Abstract
Extended-spectrum β-lactamases-producing Escherichia coli (ESBL-EC) pose a serious threat. Moreover, widespread antimicrobial use in Egypt increased the prevalence of antimicrobial resistance (AMR). In this study, whole-genome sequencing (WGS) using the Illumina NovaSeq 6000 was performed on two isolates (UPE7 and UPE139) recovered from participants with urinary tract infections to characterize their resistomes and virulomes. Antibiotic resistance and virulence genes of the two clinical E. coli strains were predicted using computational analysis tools. Several virulence traits and antibiotic resistance genes (ARGs) were identified. Strain UPE7 harbored blaTEM−1B, blaCTM−X−15, blaCMY−2, and strain UPE139 revealed the presence of blaOXA−244, blaTEM−12, blaTEM−82, and blaCTM−X−15 rending the resistance phenotype. The presence of mobile genetic elements adjacent to ARGs thereby suggests their potential for dissemination through horizontal gene transfer. Furthermore, the serotyping in silico investigation revealed that E. coli UPE7 and UPE139 serotypes were O8:H9 and O9:H30, respectively. Notably, key mutations in the gyrA, parC, and parE genes were predicted, consistent with their confirmed resistance to levofloxacin. These findings emphasize the importance of genomic surveillance to guide antimicrobial therapy and monitor emerging high-risk clones, and they support the need for larger-scale genomic studies to improve epidemiological understanding and clinical relevance.
Similar content being viewed by others
Introduction
Antibiotic resistance (AMR) is a major concern in modern healthcare since it possesses the potential to spread worldwide and has limited treatment options1. Poverty, poor sanitation, easily available antibiotics without prescription, and ignorant clinical practice are factors that aid in the spread of multidrug-resistant (MDR) microbial strains in community and hospital settings2. In addition, antibiotics are extensively employed in agriculture to enhance crop yields and in animal husbandry, particularly in cattle farming, where they are administered for therapeutic, prophylactic, and metaphylactic purposes, as well as at controlled concentrations in animal feed as growth promoters to prevent disease and safeguard animal health3,4. Therefore, the extensive use of antibiotics outside human medicine has accelerated the emergence and dissemination of MDR bacteria, representing an escalating threat to both animal and public health4. New examples of AMR are regularly reported, and the time needed for bacteria to become resistant to newly introduced antibiotics is getting shorter5.
Escherichia coli was listed among the World Health Organization (WHO) priority organisms that cause critical infections and require the formulation of new treatments6. Remarkably, the resistance of E. coli strains to third-generation antibiotics is mainly associated with the production of extended-spectrum β-lactamases (ESBLs) globally7. In the last decade, ESBL-EC infections have become challenging because of the rapid pandemic spread of antimicrobial-resistant clones and limited treatment options that put the patient’s life in danger8,9. Furthermore, ESBL-EC is often linked to an increased incidence of nosocomial infections in hospitals10. E. coli isolates may carry multiple antibiotic resistance genes carried on mobile genetic elements (MGEs), including plasmids, transposons, integrons, and insertion sequences (IS), which can spread to compatible neighboring species via horizontal gene transfer (HGT)11,12. In addition, E. coli harbors diverse virulence factors (adhesion-associated fimbriae, hemolysin, siderophores, flagella, capsular polysaccharides, and outer membrane proteins) that contribute to distinct aspects of pathogenicity13. The combined action of these determinants enables the bacterium to establish infection, persist with the host, and evade defense mechanisms14.
Whole genome sequencing (WGS) has emerged as a feasible approach for detecting antibiotic resistance. Indeed, comprehensive genetic analysis via next-generation sequencing (NGS) enables high-resolution genotyping of clinically significant MDR strains and facilitates targeted antibiotic usage directly guided by genetic sequence15. In addition, NGS can study the evolutionary relationship of MDR strains from different geographical regions. Moreover, it is a helpful tool for tracking MGEs, identifying emerging resistance mechanisms, and uncovering chromosomal mutations10,16.
In this study, two MDR E. coli strains, UPE7 and UPE139, were isolated from patients with urinary tract infections (UTIs) at Mansoura University Hospital (Egypt). To characterize the two uropathogenic E. coli strains in-depth, WGS using Illumina sequencing was conducted. Additionally, MLST, serotyping, and phylotyping were used to investigate the clonal relatedness of the isolates. Genome assemblies of UPE7 and UPE139 were screened for the presence of ARGs, MGEs, and virulence factors on the genomic level.
Results
Antimicrobial resistance profile
The two isolates, UPE7 and UPE139, were identified from patients with UTI in Egypt and revealed extensive resistance patterns (Table 1). Among aminoglycosides, both isolates exhibited high-level resistance to gentamicin (≥ 32 µg/mL for UPE7; ≥16 µg/mL for UPE139) and amikacin (≥ 64 µg/mL for both). UPE7 and UPE139 strains were resistant to penicillins and β-lactams, including ampicillin-sulbactam, cefoperazone-sulbactam, and piperacillin-tazobactam, as well as extended-spectrum cephalosporins such as ceftazidime, cefotaxime, and cefepime. Carbapenems, including ertapenem, imipenem, and meropenem, also demonstrated elevated MIC values, confirming carbapenem resistance in both isolates. Resistance extended to fluoroquinolones, with high MIC values reported to levofloxacin (≥ 16 µg/mL for UPE7; ≥8 µg/mL for UPE139). Additionally, strain UPE139 revealed intermediate resistance to chloramphenicol, whereas polymyxin B exhibited resistance in UPE7 (8 µg/mL) but intermediate resistance in UPE139. Both strains were resistant to the sulfonamide antibiotic, trimethoprim-sulfamethoxazole.
General genomic features of E. coli strains UPE7 and UPE139
According to VITEK biochemical identification and genomic characterization, the two isolates were identified as Escherichia coli. The predicted genome sizes were 4,828,053 bp and 5,096,432 bp for UPE7 and UPE139, respectively. The G + C contents for both isolates were about 50.6%. Moreover, WGS yielded 681,534 and 992,028 reads for UPE7 and UPE139, respectively. Genomic features, including coding sequence (CDS), rRNA, tRNA, and CRISPR array numbers, are listed in Table 2. The whole genome shotgun projects of UPE7 and UPE139 were submitted to the National Biotechnology Information Centre (NCBI) GenBank database with the accessions JBHWTZ000000000 and JBHWUA000000000, respectively.
