Abstract
Background
Complete blood count (CBC) testing is critical for diagnosing and monitoring hematological conditions, including leukemia. However, conventional analyzers require specialized infrastructure and trained personnel, limiting access in resource-constrained settings. Point-of-care hematology devices provide a promising alternative by delivering rapid results with minimal complexity. This study aimed to evaluate the analytical performance of the Hilab Lens versus XE-2100.
Methods
Blood samples (n = 555) were analyzed using both devices at Erastinho Hospital, a pediatric oncology center. Device performance was assessed through precision testing, method comparison, sample type equivalence, evaluation of potential interferences, and caregiver satisfaction surveys. Agreement and correlation were evaluated using Bland–Altman plots and Passing–Bablok regression.
Results
Here we show that the Hilab Lens demonstrates high precision and consistency, meeting European Federation of Clinical Chemistry and Laboratory Medicine (EFLM). Method comparison revealed strong correlations (key analytes r ≥ 0.95), minimal bias, and robust agreement with the reference analyzer across all CBC parameters. The Hilab Lens achieved high accuracy in flagging studies (≥ 98.8%), particularly for band neutrophils and immature cells. No significant interferences were detected (p > 0.5). Caregivers reported high satisfaction with the test (mean score 4.65 on a 5-point scale).
Conclusions
These results support the Hilab Lens as a reliable and accessible alternative to conventional hematology analyzers. Its ease of use and rapid turnaround make it suitable for environments where timely CBC testing is essential. The high acceptance observed in pediatric oncology suggests its potential for broader implementation in clinical practice.
Plain language summary
Blood tests are crucial for diagnosing and monitoring several diseases, but traditional laboratory machines require specialized facilities and trained staff, making them difficult to access in many regions. In this study, we tested a small and portable device called the Hilab Lens. It was used with children receiving cancer treatment, and the results were compared with those from a standard hospital machine.
As a result, the devices provided comparable results, with Hilab Lens being faster and cheaper. These findings suggest that this POC device could help bring essential blood testing to more people, especially in areas with limited medical resources, improving access to timely diagnosis and treatment.
Similar content being viewed by others
Introduction
Monitoring treatment responses in leukemia relies on complete blood counts (CBCs), which provide critical insights into hematologic response.1 This test is also essential for diagnosing anemia, various types of cancer, infections, acute bleeding, allergies, and immune deficiencies, making it an invaluable tool in clinical practice, particularly in pediatrics. CBC offers a comprehensive evaluation of the production and immune function of the white blood cells (WBC). Beyond total WBC counts, the differential analysis identifies their subtypes, such as neutrophils (NEU), monocytes (MON), lymphocytes (LYM), eosinophils (EOS), basophils (BAS), and immature cells. These subpopulations provide critical diagnostic and prognostic information, such as neutrophilia in bacterial infections or lymphocytosis in viral infections and hematological malignancies. Additionally, the test assesses red blood cell (RBC) counts, hemoglobin (HB) levels, and hematocrit (HT) values to evaluate oxygen-carrying capacity. Red blood cell indices—including mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and red cell distribution width (RDW)—offer insights into cell morphology and hemoglobin content, aiding in the differentiation of anemia types. Platelet counts (PLA) are also included in the test, allowing hemostasis, thrombosis, and wound healing evaluation.2
Global cancer statistics from 2022 indicate ~20 million new cases and around 10 million cancer-related deaths.3 Among these, hematological malignancies represent a significant and growing burden across all age groups. According to the World Health Organization, approximately 400,000 children and adolescents aged 0–19 years are diagnosed with cancer annually. In the United States, cancer is the leading cause of death by disease after infancy among children. There is an estimation that, in 2025, a total of 14,910 children and adolescents ages 0 to 19 will be diagnosed with cancer, and 1,590 will die of the disease. While >80% of pediatric cancer patients in high-income countries achieve remission due to early diagnosis and comprehensive care, this rate drops below 30% in low- and middle-income countries. Barriers such as limited diagnostic access, misdiagnosis or delayed diagnosis, treatment abandonment, toxicity-related mortality, and relapse contribute to these disparities.4 Given these challenges, timely and accurate hematologic testing is crucial for early detection, treatment monitoring, and improving survival rates in pediatric oncology.5,6,7
Hematology analyzers integrate multiple technologies, including spectrophotometry, impedance, conductivity measurement, and flow cytometry, to ensure precise cell counting and hematimetric indices calculation. Despite their high accuracy, these analyzers face challenges when processing abnormal blood samples, often requiring manual microscopic review to resolve flagged results.8 Additionally, their reliance on complex infrastructure, specialized maintenance, and trained personnel restricts their availability to large medical centers. This centralization creates significant barriers to early diagnosis and continuous monitoring of hematological disorders, particularly in resource-limited settings, contributing to global disparities in morbidity and mortality.
Point-of-care (POC) testing has emerged as a promising alternative to address these limitations. Typically obtained via a fingerstick, capillary sampling is less invasive and easier to perform. POC devices also deliver rapid results, enabling timely clinical decisions without requiring centralized laboratory facilities. This decentralization is particularly beneficial in regions where traditional laboratory services are scarce or inaccessible. Furthermore, POC testing facilitates frequent monitoring, which is critical for pediatric oncology patients undergoing intensive treatment protocols. By reducing dependence on complex infrastructure and specialized personnel, these diagnostic tools expand healthcare access and improve patient outcomes.9
Given the critical role of CBC testing in evaluating hematologic response and diagnosing hematological malignancies, it is essential that hematology analyzers produce comparable results and can be used interchangeably across laboratories. To ensure analytical reliability, the Clinical and Laboratory Standards Institute (CLSI) mandates that validation must first be conducted by the manufacturer under optimal conditions, followed by verification within clinical laboratories to confirm performance under real-world conditions.10,11
In this context, this study aims to perform a comprehensive analytical assessment comparing the Hilab Lens (Hi Technologies, Brazil), a POC hematology device, with the Sysmex XE-2100 (Sysmex Corporation, Japan), a widely used reference analyzer in clinical laboratories. The study evaluated key performance parameters such as precision, method comparison, sample type equivalence, potential interfering factors, and overall user experience at Erastinho Hospital (Curitiba, Parana, Brazil), a specialized pediatric oncology center dedicated to the diagnosis, treatment, and multidisciplinary care of children and adolescents with cancer.
Methods
The Hilab Lens
Figure 1, Supplementary Video 1 and Supplementary note 1 provide a comprehensive overview of the Hilab Lens device, diagnostic test, and its operational workflow for CBC analysis in a POC setting.
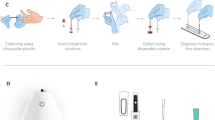
A The Hilab Lens design. B Key components of the device including NFC reader, sensor, sample illumination assembly, magnets for analysis position registration, and an optical assembly equipped with an objective and image sensor. Accessories included with the device: tube and capsule support, calibration capsule, and a fixed-volume pipette (10 µL). C The diagnostic kit components: an analysis capsule, reagent solutions, and NFC card that stores essential data such as calibration curves, expiration date, and traceability information. D Analysis capsule overview: analysis of RBC and PLA (1), WBC (2), and HB (3). E The step-by-step testing workflow, including patient registration (Specialist Portal), assembly of the analysis capsule, blood collection and dilution, homogenization, dispensing, reading (with AI), and result generation (Specialist Portal). F AI functionalities of the Hilab Lens, covering: calibration step; image processing and cell detection of RBC/PLA series (1st chamber); Fine-focus adjustments used for WBC series, enabling differential WBC analysis (2nd chamber). G Specialist Portal: Cloud-Based system utilized by the Hilab Lens.
