Abstract
Ceftazidime-avibactam (CZA) is currently one of the last resorts used to treat infections caused by carbapenem-resistant Enterobacteriaceae and Pseudomonas aeruginosa. However, KPC variants have become the main mechanism mediating CZA resistance in KPC-producing gram-negative bacteria after increasing the application of CZA. Our previous study revealed that CZA-resistant KPC-33 had emerged in carbapenem-resistant P. aeruginosa (CRPA) and had resulted in death due to hypervirulence and extensive drug resistance; however, the evolutionary path of KPC-33-producing CRPA has not been investigated. Here, we observed the emergence of blaKPC-33 in CRPA under drug pressure, leading to resistance to CZA. We further elucidated the pathway of resistance development due to blaKPC mutations in P. aeruginosa. Three KPC-producing P. aeruginosa (KPC-PA) strains (including one blaKPC-33-positive strain and two blaKPC-2-positive strains) were successively isolated from a hospitalized patient. The blaKPC-33-positive CZA-resistant strain SRPA0656 (CZA MIC >128 μg/mL, imipenem MIC = 32 μg/mL) was isolated after the blaKPC-2-positive P. aeruginosa SRP2863 (CZA MIC = 1 μg/mL, imipenem MIC >128 μg/mL) was treated with CZA. The subsequent use of carbapenems to treat the infection led to the re-emergence of the KPC-2-producing strain SRPA3703. Additionally, we collected four other KPC-33-producing P. aeruginosa strains. Antimicrobial susceptibility testing revealed that all the KPC-33-bearing P. aeruginosa strains in this study were multidrug-resistant but susceptible to colistin and amikacin. Whole-genome sequencing indicated that blaKPC-33 was located on two Tn4401-like transposons contained in the plasmids and that most of these plasmids could be transferred into P. aeruginosa PAO1Rif isolates. Growth rate determination demonstrated that the relative growth rate of P. aeruginosa harboring blaKPC-33 was faster than that of P. aeruginosa harboring blaKPC-2 in the logarithmic phase. Global phylogenetic analysis revealed that most KPC-PA strains were isolated from China and the USA. MLST revealed that the most common ST in KPC-PA was ST463, which was detected only in China, and that all the strains carried blaKPC-2 or its derivatives. These results indicated that the use of CZA for the treatment of KPC-2-producing P. aeruginosa may have contributed to the evolution of KPC-33. The widespread dissemination of KPC-PA (especially the ST463) and Tn4401 transposons may increase the spread of CRPA isolates carrying blaKPC-33. Close attention to the development of resistance to CZA during clinical treatment of CRPA infection and monitoring CZA-resistant strains is necessary to prevent further spread.
Similar content being viewed by others
Introduction
Pseudomonas aeruginosa is a common pathogen of nosocomial infection, with a large number of intrinsic antibiotic resistance genes and exogenously acquired resistance genes in the genome, increasing the difficulty of clinical treatment1,2. Recently, the WHO updated the list of bacterial priority pathogens, moving carbapenem-resistant P. aeruginosa (CRPA) from the “critical” to the “high” category, but innovative antibiotics targeting CRPA are still needed3. There are several main mechanisms of carbapenem resistance in P. aeruginosa, including the loss of the OprD protein, the overexpression of efflux pumps and/or AmpC-lactamases and the production of carbapenemase4. Among these, the most common is the acquisition of carbapenemase, and KPC-2 and VIM-2 were the most prevalent carbapenemases in P. aeruginosa worldwide at present5. Notably, up to 40% of carbapenemase-producing P. aeruginosa isolates harboring blaKPC-2 have been identified in China5.
Avibactam is a novel enzyme inhibitor with favorable efficacy in KPC-producing Enterobacteriaceae and P. aeruginosa when combined with ceftazidime6,7,8. However, with the widespread use of ceftazidime-avibactam (CZA) in clinical treatment, resistance to CZA has also emerged, and the susceptibility of CRPA to CZA has reached 38.2% globally as of 20219. CZA resistance in P. aeruginosa is associated with the production of metallo-β-lactamases (MBLs), changes in the Ω-loop of the AmpC enzyme, mutations or multiple copies of the KPC enzyme, and/or the overexpression of efflux pumps10,11. Among these, blaKPC mutations play a major role in the CZA resistance mechanism12. To date, the NCBI GenBank database lists 229 blaKPC variants, and compared with wild-type blaKPC (blaKPC-2 and blaKPC-3), blaKPC variants usually undergo amino acid substitution, insertion, or deletion12. These variants were frequently observed in Enterobacteriaceae bacteria but are less common in P. aeruginosa13,14. KPC-33, one of the most common variants, was commonly observed in Klebsiella pneumoniae and induces alterations in the hydrogen bonding structure of the omega-loop in KPC, resulting in resistance to CZA12. Compared with KPC-2, KPC-33 has a D179Y mutation in the Ω-loop, which reduces the inhibition of avibactam and enhances the efficiency of ceftazidime hydrolysis15.
The emergence of KPC-33 in P. aeruginosa was reported in our previous study16; however, the evolutionary path of blaKPC-33 in the CRPA has not been investigated. In this study, we observed the emergence of blaKPC-33 in the CRPA under drug pressure, leading to the emergence of resistance to CZA. We further elucidated the evolutionary trajectory of blaKPC mutations in P. aeruginosa. We also analyzed the genetic characteristics of the KPC-33-producing CRPA and the global phylogeography of KPC-producing P. aeruginosa.
