ABSTRACT
Chronic elevations in interleukin-6 (IL6) signaling have been shown to exacerbate features of cardiometabolic disease. A common variant in the IL6 promoter (location -174 G/C, identifier rs1800795) is associated with increased circulating IL6, and increased cardiometabolic disease incidence in some populations. This study’s objective was to isolate the impact of this gene variant on cardiometabolic responses to metabolic stress, using knock-in mice with a GG wildtype or variant CC genotype for the murine homolog of rs1800795. Male and female IL6 variant CC mice on a high fat diet exhibited enhanced systemic IL6 levels but similar weight gain, energy expenditure, adipose tissue inflammation, glucose homeostasis, and cardiac function relative to control GG mice. Sex differences in the effect of the IL6 variant on cardiomyocyte dimensions were observed, with male variant mice exhibiting smaller cardiomyocyte volume, and female variant mice exhibiting larger cardiomyocyte volume with smaller raw heart mass relative to control GG mice. These findings suggest that, in a controlled experimental setting, the IL6 promoter variant (−174 G/C) does not increase susceptibility to cardiometabolic disease. Further work is required to understand the mechanistic link between this IL6 variant and associated increased cardiometabolic risk observed in population studies.
Similar content being viewed by others
Introduction
Cardiometabolic disease affects approximately 20–25% of the adult population worldwide1, with genetic variation playing a significant role in influencing disease onset and progression2. Genetic variants implicated in cardiometabolic and cardiovascular disease are increasingly being targeted in precision medicine for improved diagnosis, screening, and disease management3. Interleukin-6 (IL6) is a cytokine with pro-inflammatory functions4. Increased circulating adipose-derived IL6 levels induce proliferation of adipose tissue macrophages5,6, associated with adipose tissue expansion and systemic low-grade chronic inflammation, a key factor in the development of insulin resistance7,8,9, and atherosclerotic plaques10. Increased levels of circulating IL6 have additionally been associated with cardiomyocyte hypertrophy11 and decreased cardiac contractile function12. In population studies, chronically elevated IL6 levels have been linked to increased mortality from cardiovascular disease13,14.
A common variant in the IL6 promoter region (−174 G/C, rs1800795) has up to 42% prevalence15 and is associated with increased circulating IL6 in patients with cardiovascular disease and in healthy subjects16. This IL6 variant has been associated with increased prevalence of elevated body mass index and increased risk of developing type 2 diabetes17,18,19. Further, the risk of developing coronary artery disease is increased by 16% (for GC heterozygotes) and 48% (for CC homozygotes) in a meta-analysis of multi-ethnic studies20. In a recent study of >1000 cardiovascular disease cases in a Caucasian population, the homozygous CC genotype was associated with an increased risk of death from adverse cardiovascular events over a 10-year monitoring period21. In contrast, another recent meta-analysis suggested that the IL6 variant C allele is associated with a decreased susceptibility to type 2 diabetes in Asian populations22. Taken together, these studies suggest that the IL6 variant may be a significant risk allele in the pathogenesis of cardiometabolic disease. Because an association has not yet been fully elucidated from existing human studies, this highlights the need for animal studies to determine a potential causative link.
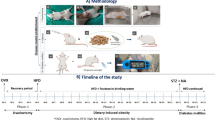
We have previously reported that the IL6 variant C allele heightens production and secretion of IL6 under acute exercise conditions in novel knock-in mice with a wild-type GG or variant CC genotype for the murine homolog (Il6) of rs180079523. There is additional evidence that the IL6 gene displays increased promoter activity when the C allele variant is expressed in fibroblast cells24. We suspect that the IL6 variant C allele may increase production and/or secretion of IL6 under chronic metabolic stress conditions and thus contribute to the development of cardiometabolic disease. Therefore, the aim of our study was to utilize our knock-in mouse model to examine the effects of the Il6 variant, in a tightly controlled experimental setting, on cardiometabolic responses to a high fat diet (HFD). We hypothesized that under HFD conditions, mice with the variant C allele would display greater impairment in cardiometabolic health due to alterations in IL6 signaling.
RESULTS
The Il6 variant CC genotype does not increase weight gain or impair body composition in HFD mice
Due to reported associations between the IL6 variant and increased body mass index (BMI) in human studies17,18,19, we investigated whether mice with the CC genotype gained weight at a greater rate than GG genotype when fed a high-fat diet (HFD). There was no effect of genotype on cumulative weight gain over the 10-week HFD intervention in male or female mice (Fig. 1A). At the end of the HFD intervention, % total weight gain and % fat mass were assessed with no genotype effect observed in either measure for males or females (Fig. 1B, C). Additionally, no differences in fat distribution were observed with the IL6 variant in either male or female mice, as subcutaneous and gonadal (visceral) fat masses were similar between CC and GG mice (Supplementary Table 1). Weights of liver, gastrocnemius, and tibialis anterior muscles additionally remained similar between genotypes (Fig. 1D). These data suggest that the Il6 variant does not exacerbate body weight or fat mass gain in mice on an HFD.
