Abstract
Background:
Understanding the temporal trends of blood-based biomarkers and their associations with brain structure is crucial for early detection and intervention in psychiatric disorders. This study aimed to explore these trends in the decade before and after diagnosis, along with the cross-sectional relationships with brain structures.
Methods:
Utilizing UK Biobank data, we conducted a nested case-control analysis of individuals aged 40-69 years diagnosed with anxiety (n = 27,216), bipolar disorder (n = 1325), depression (n = 36,570), or schizophrenia (n = 1478) within 10 years of baseline. We used multivariable linear regression to analyze temporal trends and brain structure associations for 31 blood cell counts, 28 biochemistry markers, and 168 serum metabolites.
Results:
Here we show that compared to controls, significant temporal divergence is observed in 39, 6, 55, and 12 blood-based markers for anxiety, bipolar disorder, depression, and schizophrenia, respectively. Common biomarkers like cystatin C, red blood cells, hemoglobin, hematocrit, and total bilirubin are identified. Biomarkers cluster into groups with either linear or non-linear trends. Among the linearly changing biomarkers, some have a widening difference from controls while others have a narrowing one. For example, in the case of depression, HDL-TG demonstrates an increasing disparity over time, while cholesterol exhibits a decreasing trend. Non-linear clusters often show reversals around diagnosis, indicating potential treatment effects. Differential associations are found between biomarkers and brain regions, including the orbitofrontal cortex, hippocampus, and accumbens.
Conclusions:
This study reveals the temporal trends of blood-based biomarkers in psychiatric disorders and their correlations with brain structure, aiding early detection and potentially enhancing clinical outcomes.
Plain language summary
This study aimed to understand whether components of the blood change over time in people with psychiatric disorders such as anxiety, bipolar disorder, depression, and schizophrenia. We used statistical methods to compare components of the blood between people with psychiatric disorders and controls. We found that certain blood markers changed differently in people with psychiatric disorders. Some markers show changes around the time of diagnosis, potentially linked to treatment effects. The markers could help detect the presence of a psychiatric disorder earlier and guide the choice of treatments.
Similar content being viewed by others
Introduction
More than 14% of adults aged 60 and above suffer from a psychiatric disorder, and the prevalence increases with age1,2. Considering the rapid aging of the global population3,4, it is expected that a growing number of elderly individuals will experience psychiatric disorders in the coming years. Psychiatric disorders are often accompanied by a multitude of debilitating symptoms5, including emotional dissonance, cognition decline, motor dysfunction and disturbances of the metabolic and immune system6,7,8,9,10,11. These alterations not only have an impact on health in their own right but also increase the risk of other adverse health outcomes, encompassing neurodegenerative disorders12, cardiovascular disease13,14 and cognitive impairment2. They can also result in a substantial reduction in life expectancy (10–20 years)15,16,17. Early intervention at the onset of psychiatric disorders can improve a range of outcomes18. However, older adults frequently face insufficient recognition and undertreatment of their psychiatric health conditions.
Recent evidence has revealed that metabolic markers in psychiatric disorders change compared with controls and differ significantly between different psychiatric disorders19,20,21,22. There are also studies that identified causal roles for certain metabolites in psychiatric disorders, suggesting that metabolic dysregulation contributes to the development of psychiatric disorders11,23,24. Various immune biomarkers, such as C-reactive protein (CRP) and immune cell count, have also been associated with psychiatric disorders25,26,27,28. However, a comprehensive understanding of the temporal trends of these blood-based biomarkers throughout the pre- and post-diagnostic phases of various psychiatric disorders is currently lacking. Understanding these trends is crucial because it can reveal early signs of psychiatric disorders, allowing for timely interventions. Traditional case-control or causal studies often focus on a single point in time, whereas examining the temporal trends can provide insights into the progression and underlying mechanisms of these disorders. Additionally, the association between different blood-based biomarkers and brain imaging findings remains uninvestigated. Understanding their relationship could reveal which biomarkers can be intervened on to reduce the impact of the disease on the brain.
This study leverages data from the UK Biobank (UKB)29 to examine temporal trends of blood-based biomarkers in psychiatric disorders. The cohort includes over 500,000 middle-aged to elderly adults with comprehensive measurements of blood cell counts, biochemical markers, and metabolomic profiles, including participants diagnosed with anxiety, bipolar disorder, depression, and schizophrenia. Through nested case-control analysis, we investigate temporal divergence patterns of these biomarkers relative to controls across pre- and post-diagnostic phases, while assessing their differential associations with brain structure using neuroimaging data in a case-control design. Our findings demonstrate that identified biomarkers exhibit linear or non-linear temporal trajectories, with non-linear patterns frequently reversing near diagnosis, potentially reflecting treatment effects. Furthermore, specific biomarkers show significant differential associations with neuroanatomical measures in brain regions, such as the orbitofrontal cortex and hippocampus. Collectively, these results reveal complex temporal trends of peripheral biomarkers in psychiatric disorders and their differential associations with brain structure compared to controls.
Methods
Study cohort
The data employed in this study were retrieved from the UKB29. The UKB is a large-scale prospective cohort study. It encompasses over 500,000 participants from 22 assessment centers. These participants were aged between 40 and 69 years at the baseline, which occurred from March 2006 to October 201029. The dataset encompasses comprehensive information on participants’ demographic, behavioral, blood, neuroimaging assessments, and health-related outcomes. All these data were preprocessed prior to the analyses of the study.
This study was conducted using the UKB resource under application number 19542. Ethics approval for the UKB study was obtained from the North West Haydock Research Ethics Committee (REC) (REC Reference: 21/NW/0157). All participants provided written informed consent, emphasizing the voluntary nature of participation and the freedom to withdraw at any time without explanation.
Outcome ascertainment
We explored four psychiatric disorders, including anxiety, depression disorders, bipolar disorders, and schizophrenia. Diagnosis and recorded dates were ascertained and classified according to the International Classification of Diseases (ICD), Tenth Revision (ICD-10), specifically code G20, extracted from first occurrences data (UKB category 1712) which is identified through linkage to primary care (category 3000), hospital inpatient (category 2000), self-reported medical conditions (UKB field 20,002) and death register records (field 40,001 and 40,002). The categories of psychiatric disorders were described as codes F10-19, F20, F30–F39, etc. in ICD-10 (Supplementary Table 1). The date of any earliest recorded diagnosis listed above was leveraged as the date of diagnosis.
In this study, cases indexed only as self-reported sources and lacking metabolomic data were excluded. Additionally, to avoid potential sample bias, individuals who were diagnosed with psychiatric disorders more than ten years before or after the baseline visit were also excluded. The flow chart of eligible standards was demonstrated in Supplementary Fig. 1.
