Abstract
The occurrence of treatment resistance in women with postpartum depression (PPD) and risk factors for treatment resistance remain less studied. This study aimed to determine the rate of treatment resistance and the associated risk factors among women with PPD in a nationwide setting. Here we conducted a nationwide register-based cohort study of 58,618 patients with a first-ever PPD during 2006–2021 in Sweden. Information on demographics, pregnancy characteristics, pre-existing physical and psychiatric conditions and treatment was retrieved from Swedish national registers. The outcome was treatment-resistant PPD (TRPPD) within 1 year following PPD diagnosis. Associations between potential risk factors and TRPPD were assessed using multivariable Poisson regression. Among the 58,618 patients with PPD, 3,522 (6.0%) met the criteria for TRPPD during 1 year after PPD diagnosis. Lower educational level, lower household income, being non-cohabiting, smoking in early pregnancy, delivery by cesarean section, pre-existing physical conditions and pre-existing psychiatric disorders were significantly associated with a higher risk of TRPPD. In addition, patients with two births (versus primiparity) or with a prior premenstrual disorder had a lower risk of TRPPD. Treatment resistance in patients with PPD is common and is notably associated with specific demographic and clinical profiles. These findings may provide grounds for practical risk assessment at PPD diagnosis and highlight the need for personalized management strategies.
Similar content being viewed by others
Main
Postpartum depression (PPD) impacts millions of new mothers worldwide, with approximately one out of five mothers experiencing a depressive episode within the first months after delivery1. PPD has substantial influence on the affected patients in both the short and the long run2, resulting in negative consequences (for example, increased suicidal behavior3 and premature death4) and increased societal burden5. Psychotherapy is the first-line treatment for mild PPD, while pharmacotherapy is often indicated for patients with moderate and severe symptoms6. Although selective serotonin reuptake inhibitors are the most prescribed and first choice medications for PPD6,7, evidence from randomized clinical trials8 shows that the response rates are 43%–87% and remission rates are 37%–65% after 6–8 weeks of treatment.
Most existing studies define treatment resistance as no symptom remission after two or more pharmacologically different antidepressant treatments at an adequate dose and duration9. Clinical studies on non-perinatal depression report that a substantial number of patients10 (~30%) develop treatment resistance, while epidemiological studies indicate 11%–15% of patients are classified with treatment resistance7,11,12. Treatment-resistant depression has been linked to negative personal and societal outcomes11,12,13 and excess mortality12,14. Varying factors, including comorbid psychiatric conditions, sociodemographic vulnerability (for example, younger age, unemployment), medical conditions and greater severity of depression, have been associated with treatment-resistant depression15.
By contrast, limited data exist regarding how often treatment resistance occurs in patients with PPD, a subset of depression that may involve distinct pathophysiological mechanisms16, such as reproductive hormonal fluctuations17. In addition, PPD is often undertreated6,18 due to concerns about the potential adverse effects of pharmacotherapy on the infant, potentially leading to different clinical profiles and courses compared with non-perinatal depression. In addition, the clinical prognosis of PPD might be attributable to pregnancy characteristics and outcomes19,20,21, which are not relevant in the context of non-perinatal depression. The exclusive report, to our knowledge, a descriptive study in the United States with 3.2 million pregnant women, reported that about 5% of the patients with perinatal depression developed treatment resistance within 1 year22. However, the rates of PPD (2.5%) and treatment resistance in this study may have been underestimated as the study population consisted of only patients with commercial insurance, a population with better socioeconomic status22. Given the high disease burden of PPD3,4 and the occurrence of treatment resistance, which potentially leads to worse prognosis, it is critical to identify risk factors that influence the treatment response. However, to our knowledge, risk factors for treatment resistance have not been well investigated.
To address these knowledge gaps, we leveraged a national register-based cohort of women diagnosed with PPD in Sweden to assess the prevalence of treatment resistance and to identify potential risk factors associated with treatment resistance.
