Abstract
Mild cognitive impairment (MCI) is a critical window for dementia prevention, yet integrating evidence-based interventions into community care remains a challenge. We developed and evaluated a 24-week community-based health management intervention in Shanghai primary care using a Sequential Multiple Assignment Randomized Trial (SMART). Ninety-three participants were initially randomized to cognitive training with in-person Tai Chi (CT+InTC), virtual reality Tai Chi (CT + VRTC), or a health education group. Non-responders in the intervention arms were re-randomized to either dose escalation or switch formats. Guided by a proposed integrated framework combining the Social Ecological Model and the Consolidated Framework for Implementation Research (SEM–CFIR), the intervention included digital screening, app-based training, culturally adapted Tai Chi, and WeChat peer support. Five of six adaptive pathways improved cognition, with the largest gains in the double-dose CT+InTC group. Compared with the health education group, the intervention improved Montreal Cognitive Assessment (MoCA) by 3.29 points and Memory Guard scores by 3.06 points. In adjusted difference-in-differences models, both high individual attendance and strong community partnership significantly moderated intervention outcomes, with additive and synergistic effects on cognitive improvement. This SMART, multilevel approach was feasible and effective, offering a replicable model for dementia prevention in older adults with MCI.
Similar content being viewed by others
Introduction
Cognitive impairment, including mild cognitive impairment (MCI) and dementia, has become a pressing global public health challenge, with profound implications for healthcare systems, long-term care, and population well-being. Currently, more than 57 million people worldwide are living with dementia, and this number is projected to triple by 2050 as populations age1. Recognizing this escalating burden, the World Health Organization (WHO) has launched a global action plan, calling on countries to prioritize dementia prevention and control as an urgent public health imperative2,3. MCI is an intermediate stage between normal cognitive aging and dementia, marked by detectable cognitive decline that does not significantly impair daily functioning4. From 89 studies with a mean follow up of 5.2 years, conversion risk was 41.5 percent in clinic referred cohorts and 27.0 percent in population based cohorts, with Alzheimer’s dementia the most common outcome5. The average annual progression rate is between five and fifteen percent6. Early identification and intervention for individuals with MCI are therefore considered critical strategies to delay or prevent progression to dementia7. In recent years, community-based management has gained global recognition as an effective approach to facilitate early detection and intervention among older adults at risk of cognitive decline8,9.
Globally, the prevalence of MCI in older adults is estimated at 15.56–19.70%10,11. China, one of the fastest aging countries, reports a prevalence of 15.5% among adults aged 60 years and older. Within China, rates are higher in women than in men and higher in rural than in urban settings; among rural older adults the prevalence is approximately 26.48%12. In recent years, China has introduced multiple dementia prevention and control policies, practice guidelines, and expert consensus statements, emphasizing the goals of “early prevention, early detection, and early intervention”13,14,15,16. The National Action Plan for Dementia 2024–203013 assigns primary care to lead cognitive screening from age 65, expand workforce training, and deliver early intervention that includes health education, lifestyle guidance, risk factor management and cognitive training, with participation of social workers and volunteers; it also strengthens two way referral between community services and hospitals and promotes memory clinics and cognitive disorder services. However, within the primary healthcare system led by community health service centers, the identification and management of MCI have yet to be effectively implemented17,18. Primary care faces multiple challenges, including inadequate workforce capacity, a lack of standardized screening and intervention protocols for MCI management, insufficient training of healthcare providers, underdeveloped cross-sector collaboration mechanisms, low levels of health literacy and willingness to engage among residents, and poor integration of medical and social resources17. These multifaceted barriers intersect, hindering the effective adoption, scale-up, and sustainable implementation of community-based MCI health management.
Compared with cognitively normal older adults, individuals with MCI have approximately a 3.3-fold higher risk of incident dementia19. Modeling studies suggest that delaying the onset or progression of dementia by one year could avert nearly 9.2 million cases by 205020. In the UK Biobank (n = 344,324, 15-year follow-up), lifestyle, comorbidities, and socioeconomic status were the major contributors to dementia risk, with an estimated 47.0–72.6% of dementia being preventable—lifestyle accounting for the largest share21. Beyond non-modifiable factors (age, socioeconomic status, ApoE ε4), cognitive training and lifestyle modification have been associated with reversion from MCI to normal cognition. Among non-pharmacological strategies, physical exercise is recommended by multiple guidelines, expert consensuses, and epidemiological studies as a key measure for MCI management22. Synthesized evidence indicates that structured exercise and healthier lifestyles are associated with improvements in global cognition and executive functions and are included in risk-reduction recommendations1. However, some exercise modalities are high-intensity, physically demanding, or monotonous, limiting long-term attendance in older adults and constraining real-world implementability. This highlights the need for safe, low-impact, socially engaging, and culturally acceptable options. Tai Chi offers a culturally congruent solution in China: its slow, continuous movements entail postural control and sustained attentional shifting, placing demands on balance and executive functions23. Recent systematic review and meta-analysis focused on older adults with MCI found that Tai Chi improved executive function, episodic memory, visuospatial function and global cognition, with a total training dose >1,500 minutes associated with greater gains suggesting a dose–response relationship24,25. These features make Tai Chi a pragmatic and scalable exercise component for community programs in China.
Currently, research on the health management of MCI has predominantly focused on evaluating intervention effects, particularly the efficacy of non-pharmacological interventions, such as physical exercise and cognitive training26,27. Such studies are largely situated within the domain of behavioral interventions; however, behavior change is a dynamic and complex process shaped by the interplay of multi-level and multi-dimensional factors. A substantial body of population-based research has accumulated rich scientific evidence—ranging from analyses of influencing factors to explorations of underlying mechanisms, and from cross-sectional surveys to intervention cohort studies28,29,30. Nevertheless, the majority of these studies have focused on individual-level outcome changes and remain confined within a singular “biomedical” perspective. Systematic investigations into higher-level factors—such as stakeholders, organizational contexts, and interpersonal relationships—are relatively scarce, with limited integration of contextual factors, implementation processes, and sustainability considerations into a comprehensive, multi-dimensional exploration.
A critical issue in the current research on MCI health management in older adults, both domestically and internationally, is the predominant focus on “data outcomes” rather than the practical implementation of interventions in real-world settings. In community contexts, intervention studies often employ strict controls over environmental and individual variables to minimize confounding and ensure internal validity. Although such controlled conditions are essential for establishing intervention efficacy, they may compromise the applicability and generalizability of the findings to real-world practice. Health management strategies that are divorced from the practical setting and the sociocultural context of the target population often experience reduced effectiveness in actual implementation31. Evidence-based interventions, regardless of their demonstrated efficacy, may not directly translate into feasible and actionable practices, thereby creating a persistent “evidence-to-practice” gap32. This limitation constrains the development of effective, scalable, and sustainable implementation strategies for community-based MCI health management in older adults.
To address this gap, implementation science offers a robust theoretical and methodological foundation. By emphasizing a multilevel, multidimensional contextual approach, implementation science systematically examines how interventions are adopted, implemented, scaled, and sustained within real-world settings33,34. The Social Ecological Model provides a comprehensive framework for understanding the interplay of individual, interpersonal, organizational, and community-level determinants35. By integrating these perspectives, the model enables a holistic analysis of multilevel influences on implementation outcomes. Guided by this framework, the present study systematically analyzes the multilevel and multidimensional challenges and barriers to implementing community-based MCI health management. The study aims to generate actionable recommendations to enhance cross-sector collaboration and promote continuous improvement in community-based dementia prevention efforts.