Determination of serotypes, sequence type, and phylogroup
In silico serotyping identified E. coli UPE7 as O8:H9, based on the presence of wzt and wzy alleles (O8) and the fliC gene (H9). Similarly, UPE139 was assigned to O9:H30, carrying wzm and wzt alleles (O9) and the fliC gene (H30). Furthermore, MLST allelic analysis was performed based on the allelic combination of seven genes, and the results revealed no clonal relationship between the two isolates assigned to different sequence types. Based on the Pasteur (Achtman) scheme, the sequence type assigned the isolates UPE7 and UPE139 to ST692 (ST410) and ST650 (ST361), respectively. In addition, the Clermont E. coli phylotyping assigned UPE7 and UPE139 to phylogroup C (fdm, trpA, gpC, ybgD) and phylogroup A (fdm, cfaB, ybgD), respectively.
Detection of virulence factors
Screening the uropathogenic E. coli strains UPE7 and UPE139 genome sequences revealed the occurrence of a diverse array of virulence genes associated with adhesion, host cell invasion, iron acquisition, and persistence in the urinary tract. Strain UPE7 carried genes encoding fimbrial and adhesion factors, including fimH (type 1 fimbriae) and csgA (curlin major subunit CsgA), fdeC (intimin-like adhesion FdeC), IpfA (long polar fimbriae), and the yehABCD fimbrial cluster, which enhance adhesion and biofilm formation. Additionally, hlyE (hemolysin) and hha (hemolysin express modulator), nlpI (lipoprotein precursor), and terC (tellurium ion resistance protein). In comparison, UPE139 harbored several overlapping factors, including fimH, csgA, fdeC, nlpI, hylE, and the yehABC fimbrial cluster, but also possessed additional determinants, capU (hexosyltransferase homolog linked to capsule synthesis), shiA (associated with the Shigella SHI-2 pathogenicity island), sitABCD (iron transport), tia (Tia invasion determinant), and traT (outer membrane protein complement resistance).
Detection of antimicrobial resistance genes (ARGs)
Several ARGs conferring resistance to multiple antibiotic classes were identified in the UPE7 and UPE139 genome sequences, demonstrating a correlation between phenotypic resistance and underlying genetic determinants. The observed resistance to cephalosporins and β-lactams was confirmed by detection of ARGs, including blaTEM−1B, blaCMY−2, and blaCTM−X−15 in the UPE7 isolate, as well as blaTEM−12, blaTEM−82, and blaCTM−X−15 in UPE139. Moreover, high-level aminoglycoside resistance in both isolates was explained by the presence of aminoglycoside genes such as aph(3’’)-Ib, aph(6)-Id, and aadA5. Resistance to tetracyclines, quinolones, macrolides, sulfonamides, and trimethoprim was consistent with the detection of tetA, tetD, tetR, qnrS1, mphA, mdfA, sul1, sul2, and dfrA17 in both genome sequences. In addition, the resistance of the UPE139 strain to carbapenems was associated with the occurrence of blaOXA−244, a member of the OXA-48-like carbapenemase, whereas no carbapenemase resistance genes were detected in the UPE7 genome sequence. In particular, resistance extended to fluoroquinolones is related to mutations in quinolone resistance-determining regions (gyrA, parC, parE, qnr). In addition, both strains revealed regulatory and efflux-associated genes such as acrR, mdtK, and marR, which reduce intracellular quinolone concentrations through active efflux. The presence of the arnABCDTEF operon in both UPE7 and UPE139 was associated with resistance to polymyxin B (Fig. 1; Table 3).
Antimicrobial resistance genes predicted from the whole-genome sequence data of (A) E. coli UPE7 and (B) E. coli UPE139 using CARD-RGI (https://card.mcmaster.ca/analyze/rgi). ARO represents the Antibiotic Resistance Ontology, and the surrounding colored segments represent the detection confidence level of AMR genes. Green color (Perfect) indicates AMR genes perfectly matching reference sequences in the database. Yellow color (Strict) indicates that AMR genes are closely related to known AMR, with some variations. Red (Loose) indicated low-confidence matches.
The analysis of the UPE7 genome revealed the presence of ESBL genes near mobile genetic elements, such as insertion sequences and transposons. The investigation revealed that blaTEM−1B is located between the TnpA transposon. Moreover, the blaCMY−2-containing region revealed the occurrence of this gene chromosomally within the cassette comprising ISEcp1-blaCMY−2-orf-IS200-orf-orf-IS3. In addition, the blaCTX−M−15 gene was bracketed by transposon TnpA and the insertion sequence IS6 (Fig. 2A). Meanwhile, the analysis of ESBL-related genes in the UPE139 genome sequence revealed the presence of the blaCTX−M−15 gene associated with transposon TnpA and IS2. Notably, blaOXA−244 was positioned between two insertion sequences (IS1). Moreover, blaTEM−12 and blaTEM−82 were found to be associated with transposon Tn3 and insertion sequence IS6, respectively (Fig. 2B). Therefore, the WGS-based resistance profile confirmed that UPE7 and UPE139 were multidrug-resistant strains.
Schematic representation of ARGs and genetic mobile elements detected in (A) UPE7 and (B) UPE139. The colors represented: ARGs (Red), Transposase-related genes (green), Insertion sequence elements (Blue, Yellow, Orange).
Investigating chromosomal mutations
In this study, the UPE7 and UPE139 isolates were phenotypically resistant to levofloxacin, a fluoroquinolone antibiotic. Therefore, the chromosomal mutations in DNA gyrase (gyrA) and topoisomerase IV (parC and parE) were investigated. Notably, two types of gyrA mutations (S83L and D87N), one parC mutation (S80I), and one parE mutation (S458A) were detected in UPE7 and UPE139 genome sequences.
Prediction of mobile genetic elements (MGEs)
Screening MGEs conferred the presence of numerous genes associated with the mobilization of ARGs in UPE7 and UPE139 strains. E. coli UPE7 genome sequence exhibited plasmid replicons, namely Col(BS512), IncFIA, IncFIB (AP001918), IncFII (pAMA1167-NDM-5) and IncQ1. Moreover, the UPE139 genome sequence revealed the presence of plasmid replicons: IncFII, IncFIA, IncFIB, IncQ1, and IncY. Plasmid-mediated quinolone resistance (PMQR) gene qnrS1 and ESBL gene blaCTX−M−15 were present in the IncFII plasmid in UPE7 and UPE139 genomes. In addition, several IS and transposable elements were detected in the two strains. UPE7 and UPE139 genome sequences revealed the presence of Insertion sequences ISEcp1, belonging to the IS1380 family transposase, and IS200/IS605. Furthermore, IS3 and IS6 were found at a relatively high frequency in the genomes of the two isolates.
