Abstract
Retinal imaging is essential for diagnosing and treating retinal diseases. However, the lack of standardized and realistic optical phantoms limits the calibration and validation of imaging systems. Here we developed a realistic retinal phantom based on the concept of a super phantom designed to replicate the morphological and functional characteristics of the human retina. The phantom comprises a 13-layered structure, microfluidic channels to emulate vascular networks in the human retina, and fluorescent microbeads to replicate retinal autofluorescence. We validated the measurement of axial resolution, depth range, and field-of-view of optical coherence tomography (OCT) using the phantom. In addition, we confirmed the utility of the phantom across multiple ophthalmic imaging modalities, including OCT, OCT angiography, fundus autofluorescence, fluorescein angiography, and indocyanine green angiography. Imaging systems will be able to enhance the diagnosis and therapeutic monitoring of retinal diseases by standardizing imaging systems with this phantom and improving both the qualitative analysis and the accuracy of quantitative parameters, such as retinal thickness.
Similar content being viewed by others
Introduction
Blindness caused by retinal diseases, such as diabetic retinopathy, age-related macular degeneration, macular edema, and retinoblastoma, remains a major global health challenge. These conditions often cause structural changes such as retinal thickness variations, and vascular abnormalities including vascular wall damage and neovascularization1,2. Early diagnosis and continuous monitoring of these diseases are critical for preventing irreversible vision loss. Recently, research on gene and cell therapies has been actively conducted for therapeutic monitoring of genetic retinal diseases3,4. As a result, retinal imaging has become indispensable for diagnosing, tracking the progression and therapeutic monitoring of retinal diseases.
The current retinal imaging modalities provide extensive insights into the structural and functional changes that occur in a range of diseases. Fundus photography and scanning laser ophthalmoscopy (SLO) capture two-dimensional morphological images, whereas fluorescein angiography (FA) and indocyanine green angiography (ICGA) visualize the retinal vasculature using exogenous contrast agents5,6,7,8,9,10. Fundus autofluorescence (FAF) leverages the natural fluorescence of lipofuscin in the retinal pigment epithelium (RPE) to provide data on the structural and metabolic features of the retina11,12. Optical coherence tomography (OCT), introduced in 1991, has become a cornerstone in ophthalmic diagnostics, offering micrometer-scale, cross-sectional, and three-dimensional imaging of the retinal layers13,14. OCT angiography (OCTA), an extension of OCT, enables depth-resolved imaging of vascular networks without using contrast agents15,16.
Recent research advances have greatly enhanced the precision of retinal imaging technologies, enabling detailed multifunctional visualization of retinal structures and vasculature17. For instance, multimodal imaging modalities, such as OCT combined with SLO, now offer imaging over a 100-degree field of view (FOV). However, these advances underscore the critical need for standardized evaluation tools for validating imaging quality, assessing system performance, and ensuring reliable diagnostic outcomes. Optical phantoms have emerged as effective and reproducible tools that exhibit great consistency and adaptability, and facilitate ethical compliance when using animal or human samples18,19. Beyond system validation, phantoms are increasingly being recognized as essential for operator training, device calibration, and preclinical development of novel imaging technologies.
Despite these advantages, the existing retinal phantoms have notable limitations. Most phantoms are either overly simplified or tailored to specific imaging modalities, lacking the complexity of replicating the multilayered structure and functional properties of the human retina. Recent efforts to improve the design of optical phantoms have included features such as multilayered films, microfluidic vascular networks, and curved surfaces. Phantoms with specific features, such as six-layered models for the comparison of retinal nerve fiber layer thickness on OCT (Kinkelder et al.20) and foveal pits for the evaluation of OCT accuracy (Baxi et al.21), have been introduced in previous studies. Other studies were focused on the fabrication of phantoms that mimic specific features of the retina, such as healthy and diseased retinal phantoms that replicate the foveal shape22, and an optic nerve head (ONH) phantom used for OCT-based measurements23. Cao et al.24 constructed a model eye that comprises an artificial eye frame with a multilayer film that mimics the retina for evaluation of the axial resolution of OCT images.
In addition to structural phantoms, functional phantoms used for evaluating angiography systems have been developed. Flow phantoms composed of agar or gelatin-mixed scattering particles and glass or silicon capillary tubes to represent fluid flow were predominantly utilized in previous studies25,26,27,28,29,30. These phantoms are used to calculate and assess the accuracy of OCTA or retinal vessel oximetry. Luu et al.31 devised a microfluidics-based phantom that emulates the realistic superficial vasculature networks. Hayakawa et al.32 developed a phantom that mimics the retinal curvature with embedded microchannels for simulating vitreous surgery. Zhao et al.33 developed a multilayered retinal phantom with vascular networks using 3D printing and spin-coating techniques. Notably, these existing phantoms were generally focused on either the structural or functional characteristics of the retina, which limits their utility for comprehensive evaluations of multimodal imaging systems.
To overcome these limitations, we aimed to build upon our previous work34 and fabricate a more realistic retinal phantom based on the concept of the super phantom—advanced models introduced by Manhar et al.35 that replicate both the morphological and functional characteristics of the target sample. Our phantom features a 13-layered structure created using spin-coating, microfluidic channels that mimic retinal vascular networks, and fluorescent microbeads to simulate autofluorescence. The phantom was designed for use across multiple imaging modalities, including OCT, OCTA, FA, ICGA, and FAF, and provides a versatile platform for system validation and operator training. Unlike previous iterations, this phantom incorporates a realistic curvature and functional properties, enabling more accurate approximation of the human retinal anatomy and physiology. In this article, we describe the design, fabrication, and validation of this super phantom. By demonstrating its applicability in the measurement of axial resolution, depth range, and field-of-view, we established the utility of the phantom as a valuable tool that has the potential to bridge critical gaps in diagnostic technology by facilitating the standardization of imaging system evaluations, improvement of diagnostic accuracy, and development of next-generation ophthalmic imaging technologies.
Methods
Development of the retinal phantom
To develop a realistic retinal phantom, we replicated the layers and curvature of the retina, improving upon our previous design. FA and ICGA images were used as a reference to modify the microfluidic channels to better mimic the retinal blood vessel networks. Retinal autofluorescence was simulated using fluorescent microbeads (Fluorescent Microspheres; Cospheric LLC, Goleta, USA). The phantom was fabricated using polydimethylsiloxane (PDMS; Sylgard 184 kit, Dow Corning, USA) and titanium dioxide (TiO₂, 718467, Sigma-Aldrich, USA). The TiO₂ concentration in the PDMS was optimized to match retinal image scattering, and the thickness of each layer was determined through analysis of human retinal OCT images36,37,38,39,40.