Results
The development of clinical resistance in KPC-33-producing CRPA
An 86-year-old male was admitted to the hospital due to emphysema and fever after kyphoplasty. During the early stage of treatment, the patient was treated with meropenem, piperacillin‒tazobactam (PIP/TZP), and colistin; however, the inflammatory indices did not significantly improve. A carbapenem-resistant but CZA-susceptible P. aeruginosa strain (SRPA2863, KPC-2 positive) was isolated from the patient’s sputum on Day 46 after hospital admission. Owing to poor therapeutic effects, CZA (2.5 g q8h) and imipenem cilastatin sodium (2 g q8h) were used for anti-infective treatment. On Day 71, the patient’s condition had not improved, and a CZA-resistant P. aeruginosa strain (SRPA0656, KPC-33 positive) was isolated from his sputum. Consequently, PIP/TZP (4.5 g q8h) with colistin was used as a therapy. Considering the fluctuating inflammatory indices, PIP/TZP was discontinued, and imipenem cilastatin sodium (2 g q8h) was given. Unfortunately, the KPC-2-producing CRPA(SRPA3703) was isolated from sputum on Day 92. Despite continuous adjustments in the treatment regimen, the patient’s condition was not controlled, and he ultimately died as a result of respiratory failure and septic shock. The duration of CRPA during the course of antibiotic therapy is summarized in Fig. 1.
The light-yellow bar represents imipenem, the purple bar represents meropenem, the gray bar represents ceftazidime-avibactam, the green bar represents colistin, and the blue bar represents piperacillin-tazobactam. The black arrows indicate the time of P. aeruginosa isolation. IPM imipenem, MEM meropenem, PIP/TZP piperacillin-tazobactam, CST colistin, CZA ceftazidime-avibactam.
Antimicrobial susceptibility testing (AST) revealed that the three CRPA strains were resistant to cephalosporins and carbapenems but susceptible to amikacin, colistin, and CZA (except for the blaKPC-33-positive strain SRPA0656, which was susceptible to amikacin and colistin but resistant to CZA) (Table 1). In addition, whole-genome sequencing (WGS) was performed to determine the genetic relationships among the three CRPA strains. MLST results revealed that all strains belonged to the same ST, ST463. SNP counts further showed that the three CRPA strains were highly homologous, with small SNPs (ranging from 4 to 8 SNPs) identified via whole-genome SNP analysis. In summary, we isolated three CRPA strains on Days 46, 71, and 92 after patient admission. The KPC-2-producing P. aeruginosa strains (SRPA2863 and SRPA3703) isolated after carbapenem treatment displayed CZA susceptibility but carbapenem resistance (CZA MIC = 1 μg/mL, imipenem MIC >128 μg/mL). In contrast, SRPA0656 (isolated 26 days after CZA treatment) had increased susceptibility to carbapenems and resistance to CZA (CZA MIC >128 μg/mL, imipenem MIC = 32 μg/mL).
Characteristics of KPC-33-producing CRPA isolates
To further characterize the KPC-33-producing CRPA isolates, four additional strains were collected. Five KPC-33-producing CRPA strains were isolated from different patients, including four from sputum and one from blood (Supplementary Table 1). The results of antimicrobial susceptibility determination revealed that all KPC-33-producing CRPA strains were resistant to ceftazidime, cefepime, meropenem, imipenem, ciprofloxacin, and CZA, but susceptible to colistin and amikacin, indicating that these strains were multidrug-resistant (MDR) P. aeruginosa (Table 1).
WGS analysis further revealed that the KPC-33-producing CRPA isolates identified in this study could be categorized into two STs (ST463 and ST1076). The chromosomes of these strains were ~7 Mb in length, and the plasmids were of various sizes (Supplementary Table 2). We further confirmed that a total of eight antibiotic resistance genes (ARGs) confer resistance to five classes of antimicrobials, including those conferring resistance to β-lactams (blaKPC-33, blaPDC-374 and blaOXA486/395), aminoglycosides (aph(3’)-IIa), fosfomycin (fosA), fluoroquinolones (crp), and phenicols (catB7). The blaKPC-33 genes were localized on the plasmid in all the strains.
Characteristics of the plasmid carrying bla KPC-33 in P. aeruginosa
The WGS results revealed that blaKPC-33 was located on two different plasmids, type I and type II (Fig. 2 and Supplementary Table 2)17. The four plasmids harboring blaKPC-33 were identified as an uncategorized type (type I) (Fig. 2B). The complete sequences of pKPC33-ZYPA54 and pSYPA07-KPC-33 were obtained by using nanopore technology, and the plasmid contigs of pSRPA0656 and pSRPA5504 were highly matched with those of pSYPA07-KPC-33 and pKPC33-ZYPA54. All type I plasmids were highly similar to pZYPA01 (GenBank accession no. MZ050803.1, 100% query coverage and 100% nucleotide identity) and pR20-48 (GenBank accession no. CP138392.1, 99–100% query coverage, and 100% nucleotide identity) from clinical P. aeruginosa in China (Fig. 2B). BLASTn results revealed that the type II plasmid pSPPA1308 was highly identical to the backbone sequence of the plasmid pOZ176 (GenBank accession no. KC543497.1) with 94% query coverage and 100% nucleotide identity (Fig. 2A), the first IncP-2 plasmid to be identified18. Additionally, pSPPA1308 was similar to pLHL1-KPC-3 (GenBank accession no. CP081203.1) and the P9W plasmid unnamed1 (GenBank accession no. CP081203.1), which carry blaKPC-3 and blaKPC-2, respectively, with 100% query coverage and 100% nucleotide identity. Conjugation experiments were performed to confirm that the blaKPC-33-carrying plasmids pSRPA1308 and pSRPA5504 were successfully transferred into rifampicin-resistant PAO1, indicating that blaKPC-33 was located in a transferable plasmid. Compared with those of the PAO1Rif recipient strain, the resistance of the transconjugants to ceftazidime, cefepime, meropenem, imipenem, and CZA increased to varying degrees (Table 1). Notably, the elevated level of resistance to CZA in transconjugants was much greater than that in carbapenems.
