Abstract
Red cell distribution width (RDW) and glycated hemoglobin index (HGI) is considered an important tool for assessing the prognosis of sepsis patients, closely related to the risk of mortality associated with sepsis. This study investigates the association between the HGI combined with RDW and the risk of in-hospital mortality in patients with sepsis. We analyzed data from 13,726 sepsis patients who were admitted to the intensive care unit (ICU) for more than 24 h, sourced from the American Medical Information Mart for Intensive Care (MIMIC-IV) database. Kaplan-Meier survival curves and multivariable Cox regression analyses were employed to assess the impact of various variables on patient outcomes, stratified by quartiles of HGI and RDW. Additionally, restricted cubic spline (RCS) analysis was utilized to explore how changes in HGI and RDW might influence the studied outcomes. The results indicated that the highest quartile (Q4) of the combined metrics significantly increased in-hospital mortality compared to the lowest quartile (Q1) (p < 0.0001). Multivariable Cox regression analysis revealed that patients in Q4 faced the highest risk of in-hospital mortality (hazard ratio: 1.22, 95% confidence interval: [1.10–1.36], p < 0.001). RCS analysis demonstrated a nonlinear relationship between HGI-RDW and the risk of adverse outcomes. Further analysis identified significantly elevated risks in patients over 65 years old, those who were widowed, those receiving macrolide antibiotics, and those with congestive heart failure or severe liver disease. In conclusion, elevated levels of the combined HGI and RDW metrics are independent risk factors for adverse outcomes in critically ill patients with sepsis, associated with increased mortality.
Similar content being viewed by others
Introduction
Caused by an abnormal immune response to infection, sepsis could be a fatal condition with potential organ dysfunction and severe complications1. It remains a global challenge to be tackled in critical care medicine and also one of the biggest killers in the intensive care unit (ICU)2. Each year witnesses more than 48.9 million new cases of sepsis, accounting for 20% of all deaths worldwide3. The slow decline of incidence and mortality rates achieved through advances in critical care4,5 adds urgency to the identification of reliable risk factors for improved outcomes and early preventive strategies.
Insulin resistance (IR) characterized by diminished responsiveness of peripheral tissues to insulin is frequently observed in sepsis patients. This pathological state is often associated with elevated insulin levels and sensitive glucose fluctuations, both of which may contribute to increased mortality risk6. Some studies have indicated a correlation between poor glycemic control and an increased risk of severe infections, with glycated hemoglobin A1c (HbA1c) > 53 mmol/L (7.0%)7,8. However, the relationship between HbA1 and sepsis outcomes remains controversial. Balintescu A et al.9 observed a J-shaped HbA1c-the risk of sepsis relationship, without a direct correlation between HbA1c levels and mortality in sepsis. Additionally, lower HbA1c levels have been associated with an increased susceptibility to infection and sepsis in diabetic patients. All these add weight to the conclusion that HbA1c alone may not serve as a robust predictor of sepsis prognosis or mortality.
The Hemoglobin Glycation Index (HGI), which represents individual variations in hemoglobin glycation, is an emerging marker to assess glycemic control in patients with diabetes10. HGI is calculated as the difference between the measured HbA1c and the predicted HbA1c11. As a potential marker of glucose variability, HGI is correlated with insulin resistance, diabetes-related complications, and adverse outcomes in patients in critical condition suffering from coronary artery disease12,13,14.
Red cell distribution width (RDW) measures variability in red blood cell size15. It reflects anemia caused by chronic disease, iron metabolism dysfunction, and impaired erythropoiesis, often mediated by various cytokines16,17. Among patients in critical states, elevated RDW is commonly observed, particularly in ICU admissions and inflammatory states such as sepsis18. Elevated RDW may be related to chronic inflammation and oxidative stress associated with sepsis. Though RDW was identified as an independent marker of mortality in critically ill sepsis patients19,20, the combined prognostic utility of RDW and HGI in assessing mortality risk remains unexplored.
In this context, the analysis of HGI-RDW combined metric might enable a comprehensive assessment of metabolic dysregulation, inflammatory responses, and prognostic risks. Therefore, exploring the combined effect of HGI and RDW on sepsis-related mortality is the research objective, with the goal of improving risk stratification and providing a theoretical basis for personalized prevention and management strategies in this population.