Results
Participant Flow
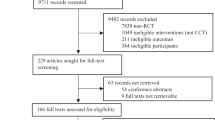
A total of 93 participants were enrolled and randomized into the study. Among them, 26 were assigned to the health education group, 33 to the cognitive training plus in-person Tai Chi (CT + InTC) group, and 34 to the cognitive training plus virtual reality Tai Chi (CT + VRTC) group (Fig. 1).
The flow of participants throughout the study.
During the first 12-week intervention stage, no participants dropped out. In the second stage, five participants withdrew from the intervention groups (7.5% attrition), and six withdrew from the health education group (23.1% attrition). Overall, 82 participants completed the study, resulting in a participation rate of 88.2%.
In addition, There were no significant differences in demographic characteristics between those who completed the intervention (n = 62) and the total screened MCI population (n = 123) in age, sex, education level, or household income (P > 0.05), indicating that the intervention sample was representative of the broader community population.
Baseline characteristics
A total of 82 community-dwelling older adults with MCI were included in the final analysis, comprising 20 participants in the health education group, 30 participants in the cognitive training plus in-person Tai Chi group (CT + InTC), and 32 participants in the cognitive training plus virtual reality Tai Chi group (CT + VRTC). Among them, 29 were male (35.37%) and 53 were female (64.63%), with a mean age of 71.33 ± 6.47 years. Ten participants (12.20%) lived alone, the mean body mass index (BMI) was 24.03 ± 2.75 kg/m², and the mean years of education were 10.54 ± 2.93 years.
At baseline, the mean MoCA score was 22.17 ± 2.14, and the mean Memory Guard Scale score was 27.59 ± 3.53. The mean GDS-15 score was 2.74 ± 1.95. The mean Social Support Rating Scale (SSRS) score was 36.79 ± 6.82.
One-way ANOVA and chi-square tests were performed to compare baseline demographic and clinical characteristics across the three groups. No statistically significant differences were found in sex, age, years of education, living status, BMI, household income, prevalence of chronic diseases, cognitive function (MoCA, Memory Guard Scale), depressive symptoms (GDS-15), social support (SSRS), or sarcopenia-related measures (P > 0.05). Detailed baseline characteristics are presented in Table 1.
Intervention Effectiveness
All older adults with MCI who participated in the intervention were defined as the intervention group. Results from the linear mixed-effects model showed that the intervention group achieved significantly greater improvements in cognitive function compared to the health education group (Table 2). Specifically, the intervention group demonstrated a mean increase in MoCA scores of 3.29 points (95% CI: 2.56, 4.02; P < 0.001) relative to the health education group. Similarly, the intervention group exhibited a higher increase in Memory Guard scores, with a mean difference of 3.06 points (95% CI: 2.02, 4.10; P < 0.001).
In the intervention group, six distinct treatment pathways were formed under the SMART design based on participants’ response status after the first-stage intervention (Table 3). Linear mixed models were used to evaluate cognitive changes across these pathways. Results showed significant improvements in cognitive function scores in five of the six pathways, except for the CT + VRTC → 2CT + 2VRTC group.
Intervention Delivery and Attendance
All Tai Chi sessions, whether delivered as Tai Chi or VR Tai Chi, were implemented exactly as scheduled, with a frequency of once or twice per week, achieving 100% attendance to the intervention plan. Cognitive training tasks were automatically delivered daily by the system, with a 100% task delivery rate. Health education sessions organized by the intervention team were also fully completed as scheduled (100% completion rate).
Attendance for Tai Chi sessions was recorded weekly by community volunteers, while attendance to cognitive training was monitored through backend data from the mobile application. Among participants, 45 (72.58%) achieved the prescribed cognitive training frequency, and 44 (70.97%) met the prescribed Tai Chi session attendance. Notably, 36 participants (58.06%) successfully adhered to both the cognitive training and Tai Chi exercise as planned.
Provider Feedback
Following the intervention, structured interviews and surveys were conducted with 12 service providers involved in implementation, including 3 community physicians, 3 community nurses, and 6 community volunteers, to assess feasibility, acceptability, and implementation challenges.
Overall, 10 providers (83.3%) reported that the intervention could be integrated into routine practice with minor adjustments. Providers generally agreed that the digital screening tool and app-based cognitive training reduced workload and improved standardization. However, 7 providers (58.3%) noted that initial training on digital tools required more time and support than anticipated, and 5 providers (41.7%) reported that some older adults needed additional technical assistance during VR Tai Chi sessions.
Facilitators included strong team collaboration, clear role delineation, and effective communication via WeChat groups. Reported barriers included competing clinical demands, limited time to monitor participant attendance, and varying levels of digital literacy among older adults.
Providers recommended adding a dedicated coordinator and offering regular training sessions to further enhance sustainability and implementation efficiency.
Interaction Effects of Multi-Level Socio-Ecological Factors on Intervention Effectiveness
To further examine the influence of individual- and organizational-level factors on intervention effectiveness, this study incorporated two key variables: individual attendance (i.e., attendance rates to cognitive training and Tai Chi sessions) and community partnership strength (i.e., the mean partnership score among community stakeholders involved in MCI health management). A DID analysis was performed across adjusted models (Table 4). The main DID effect on Memory Guard was 2.458 (adjusted p = 0.033; adjusted 95% CI: 0.204 to 4.713). For two way moderation, Participants with high attendance demonstrated an additional gain of 3.401 points compared to those with low attendance (adjusted p = 0.018; adjusted 95% CI: 0.500 to 6.301). Those from communities with a high organizational environment showed an additional increase of 2.407 points relative to those from low partnership settings (adjusted p = 0.023; adjusted 95% CI: 0.339 to 4.476). For the three-way interaction, participants with both high attendance and a high organizational environment achieved an additional gain of 3.289 points (adjusted p = 0.015; adjusted 95% CI: 0.467 to 6.111), while the other three-way contrasts were not statistically significant after multiple comparison correction. These findings highlight the significant moderating roles of both individual- and organizational-level factors in shaping intervention outcomes and underscore the necessity of integrating multi-level strategies to optimize cognitive health interventions in community settings.
Secondary Outcomes
The intervention group had a higher increase in SSRS scores (mean difference: 1.60; 95% CI: 0.36 to 2.83; p = 0.012) and EQ-VAS scores (mean difference: 4.56; 95% CI: 2.08 to 7.04; P < 0.001). Additionally, the intervention group showed a greater reduction in depressive symptoms, as reflected by lower GDS-15 scores compared to the health education group.
