving neuronal integrity and limiting oxidative damage21.
Effect of Fe-NPs on ALT, AST, LDH and MDH
Figure 7A,B and 8 summarize the activities of ALT, AST, LDH, and MDH in the liver and gill tissues of Pangasianodon hypophthalmus reared under control conditions and subjected to combined cadmium and high temperature (Cd + T) stress for 40 days. The activity of ALT in the liver was significantly reduced (p = 0.013) in fish supplemented with Fe-NPs at 10 mg kg−1 diet. Similarly, ALT activity in the gills was significantly lowered (p = 0.011) in groups receiving Fe-NPs at both 10 and 15 mg kg−1 diets under stress conditions, compared to the control and Cd + T groups. AST activity in both liver (p = 0.017) and gill (p = 0.001) tissues was markedly elevated in fish exposed to Cd + T stress and fed a control diet. However, supplementation with Fe-NPs at 10 mg kg−1 significantly reduced AST activity under stress conditions compared to other groups (Fig. 7A–B). In addition, LDH activity in liver (p = 0.0015) and gill (p = 0.0011) tissues was significantly decreased in the group fed Fe-NPs at 10 mg kg−1 diet under stress conditions compared to the control and Cd + T groups. Similarly, MDH activity in the liver was significantly reduced with Fe-NPs supplementation at 10 mg kg−1 diet. In gill tissue, MDH activity was significantly reduced (p = 0.014) in the group fed 10 mg kg−1 Fe-NPs, followed by the 15 mg kg−1 Fe-NPs group, compared to control and Cd + T-exposed groups. Notably, both LDH and MDH activities were significantly elevated in fish concurrently exposed to Cd + T and fed a control diet, highlighting the detrimental effect of the combined stress (Fig. 8).
(A,B) Potential role of Fe-NPs diets on ALT and AST in liver and gill tissues of fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
(A,B) Potential role of Fe-NPs diets on LDH and MDH in liver and gill tissues of fish reared under control or cadmium and high temperature stress for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
The activities of protein metabolic enzymes such as ALT and AST in liver and gill tissues were elevated under concurrent exposure to cadmium and high temperature (Cd + T) stress. This elevation may be attributed to disruptions in amino acid metabolism82. Increased levels of ALT and AST are recognized indicators of tissue damage, particularly in the liver83,84. Additionally, the elevated activities of LDH and MDH observed under Cd + T stress suggest increased physiological stress in fish, often associated with reduced oxygen availability85. Under such stress, anaerobic metabolic pathways become more active, leading to elevated lactate concentrations in tissues85,86. The increased LDH activity may also reflect metabolic disturbances and reduce nutrient conversion efficiency. Interestingly, dietary supplementation with Fe-NPs significantly reduced ALT and AST activities, suggesting a protective role against Cd + T induced stress. This reduction could be due to the ability of Fe-NPs to mitigate cellular degeneration and necrosis87. Iron plays an essential role in maintaining fish health and enhancing resistance to environmental stressors88. Moreover, Fe-NPs improve antioxidant capacity, which helps scavenge harmful free radicals and alleviates oxidative damage, thereby protecting liver function and enhancing immune response in fish exposed to Cd + T stress89.
Effect of Fe-NPs on bioaccumulation of cd in water and fish tissues
The concentration of cadmium (Cd) in the experimental water samples ranged from 0.2 µg L−1 in the control group to 1480 µg L−1 in the group concurrently exposed to Cd and high temperature (Cd + T) and fed a control diet. In comparison, the Cd concentrations were reduced to 236 µg L−1 and 210 µg L−1 in the groups supplemented with Fe-NPs at 10 and 15 mg kg−1 diet, respectively, under Cd + T stress. Similarly, Cd bioaccumulation in muscle, liver, gill, kidney, and brain tissues was assessed at the end of the 40-day experimental period. The highest Cd accumulation was observed in the kidney (14.11 mg kg−1) and liver (12.52 mg kg−1), followed by gill (6.15 mg kg−1), muscle (1.35 mg kg−1), and brain (0.65 mg kg−1) tissues. Notably, dietary supplementation with Fe-NPs at both 10 and 15 mg kg−1 significantly reduced Cd bioaccumulation across all examined tissues (Table 5).
