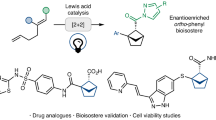
Abstract
Bioisosteric replacement of aromatic and heteroaromatic rings with bridged bicyclic hydrocarbons is an important strategy in drug discovery. Intramolecular [2+2] cycloadditions of unconjugated dienes can provide a route to such motifs but are governed by the ‘rule-of-five’, which dictates that five-membered rings are preferentially formed, limiting access to alternative ring sizes. Here we introduce a visible-light-mediated intramolecular [2 + 2] cycloaddition of aza-1,6-dienes that leverages radical stabilization strategies to enable the selective formation of bridged bicycles over typically favoured fused bicycles. This approach generates previously elusive 6-azabicyclo[3.1.1]heptanes with facile substitution at every position around the ring. Exit vector analysis and comparison of the physicochemical and pharmacological properties of a 6-azabicyclo[3.1.1]heptane analogue of a piperazine-based drug demonstrate the potential application of this scaffold in medicinal chemistry. The methodology enables access to new chemical space, with implications for drug discovery and beyond.

Similar content being viewed by others
Main
Developing synthetic methodologies to construct complex, rigid, three-dimensional molecular architectures is a major focus in contemporary organic synthesis. This is because C(sp3)-rich polycyclic scaffolds have found widespread application in medicinal chemistry as replacements for planar aromatic rings, which can provide drug candidates with improved physicochemical and pharmacokinetic properties1,2,3. For example, structurally rigid bicyclo[n.1.1]alkanes have emerged as effective bioisosteres of benzene rings, with numerous reports of the use of bicyclo[1.1.1]pentane (BCP)3,4,5,6,7,8,9,10,11,12, bicyclo[2.1.1]hexane (BCHex)3,8,9,12,13 and bicyclo[3.1.1]heptane (BCHep)9,12,13,14 groups (Fig. 1a (A.1)). However, analogous isosteric mimetics of heteroaromatic compounds remain underexplored, despite the prevalence of nitrogen heterocycles in Food and Drug Administration-approved small-molecule drugs15,16,17. A notable study by Mykhailiuk and coworkers demonstrated that replacing the 3,5-disubstituted pyridine ring in the antihistamine rupatidine with a 3-azabicyclo[3.1.1]heptane (3-N-BCHep) improved solubility, lipophilicity and metabolic stability, underscoring the potential of this motif in medicinal chemistry18 (Fig. 1a (A.2)). Since then, substituted N-BCHep scaffolds have gained attention as bioisosteres of nitrogen heterocycles, with several studies focusing on 2-N-BCHep19,20,21,22,23,24 and 3-N-BCHep18,20,25,26,27,28,29,30,31 derivatives (Fig. 1a (A.2)). By contrast, there are limited examples of the use of substituted 6-N-BCHeps32,33,34, wherein the nitrogen atom is incorporated into the four-membered ring of the bicycle (Fig. 1a (A.3)). This modification reduces the basicity of the nitrogen and strengthens the adjacent C–H bonds33,35,36, which could offer distinct electronic properties and improved metabolic stability compared with other N-BCHep isomers. Furthermore, comparison of various geometric descriptors, including exit vectors, revealed good similarities between the 6-N-BCHeps and their aromatic counterparts (Fig. 1b). However, access to substituted 6-N-BCHeps is limited by a lack of general methodologies for their synthesis32, so there are scant data concerning their suitability as bioisosteres of nitrogen heterocycles33,34,37.
a, Three-dimensional (3D) bioisosteres of substituted (hetero)aromatic rings. b, Exit vector analyses of N-heterocycles. D2-3, distance between carbon atoms 2 and 3; θ, angle between C2–C3 atoms and H1–C2; φ, angle between H4–C3 and H1–C2. c, Selectivity in intramolecular aza-Paternò–Büchi reactions39,47,48. d, DFT calculations: breaking the rule-of-five. Fsp3, fraction of sp3-hybridized carbon atoms; Ts, p-toluenesulfonyl; TS, transition state; k, rate constant.