Discussion
This study established an integrated theoretical framework by combining the SEM35 and the CFIR36,37 to systematically guide the development and implementation of a dual-task intervention for community-dwelling older adults with MCI. The SEM-CFIR framework not only provided a comprehensive perspective across individual, interpersonal, organizational, and policy levels, but also incorporated public involvement throughout the intervention design and implementation process, ensuring the intervention was needs-driven and culturally appropriate. By actively engaging community stakeholders and older adult participants, the study systematically identified multilevel facilitators and barriers to implementation, and offered structured theoretical support for co-designing intervention strategies, mobilizing stakeholders, and fostering cross-sector collaboration. The study’s findings demonstrate the practical applicability and effectiveness of the SEM-CFIR framework in real-world community interventions, highlighting its value as a bridging framework that connects theory with practice while promoting public involvement in scientific research.
Unlike previous implementation studies employing frameworks, such as RE-AIM38 or i-PARIHS39 which typically emphasize discrete implementation outcomes or contextual elements, the integrated SEM-CFIR framework in this study uniquely highlighted the dynamic interactions across individual, interpersonal, organizational, and broader community levels. By explicitly incorporating public involvement and continuous stakeholder engagement40, this framework provided nuanced insights into how multilevel contextual determinants synergistically influenced intervention fidelity, acceptability, and sustainability, contributing to a deeper understanding of complex intervention processes in community settings. Furthermore, the deliberate incorporation of public involvement strategies, such as preference surveys and structured stakeholder interviews was integral in tailoring interventions to the specific sociocultural contexts of older adults with MCI. Such co-design approaches not only enhanced intervention acceptability but also fostered higher attendance by ensuring alignment with participant preferences and real-world conditions. Our findings suggest that meaningful public involvement is an essential component of effective implementation science practices, providing a robust pathway for enhancing the relevance, uptake, and longevity of community-based health interventions.
Multiple randomized controlled trials have confirmed that non-pharmacological interventions are an important strategy for managing MCI41,42. However, even evidence-based interventions may vary in their optimal sequence and dosage across individuals, and not all patients exhibit consistent responses; in some cases, mismatched interventions may even reduce attendance43,44. In this study, we incorporated a SMART design into the intervention protocol, allowing for adaptive modifications for participants who did not respond after the first-stage intervention. The results showed that among the multiple intervention pathways generated under the SMART design, all pathways except for the “CT + VRTC → 2CT + 2VRTC” pathway achieved significant improvements in cognitive function.
Furthermore, the study achieved favorable results in terms of intervention feasibility and process evaluation. The intervention protocol was implemented with 100% fidelity; attendance rates were 72.58% for cognitive training, 70.97% for Tai Chi training, and 58.06% for dual-task training. The loss-to-follow-up rate in the intervention group was 7.46%, notably lower than that in the health education group (23.08%). These findings indicate that the intervention, when coupled with implementation strategies, was feasible, acceptable, and well-received within the community setting, underscoring the value of integrating an implementation science framework into intervention development and optimization.
This study employed a dual-task training model combining cognitive training with Tai Chi exercise. Tai Chi, a traditional Chinese practice widely embraced by older adults, emphasizes fluid and progressive movements while promoting flexibility, balance, and coordination. The findings of Li et al.45 are consistent with those of this study; in their research, 304 older adults with MCI were randomized into three groups: cognitive training combined with Tai Chi, Tai Chi exercise alone, and stretching exercise. The intervention was delivered twice per week for 60 minutes per session over 24 weeks. Their results showed that the dual-task training led to significantly greater improvements in MoCA scores. Compared to single-task interventions, dual-task training not only increased cognitive load but also provided broader and more diverse cognitive stimulation, supporting the development of multiple cognitive domains. Overall, dual-task training more closely mirrors the multitasking demands of daily life, thereby better meeting the complex cognitive needs of older adults46.
Randomized evidence in older adults with MCI shows that Tai Chi improves cognition and elevates plasma brain-derived neurotrophic factor, indicating an exercise-induced trophic milieu supportive of plastic change47. Resting-state MRI studies report strengthened hippocampus–medial prefrontal connectivity after longitudinal Tai Chi practice alongside memory gains, consistent with network-level consolidation48. Cerebrovascular studies further suggest improvements in cerebral blood-flow indices with Tai Chi, providing a hemodynamic substrate for cognitively demanding activities49. In turn, cognitive training provides guidance, selecting, stabilizing and integrating newly modifiable synapses and newly generated neurons into functional circuits50. Because several of these trophic signals are transient following exercise, delivering cognitive engagement in close temporal proximity may magnify consolidation, and some studies suggest order effects51,52. The strength and consistency of these synergies likely depend on exercise intensity, dose and attendance, and the evidence base remains heterogeneous. Accordingly, the optimal way to combine modalities, including whether delivery should be simultaneous or sequential and which ordering is most effective, remains an open question and motivates direct comparison trials that jointly optimize efficacy, attendance and real world feasibility. Our program integrates a culturally familiar physical practice with structured cognitive training in a community setting; although temporal order was not experimentally manipulated, the design is intended to harness complementary biological pathways while maintaining implementability.
In addition, this study innovatively incorporated immersive VR Tai Chi training. This VR-based intervention provided older adults with an experience akin to being in a real-life environment through vivid and realistic virtual scenes, soothing background music, and visual guidance via Tai Chi diagrams. Our findings showed that after 12 weeks of intervention, the CT + VRTC group demonstrated superior cognitive improvements compared to the CT+InTC group. Similarly, Liao et al.53 conducted a randomized trial involving 34 community-dwelling older adults with MCI, allocating them to either a “VR physical exercise + cognitive training” group or a “conventional physical exercise + cognitive training” group, with sessions held three times per week for 60 minutes each over 12 weeks. Their results demonstrated greater overall cognitive improvements in the VR group compared to the conventional group. Furthermore, their study found that participants in the VR group exhibited reduced prefrontal cortex activation and improved neural efficiency in post-intervention hemodynamic assessments. These findings suggest that VR-based interventions, empowered by digital technology, hold potential advantages for improving cognitive function among community-dwelling older adults with MCI.
Further analysis revealed significant interaction effects across multiple levels of the SEM on intervention outcomes. Difference-in-differences analysis indicated that both individual attendance and organizational-level community partnership scores were independently associated with improvements in cognitive function. Participants with high attendance and strong community partnerships experienced the greatest cognitive gains (net increase of 6.516 points on the Memory Guard Scale). These findings align with the core premise of the SEM54, which posits that health behaviors are shaped not only by individual factors but also by interpersonal, organizational, and policy-level influences.
The value of the SEM lies in its emphasis on multi-level and multidimensional determinants and the dynamic interactions between levels35,54. Our study confirmed that internal motivation particularly attendance is a key determinant of intervention success. Previous studies have shown that approximately three-quarters of MCI participants initially agree to participate, but an average of 15% drop out each month, with the highest attrition occurring in the fourth month55. Factors, such as sex, marital status, education, disease awareness, health beliefs, self-efficacy, social support, intervention format and efficacy, and contextual factors have all been shown to influence attendance56. Therefore, enhancing social support and building strong stakeholder partnerships are essential strategies to foster intrinsic motivation and promote “re-socialization” among older adults with MCI in community settings.
