Abstract
This study evaluated the feasibility of a novel dynamic conformal arc therapy (DCAT) plan, based on segment shape optimization (SSO) and variable dose rate (VDR), versus volumetric modulated arc therapy (VMAT) for stereotactic body radiotherapy (SBRT) in stage I/II centrally located non-small-cell lung cancer (NSCLC) per RTOG 0813. Twenty-five patients with PTV < 70 cc were retrospectively analyzed, with both plans using identical parameters in Monaco. Both plans met RTOG 0813 criteria. DCAT showed increased ipsilateral and total lung dose but no significant differences in most other organs at risk (OARs) (P > 0.05). DCAT significantly reduced segments (median 19.38%), monitor units (MUs, median 23.27%), and beam-on time (BOT, median 8.80 s; P < 0.05). Its γ passing rate (2%, 1 mm) was higher (93.70% vs. VMAT 91.90%, P < 0.05). Larger PTV volumes improved DCAT’s heterogeneity index (HI) and segment advantages but worsened R50% vs. VMAT. Novel DCAT is feasible for SBRT in such NSCLC, especially with small PTV, offering shorter BOT, higher delivery accuracy, and comparable OARs sparing to VMAT.
Similar content being viewed by others
For medically inoperable patients with node-negative and centrally located non-small-cell lung cancer (NSCLC), stereotactic body radiotherapy (SBRT) is emerging as a standard of care with high cure rates and low treatment-related toxicity1,2,3,4. The characteristics of SBRT include fewer fractions, higher doses per fraction, high conformity, and accurate delivery5. For patients with stage I/II centrally located NSCLC, SBRT has become a curable option based on several randomized clinical trials6,7,8,9. Centrally located NSCLC is close to vital structures such as the bronchus, esophagus, great vessels, and brachial plexus, which may be damaged by the high dose of SBRT, thus failing to achieve optimal tumor control. Therefore, SBRT plans for centrally located NSCLC need to balance the need for tumor control with minimizing the risk of radiotherapy toxicity6.
Traditionally, to achieve high plan quality, many lung cancer SBRT plans use VMAT technique10,11. VMAT is a technique that provides a highly conformal dose distribution, delivering a continuous intensity-modulated beam as the gantry rotates around the patient12. Of all radiotherapy techniques, VMAT has the best planning quality and can be used as a reference for other radiotherapy techniques13,14. During the execution of a VMAT plan, the gantry’s rotational speed, the multi-leaf collimator’s (MLC’s) aperture, and the dose rate are all adjusted simultaneously15. Many studies have reported comparisons between VMAT and other techniques, confirming that VMAT plans can reduce treatment time and provide better organs at risk (OARs) sparing without affecting the dose coverage16,17,18,19. However, for lung SBRT treatment, highly modulated VMAT plans are susceptible to minor field dosimetry errors20 and interplay effects21,22, resulting in errors in dose calculation and dose delivery23. Especially under free-breathing conditions, interplay effects can cause dose deviations within and at the edges of the target that can reach a maximum of 15–20% 22. A more severe interplay effect occurs when combining FFF beams and the hypofractionated SBRT treatment method24,25. In contrast, the DCAT plans can be less affected by interplay effect due to the characteristics of fewer MLC leaves blocking the target and low modulation degree26,27,28,29,30,31.
Conventional DCAT plans rely on the dynamic motion of multi-leaf collimator (MLC) leaves and gantry rotation to maintain beam shape conformity to the target during gantry rotation, which results in insufficient dose modulation capability32,33. To address this limitation and enhance dose modulation, the Monaco TPS has implemented several improvements to conventional DCAT plans via segment shape optimization (SSO) and variable dose rate (VDR). SSO allows the MLC to move within 5 mm inside and outside the target, thus effectively reducing the segments and shortening the delivery time. VDR allows the accelerator to reduce the dose rate where a limited dose is needed. With both tools, Monaco TPS can improve the plan quality while maintaining the advantages of the conventional DCAT plans, resulting in better OARs sparing26,34.
Recently, FFF beams have increasingly begun to be applied clinically in SBRT treatment for NSCLC due to higher dose rates and superior dosimetric advantages35,36,37. FFF beams can significantly shorten the beam-on time (BOT) due to higher dose rates, thereby improving patient comfort and reducing dose output uncertainty due to less collimator scatter and electron contamination38,39,40.
Although some scholars have investigated the application of DCAT plans in the SBRT treatment of peripheral lung cancer41,42, data on plan quality, BOT, and γ passing rates of DCAT plans using FFF beams when applied to the SBRT treatment of stage I/II centrally located NSCLC have been rarely reported. For this reason, this paper analyzes the application of SSO- and VDR-based FFF-DCAT plans in the SBRT treatment of stage I/II centrally located NSCLC using the new version of Monaco TPS (version 6.0), aiming to provide a reference for clinical application.
Materials and methods
Patient cohort
We retrospectively selected 25 consecutive patients with stage I/II centrally located NSCLC treated with VMAT-SBRT after obtaining approval from our institutional ethics committee. Written informed consent was obtained from the patients for the use of their anonymized data in the present study. All methods were preformed in accordance with the following relevant guidelines and regulations. The patient stages were 10 T1N0M0 and 15 T2N0M0; patients’ ages ranged from 47 to 71 years; 17 were male, and eight were female; and the targets were all located in the lower lobes of the lungs, with 11 in the left lower lobe and 14 in the right lower lobe. Based on the RTOG 0236 and 0813 protocols1,43, the screened patients were required to fulfill the following conditions: targets (planning target volume, PTV) no larger than 5 cm and located outside the 0.5-cm area around the proximal bronchial tree (PBT) and within the 2-cm area or immediately adjacent to the mediastinal or pericardial pleura. Exclude patients in whom the extent of the tumor cannot be defined on CT (e.g., solid lesions around the tumor or lung atelectasis) and patients with ultracentral lung tumors44,45.