Results
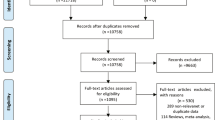
We identified 64,150 patients with a diagnosis of first-ever PPD. After excluding 5,441 patients with a record of antepartum depression and 91 with psychosis, bipolar disorder or dementia between childbirth and PPD diagnosis, 58,618 patients remained for the final analysis (Fig. 1). Among them (mean age 30.8 years, s.d. 5.3 years), 50,679 (86.5%) had received treatments (antidepressants or add-on medications or electroconvulsive therapy (ECT) or repetitive transcranial magnetic stimulation (rTMS)), and 3,522 (6.0%) fulfilled the criteria of TRPPD within 1 year after PPD diagnosis.
Pregnant women were identified from the Swedish Medical Birth Register. Depression was ascertained from the National Patient Register (Inpatient/Outpatient specialized care), Prescribed Drug Register and five regional primary care centers.
Demographic and lifestyle factors
Compared with patients without TRPPD, patients with TRPPD were more likely to be born outside Sweden (adjusted risk ratio (RR) for Europe versus Sweden: 1.25, 95% confidence interval (CI) 1.09–1.42; non-Europe versus Sweden: 1.15, 95% CI 1.04–1.28) and reside in the north of the country (northern versus southern: 1.17, 1.05–1.29), and were less likely to be diagnosed with PPD after 2015 (0.85, 0.78–0.93; Table 1). Patients with lower educational attainment (<9 versus >12 y: 1.61, 1.45–1.79) or lower household income level (lowest 20% versus top 20%: 1.35, 1.20–1.52) and being non-cohabiting (1.27, 1.15–1.42) had higher risks of TRPPD. The risk of TRPPD was also higher in patients who were smoking in early pregnancy (RRs for 1–9 cigarettes day−1: 1.45, 1.30–1.63; ≥10 cigarettes day−1: 1.53, 1.28–1.84) than in non-smoking patients (Table 1).
Pregnancy characteristics
We observed a lower risk of TRPPD in patients having two births (RR 0.89, 0.82–0.96) but not in patients having three or more births, compared with primiparous patients (Table 2). Patients with cesarean section (RR 1.15, 1.07–1.25) and patients with a preterm delivery (32–36 weeks; RR 1.23, 1.07–1.40) were at a higher risk of TRPPD. No associations were noted for other pregnancy outcomes, including multiple gestation, birth weight, Apgar score at 5 min and stillbirth (Table 2).
Physical and psychiatric conditions existing before PPD
Pre-existing physical conditions (RRs for Charlson comorbidity index = 1: 1.28, 1.16–1.42; Charlson comorbidity index ≥ 2: 1.42, 1.17–1.73) were associated with higher risks of TRPPD, but no association was found with hypertensive disorders or diabetes when assessed separately (Table 3). In addition, patients with any pre-existing psychiatric disorder were at a higher risk of TRPPD, with RR of 2.21 (2.06–2.37). The RRs for specific types of psychiatric disorders ranged from 1.69 (1.53–1.87) for substance abuse to 8.49 (7.58–9.52) for psychotic disorders (Table 3). Patients with a prior premenstrual disorder had a lower risk of TRPPD (0.71, 0.59–0.86).
Sensitivity analyses
We observed comparable associations when restricted to PPD patients without any pre-existing psychiatric disorders, except that the association with having two births was no longer present, while a higher risk of TRPPD was noted among patients with three or more births (Supplementary Table 1). Similar results were obtained when restricting the analysis to PPD ascertained through clinical diagnosis only or restricting to counties with primary care data available (Supplementary Table 2). Using the two alternative definitions of TRPPD also yielded comparable results (Supplementary Table 2). Last, in the mutually adjusted model, we found all primary factors remained statistically significant despite the associations being somewhat attenuated (Supplementary Table 2).
Discussion
In this large, population-based cohort of patients with first-ever diagnosed PPD, we found the occurrence of treatment resistance to be common in patients with PPD. Moreover, our study pinpoints that multiple demographic and clinical profiles, including lower educational level, lower income, cigarette smoking, primiparity, cesarean section, preterm delivery (32–36 weeks) and pre-existing physical and psychiatric conditions, are independently associated with a higher risk of developing TRPPD.