At the organizational level, community partnership scores—developed from the perspectives of MCI health management stakeholders—revealed that lower partnership levels were associated with poorer intervention outcomes. As the intervention requires coordination among general practitioners, community workers, and volunteers, weak partnership intentions may compromise communication, responsibility sharing, and collaborative execution17. Previous research has shown that community partnership functions as a mediating factor between perceived organizational barriers and behavioral intention57,58. Lower levels of partnership not only reflect weaker collaborative foundations but also result in diminished motivation to engage in health management behaviors, ultimately undermining intervention effectiveness. Community partnership moderated cognitive gains in the DID analysis. This is consistent with CFIR constructs, such as networks and communication, implementation climate and readiness for implementation, and with the SEM at the organizational levels37,54,59. Stronger partnership plausibly improves reach, attendance and fidelity through social support, higher organizational readiness, clear roles and routine two way communication. Reviews of community engagement report better recruitment, retention and intervention fit under genuine partnership60,61. Accordingly, partnership should be treated as an active ingredient; plans should specify stakeholder mapping, shared goals, role clarity and regular communication.
Taken together, this study reinforces the utility of the SEM in elucidating the multilevel drivers of health behavior and highlights that intervention success is determined not only by the precision of the intervention strategy itself but also by the interplay of individual motivation, interpersonal support, and organizational readiness. Even when interventions are well-matched to individual needs, the absence of robust multilevel support systems may significantly limit their effectiveness. Based on the findings and implementation processes described above, we developed an integrated community-based MCI health management framework (Fig. 2).
The model highlights the systematic integration of digital screening and diagnostic tools, adaptive dual-task intervention strategies informed by the SMART design, and a supportive community service network leveraging digital health technologies. This framework embodies a comprehensive approach aligned with the SEM-CFIR model, emphasizing multi-level stakeholder collaboration, targeted public involvement, and adaptive intervention tailoring to maximize acceptability, feasibility, and sustained health outcomes in real-world community settings.
This study has several limitations. First, the sample was drawn from a limited setting (a specific community or region with a relatively small number of participants, especially in the health education group), which may constrain the generalizability of the findings to other populations or healthcare contexts. In addition, the analysis included 82 participants and, for a SMART with six adaptive pathways, this number is small and reduces power for subgroup comparisons. Loss to follow up was higher in the health education group than in the intervention group, which may bias between group estimates. Accordingly, pathway findings are exploratory and should be confirmed in larger multisite studies. Second, the intervention design and implementation relied on community resources (e.g. trained volunteers and VR equipment) and was not blinded, potentially introducing selection or performance biases and limiting reproducibility in less-equipped settings. Future work will adapt the program to local conditions by mapping available resources and service pathways, integrating delivery into existing primary care workflows, co designing culturally and linguistically appropriate materials with stakeholders, defining a minimal core package with clear dose and schedule, and providing standardized training. Third, the follow-up duration was 24 weeks, so we could not observe long-term sustainability of the cognitive improvements or capture any delayed effects; a longer-term follow-up would be needed to confirm whether the benefits are maintained over time. Finally, while we evaluated key factors at the individual and organizational levels, other unmeasured contextual variables may have influenced the outcomes. We did not collect detailed provider characteristics or measures of inter institutional coordination, which may moderate implementation; future studies will collect these variables and examine their moderating effects on outcome. In addition, we did not conduct an economic evaluation or an evaluation of scale up potential; future work will collect resource and cost data, evaluate cost effectiveness and budget impact, and assess scale up across sites. These limitations suggest caution in interpreting the results, and highlight the need for further studies with larger, more diverse samples, extended follow-up periods, and refined methodologies to validate and extend our findings. Additionally, despite the comprehensive SEM-CFIR integration, certain potential influencing factors, such as detailed provider characteristics (e.g., years of experience, attitudes towards technology), were not explicitly measured. These factors might further moderate implementation outcomes, suggesting future research should incorporate more granular assessments of provider-level determinants.
This study demonstrated the feasibility and effectiveness of a stakeholder-informed, adaptively designed behavioral intervention for older adults with MCI within community-based primary care settings. The intervention integrated culturally tailored Tai Chi training with intelligent cognitive training tools, addressing key challenges related to engagement, accessibility, and sustainability. The application of a SMART design enabled real-time adaptation of intervention pathways based on individual responsiveness, thereby enhancing both personalization and implementation efficiency. The findings highlight the critical role of participatory implementation strategies, digital augmentation, and structured community coordination in optimizing complex health interventions for cognitively vulnerable populations. The study provides implementation-relevant evidence to support dementia prevention and healthy aging initiatives, especially within primary care systems and under-resourced community settings.
Methods
Theoretical Framework and Study Design
This study adopts an implementation science research design, guided by the Social Ecological Model (SEM) and the Consolidated Framework for Implementation Research (CFIR)36,54, to develop and implement a community-based health management intervention for MCI within a multilevel and multidimensional contextual framework. A nested mixed-methods design was employed, integrating qualitative and quantitative approaches across four stages: needs assessment, intervention development, intervention implementation, and process evaluation.
In the needs assessment stage, qualitative interview guides and survey instruments were developed based on CFIR-SEM framework to collect multilevel data on the needs, barriers, and facilitators identified by service providers, managers, and older adults. Findings from the needs assessment informed the identification of key contextual determinants and were used to develop intervention content and implementation strategies tailored to the specific community context, enhancing feasibility and contextual fit.
During the intervention implementation stage, a Sequential Multiple Assignment Randomized Trial (SMART) design was employed to dynamically adjust intervention pathways based on participant responses and contextual feedback, enabling personalized and adaptive intervention delivery43. The study evaluated both clinical outcomes (cognitive function improvement) and implementation outcomes, including feasibility, reach, attendance, and sustainability.
Process evaluation was conducted to systematically identify key contextual factors influencing implementation. Additionally, the study explored how individual-level factors and organizational-level factors moderated intervention outcomes. This research provides empirical evidence and practical insights to support the scalable and sustainable implementation of MCI health management interventions in community settings (Fig. 3).
This framework illustrates how multilevel contextual determinants—including organizational, interpersonal, and individual domains—interact dynamically to influence the development, execution, and outcomes of community-based adaptive interventions. The implementation process domain details specific steps, from stakeholder engagement and contextualized strategy development to adaptive execution and data-driven refinement, emphasizing iterative feedback mechanisms for optimizing intervention effectiveness, acceptability, and sustainability.
Participants & Setting
All participants in this study were recruited from a prior community-based cognitive screening program conducted in Shanghai62, targeting older adults aged 60 years and above. Recruitment for the present trial took place from 1 April 2023 to 31 July 2023, and the intervention was delivered from May 2023 through December 2023. Building on that screening, individuals identified as at high risk by the digital tool underwent standardized clinical assessment by neurologists, who confirmed MCI.
Inclusion criteria were: (1) clinically diagnosed with MCI63; (2) no regular participation in Tai Chi, Baduanjin, or similar exercise programs in the past three months; (3) voluntary participation with signed informed consent; and (4) basic proficiency in operating a smartphone, assessed by participant self report. Exclusion criteria included: (1) cognitive impairment due to non-degenerative causes (e.g., cerebrovascular disease); (2) a history of neurological disorders (e.g., Parkinson’s disease); (3) severe psychiatric illness; (4) contraindications to exercise, color blindness, or other conditions impairing ability to participate; (5) current participation in other rehabilitation therapies; (6) unwillingness to undergo randomization or complete the 24-week intervention and follow-up; and (7) any other condition deemed by the research team to preclude study completion. Individuals meeting any exclusion criterion were not eligible for the study.
