Abstract
The impact of allergic diseases on bone loss remains unclear because it is considered to result from the use of corticosteroids to ameliorate allergic inflammation. To explore the effects of allergic diseases on bone metabolism, we investigated long bones in ovalbumin (OVA)-induced asthmatic mice. Compared with that of vehicle-treated controls, the bone mass of OVA-induced asthmatic mice was lower. We found fewer mature osteoblasts on the surfaces of the cortical and trabecular bones of allergen-sensitized mice than in those of vehicle-treated mice. Piezo1 and Piezo2 were located in mature osteoblasts, and their expression levels decrease during osteocytogenesis. Compared with that in non-asthmatic mice, the expression levels of both Piezo1 and Piezo2 were suppressed in asthmatic mice. The Piezo1 agonist reversed OVA-induced bone loss, with increased osteogenesis and type I collagen (Col1) expression. Furthermore, compared with control mice, Piezo2 heterozygous deletion mice with OVA presented a decreased bone volume. Our data suggest that asthma could induce osteopenia and that Piezo channel modulation could be a possible way to prevent bone loss induced by allergic asthma.

Similar content being viewed by others
Introduction
Recent advances in the understanding of bone metabolism have revealed that in addition to the cooperation between osteoblasts and osteoclasts in bone formation and bone loss, diverse modulatory systems contribute to bone homeostasis1. Asthma is a major noncommunicable airway disease worldwide that affects all ages and is the most common chronic disease among children. Bone health in asthmatic patients has received attention because people with asthma might have reduced bone mineral density and an increased risk of bone fracture, which has been associated with oral or inhaled glucocorticoid use2,3. Preserving bone strength and reducing fractures are important public health goals because fractures are associated with increased morbidity and mortality. As atopic asthma is a common disease affecting various age groups, the association between asthma and bone metabolism could have a significant impact on health sustainability. We previously demonstrated that rats with ovalbumin (OVA)-induced atopic diathesis presented increased eosinophil infiltrates in the alveolar bone marrow and increased osteoclastogenesis4. Furthermore, during orthodontic tooth movement, the rats with OVA-induced atopic diathesis showed greater tooth movement and tooth root resorption than did the vehicle-treated rats. Recently, a UK population-based matched cohort study5 revealed that patients with asthma had a higher risk of osteoporosis and fragility fractures of the vertebra and wrist, which was independent of age, sex, and oral corticosteroid use. However, the direct impact of atopic allergic diseases on bone metabolism has not been clearly demonstrated.
In accordance with a variety of environmental stimuli, bone continuously remodels with a delicate balance between bone formation and resorption through the coordinated functions of bone cells: mesenchymal-derived osteoblasts and osteocytes in the bone matrix and bone marrow-derived osteoclasts. Maintaining bone tissue quantity and the integrity of the bone structure is considered to depend on the balance of the supply of an appropriate number of osteoblasts, osteoblast precursors to mature osteoblasts differentiation, and subsequent transformation into osteocytes, creating a suitable major bone matrix which includes type I collagen (Col1)6,7. Physiological bone formation is known to be associated with mechanically gated ion channels, i.e., Piezo channels8. Piezo1 is reported to play a major role in osteoblastic mechanosensing9 and long bone morphogenesis10,11. Nevertheless, the pathophysiological role of Piezo channels in bone cells and their subcellular localization remain to be elucidated.
Here, we aimed to examine whether asthma affects bone mass and the bone matrix and whether it affects the structure and metabolism of bone cells in individual mice. We found less bone mass in the femurs of the mice with OVA-induced asthma than in those of the control mice. Asthmatic mice presented suppressed expression of Piezo channels and decreased Col1 in the bone matrix. Pharmacological activation of Piezo1 reversed the decreases in bone volume and bone matrix production induced by OVA sensitization. Our study provides evidence that systemic allergies affect bone metabolism and provides molecular insight into the participation of Piezo channels in bone matrix formation.
Results
OVA-induced asthma led to bone loss
We used the ovalbumin (OVA) mouse model of allergic asthma12 (Fig. 1A). The animals were divided into three experimental groups: O + A: immunized with OVA as an antigen and aluminum gel (Alum) as an adjuvant. Alum only and vehicle (PBS) only were used as controls. Histological observations of the lungs via periodic acid–Schiff (PAS) staining confirmed distinct pulmonary inflammation, such as bronchial epithelial hyperplasia, goblet cell hyperplasia, and an increased number of infiltrating inflammatory cells (Fig. 1B and Supplementary Fig. 1A). The OVA-induced asthma model is known as a Th2 response that induces eosinophil infiltration. We detected a greater number of eosinophils in the lung tissue (Supplementary Fig. 1A) and in the bone marrow of the tibia (Fig. 1C). We did not find a significant difference in body weight among the groups (Supplementary Fig. 1B).
A Scheme of experiments involving OVA-induced asthma model mice. B Light microscopy images of representative PAS-stained lung sections. A significantly greater number of PAS-stained goblet cells were observed in the bronchi of O + A mice than in those of control mice. Scale bar: 50 µm. Quantitative analysis of the PAS-positive area in the bronchus. n = 3 male mice per group. C Giemsa-stained distal femurs. Compared with those of the control mice, the bone marrow of the O + A mice presented more eosinophils. The right panel shows a higher magnification image of the bone marrow with eosinophils adjacent to osteoblasts (Ob) on the cortical bone surface (CB). Scale bars: 20 µm and 5 µm. D Left image showing the overview of the volume of interest (green) of the femur. GP: growth plate. The yellow rectangle indicates the area of the representative images of 3D reconstructed bone shown in the right panels. Scale bars: 50 µm, 250 µm, 100 µm. E Quantitative analysis of the trabecular bone volume (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), trabecular bone pattern factor (Tb.Pf), and structure model index (SMI). n = 5 male mice per group. The data are presented as the median with interquartile range (IQR); one-way ANOVA with Tukey’s post hoc test was used. *p < 0.05, **p < 0.01, ***p < 0.001 compared with the control groups.
To assess the effect of asthma on bone mass, we selected the volume of interest (VOI) as the primary spongiosa of the distal femur just under the growth plate cartilage at a voxel resolution of 1 μm3 by microcomputed tomography (µCT) because the region is considered active bone formation and resorption, which involves intense distribution of mature osteoblasts and osteoclasts13 (see below). Compared with the Alum or PBS groups, the O + A group displayed a lower bone volume (BV/TV), thinner trabecular bone thickness (Tb.Th), higher trabecular pattern factor (Tb.Pf), and structure model index (SMI) (Fig. 1D, E). To explore whether bone metabolism is affected by asthma, we histomorphometrically compared the images stained with alkaline phosphatase (ALP) and tartrate-resistant acid phosphatase (TRAP). Compared with those in the PBS and Alum groups, a smaller trabecular bone surface was covered by ALP-positive cells in the femurs of the O + A group (Fig. 2A). We found more multinucleated TRAP-positive cells on the surface of the trabecular bone of the primary spongiosa in the O + A group than in the PBS and Alum groups (Supplementary Fig. 1C), and the number of osteoclasts per bone surface was greater in the O + A group than in the other two groups (Supplementary Fig. 1D).