Design and fabrication of the microfluidic channels
Three vascular networks—superficial vessels (SV), deep vessels (DV), and choroidal vessels (CV)—were replicated using microfluidic channels (Fig. 1).
-
Superficial vessels (Ch1): The first microfluidic channel (Ch1) was designed to mimic the superficial retinal vessels based on FA images of a normal retina41,42,43. Data on the vessel regions were extracted and processed into binary images (Supplementary Fig. 1a, Step 1). Disconnected segments were connected to ensure continuous fluid flow (Supplementary Fig. 1a, Step 2). The channel was designed to have an overall diameter of 16 mm (Supplementary Fig. 1a, Step 3), with the fovea and ONH spaced 4.5 mm apart (Fig. 1a)44.
-
Deep vessels (Ch2): The second microfluidic channel (Ch2) represents the deep vessels, which consists of small capillaries. A honeycomb-shaped design with a channel width of 20 µm was chosen to simplify the intricate capillary network (Fig. 1b). A hexagonal region with a 0.7 mm diameter in the center was left unfilled to replicate the avascular foveal zone.
-
Choroidal vessels (Ch3): The third microfluidic channel (Ch3) was designed based on diagrams and ICGA images of the choroidal vasculature45,46. The channel was designed to replicate the four vortex veins (Supplementary Fig. 1b, Step 1) and aligned with the ONH region of Ch1 (Fig. 1c; Supplementary Fig. 1b, Step 2). Notably, the number of vortex veins in the retina typically ranges from four to eight, with four or five being predominant in the normal population, as indicated in ICGA studies47.
a Microfluidic channel design for the superficial retinal vessels (Ch1). b Microfluidic channel design for the deep vessels (Ch2). c Microfluidic channel design for the choroidal vessels (Ch3). d Superimposed images of all the channels.
The three channels were merged to align with the avascular region and interconnected through holes (Fig. 1d). As shown in Fig. 1a–c, photomasks with design patterns were used to define the microfluidic channels and were subsequently subjected to photolithography34. Each microfluidic channel was patterned on a 4-inch silicon wafer, which served as a mold for fabricating the channels. The heights of Ch1 and Ch2 were adjusted to 20 µm to match the inner diameter of the human retinal layers, where each vessel predominantly resides36,37,38,39,40. The height of Ch3, corresponding to the choroid and containing larger blood vessels, was designed to be 35 µm48. Additionally, to improve the durability and usability of the phantom, the minimum channel width was set to 20 µm, as narrower widths would make it difficult to extract the PDMS from the wafer mold. We fabricated eight microfluidic channel blocks for each channel and measured their average height and width using white light scanning interferometry (WSI; NV-2400, NanoSystem Co., Korea). The height of the channel through which fluid will flow was determined based on the target thickness of the photoresist layer (SU-8 2000 Series, Kayaku Advanced Materials, USA). The thickness from the surface of the thin film to the top of each channel was defined by the applied load after pouring the PDMS-TiO2 mixture into the wafer mold.
Multilayered thin films and retinal curvature
Two types of multilayered thin films (ML1 and ML2) were fabricated using spin-coating to replicate the intricate structure of the retina, as described in our previous studies34,41,42. The detailed configuration of the phantom is shown in the schematic in Fig. 2.
-
First Multilayer (ML1): ML1 comprises three layers that correspond to the ganglion cell layer, inner plexiform layer, and inner nuclear layer, which are positioned between the nerve fiber layer (NFL) and the outer plexiform layer (OPL).
-
Second Multilayer (ML2): ML2 represents the hyperreflective outer retinal band (HORB), which consists of four distinct highly reflective bands situated beneath the outer nuclear layer (ONL)49,50,51,52:
-
1.
External limiting membrane (Band 1)
-
2.
Boundary between the inner segment and outer segment (OS) of the photoreceptor layer (Band 2)
-
3.
OS tips or Verhoeff membrane (Band 3)
-
4.
Retinal pigment epithelium (RPE; Band 4)
The retinal phantom consists of two different multilayers, three single layers containing microfluidics channels, and a base plate with a retinal curvature. NFL nerve fiber layer, GCL ganglion cell layer, IPL inner plexiform layer, INL inner nuclear layer, OPL outer plexiform layer, ONL outer nuclear layer, ELM external limiting membrane, IS inner segment, OS outer segment, RPE retinal pigment epithelium, SV superficial vessels, DV deep vessels, CV choroidal vessels.
ML2 emulates the ONL and HORB, representing a total of eight layers below the OPL.
To replicate the autofluorescence properties of lipofuscin found in the RPE, orange-yellow fluorescent microbeads with diameters of 1–5 µm were incorporated into the PDMS-TiO₂ mixture at a weight ratio of 0.5%. Lipofuscin, a naturally occurring component in the RPE with a maximum particle size of 3–5 µm, exhibits yellow autofluorescence when stimulated by near-ultraviolet or blue light53. The fluorescent beads, with peak excitation and emission wavelengths of 460 nm and 594 nm, respectively, were mixed into the PDMS solution to mimic the autofluorescence property of lipofuscin. The mixing ratio and bead distribution were optimized using a fluorescence microscope system and a vision camera (Supplementary Fig. 2). A 488 nm laser diode (LP488-SF20, Thorlabs, Newton, NJ) was employed to excite the fluorescent beads, while a dichroic mirror (#86-334, Edmund Optics, Barrington, NJ) and an emission filter (#67-034, Edmund Optics, Barrington, NJ) were used to isolate the emitted light.
ML1 was fabricated on a 6-inch glass substrate treated with hydrophobic silane to improve adhesion and facilitate layer curing. ML2 was spin-coated onto a 6-inch silicon wafer, which reduced the load on the spin-coating machine and allowed for higher spin speeds to achieve thinner bands. Unlike the glass substrate, the silicon wafer allowed the cured PDMS thin film to be detached without a silane coating. Despite the differences in the substrate materials used, ML1 and ML2 were fabricated using the same procedure, which included iterative spin-coating, curing, and thickness measurement. These processes were repeated three times for ML1 and eight times for ML2 to achieve the desired multilayer structure.
The base plate, designed to replicate the choroid, was produced by casting a PDMS-TiO₂ mixture (1.0% concentration) in a convex mold with a diameter of 23 cm, representing the average retinal curvature of the human eye54. The resulting base plate, which had a thickness exceeding 5 mm, was designed to facilitate the insertion of inlet and outlet tubes. The two multilayers (ML1 and ML2) and three microfluidic channel layers were affixed to the curved base plate, creating a retinal phantom that accurately replicates both the layered structure and curvature of the retina.