Discontinuation criteria were: (1) Occurrence during the study of serious safety events or newly diagnosed conditions requiring surgical or other intensive treatments (e.g., malignancy), making continued participation inappropriate; (2) Voluntary withdrawal of consent by the participant at any time. Elimination criteria were: (1) Participants mistakenly enrolled who did not actually meet the inclusion criteria; (2) Participants with incomplete data or records that precluded assessment of primary outcomes or safety; (3) Participants failing to engage in the prescribed intervention training in accordance with the protocol. Withdrawal criteria were: Participants who met the eligibility criteria and provided informed consent but failed to complete the scheduled intervention plan, withdrew, or were lost to follow-up during the study period, resulting in uncertainty about outcomes, were classified as withdrawn cases. For all withdrawals, study staff made every effort to contact participants to encourage continued engagement. If participants declined to resume, the reasons for withdrawal were documented in detail in the study records.
The intervention was implemented within the primary healthcare system led by community health service centers, integrating medical care, rehabilitation, and health education resources. It was conducted across three urban communities located in Huangpu District, Pudong New Area, and Putuo District of Shanghai. Intervention sites included community health centers and neighborhood committee activity rooms. These districts were selected to reflect diverse community contexts in terms of socioeconomic status and healthcare infrastructure. Before contacting participants, participating centers were selected. This study team then convened a training meeting with site leadership and stakeholders to review the intervention content, standard operating procedures, role assignments, and referral workflows. Site specific WeChat groups were created for ongoing coordination. Providers subsequently ran awareness activities at their facilities to inform eligible residents. Interested residents attended two orientation sessions, during which staff explained study aims and procedures, completed screening, obtained written informed consent, and provided hands on onboarding to the digital cognitive program and the Tai Chi classes. Family members or caregivers were encouraged to attend these sessions to support technology use and attendance. Each site’s WeChat group was used to deliver reminders, share materials, answer questions, and monitor engagement. Full operational details are provided in Supplementary Appendix 1. This study was approved by the Ethics Committee of Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (No. KY2023-24). Written informed consent was obtained from all participants. This trial was registered with Chinese Clinical Trial Registry in January 2021, ChiCTR2100042748.
SMART Intervention Design
Based on the best available empirical evidence and practice guidelines both domestically and internationally4,29,64,65,66, and taking into account the feasibility of community-based implementation. The intervention design was developed based on the findings from implementation strategy activities, including a public involvement survey and qualitative assessments with stakeholders. These activities identified key preferences, needs, and contextual factors that informed the content and structure of the intervention.
This study designed a dual-task intervention combining cognitive training with Tai Chi exercise, using a SMART design (Fig. 4). The intervention lasted 24 weeks, divided into two 12-week stages. The initial intervention protocol consisted of 1 hour per week of cognitive training and 1 hour per week of Tai Chi exercise, delivered either in virtual reality (VR)-based or in-person formats. The in-person Tai Chi sessions were delivered in a group setting, fostering social interaction and attendance. In contrast, the virtual reality (VR) Tai Chi was practiced individually, with peer support provided via a group chat. Participants were randomly assigned to one of three groups: cognitive training plus Tai Chi (CT + InTC), cognitive training plus VR Tai Chi (CT + VRTC) and health education group. Tai Chi training used the 24 form simplified routine, delivered either in person in groups or guided in virtual reality. Cognitive training comprised adaptive app based exercises across memory, attention, executive function, logical reasoning, visuospatial skills and reaction speed, delivered as brief daily tasks. All three study groups received the same health education sessions.
SMART Intervention Design.
At the end of the first stage, intervention response was evaluated based on changes in cognitive function and participants’ self-reported perceptions of effectiveness. Based on response status, intervention protocols were adapted in the second stage as follows: responders in each group continued their original protocol; non-responders in the CT + InTC group were re-randomized to either CT + VRTC or a doubled-dose CT + InTC regimen (2 hours/week for both cognitive training and Tai Chi); non-responders in the CT + VRTC group were re-randomized to either CT + InTC or a doubled-dose CT + VRTC. We implemented an adaptive component to promptly adjust care for early non-responders by escalating dose or switching modality. This design also permits estimation of the most effective second-stage strategy conditional on early non-response. This SMART design enabled individualized and adaptive intervention pathways based on participant responsiveness, with the goal of enhancing both intervention effectiveness and sustainability. The health education group was included to evaluate the comparative effectiveness of the SMART intervention framework.
This study employed a SMART design. Based on a precision‑based sample size estimation approach for pilot SMART67, we calculated that at least 80 participants were required to estimate the marginal mean outcomes under each embedded dynamic treatment regime within ±0.45 standard deviations, assuming a 60% response rate and a two‑sided α of 0.05. Participants were randomized at enrollment using a computer‑generated sequence with equal allocation probability across the three study groups. Recruitment proceeded on a rolling basis. To ensure valid comparisons across all three groups, the initial allocation ensured that each group reached the minimum required sample size (n ≥ 26). The remaining participants were allocated to the two adaptive intervention groups to meet second-stage SMART requirements, resulting in final group sizes of 34 in CT+InTC, 33 in CT + VRTC, and 26 in the health education group. Allocation concealment was maintained, with assignments released only after eligibility confirmation. Additional methodological details are provided in Supplementary Appendix 1.
Implementation strategy
The implementation strategies in this study were systematically developed from empirical work comprising in depth semi structured interviews with 14 community healthcare providers and administrators17, and structured questionnaires from 123 community dwelling older adults with cognitive impairment. Interview data were analyzed thematically with reference to the CFIR framework, and questionnaire data were summarized using descriptive statistics. Drawing on these sources, the study identified multilevel contextual determinants at the individual level (intervention efficacy and attendance), the interpersonal level (social support), and the organizational level (resource availability and cross sector collaboration) that acted as barriers and facilitators to implementation.
Building on these insights, the study not only adapted intervention components, such as selecting Tai Chi as a culturally appropriate exercise modality, incorporating WeChat-based peer support, and developing tailored health education content, but also translated these findings into a standardized, operational, and replicable set of implementation strategies. These strategies specified stakeholder roles and responsibilities, resource mobilization mechanisms, intervention workflows, and process monitoring procedures, creating a structured pathway across individual, organizational, and community levels.
In addition to qualitative findings, the study incorporated two quantitative assessments—including a public involvement survey among older adults with MCI and a structured survey among healthcare providers—to further inform strategy development. First, a structured survey using the Partnership Analysis Tool57 (developed by VicHealth) was administered to community healthcare providers and community workers to evaluate cross-sector partnerships across seven dimensions: importance of partnerships, partner selection, effectiveness, collaborative planning, action implementation, implementation barriers, and sustainability. The survey revealed a mean partnership score of 64.38 ± 17.57, indicating low partnership strength and limited cross-sector collaboration capacity within community settings. Second, a public involvement survey was conducted with 123 older adults diagnosed with MCI to assess their preferences and acceptance of non-pharmacological interventions. Results showed that 91 participants (73.98%) were willing to adhere to at least two sessions of exercise per week, while 79 participants (64.23%) expressed willingness to adhere to five to six sessions of cognitive training per week. The detailed survey instruments used in this study are provided in the Supplementary Appendix 1.