A pSPRA1308 was used as the reference plasmid to perform the genome alignment with P9W plasmid unnamed1, pLHLl-KPC-3, and pOZ176. B pZYPA54-KPC-33 was used as the reference plasmid to perform the genome alignment of pSYPA07-KPC-33, pSRPA0656, pSRPA5504, pR20-24, and pZYPA01. The colored circles from the inside to the outside represent each plasmid, as shown in the right column. The solid regions demonstrate sequence similarity, whereas the gaps represent regions lacking sequence similarity.
The genetic context of bla KPC-33
WGS analysis showed that the blaKPC-33 genes were located in two Tn4401-like transposons, with an identical core genetic platform, namely Tn6296 (∆ISKpn27–blaKPC-33–∆ISKpn6–korC-klcA). The Type I Tn4401-like transposons (IS26-∆ISKpn27–blaKPC-33–∆ISKpn6–korC-klcA-IS26-TpnA-IS26-IS26) were identical in the type I plasmid (Fig. 3). Four copies of IS26 (three intact and one truncated) were found in the blaKPC region from pSRPA5504, pSRPA0656, and pKPC33-ZYPA54, with one overlapping IS26 in the two IS26 units (IS26-Tn6376-IS26 unite and IS26-blaKPC-IS26 unite) (Fig. 3). Interestingly, in pSYPA07-KPC-33, the blaKPC core platform was translocated by IS26, resulting in the truncation of Tn6373 and forming a genetic context of IS26-∆ISKpn27–blaKPC-33–∆ISKpn6–korC-klcA-IS26 (Fig. 3). In the pSPPA1308 plasmid, Type II Tn4401-like transposons were composed of IS6100-∆ISKpn27–blaKPC-33–∆ISKpn6–korC-klcA-TpnR-TpnA. However, Tn6296 was truncated by the tnpR module of Tn1403 and IS6100, resulting in the formation of the ∆Tn1403-∆Tn6296-IS6100 region (Fig. 3).
Comparison of the genetic context of the blaKPC region from pSRPA1308, the blaKPC region from pSRPA5504, and the blaKPC region from pSRPA0656, pKPC33-ZYPA54, pSYPA07-KPC-33, and pZYPA01. The gray shading and squares indicate homologies between the corresponding genetic loci on each plasmid. Arrows indicate open reading frames, with arrowheads indicating the direction of transcription: red, antibiotic resistance-encoding genes; blue, transposon- and integron-associated genes; other genes are shown by gray arrows.
KPC-33-mediated resistance to CZA and the growth rate of isolates carrying bla KPC-33
Cloning experiments were performed to investigate whether blaKPC-33 was responsible for CZA resistance in P. aeruginosa. blaKPC-2 and blaKPC-33 were expressed successfully in P. aeruginosa PAO1. The P. aeruginosa PAO1 strain carrying pGK-KPC-2 was resistant to ceftazidime, cefepime, meropenem, and imipenem, but was susceptible to CZA. In contrast, the P. aeruginosa PAO1 transformant carrying pGK-KPC-33 exhibited resistance to ceftazidime, cefepime, and CZA, but was susceptible to meropenem and imipenem (Table 1). Overall, these results indicate that blaKPC-33 is responsible for CZA resistance in P. aeruginosa.
To clarify the effect of blaKPC-33 on the growth rate of P. aeruginosa, we determined the growth curves of clinical KPC-PA strains and clonal strains in the absence of antibiotic pressure (Supplementary Fig. 2A–D). The relative growth rate of PAO1-pGK-KPC-33 was ~4.0% faster than that of PAO1-pGK-KPC-2 in logarithmic phase (0.9054 ± 0.005 vs. 0.8692 ± 0.027) (Supplementary Fig. 2A). The relative growth rate of SRPA0656 was 5.7% faster than that of SRPA3703 (0.6951 ± 0.012 vs. 0.6573 ± 0.010) and 5.8% faster than that of SRPA2863 (0.6951 ± 0.012 vs. 0.6570 ± 0.017) in clinical KPC-PA. The doubling time of strains carrying blaKPC-33 during the logarithmic phase was 3.4–10.7% shorter than that of strains carrying blaKPC-2 (Supplementary Fig. 2B). However, there was no significant difference between KPC-2-producing strains and KPC-33-producing strains in the stationary phase. Overall, in P. aeruginosa, strains carrying blaKPC-33 exhibit better growth during the logarithmic phase compared to strains carrying blaKPC-2.