In this study, the proportion of patients with PPD who fulfilled the predefined TRPPD was 6.0%. Even with stricter criteria applied in our study, the proportion of treatment resistance was higher than the proportion reported in a previous study conducted in the United States22. However, the numbers of peripartum depression (2.5%) and treatment resistance (5.0%) in the US study might have been underestimated because the study included only insured people and excluded women with prior psychosis, bipolar disorder or dementia22. By contrast, the proportion of treatment resistance in our study was lower than that reported among patients with non-perinatal depression in population-based studies in Sweden12 (13%) and Denmark7 (15%), and even lower than the proportions reported in clinical trials10 (30%). Several factors may explain the lower treatment resistance in PPD compared with non-perinatal depression. First, compared with patients with non-perinatal depression, some patients with PPD may hesitate to continue or initiate pharmacotherapy or choose psychotherapy over pharmacotherapy due to concerns of potential harmful effects on the infant through breastfeeding6; therefore, PPD patients may receive less pharmacotherapy during the study period. Indeed, our data showed that 13.5% of patients did not receive any pharmacological or ECT/rTMS treatment during the postpartum year. Thus, the low proportion might indicate that women with PPD are somewhat untreated or undertreated, which is supportive in previous reports6,23. Second, in 2010, a screening for PPD was introduced in Swedish national guidelines. Therefore, patients diagnosed from 2010 onward might have less severe symptoms; and timely treatments/interventions which, if successful, may contribute to the lower treatment resistance in our study. Our results indicate lower risks of TRPPD in patients diagnosed after 2010 than before 2010, which also supports this explanation. In addition, compared with previous studies in which patients were ascertained through inpatient/outpatient care diagnosis7,12, our study including primary care diagnosis may have captured more mild or moderate cases, leading to lower treatment resistance.
We found that several demographic factors, including lower educational attainment or household income, being non-cohabiting, being born outside Sweden and living in northern Sweden, were associated with higher risk of TRPPD. One potential interpretation is that socioeconomically disadvantaged patients may have less accessibility to healthcare resources and social support, resulting in deteriorating disease severity and treatment response. Similar findings have been observed for non-perinatal depression in previous studies24. For example, Gronemann et al.25 found that patients with depression becoming unemployed were at higher risk of treatment resistance. In addition, substantial evidence has indicated that low socioeconomic status was associated with elevated levels of depressive symptoms26,27, which also supports our findings.
Moreover, the results of higher risk of TRPPD among patients who were smoking is in line with findings in people with non-perinatal depression24. Possibly, smoking individuals have more severe depressive symptoms28, or smokers may receive bupropion for smoking cessation29; thus, they might receive more antidepressants while not being depressed. Besides, smoking is considered an indicator of unfavorable socioeconomic status in the Swedish female population30. These factors may explain the higher risk of TRPPD among smoking patients. It is also possible that smoking may interact with drug metabolism and affect treatment effect31, leading to more antidepressant treatments.
Although some studies indicate that multiparity may predict prolonged depressive symptoms19, most studies indicate that multiparity is associated with lower risk of PPD32. By contrast, in our data, PPD patients with two births (the most common type in Sweden) had a lower risk of TRPPD but not among patients with three or more births. However, in the sensitivity analysis excluding patients with prior psychiatric disorders, as well as in the mutually adjusted analysis, a higher risk of TRPPD was observed only among patients with three or more births. Thus, more research is needed to corroborate our findings on the link between parity and TRPPD. In addition, PPD patients who gave birth through cesarean section were more likely to develop PPD33. Cesarean section, particularly acute cesarean section, can lead to a negative delivery experience20, which may drive these patients to more severe symptoms of postpartum depression21. Preterm delivery at 32–36 weeks was associated with a higher risk of TRPPD. It is plausible that preterm birth entails higher risk of neonatal complications and stressful experiences such as admission to neonatal intensive care units34, which may contribute to worse prognosis of PPD. However, very preterm birth (22–31 weeks) was not associated with TRPPD, which argues against such explanation. In addition, low birth weight, another risk factor for neonatal complications, was not associated with TRPPD. We did not note a material difference in treatment response between patients with a live birth and those with a stillbirth, although the etiology of PPD may differ between these two groups, as depression following a stillbirth may be more closely related to grief over the loss.
Our finding on the higher risk of TRPPD among patients with previous physical conditions is also in line with studies on non-perinatal depression15,35. This could be attributed to the chronic inflammation, or altered hepatic or renal functions, or drug interactions with antidepressant due to these physical comorbidities31. Recent findings indicate non-responders to antidepressant treatment have higher inflammatory levels compared with responders36. Of note, we did not observe a higher risk of TRPPD among patients with gestational diabetes or pre-eclampsia, although mothers with such conditions generally have a higher rate of PPD32,37.