A Representative light microscopy images of alkaline phosphatase (ALP) staining. B–D Immunofluorescence images of Osterix (Osx), type I collagen (Col1), and podoplanin. The inset image in (D) shows an enlarged view of the osteoblasts on the bone surface. The expression of ALP, Osterix, Col1, and podoplanin around the trabecular bone decreased in O + A mice. GP: growth plate, Tb: trabecular bone. The dashed lines demarcate the boundary of the growth plate or trabecular bone surface. The graphs on the right of (A–D) show the results of the quantitative analysis of the staining intensities. E, F Immunoblot analysis of Osx (E) and Col1 (F) protein expression in distal femurs. The quantified protein levels were normalized to those of TBP or GAPDH. n = 3–4 male mice per group. The data are presented as the median with IQR. One-way ANOVA with Tukey’s post hoc test was used. *p < 0.05, **p < 0.01, ***p < 0.001 compared with the control groups. Scale bars: 200 µm (A), 50 µm (B: upper, C, D), 20 µm (B: lower).
To assess whether OVA sensitization affects osteogenesis, we compared the expression of Osterix (Osx), a key osteoblastic transcription factor14. The number of Osx-immunoreactive cells on the trabecular bone was significantly lower in the O + A group than in the PBS or Alum groups (Fig. 2B). Next, we examined the expression levels of Col1, a fundamental bone matrix, and podoplanin, a transmembrane glycoprotein considered critical in early osteoblast‒osteocyte transitions. Compared with that in the PBS and Alum groups, Col1 expression in the trabecular bone of the femur was lower in the O + A group (Fig. 2C). We found that some of the osteoblasts and early osteocytes in the bone matrix were immunoreactive for podoplanin, which is consistent with previous studies15. (Fig. 2D). The expression of podoplanin was also lower in the trabecular and cortical bones of the O + A group than in those of the control group (Fig. 2D). Semaphorin 3A (Sema3A), a membrane-associated secretory protein, has been reported to have osteoprotective functions, with new bone formation and suppression of osteoclastogenesis16,17. The expression of Sema3A in the bone of the O + A group was lower than that in the control group (Supplementary Fig. 2A). Immunohistochemistry revealed that Sema3A was located at similar intensities in osteoblasts and osteoclasts at the surface of the trabecular bone of the femurs in the PBS group. However, in the O + A group, Sema3A immunoreactivity was suppressed in osteoblasts compared with that in the PBS group (Supplementary Fig. 2A, B). Immunoblotting confirmed the decreased amounts of Osx, Col1, and Sema3A in the femurs of the O + A group compared with those in the controls (Fig. 2E, F and Supplementary Fig. 2C). These data indicated that osteogenesis was suppressed in the long bones of OVA-induced asthma mice.
Suppressed Piezo channel expression in the bones of OVA-induced mice
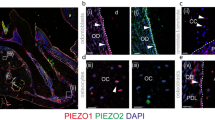
Recently, the mechanosensitive cation channels Piezo1 and Piezo2 have been reported to play significant roles in osteogenesis and bone tissue formation9,10,11,18,19. We generated antibodies for immunohistochemistry. Antibodies against Piezo1 or Piezo2 recognized the membrane and cytoplasmic fractions of Piezo1- or Piezo2-transfected HEK293 cells, respectively (Supplementary Fig. 3A). The Piezo1 antibody did not recognize Piezo2, and vice versa. We did not find any immunofluorescent signals when the antibodies were preincubated with the antigenic peptides (Supplementary Fig. 3B). In the bone tissues, the Piezo1 antibody was almost colocalized with the commercially available antibody, which was validated in both the knockdown cells20 and the knockout tissues21 (Supplementary Fig. 3C). We used our antibody because our method resulted in less background staining in bone tissues. In the bone tissues of Piezo2-GFP knock-in mice22, the immunohistochemical signals of our Piezo2 antibody almost colocalized with those of the anti-GFP antibody (Supplementary Fig. 3D). In the femur, we found marked Piezo1- and Piezo2-labeled osteoblasts located on the surfaces of the primary spongiosa and the cortical bone rather than the secondary spongiosa (Fig. 3A). Piezo1 and Piezo2 were coexpressed extensively in mature osteoblasts on the surface of the bone matrix, as shown by nuclear Osx labeling (Supplementary Fig. 3E).
A Immunofluorescence images of Piezo1 (magenta) and Piezo2 (green) in the femurs of WT mice. Scale bar: 20 µm. The right panels show optical sections from Airyscan superresolution images merged with differential interference contrast (DIC) images in the area indicated by white rectangles. Mature osteoblasts (Ob), osteocytes (Ocy) and osteoclasts (Oc) expressing Piezo1 and Piezo2. Scale bar: 5 µm. B, D Piezo1 (magenta) and Piezo2 (green) immunofluorescence images of trabecular (upper) and cortical (lower) bones in the distal femur from each group. Scale bars: 50 µm. C Piezo1 fluorescence intensity and number of Piezo1-positive surfaces per trabecular bone surface (Piezo1+. S/BS) were significantly lower than those in the controls. E Piezo2 fluorescence intensity and number of Piezo2-positive surfaces per trabecular bone surface (Piezo2+ S/BS) were significantly lower than those in the controls. F Immunoblot analyses of Piezo1 and Piezo2 expression in the femur. Graphs showing the results of the quantitative analysis of the band intensities. n = 3–4 male mice per group. The data are presented as the median with IQR. One-way ANOVA with Tukey’s post hoc test was used. *p < 0.05, **p < 0.01, ***p < 0.001 compared with the control groups.
Immunoreactivity of Piezo1 and Piezo2 was conspicuous in mature osteoblasts, osteoblastic osteocytes, and osteoid osteocytes and weak in osteocytes (Fig. 3A). Osteoclasts were also labeled with Piezo1 and Piezo2, but their expression was relatively low compared with that of osteoblasts (Fig. 3A). We compared the expression of Piezo1 and Piezo2 in osteoblasts among the experimental groups. The expression of Piezo1 and Piezo2 in osteoblasts was clearly diminished in the femurs of the O + A group (Fig. 3B, D). The Piezo1 and Piezo2 intensities and the numbers of Piezo1- and Piezo2-immunoreactive cells at the trabecular bone surface were significantly lower in the O + A group than in the Alum or PBS group (Fig. 3C, E). Immunoblotting of proteins extracted from the metaphysis of the femur confirmed the lower Piezo1 and Piezo2 levels in the O + A group than in the PBS group (Fig. 3F). These data demonstrated that the bones of asthmatic mice have less Piezo1 and Piezo2 than those of control mice.