The most convergent route to substituted 2-N-BCHep and 3-N-BCHep derivatives is through intermolecular [3 + 2] cycloadditions of bridgehead-substituted bicyclo[1.1.0]butanes with nitrogen-containing 1,3-dipoles19,20,21,22,23,24,25,26,27,28,31 (Fig. 1a (A.2)). However, this method is unsuitable for the synthesis of 6-N-BCHeps because it requires replacement of the bicyclo[1.1.0]butanes with 2-azabicyclo[1.1.0]butanes, which have not previously been isolated due to their instability38. An alternative approach to azabicyclo[n.1.1]alkanes is intramolecular [2 + 2] photocycloadditions of acyclic unconjugated aza-dienes (aza-Paternò–Büchi) using energy transfer (EnT) photocatalysis. This method has been successfully applied to the synthesis of 5-azabicyclo[2.1.1]hexane (5-N-BCHex) rings from aza-1,5-dienes39 (Fig. 1c, top), but there are no reports of analogous reactions of aza-1,6-dienes providing 6-N-BCHeps. Intramolecular [2 + 2] photocycloadditions of unconjugated dienes are generally constrained by an empirical rule known as the ‘rule-of-five’, which dictates that the 1,2-biradical formed upon photoexcitation of the substrate undergoes kinetically favoured 5-exo-trig cyclization to form a five-membered ring40,41,42. This results in the generation of bridged BCHex products from 1,5-dienes, whereas 1,6-dienes selectively cyclize to give fused five-membered ring systems instead of bridged six-membered BCHeps. Although a limited number of reports in which geometric restrictions on the 1,6-diene substrates can bias the selectivity towards the BCHep scaffold43,44,45,46, no general methods for selectivity control or analogous syntheses of 6-N-BCHeps exist. Indeed, Schindler and coworkers observed that the intramolecular [2 + 2] photocycloadditions of aza-1,6-dienes (hex-5-enal oximes) afforded fused azetidine products, consistent with the rule-of-five47 (Fig. 1c, middle). Nonetheless, we recognized that using readily accessible aza-1,6-dienes as substrates in [2 + 2] cycloadditions could provide an efficient strategy for constructing multisubstituted 6-N-BCHeps and could also enable access to previously elusive substituted 3,6-diazabicyclo[3.1.1]heptane (3,6-N2-BCHep) and 3-oxa-6-azabicyclo[3.1.1]heptane (3-O-6-N-BCHep) products through simple substrate modification. Therefore, we sought to design an intramolecular photocycloaddition that defies the rule-of-five.
Achieving selective 6-N-BCHep formation in [2 + 2] photocycloadditions of aza-1,6-dienes 1 requires the alkene 1,2-biradical in 1* to react with the imine via 6-exo-trig cyclization at C1 instead of 5-exo-trig cyclization at C2 (Fig. 1c, bottom). This is challenging due to the disparity in rates of ring formation, with 5-exo-trig cyclization of non-stabilized alkyl radicals onto hydrazones reported to be more than 100 times faster than 6-exo-trig cyclization48 (Fig. 1c, middle). We hypothesized that a radical stabilization strategy could be used to overturn this inherent kinetic preference, wherein the introduction of a radical-stabilizing group at C2 of the alkene in 1 could reduce the rate of cyclization at C2 of the 1,2-biradical intermediate 1*, resulting in selective 6-exo-trig cyclization of the more reactive C1 position. In addition, the group at C2 would stabilize the resulting 1,4-biradical intermediate, thus providing a thermodynamic driving force for the formation of the 6-membered ring intermediate 1ʹ, before radical recombination yields the bridged bicyclic product 2. Support for this hypothesis was provided by a density functional theory (DFT) study of the transition state energies of two cyclization pathways of 1,2-biradical 3*, the precursor to 3,6-N2-BCHep 4 (Fig. 1d). Cyclization of C2-methylated substrate 3ʹ led to the expected 5-exo-trig cyclization (see Supplementary Section 5 for DFT results), whereas introducing a phenyl ring as a radical-stabilizing group at C2 kinetically suppressed this undesired pathway, thereby enabling the anti-rule-of-five 6-exo-trig cyclization. Interestingly, this same selectivity reversal effect is not observed for the analogous 1,6-dienes49,50,51, highlighting the importance of the presence of the oxime.
Here, we report the successful implementation of this anti-rule-of-five intramolecular [2 + 2] photocycloaddition of aza-1,6-dienes using EnT photocatalysis with visible light, which provides a general strategy for the synthesis of substituted 6-N-BCHeps, 3,6-N2-BCHeps and 3-O-6-N-BCHeps. These structurally rigid motifs exhibit well-defined exit vectors and could therefore serve as bioisosteres for a range of nitrogen-containing heterocycles, including both aromatic and saturated systems. Moreover, the rich downstream chemistry offered by these compounds further underscores their synthetic utility in accessing structurally complex molecules. This allowed facile preparation of a 3,6-N2-BCHep analogue of a piperazine-based drug, whose physicochemical properties were tested to evaluate the potential of these scaffolds as bioisosteres in medicinal chemistry.