Informed by the SEM-CFIR, these findings guided the formulation of a comprehensive, multi-level set of implementation strategies. The aim was to enhance the accessibility, acceptability, attendance, and sustainability of community-based MCI health management interventions, while addressing the complex contextual challenges inherent in real-world primary care settings. The key strategies are summarized in Table 5.
The implementation strategies developed were systematically informed by qualitative insights and quantitative assessments from stakeholder-driven interviews and structured surveys (Table 5). For example, qualitative feedback from healthcare providers identifying barriers, such as ‘insufficient staffing,’ ‘lack of standard procedures,’ and ‘time constraints’ led to the integration of digital cognitive tools (including digital screening and app-based cognitive training) to reduce provider workload and standardize intervention delivery. Participant preferences for culturally familiar activities informed the selection of Tai Chi as the primary exercise modality. Additionally, structured WeChat communication groups were established in response to expressed needs for continuous peer support among older adults and community workers. These operational steps were standardized into a replicable implementation protocol detailing specific roles, workflows, and sustainability measures, ultimately enhancing intervention acceptability, feasibility, and contextual fit.
Trial Outcomes
The primary outcome of this study is the improvement in cognitive function among community-dwelling older adults with MCI, assessed using the Montreal Cognitive Assessment (MoCA) and the Memory Guard Scale at baseline, 12 weeks, and 24 weeks. We selected the MoCA because it was specifically developed to detect MCI and showed higher sensitivity than brief screens, such as the MMSE across diverse settings and populations68. Its psychometric performance in multiethnic/Asian cohorts is well documented69, and importantly, MoCA has demonstrated responsiveness to change in MCI intervention trials24. Besides, we included Memory Guard, an internally developed and validated computerized neuropsychological assessment for older adults. Delivered via mobile/tablet with randomized, gamified clinic-visit tasks, it captures multiple domains and time-based features; in community validation against MoCA, it showed high diagnostic performance for MCI with accuracy 93.75 percent, area under the curve 0.923, sensitivity 91.67 percent, and specificity 95.45 percent70. By complementing MoCA’s global score with a memory-focused, repeatable measure, Memory Guard serves as a low-burden secondary outcome in our study. In addition, the primary outcome includes an analysis of how individual-level and organizational-level factors influence intervention effectiveness, aiming to explore the moderating effects of key contextual elements on cognitive outcomes.
The secondary outcomes focus on evaluating the implementation process, guided by the “Process” domain of the CFIR framework and a socio-ecological perspective at both individual and organizational levels. Key implementation outcomes include implementation fidelity (whether weekly intervention sessions were delivered as planned), attendance (the proportion of scheduled sessions completed by participhants), reach (the proportion of eligible individuals who were recruited and initiated the intervention), and acceptability and satisfaction (assessed via a post-intervention questionnaire). Data sources for the implementation evaluation include intervention logs, standardized checklists completed by the research team and community volunteers, weekly participant-completed training logs, and satisfaction surveys conducted after the intervention.
Additional secondary health outcomes include depressive symptoms (measured by the Geriatric Depression Scale-15)71, muscle function, and quality of life (measured by the EQ-VAS)72, evaluated at baseline and 24 weeks.
Data Analysis
Statistical analyses were conducted using SPSS 26.0 software. Categorical variables were presented as counts and percentages (%), whereas continuous variables were summarized using means ± standard deviations (SD) or medians. For continuous data meeting assumptions of normality, analysis of variance (ANOVA) was utilized for between-group comparisons; otherwise, the Kruskal-Wallis test was applied.
To rigorously evaluate the intervention effects while controlling for potential confounders, linear mixed models (LMMs) were employed. Specifically, demographic and baseline characteristics including sex, age, living alone status, average household annual income, education level, and chronic conditions were incorporated as fixed effects in the model to assess between-group differences in outcome measures before and after intervention. Additionally, within-group analyses were conducted using LMMs to compare outcome changes across different adaptive intervention pathways established by the SMART design.
Following the statistical framework proposed by Lu et al, we implemented a weighted-and-replicated estimation approach combined with difference-in-differences (DID) analysis to compare embedded DTRs using repeated measures spanning multiple treatment stages73. This integration provides doubly robust protection against model misspecification: SMART-specific inverse probability weights correct for differential selection probabilities across treatment sequences, while the DID structure adjusts for time-invariant confounding and secular trends, yielding unbiased estimates under the sequential randomization and consistency assumptions. Participants were initially randomized with equal probability at the first stage. At the second stage, treatment allocation probabilities varied by initial response status: responders and education group continued their initial intervention without re-randomization, while non-responders were re-randomized to alternative interventions with equal probability. Individual-level weights were calculated as the inverse probability of receiving the observed treatment sequence. These weights were subsequently normalized by the ratio of total sample size to the sum of all weights to maintain the original sample size in weighted analyses. This weighting strategy ensures unbiased estimation of marginal mean trajectories under each embedded DTR by appropriately accounting for differential selection probabilities across treatment sequences. Given the multiple pairwise comparisons generated by moderation analyses, we implemented the Šidák correction to control the family-wise error rate74.
Data availability
The datasets generated and analyzed during the current study are not publicly available due to participant privacy and confidentiality agreements but are available from the corresponding author on reasonable request.
References
Livingston, G. et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet 404, 572–628 (2024).
Risk Reduction of Cognitive Decline and Dementia: WHO Guidelines. (World Health Organization, 2019).
WHO.Global health observatory: dementia standards/guidelines/protocols, https://www.who.int/data/gho/data/themes/global-dementia-observatory-gdo/diagnosis-treatment-and-care/dementia-standards-guidelines-protocols
Petersen, R. C. et al. Practice guideline update summary: mild cognitive impairment: report of the guideline development, dissemination, and implementation subcommittee of the American Academy of Neurology. Neurology 90, 126–135 (2018).
Salemme, S. et al. The prognosis of mild cognitive impairment: a systematic review and meta-analysis. Alzheimers Dement 17, e70074 (2025).
Dunne, R. A. et al. Mild cognitive impairment: the Manchester consensus. Age Ageing 50, 72–80 (2021).
Sanz-Blasco, R. et al. Transition from mild cognitive impairment to normal cognition: Determining the predictors of reversion with multi-state Markov models. Alzheimers Dement 18, 1177–1185 (2022).
Powell C., Tomlinson J., Quinn C., Fylan B. Interventions for self-management of medicines for community-dwelling people with dementia and mild cognitive impairment and their family carers: a systematic review. Age Ageing 51, afac089 (2022).
Woodward, M. et al. Nationally informed recommendations on approaching the detection, assessment, and management of mild cognitive impairment. J. Alzheimers Dis.89, 803–809 (2022).
Bai W., et al Worldwide prevalence of mild cognitive impairment among community dwellers aged 50 years and older: a meta-analysis and systematic review of epidemiology studies. Age Ageing 51, afac173 (2022).
Song, W. X. et al. Evidence from a meta-analysis and systematic review reveals the global prevalence of mild cognitive impairment. Front. Aging Neurosci. 15, 1227112 (2023).