Distinct subcellular localization of Piezo1 and Piezo2 in mature osteoblasts and their decrease in asthmatic mice
Piezo1 has been reported to be vitally important for the differentiation of mesenchymal stem cells into osteoblast lineage cells23. Piezo2 has been reported to play a part in bone formation11,19. However, their subcellular localization in mature osteoblasts on the bone surface, which plays a major role in the production of the bone matrix, remains to be elucidated. Airyscan superresolution imaging revealed that Piezo1 and Piezo2 were located on the cell surface in a distinct manner (Supplementary Fig. 4A). Moreover, multisubcellular organelle marker staining revealed that Piezo1 predominantly colocalized with calnexin and KDEL, both of which are endoplasmic reticulum (ER) markers (Fig. 4A and Supplementary Fig. 4B). Piezo1 was partially associated with CD63 (a marker for late endosomes, lysosomes and secretory vesicles called exosomes), GM130 (a Golgi marker), EEA1 (an endosome marker) and Lamp1 (a lysosomal marker) (Supplementary Fig. 4B). On the other hand, Piezo2 immunoreactivity in osteoblasts was characterized as a sphere adjacent to the nucleus, with GM130 labeling reported as a Golgi ribbon24,25 (Fig. 4A and Supplementary Fig. 4C). Piezo2 was also localized to a small extent with EEA1, KDEL, and Lamp1 (Supplementary Fig. 4C). Ultrastructural observation by transmission electron microscopy confirmed the localization of Piezo1 on the cell surface membrane and the ER membrane, whereas Piezo2 was localized on the cell surface membrane and areas composed of assemblies of the Golgi apparatus (Fig. 4B and Supplementary Fig. 5A). Airyscan superresolution imaging revealed that Piezo2 was adjacent to Col1 at the periphery of the Golgi area and in the osteoid, a newly formed bone matrix (Fig. 4C). Mature osteoblasts harboring Piezo1 and Piezo2 extended many cellular processes into the osteoid with filamentous actin (Supplementary Fig. 5B, D). These cytoplasmic processes had punctate Piezo1 and Piezo2 immunoreactivities in the cellular processes. Piezo1 was localized on actin filaments in the cytoplasmic processes (Supplementary Fig. 5B, C). A greater amount of Piezo2 colocalized on actin filaments than that of Piezo1(Supplementary Fig. 5D, E). Unlike Piezo1, many Piezo2 puncta were localized in the osteoid, some of which colocalized with CD63 (Supplementary Fig. 4C). Ultrastructurally, Piezo2-labeled cytoplasmic processes protrude into the osteoid. Some of the Piezo2-labeled cytoplasmic processes were proximate to the collagen fibrils (Supplementary Fig. 5A). Punctate signals representing Piezo1 and Piezo2 were also observed on a complex network of actin in the bone matrix connected to osteoblastic osteocytes, osteocytic osteoblasts, osteoid osteocytes, and osteocytes known as the osteocytic lacunar canalicular system (Supplementary Fig. 5B, D).
A Airyscan superresolution images of mature osteoblasts on the bone surface of distal femoral metaphysis from WT mice. Piezo1 (green) colocalized with calnexin (magenta), and Piezo2 (green) colocalized with GM130 (magenta) was observed. Scale bar: 5 µm. B Immunoelectron microscopy images of osteoblasts in the femur. Elevated electron densities with diaminobenzidine (DAB)-positive immunoreactivities were detected in rough endoplasmic reticulum (rER) membranes with the Piezo1 antibody and in Golgi membranes with the Piezo2 antibody. Note the lack of DAB reaction products in the Golgi and nuclear membranes in the former and that in rER and nuclear membranes in the latter, respectively. N: nucleus, m: mitochondria, G: Golgi stacks. Scale bar: 1 µm. C Airyscan superresolution images of optical sections obtained at z-intervals of 160 nm and processed by Airyscan Joint Deconvolution. Osteoblasts were stained with Piezo2 (green) and Col1 (magenta) in the femurs of the PBS group. Scale bar: 5 µm. The lower panels show higher-magnification images of the yellow rectangles. Scale bar: 1 µm. Line plots along white arrowheads show the localization of Piezo2 and Col1 puncta in the Golgi area (i) and osteoid (ii). A.U.: arbitrary units. D Representative immunofluorescence images of calnexin and GM130 in the groups. Scale bars: 50 µm, 10 µm. E Fluorescence intensity of calnexin and GM130 in the trabecular region of the femur. F Airyscan superresolution images of osteoblasts immunostained with Piezo1 (green) and calnexin (magenta) (upper panels), and Piezo2 (green) and GM130 (magenta) (lower panels), showing less immunolabelling in the O + A group than in the control group. Scale bar: 5 µm. G Quantitative analysis of the weighted colocalization (as a percentage) of calnexin with Piezo1 and GM130 with Piezo2 in femur osteoblasts. H Airyscan superresolution images of Col1 (green) and GM130 (magenta) immunofluorescence in osteoblasts from the femur. Scale bar: 5 µm. I Quantitative analysis of Col1 fluorescence intensity and weighted colocalization (as a percentage) of Col1 with GM130 in femur osteoblasts. n = 3 male mice per group. The data are presented as the median with IQR. One-way ANOVA with Tukey’s post hoc test. *p < 0.05, **p < 0.01, ***p < 0.001 compared with the control groups.
We next explored whether OVA sensitization affects the amount of osteoblastic ER and Golgi, a substantial compartment of bone matrix synthesis, because we detected less Col1 in the bone tissues of the O + A mice than in those of the control mice (Fig. 2C). The labeling and intensity of calnexin and GM130 were lower in the mature osteoblasts of the O + A group than in those of the control group (Fig. 4D, E). Furthermore, the colocalization of Piezo1, calnexin, Piezo2, and GM130 was clearly diminished in the O + A group (Fig. 4F, G). In mature osteoblasts, Col1 was localized in the GM130-positive Golgi area, and Col1 was more intense in the periphery of the Golgi area (Fig. 4H). The intensity and area of colocalization of Col1 and GM130 were lower in the mature O + A osteoblasts than in the control osteoblasts (Fig. 4H, I).
Piezo1 and Piezo2 affect OVA-induced bone loss
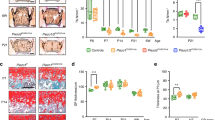
Finally, to determine whether activation of Piezo1 is able to prevent bone loss caused by OVA sensitization, we applied the Piezo1 agonist Yoda1 to O + A mice (Fig. 5A). In the distal femur, the bone volume of Yoda1-treated O + A mice was comparable to that of no-OVA no-Yoda1-treated mice (Fig. 5B). Compared with no-OVA no-Yoda1-treated mice, no-OVA Yoda1-treated mice did not have significantly greater bone volume (Fig. 5B). ALP activity was greater in Yoda1-treated O + A mice (Fig. 5C), whereas TRAP activity was lower in Yoda1-treated O + A mice (Fig. 5D) than in no-Yoda1-treated O + A mice. Additionally, the expression of Osx and Col1 in the bone tissues was elevated by Yoda1 treatment (Fig. 5E, F). The height of mature osteoblasts was greater than that of control osteoblasts, and mature osteoblasts were distributed on the bone surface of Yoda1-treated mice. Furthermore, we detected greater expression of calnexin in Yoda1-treated mice than in control mice (Fig. 5G), but the expression of GM130 was similar between these two groups (Fig. 5H). Our results demonstrated that the activation of Yoda1 resistance to OVA-induced bone loss resulted from the activation of osteoblasts.