Results and discussion
Reaction design and optimization
To evaluate our hypothesis, aza-1,6-diene 3 containing an oxime and radical-stabilizing phenyl ring at C2 was chosen as the model cycloaddition precursor (Table 1). After evaluation of various parameters, we found that this reaction was indeed viable. Optimum conditions required the use of [Ir(dF[CF3]ppy)2(dtbbpy)]PF6 (1 mol%) as the photocatalyst under 427-nm light, in acetonitrile (0.1 M) at room temperature, which gave 3,6-N2-BCHep 4 in 68% isolated yield together with 13% of the fused product 4a (entry 1). This showed that the intramolecular [2 + 2] photocycloaddition of aza-1,6-dienes could undergo the anti-rule-of-five pathway through tuning the reactivity of the 1,2-biradicals (1*; Fig. 1c). Alternative photocatalysts with different redox potentials and triplet energies were tested (entries 2–5), and a correlation was found between the yield and triplet energy of the photocatalyst, but not with redox potentials, indicating that the reaction probably occurs through triplet EnT rather than a photoredox process52. The reaction was found to be relatively insensitive to solvent polarity (Supplementary Table 1), which again supports an EnT mechanism, where a neutral encounter complex rather than charge separation is involved53. Although thioxanthone, which was discovered to be similarly as effective at promoting the desired reaction of substrate 3, represents a cost-effective and practical alternative photosensitizer, it was deemed to be less general in its reactivity with a broad array of substrates. Finally, control experiments showed that both light and photocatalyst were necessary (entries 6 and 7).
Substrate scope
Having established the optimized conditions, we then explored the substrate scope (Fig. 2). Variation of the aromatic alkene substituent demonstrated that electron-deficient (6–11) and electron-rich phenyl rings (12 and 13) as well as heteroaromatics (14–17) could be used, giving the corresponding 3,6-N2-BCHeps in moderate-to-good yields. The reaction was successfully scaled up to gram scale with lower catalyst loading (0.5 mol%), giving the product 4 in similar yield (61% versus 68%). The reaction was further extended to incorporate aromatic fragments of six marketed pharmaceuticals (18–23), highlighting the broad functional group tolerance of the photocycloaddition. In all these cases, the fused bicycle side-products were either isolated in low yields or not observed at all (see Supplementary Section 1.11 for details). Importantly, the [2 + 2] photocycloaddition was not limited to aromatic alkenes and could be extended to dienes (24–26)54, which displayed complete regioselectivity for the formation of the bridged bicycle over the fused bicycle. Enones could also be used (27 and 28)55, although they were lower-yielding and returned substantial quantities of starting material (20–40%). Modifications to the tether between the styrene and oxime were also explored, including other N-protecting groups (29 and 30), oxygen (31 and 32) and all-carbon linkers (33). Furthermore, 2,3-amide (34), 3,4-amide (35 and 37) and 2,3,4-imide (36 and 38–44) tethers could be used, delivering the bridged bicycles in high yields. Using amide and imide tethers also enabled the bridgehead position (C5) to be substituted with various (hetero)alkyl or aryl groups (37–44), with examples including pharmaceutically relevant heterocycles (42 and 43). In addition to O-methyl oximes, it was found that a broader range of oximes (47 and 48) and hydrazones (49–52) underwent successful cycloaddition. While the parent unsubstituted (N–OH) oxime reacted efficiently, the N–OH product 45 was found to be unstable and rearranged to the [3.2.1] bicycle 46. However, in situ O-acylation prevented this rearrangement and allowed isolation of the stable acetoxy derivative 47 in good yield.
Reactions were performed with a mixture of E/Z oximes (or hydrazones) as the substrates, using 1 mol% [Ir(dF[CF3]ppy)2(dtbbpy)]PF6 in MeCN (0.1 M) under blue LED (427 nm) irradiation for 1–4 h. Yields are of isolated products. aThe reaction time is 20 h. b0.5 mol% [Ir(dF[CF3]ppy)2(dtbbpy)]PF6 was used. cThioxanthone (10 mol%) was used as the photocatalyst under 390-nm LED irradiation. d5 h reaction time, and 22% of the substrate was recovered. e24 h reaction time, and 41% of the substrate was recovered. fCH2Cl2 (0.1 M) as the solvent, then acylation using Ac2O. gDMSO (0.1 M) as the solvent. i-Pr, iso-propyl; Bn, benzyl; Ac, acetyl; DMSO, dimethyl sulfoxide.