Phylogenetic analysis of KPC-33-positive P. aeruginosa strains globally
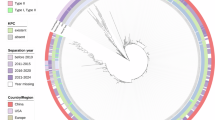
A phylogenetic tree was constructed based on the core genomic SNPs of nine strains of clinical P. aeruginosa carrying blaKPC-33 (five from this study and four from the NCBI database) (Fig. 4A). The core genomes of the nine isolates were identified with a total of 53,450 SNPs (Supplementary Fig. 1). The phylogenetic tree revealed that the KPC-33-producing P. aeruginosa strains were distributed mainly in China (88.9%, 8/9) and Chile (11.1%, 1/9) (Fig. 4A). All P. aeruginosa strains harboring blaKPC-33 had seven or more resistance genes.
A Phylogenetic relationships of KPC-33 producing P. aeruginosa in this study and NCBI database. The blue squares represent the corresponding drug-resistance genes. B Phylogenetic tree of 714 KPC-PA isolates collected from this study and NCBI database. The red-marked genomes were collected clinically in this study.
Based on the phylogenetic tree, KPC-33-producing P. aeruginosa isolates could be categorized into three clades, and four STs, namely, ST463, ST235, ST1076, and ST654, were identified (Fig. 4A). In previous studies, SNPs <45 were defined as clonal dissemination; otherwise, they were defined as nonclonal dissemination19. Notably, there were few SNPs (4–39 SNPs) between the clinical strains SYPA07, ZYPA54, PA0107, R19-73, and SRPA0656, indicating a close phylogenetic relationship. SYPA07, ZYPA54, PA0107, and R19-73 originated from the same hospital, whereas SRPA0656 originated from a different hospital in the same province. These findings suggest that KPC-33-producing CRPA has been transmitted from hospital to hospital in certain regions.
Phylogeographic and phylogenetic analysis of KPC-producing CRPA strains globally
To investigate the phylogeographic relationships of the KPC-producing CRPA strains, basic information on a total of 714 KPC-producing CRPA strains was obtained from this study and the NCBI database (Supplementary Table 3). Most of the KPC-producing CRPA strains were isolated from Asia (64.1%, 458/714), followed by North America (17.51%, 125/714), South America (16.11%, 115/714), Europe (2.00%, 14/714), and other regions (0.28%, 2/714) (Fig. 5A). KPC-producing CRPA strains have been reported in 17 countries worldwide, with the majority of isolates detected in China (63.02%, 452/714), followed by the USA (17.29%, 124/717) and Colombia (7.53%, 54/717) (Fig. 5B). KPC-producing CRPA strains have been reported almost every year since 2007, particularly in China. Notably, KPC variants (KPC-33, KPC-31, KPC-35, KPC-90, and KPC-113) responsible for CZA resistance emerged after the Food and Drug Administration approved CZA for marketing in 2015. KPC-2-producing P. aeruginosa predominated among the KPC-producing CRPA strains (93.28%, 666/714), followed by KPC-3-producing P. aeruginosa (2.52%,18/714). Moreover, most of these KPC-2-producing CRPA strains were isolated from China (65.47%, 436/666), the USA (14.86%, 99/666) and South America (15.47%, 103/666) (Fig. 5B). MLST analysis showed that the 714 KPC-producing CRPA strains could be categorized into 52 different STs (Fig. 4B). The most common STs were ST463 (45.66%, 326/714), followed by ST235 (9.80%, 70/714).
To trace the phylogenetic relationships between the CRPA strains carrying blaKPC-33 in this study and other KPC-producing CRPA strains globally, a phylogenetic tree was constructed using the assembled genomes of 714 KPC-producing CRPA strains from this study and the NCBI database (Fig. 4B and Supplementary Table 3). The phylogenetic tree showed that the KPC-producing CRPA strains could be categorized into five clades, and the seven clinical isolates in this study were distributed in Clade 1 and Clade 2. The phylogenetic tree further revealed that the STs of the KPC-producing CRPA strains isolated in China were diverse and dominated by ST463. Notably, the ST463 KPC-producing CRPA strains were detected only in China, and all the ST463 P. aeruginosa strains carried blaKPC-2 or its variants.
Discussion
In our previous study16, blaKPC-33 emerged from the hypervirulent ST463 CRPA, but there was no clear evidence to indicate the source of blaKPC-33. Here, we isolated KPC-2-producing CRPA strains (SRPA2863 and SRA3703) after carbapenem treatment, while KPC-33-producing CRPA strains (SRPA0656) were isolated after 26 days of treatment with CZA. The blaKPC-33 variant caused by the D179Y mutation of blaKPC-2 was driven mainly by the presence of CZA. Later, the strain was recovered to KPC-2 after carbapenem treatment. The patient had a history of CZA treatment, which indicates that KPC-33 was mutated from the wild-type KPC-2 in P. aeruginosa strains after CZA exposure. Available studies also suggest that CZA may be an independent risk factor for the emergence of KPC variants20,21,22 in bacteria. In this study, we further elucidated the pathway by which blaKPC-2 evolved into blaKPC-33 in ST463 CRPA strains, leading to CZA resistance.
