Abstract
Background
Viral and infectious illnesses can exert profound and enduring effects on population health and well-being. In the aftermath of SARS-CoV-2 infection, post-acute sequelae, collectively referred to as Long COVID, have emerged as a major global health challenge, affecting more than 400 million people and contributing to estimated annual economic costs exceeding $1 trillion.
Scope of the review
Long COVID encompasses a wide and heterogeneous spectrum of debilitating symptoms, including cognitive dysfunction, sleep disturbances, severe fatigue, and post-exertional malaise. Despite its substantial burden, fundamental uncertainties remain regarding its underlying pathophysiology, the development of robust diagnostic criteria, and the identification of effective therapeutic options.
Key insights
This review synthesises current evidence on the biological mechanisms thought to contribute to Long COVID, spanning immune dysregulation, viral persistence, autonomic dysfunction, microvascular pathology, and other emerging hypotheses. We examine advances and limitations in contemporary diagnostic approaches and critically appraise existing treatment strategies, highlighting inconsistencies and gaps that hinder clinical consensus.
Implications
By integrating interdisciplinary insights, this review underscores the urgent need for mechanistic clarity, validated diagnostic frameworks, and rigorously evaluated treatment pathways. Addressing these gaps will be essential to developing effective, evidence-based management strategies and mitigating the long-term impact of Long COVID on global health.
Plain language summary
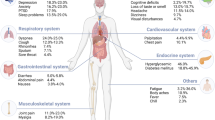
COVID-19 has caused millions of deaths worldwide, but its impact goes beyond the initial infection. Some people develop Long COVID, a condition where symptoms persist or appear months after infection, even in mild or asymptomatic cases. Long COVID affects multiple body systems and can involve over 200 symptoms, often resembling chronic fatigue syndrome. Its causes remain unclear, and there are no proven treatments or reliable diagnostic tests. Research into blood biomarkers and symptom patterns holds promise, but progress is slow due to the complexity of the condition. This review explores current knowledge, gaps, and future directions for research, diagnosis, and treatment.
Similar content being viewed by others
Introduction
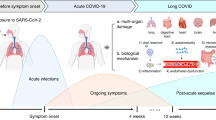
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus responsible for acute COVID-19, has caused over seven million deaths worldwide1. However, the burden of COVID-19 extends beyond the acute phase, with an estimated excess of 15 million deaths globally linked to SARS-CoV-2-related complications2. Following infection, a subset of individuals do not fully recover but instead develop new, persistent, or episodic symptoms that significantly impair their quality of life and disrupt daily functioning3,4. The risk of developing Long COVID increases with the severity of the initial illness. A population-based cohort study found that individuals with mild to moderately severe COVID-19 were three times more likely to develop Long COVID than asymptomatic individuals. In contrast, those with severe or critical illness had nearly a tenfold increased risk5. Importantly, even asymptomatic infection can result in long-term sequelae6. These persistent symptoms are referred to by various terms, including ongoing symptomatic COVID-19, post-COVID-19 condition or syndrome, and post-acute sequelae of COVID-19 (PASC). However, the most widely recognised term, originating from the patient community, is Long COVID7,8,9.
Definitions of Long COVID have been revised multiple times, reflecting both limited understanding of its underlying mechanisms and the growing recognition that it encompasses several subtypes10. Current definition is persistence or emergence of new symptoms three months after initial SARS-CoV-2 infection, lasting for at least two months and not explained by an alternative diagnosis11. Long COVID is a complex, multisystem condition associated with over 200 reported symptoms, ranging in severity from mild to debilitating presentations12. In some cases, the illness follows an episodic pattern, but its overall clinical course and trajectory remain poorly understood. Notably, there is substantial symptomatic and clinical overlap with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), another condition often triggered by viral infection13.
Currently, no curative treatments for Long COVID have been established, mainly due to its poorly understood and likely heterogeneous pathophysiological basis. Progress has also been hampered by the absence of clear diagnostic criteria and validated biomarkers. Although research has advanced our understanding of potential mechanisms underlying Long COVID, significant gaps remain in the knowledge required to develop practical diagnostic tools, treatments, and management strategies. Anecdotal evidence from several pharmacological trials suggests potential therapeutic benefit; however, these findings have yet to be rigorously tested in large-scale, randomised controlled trials. Furthermore, the limited availability of tools to stratify patients, such as predictive biomarkers or detailed clinical and physiological assessments, continues to impede the design and implementation of effective clinical trial programmes.
Identifying blood-based biomarkers for the diagnosis and treatment of Long COVID has been a key research priority; however, progress has been limited by the condition’s heterogeneous nature. Inconsistencies across studies, such as variation in which cyto-/chemokines are examined, differences in assay sensitivity, and a lack of standardisation in sample collection, have further hindered biomarker discovery. Important variables, including illness duration, time of day or season of sample collection14, and the presence of pre-analytical stressors (especially in individuals prone to post-exertional malaise [PEM] or post-exertional symptom exacerbation [PESE]), are rarely accounted for. A systematic review by Thomas et al.15 identified several candidate blood biomarkers associated with Long COVID, reflecting immunological and inflammatory dysfunction, endothelial/vascular impairment, metabolic disruption, and clotting abnormalities. However, no disease-specific biomarker or set of biomarkers has yet been validated, and distinct subtypes of Long COVID may likely require different diagnostic signatures. Proteomic analyses measuring over 6500 proteins at various post-infection time points have revealed transient complement system alterations in individuals with Long COVID, which tend to normalise in those who recover naturally. Given the complexity of the condition, future research may need to focus on subtype-specific biomarker profiles to enable patient stratification and guide targeted treatment strategies. Emerging machine learning approaches, such as those described by Liew et al.16, have begun to link symptom clusters with specific biomarker patterns, offering promising avenues to refine diagnostics and therapeutic approaches.
In this review, we synthesise clinical literature on Long COVID to explore current understanding of its prevalence, pathophysiology, and potential treatment options. Where relevant, we also incorporate comparative insights from studies on ME/CFS and Lyme disease, conditions that share overlapping features of post-infection-associated symptoms. Priority was given to studies employing stringent diagnostic and inclusion criteria to minimise confounding from factors such as hospitalisation during acute infection, pre-existing comorbidities, and unrelated etiologies. This review aims to identify key knowledge gaps and highlight new directions for therapeutic development that may guide future collaborative research efforts.
Prevalence and economic burden of long COVID
The global prevalence of Long COVID remains uncertain, largely due to the absence of standardised diagnostic criteria, inconsistent public health surveillance, and regional differences in pandemic dynamics. Recent studies estimate that between 65 and 400 million people worldwide have experienced persistent symptoms following confirmed SARS-CoV-2 infection7,17. Despite this wide range, these figures are likely conservative, and the true global health burden is almost certainly higher. The lack of diagnostic clarity often results in patients consulting multiple healthcare providers, many of whom may lack the expertise to manage the condition effectively. Collectively, this leads to increased medical costs and delayed access to care. Additionally, the inability of many individuals with Long COVID to work contributes significantly to both economic losses and broader societal impacts. While precise estimates of the global economic burden are still emerging, preliminary analyses suggest it could exceed $1 trillion annually17. As research advances, the full scope of Long COVID’s impact on global economies, healthcare systems, and population health will become increasingly clear18.
Theories of long COVID pathophysiology
Long COVID is a sequela of SARS-CoV-2 infection and a unique condition driven by complex, interacting mechanisms (Fig. 1), resulting in a wide range of symptoms that significantly impact quality of life19. Here, we provide a comprehensive overview of the leading hypotheses regarding the underlying pathophysiology of Long COVID. Emerging empirical evidence implicates several interconnected mechanisms, including immune dysregulation (such as autoimmunity), gut microbiome dysbiosis, coagulopathies, and viral persistence. Additional contributing factors include vascular abnormalities, endothelial dysfunction, autonomic nervous system dysregulation, and neuronal impairments. Together, these findings suggest that Long COVID is a multifactorial condition involving complex, systemic disruptions across immune, vascular, and neurological domains.
A graphic representation of the known pathophysiology of Long COVID by M.Faghy created in BioRender (https://biorender.com/shortURL) is licensed under CC BY 4.0.
Immunological dysfunction
Circulating cytokines
The immune system plays a crucial role in combating acute infections and has been a major focus of research aimed at understanding the pathological mechanisms underlying Long COVID. First, we describe alterations in circulating cytokines observed in patients with Long COVID, before discussing recent evidence suggesting altered immune cell function and autoimmunity.
Inflammatory cytokines and chemokines are dysregulated over a period of 7–14 months in individuals with Long COVID20,21, although significant variability exists between patients. Among these, interleukin-6 (IL-6) has received considerable attention in soluble immune analyses. IL-6 is produced by various cell types, including macrophages, lymphocytes, skeletal muscle, and endothelial cells, and resident cells of the brain22, which plays a major role in the proinflammatory response during the acute phase of infection22. It also serves as a prognostic marker for adverse disease outcomes following acute infection. It can be influenced by comorbidities such as cardiovascular disease and type 2 diabetes mellitus, as well as psychological stress and acute exercise23,24.
Giannitrapani et al.25 demonstrated that abnormally high IL-6 levels during acute infection were associated with an increased risk of developing Long COVID and a decline in mobility after 12 months. Similarly, Son et al.26 reported strong associations between IL-6, tumour necrosis factor-α (TNF-α), and vascular cell adhesion molecule 1 (VCAM-1) at 6 months post-infection with overall Long COVID symptoms. However, IL-6 levels did not correlate with specific symptoms such as fatigue, cough, or dyspnoea up to 12 months after infection. Attempts to identify biochemical risk factors for Long COVID have proposed a cytokine triad consisting of IL-6, interleukin-1 beta (IL-1β), and TNF-α27. Patel et al.28 identified angiopoietin 1 (Ang1) and platelet P-selectin as key vascular biomarkers in Long COVID using machine learning approaches29. Whether these markers, alone or in combination with others, can serve as specific diagnostic tools for Long COVID requires further study.
Altered T-cell recognition of SARS-CoV-2 antigens or broader T-cell dysfunction in Long COVID remains an area of considerable debate30. Antiviral immune responsiveness, including the expression of immune exhaustion markers, is likely linked to the presence of a long-term viral antigen reservoir31,33. One study reported generalised functional exhaustion of CD8 + T cells in Long COVID patients, demonstrated by extremely low levels of CD8+ interferon-gamma (IFN-γ) and TNF-α production in response to nonspecific stimulants, with similar findings observed in ME/CFS34. Significant uncertainties remain regarding whether viral persistence results from insufficient adaptive immunity or if it paradoxically drives excessive adaptive immune responses, as well as the extent to which persistence reflects active viral replication versus latency. Given this complexity, common inflammatory cytokine markers lack specificity for Long COVID. A more detailed understanding of the roles these cytokines play in the pathophysiology of Long COVID is essential to advancing diagnostic and therapeutic strategies.