Phantom assembly
The assembly of the retinal phantom involved bonding the single-layered thin films containing the microfluidic channels with the multilayered thin films after oxygen plasma treatment. The upper layer of the channel at the inlet port of Ch1 was thin, raising concerns regarding swelling or bursting under fluid pressure. To prevent this, an additional transparent PDMS layer was attached to the top of Ch1. This PDMS layer was fabricated by spin-coating a silane coating on a glass substrate, which acted as a pressure-resistant substrate during the production of Ch1. During the curing process, the PDMS layer came into direct contact with the PDMS mixture of Ch1 (Fig. 3a).
a Production of a transparent layer to prevent Ch1 from bursting. b Create an inlet and outlet holes for fluid flow and attach two multilayers and three channels. c Structure containing two multilayers and three channels is cut to 34 mm after attaching the curved base plate. d Attach a transparent PDMS ring pattern to prevent fluid leakage from Ch1. e Attach the tubes to the inlet and outlet ports.
As shown in Fig. 3b, the two multilayers and three single layers with microfluidic channels were punched to create inlet and outlet holes for fluid flow. These layers were then aligned and bonded through the through-holes to interconnect the channels. The assembled multilayers and channels were attached to the curved base plate and cut to a diameter of 34 mm using a cutting punch (Fig. 3c). A 2-mm thick transparent PDMS ring was added to seal the holes in Ch1 (Fig. 3d). The inner and outer diameters of the ring were 18 mm and 34 mm, respectively. Fluorinated ethylene propylene (FEP) tubes (1521 L, IDEX Health & Science LLC., USA) with an inner diameter of 1/16 inch and an outer diameter of 1/8 inch were connected to the inlet and outlet ports (Fig. 3e). A waterproof silicone bond was applied to secure the tubes, which were cured for 24 h before use.
After assembly, the retinal phantom was placed in a housing with a lens designed for ophthalmic imaging. The distance between the lens and the phantom was simulated by considering the refractive index of the vitreous humor. In accordance with the International Organization for Standardization (ISO) 16,971 recommendations, the lens attached to the front of the housing had a posterior focal length of ~17 mm (58D) at the air-lens-solution interface55. Once the phantom was inserted into the housing, the vacant area was filled with a simulated vitreous humor solution (BZ277; Biochemazone, Alberta, Canada) using a syringe to replicate the vitreous humor in the eye.
A Luer-lock syringe was attached to a tube connected to the phantom to provide fluid flow inside the phantom. For OCTA imaging, a blood-mimicking fluid was created by mixing red ink with a diluted 2% intralipid solution in deionized water, which was then injected into the phantom. For FA and ICGA imaging, solutions were prepared using fluorescein and ICG powder at concentrations suitable for human conditions. These solutions were combined with red ink to simulate blood before being injected into the phantom. For FA, ICGA, and FAF imaging, which do not require continuous fluid flow, the phantom was sealed after fluid injection to prevent leakage.
Imaging system
We used a custom-built OCT system56,57 and OCTA processing to verify the performance of our retinal phantom. The OCT system utilizes a superluminescent diode light source with a center wavelength (λc) of 849 nm and a −3 dB bandwidth (Δλ) of 100 nm (BLM2-D-840-B-I-10, SUPERUM, Ireland), yielding a theoretical axial resolution (Δz) of 3.2 µm in air, calculated as:
The collimated light beam was directed into the phantom using 2-D galvanometer scanners, a tube lens, and a scan lens, with an optical power of ~780 µW at the phantom’s surface. The combined light reflected from the reference and sample arms was directed to the spectrometer at an A-line rate of 110 kHz.
A spectrometer that includes a line-scan camera with 4096 pixels and a Camera Link interface (spL4096-70 km, Basler, Germany) was used in previous studies56,57. In the present study, we used an updated spectrometer with a 2048-pixel line-scan camera and a USB 3.0 interface (October Plus, Teledyne E2V, UK). Consequently, the axial resolution and depth range were recalibrated using a BK7 glass sample in accordance with ISO 16971 standards. The BK7 glass had a refractive index of 1.5098 at 850 nm and a thickness of 995.71 ± 0.05 µm, measured using a reference gauge block and comparator34. The recalibrated axial resolution in air was measured as 3.36 µm, with a depth range of 1.96 mm.
OCTA images were acquired using the split-spectrum amplitude-decorrelation angiography algorithm58, which divides the spectra into four parts and averages five A-lines for noise reduction. Additionally, clinical ophthalmic imaging systems that incorporate OCTA and multicolor SLO were used to acquire OCT, OCTA, FAF, FA, and ICGA images of the phantom. These systems offer an ultrawide FOV, ensuring comprehensive visualization of the structure and vascular networks of the phantom.
Results
Retinal phantom: morphological and functional properties
We successfully designed and manufactured a retinal phantom mount, supporting arms, and a holder to secure the phantom in a clinical ophthalmic imaging instrument (Fig. 4). The phantom was connected to a syringe pump via FEP tubes and a Luer-lock syringe, allowing for controlled fluid injection (Fig. 4a). In addition, the phantom can be embedded into the chin rest of the clinical instrument (Fig. 4b).
a The phantom connected to a syringe pump. b The retinal phantom embedded into the chin rest of a clinical ophthalmic imaging instrument (OCT combined with multicolor SLO).
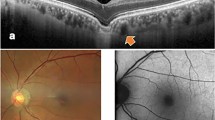
The retinal phantom effectively replicated the morphological and functional characteristics of the human retina. As shown in Fig. 5a, cross-sectional OCT images acquired using a commercial clinical system demonstrate that the phantom reproduces key structural features, including the relative thickness and signal profile across 13 retinal layers. The human retina image used for comparison was obtained from the publicly available OCTDL dataset published by Kulyabin et al.59. Thickness measurements were obtained using six phantoms, with the measurements taken at four points per phantom (Fig. 5b). The segmentation of the NFL (green line) and RPE (red line) (Fig. 6a) confirmed that the target thickness value for each layer was achieved with a standard deviation of ≤2.17 µm, indicating high reproducibility across multiple phantoms. Using this segmentation data, an en-face thickness color map (0–500 µm) was generated to validate the consistency of the phantom’s thickness (Supplementary Fig. 3). All layers were attached to the base plate with a retinal curvature, allowing images with similar curvatures to be obtained without additional correction.
a Comparison of a human retina OCT image (retina) and a retinal phantom OCT image (right). The human retina image was obtained from the publicly available OCTDL dataset published by Kulyabin et al.59. Red arrowheads indicate the vitreous membrane surrounding the vitreous humor. The phantom image shows 13 distinguishable layers including the nerve fiber layer (NFL), ganglion cell layer (GCL), inner plexiform layer (IPL), inner nuclear layer (INL), outer plexiform layer (OPL), outer nuclear layer (ONL), and choroid, as well as four hyperreflective bands. Scale bar is 100 μm. b Thickness measurements for each retinal layer of the phantom, based on 24 samples (six phantoms × four positions per phantom). Data are presented as mean ± standard deviation. The original measurement data can be found in Supplementary Data 1. Each scale bar represents 100 μm. The standard deviation was less than 2.17 μm.
a Cross-sectional OCT image of the retinal phantom. Ch1, the first microfluidic channel, mimics superficial vessels. Ch2 and Ch3 are the second and third microfluidic channels and replicate the deep and choroidal vessels, respectively. Scale bar is 100 μm. b Result of the axial resolution measurement from the line spread function (yellow line) in (a) after zero-padding from the original axial resolution measurement data in Supplementary Data 2.
