Abstract
The emergence of wearable devices capable of continuous biomarker monitoring has significantly transformed the traditional landscape of healthcare, offering real-time insights into physiological changes, enabling early detection of disease, and facilitating individualized therapeutic. Nucleic acid aptamers, as a highly sensitive biorecognition elements with programmable properties, provide an innovative solution for building a new generation of continuous monitoring system. This review presents recent developments in aptamers-based wearable electrochemical sensors for continuous monitoring of biomarkers. Firstly, the core advantages of aptamers in continuous monitoring are summarized, including unique reversible binding properties, excellent biostability and engineerable modification ability, which make them ideal recognition elements for building real-time biosensing interfaces. Secondly, this review provides insights into the design principles of aptamers-based electrochemical sensors, including the characterization of aptamers, the immobilization methods on the electrode surface and the sensing strategies of sensors. Besides, this review highlights recent research advances in the field, focusing on the design of different types of sensors as well as their applications. Specifically, according to the difference target acquisition methods, the type of sensors is classified into non-invasive sensors, which involve monitoring biomarkers present in accessible biofluids such as sweat, saliva and wound exudate, and low-invasive sensors utilizing microneedle patch technology to sample biomarkers within interstitial fluids. Finally, this review introduces the challenges and discuss potential solutions for future development of aptamers-based wearable biosensors, including how to achieve effective biomarkers detection, ensure reversible binding response and ensure long-term stability, etc. This review provides useful reference for the development of aptamers-based electrochemical continuous monitoring technology in healthcare applications.

Similar content being viewed by others
Introduction
Biomarkers are biochemical indicators that reflect changes in the structure or function of biological systems at the level of organs, tissues, cells, and subcellular components. These indicators play a crucial role in disease diagnosis, disease staging, and the evaluation of the safety and efficacy of novel drugs or therapeutic interventions1. Biomarkers encompass a diverse range of molecular forms, broadly categorized by their chemical nature into proteins, peptides, nucleic acids, and small-molecule metabolites. For example, inflammatory factors serve as biomarkers for reflect the inflammatory state in the body and evaluate disease progression2. β-amyloid is closely implicated in the pathogenesis of Alzheimer’s disease3,4. Circulating tumor DNA has been applied in liquid biopsies and the monitoring of tumor treatment efficacy5, and creatinine is routinely used to assess renal functions6. Currently, biomarker detection in clinical practice primarily relies on venous blood sampling, coupled with various in vitro assays, such as immunological assays, liquid chromatography, mass spectrometry, and PCR detecting system7,8. These methods have the advantages of good accuracy and high sensitivity, but the in vitro testing paradigm also faces some challenges, including the requirement for complex invasive sampling procedures, time-consuming workflows, and difficulties in achieving real-time analysis.
Compared to traditional clinical single-point measurements, continuous biomarkers monitoring enables the capture of subtle and transient fluctuations of physiological indicators, which can provide a real-time assessment of disease state and progression and offer a more timely and comprehensive reference for clinical decision-making1,9,10. For example, for individuals with diabetes mellitus, the amplitude and frequency of insulin level fluctuations are abnormally elevated, exhibiting a complex and dynamic relationship with blood glucose levels. Continuous monitoring of insulin is therefore crucial to prevent transient hyperglycemia or hypoglycemia from masking underlying insulin resistance or secretory dysfunctions11. Current methods for continuous biomarker monitoring primarily utilize sensors functionalized with antibodies or enzymes. Antibody-based sensors demonstrate excellent specificity and sensitivity12,13. However, as protein molecules, antibodies exhibit inherent limitations in stability and susceptibility to denaturation in vivo. Furthermore, the antigen-antibody binding process is often irreversible, which greatly limits the regeneration of the sensor and affects the continuity of the sensing process. Enzyme-modified sensors rely on the catalytic activity of enzymes for target detection, offering a partial solution to the limitations of antibody-based sensors in continuous monitoring14,15. However, the high specificity in enzyme and substrate interactions restricts the broader applicability of these sensors to a wider range of analytes. Therefore, the development of a stable, widely applicable, and reproducible sensor for continuous in situ monitoring of biomarkers is critical16,17.
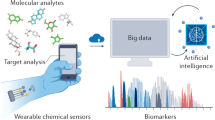
Aptamers mainly refer to nucleic acid aptamers, which are single-stranded oligonucleotide molecules selected through the systematic evolution of ligands by exponential enrichment (SELEX) process, which have the advantages such as facile chemical synthesis, controllable three-dimensional conformation, excellent thermal stability, high target affinity, and reversible binding kinetics18,19. Among them, aptamers are widely used as biorecognition elements in sensors by taking advantage of their high specificity for targets, and achieve efficient target binding to a diverse range of analytes, including ions, small molecules, hormones, proteins, and even cells20,21,22. The binding process primarily through non-covalent interactions such as hydrogen bonding, electrostatic forces, hydrophobic interactions, and π-π stacking23,24. In analytical applications, aptamer-based sensors can avoid the need for complex sample pretreatment steps associated with traditional analytical methods, enabling direct analysis of target analytes in complex biological matrices like serum, whole blood, sweat, and interstitial fluid (ISF)25,26,27. This significantly reduces the analysis time and operational complexity28. Furthermore, the dynamic and reversible binding characteristics between aptamers and their targets enable sensor regeneration, which is essential to construct continuous monitoring platforms with high temporal resolution29,30,31. Consequently, aptamer-based sensors are capable of tracking real-time fluctuations in biomarker concentrations, providing more accurate and comprehensive information for early disease warning, treatment efficacy assessment, and personalized health management (Fig. 1)32,33,34,35.
Overview of aptamers-based wearable electrochemical sensors for continuous monitoring applications, including drug metabolism monitoring, wound management, disease management and prevention, and health monitoring
At the same time, achieving long-term, stable, and accurate in vivo continuous monitoring of biomarkers using aptamer-based sensors presents some challenges17,36. Firstly, it remains a key challenge to achieve efficient capture of biomarkers in ISF or blood in a minimally or even non-invasive manner, especially for substances with low concentration levels. Secondly, while the binding of aptamers to their targets is theoretically reversible, achieving signal reversibility and complete recovery to baseline levels faces significant technical bottlenecks in practical applications, which restricts the reusability of the aptamer-based sensors and their long-term monitoring performance. Furthermore, the binding process between aptamers and targets requires a certain amount of time, and the binding rate directly impacts the sensor’s responsiveness, particularly in continuous monitoring situations that require rapid response times. In addition to the previously mentioned issues, the practical application of in vivo sensors is limited by other factors, including long-term stability, biocompatibility, and signal drift35,37,38. Therefore, improving efficient target capture based on minimally or noninvasive techniques, exploring sensor regeneration based on reversible binding between the aptamers and the targets, optimizing the response speed of the sensor, and enhancing the long-term stability and biocompatibility are essential for advancing the utility and the reliability of aptamer-based continuous monitoring sensors.
This review discusses recent advances in aptamer-based electrochemical sensors for continuous monitoring of biomarkers. Firstly, the core advantages of aptamers for continuous monitoring are summarized, highlighting their unique reversible binding properties, excellent biostability, and versatile engineering capabilities, which make them ideal recognition elements for constructing real-time biosensing interfaces. Next, the design principles in aptamer-based electrochemical sensors are discussed, including aptamer immobilization strategies and diverse sensing methodologies. We then present recent research advances in aptamer-based wearable electrochemical sensors, discussing continuous monitoring strategies categorized by biomarker acquisition method. These include non-invasive approaches, which emphasize in situ monitoring of biomarkers in easily accessible biofluids such as sweat, saliva, and wound exudate, and low-invasive methods, which commonly relies on microneedle patch systems for efficient sampling of ISF. Furthermore, the challenges and opportunities of aptamer-based electrochemical sensors are discussed, and solutions for designing next-generation sensors are explored, illustrating their future development potential in the field of medical engineering. In summary, this review aims to provide valuable insights and inspirations for continuous monitoring studies of biomarkers, thereby promoting the application of aptamer-based wearable electrochemical sensors in the biomedical field.
Screening and characteristics of aptamers
As important biorecognition elements, both aptamers and antibodies exhibit high affinity for their targets. However, antibodies, being immunoglobulins, possess certain limitations in practical applications. First, antibodies are usually obtained relies on animal immune systems, which are complex, costly, and only applies to substances that are immunogenic. Furthermore, inherent batch-to-batch variability in antibodies production can compromise reproducibility, and the proteinaceous nature of antibodies presents challenges for controlled chemical modification. Their three-dimensional structure of the antibodies is also susceptible to denaturation by environmental factors, which affects their stability and results in the loss of specificity and affinity for the target antigen39. Compared to antibodies, aptamers well overcome the limitations mentioned above and show greater advantages in the sensing process. As aptamers are selected in vitro under controlled conditions, their target recognition is unconstrained by immunogenicity, thus broadening their applicability. Moreover, in vitro selection ensures minimal batch-to-batch variation and high product uniformity. Finally, aptamers can be readily modified or conjugated with functional groups, facilitating the development of diverse detection strategies19,40.
Screening of aptamers
Aptamers are functional oligonucleotide molecules selected through the Systematic Evolution of Ligands by Exponential enrichment (SELEX) technique, which is an iterative selection method designed to identify nucleic acid sequences exhibiting high affinity for a specific target molecule. The process begins by constructing a diverse library of random nucleic acid molecules, followed by iterative cycles of incubation with the target, separation of binding complexes, PCR amplification, and conversion to single-stranded nucleic acids, and progressively enriching aptamers exhibiting high affinity for the target. (Fig. 2a)41,42. A key characteristic of SELEX is in vitro operational mode, which avoids the limitations imposed by individual variations in animal immune systems and allows the selection of aptamers against molecules that are difficult to target with traditional antibodies, such as toxins. Concurrently, the highly tunable selection conditions enable researchers to optimize key performance indicators of the aptamers according to specific application requirements, such as binding kinetics and conformational transition thresholds.