Pre-existing psychiatric conditions have been repeatedly and consistently associated with increased risk of treatment resistance in patients with non-perinatal depression15,38. In the study by Cepeda et al.22, women with perinatal depression who developed treatment resistance had higher rates of psychiatric comorbidities in the previous year than those who did not develop treatment resistance. However, the result is from descriptive comparisons without confounding adjustment22. In this study, we illustrated that pre-existing psychiatric conditions, both any psychiatric disorder and a range of specific psychiatric disorders, were associated with a higher risk of TRPPD. Patients with pre-existing psychiatric conditions might have received antidepressants to treat depression or conditions other than depression, and therefore could have become somewhat tolerant to antidepressants intended for their PPD39. By contrast, patients with prior premenstrual disorders were at a lower risk of TRPPD. This subgroup of patients are sensitive to normal changes of gonadal steroid levels40 and may represent a ‘hormone-sensitive’ phenotype of PPD17. Theoretically, in this subgroup, depressive symptoms are triggered by the normal drop of gonadal hormones after delivery, but symptoms may remit when hormone levels begin rising again. Future studies, however, are needed to elucidate the role of hormonal factors in treatment response of PPD. It is worth noting that when patients with a psychiatric history were excluded, we saw similar results for risk factors, including socioeconomic status, parity and prior physical comorbidities. This suggests these risk factors are independent of prior psychiatric conditions.
Of note, we found that most factors associated with TRPPD in our study overlap with the psychosocial risk factors for PPD. A probable explanation could be that PPD patients with these factors are more likely to develop more severe depression or require more intensive treatment due to pre-existing mental conditions, which increases their chances of developing treatment resistance or being classified as treatment resistant.
Several strengths of our study assure robustness of our findings. The nationwide population-based sample together with high-quality, prospectively collected healthcare register data enabled a representative sample of treatment resistance among women with PPD and a comprehensive assessment of various risk factors for TRPPD. Our study has weaknesses. First, in Sweden, depression is often managed in primary care41. While we used antidepressant prescriptions for identification, we might have missed mild PPD cases who were not prescribed pharmacotherapy as we only had primary care data from five counties. Yet this concern was addressed by the almost identical results when repeating the analysis using data from only these counties. Second, psychotherapy is the first-line treatment for mild to moderate depression in Sweden. However, due to lacking information on psychotherapy, the influence of psychotherapy on treatment response was not considered in our study. Future studies incorporating both psychotherapy and pharmacotherapy are highly warranted. Third, we may have some degree of misclassification of treatment resistance as our definition did not consider the dose of treatment. However, largely comparable results were obtained when utilizing two alternative definitions of treatment resistance. In addition, misclassification of treatment resistance may occur among patients with prior psychiatric disorders as these patients could restart or continue treatments for their prior illness during the postpartum period, leading to overestimation of TRPPD. The concern, however, was partially alleviated as we obtained similar results when excluding PPD patients who had any pre-existing psychiatric conditions. Last, our results might not be generalized to populations outside Sweden where different healthcare service systems apply.
In conclusion, based on a nationwide sample, our study shows that the prevalence of treatment resistance was common in patients with PPD. We found primiparous patients with lower socioeconomic status, cigarette smoking and pre-existing physical and psychiatric conditions are at a higher risk of developing treatment resistance. These findings may provide grounds for practical risk assessment at PPD diagnosis and highlight the need for personalized management strategies (for example, actively monitoring treatment response to aid earlier medication adjustments), particularly for patients identified as at risk for worse prognosis.
Methods
Data resource
This study was based on nationwide Swedish population and healthcare registers. Individuals were linked through the unique personal identification number that is assigned to every resident in Sweden. The Medical Birth Register (MBR)42 collects nationwide data on pregnancy, delivery and neonatal characteristics in Sweden from 1973. The National Patient Register43 includes nationwide information on inpatient specialized care since 1987 and on hospital-based outpatient specialized care since 2001 (coverage >80%). The Prescribed Drug Register44 includes all prescribed drugs dispensed at pharmacies since July 2005. We also included primary care data from five counties (Stockholm, Skåne, Uppsala, Värmland and Västra Götaland), which accounts for 58%–62% of women of reproductive age in Sweden during 2001–2021. The Longitudinal Integration Database for Health Insurance and Labor Market45 contains sociodemographic information, for example, income and education level, on a yearly basis for Swedish residents aged ≥16 years since 1990.