To further assess structural precision, the average height and width were measured at both the widest and narrowest positions of each channel (Supplementary Fig. 4). The measured heights of Ch1, Ch2, and Ch3 were 18.94 ± 0.36, 17.84 ± 0.31, and 33.25 ± 0.39 µm, respectively, which were slightly lower than the intended values. The widest channel in Ch1, corresponding to the temporal artery and vein of the retina, measured 225.35 ± 0.05 µm, while the narrowest channel at the foveal pit measured 30.27 ± 0.25 µm. The channel width in Ch2, corresponding to the honeycomb structure, was consistent at 18.72 ± 0.35 µm. The widest and narrowest channels in Ch3 measured 134.88 ± 0.52 and 23.38 ± 0.05 µm, respectively.
A light-scattering fluid was injected into the phantom to simulate blood flow within the microfluidic channels. The light intensity in Ch3 was reduced due to transmission limitations, a behavior consistent with that of choroidal vessels in the human retina. The Ch3 showed a slightly lower intensity than the other channels, mimicking the light transmission characteristics of human retinal blood vessels (Fig. 6a). Fluorescent microbeads were incorporated into the RPE layer to match the fluorescence intensity of the retina. These microbeads, which were evenly distributed without clumping, emitted yellow-orange light when visualized using a vision camera and fluorescence microscope (Supplementary Fig. 2), further validating the functional accuracy of the phantom.
System validation: depth range and axial resolution of OCT
A cross-sectional image of the phantom obtained using the custom-built OCT system confirmed the presence of 13 distinct layers, including four high-intensity bands in the ONL layer (Fig. 6a). Despite the retinal curvature of the phantom, all the layers and microfluidic channels remained fixed and unchanged.
The image depth range (DT) was calculated using the following equation:
where Tp is the phantom thickness, N is the total number of image pixels, Np is the number of pixels corresponding to the phantom thickness, and Rp is the refractive index of the phantom. The depth range of the OCT system was measured as 1.964 mm using the phantom, which closely matched the 1.96 mm depth range measured using BK7 glass.
By measuring the depth range of an OCT image, the interface between the vitreous humor and the transparent layer of the phantom (white arrows in Fig. 6a) can serve as a reference point for measurement of axial resolution. The point spread function (Fig. 6b) from the yellow line in Fig. 6a was used to obtain an axial resolution measurement of 2.5 µm in the vitreous humor (n = 1.34), equivalent to 3.35 µm in air.
System validation: field of view
FOV is a critical parameter used for evaluating the performance of retinal imaging systems. The process of measuring FOV using the retinal phantom in a custom-built OCT system is shown in Fig. 7. The design of the microfluidic channels was compared with that of the OCT fundus image of the phantom to validate the FOV of the system.
a Enlarged version of the design of the microfluidic channels shown in Fig. 1a. b OCT fundus image of the retinal phantom. c Superimposed images (a, b). d The result of the FOV measurement. Each scale bar is 2 mm.
An enlarged version of the channel design depicted in Fig. 1a is shown in Fig. 7a, whereas the OCT fundus image projected onto the x-y plane from the 3D volumetric OCT data is presented in Fig. 7b. When superimposed (Fig. 7c), the channel design and the OCT fundus image exhibited a high degree of correspondence, confirming the accuracy of the field measurements of the OCT system. Using the fixed 4.5 mm distance between the ONH and the foveal center in the phantom design, the FOV of the custom-built OCT system was determined to be 10.2 mm (Fig. 7d). This measurement aligns with the standards defined by ISO 1094060 and ISO 1697155, thereby verifying that the OCT system provides a precise and reliable FOV for retinal imaging.
Although this method was demonstrated using OCT, it is can be applied to other retinal imaging systems, such as OCTA, FA, ICGA, and FAF. The precisely designed structure of the phantom, including the fixed distance between the ONH and foveal center, enables accurate FOV validation across different imaging modalities. The FOV of each modality can be effectively evaluated by superimposing the phantom’s design with fundus or en-face images obtained from the system. This versatility underscores the utility of the phantom in the development and standardization of a wide range of ophthalmic imaging instruments.
System validation: OCT angiography algorithm
To validate the OCTA algorithm, the retinal phantom was used to simulate fluid flow and assess the structure and connectivity of the microfluidic channels. OCTA en-face images of the three microfluidic channels were processed, along with a maximum amplitude projection (MAP) image, using a depth color map (Fig. 8a). These MAP images, computed with depths ranging from 0 µm (top of Ch1) to 350 µm in the phantom, provided clear and detailed visualizations of the upper (Ch1), middle (Ch2), and lower (Ch3) channels, as well as the ONH and fovea regions. The honeycomb structure of Ch2 was seamlessly connected, confirming the integrity of the microfluidic network of the phantom.
a En-face OCTA images of each channel obtained using the custom-built OCT. These OCTA images were computed using the SSADA algorithm. The MAP images depict a range from 0 (top of Ch1) to 350 μm. b Test conducted to assess the channel structure, fluid leakage, and reusability of the phantom. Each scale bar is 2 mm.
Further analysis of the OCTA images of Ch3 revealed black dots in the ONH region corresponding to the pillars designed to prevent channel collapse. Shading artifacts observed in Ch2 were attributed to shadowing effects caused by Ch1. Despite these minor artifacts, the OCTA algorithm effectively reconstructed the channel geometry and depth-resolved features, demonstrating its robustness in accurately visualizing the vascular network of the phantom.
The vascular connectivity visualized by the OCTA algorithm was assessed. The algorithm successfully visualized continuous fluid flow through the microfluidic channels, confirming the connectivity and structural integrity of the network across all layers. These results validate the ability of the algorithm to detect and analyze vascular structures and confirm its applicability in the evaluation of retinal imaging systems.