Positioning and contouring
The positioning was applied on a 16-row big bore 4DCT scanner (Philips Medical Systems, Cleveland, OH), and the maximum intensity projection (MIP)-CT and the average intensity projection (AIP)-CT were generated subsequently. The thickness of CT images is 3 mm.
All patients were placed in the supine position with arms crossed over the head. The thoracic region was immobilized using a thermoplastic film (Klarity Medical Equipment Co., Ltd., Guangzhou, China). The scan scope included all OARs to be evaluated. The recommended range was from the upper edge of the cricoid cartilage to the upper edge of the vertebral body of lumbar 2, which was at least 10 cm above the upper and lower boundaries of the target. The internal target volume (ITV) was delineated on the MIP-CT by a radiation oncologist with expertise in lung SBRT. Based on the 4DCT, all patients selected had a breathing amplitude of < 1 cm in the three-dimensional direction. The planning target volume (PTV) was created by adding an isotropic 5 mm margin to the ITV according to the RTOG protocols1,46. The AIP-CT was used for planning.
The ITV diameter was 2.04 ± 0.65 cm (range: 1.08–3.69 cm), and the volume was 11.90 ± 6.70 cc (range: 2.34–27.56 cc). The PTV volume was 29.49 ± 13.57 cc (range: 5.82–61.41 cc), with a maximum PTV volume of less than 70 cc. The OARs included the ipsilateral lung and all (excluding ITV), ipsilateral PBT, spinal cord, esophagus, great vessels, heart, ipsilateral brachial plexus, and skin.
Planning
All plans were designed in Monaco TPS (V6.0, Elekta Solution AB, Kungstensgatan 18, Stockholm, Sweden) using the XVMC (X-ray Voxelized Monte Carlo) algorithm26 with 6 MV-FFF beams. The plans were applied using an Infinity accelerator with an Agility collimator (5 mm MLC, 160 leaves).
Based on the RTOG 0813 protocol, the treatment prescription for this experiment was five fractions every second to third day over 1.5 to 2 weeks at a total dose of 50 Gy, with a single dose of 10 Gy/fx, and an equivalent biological dose of 100 Gy when the tumor α/β = 10 Gy.
The DCAT and VMAT plans using three noncoplanar single arcs were redesigned for each patient. Two 210° arcs (table angle 0°) on the ipsilateral side, plus a 60° anterior arc (table angle 90°), were used, with the isocenter placed at the center of the PTV. The three single arcs for all plans used different collimator angles between ± 45° to minimize the tongue-and-groove effect47. The target margin was selected to be 0–1 mm, the angular increment was set to 10°, SSO and VDR were selected, and a 2.0 mm dose grid resolution and a 1% statistical uncertainty per plan were selected. All plans were designed so that the dose limits for the ipsilateral lung and ipsilateral PBT needed to be met first or even superior to the target dose coverage. All plans were rescaled to match the coverage requirement of 95% of the PTV covered by prescription dose and 99% of the ITV covered by prescription dose and to ensure that all hot spots (between 120% and 150%) were located within the ITV. If OARs overlap the PTV, the minimum dose for the PTV and ITV should be at least 70% and 90% of the prescription dose, respectively.
Plan evaluation
Plan quality was evaluated based on different dosimetric indices. PTV was evaluated using D98%, D2%, and Dmean, where D98%, D2%, and Dmean represent the minimum, maximum, and average PTV dose, respectively. According to RTOG 0813 protocol, D2% was < 86.0 Gy at PTV volume = 70 cc, D2% was < 77.0 Gy at PTV volume = 50 cc, and D2% was < 57.0 Gy at PTV volume < 10 cc, which means that D2% decreased with decreasing PTV volume.
The Conformity Index (CI)48 and Heterogeneity Index (HI)49 were used to evaluate high dose spillage and target dose homogeneity between the 2-group plans. They were referring to the RTOG 0813 protocol and plan quality. CI = PIV/TV, where PIV is the prescription isodose volume, and TV is the target volume. HI = D5%/D95%, where D95% and D5% are the minimum doses to the hottest 95% and 5% of the PTV. The values of CI and HI are ≥ 1, where CI closer to 1 is better, and HI < 1.6 is sufficient. According to RTOG 0813 protocol, CI should be < 1.5 and preferably < 1.2.
D2cm and R50% were used to assess intermediate dose spillage for both groups. Where D2cm records the maximum dose at any point ≥ 2 cm from the PTV in any direction, R50% represents the ratio of the volume encircled by the 50% prescription isodose line to the PTV volume, with R50% increasing as the PTV volume decreases. According to RTOG 0813 protocol, R50% was < 4.8 at PTV volume = 70 cc, R50% was < 5.0 at PTV volume = 50 cc, and R50% was < 7.5 at PTV volume < 10 cc. The planning goals of targets are shown in Table 1.
The OARs were compared in compliance with the requirements of RTOG 0813 protocol and other literature50,51, including lung V5Gy, V20Gy, spinal cord V22.5 Gy and V13.5 Gy, ipsilateral PBT V18Gy, esophagus V27.5 Gy, heart V32Gy, great vessels V47Gy, ipsilateral brachial plexus V30Gy, and skin V30Gy, and Dmax for all OARs except lungs. The planning goals of OARs are shown in Table 2.
Plan complexity was assessed using segments and MUs, which positively correlate with plan complexity52. BOT was also recorded for the 2-group plans when measuring the γ passing rates using SRS MapCHECK (equipped with the StereoPHAN module) (Sun Nuclear, Melbourne, FL). Plan delivery accuracy was expressed using the γ passing rates. Based on the AAPM TG 218 report and other literature53,54 and considering the actual situation in the department, the qualified standards of the γ passing rates under different acquisition parameters are shown in Table 3. The acquisition parameters were 2%/2 mm, 2%/1 mm, and 1%/2 mm, excluding data below 10% of the maximum dose.