Study design and study population
We conducted a nationwide, register-based cohort study leveraging a range of national health registers in Sweden. We first identified 1,042,423 women with 1,784,326 pregnancies from the MBR who gave birth during 2006–2021 in Sweden. After excluding 24,997 duplicate records of the same pregnancy due to multiple births and 31,090 pregnancies with erroneous records (that is, 31,083 possibly wrong identification, 7 died before childbirth), the study base consisted of 1,728,239 pregnancies from 1,022,698 women. We identified women with PPD from the study base and included only their first-ever PPD in cases of multiple PPD records. Further, we excluded patients with a record of antepartum depression for the pregnancy studied. Patients with psychosis, bipolar disorder or dementia between childbirth and PPD diagnosis were also excluded to ensure a sample of patients with definitive depression diagnosis.
The study was approved by the Swedish Ethics Review Authority (2018/1515-31 and 2021-02775). Informed consent from each participant was waived by Swedish law when using register-based data.
Ascertainment of PPD
In line with previous studies46, we defined PPD as the first record of depression diagnosis in both specialist care (nationwide) and primary care (five counties available), or as a first filled prescription of antidepressants, during the first year after birth (identification codes are listed in Supplementary Table 3). While women with PPD can have a prior depression, it was the first identified PPD that was included in our analysis. Although in diagnostic definition47, PPD is defined as a depressive episode within weeks after childbirth, the timing is often extended to encompass the first year postpartum in research settings as the clinical diagnosis may be substantially delayed.
Ascertainment of TRPPD
The outcome was treatment resistance occurring within 1 year following PPD diagnosis, that is, TRPPD. There is no consensus on definition of treatment-resistant depression. In addition, it is challenging to determine the reasons for medication changes or discontinuation in registry data as they could be due to side effects, lack of effectiveness or disease remission. Therefore, we adapted a treatment-resistant depression definition in previous studies22, which counts the number of antidepressants (that is, ≥3 different antidepressants) during 12 months. This definition has been proved to have better performance in discriminating individuals with and without evidence of treatment resistance than definitions that are based on adequacy of treatment dose and duration, and the definition achieves consistent performance across databases48. Besides, following Lundberg et al.11, we also considered use of add-on medication (lithium, risperidone, olanzapine, aripiprazole and quetiapine ( > 100 mg), which are recommended by guidelines for treatment-resistant depression49), or ECT or rTMS as treatment resistance. Briefly, we defined TRPPD as use of ≥3 different antidepressants, or add-on medication, or ECT or rTMS during the first year after the PPD diagnosis. Identification codes and data sources are listed in Supplementary Table 3.
Potential risk factors for TRPPD
We sourced registers for demographics, lifestyle factors and pregnancy characteristics as potential risk factors. Maternal educational attainment and household income were obtained from the Longitudinal Integration Database for Health Insurance and Labor Market. Using the MBR, we retrieved information on maternal age, civil status, maternal birth country, maternal residency in Sweden, calendar year at delivery, and cigarette smoking, snus use and body mass index in early pregnancy. Pregnancy characteristics and pregnancy outcomes, including parity, multiple gestation, delivery mode, gestational length, birth weight, Apgar score at 5 min, stillbirth and gestational hypertensive or diabetic disorders, were also obtained from the MBR.
We considered pre-existing physical and psychiatric conditions (including any or specific categories of psychiatric disorders) before PPD diagnosis as potential risk factors and identified them from the National Patient Register. An adapted Charlson comorbidity index for register-based research in Sweden (we excluded diabetes as this category was listed separately in this study) was used as a proxy for physical comorbidity burden50. An index of zero indicates no comorbidity; the higher the index, the greater the comorbidity burden. Identification codes of psychiatric conditions are listed in Supplementary Table 3.