Materials and methods
Study population
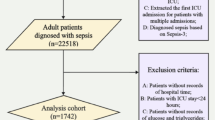
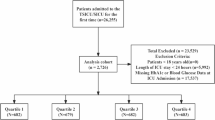
Data of eligible adult patients in the Medical Information Mart for Intensive Care IV (MIMIC-IV) database were obtained, covering the period from 2008 to 2019, including data on HbA1c, fasting blood glucose (FBG), RDW, and sepsis (sequential organ failure assessment [SOFA] ≥ 2). We included adult patients (≥ 18 years) with sepsis who were admitted to the ICU for the first time and stayed for more than 24 h. What’s more, HbA1c, FBG, and RDW data are all within seven days of the patient’s diagnosis. The exclusion criteria were: (1) ICU stay < 24 h, and (2) patients without HbA1c, FBG, or RDW data. The MIMIC-IV database was approved by the Institutional Review Board of Beth Israel Deaconess Medical Center and the Massachusetts Institute of Technology. The patient selection process is illustrated in Fig. 1.
Flowchart of data selection.
Data collection
Clinical and demographic data within the first 24 h after ICU admission were extracted from the MIMIC-IV database using PostgreSQL (version 14.2). The variables collected included demographic characteristics (age, gender, race, marital status), vital signs (heart rate, systolic blood pressure (SBP) diastolic blood pressure (DBP), mean blood pressure (MBP), temperature, peripheral capillary oxygen saturation (SPO2)), common comorbidities (diabetes, severe liver disease, chronic pulmonary disease (CPD), congestive heart failure (CHF), cerebrovascular disease, myocardial infarction, hypertension), and survival outcomes. Additional data on laboratory parameters, including red blood cell (RBC), white blood cell (WBC), hemoglobin, hematocrit, mean corpuscular volume (MCV), estimated glomerular filtration rate (eGFR), international normalized ratio (INR), prothrombin time (PT), partial thromboplastin time (PTT) were obtained. Severity scores, such as acute physiology score III (APSIII), simplified acute physiology score II (SAPSII), systemic inflammatory response syndrome (SIRS), and SOFA score, were also recorded, along with data on medications and interventions, including antibiotics.
The eGFR was calculated as follows: (for males, multiply by 1)
All comorbidities were identified using the International Classification of Diseases (ICD-9 or ICD-10) codes. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational research. If a patient was admitted to the ICU multiple times, only the first admission data were recorded and analyzed.
Definition of combined metrics and study outcomes
HGI was defined as the gap between the observed HbA1c and the expected HbA1c levels. HGI = actual HbA1c − predicted HbA1c. The predicted HbA1c was calculated using FBG or mean glucose levels, based on the equation (Fig. 2):
illustrates the linear relationship between HbA1c and FBG.
The actual HbA1c values were obtained through laboratory measurements. The HGI was classified into two levels: low (−4.269 ≤ HGI≤−0.427) and high (−0.427 < HGI ≤ 13.538)(based on median). RDW was also classified into low (11.1 ≤ RDW ≤ 14.7) and high (14.7 < RDW ≤ 40.7) levels (based on median). The combined metric was stratified into quartiles as follows: Q1 – low HGI and low RDW, Q2 – low HGI and high RDW, Q3 – high HGI and low RDW, and Q4 – high HGI and high RDW, with a primary outcome of as sepsis-related in-hospital mortality.
Statistical methods
Statistical analyses were performed using R version 4.3.1. Data with normal distributions were presented as means ± standard deviation (x̄ ± s), while non-normally distributed data were reported as medians (interquartile range) [M(QL, QU)], with comparisons analyzed by the Mann-Whitney U test. Group comparisons were made using the chi-square test. Patients were assigned into four subgroups according to quartiles, which were analyzed as both continuous and categorical variables. Q1 served as the reference group. Kaplan-Meier(KM) survival curves and Cox proportional hazards models were employed to illustrate cumulative incidence by quartile and the hazard ratios (HR) of HGI and RDW as risk factors for the outcome events respectively. Restricted cubic spline (RCS) curves were used to explore nonlinear relationships between HGI and RDW as continuous variables and outcomes. A forest plot was generated to visualize the association between the combined metric and sepsis-related mortality. Additionally, receiver operating characteristic (ROC) curves were utilized to validate the predictive accuracy. All descriptive analyses were two-sided, with p < 0.05 considered statistically significant.