Comparative analysis
A comparative genomic analysis involving UPE7, UPE139, and ten E. coli strains isolated from patients in Egypt was performed (Fig. 3). The genomic analyses of the annotated sequenced genomes revealed a variety of phylogroups, serotypes, and distributions of antibiotic resistance genes. The strains demonstrated combinations of ESBL genes, including the most predominant gene blaCTX−M15, and carbapenem genes such as blaNDM−1 and blaNDM−5. Notably, the four strains in this study harbored the blaCMY−2 gene, and one strain harbored the blaOXA−244 gene. The studied strains revealed the occurrence of one or more aminoglycoside genes. Most of the investigated strains revealed the presence of the macrolide resistance genes (mphA and mdfA), which confer macrolide resistance. Moreover, most of the isolates showed quinolone resistance-determining regions (gyrA, parC, parE), as well as the presence of qnrS1, which confers plasmid-mediated quinolone resistance.
Heatmap represents antibiotic resistance genes in E. coli UPE7, E. coli UPE139, and ten selected uropathogenic E. coli strains using the ARG detection method available through the BV-BRC web server. The purple and yellow colors represent present and absent genes, respectively.
Pan-genome analysis revealed 5,367 orthologous gene clusters identified across the analyzed E. coli genome sequences, of which 3,352 shared orthologous gene clusters were identified among the E. coli strains under investigation (Fig. 4A). Notably, some gene families are involved in critical biological processes. Furthermore, the phylogenetic tree based on whole-genome sequencing revealed the close relation between UPE139 and HR135 (accession number QXNW00000000), a phylogroup A strain, which was previously isolated from UTI in Egypt. Moreover, UPE7 was clustered with SM_2771 (accession number JAJKQA000000000) and K12 strain 71 (accession number SSTS00000000), belonging to phylogroup C and sequence type ST692 (Fig. 4B). E. coli strains within the same phylogroup displayed varied levels of ARGs.
(A) Upset figure showing the common genes in UPE7, UPE139, and other selected E. coli genomes, indicating the number of shared and unique protein clusters and unique genes. (B) Whole genome-based phylogenetic tree of UPE7 and UPE139 isolates, constructed based on the presence/absence of orthologous gene clusters identified using the pan genome analysis tool.
Discussion
According to the WHO, ESBL-producing Enterobacteriaceae pose a significant risk to human health17. Therefore, pathogen surveillance using genomic approaches helps to understand the dynamics, and the results are important for controlling the spread of resistant bacteria18. Consequently, identifying resistance traits through WGS is significant in improving diagnosis and innovative regimes for treating patients with UTI19,20. In the current study, the resistance genotypes of UPE7 and UPE139 strains corresponded to the phenotypic resistance of antibiotics under investigation, reflecting the correlation between the high resistance rate and the frequent prescription of these antibiotics in the treatment of UTI in Egyptian healthcare21.
In Egypt, several findings have demonstrated a higher prevalence of AMR among carbapenem-resistant and ESBL-EC, highlighting the significance of understanding the molecular mechanisms of resistance22. The rapid emergence of MDR strains of E. coli is a serious concern in developing countries. These high observed rates in cephalosporins and carbapenemes are attributed to antibiotic misuse and extensive usage of third-generation antimicrobials, particularly in Egyptian hospitals23,24. Furthermore, various factors, including poor hygiene, antibiotics without prescription, and a lack of rapid diagnostic methods, promote the emergence of these ESBL-producing isolates25.
The sequence type UPE7 was ST410, according to Achtman’s scheme. This sequence type was previously identified in E. coli EC13655, isolated from a hospitalized patient in Egypt21. Furthermore, the uropathogenic E. coli NGCE33 strain (ST410 clone) containing an array of β-lactamase genes was previously detected in Bangladesh26. E. coli of the ST410 lineage is an emerging MDR high-risk clone that has shown an increasing worldwide spread and revealed non-susceptibility to carbapenems, cephalosporins, fluoroquinolones, aminoglycosides, and tetracyclines27,28. However, UPE139 was ST361, according to Achtman’s scheme. A previous study revealed that E. coli ST361 was a key player in the dissemination of carbapenem resistance in France, due to the chromosomal localization of blaOXA−24429. In addition, the clone ST361 was previously detected in seven patients in Denmark in May 201830. Therefore, it is essential to understand the potential threats of these high-risk clones, use effective antibiotics, and take precautions against their spread.
Clermont phylotyping plays a crucial role in identifying the isolates and developing new therapeutic agents. Phylogenetic group A is usually associated with commensal strains, suggesting that the gastrointestinal tract is the primary source of E. coli strains that colonize the urinary tract31,32. Most E. coli isolates belong to phylogroup A and possess multiple ARGs and fewer virulence determinants21. Moreover, phylogroup C possesses a high rate of MDR E. coli with lower motility33.
Investigating the virulome of E. coli strains UPE7 and UPE139 more in depth through WGS revealed a diverse set of virulence determinants that underline their pathogenic potential and ability to persist in the urinary tract environment. Both strains carried fimbrial and adhesion-related genes, which are strongly associated with biofilm formation34. The presence of sitABCD, an iron uptake system that acquires iron to boost their pathogenicity potential, tends to restrict iron bioavailability, a traditional host defense mechanism against bacterial invasion35. Moreover, the presence of hemolysin genes in both isolates is associated with disease severity in UTI and indicates the cytotoxic activity against a broad range of cells36. The identification of shiA, typically associated with the Shigella SHI-2 pathogenicity island, points toward horizontal gene transfer events that may have expanded UPE139’s virulence repertoire37. These findings suggest that while both UPE7 and UPE139 exhibit core uropathogenic-associated virulence traits, UPE139 appears to possess a more complex virulence profile, which may confer increased adaptability and pathogenic potential in the urinary tract.
CTX-M-type ESBLs producing E. coli are associated with the international dissemination of MDR high-risk clones38. Several studies have revealed that blaCTX−M−15, one of the most prevalent genotypes, is widely found in E. coli and rapidly disseminates within patients with urinary tract infections39,40. A previous study reported that blaCTX−M−15 was predominant and present in 68.4% of urinary ESBL-producing E. coli isolates in Minia University Hospital (Minia, Egypt)41. In addition, the location of the blaCTX−M−15 gene adjacent to IS26 (which belongs to the IS6 family) might be involved in their mobilization42. Over the past two decades, CMY-2-like enzymes have been the most commonly observed in ESBL-EC43. In this study, the blaCMY−2 gene was flanked by a single copy of insertion sequence ISEcp1, which facilitates mobilization of the adjacent ARG sequence by a one-end transposition mechanism21. Studies have reported the occurrence of CMY-2 in E. coli (strain UEC59) isolated from urinary tract infection patients in Pakistan, and E. coli isolates in Mexico44,45. The blaCMY−2 gene was recently predicted in 10% of tested E. coli strains isolated from hospitalized patients in Alexandria Main University Hospital (Alexandria, Egypt)21.