Hardware
The Hilab Lens (National Health Surveillance Agency (ANVISA) registration no. 80583710047) is a compact, battery-operated, POC hematology analyzer designed for rapid blood analysis through a dual-stage optical and spectrophotometric reading system (Fig. 1A). The analytical process begins with a self-calibration step that requires a dedicated calibration capsule with an incorporated Fuchs-Rosenthal grid for microscopy (Fig. 1B). The calibration procedure is fully automated, ensuring consistent performance, and is required every 24 h. During sample analysis, the device performs the reading in two sequential stages. At the first analytical step, it performs WBC classification and quantification, capturing high-resolution microscopic images from the analysis capsule (2nd chamber) containing the diluted blood sample. The second analysis stage simultaneously evaluates RBC, PLA, and HB using two distinct methodologies. Through optical microscopy (1st chamber), the device performs the RBC and PLA counts, similar to the WBC analysis, ensuring morphological assessment and numerical quantification. Alongside the microscopy, the device employs an 11-channel spectrophotometer and a controlled LED array to perform absorbance-based quantification of HB (3rd chamber).
Diagnostic kit and test operation
The diagnostic kit comprises a reading card, an analysis capsule, and three reagent solutions (Fig. 1C; ANVISA registration no. 80583710046). Supplementary materials include a fixed-volume pipette, disposable tips, tubes, and capsule support. To carry out the test, first, the test operator positions the analysis capsule on the capsule support (Fig. 1D) and places the tubes in the support with their caps open. Then, the operator aspirates blood samples (collected via venous or capillary methods) using a single-volume (10 μl) pipette. The blood samples should be individually dispensed into tubes (1, 2, and 3; 10 μl/each). After this, the operator homogenizes the blood with the reagent solutions by inverting the tubes and returning them to their upright position for 30 s. Finally, 10 μl of solutions from tubes 1, 2, and 3 are transferred into chambers 1, 2, and 3 of the analysis capsule, respectively. Next, the operator swipes the reading card across the upper back portion of the Hilab Lens reader until the LED changes from green to pulsating blue. Finally, the test operator inserts the analysis capsule into the device to the first stage (marked “1”; 2nd chamber reading). After receiving a command from the Specialist Portal (~5 min), the operator pushes to the second stage (marked “2”; 1st and 3rd chambers reading). The device takes ~10 min to read the analysis capsule, and the complete process takes ~25 min (Fig. 1E).
A.I
A series of custom-built AI algorithms compose the Hilab AI system, including Convolutional Neural Networks (CNNs), Transformers, and Multi-Layer Perceptrons (MLPs). This tailored approach allows the system to address domain-specific challenges effectively while maintaining robust performance across various use cases. Hematologists’ insights and annotations are used to construct the dataset for AI training, ensuring that models are grounded in expert knowledge and clinical accuracy. Regarding cell detection, to achieve optimal performance, the Hilab AI models leverage state-of-the-art (SotA) methodologies for object detection, classification, and anomaly detection. The AI models were designed for detecting and classifying blood cells (Fig. 1F), with separate models developed for RBC/PLA (RBC + PLA Series) and WBC (WBC Series). Since a single image can contain thousands of small cells, the Hilab Lens divides each image into a grid and processes each sub-image using the YOLO (you only look once) deep learning approach.12 Finally, the Hilab Lens validates the results generated by the AI models by comparing them to the gold-standard outcomes.
Cloud System: Specialist Portal
The Hilab Specialist Portal (Fig. 1E), accessible from multiple devices, is a cloud-based interactive platform designed to facilitate the patient registration, operation, and result validation of the Hilab Lens device. Since the Hilab Lens does not have an integrated display, this platform is the primary interface between the test operator and the device. The platform maintains the entire traceability of all processes, including test batch information, device identification, operator credentials, and patient exam history, ensuring regulatory compliance and data integrity. The Specialist Portal also offers a range of essential features, including device calibration verification, patient registration, access to test instructions (IFU), and a historical database of previously validated reports. It allows operators to review the calibration history of the device, access training materials, and contact the technical support team for real-time assistance. Also, a 24/7 support service is available to address operational concerns, ensure continuous system functionality, and guarantee timely report delivery.
Test principle
The Lens CBC test employs optical microscopy and spectrophotometry methodologies for blood analysis. Reagent solutions 1 and 2 have distinct affinities for cellular structures and enable blood cell identification. HB quantification, on the other hand, is achieved using reagent solution 3, composed of salts that react with RBCs, promoting their lysis and subsequent conversion of HB to methemoglobin. The Hilab Lens utilizes AI to digitize the sample, count blood cells, quantify HB, and infer the concentration of red cell indices (HT, MCV, and MCH) based on previous studies.13 The AI also evaluates the sample regarding the flag’s presence. The Hilab Lens sends the data to the device’s software (Specialist Portal), where the test operator may assess and approve it. Finally, the test release results for 20 parameters of the CBC test: Quantitative analysis of RBC, HB, HT, MCV, MCH, WBC, NEU#, NEU%, LYM#, LYM%, MON#, MON%, EOS/BAS#, EOS/BAS% (quantified together); and qualitative analysis of RDW (Anisocytosis), mean platelet volume (MPV), band neutrophils, immature cells, and poikilocytosis. Hi technologies® patented the device, the cloud system, and all reagent solutions. According to the clinical practice adopted by the test manufacturer, the Lens CBC test includes both the total and differential WBC count.
Study design
This prospective and cross-sectional study was conducted in accordance with the ethical principles of the Declaration of Helsinki. The study protocol was approved by the Research Ethics Committee of the Paranaense League Against Cancer (CAAE number 49961421.3.1001.0098). A team of researchers from the Hilab company performed the study invitation at Erastinho Hospital (Curitiba, Parana, Brazil), a specialized pediatric oncology center dedicated to the diagnosis, treatment, and comprehensive care of children and adolescents with cancer. Study invitations were extended to patients and their legal guardians, offering participation via venous blood donation or combined venous and capillary collection.
A total of 674 pediatric patients were screened for eligibility. After exclusions due to refusal or unmet criteria, 562 participants were included and allocated by convenience into three study segments: precision study, method comparison (with interference and flagging analysis), and venous vs. capillary comparison. A detailed participant flow diagram is presented in Fig. 2. The study population included children and adolescents with varying hematological profiles, including those undergoing active cancer treatment or in remission, allowing assessment of the Hilab Lens performance across clinically diverse scenarios. All methods followed applicable ethical and biosafety standards.14 Informed consent was obtained from all participants or their legal guardians. Venous blood samples were analyzed using the standard hematology analyzer (Sysmex XE-2100) by the hospital’s clinical lab team, while Hilab Lens tests were performed independently by Hilab professionals. A third blinded team analyzed and compared the results to ensure unbiased evaluation.
A total of 674 children were invited to participate. Of these, 112 were excluded. The remaining 562 participants were allocated by convenience into different study segments: (1) Precision Study: Samples from 12 patients (n = 3 samples/analyte) were selected based on clinically relevant concentration ranges for RBC, PLA, WBC, and HB; (2) Method Comparison Study: 495 patients were selected for method comparison, being two were excluded. Among the remaining 493 samples, 33 presenting aspects considered potential interfering factors by previous studies were assigned to the interference study. The remaining 460 samples were analyzed in the method comparison study, parallelly with flagging study evaluation; (3) Venous vs. Capillary Comparison: 105 patients met the eligibility criteria for this comparison, but 55 were excluded. The remaining 50 patients underwent capillary blood collection and were included in the venous vs. capillary comparison study, parallelly with the assessment of patient preference study.