Synthetic transformations and applications
Having shown the broad range of 6-N-BCHeps that could be accessed using this anti-rule-of-five photocycloaddition of aza-1,6-dienes, we were interested in extending the diversity of accessible products through further derivatisation. Vinyl 3,6-N2-BCHep 26 is a valuable substrate for diversification because of the synthetic utility of the vinyl group (Fig. 3, left). Importantly, the synthesis of 26 was scalable, with an 85% yield obtained when the cycloaddition was performed on a gram scale. We subsequently demonstrated that the vinyl group in 26 could be hydrogenated to give alkyl-substituted 3,6-N2-BCHep 53 and subjected to hydroboration/oxidation to give alcohol 54. Ozonolysis followed by amination, reduction or oxidation provided oxime 55, alcohol 56 and carboxylic acid 58, respectively. In addition, methylated product 57 could be accessed by sulfonylation and reduction of alcohol 56. Finally, derivatization of carboxylic acid 58 by amidation and reductive decarboxylation allowed the generation of hydroxamic acid 59 and the unsubstituted 3,6-N2-BCHep skeleton 60.
Diversification of products 26 and 61 using a wide variety of conditions. aPd/C, H2. b9-Borabicyclo(3.3.1)nonane (9-BBN), then H2O2, NaOH. cO3, Me2S, then NH2OBn·HCl, NaOAc. dO3, then NaBH4. e4-(Trifluoromethyl)benzenesulfonyl chloride, Et3N, 4-dimethylaminopyridine (DMAP), then NaBH4. fO3, Me2S, then NaH2PO4, 2-methyl-2-butene, NaClO2. gOxalyl chloride, then NH2OH·HCl, K2CO3. hN-hydroxyphthalimide, N,N′-diisopropylcarbodiimide, DMAP, then [Ir(ppy)2(dtbbpy)]PF6, Et3N, 1,4-cyclohexadiene, 456-nm blue LEDs. iIron powder, NH4Cl. jPhenyl isocyanate. kPhenyl isothiocyanate. lp-Toluenesulfonyl chloride, Et3N, DMAP. mN,N-dimethylsulfamoyl chloride, Et3N, DMAP. nDi-tert-butyl dicarbonate, DMAP. op-Toluoyl chloride, Et3N, DMAP. Tol, 4-methylphenyl; Boc, tert-butoxycarbonyl.
Further product diversification was possible through reductive cleavage of the oxime-derived N–O bond and functionalization of the resulting secondary amine (Fig. 3, right). While reduction of the N–OMe bond in 4 was not possible due to the sensitivity of the aryl-azetidine group, the more polarized N–O bond in acetoxy derivative 47 was readily cleaved under mild reducing conditions (iron powder)56, providing amine 61 in 85% yield. This enabled high-yielding derivatization to generate urea 62, thiourea 63, sulfonamide 64, sulfamide 65, carbamate 66 and amide 67, demonstrating the broad range of functional groups that could be incorporated onto the 3,6-N2-BCHeps.
To determine whether the 3,6-N2-BCHep scaffolds are suitable for use in bioactive molecules, 68 was prepared as an isosteric analogue of the matrix metalloproteinase inhibitor DB04232 and subjected to analysis57 (Fig. 4). The physicochemical and pharmacological properties of 68 were assessed in comparison with DB04232. Interestingly, although the isosteric analogue has a greater molecular weight (MW) than DB04232, it has reduced lipophilicity (logP). This counterintuitive reduction in lipophilicity has been observed previously and ascribed to an increase in solvent-exposed polar surface area resulting from conformational constraints58. Apart from this positive attribute, 68 exhibited similar intrinsic clearance (CLint) in human liver microsomes (in vitro human CLint, ml min−1 kg−1) and had a similar half-life (T1/2) compared with DB04232. These findings indicate that 68 is at least as metabolically stable as DB04232, demonstrating the potential for application of 3,6-N2-BCHep analogues as isosteres of frequently used heterocycles in drug candidates. However, further studies on these systems, including efficacy analysis and comparisons with unsaturated analogues, are required before the prospective utility of these systems can be fully evaluated.
aDefined as logP at pH 7.4; determined by high-performance liquid chromatography (HPLC) analysis. bDefined as metabolic half-life. cDefined as microsome intrinsic clearance. dDefined as hepatic intrinsic clearance. eDetermined at 60 min in the presence of human liver microsomes with NADPH (reduced nicotinamide adenine dinucleotide phosphate). Cbz, benzyloxycarbonyl.