Psychiatric assessments
The patient health questionnaire-430 (PHQ-4) and the Eysenck personality questionnaire-revised (EPQ-R)31 were utilized to evaluate self-reported symptoms of participants. The PHQ-4 instrument combines a 2-item depression scale (PHQ-2) and a 2-item anxiety scale (general anxiety disorder (GAD)−2). Both subscales employ a 4-point Likert-type scale. Participants need to rate the frequency of experiencing the symptoms described in the items over the past two weeks. By adding up the scores from these two subscales, we obtained a PHQ-4 total score. This score acts as an indicator of the severity of depressive and anxiety symptoms. Higher scores indicate greater symptom intensity.
Additionally, we utilized the EPQ-R, focusing specifically on the neuroticism dimension, which comprises 12 items. Participants’ responses to these questions, with ‘Yes’ answers contributing to the neuroticism score, are aggregated into a single integer for each individual, providing a quantitative measure of neuroticism traits. The specific items used to assess each symptom are provided in Supplementary Table 2.
Blood-based biomarker measurements
Blood samples of participants were collected at baseline (2006–2010) by the UKB. The processing details and quality control checks of blood samples can be found on the UKB website (https://www.ukbiobank.ac.uk/enable-your-research/about-our-data/biomarker-data). In this research, 31 blood cell counts, 28 biochemistry markers, and 168 serum metabolites were utilized. A full list of abbreviations and Field ID of for all blood-based markers in this study is provided in Supplementary Data 1.
Standard hematological assays, such as full blood counts and related parameters, were performed on fresh whole blood within 24 h of blood collection for all participants. Multiple immunological assays and biological chemistry analyzers were employed to measure blood biochemistry markers. The details thereof are provided on the UKB website (https://www.ukbiobank.ac.uk/enable-your-research/about-our-data/biomarker-data). The metabolic biomarker profiling of participants from the UKB is being carried out by Nightingale Health Ltd32,33,34. This profiling is based on high-throughput nuclear magnetic resonance. To minimize technical variability that could confound our results, raw data were carefully processed, taking into account factors, such as shipping batch, 96-well plate layout, well position, aliquoting robot, and aliquot tip, all before analysis by Nightingale Health Plc33,34. A total of 168 metabolic measures in absolute levels are quantified for each EDTA plasma sample. These measures encompass specific aspects of lipoprotein lipids across 14 subclasses, fatty acid profiles and compositions, cholesterol metabolism, and a diverse range of low-molecular weight metabolites. The latter include amino acids, metabolites associated with glycolysis, and ketone bodies.
Neuroimaging phenotypes
The neuroimaging phenotypes used in this study were broadly categorized into two groups: T1-weighted MRI for quantifying cortical and subcortical volumes, and diffusion tensor imaging (DTI) for assessing white matter microstructure. All neuroimaging data originated from the UKB Imaging Database. These data were processed before any analyses. The detailed protocol is available at https://biobank.ctsu.ox.ac.uk/crystal/refer.cgi?id=2367.
T1-Weighted MRI
T1-weighted MRI data were acquired using a 3 T Siemens Skyra scanner with the following parameters: 1 × 1 × 1 mm³ resolution, 208 × 256 × 256-pixel field of view, 5 min duration, and acceleration factor (iPAT) of 2. Pre-scan normalization was also applied (https://biobank.ndph.ox.ac.uk/showcase/refer.cgi?id=1977). The UKB pre-processed and quality-controlled the images using FMRIB Software Library packages following a standard protocol35. The data is now nonlinearly warped to MNI152 space using FNIRT (FMRIB’s Nonlinear Image Registration Tool36,37). A standard-space brain mask is then back-transformed into the space of the T1, and applied to the T1 image to generate a brain-extracted T1. Next, tissue-type segmentation is applied using FMRIB’s Automated Segmentation Tool38 gray matter and white matter (T1_brain_seg) and so on. This processing is also used to generate a fully bias-field-corrected version of the brain-extracted T1. The T1 images were also processed with FreeSurfer. Surface atlases were used to extract IDPs relating to the volume of standard atlas regions39,40. Subcortical regions were extracted via FreeSurfer’s aseg tool41. Quality control checks were performed using the Qoala-T approach42.
Diffusion tensor imaging
Water molecule movement within the local tissue environment was measured using DTI. Metrics reflecting white matter integrity and microstructural properties were calculated, including fractional anisotropy (FA), mean diffusivity (MD), and intracellular volume fraction (ICVF). FA and MD were derived from the DTI fitting tool DTIFIT using the b = 1000 shell (50 directions). ICVF was obtained after processing the full two-shell DTI data using the neurite orientation dispersion and density imaging43 modeling with the accelerated microstructure imaging via convex optimization tool44.
In the analysis, individuals with conditions confounding the assessment of brain atrophy were excluded. These conditions included brain tumor, neurodegenerative disease, cerebrovascular diseases, cerebral infarction, epilepsy, multiple sclerosis, encephalitis, or brain abscess, which were identified using ICD-10 codes and self-reported diseases (Supplementary Table 3).
Nested case-control study
We conducted a nested case-control study within the UKB cohort, adeptly harnessing the merits of both cohort and case-control study designs. A nested case-control study, essentially a case-control study nested within a cohort, combines the strengths of both methodologies to offer a robust epidemiological research framework10,45,46. This involved initially forming a cohort of participants, tracking them over an extended period, and subsequently identifying within this cohort. In this study, individuals with anxiety, depression, bipolar disorder, or schizophrenia were identified as cases. Controls, on the other hand, were selected from those without any blood or immune disorders, mental health or behavioral issues, infectious diseases, or nervous system disorders at baseline. For each case identified, we matched approximately three controls based on various demographic and lifestyle factors, including age, sex, ethnicity, years of education, body mass index (BMI), socioeconomic status, alcohol consumption status, and smoking status, utilizing the Matchit R package47. To analyze the temporal trends of the disease, we calculated the years since clinical diagnosis for each case by subtracting the baseline date from the diagnosis date. For controls, we assigned a pseudo-diagnosis date that mirrored the diagnosis date of their matched case. This arrangement yielded a comprehensive dataset of matched case-control pairs, spanning consecutive time frames before and after diagnosis. This unique dataset allowed us to delve into the temporal divergence between cases and controls over time, specifically examining the divergence patterns across various psychiatric assessments and blood-based biomarkers. The study design was displayed in Fig. 1.