Channel geometry, fluid leakage, and reusability assessment
A 2% intralipid solution mixed with red ink was injected into the phantom over an extended period to evaluate the channel geometry, potential fluid leakage, and reusability of the phantom (Fig. 8b). The ink solution, which was delivered via a syringe pump, flowed sequentially through Ch3, Ch2, and Ch1 before exiting through the outlet tube (Supplementary Movie 1). Owing to the complex parallel connection between Ch1 and Ch3, the fluid required ~5–10 min to traverse all the channels. Once filled, the fluid exhibited stable flow for over 5 h without interlayer leakage or structural damage to the channels, as long as the pressure remained within a non-destructive range. After the fluid injection, ethanol was used to clean the phantom for reuse. The ethanol flowed along the same path as the ink solution and effectively washed out the residual fluid. After cleaning, the structure and channel connectivity of the phantom remained intact, demonstrating its durability and reusability over multiple sessions. These results confirmed the robustness and suitability of the phantom for repeated use under various experimental conditions.
Comprehensive validation of retinal imaging modalities using the phantom
We obtained en-face images using OCTA, ICGA, FA, and FAF to evaluate the compatibility of the retinal phantom with various clinical ophthalmic imaging instruments (Fig. 9). The results demonstrated that the phantom effectively replicated the key features of retinal imaging, confirming its versatility across multiple modalities.
a OCTA image b ICGA image c FA image d FAF image. Each scale bar is 2 mm.
In the OCTA image, all channel layers were well-distinguished, similar to the MAP image obtained using the custom-built OCT system (Fig. 8a). The honeycomb structure of Ch2 was particularly clear, highlighting the detailed vascular network of the phantom.
For FA and ICGA, the static regions mimicking tissue appeared dark, whereas the microchannels replicating blood vessels emitted light, which is consistent with angiographic imaging of the retina. The fluorescence emitted from the microfluidic channels injected with ICG and fluorescein closely resembled that observed in human retinal angiography images. In the ICGA image, Ch3 appeared above Ch1 and Ch2 because of the system software settings, reflecting depth-dependent imaging characteristics. Although Ch2 and Ch3 were less visible in the FA image, FA provided a broader FOV than the custom-built OCT system, delivering a more comprehensive image of Ch1.
FAF imaging is used to detect and capture naturally occurring fluorophores. The RPE layer emits light in the yellow-orange spectrum, whereas blood absorbs light, causing blood vessels to appear dark. The phantom consistently replicated these features; areas where red ink was injected into the channels appeared dark, whereas other regions emitted bright fluorescence (bottom right in Fig. 9). The fluorescent microbeads in the RPE layer above Ch3 contributed to the strong autofluorescence, rendering Ch3 invisible in FAF images, whereas Ch1 and Ch2 appeared as dark regions owing to absorption of light by the injected fluid.
These results confirmed the ability of the phantom to emulate both the structural and functional characteristics of the retina, demonstrating its compatibility with various imaging modalities and its utility for system development and validation.
Discussion
Phantoms that mimic tissues are invaluable tools for comparing and evaluating imaging instruments because of their consistency and insensitivity to experimental environments61,62,63,64. The concept of a super phantom, which entails combining the physical and functional properties of the target organ or tissue, represents an important step in the development of phantoms35. These phantoms replicate anatomical and optical properties while simulating physiological processes such as blood flow and tissue motion. With the commercialization of multimodal ophthalmic imaging systems, such as OCT/OCTA combined with fundus photography or multicolor SLO, there is an increasing need for multifunctional phantoms that support multiple modalities to replace traditional single-function phantoms.
Retinal phantoms have evolved incrementally over the years (Supplementary Table 1). Early phantom designs were focused on replicating retinal morphology using stacked silicone films or laser-etched foveal and optic nerve shapes20,21,22,23,24. Although these phantoms are effective for structural imaging, they lack vascular functionality and are unsuitable for angiographic imaging systems. Fluidic phantoms that incorporate capillary tubes or microfluidic channels allow for the simulation of retinal blood flow, enabling the validation of imaging techniques such as angiography and retinal oximetry25,26,27,28,29,30,31,32. However, these designs do not replicate the multi-layered structure required for advanced diagnostic systems such as OCT. Recent advancements have addressed these limitations by integrating both morphological and functional properties, thereby enabling phantoms to support a wider range of imaging modalities33,34,41,42.
In this study, we developed a super phantom that uniquely integrates structural and functional features to accurately replicate the human retina. Unlike previous phantoms that were primarily focused on either structural or functional imaging, our phantom combines both capabilities in a single, reproducible model. It includes 13 distinct retinal layers, three microfluidic channels (SV, DV, and CV) for angiographic imaging, and fluorescent microbeads in the RPE layer for FAF imaging. The layers are attached to a curved substrate, enabling the phantom to emulate the natural curvature of the retina without requiring lens correction. This comprehensive design allows for the generation of cross-sectional and thickness map images similar to those of the human retina and facilitates multimodal imaging, including OCTA, FA, ICGA, and FAF. These features establish the phantom as a pioneer tool for validation and performance assessment of ophthalmic imaging systems.
The innovative integration of structural and functional elements in the phantom positions it as a transformative solution for advancing the development of imaging systems. In addition, its ability to replicate both retinal morphology and vascular functionality provides a comprehensive platform for the evaluation and standardization of multimodal imaging instruments. For instance, the consistent thickness and vascular connectivity of the phantom offer a reliable reference for calibration of imaging systems, ensuring consistent diagnostic output across devices. Additionally, its uniform image data make it an ideal training set for AI-based algorithms such as retinal layer segmentation in OCT and vascular segmentation in fundus images. This application is particularly critical as deep learning is becoming increasingly integral to ophthalmic diagnostics, requiring robust and standardized training datasets.
However, despite its structural and functional advantages, the phantom has certain limitations, particularly in relation to imaging artifacts that may arise from interactions with specific imaging modalities. No apparent artifacts were observed in OCT (which employs an interferometer) or in FA, ICGA, and FAF (which use fluorescent filters). In contrast, eliminating surface reflections from the PDMS was challenging during color fundus photography, which does not use fluorescent filters, resulting in reflection artifacts. Additionally, when filling the phantom housing with a vitreous humor solution to mimic the vitreous body, the introduction of air bubbles during injection led to imaging artifacts. Therefore, care must be taken to avoid air bubble formation during insertion of the solution to prevent such artifacts.