Statistical analysis
Main analysis
We calculated the absolute risk of TRPPD using the number of TRPPD divided by total number of PPD patients during 1 year after PPD diagnosis. We used Poisson regression models to assess the potential risk factors associated with TRPPD by estimating RRs and 95% CIs, contrasting the absolute risk of TRPPD in the exposed group with that in the reference group. We started with univariable analysis (Model 1) followed by multivariable analysis (Model 2), which adjusted for maternal age, educational level, calendar year, residential region, maternal country of birth, parity and multiple gestation, whenever applicable. To address the concern of multiple testing as many risk factors were being tested, we collected the P values of all estimates and produced corresponding corrected P values using the false discovery rate (that is, Benjamini-Hochberg) method.
Sensitivity analyses
We performed several sensitivity analyses to test the robustness of our findings. First, a prior psychiatric condition may be an indication for treatments, including antidepressants, leading to being more likely to fulfill the criteria of TRPPD. Therefore, we limited the analyses to patients without a history of any psychiatric disorders. Second, some PPD cases were identified through antidepressants, which could have been prescribed for other psychiatric conditions (for example, anxiety disorders). We thus restricted the analysis to patients with a clinically confirmed diagnosis of PPD only. Third, we limited the analysis to counties with primary care data. Fourth, we used an alternative definition of TRPPD requiring at least one antidepressant before the add-on medication or ECT/rTMS: namely, use of ≥3 different antidepressants, or first antidepressant + add-on medication, or first antidepressant + ECT/rTMS during the year after the PPD diagnosis. Fifth, to consider potential misclassification of treatment resistance due to medication switches caused by side effects, we used an alternative TRPPD definition that accounted for duration of treatment. In this alternative definition, to count as an eligible drug trial during a treatment period (the time between two prescriptions for the same drug within 120 days51), another antidepressant or add-on medication had to be prescribed more than 28 days after the previous treatment started and last at least 28 days. Time-interval criteria were not applied to ECT or rTMS. Last, some of the risk factors studied might correlate with each other. We therefore included all factors in an additional model for mutual adjustment and assessed the independent factors associated with treatment resistance.
Data were processed and analyzed using SAS (version 9.4). All tests were two sided, and P < 0.05 was considered statistically significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Due to privacy protection measures, such as the General Data Protection Regulation (GDPR), the registers’ data are not publicly accessible. Researchers who are interested in replicating this study can apply for access to individual-level data through Statistics Sweden (https://www.scb.se/en/services/ordering-data-and-statistics/ordering-microdata/). Access to data on patient health can be applied for through Socialstyrelsen (https://www.socialstyrelsen.se/en/statistics-and-data/registers/).
Code availability
Analysis coding is available via GitHub at https://github.com/YufChe/TRPPD.
References
Wang, Z. et al. Mapping global prevalence of depression among postpartum women. Transl. Psychiatry 11, 543 (2021).
Slomian, J. et al. Consequences of maternal postpartum depression: a systematic review of maternal and infant outcomes. Womens Health 15, 1745506519844044 (2019).
Yu, H. et al. Perinatal depression and risk of suicidal behavior. JAMA Netw. Open 7, e2350897 (2024).
Hagatulah, N. et al. Perinatal depression and risk of mortality: nationwide, register based study in Sweden. Br. Med. J. 384, e075462 (2024).
Epperson, C. N. et al. Healthcare resource utilization and costs associated with postpartum depression among commercially insured households. Curr. Med. Res. Opin. 36, 1707–1716 (2020).
Moore Simas, T. A., Whelan, A. & Byatt, N. Postpartum depression—new screening recommendations and treatments. JAMA 330, 2295–2296 (2023).
Gronemann, F. H. et al. Treatment patterns in patients with treatment-resistant depression in Danish patients with major depressive disorder. J. Affect. Disord. 287, 204–213 (2021).
De Crescenzo, F. et al. Selective serotonin reuptake inhibitors (SSRIs) for post-partum depression (PPD): a systematic review of randomized clinical trials. J. Affect. Disord. 152–154, 39–44 (2014).
Brown, S. et al. Current and common definitions of treatment-resistant depression: findings from a systematic review and qualitative interviews. Can. J. Psychiatry 64, 380–387 (2019).
Rush, A. J. et al. STAR*D: revising conventional wisdom. CNS Drugs 23, 627–647 (2009).
Lundberg, J. et al. Association of treatment-resistant depression with patient outcomes and health care resource utilization in a population-wide study. JAMA Psychiatry 80, 167–175 (2023).