OXA-244 carbapenemase was detected in the E. coli UPE139 genome sequence. It differs from OXA-48 by a single amino acid substitution, showing reduced activity against carbapenem antibiotics46. A previous study reported the presence of blaOXA−244 in the genome of MDR E. coli strain HR14_AS, recovered in December 2014 from a wound pus swab of a male patient in Egypt47. Similar to our results, blaOXA−244 is chromosomally integrated into an ISR1-related Tn6237 transposon, which is responsible for the dissemination of OXA-244-producing E. coli48. Despite the absence of known carbapenemase-encoding genes in the genome sequence of UPE7, the isolate revealed resistance to carbapenems. This result is in agreement with a previous study, which reported that imipenem-resistant E. coli ECV219 did not carry genes associated with carbapenem resistance49. This suggests the involvement of an alternative resistance mechanism, including inducible overexpression of chromosomal cephalosporinases, such as AmpC (e.g., CMY-2), combined with porin loss that can result in clinically significant carbapenem resistance50,51.
Healthcare-associated infections caused by isolates with mcr genes that contribute to colistin resistance are associated with increased morbidity and mortality rates, as these plasmid-borne genes facilitate horizontal transfer between bacterial species, promoting their rapid global dissemination24. In this study, although the genome sequences of UPE7 and UPE139 were mcr-negative, both isolates exhibited phenotypic resistance to polymyxin. This indicates that polymyxin B resistance is mediated by chromosomally encoded mechanisms. Polymyxins belong to a group of cationic antimicrobial peptides, and they owe their antimicrobial action to binding to lipid A, a component of the negatively charged lipopolysaccharide (LPS) in the outer membrane of Gram-negative strains, leading to cell lysis52,53. Moreover, resistance to colistin in E. coli can be acquired through chromosomal point mutation, particularly in genes encoding two-component regulatory systems, such as PhoPQ and PmrAB54,55. These mutations result in the expression of the arnBCDTEF operon, which leads to lipopolysaccharide modification via the addition of 4-amino-4-deoxy-l-arabinose (l-Ara4N) to lipid A, thereby reducing the net negative charge of the outer membrane and conferring resistance to polymyxins in E. coli56.
Notably, gyrA (S83L, D87N), parC (S80I), and parE (S458A) mutations were reported in UPE7 and UPE139 isolates. Fluoroquinolone resistance is mainly caused by point mutations in genes encoding DNA gyrase and topoisomerase IV, such as gyrA, gyrB, parC, and parE in quinolone resistance-determining regions57. These types of mutation are associated with high resistance levels to fluoroquinolones in MDR E. coli strains58. In addition, resistance to fluoroquinolones is dependent on the capacity of bacteria to reduce drug accumulation through different efflux systems59. A previous study in Thailand reported that E. coli urinary isolates have a high percentage of resistance against levofloxacin, greater than 90%60. Moreover, a recent study conducted in Egypt demonstrated that 58.7% of isolates of Gram-negative clinical pathogens have at least one quinolone resistance (qnr) gene61.
Studying MGEs is significant in understanding and suggesting the mobility dynamics of ARGs among pathogenic strains. The isolates UPE7 and UPE139 genome sequences harbor multiple plasmid replicons. Previous studies have denoted that IncF plasmids are associated with highly drug-resistant properties and may likely cause long-term resistance and pathogenicity in patients and hospital environments10. This agreed with a previous study from Egypt that demonstrated the presence of IncF replicon types in MDR E. coli isolates62. Similar to our results, previous studies have shown that plasmids expressing the ESBL phenotype also carry genes encoding resistance to antibiotics such as tetracyclines, fluoroquinolones, and aminoglycosides63,64. In agreement with a previous study, the Tn3 transposon harbors the β-lactamase TEM gene, which is transmitted between plasmids of different replicon types39.
This study was limited to only two E. coli isolates, which restricts the extent to which the findings can be generalized to the broader population of uropathogenic E. coli strains in Egypt. Although these isolates were selected based on distinct antimicrobial resistance profiles and prior epidemiological relevance, they do not encompass the full genetic and phenotypic diversity present in clinical settings. To address these limitations, future studies should include a larger and more representative collection of isolates, combined with complementary phenotypic and transcriptomic analyses, to provide a more comprehensive understanding of resistance mechanisms and their clinical implications.
Methods
Sample collection and characterization
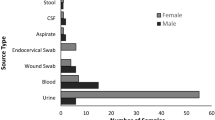
A hundred clinical specimens were collected from females whose ages ranged from 20 to 40 years in a tertiary hospital (Mansoura, Egypt). The study was approved by the University Institutional Review Board of Mansoura University (IRB: R.25.01.3002). Ethical considerations, including personal data privacy, were considered during all study steps. Isolation was performed on MacConkey agar plates (MAC; SRL, India), followed by bacterial purification and conventional identification using the VITEK 2 system (BioMérieux, France) available in the Clinical Pathology Department, Faculty of Medicine. Pure cultures were stored at −20 °C in tryptic soy broth (TSB; SRL, India) supplemented with 50% (v/v) glycerol for further phenotypic and genotypic characterization.
Determination of minimum inhibitory concentration (MIC)
The minimum inhibitory concentration of the isolates was determined using the DL microbial AST system (Zhuhai DL Biotech, China; https://en.medicaldl.com). The results were obtained after 24 h against 24 antimicrobial agents, including gentamicin, amikacin, ampicillin-sulbactam, ceftazidime-clavulanate, cefotaxime-clavulanate, cefoperazone-sulbactam, piperacillin-tazobactam, ertapenem, imipenem, meropenem, cefazolin, cefuroxime, cefotaxime, ceftazidime, cefepime, ampicillin, cefoxitin, levofloxacin, azithromycin, chloramphenicol, polymyxin B, minocycline, tigecycline, and trimethoprim-sulfamethoxazole. The minimum inhibitory concentrations (MIC, µg/mL) of the isolates were interpreted as susceptible (S), intermediate (I), or resistant (R) according to Clinical and Laboratory Standards Institute (CLSI) guidelines65. UPE7 and UPE139 strains were categorized phenotypically as MDR and positive for ESBL based on the resistance profiles. Thus, the two strains were subjected to a comprehensive genomic investigation.