This figure presents a nested case-control study using the UKB dataset. The solid horizontal bars indicate the estimated duration from blood biomarker assessments to the clinical diagnosis of psychiatric disorders. They start from baseline visits and reach the dates of diagnosis, or vice versa. For the purpose of analysis, we aligned the patients based on the diagnosis dates. On the new timeline, the data collected at the baseline became the data collected at different time periods before and after the diagnosis. We used a color gradient from dark blue (representing zero years to diagnosis) to light blue (indicating ten years to diagnosis). For each identified case, we matched ~3 controls. For the controls, their corresponding duration was matched to that of the individuals with psychiatric disorders. Subsequently, we applied the locally estimated scatterplot smoothing (LOESS) technique to visualize the temporal variations in psychiatric assessments and blood-based biomarkers related to the timing of disease duration. To reveal the specific temporal patterns of blood-based markers that change during disease progression, we clustered them into different groups based on their temporal trends. Finally, we tested the differential associations between the identified blood-based biomarkers and brain structures within the case-control framework, using a multivariable linear regression model for statistical analysis.
Statistics and reproducibility
Statistical analysis of the temporal divergence trend in biomarkers for psychiatric disorders
Our primary objective was to identify the temporal divergence trend in blood-based markers between individuals with psychiatric disorders and controls. We used a multivariable linear regression model to compare each variable of interest between the two groups. To evaluate the effect of disease on blood markers, as well as the linear and nonlinear changes of blood markers with time to diagnosis, we included group and its interactions with time and the square of time as independent variables, with blood-based markers as the response variable48. Testing the effect of group allowed us to determine the differences in blood markers between cases and controls independently of time. Evaluating the interaction between group and time enabled us to identify the divergence in the linear trajectories of blood markers between cases and controls. Assessing the interaction between group and time squared helped us understand the differences in nonlinear trajectories of blood markers between cases and controls. Other independent variables included age, sex, 5-year age indicators by sex interactions, self-identified ethnicity, self-identified ethnicity by sex interactions, study site, years of education, Townsend deprivation index (TDI), study site, BMI, drinking status, and smoking status, fasting time (one indicator per fasting time, except a single indicator for >18 h and for 0 or 1 h), estimated sample dilution factor (icosatiles), icosatiles of time of sampling during the day, month of assessment (indicators for each month of participation, with the exception that all of 2006 and August through October of 2010 were assigned a single indicator), and day of assay (one indicator per day the assay was performed). Ethnicity was categorized as white, asian, black, and other ethnicity (e.g., mixed ethnicity or other ethnicity).
Continuous variables were normalized before regression, and categorical variables were made dummy variables. Missing values were not included in the analysis. P values for group and its interactions with time and the square of time were corrected for multiple comparisons using the Bonferroni method, respectively. Blood-based markers with any corrected p-values less than 0.01 were used for temporal trends visualization. The analysis was performed using the StatsModel (v0.11.1) and ScikitLearn (v0.24.1) packages in Python (v3.9).
Trajectory analysis
To visualize the temporal trends of blood-based markers in psychiatric disorders, we employed the LOESS49 method to assess linear or non-linear patterns in blood-based marker evolution over time. To ensure that each assessment’s values were easily comparable, blood-based marker values for psychiatric disorders were transformed using z-scores relative to controls, adjusting for the aforementioned covariates. The predicted trajectories over time were subsequently divided into groups through hierarchical clustering. This was accomplished by utilizing the hclust function from the R stats package, employing the complete method47.
Statistical analysis of biomarker-brain structure associations
Given that blood-based biomarkers and imaging-derived metrics required adjustment for different covariate sets, we first performed separate regression analyses for each domain prior to the differential associations analyses. For blood-based biomarkers, covariates included fasting time, estimated sample dilution factor, time of blood sample collection (as described above), and baseline recruitment site. For imaging-derived measures, covariates comprised intracranial head size (to account for global brain volume effects) and imaging site. Residuals from these regressions—reflecting the variance in biomarkers and brain structures independent of their respective covariates—were then used in subsequent models to examine group-specific associations, ensuring that technical and demographic confounders were systematically removed.
To investigate differential associations between the identified blood-based biomarkers and specific brain structures in a case-control framework, we utilized a multivariable linear regression model. This model included interactions between the group factor (case vs. control) and each blood-based biomarker, alongside common covariates: age, sex, age by sex interactions, self-identified ethnicity, self-identified ethnicity by sex interactions, years of education, TDI, BMI, drinking status, and smoking status. The dependent variables were the brain structural measures of interest (e.g., regional volumes, DTI metrics). This approach allowed us to test whether the relationships between biomarkers and brain structures differed significantly between cases and controls. In this study, all tests were carried out on a two-sided basis.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Results
Study populations
In this study, 56,535 individuals diagnosed with psychiatric disorders were included, with a median age of 56.00 [IQR 49.00–62.00]. Among them, 20,520 (36.30%) were male, and the majority (95.33%) had white ethnicity. The sample included 27,216 individuals diagnosed with anxiety, 1325 with bipolar disorder, 36,570 with depression, and 1478 with schizophrenia. Nearly three controls were matched for each case. Therefore, a total of 206,483 individuals were included in the main analyses. Table 1 presents the demographic information of these participants. Similar demographic statistics were observed between the case and control groups.
Temporal divergence of psychiatric assessments trends in psychiatric disorders compared to controls
The results of two psychiatric assessments indicate a significant temporal divergence trend between individuals with psychiatric disorders and controls. Among the four psychiatric disorders, neuroticism scores in affected individuals showed a consistent increase in the ten years preceding diagnosis, compared to controls (Fig. 2). In cases of anxiety and depression, neuroticism scores continued to increase after diagnosis. In contrast, for individuals with schizophrenia and bipolar disorder, neuroticism scores began to decrease around the time of diagnosis. Among the four psychiatric disorders, the temporal trend of the PHQ-4 scores presented an inverted U-shaped pattern when compared with the control group (Fig. 2).
The temporal trends of the PHQ-4 score and the neuroticism score were analyzed using a LOESS regression model on z-scores across time, both before and after the diagnosis of four psychiatric disorders. The number of participants included in the analysis for each disorder— anxiety, bipolar disorder, depression, and schizophrenia—was 27,216, 1325, 36,570, and 1478, respectively. Before being transformed using z-scores relative to controls, psychiatric assessments for psychiatric disorders were adjusted for age, gender, ethnicity, educational years, the Townsend Deprivation Index, drinking status, and smoking status.