To evaluate the advantages and disadvantages of DCAT and VMAT plans at different PTV volumes, the DCAT/VMAT ratios were calculated for some of the plan quality parameters, and ratios less than 1 indicate that the corresponding parameters of the DCAT plans are better than those of the VMAT plans, as shown in Table 4; Fig. 5.
Statistical analysis
Statistical analyses were performed by comparing the novel DCAT with VMAT using the paired nonparametric Wilcoxon signed-rank test (IBM SPSS Statistics for Windows, Version 23.0; IBM Corp., Armonk, NY, USA). Data were presented as Median (IQR).
Spearman’s rank correlation coefficient (denoted as ρ) was used to assess correlations between DCAT/VMAT ratios and the PTV volume. The correlation coefficient (ρ) ranges from − 1 to 1, with absolute values closer to 1 indicating a stronger correlation. A 95% confidence interval was applied, and a two-tailed P < 0.05 was considered statistically significant.
Results
Table 1 summarizes the statistical analyses of the dosimetric parameters of the 2-group plans for targets (mainly the PTV), and Fig. 1 compares selected target parameters for the 2-group plans. When normalizing all plans to 100% of the prescribed dose covering 95% of the PTV, they all met the dose limits described in the RTOG 0813 protocol. The DCAT group had more considerable high and intermediate dose spillage than the VMAT group, as evidenced by a more significant CI (1.15 vs. 1.13, P < 0.05) than the VMAT group and greater D2cm (52.18 Gy vs. 50.68 Gy, P < 0.001) and R50% (4.86 vs. 4.52, P < 0.001) than the VMAT group but judging from the absolute values, the differences in CI, D2cm, and R50% between the 2-group plans were not that big (exceeding 1.74%, 2.87%, and 6.70%, respectively). The dose homogeneity in PTV was better in the DCAT group, as evidenced by the fact that the HI (1.24 vs. 1.13, P < 0.05) and D2% (62.60 Gy vs. 69.07 Gy, P = 0.001) of the PTV were smaller than those of the corresponding VMAT group, and the D98% (44.54 Gy vs. 43.10 Gy, P < 0.05) of the PTV was bigger than that of the VMAT group. Figure 2 shows the transverse and coronal dose distributions, as well as the DVH for the DCAT plan and the VMAT plan in one case, and the DCAT plan had a more uniform target dose (less volume of 5750 cGy, comparable volume of prescription dose) and comparable OARs dose compared to the VMAT plan.
Comparison of PTV dosimetric parameters (A, CI; B, HI; C, D2cm; D, R50%) between the 2-group plans (the blue triangles represent DCAT plans, and the red squares show VMAT plans). The results show that most HI for DCAT plans were lower than its corresponding VMAT plans. Otherwise, the DCAT plans did not differ significantly in absolute values from the VMAT plans regarding CI, D2cm, and R50%. Abbreviations: PTV planning target volume, CI conformity index, HI homogeneity index, DCAT dynamic conformal arc therapy, VMAT volumetric-modulated arc therapy, D2cm the maximum dose at any point ≥ 2 cm from the PTV in any direction, R50% the ratio of the volume encircled by the 50% prescription isodose line to the PTV volume.
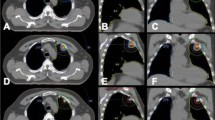
Comparison of DCAT and VMAT plans for a typical patient. The upper and middle panels show isodose distributions for the DCAT (left) and VMAT (right) plans. Comparable isodose distributions were obtained with the DCAT plan. The OARs include ipsilateral lung, contralateral lung, spinal cord, ipsilateral PBT, esophagus, heart, great vessels, ipsilateral brachial plexus, and skin. The lower panel shows the DVH comparison for both plans. The solid line represents the DCAT plan, and the dashed line shows the VMAT plan (red, ITV; blue, PTV; green, ipsilateral lung; yellow, spinal cord; cyan, ipsilateral PBT; pink, heart; carmine, great vessels; other OARs not shown). DCAT plan had a more uniform target dose and comparable OAR dose to the VMAT plan. Abbreviations: ITV internal target volume, PTV planning target volume, PBT proximal bronchial tree, DCAT dynamic conformal arc therapy, VMAT volumetric-modulated arc therapy, DVH dose volume histogram.
Table 2 lists the dosimetric differences in OARs for the 2-group plans. Dosimetric differences for most OARs were insignificant except for the ipsilateral lung, lung all, and the ipsilateral PBT V18Gy. Figure 3 illustrates some of the OARs’ parameters for the 2-group plans. Although dosimetric parameters of some OARs were statistically different (P < 0.001), for example, the median increment of V5Gy in the ipsilateral lung was 8.13% for the DCAT group separately. The median increase of V5Gy in the lung all for the DCAT group was 5.81%. However, combining the information in Table 2; Fig. 3, all dosimetric parameters of the 2-group plans met the RTOG 0813 protocol. Since the distance between the target and the ipsilateral PBT was less than 2 cm, the V18Gy of the ipsilateral PBT in the VMAT group was inferior to that in the DCAT group (P < 0.05). Data for some OARs are not presented in Table 2 (spinal cord V22.5 Gy, V13.5 Gy, esophagus V27.5 Gy, great vessels V47Gy, ipsilateral brachial plexus V30Gy, and skin V30Gy), because most values of them were 0.