Whole-genome sequencing, assembly, and functional annotation
Genomic DNA from E. coli UPE7 and UPE139 was extracted using the GeneJET™ Genomic DNA Purification Kit (Thermo Fisher Scientific, UK) following the manufacturer’s instructions. Library preparation was carried out on a Hamilton Microlab STAR automated liquid handling system (Hamilton Bonaduz AG, Switzerland) using the Nextera XT Library Prep Kit (Illumina, UK). Genome sequencing was outsourced by MicrobesNG (Birmingham, UK; http://microbesng.uk), on the Illumina NovaSeq 6000 platform (Illumina, USA) using a 250 bp paired-end protocol with 30X sequence coverage. The sequencing adaptors and low-quality reads were trimmed using Trimmomatic (Version 0.30) with a sliding cut-off of Q1566. Subsequently, de novo genome assembly was performed using SPAdes software (Version 3.7.0)67. The newly sequenced genomes were annotated using Prokka software (Version 1.11) and the nucleotide Basic Local Alignment Search Tool (BLASTn) available through the NCBI Prokaryotic Genome Annotation Pipeline (PGAP)68,69.
Determination of serotypes, sequence type (MLST), and phylogroup
The serotypes of E. coli strains were determined with SeroTypeFinder 2.0 provided by the Centre for Genomic Epidemiology (CGE; https://cge.cbs.dtu.dk/services/SerotypeFinder/) for the identification of somatic (O) and flagellar (H) antigens. Multi-locus sequence type (MLST) was determined using the Institute Pasteur web server (https://bigsdb.pasteur.fr)70. The phylogroups of UPE7 and UPE139 were determined using Clermont typing71.
Identification of virulence, mutations, and antimicrobial resistance genes
Virulence factors in E. coli UPE7 and UPE139 genome sequences were investigated using the VirulenceFinder-2.0 server (https://cge.food.dtu.dk/services/VirulenceFinder/), available through CGE. Antimicrobial resistance genes (ARGs) were identified in the UPE7 and UPE139 genome sequences using the Bacterial and Viral Bioinformatics Resource Centre (BV-BRC; https://www.bv-brc.org/) webserver (Version 3.39.10)72 and Comprehensive Antibiotic Resistance Database (CARD-RGI; https://card.mcmaster.ca/) with 90% identity and ≥ 60% coverage thresholds73. Chromosomal mutations mediating AMR were analyzed using the ResFinder 4.6.0 server74 available through CGE.
Identification of plasmids and insertion sequences (IS)
The genome sequences of the two E. coli strains were investigated for plasmids using the PlasmidFinder (Version 2.0) online tool75. Plasmids of the IncF type were subtyped by the plasmid MLST webserver (https://pubmlst.org/organisms/plasmid-mlst). Insertion sequences (IS) were determined using ISfinder (https://www-is.biotoul.fr) using BLASTn (Version 2.2.31+)76.
Comparative genomic analyses
A total of ten genome sequences associated with urinary tract infections were selected based on their geographic relevance to Egypt, and the sequences were retrieved from the NCBI database (Table 4). Furthermore, comparative genomics analysis was performed using OrthoVenn3 (https://orthovenn3.bioinfotoolkits.net) and the Integrated Prokaryotes Genome and Pan-Genome Analysis service (IPGA Version 1.09; https://nmdc.cn/ipga/)77,78. A whole genome-based phylogenetic tree of UPE7 and UPE139 isolates, and other E. coli strains, was created using the IPGA web server78.
Conclusions
This study offers insights into the prevalence of antibiotic resistance mechanisms and mobile genetic elements in two E. coli strains (UPE7 and UPE139) isolated from patients with urinary tract infections in Egypt. The clinical isolates were identified as multidrug-resistant, exhibiting resistance to multiple classes of antimicrobials. Genomic analysis of MGEs highlighted their role in the rapid dissemination of MDR traits across different clonal types of pathogenic E. coli. These findings underscore the importance of combining phenotypic testing with genomic approaches to guide the treatment of urinary tract infections and inform effective control strategies. However, the limited number of isolates analyzed, future studies involving larger and more diverse collections of clinical strains are essential to validate these results and capture broader resistance patterns. Moreover, subsequent investigations will include plasmid sequencing to further explore the presence and transferability of resistance genes. Additionally, expanded genomic surveillance across different geographic regions and healthcare settings will be critical for tracking the emergence and dissemination of high-risk clones, identifying resistance genes, and guiding antibiotic stewardship efforts.
Data availability
The whole genome sequences of *E. coli* UPE7 and *E. coli* UPE139 were deposited at the DDBJ/ENA/GenBank database under the accession numbers JBHWTZ000000000 and JBHWUA000000000, respectively.
References
Chinemerem Nwobodo, D. et al. Antibiotic resistance: The challenges and some emerging strategies for tackling a global menace. J. Clin. Lab. Anal. 36, e24655. https://doi.org/10.1002/jcla.24655 (2022).
Endale, H., Mathewos, M. & Abdeta, D. Potential causes of the spread of antimicrobial resistance and preventive measures in one health perspective Review. Infect. Drug Resist. 16, 7515–7545. https://doi.org/10.2147/IDR.S428837 (2023).
Mann, A., Nehra, K., Rana, J. S. & Dahiya, T. Antibiotic resistance in agriculture: Perspectives on upcoming strategies to overcome upsurge in resistance. Curr. Res. Microb. Sci. 2, 100030. https://doi.org/10.1016/j.crmicr.2021.100030 (2021).
Matheou, A. et al. Antibiotic Use in Livestock Farming: A Driver of Multidrug Resistance? Microorganisms 13, 779. https://doi.org/10.3390/microorganisms13040779 (2025).
Rosini, R., Nicchi, S., Pizza, M. & Rappuoli, R. Vaccines against antimicrobial resistance. Front. Immunol. 11, 1048. https://doi.org/10.3389/fimmu.2020.01048 (2020).
World Health Organization. WHO publishes list of bacteria for which new antibiotics are urgently needed. (2017).
Pfeifer, Y., Cullik, A. & Witte, W. Resistance to cephalosporins and carbapenems in Gram-negative bacterial pathogens. Int. J. Med. Microbiol. 300, 371–379. https://doi.org/10.1016/j.ijmm.2010.04.005 (2010).
Kettani Halabi, M. et al. Antibiotic resistance pattern of extended-spectrum beta-lactamase-producing Escherichia coli isolated from patients with urinary tract infection in Morocco. Front. Cell. Infect. Microbiol. 11, 720701. https://doi.org/10.3389/fcimb.2021.720701 (2021).
Hrovat, K., Molan, K., Seme, K. & Ambrožič Avguštin, J. Molecular characterization of extended-spectrum β-lactamase-producing Escherichia coli isolated from lower respiratory tract samples between 2002 and 2019 in the Central Slovenia region. Ann. Clin. Microbiol. Antimicrob. 23, 6. https://doi.org/10.1186/s12941-023-00664-1 (2024).
Moniruzzaman, M. et al. Multidrug-resistant Escherichia coli isolated from patients and surrounding hospital environments in Bangladesh: A molecular approach for the determination of pathogenicity and resistance. Heliyon 9, e22109. https://doi.org/10.1016/j.heliyon.2023.e22109 (2023).