Reutfors, J. et al. Mortality in treatment-resistant unipolar depression: a register-based cohort study in Sweden. J. Affect. Disord. 238, 674–679 (2018).
Johnston, K. M. et al. The burden of treatment-resistant depression: a systematic review of the economic and quality of life literature. J. Affect. Disord. 242, 195–210 (2019).
Brenner, P. et al. Excess deaths in treatment-resistant depression. Ther. Adv. Psychopharmacol. 11, 20451253211006508 (2021).
O’Connor, S. J. et al. Predictors and risk factors of treatment-resistant depression: a systematic review. J. Clin. Psychiatry 85, 23r14885 (2023).
Batt, M. M. et al. Is postpartum depression different from depression occurring outside of the perinatal period? A review of the evidence. Focus (Am. Psychiatr. Publ.) 18, 106–119 (2020).
Schiller, C. E., Meltzer-Brody, S. & Rubinow, D. R. The role of reproductive hormones in postpartum depression. CNS Spectr. 20, 48–59 (2015).
Byatt, N. et al. Enhancing participation in depression care in outpatient perinatal care settings: a systematic review. Obstet. Gynecol. 126, 1048–1058 (2015).
Gidén, K. et al. Remission or persistence? A prediction tool to identify women at risk for long-term depressive symptoms postpartum. Depress. Anxiety 2024, 7734542 (2024).
Eckerdal, P. et al. Delineating the association between mode of delivery and postpartum depression symptoms: a longitudinal study. Acta Obstet. Gynecol. Scand. 97, 301–311 (2018).
Ilska, M. et al. Vaginal delivery or caesarean section—severity of early symptoms of postpartum depression and assessment of pain in Polish women in the early puerperium. Midwifery 87, 102731 (2020).
Cepeda, M. S., Kern, D. M. & Nicholson, S. Treatment resistant depression in women with peripartum depression. BMC Pregnancy Childbirth 19, 323 (2019).
Treatment and management of mental health conditions during pregnancy and postpartum: ACOG clinical practice guideline no. 5. Obstet. Gynecol. 141, 1262–1288 (2023).
Perlman, K. et al. A systematic meta-review of predictors of antidepressant treatment outcome in major depressive disorder. J. Affect. Disord. 243, 503–515 (2019).
Gronemann, F. H. et al. Socio-demographic and clinical risk factors of treatment-resistant depression: a Danish population-based cohort study. J. Affect. Disord. 261, 221–229 (2020).
Korous, K. M. et al. Socioeconomic status and depressive symptoms: an individual-participant data meta-analysis on range restriction and measurement in the United States. J. Affect. Disord. 314, 50–58 (2022).
Madigan, A. & Daly, M. Socioeconomic status and depressive symptoms and suicidality: the role of subjective social status. J. Affect. Disord. 326, 36–43 (2023).
Fluharty, M. et al. The association of cigarette smoking with depression and anxiety: a systematic review. Nicotine Tob. Res. 19, 3–13 (2017).
Holm, K. J. & Spencer, C. M. Bupropion: a review of its use in the management of smoking cessation. Drugs 59, 1007–1024 (2000).
Engström, K., Magnusson, C. & Galanti, M. R. Socio-demographic, lifestyle and health characteristics among SNUS users and dual tobacco users in Stockholm County, Sweden. BMC Public Health 10, 619 (2010).
El-Hage, W. et al. Mechanisms of antidepressant resistance. Front. Pharmacol. 4, 146 (2013).
Zhao, X. H. & Zhang, Z. H. Risk factors for postpartum depression: an evidence-based systematic review of systematic reviews and meta-analyses. Asian J. Psychiatry 53, 102353 (2020).
Xu, H. et al. Cesarean section and risk of postpartum depression: a meta-analysis. J. Psychosom. Res. 97, 118–126 (2017).
Ward, R. M. & Beachy, J. C. Neonatal complications following preterm birth. BJOG 110, 8–16 (2003).
Iosifescu, D. V., Bankier, B. & Fava, M. Impact of medical comorbid disease on antidepressant treatment of major depressive disorder. Curr. Psychiatry Rep. 6, 193–201 (2004).
Haroon, E. et al. Antidepressant treatment resistance is associated with increased inflammatory markers in patients with major depressive disorder. Psychoneuroendocrinology 95, 43–49 (2018).