A Yoda1 administration schedule in asthma model mice. B Representative 3D-rendered µCT images of primary spongiosa in the proximal tibia metaphysis from mice in the PBS or O + A group with or without Yoda1 treatment. The graph shows the quantitative analysis of the trabecular bone volume (BV/TV). n = 3 male mice per group. Representative microscopy images of ALP (C) and TRAP (D) staining and immunofluorescence images of Osterix (E) and type I collagen (F) and Airyscan superresolution images of calnexin (G) and GM130 (H) immunostaining of femurs from O + A mice and O + A+Yoda1 mice. Histomorphometric analyses of ALP- or TRAP-positive surfaces per bone surface and the fluorescence intensities of Osx, Col1, calnexin, and GM130 in the trabecular region of the femur are shown on the right of each panel. I Representative 3D-rendered µCT images of primary spongiosa in the proximal tibia metaphysis from WT or Piezo2+/− mice in the PBS or O + A groups. n = 5 female mice per group. The data are presented as the median with IQR. One-way ANOVA with Tukey’s post hoc test. *p < 0.05 compared with the control groups. Scale bars: 200 µm (B, C, F), 100 µm (I), 20 µm (D, E), and 5 µm (G, H).
To explore the effect of Piezo2 on OVA-induced bone loss, we compared Piezo2 heterozygously deleted (Piezo2+/−) mice. Similar bone volumes were found in wild-type and Piezo2+/− mice. However, after OVA treatment, the bone volume of Piezo2+/− mice was clearly smaller than that of PBS-treated mice (Fig. 5I). These results suggest that the bone loss observed in asthmatic mice is associated with Piezo1 and Piezo2.
Discussion
Osteoporosis is a silent disease, and osteoporosis-related fractures have a substantial impact on individual patients. Patients with allergic asthma are at increased risk of developing osteoporosis3,26. The increased risk has been regarded as a type of corticosteroid-induced osteoporosis, and the direct involvement of allergic asthma in bone metabolism has not been clearly demonstrated. We observed the loss of long bones in OVA-induced allergic asthma model mice, a general asthma mouse model, suggesting that allergic asthma affects bone metabolism. We asked whether aluminum, which is generally used as an adjuvant for asthma models, affected bone loss in our experimental sets because aluminum is a well-known inhibitor of bone mineralization and phosphate absorption27. Since the results of the Alum group were similar to those of the PBS group, we concluded that asthma itself affected bone loss. The reason why osteoclastogenesis occurred in bone following injection with a small amount of aluminum gel compared with injection with PBS remained to be elucidated. We previously demonstrated decreased bone and increased orthodontic tooth resorption in OVA-induced atopic diathesis rats4. The presumption that allergic diseases aggravate osteoporotic fracture is supported by the large cohort study by Lowe et al.28, which included patients with atopic eczema, and Chalitsios et al.5, which reported that patients with asthma have increased fracture risk, particularly major osteoporotic fragility fractures. Lowe et al.28 reported that fracture risk increased with increasing eczema severity, and furthermore, the association persisted after oral glucocorticoid adjustment. Overall, we speculate that atopic allergies affect bone volume and quality.
To explore the OVA-induced effect on bone volume, we chose the ROI and VOI as the spongiosa just beneath the growth plate cartilage, which is known as an active region of bone formation and resorption13. We also observed that ALP-, Osx-, and podoplanin-labeled mature osteoblasts and TRAP-positive osteoclasts were clearly aligned on the surface of the trabecular bone on the spongiosa just beneath the growth plate cartilage. We assumed that we could visualize early bone loss as an onset because the ROI and VOI have dynamic metabolism with analyses by 1 µm high-resolution µCT. On the surface of the trabecular bone of O + A mice compared with that of control mice, we detected suppressed osteogenesis, as demonstrated by fewer ALP- and Osterix-positive cells, and increased osteoclastogenesis, as demonstrated by a greater number of TRAP-positive cells. The decrease in Col1 in O + A bone suggested impaired bone matrix production, and the decrease in podoplanin in O + A bone suggested suppressed differentiation from osteoblasts to osteocytes. We detected an increased number of eosinophils not only in the lungs but also in the bone marrow of OVA-induced mice. In OVA-induced atopic diathesis rats, inflammatory cytokine and leukotriene levels in bone tissues are elevated, and the number of eosinophils in the bone marrow is increased4. Modulation of the immune system with inflammatory mediators may affect osteogenesis and osteoclastogenesis in O + A bone. Additionally, one possible explanation for the lower amount of bone in O + A mice could be the lower Sema3A expression in osteoblasts than in PBS-treated mice. Osteoblastic Sema3A plays a protective role against bone resorption16,17 along with neuronal Sema3A29. As Sema3A is reportedly associated with asthmatic airway inflammation30, Sema3A might be a pharmacological target for the treatment of asthma and bone loss. The molecular mechanisms modulating the differentiation of bone cells remain to be elucidated further.
The expression of Piezo1 and Piezo2 in bone cells has been previously reported, but their precise protein localization has not been clearly identified. We carefully evaluated the specificity of the antibodies by immunoblotting and immunohistochemistry. We concluded that our antibodies, with our protocols, demonstrated that mature osteoblasts had prominent Piezo1 and Piezo2 expression in bone tissues in vivo. Under superresolution microscopy, slight differences in localization were detected between the anti-GFP antibody and the Piezo2 antibody, suggesting that the fusion protein slightly modified the protein. We employed our antibodies because our goal was to determine the in situ localization of Piezo1 and Piezo2. We found that Piezo1 and Piezo2 were expressed in preosteoblasts, osteoblasts, and osteocytes, and both were the most prominent in mature osteoblasts on the bone surface, both in the trabecular and cortical bones of the tibia and femur, suggesting a pivotal role in mature osteoblasts. Piezo1 and Piezo2 are distinctly labeled at the surface of the plasma membrane and on the cytoplasmic processes of mature osteoblasts. We also detected Piezo1 and Piezo2 puncta on the actin filaments in the cytoplasmic processes of the osteoid and in the osteocytic lacunar–canalicular system of osteocytes and bone-lining osteoblasts. Piezo1 and Piezo2 may be suitable for sensing mechanical or chemical bone marrow and the bone matrix environment. Additionally, Piezo2 vesicles, which are not Piezo1 vesicles but rather abundant, are localized in the osteoid, the newly formed bone matrix, and the cytoplasmic processes extending towards the osteoid. Additionally, the cytoplasmic processes associated with Piezo2 are connected with the cytoplasmic processes of osteocytes in the bone matrix. Ultrastructurally, we confirmed that Piezo2-labeled cytoplasmic processes extended along the collagen fibers. These structural characteristics are consistent with three-dimensional observations via FIB-SEM, which revealed that the cytoplasmic process in the transverse process twists around collagen fibers in the osteoid31. Piezo2 is a well-known light touch sensor in mice22,32,33 and humans34 that senses matrix stiffness35. Based on these Piezo2 functions, we speculate that the effects of Piezo2 on the cytoplasmic processes and vesicles in the osteoid may be able to be used to monitor the quality or contents of the bone matrix, after which osteoblasts modulate their role to adjust the quality and quantity of the extracellular matrix components and mineralization. This idea is supported by the finding that Prrx1-mediated Piezo1 and Piezo2 double-knockout mice presented lower levels of Col1 in the serum and cortical bone than Piezo1 single-knockout mice did11. Additionally, bone marrow-derived mesenchymal stem cells sense extracellular matrix stiffness via Piezo1 and Piezo211. Piezo1, Piezo1 or Piezo2 regulate osteogenesis9,11,18. Furthermore, we speculate that Piezo1 and Piezo2 in preosteoblasts, osteoblasts, and osteocytes might play a part in self-renewal and cell fate definition in response to the microenvironment of the osteoid-like intestinal stem cell niche demonstrated in a recent study36. How osteoblastic cytoplasmic processes sense the mechanical environment of the bone matrix via Piezo channels and then translate into the regulation of osteogenic differentiation is still a subject of future research.