Cadmium (Cd) bioaccumulation in various fish tissues such asliver, gill, muscle, kidney, and brain reflect the extent of metal bioavailability in the aquatic environment90. In the present study, Cd accumulation was notably higher in the liver and kidney, indicating that the rate of Cd uptake exceeded its elimination, consistent with earlier findings91. This accumulation pattern also corresponded with reduced digestive enzyme activity in the intestine, suggesting that Cd toxicity impairs gastrointestinal function and results in a “spill-over” effect, thereby exacerbating systemic toxicity92. Cd uptake in gill tissue occurs primarily via passive diffusion and subsequent binding to metallothionein-like proteins (MTLPs), which serve a protective role93. Furthermore, fish may detoxify Cd through conversion into less biologically active forms via endogenous detoxification pathways94. Remarkably, dietary supplementation with Fe-NPs effectively reduced Cd accumulation across all tissues, highlighting their potential role in enhancing metal detoxification. To the best of our knowledge, this is the first report demonstrating a significant reduction in Cd bioaccumulation in multiple tissues of fish through the dietary inclusion of Fe-NPs.
Conclusion
The present investigation addresses pressing environmental concerns such as cadmium pollution and high temperature stress. Dietary supplementation with iron-nanoparticles (Fe-NPs) showed promising results for mitigation of cadmium and high temperature stress in fish. Fe-NPs containing diets also modulate stress-responsive gene expression, thereby enhancing the resilience of fish to these environmental challenges. Specifically, Fe-NPs supplementation at 10 mg kg−1 was effective in promoting cadmium detoxification across various fish tissues. Moreover, Fe-NPs at this level significantly improved the expression of genes associated with growth performance, antioxidant defense, immune response, and apoptosis regulation. Collectively, these findings highlight the potential of Fe-NPs supplementation as a strategic intervention to bolster fish health and performance under the compounded stressors of pollution and climate change.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- APHA:
-
American Public Health Association
- AOAC:
-
Association of Official Agricultural Chemist
- Cd:
-
Cadmium
- CMC:
-
Carboxymethyl cellulose
- CAS 3a :
-
Caspase 3a
- CAT:
-
Catalase
- CP:
-
Crude protein
- DGI:
-
Daily growth index
- ELISA:
-
Enzyme-linked immunosorbent assay
- EE:
-
Ether extract
- FCR:
-
Feed conversion ratio
- FAO:
-
Food and Agriculture Organization
- GPx:
-
Glutathione peroxidase
- GHR1 :
-
Growth hormone regulator 1
- GH :
-
Growth hormone
- HSP70 :
-
Heat shock protein
- T:
-
High temperature
- iNOS :
-
Inducible nitric oxide synthase
- ICPMS:
-
Inductively Coupled Plasma Mass Spectrometry
- Fe-NPs:
-
Iron nanoparticles
- Fe:
-
Iron
- LC50 :
-
Lethal concentration
- MYST:
-
Myostatin
- NFkB:
-
Nuclear factor kappa B
- PER:
-
Protein efficiency ratio
- RFI:
-
Relative feed intake
- RAC:
-
Research Advisory Committee
- SGR:
-
Specific growth rate
- SOD:
-
Superoxide dismutase
- USFDA:
-
United States Food and Drug Administration
- TAN:
-
Total ammonia nitrogen
- TNFα :
-
Tumor necrosis factor
- WHO:
-
World Health Organization
References
Kumar, N., Chandan, N. K., Bhushan, S., Singh, D. K. & Kumar, S. Health risk assessment and metal contamination in fish, water and soil sediments in the East Kolkata Wetlands, India, Ramsar site. Sci. Rep. 13(1), 1546 (2024).
IPCC. in Climate Change 2013: the Physical Science Basis. Contribution of Working Group I To the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Stocker. 1535 (eds Stocker, T. F. D. et al.) (Cambridge University Press, 2013).
Paul, S. et al. Evaluation of water quality and toxicity after exposure of lead nitrate in freshwater fish, major source of water pollution. Egypt. J. Aquat. Res. 45(4), 345–351 (2019).
Mashhadikhan, S., Amooghin, A. E., Sanaeepur, H. & Shirazi, M. M. A. A critical review on cadmium recovery from wastewater towards environmental sustainability. Desalination 535, 115815 (2022).
Othmani, A. et al. A comprehensive review on green perspectives of electrocoagulation integrated with advanced processes for effective pollutants removal from water environment. Environ. Res. 215(1), 114294 (2022).