Eligibility criteria
Participants eligible for the study were children and adolescents aged 1−18 years of any sex, with active cancer or in remission state, under follow-up at Erastinho Hospital. Additional inclusion criteria required a prior medical prescription for a CBC test on the day of the invitation. The study included patients regardless of pre-analytical factors that could interfere with CBC parameters and whether or not they were fasting at the time of collection.
For participants donating only venous blood, exclusion criteria included: Pre-analytical factors that rendered analysis impossible by the conventional method, such as clotted samples, incorrect collection tubes, or insufficient blood volume; Compromised venous blood collection; and Lack of legal guardian authorization. For participants donating both venous and capillary blood, the study applied the same exclusion criteria, with the addition of: Compromised capillary blood collection; Lack of medical authorization for capillary sampling; Patients referred for hematopoietic stem cell transplantation (HSCT) or hospitalization after CBC results; Insufficient capillary blood volume.
Sample population
The study encompassed samples (n = 555) from patients between 2−18 years old (2–3 y: 11.5%; 4−7 y: 31.6%; 8−11 y: 25.5%; 12−15 y: 19%; 16−18 y: 12.4%), including boys (62.2%) and girls (37.8%). The blood samples enclosed patients with active cancer (73.3%) and in remission stage (26.7%), with (63.4%) or without (36.6%) pathological conditions associated (Cardiovascular diseases (61.4%); Respiratory diseases (16.5%); Mental disorders (11.4%); Autoimmune Diseases (5.7%); Nervous system disorders (4.4%); Diabetes (0.6%)). Within patients with active cancer, the cancer types encompassed leukemia (79.5%; Acute Lymphocytic Leukemia (ALL); Acute Myelogenous Leukemia (AML)), Lymphoma (6.9%; Hodgkin and non-Hodgkin lymphoma), Pilocytic Astrocytoma (3.8%), Sarcoma (1%), and blastomas (8%; Medulloblastoma; Neuroblastoma; Retinoblastoma; Nephroblastoma).
Precision
The accuracy assessment of the point-of-care device followed the CLSI EP15-A3 guidelines. Due to the incompatibility of the commercial hematological controls with the Hilab Lens reagent solutions, twelve (3/analyte) whole blood samples (collected in tubes containing K3EDTA; Vacuette®, Greiner Bio-One, Brazil) were selected to cover clinically relevant concentrations of RBC, PLA, WBC, and HB. Each sample underwent ten consecutive measurements in the Hilab Lens for repeatability assessment. For reproducibility evaluation, the tests were performed again by two different operators. The conventional hematology analyzer determined the sample’s reference values, and the obtained data underwent statistical analysis, evaluating the acceptability criteria established by the European Federation of Clinical Chemistry and Laboratory Medicine (EFLM; Biological variation; Higher CI limit). This study also performed the data evaluation through the Gage R&R method, considering the valuable information provided by this analysis15 as separate variations due to the tool (device) and the operator (test operator). For EP15-A3 analysis, average, standard deviation (SD), CV (coefficient of variation; Intra and Inter), Random Error, Total Error, and Bias were calculated. For Gage R&R analysis, degree of freedom (DF), Sum of squares (SS), Mean squares (MS), F-value (F), P-value (P), Variance components, Study Var (%SV), and SD were calculated and shown.
Method comparison
The method comparison study followed CLIA guideline EP09, evaluating the performance of the Hilab Lens against a conventional hematology analyzer (Sysmex XE-2100; Sysmex Corporation, Japan), widely used in clinical laboratories. The health professionals collected four hundred and sixty venous blood samples in tubes coated with K3EDTA (Vacuette®, Greiner Bio-One, Brazil). Each sample was analyzed using the conventional hematology analyzer (Reference values) and the Hilab Lens. The researchers kept the samples under constant refrigeration (2–8 °C) until analysis, and the maximum time between the Hilab Lens and conventional device analysis was 4 h.
To obtain a visual review of the data and determine the underlying variability characteristics of the relationship between methodologies, a Passing-Bablok regression and a Bland-Altman plot for each CBC analyte were performed. Also, to evaluate the Hilab Lens accuracy, each result was classified as either within or outside the reference range, followed by a confusion matrix construction. The quantitative analysis encompassed the standard Error (SE), p-value of the Student t-test (two-sided), upper limit of agreement (ULoA), bias, lower limit of agreement (LLoA), correlation coefficients (Pearson’s r), intercept, and slope. For qualitative analysis, accuracy, specificity, sensibility, Cohen’s Kappa, Positive Predictive Value (PV +), Negative Predictive Value (PV -), Positive Likelihood Ratio (LR+), and Negative Likelihood Ratio (LR -) were calculated and shown.
Flagging study
After the venous whole blood collection for method comparison study, a blood smear of each sample (n = 460) was prepared following the standard protocol (reference16 May–Grunwald–Giemsa; Laborclin; ANVISA registration no. 10097010105) and evaluated in an optical microscope (NO115T1, Global Trade Technology, São Paulo, Brazil) by a trained hematologist. The qualitative analysis encompassed the presence of all poikilocytosis types (Schistocytes, Stomatocytes, Echinocytes, Acanthocytes, Codocytes, Elliptocytes, Dacrocytes, and Drepanocytes), RBC and PLA clumps, anisocytosis, abnormal quantity of band neutrophils, and immature cells in Hilab Lens and conventional blood smear technique. The study used the morphologic criteria recommended by the International Society for Laboratory Hematology17 to identify abnormal cells. The statistical analysis encompassed accuracy, specificity, sensibility, Cohen’s Kappa, PV + , PV -, LR +, and LR -.
Interference study
The interference study encompassed samples presenting aspects considered potential interfering factors for HB values,18 analyzing thirty-three venous blood samples (K3EDTA; Vacuette®, Greiner Bio-One; Brazil) through the conventional hematology analyzer and the Hilab Lens. The study evaluated the obtained results through SE, Pearson correlation (r), Slope, Intercept, ULoA, bias, LLoA, and p-value of the Student T-test (two-sided) of each potential interference.
Venous vs. capillary comparison
To assess the equivalence between venous and capillary blood samples, paired samples from 50 volunteers were analyzed using the Hilab Lens device. Fresh capillary blood was collected via fingerstick using a 21 G single-use safety lancet (no anticoagulant), following international guidelines for capillary blood collection.19 The procedure was conducted on the lateral surface of the distal phalanx of the middle or ring finger after skin antisepsis with 70% isopropyl alcohol and complete drying. The first drop of blood was discarded to avoid tissue fluid contamination, and subsequent drops were collected without applying excessive pressure to the puncture site, minimizing the risk of hemolysis or dilution with interstitial fluid. All operators were trained in capillary blood collection according to these standards to ensure procedural consistency. Venous blood was drawn into sterile, single-use tubes coated with K₃EDTA anticoagulant (Vacuette®, Greiner Bio-One, Brazil), following the conventional methodology.19 To visually inspect the data and characterize the variability between sample types, Passing-Bablok regression and Bland-Altman plots were generated for each CBC analyte. Statistical evaluation included standard error (SE), Student’s t-test p-value, upper and lower limits of agreement (ULoA and LLoA), bias, Pearson’s correlation coefficient (r), intercept, and slope.
Evaluation of patient preferences
Considering the broad age range and cognitive differences among patients, the legal guardians of 50 children undergoing capillary blood collection (Venous vs. Capillary Comparison study) were invited to complete an anonymous patient satisfaction survey to assess patient preferences. Before the fingerstick collection, legal guardians answered a questionnaire evaluating their acceptance of POC tests, focusing on confidence, motivation, time to result, convenience, transportation time, and costs. To assess perceptions of the Hilab Lens in pediatric care, a similar questionnaire was administered after blood collection, covering aspects of the fingerstick experience and overall test perception (Supplementary Fig. 2). The questionnaire was developed based on previous studies,20 with satisfaction scores rated from 1 to 5 (5 being the highest satisfaction), and responses were stored anonymously. Additionally, the questionnaire included an optional open-ended question for participants to provide additional comments. Statistical analysis included calculating the mean satisfaction score and generating bar charts to display response frequencies.