Piezo1 is known to play a significant role in endochondral ossification11,19. Osteoblastic Piezo1 plays a crucial role, as clearly demonstrated by studies in Dmp1-Cre;Piezo1f/f mice18, Prx1-Cre-mediated9, Prrx1-Cre-mediated11, and Runx2-Cre-mediated Piezo1 deletion mice19. The conspicuous Piezo1 expression of mature osteoblasts and asthmatic bone loss with suppressed Piezo1 expression in our study are consistent with the significant role of Piezo1 in osteoblasts reported in previous studies. By performing subcellular marker labeling under a superresolution microscope, we unexpectedly found that Piezo1 was clearly localized in the ER membrane and that Piezo2 was localized in the Golgi membrane, which was confirmed by transmission electron microscopic observation. It seems reasonable that not only the surface plasma membrane but also the ER and Golgi membranes respond to mechanical stimuli and possibly modulate processing or protein transport. In osteoblasts, Piezo1-immunoreactive ER and Piezo2-immunoreactive Golgi were suppressed in asthmatic mice, suggesting that systemic asthma conditions affect osteogenesis and protein synthesis in osteoblasts. Furthermore, the vital role of Piezo1 was confirmed by the finding that systemic Yoda1 administration prevented the bone loss induced by OVA. We did not find a significant difference in the bone volume of Yoda1-treated and PBS-treated mice, although the same dose and administration period were used by Li et al.18. This difference might be due to differences in age and sex, as Li et al.18 reported differences in 4-month-old female mice. After Yoda1 was applied to OVA-treated mice, we detected enhanced osteogenesis by ALP- and Osterix-labeled osteoblasts and increased calnexin ER staining. Additionally, Yoda1 increased Col1 labeling and resistance to OVA-induced osteoclastogenesis, suggesting that the activation of Piezo1 could be a possible target for combatting allergy-associated bone loss. However, as Yoda1-treated mice have been reported to exhibit itch-related behavior in the skin37, Yoda1 application might not be a viable option. Recently developed Piezo1 agonists could be possible targets38. Further studies are needed to identify effective pharmaceuticals for Piezo1 activation and the timing of Piezo1 activation, such as physical exercise.
In asthmatic mice, we found that suppressed osteogenesis and decreased expression of Piezo1 and Piezo2 in mature osteoblasts, and that the decrease in osteogenesis was reversed by Yoda1 treatment. Along with the results showing less bone in OVA-treated Piezo2 heterozygously deleted mice than in PBS control mice, these findings suggest that both Piezo1 and Piezo2 affect bone metabolism and bone matrix formation. Although the function of Piezo1 in osteogenesis is well established, genetic mutation of Piezo1 in bone deformation is not as apparent in humans as in patients with Piezo2 mutation, who present with apparent bone deformity39. Previous studies on the downstream signals of Piezo1 and Piezo2, such as the β-catenin pathway9,11, have revealed excellent results; however, our knowledge of the functions of Piezo1 and Piezo2 is limited. Studying the subcellular functions of Piezo channels, such as the ER, Golgi, or cytoskeleton, may reveal the unknown functions of Piezo channels.
Our data provide a new link between asthma and bone loss without glucocorticoids and highlight the influence on osteogenesis. Targeting communication through chronic allergic diseases and bone metabolism through Piezo1 and Piezo2 or other potentially unknown molecular mechanisms may be an effective approach to preserve appropriate bone volume and quality.
Methods
Mice
Wild-type C57BL/6N mice were purchased from SLC Japan, Inc. (Hamamatsu, Japan). Piezo2-GFP-IRES-Cre mice (Piezo2tm1.1(cre)Apat (MGI:5574478)) and Piezo2+/− mice (Piezo2tm2.2Apat (MGI:5574479))22 were kind gifts from Prof. Keiko Nonomura and Prof. Ardem Patapoutian. All the mice were housed in the specific pathogen-free animal facility at Saga University with a 12-h light cycle and free access to water and food (CE-2; CLEA Japan, Inc., Tokyo, Japan). All animal experimental procedures were approved by the Animal Care and Use Committee of Saga University with No. 28–030–6, G2020–01–07, A2020–009–1, and A2023–026–0. We have complied with all relevant ethical regulations for animal use. The care of the animals was in accordance with institutional guidelines. Age-matched male and female mice aged 6–9 weeks were used for experiments. All mice were randomly selected for experiments.
Allergic asthma model
The allergic asthma model was generated as previously described40. The mice were intraperitoneally sensitized with 50 μg of ovalbumin (OVA) (A5503, Sigma‒Aldrich, St. Louis, MO, USA) containing 2 μl of aluminum gel (Alum) (LG-6000, LSL, Japan) diluted in 200 µl of phosphate-buffered saline (PBS) (10 ml/kg body weight) on days 0, 7, and 14 (Fig. 1A). The mice were intranasally challenged with 2.5 mg/ml OVA solution for four consecutive days. PBS- or Alum- and PBS-sensitized mice were used as controls (Fig. 1A). For Yoda1-treated mice, we intraperitoneally injected Yoda1 (SML1558, Sigma, St. Louis, MO) into PBS- or OVA+Alum (O + A)-sensitized mice at days 1–5 and 8–12 (Fig. 5A). Yoda1 was dissolved in 40 mM DMSO as a stock, diluted in 5% ethanol and administered intraperitoneally at a dose of 5 µmol/kg body weight.
Histology and histochemistry
The mice were deeply anaesthetized with a mixture of midazolam (4 mg/kg, 042824; Maruishi), butorphanol tartrate (5 mg/kg, VETLI5; Meiji Seika Pharma), and hydrochloric acid medetomidine (0.3 mg/kg, 9021; Kyoritsu Seiyaku) and then intracardially perfused with heparinized 0.1 M PBS, followed by 4% paraformaldehyde in 0.1 M phosphate buffer. The lungs, femurs, and tibias were removed and postfixed overnight at 4 °C. Femoral or tibial tissues were decalcified in 10% ethylenediaminetetraacetic acid for ten days. After cryoprotection with 20% sucrose overnight, the tissues were embedded in Tissue-Tek OCT compound (Sakura Finetek Japan, Tokyo, Japan). Five-micron-thick lung sections were cut with an FSE cryostat (Thermo Fisher Scientific, Waltham, MA, US) and placed on microslide glass (Frontier FRC-01, Matsunami Glass Ind., Ltd., Kishiwada, Japan). The sections were stained with periodic acid-Schiff (PAS) and Giemsa solutions. For alkaline phosphatase (ALP) staining, longitudinal 5 µm thick sections of bones were incubated with 5% N,N-dimethylformamide (045-02916; Wako Pure Chemical Industries), 1 mg/ml naphthol AS-MX phosphate (N4875-1G; Sigma‒Aldrich), and 1 mg/ml fast blue BB salt (066-05461; Wako Pure Chemical Industries) in 0.1 mM Tris-HCl (pH 9.2) at 37 °C for 30 min. TRAP staining was performed using an acid phosphatase leukocyte kit (387 A; Sigma‒Aldrich). Images were taken with an Axio Imager M2 microscope (Carl Zeiss) or a Nanozoomer S60 microscope (Hamamatsu Photonics K.K., Shizuoka, Japan).