Results
Baseline characteristics
13,726 sepsis patients were included in this study. The majority were over 65 years old (57.66%), male (60.56%), white race (65.47%), married (47.79%), and without a history of myocardial infarction (81.32%), cerebrovascular disease (83.07%), CPD (76.37%), CHF (69.27%), severe liver disease (95.43%), or diabetes (69.42%). However, hypertension was prevalent among patients(63.81%). As shown in the stratified results (Table 1), in reference to the low HGI-RDW group, significant fluctuations in certain biochemical indicators were observed in the high HGI-RDW group. Notably, the eGFR exhibited the most significant decline, indicating renal impairment (Q4: 15.569 vs. Q1: 45.979). Additionally, APSIII increased (Q4: 49.000 [37.000, 64.000] vs. Q1: 37.000 [27.000, 51.000]) and SAPSII scores also rose (Q4: 40.000 [32.000, 49.000] vs. Q1: 35.000 [28.000, 43.000]), suggesting an increase in disease severity. Furthermore, patients in the high HGI-RDW group were more prone to CHF (Q4: 43.56% vs. Q1: 19.65%) and diabetes (Q4: 47.07% vs. Q1: 10.30%). (Baseline characteristics based on HGI and RDW classifications were detailed in Tables S1 and S2.)
KM survival curves and cumulative incidence curves
Based on the quartiles of the HGI-RDW combined metric, the log-rank test was used to compare overall survival distributions among the four groups. The incidence of primary outcomes between groups was illustrated using KM survival analysis curves (Fig. 3) (in-hospital mortality curves for the individual HGI and RDW metric were presented in Fig. S1 and Fig. S2). Patients in the lowest quartile (Q1) had significantly higher survival rates than those in the other groups, while the Q4 group exhibited the lowest survival rate. The inter-group differences were significant (p < 0.0001), indicating that lower levels of both HGI and RDW are associated with better survival in sepsis patients. Conversely, elevated levels of both HGI and RDW had a detrimental impact on patient prognosis. Moreover, The combined metric (HGI-RDW) had a more pronounced effect on mortality than the individual metrics alone. At 50, 100, and 150 days, sepsis patients with high HGI-RDW combined metric had lower survival rates compared to those with only high HGI or RDW. In contrast, patients with low HGI-RDW combined metric had higher survival rates than those with low level of either metric alone.
illustrates the in-hospital mortality curves for the combined metric.
Multivariable Cox regression models
Multivariable Cox regression analysis (Table 2) indicated that in the unadjusted model (Model 1), patients with high HGI-RDW combined metrics faced a higher risk of adverse outcomes in relation to the low-level group (HR = 1.47, 95% confidence interval [CI]: [1.36–1.59], p < 0.001). After adjusting for demographic characteristics (Model 2), adverse outcome risk remains high for the high HGI-RDW combined metrics group [Q4: HR = 1.39, 95% CI (1.28–1.50), p < 0.001]. This trend persisted in Model 3, the fully adjusted model, indicating that high HGI-RDW combined metrics were associated with a higher risk of adverse outcomes in sepsis patients [HR = 1.22, 95% CI (1.10–1.36), p < 0.001]. Additionally, the high HGI-RDW combined metrics group was observed to have a higher risk than another single metric group [HGI-RDW (HR) = 1.39 > RDW (HR) = 1.13 > HGI (HR) = 1.08]. The relationships between sepsis outcomes and single HGI and RDW metric were detailed in Tables S3 and S4.
RCS curves
RCS analysis was conducted across Models 1, 2, and 3 to examine the continuous relationship between HGI, RDW, and in-hospital mortality. As shown in Fig. 4 and Fig. 5, although HGI and RDW did not exhibit a completely positive linear correlation with in-hospital mortality, the HR consistently remained above 1, indicating an association between high HGI and RDW levels and increased in-hospital mortality (P for nonlinear < 0.001).
RCS analysis of HGI and sepsis risk A. RCS curve analysis based on Model (1) B. RCS curve analysis based on Model (2) C. RCS curve analysis based on Model 3.