Immune cell alterations
Recent literature on Long COVID has increasingly focused on alterations in immune cell function and responses to pathogens30,33. However, it remains unclear whether these immune changes are a primary driver of disease pathogenesis, such as through autoimmunity, or a secondary response to other underlying factors like viral persistence, chronic/latent viral reactivation, or microthrombosis, or perhaps a combination of both. Patterson et al.35 report that T-cell activation, marked by elevated IFN-γ and interleukin-2 (IL-2), alongside impaired T-cell recruitment, suggests a dysfunctional antiviral response. Dysregulation of CD4+ and CD8 + T cells in this context may indicate T-cell exhaustion. Su et al.36 observed an expansion of cytotoxic T cells in individuals exhibiting the gastrointestinal Long COVID phenotype 2–3 months after infection. Additionally, stimulation with PMA and ionomycin has been shown to increase intracellular production of IL-2, IL-4, IL-6, IFN-γ, and TNF-α in both CD4+ and CD8 + T cells in people with Long COVID37. However, this pattern was not observed in CD8 + T cells from patients with ME/CFS, where IFN-γ and TNF-α responses were absent34. Evidence also implicates monocytes in Long COVID pathogenesis35,38. For instance, Scott et al.39 identified distinct monocyte signatures, such as expression of prostaglandin-generating enzyme cyclooxygenase-2 (COX-2), interleukin-8 receptor beta (IL-8Rβ, also known as CXCR2), C-X-C chemokine receptor type 6 (CXCR6), and the adhesion molecule P-selectin glycoprotein ligand-1 (PSGL-1), which appear to be associated with fatigue and dyspnoea in Long COVID patients.
Autoimmunity
Epidemiological and immunological studies have linked viral and bacterial infections, including SARS-CoV-2, to an increased risk of autoimmune diseases such as vasculitis, type 1 diabetes, and inflammatory bowel disease30,40,41,42. This risk was most pronounced early in the pandemic (2020–2021), particularly following severe infections with pre-Delta variants and among unvaccinated populations43,44,45. More recent data suggest a reduced risk with Omicron variants and vaccination46. Long COVID may involve autoimmune dysregulation triggered by prolonged immune activation involving B and T cells30,47,48, with latent virus reactivation, especially Epstein-Barr virus (EBV), known to be implicated in multiple sclerosis, potentially contributing49. Although autoimmune diseases arise from genetic and environmental factors50, the role of COVID-19–induced autoimmunity in Long COVID pathophysiology remains unclear.
Autoantibodies (AABs) have been investigated as biomarkers for acute and post-acute COVID-19. Neutralising AABs against type I interferons (IFNs), prevalent in men, older adults, and fatal COVID-19 cases, correlate with disease severity and may persist post-infection51,52,53. However, longitudinal studies indicate limited persistence of these AABs beyond 6–8 months and inconsistent associations with Long COVID symptoms36,54. Some reports link baseline IFN-α2 AABs with respiratory symptoms54, and implicate AABs targeting type III IFN (IFN-λ)55.
Antinuclear antibodies (ANA) and extractable nuclear antigen (ENA) AABs are frequently detected in Long COVID patients up to 12 months post-infection, particularly against SS-B/La and U1-snRNP26. These autoantibodies show variable associations with fatigue, dyspnoea, and neurocognitive symptoms56. Dysregulated AABs targeting G-protein-coupled receptors (GPCRs), involved in autonomic nervous system function, are strongly linked to symptom severity and patterns57,58,59. In Long COVID patients with biopsy-confirmed small fibre neuropathy, 25% exhibited autoantibodies against gangliosides regulating GPCR activity60, although other studies report no persistent AABs in similar cohorts61.
Variability in study design, including cohort selection, timing of sampling, assay methods, and range of AAB targets, likely contributes to inconsistent findings. Supporting this, a conformationally sensitive protein array screening ~20,000 human proteins identified elevated AABs against PITX2 and FBXO2 in Long COVID patients ~14 months post-infection, correlating with palpitations and cognitive disturbances62. Other AABs associated with Long COVID include those targeting thyroid peroxidase, thyroglobulin, IL-2, and CD8B55,63. Notably, some AABs may be protective; for example, anti-chemokine AABs were inversely correlated with Long COVID development at 1 year64. To clarify the predictive role of autoantibodies, longitudinal studies with serial sampling, detailed phenotyping, larger cohorts, and optimised assays are essential. Improved methodologies and replication will aid in identifying patient subgroups likely to benefit from AAB-targeted or immunomodulatory treatments. The detection of autoantibodies linked to autonomic, neurocognitive, immune, mitochondrial, and vascular dysfunction has spurred early trials employing plasmapheresis, intravenous immunoglobulin (IVIg), and other immunomodulators.
Therapeutic apheresis techniques such as INUSpheresis and immunoadsorption, which remove circulating autoantibodies, are under investigation for Long COVID65,66 and have shown promise in ME/CFS67. Biomarker analyses suggest that reductions in neurotransmitter AABs, lipids, inflammatory markers, and fibrinogen correlate with clinical improvement and may help identify likely responders68. These findings emphasise the need for a precision medicine approach that tailors therapies based on predictive clinical and biomarker profiles, especially given a recent randomised trial of plasma exchange showed safety but no efficacy for Long COVID symptoms, quality of life, or function69.
Anti-GPCR antibody therapies are a promising avenue, although their efficacy and safety require further validation. Preliminary reports suggest that intravenous immunoglobulin (IVIg), commonly used to treat autoimmune disorders and thought to act partly through anti-idiotypic antibodies that neutralise autoantigens, autoantibodies, and cytokines, may alleviate symptoms of Long COVID70. Small case series and retrospective studies report symptomatic improvements, including fatigue and cognitive deficits, following IVIg treatment, with ongoing clinical trials currently evaluating its efficacy70,71,72.
Emerging immunomodulators such as BC007, an aptamer neutralising functional GPCR autoantibodies, have shown potential in early trials for improving fatigue and microcirculatory dysfunction in Long COVID subsets73,74. Similarly, low-dose naltrexone (LDN), which modulates immune responses by attenuating microglial activation and proinflammatory cytokine production, possibly via non-opioid pathways including TRPM3 channel regulation, has shown preliminary promise. Retrospective reviews and observational studies report improvements in fatigue, PEM, sleep disturbances, and quality of life among Long COVID patients treated with LDN, warranting further controlled trials incorporating predictive clinical and biomarker criteria75,76,77,78,79. A retrospective review of 59 Long COVID patients treated off-label with LDN reported improvements in fatigue, PEM, unrefreshing and disrupted sleep, and overall functional status80. Additional observational studies have similarly reported symptom improvement, enhanced well-being, and better quality of life in subsets of Long COVID patients77,78,79. Further research is needed to identify clinical and biomarker predictors of treatment response and to incorporate these into the design of randomised controlled trials.
Despite growing evidence, the mechanisms linking SARS-CoV-2 infection to autoimmunity remain incompletely understood. Proposed pathways include immune dysregulation involving inflammation, immune exhaustion, and expansion of naïve B cells producing autoantibodies81; persistent viral antigen exposure; direct activation of autoreactive plasmablasts; molecular mimicry potentially via amyloidogenic cross-seeding82; gut microbiome dysbiosis83; and reactivation of latent herpesviruses, particularly EBV, possibly mediated by epitope homology84. The contribution of Long COVID-associated autoantibodies to cellular and tissue injury across organ systems, including their functional effects, requires further elucidation. Future research should identify specific autoantigen targets and assess homology with microbial and viral proteins, with trials focusing on patient subsets defined by autoantibody profiles and clinical phenotypes to optimise therapeutic outcomes.
Viral persistence/chronic viral reactivation
The persistence of live (chronic productive infection or defective viral persistence) or reactivated viruses (latent infection) is an area of great importance in increasing the pathological understanding of Long COVID. Despite symptomatic resolution, some people are not able to fully clear a virus (Fig. 2), and the continued presence of a virus or parts of a virus is linked to increased antigen detection85. Similarities have been observed with single-stranded DNA viruses, such as Ebola virus, Zika virus, enterovirus, and measles31,86. Research has identified persistent SARS-CoV-2 proteins and microthromboses in tissue, including skeletal muscle, and autopsy studies30,31,87. The gastrointestinal tract, however, has undoubtedly been the most studied site for SARS-CoV-2 reservoirs, and viral antigen profiles have also been found in stool samples post-SARS-CoV-2 infection88, irrespective of a persistent symptom profile89,90,91,92. In other words, individuals vary considerably in their ability to resist both initial and post-infection virions and their products.
Mechanistic insight into the role of viral persistence across different systems and tissues in the context of Long COVID, image published by Chen et al.108.
The evidence and role of viral persistence in the pathophysiology of chronic disease were established long before SARS-CoV-2 and Long COVID93,94. Viral persistence and viral reservoirs can play an important role in Long COVID and, more broadly, chronic disease presentation. Several trials of antiviral medications are ongoing and show promise in reducing the symptom burden of Long COVID. However, combined therapeutic approaches are likely needed to address the broader pathologies of Long COVID.
Proal et al.32, concluded that SARS-CoV-2 ribonucleic acid (RNA) and protein are present in a range of tissue types collected weeks or months after acute COVID-19, including in brain regions, lymph nodes, the thorax, sciatic nerve, ocular tissue, the cervical spinal cord, the brainstem, and the olfactory nerve95. The brain and the brainstem are particularly vulnerable to acute and chronic damage from various sources, particularly viral invasion, inflammation, and vascular activation96. Data from single molecular arrays enable high-sensitivity detection of low-abundance proteins, including SARS-CoV-2 antigens in serum. One year post-infection, ~60% of people with Long COVID demonstrated the presence of SARS-CoV-2 spike proteins and a greater number of organ systems involved in symptoms that were not evident in controls97,98,99. Whole-body positron emission tomography on twenty-four people with Long COVID100 demonstrated increased T-cell activation in several anatomical regions compared to pre-pandemic controls. There was increased uptake in the brainstem, spinal cord, bone marrow, nasopharyngeal and hilar lymphoid tissue, cardiopulmonary tissues, and gut wall. Following in situ hybridisation, SARS-CoV-2 RNA and immunohistochemical studies were completed in a subset of participants with Long COVID symptoms. Elevated cellular SARS-CoV-2 RNA in rectosigmoid lamina propria tissue was observed in all participants, confirming that viral persistence could be associated with long-term immunological perturbations in Long COVID.
Greater consideration of chronic viral reactivation is also warranted, given the growing evidence that supports a potential contributing role in the pathophysiology of Long COVID. Several studies have highlighted the reactivation of latent viruses, including EBV, human herpesviruses (e.g., HHV-6), and cytomegalovirus (CMV), in individuals with post-acute sequelae of SARS-CoV-2 infection. Peluso et al.101 and Maguire et al.102 identified serological and transcriptional signatures consistent with viral reactivation in Long COVID cohorts. Similarly, Vojdani et al.103 and Bernal et al.104 provide immunological and molecular evidence suggesting that reactivation of latent viral reservoirs may contribute to ongoing inflammation, immune dysregulation, and multisystem symptoms observed in Long COVID These findings support the hypothesis that chronic viral reactivation, alongside viral persistence, may be a key driver of sustained pathology in a subset of patients.
While the pathophysiologic evidence of viral persistence/chronic viral reactivation is mounting, further studies are needed into the exact mechanisms in the pathology of Long COVID. Particularly, the observation that previously infected individuals without Long COVID symptoms also have remnants of viral proteins in tissue87 and plasma105 suggests possible additional contributing factors. However, it must be verified utilising more advanced methodologies to better understand the impact that persistent pathogens may be having on host tissue. For instance, spatial transcriptomics is a relatively novel technique that enables the mapping of gene expression within the spatial context of biopsied tissues whilst preserving the intricate architecture and cellular interactions of the sample. In acute SARS-CoV-2 research, this approach has already proven to be invaluable in uncovering the spatial landscape of lung pathology during COVID-19 progression106. In Long COVID, techniques such as spatial transcriptomics can be applied to better quantify and understand which tissues are most likely to host viral reservoirs. These localised immune responses are triggered by persistent pathogens and viral remnants in vulnerable individuals, and how these processes may also reactivate latent pathogens32,107,108. The implementation of advanced techniques such as spatial transcriptomics allows researchers to go beyond the simple identification of persistent pathogens or viral remnants in study participants and utilise preserved spatial context to explore the complex interplay between viral persistence, immune responses, and latent viral reactivation in Long COVID.