A Total weight gain of male and female mice during 10 weeks of HFD feeding. B Percentage gain in total body weight of male and female mice following the 10 week HFD intervention. C Percentage gain in fat mass of male and female mice following the 10 week HFD intervention (D) Tissue weights of male and female mice following the 10 week HFD intervention. Data are presented as individual values, mean ± SEM. n = 12–21 per group. Data were analyzed by mixed effects model (A) and Student’s T-test (B–D).
The Il6 variant CC genotype does not impair energy expenditure or substrate metabolism in HFD mice
We previously reported that the Il6 variant CC genotype does not alter energy expenditure or substrate metabolism in young chow-fed mice23, – but we speculated that this may be altered in response to a HFD intervention. Energy expenditure over 48 h, when adjusted for body weight, was similar between genotypes in both male (Fig. 2A) and female (Fig. 2B) mice. Similarly, no effect of genotype was observed in whole-body substrate metabolism (measured in respiratory exchange ratio of carbohydrate to fatty acid oxidation) in mice of both sexes (Fig. 2C, D). Collectively, this suggests that the variant is unlikely to alter either of these metabolic parameters in isolation.
A Energy expenditure over 48 h time course for male mice adjusted for body weight + average energy expenditure per light-dark cycle. B Energy expenditure over 48 h time course for female mice adjusted for body weight + average energy expenditure per light-dark cycle. C Respiratory exchange ratios (RER) over 48 h time course (males) + average RER per light-dark cycle (D) RER over 48 h time course (females) + average RER per light-dark cycle. Data are presented as mean ± SEM. n = 8–10 per group. Data were analyzed by two-way ANOVA with Tukey’s multiple comparison tests.
Il6 variant CC genotype increases systemic IL6 levels but does not exacerbate adipose tissue inflammation in HFD mice of either sex
We previously reported that the Il6 variant did not alter basal circulating IL6 in sedentary chow-fed mice23. Given that HFD has been reported to increase IL625, we theorized that an effect of the IL6 variant on IL6 levels may emerge in the HFD setting of heightened systemic and tissue inflammation. Circulating IL6 concentrations were elevated in HFD male and female variant mice (Fig. 3A), similar to prior evidence that the variant enhanced muscle Il6 gene transcription and plasma IL6 levels following exercise23. However, no effect of genotype was observed on subcutaneous and gonadal populations of myeloid cells or macrophages in male or female mice (Fig. 3B, C). Additionally, no effect on the ratio of M1:M2 macrophages was observed between genotypes in subcutaneous and gonadal fat pads of male or female mice (Fig. 3D). Overall, these data suggest that the Il6 variant may increase systemic IL6 levels in HFD-fed mice but does not impact adipose tissue inflammation in HFD-fed mice.
A IL6 concentration (pg/ml) in the plasma of male and female HFD mice. B Total adipose myeloid cell population (% of CD45+ parent cell population) in subcutaneous (sub) and gonadal fat pads of male and female HFD mice. C Total macrophage cell population (% of myeloid cell lineage) in subcutaneous (sub) and gonadal fat pads of male and female HFD mice. D M1 (pro-inflammatory) to M2 (anti-inflammatory) ratio of macrophages per gram of individual fat pad weight in subcutaneous (sub) and gonadal fat pads of male and female HFD mice. Data are presented as individual values, mean ± SEM. n = 8–10 per group. Data were analyzed by Mann–Whitney U-test (A) and Student’s T-test (B–D). Significance was determined at *p < 0.05, **p < 0.01 and ***p < 0.001.
Glucose homeostasis is not impaired by the Il6 variant CC genotype in HFD mice
Given the reported associations between the Il6 variant and impaired glucose tolerance, we investigated the possible effects of the Il6 variant on glucose homeostasis in mice on an HFD. Following a 6 h fast, similar fasting blood glucose levels were observed between genotypes in both male and female mice (Table 1). No differences in glucose or insulin tolerance were observed between genotypes of male or female mice (Table 1, Fig. 4A, B). These data suggest that the Il6 variant does not exacerbate HFD-induced glucose or insulin handling in either male or female mice.
A Glucose tolerance curves for male and female mice. B Insulin tolerance curves for male and female mice. n = 12–21 per group. Data are presented as mean ± SEM.
Cardiac function is not impaired by the Il6 variant CC genotype in HFD mice
Human studies have shown that the IL6 variant has been associated with an increased risk of poor cardiovascular prognosis21, so we investigated the effect of the Il6 variant on cardiac function parameters in HFD mice. Overall, cardiac output and stroke volume remained consistent between genotypes in both males and females (Table 2). Systolic function was unaffected by Il6 genotype, with no differences observed in left ventricular ejection fraction, fractional shortening, or end-systolic volume in both males and females (Table 2). Similarly, diastolic function was also unaffected by Il6 genotype, with no differences observed in the E/e’ ratio of early mitral valve blood flow (E wave) to early mitral annulus tissue velocity (e’ wave), or in end-diastolic volume in both males and females (Table 2). These data suggest that the Il6 variant does not exacerbate HFD-induced cardiac dysfunction in either male or female mice.