Abalaka, S. E. et al. Toxicopathological effects of acute cadmium chloride exposure of African Catfish, Clarias gariepinus. Oxicol. Environ. Health Sci. 14, 25–32 (2021).
Genchi, G., Sinicropi, M. S., Lauria, G., Carocci, A. & Catalano, A. The effects of cadmium toxicity. Int. J. Environ. Res. Public. Health. 17(11), 3782 (2020).
Zhong, W. et al. Effect of 24-epibrassinolide on reactive oxygen species and antioxidative defense systems in tall fescue plants under lead stress. Ecotoxicol. Environ. Saf. 187, 109831 (2020).
Liu, Y. et al. Toxic effects of cadmium on fish. Toxics 10(10), 622 (2022).
Das, S., Kar, I. & Kumar Patra, A. Cadmium induced bioaccumulation, histopathology, gene regulation in fish and its amelioration-A review. J. Trace Elem. Med. Biol. 79, 127202 (2023).
Abdel-Tawwab et al. The protective efficacy of dual dietary Rosemary plus cinnamon mix against lead nitrate-induced immune suppression, genotoxicity, and oxidant/antioxidant status in nile tilapia fingerlings. Aquacult. Int. 32, 4009–4029 (2024).
Hamed, H. S. et al. Effect of dietary Moringa Oleifera leaves nanoparticles on growth Performance, Physiological, immunological Responses, and liver antioxidant biomarkers in nile tilapia (Oreochromis niloticus) against zinc oxide nanoparticles toxicity. Fishes 7, 360 (2022).
Kumar, N., Singh, D. K., Bhushan, S. & Jamwal, A. Mitigating multiple stresses in Pangasianodon hypophthalmus with a novel dietary mixture of selenium nanoparticles and Omega-3-fatty acid. Sci. Rep. 11(1), 19429 (2021).
Elabd, H. et al. Nano-Curcumin/Chitosan modulates Growth, Biochemical, Immune, and antioxidative Profiles, and the expression of related genes in nile tilapia, Oreochromis niloticus. Fishes 8, 333 (2023).
Elabd, H. et al. Dietary deacetylated Chitin nanoparticles confer protection against Diazinon toxicity in male African catfish: evaluation of immune-biochemical, antioxidant, and reproductive profiles. Fish. Physiol. Biochem. 51, 32 (2025).
National Research Council (NRC). Nutrient Requirements of Fish and Shrimp (National Academy, 2011).
Guo, Y-L. et al. The decreased growth performance and impaired immune function and structural integrity by dietary iron deficiency or excess are associated with TOR, NF-κB, p38MAPK, Nrf2 and MLCK signaling in head kidney, spleen and skin of grass carp (Ctenopharyngodon idella). Fish. Shellfish Immunol. 65, 145–168 (2017).
Guo, Y. L. et al. The impaired immune function and structural integrity by dietary iron deficiency or excess in gill of fish after infection with flavobacterium columnare: regulation of NF-κB, TOR, JNK, p38MAPK, Nrf2 and MLCK signalling. Fish. Shellfish Immunol. 74, 593–608 (2018).
Mahboub, H. H. et al. Adsorptivity of mercury on magnetite nano-particles and their influences on growth, economical, hemato-biochemical, histological parameters and bioaccumulation in nile tilapia (Oreochromis niloticus). Aquat. Toxicol. 235, 105828 (2021).
Wang, H. et al. Comparison of copper bioavailability in copper-methionine, nano-copper oxide and copper sulfate additives in the diet of Russian sturgeon acipenser gueldenstaedtii. Aquaculture 482, 146–154 (2018).
Kumar, N., Thorat, S. T., Gunaware, M. A., Kumar, P. & Reddy, K. S. Unraveling gene regulation mechanisms in fish: insights into multistress responses and mitigation through iron nanoparticles. Front. Immunol. Comp. Immunol. 15–2024. https://doi.org/10.3389/fimmu.2024.1410150 (2024).
Hayyan, M., Hashim, M. A. & AlNashef, I. M. Superoxide ion: generation and chemical implications. Chem. Rev. 116(5), 3029–3085 (2016).
Xie, D. et al. Elevated temperature as a dominant driver to aggravate cadmium toxicity: investigations through toxicokinetics and omics. J. Hazard. Mater. 474, 134789 (2024).
Kumar, N. et al. Immuno-protective role of biologically synthesized dietary selenium nanoparticles against multiple stressors in Pangasinodon hypophthalmus. Fish. Shellfish Immunol. 78, 289–298 (2018).