Endothelial pathology, platelet hyperactivation, and clotting pathologies
Persistent endothelial cell damage, clotting abnormalities, and increased platelet activation are central pathological processes often linked to viral or spike protein persistence, latent virus reactivation, and immune-related factors in people with Long COVID7,30,33,109,110,111,112. The glycocalyx matrix in the capillary endothelium, which serves as a fluid barrier, can be shed due to heightened inflammatory mediators, resulting in significant alterations in microvascular resistance and capillary blood flow113. SDC-1 has emerged as a reliable marker for assessing glycocalyx injury disease114. This study highlights endothelial damage in convalescent COVID-19 patients with mild disease progression, suggesting that glycocalyx injury can persist even in the absence of severe disease114.
Capillary rarefaction, which refers to the reduction in the number or density of capillaries within a given tissue or organ, leads to impaired microcirculation and reduced oxygen and nutrient delivery. It is commonly observed in conditions associated with chronic inflammation, endothelial dysfunction, and vascular abnormalities, such as hypertension and diabetes. Capillary rarefaction can result from capillary degeneration, impaired angiogenesis, or structural remodelling of the microvasculature. It has also been observed in Long COVID patients via sublingual video microscopy115, further indicating endothelial dysfunction or damage. Endothelial cells may also exhibit apoptotic behaviour long after infection, disrupting communication between endothelial cells and vascular smooth muscle cells113, further contributing to disease pathology. Moreover, elevated levels of endothelin 1 (ET-1) have been detected in Long COVID patients five months after acute infection, in contrast to those who had recovered from COVID-19 and healthy individuals110. These increased ET-1 levels likely reflect vascular injury and the activation of the intrinsic coagulation pathway116.
Various pathologies leading to endothelial damage promote platelet adhesion and coagulation, impairing organ function and perpetuating platelet hyperactivation through upregulated expression of inflammatory and adhesion molecules117. Endothelial damage leads to the release of intracellular contents, including von Willebrand factor (vWF) and Factor VIII (FVIII), into the circulation118, contributing to the formation of anomalous fibrin(ogen) amyloid deposits or microclots (Fig. 3), which in turn contribute to tissue hypoxia and further injury119, as well as multiple other observables82,119,120. Microthrombi have been identified in post-mortem studies of COVID-19 patients121 and individuals with Long COVID have anomalous microclots in circulation112,122. The persistent endothelial inflammation, the presence of anomalous (insoluble) microclot deposits, platelet hyperactivation, and their association with chronic Long COVID symptoms are well-documented7,30,123. These studies underscore the role of microthrombogenesis in Long COVID. Anomalous deposits, like other amyloid proteins, exhibit greater resistance to fibrinolysis and have also been detected in skeletal tissue87, suggesting they could contribute to Long COVID pathophysiology82,119,122,123. Recent evidence during repeated submaximal exercise demonstrates fragmentation of microclots, which is linked to increased inflammation and impaired oxygen transport in people with Long COVID124. These factors can provide a plausible explanation for diverse tissue-specific dysfunctions observed in Long COVID.
Multiple factors contribute to endothelial dysfunction and damage in Long COVID, with (1) viral persistence being a key driver of endothelial injury. (2) Endothelial damage leads to (3) the release of intracellular components, such as vWF and FVIII, into circulation, which in turn contributes to the formation of anomalous microclot deposits (7). Elevated levels of ET-1 (4) and SDC-1 (5) serve as markers of vascular and glycocalyx injury, respectively. Platelet hyperactivation is a well-established feature of Long COVID, with endothelial damage further promoting platelet adhesion (6) and coagulation. Additionally, the finding of capillary rarefaction in Long COVID may also indicate vascular damage. (8) Research has shown that the spike protein of SARS-CoV-2 directly induces the formation of anomalous fibrin(ogen) deposits in purified soluble fibrinogen (fibrinaloids)204. Spike protein can also induce anomalous deposits in plasma from healthy individuals. The extent of amyloidogenicity might be related to virulence205. It is therefore evident that the formation of these deposits is on the disease pathway. By A. Kruger created in BioRender (https://biorender.com/shortURL) is licensed under CC BY 4.0.
Mechanistic insights into the most common symptoms of long COVID
Next, we discuss the primary symptoms of Long COVID and deepen the discussion of the pathophysiology of each symptom, linking it to the proposed underlying causes of Long COVID described above. Since most research has focused on postural orthostatic tachycardia syndrome (POTS), PEM/PESE, respiratory abnormalities, cognitive dysfunction and sleep disturbances, we decided to focus on these primary symptoms but acknowledge that this list is not exclusive and that there is a broad range of symptoms that vastly impact the lives of people with Long COVID.
Neurological manifestations of long COVID
Dysautonomia and POTS
Orthostatic intolerance, including POTS and orthostatic hypotension, is reported in around half of people with Long COVID125. There is a plausible link between damage to the autonomic nervous system and Long COVID symptoms, but the exact pathophysiology remains unknown. Orthostatic intolerance is a hallmark feature of Long COVID dysautonomia. Upon standing, there is an instantaneous shift of ~0.5–1 L of blood to the capacitance vessels in the lower extremities and splanchnic circulation126,127. An additional 10-25% of the plasma volume is driven out of the vasculature into the interstitial space127,128. A well-functioning autonomic nervous system detects these hemodynamic changes. It invokes immediate auto-compensatory mechanisms, resulting in peripheral vasoconstriction and an increase in systemic vascular resistance to maintain venous return to the heart127,129. However, in Long COVID, it is thought that the autonomic nervous system may not detect and respond to gravitational changes correctly, resulting in the pooling of venous blood in the lower limbs when standing up, which may lead to an exaggerated increase in heart rate to maintain cardiac output. The ability of anomalous microclots to induce hypoxia provides a plausible mechanistic explanation for POTS. What the heart may be doing is detecting the hypoxia and having a compensatory increase in the heart rate120.
POTS is likely underpinned by multiple aetiologies that converge to produce a similar clinical phenotype, often overlapping with other well-defined syndromes such as ME/CFS and post-treatment Lyme disease130. A primary reduction in circulating plasma volume (hypovolemic POTS) will lead to reflex sympathetic vasoconstriction to maintain blood pressure at the cost of an increased heart rate, which has been reported in ~70% of people with Long COVID131. Therapeutic strategies that aim to increase the circulating plasma volume, such as increased salt and water intake and the use of desmopressin, can improve symptoms. A patchy denervation of the small nerve fibres to the blood vessels in the extremities has been observed in up to 20%-40% of people with Long COVID (neuropathic POTS)125. This small fibre neuropathy may result from damage through a viral or autoimmune insult132,133. Consequently, an abnormal sympathetic tone reduces the ability to vasoconstrict, causing compensatory tachycardia.
Abnormal sweating, bladder control problems, gastric issues, skin discolouration, increased venous pooling in the legs, as well as syncope and palpitations are commonly observed. Many people with POTS describe “adrenaline surges” and are found to have elevated levels of circulating plasma norepinephrine (Hyperadrenergic POTS). Norepinephrine levels measured during orthostasis may be up to four times higher in POTS patients compared to healthy controls133. Some people have also been identified as having a genetic loss-of-function mutation of the norepinephrine transporter, which is responsible for norepinephrine clearance, increasing circulating levels of norepinephrine at rest and during standing up. It might explain why many people feel tremulous and anxious in response to innocuous stimuli. The underlying factors that contribute to POTS in Long COVID are currently poorly understood.
A dysfunctional autonomous nervous system (dysautonomia), disordered tissue perfusion, endothelial inflammation and dysfunction and/or chronic inflammation might all contribute. The endothelial glycocalyx may be damaged, increasing the endothelial permeability113, and causing plasma to leak into the interstitial fluid134,135; however, more research is required to test this hypothesis. Additionally, the autonomic nervous system dysfunction likely contributes to the development of POTS. Indeed, the autonomic nervous system dysfunction has been implicated in the pathophysiology of Long COVID more generally. People with Long COVID showed significant diaphragmatic dysfunction and reductions in maximum inspiratory pressure, implicating the phrenic nerve in respiratory muscle weakness136,137. Damage to endothelial cells surrounding the vagus nerve, which carries sympathetic and parasympathetic fibres of the autonomic nervous system, and/or inflammatory immune cell infiltration into the vagus nerve, might affect vagal nerve activity and contribute to dysautonomia138,139,140. A recent post-mortem study detected SARS-CoV-2 remnants in the vagus nerve, suggesting that viral infiltration has the potential to affect nerve function139. Direct evidence of such a hypothesis is lacking and requires more detailed studies.
A link between inflammation and dysautonomia in Long COVID has been hypothesised. The inflammatory vagus nerve reflex is integral in regulating inflammation homeostasis via a cholinergic neural reflex mechanism. This inflammatory reflex controls innate immune responses and inflammation during pathogen invasion and tissue injury141. Efferent vagal fibres from the dorsal motor nucleus inhibit cytokine storms via cholinergic pathways142. Therapeutic agents targeting the efferent vagal nerve fibres, therefore, may not only help relieve disabling symptoms of dysautonomia but also be potentially disease-modifying by modulating chronic inflammation in people with Long COVID. Whether treatments targeting the vagus nerve will help with reducing dysautonomia, POTS, and other Long COVID-related symptoms represents an important area of future study.
Cognitive dysfunction and brain fog
Many of the neurological and neuropsychiatric symptoms seen in individuals with Long COVID appear to involve neuroinflammatory cascades triggered by systemic inflammation. This inflammation may contribute to the disruption of the blood-brain barrier (BBB) or enable direct viral neuroinvasion, particularly via infection of the olfactory bulb (Fig. 4). Immune system dysregulation, in conjunction with systemic vascular impairment, has been observed in individuals experiencing COVID-related cognitive dysfunction. These findings suggest that BBB leakage and post-COVID cognitive dysfunction may be interrelated143. In addition, the accumulation of viral antigens and microglial dysfunction has been identified alongside significant vascular inflammation in the brains of deceased COVID-19 patients144.
Humans and macaques develop similar viral diseases and share common histopathological markers, such as (1) infection of neurons and glial cells, detected by colocalization with different viral markers (SARS-CoV-2 spike and nucleocapsid proteins, double-stranded RNA). (2) Disruption of the blood-brain barrier (BBB), with viral presence associated with vascular abnormalities. (3) astrocyte-associated neuroinflammatory response. By D. Beckman, created in BioRender (https://biorender.com/shortURL) is licensed under CC BY 4.0.
Whilst aged individuals are particularly vulnerable to COVID-19 complications, it is widely acknowledged that younger patients can also be adversely affected, including through long-term neurological symptoms. Early in the pandemic, some studies detected neuroinflammatory responses without evidence of active viral presence in the brain145. However, later research identified persistent SARS-CoV-2 RNA and confirmed viral neuroinvasion in human, monkey, and mouse brains95,146,147,148. The neuroinvasive potential of coronaviruses had already been established in previous strains following airway exposure, with common entry routes including the olfactory nerve, retrograde transport via cranial nerves, or infiltration through the bloodstream or infected immune cells149,150,151,152. SARS-CoV-2 spike and nucleocapsid proteins, as well as viral RNA, have been found in brain tissue and across the skull–meninges–brain axis from days to even years after acute infection95,146,153. Persistent viral presence in brain tissue may contribute to prolonged neurological symptoms in Long COVID, potentially through mechanisms such as impaired serotonin transmission154. Notably, both cerebral organoids and post-mortem brain tissue from individuals with COVID-19 have shown that SARS-CoV-2 can disrupt synaptic homeostasis and increase the expression of synaptic proteins155,156.