The Il6 variant has differential effects on cardiomyocyte size in male and female HFD mice
Pathological cardiac hypertrophy occurs in response to adverse metabolic changes in the heart and generally precedes heart failure26. Although we did not observe an effect of Il6 variant genotype on functional changes in the heart in HFD mice, we sought to determine the effect of the variant on changes in cardiac morphology that may be preceding functional change. Surprisingly, the Il6 variant elicited differential effects on heart and cardiomyocyte size in males and females. Overall, raw heart weights were smaller in variant CC female mice (Fig. 5B), but remained consistent between genotypes when adjusted for body weight (Fig. 5C). This, however, was contradicted by an increase in cardiomyocyte area, length, width and volume (Fig. 5D, Table 3). Increased cardiac Il6 mRNA expression was observed in female CC variant mice, a trend that was also observed in male CC variant mice but did not reach statistical significance (p = 0.0521, Fig. 5E). We therefore estimated that female CC mice hearts contain fewer numbers of cardiomyocytes (~10.4 million) than wild-type GG mice (~14 million) (Table 3). In the variant CC males, however, total heart weights and left ventricular mass remained similar with wild-type GG males (Table 3), but a decrease was observed in cardiomyocyte area, length and volume (Fig. 5, Table 3). We therefore estimated that male CC mice hearts contain greater numbers of cardiomyocytes than wild-type GG mice (Table 3).
A Representative images of WGA-stained cardiomyocytes of male and female GG and CC mice. B Total heart weights (raw values) of male and female GG and CC mice. C Total heart weights (mg/g body weight) of male and female GG and CC mice. D Volume of cardiomyocytes of male and female GG and CC mice, calculated by 7.59 × 10-3 ×length (µm) × width (µm). E Cardiac IL6 mRNA expression levels in male and female GG and CC mice. Data are presented as individual values ± SEM. n = 20–40 images derived from 3 animals per group. Data were analyzed by Student’s T-test. Significance was determined at *p < 0.05 and **p < 0.01
Discussion
The Il6 -174 G/C promoter variant has been shown to associate with increased incidence of cardiometabolic disease. In this study, we utilized a murine model of the IL6 -174 G/C promoter variant23 to examine its effect on cardiometabolic health in response to chronic metabolic stress in a tightly controlled experimental setting. We provide the first evidence that while the Il6 promoter variant has some effect on systemic IL6 levels and cardiac morphology, it has very little impact on the key features of cardiometabolic disease. Variant and control mice exhibited a similar effect of the HFD on weight gain, energy expenditure, substrate utilization, adipose tissue inflammation, glucose homeostasis, and cardiac function. These findings indicate that the Il6 -174 G/C genotype in isolation is unlikely to be a significant risk factor for cardiometabolic disease and the association between disease risk and the Il6 variant observed in human studies is more complex.
We observed interesting sex differences in cardiac hypertrophy in response to the HFD in male and female variant CC mice. In males, cardiomyocytes were smaller on average in variant mice, suggesting that these mice were protected from HFD-induced cardiomyocyte hypertrophy. Meanwhile, total heart weight and left ventricular mass remained consistent between genotypes, so we estimated that variant CC males had greater cardiomyocyte numbers on average. It has previously been demonstrated that HFD feeding induces apoptosis of cardiomyocytes through the inhibition of normal autophagy processes27. Although neither apoptosis nor fibrosis were evaluated in the present study, our estimates of cardiomyocyte number suggests that variant CC males may be protected against HFD-induced apoptosis and further investigation is warranted. Conversely, the opposite effect was observed in variant females, with larger cardiomyocytes but smaller hearts corresponding to fewer cardiomyocytes. Interestingly, estrogen signaling has been shown to be protective against cardiac hypertrophy, apoptosis and fibrosis through expression of estrogen receptors on the surface of cardiomyocytes28,29. Thus, our observation that female IL6 variant mice displayed cardiomyocyte hypertrophy relative to control mice could be explained by a potential effect of the IL6 variant on downregulation of estrogen signaling. A link between decreased estrogen signaling and increased IL6 levels and cardiovascular disease has been identified30,31, which suggests that increased circulating and cardiac IL6 in the variant CC females may be offsetting some of the protective effects of estrogen signaling on the cardiomyocytes. Further investigation is required to confirm this speculative mechanism. Thus, variant CC females may be predisposed to both HFD-induced cardiomyocyte hypertrophy and apoptosis, offsetting the increased cell size with increased cell death and resulting in smaller heart size. Increased IL6 signaling is known to promote cardiac hypertrophy through increased oxidative stress and mitochondrial dysfunction caused by continuous activation of the gp130/STAT3 signaling pathway32. Additionally, cardiomyocytes have been demonstrated to produce IL6 in response to hypoxic and ischemic stress33,34. Both circulating and cardiac IL6 were elevated in the Il6 variant mice (although cardiac Il6 mRNA did not reach significance in males) which may underlie the cardiac remodeling observed. Quantification of cardiac-specific macrophages, apoptosis or fibrosis were not performed in this study but would provide valuable insight into any effects of the increased systemic IL6 signaling in cardiac inflammatory responses. Further research is warranted to assess the effect of this variant on inflammatory responses in the heart. However, these observed differences in cardiac hypertrophy in male and female variant mice were insufficient to induce changes in cardiac function, with both systolic and diastolic function remaining consistent between genotypes in mice of both sexes after 10 weeks on the HFD intervention. This is likely due to no observable differences in left ventricle dimensions (end-diastolic and end-systolic volumes) between genotypes that would likely have functional effects. Overall, these findings suggest that the Il6 variant may alter cellular numbers and morphology in the heart sex-specifically, possibly due to changes in intra-cardiac IL6 signaling, and provides an important lead for further investigation.