Microcomputed tomography analysis
The right femurs and tibias were removed from the surrounding soft tissues and stored in 70% ethanol at 4 °C. The distal femurs or proximal tibias were scanned with a SkyScan 1272 microcomputed tomography (µCT) system (Bruker, Kontich, Belgium) and analyzed using CTan software (version 1.20.8.0, Bruker). The primary spongiosa region was extracted by segmenting the epiphysis, growth plate, and diaphysis from the distal femur or proximal tibia. The interface between the growth plate and the metaphysis was established as the reference plane, with the primary spongiosa region defined as the area extending 150 µm (for the femur) or 120 µm (for the tibia) from this reference plane towards the diaphysis. The trabecular bone volume (BV/TV), trabecular thickness (Tb.Th), trabecular number (Tb.N), trabecular separation (Tb.Sp), trabecular bone pattern factor (Tb.Pf), and structure model index (SMI) were analyzed. 3D-rendered images of the VOI were produced by CTvox software (version 3.3.1, Bruker).
Antibody generation
The guinea pig polyclonal anti-mouse Piezo1 antibody was raised against a peptide corresponding to mouse Piezo1 (residues 1759 to 1773: VLRRYENKPYFPPRI). A rabbit polyclonal anti-mouse Piezo2 antibody was raised against a peptide corresponding to mouse Piezo2 (residues 401 to 413: SMTQDDYKPSDGL). Both antibodies were affinity-purified with the peptide. HEK293 cells transfected with either mouse Piezo1 or Piezo2 (kind gifts from Prof. Makoto Tominaga and Dr. Yasunori Takayama) were used to assess antibody specificity. Immunoblotting revealed that the protein fractions of the membrane or cytosol of the transfected cells reacted with Piezo1 or Piezo2, respectively (Supplementary Fig. 3A). Immunofluorescent controls involved the use of no primary or secondary antibody or the replacement of the primary antibody with preabsorbed with the control peptide at 10−4–10−6 M (Supplementary Fig. 3B).
Immunofluorescence staining
Longitudinal serial femoral sections at a thickness of 5 μm were permeabilized with 0.3% Triton X-100 diluted in PBS for 10 min. After incubation in 5% normal donkey serum, 1% bovine serum albumin, or 0.05% sodium azide for 45 min at room temperature, the sections were incubated with primary antibodies overnight at 4 °C. For mouse antibodies, samples were processed with Mouse-on-Mouse (M.O.M.) mouse Ig blocking reagent (BMK-2202, Vector Laboratories, Burlingame, CA, US) for 1 h at room temperature and incubated with primary antibodies overnight at 4 °C. The primary antibodies used were as follows: Piezo1 (1 µg/ml), Piezo2 (1 µg/ml), Osterix (1:4000, ab22552, Abcam), type I collagen (1 µg/ml)41, podoplanin (1:1000, AF3244, R&D Systems), semaphorin 3 A (1 µg/ml, ab23393, Abcam), Piezo1 (1:400, 15939-1, Proteintech), Piezo1 (1:400, NBP1-78446SS, Novus), KDEL (1:200, M181-3, MBL), GM130 (1:200, BD 610822, BD Pharmingen), calnexin (1:3000, ab22595, Abcam), CD63 (1:600, BD 564221, BD Pharmingen), Lamp-1 (1:200, BD 553792, BD Pharmingen) and EEA1 (1:200, M176-3, MBL). After primary antibody incubation, the sections were washed and incubated with secondary antibodies for 1 h at room temperature. The secondary antibodies used were Alexa Fluor 488-conjugated donkey anti-rabbit IgG (1:200, 711-545-152, Jackson ImmunoResearch Laboratories), Alexa Fluor 594-conjugated donkey anti-guinea pig IgG (1:200, 706-585-148, Jackson ImmunoResearch Laboratories), Alexa Fluor 594-conjugated donkey anti-rat IgG (1:200, A21209, Thermo Fisher Scientific), and DyLight 649-conjugated donkey anti-mouse IgG (1:400, ab96878, Abcam). Filamentous actin was stained with Phalloidin-iFluor647 Reagent (1:1000, ab176759, Abcam) for 30 min. Nuclei were counterstained with 4’,6-diamidino-2-phenylindole dihydrochloride (DAPI) (1:1000, 340-07971, Dojindo Laboratories) for 5 min. The sections were mounted, and images were taken with a benchtop spinning disc confocal microscope BC43 (Oxford Instruments) with a Plan-Apochromat 20× objective (Nikon, Japan) or a confocal scanning microscope LSM800 with an Airyscan module (Carl Zeiss, Oberkochen, Germany) with a Plan-Apochromat 20× objective or Plan-Apochromat 63× oil immersion objective. Z stack images were taken in 16-bit mode using unidirectional scanning, with a digital zoom of 2.0, a field of view of 50 × 50 µm, and pixel scaling of (x, y, z) 29 × 29 × 160 nm. Superresolution images with XY resolution of ~120 nm and Z resolution of ~350 nm42 were obtained by Airyscan processing of the raw images with automatically determined superresolution parameters using ZEN Blue 3.5 software (Carl Zeiss). For Fig. 4A, Supplementary Fig. 4A–C, and Supplementary Fig. 5B–E, the raw images were processed using automatic parameters with additional 0.5 manual adjustments per channel. For Fig. 4C, images with XY resolution 90 nm and Z resolution of 270 nm43 were obtained by Airyscan Joint Deconvolution conducted at 5 iterations for DAPI, 10 iterations for Piezo2, and 20 iterations for Col1 signals. We confirmed that the present configuration showed little or no chromatic aberration by imaging 0.2-µm-diameter Tetraspeck microspheres (Invitrogen).