In summary, current evidence suggests that neurological complications following COVID-19 may arise from systemic inflammation leading to neuroinflammation, and in some cases, from direct viral infection and its inflammatory consequences. Clarifying the pathways of viral entry and the mechanisms of cellular damage will be crucial for understanding and treating Long COVID’s neurological outcomes.
Respiratory symptoms
Pulmonary abnormalities are frequently reported in patients with Long COVID157 and may arise from chronic airway inflammation, pulmonary endothelialitis, fibrosis158, restrictive parenchymal physiology159, dysfunctional breathing (e.g., due to dysautonomia), chest tightness, or dyspnoea158. Whether these changes reflect a core feature of Long COVID or result from broader systemic pathophysiology remains uncertain. Impaired lung function may also be driven by hemodynamic disturbances in pulmonary circulation, leading to reduced alveolar gas exchange160,161. Dhawan et al.162 identified signs of pulmonary small-vessel disease and parenchymal involvement. Furthermore, paired expiratory axial CT scans often reveal widespread lobular and regional low-attenuation areas indicative of air trapping, a contributor to ventilation/perfusion (V̇/Q) mismatch and reduced exercise tolerance. Patients presenting with persistent respiratory symptoms may benefit from advanced imaging modalities, such as CT pulmonary angiography with dual-energy sequencing, which can support both diagnosis and mechanistic understanding163. These techniques could help characterise small-vessel perfusion abnormalities and clarify the long-term pulmonary vascular sequelae of COVID-19 and Long COVID162.
Fatigue and PEM
Chronic fatigue is one of the most prevalent and debilitating symptoms reported by individuals with Long COVID, characterised by an inability to perform physical or cognitive tasks that would typically fall within their normal energy capacity. Approximately 50%-80% of patients also experience PEM, a hallmark feature involving the worsening or emergence of new symptoms following physical, mental, or cognitive exertion that exceeds a highly individual and variable threshold164 (Fig. 5). The onset of PEM typically occurs within 48 hours of exertion and may persist for days, weeks, or even months165. There is substantial overlap between the clinical presentation and proposed pathophysiological mechanisms of Long COVID and ME/CFS, in which PEM is a core diagnostic criteria166. Given the strong association between exertion and symptom exacerbation, many patients identify PEM as a primary contributor to their reduced functional capacity and diminished quality of life. As such, patients often remain physically inactive to avoid PEM. Interventions such as graded exercise therapy, designed to increase activity levels over time, have been linked to adverse outcomes in this population and are considered high-risk for inducing PEM167. Despite these data on risks of PEM/PESE, the role and appropriateness of physical activity and exercise training remain widely debated for people with Long COVID and ME/CFS164,168.
A graphic summary of the known causes and contributors to PEM/PESE in people with Long COVID.
The exact pathophysiological mechanisms underlying PEM remain unclear. However, current evidence suggests that the mechanisms responsible for reduced exercise capacity and excessive fatigue in Long COVID differ from those driving PEM specifically87. Individuals with Long COVID consistently exhibit lower maximal oxygen uptake and peak power output during cycle ergometer testing compared to age- and sex-matched controls87,169. These deficits are likely attributable to peripheral impairments rather than significant pulmonary or cardiac dysfunction. Structural and morphological changes in skeletal muscle appear to play a significant role in this reduced exercise tolerance and persistent fatigue87,170. Recent literature has identified five key mechanisms contributing to skeletal muscle dysfunction in Long COVID: local hypoxia (including impaired oxygen delivery and potential reactive oxygen species production), physical deconditioning, electrophysiological abnormalities, autoimmune processes, and central fatigue (see ref. 123 for more details).
Skeletal muscle changes have also been associated with the earlier onset of fatigue, with some alterations potentially resulting from repeated episodes of PEM87. Additionally, capillary abnormalities, endothelial dysfunction, and microclot formation/fragmentation may impair oxygen delivery, although the exact mechanisms behind endothelial dysfunction in Long COVID remain unknown. Muscle fibres in individuals with Long COVID tend to be more glycolytic and exhibit reduced mitochondrial respiration171. Biomarkers indicate decreased mitochondrial content, biogenesis, and function, including diminished cytochrome c oxidase activity, subsarcolemmal mitochondrial accumulation, and disrupted cristae architecture171,172. These mitochondrial impairments contribute to the accelerated development of fatigue. Moreover, abnormalities in fatty acid oxidation during exercise have been reported173,174, along with exaggerated blood lactate accumulation. While the cause of this lactate buildup remains unclear, it may reflect a very low ventilatory threshold in some patients and is consistent with a pattern of chronic ischaemia-reperfusion injury119.
Mitochondrial dysfunction may play a central role in the pathophysiology of Long COVID, given its widespread impact on cellular and systemic function175. SARS-CoV-2 has been shown to bind to mitochondria and utilise ATP to support viral replication176. Nasopharyngeal samples from individuals with acute SARS-CoV-2 infection reveal impaired transcription of both nuclear and mitochondrial genes, which may trigger an antiviral immune response while simultaneously disrupting mitochondrial function177. The Wiskott-Aldrich Syndrome Protein Family Member 3 (WASF3) has been implicated in the disrupted formation of mitochondrial respiratory supercomplexes and impaired respiration in ME/CFS. It is also associated with endoplasmic reticulum stress in skeletal muscle178. However, the precise role of WASF3 or other mitochondrial abnormalities in contributing to fatigue and exercise intolerance in Long COVID remains unclear. Developing pharmacological treatments aimed at restoring mitochondrial function may hold promise for alleviating fatigue and preventing PEM. However, such therapies must target the specific mitochondrial pathologies involved, and further research is needed to guide these interventions. Notably, acute exertion beyond the threshold that triggers PEM initiates a cascade of symptoms, the origins of which remain unknown. This cascade is not restricted to skeletal muscle involvement, as mental or cognitive exertion and even emotional stress can also precipitate PEM or PESE. It suggests that a blood-borne or neurologically mediated factor may initiate PEM, although more research is required to identify the underlying mechanisms.
PEM may be partly explained by cellular damage caused by reactive oxygen species (ROS) in the context of ischaemia-reperfusion injury119. Local skeletal muscle changes following the onset of PEM include further deterioration in mitochondrial respiration, compounding the mitochondrial abnormalities already observed in individuals with Long COVID87. Evidence of exercise-induced muscle damage has been reported in muscle biopsies both before172 and following maximal exercise in people with Long COVID87. Additionally, immune-mediated structural changes in the skeletal muscle microvasculature may contribute to exercise-induced fatigue and muscle pain179. As skeletal muscle pain is often triggered by local inflammation and the release of cytokines by infiltrating immune cells, these mechanisms may help explain the pain experienced after exertion that exceeds the PEM threshold175,176,177,178.
Currently, there are no established cures or pharmacological treatments for PEM. Therefore, effective symptom management strategies, such as pacing, are essential. While there is emerging evidence supporting the role of physical therapy in managing PEM (REF), there remains no compelling evidence across post-acute infectious syndromes that graded exercise therapy (GET) is effective in restoring pre-infection levels of function180. Prior studies supporting GET have been subject to extensive critique181,182,183,184, due to issues such as vague inclusion criteria, high drop-out rates, unclear exercise prescriptions, and failure to measure PEM-related outcomes. Until further research is available that can outline the biomechanistic causes of PEM, the World Health Organisation185, Centres for Disease Control186, National Institute for Health and Care Excellence187,188, and World Physiotherapy189 Guidelines explicitly do not recommend graded exercise therapy as a treatment for people with Long COVID.
In the absence of effective medications to raise the PEM threshold or shorten its duration, the most widely used strategy for managing PEM is pacing. Pacing involves encouraging individuals to remain as active as possible without exceeding their PEM threshold190, by organising daily activities within their established functional limits191. This strategy aims to prevent and reduce the frequency and severity of PEM episodes. To be effective, pacing strategies must be highly individualised, accounting for the fluctuating and episodic nature of Long COVID. Incorporating objective biophysical monitoring tools, such as blood lactate levels192, heart rate, and heart rate variability, can support the development of personalised pacing plans193. These physiological markers can be integrated into daily routines and offer near real-time feedback194, helping individuals remain within their energy envelope and avoid triggering PEM. While pacing does not provide a cure, it offers a practical approach to improving functional status, quality of life, and fatigue management in the absence of pharmacological treatments. However, despite its widespread use, the efficacy of pacing as a management strategy for PEM has not yet been rigorously studied, highlighting an urgent need for further research.
Sleep disturbances in long COVID
People with acute COVID-19, particularly those who experienced more severe symptoms during the initial infection, and those with Long COVID, frequently report excessive daytime sleepiness, low mood, and a diminished quality of life195. Hospitalisation for COVID-19 is associated with a significantly increased risk of breathlessness and sleep disorders compared to non-hospitalised individuals. In a study involving 2,320 participants, those who were not hospitalised showed a higher risk of circadian rhythm sleep disorders196. Mediation analysis indicated that anxiety accounted for 18–39% of the observed effects, while muscle weakness contributed 27–41%. One of the most comprehensive multinational studies on sleep quality in COVID-19 survivors identified at least six separate domains of sleep disturbance and confirmed that the severity of the initial infection was an independent predictor of poor sleep outcomes197. Despite these findings, there remains a critical need to better understand the underlying pathology of self-reported sleep disturbances and insomnia in Long COVID.
Objective assessments, such as polysomnography, should be more widely utilised. These have shown a marked reduction in slow-wave sleep and the presence of both sleep-onset and sleep-maintenance insomnia, often characterised by prolonged periods of wakefulness after sleep onset198. Of particular concern is the presence of REM sleep behaviour disorder (RBD), a parasomnia involving dream-enactment behaviours due to the loss of REM sleep atonia199. RBD and REM sleep without atonia are both associated with alpha-synucleinopathies and are known risk factors for developing neurodegenerative diseases such as Parkinson’s disease200. A prospective cohort study of individuals with Long COVID reported excessive daytime sleepiness, including central hypersomnolence and new-onset narcolepsy, in 3.2% of participants. This study also noted a signal suggesting an increased risk of central hypersomnolence syndromes and narcolepsy in this population201. The importance of sleep is well documented, but research on Long COVID is currently still in its infancy. Still, the impact of sleepiness in people with Long COVID on physical and mental well-being, functional status, and quality of life should be better studied. Whilst the importance of sleep for health is well-established, research into sleep disturbances in Long COVID remains in its early stages. Further investigation is urgently needed to better understand how sleep dysfunction affects physical and mental well-being, functional capacity, and overall quality of life in people with Long COVID.
Guidance for clinical practice
Long COVID continues to represent a significant unmet clinical need and is contributing to rising healthcare costs on a global scale. While advancing our understanding of the underlying pathophysiology remains central to addressing this condition, the development of precision or personalised medicine approaches is unlikely to progress meaningfully without increased research efforts aimed at uncovering the biological mechanisms of this novel disease. In parallel with deeper pathological insight, there is an urgent need to expand access to diagnostic testing that can support clinical decision-making in Long COVID. Where symptom profiles and underlying pathologies overlap with other conditions, there is potential to repurpose existing licensed medications, an approach that should be evaluated at scale.
Effective symptom management strategies must increasingly leverage wearable technologies capable of tracking biomarkers such as heart rate variability (HRV). These tools, when combined with artificial intelligence and machine learning, can deliver real-time, objective feedback to both patients and clinicians. Where feasible, integrating this physiological data with biological insights will allow for the refinement of treatment strategies and ultimately improve patient outcomes.