Statistics and reproducibility
The sample size was calculated according to Chow et al.21 based on the equivalence (method comparison and flagging studies) or non-inferiority (Venous vs. Capillary comparison and Evaluation of patient preferences studies) tests. CLSI (EP15-A3) definitions supported the sample size calculation for precision study. The Kolmogorov–Smirnov normality test was applied to ensure that the data met the criteria for performing the parametric tests. The method comparison study used Passing-Bablok regression due to its robustness against measurement errors, providing an estimation of proportional and systematic differences. Also, the Bland-Altman plots evaluated the agreement and systematic bias between the Hilab Lens and the Sysmex XE-2100. In the precision test, the Gage R&R method was employed to quantify the contribution of variability sources (device, operator, and sample) and to ensure analytical reliability. For all analyses, the significance level was set at p ≤ 0.05, no p-value adjustment for multiple tests was necessary, and data encompassed the 95% confidence interval (95% IC). All tests were double-blinded, analyzed, and plotted using the R software (version 4.2.1) statistics package analysis.
Results
Precision
The Hilab Lens device demonstrated satisfactory analytical precision for all evaluated analytes, in accordance with the acceptance criteria proposed by the EFLM (Biological Variation, higher confidence interval). As shown in Table 1, intra-assay CVs ranged from 2.39% to 2.78% for RBC, 2.08% to 3.10% for HB, 5.04% to 6.48% for PLA, and 6.38% to 10.56% for WBC. Inter-assay CVs were consistently low, with maximum values of 1.65% for RBC, 1.20% for HB, 5.54% for PLA, and 3.30% for WBC. Total error remained within the allowable thresholds for all analytes. Although HB’s intra-assay CV slightly exceeded EFLM (higher CI) specifications (3.1% versus 2.7%), the total error and bias remained within the derived TAE. Additionally, Gage R&R analysis (Supplementary Table 2) confirmed that ≥98% of the total variation was due to part-to-part variability, while the contribution from the measurement system was minimal (≤1.88% across all analytes). These findings reinforce the high precision and analytical robustness of the Hilab Lens for use in POC hematological testing.
Method comparison
The comparison between the Hilab Lens and the conventional hematology analyzer revealed a high level of agreement across all evaluated analytes. Pearson’s correlation coefficients ranged from 0.73 (MCV) to 0.99 (PLA and WBC), with statistically significant associations (p < 0.001), indicating strong linear relationships between methods. Passing-Bablok regression confirmed linearity for most parameters, with slope values close to 1.00 and narrow confidence intervals. The Bland-Altman analysis indicated that a significant percentage of results fell within the total allowable error, reinforcing the clinical applicability of the device. Minimal bias and acceptable ULoA (95% CI) and LLoA (95% CI) were observed for all analytes. Furthermore, no statistically significant differences were found between methodologies for any analyte (p > 0.5; Fig. 3; Supplementary Table 3).
Passing-Bablok (A−K) and Bland–Altman (L−V) plots related to method comparison study between the Hilab Lens and Sysmex XE-2100 for Erythrocytes (A, L), Platelets (B, M), Hemoglobin (C, N), Hematocrit (D, O), MCV (E, P), MCH (F, Q), Total Leukocytes (G, R), Neutrophils (#; H, S), Lymphocytes (#; I, T), Monocytes (#; J, U) and Eosinophils/Basophils (#; K, V). Graphs also present Pearson correlation, Intercept (95% CI), Slope (95% CI), Student T-test p-value (two-sided), standard error (SE), bias, upper limit of agreement (ULoA), and lower limit of agreement (LLoA) for each analyte. Green and red dotted lines indicate expected values for healthy individuals and identity lines, respectively. Blue and black solid lines demonstrate the regression and equity lines, respectively (number of patients = 460).
In the qualitative classification of CBC results (Supplementary Table 1), Hilab Lens demonstrated excellent diagnostic performance. Accuracy exceeded 98.9% for all analytes, with sensitivity and specificity ranging from 98.5% to 99.9%. Cohen’s Kappa coefficients were above 0.95 across the board, indicating almost perfect agreement. Positive predictive values (PV + ) were >97%, while likelihood ratios confirmed high diagnostic strength (LR+ up to 9869.3; LR– as low as 0.003).
Flagging study
The Hilab Lens demonstrated excellent performance in detecting hematological morphological abnormalities compared to conventional optical microscopy. RBC agglutination and PLA aggregation showed perfect agreement, with 100% accuracy. Detection of band neutrophils and immature cells yielded slightly lower sensitivity (94.4% and 94.9%, respectively), while maintaining high accuracy (99.6% and 98.8%). For poikilocytosis and anisocytosis detection also showed strong performance. Positive likelihood ratios (LR + ) were particularly high for anisocytosis, band neutrophils, and immature cells, while negative likelihood ratios (LR–) remained below 0.05 for all alterations, confirming the system’s high diagnostic reliability (Table 2). Also, all samples classified as “normal” by the Hilab Lens were also reviewed by trained hematologists using microscopy, and no instances of missed significant abnormalities (e.g., blasts or dysplastic forms) were observed during this evaluation.
Beyond statistical performance, morphological evaluation (Fig. 4) revealed a high degree of visual similarity between the traditional blood smear (BS) and the Hilab Lens (HL) liquid medium, reinforcing the consistency of the system in capturing key hematological features. RBC abnormalities, such as anisocytosis and poikilocytosis, were identical in both methods (Fig. 4A). Likewise, red cell agglutination and PLA aggregates displayed comparable structural characteristics in both BS and HL (Fig. 4B-E). For immature WBCs evaluation, exhibited analogous staining patterns and nuclear characteristics when comparing HL and BS (Fig. 4H-I). These images are presented for illustrative purposes only, to demonstrate the morphological preservation and contrast between mature and immature cells, and RBC/PLA alterations between methodologies. Quantitative performance data related are presented separately in Table 2.
Panel (A) shows RBCs analyzed by the Hilab Lens from a patient with significant hematological alterations, highlighting anisocytosis and different types of poikilocytosis. Panels (B, C) compare RBC clumps observed in BS and HL, respectively. Similarly, Panels (D, E) illustrate platelet aggregates in both methods. Panels (F, G) display different types of mature WBC as observed in the BS and HL, respectively. Panels (H, I) illustrate immature WBC, comparing their morphology in the BS and in the HL liquid medium. BS were stained with May–Grunwald–Giemsa dye, and images were captured using a Global Optics (NO115T1, Global Trade Technology, São Paulo, Brazil) optical microscope (original magnification × 1000).
Interference study
The Hilab Lens maintained strong correlation with the reference method under all evaluated interfering conditions (Table 3), with Pearson correlation coefficients (r) between 0.97 and 0.99. In samples with hemoglobinopathies and high turbidity, the device achieved r = 0.99, with slopes of 0.93 and 0.98, respectively, and negligible bias (0.01 and 0.09 g/dL). For samples collected via central venous catheter, the correlation remained high (r = 0.97), though with a slope of 1.13 and a slightly higher bias (0.36 g/dL), and broader limits of agreement (–1.21 to 1.93 g/dL). In the presence of EDTA excess, the system showed r = 0.97, a slope of 1.06, and a bias of 0.39 g/dL. Across all conditions, no statistically significant differences were observed when compared to the reference method (p > 0.5), supporting the robustness of the Hilab Lens against potential interferences.