Schematic illustration of aptamer selection processes. a Systematic Evolution of Ligands by Exponential Enrichment (SELEX). b A microfluidic SELEX platform for efficient selection. Reproduced with permission from Elsevier (2025)43. c A high-dimensional microfluidic approach for selecting aptamers with controllable binding affinity. Reproduced with permission from Springer Nature (2023)46. d A precise and systematic aptamers selection precision-SELEX strategy based on magnetic bead separation technology. Reproduced with permission from Elsevier (2024)47. e A method for dynamically selecting high-affinity aptamers using a magnetic-activated continuous deflection microfluidic chip. Reproduced with permission from The Royal Society of Chemistry (2024)45. f A one-step aptamers enrichment platform based on a three-dimensional, contamination-free porous hydrogel. Reproduced with permission from Springer Nature (2023)48
Furthermore, the modular nature of the SELEX process facilitates its integration with automated platforms. For instance, microfluidic chip technology and magnetic beads separation techniques are often used to integrate with SELEX for automated and high-throughput screening43,44,45. Optimized designs of microfluidic chips often aim to prevent cross-contamination between reaction zones by creating separate inlets and reaction chambers. In SELEX processes, the common strategies are usually to immobilize the target in a specific region of the microfluidic chip and precisely controlling fluid flow to coordinate the entire selection process (Fig. 2b, c)44. Similarly, magnetic beads separation techniques utilize magnetic beads as a solid carrier for target immobilization and leverage magnetic forces to efficiently perform separation steps during the selection process (Fig. 2d, e)45. The combination of SELEX processes with an automated platform can significantly reduce screening time, improve screening efficiency, and enable real-time monitoring of the selection efficiency for each round. Following over three decades of technological advancement, SELEX has matured into a well-established technology and a standardized method for aptamer generation. By combining SELEX with more emerging technologies, it is expected that more accurate and efficient aptamer screening platforms will be developed46,47,48.
Specificity of aptamers
As crucial biorecognition elements, the main characteristics of aptamers are their high specificity, which primarily from the precise matching of their structure to the surface of the targets. The SELEX in vitro selection process is a directed evolution process. Through multiple rounds of selection and enrichment, sequences with weaker binding affinity to the target are progressively eliminated, resulting in aptamer sequences that are highly complementary to the target49. Besides, upon aptamer-target binding, the target often induces a conformational change in the aptamer and further enhances the binding affinity, allowing the aptamer structure to better adapt to the target50. Moreover, the high affinity of aptamers also originates from their complex and diverse three-dimensional conformations. Unlike linear nucleic acid chains, aptamers can self-assemble through non-covalent interactions to form a variety of complex spatial structures, such as stem-loop, hairpin, pseudoknots and G-quadruplex, which provide versatile binding modes for target recognition51,52. The diversity and dynamic nature of aptamers three-dimensional structures are fundamental to their high-affinity binding. Finally, the interaction between aptamer and target is not a singular force but involves a synergistic effect of various non-covalent interactions, including hydrogen bonds, electrostatic interactions, hydrophobic interactions and van der Waals forces, creating a robust binding interface, enhancing the stability and affinity and enabling the aptamer to efficiently recognize and bind to the target24.
For instance, using nuclear magnetic resonance methods, Xu et al. presented the first high-resolution nuclear magnetic resonance structure of the complex between aflatoxin B1 (AFB1) and its 26-mer DNA aptamer (AF26). AFB1 binds to the 16-residue loop region of the aptamer, inducing it to fold into a compact structure through the assembly of two bulges and a hairpin structure. AFB1 is tightly enclosed within the cavity formed by the bulges and hairpin, fixed between G·C base pair, G·G·C triple and multiple T bases through strong π-π stacking, hydrophobic interactions and donor atom-π interactions, respectively53. Subsequently, the same team elucidated the high-resolution structure of the aptamer-ochratoxin analogues A (OTA) complex in solution, finding that OTA can induce the aptamer to fold into a duplex-quadruplex structural scaffold stabilized by Mg2+ and Na+ ions. Unlike the commonly found model of small molecule ligands binding to G-quadruplexes through direct stacking with the G-tetrad plane, OTA does not directly interact with the G-quadruplex but binds to the junction between the double helix and G-quadruplex structure through π-π stacking, halogen bonding and hydrophobic interactions54. Therefore, based on the coordinated action of the above factors, aptamers exhibit excellent specificity, which is the key to the application in the field of biosensing and ensures the accuracy of detection and the reliability of research.
Modifiability of aptamers
Aptamers are easy to modify, which is one of the other outstanding characteristics of aptamers over antibodies and other biological recognition elements. This property stems from the established principles of nucleic acid chemistry and the efficient synthetic accessibility of oligonucleotides55,56. The ease of chemical modification allows for precise control over aptamer function, expanding their utility in biosensing applications. Current research in aptamer modification focuses on conjugating functional groups or nanomaterials to aptamers, predominantly through terminal modifications. This strategy achieves two key objectives of the sensor design, on the one hand, efficient immobilization of the aptamer on the electrode surface and on the other hand, amplification of the detected signal during the detection process. The realization of these two key steps has driven the increasing use of aptamers in the field of biosensors and is expected to lead to the development of sensors with better detection performance.
Immobilization of aptamers on electrode surface
Effective immobilization of the aptamers on the electrode surface is the basis for signal sensing. In electrochemical sensors, depending on the different functional groups used for terminal modification of the aptamers, the immobilization methods can be categorized into physical adsorption method, gold-sulfur self-assembled method, covalent bond method and the biotin-streptavidin affinity method (Fig. 3a)57. Physical adsorption is a simple and convenient method, as it does not require any modification of the aptamers. Furthermore, due to the negative charge carried by the phosphate skeleton, aptamers can be immobilized through electrostatic force. For example, positively charged chitosan can be used to pre-modify the electrode surface, and the negatively charged aptamer interacts with the positively charged chitosan, resulting in immobilization on the electrode surface. Based on this principle, Salma et al. developed a platform for nucleic acid detection using chitosan and chitosan coated gold nanoparticles (AuNPs). Due to the overall zeta potential of chitosan, it possesses the ability to bind DNA. In negative samples, free chitosan conjugated with non-target DNA prevents AuNPs-DNA interactions, whereas, in positive samples, the amplified DNA saturates the free chitosan, leading to AuNPs aggregation58. The advantage of physical adsorption method is simple operation. However, the binding between the aptamers and the electrode surface is weak, which is prone to release and the orientation of aptamers fixed is irregular, affecting the recognition between the aptamer and the target, and resulting in low sensitivity. With the emergence of other alternative aptamers immobilization methods, the focus of research has gradually shifted away from physical adsorption.
Aptamers immobilization strategies on electrode surfaces. a General principles of aptamers immobilization on electrode surfaces. b Aptamers immobilization on a gold electrode surface via Au-S bonds. Reproduced with permission from Elsevier (2024)98. c Aptamers immobilization on gold nanoparticle-modified electrode surface via Au-S bonds. Reproduced with permission from Elsevier (2025)63. d Aptamers immobilization on electrode surface using the biotin-streptavidin system. Reproduced with permission from American Chemical Society (2010)67
Self-assembly of thiol-modified aptamers on gold surfaces via Au-S bonds to form ordered aptamer monolayers is a commonly employed strategy for constructing aptamer-sensing interfaces. Wu et al. used magnetron sputtering to prepare a uniform gold layer on a microneedle array, using a self-assembly strategy, successfully immobilized aptamers targeting antibiotics onto the microneedle electrode surface59. Gao et al. also employed the gold-sulfur self-assembled method to successfully immobilize aptamers for IL-6, IL-8, TGF-β1, and Staphylococcus aureus on gold electrode surfaces, enabling the simultaneous detection of these biomarkers (Fig. 3b)60. The Au-S bond self-assembly method is operationally simple and does not require chemical modification of the electrode surface. Furthermore, while this method is not only suitable for aptamer modification on gold electrode surfaces, thiol-modified aptamers can also be readily immobilized on the surfaces of gold nanomaterials such as AuNPs and gold nanorods (Fig. 3c). To further enhance sensing performance, Cui et al. modified the working electrode with AuNPs and MXene. In this sensor, capture single-stranded DNA (ssDNA) was modified with a thiol group at its terminus. The ssDNA was immobilized on the working electrode surface through the interaction between the thiol group and the AuNPs. This study showed that, compared to a conventional gold electrode, large specific surface area of AuNPs increased the effective electrode area, thereby enhancing the aptamers' loading capacity. The ultra-high conductivity of MXene helps to increase the charge transfer rate. The combination of both effectively improves the detection sensitivity61. Similarly, Lin et al. modified microneedle electrodes with AuNPs for efficient aptamer immobilization and signal transduction, achieving aptamer-based drug sensing. This coating minimizes substrate impurities and provides a high-quality surface conducive to strong aptamer binding. Furthermore, the nanostructured morphology of the AuNPs effectively increases the surface area available for aptamer immobilization, thereby enhancing the signal-to-noise ratio of the measurements. On this basis, based on thiol-modified aptamers, we can achieve efficient immobilization of aptamers and effective drug sensing62,63. It is important to note that, due to the relatively large spacing between sulfur atoms in the aptamer monolayer formed by Au-S bond self-assembly, it is difficult to form a tightly packed modification layer, which can lead to a certain proportion of non-specific adsorption. Therefore, it is necessary to block the remaining vacant sites on the electrode surface after aptamer assembly.
The covalent bond method involves the interaction of aptamers with a terminal modified chemical group (most commonly amino group) with the corresponding chemical group (hydroxyl, carboxyl, amino, etc.) on the surface of the electrode surface and the formation of an ordered film, which is driven by the covalent binding force of the chemical bond. In contrast to the gold-sulfur self-assembled method, this process typically requires pretreatment of the electrode to introduce the necessary activating groups64. For example, Muamer et al. modified the terminus of an insulin-specific aptamer with an amino group and activated the carboxyl functional groups on the electrode surface using N-ethyl-N’-(3-dimethylaminopropyl) carbodiimide/N-hydroxysuccinimide (EDC/NHS). By relying on the covalent bond formation between the amino and carboxyl groups, they immobilized the aptamers on the electrode surface65. This covalent bond method optimizes the distribution and orientation of aptamers on the electrode surface and reduces non-specific adsorption. However, this process requires chemical modification of both the aptamer and the electrode surface, which makes the steps relatively elaborate, and the potential side effects of chemical reagents added during the coupling process should also be considered66.
The biotin-streptavidin affinity method uses the strong interaction between biotin and streptavidin to immobilize aptamers on electrode surfaces. This system, representing one of the highest affinity interactions known in nature, allows for efficient aptamer immobilization by modifying the aptamers with biotin and immobilizing streptavidin on the electrode surface. For example, Liu et al. reported a stable biosensor based on streptavidin-biotin interaction. Amino-modified biotin was immobilized on carboxyl-modified single-walled carbon nanotubes using EDC/NHS coupling. Subsequently, aptamers modified with streptavidin were bound to the nanotubes via streptavidin-biotin interactions. While this method employs carbodiimide chemistry for stable covalent streptavidin immobilization, this additional step increases the complexity of the fabrication process and may not be strictly necessary (Fig. 3d)67. While the strong biotin-streptavidin force facilitates efficient aptamer immobilization, this method necessitates simultaneous modification of both the aptamers and the electrode surface, which may increase experimental costs and operational complexity.