RCS analysis of RDW and sepsis risk A. RCS curve analysis based on Model (1) B. RCS curve analysis based on Model (2) C. RCS curve analysis based on Model 3.
Multivariable Cox regression forest plot
To further investigate the risk relationship between the HGI-RDW combined metric and sepsis mortality outcomes across different variables, comparisons of HRs among various factors were conducted using the Wald test within a multivariable Cox regression model (Fig. 6). Among sepsis patients, those aged ≤ 65 years (p < 0.001, HR: 0.62, 95% CI: [0.58, 0.66]) had a lower hazard ratio (HR < 1) compared to patients aged > 65 years (reference group, HR = 1), indicating a higher risk for adverse outcomes in the older group. Additionally, widowed patients (p < 0.001, HR: 1.38, 95% CI: [1.22, 1.55]), patients treated with macrolide antibiotics (p < 0.001, HR: 1.31, 95% CI: [1.14, 1.51]), those with CHF (p < 0.001, HR: 1.22, 95% CI: [1.15, 1.30]), and patients with severe liver disease (p < 0.001, HR: 1.32, 95% CI: [1.18, 1.47]) also demonstrated significant risk. .Similar trends were observed when analyzing individual HGI and RDW metric (Fig. S3 and Fig. S4).
Cox regression forest plot of HGI-RDW combined metric and mortality outcomes in sepsis patients.
ROC curves
According to ROC analysis results, the area under the ROC curve (AUC) was 0.75 (95%CI: 0.699–0.8), demonstrating the efficacy of HGI-RDW combined metric in mortality prediction, suggesting a strong correlation with sepsis-related outcomes (Fig. 7).
ROC curve.
Discussion
As the first to explore the relationship between the combined metric of HGI and RDW and in-hospital mortality rate in sepsis patients, this study found that the HGI-RDW levels are significantly associated with the risk of mortality in sepsis patients, with higher combined values indicating worse long-term survival. Even after adjusting for confounding factors, mortality risk remained higher in patients with elevated HGI-RDW metric, especially among those over 65, widowed patients, those treated with macrolide antibiotics, and patients with CHF or severe liver disease. These findings highlight the potential of using HGI-RDW combined metric as independent predictor for critically ill sepsis patients.
In recent years, with a deeper understanding of the pathophysiology of sepsis, increasing attention has been paid to the interactions between inflammation, immune responses, metabolic disorders, and organ dysfunction21,22. HGI that reflects the variability in HbA1c levels is typically used to assess glycemic control in diabetic patients while bearing some relation with inflammatory responses and metabolic disturbances in sepsis patients12. IR and hyperglycemia induced by acute stress are commonly observed in sepsis patients23. For critically ill sepsis patients without diabetes, they face a challenging metabolic environment due to excessive counter-regulatory hormones (e.g., glucagon, catecholamines, growth hormone) and cytokines (e.g., IL-1, IL-6, TNF-α), which, coupled with the use of exogenous catecholamines and glucose, result in relative insulin deficiency24. In this study, the majority of sepsis patients did not have diabetes, and higher HGI levels were positively correlated with increased mortality. Moreover, patients with high HGI were more prone to diabetes and CHF, consistent with existing research linking HGI to heart failure risk in hypertensive populations25. HGI, therefore, is not only a prognostic marker for diabetes but also holds value in assessing risk among non-diabetic sepsis patients. Additionally, sepsis patients are often susceptible to acute stress responses, which may present as short-term fluctuations that can affect the reliability of single glucose measurements26. Therefore, compared to traditional FBG metric, HGI may provide a more stable representation of glycemic control over months, providing a more reliable marker of patient risk compared to FBG alone.
RDW, as a marker of variability in red blood cell volume, is a common blood test parameter. Elevated RDW may be closely related to erythropoiesis disorders, chronic inflammation, and oxidative stress, and is associated with poor prognosis in various chronic diseases, such as cardiovascular disease, diabetes, and chronic kidney disease27,28,29. The results of this study align with these observations, as sepsis patients with high RDW exhibited significantly higher in-hospital mortality rates. An elevated RDW may reflect the intensity of the inflammatory response and the burden of chronic disease in sepsis patients30. Sepsis patients typically experience severe systemic inflammatory responses, leading to iron metabolism disorders and suppression of erythropoiesis31. Furthermore, oxidative stress and systemic inflammation may increase red blood cell size variability by damaging the structure and function of red blood cell membranes, thereby elevating RDW and exacerbating prognosis32,33.