Despite the observed alterations in cardiac hypertrophy, we detected very little impact of the Il6 variant in other cardiometabolic health parameters measured in male and female mice. Interestingly, circulating IL6 was elevated in male and female variant mice, but this did not have a measurable impact on adipose tissue inflammation, weight gain, energy expenditure, glucose intolerance or cardiac function. We have previously reported that the Il6 variant upregulates production and secretion of IL6 in muscle in response to exercise23, therefore, it is likely that in a high fat diet setting of elevated IL6 production25,35,36,37, the variant further enhances the production and secretion of IL6 from adipose tissue, but not to a degree where this has measurable effects on the adipose inflammatory response. It is worth noting that male variant mice exhibited greater circulating IL6 levels than females (averaging around 10-fold greater). This is consistent with previous associations between male sex and increased circulating IL6 levels in response to inflammation reported in human studies38,39. Our observations support the associations reported in existing literature between the human IL6 -174 G/C promoter variant and increases in circulating IL6 but contradict the association between this variant and increased prevalence of cardiometabolic disease. Two recent Mendelian randomization studies identified 8 and 34 independent genetic variants in the IL6 and soluble IL-6 receptor (IL6R) gene respectively that potentially associate with cardiovascular disease susceptibility14,40. We therefore speculate that the IL6 -174 G/C promoter variant may be a marker rather than a causative variant, and in isolation it is unlikely to be a significant risk allele for the development of cardiometabolic or cardiovascular disease. However, additive effects of other SNPs in the IL6 or IL6R genes may result in more dramatic alterations in IL6 signaling in response to chronic metabolic stress. Furthermore, some of the associations reported are ethnicity-specific, with the IL6 -174 G/C promoter variant being shown to have a detrimental effect on cardiometabolic risk in Caucasian populations21, but contradictorily a protective effect in Asian populations22. Our observation that the male variant mice had a decreased M1:M2 ratio in the subcutaneous fat could hint at a potential protective effect of this variant under HFD conditions. However, as we did not observe changes in weight gain, glucose homeostasis or cardiac function, we cannot establish whether there is an overall benefit of the variant on cardiometabolic health in our model. Therefore, further research is warranted to investigate potential additive effects of different risk alleles in IL6 and IL6R on prevalence of these diseases.
In our model, cardiomyocyte volume was calculated using a pre-determined rendered cell volume constant derived from studies using rat cardiomyocytes41. We have extrapolated this calculation for use in our mice hearts as we did not have access to 3D cell culture imaging. The average number of cardiomyocytes was then estimated using average total heart weight adjusted for a pre-determined value that up to 85% of an adult mouse heart volume is composed from myocytes42. Because we are using two different assumptions to estimate the number of myocytes in an ‘average’ heart per genotype and sex, and our calculation does not account for any fibrosis or apoptosis that may be present in these hearts, we are limited in the conclusions that can be drawn from these values. Given that a key finding of this study relates to myocardial and cardiomyocyte structural remodeling, assessing markers of cell death and collagen deposition should be prioritized in further characterizing the effect of the IL6 variant on the cardiac response to HFD. Our model is additionally limited by the length of time that the mice were exposed to the HFD intervention. 10–12 weeks of HFD feeding is well documented to be sufficient in inducing obesity, insulin resistance and cardiac dysfunction in C57 mice43,44. None of these parameters were affected by Il6 variant genotype in our mice, but we did observe effects of the Il6 variant on cardiac hypertrophy and circulating IL6. Thus, we speculate that ongoing extended HFD intervention may potentially result in cumulative structural and functional impacts in the hearts of male and female mice that are altered by Il6 variant genotype.