Li, W., Ma, J., Sun, X., Liu, M. & Wang, H. Antimicrobial resistance and molecular characterization of gene cassettes from class 1 integrons in Escherichia coli strains. Microb. Drug Resist. 28, 413–418. https://doi.org/10.1089/mdr.2021.0172 (2022).
Whelan, S., Bottacini, F., Buttimer, C., Finn, K. & Lucey, B. Whole genome sequencing of uropathogenic E. coli from Ireland reveals diverse resistance mechanisms and strong correlation with phenotypic (EUCAST) susceptibility testing. Infect. Genet. Evol. 121, 105600. https://doi.org/10.1016/j.meegid.2024.105600 (2024).
Sora, V. M. et al. Extraintestinal pathogenic Escherichia coli: virulence factors and antibiotic resistance. Pathogens 10, 1355. https://doi.org/10.3390/pathogens10111355 (2021).
Shah, C., Baral, R., Bartaula, B. & Shrestha, L. B. Virulence factors of uropathogenic Escherichia coli (UPEC) and correlation with antimicrobial resistance. BMC Microbiol. 19, 204. https://doi.org/10.1186/s12866-019-1587-3 (2019).
Yamin, D. et al. Yusof, N. Y. Current and future technologies for the detection of antibiotic-resistant bacteria. Diagnostics (Basel). 13, 3246. https://doi.org/10.3390/diagnostics13203246 (2023).
Morales, G. et al. The role of mobile genetic elements in virulence factor carriage from symptomatic and asymptomatic cases of Escherichia coli bacteriuria. Microbiol. Spectr. 11, e0471022. https://doi.org/10.1128/spectrum.04710-22 (2023).
Tacconelli, E. et al. Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 18, 318–327. https://doi.org/10.1016/S1473-3099(17)30753-3 (2018).
Garza-Ramos, U. et al. Garza-González, E. Whole genome analysis of Gram-negative bacteria using the EPISEQ CS application and other bioinformatic platforms. J. Glob Antimicrob. Resist. 33, 61–71. https://doi.org/10.1016/j.jgar.2023.02.026 (2023).
Ahmed, M. Y. et al. Whole-genome sequencing of multidrug-resistant Escherichia coli causing urinary tract infection in an immunocompromised patient: a case report. J. Med. Case Rep. 18, 326. https://doi.org/10.1186/s13256-024-04663-4 (2024).
Flores-Oropeza, M. A. et al. Comparative genomic analysis of uropathogenic Escherichia coli strains from women with recurrent urinary tract infection. Front. Microbiol. 14, 1340427. https://doi.org/10.3389/fmicb.2023.1340427 (2024).
Mohamed, N. M., Zakaria, A. S. & Edward, E. A. Genomic characterization of international high-risk clone ST410 Escherichia coli co-harbouring ESBL-encoding genes and blaNDM-5 on IncFIA/IncFIB/IncFII/IncQ1 multireplicon plasmid and carrying a chromosome-borne blaCMY-2 from Egypt. Antibiot. (Basel). 11, 1031. https://doi.org/10.3390/antibiotics11081031 (2022).
El-Kholy, A. et al. Landscape of multidrug-resistant gram-negative infections in Egypt: Survey and Literature Review. Infect. Drug Resist. 14, 1905–1920. https://doi.org/10.2147/IDR.S298920 (2021).
Mohamed, E. S., Khairy, R. M. M. & Abdelrahim, S. S. Prevalence and molecular characteristics of ESBL and AmpC β -lactamase producing Enterobacteriaceae strains isolated from UTIs in Egypt. Antimicrob. Resist. Infect. Control. 9, 198. https://doi.org/10.1186/s13756-020-00856-w (2020).
Bakleh, M. Z., Kohailan, M., Marwan, M. & Alhaj Sulaiman A. A Systematic review and comprehensive Analysis of mcr gene prevalence in bacterial isolates in arab countries. Antibiot. (Basel). 13, 958. https://doi.org/10.3390/antibiotics13100958 (2024).
Chong, Y., Shimoda, S. & Shimono, N. Current epidemiology, genetic evolution and clinical impact of extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella pneumonia. Infect. Genet. Evol. 61, 185–188. https://doi.org/10.1016/j.meegid.2018.04.005 (2018).
Hossain, M. et al. Genotype-phenotype correlation of β-lactamase-producing uropathogenic Escherichia coli (UPEC) strains from Bangladesh. Sci. Rep. 10, 14549. https://doi.org/10.1038/s41598-020-71213-5 (2020).
Roer, L., Overballe-Petersen, S., Hansen, F., Schønning, K., Wang, M., Røder, B. L.,Hansen, D. S., Justesen, U. S., Andersen, L. P., Fulgsang-Damgaard, D., Hopkins, K.L., Woodford, N., Falgenhauer, L., Chakraborty, T., Samuelsen, Ø., Sjöström, K., Johannesen,T. B., Ng, K., Nielsen, J., Ethelberg, S., … Hasman, H. Escherichia coli sequence type 410 is causing new international high-risk clones. mSphere. 3, e00337-18 (2018). doi:10.1128/mSphere.00337-18.
Pitout, J. D. D., Peirano, G., Matsumura, Y., DeVinney, R. & Chen, L. Escherichia coli sequence type 410 with carbapenemases: a paradigm shift within E. coli toward multidrug resistance. Antimicrob. Agents Chemother. 68, e0133923. https://doi.org/10.1128/aac.01339-23 (2024).
Emeraud, C. et al. Emergence and polyclonal dissemination of OXA-244-producing Escherichia coli, France. Emerg. Infect. Dis. 27, 1206–1210. https://doi.org/10.3201/eid2704.204459 (2021).
Hammerum, A. M. et al. Surveillance of OXA-244-producing Escherichia coli and epidemiologic investigation of cases, Denmark, January 2016 to August 2019. Euro. Surveill. 25, 1900742. https://doi.org/10.2807/1560-7917.ES.2020.25.18.1900742 (2020).
López-Banda, D. A. et al. Identification of virulence factor genes in Escherichia coli isolates from women with urinary tract infection in Mexico. Biomed. Res. Int. 2014 (959206). https://doi.org/10.1155/2014/959206 (2014).
Khairy, R. M., Mohamed, E. S., Ghany, A., Abdelrahim, S. S. & H. M. & Phylogenetic classification and virulence gene profiles of uropathogenic E. coli and diarrhegenic E. coli strains isolated from community-acquired infections. PLoS One. 14, e0222441. https://doi.org/10.1371/journal.pone.0222441 (2019).