Caropreso, L. et al. Preeclampsia as a risk factor for postpartum depression and psychosis: a systematic review and meta-analysis. Arch. Womens Ment. Health 23, 493–505 (2020).
Cepeda, M. S., Reps, J. & Ryan, P. Finding factors that predict treatment-resistant depression: results of a cohort study. Depress. Anxiety 35, 668–673 (2018).
Kinrys, G. et al. Tachyphylaxis in major depressive disorder: a review of the current state of research. J. Affect. Disord. 245, 488–497 (2019).
Schmidt, P. J. et al. Differential behavioral effects of gonadal steroids in women with and in those without premenstrual syndrome. N. Engl. J. Med. 338, 209–216 (1998).
Tylee, A. & Jones, R. Managing depression in primary care. Br. Med. J. 330, 800–801 (2005).
Cnattingius, S. et al. The Swedish medical birth register during five decades: documentation of the content and quality of the register. Eur. J. Epidemiol. 38, 109–120 (2023).
Ludvigsson, J. F. et al. External review and validation of the Swedish national inpatient register. BMC Public Health 11, 450 (2011).
Wettermark, B. et al. The new Swedish Prescribed Drug Register—opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol. Drug Saf. 16, 726–735 (2007).
Ludvigsson, J. F. et al. The longitudinal integrated database for health insurance and labour market studies (LISA) and its use in medical research. Eur. J. Epidemiol. 34, 423–437 (2019).
Larsen, S. V. et al. Depression associated with hormonal contraceptive use as a risk indicator for postpartum depression. JAMA Psychiatry 80, 682–689 (2023).
American Psychiatric Association, DSM-5 Task Force. Diagnostic and Statistical Manual of Mental Disorders: DSM-5 5th edn (APA, 2013).
Cepeda, M. S. et al. Finding treatment-resistant depression in real-world data: how a data-driven approach compares with expert-based heuristics. Depress. Anxiety 35, 220–228 (2018).
Dold, M. & Kasper, S. Evidence-based pharmacotherapy of treatment-resistant unipolar depression. Int. J. Psychiatry Clin. Pract. 21, 13–23 (2017).
Ludvigsson, J. F. et al. Adaptation of the Charlson Comorbidity Index for register-based research in Sweden. Clin. Epidemiol. 13, 21–41 (2021).
Fazel, S. et al. Antipsychotics, mood stabilisers, and risk of violent crime. Lancet 384, 1206–1214 (2014).
Acknowledgements
The work was supported by Karolinska Institutet Research Foundation to Y.C. (grant number 2024-02707) and E.B. (grant number 2022-01548); the European Research Council to D.L. (grant number 101165552); the Swedish Research Council (Vetenskapsrådet) to D.L. (grant number 2024-02592); the Swedish Research Council for Health, Working Life and Welfare (FORTE) to D.L. (grant number 2020-00971 and 2023-00399); and Karolinska Institutet Strategic Research Area in Epidemiology and Biostatistics to D.L. Y.L. is supported by the US NIMH R01 (grant number MH123724), the European Union’s Horizon 2020 research and innovation program (grant number 964874), the Swedish Research Council (Vetenskapsrådet, grant number 2021-02615) and the European Research Council (grant number 101042183). Researchers are independent of the funders. The funding has no role in the design of the study and collection, analysis and interpretation of data and in writing the manuscript.
Funding
Open access funding provided by Karolinska Institute.
Author information
Authors and Affiliations
Contributions
Y.C. and D.L. conceived the study. Y.C. and D.L. prepared and analyzed the data. Y.C. drafted the paper. Y.C., E.B., M.B., E.E.J., E.F., Y.L., A.S. and D.L. interpreted the results, reviewed the paper and approved the decision to submit the paper. Y.C., E.B., Y.L. and D.L. obtained funding. Y.C., E.B. and E.E.J. provided administrative, technical or material support. D.L. supervised the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Sofie Egsgaard, Dolores Marín-Morales and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–3.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Y., Bränn, E., Bendix, M. et al. Risk factors for treatment resistance among women with postpartum depression in a nationwide study. Nat. Mental Health 4, 288–297 (2026). https://doi.org/10.1038/s44220-026-00587-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44220-026-00587-8