Due to the facile modification of aptamers, their immobilization on electrode surfaces is now a relatively well-established technique. In practical applications, the optimal immobilization method should be selected with careful consideration of the detection requirements and the properties of the electrode material to ensure efficient acquisition of electrochemical signals. Furthermore, to minimize the influence of interfering substances present in biological matrices and to enhance sensor specificity, effective blocking of the electrode surface is crucial. Commonly used blocking reagents include 6-hydroxy-1-hexanethiol, polyethylene glycol, and bovine serum albumin etc. A rational selection of blocking reagents should be made based on the specific application situation, the physicochemical properties of the target biomarker, and the aptamer immobilization strategy, to achieve optimal blocking performance.
Amplification of detection signal
Beyond facilitating efficient aptamer immobilization, another significant application of modified aptamers lies in meeting practical detection requirements and enhancing detection sensitivity. In electrochemical sensors, aptamers can be precisely conjugated with redox-active molecules, nanoparticles, and enzymes to create highly sensitive, selective, and stable detection platforms68. Among them, redox-active molecules are most widely used in electrochemical detection, and their reversible electron transfer characteristics can be directly converted into electrical signals to realize precise identification and quantitative analysis of targets. Methylene blue (MB), one of the representative redox-active molecules, has advantages in electrochemical performance, structural stability, and compatibility with aptamers, which makes it an ideal choice for constructing various aptamer-based wearable electrochemical sensors. For example, Lin et al. modified the terminus of an aptamer with MB as a signal reporter molecule for continuous sensing of drug binding62. Similarly, Gao et al. modified a synthesized aptamer sequence with a thiol group at one end for covalent binding to AuNPs and with MB as a redox probe at the other end to achieve high-sensitivity sensing60. Yang et al. rationally designed aptamer sequences to undergo desired binding-induced conformational changes and modified them with MB for measuring malaria biomarker lactate dehydrogenase69.
In addition to MB, ferrocene molecule (Fc) and quinone compounds are also commonly used redox-active molecules that play important roles in electrochemical sensing. Zargartalebi et al. reported a sensor for continuous protein monitoring. This sensor employed a rigid, electrode-bound double-stranded DNA scaffold on the electrode surface. One strand of the scaffold was modified with a protein-specific aptamer sequence, while the other strand was modified with Fc as an electron donor to enable protein monitoring70. Jiang et al. developed an Fc-DNA2 complex as an electrochemical signal-responsive probe. The competitive binding between alternariol (AOH) and Fc-DNA2 to the AOH aptamer results in the change of electrochemical signal, which realizes the highly sensitive detection of AOH71. Liu et al. simultaneously modified a complementary DNA (cDNA) probe for acetamide (ACE) with both Fc and MB. The synergistic effect of the two redox-active molecules effectively reduced systematic errors and background noise, enhancing the reliability of the detection72. Modification of aptamers with redox-active molecules significantly enhance the signal transduction ability and achieves label-free and highly sensitive target detection through the synergistic effect of molecular recognition and electrochemical response, which provides a new idea for the construction of high-efficiency biosensors.
In recent years, the electrochemical activity of some metal nanoparticles and metal oxides has been explored and developed73,74. However, most metal nanoparticles are unstable and easily oxidized by atmospheric oxygen or decomposed by light, while metal oxides often have large particle sizes and poor biocompatibility. These drawbacks significantly limit the application of such electroactive materials. Research has found that embedding biomaterials within metal nanoparticles can effectively improve their physicochemical properties, such as easy oxidation, easy decomposition, and poor solubility, while preserving the electrochemical activity of the core material. For example, Huang et al. synthesized two types of core-shell nanomaterials, Ag-Au core-shell nanoparticles (Ag@AuNPs) and Cu2O-Au core-shell nanoparticles (Cu2O@AuNPs). Based on these two materials, two novel electroactive signal probes were prepared, which were modified with acetamiprid and malathion aptamers, respectively, while the electrochemical signals of acetamiprid and malathion were detected simultaneously75. The high conductivity, catalytic activity, and large specific surface area of nanomaterials show significant advantages in electrochemical sensor design. Moreover, with the continuous optimization of material preparation technology, nanomaterials can realize controllable synthesis and property modulation, which enhances the biorecognition efficiency of the sensing interface and provides a new way to construct highly sensitive and stable sensor devices.
Furthermore, aptamers can also be conjugated with biomacromolecules such as enzymes, for example, horseradish peroxidase and alkaline phosphatase76. These aptamers-enzyme complexes are more frequently used in electrochemical immunoassays. By combining the high target specificity of aptamers with the efficient catalytic properties of enzymes, the aptamer-target binding event is transformed into an amplified electrochemical signal through enzymatic reactions, enabling highly sensitive detection of the target analyte. Compared with the traditional enzyme-linked immunosorbent assay (ELISA), enzyme-based aptamer methods have made promising development, as they achieve precise capture of the target through the high affinity of the aptamers, and utilize the enzyme-catalyzed signal amplification strategy to enhance the detection sensitivity. However, such methods still rely on a multi-step enzymatic reaction system and require precise control of parameters such as reaction temperature, pH, and incubation time, resulting in high operational complexity and limited dynamic response range, which are difficult to meet the demands of in situ continuous monitoring in electrochemical sensing systems.
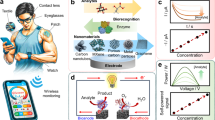
Electrochemical sensing strategy based on aptamers
Aptamers as recognition elements of biosensors, their specific binding to target is the foundation for constructing highly selective biosensing platforms. However, the aptamer-target binding event is often challenging to directly detect and quantify. Consequently, an efficient signal transduction strategy is crucial for converting this binding event into a readily detectable and quantifiable signal. In aptamers-based wearable electrochemical sensors, diverse signal transduction mechanisms are utilized to achieve sensitive and rapid detection of target analytes. Among these, signal transduction strategies based on changes in electrode surface impedance and the use of electrochemically active substances are the two most widely adopted and extensively investigated. The subsequent sections will elaborate on the principles, characteristics, advantages, and limitations of these two signal transduction strategies and analyze their application in the biomedical field.
Signal transduction based on changes in electrode surface impedance
In aptamers-based wearable electrochemical sensors, a significant signal transduction strategy relies on changes in electrode surface impedance (Fig. 4a). The core of this method lies in the formation of specific electrochemical interface characteristics when aptamers are immobilized on the electrode surface. Specifically, aptamer-target complex formation can modify the thickness or density of the aptamer layer or the adsorbed target layer, which influences the diffusion of ions and electrons to the electrode surface, thereby affecting the charge transfer process and electrochemical reaction resistance77. Several recent studies have validated the effectiveness of this strategy. Masoumeh et al. immobilized Staphylococcus aureus aptamers on a nanocomposite-modified screen-printed electrode and used differential pulse voltammetry to quantitatively analyze the electrochemical signals in the presence and absence of the bacteria78. Muamer et al. fabricated a microneedle array-based biosensor for insulin detection, where gold-modified microneedle arrays were used for aptamer immobilization, and changes in the electrochemical impedance spectroscopy (EIS) signal were recorded to reflect the aptamer-insulin binding process (Fig. 4d)65. Kwon et al. proposed a simple and cost-effective electrochemical SELEX monitoring method, in which the binding reaction between cells and aptamers is translated into an electrical signal and analyzed using EIS79. This method eliminates the need for expensive equipment and specific labels such as fluorescent dyes or enzymes, which simplifies the detection process and reduces associated costs, making it an attractive method for continuous monitoring of target. However, this approach faces limitations, especially in terms of its sensitivity. The impedance change induced by a low-concentration target can be weak and challenging to detect effectively. Furthermore, non-specific adsorption of impurities or structurally similar molecules in biological samples onto the electrode surface can introduce significant background interference.
Illustration of various aptamers-based electrochemical sensing modalities. a Changes in electrode surface impedance. b Direct capture signal transduction using electrochemically active substances. c Competitive release signal transduction using electrochemically active substances. d Aptamer-based sensor to perform electrochemical quantification of insulin in ISF using impedance changes on the electrode surface. Reproduced with permission from Elsevier (2025)65. e Aptamer-based direct capture strategy for continuous antibiotic monitoring. Reproduced with permission from American Association for the Advancement of Science (2022)62. f In situ sweat estradiol detection via target-induced displacement of aptamer chains. Reproduced with permission from Springer Nature (2023)61
Signal transduction based on electrochemically active substances
Signal transduction using electrochemically active probes is a significant strategy that relies on the modifiability of aptamers, as previously discussed. This approach involves conjugating an electrochemically active probe acting as a signal probe to the aptamer. Upon aptamer binding to the target, measurable changes occur in the redox behavior of the electrochemically active substance on the electrode surface, enabling quantitative analysis of the target. The primary detection methods employed in this strategy include direct capture mode and competitive release mode. In the direct capture mode, the electrochemically active probes are directly modified with the aptamers. When the aptamer specific binding with the target, a conformational change in the aptamer is induced, which results in a change in the distance between the labeled electrochemically active probes and the electrode surface. This change in distance directly affects the redox reaction kinetics and electrochemical signal intensity of the electrochemically active substances at the electrode surface (Fig. 4b). Based on this detection method, Gao et al. achieved the simultaneous detection of multiple biomarkers in wound exudate60. And Lin et al. enabled kinetic monitoring of drug metabolism in ISF (Fig. 4e)62. This unique sensing mechanism provides a wide range of applications and offers a practical and effective detection strategy for aptamer-based electrochemical continuous monitoring.
In the competitive release mode, an aptamer-complementary nucleic acid chain containing an electrochemically active probe is synthesized, and first assembled on the electrode surface. When no target exists, the electrochemically active probes are bound to the electrode surface, exhibiting a high or characteristic electrochemical signal. And when the target is present in the sample, it competitively binds to the aptamer, and the interaction between the aptamer and its complementary strand is disrupted, leading to the release of the electrochemically active probes-labeled complementary chain from the complex and its detachment from the electrode surface. This release process is competitive, and the released electrochemically active probes diffuse into the solution, resulting in a decrease of the probes on the electrode surface (Fig. 4c). Alternatively, the free electrochemically active probes can undergo electrochemical reactions on the electrode surface, generating or altering the electrochemical signal.