Moving long COVID research forward
While international and collaborative research efforts have advanced the pathophysiological understanding of Long COVID, many areas still require further investigation before effective management and curative treatments can be integrated into standard clinical care. Although numerous multicentre trials have enhanced our pathological insights. Notably, in the United States, NIH RECOVER-TLC (Researching COVID to Enhance Recovery-Treating Long COVID), an ongoing programme sponsored by the National Institute of Allergy and Infectious Diseases, which aims to identify and pilot the most promising treatment avenues for specific Long COVID subsets by fostering integration of data from pathobiological studies with clinical data from observational cohorts cohorts202. However, there remains a significant gap in mechanistic understanding, which has hindered the development of clear diagnostic criteria applicable to clinical practice and service delivery pathways203.
Until the key mechanisms of Long COVID are elucidated and curative therapies developed, individuals living with this episodic disability must have access to high-quality, evidence-based, condition-specific interventions aimed at alleviating and managing their symptoms. To support these global efforts, sustained commitment and investment from governments, policymakers, and research funding bodies are essential to prevent the emergence of substantial public health and socioeconomic burdens. Accordingly, in Table 1, we propose key research priorities necessary for advancing mechanistic knowledge, which can inform the development of condition-specific diagnostic approaches and care pathways that incorporate effective treatment and management strategies.
Conclusion
Long COVID represents a significant and complex global health challenge, characterised by a wide spectrum of persistent symptoms driven by multifactorial pathophysiological mechanisms. Although research has advanced considerably, critical gaps remain in identifying definitive diagnostic biomarkers, fully understanding disease mechanisms, and developing effective treatments. The intricate interplay between immune dysregulation, endothelial dysfunction, viral persistence, and coagulation abnormalities underscores the necessity for interdisciplinary research to unravel the underlying pathways of Long COVID. Future research must prioritise large-scale, rigorously controlled studies to establish pathology-specific biomarker profiles, refine diagnostic criteria, and develop targeted therapeutic interventions. Addressing the heterogeneity of Long COVID through precision medicine approaches will be crucial for improving patient stratification and optimising treatment strategies. Moreover, sustained investment and international collaboration are essential to alleviate the long-term health and socioeconomic impacts of this condition. Until curative therapies emerge, emphasis on evidence-based symptom management, early diagnosis, and patient-centred care remains vital to enhancing quality of life and reducing disease burden. As Long COVID research stands at a critical crossroads, bridging these knowledge gaps will not only revolutionise care for those affected but also provide valuable insights into other post-viral syndromes and chronic inflammatory diseases.
References
Worldometer. COVID - Coronavirus Statistics - Worldometer. https://www.worldometers.info/coronavirus/.
Msemburi, W. et al. The WHO estimates of excess mortality associated with the COVID-19 pandemic. Nature 613, 130–137 (2023).
Thomas, C. et al. Lived experience of patients with Long COVID: a qualitative study in the UK. BMJ Open 13, e068481 (2023).
Owen, R. et al. Acute COVID-19, the Lived Experience, and Lessons to Learn for Future Pandemics. Disaster Med. Public Health Prep. 17, e534 (2023).
Kogevinas, M. et al. Risk, determinants, and persistence of long-COVID in a population-based cohort study in Catalonia. BMC Med. 23, 140 (2025). In a prospective population-based cohort of 2,764 SARS-CoV-2-infected adults from Catalonia, Risk, determinants, and persistence of long-COVID in a population-based cohort study in Catalonia found that 23% developed long-COVID symptoms between 2021–2023, more than half of whom still had symptoms after two years, and that risk was significantly elevated in women, younger individuals, those with severe acute infection, obesity, asthma/COPD or mental-health disorders, while prior vaccination.
Ma, Y. et al. Long-term consequences of asymptomatic SARS-CoV-2 infection: a systematic review and meta-analysis. Int. J. Environ. Res. Public. Health 20, 1613 (2023).
Davis, H. E., McCorkell, L., Vogel, J. M. & Topol, E. J. Long COVID: major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 21, 133–146 (2023).
Al-Aly, Z. & Topol, E. Solving the puzzle of long Covid. Science 383, 830–832 (2024).
Callard, F. & Perego, E. How and why patients made Long Covid. Soc. Sci. Med. 268, 113426 (2021).
Greenhalgh, T., Sivan, M., Perlowski, A. & Nikolich, J. Ž. Long COVID: a clinical update. Lancet 404, 707-724 (2024).
Organisation, W. H. A clinical case definition of post COVID-19 condition by a Delphi consensus, 6 October 2021. https://www.who.int/publications/i/item/WHO-2019-nCoV-Post_COVID-19_condition-Clinical_case_definition-2021.1 (2021).
Kim, Y., Bae, S., Chang, H.-H. & Kim, S.-W. Long COVID prevalence and impact on quality of life 2 years after acute COVID-19. Sci. Rep. 13, 11207 (2023). Seventy-one percent of participants reported at least one long-COVID symptom at 24 months post-infection, and around one-third continued to experience reduced neuropsychiatric quality of life despite improvement over time, highlighting the long-term and ongoing impact of even mild acute COVID‑19 on survivors’ daily living.
Dehlia, A. & Guthridge, M. A. The persistence of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) after SARS-CoV-2 infection: a systematic review and meta-analysis. J. Infect. 89, 106297 (2024).
Hornig, M. et al. Distinct plasma immune signatures in ME/CFS are present early in the course of illness. Sci. Adv. 1, e1400121 (2015).
Thomas, C. et al. Blood biomarkers of long COVID: a systematic review. Mol. Diagn. Ther. 28, 537–574 (2024).
Liew, F. et al. Large-scale phenotyping of patients with long COVID post-hospitalization reveals mechanistic subtypes of disease. Nat. Immunol. 25, 607–621 (2024).
Al-Aly, Z. et al. Long COVID science, research and policy | Nature Medicine. https://www.nature.com/articles/s41591-024-03173-6 (2024).
Faghy, M. A. et al. Is Long COVID the next global health crisis? J. Glob. Health (In Press).
Faghy, M. A. et al. COVID-19 patients require multi-disciplinary rehabilitation approaches to address persisting symptom profiles and restore pre-COVID quality of life. Expert Rev. Respir. Med. 16, 595–600 (2022).
Zhang, S. et al. SARS-CoV-2 binds platelet ACE2 to enhance thrombosis in COVID-19. J. Hematol. Oncol. J. Hematol. Oncol. 13, 120 (2020).
Klein, J. et al. Distinguishing features of Long COVID identified through immune profiling. Nature 623, 139–148 (2023). Individuals with long COVID exhibit distinct immune-profile signatures, such as altered myeloid and lymphocyte populations, elevated SARS-CoV-2 and reactivated Epstein–Barr virus antibody responses, and reduced cortisol levels, which together may serve as biomarkers and provide insight into the pathobiology of the condition.
Montazersaheb, S. et al. COVID-19 infection: an overview on cytokine storm and related interventions. Virol. J. 19, 92 (2022).
Del Giudice, M. & Gangestad, S. W. Rethinking IL-6 and CRP: Why they are more than inflammatory biomarkers, and why it matters. Brain. Behav. Immun. 70, 61–75 (2018).
Friedman, E. M., Hayney, M., Love, G. D., Singer, B. H. & Ryff, C. D. Plasma interleukin-6 and soluble IL-6 receptors are associated with psychological well-being in aging women. Health Psychol. 26, 305 (2007).
Giannitrapani, L. et al. Can baseline IL-6 levels predict long COVID in subjects hospitalized for SARS-CoV-2 disease?. Int. J. Mol. Sci. 24, 1731 (2023).
Son, K. et al. Circulating anti-nuclear autoantibodies in COVID-19 survivors predict long COVID symptoms. Eur. Respir. J. 61, 2200970 (2023).
Schultheiß, C. et al. The IL-1β, IL-6, and TNF cytokine triad is associated with post-acute sequelae of COVID-19. Cell Rep. Med. 3, 100663 (2022).
Patel, M. A. et al. Elevated vascular transformation blood biomarkers in Long-COVID indicate angiogenesis as a key pathophysiological mechanism. Mol. Med. 28, 122 (2022).
Patel, M. A. et al. Organ and cell-specific biomarkers of Long-COVID identified with targeted proteomics and machine learning. Mol. Med. 29, 26 (2023).
Altmann, D. M., Whettlock, E. M., Liu, S., Arachchillage, D. J. & Boyton, R. J. The immunology of long COVID. Nat. Rev. Immunol. 1, 17 (2023).
Proal, A. D. et al. SARS-CoV-2 reservoir in post-acute sequelae of COVID-19 (PASC). Nat. Immunol. 1–12 (2023). Evidence that SARS-CoV-2 RNA and/or proteins persist in tissues long after acute infection, proposes that such viral reservoirs may drive chronic symptoms via immune, coagulation and microbiome dysregulation, and underscores the importance of targeting viral persistence in the search for therapies for Long COVID.
Proal, A. D. et al. Targeting the SARS-CoV-2 reservoir in long COVID. Lancet Infect. Dis. 25, e294–e306 (2025).
Proal, A. D. & VanElzakker, M. B. Long COVID or post-acute sequelae of COVID-19 (PASC): an overview of biological factors that may contribute to persistent symptoms. Front. Microbiol. 12, 1494 (2021).
Gil, A. et al. Identification of CD8 T-cell dysfunction associated with symptoms in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and Long COVID and treatment with a nebulized antioxidant/anti-pathogen agent in a retrospective case series. Brain Behav. Immun. Health 36, 100720 (2024).
Patterson, B. K. et al. Persistence of SARS CoV-2 S1 protein in CD16+ monocytes in post-acute sequelae of COVID-19 (PASC) up to 15 months post-infection. Front. Immunol. 12, 5526 (2022).
Su, Y. et al. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell 185, 881–895 (2022).
Santopaolo, M. et al. Prolonged T-cell activation and long COVID symptoms independently associate with severe COVID-19 at 3 months. Elife 12, e85009 (2023).
Patterson, B. K. et al. Immune-based prediction of COVID-19 severity and chronicity decoded using machine learning. Front. Immunol. 12, 2520 (2021).
Scott, N. A. et al. Monocyte migration profiles define disease severity in acute COVID-19 and unique features of long COVID. Eur. Respir. J. 61, 2202226 (2023).
Fujinami, R. S. Viruses and autoimmune disease–two sides of the same coin?. TRENDS Microbiol. 9, 377–381 (2001).
Sfriso, P. et al. Infections and autoimmunity: the multifaceted relationship. J. Leukoc. Biol. 87, 385–395 (2010).
Sharma, C. & Bayry, J. High risk of autoimmune diseases after COVID-19. Nat. Rev. Rheumatol. 1–2 https://doi.org/10.1038/s41584-023-00964-y (2023).
Wuller, S. et al. Severity of acute SARS-CoV-2 infection and risk of new-onset autoimmune disease: A RECOVER initiative study in nationwide U.S. cohorts. PloS One 20, e0324513 (2025). Individuals who experienced more severe acute SARS‑CoV‑2 infection had a significantly higher risk of developing new autoimmune diseases, such as thyroid disease, psoriasis/psoriatic arthritis, inflammatory bowel disease, and (in children) type 1 diabetes, within up to two years post‑infection, highlighting the severity of initial COVID‑19 illness as a potential predictor and modifiable risk factor for autoimmune sequelae.