In conclusion, the Il6 promoter variant increases circulating IL6 and alters cardiac hypertrophy in a sex-specific manner but has very little effect on other parameters of cardiometabolic health in both male and female mice under 10-week HFD conditions. This indicates that the IL6 -174 G/C genotype is unlikely to be a significant risk factor for cardiometabolic disease in isolation and other factors (eg additional variants) may play a role in the observed increased disease risk observed in human studies.
Methods
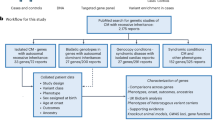
Experimental animals
All animal experiments were approved by the University of Auckland Ethics Committee (AEC Approval Number 2280) and complied with the guidelines and regulations of the Code of Practice for the Care and Use of Animals for Scientific Purposes. Knock-in mice with wild-type GG and variant CC genotypes of a C57Bl/6 J background strain were developed by the Rodent Genome Engineering Resources, Vernon Jansen Unit (VJU) at the University of Auckland, as previously described23. Animals were housed in a temperature-controlled environment at 22 °C with 12-h light/dark cycles. Upon weaning until 10 weeks of age, animals were fed chow diet (TekLad Madison, WI USA) and water was provided ad libitum. At 10 weeks of age, animals were randomly assigned to experimental groups and were fed a high-fat diet (60% fat by calorie content; SF13-092; Specialty Feeds, Australia) ad libitum along with water. All in vivo phenotyping was performed once mice had been on the high-fat diet intervention for 10 weeks, with a minimum of 48 h recovery time in between each phenotyping test. All phenotyping tests were conducted during the light cycle (between 6am and 6pm).
Body composition
Body composition (fat mass) of the mice was assessed using an EchoMRI (LLC, Houston, TX, USA).
Indirect calorimetry
Indirect calorimetry to assess energy expenditure and respiratory exchange ratio was undertaken in individually housed mice using a Promethion Core metabolic cage system (Sable Systems Las Vegas, NV USA). Mice were acclimatized for 48 h before data were collected for 48 h. Mice had ad libitum access to tap water and their study diet throughout both acclimatization and data recording phases. All data were recorded continuously from 12 cages simultaneously and processed into 15 min averages using MacroInterpreter v.2.38.
Glucose and insulin tolerance tests
Insulin (ITT) and glucose (GTT) tolerance tests were performed in 6 h fasted mice, by intraperitoneally injecting a bolus of insulin (0.75 mU/g body weight) or oral gavage of D-glucose (1.5 mg/g body weight). Blood samples were collected from tail vein bleeds at time points 0, 15, 30, 45, 60, 90, 120 min post insulin/glucose bolus, and blood glucose was determined using a handheld glucometer (Accu-chek performa; Roche, Basal, Switzerland).
Echocardiography
Echocardiography was performed following 10 weeks of dietary intervention. The mice were anaesthetized with isoflurane (4%, Baxter Healthcare) mixed with oxygen and room air, and maintained with 1.5–2.5% isoflurane for the duration of the echocardiographic assessment (<45 min per animal). Transthoracic echocardiography was performed using the VEVO LAZR-X 3100 with a MX400 (21–44 Hz) linear array transducer coupled to a digital ultrasound system (FUJIFILM Visual Sonics). Mice were secured to a warm platform containing ECG lead pads throughout the procedure. All analyses were performed by an investigator blinded to sex, genotype, and exercise, using Vevo LAB software version 5.6.1 (FUJIFILM Visual Sonics).
M-mode echocardiography imaging
Left ventricular (LV) M-mode two-dimensional echocardiography was performed in a parasternal short-axis view at the mid papillary level to measure LV wall and chamber dimensions. LV volumes were derived from dimensions using the Teichholz equation (end diastolic volume (EDV) = (7/(2.4 + LV internal diameter at end-diastole (LVIDd))) x LVIDd; end systolic volume (ESV) = (7/(2.4 + LVID at end-systole (LVIDs))) x LVIDs). Systolic function parameters: % ejection fraction ((EDV – ESV)/EDV) x 100), and % fractional shortening ((LVIDd - LVIDs)/(LVIDd) x100). At least 3 consecutive cardiac cycles were sampled per cineloop image and 3 images per animal were analyzed and averaged.
Pulse wave Doppler and tissue Doppler imaging
Pulse wave Doppler and tissue Doppler imaging were acquired from the apical 4 chamber view to assess LV diastolic function parameters: velocity of mitral inflow during early passive filling (E) and velocity of mitral annulus during early passive filling (e’) and E-wave deceleration time. At least 3 cardiac cycles were sampled per cineloop image and 2–4 images per animal were analyzed and averaged.