KPC-33, as the most prevalent KPC-2 variant, was first identified in K. pneumoniae from Italy and then appeared in China and Greece22,23. KPC-33 is a single amino acid substitution at position 179 in the -ring, which increases the affinity of ceftazidime and attenuates the inhibitory effect of avibactam. This variation results in altered susceptibility of the bacteria to CZA and carbapenems: the “see-saw” effect15. AST revealed that, compared with the SRPA2863 and SRA3703 isolates, SRPA0656 exhibited >128-fold increase in the MIC of CZA and >2-fold decrease in the MIC of carbapenem. However, different from K. pneumoniae carrying blaKPC-3322, the KPC-33-producing CRPA strains in this study remained carbapenem-resistant, which may be related to the inactivation of OprD and the overexpression of efflux pumps in P. aeruginosa24,25. Furthermore, our study found that P. aeruginosa carrying blaKPC-33 exhibited better growth performance than those carrying blaKPC-2 during the logarithmic phase, which may also be an important reason for the continued emergence of KPC-33 mutants. Consequently, for KPC-producing CRPA strains under CZA stress, there is a high risk of developing resistance to both carbapenems and CZA, which may greatly limit the treatment options. Novel β-lactam‒β-lactamase inhibitor combinations, such as imipenem‒relebactam and meropenem‒vaborbactam, have been shown to be effective in vitro against KPC-producing Enterobacteriaceae, including KPC-33-producing K. pneumoniae. CZA in combination with other antibiotics has also been shown to have a favorable effect, and there have been reports that CZA being combined with aztreonam to successfully treat KPC-33-producing K. pneumoniae infections26. Nonetheless, we cannot ignore the risk of P. aeruginosa harboring blaKPC-33. Regular microbial culture and genotyping is needed to monitor dynamic changes in blaKPC in patients and adjust the treatment plan in a timely manner during the treatment of KPC-producing CRPA strains with CZA.
Consistent with Hu’s study27, we observed two blaKPC-bearing plasmid types. The type II plasmid is a common type of P. aeruginosa plasmid that is usually over 300 kb in size and has a narrow host range28. As previously reported27,29,30, the type I plasmid was closely related to the ST463 clones and always carried blaKPC-2. Although the T4SS was present in the type I plasmid, conjugation experiments demonstrated that it could not be transferred, which may be due to the absence of other mobile elements. In addition, blaKPC-33 is located in a different type of plasmid, but the core genetic platform of blaKPC-33, named Tn6296 with a structure of ∆ISKpn27–blaKPC-33–∆ISKpn6, was not changed and played an important role in the transfer of blaKPC. Compared with the pZYPA01 plasmid, the IS26-blaKPC-IS26 unit of the plasmid pKPC33-SYPA07 was inverted, and the IS26-Tn6373-IS26 unit lost IS26 and TpnA, which likely occurred due to the IS26-mediated inversion event. Moreover, IS26 can be used to form multicopy resistance genes by direct “copy and paste” or via the insertion of a “translocatable unit”31. Previous studies have indicated that IS26 mediates the formation of two copies of blaKPC-232. Briefly, the type II plasmid, as well as the Tn4401 transposons, may, contribute to the spread of blaKPC-33, leading to serious challenges in the prevention of extensive drug resistance in P. aeruginosa.
In addition, we collected information on P. aeruginosa strains harboring blaKPC-33 worldwide, all of which were isolated in China and Chile. Coincidentally, the global geographic analysis revealed high isolation rates of KPC-producing CRPA strains in both China and Chile, which is also consistent with other studies32,33. ST463 P. aeruginosa was the most prevalent type of KPC-producing CRPA strain in China, and the high carriage rate of blaKPC-2 offers additional possibilities for the emergence of KPC variants, which may explain why P. aeruginosa carrying blaKPC-33 was detected more frequently in China. In addition, there was evidence that the ST463 clone carries both exoU and exoS, which may be important reasons for the poor prognosis of patients30. The ST654 and ST235 clones, among the top ten global high-risk clones of P. aeruginosa, were associated with different acquired β-lactamases, IMP, and VIM MBLs in particular34. ST235 and ST643 KPC-producing CRPA strains are also prevalent worldwide and may have contributed to the spread of blaKPC to some extent. Therefore, we still need to strengthen surveillance to prevent the spread of high-risk clones carrying blaKPC, especially ST463 P. aeruginosa, which may become a future threat.
In summary, we report the global phylogeography and genetic characterization of carbapenem- and CZA-resistant KPC-33-producing CRPA strains. The use of CZA and the relatively high growth rate of blaKPC-33 may be important for inducing blaKPC-33 mutations in CRPA strains carrying blaKPC-2. In addition, the type II plasmid, as well as the Tn4401 transposons, may contribute to the spread of blaKPC-33 in CRPA strains, further limiting treatment options. Moreover, blaKPC-33 has emerged in high-risk clone ST463 CRPA strains. Therefore, it is necessary to monitor the resistance phenotypes of KPC-producing CRPA strains regularly and adjust antibiotic regimens in a timely manner when CZA is used to treat KPC-2-producing CRPA strains in the clinic.
Methods
Isolate data
We successively isolated KPC-33- and KPC-2-producing P. aeruginosa from a patient during treatment, including two KPC-2-producing strains (isolated on 26 August 2023 and 13 October 2023) and one KPC-33-producing strain (isolated on 21 September 2023). To study the prevalence of KPC-33-producing P. aeruginosa, four other P. aeruginosa strains carrying blaKPC-33 were collected from two tertiary hospitals in Zhejiang, China. These strains were identified using the MALDI-TOF MS system (bioMérieux, Marcy l’Etoile, France). The primers KPC-F and KPC-R were used to sequence the blaKPC genes (Supplementary Table 4). This study was conducted in accordance with the Declaration of Helsinki and had been reviewed and approved by the Research Ethics Committee of Zhejiang Provincial People’s Hospital (Approval no.: QT2024188).