Venous vs. capillary comparison
The comparative analysis between capillary and venous samples demonstrated strong agreement across all analytes. Passing-Bablok regression confirmed linearity, with slopes close to 1 and intercepts within acceptable ranges (e.g., RBC: slope = 0.97 [0.877–1.087]; PLA: 0.999 [0.885–1.117]; HB: 0.96 [0.802–1.107]). Pearson correlation coefficients further supported this, ranging from 0.81 to 0.97 (e.g., WBC: r = 0.973 [0.952–0.984]; RBC: r = 0.956 [0.923–0.975]; HB: r = 0.902 [0.833–0.943]; p < 0.001 for all), confirming strong linear association between sample types (Fig. 5; Supplementary Table 4).
Passing-Bablok plots related to effect of sample type study between venous and capillary blood samples for Erythrocytes (A), Platelets (B), Hemoglobin (C), Hematocrit (D), MCV (E), MCH (F), Total Leukocytes (G), Neutrophils (#; H), Lymphocytes (#; I), Monocytes (#; J) and Eosinophils/Basophils (#; K). Graphs also present Pearson correlation, Intercept (95% CI) and Slope (95% CI) for each analyte. Green dotted lines indicate the healthy individuals’ expected values. Blue solid lines and red dotted lines demonstrate the regression and identity lines, respectively (number of patients = 50).
Bland-Altman analysis (Supplementary Fig. 1, Supplementary Table 4) revealed minimal bias and clinically acceptable agreement limits for all parameters (e.g., HB: bias = –0.30 g/dL [–0.53 to –0.07], limits = –1.88 to 1.29 g/dL; RBC: bias = –68,285, limits = –518,153 to 381,582), reinforcing equivalence between matrices. No statistically significant differences were observed in any analyte when comparing capillary and venous samples (e.g., HB: SE = 0.26; p = 0.42; WBC: SE = 421.67; p = 0.83) analyzed in the Hilab Lens device.
Evaluation of patient preferences
All legal guardians (n = 50) completed the questionnaire at the anonymous collection site. The mean satisfaction score on a scale of 1 (Disagree) to 5 (Agree) for their acceptability with POC tests was 4.54 (before the fingerprick collection) and 4.76 (after blood collection) for Hilab Lens perception in pediatric care. Only one legal guardian included additional comments, reporting that there was strangeness regarding the capillary collection due to the autism of the child.
Before the blood collection and test execution, participants expressed a high level of motivation regarding the potential of POC tests for monitoring their children’s health conditions, with 94% agreeing or somewhat agreeing that POC tests would encourage them to be more proactive about health management (Fig. 6A). Additionally, 50% of participants trusted venous blood samples more than capillary blood samples, whereas 48% expressed equal trust in both sample types (Fig. 6B). Convenience was another key aspect, with 92% agreeing that it would be better to discuss the test results immediately with a physician rather than return for a second appointment (Fig. 6C). The reduction in logistical burdens was also valued, as 80% of respondents agreed or somewhat agreed that not having to go to an external laboratory for testing would be more convenient (Fig. 6D), and 98% recognized that external laboratory visits involved additional time and transportation costs (Fig. 6E). Finally, 90% of guardians agreed that receiving test results at home or in a clinical setting could streamline medical decision-making (Fig. 6F).
A−F illustrate results collected through the first questionnaire, performed before the blood collection and CBC test operation, capturing participants’ motivation, trust in sample types, convenience, and the importance of rapid results. G−J show results collected through the second questionnaire, performed after the test execution, reflecting comfort with capillary blood collection, perceived effectiveness, the suitability of POC testing for children, and overall satisfaction (number of patients = 50).
After the capillary blood collection and test execution, participants evaluated their experiences with the procedure and the Hilab Lens device. Regarding the capillary blood collection process, 98% of respondents described the procedure as either very effective (52%) or effective (44%), with only 4% considering it somewhat effective and none rating it as ineffective (Fig. 6H). The Hilab Lens device was perceived positively, with 96% of guardians agreeing that performing a CBC via a POC test was an advantageous alternative for children (Fig. 6I). When considering the overall experience with the Hilab Lens and the CBC test execution, 98% of participants rated their satisfaction as excellent (52%) or good (46%), with no participants reporting dissatisfaction (Fig. 6J).
Discussion
The initial iteration of the Hilab System, comprising the Hilab Lens and Hilab Flow devices, was developed to provide comprehensive CBC analyses at the point of care. Clinical validation studies demonstrated accurate and reliable results, comparing this system with conventional hematology analyzers. However, the necessity of operating two separate devices and managing distinct diagnostic kits for microscopy and colorimetry posed challenges related to usability and workflow efficiency.22,23 In response to these challenges, the Hilab optimized the Hilab Lens into a single device capable of performing optical microscopy and spectrophotometric analyses in a single workflow.
The present study was designed to evaluate the performance of this optimized Hilab Lens in comparison to the Sysmex XE-2100 methodology and traditional blood smear technique, both of which were used as reference methods in this study for CBC testing,8 evaluating key analytical aspects such as precision, method comparison, sample type equivalence, potential interfering factors, and user experience. The study population comprised pediatric patients from Erastinho Hospital, the first dedicated pediatric oncology hospital in Parana, Brazil, underscoring the device’s applicability in specialized clinical settings focused on childhood cancer care. The heterogeneity of samples ensured a broad spectrum of hematological profiles, allowing a comprehensive evaluation of the Hilab Lens’ performance across distinct clinical scenarios.
The precision evaluation of the Hilab Lens included assessments of repeatability (intra-assay precision) and reproducibility (inter-assay precision) for RBC, HB, PLA, and WBC. Considering the higher variability observed for POC instruments, the study utilized the “higher CI limit” criteria (EFLM Biological Variation Database) to calculate the acceptable limits of CV, Bias, and Total Error for all analytes. Although the intra-assay CV for HB slightly exceeded the EFLM criteria, all analytes demonstrated acceptable performance in terms of CV, total error, and bias (Table 1). According to CLSI EP15-A3 guidelines, CV should not be interpreted in isolation; when it exceeds the recommended threshold, acceptable values for total error and bias may justify the result. In this context, the observed values for HB remain compliant with CLSI standards. Additionally, a Gage R&R analysis was conducted to evaluate sources of measurement variability in the Hilab Lens, including repeatability (within-device precision), reproducibility (operator-related variability), and part-to-part variation (biological variability among samples; Supplementary Table 2). Part-to-part variability was the dominant source, accounting for ≥98% of total variance across all analytes. This finding indicates that most variability arises from intrinsic biological differences rather than inconsistencies in measurement.24 Reproducibility showed minimal impact for all parameters, supporting the robustness of the method in point-of-care applications and aligning with performance standards observed in established hematology analyzers.25
The method comparison between the Hilab Lens and the conventional hematology analyzer demonstrated strong concordance across key hematological parameters. Specifically, RBC, PLA, HB, WBC, NEU, LYM, and MON showed high Pearson correlation coefficients (r = 0.95–0.99), and the Passing-Bablok regression analyses yielded intercepts and slopes with 95% confidence intervals encompassing 0 and 1, respectively (Fig. 3; Supplementary Table 3). These results indicate the absence of significant constant or proportional bias between the methods.26 This performance is consistent with previous studies, such as the comparison between the Mindray BC-5180 and the Sysmex XN-1000, which also reported strong correlation and negligible bias across similar analytes.25
For estimated parameters, such as HT, MCV, and MCH, the regression analyses revealed intercepts and slopes with 95% CI not encompassing 0 and 1, suggesting potential systematic differences. This phenomenon has been observed in other clinical studies, for instance, DxH 900 (Beckman Coulter), Yumizen H2500 (Horiba), Dirui BF-7200, Alinity HQ (Abbott), and XN20 system (Sysmex) comparison, where calculated parameters showed higher statistical variance, attributed to differences in measurement principles (e.g., impedance vs. optical), calibration standards, and proprietary algorithms.27,28,29,30 Despite these statistical discrepancies, the qualitative analysis of the Hilab Lens demonstrated high accuracy, specificity, and sensitivity, indicating that these differences do not impact clinical decision-making. Regarding EOS/BAS counts, although the regression parameters did not meet the stringent criteria set by CLSI, this challenge is not unique to the Hilab Lens. Other conventional analyzers, such as the Alinity HQ and DxH 900, have also faced difficulties in achieving these criteria for EOS and BAS counts due to the low prevalence of these cells in the bloodstream.26,31 Nevertheless, high accuracy values for this parameter were observed in the qualitative analysis of the Hilab device, supporting its clinical reliability.