Cong, L. et al. Mild cognitive impairment among rural-dwelling older adults in China: a community-based study. Alzheimers Dement. 19, 56–66 (2023).
Notice on the Issuance of the National Action Plan for Addressing Dementia in Older Adults (2024–2030) [https://www.gov.cn/zhengce/zhengceku/202501/content_6996231.htm]
Zhi, N. et al. The China Alzheimer Report 2025. Gen. Psychiatr. 38, e102020 (2025).
Notice on Issuing the Implementation Plan for Deepening the Development of Dementia-Friendly Communities (2025) [https://www.shanghai.gov.cn/gwk/search/content/ee48787ab70746659348ae16e8a217a2], 2030
Impairment CSoDaC Chinese expert consensus on the diagnosis and treatment of mild cognitive impairment due to Alzheimer′s disease 2024. Chin. J. Neurol. 57, 715–737 (2024).
Bian, D. et al. Determinants influencing the implementation of community health management for cognitive impairment: a qualitative study. BMC Geriatr. 25, 246 (2025).
Lu, Y., Liu, C., Fawkes, S., Wang, Z. & Yu, D. Knowledge, attitudes, and practice of general practitioners toward community detection and management of mild cognitive impairment: a cross-sectional study in Shanghai, China. BMC Prim. Care 23, 114 (2022).
Petersen, R. C. et al. Author response: practice guideline update summary: mild cognitive impairment: report of the guideline development, dissemination, and implementation subcommittee of the American Academy of Neurology. Neurology 91, 373–374 (2018).
Brookmeyer, R., Johnson, E., Ziegler-Graham, K. & Arrighi, H. M. Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement 3, 186–191 (2007).
Zhang, Y. et al. Identifying modifiable factors and their joint effect on dementia risk in the UK Biobank. Nat. Hum. Behav. 7, 1185–1195 (2023).
Li, W. et al. Cost-effectiveness of physical activity interventions for prevention and management of cognitive decline and dementia-a systematic review. Alzheimer's Res. Ther. 15, 159 (2023).
Siu, P. M. et al. Effects of Tai Chi or exercise on sleep in older adults with insomnia: a randomized clinical trial. JAMA Netw. Open 4, e2037199 (2021).
Chen, H. et al. Effectiveness of Tai Chi on cognitive function among older adults with mild cognitive impairment: a systematic review and meta-analysis of randomized controlled trials. Aging Ment. Health 28, 285–293 (2024).
Wang, W. T. & Wang, H. Effects of Tai Chi Chuan on cognitive function in adults 60 years or older with mild cognitive impairment: a systematic review and meta-analysis. Front Physiol. 16, 1556622 (2025).
Rosenberg, A., Mangialasche, F., Ngandu, T., Solomon, A. & Kivipelto, M. Multidomain interventions to prevent cognitive impairment, Alzheimer’s Disease, and dementia: from finger to world-wide fingers. J. Prev. Alzheimers Dis. 7, 29–36 (2020).
Xue, D., Li, P. W. C., Yu, D. S. F. & Lin, R. S. Y. Combined exercise and cognitive interventions for adults with mild cognitive impairment and dementia: a systematic review and network meta-analysis. Int J. Nurs. Stud. 147, 104592 (2023).
Gavelin, H. M. et al. Combined physical and cognitive training for older adults with and without cognitive impairment: a systematic review and network meta-analysis of randomized controlled trials. Ageing Res Rev. 66, 101232 (2021).
Huang, X. et al. Comparative efficacy of various exercise interventions on cognitive function in patients with mild cognitive impairment or dementia: a systematic review and network meta-analysis. J. Sport Health Sci. 11, 212–223 (2022).
Reparaz-Escudero, I., Izquierdo, M., Bischoff-Ferrari, H. A., Martinez-Lage, P. & Saez de Asteasu, M. L. Effect of long-term physical exercise and multidomain interventions on cognitive function and the risk of mild cognitive impairment and dementia in older adults: a systematic review with meta-analysis. Ageing Res. Rev. 100, 102463 (2024).
Grol, R. Successes and failures in the implementation of evidence-based guidelines for clinical practice. Med. Care 39, II46–II54 (2001).
Rubin, R. It takes an average of 17 years for evidence to change practice-the burgeoning field of implementation science seeks to speed things up. JAMA 329, 1333–1336 (2023).
Neta, G. et al. Advancing climate change health adaptation through implementation science. Lancet Planet Health 6, e909–e918 (2022).
Chays-Amania, A., Schwingrouber, J. & Colson, S. Using implementation science to implement evidence-based practice: a discursive paper. J. Adv. Nurs. 81, 8050–8061 (2024).
Holt-Lunstad, J. Why social relationships are important for physical health: a systems approach to understanding and modifying risk and protection. Annu. Rev. Psychol. 69, 437–458 (2018).
Damschroder, L. J. et al. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 4, 50 (2009).
Damschroder, L. J., Reardon, C. M., Widerquist, M. A. O. & Lowery, J. The updated consolidated framework for implementation research based on user feedback. Implement Sci. 17, 75 (2022).
Glasgow, R. E., Vogt, T. M. & Boles, S. M. Evaluating the public health impact of health promotion interventions: the RE-AIM framework. Am. J. Public Health 89, 1322–1327 (1999).
Duan, Y. et al. Conceptual and relational advances of the PARIHS and i-PARIHS frameworks over the last decade: a critical interpretive synthesis. Implement Sci. 17, 78 (2022).
Nissen, S., Karlsson, A. W. & Norgaard, B. Evaluation tools for patient and public involvement (PPI) in health research: a scoping review. Patient (Springer, 2025).
Ye, Y. et al. Efficacy of technology-based cognitive and exercise interventions for mild cognitive impairment: a systematic review, network meta-analysis, and meta-regression of randomized controlled trials. Ageing Res. Rev. 100, 102438 (2024).
Ding, Z., Leung, P. Y., Lee, T. L. & Chan, A. S. Effectiveness of lifestyle medicine on cognitive functions in mild cognitive impairments and dementia: a systematic review on randomized controlled trials. Ageing Res. Rev. 86, 101886 (2023).
Kidwell, K. M. & Almirall, D. Sequential, multiple assignment, randomized trial designs. JAMA 329, 336–337 (2023).
Brauner J. M., et al. Inferring the effectiveness of government interventions against COVID-19. Science 371, 9338 (2021).
Li, F., Harmer, P., Eckstrom, E., Fitzgerald, K. & Winters-Stone, K. Clinical effectiveness of cognitively enhanced tai ji quan training on global cognition and dual-task performance during walking in older adults with mild cognitive impairment or self-reported memory concerns : a randomized controlled trial. Ann. Intern. Med. 176, 1498–1507 (2023).
Ali, N. et al. The effects of dual-task training on cognitive and physical functions in older adults with cognitive impairment; a systematic review and meta-analysis. J. Prev. Alzheimers Dis. 9, 359–370 (2022).
Sungkarat, S., Boripuntakul, S., Kumfu, S., Lord, S. R. & Chattipakorn, N. Tai Chi improves cognition and plasma BDNF in older adults with mild cognitive impairment: a randomized controlled trial. Neurorehabil. Neural Repair 32, 142–149 (2018).