For example, Liu et al. developed a dual-mode sensor based on a competitive mechanism. A complementary DNA (cDNA) probe for acetamiprid (ACE) aptamer, simultaneously modified with MB and Fc, was pre-bound to the ACE aptamer. When the target ACE was present, it competed with the cDNA probe for binding to the ACE aptamer, leading to a decrease in the electrochemical detection signal72. Similarly, Cui et al. reported an aptamer-based electrochemical sensor for the indirect detection of estradiol. This flexible “parallel face-to-face structure” sensor comprised a biorecognition interface (modified with an estradiol aptamer and MB-labeled complementary DNA (MB-ssDNA)) and a AuNPs-MXene modified working electrode (immobilized with capture ssDNA) for indirect target detection. In the presence of the target, the estradiol molecules competed with the MB-ssDNA for binding to the estradiol aptamer. Due to the high affinity of the aptamer for the target, estradiol molecules bound to the estradiol aptamer, leading to the release of the redox probe MB-ssDNA molecules from the biorecognition interface, which were then captured by the working electrode via hybridization with the complementary DNA (Fig. 4f)61.
Signal transduction strategies based on electrochemically active probes show unique advantages in the field of biosensing. Compared to the methods that rely on impedance changes at the electrode interface, this strategy can significantly increase the sensitivity through the inherent electrochemical response of redox-active probes. At the same time, this method eliminates the need for complex sensing design, which significantly simplifies the detection process, and combined with the miniaturized electrode design, it supports dynamic real-time monitoring, which has significant cost advantages in immediate clinical detection and online analysis. However, this strategy also has some limitations, including the possibility that electroactive labels may reduce the binding affinity of the target due to steric effects, and the susceptibility of the active probes to interference by redox substances in the physiological environment.
Application of continuous monitoring electrochemical sensors based on aptamers
Development of aptamer-based electrochemical detection methods
Kevin W. Plaxco’s reported the use of aptamers as molecular recognition elements for detecting thrombin. In this study, the proof-of-concept validation of aptamers binding electrochemical method for detecting target was conducted. By using changes in electrochemical signals upon aptamer binding to thrombin, they successfully achieved thrombin detection. This work demonstrated the potential of aptamers in biosensing and established a foundation for future development of aptamer-based sensors80. Subsequently, researchers began exploring aptamers' immobilization on diverse substrate materials and investigating various signaling pathways. Due to their inherent advantages of ease of operation, relatively low cost, and easily to miniaturization and integration, electrochemical methods have received widespread attention, and have gradually become one of the mainstream research directions in the field of aptamer biosensors81,82. This early stage of research yielded numerous proof-of-concept studies, confirming the feasibility of utilizing aptamers for electrochemical biosensing. However, the performance, stability, and selectivity of aptamer-based sensors required improvement, which limited their broader application at the time.
With the maturity of aptamer selection technologies and the rapid development of nanomaterial technology, aptamer-based electrochemical sensors have entered a phase of accelerated development. The nanomaterials such as nanoparticles, carbon nanotubes, and reduced graphene oxide, which usually have high specific surface area, excellent electrical conductivity, good biocompatibility, and unique optical properties, are used for modification of sensing electrodes in order to achieve an increase in the specific surface area of the electrodes, electrical conductivity of the electrodes, and the immobilization efficiency of the aptamers83,84. For example, Masoumeh et al. synthesized a nanocomposite of Ag nanoparticles, chitosan, graphene quantum dots, and nitrogen-doped TiO2 nanoparticles (Ag-Cs-Gr QDs/NTiO2) and used it to modify the surface of the screen-printed carbon electrode as a platform for aptamer attachment. The monitoring of the actual performance of the aptamer sensor in human serum samples shows that it is superior to traditional methods (Fig. 5a)78. Lu et al. developed a nano Au decorated nitrogen-deficient boron-doped carbon nitride (Au/BCN) nanocomposite. Nano Au was modified in BCN to further enhance light absorption due to its unique surface plasmon resonance effect. Meanwhile, nano Au acts as a bridge between BCN and the aptamer, fixing the aptamer through thiol-gold bonding. The detection accuracy and feasibility of the sensor modified with the nanomaterials indicate that it is a promising analytical method (Fig. 5b)85.
Application of aptamers-based electrochemical sensor. a Constructing aptamers-based electrochemical sensor for the analysis of Staphylococcus aureus by modifying the screen-printed carbon electrode with aptamer/Ag-Cs-Gr QDs/TiO2. Reproduced with permission from Elsevier (2022)78. b Photoelectrochemical sensor of oxytetracycline based on Au-decorated nitrogen-deficient boron-doped carbon nitride (Au/BCN) nanocomposites. Reproduced with permission from Elsevier (2023)85. c Electrochemical aptamer sensor based on core-shell nanomaterial labeling for simultaneous detection of imidacloprid and malathion. Reproduced with permission from Elsevier (2023)75. d Constructing an aptamers-based electrochemical sensor for the analysis of AOH by modifying electrode with Ru MOF/ Cu@Au. Reproduced with permission from Elsevier (2023)71. e Electrochemical and electrochemiluminescent dual-mode aptamer sensor based on self-enhancing bimetallic luminescent MOF. Reproduced with permission from Elsevier (2024)72. f Molecularly imprinted polymers-aptamer electrochemical sensor based on double recognition strategy for chloramphenicol detection. Reproduced with permission from Elsevier (2024)87
Concurrently, modification strategies have been intensively researched to functionalize aptamers according to specific assay requirements86. Huang et al. synthesized two types of core-shell nanomaterials and used them for aptamer labeling. The sensor based on modified aptamer showed high sensitivity and low detection limit for target determination (Fig. 5c)75. In the realm of signal transduction, researchers have devised numerous novel signal transduction modes, encompassing electrochemiluminescence and others. Jiang et al. reported a novel dual-mode aptamer sensor. Ru MOF/Cu@AuNPs serve as ECL substrate platforms for detecting AOH. Compared with conventional aptamer sensors with a single detection signal, this dual-mode aptamer sensor can simultaneously detect ECL and EC signals, improving the accuracy of the results (Fig. 5d–f)71,87. At this stage, aptamer-based electrochemical sensors have demonstrated substantial performance enhancements and are increasingly being implemented in practical detection scenarios, including environmental monitoring, food safety testing, and disease diagnostics88.
In recent years, the development of aptamer-based electrochemical sensors has exhibited a trend towards diversification. Researchers are actively exploring the use of aptamer-based electrochemical sensors for real-time and continuous in vivo monitoring of physiological environments89. Concurrently, aptamer-based electrochemical sensors are being integrated with other biomedical technologies, such as wearable devices, flexible electronics, and bioprinting, to develop novel continuous monitoring systems. Furthermore, the application of aptamers in emerging fields like biocomputing and bioimaging is attracting increasing attention90. At this stage, aptamer-based electrochemical sensors have not only achieved significant advances in fundamental research but also demonstrated substantial application potential in areas such as clinical diagnostics, personalized medicine, and sports performance monitoring.
As an emerging technological platform in health monitoring, aptamer-based wearable electrochemical sensors enable real-time, continuous monitoring of target biomarkers via minimally invasive or non-invasive sampling methods91,92. This approach significantly overcomes the invasive nature and operational complexity of traditional blood testing. By capturing specific targets within alternative biological fluids, the sensors can dynamically reflect the body’s metabolic state, providing molecular information with high spatial and temporal resolution for chronic disease management and stress monitoring93,94. While aptamer-based wearable electrochemical sensors present opportunities for innovative applications, they also pose unique design challenges. Key considerations in the development of these sensors include the stability of the aptamers in biological fluids, the establishment of reliable and applicable signal transduction mechanisms, and the integration of data processing and communication technologies for real-time data analysis and feedback. Furthermore, the sensitive detection of low-concentration biomarkers in alternative biological fluids is critical. The design of aptamer-based wearable electrochemical sensors must also prioritize user comfort and convenience. Ensure these sensors' portability, flexibility, and the ability to achieve continuous monitoring without disrupting daily activities. Addressing these challenges necessitates the integration of innovative materials, biotechnology, and electronic design strategies to construct efficient and practical aptamer-based wearable electrochemical sensors. With the gradual resolution of these challenges, the aptamer-based wearable electrochemical sensors have great potential in achieving health monitoring and personalized medicine. And it is expected to achieve seamless integration of physiological information with daily life95.
Non-invasive continuous biomarker monitoring based on aptamers electrochemical sensors
The readily accessible secretions, such as sweat, saliva, and tears, undergo significant filtration from their blood and ISF sources, resulting in biomarker concentrations substantially lower than those found in blood92. To address this limitation, researchers have developed a flexible field-effect transistor (FET) biosensor array, which utilizes previously unreported cortisol aptamers coupled with a nanometer-thin-film In2O3 FET. Upon capture by the aptamers, the binding of cortisol molecules is transduced into an electrical signal by the FET. The sensing system utilizes label-free, autonomous, and wireless detection technology with an extremely low cortisol detection limit (1 pM) and was applied to take readings in the morning and evening to monitor cortisol levels in sweat. Furthermore, the aptamer-based wearable electrochemical sensors have been successfully regenerated and employed for continuous analysis and monitoring. The research indicates that gate voltage sweeps versus static gate-voltage bias produce different sensor behaviors. While the sensing mechanism of the aptamer-FET relies on surface charge redistribution resulting from target-induced aptamer conformational changes, the gate voltage also influences the aptamer conformation. For instance, altering the gate voltage can induce a low-affinity state in the aptamer, facilitating target release and enabling sensor regeneration. Building upon this research, the validation of smartwatches based on aptamer-FET arrays has been demonstrated, enabling seamless and real-time cortisol sweat sensing (Fig. 6a)96.
Non-invasive nucleic acid aptamer-based sensors. a Non-invasive cortisol monitoring system based on flexible FET biosensors. Reproduced with permission from American Association for the Advancement of Science (2022)96. b, c Wearable aptamer nano-biosensors for noninvasive female hormone monitoring. Reproduced with permission from Springer Nature (2023)61. b Photograph of wearable aptamer nano-biosensor. c Schematic of the sensor regeneration process. d, e Flexible multiplexed aptamer sensors for immediate in situ wound monitoring. Reproduced with permission from American Association for the Advancement of Science (2021)60. d Schematic of the immunosensor for detection of TNF-α, IL-6, IL-8, TGF-β1, S. aureus, pH, and temperature. e Illustration of thin and soft dressing for biomarker analysis, which maintains normal skin function by letting oxygen in and moisture vapor out
Beyond cortisol, continuous monitoring of female hormone levels can also be achieved using sweat-based biosensors. Female hormone plays a critical role in regulating all aspects of reproductive activity, making it essential for reproductive processes and women's health. Cui et al. reported a wearable, skin-adherent nano-biosensor based on target-induced strand displacement for in situ monitoring of estradiol in sweat. The sensor allows for an ultra-low detection limit of 0.14 pM, and based on this, a human study was performed on two healthy women over two consecutive menstrual cycles. Wearable technology for real-time in situ estradiol analysis was evaluated in vitro on day 5, day 13, and day 20 during a menstrual cycle in three female participants, and the measured results were as expected. The estradiol sensor can be readily regenerated under deionized water or acidic conditions for repeated estradiol quantification. After successful regeneration, the estradiol sensor exhibited a signal drift of only 6.2% after five cycles of repetitive measurement and regeneration. The combination of reagent-free, amplification-free, and “signal-on” detection methods with the AuNPs-MXene-based working electrode enables reliable detection sensitivity and an ultralow detection limit. This fully integrated system autonomously induces sweat at rest via iontophoresis, performs precise microfluidic sweat sampling controlled by capillary bursting valves, and facilitates real-time estradiol analysis and calibration. Therefore, the device is capable of collecting multivariate information, integrating signal processing and wireless communication with a user-friendly interface. This enables convenient at-home reproductive hormone monitoring and can be reconfigured for broader biomarker monitoring applications in personalized medicine (Fig. 6b, c)61.