Antimicrobial susceptibility testing
The minimal inhibitory concentrations (MICs) of the strains in this study were determined by the broth microdilution method. A total of eight antibiotics were tested, including cefepime, ceftazidime, imipenem, meropenem, amikacin, ciprofloxacin, colistin, and CZA. The results were interpreted according to the breakpoints recommended by the 2024 Clinical and Laboratory Standards Institute (CLSI) guidelines35, except for colistin, which was interpreted according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines (http://www.eucast.org/clinical_-breakpoints). P. aeruginosa ATCC 27853 was used as the quality control strain.
Whole-genome sequencing and analysis
Extraction of the Genomic DNA of isolates in this study was performed with QIAamp DNA MiniKit (Qiagen, Valencia, CA, USA). Oxford Nanopore (MinION system, Nanopore, Oxford, UK) and Illumina sequencing (NovaSeq system, Illumina Inc, San Diego, USA) were used respectively for genome sequencing and followed by de novo assembly with Unicycler v0.4.836. Prokka 1.11 was used to annotate the location and function of the genes37. Multilocus sequence type (MLST) was determined by using the MLST 2.1 (https://github.com/tseemann/mlst). ResFinder 4.1 and PlasmidFinder 1.3 at the Center for Genomic Epidemiology (http://www.genomicepidemiology.org/) were used to identify resistance genes and plasmid types. BLASTn v2.4.0 searches were conducted to find similar plasmids in this study38. The circular genome map and comparative genome map were completed by BRIG v0.95 and Easyfig v2.2.339,40.
Cloning experiments
Cloning experiments were conducted according to the methods described in a previous study41. Briefly, the wild-type blaKPC-2 gene and blaKPC-33 gene sequences containing the wild promoter were amplified from SRPA1308 and SRPA3703 as templates, respectively. The purified product was ligated with the pGK-1900 vector and transformed into E. coli DH5α. The recombinant plasmids pGK-KPC-33 and pGK-KPC-2 were also transferred into the PAO1 strain by electrochemical transformation experiments, respectively. Transformants were selected from Luria-Bertani (LB) agar plates supplemented with 50 μg/mL gentamicin, and further confirmed by PCR and Sanger sequencing. Primers were listed in Supplementary Table 4.
Plasmid conjugation experiments
According to previous studies42, the KPC-33-producing CRPA was used as the donor strain, and the rifampin-resistant P. aeruginosa PAO1Rif was used as the recipient strain for the conjugation assay. Bacterial suspensions of donor strain and PAO1Rif in the logarithmic phase of growth were 1:1 mixed and dropped onto filter membranes for overnight culture. Overnight cultures were screened on a selection medium (containing 16 μg/mL CZA and 800 μg/mL rifampin). The transconjugants harboring target plasmids were confirmed by PCR and antimicrobial susceptibility. Primers were listed in Supplementary Table 4.
Growth rate determination
We determined the growth rate of clinical and clonal strains in this study to investigate their fitness cost. As in the previous study43, three monoclones of clinical and clonal strains were grown overnight, and 200 μL of the overnight culture diluted at 1: 100 was added to a flat-bottom 100-well plate. Each overnight culture was repeated three times. The optical density of each culture at 600 nm was measured every 5 min for 20 h by Bioscreen C analyzer (Oy Growth Curves Ab.Ltd., Finland). The growth rate based on OD600 curves was calculated by an R script44. The growth rate results were analyzed using of one-way analysis of variance (ANOVA) as available in GraphPad Prism version 9. An adjusted P value <0.05 was considered significant.
Phylogenetic analysis
To further trace the phylogenetic relationship between blaKPC-33 in this study and blaKPC-positive P. aeruginosa isolates worldwide and to monitor the global phylogeography of KPC-PA, we evaluated the GenBank (https://www.ncbi.nlm.nih.gov/datasets/ genome, accessed December 19, 2023) for all available 27,716 P. aeruginosa genomes. And 707 assembled P. aeruginosa carrying blaKPC were screened. Phylogenetic trees were constructed using Roary and FastTree based on SNPs in the core genome and further visualized using iTOL v6 (https://itol.embl.de). ChiPlot (https://www.chiplot.online/map_plot.html) and Inkscape-1.3.2 was used for global geographic distribution mapping.
Data Availability
Sequence data that support the findings of this study have been deposited in the GenBank nucleotide database with the primary accession code PRJNA1120349 and PRJNA790682.
References
Jurado-Martín, I., Sainz-Mejías, M. & McClean, S. Pseudomonas aeruginosa: An audacious pathogen with an adaptable arsenal of virulence factors. Int. J. Mol. Sci. 22, 3128 (2021).
Botelho, J., Grosso, F. & Peixe, L. Antibiotic resistance in Pseudomonas aeruginosa - Mechanisms, epidemiology and evolution. Drug Resist. Updat. 44, 100640 (2019).
WHO. WHO Bacterial Priority Pathogens List, 2024: bacterial pathogens of public health importance to guide research, development and strategies to prevent and control antimicrobial resistance. (2024).