Tao, J. et al. Increased hippocampus-medial prefrontal cortex resting-state functional connectivity and memory function after TAI CHI CHUAN practice in elder adults. Front. Aging Neurosci. 8, 25 (2016).
Li, L. et al. Tai Chi exercise improves age-associated decline in cerebrovascular function: a cross-sectional study. BMC Geriatr. 21, 293 (2021).
Herold, F., Hamacher, D., Schega, L. & Muller, N. G. Thinking while moving or moving while thinking - concepts of motor-cognitive training for cognitive performance enhancement. Front. Aging Neurosci. 10, 228 (2018).
Nilsson, J. et al. Acute increases in brain-derived neurotrophic factor in plasma following physical exercise relates to subsequent learning in older adults. Sci. Rep. 10, 4395 (2020).
Fissler, P., Kuster, O., Schlee, W. & Kolassa, I. T. Novelty interventions to enhance broad cognitive abilities and prevent dementia: synergistic approaches for the facilitation of positive plastic change. Prog. Brain Res. 207, 403–434 (2013).
Liao, Y. Y., Tseng, H. Y., Lin, Y. J., Wang, C. J. & Hsu, W. C. Using virtual reality-based training to improve cognitive function, instrumental activities of daily living and neural efficiency in older adults with mild cognitive impairment. Eur. J. Phys. Rehabil. Med. 56, 47–57 (2020).
Golden, S. D. & Earp, J. A. Social ecological approaches to individuals and their contexts: twenty years of health education & behavior health promotion interventions. Health Educ. Behav. 39, 364–372 (2012).
Stiggelbout, M., Hopman-Rock, M., Tak, E., Lechner, L. & van Mechelen, W. Dropout from exercise programs for seniors: a prospective cohort study. J. Aging Phys. Act. 13, 406–421 (2005).
ZHEN et al. Research progress on influencing factors of cognitive management compliance among people with mild cognitive impairment in the community. Chin. Gen. Pract. 23, 1322–1326 (2020).
Loban, E., Scott, C., Lewis, V. & Haggerty, J. Measuring partnership synergy and functioning: multi-stakeholder collaboration in primary health care. PLoS One 16, e0252299 (2021).
Hoekstra, F. et al. Panel SCIGPC, Gainforth HL: a review of reviews on principles, strategies, outcomes and impacts of research partnerships approaches: a first step in synthesising the research partnership literature. Health Res. Policy Syst. 18, 51 (2020).
McLeroy, K. R., Bibeau, D., Steckler, A. & Glanz, K. An ecological perspective on health promotion programs. Health Educ. Q 15, 351–377 (1988).
Weiner, B. J. A theory of organizational readiness for change. Implement Sci. 4, 67 (2009).
Julian McFarlane, S., Occa, A., Peng, W., Awonuga, O. & Morgan, S. E. Community-based participatory research (CBPR) to enhance participation of racial/ethnic minorities in clinical trials: a 10-year systematic review. Health Commun. 37, 1075–1092 (2022).
Bian, D. et al. Relationship between social support, sarcopenia, and cognitive impairment in chinese community-dwelling older adults. J. Nutr. Health Aging 27, 726–733 (2023).
Chinese Dementia and Cognitive Impairment Diagnosis and Treatment Guideline Writing Group Cognitive Disorder Diseases Professional Committee of the Neurology Branch of Chinese Medical Doctor Association: Chinese guidelines for the diagnosis and treatment of dementia and cognitive impairment (V): Diagnosis and treatment of mild cognitive impairment. Natl. Med. J. China 98, 1294–1301 (2018).
Geriatric Nursing, C. hina & XSoN, A. lliance Central South University: clinical practice guideline for non-pharmacological interventions in older adults with cognitive decline: physical activity. Chin. Gen. Pract. 2023 26, 1927–1937 (1971).
Sherman, D. S., Mauser, J., Nuno, M. & Sherzai, D. The Efficacy of cognitive intervention in mild cognitive impairment (MCI): a meta-analysis of outcomes on neuropsychological measures. Neuropsychol. Rev. 27, 440–484 (2017).
Karssemeijer, E. G. A. et al. Positive effects of combined cognitive and physical exercise training on cognitive function in older adults with mild cognitive impairment or dementia: a meta-analysis. Ageing Res Rev. 40, 75–83 (2017).
Yan, X., Ghosh, P. & Chakraborty, B. Sample size calculation based on precision for pilot sequential multiple assignment randomized trial (SMART). Biom. J. 63, 247–271 (2021).
Jia, X. et al. A comparison of the Mini-Mental State Examination (MMSE) with the Montreal Cognitive Assessment (MoCA) for mild cognitive impairment screening in Chinese middle-aged and older population: a cross-sectional study. BMC Psychiatry 21, 485 (2021).
Chen, K. L. et al. Validation of the Chinese version of Montreal cognitive assessment basic for screening mild cognitive impairment. J. Am. Geriatr. Soc. 64, e285–e290 (2016).
Xiao, Y. et al. Development and validity of computerized neuropsychological assessment devices for screening mild cognitive impairment: ensemble of models with feature space heterogeneity and retrieval practice effect. J. Biomed. Inf. 131, 104108 (2022).
Park, S. H. & Kwak, M. J. Performance of the geriatric depression scale-15 with older adults aged over 65 years: an updated review 2000-2019. Clin. Gerontol. 44, 83–96 (2021).
Luo, N. et al. Estimating an EQ-5D-5L value set for China. Value Health 20, 662–669 (2017).
Lu, X. et al. Comparing dynamic treatment regimes using repeated-measures outcomes: modeling considerations in SMART studies. Stat. Med. 35, 1595–1615 (2016).
Šidák, Z. Rectangular confidence regions for the means of multivariate normal distributions. J. Am. Stat. Assoc. 62, 626–633 (1967).
Acknowledgements
The authors would like to thank the community health service centers, the neighborhood committees, and all the healthcare professionals, Tai Chi instructors, and community volunteers who contributed to the design, implementation, and monitoring of the intervention. We are also grateful to the older adult participants and their families for their time and engagement throughout the study. Special thanks to the VR technology support team for providing training and technical assistance. We acknowledge Shanghai Thoven Intelligent Technology Company Ltd. (Shanghai, China) for the provision of the cognitive training management system used in this research. We also thank the Tai Chi Association of Pudong New District, Shanghai, for their support in designing and delivering the Tai Chi training sessions. This research was suppported by the National Natural Science Foudation of China (82304114); Yunnan Key Research Program (202402AD080004). The funders played no role in the design, analysis, and writing of the study.
Author information
Authors and Affiliations
Contributions
GL and CS led the project as the principal investigator. DB executed the project. XL, WG, PL, FF, and LW coordinated participant recruitment and intervention delivery. DB, JZ, CY and XH conducted data management and analysis. DB, CS and GL led quality control and monitoring throughout the study. DB and JZ drafted the initial manuscript. All authors agree to be personally accountable for their own contributions and to the accuracy and integrity of all parts of the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bian, D., Zhang, J., Yu, C. et al. Adaptive community care for mild cognitive impairment: a SMART randomized trial with cognitive and implementation outcomes. npj Dement. 2, 7 (2026). https://doi.org/10.1038/s44400-025-00052-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44400-025-00052-w