Correlative confocal laser scanning light microscopy (CLSM)–electron microscopy (EM)
The CLSM–EM method was performed according to the previous methods with slight modifications44,45,46. Serial 50-μm-thick sections were cut from decalcified femur using a vibrating microtome (Dosaka TTK-3000W). After cryoprotection with 25% sucrose in PBS, the sections were quickly frozen in liquid nitrogen vapor and rapidly thawed with 25% sucrose in PBS. The sections were incubated with 5% normal donkey serum (Jackson ImmunoResearch Laboratories) and 0.1% sodium azide in PBS for 1 h, followed by incubation with anti-Piezo1 (1:300) or anti-Piezo2 (1:2000) antibody at 4 °C for 4 days. The incubating medium did not contain any detergent, such as Triton-X100. After being rinsed several times in PBS, the sections were incubated with biotinylated anti-guinea pig IgG (1:250; Jackson ImmunoResearch Laboratories) or biotinylated anti-rabbit IgG (1:250; Jackson ImmunoResearch Laboratories) for Piezo1 and Piezo2 labeling, respectively, at 4 °C overnight, followed by incubation with Rhodamine red-conjugated streptavidin (1:500, Jackson ImmunoResearch) at 4 °C overnight. Hoechst33342 (1:1000) and phalloidin 647 (1:1000) were added to the solution during the last 30 min of incubation. Immunostained sections were rinsed several times, mounted in Vectashield (Vector Laboratories), and examined using a confocal laser-scanning light microscope (CLSM; model C2, Nikon) with a 60× objective (Plan Apo, ND = 1.40, Nikon). After CLSM, the coverslips were gently removed, and the sections were subjected to the ABC method (Vector Laboratories) using the DAB peroxidase reaction to visualize Piezo1 or Piezo2 antibodies. After the reaction, the sections were rinsed and treated with 1% OsO4 in 0.1 M phosphate buffer, en bloc staining with 1.5% uranyl acetate, dehydration in ethanol, and embedding in Epon-Araldite. The Piezo1- or Piezo2-immunolabelled osteoblasts on the trabecular or cortical bone observed by CLSM were examined by EM (HT7700, Hitachi).
Western blot analysis
Western blot analysis was performed as previously described47. Briefly, protein extracts from tissues were lysed in lysis buffer (50 mM Tris-HCl, 150 mM NaCl, 1% Triton X-100, 0.5% NP-40) containing protease inhibitor (Nacalai Tesque, Kyoto, Japan) and phosphatase inhibitor (PhosSTOP; Roche, Basel, Switzerland) for 10 min on ice. The protein extracts were subsequently centrifuged at 14,000 rpm at 4 °C for 20 min. The supernatants were collected, and the protein concentration was quantified with a Pierce BCA protein assay kit (23225, Thermo Fisher Scientific). Proteins were separated on 4–15% Mini-PROTEAN TGX gels (Bio-Rad Laboratories, Hercules, CA, US) and transferred to PVDF membranes (Immobilon P, Merck Millipore, Burlington, MA, US). The membranes were blocked with 5% bovine serum albumin for 1 h at room temperature and then incubated with primary antibodies overnight at 4 °C. The primary antibodies used were as follows: Piezo1 (1 µg/ml), Piezo2 (1 µg/ml), Osterix (1:4000, ab22552, Abcam), type I collagen (1 µg/ml), semaphorin 3 A (1 µg/ml, ab23393, Abcam), GAPDH (1:2000, sc25778, Santa Cruz Biotechnology), TBP (TATA-binding protein, 1:2000, #8515, Cell Signaling Technology), and pan-actin (1:2000, 12748, Cell Signaling Technology). After being washed three times with Tris-buffered saline with 0.1% Tween 20, the PVDF membranes were incubated with secondary antibodies for 1 h at room temperature. The secondary antibodies used were donkey anti-rabbit IgG (1:4000, NA934, GE Healthcare) and goat anti-guinea pig IgG (1:4000, 106-035-003, Jackson ImmunoResearch Laboratories). Immunoreactive bands were visualized using ECL Prime Western blotting detection reagent (RPN2232, GE Healthcare, Chicago, IL, US) and analyzed via a FUSION-FX7 imaging system (Vilber, Collégien, France).
Image analysis
The trabecular region of the distal femur extending 1.0 mm from the growth plate towards the diaphysis was measured. The osteoblast surface/bone surface (Ob. S/BS) from the ALP-stained images and the osteoclast surface/bone surface (Oc. S/BS) from the TRAP-stained images were quantified using ImageJ software (v2.14.0). The intensity of immunofluorescence and the number of Osx-positive cells and Piezo1- or Piezo2-positive surfaces per bone surface were measured by ImageJ. The intensity profile of immunofluorescence and the colocalization of immunoreactivities were determined with ZEN Blue 3.5 (Carl Zeiss).
Statistics and reproducibility
All the experiments were conducted at least three times independently under the same conditions. For data measurements, individual mice were used as experiment units. Graphical presentation was performed using JMP Pro 15 (SAS Institute Inc.) or GraphPad Prism 9.5.1 (GraphPad Software Inc.). Statistical differences were analyzed by one-way ANOVA with Tukey’s post hoc test for three groups using JMP Pro 15. The data are shown as the median with interquartile range (IQR) without exclusion, and differences were considered significant at p < 0.05.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article. All source data underlying graphs can be obtained in Supplementary data and unprocessed Western blots in Supplementary Fig.6.
References
Sims, N. A. & Martin, T. J. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep. 3, 481 (2014).
Shaheen, M. S. & Silverberg, J. I. Association of asthma with osteopenia, osteoporosis, osteomalacia, and fractures. Allergy Asthma Proc. 41, 112–119 (2020).
van Staa, T. P., Leufkens, H. G. & Cooper, C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos. Int. 13, 777–787 (2002).
Murata, N. et al. Effect of allergen sensitization on external root resorption. J. Dent. Res. 92, 641–647 (2013).
Chalitsios, C. V., McKeever, T. M. & Shaw, D. E. Incidence of osteoporosis and fragility fractures in asthma: a UK population-based matched cohort study. Eur. Respir. J. 57 https://doi.org/10.1183/13993003.01251-2020 (2021).
Florencio-Silva, R., Sasso, G. R., Sasso-Cerri, E., Simões, M. J. & Cerri, P. S. Biology of bone tissue: structure, function, and factors that influence bone cells. Biomed. Res. Int. 2015, 421746 (2015).
Bolamperti, S., Villa, I. & Rubinacci, A. Bone remodeling: an operational process ensuring survival and bone mechanical competence. Bone Res. 10, 48 (2022).
Coste, B. et al. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 330, 55–60 (2010).
Wang, L. et al. Mechanical sensing protein PIEZO1 regulates bone homeostasis via osteoblast-osteoclast crosstalk. Nat. Commun. 11, 282 (2020).
Sun, W. et al. The mechanosensitive Piezo1 channel is required for bone formation. Elife 8 https://doi.org/10.7554/eLife.47454 (2019).
Zhou, T. et al. Piezo1/2 mediate mechanotransduction essential for bone formation through concerted activation of NFAT-YAP1-ss-catenin. Elife 9 https://doi.org/10.7554/eLife.52779 (2020).
Caceres, A. I. et al. A sensory neuronal ion channel essential for airway inflammation and hyperreactivity in asthma. Proc. Natl. Acad. Sci. USA 106, 9099–9104 (2009).
Shen, V. et al. Short-term immobilization-induced cancellous bone loss is limited to regions undergoing high turnover and/or modeling in mature rats. Bone 21, 71–78 (1997).
Nakashima, K. et al. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 108, 17–29 (2002).
Nagai, T. et al. Immunocytochemical assessment of cell differentiation of podoplanin-positive osteoblasts into osteocytes in murine bone. Histochem. Cell Biol. 155, 369–380 (2021).
Hayashi, M. et al. Osteoprotection by semaphorin 3A. Nature 485, 69–74 (2012).