This study is the first to observe the impact of combined metric of HGI and RDW on the prognosis of sepsis patients, with HGI representing long-term glucose exposures and metabolic control, making it more suitable for disease prognostic evaluation. While RDW reflects the quality of erythropoiesis and the extent of the inflammatory response. Elevated HGI may indicate metabolic dysregulation and heightened inflammatory responses, while RDW, by illustrating heterogeneity in red blood cell size, further reveals the profound impacts of chronic inflammation and oxidative stress. Compared to traditional scoring systems such as SOFA and acute physiology and chronic health evaluation (APACHE), HGI captures chronic metabolic abnormalities, while RDW reflects acute inflammatory states, together constructing a more comprehensive risk assessment framework. Furthermore, traditional scoring systems often do not fully account for glucose metabolism abnormalities and dynamic changes in hematological parameters. In contrast, the HGI-RDW combined metric provides more targeted information, enhancing the sensitivity and specificity of prognostic evaluations. In sepsis patients, this metric offers a comprehensive view of a patient’s metabolic and inflammatory status. The combined effects of both metrics, through exacerbating systemic inflammation and oxidative stress, impairing organ microcirculation and function, ultimately lead to increased mortality34,35. Long-term glucose metabolic abnormalities may increase glucose metabolism via the polyol pathway, exacerbating non-enzymatic glycation of proteins and lipids, leading to the formation of advanced glycation end products (AGEs), which further induce oxidative stress and inflammation36. Additionally, oxidative stress results in increased free radical production, weakening the antioxidant defenses of endothelial cells and causing mitochondria to produce excess reactive oxygen species (ROS), directly damaging cell membranes and DNA, leading to endothelial dysfunction37. These mechanisms together aggravate organ injury and functional decline in sepsis patients, further leading to adverse prognoses. In this study, the combination of high HGI and RDW had a more pronounced impact on the survival rate than either metric alone. Maintaining low levels of both HGI and RDW often indicated a higher survival rate. Notably, the presence of isolated high RDW and HGI levels did not result in a significant decrease in eGFR, whereas the combined high levels of RDW and HGI led to a reduction in eGFR to 15.569 mL/min, suggesting further renal impairment in sepsis patients. This infers that elevated combined HGI and RDW may exert a stronger damaging effect on the endothelial lining, exacerbating glomerular endothelial dysfunction and glomerulosclerosis, consequently leading to a significant decline in eGFR38. This effect involves the synergistic destruction of vascular endothelial cells by long-term glucose metabolic abnormalities and oxidative stress, resulting in impaired renal microcirculation and significantly reduced renal function36,39. Thus, monitoring the combined metric of HGI and RDW may provide more sensitive insights into renal function in sepsis, potentially helping to identify high-risk patients who require closer monitoring and early intervention.
The innovation of this study lies in combining the metrics of HGI and RDW to predict prognosis in sepsis patients. The findings reveal that the combined metric outperforms single markers in predicting mortality, particularly in patients with both elevated HGI and RDW. This combined metric offers a comprehensive assessment method, aiding in the early identification of high-risk patients and enabling timely intervention. However, this study has certain limitations. First, the study analyzed only included the initial monitored parameter following ICU admission, without considering dynamic changes in these metrics. Second, as a retrospective study, although various confounding factors were adjusted for in the model, unmeasured confounding factors may still influence the results. Third, the data was drawn from a single database, and external validation of the findings requires further multicenter studies.
Conclusion
This study is the first to explore the prognostic value of combining HGI and RDW metrics in critically ill sepsis patients. The results indicate that elevated HGI and RDW levels are significantly associated with a higher risk of in-hospital mortality. Furthermore, compared to single metrics, the combined metric better reflects the prognostic status of sepsis patients. Based on the findings of this study, the HGI-RDW combined metric not only provides a new perspective for risk stratification in sepsis patients but also guides the development of personalized treatment plans. For high-risk patients, strict glycemic control and targeted anti-inflammatory therapies may improve their prognosis and reduce mortality.