Conclusions
In this study, we have introduced a visible-light-mediated intramolecular [2 + 2] cycloaddition of aza-1,6-dienes, a synthetic approach that defies the long-standing rule-of-five in radical cyclization. By exploiting differential radical stabilization, we demonstrated that this strategy can be used to access a diverse range of substituted 6-azabicyclo[3.1.1]heptane (6-N-BCHep) scaffolds, a previously challenging to access class of bridged nitrogen heterocycles. The methodology demonstrates broad functional group tolerance and enables selective substitution at multiple positions, expanding the chemical space for medicinal chemistry applications. Moreover, preliminary studies on the newly synthesized 6-N-BCHeps show their potential as bioisosteres, offering similar or enhanced pharmacokinetic properties compared with their saturated counterparts. This work not only provides a synthetic tool for accessing complex heterocycles but also contributes to the ongoing exploration of three-dimensional chemical space, paving the way for future developments in medicinal chemistry and beyond.
Methods
General procedure for intramolecular [2 + 2] cyclization
An oven-dried round-bottom flask containing aza-1,6-dienes (1.0 equiv.) and [Ir(dF(CF3)ppy)2(dtbbpy)](PF6) (1 mol%) was sealed and subjected to three N2 evacuation–refill cycles before anhydrous solvent (0.1 M) was added. The flask was placed at a distance of approximately 1 cm from a 40 W KSPR160L-427 nm (or 390 nm) Kessil light (100% intensity), and the reaction was stirred under continuous irradiation under a nitrogen atmosphere for 1–4 h until complete (judged by thin-layer chromatography analysis). The solvent was removed in vacuo, and the crude product was purified by flash column chromatography.
Data availability
The X-ray crystallographic coordinates for structures 36 and 62 reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers 2451817 and 2451818, respectively. These data can be obtained free of charge via The Cambridge Crystallographic Data Centre at www.ccdc.cam.ac.uk/data_request/cif. All other data are available in the article or its Supplementary Information. A preprint version of this work was previously posted on ChemRxiv at https://doi.org/10.26434/chemrxiv-2025-285mh.
References
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Lovering, F. Escape from Flatland 2: complexity and promiscuity. MedChemComm 4, 515–519 (2013).
Mykhailiuk, P. K. Saturated bioisosteres of benzene: where to go next? Org. Biomol. Chem. 17, 2839–2849 (2019).
Kanazawa, J. & Uchiyama, M. Recent advances in the synthetic chemistry of bicyclo[1.1.1]pentane. Synlett 30, 1–11 (2019).
He, F.-S., Xie, S., Yao, Y. & Wu, J. Recent advances in the applications of [1.1.1]propellane in organic synthesis. Chin. Chem. Lett. 31, 3065–3072 (2020).
Ma, X. & Nhat Pham, L. Selected topics in the syntheses of bicyclo[1.1.1]pentane (BCP) analogues. Asian J. Org. Chem. 9, 8–22 (2020).
Anderson, J. M., Measom, N. D., Murphy, J. A. & Poole, D. L. Bridge functionalisation of bicyclo[1.1.1]pentane derivatives. Angew. Chem. Int. Ed. 60, 24754–24769 (2021).
Subbaiah, M. A. M. & Meanwell, N. A. Bioisosteres of the phenyl ring: recent strategic applications in lead optimization and drug design. J. Med. Chem. 64, 14046–14128 (2021).
Bellotti, P. & Glorius, F. Strain-release photocatalysis. J. Am. Chem. Soc. 145, 20716–20732 (2023).
Macreadie, L. K., Idrees, K. B., Smoljan, C. S. & Farha, O. K. Expanding linker dimensionality in metal–organic frameworks for sub-ångstrom pore control for separation applications. Angew. Chem. Int. Ed. 62, e202304094 (2023).