Most analytes demonstrated minimal bias and acceptable limits of agreement in the Bland-Altman analysis, supporting the clinical interchangeability between the Hilab Lens and the conventional method (Fig. 2). Low bias values for HB, WBC, PLA, RBC, NEU, LYM, and MON reinforce the consistency and reliability of the Hilab Lens. Although HT, MCV, and MCH showed wider limits of agreement, no statistically significant differences were observed in the Student’s t-test (p > 0.5 for all analytes), indicating that such variability does not translate into meaningful clinical impact. Similarly, the qualitative classification analysis (Supplementary Table 1) reinforces the clinical reliability of the Hilab Lens. Accuracy, specificity, and sensitivity exceeded 98.9% for all parameters, with Cohen’s Kappa coefficients above 0.95, indicating near-perfect agreement with a reference method. Positive predictive values (PV + ) above 97%, along with LR+ ranging from 120.9 to 9869.3 and LR– between 0.003 and 0.100, demonstrate strong diagnostic performance. These findings are consistent with previous validation studies,26,31 supporting the method’s robustness even in the presence of statistical variation in calculated indices.
A central concern in clinical laboratories is the effectiveness of flagging detection by automated hematological analyzers. The specificity of automated hematology analyzers is often unsatisfactory, frequently generating false positives results. Prior literature highlights that automated hematological devices—especially those based on impedance technology—may misclassify PLA aggregates as small RBC or fail to accurately detect macrothrombocytopenia. These events trigger unnecessary manual smear reviews by trained hematologists. While microscopy is considered the gold standard methodology for cell review, this technique is labor-intensive and subject to inter-observer variability, making it challenging for high-throughput clinical workflows.29,30,31,32,33
Considering the false results associated with conventional hematological devices for flagging detection, for this parameter this study adopted conventional microscopy as the reference method to evaluate the Hilab Lens performance, as previously established in literature.33 As a result, the Hilab optical-based system demonstrated robust agreement with standard technique in identifying key hematological disturbances, for all evaluated parameters (Table 2). Regarding anisocytosis, although the Hilab Lens does not provide a quantitative measurement, it is noteworthy that automated hematology analyzers may present limitations in accurately quantifying RDW in the presence of microcytes or RBC agglutinates, leading to potential misclassification.18,29,30,31,32,33 In contrast, the Hilab Lens’s morphological approach offers more reliable detection in such scenarios. The detection of band neutrophils and immature cells exhibited high overall accuracy (99.6% and 98.8%, respectively) but the lowest sensitivity among the flagged parameters (94.4% and 94.9%). Notably, these lower sensitivity values were exclusively observed in patients with WBC values below 1,500/µL. In such leukopenic conditions, the smaller evaluation area of the Hilab Lens, compared to the traditional blood smear, affects the test sensitivity. However, the clinical significance of detecting immature cells diminishes in these cases.34 These findings support the clinical applicability of the Hilab Lens in environments where traditional smear analysis may not be accessible, reinforcing its potential to streamline morphological evaluation without compromising diagnostic accuracy.
The visual evaluation further reinforced the high concordance between the liquid medium of the Hilab Lens and the pattern observed in the traditional blood smear technique. RBC abnormalities, including anisocytosis and poikilocytosis, were similarly detected by both methodologies. Mature and immature WBCs retained well-preserved nuclear and cytoplasmic features in both techniques, with consistent staining characteristics. For Hilab Lens, immature cells tend to acquire a pinker hue, while mature WBCs stain more prominently in purple—a phenomenon consistent with cytochemical staining variations observed in conventional hematology (Fig. 4). Although Fig. 4 illustrates the morphological detail captured by the Hilab Lens, it was not intended to demonstrate diagnostic accuracy for specific WBC abnormalities, such as blasts. In the context of this study, the term “immature cells” refers to any white blood cells exhibiting morphological features of immaturity, including promyelocytes, myelocytes, metamyelocytes, and lymphoid/myeloid blasts, as identified by trained hematologists during conventional microscopy review. While these subtypes hold distinct clinical significance, the current version of the Hilab Lens does not attempt to subclassify immature WBCs. Instead, it provides a flag indicating the general presence of immature cells, which serves as a critical alert for further clinical investigation. This definition was adopted to align with the device’s present capabilities and the scope of validation conducted in this study. However, the high morphological similarity between its liquid medium and traditional blood smears suggests that future iterations may integrate this functionality through further AI-based training.
Considering the Hilab Lens optimization to encompass the spectrophotometric technique, the interference study (Table 3) evaluated the robustness of the device in measuring HB levels across various potential interfering conditions.35 The results demonstrated strong correlations with the conventional hematology analyzer across all tested conditions, with Pearson correlation coefficients (r) ranging from 0.97 to 0.99. High correlation values in samples with hemoglobinopathies suggest that the device accurately measures the analyte even for abnormal protein variants. For samples exhibiting EDTA excess and high turbidity, often due to elevated lipid or protein levels, the Hilab Lens also showed minimal bias, indicating that the device effectively mitigates the impact of these interferences on HB measurements. Regarding samples collected via central venous catheter, the slope and a bias indicate a slight proportional difference, but no statistical differences were observed (p > 0.5) for HB results. This variability may stem from factors such as hemodilution or contamination during sample collection, which are known to affect laboratory results.36
In pediatric oncology, difficult venous access due to small veins and treatment-related vascular changes makes blood collection technically challenging and distressing. Similarly, elderly patients often present fragile veins and comorbidities that complicate blood collection, and hospitalized patients undergoing daily venous blood sampling can experience significant blood loss, with an increased risk of iatrogenic anemia.37,38,39 The comparative analysis of Hilab Lens for venous and capillary blood samples demonstrated a strong correlation across all CBC parameters. Passing-Bablok regression further confirmed the high level of agreement for all analytes, and the Student’s t-test indicated no statistically significant differences between samples for all parameters (Fig. 5; Supplementary Fig. 1, Supplementary Table 4).
Although previous studies have reported differences between capillary and venous blood samples in CBC parameters,2,37 other studies did not observe significant differences between capillary and venous samples.22,23,40,41,42 This consistency could be attributed to standardized sample collection procedures, minimizing pre-analytical variability, and a study population with stable physiological conditions, reducing the impact of factors that might cause discrepancies in other studies. Thus, these data support capillary blood collection as a viable alternative for CBC analysis, offering a less invasive approach while maintaining analytical reliability.