Comparison of OARs dosimetric parameters (A, B, ipsilateral lung; C, D, lung all; E, F, ipsilateral PBT) between the 2-group plans (the blue triangles represent DCAT plans, and the red squares show VMAT plans). The results show that despite the above parameters for DCAT plans being more significant than its corresponding VMAT plans, they were still somewhat comparable. Abbreviations: PBT proximal bronchial tree, DCAT dynamic conformal arc therapy, VMAT volumetric-modulated arc therapy.
Regarding the plan complexity, the DCAT group showed a significant reduction in segments and MUs compared to the VMAT group (19.38% reduction in segments on median number and 23.27% reduction in MUs on average, P < 0.05) as shown in Table 3; Fig. 4. The median BOT in the DCAT group (161.00 s) was less than that in the VMAT group (169.80 s) by 8.80 s (P < 0.05). The γ passing rates under different acquisition parameters (2%/2 mm, 2%/1 mm, and 1%/2 mm) showed that both groups had a high dose delivery accuracy. The DCAT group had an apparent advantage under the most stringent acquisition parameters (2%/1 mm, DCAT 93.70% vs. VMAT 91.90%; P < 0.05), with an median increase of 1.80% and a maximum increase of 8.60%.
Comparison of dosimetric parameters (A, Segments; B, MUs; C, BOT; D, γ passing rates) between the 2-group plans (the blue triangles represent DCAT plans, and the red squares show VMAT plans). The results show that the above parameters for DCAT plans were more advantageous than its corresponding VMAT plans. Abbreviations: DCAT dynamic conformal arc therapy, VMAT volumetric-modulated arc therapy, MUs monitor units, BOT beam-on time.
The target (PTV) volume also influences DCAT plan quality. We selected parameters with statistical differences in the plan quality and evaluated the correlation between the corresponding DCAT/VMAT ratios and the target volume. As can be seen in Table 4, only HIDCAT/HIVMAT, R50%DCAT/R50%VMAT, and SegmentsDCAT/SegmentsVMAT had moderately correlations with PTV volume (ρ = −0.498, 0.550, −0.53, P < 0.05). Figure 5 shows a histogram of the statistically significant DCAT/VMAT ratio in relation to PTV volume, where the trend of change in HI is relatively slight and not very significant. Table 4; Fig. 5 show that with the increase in PTV volume, the HI and the segments of the DCAT plans exhibit a weak advantage, and the R50% of the DCAT plans is getting closer and closer to that of the VMAT plans with decreasing PTV volume.
Histogram of DCAT/VMAT ratios (A, HI; B, R50%; C, Segments) versus PTV volume. There was a significant correlation between the DCAT/VMAT ratios of the above parameters and PTV volume (ρ = −0.498, 0.550, −0.53, P < 0.05). Abbreviations: HI homogeneity index, DCAT dynamic conformal arc therapy, VMAT volumetric-modulated arc therapy, PTV planning target volume, R50% the ratio of the volume encircled by the 50% prescription isodose line to the PTV volume.
Discussion
Based on the RTOG 0813 protocol, we compared the plan quality, BOT, and γ passing rates between DCAT plans and VMAT plans in stage I/II centrally located NSCLC patients with PTV volume < 70 cc and analyzed the feasibility of DCAT plans to conduct SBRT treatment (50 Gy/5fx) under certain conditions (PTV volume < 70 cc). The results showed that all evaluation parameters of the DCAT plans met the RTOG 0813 protocol. Compared with the VMAT plans, most target parameters of the DCAT plans were close to their counterparts in the VMAT plans (e.g., CI, D2cm, and R50%, etc.), and some of the target parameters, such as the HI (including D2% and D98%), were superior to those of the VMAT plans. For all patients, the DCAT plans provided similar OARs sparing and were far below the requirements of the RTOG 0813 protocol. The advantage of the DCAT plans is that, due to the low modulation degree, fewer segments and MUs are required to deliver the same prescribed dose55, as shown in Table 3. This results in two benefits: firstly, the BOT was significantly shorter (median reduction of 8.80 s, P < 0.05), and second, the gamma passing rates (2%/1 mm) of the DCAT plans were significantly higher (P < 0.05). These benefits improved patient comfort on the one hand and plan delivery accuracy on the other56,57. As the target volume increased, the advantages of the DCAT plans became more and more significant in HI and segments while gradually inferior to the VMAT plans in R50%.
SBRT treatment of centrally located NSCLC is much more complex than peripheral NSCLC58. The common side effects include fatal radiation pneumonitis in patients with poor respiratory function, particularly those with underlying interstitial lung disease59. Therefore, the doses to healthy lungs need to be strictly controlled. In this paper, the ipsilateral lung parameters (V5Gy, V20Gy) and lung all parameters (V5Gy, V20Gy) in the DCAT group were significantly higher than those in the VMAT group (P < 0.001). Nevertheless, the lung dosimetric indices of both groups were significantly lower than the requirements of the RTOG 0813 protocol. More importantly, significant differences in dosimetry do not necessarily lead to significant clinical organ toxicity, especially when the dosimetric parameters are below safety standards.
Fatal hemoptysis occurs when the trachea and bronchus are irradiated at doses higher than the safety standard60. All targets in this paper were within 1 cm of the trachea and bronchus. However, there was no overlapping volume, so the tracheal and bronchial doses met the safety criteria (referring to Dmax, V18Gy in Table 2). As for fatal esophageal ulcers and brachial plexopathy, both need no discussion because the corresponding OAR doses were too low. The doses for the remaining OARs were well below the RTOG 0813 protocol. Therefore, no acute or late radiotoxicity corresponding to the implementation of the DCAT plans is expected28.