Shire, B. R. & Anderson, E. A. Conquering the synthesis and functionalization of bicyclo[1.1.1]pentanes. JACS Au 3, 1539–1553 (2023).
Tsien, J., Hu, C., Merchant, R. R. & Qin, T. Three-dimensional saturated C(sp3)-rich bioisosteres for benzene. Nat. Rev. Chem. 8, 605–627 (2024).
Meanwell, N. A. Applications of bioisosteres in the design of biologically active compounds. J. Agric. Food Chem. 71, 18087–18122 (2023).
Frank, N. et al. Synthesis of meta-substituted arene bioisosteres from [3.1.1]propellane. Nature 611, 721–726 (2022).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014).
Shearer, J., Castro, J. L., Lawson, A. D. G., MacCoss, M. & Taylor, R. D. Rings in clinical trials and drugs: present and future. J. Med. Chem. 65, 8699–8712 (2022).
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the nitrogen heterocycle compositions and properties of U.S. FDA-approved pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Dibchak, D. et al. General synthesis of 3-azabicyclo[3.1.1]heptanes and evaluation of their properties as saturated isosteres. Angew. Chem. Int. Ed. 62, e202304246 (2023).
Dutta, S., Daniliuc, C. G., Mück-Lichtenfeld, C. & Studer, A. Formal [2σ + 2σ]-cycloaddition of aziridines with bicyclo[1.1.0]butanes: access to enantiopure 2-azabicyclo[3.1.1]heptane derivatives. J. Am. Chem. Soc. 146, 27204–27212 (2024).
Lin, Z., Ren, H., Lin, X., Yu, X. & Zheng, J. Synthesis of azabicyclo[3.1.1]heptenes enabled by catalyst-controlled annulations of bicyclo[1.1.0]butanes with vinyl azides. J. Am. Chem. Soc. 146, 18565–18575 (2024).
Liu, Y. et al. Pyridine-boryl radical-catalyzed [3π + 2σ] cycloaddition for the synthesis of pyridine isosteres. Chem 10, 3699–3708 (2024).
Zhang, J., Su, J.-Y., Zheng, H., Li, H. & Deng, W.-P. In(OTf)3-catalyzed (3 + 3) dipolar cyclization of bicyclo[1.1.0]butanes with N-nucleophilic 1,3-dipoles: access to 2,3-diazabicyclo[3.1.1]heptanes, 2,3-diazabicyclo[3.1.1]heptenes, and enantiopure 2-azabicyclo[3.1.1]heptanes. ACS Catal. 14, 17837–17849 (2024).
George, M., Mindner, J., Wittmer, S., Knyazev, D. A. & Werz, D. B. Double strain-release (3+3)-cycloaddition: Lewis acid catalyzed reaction of bicyclobutane carboxylates and aziridines. Chem. Eur. J. 31, e202404099 (2025).
Zhu, S. et al. FeCl3-catalyzed intermolecular formal [8π+2σ] cycloaddition of azaheptafulvene with bicyclo[1.1.0]butanes for the synthesis of cycloheptatriene-fused 2-azabicyclo[3.1.1]heptanes. Org. Lett. 27, 3831–3837 (2025).
Dhake, K. et al. Diastereoselective dearomative cycloaddition of bicyclobutanes with pyridinium ylides: a modular approach to multisubstituted azabicyclo[3.1.1]heptanes. Chem. Commun. 60, 13008–13011 (2024).
Hu, S., Gao, Y., Pan, Y., Ni, D. & Deng, L. Modular synthesis of azidobicyclo[2.1.1]hexanes via (3 + 2) annulation of α-substituted vinyl azides and bicyclo[1.1.0]butanes. J. Org. Chem. 89, 15151–15157 (2024).
Jiang, Q. et al. Lewis-acid-catalyzed dearomative [4π + 2σ] cycloaddition of bicyclobutanes with isoquinolinium methylides for the synthesis of ring-fused azabicyclo[3.1.1]heptanes. Org. Lett. 26, 9311–9315 (2024).
Liang, Y., Nematswerani, R., Daniliuc, C. G. & Glorius, F. Silver-enabled cycloaddition of bicyclobutanes with isocyanides for the synthesis of polysubstituted 3-azabicyclo[3.1.1]heptanes. Angew. Chem. Int. Ed. 63, e202402730 (2024).
Lysenko, V. et al. Multigram synthesis of 3-azabicyclo[3.1.1]heptane derivatives including bicyclic thalidomide analogs. Eur. J. Org. Chem. 28, e202400938 (2024).