Tissue and plasma collection
Mice were euthanized via CO2 inhalation. Blood was collected via cardiac puncture (immediately following CO2 euthanasia) into tubes containing EDTA and plasma was separated from whole blood by centrifuging at 6000 g for 5 min. Tissues were rapidly dissected and weighed. Hearts were washed in Kreb’s heart buffer (146 mM NaCl, 4.69 mM KCl, 0.35 mM NaH2PO4, 1.05 mM MgSO4, 10 mM HEPES, 11.1 mM D-Glucose, pH 7.40) and the cardiac mid-sections were dissected and fixed in 4% of paraformaldehyde (Sigma, cat#1.00496). Subcutaneous and gonadal fat pads were collected in cold PBS.
Plasma analysis
Plasma IL6 concentrations were determined using a pre-coated Invitrogen Mouse IL6 Enzyme-Linked Immunosorbence Assay (ELISA) High Sensitivity Kit (ThermoFisher Scientific, Waltham, MA USA) according to the manufacturer’s instructions.
Flow cytometry
To isolate the stromal vascular fraction, visceral (gonadal) and subcutaneous fat pads were excised and chopped into fine pieces in 2 ml of PBS, incubated in 6 ml of digestion buffer (2 mg collagenase B, 0.1 mM CaCl, 0.5% bovine serum albumin, 0.1 mg DNAase in PBS) for 60 min at 37 °C, 250 rpm, and washed through a 100 μm filter with FACS buffer (PBS, 2% calf serum, 0.25 M EDTA). The suspension was pelleted by centrifugation (500 g, 10 mins, 4 °C) and the resulting cell pellet was lysed in red blood cell (RBC) lysing buffer for 5 min to remove residual RBCs, and washed twice in FACS buffer by centrifuging at 300 g for 5 min. The immune cell-enriched pellet was resuspended in 50 μL of FACS buffer containing antibodies (Supplemental Table 1) and incubated on ice for 30 min (light protected). Excess unbound antibody was removed by washing twice in FACS buffer by centrifuging at 300 g for 5 min. The final pellet was then suspended in FACS buffer before being analyzed on a Northern Lights Gating strategy is outlined in Supplemental Fig. 1. Data was analyzed using FlowJo V 10.4.2.
Histology
Cardiac mid-sections were fixed in 4% of paraformaldehyde (Sigma, cat#1.00496) and cryopreserved by soaking in 10% sucrose until tissues sunk, then 20% sucrose and then in 30% sucrose overnight prior to optimal cutting temperature compound (OCT) embedding. Sections were cut (10 μm sections) and stained with Wheat Germ Agglutinin - Alexa Fluor™ 594 (WGA-AF594) conjugate 1:100 (Sigma, cat#W11262). Tissue sections were imaged using a Zeiss LSM 710 inverted confocal microscope under 40x oil magnification. Cardiomyocyte dimensions were traced using ImageJ software (NIH, Bethesda, USA). Cardiomyocyte volume was estimated using a previously determined volume constant of 7.59 × 10-3 for rat myocytes41 that we extrapolated for use in mice and adjusted for the value that up to 85% of an adult mouse heart volume is composed from myocytes42.
Real-time polymerase chain reaction
mRNA was isolated from frozen cardiac tissue using Quick-RNA MiniPrep Plus (Zymo Research Irvine, CA, USA) following the manufacturer’s instructions. RNA concentration and quality were determined by spectrophotometry (Nanodrop One; ThermoFisher Scientific, Waltham, MA, USA) on mRNA from cardiac tissue. mRNA was reverse transcribed using the High Capacity cDNA Reverse Transcription kit (Applied Biosystems, Foster City, CA, USA). Quantitative real-time PCR was performed on a QuantStudio 6 PCR System using the SYBR Select Master Mix (Applied Biosystems, Foster City, CA, USA). Reactions were performed in triplicate and relative quantification achieved using the DDCt method with B2m (β-2 microglobulin) expression as an internal housekeeping control. All mRNA fold changes are expressed as relative to the reference male & female ‘GG control’ groups respectively. Primer sequences are listed in Supplementary Table 3.
Statistical analysis
Statistical analysis was performed using Prism v.8.0.0 (GraphPad Software Incorporated San Diego, CA USA) with statistical significance determined as P < 0.05 and checked for normal distribution where appropriate using the Shapiro-Wilk test. Specific statistical analysis used is indicated in the figure legends.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Khan, A. R. et al. The promising frontier of cardiometabolic syndrome: a new paradigm in cardiology. Cureus 15, e45542 (2023).
Tada, H., Fujino, N., Hayashi, K., Kawashiri, M. A. & Takamura, M. Human genetics and its impact on cardiovascular disease. J. Cardiol. 79, 233–239 (2022).
Semsarian, C. et al. Precision medicine in cardiovascular disease: genetics and impact on phenotypes: JACC focus seminar 1/5. J. Am. Coll. Cardiol. 77, 2517–2530 (2021).
Vozarova, B. et al. Circulating interleukin-6 in relation to adiposity, insulin action, and insulin secretion. Obes. Res. 9, 414 (2001).