The advantages of the DCAT plans in treating lung cancer are mainly shorter BOT and higher dose delivery accuracy61,62. Therefore, the preferred treatment technique in some radiotherapy centers for SBRT treatment is DCAT63. In this paper, the median reduction of segments in the DCAT group was 19.38%, and the median reduction of MUs was 23.27%, and the differences were statistically significant (P < 0.05). The average BOT in the DCAT group (161.00 s) was less than that in the VMAT group (169.80 s) by 8.80 s (P < 0.05). The segments of the DCAT plans based on the SSO and VDR can move in a short range inside and outside the targets, thus possessing a specific dose modification ability in the targets, which can be regarded as the most uncomplicated VMAT plans26, and thus have both the intensity modulation ability close to that of the VMAT plans, and a simpler dose delivery. The γ passing rates of the 2-group plans were following the AAPM TG 218 report53, as shown in Table 3; Fig. 4. However, the DCAT plans had higher dose delivery accuracy, which was demonstrated by the more stringent γ passing rates acquisition parameters (2%/1 mm, DCAT 93.70% vs. VMAT 91.90%; P < 0.05), with a median γ passing rates improvement of 1.80% and a maximum improvement of 8.60%. This is because all segments of the DCAT plans have regular shapes and large sizes26,27,28,29,34 and do not have extensive blocking of the central target area or large ranges of leaf motion distances, which reduce computational uncertainty and lower the requirement for MLC position accuracy26. In short, the DCAT plans can reduce delivery uncertainties associated with small-field dosimetry errors and interplay effects in small lesions, while simplifying the process of treatment planning41,42,64,65.
The DCAT plans’ performance at different target volumes is also a matter of interest in this paper. It has been reported in the literature66 that DCAT can achieve comparable plan quality to VMAT in early-stage peripheral lung cancer patients with PTV < 95 cc. However, there are rare reports in stage I/II centrally located lung cancer about DCAT plans. As shown in Table 4; Fig. 5, the ratios of HIDCAT/HIVMAT and SegmentsDCAT/SegmentsVMAT became smaller and smaller as the target volume increased, suggesting that the HI and segments of the DCAT plans had an increasing advantage relative to the VMAT plans (P < 0.05); while the R50%DCAT/R50%VMAT ratios were getting bigger and bigger, indicating that the R50% of the DCAT plans had better performance in small target volume (P < 0.05). Given that SBRT therapy requires a faster dose fall-off outside the target volume, DCAT plans appear to have more advantages in small-volume targets.
The application of the DCAT plans in this paper has the following prerequisites. Firstly, the small sample size results in a lack of persuasiveness and significant trends in the statistical analysis of some data, such as the HI. Secondly, the PTV volume is < 70 cc and the target volume dramatically influences the choice between the DCAT and VMAT plans63. Thirdly, the 3DCRT plans also play an important role in lung cancer SBRT therapy67, and a comprehensive comparison of their advantages and disadvantages with arc therapy will be presented in another paper. Finally, although this paper discusses the differences in OAR doses, it does not address the clinical effects resulting from these dose differences.
Conclusions
The results of this paper indicate that the novel DCAT plans have high feasibility in the SBRT treatment of stage I/II centrally located NSCLC patients within a specific range of target volume (PTV < 70 cc). Although VMAT plans have certain advantages in OARs sparing, such differences are not significant in most OARs. Meanwhile, DCAT plans feature shorter BOT and higher γ passing rates, which means DCAT plans can improve patient comfort and the dose delivery accuracy.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- DCAT:
-
dynamic conformal arc therapy
- SSO:
-
segment shape optimization
- VDR:
-
variable dose rate
- NSCLC:
-
non-small-cell lung cancer
- PTV:
-
planning target volume
- VMAT:
-
volumetric-modulated arc therapy
- BOT:
-
beam-on time
- OARs:
-
organs at risk
- Mus:
-
monitor units
- HI:
-
homogeneity index
- R50% :
-
the ratio of the volume encircled by the 50% prescription isodose line to the PTV volume
- SBRT:
-
stereotactic body radiotherapy
- FFF:
-
flattening-filter free
- MLC:
-
multi-leaf collimator
- TPS:
-
treatment planning system
- ITV:
-
internal target volume
- AIP:
-
average intensity projection
- MIP:
-
maximum intensity projection
- XVMC:
-
X-ray Voxelized Monte Carlo
- PBT:
-
proximal bronchial tree
- CI:
-
conformity index
- D2cm :
-
the maximum dose at any point ≥ 2 cm from the PTV in any direction
- AAPM:
-
American Association of Physicists in Medicine
- SD:
-
standard deviation
- DVH:
-
dose volume histogram
- N/A:
-
not applicable
References
Bezjak, A. et al. Safety and efficacy of a Five-Fraction stereotactic body radiotherapy schedule for centrally located Non-Small-Cell lung cancer: NRG Oncology/RTOG 0813 trial. J. Clin. Oncol. 37, 1316–1325. https://doi.org/10.1200/jco.18.00622 (2019).
Senthi, S., Haasbeek, C. J., Slotman, B. J. & Senan, S. Outcomes of stereotactic ablative radiotherapy for central lung tumours: a systematic review. Radiother Oncol. 106, 276–282. https://doi.org/10.1016/j.radonc.2013.01.004 (2013).
Modh, A. et al. Local control and toxicity in a large cohort of central lung tumors treated with stereotactic body radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 90, 1168–1176. https://doi.org/10.1016/j.ijrobp.2014.08.008 (2014).
Mangona, V. S. et al. Toxicity after central versus peripheral lung stereotactic body radiation therapy: a propensity score matched-pair analysis. Int. J. Radiat. Oncol. Biol. Phys. 91, 124–132. https://doi.org/10.1016/j.ijrobp.2014.08.345 (2015).
Videtic, G. M. M. et al. Stereotactic body radiation therapy for early-stage non-small cell lung cancer: executive summary of an ASTRO Evidence-Based guideline. Pract. Radiat. Oncol. 7, 295–301. https://doi.org/10.1016/j.prro.2017.04.014 (2017).