Revie, R. I., Whitaker, B. J., Paul, B., Smith, R. C. & Anderson, E. A. Synthesis of heterocycle-substituted bicyclo[3.1.1]heptanes and aza-bicyclo[3.1.1]heptanes via photocatalytic Minisci reaction. Org. Lett. 26, 2843–2846 (2024).
Wang, X., Gao, R. & Li, X. Catalytic asymmetric construction of chiral polysubstituted 3-azabicyclo[3.1.1]heptanes by copper-catalyzed stereoselective formal [4π+2σ] cycloaddition. J. Am. Chem. Soc. 146, 21069–21077 (2024).
Zhao, J., Zhao, X.-J., Cao, P., Liu, J.-K. & Wu, B. Polycyclic azetidines and pyrrolidines via palladium-catalyzed intramolecular amination of unactivated C(sp3)–H bonds. Org. Lett. 19, 4880–4883 (2017).
Chernykh, A. V., Vashchenko, B. V., Shishkina, S. V., Volochnyuk, D. M. & Grygorenko, O. O. 3-Substituted 6-azabicyclo[3.1.1]heptanes: nonclassical piperidine isosteres for drug discovery. J. Org. Chem. 89, 10440–10450 (2024).
Chernykh, A. V. et al. Fluorine-containing 6-azabicyclo[3.1.1]heptanes: bicyclic piperidine analogs for drug discovery. Eur. J. Org. Chem. 28, e202500022 (2025).
St. Jean, D. J. Jr & Fotsch, C. Mitigating heterocycle metabolism in drug discovery. J. Med. Chem. 55, 6002–6020 (2012).
Obach, R. S. et al. Strategies toward optimization of the metabolism of a series of serotonin-4 partial agonists: investigation of azetidines as piperidine isosteres. Xenobiotica 46, 1112–1121 (2016).
Zhang, W. et al. Discovery of (2S)-N-(6-cyano-5-(trifluoromethyl)pyridin-3-yl)-3-(6-(4-cyanophenyl)-3,6-diazabicyclo[3.1.1]heptan-3-yl)-2-hydroxy-2-methylpropanamide as a highly potent and selective topical androgen receptor antagonist for androgenetic alopecia treatment. J. Med. Chem. 67, 322–348 (2024).
Rágyanszki, A., Fiser, B., Lee-Ruff, E. & Liebman, J. F. Strained small nitrogen heterocycles-azabicyclobutanes and azirines. ChemistrySelect 8, e202301405 (2023).
Rigotti, T. & Bach, T. Bicyclo[2.1.1]hexanes by visible light-driven intramolecular crossed [2 + 2] photocycloadditions. Org. Lett. 24, 8821–8825 (2022).
Liu, R. S. H. & Hammond, G. S. Photosensitized internal addition of dienes to olefins. J. Am. Chem. Soc. 89, 4936–4944 (1967).
Srinivasan, R. & Carlough, K. H. Mercury(3P1) photosensitized internal cycloaddition reactions in 1,4-, 1,5-, and 1,6-dienes. J. Am. Chem. Soc. 89, 4932–4936 (1967).
Gleiter, R. & Sander, W. Light-induced [2+2] cycloaddition reactions of nonconjugated dienes—the effect of through-bond interaction. Angew. Chem. Int. Ed. 24, 566–568 (1985).
Zhao, J. et al. Intramolecular crossed [2+2] photocycloaddition through visible light-induced energy transfer. J. Am. Chem. Soc. 139, 9807–9810 (2017).
Kerckhoffs, A. et al. Synthesis of aryl-fused bicyclo[3.1.1]heptanes (BCHeps) and validation as naphthyl bioisosteres. Preprint at ChemRxiv https://doi.org/10.26434/chemrxiv-2024-t0t94 (2024).
Dethe, D. H., Sharma, N. & Juyal, S. Concise enantioselective total syntheses of rearranged ent-trachylobane diterpenoids (–)-Wallichanols A and B. Angew. Chem. Int. Ed. 64, e202505766 (2025).
Li, M., Tao, L.-Y., Gu, X.-T., Wei, Y. & Shi, M. Intramolecular crossed [2+2] cycloaddition through visible-light triplet sensitization: expedient access to bridged bicyclic alkene. Adv. Synth. Catal. 367, e202401402 (2025).