Effective wound management, encompassing both monitoring and treatment of the wound healing process, can reduce hospitalization time, accelerate wound closure, and contribute to advancements in medical research. Real-time wound monitoring is critical. Because it provides timely feedback on the wound’s condition and enables prompt and on-demand treatment, thereby minimizing the likelihood of infection. Gao et al. reported a flexible multiplexed immunosensing platform, named VeCare. The platform is designed for real-time quantitative assessment of the wound microenvironment. The integration of biomimetic passive microfluidic collectors facilitates efficient wound exudate collection, enabling accurate and effective in situ measurements. This sensor overcomes the limitations of traditional single-biomarker detection by quantifying a broad spectrum of healing-related biomarkers using nucleic acid aptamer sensor arrays. Specifically, the immunosensor array enables synchronous quantitative evaluation of multiple clinically relevant biomarkers, including TNF-α, IL-6, IL-8, TGF-β1, pH, temperature, and Staphylococcus aureus within minutes. The technology has demonstrated its in situ multiple wound monitoring capabilities in a rat model and in patients with lower extremity venous ulcers. Longitudinal wound monitoring was first performed in a mouse model, and then, for clinical application, wound exudates were collected weekly from 5 consecutive patients with unhealed venous ulcers and assessed using the VeCare platform. The results of the experiments showed that the immunosensor allowed in situ multi-biomarker analysis of wound fluid during wound healing. The platform is scalable, readily adaptable, and suitable for a wide range of potential applications, which has considerable potential in personalized wound management and improved chronic wound healing outcomes (Fig. 6d, e)60.
Aptamers-based wearable electrochemical sensors offer the potential for non-invasive biomarker monitoring in alternative biological fluids, such as sweat, saliva, tears, and wound exudate. This approach provides an alternative to traditional invasive blood testing and facilitates real-time, continuous tracking of physiological and psychological states. This is a significant advancement in diagnostics and has the potential to revolutionize individualized forms of home health monitoring. However, biosensing in alternative secretions faces several challenges, particularly in ensuring accuracy and reliability. Mechanical strain, temperature variations, pH fluctuations, and biological contamination are potentially important factors that can influence the long-term performance of these sensors. For instance, mechanical strain generated by daily activities can affect the sensor readings, temperature changes can affect the biochemical reactions underlying biomarker detection, and pH fluctuations stemming from individual variations may also influence sensor functionality. Furthermore, given the low concentrations of biomarkers in alternative secretions, optimizing signal transduction mechanisms to minimize background current and enhance sensitivity represents a critical direction for the development of non-invasive aptamer-based wearable electrochemical sensors.
Low-invasive continuous biomarker monitoring based on aptamers electrochemical sensors
Microneedle patch technology offers a minimally invasive and effective approach to invasive continuous monitoring based on nucleic acid aptamers. This technology employs a microneedle array to penetrate the stratum corneum of the skin, enabling painless or minimally painful access to subcutaneous ISF for real-time and continuous monitoring of multiple biomarkers. Microneedle patches mitigate the discomfort and potential infection risks associated with vascular puncture, thereby improving patient compliance and comfort97. Furthermore, compared to non-invasive continuous monitoring methods, minimally invasive microneedle patches effectively bypass the skin’s barrier function, significantly enhancing the extraction efficiency and bioavailability of biomarkers. Consequently, these sensors exhibit notable advantages in detection sensitivity, response speed, stability, and resistance to interference, greatly expanding their applicability in the biomedical field.
Wu et al. developed a microneedle electrochemical biosensor array for continuous monitoring of drug kinetics. This platform uses the high affinity and specificity of aptamers for molecular recognition and provides initial validation of aptamer-based microneedle sensor arrays for real-time, continuous drug monitoring. The monitoring performance of the sensing system was first characterized in vitro using a flow system, followed by in vivo validation via application of the microneedle sensor array to rat skin for continuous monitoring of antibiotic concentrations after intravenous injection. This work represents a successful application of aptamer-based microneedle array sensors for in vivo sensing and establishes a promising platform for the future translation in preclinical animal models, significantly advancing their application and development in this field (Fig. 7a, b)59. Lin et al. fabricated a high-quality gold working electrode by depositing AuNPs onto the surface of microneedles. This improvement achieved effective immobilization of aptamers and efficient electrochemical signal transduction. This method utilizes clinically optimized microneedles, and the entire process only requires a single-step AuNPs deposition and aptamer coating to economically and efficiently prepare high-performance working electrodes for transdermal sensing. The developed sensor was then applied to the dorsal skin of rats for continuous monitoring of tobramycin in ISF. The vancomycin profile captured in the ISF showed a similar drug distribution/elimination pattern as that observed in previously reported vancomycin studies, suggesting that the microneedle patch can be utilized for the real-time continuous ISF drug detection. Following validation, this method was shown to be suitable for detecting various ISF analytes and is particularly well-suited for monitoring antibiotic drugs with narrow therapeutic windows, confirming its potential clinical utility for drug exposure monitoring (Fig. 7c, d)62.
Aptamer-based electrochemical sensors for small molecule monitoring in ISF using microneedle patches. a, b Aptamer sensors for continuous, real-time drug monitoring. Reproduced with permission from American Chemical Society (2022)59. a Schematic diagram of wearable microneedle sensor patch designed for painless molecular tracking in research animals. b Photograph of application of microneedle patch on rat back skin. c, d Aptamer sensors for precise drug delivery in narrow therapeutic windows. Reproduced with permission from American Association for the Advancement of Science (2022)62. c Wearable microneedle-based electrochemical aptamer biosensing for precision dosing of drugs. d Images of assembled aptamer-based microneedle patch. e A magnetic-assisted fixation aptamer microneedle sensor array for pharmacokinetic measurements in ISF. Reproduced with permission from Elsevier (2024)98. f An integrated system combining biocompatible hydrogels and microneedle arrays for biosensing of blood glucose and lactate. Reproduced with permission from Wiley (2024)99
The stability is a critical consideration when applying microneedle patches to rodent skin. This issue is primarily attributed to the rheological properties of the rodent skin, where tissue contraction following microneedle insertion can cause the microneedles to discharge from the skin. This sensor retraction can lead to a loss of electrical contact between the working and reference electrodes, potentially resulting in irreversible sensor damage. To address this challenge, Maria et al. proposed an innovative magnetic-assisted fixation method, which employs magnetic force to firmly secure the microneedle sensor array to the skin of living rodents, preventing microneedle retraction under minor pressure. Specifically, this method involves implanting a metallic plate under the subcutaneous tissue of the rodent and integrating a ring-shaped permanent magnet onto the microneedle patch platform. Utilizing this novel magnetic support device enabled continuous monitoring of therapeutic tobramycin in the animal’s skin ISF. The wearable microneedle patch design reported here achieves stable baseline measurement of electrochemical biological affinity signals through magnetic-assisted skin attachment method, with drift comparable to other published monitoring sensors. At the same time, based on this stable signal baseline, molecular tracking for drift correction in dermis ISF is achieved for the first time using an in vivo microneedle patch sensor array combined with the kinetic differential measurements. By collecting real-time and serial data on the device, the correction of manufacturing variations between sensors, amplification of sensor signal gain, and correction of drift can be achieved, ultimately obtaining accurate pharmacokinetic measurement results (Fig. 7e)98. In a departure from traditional solid microneedles, Fatemeh et al. utilized methacrylic hyaluronic acid (MeHA) hydrogel microneedle (HMN) arrays. These HMN patches swell upon insertion into the skin, facilitating the diffusion of the target analytes towards an aptamer-functionalized electrode. To eliminate potential probe damage during microneedle insertion, the aptamer was not directly placed in the ISF. Instead, the HMN patches are attached to three-electrode electrochemical chips via a hybrid integration. Specifically, the researchers employed a thin of highly swellable hydrogel to adhere the HMN patches to the electrode surface. The implementation of MeHA HMNs effectively addresses the stability challenges encountered with traditional solid microneedle patches, providing a biocompatible, flexible, and mechanically robust medium for ISF extraction and sensing (Fig. 7f)99.
Compared to small molecules, large biomolecules often exhibit higher disease specificity and provide more comprehensive pathophysiological information. Continuous in vivo monitoring of proteins is crucial for real-time disease management and prevention. While sensors for monitoring small molecules, such as glucose, have been available for over a decade, real-time analysis of proteins remains a significant challenge. The inherently low physiological levels of proteins require sensors to have high affinity. However, the excessively tight binding between high-affinity sensors and their target results in prohibitively long dissociation times, hindering their application for real-time tracking of decreasing protein levels. Beyond the detection of small molecules, aptamer-based microneedle patch sensors have demonstrated potential for detecting large molecules in ISF. By designing aptamers that specifically recognize large molecular targets and integrating them into minimally invasive sensor platforms, it is possible to achieve precise monitoring of complex biological entities, such as proteins, peptides, and exosomes. This approach offers novel technological tools for early disease diagnosis, prognostic assessment, and personalized therapy.
Muamer et al. developed a biosensor based on a high-density polymer microneedle array (MNA) for detecting insulin in ISF. This biosensor consists of a gold-coated polymer MNA modified with an insulin-specific aptamer, which was designed to enable both extraction and electrochemical quantification of insulin from ISF. In vitro testing of the microneedle biosensor in artificial ISF demonstrated high detection selectivity for insulin, with a linear response between 0.01 nM and 4 nM, encompassing both physiological and pathological concentration ranges. To further validate the feasibility of this approach in vivo, the researchers applied the MNA-based biosensing platform to mouse skin. The MNA effectively penetrated the skin to access the ISF, enabling insulin extraction and electrochemical measurements, which are sensitive enough to accurately assess insulin levels within the ISF. (Fig. 8a, b)65. Beyond insulin detection, this aptamer-based biosensing platform for biomarker extraction and quantification holds promise for transforming protein biomarker detection from traditional laboratory settings to personalized point-of-care testing environments.