To address the issue of overestimated NFL thickness in previous phantom designs34, we incorporated a transparent layer into our phantom that reduced the thickness of the NFL from 102.42 μm to 26.05 μm while preventing channel rupture under high pressure. The transparent layer also facilitates axial resolution measurements in OCT by creating a reflective interface with the simulated vitreous humor. Furthermore, this layer mimics the vitreous membrane in cross-sectional OCT images, enhancing the anatomical accuracy of the phantom and reinforcing its role as a diagnostic reference tool.
Despite the advanced features of the phantom, it has certain deliberate design limitations to enhance stability, reusability, and reproducibility. For example, the curved morphology of the ONH and fovea was not replicated to prevent structural deformation and maintain fluid channel connectivity. Spin-coating was used as the primary fabrication method to minimize thickness variations, ensuring consistent production while sacrificing the ability to replicate highly irregular layers. In addition, the minimum channel width was set to 20 µm and measured to be 18.72 ± 0.52 µm, as narrower widths made it difficult to extract the PDMS from the wafer mold. Although this width is larger than the typical diameter of human retinal capillaries (5–10 µm), the fabricated channels exhibited high dimensional consistency, with a small standard deviation. These choices enhanced the durability and usability of the phantom for repeated experiments, making it a practical for real-world applications. This trade-off allowed for robust and reusable phantom fabrication but limited the anatomical fidelity at the microcapillary level. Nonetheless, the vascular design incorporated key anatomical features, including the temporal arcade and foveal avascular zone, in order to provide a functional representation of major retinal vessels. Replicating the full complexity of retinal microvasculature remains a technical challenge, and future efforts should explore advanced fabrication methods, such as high-resolution 3D printing or lithographic techniques, to achieve finer and more anatomically accurate vascular networks.
In addition, future research directions for this phantom include several enhancements that could further expand its utility. Incorporating dynamic fluid flow at variable rates or pulsatile motion can better replicate physiological blood flow conditions. We attempted to measure the flow velocity within the microfluidic channels using Doppler ultrasonography, a common technique utilized in commercial imaging equipment. However, the measurements were limited due to insufficient resolution, preventing accurate assessment of flow velocity in the phantom and access to an imaging system capable of evaluating flow velocity within the microfluidic channels was not available. Future research should focus on integrating calibrated Doppler OCT to enable quantitative flow velocity measurements within the phantom. In addition, adding curved ONH and foveal structures without compromising stability will improve anatomical accuracy and support a wider range of diagnostic applications. Furthermore, calibrating the phantom to simulate oxygen saturation levels in retinal blood vessels could turn it into a powerful tool for evaluating angiography systems and oximetry testing65,66. Extending these capabilities could position the phantom as a universal standard for the validation of retinal imaging systems and development of algorithms.
This retinal phantom was developed to provide consistent reference data to support the calibration of various imaging systems. Such a phantom can serve as a benchmark for system evaluation and standardization. Previous reports have demonstrated variability across OCT devices (Kinkelder et al.20), underscoring the need for calibration tools for harmonizing device outputs. By providing known structural and optical properties, the phantom enables validation of image analysis algorithms and supports the development of standardized imaging protocols19,35. In addition to calibration, the phantom can also serve as an educational tool for training imaging techniques. While the present study focused on the phantom’s design and validation, further research is warranted to quantitatively evaluate its contribution to imaging performance across different devices and modalities.
This study established our retinal phantom as a cornerstone that can advance diagnostic technologies by bridging critical gaps in the validation of multimodal imaging. Its innovative design, versatile applications, and potential for future enhancements make it a transformative tool for clinical and industrial applications. As imaging technologies continue to evolve, this phantom will offer a robust platform for setting new benchmarks for retinal imaging.
CONCLUSION
This study presents the development of a comprehensive retinal phantom that bridges critical gaps in multimodal ophthalmic imaging validation. By integrating both morphological and functional characteristics of the human retina, the phantom establishes a novel standard for system evaluation and calibration. Constructed using PDMS and TiO2 powder, the phantom replicates the layered structure and curvature of the retina with high accuracy. It includes thirteen distinct retinal layers, modeled through spin-coated multilayered thin films, and features three vascular networks—superficial, deep, and choroidal vessels—designed from conventional FA and ICGA images. The incorporation of fluorescent microbeads into the RPE layer further enables FAF imaging, replicating retinal autofluorescence properties.
The versatility of the phantom was validated using various imaging modalities, including OCT, OCTA, FA, ICGA, and FAF, and by employing both a custom-built OCT system and a clinical ultrawide FOV OCT system combined with multicolor SLO. The phantom accurately mimicked cross-sectional retinal images, vascular connectivity, and fluorescence characteristics, demonstrating its compatibility across multiple imaging systems. Additionally, it facilitated the measurement of key system parameters, such as axial resolution, depth range, FOV calibration, and fluorescence image quality, proving its utility as a benchmark tool for performance assessment.
By addressing the limitations of previous retinal phantoms, the model developed in this study provides a practical approach to combining structural and functional imaging validation into a single reproducible tool. In addition, it offers consistent and reliable data for calibrating imaging devices, validating new algorithms, and training AI models for retinal diagnostics. The uniformity and stability of the phantom ensure its reusability, making it a practical resource for both research and clinical applications.
Future developments that could enhance the capabilities of the phantom include integration of dynamic fluid flow, simulation of retinal oxygen saturation, and inclusion of more complex anatomical features, such as irregular foveal or ONH morphology. These improvements will further expand the applicability of the phantom and solidify its role as a cornerstone in advancing ophthalmic imaging technologies.
This retinal phantom developed in this study not only serves as a versatile tool for validation of imaging systems but also contributes to the establishment of new benchmarks for standardization of retinal imaging system. As the phantom was developed by combining innovation, reproducibility, and practicality, it serves as a transformative tool for clinical diagnostics, development of imaging systems, and optimization of algorithms.
Data availability
The experiment data supporting this study can be provided upon reasonable request by contacting the corresponding author. The OCT image of the human retina shown in Fig. 5a was obtained from the publicly available OCTDL dataset (image number: no_1391081_3) published by Kulyabin et al.59, which is accessible via https://arxiv.org/abs/2312.08255 and https://doi.org/10.1038/s41597-024-03182-7.
References
Abràmoff, M. D., Garvin, M. K. & Sonka, M. Retinal imaging and image analysis. IEEE Rev. Biomed. Eng. 3, 169–208 (2010).
Badar, M., Haris, M. & Fatima, A. Application of deep learning for retinal image analysis: a review. Comput. Sci. Rev. 35, 100203 (2020).
Drag, S., Dotiwala, F. & Upadhyay, A. K. Gene therapy for retinal degenerative diseases: progress, challenges, and future directions. Investig. Ophthalmol. Vis. Sci. 64, 39 (2023).