Bang, A. & Bezjak, A. Stereotactic body radiotherapy for centrally located stage I non-small cell lung cancer. Transl Lung Cancer Res. 8, 58–69. https://doi.org/10.21037/tlcr.2018.10.07 (2019).
Kepka, L. & Socha, J. Dose and fractionation schedules in radiotherapy for non-small cell lung cancer. Transl Lung Cancer Res. 10, 1969–1982. https://doi.org/10.21037/tlcr-20-253 (2021).
Chang, J. Y., Bezjak, A. & Mornex, F. Stereotactic ablative radiotherapy for centrally located early stage non-small-cell lung cancer: what we have learned. J. Thorac. Oncol. 10, 577–585. https://doi.org/10.1097/jto.0000000000000453 (2015).
Davis, J. N. et al. Stereotactic body radiotherapy for centrally located early-stage non-small cell lung cancer or lung metastases from the RSSearch(®) patient registry. Radiat. Oncol. 10, 113. https://doi.org/10.1186/s13014-015-0417-5 (2015).
Hamilton, T. et al. Lung SBRT treatment planning: a study of VMAT Arc selection guided by collision check software. Med. Dosim. 48, 82–89. https://doi.org/10.1016/j.meddos.2023.01.003 (2023).
Ye, W. et al. Dosimetric investigation of couch rotation angles in non-coplanar VMAT plans for lung cancer SBRT. Front. Oncol. 14, 1454676. https://doi.org/10.3389/fonc.2024.1454676 (2024).
Otto, K. Volumetric modulated Arc therapy: IMRT in a single gantry Arc. Med. Phys. 35, 310–317. https://doi.org/10.1118/1.2818738 (2008).
Ong, C. L. et al. Stereotactic radiotherapy for peripheral lung tumors: a comparison of volumetric modulated Arc therapy with 3 other delivery techniques. Radiother Oncol. 97, 437–442. https://doi.org/10.1016/j.radonc.2010.09.027 (2010).
Dobler, B. et al. Re-irradiating spinal column metastases using IMRT and VMAT with and without flattening filter - a treatment planning study. Radiat. Oncol. 11, 33. https://doi.org/10.1186/s13014-016-0603-0 (2016).
Nguyen, D. et al. A comprehensive formulation for volumetric modulated Arc therapy planning. Med. Phys. 43, 4263. https://doi.org/10.1118/1.4953832 (2016).
Pigorsch, S. U. et al. Report on planning comparison of VMAT, IMRT and helical tomotherapy for the ESCALOX-trial pre-study. Radiat. Oncol. 15, 253. https://doi.org/10.1186/s13014-020-01693-2 (2020).
Vanetti, E. et al. Volumetric modulated Arc radiotherapy for carcinomas of the oro-pharynx, hypo-pharynx and larynx: a treatment planning comparison with fixed field IMRT. Radiother Oncol. 92, 111–117. https://doi.org/10.1016/j.radonc.2008.12.008 (2009).
Jensen, C. A., Roa, A. M. A., Johansen, M., Lund, J. & Frengen, J. Robustness of VMAT and 3DCRT plans toward setup errors in radiation therapy of locally advanced left-sided breast cancer with DIBH. Phys. Med. 45, 12–18. https://doi.org/10.1016/j.ejmp.2017.11.019 (2018).
Sapkaroski, D., Osborne, C. & Knight, K. A. A review of stereotactic body radiotherapy - is volumetric modulated Arc therapy the answer? J. Med. Radiat. Sci. 62, 142–151. https://doi.org/10.1002/jmrs.108 (2015).
Wolfs, C. J. A., Swinnen, A. C. C., Nijsten, S. & Verhaegen, F. Should dose from small fields be limited for dose verification procedures? Uncertainty versus small field dose in VMAT treatments. Phys. Med. Biol. 63, 20nt01. https://doi.org/10.1088/1361-6560/aae338 (2018).
Leste, J. et al. A study of the interplay effect for VMAT SBRT using a four-axes motion Phantom. J. Appl. Clin. Med. Phys. 21, 208–215. https://doi.org/10.1002/acm2.12947 (2020).
Edvardsson, A., Nordström, F., Ceberg, C. & Ceberg, S. Motion induced interplay effects for VMAT radiotherapy. Phys. Med. Biol. 63, 085012. https://doi.org/10.1088/1361-6560/aab957 (2018).
Masi, L., Doro, R., Favuzza, V., Cipressi, S. & Livi, L. Impact of plan parameters on the dosimetric accuracy of volumetric modulated Arc therapy. Med. Phys. 40, 071718. https://doi.org/10.1118/1.4810969 (2013).
Ong, C. L., Dahele, M., Slotman, B. J. & Verbakel, W. F. Dosimetric impact of the interplay effect during stereotactic lung radiation therapy delivery using flattening filter-free beams and volumetric modulated Arc therapy. Int. J. Radiat. Oncol. Biol. Phys. 86, 743–748. https://doi.org/10.1016/j.ijrobp.2013.03.038 (2013).
Hrbacek, J., Lang, S., Graydon, S. N., Klöck, S. & Riesterer, O. Dosimetric comparison of flattened and unflattened beams for stereotactic ablative radiotherapy of stage I non-small cell lung cancer. Med. Phys. 41, 031709. https://doi.org/10.1118/1.4866231 (2014).
Clements, M., Schupp, N., Tattersall, M., Brown, A. & Larson, R. Monaco treatment planning system tools and optimization processes. Med. Dosim. 43, 106–117. https://doi.org/10.1016/j.meddos.2018.02.005 (2018).
Lee, S. et al. Dosimetric benefits of dynamic conformal Arc therapy-combined with active breath-hold in lung stereotactic body radiotherapy. Med. Dosim. 47, 54–60. https://doi.org/10.1016/j.meddos.2021.08.004 (2022).