Kumar, N. et al. Nano–zinc enhances gene regulation of non–specific immunity and antioxidative status to mitigate multiple stresses in fish. Sci. Rep. 13(1), 5015 (2023).
Teunen, L. et al. Effect of abiotic factors and environmental concentrations on the bioaccumulation of persistent organic and inorganic compounds to freshwater fish and mussels. Sci. Total Environ. 799, 149448 (2021).
APHA-AWWA-WEF. In Standard Methods for the Estimation of Water and Waste Water. 20th edn (eds Clesceri, L. S., Greenberg, A. E. & Eaton, A. D.) (American Public Health Association, 1998).
Ganeshkar, P. et al. Eco-friendly nano-copper synthesis: gene and enzyme modulation for combating cadmium and ammonia toxicity in fish. Biol. Trace Elem. Res. 203, 5657–5675 (2025).
Kumar, N. Dietary riboflavin enhances immunity and anti-oxidative status against arsenic and high temperature in Pangasianodon hypophthalmus. Aquaculture 533, 736209 (2021).
AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists 16th edn 31–65 (AOAC International, 1995).
Halver, J. E. The nutritional requirements of cultivated warm water and cold water fish species. In Report of the FAO Technical Conference on Aquaculture, Kyoto, Japan, 26 May–2 June. FAO Fisheries Report No. 188 FI/ R188 (En) 9 (1976).
Kumar, N., Kumar, P. & Reddy, K. S. Magical role of iron nanoparticles for enhancement of thermal efficiency and gene regulation of fish in response to multiple stresses. Fish. Shellfish Immunol. 154, 109949 (2024).
Kumar, N., Krishnani, K. K., Gupta, S. K. & Singh, N. P. Selenium nanoparticles enhanced thermal tolerance and maintain cellular stress protection of pangasius hypophthalmus reared under lead and high temperature. Res. Physiol. Neurobiol. 246, 107–116 (2017).
Lowry, O. H., Ronebrough, N. J., Farr, A. L. & Randall, R. J. Protein measurement with Folin phenol reagent. J. Biol. Chem. 193, 265–276 (1951).
Mulla, A. N. et al. Curcumin as a protective agent against chromium and ammonia toxicity using molecular and biochemical approaches in fish. Sci. Rep. 15(1), 12023 (2025).
Takahara, S. et al. Hypocatalesemia, a new generis carrier state. J. Clin. Investig. 29, 610–619 (1960).
Misra, H. P. & Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 247, 3170–3175 (1972).
Habing, W. H., Pabst, M. N., Bjacoby, W. & Glutathion, S. Transferase, the first enzymatic step in Mercatpopunc acid formation. J. Biol. Chem. 249, 7130–7138 (1974).
Hestrin, S. Modified by Augustinsson, 1957. The reaction of acetyl choline esters and other carboxylic acid derivatives with hydroxyline and its analytical application. J. Bio. Chem. 180, 249–261 (1949).
Wootton, I. D. P. Microanalysis. In Medical Biochemistry (ed. J. Churchill) 4th edn, 101–107 (1964).
Wroblewski, L. & LaDue, J. S. Lactic dehydrogenase activity in blood. Proc. Soc. Exp. Biol. Med. 90, 210–213 (1955).
Ochoa, S. Malic dehydrogenase and ‘malic’ enzyme. In Methods of Enzymology (eds Coloric, S. P. & Kaplan, N.) ( Vol I 735–745 (Academic, (1955).
Pfaf, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucl. Acids Res. 29(9), e45 (2001).
Kumar, N., Krishnani, K. K., Meena, K. K., Gupta, S. K. & Singh, N. P. Oxidative and cellular metabolic stress of Oreochromis mossambicus as biomarkers indicators of trace element contaminants. Chemosphere 171, 265–274 (2017).
Kumar, N., Krishnani, K. K., Gupta, S. K. & Singh, N. P. Cellular stress and histopathological tools used as biomarkers in Oreochromis mossambicus for assessing metal contamination. Environ. Toxicol. Pharmacol. 49, 137–147 (2017).
Hussain, S. M. et al. Evaluation of growth, nutrient absorption, body composition and blood indices under dietary exposure of iron oxide nanoparticles in common carp (Cyprinus carpio). J. Anim. Physiol. Anim. Nutr. 108(2), 366–373 (2024).