Surmi, B. K. & Hasty, A. H. Macrophage infiltration into adipose tissue: initiation, propagation and remodeling. Future Lipidol. 3, 545–556 (2008).
Braune, J. et al. IL-6 regulates M2 polarization and local proliferation of adipose tissue macrophages in obesity. J. Immunol. 198, 2927–2934 (2017).
Bastard, J. P. et al. Adipose tissue IL-6 content correlates with resistance to insulin activation of glucose uptake both in vivo and in vitro. J. Clin. Endocrinol. Metab. 87, 2084–2089 (2002).
Rotter, V., Nagaev, I. & Smith, U. Interleukin-6 (IL-6) induces insulin resistance in 3T3-L1 adipocytes and is, like IL-8 and tumor necrosis factor-alpha, overexpressed in human fat cells from insulin-resistant subjects. J. Biol. Chem. 278, 45777–45784 (2003).
Trujillo, M. E. et al. Interleukin-6 regulates human adipose tissue lipid metabolism and leptin production in vitro. J. Clin. Endocrinol. Metab. 89, 5577–5582 (2004).
Reiss, A. B., Siegart, N. M. & De Leon, J. Interleukin-6 in atherosclerosis: atherogenic or atheroprotective?. Clin. Lipidol. 12, 14–23 (2017).
Wollert, K. C. et al. Cardiotrophin-1 activates a distinct form of cardiac muscle cell hypertrophy: assembly of sarcomeric units in series via gp130/leukemia inhibitory factor receptor-dependent pathways. J. Biol. Chem. 271, 9535–9545 (1996).
Fontes, J. A., Rose, N. R. & Čiháková, D. The varying faces of IL-6: from cardiac protection to cardiac failure. Cytokine 74, 62–68 (2015).
Lindmark, E., Diderholm, E., Wallentin, L. & Siegbahn, A. Relationship between interleukin 6 and mortality in patients with unstable coronary artery disease: effects of an early invasive or noninvasive strategy. Jama 286, 2107–2113 (2001).
Rosa, M. et al. A Mendelian randomization study of IL6 signaling in cardiovascular diseases, immune-related disorders and longevity. NPJ Genom. Med. 4, 23 (2019).
GNOMAD. SNV: 7-22727026 -C-G(GRCh38). 2024 [cited 2025; Available from: https://gnomad.broadinstitute.org/variant/7-22727026-C-G?dataset=gnomad_r4.
Rai, H. et al. Association of interleukin 6 -174 G/C polymorphism with coronary artery disease and circulating IL-6 levels: a systematic review and meta-analysis. Inflamm. Res. 70, 1075–1087 (2021).
Kubaszek, A. et al. Promoter polymorphisms of the TNF-alpha (G-308A) and IL-6 (C-174G) genes predict the conversion from impaired glucose tolerance to type 2 diabetes: the Finnish. Diab. Prev. Study Diab. 52, 1872–1876 (2003).
Möhlig, M. et al. Body mass index and C-174G interleukin-6 promoter polymorphism interact in predicting type 2 diabetes. J. Clin. Endocrinol. Metab. 89, 1885–1890 (2004).
Testa, R. et al. Interleukin-6-174 G > C polymorphism affects the association between IL-6 plasma levels and insulin resistance in type 2 diabetic patients. Diab. Res. Clin. Pract. 71, 299–305 (2006).
Salari, N. et al. The effect of polymorphisms (174G> C and 572C> G) on the Interleukin-6 gene in coronary artery disease: a systematic review and meta-analysis. Genes Environ. 43, 1 (2021).
Schulz, S., Rehm, S., Schlitt, A., Bitter, K. & Reichert, S. The interleukin 6 protein level as well as a genetic variants, (rs1800795, rs1800797) are associated with adverse cardiovascular outcomes within 10-Years Follow-Up. Cells 12, 2722 (2023).
Cheng, Z., Zhang, C. & Mi, Y. IL-6 gene rs1800795 polymorphism and diabetes mellitus: a comprehensive analysis involving 42,150 participants from a meta-analysis. Diabetol. Metab. Syndr. 14, 95 (2022).
Watson, L. E. et al. An IL-6 promoter variant (−174 G/C) augments IL-6 production and alters skeletal muscle transcription in response to exercise in mice. J. Appl. Physiol. 138, 213–225 (2024).
Noss, E. H., Nguyen, H. N., Chang, S. K., Watts, G. F. & Brenner, M. B. Genetic polymorphism directs IL-6 expression in fibroblasts but not selected other cell types. Proc. Natl. Acad. Sci. USA 112, 14948–14953 (2015).
Ko, H. J. et al. Nutrient stress activates inflammation and reduces glucose metabolism by suppressing AMP-activated protein kinase in the heart. Diabetes 58, 2536–2546 (2009).