Horcajada, J. P. et al. Epidemiology and treatment of multidrug-resistant and extensively drug-resistant Pseudomonas aeruginosa infections. Clin. Microbiol Rev. 32, e00031–19 (2019).
Reyes, J. et al. Global epidemiology and clinical outcomes of carbapenem-resistant Pseudomonas aeruginosa and associated carbapenemases (POP): a prospective cohort study. Lancet Microbe 4, e159–e170 (2023).
Levasseur, P. et al. In vitro antibacterial activity of the ceftazidime-avibactam combination against Enterobacteriaceae, including strains with well-characterized β-lactamases. Antimicrob. Agents Chemother. 59, 1931–1934 (2015).
Kazmierczak, K. M., de Jonge, B. L. M., Stone, G. G. & Sahm, D. F. In vitro activity of ceftazidime/avibactam against isolates of Pseudomonas aeruginosa collected in European countries: INFORM global surveillance 2012-15. J. Antimicrob. Chemother. 73, 2777–2781 (2018).
Kazmierczak, K. M. et al. Global dissemination of blaKPC into bacterial species beyond Klebsiella pneumoniae and in vitro susceptibility to ceftazidime-avibactam and aztreonam-avibactam. Antimicrob. Agents Chemother. 60, 4490–4500 (2016).
Lee, Y. L., Ko, W. C. & Hsueh, P. R. In vitro activities of ceftazidime-avibactam, ceftolozane-tazobactam, meropenem-vaborbactam and other comparators against Pseudomonas aeruginosa isolates with discrepant resistance to carbapenems: data from the Antimicrobial Testing Leadership and Surveillance (ATLAS) program, 2012-2021. Int. J. Antimicrob. Agents 62, 106867 (2023).
Winkler, M. L., Papp-Wallace, K. M. & Bonomo, R. A. Activity of ceftazidime/avibactam against isogenic strains of Escherichia coli containing KPC and SHV β-lactamases with single amino acid substitutions in the Ω-loop. J. Antimicrob. Chemother. 70, 2279–2286 (2015).
Sanz-García, F., Hernando-Amado, S. & Martínez, J. L. Mutation-driven evolution of Pseudomonas aeruginosa in the presence of either ceftazidime or ceftazidime-avibactam. Antimicrob. Agents Chemother. 62, e01379–18 (2018).
Ding, L. et al. Klebsiella pneumoniae carbapenemase variants: the new threat to global public health. Clin. Microbiol. Rev. 36, e0000823 (2023).
Tu, Y. et al. Emergence of a KPC-90 variant that confers resistance to ceftazidime-avibactam in an ST463 carbapenem-resistant Pseudomonas aeruginosa strain. Microbiol. Spectr. 10, e0186921 (2022).
Yang, Q. et al. A novel KPC-113 variant conferring carbapenem and ceftazidime-avibactam resistance in a multidrug-resistant Pseudomonas aeruginosa isolate. Clin. Microbiol. Infect. 29, 387.e7–.e14 (2023).
García, P. et al. Acquisition of resistance to ceftazidime-avibactam during infection treatment in Pseudomonas aeruginosa through D179Y mutation in one of two blaKPC-2 gene copies without losing carbapenem resistance. Front. Cell Infect. Microbiol. 12, 981792 (2022).
Zhang, P. et al. Emergence of blaKPC-33-harboring hypervirulent ST463 Pseudomonas aeruginosa causing fatal infections in China. J. Infect. 85, e86–e88 (2022).
Zhu, Y. et al. Emergence of ceftazidime- and avibactam-resistant Klebsiella pneumoniae carbapenemase-producing Pseudomonas aeruginosa in China. mSystems 6, e0078721 (2021).
Xiong, J. et al. Complete sequence of pOZ176, a 500-kilobase IncP-2 plasmid encoding IMP-9-mediated carbapenem resistance, from outbreak isolate Pseudomonas aeruginosa 96. Antimicrob. Agents Chemother. 57, 3775–3782 (2013).
Han, X. et al. Epidemiological characteristics of OXA-232-producing carbapenem-resistant Klebsiella pneumoniae strains isolated during nosocomial clonal spread associated with environmental Colonization. Microbiol. Spectr. 10, e0257221 (2022).
Corcione, S. et al. Ceftazidime-avibactam (C/A) resistant, meropenem sensitive KPC-producing Klebsiella pneumoniae in ICU setting: We are what we are treated with? Int. J. Mol. Sci. 24, 4767 (2023).
Shields, R. K. et al. Emergence of ceftazidime-avibactam resistance due to plasmid-borne blaKPC-3 mutations during treatment of carbapenem-resistant Klebsiella pneumoniae Infections. Antimicrob. Agents Chemother. 61, e02097-16 (2017).
Shi, Q. et al. Emergence and recovery of ceftazidime-avibactam resistance in blaKPC-33-harboring Klebsiella pneumoniae sequence type 11 isolates in China. Clin. Infect. Dis. 71, S436–s9 (2020).
Galani, I. et al. Emergence of ceftazidime-avibactam resistance through distinct genomic adaptations in KPC-2-producing Klebsiella pneumoniae of sequence type 39 during treatment. Eur. J. Clin. Microbiol. Infect. Dis. 40, 219–224 (2021).
Lorusso, et al. Role of Efflux pumps on antimicrobial resistance in Pseudomonas aeruginosa. Int. J. Mol. Sci. 23, 15779 (2022).