Khaboushan, A. S. et al. Cell therapy for retinal degenerative disorders: a systematic review and three-level meta-analysis. J. Transl. Med. 22, 227 (2024).
Sharp, P. F. & Manivannan, A. The scanning laser ophthalmoscope. Phys. Med. Biol. 42, 951 (1997).
Mainster, M. A., Desmettre, T., Querques, G., Turner, P. L. & Ledesma-Gil, G. Scanning laser ophthalmoscopy retroillumination: applications and illusions. Int. J. Retin. Vitr. 8, 71 (2022).
Pinhas, A. et al. In vivo imaging of human retinal microvasculature using adaptive optics scanning light ophthalmoscope fluorescein angiography. Biomed. Opt. Express 4, 1305–1317 (2013).
Spaide, R. F., Klancnik, J. M. & Cooney, M. J. Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol. 133, 45–50 (2015).
Slakter, J. S., Yannuzzi, L. A., Guyer, D. R., Sorenson, J. A. & Orlock, D. A. Indocyanine-green angiography. Curr. Opin. Ophthalmol. 6, 25–32 (1995).
Desmettre, T., Devoisselle, J. M. & Mordon, S. Fluorescence properties and metabolic features of indocyanine green (ICG) as related to angiography. Surv. Ophthalmol. 45, 15–27 (2000).
Schmitz-Valckenberg, S., Holz, F. G., Bird, A. C. & Spaide, R. F. Fundus autofluorescence imaging: review and perspectives. Retina 28, 385–409 (2008).
Schmitz-Valckenberg, S. et al. Fundus autofluorescence imaging. Prog. Retin. Eye Res. 81, 100893 (2021).
Huang, D. et al. Optical coherence tomography. Science 254, 1178–1181 (1991).
Drexler, W. & Fujimoto, J. G. Optical Coherence Tomography: Technology and Applications. (Springer Science & Business Media, 2008).
De Carlo, T. E., Romano, A., Waheed, N. K. & Duker, J. S. A review of optical coherence tomography angiography (OCTA). Int. J. Retin. Vitr. 1, 1–15 (2015).
Spaide, R. F., Fujimoto, J. G., Waheed, N. K., Sadda, S. R. & Staurenghi, G. Optical coherence tomography angiography. Prog. Retin. Eye Res. 64, 1–55 (2018).
Ilginis, T., Clarke, J. & Patel, P. J. Ophthalmic imaging. Br. Med. Bull. 111, 77–78 (2014).
Kwon, I. H., Kim, H.-S., Kim, D. Y., Lee, H.-J. & Lee, S.-W. Utilizing optical phantoms for biomedical-optics technology: Recent advances and challenges. Curr. Opt. Photonics 8, 327–344 (2024).
Park, K. S. et al. Phase stable swept-source optical coherence tomography with active mode-locking laser for contrast enhancements of retinal angiography. Sci. Rep. 11, 16636 (2021).
de Kinkelder, R., de Bruin, D. M., Verbraak, F. D., van Leeuwen, T. G. & Faber, D. J. Comparison of retinal nerve fiber layer thickness measurements by spectral-domain optical coherence tomography systems using a phantom eye model. J. Biophotonics 6, 314–320 (2013).
Baxi, J. et al. Retina-simulating phantom for optical coherence tomography. J. Biomed. Opt. 19, 021106 (2014).
Lee, G. C., Smith, G. T., Agrawal, M., Leng, T. & Ellerbee, A. K. Fabrication of healthy and disease-mimicking retinal phantoms with tapered foveal pits for optical coherence tomography. J. Biomed. Opt. 20, 085004 (2015).
Agrawal, A. et al. Optic nerve head measurements with optical coherence tomography: a phantom-based study reveals differences among clinical devices. Investig. Ophthalmol. Vis. Sci. 57, OCT413–OCT420 (2016).
Cao, Z. et al. Model eyes with curved multilayer structure for the axial resolution evaluation of an ophthalmic optical coherence tomography device. J. Innov. Opt. Health Sci. 11, 1850013 (2018).
Chen, Z., Milner, T. E., Dave, D. & Nelson, J. S. Optical Doppler tomographic imaging of fluid flow velocity in highly scattering media. Opt. Lett. 22, 64–66 (1997).
Yazdanfar, S. & Izatt, J. A. Self-referenced Doppler optical coherence tomography. Opt. Lett. 27, 2085–2087 (2002).
Mordant, D. J. et al. Validation of human whole blood oximetry, using a hyperspectral fundus camera with a model eye. Investig. Ophthalmol. Vis. Sci. 52, 2851–2859 (2011).
Jeong, H. et al. Ultra-fast displaying spectral domain optical Doppler tomography system using a graphics processing unit. Sensors 12, 6920–6929 (2012).
Tokayer, J., Jia, Y., Dhalla, A.-H. & Huang, D. Blood flow velocity quantification using split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Biomed. Opt. Express 4, 1909–1924 (2013).
Damodaran, M., Amelink, A. & De Boer, J. F. Optimal wavelengths for subdiffuse scanning laser oximetry of the human retina. J. Biomed. Opt. 23, 086003 (2018).
Luu, L., Roman, P. A., Mathews, S. A. & Ramella-Roman, J. C. Microfluidics based phantoms of superficial vascular network. Biomed. Opt. Express 3, 1350–1364 (2012).
Hayakawa, T. et al. Retinal vessel model fabricated on a curved surface structure for a simulation of microcannulation. ROBOMECH J. 3, 1–8 (2016).
Zhao, X. et al. Rapid prototyping of a retinal multivascular network phantom for optical retinal vascular imaging equipment evaluation. Biomed. Opt. Express 15, 4253–4263 (2024).
Lee, H.-J., Samiudin, N. M., Lee, T. G., Doh, I. & Lee, S.-W. Retina phantom for the evaluation of optical coherence tomography angiography based on microfluidic channels. Biomed. Opt. Express 10, 5535–5548 (2019).
Manohar, S., Sechopoulos, I., Anastasio, M. A., Maier-Hein, L. & Gupta, R. Super phantoms: advanced models for testing medical imaging technologies. Commun. Eng. 3, 73 (2024).
Demirkaya, N. et al. Effect of age on individual retinal layer thickness in normal eyes as measured with spectral-domain optical coherence tomography. Investig. Ophthalmol. Vis. Sci. 54, 4934–4940 (2013).
den Haan, J. et al. Retinal thickness correlates with parietal cortical atrophy in early-onset Alzheimer’s disease and controls. Alzheimer Dement.10, 49–55 (2018).
Chua, J. et al. Age-related changes of individual macular retinal layers among Asians. Sci. Rep. 9, 20352 (2019).