Negishi-Koga, T. & Takayanagi, H. Bone cell communication factors and Semaphorins. Bonekey Rep. 1, 183 (2012).
Li, X. et al. Stimulation of Piezo1 by mechanical signals promotes bone anabolism. Elife 8 https://doi.org/10.7554/eLife.49631 (2019).
Hendrickx, G. et al. Piezo1 inactivation in chondrocytes impairs trabecular bone formation. J. Bone Min. Res. 36, 369–384 (2021).
Zhang, G., Li, X., Wu, L. & Qin, Y. X. Piezo1 channel activation in response to mechanobiological acoustic radiation force in osteoblastic cells. Bone Res. 9, 16 (2021).
Sugisawa, E. et al. RNA sensing by gut piezo1 is essential for systemic serotonin synthesis. Cell 182, 609–624.e621 (2020).
Woo, S. H. et al. Piezo2 is required for Merkel-cell mechanotransduction. Nature 509, 622–626 (2014).
Sugimoto, A. et al. Piezo type mechanosensitive ion channel component 1 functions as a regulator of the cell fate determination of mesenchymal stem cells. Sci. Rep. 7, 17696 (2017).
Wei, C. et al. Calcium flickers steer cell migration. Nature 457, 901–905 (2009).
Saraste, J. & Prydz, K. A new look at the functional organization of the Golgi ribbon. Front. Cell Dev. Biol. 7, 171 (2019).
Barry, L. E. et al. Age and sex associations with systemic corticosteroid-induced morbidity in asthma. J. Allergy Clin. Immunol. Pract. 6, 2014–2023.e2012 (2018).
Zafar, T. A., Teegarden, D., Ashendel, C., Dunn, M. A. & Weaver, C. M. Aluminum negatively impacts calcium utilization and bone in calcium-deficient rats. Nutr. Res. 24, 243–259 (2004).
Lowe, K. E. et al. Atopic eczema and fracture risk in adults: A population-based cohort study. J. Allergy Clin. Immunol. 145, 563–571.e568 (2020).
Li, Z. et al. The role of semaphorin 3A in bone remodeling. Front Cell Neurosci. 11, 40 (2017).
Adi, S. D. et al. Semaphorin 3A is effective in reducing both inflammation and angiogenesis in a mouse model of bronchial asthma. Front. Immunol. 10, 550 (2019).
Yamamoto, T. et al. Structure and formation of the twisted plywood pattern of collagen fibrils in rat lamellar bone. J. Electron Microsc61, 113–121 (2012).
Ranade, S. S. et al. Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature 516, 121–125 (2014).
Maksimovic, S. et al. Epidermal Merkel cells are mechanosensory cells that tune mammalian touch receptors. Nature 509, 617–621 (2014).
Szczot, M. et al. Cell-type-specific splicing of piezo2 regulates mechanotransduction. Cell Rep. 21, 2760–2771 (2017).
Pardo-Pastor, C. et al. Piezo2 channel regulates RhoA and actin cytoskeleton to promote cell mechanobiological responses. Proc. Natl. Acad. Sci. USA 115, 1925–1930 (2018).
Baghdadi, M. B. et al. PIEZO-dependent mechanosensing is essential for intestinal stem cell fate decision and maintenance. Science 386, eadj7615 (2024).
Hill, R. Z., Loud, M. C., Dubin, A. E., Peet, B. & Patapoutian, A. PIEZO1 transduces mechanical itch in mice. Nature 607, 104–110 (2022).
Hao, R. et al. A novel Piezo1 agonist promoting mesenchymal stem cell proliferation and osteogenesis to attenuate disuse osteoporosis. Small Sci. 4, 2400061 (2024).
Chesler, A. T. et al. The role of PIEZO2 in human mechanosensation. N. Engl. J. Med. 375, 1355–1364 (2016).
Cao, A. et al. Transient receptor potential channel vanilloid 1 contributes to facial mechanical hypersensitivity in a mouse model of atopic asthma. Lab. Investig. 103, 100149 (2023).
Ohsaki, Y., Nagata, K. & Kurisu, K. Localization of types I and III collagen and fibronectin in the developing mouse palatal shelves. Acta Anat.153, 161–167 (1995).
Huff, J. The Airyscan detector from ZEISS: confocal imaging with improved signal-to-noise ratio and super-resolution. Nat. Methods 12, i–ii (2015).
Prazeres da Costa O. et al. A practical guide of deconvolution. (Carl Zeiss) https://pages.zeiss.com/rs/896-XMS-794/images/ZEISS-Microscopy_A-Practical-Guide-of-Deconvolution.pdf (2021).
Fukuda, T. Structural organization of the dendritic reticulum linked by gap junctions in layer 4 of the visual cortex. Neuroscience 340, 76–90 (2017).
Ogata, S., Miyamoto, Y., Shigematsu, N., Esumi, S. & Fukuda, T. The tail of the mouse striatum contains a novel large type of gabaergic neuron incorporated in a unique disinhibitory pathway that relays auditory signals to subcortical nuclei. J. Neurosci. 42, 8078–8094 (2022).
Shigematsu, N., Nishi, A. & Fukuda, T. Gap junctions interconnect different subtypes of parvalbumin-positive interneurons in barrels and septa with connectivity unique to each subtype. Cereb. Cortex 29, 1414–1429 (2019).
Aijima, R. et al. The thermosensitive TRPV3 channel contributes to rapid wound healing in oral epithelia. Faseb J. 29, 182–192 (2015).
Acknowledgements
The authors thank the Division of Biological Resources and Development, Analytical Research Center for Experimental Sciences of Saga University for animal care, and the Division of Instrumental Analysis, Analytical Research Center for Experimental Sciences of Saga University for their technical assistance. This work was supported by grants from the Japan Society for the Promotion of Science (JSPS) KAKENHI grant number JP20K21683 as Grant-in-Aid for Challenging Research (Exploratory) to M.A.K., JP24K23594 as Grant-in-Aid for Research Activity Start-up to W.G., and from the Japanese Society for Bone and Mineral Research as the JSBMR Frontier Scientist Grant to M.A.K.
Author information
Authors and Affiliations
Contributions
Weiqi Gao: methodology, software, validation, formal analysis, investigation, data curation, writing—original draft preparation, visualization, funding acquisition, Takeshi Sawada: methodology, software, investigation, data curation, visualization, Ailin Cao: methodology, investigation; Reiko U. Yoshimoto: investigation, resources, writing; Yu Yamaguchi: investigation, resources, data curation; Yuki Takahashi: investigation, data curation, Takaichi Fukuda: investigation, data curation, Yasuyoshi Ohsaki: resources, validation, Reona Aijima: investigation, resources, data curation, Tomoko Kadowaki: resources, data curation, Takayuki Tsukuba: Resources, data curation, Mizuho A. Kido: conceptualization, methodology, resources, writing— original draft preparation, review and editing, visualization, supervision; project administration; funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Patrycja Nejman-Gryz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Marco Fritzsche and Joao de Sousa Valente. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, W., Sawada, T., Cao, A. et al. Ovalbumin-induced asthma leads to bone loss with Piezo channel suppression in mice. Commun Biol 8, 1309 (2025). https://doi.org/10.1038/s42003-025-08753-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-08753-x