Wee, L. E. et al. Autoimmune sequelae after delta or omicron variant SARS-CoV-2 infection in a highly vaccinated cohort. JAMA Netw. Open 7, e2430983–e2430983 (2024).
Chang, R. et al. Risk of autoimmune diseases in patients with COVID-19: a retrospective cohort study. EClinicalMedicine 56, (2023).
Hileman, C. O., Malakooti, S. K., Patil, N., Singer, N. G. & McComsey, G. A. New-onset autoimmune disease after COVID-19. Front. Immunol. 15, (2024).
Dotan, A. & Shoenfeld, Y. Chapter 13 - Autoimmunity on the Rise: COVID-19 as a Trigger of Autoimmunity. in Infection and Autoimmunity (Third Edition) (eds Mahroum, N., Watad, A. & Shoenfeld, Y.) 247–257 (Academic Press, Amsterdam, 2024). https://doi.org/10.1016/B978-0-323-99130-8.00030-1.
Wekerle, H. & Hohlfeld, R. Molecular mimicry in multiple sclerosis. N. Engl. J. Med. 349, 185–185 (2003).
Talwar, S., Harker, J. A., Openshaw, P. J. M. & Thwaites, R. S. Autoimmunity in long COVID. J. Allergy Clin. Immunol. 155, 1082–1094 (2025).
Weinstock, L. B. et al. Mast cell activation symptoms are prevalent in Long-COVID. Int. J. Infect. Dis. 112, 217–226 (2021).
Bastard, P. et al. Human autoantibodies neutralizing type I IFNs: From 1981 to 2023. Immunol. Rev. 322, 98–112 (2024).
Bastard, P. et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science 370, eabd4585 (2020).
Mouton, W. et al. Immunological and Clinical Markers of Post-acute Sequelae of COVID-19: Insights from Mild and Severe Cases 6 Months Post-infection. Eur. J. Immunol. 55, e51948 (2025). Whilst most humoral, viral, and tissue‑damage biomarkers at 6–9 months after acute COVID‑19 were driven by initial illness severity rather than the presence of post‑acute sequelae (PASC), individuals with PASC exhibited distinctive T‑cell signatures, specifically higher cytokine‑secreting CD4⁺/CD8⁺ memory T cells and increased terminally differentiated CD45RA⁺ TEMRA cells responsive to SARS‑CoV‑2 nucleocapsid/membrane proteins, suggesting a potential immunological marker set for long‑term symptom persistence.
Jernbom, A. F. et al. Prevalent and persistent new-onset autoantibodies in mild to severe COVID-19. Nat. Commun. 15, 8941 (2024).
Rojas, M. et al. Autoimmunity is a hallmark of post-COVID syndrome. J. Transl. Med. 20, 129 (2022).
Seeßle, J. et al. Persistent Symptoms in Adult Patients 1 Year After Coronavirus Disease 2019 (COVID-19): a Prospective Cohort Study. Clin. Infect. Dis. 74, 1191–1198 (2022).
Sotzny, F. et al. Dysregulated autoantibodies targeting vaso- and immunoregulatory receptors in Post COVID Syndrome correlate with symptom severity. Front. Immunol. 13, (2022). Individuals with post‑COVID syndrome (PCS) exhibited altered serum levels of autoantibodies against vaso‑ and immunoregulatory G‑protein‑coupled receptors (GPCRs). Notably including those targeting ADRB2, STAB1 and ADRA2A, with several of these autoantibody levels correlating with symptom severity (e.g., fatigue and vasomotor symptoms) and thus implicating immune‑ and vascular‑dysregulation pathways as potential contributors to PCS.
Hofmann, S. et al. Functional Autoantibodies Targeting G-Protein-Coupled Receptors and Their Clinical Phenotype in Patients with Long-COVID. Int. J. Mol. Sci. 26, 6746 (2025).
Wallukat, G. et al. Functional autoantibodies against G-protein coupled receptors in patients with persistent Long-COVID-19 symptoms. J. Transl. Autoimmun. 4, 100100 (2021).
Maguire, C. et al. Analysis of 977 Long COVID Patients Reveals Prevalent Neuropathy and Association with Anti-Ganglioside Antibodies. MedRxiv Prepr. Serv. Health Sci. 2025.03.04.25323101 https://doi.org/10.1101/2025.03.04.25323101 (2025).
Gavrilova, N. Y. et al. Autoantigens of Small Nerve Fibers and Human Coronavirus Antigens: Is There a Possibility for Molecular Mimicry?. Curr. Microbiol. 81, 366 (2024).
Hatayama, Y. et al. Identification of Putative Serum Autoantibodies Associated with Post-Acute Sequelae of COVID-19 via Comprehensive Protein Array Analysis. Int. J. Mol. Sci. 26, 1751 (2025).
Matula, Z. et al. High prevalence of long COVID in anti-TPO positive euthyroid individuals with strongly elevated SARS-CoV-2-specific T cell responses and moderately raised anti-spike IgG levels 23 months post-infection. Front. Immunol. 15, 1448659 (2024).
Muri, J. et al. Autoantibodies against chemokines post-SARS-CoV-2 infection correlate with disease course. Nat. Immunol. 24, 604–611 (2023).
Scheibenbogen, C. et al. Immunoadsorption to remove ß2 adrenergic receptor antibodies in chronic fatigue syndrome CFS/ME. PLoS One 13, e0193672 (2018).
Stein, E. et al. Efficacy of repeated immunoadsorption in patients with post-COVID myalgic encephalomyelitis/chronic fatigue syndrome and elevated β2-adrenergic receptor autoantibodies: a prospective cohort study. Lancet Reg. Heal. 49, 101161 (2025).
Dorst, J. Apheresis in neurological disorders. J. Clin. Med. 9, 3211 (2020).
Achleitner, M. et al. Clinical improvement of Long-COVID is associated with reduction in autoantibodies, lipids, and inflammation following therapeutic apheresis. Mol. Psychiatry 28, 2872–2877 (2023).
España-Cueto, S. et al. Plasma exchange therapy for the post COVID-19 condition: a phase II, double-blind, placebo-controlled, randomized trial. Nat. Commun. 16, 1929 (2025).
Morse, B. A., Motovilov, K., Michael Brode, W., Michael Tee, F. & Melamed, E. A review of intravenous immunoglobulin in the treatment of neuroimmune conditions, acute COVID-19 infection, and post-acute sequelae of COVID-19 Syndrome. Brain. Behav. Immun. 123, 725–738 (2025).
Thompson, J. S., Thornton, A. C., Ainger, T. & Garvy, B. A. Long-term high-dose immunoglobulin successfully treats Long COVID patients with pulmonary, neurologic, and cardiologic symptoms. Front. Immunol. 13, 1033651 (2022).
Hogeweg, M. et al. Intravenous immunoglobulins in the treatment of post-COVID: a case-control study. J. Intern. Med. 293, 656–658 (2023).
Hohberger, B. et al. Case report: neutralization of autoantibodies targeting G-protein-coupled receptors improves capillary impairment and fatigue symptoms after COVID-19 infection. Front. Med. 8, 754667 (2021).
Hohberger, B. et al. Safety, tolerability and clinical effects of rovunaptabin, also known as BC007 on fatigue and quality of life in patients with Post-COVID syndrome (reCOVer): a prospective, exploratory, placebo-controlled, double-blind, randomised phase IIa clinical trial (RCT). EClinicalMedicine 86, 103358 (2025).
Bonilla, H. et al. Therapeutic trials for long COVID-19: a call to action from the interventions taskforce of the RECOVER initiative. Front. Immunol. 14, 1129459 (2023).
Li, Z., You, Y., Griffin, N., Feng, J. & Shan, F. Low-dose naltrexone (LDN): A promising treatment in immune-related diseases and cancer therapy. Int. Immunopharmacol. 61, 178–184 (2018).
Tamariz, L., Bast, E., Klimas, N. & Palacio, A. Low-dose naltrexone improves post-COVID-19 condition symptoms. Clin. Ther. 46, e101–e106 (2024).
Isman, A. et al. Low-dose naltrexone and NAD+ for the treatment of patients with persistent fatigue symptoms after COVID-19. Brain Behav. Immun. Health 36, 100733 (2024).
O’Kelly, B. et al. Safety and efficacy of low dose naltrexone in a long covid cohort; an interventional pre-post study. Brain Behav. Immun. Health 24, 100485 (2022).
Bonilla, H. et al. Low-dose naltrexone use for the management of post-acute sequelae of COVID-19. Int. Immunopharmacol. 124, 110966 (2023).
Acosta-Ampudia, Y. et al. Persistent autoimmune activation and proinflammatory state in post-Coronavirus disease 2019 syndrome. J. Infect. Dis. 225, 2155–2162 (2022).
Kell, D. B. & Pretorius, E. Are fibrinaloid microclots a cause of autoimmunity in long Covid and other post-infection diseases?. Biochem. J. 480, 1217–1240 (2023).
Hromić-Jahjefendić, A. et al. The intersection of microbiome and autoimmunity in long COVID-19: current insights and future directions. Cytokine Growth Factor Rev. 82, 43–54 (2025).
Adiguzel, Y., Bogdanos, D. P. & Shoenfeld, Y. Molecular/antigenic mimicry and immunological cross-reactivity explains SARS-CoV-2-induced autoimmunity. Autoimmun. Rev. 24, 103811 (2025).
Peluso, M. J. et al. Plasma-based antigen persistence in the post-acute phase of COVID-19. Lancet Infect. Dis. 24, e345–e347 (2024). SARS‑CoV‑2 antigens (notably spike and nucleocapsid) were detectable in plasma up to 14 months after acute infection at significantly higher rates than in pre‑pandemic controls. Providing compelling evidence for persistent viral antigenemia in the post‑acute phase of COVID‑19 and underscoring a potential mechanism for ongoing symptomatology in long COVID.
Choutka, J., Jansari, V., Hornig, M. & Iwasaki, A. Unexplained post-acute infection syndromes. Nat. Med. 28, 911–923 (2022).
Appelman, B. et al. Muscle abnormalities worsen after post-exertional malaise in long COVID. Nat. Commun. 15, 17 (2024). Patients with Long COVID showing post‑exertional malaise (PEM) exhibited marked skeletal‑muscle abnormalities, including increased glycolytic fibre composition, reduced mitochondrial oxidative‑phosphorylation capacity, amyloid‑containing deposit accumulation and exercise‑induced myopathy. These abnormalities worsened following an exercise challenge, thereby offering direct mechanistic insight into how PEM may contribute to impaired functional capacity in Long COVID.
Hawley, H. B. Long COVID: clinical findings, pathology, and endothelial molecular mechanisms. Am. J. Med. https://www.sciencedirect.com/science/article/pii/S0002934323005399 (2023).
Wu, Y. et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol. Hepatol. 5, 434–435 (2020).
Cheung, C. C. L. et al. Residual SARS-CoV-2 viral antigens detected in GI and hepatic tissues from five recovered patients with COVID-19. Gut 71, 226–229 (2021).
Gaebler, C. et al. Evolution of antibody immunity to SARS-CoV-2. Nature 591, 639–644 (2021).
Natarajan, A. et al. Gastrointestinal symptoms and fecal shedding of SARS-CoV-2 RNA suggest prolonged gastrointestinal infection. Med 3, 371–387.e9 (2022).
Scott, J. T. et al. Post-Ebola syndrome, Sierra Leone. Emerg. Infect. Dis. 22, 641 (2016).
Dokubo, E. K. et al. Persistence of Ebola virus after the end of widespread transmission in Liberia: an outbreak report. Lancet Infect. Dis. 18, 1015–1024 (2018).