Data availability
The datasets analysed during the current study are available in the MIMIC-IV database (MIMIC-IV v3.0 (physionet.org)).
References
Jacobi, J. The pathophysiology of sepsis – 2021 update: part 2, organ dysfunction and assessment[J]. Am. J. Health Syst. Pharm. 79 (6), 424–436 (2022).
Wang, Z. et al. The relationship between hematocrit and serum albumin levels difference and mortality in elderly sepsis patients in intensive care units-a retrospective study based on two large database[J]. BMC Infect. Dis. 22 (1), 629 (2022).
Rudd, K. E. et al. Global, regional, and National sepsis incidence and mortality, 1990–2017: analysis for the global burden of disease Study[J]. Lancet 395 (10219), 200–211 (2020).
Rhee, C. et al. Incidence and trends of sepsis in US hospitals using clinical vs claims data, 2009–2014[J]. JAMA 318 (13), 1241–1249 (2017).
Alvaro-Meca, A. et al. Epidemiological trends of sepsis in the twenty-first century (2000–2013): an analysis of incidence, mortality, and associated costs in Spain[J]. Popul. Health Metr. 16 (1), 4 (2018).
Li, X. et al. Acute glycemic variability and risk of mortality in patients with sepsis: a meta-analysis[J]. Diabetol. Metab. Syndr. 14 (1), 59 (2022).
McKane, C. K. et al. Diabetes mellitus and community-acquired bloodstream infections in the critically ill[J]. J. Crit. Care. 29 (1), 70–76 (2014).
Dronge, A. S. et al. Long-term glycemic control and postoperative infectious complications[J]. Arch. Surg. 141 (4), 375–380 (2006).
Balintescu, A. et al. Glycaemic control and sepsis risk in adults with type 1 diabetes[J]. Diabetes Obes. Metab. 25 (7), 1942–1949 (2023).
Hempe, J. M. et al. The hemoglobin glycation index identifies subpopulations with harms or benefits from intensive treatment in the ACCORD trial[J]. Diabetes Care. 38 (6), 1067–1074 (2015).
Twomey, P. J. et al. Biological variation in HbA1c predicts risk of retinopathy and nephropathy in type 1 diabetes[J]. Diabetes Care. 27 (10), 2569–2570 (2004).
Kim, W. et al. Hemoglobin glycation index is associated with incident chronic kidney disease in subjects with impaired glucose metabolism: A 10-year longitudinal cohort study[J]. J. Diabetes Complications. 35 (1), 107760 (2021).
Wei, X. et al. Risk analysis of the association between different hemoglobin glycation index and poor prognosis in critical patients with coronary heart disease-A study based on the MIMIC-IV database[J]. Cardiovasc. Diabetol. 23 (1), 113 (2024).
Marini, M. A. et al. Association between hemoglobin glycation index with insulin resistance and carotid atherosclerosis in non-diabetic individuals[J]. PLoS One. 12 (4), e0175547 (2017).
Aslan, D. et al. Importance of RDW value in differential diagnosis of hypochrome anemias[J]. Am. J. Hematol. 69 (1), 31–33 (2002).
Weiss, G. & Goodnough, L. T. Anemia of chronic disease[J]. N Engl. J. Med. 352 (10), 1011–1023 (2005).
Weiss, G., Ganz, T. & Goodnough, L. T. Anemia of inflammation[J]. Blood 133 (1), 40–50 (2019).
Scharte, M. & Fink, M. P. Red blood cell physiology in critical illness[J]. Crit. Care Med. 31 (12 Suppl), S651–S657 (2003).
Moreno-Torres, V. et al. Red blood cell distribution width as prognostic factor in sepsis: A new use for a classical parameter[J]. J. Crit. Care. 71, 154069 (2022).
Dankl, D. et al. Red cell distribution width is independently associated with mortality in sepsis[J]. Med. Princ Pract. 31 (2), 187–194 (2022).
Fenner, B. P. et al. Immunological endotyping of chronic critical illness after severe sepsis[J]. Front. Med. (Lausanne). 7, 616694 (2020).
Nakamori, Y., Park, E. J. & Shimaoka, M. Immune deregulation in sepsis and septic shock: reversing immune paralysis by targeting PD-1/PD-L1 pathway[J]. Front. Immunol. 11, 624279 (2020).