Identified blood-based biomarkers in psychiatric disorders compared with controls
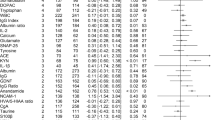
After applying a multivariable linear regression model, conducting multiple corrections, and adjusting for covariates, we identified 39, 6, 55, and 12 blood—based biomarkers that exhibited divergent temporal trends compared to the control group for anxiety, bipolar disorder, depression, and schizophrenia, respectively (corrected P < 0.01; Fig. 3 and Supplementary Data 2–5). Among these psychiatric disorders, there were some common markers. For instance, cystatin C levels were elevated in all four disorders, whereas red blood cells (RBC), hemoglobin concentration (Hb), hematocrit percentage (Hct), and total bilirubin levels were decreased in all of them. Some blood markers were shared among different disorders and showed consistent directions of change. For example, alkaline phosphatase (ALP), CRP, gamma-gamma-glutamyl transferase (GGT), mean reticulocyte volume (MRV), mean sphered cell volume (MSCV), and neutrophil percentage (NEUT-P) were all elevated in depression, anxiety, and schizophrenia. Lymphocyte percentage (LYMP-P) was significantly decreased in anxiety, bipolar disorder, and depression. Vitamin D and mean corpuscular hemoglobin concentration (MCHC) were significantly decreased in both depression and schizophrenia. A specific number of metabolic markers were found in both depression and anxiety, with some of them being shared, such as low-density lipoprotein cholesterol (LDL-C), LDL-free cholesterol (FC), and intermediate-density lipoprotein cholesterol. Furthermore, specific triglyceride-related metabolic markers were identified in cases of depression.
The Y-axis displays the beta values, representing the effect size of differences for various blood-based biomarkers from controls for each disorder. Regarding the beta values of the interaction between the group and time, please refer to Supplementary Data 2–5. The X-axis categorizes these biomarkers into different types. Statistically significant biomarkers are labeled with their names. Panels a, b, c and d correspond to anxiety disorder (n = 26,839), bipolar disorder (n = 1295), depression (n = 36,088), and schizophrenia (n = 1440), respectively.
Temporal divergence of blood-based biomarker trends in psychiatric disorders compared to controls
The identified blood-based markers for anxiety, depression, bipolar disorder, and schizophrenia were respectively grouped into five, four, nine, and four clusters according to their trajectory (Supplementary Fig. 2 and Supplementary Data 6). These markers showed significant linear or non-linear divergence compared to controls.
In anxiety disorder, five clusters of blood-based markers were identified (Fig. 4 and Supplementary Fig. 3). Three clusters consistently showed higher levels than controls throughout the observation period, while two clusters remained persistently lower. Cluster 1, which included GGT and cystatin C, demonstrated a gradual reduction in differences from controls as the disease progressed. The second cluster, containing mean corpuscular volume (MCV), RDW, NEUT-C, NEUT-P, MRV, MSCV, immature reticulocyte fraction (IRF), high light scatter reticulocyte, ALP, aspartate aminotransferase (AST), and CRP, also showed a decreasing difference, though less pronounced. Cluster 3 consisted solely of triglycerides, whose difference trajectory exhibited a non-linear upward trend as the disease advanced. Clusters 4 and 5 remained consistently lower than controls, following a slightly positive U-shaped curve with the inflection point near the time of diagnosis. The majority of these markers within Clusters 4 and 5 were associated with metabolism.
The temporal trends of blood-based markers that showed significant divergence from controls were presented. The details of the statistics of identified blood-based markers of anxiety disorder were provided in Supplementary Data 2. Each significant biomarker was analyzed using a LOESS regression model on z-scores across time, both before and after the diagnosis. Before being transformed using z-scores relative to controls, blood-based markers were adjusted for covariates. Then, biomarkers were grouped based on their similarity of trajectories, and bold lines represent the average temporal trend of each group. LOESS, locally estimated scatterplot smoothing.
For bipolar disorder, four clusters of blood-based biomarkers were identified (Fig. 5 and Supplementary Fig. 4). Only cystatin C in cluster 1 remained consistently above control levels throughout the observation period, following a slightly inverted U-shaped trajectory. The markers in Clusters 2–4 were consistently lower than controls. Cluster 2, including Hct, RBC, and Hb, exhibited a positive U-shaped curve. Cluster 3 contained only total bilirubin, with differences from controls continuously expanding as the disease progressed. Cluster 4, containing only LYMP-P, demonstrated a continuous reduction in the difference from controls during the observation period.
The temporal trends of blood-based markers that showed significant divergence from controls were presented. The details of the statistics of identified blood-based markers of bipolar disorder were provided in Supplementary Data 3. Each significant biomarker was analyzed using a LOESS regression model on z-scores across time, both before and after the diagnosis. Before being transformed using z-scores relative to controls, blood-based markers were adjusted for covariates. Then, biomarkers were grouped based on their similarity of trajectories, and bold lines represent the average temporal trend of each group. LOESS, locally estimated scatterplot smoothing.
In depression, nine clusters of blood-based biomarkers were identified (Fig. 6 and Supplementary Fig. 5). Four clusters remained consistently higher than controls, while five clusters were persistently lower. Cluster 1, which included GlycA, IRF, and certain triglyceride-related metabolic markers, was overall higher than controls, with the difference progressively widening as the disease advanced. Clusters 2 to 4 exhibited differences above control levels throughout the observation period, albeit with less conspicuous temporal variation, including markers, such as mean corpuscular hemoglobin (MCH), ALT, Cystatin C, MCV, CRP, and ALP, among others. Clusters 5 to 6 remained lower than controls, also with relatively subtle temporal changes, encompassing markers like Apolipoprotein A, Urate, LYMP-P, insulin-like growth factor 1, and vitamin D, among others. Cluster 7 demonstrated a slightly positive U-shaped trajectory compared to controls, remaining consistently lower, and included RBC, Hb, Hct, and degree of unsaturation. Cluster 8 displayed an inverted U-shaped temporal pattern in biomarker levels relative to controls, with these biomarkers consistently maintaining lower concentrations throughout the observed timeframe. This pattern was particularly evident in FC-associated biomarkers, including S-LDL-FC, M-LDL-FC, L-LDL-FC, and LDL-FC. In cluster 9, although the biomarkers were lower than controls a decade before diagnosis, the difference progressively narrowed as the disease advanced, including Apo-B, LDL-C, and cholesterol.
The temporal trends of blood-based markers that showed significant divergence from controls were presented. The details of the statistics of identified blood-based markers of depression were provided in Supplementary Data 4. Each significant biomarker was analyzed using a LOESS regression model on z-scores across time, both before and after the diagnosis. Before being transformed using z-scores relative to controls, blood-based markers were adjusted for covariates. Then, biomarkers were grouped based on their similarity of trajectories, and bold lines represent the average temporal trend of each group. LOESS, locally estimated scatterplot smoothing.