Given the significant cognitive and communicative differences between younger children and adolescents, besides their ability to self-report experiences, we opted to perform the patient preferences study by administering a unique questionnaire for the legal guardians. This approach ensured a more consistent evaluation of patient acceptability by capturing the perception of the caregivers, who are directly responsible for the child’s well-being and decision-making in healthcare settings. The study revealed a high level of satisfaction with the Hilab Lens POC device, with mean scores of 4.54 for participants’ motivation; trust in the sample types; convenience; and the importance of rapid results; and 4.76 for comfort with capillary blood collection; perceived effectiveness; the suitability of POC testing for children; and overall satisfaction; following the procedure on a 5-point scale (Fig. 6). These findings align with existing literature emphasizing the POCT benefits in pediatric healthcare, such as enhancing patient satisfaction by reducing discomfort, turnaround times, the need for multiple visits, and improving the overall patient experience.43,44,45
Despite the positive perception, the study was limited to 50 participants due to critical health conditions of most patients, which highlights the challenges in conducting research within vulnerable populations. Another important consideration regarding the blood collection site is the potential infection risk in severely immunocompromised patients. Children are naturally inclined to touch various objects, so fingertip collection may be considered more susceptible to contamination by opportunistic microorganisms. Ensuring the ideal preparation of the care environment and the adequate conduct of medical procedures reduce the risks, but they must be discussed and verified with the physician before their application.46
While the Hilab Lens demonstrated strong analytical performance, this study has some limitations. First, the Hilab Lens performs a 4-part WBC differential, without distinguishing EOS and BAS individually. However, this limitation is unlikely to impact clinical interpretation significantly, as marked eosinophilia and basophilia typically present with distinct clinical features and etiologies, aiding differential diagnosis even when counted jointly.47 Additionally, although the device showed high agreement with a reference hematology analyzer under controlled conditions, external validation with other reference devices and in diverse clinical environments—such as primary care units and remote healthcare settings—is required to confirm robustness and reproducibility outside specialized centers. Another limitation is the potential variability introduced by manual pipetting; despite rigorous operator training and the use of fixed-volume pipettes, improper technique can affect sample accuracy. This highlights the need for continued operator education (Supplementary note 1) and future development of consumables or collection devices that minimize pre-analytical variability. Furthermore, pre-analytical factors such as blood sample quality—including clot formation in capillary samples—may influence results, underscoring the importance of optimizing collection protocols. Lastly, although this study was conducted in a pediatric oncology center, applicability to other populations, such as neonates, warrants further investigation. Future studies should expand validation across broader clinical scenarios, integrate real-time quality control mechanisms, and continue refining AI algorithms to enhance performance in complex hematological conditions.
Another important aspect is related to cost effectiveness. Traditional automated hematology analyzers typically require investments of USD 50,000–150,000, plus additional costs exceeding USD 20,000 for laboratory infrastructure adaptations, annual maintenance fees of USD 5,000–15,000, and reagent costs of USD 1.00–2.50 per test, in addition to requiring specialized personnel. Other point-of-care devices have acquisition costs of USD 15,000–25,000 and consumables priced around USD 7–10 per test. In contrast, the Hilab Lens offers a more accessible alternative, with a device cost below USD 3,500 and diagnostic kits priced under USD 5 per test, operating effectively in decentralized settings without the need for specialized laboratory rooms or daily maintenance by engineers. The Hilab Lens’ automatic calibration via a proprietary capsule and adaptable business model further enhance its cost-effectiveness compared to both conventional analyzers and other POC hematology platforms, making it especially suitable for primary care facilities and low-resource environments.
In summary, the Hilab Lens demonstrated strong analytical performance, comparable to golden standard techniques, while offering the advantage of rapid, decentralized testing. Its ease of use, minimal infrastructure requirements, and AI-powered analysis make it a valuable alternative for hematology testing in resource-limited settings. The high user acceptance among caregivers also highlights its potential for broader adoption in pediatric care. Future work should focus on expanding its clinical applications and optimizing its usability in non-specialized healthcare settings. These findings support the implementation of AI-driven point-of-care hematology devices as a transformative tool in modern diagnostics, enhancing accessibility and improving patient outcomes.
Data availability
The datasets generated and analyzed during this study are available from the corresponding author upon request. Source data underlying the figures are provided as Supplementary Data. Any additional information related to the study is available upon request, in accordance with institutional and ethical guidelines.
References
Deininger, M. W. Molecular monitoring in CML and the prospects for treatment-free remissions. Hematol. Am. Soc. Hematol. Educ. Prog. 2015, 257–263 (2015).
Hongxiang, X. et al. Consistency analysis of two fingertip capillary blood sampling methods for complete blood count. Sci. Rep. 14, 15011 (2024).
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Steliarova-Foucher, E. et al. International incidence of childhood cancer, 2001-10: a population-based registry study. Lancet Oncol. 18, 719–731 (2017).
Syed-Abdul, S. et al. Artificial intelligence based models for screening of hematologic malignancies using cell population data. Sci. Rep. 10, 4583 (2020).
Wu, Y. et al. Global, regional, and national childhood cancer burden, 1990-2019: An analysis based on the Global Burden of Disease Study 2019. J. Adv. Res. 40, 233–247 (2022).
Shanmugavadivel, D. et al. The childhood cancer diagnosis (CCD) study: a UK observational study to describe referral pathways and quantify diagnostic intervals in children and young people with cancer. BMJ Open 12, e058744 (2022).
Meintker, L., Ringwald, J., Rauh, M. & Krause, S. W. Comparação de contagens diferenciais automatizadas de células sanguíneas de Abbott Sapphire, Siemens Advia 120, Beckman Coulter DxH 800 e Sysmex XE-2100 em amostras normais e patológicas. Am. J. Clin. Pathol. 139, 641–650 (2013).
Kuupiel, D., Bawontuo, V. & Mashamba-Thompson, T. P. Improving the accessibility and efficiency of point-of-care diagnostics services in low- and middle-income countries: Lean and agile supply chain management. Diagnostics ((Basel)) 7, 58 (2017).
Rabinovitch, A. et al. Validation, verification, and quality assurance of automated Hematology analyzers; approved standard—second edition. Clin. Lab Stand Inst. 30, H26-A2 (2010).
Incir, S. & Palaoglu, K. E. Evaluation of the performance of sysmex XN-3100 automated hematology analyzer regarding the sysmex XE-2100 and microscopic examination. Turk. J. Biochem. 46, 29–37 (2021).
Redmon, J., Divvala, S., Girshick, R. & Farhadi, A. You only look once: unified, real-time object detection. In Proceedings of the 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), 779–788 (IEEE, 2016).
Walker, H. K., Hall, W. D. & Hurst, J. W. Clinical Methods: The History, Physical, and Laboratory Examinations 3rd edn (Butterworths, 1990).
CFR. Chapter I, Part 814—Premarket Approval of Medical Devices 4–1–22 edn (FDA: Silver Spring, MD, USA, 1986).
Franc, J. M. et al. Accuracy of a commercial large language model (ChatGPT) to perform disaster triage of simulated patients using the simple triage and rapid treatment (START) protocol: gage repeatability and reproducibility study. J. Med. Int. Res. 26, e55648 (2024).
Piaton, E. et al. Recommandations techniques et règles de bonne pratique pour la coloration de May-Grünwald-Giemsa: revue de la littérature et apport de l’assurance qualité. Ann. Pathol. 35, 294–305 (2015).
International Society Laboratories Haematology. Consensus Rules for Smear Blood Review. http://www.islh.org/web/consensus_rules.php (2020).
Gulati, G., Uppal, G. & Gong, J. Unreliable automated complete blood count results: causes, recognition, and resolution. Ann. Lab Med. 42, 515–530 (2022).
World Health Organization. WHO Guidelines on Drawing Blood: Best Practices in Phlebotomy. https://www.who.int/publications/i/item/9789241599221 (2010).
Laurence, C. O. et al. Patient satisfaction with point-of-care testing in general practice. Br. J. Gen. Pract. 60, e98–e104 (2010).