Becker, M. R., Richardson, A. D. & Schindler, C. S. Functionalized azetidines via visible light-enabled aza Paternò–Büchi reactions. Nat. Commun. 10, 5095 (2019).
Sturino, C. F. & Fallis, A. G. Rate constants for 5- and 6-exo secondary alkyl radical cyclizations onto N,N-diphenylhydrazones. J. Org. Chem. 59, 6514–6516 (1994).
Lu, Z. & Yoon, T. P. Visible light photocatalysis of [2+2] styrene cycloadditions by energy transfer. Angew. Chem. Int. Ed. 51, 10329–10332 (2012).
Mojr, V. et al. Tailoring flavins for visible light photocatalysis: organocatalytic [2+2] cycloadditions mediated by a flavin derivative and visible light. Chem. Commun. 51, 12036–12039 (2015).
Gu, X. et al. Visible-light-mediated activation of remote C(sp3)−H bonds by carbon-centered biradical via intramolecular 1,5- or 1,6-hydrogen atom transfer. Angew. Chem. Int. Ed. 63, e202409463 (2024).
Strieth-Kalthoff, F. & Glorius, F. Triplet energy transfer photocatalysis: unlocking the next level. Chem 6, 1888–1903 (2020).
Birks, J. B. Excimers and exciplexes. Nature 214, 1187–1190 (1967).
Hurtley, A. E., Lu, Z. & Yoon, T. P. [2+2] Cycloaddition of 1,3-dienes by visible light photocatalysis. Angew. Chem. Int. Ed. 53, 8991–8994 (2014).
Guo, R. et al. Strain-release [2π + 2σ] cycloadditions for the synthesis of bicyclo[2.1.1]hexanes initiated by energy transfer. J. Am. Chem. Soc. 144, 7988–7994 (2022).
Campbell, C. D. & Stewart, M. I. Reflections on the teaching practices for the reduction of nitroarenes: updating methodologies and considerations of the mechanism. J. Chem. Educ. 100, 3171–3178 (2023).
Cheng, M. et al. Design and synthesis of piperazine-based matrix metalloproteinase inhibitors. J. Med. Chem. 43, 369–380 (2000).
Degorce, S. L., Bodnarchuk, M. S., Cumming, I. A. & Scott, J. S. Lowering lipophilicity by adding carbon: one-carbon bridges of morpholines and piperazines. J. Med. Chem. 61, 8934–8943 (2018).
Acknowledgements
We thank UK Research and Innovation (UKRI) under the UK government’s Horizon Europe funding guarantee for an ERC-approved grant (EP/Y028015/1). Z.-X.Z. acknowledges support from a Leverhulme Trust Early Career Fellowship (ECF-2025-137). M.J.P.M. thanks the Bristol Chemical Synthesis Centre for Doctoral Training, funded by EPSRC (EP/S024107/1). We thank Wuxi AppTec Co., Ltd. for performing physicochemical and pharmacological studies. We thank M. Zanini and J. Tromans (University of Bristol) for assistance in the preparation of the paper. J. Wu, C. Zhu (Shanghai Jiao Tong University) and J. Yu (Tsinghua University) are warmly acknowledged for their helpful discussion. We also thank J. Wu from Shanghai Jiao Tong University for sharing laboratory facilities. The crystal structures of compounds 36 and 62 were determined by the X-ray Crystallography Service at the University of Bristol. We gratefully acknowledge H. Sparkes and J. Charmant (University of Bristol) for assistance with X-ray analysis.
Author information
Authors and Affiliations
Contributions
Z.-X.Z., A.N. and V.K.A. conceived the project; A.N. and V.K.A. directed the research, Z.-X.Z., J.L.T., A.N. and V.K.A. prepared the paper; Z.-X.Z. and K.S. performed the experimental work; M.J.T., M.J.P.M. and Y.G. performed the DFT calculations. All authors analysed the results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Peter Seavill, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Sections 1–7, Figs. 1–11 and Tables 1–7.
Supplementary Data 1
X-ray crystallographic data for compound 36, CCDC 2451817.
Supplementary Data 2
X-ray crystallographic data for compound 62, CCDC 2451818.
Supplementary Data 3 (download PDF )
DFT calculations, thermochemical data and XYZ coordinates.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, ZX., Shu, K., Tilby, M.J. et al. Breaking the ‘rule-of-five’ to access bridged bicyclic heteroaromatic bioisosteres. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-00990-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-00990-0