Uzo-God, O. C., Agarwal, A. & Singh, N. B. Effects of dietary nano and macro iron oxide (Fe2O3) on the growth, biochemical, and hematological profiles of African catfish (Clarias gariepinus) fingerlings. J Appl. Aquac 1–19 (2018).
Thangapandiyan, S., Alisha, A. S. A. & Anidha, K. Growth performance, hematological and biochemical effects of iron oxide nanoparticles in Labeo Rohita. Biocatal. Agric. Biotechnol. 25, 101582 (2020).
Sherif, A. H. & Zommara, M. A. Selenium nanoparticles ameliorate adverse impacts of aflatoxin in nile tilapia with special reference to Streptococcus agalactiae infection. Biol. Trace Elem. Res. 202(10), 4767–4777 (2024).
Sherif, A. H., Elkasef, M., Mahfouz, M. E. & Kasem, E. A. Impacts of dietary zinc oxide nanoparticles on the growth and immunity of nile tilapia could be ameliorated using Nigella sativa oil. J. Trace Elem. Med. Biol. 79, 127265 (2024).
Beisel, W. R. Single nutrients and immunity. Am. J. Clin. Nutr. 35, 417–468 (1982).
Bass, J., Oldham, J., Sharma, M. & Kambadur, R. Growth factors controlling muscle development, Domest. Anim. Endocrinol. 17, 191–197 (1999).
Prodanović, R. et al. Relationship between serum iron and insulin-like growth factor-I concentrations in 10-day-old calves. Acta Vet. Brno. 83(2), 133–137 (2014).
Gonzalez, E. M., Contreras, I. & Estrada, J. A. Effect of iron deficiency on the expression of insulin-like growth factor-II and its receptor in neuronal and glial cells. Neurología (English Edition). 29(7), 408–415 (2014).
Hashizume, T. et al. Effects of Ghrelin on growth hormone secretion from cultured adenohypophysial cells in pigs. Domest. Anim. Endocrinol. 24(3), 209–218 (2003).
Ostling, P., Bjork, J. K., Roos-Mattjus, P., Mezger, V. & Sistonen, L. Heat shock factor 2 (HSF2) contributes to inducible expression of Hsp genes through interplay with HSF1. J. Biol. Chem. 282, 7077–7086 (2007).
Guo, S-N., Zhenga, J-L., Yuan, S-S. & Zhu, Q-L. Effects of heat and cadmium exposure on stress-related responses in the liver of female zebrafish: heat increases cadmium toxicity. Sci. Total Environ. 618, 1363–1370 (2018).
Ramyaa, P., Krishnaswamy, R. & Padma, V. V. Quercetin modulates OTA-induced oxidative stress and redox signalling in HepG2 cells-up regulation of Nrf2 expression and down regulation of NF-κB and COX-2. Biochim. Biophys. Acta. 1840, 681–692 (2013).
Sandhu, N. & Vijayan, M. M. Cadmium-mediated disruption of cortisol biosynthesis involves suppression of corticosteroidogenic genes in rainbow trout. Aquat. Toxicol. 103, 92–100 (2011).
Vos, M. J., Hageman, J., Carra, S. & Kampinga, H. H. Structural and functional diversities between members of the human HSPB, HSPH, HSPA, and DNAJ chaperone families. Biochemistry 47, 7001–7011 (2008).
Desai, N. S., Agarwal, A. A. & Uplap, S. S. HSP: evolved and conserved proteins, structure and sequence studies. Int. J. Bioinform Res. 2, 67–87 (2010).
Ali, A., Bharadwaj, S., O’Carroll, R. & Ovsenek, N. HSP90 interacts with and regulates the activity of heat shock factor 1 in xenopus oocytes. Mol. Cell. Biol. 18, 4949–4960 (1998).
Zou, J., Guo, Y., Guettouche, T., Smith, D. F. & Voellmy, R. Repression of heat shock transcription factor HSF1 by HSP90 (HSP90 complex) that forms a stress sensitive complex with HSF1. Cell 94, 471–480 (1998).
Dendorfer, A. et al. Deferoxamine induces prolonged cardiac preconditioning via accumulation of oxygen radicals. Free Radic Biol. Med. 38, 117–124 (2005).
Hahn, J. Y. et al. red i.; beta-Catenin overexpression reduces myocardial infarct size through differential effects on cardiomyocytes and cardiac fibroblasts. J. Biol. Chem. 281, 30979–30989 (2006).