Sherrard, L. J. et al. Emergence and impact of oprD mutations in Pseudomonas aeruginosa strains in cystic fibrosis. J. Cyst. Fibros. 21, e35–e43 (2022).
Zhou, J. et al. Emergence of ceftazidime-avibactam resistance in blaKPC-33-harbouring ST11 Klebsiella pneumoniae in a paediatric patient. Int. J. Antimicrob. Agents 63, 107163 (2024).
Hu, Y. et al. Emergence and expansion of a carbapenem-resistant Pseudomonas aeruginosa clone are associated with plasmid-borne blaKPC-2 and virulence-related genes. mSystems 6, e00154-21 (2021).
Dong, N. et al. Emergence of an extensive drug resistant Pseudomonas aeruginosa strain of chicken origin carrying blaIMP-45, tet(X6), and tmexCD3-toprJ3 on an Inc(pRBL16) plasmid. Microbiol. Spectr. 10, e0228322 (2022).
Zhang, P. et al. Pseudomonas aeruginosa high-risk sequence type 463 co-producing KPC-2 and AFM-1 carbapenemases, China, 2020-2022. Emerg. Infect. Dis. 29, 2136–2140 (2023).
Hu, H. et al. Bloodstream infections caused by Klebsiella pneumoniae carbapenemase-producing P. aeruginosa sequence type 463, associated with high mortality rates in China: a retrospective cohort study. Front. Cell Infect. Microbiol. 11, 756782 (2021).
Partridge, S. R., Kwong, S. M., Firth, N. & Jensen, S. O. Mobile genetic elements associated with antimicrobial resistance. Clin. Microbiol. Rev. 31, e00088-17 (2018).
Fang, Y. et al. An XDR Pseudomonas aeruginosa ST463 strain with an IncP-2 plasmid containing a novel transposon Tn6485f encoding blaIMP-45 and blaAFM- and a second plasmid with two copies of bla(KPC-2). Microbiol. Spectr. 11, e0446222 (2023).
Gaete, M. E. et al. Carbapenemases in Pseudomonas aeruginosa with decreased susceptibility to carbapenems after a decade: from VIM to KPC. Rev. Chil. Infectol. 37, 389–394 (2020).
Oliver, A., Mulet, X., López-Causapé, C. & Juan, C. The increasing threat of Pseudomonas aeruginosa high-risk clones. Drug Resist. Updat. 21-22, 41–59 (2015).
CLSI. Performance Standards for Antimicrobial Susceptibility Testing 34th edn. (Clinical and Laboratory Standards Institute, 2024).
Wick, R. R., Judd, L. M., Gorrie, C. L. & Holt, K. E. Unicycler: resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput. Biol. 13, e1005595 (2017).
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069 (2014).
Zhang, Z., Schwartz, S., Wagner, L. & Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol. 7, 203–214 (2000).
Alikhan, N. F., Petty, N. K., Ben Zakour, N. L. & Beatson, S. A. BLAST ring image generator (BRIG): simple prokaryote genome comparisons. BMC Genomics 12, 402 (2011).
Sullivan, M. J., Petty, N. K. & Beatson, S. A. Easyfig: a genome comparison visualizer. Bioinformatics 27, 1009–1010 (2011).
Li, X. et al. Genomic epidemiology and ceftazidime-avibactam high-level resistance mechanisms of Pseudomonas aeruginosa in China from 2010-2022. Emerg. Microbes Infect. 13, 2324068 (2024).
Zhou, L. et al. Characterization of a novel Tn6485h transposon carrying both blaIMP-45 and blaAFM-1 integrated into the IncP-2 plasmid in a carbapenem-resistant Pseudomonas aeruginosa. J. Glob. Antimicrob. Resist. 35, 307–313 (2023).
Zhang, P. et al. In vivo acquisition of blaKPC-2 with low biological cost in blaAFM-1-harboring ST463 hypervirulent Pseudomonas aeruginosa from a patient with hematologic malignancy. J. Glob. Antimicrob. Resist. 31, 189–195 (2022).
Freddolino, P. L., Goodarzi, H. & Tavazoie, S. Fitness landscape transformation through a single amino acid change in the rho terminator. PLoS Genet. 5, e1002744 (2012).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (82472323 and 82172306), the National Health Commission Scientific Research Fund-Zhejiang Provincial Major Health Science and Technology Plan Project (WKJ-ZJ-2414). We thank Professor Ruichao Li (Yangzhou University) for his technical support.
Author information
Authors and Affiliations
Contributions
Conceptualization: X.L., M.C., Y.T., and Y.Y.; data curation: X.L., M.C., and Y.T.; formal analysis: L.Z., J.Y., Y.H., and Y.Z.; investigation: X.L. and M.H.; methodology: L.Z., Y.Z., X.Z., and Y.H.; validation: X.L. and Y.T.; visualization: L.Z. and J.Y.; writing—original draft: L.Z. and J.Y.; writing—review and editing: X.L., H.L., J.H., and X.Z.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, L., Yao, J., Zhang, Y. et al. Global phylogeography and genetic characterization of carbapenem and ceftazidime-avibactam resistant KPC-33-producing Pseudomonas aeruginosa. npj Antimicrob Resist 3, 3 (2025). https://doi.org/10.1038/s44259-024-00073-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44259-024-00073-0