Marik, P. E. & Raghavan, M. Stress-hyperglycemia, insulin and Immunomodulation in sepsis[J]. Intensive Care Med. 30 (5), 748–756 (2004).
McCowen, K. C., Malhotra, A. & Bistrian, B. R. Stress-induced hyperglycemia[J]. Crit. Care Clin. 17 (1), 107–124 (2001).
Wang, R. et al. Association of triglyceride glucose-body mass index and hemoglobin glycation index with heart failure prevalence in hypertensive populations: a study across different glucose metabolism status[J]. Lipids Health Dis. 23 (1), 53 (2024).
Li, L. et al. Association of stress hyperglycemia ratio and mortality in patients with sepsis: results from 13,199 patients. Infection, (2024).
Lu, Y. A. et al. Red cell distribution width associated with adverse cardiovascular outcomes in patients with chronic kidney disease[J]. BMC Nephrol. 18 (1), 361 (2017).
Deng, X. et al. Red blood cell distribution width is associated with adverse kidney outcomes in patients with chronic kidney disease[J]. Front. Med. (Lausanne). 9, 877220 (2022).
Hong, J. et al. Impact of red cell distribution width and red cell distribution width/albumin ratio on all-cause mortality in patients with type 2 diabetes and foot ulcers: a retrospective cohort study[J]. Cardiovasc. Diabetol. 21 (1), 91 (2022).
Liu, J. et al. Association of red cell distribution width-to-platelet ratio and mortality in patients with sepsis[J]. Mediators Inflamm. 2022, 4915887 (2022).
Brandtner, A. et al. Linkage of alterations in systemic iron homeostasis to patients’ outcome in sepsis: a prospective study[J]. J. Intensive Care. 8, 76 (2020).
Orrico, F. et al. Oxidative stress in healthy and pathological red blood cells[J]. Biomolecules 13 (8), 1262 (2023).
Nakamura, N. et al. Utilizing red blood cell distribution width (RDW) as a reliable biomarker to predict treatment effects after chimeric antigen receptor T cell therapy[J]. Clin. Exp. Med. 24 (1), 105 (2024).
Gao, C. & Peng, L. Association and prediction of red blood cell distribution width to albumin ratio in all-cause mortality of acute kidney injury in critically ill patients[J]. Front. Med. (Lausanne). 10, 1047933 (2023).
Hempe, J. M. et al. High and low hemoglobin glycation phenotypes in type 1 diabetes: a challenge for interpretation of glycemic control[J]. J. Diabetes Complications. 16 (5), 313–320 (2002).
Brownlee, M. Biochemistry and molecular cell biology of diabetic complications[J]. Nature 414 (6865), 813–820 (2001).
Goh, S. Y. & Cooper, M. E. Clinical review: the role of advanced glycation end products in progression and complications of diabetes[J]. J. Clin. Endocrinol. Metab. 93 (4), 1143–1152 (2008).
Xu, Y. & Qi, W. Association between red cell distribution width to albumin ratio and acute kidney injury in patients with sepsis: a MIMIC population-based study[J]. Int. Urol. Nephrol. 55 (11), 2943–2950 (2023).
Wada, J. & Makino, H. Inflammation and the pathogenesis of diabetic nephropathy[J]. Clin. Sci. (Lond). 124 (3), 139–152 (2013).
Funding
This study was funded by the Zhejiang Provincial Health Science and Technology Program Project (2022KY1442).
Author information
Authors and Affiliations
Contributions
Huan Xu and Luxiang Liu designed and conceptualized this study, Huan Xu analyzed the data and wrote the manuscript, Jiaojian Lv and Feifei Lin drew the images, and Luxiang Liu checked manuscript .
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
The MIMIC-IV database has obtained approval from the Institutional Review Boards of Beth Israel Deaconess Medical Center and the Massachusetts Institute of Technology.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, H., Lv, J., Lin, F. et al. Combined glycated hemoglobin index and red cell distribution width predict in-hospital mortality in critically ill sepsis patients based on MIMIC-IV analysis. Sci Rep 15, 12266 (2025). https://doi.org/10.1038/s41598-025-94179-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94179-8