In schizophrenia, the significant temporal divergence trends of the four clusters all conformed to U-shaped curves (Fig. 7 and Supplementary Fig. 6). Clusters 1 and 2 were above control levels throughout the observation period, with an overall inverted U-shaped change trajectory. Cluster 1 included GGT and RDW, while Cluster 2 comprised MRV, CRP, cystatin C, and NEUT-C. Notably, although Cluster 2 as a whole exhibited an inverted U-shaped trajectory, NEUT-C followed a positive U-shaped curve, yet its difference range was remarkably close to that of the other markers in Cluster 2. The trajectories of clusters 3 and 4 were both positive U-shaped, with cluster 3 displaying a slightly larger amplitude of change. Cluster 3 contained markers such as RBC, Hct, Hb, and Vitamin D, while cluster 4 encompassed MCHC and total bilirubin.
The temporal trends of blood-based markers that showed significant divergence from controls were presented. The details of the statistics of identified blood-based markers of schizophrenia were provided in Supplementary Data 5. Each significant biomarker was analyzed using a LOESS regression model on z-scores across time, both before and after the diagnosis. Before being transformed using z-scores relative to controls, blood-based markers were adjusted for covariates. Then, biomarkers were grouped based on their similarity of trajectories, and bold lines represent the average temporal trend of each group. LOESS, locally estimated scatterplot smoothing.
Identified blood-based markers were differentially associated with brain structures between case and control
To examine whether alterations in blood-based biomarkers correlate with changes in brain structure, we investigated differential associations between these biomarkers and brain structures using a case-control design. Our analysis identified significant differential associations between blood-based markers and brain structures in individuals with psychiatric disorders compared to controls (corrected P < 0.01; Fig. 8). Additional associations between blood-based markers and brain structures within each psychiatric group are detailed in Supplementary Figs. 7–10.
a Differential associations between the identified blood-based biomarkers and brain volume in a case-control study. b Differential associations between the identified blood-based biomarkers and the microstructural properties of white matter in a case-control study. Neuroimaging metrics for both hemispheres were averaged prior to conducting analyses. The number of participants included in the analysis for each disorder— anxiety, bipolar disorder, depression, and schizophrenia—was 7176, 315, 9633, and 282, respectively. *, p_bfi <0.05; **, p_bfi <0.01; ***, p_bfi <0.005. AR acoustic radiation, ATR anterior thalamic radiation, CGPOC cingulate gyrus part of cingulum, CT corticospinal tract, FA fractional anisotropy, FMA forceps major, FMI forceps minor, ICVF intra-cellular volume fraction, IFF inferior front-occipital fasciculus, ILF inferior longitudinal fasciculus, MD mean diffusivity, ML medial lemniscus, MC middle cerebellar peduncle, PHPC parahippocampal part of cingulum, PTR posterior thalamic radiation, SLF superior longitudinal fasciculus, STR superior thalamic radiation, UF uncinate fasciculus. AX anxiety, BIP bipolar disorder, DEP depression, SCH Schizophrenia.
In anxiety disorder versus controls, four blood markers (CRP, IRF, NEUT-C, RBC) and two blood markers (AST, MCV) demonstrated differential correlations with brain volume and DTI measures, respectively. Notably, the volume of medial orbitofrontal cortex and middle temporal regions, implicated in anxiety disorders50,51, demonstrated differential correlations with CRP and RBC between anxiety disorders and controls, respectively.
In bipolar disorder versus controls, three blood markers (cystatin C, LYMP-P, RBC) showed differential correlations with brain volume measures, whereas another set of three markers (cystatin C, Hct, RBC) demonstrated differential correlations with DTI measures. Both cystatin C and RBC were found to exhibit significant differential correlations with brain volume and DTI measures. The brain regions with differential correlations to cystatin C were the middle temporal and lateral orbitofrontal cortices, while the region related to RBC was the entorhinal cortex.
In depression versus controls, five blood markers (HbA1c, HDL-TG, IRF, degree of unsaturation, XL-HDL-TG) and one blood marker (MCH) demonstrated differential correlations with brain regional volumes and DTI measures, respectively. Notably, the volume of the accumbens, a core brain region in the reward system52, demonstrated differential correlations with HDL-TG, degree of unsaturation, and XL-HDL-TG between cases and controls.
In schizophrenia versus controls, one blood marker (vitamin D) and two blood markers (MCHC, RDW) demonstrated differential correlations with brain regional volumes and DTI measures, respectively. Vitamin D demonstrated differential correlations with the volumes of the hippocampus and insula between cases and controls.
Discussion
In this study, we investigated the temporal trends of psychiatric assessments and blood-based biomarkers over a decade leading up to and following the diagnosis of psychiatric disorders, in comparison with matched controls. Our results indicate that 39, 6, 55, and 12 blood-based markers exhibited significant divergence in anxiety, bipolar disorder, depression, and schizophrenia, respectively. Common markers were identified, such as cystatin C, RBC, Hb, and Hct, which exhibited similar temporal patterns. These biomarkers clustered into groups characterized by either linear or non-linear temporal trajectories. Among the linearly changing biomarkers, some showed an increasing difference from controls over time, such as triglycerides and related markers in depression, while others, like LYMP-P in bipolar disorder, demonstrated a decreasing difference from controls over time. Notably, clusters demonstrating non-linear trends frequently displayed reversals around the time of diagnosis, indicating potential post-treatment improvements. This is especially evident in schizophrenia, as exemplified by the temporal trends of biomarkers including GGT, CRP, RDW, and MCHC. Additionally, our analysis revealed differential associations between these biomarkers and brain structures, specifically highlighting regions such as the orbitofrontal cortex, hippocampus, and accumbens, which are implicated in the pathophysiology of these disorders. These findings suggest that blood-based biomarkers could potentially aid in the early detection of various psychiatric disorders and inform the design of therapeutic clinical trials aimed at mitigating their impact on brain structure.
Neuroticism has been suggested to serve as an efficient marker of nonspecific general risk for psychiatric disorders53. We observed an upward trend of psychiatric assessments in four different psychiatric disorders prior to diagnosis, indicating a worsening of symptoms as the disease progressed. For cases with anxiety and depression disorders, the neuroticism score continued to increase. However, following diagnosis, the PHQ-4 displayed a downward trend across four psychiatric disorders. In line with temporal trends of psychiatric assessments, we observed changes in several blood-based markers that occurred up to a decade before diagnosis, presenting an inverted or positive U-shaped trend. This finding raises the possibility that these changes alleviate after treatment, or changes of these markers may be compensatory responses to developing illness54,55,56.