Mesny, E. et al. Clinical outcomes and lung toxicities after lung SABR using dynamic conformal Arc therapy: a single-institution cohort study. Radiat. Oncol. 18, 36. https://doi.org/10.1186/s13014-023-02227-2 (2023).
Gauer, T., Sothmann, T., Blanck, O., Petersen, C. & Werner, R. Under-reported dosimetry errors due to interplay effects during VMAT dose delivery in extreme hypofractionated stereotactic radiotherapy. Strahlenther Onkol. 194, 570–579. https://doi.org/10.1007/s00066-018-1264-8 (2018).
Ku, E. et al. SU-F‐T‐112: long‐term follow‐up of NSCLC patients treated with lung SBRT using the modified conformal Arc (MDCA) planning technique. Med. Phys. 43, 3487–3487 (2016).
Bokrantz, R., Wedenberg, M. & Sandwall, P. Dynamic conformal Arcs for lung stereotactic body radiation therapy: A comparison with volumetric-modulated Arc therapy. J. Appl. Clin. Med. Phys. 21, 103–109. https://doi.org/10.1002/acm2.12800 (2020).
Piermattei, A. et al. Dynamic conformal Arc therapy: transmitted signal in vivo dosimetry. Med. Phys. 35, 1830–1839. https://doi.org/10.1118/1.2900718 (2008).
Lee, J. N. et al. A dosimetric comparison of 3D DCAT vs VMAT for palliative and early-stage liver lesions using eclipse TPS. Med. Dosim. 47, 252–257. https://doi.org/10.1016/j.meddos.2022.04.002 (2022).
Kim, S. et al. Negative margin technique - a novel planning strategy to improve dose conformation in SBRT using dynamic conformal Arc delivery. J. Appl. Clin. Med. Phys. 14, 79–89. https://doi.org/10.1120/jacmp.v14i5.4283 (2013).
Vassiliev, O. N. et al. Radiotherapy of lung cancers: FFF beams improve dose coverage at tumor periphery compromised by electronic disequilibrium. Phys. Med. Biol. 63, 195007. https://doi.org/10.1088/1361-6560/aadf7d (2018).
Nakano, H. et al. Radiobiological effects of flattening filter-free photon beams on A549 non-small-cell lung cancer cells. J. Radiat. Res. 59, 442–445. https://doi.org/10.1093/jrr/rry041 (2018).
Pokhrel, D., Halfman, M. & Sanford, L. FFF-VMAT for SBRT of lung lesions: improves dose coverage at tumor-lung interface compared to flattened beams. J. Appl. Clin. Med. Phys. 21, 26–35. https://doi.org/10.1002/acm2.12764 (2020).
Benedict, S. H. et al. Stereotactic body radiation therapy: the report of AAPM task group 101. Med. Phys. 37, 4078–4101. https://doi.org/10.1118/1.3438081 (2010).
Vassiliev, O. N. et al. Dosimetric properties of photon beams from a flattening filter free clinical accelerator. Phys. Med. Biol. 51, 1907–1917. https://doi.org/10.1088/0031-9155/51/7/019 (2006).
Tambe, N., Fryer, A., Marsden, J., Moore, C. & Beavis, A. Determination of clinically appropriate flattening filter free (FFF) energy for treating lung SABR using treatment plans and delivery measurements. Biomedical Phys. Eng. Express. 2, 065016 (2016).
Ross, C. C. et al. A novel modified dynamic conformal Arc technique for treatment of peripheral lung tumors using stereotactic body radiation therapy. Pract. Radiat. Oncol. 1, 126–134 (2011).
Pokhrel, D., Visak, J. & Sanford, L. A novel and clinically useful dynamic conformal Arc (DCA)-based VMAT planning technique for lung SBRT. J. Appl. Clin. Med. Phys. 21, 29–38. https://doi.org/10.1002/acm2.12878 (2020).
Timmerman, R. et al. Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J. Clin. Oncol. 24, 4833–4839. https://doi.org/10.1200/jco.2006.07.5937 (2006).
Raman, S. et al. Ultracentral tumors treated with stereotactic body radiotherapy: Single-Institution experience. Clin. Lung Cancer. 19, e803–e810. https://doi.org/10.1016/j.cllc.2018.06.001 (2018).
Lindberg, K. et al. The HILUS-Trial-a prospective nordic multicenter phase 2 study of ultracentral lung tumors treated with stereotactic body radiotherapy. J. Thorac. Oncol. 16, 1200–1210. https://doi.org/10.1016/j.jtho.2021.03.019 (2021).
Videtic, G. M. et al. A randomized phase 2 study comparing 2 stereotactic body radiation therapy schedules for medically inoperable patients with stage I peripheral Non-Small cell lung cancer: NRG oncology RTOG 0915 (NCCTG N0927). Int. J. Radiat. Oncol. Biol. Phys. 93, 757–764. https://doi.org/10.1016/j.ijrobp.2015.07.2260 (2015).
Potter, N. J. et al. Beam flatness modulation for a flattening filter free photon beam utilizing a novel direct leaf trajectory optimization model. J. Appl. Clin. Med. Phys. 21, 142–152. https://doi.org/10.1002/acm2.12837 (2020).
Shaw, E. et al. Radiation therapy oncology group: radiosurgery quality assurance guidelines. Int. J. Radiat. Oncol. Biol. Phys. 27, 1231–1239. https://doi.org/10.1016/0360-3016(93)90548-a (1993).
Ronaldson, J. P., Bennett, H., Roberts, J., Ronaldson, A. J. & Cousins, A. T. A dosimetric comparison of flattening filter free and conventional VMAT treatments for some common cancer sites. Phys. Eng. Sci. Med. 43, 719–725. https://doi.org/10.1007/s13246-020-00877-0 (2020).