Brandl, C. et al. Retinal layer thicknesses in early age-related macular degeneration: results from the German AugUR Study. Investig. Ophthalmol. Vis. Sci. 60, 1581–1594 (2019).
Jáñez-Escalada, L. et al. Spatial analysis of thickness changes in ten retinal layers of Alzheimer’s disease patients based on optical coherence tomography. Sci. Rep. 9, 13000 (2019).
Lee, H.-J., Lee, S.-W., Samiudin, N. M. & Doh, I. Retinal phantom mimicking superficial vascular netwoarks and full retinal layers for OCT and OCT-angiography. Proc. SPIE 11630, 116300N (2021).
Lee, H.-J., Lee, S.-W., Samiudin, N. M. & Doh, I. Retinal phantom applicable to OCT, OCT-angiography, and fundus angiography. Proc. SPIE 11633, 116330B (2021).
Srinidhi, C. L., Aparna, P. & Rajan, J. Recent advancements in retinal vessel segmentation. J. Med. Syst. 41, 1–22 (2017).
Jonas, R. A. et al. Optic disc-fovea distance, axial length and parapapillary zones. The Beijing Eye Study 2011. PLoS ONE 10, e0138701 (2015).
Hayreh, S. S. In vivo choroidal circulation and its watershed zones. Eye 4, 273–289 (1990).
Spaide, R. F. et al. Venous overload choroidopathy: a hypothetical framework for central serous chorioretinopathy and allied disorders. Prog. Ret. Eye Res. 86, 100973 (2022).
Verma, A., Bacci, T., Sarraf, D., Freund, K. B. & Sadda, S. R. Vortex vein imaging: what can it tell us? Clin. Ophthalmol. 15, 3321–3331 (2021).
Branchini, L. A. et al. Analysis of choroidal morphologic features and vasculature in healthy eyes using spectral-domain optical coherence tomography. Ophthalmology 120, 1901–1908 (2013).
Spaide, R. F. & Curcio, C. A. Anatomical correlates to the bands seen in the outer retina by optical coherence tomography: literature review and model. Retina 31, 1609–1619 (2011).
Park, J. C. et al. Objective analysis of hyperreflective outer retinal bands imaged by optical coherence tomography in patients with Stargardt disease. Investig. Ophthalmol. Vis. Sci. 56, 4662–4667 (2015).
Ross, D. H. et al. RefMoB, a reflectivity feature model-based automated method for measuring four outer retinal hyperreflective bands in optical coherence tomography. Investig. Ophthalmol. Vis. Sci. 56, 4166–4176 (2015).
Yao, X., Son, T., Kim, T.-H. & Le, D. Interpretation of anatomic correlates of outer retinal bands in optical coherence tomography. Exp. Biol. Med. 246, 2140–2150 (2021).
Singh Kushwaha, S., Patro, N. & Kumar Patro, I. A sequential study of age-related lipofuscin accumulation in hippocampus and striate cortex of rats. Ann. Neurosci. 25, 223–233 (2019).
McNabb, R. P. et al. Wide-field whole eye OCT system with demonstration of quantitative retinal curvature estimation. Biomed. Opt. Express 10, 338–355 (2019).
ISO 16971, Ophthalmic instruments—Optical coherence tomograph for the posterior segment of the human eye (International Organization for Standardization, 2015).
Lee, S.-W. et al. Ultrahigh-resolution spectral domain optical coherence tomography based on a linear-wavenumber spectrometer. J. Opt. Soc. Korea 19, 55–62 (2015).
Lee, H.-J. & Lee, S.-W. Partial spectrum detection and super-gaussian window function for ultrahigh-resolution spectral-domain optical coherence tomography with a linear-k spectrometer. Curr. Opt. Photonics 7, 73–82 (2023).
Jia, Y. et al. Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt. Express 20, 4710–4725 (2012).
Kulyabin, M. et al. OCTDL: Optical Coherence Tomography Dataset for Image-Based Deep Learning Methods. Sci. Data 11, 365 (2024).
ISO 10940, Ophthalmic instruments—Fundus cameras (International Organization for Standardization, 2009).
Gröhl, J. et al. Moving beyond simulation: data-driven quantitative photoacoustic imaging using tissue-mimicking phantoms. IEEE Trans. Med. Imaging 43, 1214–1224 (2023).
Hacker, L. et al. A copolymer-in-oil tissue-mimicking material with tuneable acoustic and optical characteristics for photoacoustic imaging phantoms. IEEE Trans. Med. Imaging 40, 3593–3603 (2021).
Cormack, J. M. et al. Focused shear wave beam propagation in tissue-mimicking phantoms. IEEE Trans. Biomed. Eng. 71, 621–630 (2024).
Kijanka, P., Vasconcelos, L., Mandrekar, J. & Urban, M. W. Evaluation of robustness of S-transform based phase velocity estimation in viscoelastic phantoms and renal transplants. IEEE Trans. Biomed. Eng. 71, 954–966 (2024).
Song, W. et al. Visible light optical coherence tomography angiography (vis-OCTA) facilitates local microvascular oximetry in the human retina. Biomed. Opt. Express 11, 4037–4051 (2020).
Gao, F., Peng, Q., Feng, X., Gao, B. & Zheng, Y. Single-wavelength blood oxygen saturation sensing with combined optical absorption and scattering. IEEE Sens. J. 16, 1943–1948 (2016).
Acknowledgements
This work was supported in part by Korea Medical Device Development Fund grants funded by the Korean government (Ministry of Science and ICT, Ministry of Trade, Industry and Energy; Ministry of Health & Welfare; Ministry of Food and Drug Safety) (Project Number: KMDF_PR_20200901_0026) and the Research Initiative Program (CRC24021-000) funded by the National Research Council of Science and Technology, Ministry of Science and ICT. This study was also supported by the Development of Measurement Standards and Technology for Biomaterials and Medical Convergence, funded by the Korea Research Institute of Standards and Science (KRISS-GP2025-0007-02 and KRISS-GP2025-0007-03).
Author information
Authors and Affiliations
Contributions
H.-J. Lee and S.-W. Lee conceptualized the project. H.-J. Lee and I. Doh developed the methodology. All the authors conducted the investigations. T. Lee and S.-W Lee acquired the funding and supervised the project. H.-J. Lee wrote the original manuscript draft. All the authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Engineering thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: [Sandra Rugonyi] and [Rosamund Daw]. [A peer review file is available.]
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, HJ., Lee, T.G., Doh, I. et al. Design and application of a realistic and multifunctional retinal phantom for standardizing ophthalmic imaging systems. Commun Eng 4, 134 (2025). https://doi.org/10.1038/s44172-025-00475-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44172-025-00475-6