Zhao, X. S., Pan, W., Bekeredjian, R. & Shohet, R. V. Endogenous endothelin-1 is required for cardiomyocyte survival in vivo. Circulation 114, 830–837 (2006).
Jiaxin, S., Shengchen, W., Yirong, C., Shuting, W. & Shu, L. Cadmium exposure induces apoptosis, inflammation and immunosuppression through cyps activation and antioxidant dysfunction in common carp neutrophils. Fish. Shellfish Immunol. 99, 284–290 (2020).
Gong, Z. G., Wang, X. Y., Wang, J. H., Fan, R. F. & Wang, L. Trehalose prevents cadmium induced hepatotoxicity by blocking Nrf2 pathway, restoring autophagy and inhibiting apoptosis. J. Inorg. Biochem. 192, 62–71 (2019).
Voss, L. et al. Impact of iron oxide nanoparticles on xenobiotic metabolism in HepaRG cells. Arch. Toxicol. 94(12), 4023–4035 (2020).
Wang, S. S. et al. Cadmium-induced apoptosis through reactive oxygen species-mediated mitochondrial oxidative stress and the JNK signaling pathway in TM3 cells, a model of mouse Leydig cells. Toxicol. Appl. Pharmacol. 368, 37–48 (2019).
Ferrari, D. M. & Soling, H. D. The protein disulphide-isomerase family: unravelling a string of folds. Biochem. J. 339, 1–10 (1999).
Solovyov, A. & Gilbert, H. F. Zinc-dependent dimerization of the folding catalyst, protein disulfide isomerase. Protein Sci. 13, 1902–1907 (2004).
Ma, J., Li, Y., Li, W. & Li, X. Hepatotoxicity of Paraquat on common carp (Cyprinus Carpio L). Sci. Total Environ. 616–617, 889–898 (2018).
Sherif, A. H., Okasha, L. A., Kassab, A. S., Abass, M. E. & Kasem, E. A. Long-term exposure to lead nitrate and zinc sulfate nile tilapia impact the Aeromonas hydrophila treatment. Mol. Biol. Rep. 51(1), 71 (2025).
Laskar, A., Eilertsen, J., Li, W. & Yuan, X-M. SPION primes THP1 derived M2 macrophages towards M1-like macrophages. Biochem. Biophys. Res. Commun. 441, 737–742 (2013).
Aliko, V., Qirjo, M., Sula, E., Morina, V. & Faggio, C. Antioxidant defense system, immune response and erythron profile modulation in gold fish, Carassius auratus, after acute manganese treatment. Fish. Shellfish Immunol. 76, 101–109 (2018).
Farombi, E. O., Adelowo, O. A. & Ajimoko, Y. R. Biomarkers of oxidative stress and heavy metal levels as indicators of environmental pollution in African Cat fish (Clarias gariepinus) from Nigeria Ogun river. Int. J. Environ. Res. Publ Health. 4(2), 158–165 (2007).
Kumar, N. et al. Acute toxicity, biochemical and histopathological responses of endosulfan in Chanos Chanos. Ecotoxicol. Environ. Saf. 131, 79–88 (2016).
Sherif, A. H. et al. The nanocomposite of chitosan-vitamin C modulates the expression of immune and antioxidant-related genes in nile tilapia stressed with lead (Pb). Aquacult. Int. 33(2), 136 (2025).
Ismail, M., Al-Naqeep, G. & Chan, K. W. Nigella sativa thymoquinone-rich fraction greatly improves plasma antioxidant capacity and expression of antioxidant genes in hypercholesterolemic rats. Free Radic Biol. Med. 48, 664–672 (2010).
Banaee, M. et al. Evaluation of single and combined effects of cadmium and micro-plastic particles on biochemical and immunological parameters of common carp (Cyprinus carpio). Chemosphere 236, 124335 (2019).
Oner, M., Atli, G. & Canli, M. Changes in serum biochemical parameters of freshwater fish Oreochromis niloticus following prolonged metal (Ag, Cd, Cr, Cu, Zn) exposures. Environ. Toxicol. Chem. Int. J. 27, 360–366 (2008).
Kumar, N., Gismondi, E. & Reddy, K. S. Copper and nanocopper toxicity using integrated biomarker response in Pangasianodon hypophthalmus. Environ. Toxicol. 39(3), 1581–1600 (2024).