Among the psychiatric disorders investigated, cystatin C57,58, RBC, Hb, Hct, and total bilirubin emerged as shared biomarkers. The concurrent decreases in RBC, Hb, and Hct strongly suggest anemia-associated pathophysiology, which may impair cerebral oxygen delivery and exacerbate neural vulnerability59. Although Cystatin C is commonly recognized as a marker of renal function, previous studies have shown that oxidative stress can upregulate its concentration60,61. Excessive reactive oxygen species generation may accelerate bilirubin catabolism, thereby reducing total bilirubin levels—a phenomenon potentially reflecting systemic antioxidant depletion in psychiatric conditions. Our findings revealed that in anxiety and bipolar disorders, RBC and cystatin C are differentially associated with the volume of the middle temporal region when comparing cases to controls. This region plays a critical role in the dynamic processing of emotion regulation62. These overlapping alterations in biomarkers emphasize the potential clinical significance of lifestyle modifications (such as diets rich in antioxidants) or pharmacological interventions for restoring redox homeostasis in psychiatric disorders.
Some of our findings from the temporal trends analysis of blood-based markers in individuals with psychiatric disorders are in line with previous studies. For instance, inflammation and inadequate vitamin D levels have been associated with an elevated risk of various psychiatric disorders26,63,64. We found that, during the observational period, the levels of CRP and NEUT-P were consistently higher, while the level of LYMP-P was lower, in cases of anxiety and depression as compared to the control group (Figs. 4 and 6). The vitamin D levels in the blood of participants with schizophrenia and depression are consistently lower than those of the control group. Previous studies have also revealed that vitamin D supplementation can enhance the total attention span and positive and negative symptoms in schizophrenia65. Hence, individuals at a high risk of schizophrenia and depression should be encouraged to be exposed to sunlight to maintain optimal vitamin D levels. Future research is requisite to further ascertain whether managing these blood markers in individuals at high risk of psychiatric disorders can mitigate the incidence of the disease66.
In the analysis of the differential associations between blood-based markers and brain structures in individuals with psychiatric disorders compared to controls, we identified both higher-order cortical regions involved in emotional processing, namely the medial orbitofrontal and lateral orbitofrontal cortices, and key limbic system regions crucial for primitive emotional responses, such as the hippocampus and nucleus accumbens (Fig. 8)62,67,68. Previous studies have consistently reported structural and functional alterations in these two set of brain regions across various psychiatric disorders50,51,67. Notably, we found that some blood-based biomarkers exhibit reversible changes, particularly in SCZ. This contrasts with neuronal damage, which is often irreversible69,70. Early monitoring and management of these biomarkers may help prevent further disease progression. Our findings underscore the multisystemic nature of psychiatric conditions, suggesting that fluctuations in blood biomarkers can impact the neural circuits implicated in these disorders, ultimately contributing to the manifestation of psychiatric symptoms71,72,73.
This study presents several strengths, including the utilization of a community-based cohort with a substantial sample size, a long follow-up period, and the comprehensive evaluation of blood-based markers. However, the results should be interpreted within the context of several limitations. First, as we used a cross-sectional dataset to explore temporal trends of blood-based markers, the ability to detect temporal trends may be limited, especially with fewer observations at certain time ranges. Despite making adjustments for age in our blood marker analyses and matching controls to cases according to age, an age difference persists among individuals with different disease durations (Supplementary Fig. 11). This age discrepancy could influence the temporal trends of these biomarkers. Future studies with longitudinal blood samples are crucial to validate the temporal trends of these markers. Second, the timing of psychiatric disorder diagnoses may not always align with the onset of symptoms74,75,76, potentially affecting the accuracy of blood-based marker trends, especially the turning points of U-shaped trends. Third, the UKB participants were predominantly white, and the psychiatric assessments were relatively simple. Future prospective studies with more diverse populations and comprehensive psychiatric assessments are required to further validate their temporal trends. Fourth, daily biological fluctuations in blood-based markers may limit the robustness of temporal trends analysis. Future studies should incorporate biological fluctuations of blood-based markers with repeated measurements of data. Fifth, it’s worth noting that the observed changes in blood-based markers may be influenced by various confounding factors, such as lifestyle habits, comorbidities, and medication use. Controlling for these factors in future studies would help to clarify the specific role of psychiatric disorders in altering blood-based markers. Finally, a few blood-based markers showed significant differences for bipolar disorder and schizophrenia compared with controls. This may be due to the small number of patients rather than indicating a minor impact of these disorders on the markers.
In conclusion, our study provides insights into the complex temporal trends of blood-based markers in individuals with psychiatric disorders and their differential association with brain structure compared to controls. These findings underscore the intricate relationship between peripheral blood and central nervous system function, revealing critical insights into the neurobiological underpinnings of psychiatric disorders. Consistent monitoring and proactive management of these biomarkers could potentially facilitate early intervention and prevent further deterioration.
Data availability
All data utilized in this study were accessed from the UKB under application number 19542. The data employed in the present study are available from UKB with restrictions imposed. Data was used under license and is, therefore, not publicly accessible. Access to the UKB data can be requested via a standard protocol (https://www.ukbiobank.ac.uk/register-apply/). The source data for Fig. 2 and Figs. 4 through 8 are available in the Supplementary Data 7. All other relevant data that support the findings of the study are available from the corresponding author upon reasonable request.
Code availability
All the scripts of the analysis are made available on GitHub (https://github.com/SnowSunshine-gqp/blood-based-biomarkers_temporal-trends) and a DOI-minting repository of the code is provided on Zenodo77.
References
Institute of Health Metrics and Evaluation. Global Health Data Exchange (GHDx). https://vizhub.healthdata.org/gbd-results/, (accessed 20 October 2023).
Kulak-Bejda, A., Bejda, G. & Waszkiewicz, N. Mental disorders, cognitive impairment and the risk of suicide in older adults. Front. Psychiatry 12, 695286 (2021).
Jarzebski, M. P. et al. Ageing and population shrinking: implications for sustainability in the urban century. npj Urban Sustain. 1, 17 (2021).
Partridge, L., Deelen, J. & Slagboom, P. E. Facing up to the global challenges of ageing. Nature 561, 45–56 (2018).
Mane-Damas, M. et al. Autoimmunity in psychotic disorders. Where we stand, challenges and opportunities. Autoimmun. Rev. 18, 102348 (2019).
Sheppes, G., Suri, G. & Gross, J. J. Emotion regulation and psychopathology. Annu. Rev. Clin. Psychol. 11, 379–405 (2015).
Hirjak, D. et al. Motor dysfunction as research domain across bipolar, obsessive-compulsive and neurodevelopmental disorders. Neurosci. Biobehav. Rev. 95, 315–335 (2018).