Stein, S. R. et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature 612, 758–763 (2022).
Yong, S. J. Persistent brainstem dysfunction in long-COVID: a hypothesis. ACS Chem. Neurosci. 12, 573–580 (2021).
Swank, Z. et al. Persistent circulating severe acute respiratory syndrome coronavirus 2 spike is associated with post-acute coronavirus disease 2019 sequelae. Clin. Infect. Dis. 76, e487–e490 (2023).
Peluso, M. J. et al. Plasma-based antigen persistence in the post-acute phase of SARS-CoV-2 infection. medRxiv 2023–10 (2023).
Craddock, V. et al. Persistent circulation of soluble and extracellular vesicle-linked Spike protein in individuals with postacute sequelae of COVID-19. J. Med. Virol. 95, e28568 (2023).
Peluso, M. J. et al. Multimodal molecular imaging reveals tissue-based T cell activation and viral RNA persistence for up to 2 years following COVID-19. medRxiv https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10418298/ (2023).
Peluso, M. J. et al. Chronic viral coinfections differentially affect the likelihood of developing long COVID. J. Clin. Invest. 133, e163669 (2023).
Maguire, C. et al. Chronic viral reactivation and associated host immune response and clinical outcomes in acute COVID-19 and post-acute sequelae of COVID-19. bioRxiv https://doi.org/10.1101/2024.11.14.622799 (2024).
Vojdani, A., Vojdani, E., Saidara, E. & Maes, M. Persistent SARS-CoV-2 infection, EBV, HHV-6 and other factors may contribute to inflammation and autoimmunity in long COVID. Viruses 15, 400 (2023).
Bernal, K. D. E. & Whitehurst, C. B. Incidence of Epstein-Barr virus reactivation is elevated in COVID-19 patients. Virus Res. 334, 199157 (2023).
Swank, Z. et al. Measurement of circulating viral antigens post-SARS-CoV-2 infection in a multicohort study. Clin. Microbiol. Infect. 30, 1599–1605 (2024).
Rendeiro, A. F. et al. The spatial landscape of lung pathology during COVID-19 progression. Nature 593, 564–569 (2021).
Cross, A. R. et al. Spatial transcriptomic characterization of COVID-19 pneumonitis identifies immune circuits related to tissue injury. JCI Insight 8, e157837 (2023).
Chen, B., Julg, B., Mohandas, S. & Bradfute, S. B. Viral persistence, reactivation, and mechanisms of long COVID. Elife 12, e86015 (2023).
Kruger, A. et al. Vascular pathogenesis in acute and long COVID: current insights and therapeutic outlook. Semin. Thromb. Hemost. 51, 256–271 (2024).
Haffke, M. et al. Endothelial dysfunction and altered endothelial biomarkers in patients with post-COVID-19 syndrome and chronic fatigue syndrome (ME/CFS). J. Transl. Med. 20, 138 (2022).
Charfeddine, S. et al. Long COVID 19 syndrome: is it related to microcirculation and endothelial dysfunction? Insights from TUN-EndCOV study. Front. Cardiovasc. Med. 8, 1702 (2021).
Kruger, A. et al. Proteomics of fibrin amyloid microclots in long COVID/post-acute sequelae of COVID-19 (PASC) shows many entrapped pro-inflammatory molecules that may also contribute to a failed fibrinolytic system. Cardiovasc. Diabetol. 21, 190 (2022).
Jarrott, B., Head, R., Pringle, K. G., Lumbers, E. R. & Martin, J. H. “LONG COVID”—a hypothesis for understanding the biological basis and pharmacological treatment strategy. Pharmacol. Res. Perspect. 10, e00911 (2022).
Vollenberg, R. et al. Indications of persistent glycocalyx damage in convalescent COVID-19 patients: a prospective multicenter study and hypothesis. Viruses 13, 2324 (2021).
Osiaevi, I. et al. Persistent capillary rarefication in long COVID syndrome. Angiogenesis 26, 53–61 (2023).
Willems, L. H. et al. Sustained inflammation, coagulation activation and elevated endothelin-1 levels without macrovascular dysfunction at 3 months after COVID-19. Thromb. Res. 209, 106–114 (2022).
Wang, C. et al. Long COVID: the nature of thrombotic sequelae determines the necessity of early anticoagulation. Front. Cell. Infect. Microbiol. 12, 861703 (2022).
Hawley, H. B. & Chang, J. C. Complement-induced endotheliopathy–associated vascular microthrombosis in coronavirus disease 2019. J. Infect. Dis. 223, 2198–2199 (2021).
Kell, D. B. & Pretorius, E. The potential role of ischaemia–reperfusion injury in chronic, relapsing diseases such as rheumatoid arthritis, long COVID, and ME/CFS: evidence, mechanisms, and therapeutic implications. Biochem. J. 479, 1653–1708 (2022).
Kell, D. B., Khan, M. A., Kane, B., Lip, G. Y. H. & Pretorius, E. Possible role of fibrinaloid microclots in postural orthostatic tachycardia syndrome (POTS): focus on long COVID. J. Pers. Med. 14, 170 (2024).
Fahmy, O. H. et al. Is microthrombosis the main pathology in coronavirus disease 2019 severity?—a systematic review of the postmortem pathologic findings. Crit. Care Explor. 3, e0427 (2021).
Pretorius, E. et al. Persistent clotting protein pathology in long COVID/post-acute sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovasc. Diabetol. 20, 172 (2021).
Kell, D. B., Laubscher, G. J. & Pretorius, E. A central role for amyloid fibrin microclots in long COVID/PASC: origins and therapeutic implications. Biochem. J. 479, 537–559 (2022).
Thomas, C. et al. Exercise-induced changes in microclotting and cytokine levels point to vascular injury and inflammation in people with long COVID. https://www.researchsquare.com/article/rs-6717727/latest (2025).
Lee, C. et al. Prevalence of orthostatic intolerance in long covid clinic patients and healthy volunteers: a multicenter study. J. Med. Virol. 96, e29486 (2024).
Smith, J. J., Porth, C. M. & Erickson, M. Hemodynamic response to the upright posture. J. Clin. Pharmacol. 34, 375–386 (1994).
Arnold, A. C., Ng, J. & Raj, S. R. Postural tachycardia syndrome–diagnosis, physiology, and prognosis. Auton. Neurosci. 215, 3–11 (2018).
Raj, S. R. et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation 111, 1574–1582 (2005).
Raj, S. R. The postural tachycardia syndrome (POTS): pathophysiology, diagnosis & management. Indian Pacing Electrophysiol. J. 6, 84 (2006).
Vernino, S. et al. Postural orthostatic tachycardia syndrome (POTS): state of the science and clinical care from a 2019 National Institutes of Health Expert Consensus Meeting-Part 1. Auton. Neurosci. 235, 102828 (2021).
Sheldon, R. S. et al. 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm 12, e41–e63 (2015).
Levine, T. D. Small fiber neuropathy: disease classification beyond pain and burning. J. Cent. Nerv. Syst. Dis. 10, 117957351877170 (2018).
Raj, S. R. Postural tachycardia syndrome (POTS). Circulation 127, 2336–2342 (2013).
Pons, S., Fodil, S., Azoulay, E. & Zafrani, L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit. Care 24, 353 (2020).
Chelazzi, C., Villa, G., Mancinelli, P., De Gaudio, A. R. & Adembri, C. Glycocalyx and sepsis-induced alterations in vascular permeability. Crit. Care 19, 26 (2015).
Finsterer, J. Re:‘Vagus nerve dysfunction in the post-COVID-19 condition’by Lladós et al. Clin. Microbiol. Infect. 30, 969–970 (2024).
Pietranis, K. A., Kostro, A. M., Dzięcioł-Anikiej, Z., Moskal-Jasińska, D. & Kuryliszyn-Moskal, A. Impact of COVID-19 on diaphragmatic function: understanding multiorgan involvement and long-term consequences. J. Clin. Med. 13, 6493 (2024).
Goldstein, D. S. Linking the extended autonomic system with the homeostat theory: new perspectives about dysautonomias. J. Pers. Med. 14, 123 (2024).
Woo, M. S. et al. Vagus nerve inflammation contributes to dysautonomia in COVID-19. Acta Neuropathol. 146, 387–394 (2023).
Andersson, U. & Tracey, K. J. Vagus nerve SARS-CoV-2 infection and inflammatory reflex dysfunction: is there a causal relationship?. J. Intern. Med. 295, 91–102 (2024).
Andersson, J. The inflammatory reflex – introduction. J. Intern. Med. 257, 122–125 (2005).
De Virgiliis, F. & Di Giovanni, S. Lung innervation in the eye of a cytokine storm: neuroimmune interactions and COVID-19. Nat. Rev. Neurol. 16, 645–652 (2020).
Greene, C. et al. Blood-brain barrier disruption and sustained systemic inflammation in individuals with long COVID-associated cognitive impairment. Nat. Neurosci. 27, 421–432 (2024). Individuals with long COVID who report cognitive impairment show persistent disruption of the blood-brain barrier (BBB), alongside sustained systemic inflammation and coagulation dysregulation, suggesting BBB dysfunction may be a key mechanistic underpinning of neurocognitive sequelae in Long COVID.
Fekete, R. et al. Microglia dysfunction, neurovascular inflammation and focal neuropathologies are linked to IL-1-and IL-6-related systemic inflammation in COVID-19. Nat. Neurosci. 28, 558–576 (2025).
Matschke, J. et al. Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. Lancet Neurol. 19, 919–929 (2020).
Beckman, D. et al. SARS-CoV-2 infects neurons and induces neuroinflammation in a non-human primate model of COVID-19. Cell Rep. 41, 111573 (2022).
Crunfli, F. et al. Morphological, cellular, and molecular basis of brain infection in COVID-19 patients. Proc. Natl. Acad. Sci. USA 119, e2200960119 (2022).
Song, E. et al. Neuroinvasion of SARS-CoV-2 in human and mouse brain. J. Exp. Med. 218, e20202135 (2021).
Dubé, M. et al. Axonal transport enables neuron-to-neuron propagation of human coronavirus. J. Virol. 92, OC43 (2018).
Jacomy, H. & Talbot, P. J. Vacuolating encephalitis in mice infected by human coronavirus OC43. Virology 315, 20–33 (2003).
Matsuda, K. et al. The vagus nerve is one route of transneural invasion for intranasally inoculated influenza A virus in mice. Vet. Pathol. 41, 101–107 (2004).
Guo Yong, G. Y., Korteweg, C., McNutt, M. A. & Gu Jiang, G. J. Pathogenetic mechanisms of severe acute respiratory syndrome. Virus Res. 133, 4–12 (2008).
Rong, Z. et al. Persistence of spike protein at the skull-meninges-brain axis may contribute to the neurological sequelae of COVID-19. Cell Host Microbe 32, 2112–2130.e10 (2024).
Wong, A. C. et al. Serotonin reduction in post-acute sequelae of viral infection. Cell 186, 4851–4867.e20 (2023).
Gelbard, H. A. & Chiang, W. SARS and synapses. Nat. Microbiol. 9, 1163–1164 (2024).
Partiot, E. et al. Brain exposure to SARS-CoV-2 virions perturbs synaptic homeostasis. Nat. Microbiol. 9, 1189–1206 (2024).
Tanaka, N. et al. Air trapping at CT: high prevalence in asymptomatic subjects with normal pulmonary function. Radiology 227, 776–785 (2003).
Cho, J. L. et al. Quantitative chest CT assessment of small airways disease in post-acute SARS-CoV-2 infection. Radiology 304, 185–192 (2022).