Kumar, N., Thorat, S. T., Chavhan, S. R. & Reddy, K. S. Potential role of dietary zinc on gene regulation of growth performance and immunity in Pangasianodon hypophthalmus against multiple stresses. Aquaculture 580, 740271 (2024).
Kumar, N. et al. Dietary Choline, betaine and lecithin mitigates endosulfan-induced stress in Labeo Rohita fingerlings. Fish. Physiol. Biochem. 38(4), 989–1000 (2012).
Kumar, N., Jadhao, S. B., Chandan, N. K., Aklakur, M. & Rana, R. S. Methyl donors potentiates growth, metabolic status and neurotransmitter enzyme in Labeo Rohita fingerlings exposed to endosulfan and temperature. Fish. Physiol. Biochem. 38(5), 1343–1353 (2012).
El-Shenawy, A. M., Gad, D. M. & Yassin, S. A. Effect of iron nanoparticles on the development of fish farm feeds. Alex J. Vet. Sci. 60(1), 102–115 (2019).
Afshari, A., Sourinejad, I., Gharaei, A., Johari, S. A. & Ghasemi, Z. The effects of diet supplementation with inorganic and nanoparticulate iron and copper on growth performance, blood biochemical parameters, antioxidant response and immune function of snow trout Schizothorax zarudnyi (Nikolskii, 1897). Aquaculture, 539, 736638 (2021).
He, K. et al. Effects of dietary nano-iron on growth, hematological parameters, immune antioxidant response, and hypoxic tolerance in juvenile largemouth bass (Micropterus salmoides). Aquaculture Rep. 33, 101759 (2023).
Chan, C. Y. & Wang, W. X. Seasonal and Spatial variations of biomarker responses of rock oysters in a coastal environment influenced by large estuary input. Environ. Pollut. 242, 1253–1265 (2018).
Amachree, D., Moody, A. J. & Handy, R. D. Comparison of intermittent and continuous exposures to cadmium in the blue mussel, mytilus edulis: accumulation and sub-lethal physiological effects. Ecotoxicol. Environ. Saf. 95, 19–26 (2013).
Chandurvelan, R., Marsden, I. D., Gaw, S. & Glover, C. N. Impairment of greenlipped mussel (Perna canaliculus) physiology by waterborne cadmium: relationship to tissue bioaccumulation and effect of exposure duration. Aquat. Toxicol. 124–125, 114–124 (2012).
Zhang, B., Shi, Z., Wang, X., Deng, S. & Lin, H. Depuration of cadmium from blue mussel (Mytilus edulis) by hydrolysis peptides and chelating metal elements. Food Res. Int. 73, 162–168 (2015).
Marasinghe Wadige, C. P. M., Taylor, A. M., Krikowa, F., Lintermans, M. & Maher, W. A. Exposure of the freshwater bivalve hyridella australis to metal contaminated sediments in the field and laboratory microcosms: metal uptake and effects. Ecotoxicology 26(3), 415–434 (2017).
Acknowledgements
This research received support from Indian Council of Agriculture Research as institutional projects (#IXX15014 and 25/S/IPP/202501). The authors extend sincere appreciation to the Director of ICAR-NIASM for providing research facilities for this work. The authors also acknowledge Vidya Pratishthan’s Arts, Commerce and Science College Baramati, Pune for providing student for Master degree dissertation.
Funding
The present work was supported by Indian Council of Agriculture Research as institutional project ((#IXX15014 and 25/S/IPP/202501).
Author information
Authors and Affiliations
Contributions
Authors Name ContributionRaveena Rajpurohit Conduct the experimentNeeraj Kumar Conceived and designed the experiments; performed the experiments; analyzed the data; contributed reagents/materials/analysis tools; wrote the paperKalpana Chandra more ResourcesPrem Kumar Data ValidationKotha Sammi Reddy Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The fish experimental facilities are registered with the Committee for the Purpose of Control and Supervision of Experiments on Animals (CCSEA), under registration number 2190/GO/RReBi/SL/2022. All experimental procedures were approved by the Institute Research Committee (IRC) and were conducted in strict accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rajpurohit, R., Kumar, N., Chandramore, K. et al. Ecofriendly synthesis of iron nanoparticles and their role in mitigation of multiple abiotic stresses in fish. Sci Rep 16, 3027 (2026). https://doi.org/10.1038/s41598-025-32925-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32925-8