Aptamers-based microneedle patch sensors for monitoring large molecules in interstitial fluid. a, b Wearable electrochemical aptamer sensor based on high-density microneedle arrays for detection of insulin in ISF. Reproduced with permission from Elsevier (2025)65. a Photograph of polymeric MNA-based electrode about wearable electrochemical biosensor based on a high-density microneedle array. b Principle and application of electrochemical biosensor based on microneedle array for insulin detection in ISF. c–e Aptamer-based active reset electrochemical sensor with high sensitivity and specificity for real-time measurement of proteins. Reproduced with permission from American Association for the Advancement of Science (2024)70. c Schematic of fabricated microneedle for actively resetting electrochemical sensor. d Principle diagram of reagentless analysis of protein biomarkers based on aptamer-based molecular pendulum. e Schematic of the implanted actively resetting electrochemical sensor mounted on a rat for real-time, in vivo cytokine monitoring
While various sensors have been developed to explore the detection of large molecules, designing sensors for even larger and more structurally complex proteins presents significant challenges. Traditional aptamers, upon binding to their target proteins, often exhibit high binding affinity and long dissociation times, making it difficult to capture real-time dynamic changes in biomarkers within blood or ISF. To address this issue, Zargartalebi et al. reported an active-reset sensor that employs a “pendulum-like” structural design, where each “pendulum” consists of a double-stranded DNA molecule. One end of the double-stranded DNA is anchored to the electrode surface, while the other end is linked to aptamer fragment that specifically binds the target protein. By changing in potential and applying electric field to the electrode to achieve rapid oscillation of the “pendulums”, the drag and inertial forces induced might cause the target protein to rapidly dissociate. This approach effectively overcomes the limitations of slow receptor dissociation in traditional affinity-based sensors. By employing an oscillation-based active sensor reset mechanism and reagentless molecular pendulum technology, the sensor achieves real-time protein measurement with high sensitivity and specificity. Experimental results demonstrate that the active sensor reset mechanism can recover to an unbound state within 1 min, which allows for continuous protein monitoring (Fig. 8c, d). In addition, the successful measurement of various proteins and molecular biomarkers with different sizes and affinities in diabetes animal models proves the versatility of the platform. Overall, the active reset mechanism proposed in this study offers a novel solution for achieving real-time, continuous monitoring of protein biomarkers in ISF and holds the potential to provide significant benefits to clinical patients, similar to current continuous glucose monitoring technologies (Fig. 8e)70.
Aptamers-based microneedle patch sensors, as an emerging biosensing technology, combine the high selectivity of aptamer recognition with the transdermal delivery and monitoring capabilities of microneedle patches, holding significant developmental importance. Compared to non-invasive biosensors, aptamer-based microneedle patch sensors have a broader range of potential applications, promising real-time, continuous, and minimally invasive monitoring of a wider array of biomarkers. This technology exhibits broad prospects in areas such as early disease diagnosis, personalized medication guidance, and remote health monitoring. Particularly in chronic disease management, aptamer-based microneedle patch sensors have the potential to improve patient comfort and adherence, thereby enhancing disease control and prognosis.
At the same time, the development of aptamer-based microneedle patch sensors still faces several challenges. Optimization of microneedle patch design and fabrication is urgently needed. This includes precise design of microneedle structures to achieve efficient transdermal penetration, stable aptamer immobilization, and repeatable large-scale production. Furthermore, improving sensor performance is critical. It is necessary to require increased sensitivity and expanded detection range, along with enhanced long-term stability and selectivity to withstand the interference of complex biological environments. In terms of biocompatibility, attention should be given to the selection and surface modification of microneedle materials to minimize local immune responses and assess potential risks associated with long-term use. Successful translation of microneedle-based sensors into clinical applications relies on continued innovation in materials science, sensor technology, and manufacturing techniques to overcome these challenges and ensure that these highly promising devices can fully realize their potential in personalized healthcare and disease management.
Design considerations of aptamer-based wearable electrochemical sensors for continuous in situ biomarker analysis
The core goal of aptamer-based wearable electrochemical sensor development is to enable continuous in situ monitoring biomarkers in biological fluids. However, accurate detection of biomarkers faces some technical challenges, including the complexity of the bio-affinity response, sensor regeneration, sensor stability, bio-contamination of the sensor surface, efficient transportation of samples on the sensor surface, etc. In this section, we will introduce the challenges and discuss potential solutions for designing the next generation of aptamer-based wearable biosensors associated with various systems and biofluids. The future research directions and perspectives in this field for accurate and continuous in situ biomarker monitoring will also be covered.
Aptamers-based biofluidic interfaces for electrochemical sensors
The performance of wearable sensors is highly dependent on how they interact with dynamic and complex biofluidic matrices. By integrating microfluidics, sensing platforms are able to efficiently collect biofluids and precisely deliver them to the surface of aptamer-based sensors. When designing aptamer-based wearable electrochemical sensing platforms, the biomarker acquisition methods and flexible device morphology, as well as efficient sample transportation, are key elements to be focused on, especially in noninvasive sensor applications. For biofluid transport, the ability of continuous sensing platforms to efficiently collect and update biofluids in a timely manner without the need for user intervention is crucial for accurately monitoring the dynamic change of biomarker concentrations. Considering the challenges of continuous long-term sensing, researchers are actively exploring the various bionic structures and the optimal design of microfluidic platforms. For example, microfluidic systems based on capillary force or negative pressure driving can precisely control the collection process of biofluidic samples. These are not only suitable for microfluidic collection and sensing in a single sensing region, but also can realize precise sampling in different sensing regions in multi-parameter sensing platforms, thus realizing the simultaneous analysis of multiple biomarkers.
Efficient monitoring of low-abundance biomarkers
Current signaling strategies based on redox-active probes provide an important technological approach for reagentless sensing of biomarkers in vivo. The aptamers undergo binding-induced conformational changes, and the molecular pendulum of redox-active probes exhibits detectable changes in the rate of electron transfer. Wearable sensors in biofluids are often required to detect low-abundance blood-related biomarkers. Aptamers-based sensors have shown excellent sensitivity in a variety of biofluids, based on the high specific binding ability of the aptamers and the efficient electron transfer of probes. When faced with the need for low-abundance biomarker detection, modification of the electrode surface by nanomaterials with high surface area and charge carrier mobility can further improve the electrode performance and enhance the signal gain. Electrodes modified with nanomaterials can now be fabricated using a variety of methods, including inkjet printing, nanocomposite deposition, and magnetron sputtering. In addition, based on the easy modification of aptamers, the detection of low-abundance biomarkers would rely on exploring safer and more effective signal amplification probes, and the modification of aptamers to achieve effective signal amplification.
Effective regeneration of aptamer-based electrochemical sensors
Aptamers screening usually relies on the SELEX technique, which favors enrichment of aptamers with high affinity to meet the sensitivity and selectivity requirements of conventional assays. However, in a continuous biosensing situation, reversible binding is necessary to capture fluctuations in the concentration of target biomarkers. Therefore, there is an urgent need to construct smart aptamer sensing systems with controlled dissociation kinetics. In the design of aptamer-based sensors, an effective balance between sensitivity and reproducibility is required. In order to realize the reversible binding of aptamer and target without significantly reducing the effective binding ability of the aptamer, the aptamer sequence can be rationally designed. Notably, traditional structure-based inverse design still faces challenges due to the complexity of the aptamer’s dynamic folding pathway and its multipoint interaction properties with the target. By cooperating with multi-parameter machine learning models, it is expected to break through the limitations of current experimental capabilities and achieve the simultaneous optimization of multiple key properties of the aptamer, such as affinity, specificity, dissociation kinetics, structure, and stability. In addition, advances in machine learning, especially by modeling molecular docking and binding kinetics, are expected to deepen our understanding of the molecular recognition mechanisms of aptamers.
Stability of aptamer-based electrochemical sensors
The stability of the aptamers-based electrochemical sensors is critical to achieve accurate and continuous measurements for biomarkers. Although aptamers-based electrochemical sensors have shown significant improvements in stability compared to antibody-based electrochemical immunosensors, they can still be affected by a number of factors, including the chemical stability of the redox-active probes, bio-contamination of the electrode surface, and the long-term reliability of the sensor performance. First, the selection of a suitable redox-active probe is a primary consideration in the design of many aptamer-based electrochemical sensors, with the aim of improving the electron transfer rate, signal quality, and overall stability. MB, a commonly used redox-active probe, has shown good stability over long periods of use under different sensor conditions. In the future, it is expected that more redox-active probes with reliable performance will be developed. Secondly, biological contamination on the electrode surface is another important factor affecting signal stability. The high content of proteins and cells in complex biofluids and the variable media conditions pose a serious challenge to the sensors, which is a major problem for bioelectronic interface. When contaminant molecules in the biofluid are adsorbed on the electrode surface, it hinders the electron transfer, which in turn affects the signal transduction of the sensor. To minimize biological contamination issues, physical and chemical barriers can be employed to protect the sensors, such as by using porous membranes, hydrogel encapsulation, polymer coatings, and nanoporous conductive substrates. Finally, the long-term reliability of sensor performance is also an important consideration. Factors such as the biocompatibility of the sensor, the resistance to stretching of sensor worn on skin surface, adhesion, and permeability may all affect the signal sensing ability to varying degrees. By rationally designing the sensor to minimize the interference of these factors, the reliability of the sensor can be improved during long-term use.
Conclusion
Aptamers are particularly compelling as biosensing elements because of their specificity, chemical resilience, ease of tailored functionalization, and the inherent reversibility of their binding interactions. These characteristics position aptamers as a transformative alternative to conventional antibodies, offering distinctive opportunities to address limitations associated with traditional biomolecular recognition elements. In clinical practice, aptamer-based wearable sensors can significantly enhance early disease diagnosis, enable dynamic monitoring of treatment effectiveness, and facilitate personalized patient management, thereby potentially improving therapeutic outcomes. Despite these promising developments, several critical issues remain unresolved. For example, achieving accurate, reversible, and stable aptamer-target interactions in vivo continues to be a central challenge. Expanding sensor applicability to a wider range of biomarkers need for a delicate balance between sensitivity of aptamer-target interactions and the practical requirements for sensor regeneration and reuse. Besides, the long-term operational stability of aptamer-based wearable electrochemical sensors within complex biological environments poses substantial difficulties. Factors including nonspecific adsorption of proteins, biofouling, mechanical stresses from continuous skin contact, and chemical degradation processes may be addressed through advanced electrode material engineering, biocompatible interface design, and innovative antifouling surface modifications.