Wu, J. et al. Variations in energy metabolism precede alterations in cardiac structure and function in hypertrophic preconditioning. Front. Cardiovasc. Med. 7, 602100 (2020).
Hsu, H.-C., Chen, C.-Y., Lee, B.-C. & Chen, M.-F. High-fat diet induces cardiomyocyte apoptosis via the inhibition of autophagy. Eur. J. Nutr. 55, 2245–2254 (2016).
Pedram, A., Razandi, M., O’Mahony, F., Lubahn, D. & Levin, E. R. Estrogen receptor-beta prevents cardiac fibrosis. Mol. Endocrinol. 24, 2152–2165 (2010).
Agrawal, P., Heimbruch, K. E. & Rao, S. Genome-wide maps of transcription regulatory elements and transcription enhancers in development and disease. Compr. Physiol. 9, 439–455 (2018).
Yang, X. P. & Reckelhoff, J. F. Estrogen, hormonal replacement therapy and cardiovascular disease. Curr. Opin. Nephrol. Hypertens. 20, 133–138 (2011).
Rachoń, D., Myśliwska, J., Suchecka-Rachoń, K., Wieckiewicz, J. & Myśliwski, A. Effects of oestrogen deprivation on interleukin-6 production by peripheral blood mononuclear cells of postmenopausal women. J. Endocrinol. 172, 387–395 (2002).
Su, J.-H. et al. Interleukin-6: a novel target for cardio-cerebrovascular diseases. Front. Pharmacol. 12, 745061 (2021).
Yamauchi-Takihara, K. et al. Hypoxic stress induces cardiac myocyte–derived interleukin-6. Circulation 91, 1520–1524 (1995).
Ancey, C. et al. Secretion of IL-6, IL-11 and lif by human cardiomyocytes in primary culture. Cytokine 18, 199–205 (2002).
Park, E. J. et al. Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell 140, 197–208 (2010).
Andrich, D. E. et al. A short-term high-fat diet alters glutathione levels and IL-6 Gene expression in oxidative skeletal muscles of young rats. Front. Physiol. 10, 372 (2019).
Kraakman, M. J. et al. Blocking IL-6 trans-signaling prevents high-fat diet-induced adipose tissue macrophage recruitment but does not improve insulin resistance. Cell Metab. 21, 403–416 (2015).
Spiker, A. M. et al. Interleukin-6 and interleukin-8 gene expressions differ between male and female patients at time of hip arthroscopy for femoroacetabular impingement syndrome. Arthrosc. Sports Med. Rehabil. 6, 100985 (2024).
Sperry, J. L. et al. Male gender is associated with excessive IL-6 expression following severe injury. J. trauma 64, 572–578 (2008).
Georgakis, M. K. et al. Associations of genetically predicted IL-6 signaling with cardiovascular disease risk across population subgroups. BMC Med. 20, 245 (2022).
Satoh, H., Delbridge, L. M., Blatter, L. A. & Bers, D. M. Surface: volume relationship in cardiac myocytes studied with confocal microscopy and membrane capacitance measurements: species-dependence and developmental effects. Biophys. J. 70, 1494–1504 (1996).
Zhou, P. & Pu, W. T. Recounting cardiac cellular composition. Circ. Res. 118, 368–370 (2016).
Siersbæk, M. S. et al. C57BL/6J substrain differences in response to high-fat diet intervention. Sci. Rep. 10, 14052 (2020).
Nguyen, S. et al. The effects of fatty acid composition on cardiac hypertrophy and function in mouse models of diet-induced obesity. J. Nutr. Biochem. 46, 137–142 (2017).
Acknowledgements
This study was funded by the Auckland University Faculty of Medical and Health Science Research Development Fund and the Maurice Wilkins Centre (MWC). T.L.M. was supported by a Rutherford Discovery Fellowship. We acknowledge the Auckland Cytometry ShaRE, Faculty of Science, University of Auckland, for providing Flow Cytometry support. In particular, we thank Thaize Chometon for her assistance in setting up the experiments.
Author information
Authors and Affiliations
Contributions
L.E.W., T.L.M., and K.M.M. conceived and designed the research. P.K. and N.F. developed the knock-in mouse line. L.E.W., M.A., C.L.M., R.F.D., and C.P.H. performed animal monitoring and in-vivo experiments. L.E.W, J.B, J.D., N.B., and R.F.D performed molecular experiments. L.E.W., T.L.M, K.M.M., R.F.D., C.P.H, M.A., K.L.W., C.P., and N.F. interpreted results of experiments. L.E.W., K.M.M., and T.L.M. drafted the manuscript. All authors approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Watson, L., Annandale, M., MacRae, C. et al. An IL6 variant -174 G-C alters cardiac hypertrophy but not cardiometabolic responses to HFD in mice. npj Metab Health Dis 4, 13 (2026). https://doi.org/10.1038/s44324-026-00107-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44324-026-00107-3