Fett, A. J. et al. Long-term changes in cognitive functioning in individuals with psychotic disorders: findings from the suffolk county mental health project. JAMA Psychiatry 77, 387–396 (2020).
Karshikoff, B., Lekander, M. & Lasselin, J. Editorial: vulnerability and protective factors for inflammation-associated somatoform and mental disorders. Brain Behav. Immun. 106, 227–230 (2022).
Chourpiliadis, C. et al. Metabolic profile and long-term risk of depression, anxiety, and stress-related disorders. JAMA Netw. Open. 7, e244525 (2024).
Davyson, E. et al. Metabolomic investigation of major depressive disorder identifies a potentially causal association with polyunsaturated fatty acids. Biol. Psychiatry 94, 630–639 (2023).
Saiz-Vazquez, O. et al. Depression as a risk factor for alzheimer’s disease: a systematic review of longitudinal meta-analyses. J. Clin. Med. 10, 1089 (2021).
Nielsen, R. E., Banner, J. & Jensen, S. E. Cardiovascular disease in patients with severe mental illness. Nat. Rev. Cardiol. 18, 136–145 (2021).
Kwapong, Y. A. et al. Association of depression and poor mental health with cardiovascular disease and suboptimal cardiovascular health among young adults in the United States. J. Am. Heart Assoc. 12, e028332 (2023).
Plana-Ripoll, O. et al. A comprehensive analysis of mortality-related health metrics associated with mental disorders: a nationwide, register-based cohort study. Lancet 394, 1827–1835 (2019).
Wykes, T. et al. Mental health research priorities for Europe. Lancet Psychiatry 2, 1036–1042 (2015).
Zhang, Z. et al. Depressive symptoms and mortality among US adults. JAMA Netw. Open 6, e2337011 (2023).
Murphy, B. P. & Brewer, W. J. Early intervention in psychosis: strengths and limitations of services. Adv. Psychiatr. Treatment 17, 401–407 (2011).
Amin, N. et al. Interplay of metabolome and gut microbiome in individuals with major depressive disorder vs control individuals. JAMA Psychiatry 80, 597–609 (2023).
Nedic Erjavec, G. et al. Short overview on metabolomic approach and redox changes in psychiatric disorders. Redox Biol. 14, 178–186 (2018).
Bot, M. et al. Metabolomics profile in depression: a pooled analysis of 230 metabolic markers in 5283 cases with depression and 10,145 controls. Biol. Psychiatry 87, 409–418 (2020).
Tomasik, J. et al. Metabolomic biomarker signatures for bipolar and unipolar depression. JAMA Psychiatry 81, 101–106 (2023).
Xiao, G. et al. Causality of genetically determined metabolites on anxiety disorders: a two-sample Mendelian randomization study. J. Transl. Med. 20, 475 (2022).
Marijnissen, R. M. et al. Metabolic dysregulation and late-life depression: a prospective study. Psychol. Med. 47, 1041–1052 (2017).
Wittenberg, G. M. et al. Major depressive disorder is associated with differential expression of innate immune and neutrophil-related gene networks in peripheral blood: a quantitative review of whole-genome transcriptional data from case-control studies. Biol. Psychiatry 88, 625–637 (2020).
Milaneschi, Y. et al. Association of inflammation with depression and anxiety: evidence for symptom-specificity and potential causality from UK Biobank and NESDA cohorts. Mol. Psychiatry 26, 7393–7402 (2021).
Haapakoski, R. et al. Cumulative meta-analysis of interleukins 6 and 1beta, tumour necrosis factor alpha and C-reactive protein in patients with major depressive disorder. Brain Behav. Immun. 49, 206–215 (2015).
Lengvenyte, A. et al. Associations of white blood cell and platelet counts with specific depressive symptom dimensions in patients with bipolar disorder: analysis of data from the FACE-BD cohort. Brain Behav. Immun. 108, 176–187 (2023).
Sudlow, C. et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Patient Health Questionnaire-4, Kroenke, K., Spitzer, R. L., Williams, J. B. W., & Löwe, B. (2009). Patient Health Questionnaire-4 (PHQ-4) [Database record]. APA PsycTests. https://doi.org/10.1037/t06168-000.
The Corsini Encyclopedia of Psychology Full Access. Eysenck Personality Questionnaire. Nilly Mor. First published: 30 January 2010. https://doi.org/10.1002/9780470479216.
Ritchie, S. C. et al. Quality control and removal of technical variation of NMR metabolic biomarker data in ~120,000 UK Biobank participants. Sci. Data 10, 64 (2023).
Soininen, P. et al. Quantitative serum nuclear magnetic resonance metabolomics in cardiovascular epidemiology and genetics. Circul. Cardiovasc. Genet. 8, 192–206 (2015).
Julkunen, H. et al. Atlas of plasma NMR biomarkers for health and disease in 118,461 individuals from the UK Biobank. Nat. Commun. 14, 604 (2023).
Jenkinson, M. et al. FSL. Neuroimage 62, 782–790 (2012).
Jesper L. R. & Andersson, M. J. A. S. S. Non-linear optimisation. Internal Technical Report TR07JA1, Oxford Centre for Functional Magnetic Resonance Imaging of the Brain, Department of Clinical Neurology, Oxford University, Oxford, UK FMRIB Technial Report TR07JA1 (2007).
Jesper L. R., Andersson, M. J. A. S. S., Non-linear registration aka Spatial normalisation. Internal Technical Report TR07JA2, Oxford Centre for Functional Magnetic Resonance Imaging of the Brain, Department of Clinical Neurology, Oxford University, Oxford, UK: FMRIB Technial Report TR07JA1 (2007).
Zhang, Y., Brady, M. & Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 20, 45–57 (2001).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980 (2006).
Fischl, B. et al. Automatically parcellating the human cerebral cortex. Cereb. Cortex 14, 11–22 (2004).
Fischl, B. et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33, 341–355 (2002).
Klapwijk, E. T. et al. Qoala-T: a supervised-learning tool for quality control of FreeSurfer segmented MRI data. Neuroimage 189, 116–129 (2019).
Zhang, H. et al. NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. Neuroimage 61, 1000–1016 (2012).
de Groot, M. et al. Improving alignment in tract-based spatial statistics: evaluation and optimization of image registration. Neuroimage 76, 400–411 (2013).
You, J. et al. Clinical trajectories preceding incident dementia up to 15 years before diagnosis: a large prospective cohort study. Mol. Psychiatry 29, 3097–3105 (2024).
Sedgwick, P. Nested case-control studies. BMJ 340, c2582 (2010).
Zhang, Z. Propensity score method: a non-parametric technique to reduce model dependence. Ann. Transl. Med. 5, 7 (2017).