This review systematically explores the recent advances and applications of aptamer-based wearable electrochemical sensors for continuous biomarker monitoring. Depending on the biomarker sampling method, the applications in different environments are discussed from both non-invasive and low-invasive perspectives. Among them, the non-invasive sampling method is mainly applied to the monitoring of biomarkers in sweat and the personalized management of wounds, while the low-invasive sampling method can directly contact the biomarkers in ISF with the assistance of the microneedle patches, which has a wider range of applications, including real-time monitoring of drug metabolism, the monitoring of chronic diseases, and prospective health management. Further research in this field can use interdisciplinary approaches, combining materials engineering, biomedical device design, and data-driven analytics, to achieve reliable in vivo sensor performance and clinical applicability. Such comprehensive strategies hold the potential to transition aptamer-based wearable sensing technology from proof-of-concept prototypes towards practical medical devices suitable for widespread clinical adoption, ultimately fostering new possibilities in precision medicine and personalized healthcare delivery. Meanwhile, the integration of these sensors with digital health platforms can further enable real-time data analytics to facilitate personalized health management and disease prevention.
References
Paek, S.-H. Real-time monitoring of biomarkers: current status and future perspectives. BioChip J. 14, 1–1 (2020).
Islam, M. A. & Masson, J.-F. Plasmonic biosensors for health monitoring: inflammation biomarker detection. ACS Sens. 10, 577–601 (2025).
Lista, S. et al. Integrative metabolomics science in Alzheimer’s disease: relevance and future perspectives. Ageing Res. Rev. 89, 101987 (2023).
Chen, X., Wei, Y., Wang, X., Liu, X. & Wang, X. An overview in interactions and biosensors of multiple biomarkers of Alzheimer’s disease. Chem. Eng. J. 508, 100704 (2025).
Han, X., Wang, J. & Sun, Y. Circulating tumor DNA as biomarkers for cancer detection. Genomics Proteomics Bioinforma. 15, 59–72 (2017).
Khaliq, N. et al. Enzyme-free detection of creatinine as a kidney dysfunction biomarker using TiO2 flow-through membranes. Nanoscale Adv. 7, 643–658 (2025).
Zirath, H. et al. Bedside immune monitoring: an automated immunoassay platform for quantification of blood biomarkers in patient serum within 20 min. Anal. Chem. 89, 4817–4823 (2017).
Zhuang, J. et al. Liposome-amplified photoelectrochemical immunoassay for highly sensitive monitoring of disease biomarkers based on a split-type strategy. Biosens. Bioelectron. 99, 230–236 (2018).
Steiger, C. et al. Dynamic monitoring of systemic biomarkers with gastric sensors. Adv. Sci. 8, 2102861 (2021).
Parolo, C. et al. Real-Time Monitoring of a Protein Biomarker. ACS Sens. 5, 1877–1881 (2020).
Liu, S., Shen, Z., Deng, L. & Liu, G. Smartphone assisted portable biochip for non-invasive simultaneous monitoring of glucose and insulin towards precise diagnosis of prediabetes/diabetes. Biosens. Bioelectron. 209, 114251 (2022).
Timilsina, S. S. et al. Biofabrication of multiplexed electrochemical immunosensors for simultaneous detection of clinical biomarkers in complex fluids. Adv. Healthcare Mater. 11, e2200589 (2022).
Chen, H. et al. The applications of electrochemical immunosensors in the detection of disease biomarkers: a review. Molecules 28, 3605 (2023).
Guo, J., Wu, S., Wang, Y. & Zhao, M. A label-free fluorescence biosensor based on a bifunctional MIL-101(Fe) nanozyme for sensitive detection of choline and acetylcholine at nanomolar level. Sens. Actuators B Chem. 312, 128021 (2020).
Yang, J. et al. Masticatory system–inspired microneedle theranostic platform for intelligent and precise diabetic management. Sci. Adv. 8, eabo6900 (2022).
Sequeira-Antunes, B. & Ferreira, H. A. Nucleic acid aptamer-based biosensors: a review. Biomedicines 11, 3201 (2023).
Ye, C., Lukas, H., Wang, M., Lee, Y. & Gao, W. Nucleic acid-based wearable and implantable electrochemical sensors. Chem. Soc. Rev. 53, 7960–7982 (2024).
Zhu, C. et al. Recent progress of SELEX methods for screening nucleic acid aptamers. Talanta 266 (2024).
Liu, B. et al. Advances of functional nucleic acids based on specific recognition:A review. Int. J. Biol. Macromol. 304, 124998 (2025).
Zhang, L. et al. Molecular elucidation of disease biomarkers at the interface of chemistry and biology. J. Am. Chem. Soc. 139, 2532–2540 (2017).
Seibold, J. M., Abeykoon, S. W., Ross, A. E. & White, R. J. Development of an electrochemical, aptamer-based sensor for dynamic detection of neuropeptide Y. ACS Sens. 8, 4504–4511 (2023).
Kumakli, H., Baldwin, M., Abeykoon, S. W. & White, R. J. Microscale, electrochemical, aptamer-based sensors for enhanced small-molecule detection at millisecond time scales. ACS Sens. 8, 4521–4530 (2023).
Rangel, A. E., Hariri, A. A., Eisenstein, M. & Soh, H. T. Engineering aptamer switches for multifunctional stimulus‐responsive nanosystems. Adv. Mater. 32, e2003704 (2020).
Zhu, C., Li, L., Yang, G. & Qu, F. Investigating the influences of random-region length on aptamer selection efficiency based on capillary electrophoresis–SELEX and high-throughput sequencing. Anal. Chem. 93, 17030–17035 (2021).
Nguyen, M.-D. et al. Truncations and in silico docking to enhance the analytical response of aptamer-based biosensors. Biosens. Bioelectron. 265, 116680 (2024).
Nguyen, M.-D. et al. Electrochemical aptamer-based biosensors for measurements in undiluted human saliva. ACS Sens. 8, 4625–4635 (2023).
Stanley, S. et al. Comprehensive aptamer-based screening identifies a spectrum of urinary biomarkers of lupus nephritis across ethnicities. Nat. Commun. 11, 2197 (2020).
Chen, L., Yang, G. & Qu, F. Advances of aptamer-based small-molecules sensors in body fluids detection. Talanta 268, 125348 (2024).
Stangherlin, S., Ding, Y. & Liu, J. Dissociation constant (Kd) measurement for small‐molecule binding aptamers: homogeneous assay methods and critical evaluations. Small Methods 7, e2401572 (2024).
Cai, S. et al. Investigations on the interface of nucleic acid aptamers and binding targets. Analyst 143, 5317–5338 (2018).
Sun, N.-N., Tinnefeld, P., Li, G.-l., He, Z.-K. & Xu, Q.-F. Aptamer melting biosensors for thousands of signaling and regenerating cycles. Biosens. Bioelectron. 271, 116998 (2025).
Biswas, G. C., Khan, M. T. M. & Das, J. Wearable nucleic acid testing platform - A perspective on rapid self-diagnosis and surveillance of infectious diseases. Biosens. Bioelectron. 226, 115115 (2023).
Dauphin-Ducharme, P. et al. Electrochemical aptamer-based sensors for improved therapeutic drug monitoring and high-precision, feedback-controlled drug delivery. ACS Sens. 4, 2832–2837 (2019).
Kim, J., Campbell, A. S., de Ávila, B. E.-F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406 (2019).
Sim, D. et al. Biomarkers and detection platforms for human health and performance monitoring: a review. Adv. Sci. 9, e2104426 (2022).
Downs, A. M. & Plaxco, K. W. Real-time, in vivo molecular monitoring using electrochemical aptamer based sensors: opportunities and challenges. ACS Sens. 7, 2823–2832 (2022).
Shaver, A. & Arroyo-Currás, N. The challenge of long-term stability for nucleic acid-based electrochemical sensors. Curr. Opin. Electrochem. 32, 100902 (2022).
Yuan, Y. & Arroyo-Currás, N. Continuous molecular monitoring in the body via nucleic acid–based electrochemical sensors: the need for statistically-powered validation. Curr. Opin. Electrochem. 39 (2023).
Yang, X., Qu, Y., Wang, C., Wu, L. & Hu, X. Development of an Enzyme-Linked Immunosorbent Assay Based on a Monoclonal Antibody for the Rapid Detection of Citrinin in Wine. Foods 14, 101305 (2024).
Thiviyanathan, V. & Gorenstein, D. G. Aptamers and the next generation of diagnostic reagents. PROTEOMICS Clin. Appl. 6, 563–573 (2012).
Ellington, A. D. & Szostak, J. W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990).
Tuerk, C. & Gold, L. Systematic evolution of ligands by exponential enrichment RNA ligands to bacteriophage T4 DNA polymerase. Science 249, 505–510 (1990).
Zhang, S. et al. Construction of a microfluidic SELEX platform for efficient screening of advanced glycation end products aptamer. Biosens. Bioelectron. 271, 117038 (2024).
Chung, Y.-D., Tsai, Y.-C., Wang, C.-H. & Lee, G.-B. Aptamer selection via versatile microfluidic platforms and their diverse applications. Lab A Chip 25, 1047–1080 (2025).
Yang, K.-Z., Wang, M., Gao, M.-Y., Wang, Y.-T. & Zhang, Z.-L. Dynamic selection of high-affinity aptamers using a magnetically activated continuous deflection microfluidic chip. Chem. Commun. 60, 2772–2775 (2024).
Chang, D. et al. A high-dimensional microfluidic approach for selection of aptamers with programmable binding affinities. Nat. Chem. 15, 773–780 (2023).
Li, Y. et al. Precision-SELEX aptamer screening for the colorimetric and fluorescent dual-readout aptasensing of AFB1 in food. Food Chem. 436, 137661 (2024).
Singh, N. K. et al. High-affinity one-step aptamer selection using a non-fouling porous hydrogel. Nat. Biotechnol. 42, 1224–1231 (2023).
Zimmer, K. J. L., Johnson, R. E., Little, H., Duhamel, J. & Manderville, R. A. Harnessing a fluorescent nucleobase surrogate for supramolecular FRET-aptamer detection and target-site mapping. ACS Sensors 10, 1822–1832 (2025).
Kalsoom, I. et al. Structure switching aptamer enhance sensitivity and specificity of photonic crystal-based sensors for RSV-G protein detection. Biosens. Bioelectron. 273, 117091 (2025).
Bae, S. W., Lee, J. S., Harms, V. M. & Murphy, W. L. Dynamic, bioresponsive hydrogels via changes in DNA aptamer conformation. Macromol. Biosci. 19, e1800353 (2018).
Qin, S.-N. et al. A nanoporous electrochemical aptamer-based sensors for rapid detection of cardiac troponin I in blood. Talanta 284, 127250 (2025).