Chow, S.-C., Shao, J., Wang, H. & Lokhnygina, Y. Sample Size Calculations in Clinical Research 3rd edn (Chapman and Hall/CRC, 2017).
Gasparin, A. T. et al. Hilab system, a new point-of-care hematology analyzer supported by the internet of things and artificial intelligence. Sci. Rep. 12, 10409 (2022).
Gasparin, A. T. et al. Hilab system device in an oncological hospital: a new clinical approach for point of care CBC test, supported by the internet of things and machine learning. Diagnostics ((Basel)) 13, 1695 (2023).
Burdick, R. K., Borror, C. M. & Montgomery, D. C. Design and Analysis of Gauge R&R Studies: Making Decisions with Confidence Intervals in Random and Mixed Effects Models. ASA-SIAM Series on Statistics and Applied Probability. https://epubs.siam.org/doi/book/10.1137/1.9780898718379 (2005).
Wang, J., Zhao, S., Su, Z. & Liu, X. Analytical comparison between two hematological analyzer systems: mindray BC-5180 vs sysmex XN-1000. J Clin Lab Anal 33, e22955 (2019).
Bilić-Zulle, L. Comparison of methods: passing and bablok regression. Biochem. Med. ((Zagreb)) 21, 49–52 (2011).
Serrando Querol, M. et al. Evaluation of the new beckmann coulter analyzer DxH 900 compared to sysmex XN20: analytical performance and flagging efficiency. Diagnostics ((Basel)) 11, 1756 (2021).
Małecka, M. & Ciepiela, O. A comparison of sysmex-XN 2000 and Yumizen H2500 automated hematology analyzers. Pract. Laboratory Med. 22, e00186 (2020).
Seyithanoğlu, M. uhammed & Tuncer, H. ilmi İsmet, Tolun, Fatma İnanç and Kilinç, Metin. “Comparison of results of two hematological analyzer systems: dirui BF-7200 and sysmex XN-1000”. Turkish J. Biochem. 49, 157–166 (2024).
Slim, C. L. & Wevers, B. A. Demmers MWHJ, Lakos G, Hoffmann JJML, Adriaansen HJ, Kooren JA, Storm H. Multicenter performance evaluation of the Abbott Alinity hq hematology analyzer. Clin. Chem. Lab Med. 57, 1988–1998 (2019).
Bayraktar, N. & Topcu, D. I. User verification of Abbott Alinity HQ hematology analyzer. Turk J. Biochem. 49, 167–177 (2024).
Bhola, R. K., Fudaly, C. & Rastogi, S. A comparative evaluation of performance of sysmex xn 3000 and horiba yumizen h2500 automated complete blood count analysers. Indian J. Hematol. Blood. Transfus. 40, 303–314 (2024).
Cornet, E., Perol, J. P. & Troussard, X. Performance evaluation and relevance of the CellaVision DM96 system in routine analysis and in patients with malignant hematological diseases. Int. J. Lab. Hematol. 30, 536–542 (2008).
Tigner, A., Ibrahim, S. A. & Murray, I. V. Histology, White Blood Cell. In: StatPearls [Internet]. Treasure Island (FL): StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK563148/ (2025).
Berkow, L. Factors affecting hemoglobin measurement. J. Clin. Monit. Comput. 27, 499−508 (2013).
Ponticelli, E. et al. Complete blood count collected via venipuncture versus peripherally inserted central catheter in hematological patients: a comparison of 2 methods. Cancer Nurs 45, E36–E42 (2022).
Becker, M. et al. Differences between capillary and venous blood counts in children—a data mining approach. Int. J. Lab Hematol. 44, 729–737 (2022).
Crocoli, A. et al. Vascular access in pediatric oncology and hematology: state of the art. Children ((Basel)) 9, 70 (2022).
Matzek, L. J. et al. A contemporary analysis of phlebotomy and iatrogenic anemia development throughout hospitalization in critically ill adults. Anesth Analg 135, 501–510 (2022).
Doeleman, M. J. H. et al. Stability and comparison of complete blood count parameters between capillary and venous blood samples. Int J Lab Hematol 45, 659–667 (2023).
Noble, L. D. et al. Painless capillary blood collection: a rapid evaluation of the onflow device. Diagnostics ((Basel)) 13, 1754 (2023).
Wisanuvej, K. et al. Comparison between blood hemoglobin concentration determined by point-of-care device and complete blood count in adult patients with dengue. PLoS Negl. Trop Dis. 15, e0009692 (2021).
Patel, K. & Suh-Lailam, B. B. Implementation of point-of-care testing in a pediatric healthcare setting. Crit. Rev. Clin. Lab Sci. 56, 239–246 (2019).
Caroline, O. L. et al. Patient satisfaction with point-of-care testing in general practice. Br. J. Gen. Pract. 60, e98–e104 (2010).
Hayes, B., Murphy, C., Crawley, A. & O’Kennedy, R. Developments in point-of-care diagnostic technology for cancer detection. Diagnostics ((Basel)) 8, 39 (2018).
Acebo, J. J. et al. Infections in immunosuppressed pediatric patients. In Pediatric Surgical Oncology (eds. Lakhoo, K., Abdelhafeez, A. H., Abib, S) (Springer, Cham, 2023);
Simon, H. U. et al. The cellular functions of eosinophils: collegium internationale allergologicum (CIA) update 2020. Int. Arch. Allergy Immunol. 181, 11–23 (2020).
Acknowledgements
The authors would like to express their gratitude to Carolina Rodrigues de Araujo Perazzoli for the creation of Fig. 1 and the graphical abstract. Her contribution in designing the visual elements significantly improved the clarity and quality of the manuscript.
Author information
Authors and Affiliations
Contributions
Aléxia Thamara Gasparin, Claudiane Isabel Franco Araujo, and Raphaela Rodrigues de Faria developed the diagnostic test kit, and were responsible for sample collection and processing. Ivan Lucas Reis Silva, Luis Felipe Hartmann and Bruno Ribeiro Medeiros developed and trained the AI models. João Victor Predebon, Luis Felipe Hartmann, Sergio Rogal, and Marcus Vinícius Mazega Figueredo developed the Hilab Lens hardware and embedded software. Bruna Hammerschmitt Gelain, Thaina Caldeiras Watanabe, Sergio Rogal, and Marcus Vinícius Mazega Figueredo developed the backend and frontend software. Olair Carlos Beltrame and Robson de Castro Coelho processed and analyzed the results obtained at Erastinho Hospital using the conventional hematology analyzer. Aléxia Thamara Gasparin and Bernardo Montesanti Machado de Almeida supervised the project and performed the statistical analyses. All authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
Aléxia Thamara Gasparin, Claudiane Isabel Franco Araujo, Raphaela Rodrigues de Faria, Ivan Lucas Reis Silva, Bruno Ribeiro Medeiros, Luis Felipe Hartmann, João Victor Predebon, Bruna Hammerschmitt Gelain, Thaina Caldeiras Watanabe, Bernardo Montesanti Machado de Almeida, Sergio Rogal, and Marcus Vinícius Mazega Figueredo are employees of Hilab, the company that developed the Hilab Lens device evaluated in this study, and therefore declare a financial competing interest. All other authors declare no competing interests. The datasets used and/or analysed during the current study are available from the corresponding. Please contact alexia.gasparin@hilab.com.br.
Peer review
Peer review information
Communications Medicine thanks Timothy Woolley, Mohammed Bensalah and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gasparin, A.T., Araujo, C.I.F., de Faria, R.R. et al. Clinical validation of the Hilab lens for AI supported point of care CBC testing in pediatric oncology. Commun Med 5, 518 (2025). https://doi.org/10.1038/s43856-025-01223-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01223-9