Leppink, J. Group-by-Time Interactions. In: Proc. Statistical Methods for Experimental Research in Education and Psychology. Springer Texts in Education (Springer, 2019).
Jacoby, W. G. Loess: a. nonparametric, graphical tool for depicting relationships between variables. Elect. Stud. 19, 577–613 (2000).
Liu, N. et al. The difference in volumetric alternations of the orbitofrontal-limbic-striatal system between major depressive disorder and anxiety disorders: a systematic review and voxel-based meta-analysis. J. Affect. Disord. 350, 65–77 (2024).
Mizzi, S. et al. Resting-state neuroimaging in social anxiety disorder: a systematic review. Mol. Psychiatry 27, 164–179 (2022).
Chen, G. et al. Distinct reward processing by subregions of the nucleus accumbens. Cell Rep. 42, 112069 (2023).
Ormel, J. et al. Neuroticism and common mental disorders: meaning and utility of a complex relationship. Clin. Psychol. Rev. 33, 686–697 (2013).
Livingston, L. A., Shah, P. & Happe, F. Compensatory strategies below the behavioural surface in autism: a qualitative study. Lancet Psychiatry 6, 766–777 (2019).
Allott, K. et al. Compensatory interventions for cognitive impairments in psychosis: a systematic review and meta-analysis. Schizophr. Bull. 46, 869–883 (2020).
Livingston, L. A. & Happe, F. Conceptualising compensation in neurodevelopmental disorders: Reflections from autism spectrum disorder. Neurosci. Biobehav. Rev. 80, 729–742 (2017).
Sun, T. et al. Associations of serum cystatin C with depressive symptoms and suicidal ideation in major depressive disorder. BMC Psychiatry 21, 576 (2021).
Li, H. et al. Cystatin C and risk of new-onset depressive symptoms among individuals with a normal creatinine-based estimated glomerular filtration rate: a prospective cohort study. Psychiatry Res. 273, 75–81 (2019).
Wang, X., Cui, L. & Ji, X. Cognitive impairment caused by hypoxia: from clinical evidences to molecular mechanisms. Metab. Brain Dis. 37, 51–66 (2022)
Nishio, C. et al. Involvement of cystatin C in oxidative stress-induced apoptosis of cultured rat CNS neurons. Brain Res. 873, 252–262 (2000).
Watanabe, S. et al. Cystatin C protects neuronal cells against mutant copper-zinc superoxide dismutase-mediated toxicity. Cell Death Dis. 5, e1497 (2014).
Wang, J. et al. The critical mediating roles of the middle temporal gyrus and ventrolateral prefrontal cortex in the dynamic processing of interpersonal emotion regulation. NeuroImage 300, 120789 (2024).
Guzek, D. et al. Association between vitamin D supplementation and mental health in healthy adults: a systematic review. J. Clin. Med. 10, 5153 (2021).
Ronaldson, A. et al. Prospective associations between vitamin D and depression in middle-aged adults: findings from the UK Biobank cohort. Psychol. Med. 52, 1866–1874 (2022).
Neriman, A., Hakan, Y. & Ozge, U. The psychotropic effect of vitamin D supplementation on schizophrenia symptoms. BMC Psychiatry 21, 309 (2021).
Drevets, W. C. et al. Immune targets for therapeutic development in depression: towards precision medicine. Nat. Rev. Drug Discov. 21, 224–244 (2022).
Zhang, B. et al. Roles of the medial and lateral orbitofrontal cortex in major depression and its treatment. Mol. Psychiatry 29, 914–928 (2024).
Rolls, E. T. et al. The orbitofrontal cortex: reward, emotion and depression. Brain Commun. 2, fcaa196 (2020).
Chen, D. et al. Neurophysiological stratification of major depressive disorder by distinct trajectories. Nat. Mental Health 1, 863–875 (2023).
Jiang, Y. et al. Neuroimaging biomarkers define neurophysiological subtypes with distinct trajectories in schizophrenia. Nat. Mental Health 1, 186–199 (2023).
Nesse, R. M. Evolutionary psychiatry: foundations, progress and challenges. World Psychiatry 22, 177–202 (2023).
Tian, Y. E. et al. Heterogeneous aging across multiple organ systems and prediction of chronic disease and mortality. Nat. Med. 29, 1221–1231 (2023).
Werner, M. C. F. et al. Immune marker levels in severe mental disorders: associations with polygenic risk scores of related mental phenotypes and psoriasis. Transl. Psychiatry 12, 38 (2022).
Wang, P. S. et al. Delays in initial treatment contact after first onset of a mental disorder. Health Serv. Res. 39, 393–415 (2004).
McLaughlin, C. G. Delays in treatment for mental disorders and health insurance coverage. Health Serv. Res. 39, 221–224 (2004).
Christiana, J. M. et al. Duration between onset and time of obtaining initial treatment among people with anxiety and mood disorders: an international survey of members of mental health patient advocate groups. Psychol. Med. 30, 693–703 (2000).
Pan, G. The analysis code for the article titled “temporal trends of blood-based markers in various psychiatric disorders and their cross-sectional brain structure associations”. Zenodo. https://doi.org/10.5281/zenodo.15532293 (2025).
Acknowledgements
We want to thank all the participants and researchers from the UKB. This study was funded by grants from the National Natural Science Foundation of China (82302312), the Natural Science Foundation of Shanghai (23ZR1406000) and 111 Project (B18015). Further, we would like to thank the support from the Shanghai Center for Brain Science and Brain-Inspired Technology, ZHANGJIANG LAB, and the State Key Laboratory of Neurobiology and Frontiers Center for Brain Science of Ministry of Education. The language of this paper has been polished by ERNIE Bot (v3.5) to enhance its readability and overall linguistic quality. The authors are responsible and accountable for the originality, accuracy, and integrity of the work. The funder had no role in study design, data collection, data analysis, data interpretation, writing of the report, or decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
L.W., J.Y., G.P. and W.C. conceived, designed, and supervised the project. Y.W., Z.Z., G.P. and J.Y. performed statistical analyses. Y.W., Z.Z. and G.P. drafted the manuscript. Y.L., J.K. and Y.W. accessed and verified the underlying data reported in the manuscript. J.Y., J.F. and W.C. contributed to the critical revision of the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Liliana Ciobanu, Rongtao Jiang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. [ Peer reviewer reports are available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, YJ., Zhou, Z., Li, YZ. et al. Temporal trends of blood-based markers in various psychiatric disorders and their cross-sectional brain structure associations. Commun Med 5, 239 (2025). https://doi.org/10.1038/s43856-025-00957-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-00957-w