Xu, G. et al. Structural basis for high-affinity recognition of aflatoxin B1 by a DNA aptamer. Nucleic Acids Res. 51, 7666–7674 (2023).
Xu, G. et al. Structural Insights into the Mechanism of High-Affinity Binding of Ochratoxin A by a DNA Aptamer. J. Am. Chem. Soc. 144, 7731–7740 (2022).
Fallah, A., Imani Fooladi, A. A., Havaei, S. A., Mahboobi, M. & Sedighian, H. Recent advances in aptamer discovery, modification and improving performance. Biochem. Biophys. Rep. 40, 101852 (2024).
Murray, MakayT. & Wetmore, StaceyD. Unlocking precision in aptamer engineering: a case study of the thrombin binding aptamer illustrates why modification size, quantity, and position matter. Nucleic Acids Res. 52, 10823–10835 (2024).
Meng, X., O’Hare, D. & Ladame, S. Surface immobilization strategies for the development of electrochemical nucleic acid sensors. Biosens. Bioelectron. 237, 115440 (2023).
Tammam, S. N. et al. Chitosan gold nanoparticles for detection of amplified nucleic acids isolated from sputum. Carbohydr. Polym. 164, 57–63 (2017).
Wu, Y. et al. Microneedle aptamer-based sensors for continuous, real-time therapeutic drug monitoring. Anal. Chem. 94, 8335–8345 (2022).
Gao, Y. et al. A flexible multiplexed immunosensor for point-of-care in situ wound monitoring. Sci. Adv. 7, eabg9614 (2021).
Ye, C. et al. A wearable aptamer nanobiosensor for non-invasive female hormone monitoring. Nat. Nanotechnol. 19, 330–337 (2023).
Lin S. et al. Wearable microneedle-based electrochemical aptamer biosensing for precision dosing of drugs with narrow therapeutic windows. Sci. Adv. 8, eabq4539 (2022).
Meng, X. et al. Engineering 3D microtip gates of all-polymer organic electrochemical transistors for rapid femtomolar nucleic-acid-based saliva testing. Biosen. Bioelectron. 273, 117170 (2025).
De Penning, S., Murphy, M. P., Kingston, T. A., Nilsen-Hamilton, M. & Shrotriya, P. Influence of crosslinker on aptamer immobilization and aptasensor sensing response for non-metallic surfaces. Biosens. Bioelectron. 270, 116933 (2025).
Dervisevic M. et al. High-density microneedle array-based wearable electrochemical biosensor for detection of insulin in interstitial fluid. Biosens. Bioelectron. 271, 116995 (2025).
Rahmani Kheyrollahi, M., Mohammadnejad, J., Eidi, A. & Jafary, H. Synthesis and in vitro study of surface-modified and anti-EGFR DNA aptamer-conjugated chitosan nanoparticles as a potential targeted drug delivery system. Heliyon 10, e38904 (2024).
Liu, Z. et al. Stable single-walled carbon nanotube-streptavidin complex for biorecognition. J. Phys. Chem. C. 114, 4345–4352 (2010).
Wang, M., Liu, H. & Fan, K. Signal amplification strategy design in nanozyme‐based biosensors for highly sensitive detection of trace biomarkers. Small Methods 7, 2301049 (2023).
Yang, Q. et al. An aptamer-based electrochemical sensor for the quantification of the malaria biomarker lactate dehydrogenase. Biosens. Bioelectron. 274, 117152 (2025).
Zargartalebi, H. et al. Active-reset protein sensors enable continuous in vivo monitoring of inflammation. Science 386, 1146–1153 (2024).
Jiang, M. et al. Construction of electrochemical and electrochemiluminescent dual-mode aptamer sensors based on ferrocene dual-functional signal probes for the sensitive detection of Alternariol. Analytica Chim. Acta 1272, 341476 (2023).
Liu, J. et al. Detection of acetamiprid residues in vegetables by a self-enhanced bimetallic luminescent MOF-based electrochemiluminescence-electrochemistry dual-mode aptasensor. Sens. Actuat. B Chem. 410, 135665 (2024).
Białas, K., Moschou, D., Marken, F. & Estrela, P. Electrochemical sensors based on metal nanoparticles with biocatalytic activity. Microchim. Acta 189, 172 (2022).
Anu Prathap, M. U., Kaur, B. & Srivastava, R. Electrochemical sensor platforms based on nanostructured metal oxides, and zeolite‐based materials. Chem. Rec. 19, 883–907 (2018).
Huang, J. et al. A novel electrochemical aptasensor based on core–shell nanomaterial labeling for simultaneous detection of acetamiprid and malathion. Food Chem. 429 (2023).
Chen, M. et al. Aptamer-based electrochemical biosensing platform for analysis of cardiac biomarkers. ACS Sens. 9, 5354–5362 (2024).
Parihar, A., Vishwakarma, P., Prajapati, P. & khan, R. Non-invasive ultra-sensitive detection of breast cancer biomarker using cerium nanoparticle functionalized graphene oxide enabled impedimetric aptasensor. Biosens. Bioelectron. 268, 116925 (2025).
Ghalkhani, M., Sohouli, E., Khaloo, S. S. & Vaziri, M. H. Architecting of an aptasensor for the staphylococcus aureus analysis by modification of the screen-printed carbon electrode with aptamer/Ag–Cs-Gr QDs/NTiO2. Chemosphere 293, 133597 (2022).
Kwon, Y. et al. Electrochemical cell-SELEX monitoring and its application to electrochemical aptasensor for colorectal cancer detection. Chem. Eng. J. 506, 159935 (2025).
Xiao, Y., Lubin, A. A., Heeger, A. J. & Plaxco, K. W. Label‐free electronic detection of thrombin in blood serum by using an aptamer‐based sensor. Angew. Chem. Int. Ed. 44, 5456–5459 (2005).
Duan, H., Tang, S.-Y., Goda, K. & Li, M. Enhancing the sensitivity and stability of electrochemical aptamer-based sensors by AuNPs@MXene nanocomposite for continuous monitoring of biomarkers. Biosens. Bioelectron. 246, 115918 (2024).
Kaur, H. & Shorie, M. Nanomaterial based aptasensors for clinical and environmental diagnostic applications. Nanoscale Adv. 1, 2123–2138 (2019).
Wang, Z. et al. Towards detection of biomarkers in the eye using an aptamer-based graphene affinity nanobiosensor. Talanta 250, 115918 (2022).
Wang, Z., Huang, C., Sun, N. & Deng, C. Advances in aptamer-based nanomaterials for separation and analysis of non-genetic biomarkers in biofluids. Sci. China Chem. 64, 932–947 (2021).
Qiao, L. et al. Ultrasensitive photoelectrochemical sensor of oxytetracycline based on nano Au decorated nitrogen-deficient boron-doped carbon nitride nanosheets. J. Environ. Chem. Eng. 11, 111547 (2023).
Kurup, C. P., Mohd-Naim, N. F. & Ahmed, M. U. Recent trends in nanomaterial-based signal amplification in electrochemical aptasensors. Crit. Rev. Biotechnol. 42, 794–812 (2021).
Geng, L. et al. Molecularly imprinted polymers-aptamer electrochemical sensor based on dual recognition strategy for high sensitivity detection of chloramphenicol. Food Chem. 437, 137933 (2024).
Wang, Y. et al. Aptamer-based electrochemical analysis platform for tumor cells and biomarkers detection. J. Electroanalytical Chem. 960, 118194 (2024).
Hu, C., Wang, L., Liu, S., Sheng, X. & Yin, L. Recent development of implantable chemical sensors utilizing flexible and biodegradable materials for biomedical applications. ACS Nano 18, 3969–3995 (2024).
Zheng, X. et al. Aptamer-functionalized nucleic acid nanotechnology for biosensing, bioimaging and cancer therapy. Nanoscale 17, 687–704 (2025).
Flynn, C. D. et al. Biomolecular sensors for advanced physiological monitoring. Nat. Rev. Bioeng. 1, 560–575 (2023).
Heikenfeld, J. et al. Accessing analytes in biofluids for peripheral biochemical monitoring. Nat. Biotechnol. 37, 407–419 (2019).
Pundir, M. et al. Emerging biotechnologies for evaluating disruption of stress, sleep, and circadian rhythm mechanism using aptamer-based detection of salivary biomarkers. Biotechnol. Adv. 59, 107961 (2022).
Nandhakumar, P. et al. Simultaneous and rapid detection of glucose and insulin: coupling enzymatic and aptamer-based assays. Anal. Chem. 96, 18806–18814 (2024).
Tu, J., Torrente‐Rodríguez, R. M., Wang, M. & Gao, W. The era of digital health: a review of portable and wearable affinity biosensors. Adv. Function. Mater. 30, 1906713 (2019).
Wang, B. et al. Wearable aptamer-field-effect transistor sensing system for noninvasive cortisol monitoring. Sci. Adv. 8, eabk0967 (2022).
Sun, H., Zheng, Y., Shi, G., Haick, H. & Zhang, M. Wearable clinic: from microneedle‐based sensors to next‐generation healthcare platforms. Small 19, e2207539 (2023).
Reynoso, M. et al. 3D-printed, aptamer-based microneedle sensor arrays using magnetic placement on live rats for pharmacokinetic measurements in interstitial fluid. Biosens. Bioelectron. 244, 115802 (2024).
Bakhshandeh, F. et al. Wearable aptalyzer integrates microneedle and electrochemical sensing for in vivo monitoring of glucose and lactate in live animals. Adv. Mater. 36, 2313743 (2024).
Acknowledgements
The authors would like to acknowledge financial support from the National Natural Science Foundation of China (Grant No. T2225010, 32171399, 32171456, 32401202), Guangdong Basic and Applied Basic Research Foundation (Grant No. 2023A1515011267, 2023A1515111139, 2025A1515010608), Science and Technology Program of Guangzhou, China (Grant No. 2024B03J0121, 2024B03J1284), Shenzhen Science and Technology Program (Grant No. RCBS20231211090558093), China Postdoctoral Science Foundation (2023TQ0386), Fundamental Research Funds for the Central Universities, Sun Yat-sen University (No. 24xkjc011), the Opening Project of The National Key Laboratory of Smart Farm Technology and Systems.
Author information
Authors and Affiliations
Contributions
Xiaotong Li, Suhang Liu, and Xinshuo Huang contributed equally.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, X., Liu, S., Huang, X. et al. Aptamers-based wearable electrochemical sensors for continuous monitoring of biomarkers in vivo. Microsyst Nanoeng 11, 241 (2025). https://doi.org/10.1038/s41378-025-00993-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41378-025-00993-5