Xhaferllari, I., Chen, J. Z., MacFarlane, M., Yu, E. & Gaede, S. Dosimetric planning study of respiratory-gated volumetric modulated Arc therapy for early-stage lung cancer with stereotactic body radiation therapy. Pract. Radiat. Oncol. 5, 156–161. https://doi.org/10.1016/j.prro.2014.08.009 (2015).
Raman, S. et al. Ultracentral tumors treated with stereotactic body radiotherapy: single-institution experience. Clin. Lung Cancer. 19, e803–e810 (2018).
Fernandez, D. J., Sick, J. T. & Fontenot, J. D. Interplay effects in highly modulated stereotactic body radiation therapy lung cases treated with volumetric modulated Arc therapy. J. Appl. Clin. Med. Phys. 21, 58–69. https://doi.org/10.1002/acm2.13028 (2020).
Miften, M. et al. Tolerance limits and methodologies for IMRT measurement-based verification QA: recommendations of AAPM task group 218. Med. Phys. 45, e53–e83. https://doi.org/10.1002/mp.12810 (2018).
Pulliam, K. B. et al. Comparison of 2D and 3D gamma analyses. Med. Phys. 41, 021710. https://doi.org/10.1118/1.4860195 (2014).
Peterlin, P., Stanič, K., Méndez, I. & Strojnik, A. Treating lung cancer with dynamic conformal Arc therapy: a dosimetric study. Radiat. Oncol. 12, 93. https://doi.org/10.1186/s13014-017-0823-y (2017).
Mittauer, K. et al. A study of IMRT planning parameters on planning efficiency, delivery efficiency, and plan quality. Med. Phys. 40, 061704. https://doi.org/10.1118/1.4803460 (2013).
Shiba, E. et al. Predictive gamma passing rate by dose uncertainty potential accumulation model. Med. Phys. 46, 999–1005. https://doi.org/10.1002/mp.13333 (2019).
Li, Y. et al. Dosimetric comparison between IMRT and VMAT in irradiation for peripheral and central lung cancer. Oncol. Lett. 15, 3735–3745. https://doi.org/10.3892/ol.2018.7732 (2018).
Nagata, Y. & Kimura, T. Stereotactic body radiotherapy (SBRT) for stage I lung cancer. Jpn J. Clin. Oncol. 48, 405–409. https://doi.org/10.1093/jjco/hyy034 (2018).
Corradetti, M. N., Haas, A. R. & Rengan, R. Central-airway necrosis after stereotactic body-radiation therapy. N Engl. J. Med. 366, 2327–2329. https://doi.org/10.1056/NEJMc1203770 (2012).
Sande, E. P. S., Roa, A., Hellebust, T. P. & A. M. & Dose deviations induced by respiratory motion for radiotherapy of lung tumors: impact of CT reconstruction, plan complexity, and fraction size. J. Appl. Clin. Med. Phys. 21, 68–79. https://doi.org/10.1002/acm2.12847 (2020).
Ge, C. et al. Effect of plan complexity on the dosimetry, delivery accuracy, and interplay effect in lung VMAT SBRT with 6 MV FFF beam. Strahlenther Onkol. 198, 744–751. https://doi.org/10.1007/s00066-022-01940-3 (2022).
Vieillevigne, L., Bessieres, S., Ouali, M. & Lanaspeze, C. Dosimetric comparison of flattened and unflattened beams for stereotactic body radiation therapy: impact of the size of the PTV on dynamic conformal Arc and volumetric modulated Arc therapy. Phys. Med. 32, 1405–1414. https://doi.org/10.1016/j.ejmp.2016.10.007 (2016).
Shi, C., Tazi, A., Fang, D. X. & Iannuzzi, C. Implementation and evaluation of modified dynamic conformal Arc (MDCA) technique for lung SBRT patients following RTOG protocols. Med. Dosim. 38, 287–290. https://doi.org/10.1016/j.meddos.2013.02.010 (2013).
Pokhrel, D., Halfman, M. & Sanford, L. A simple, yet novel hybrid-dynamic conformal Arc therapy planning via flattening filter-free beam for lung stereotactic body radiotherapy. J. Appl. Clin. Med. Phys. 21, 83–92. https://doi.org/10.1002/acm2.12868 (2020).
Liu, H. et al. Dosimetric comparison of two arc-based stereotactic body radiotherapy techniques for early-stage lung cancer. Med. Dosim. 40, 76–81. https://doi.org/10.1016/j.meddos.2014.10.004 (2015).
Cai, J., Malhotra, H. K., Orton, C. G. & Point/Counterpoint A 3D-conformal technique is better than IMRT or VMAT for lung SBRT. Med. Phys. 41, 040601. https://doi.org/10.1118/1.4856175 (2014).
Acknowledgements
This work was supported by the National Natural Science Foundation of China, (no.11965001), Henan Province Science and Technology Development Project (no.232102310091), Henan Province Medical Science and Technology Project (no. LHGJ20210407), Key Scientific Research Project of Colleges and Universities in Henan Province (no. 25B320023).
Author information
Authors and Affiliations
Contributions
YYH and JY conceived this study. YYH, JY, CW, JHY, GH, CZL and YBL reviewed and checked the data of radiotherapy and dosimetric parameters. YYH, JY, CW, JHY and GH collected and organized the data. YYH, CW, JHY and GH analyzed the data and wrote the paper. All authors reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, Y., Yang, J., Wang, C. et al. Novel DCAT plans in stereotactic body radiotherapy for stage I/II centrally located non-small-cell lung cancer. Sci Rep 16, 5197 (2026). https://doi.org/10.1038/s41598-026-35713-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-35713-0