Wang, M. C. et al. Characterization of uropathogenic Escherichia coli phylogroups associated with antimicrobial resistance, virulence factor distribution, and virulence-related phenotypes. Infect. Genet. Evol. 114, 105493. https://doi.org/10.1016/j.meegid.2023.105493 (2023).
Gagaletsios, L. A., Kikidou, E., Galbenis, C., Bitar, I. & Papagiannitsis, C. C. Exploring virulence characteristics of clinical Escherichia coli Isolates from Greece. Microorganisms 13, 1488. https://doi.org/10.3390/microorganisms13071488 (2025).
Robinson, A. E., Heffernan, J. R. & Henderson, J. P. The iron hand of uropathogenic Escherichia coli: the role of transition metal control in virulence. Future Microbiol. 13, 745–756. https://doi.org/10.2217/fmb-2017-0295 (2018).
Laura, C. R. & Rodney, A. W. Hemolysin of uropathogenic Escherichia coli: A cloak or a dagger? Biochim. Biophys. Acta. 1858, 538–545. https://doi.org/10.1016/j.bbamem.2015.08.015 (2016).
Lan, R. & Reeves, P. R. Escherichia coli in disguise: molecular origins of Shigella. Microbes Infect. 4, 1125–1132. https://doi.org/10.1016/s1286-4579(02)01637-4 (2002).
Yao, M. et al. M.Genomic Characterization of a Uropathogenic Escherichia coli ST405 Isolate Harboring blaCTX-M-15-Encoding IncFIA-FIB Plasmid, blaCTX-M-24-Encoding IncI1 Plasmid, and Phage-Like Plasmid. Front. Microbiol. 13, 845045. https://doi.org/10.3389/fmicb.2022.845045 (2022).
Carvalho, I. et al. Characterization of ESBL-producing Escherichia coli and Klebsiella pneumoniae isolated from clinical samples in a northern Portuguese Hospital: predominance of CTX-M-15 and high genetic diversity. Microorganisms. 9, (1914). (2021) https://doi.org/10.3390/microorganisms9091914
Yu, K. et al. Epidemiology and molecular characterization of CTX-M-type ESBLs producing Escherichia coli isolated from clinical settings. J. Glob Antimicrob. Resist. 36, 181–187. https://doi.org/10.1016/j.jgar.2023.11.013 (2024).
Abdelrahim, S. S., Fouad, M., Abdallah, N., Ahmed, R. F. & Zaki, S. Comparative study of CTX-M-15 producing Escherichia coli ST131 clone isolated from urinary tract infections and acute diarrhoea. Infect. Drug Resist. 14, 4027–4038. https://doi.org/10.2147/IDR.S325669 (2021).
Elgayar, F. A., Gouda, M. K., Badran, A. A. & El Halfawy, N. M. Pathogenomics analysis of high-risk clone ST147 multidrug-resistant Klebsiella pneumoniae isolated from a patient in Egypt. BMC Microbiol. 24, 256. https://doi.org/10.1186/s12866-024-03389-z (2024).
Meini, S., Tascini, C., Cei, M., Sozio, E. & Rossolini, G. M. AmpC β-lactamase-producing Enterobacterales: what a clinician should know. Infection 47, 363–375. https://doi.org/10.1007/s15010-019-01291-9 (2019).
Rafaque, Z., Dasti, J. I. & Andrews, S. C. Draft genome sequence of a uropathogenic Escherichia coli isolates (ST38 O1:H15) from Pakistan, an emerging multidrug-resistant sequence type with a high virulence profile. New. Microbes New. Infect. 27, 1–2. https://doi.org/10.1016/j.nmni.2018.10.004 (2018).
Merida-Vieyra, J., De Colsa-Ranero, A., Calderón-Castañeda, Y. & Aquino-Andrade, A. Detection of CMY-type beta-lactamases in Escherichia coli isolates from paediatric patients in a tertiary care hospital in Mexico. Antimicrob. Resist. Infect. Control. 9, 168. https://doi.org/10.1186/s13756-020-00840-4 (2020).
Oteo, J. et al. Emergence of OXA-48-producing Klebsiella pneumoniae and the novel carbapenemases OXA-244 and OXA-245 in Spain. J. Antimicrob. Chemother. 68, 317–321. https://doi.org/10.1093/jac/dks383 (2013).
Soliman, A. M. et al. Draft genome sequence of a blaNDM-1- and blaOXA-244-carrying multidrug-resistant Escherichia coli D-ST69 clinical isolate from Egypt. J. Glob Antimicrob. Resist. 22, 832–834. https://doi.org/10.1016/j.jgar.2020.07.015 (2020).
Abril, D. et al. First Report and comparative genomics analysis of a blaOXA-244-harboring Escherichia coli isolate recovered in the American continent. Antibiot. (Basel). 8, 222. https://doi.org/10.3390/antibiotics8040222 (2019).
Hoang, T. T. et al. A novel gene linked to Imipenem resistance in E. coli isolate lacking known Imipenem-resistance genes. Sci. Rep. 15, 9065. https://doi.org/10.1038/s41598-025-93587-0 (2025).
Nordmann, P., Dortet, L. & Poirel, L. Carbapenem resistance in Enterobacteriaceae: here is the storm! Trends Mol. Med. 18, 263–272. https://doi.org/10.1016/j.molmed.2012.03.003 (2012).
Wilson, H. & Török, M. E. Extended-spectrum β-lactamase-producing and carbapenemase-producing Enterobacteriaceae. Microb. Genom. 4, e000197. https://doi.org/10.1099/mgen.0.000197 (2018).
Falagas, M. E., Rafailidis, P. I. & Matthaiou, D. K. Resistance to polymyxins: Mechanisms, frequency and treatment options. Drug Resist. Updat. 13, 132–138. https://doi.org/10.1016/j.drup.2010.05.002 (2010).
Ligowska-Marzęta, M., Hancock, V., Ingmer, H. M. & Aarestrup, F. Comparison of gene expression profiles of uropathogenic Escherichia coli CFT073 after prolonged exposure to subinhibitory concentrations of different biocides. Antibiot. (Basel). 8, 167. https://doi.org/10.3390/antibiotics8040167 (2019).
Baron, S. et al. Inactivation of the arn operon and loss of aminoarabinose on lipopolysaccharide as the cause of susceptibility to colistin in an atypical clinical isolate of Proteus vulgaris. Int. J. Antimicrob. Agents. 51, 450–457. https://doi.org/10.1016/j.ijantimicag.2017.11.017 (2018).
Janssen, A. B. & van Schaik, W. Harder, better, faster, stronger: Colistin resistance mechanisms in Escherichia coli. PLoS Genet. 17, e1009262. https://doi.org/10.1371/journal.pgen.1009262 (2021).