Guler, S. A. et al. Pulmonary function and radiological features 4 months after COVID-19: first results from the national prospective observational Swiss COVID-19 lung study. Eur. Respir. J. 57, 2003690 (2021).
Kavanagh, B. P. & Hedenstierna, G. Respiratory physiology and pathophysiology. Mill. Anesth. 8, 444–472 (2015).
Wild, J. M. et al. Review of hyperpolarized pulmonary functional 129Xe MR for long-COVID. J. Magn. Reson. Imaging 59, 1120–1134 (2024).
Dhawan, R. T. et al. Beyond the clot: perfusion imaging of the pulmonary vasculature after COVID-19. Lancet Respir. Med. 9, 107–116 (2021).
Screaton, N. Commentary: pulmonary vasculopathy in post-acute-COVID-19 syndrome—insights from dual-energy CT angiography. Eur. Radiol. 33, 4698–4699 (2023).
Thaweethai, T. et al. Development of a definition of postacute sequelae of SARS-CoV-2 infection. JAMA 329, 1934–1946 (2023).
Vøllestad, N. K. & Mengshoel, A. M. Post-exertional malaise in daily life and experimental exercise models in patients with myalgic encephalomyelitis/chronic fatigue syndrome. Front. Physiol. 14, 1257557 (2023).
Medicine, I. of, Populations, B. on the H. of S. & Syndrome, C. on the D. C. for M. E. F. Beyond myalgic encephalomyelitis/chronic fatigue syndrome: redefining an illness. (National Academies Press, 2015).
Eckey, M., Li, P., Morrison, B., Davis, R. W. & Xiao, W. Patient-reported treatment outcomes in ME/CFS and long COVID. medRxiv 2024–11 (2024).
Walitt, B. et al. Deep phenotyping of post-infectious myalgic encephalomyelitis/chronic fatigue syndrome. Nat. Commun. 15, 907 (2024).
Thomas, C. et al. Submaximal 2-day cardiopulmonary exercise testing to assess exercise capacity and post-exertional symptom exacerbation in people with long COVID. Exp. Physiol. https://doi.org/10.1113/EP092415 (2025).
Charlton, B. T. et al. Skeletal muscle adaptations and post-exertional malaise in long COVID. Trends Endocrinol. Metab. 36, 614–622 (2024).
Colosio, M. et al. Structural and functional impairments of skeletal muscle in patients with postacute sequelae of SARS-CoV-2 infection. J. Appl. Physiol. 135, 902–917 (2023).
Hejbøl, E. K. et al. Myopathy as a cause of fatigue in long-term post-COVID-19 symptoms: evidence of skeletal muscle histopathology. Eur. J. Neurol. 29, 2832–2841 (2022).
de Boer, E. et al. Decreased fatty acid oxidation and altered lactate production during exercise in patients with post-acute COVID-19 syndrome. Am. J. Respir. Crit. Care Med. 205, 126–129.
Guntur, V. P. et al. Signatures of mitochondrial dysfunction and impaired fatty acid metabolism in plasma of patients with post-acute sequelae of COVID-19 (PASC). Metabolites 12, 1026 (2022).
Faghy, M. A. et al The physiologic benefits of optimizing cardiorespiratory fitness and physical activity–From the cell to systems level in a post-pandemic world. Prog. Cardiovasc. Dis. https://www.sciencedirect.com/science/article/pii/S0033062024000288?casa_token=TURJGk_Ov4MAAAAA:VTYZ3re_MZ7h0jhAEj3FBFCRZ3knHbpiKs76YseTCDa-4ac0x9RY_29i7o1NQEre5u7X8UE (2024).
Díaz-Resendiz, K. J. G. et al. Loss of mitochondrial membrane potential (Δ Ψ m) in leucocytes as post-COVID-19 sequelae. J. Leukoc. Biol. 112, 23–29 (2022).
Guarnieri, J. W. et al. Core mitochondrial genes are down-regulated during SARS-CoV-2 infection of rodent and human hosts. Sci. Transl. Med. 15, eabq1533 (2023).
Wang, P. et al. WASF3 disrupts mitochondrial respiration and may mediate exercise intolerance in myalgic encephalomyelitis/chronic fatigue syndrome. Proc. Natl. Acad. Sci. 120, e2302738120 (2023).
Aschman, T. et al. Post-COVID exercise intolerance is associated with capillary alterations and immune dysregulations in skeletal muscles. Acta Neuropathol. Commun. 11, 193 (2023).
van Rhijn-Brouwer, F. C. C.-C. et al. Graded exercise therapy should not be recommended for patients with post-exertional malaise. Nat. Rev. Cardiol. 21, 430–431 (2024).
Guide, C. T. & Goldstein, J. David Tuller responds to the PACE investigators. http://www.cfstreatmentguide.com/blog/category/pace%20trial/2 (2015).
Racaniello, V. et al. Major breakthrough on PACE trial. https://citizen-network.org/news/major-breakthrough-on-pace-trial.
Matthees, A., Kindlon, T., Maryhew, C., Stark, P. & Levin, B. No ‘Recovery’ in PACE trial, new analysis finds. https://virology.ws/2016/09/21/no-recovery-in-pace-trial-new-analysis-finds/ (2016).
Leurent, B. expert reaction to results from REGAIN trial looking at an online supervised group physical and mental health rehabilitation programme for adults with long Covid after Covid hospitalisation | Science Media Centre. https://www.sciencemediacentre.org/expert-reaction-to-results-from-regain-trial-looking-at-an-online-supervised-group-physical-and-mental-health-rehabilitation-programme-for-adults-with-long-covid-after-covid-hospitalisation/ (2024).
Organization, W. H. Clinical Management of COVID-19: Living Guideline, 13 January 2023. https://apps.who.int/iris/bitstream/handle/10665/365580/WHO-2019-nCoV-clinical-2023.1-eng.pdf (2023).
Davenport, T. E., Stevens, S. R., VanNess, J. M., Stevens, J. & Snell, C. R. Checking our blind spots: current status of research evidence summaries in ME/CFS. Br. J. Sports Med. 53, 1198–1198 (2019).
Torjesen, I. NICE cautions against using graded exercise therapy for patients recovering from COVID-19. BMJ 370, m2912 (2020).
Vink, M. & Vink-Niese, A. The updated NICE guidance exposed the serious flaws in CBT and graded exercise therapy trials for ME/CFS. Healthcare 10, 898 (2022).
World Physiotherapy. World Physiotherapy Response to COVID-19 Briefing Paper 9. Safe rehabilitation approaches for people living with Long COVID: physical activity and exercise. https://www.google.com/search?q=world+physio+guidelines+graded+exercise+therapy&sca_esv=7035ff6bfa45f4a8&rlz=1C1ONGR_enGB1012GB1012&sxsrf=ADLYWILBVImkHiI5HsE_-1J20A9KurnvMw%3A1722510955017&ei=a26rZpJk6baFsg-u7qCQBQ&oq=world+physio+guidelines+graded+exercise+th&gs_lp=Egxnd3Mtd2l6LXNlcnAiKndvcmxkIHBoeXNpbyBndWlkZWxpbmVzIGdyYWRlZCBleGVyY2lzZSB0aCoCCAAyBRAhGKABMgUQIRigATIFECEYoAFIwjFQ9AhY-SpwAngBkAEAmAGCAaAB9giqAQQxMC4yuAEDyAEA-AEBmAIOoAKjCcICChAAGLADGNYEGEfCAgcQIRigARgKwgIEECEYFZgDAIgGAZAGCJIHBDEwLjSgB5cq&sclient=gws-wiz-serp (2021).
Goudsmit, E. M., Nijs, J., Jason, L. A. & Wallman, K. E. Pacing as a strategy to improve energy management in myalgic encephalomyelitis/chronic fatigue syndrome: a consensus document. Disabil. Rehabil. 34, 1140–1147 (2012).
Jason, L. A., Evans, M., So, S., Scott, J. & Brown, A. Problems in defining post-exertional malaise. J. Prev. Interv. Community 43, 20–31 (2015).
Faghy, P. M. A., Ashton, D. R. E., McNelis, M. R., Arena, R. & Duncan, D. R. Attenuating post-exertional malaise in Myalgic encephalomyelitis/chronic fatigue syndrome and long-COVID: is blood lactate monitoring the answer?. Curr. Probl. Cardiol. 49, 102554 (2024).
Mooren, F. C. et al. Autonomic dysregulation in long-term patients suffering from post-COVID-19 Syndrome assessed by heart rate variability. Sci. Rep. 13, 15814 (2023).
Faghy, M. A. et al. Developing effective strategies to optimize physical activity and cardiorespiratory fitness in the long Covid population: the need for caution and objective assessment. Prog. Cardiovasc. Dis. https://www.sciencedirect.com/science/article/pii/S0033062024000392 (2024).
Sarkanen, T. et al. Association between hypersomnolence and the COVID-19 pandemic: the International COVID-19 Sleep Study (ICOSS). Sleep. Med. 107, 108–115 (2023).
Jackson, C. et al. Effects of sleep disturbance on dyspnoea and impaired lung function following hospital admission due to COVID-19 in the UK: a prospective multicentre cohort study. Lancet Respir. Med. https://www.thelancet.com/journals/lanres/article/PIIS2213-2600(23)00124-8/fulltext?s=09 (2023).
Alzueta, E. et al. An international study of post-COVID sleep health. Sleep. Health 8, 684–690 (2022).
Rouen, A. et al. Polysomnographic parameters in long-COVID chronic insomnia patients. Dialogues Clin. Neurosci. 25, 43–49 (2023).
Gagliano, A. et al. Artificial neural networks analysis of polysomnographic and clinical features in Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS): from sleep alteration to “Brain Fog”. Nat. Sci. Sleep 13, 1209–1224 (2021).
Gómez-Benito, M. et al. Modeling Parkinson’s disease with the alpha-synuclein protein. Front. Pharmacol. 11, 356 (2020).
Moura, A. E. F. et al. Central hypersomnia and chronic insomnia: expanding the spectrum of sleep disorders in long COVID syndrome - a prospective cohort study. BMC Neurol. 22, 417 (2022).
Breen, J. J. et al. Navigating the path forward for evidence-based management of long COVID. Nat. Immunol. 26, 536–539 (2025).
Ely, E. W., Brown, L. M. & Fineberg, H. V. Long Covid defined. N. Engl. J. Med. 391, 1746–1753 (2024).
Grobbelaar, L. M. et al. SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19. Biosci. Rep. 41, BSR20210611 (2021).
Grobbelaar, L. M. et al. Relative Hypercoagulopathy of the SARS-CoV-2 Beta and Delta Variants when Compared to the Less Severe Omicron Variants Is Related to TEG Parameters, the Extent of Fibrin Amyloid Microclots, and the Severity of Clinical Illness. Semin. Thromb. Hemost. 48, 858–868 (2022).
Acknowledgements
The authors would like to thank Dr. Amy Proal and Professor Doug Kell for their support in developing and reviewing sections throughout this manuscript. The open access charges for this manuscript were covered by generous donations of the VU Foundation and the Stichting Long COVID Nederland.
Author information
Authors and Affiliations
Contributions
M.F. conceived the idea for the manuscript. All authors were involved in the writing of sections related to their expertise and speciality. All authors reviewed and approved the manuscript before submission and supported the revision process.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Ronald Ellis and Al Ozonoff for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Faghy, M.A., Wüst, R.C.I., Altmann, D.M. et al. Current status and future perspectives on the mechanistic and pathophysiological understanding of long COVID. Commun Med 6, 255 (2026). https://doi.org/10.1038/s43856-025-01300-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01300-z