Humphrey, M. et al. Colistin resistance in Escherichia coli confers protection of the cytoplasmic but not outer membrane from the polymyxin antibiotic. Microbiol. (Reading). 167, 001104. https://doi.org/10.1099/mic.0.001104 (2021).
Fabrega, A., Madurga, S., Giralt, E. & Vila, J. Mechanism of action of and resistance to quinolones. Microb. Biotechnol. 2, 40–61. https://doi.org/10.1111/j.1751-7915.2008.00063.x (2009).
Fuzi, M., Szabo, D. & Csercsik, R. Double-serine fluoroquinolone resistance mutations advance major international clones and lineages of various multidrug-resistant bacteria. Front. Microbiol. 8, 2261. https://doi.org/10.3389/fmicb.2017.02261 (2017).
Whelan, S., Lucey, B. & Finn, K. Uropathogenic Escherichia coli (UPEC)-associated urinary tract infections: the molecular basis for challenges to effective treatment. Microorganisms 11, 2169. https://doi.org/10.3390/microorganisms11092169 (2023).
Boueroy, P. et al. A Fluoroquinolone resistance determinants in carbapenem-resistant Escherichia coli isolated from urine clinical samples in Thailand. PeerJ 11, e16401. https://doi.org/10.7717/peerj.16401 (2023).
Khalifa, H. O. et al. High prevalence of antimicrobial resistance in gram-negative bacteria isolated from clinical settings in Egypt: Recalling for judicious use of conventional antimicrobials in developing nations. Microb. Drug Resist. 25, 371–385. https://doi.org/10.1089/mdr.2018.0380 (2019).
Ramadan, H. et al. Circulation of emerging NDM-5-producing Escherichia coli among humans and dogs in Egypt. Zoonoses Public. Health. 67, 324–329. https://doi.org/10.1111/zph.12676 (2020).
Wang, J. et al. Molecular characterization of bla ESBL-harboring conjugative plasmids identified in multi-drug resistant Escherichia coli isolated from food-producing animals and healthy humans. Front. Microbiol. 4, 188. https://doi.org/10.3389/fmicb.2013.00188 (2013).
Ibekwe, A. et al. Hasan, N. Diversity of plasmids and genes encoding resistance to extended-spectrum β-Lactamase in Escherichia coli from different animal sources. Microorganisms 9, 1057. https://doi.org/10.3390/microorganisms9051057 (2021).
Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing. 31st ed. CLSI supplement M100. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087 USA, (2021).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120. https://doi.org/10.1093/bioinformatics/btu170 (2014).
Bankevich, A. et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 19, 455–477. https://doi.org/10.1089/cmb.2012.0021 (2012).
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069. https://doi.org/10.1093/bioinformatics/btu153 (2014).
Tatusova, T. et al. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 44, 6614–6624. https://doi.org/10.1093/nar/gkw569 (2016).
Jolley, K. A., Bray, J. E. & Maiden, M. C. J. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open. Res. 3, 124. https://doi.org/10.12688/wellcomeopenres.14826.1 (2018).
Clermont, O., Bonacorsi, S. & Bingen, E. Rapid and simple determination of the Escherichia coli phylogenetic group. Appl. Environ. Microbiol. 66, 4555–4558. https://doi.org/10.1128/AEM.66.10.4555-4558.2000 (2000).
Olson, R. D., Assaf, R., Brettin, T., Conrad, N., Cucinell, C., Davis, J. J., Dempsey,D. M., Dickerman, A., Dietrich, E. M., Kenyon, R. W., Kuscuoglu, M., Lefkowitz, E.J., Lu, J., Machi, D., Macken, C., Mao, C., Niewiadomska, A., Nguyen, M., Olsen, G.J., Overbeek, J. C., … Stevens, R. L. Introducing the Bacterial and Viral Bioinformatics Resource Center (BV-BRC): a resource combining PATRIC, IRD and ViPR. Nucleic Acids Res. 51, D678-D689 (2023). doi:10.1093/nar/gkac1003.
McArthur, A. G., Waglechner, N., Nizam, F., Yan, A., Azad, M. A., Baylay, A. J., Bhullar,K., Canova, M. J., De Pascale, G., Ejim, L., Kalan, L., King, A. M., Koteva, K., Morar,M., Mulvey, M. R., O’Brien, J. S., Pawlowski, A. C., Piddock, L. J., Spanogiannopoulos,P., Sutherland, A. D., … Wright, G. D. The comprehensive antibiotic resistance database. Antimicrob Agents Chemother. 57, 3348–3357 (2013). doi:10.1128/AAC.00419-13.
Bortolaia, V., Kaas, R. S., Ruppe, E., Roberts, M. C., Schwarz, S., Cattoir, V., Philippon,A., Allesoe, R. L., Rebelo, A. R., Florensa, A. F., Fagelhauer, L., Chakraborty, T.,Neumann, B., Werner, G., Bender, J. K., Stingl, K., Nguyen, M., Coppens, J., Xavier,B. B., Malhotra-Kumar, S., … Aarestrup, F. M. ResFinder 4.0 for predictions of phenotypes from genotypes. J. Antimicrob Chemother. 75, 3491–3500 (2020). doi:10.1093/jac/dkaa345.
Carattoli, A. et al. silico detection and typing of plasmids using PlasmidFinder and plasmid multilocus sequence typing. Antimicrob. Agents Chemother. 58, 3895–3903. https://doi.org/10.1128/AAC.02412-14 (2014).
Zhang, Z., Schwartz, S., Wagner, L. & Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol. 7, 203–214. https://doi.org/10.1089/10665270050081478 (2000).
Sun, J. et al. OrthoVenn3: an integrated platform for exploring and visualizing orthologous data across genomes. Nucleic Acids Res. 51, W397–W403. https://doi.org/10.1093/nar/gkad313 (2023).
Liu, D. et al. IPGA: A handy integrated prokaryotes genome and pan-genome analysis web service. iMeta 1. https://doi.org/10.1002/imt2.55 (2022). e55.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
N.E. and M.G. conceptualized and drafted the manuscript, and N.E. participated in computational analysis and figure construction. F.E. and A.A. participated in isolating the strains from patients. All authors critically reviewed the article and gave the final approval of the version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
The Institutional Review Board of Mansoura University, Egypt, approved the study (IRB: R.25.01.3002) on 21-8-2023. The study does not include any personal data, and the patients did not directly participate in this research work; informed consent was obtained from the patients. All study processes were conducted in accordance with the principles guiding research involving human participants as set out in the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El Halfawy, N.M., Gouda, M.K., Elgayar, F.A. et al. Genomic characterization of multidrug-resistant Escherichia coli strains identified from patients with urinary tract infection in Egypt. Sci Rep 16, 8958 (2026). https://doi.org/10.1038/s41598-026-40536-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40536-0