Abstract
White matter hyperintensities (WMHs) are common in Alzheimer’s disease (AD) and may contribute to cognitive impairment. However, the associations between regional WMH volumes, cognitive domains, and plasma biomarkers remain unclear. This study aimed to explore these relationships across the AD spectrum. A total of 311 participants were enrolled, including healthy controls (HC), individuals with subjective cognitive decline (SCD), mild cognitive impairment (MCI), and AD. All participants underwent comprehensive neuropsychological assessment, brain magnetic resonance imaging, and plasma biomarker analysis. WMH volumes were segmented using United Imaging software and classified based on anatomical location: juxtaventricular WMH (juxWMH), periventricular WMH (pWMH), juxtacortical WMH (jcWMH), and deep WMH (dWMH). Correlations among regional WMH volumes, plasma biomarkers, and cognitive domains were analyzed with multiple comparisons. Path analysis was used to assess potential mediation effects. Mediation analyses using bootstrapping were conducted separately in cognitively unimpaired and cognitively impaired groups to assess WMH-mediated pathways between biomarkers and cognition. Compared with HC, SCD, and MCI groups, the AD group showed significantly increased log-transformed (lg) juxWMH volumes (all P < 0.05, Bonferroni corrected). The AD group showed significantly larger lgpWMH and lgjcWMH volumes than HC, SCD and AD groups (all P < 0.05). Lower plasma Aβ42/Aβ40 ratio was associated with higher lgjuxWMH, lgpWMH, and lgjcWMH volumes. Higher lgjuxWMH volume was associated with worse memory (r = −0.16, P = 0.006), language (r = −0.34, P < 0.001), and executive function (r = 0.18, P = 0.003). Similar trends were found for lgpWMH and lgjcWMH volumes. Mediation analysis revealed that in cognitively unimpaired individuals, juxWMH and pWMH primarily mediated amyloid-cognitive associations, while in cognitively impaired patients, mediation expanded to include neuroinflammatory and neurodegeneration pathways across multiple location-specific WMH. Location-specific WMH demonstrate stage-dependent mediation patterns in AD pathophysiology, evolving from amyloid-driven changes to multi-factorial processes. These findings provide insights for developing targeted therapeutic strategies at different disease stages.
Similar content being viewed by others
Introduction
White matter hyperintensities (WMH) are defined as hyperintense signals on T2-weighted imaging (T2WI) and T2 fluid-attenuated inversion-recovery (T2-FLAIR) magnetic resonance images. WMH are relatively common in cognitively normal elderly individuals. Previous studies suggest they are associated with vascular risk factors [1], such as hypertension or diabetes, and are related to poorer cognitive performance, particularly in executive function and processing speed [2, 3]. While WMH are traditionally considered markers of cerebrovascular pathology, emerging evidence suggests their relationship with cognitive decline may be more complex, particularly in the context of neurodegenerative diseases.
The role of WMH in Alzheimer’s disease (AD) has attracted increasing attention. In patients with AD, WMH volumes appear to be larger than in cognitively normal elderly individuals [4], especially in periventricular regions[5, 6] and posterior areas. However, despite these consistent volumetric findings, the cognitive relevance of WMH in different regions controversial. Some studies suggest that periventricular white matter hyperintensities, rather than deep white matter hyperintensities, are more closely related to the pathogenesis of cognitive impairment. Recent evidence suggests that WMH topography carries pathophysiological information, with posterior WMH preferentially associated with amyloid pathology and anterior WMH more closely related to vascular risk factors [7, 8]. These findings highlight the importance of considering regional WMH distribution when investigating early AD pathology. However, other studies have reported that deep WMH burden may have a stronger association with cognitive impairment [9, 10]. This inconsistency may be partly due to the limitations of the commonly used clinical WMH scoring system - the Fazekas score. The Fazekas score is commonly used clinically to assess WMH severity, but its classification of WMH is relatively broad, and the definitions of various lesion types based on this score are not sufficiently clear, failing to reflect the complex pathology and etiology of WMH [11]. To improve anatomical specificity, Kim et al. classified WMH based on the anatomical location, etiology, and pathophysiological mechanisms into four categories: juxtacortical white matter hyperintensity (jcWMH), deep white matter hyperintensity (dWMH), periventricular white matter hyperintensity (pWMH), and juxtaventricular white matter hyperintensity (juxWMH) [12, 13].
Building on these anatomical insights, researchers have begun investigating the biological mechanisms linking WMH to AD pathology. Previous studies have investigated the relationship between WMH and cerebrospinal fluid (CSF) Aβ42 in cognitively normal individuals, suggesting potential AD-specific pathways leading to WMH. Several cross-sectional studies found that lower CSF Aβ42 levels (i.e., more abnormal) were associated with higher WMH volumes in cognitively normal elderly individuals [14, 15]. However, the relationship between plasma Aβ42 and WMH remains unclear.
Recent advances in blood-based biomarkers offer new opportunities to explore these relationships. In recent years, plasma biomarkers have become a research hotspot. For example, Teunissen and colleagues found that the plasma Aβ42/Aβ40 ratio can identify abnormal CSF Aβ and/or Aβ-PET status across clinical stages of AD [16]. Lower plasma Aβ42/Aβ40 levels are associated with cognitive impairment and an increased risk of AD development, both in cognitively unimpaired individuals or in those with subjective cognitive decline (SCD) or mild cognitive impairment (MCI) [16]. This correlation is consistent with findings from CSF Aβ and/or Aβ-PET data studies. Beyond amyloid markers, plasma biomarkers of neuroinflammation such as glial fibrillary acidic protein (GFAP) and neurodegeneration (neurofilament light chain, NFL) have also emerged as promising tools for understanding AD pathology and may provide complementary information about white matter damage.
Despite these advances, critical gaps remain in our understanding of the relationships among plasma biomarkers, anatomically-defined WMH, and cognitive function across different stages of AD. In this study, our objectives were to investigate: (1) the associations between plasma biomarkers and anatomically-defined WMH categories across the AD continuum; (2) whether location-specific WMH mediate the relationships between plasma biomarkers and cognitive function; and (3) whether these mediation patterns differ across cognitive stages. We hypothesized that plasma biomarkers and location-specific WMH are independently associated with cognitive function, with WMH serving as mediators between plasma biomarkers and cognitive outcomes. Furthermore, we anticipated that mediation patterns would evolve from simple amyloid-driven pathways in cognitively unimpaired individuals to more complex multi-pathological mechanisms involving neuroinflammation and neurodegeneration in cognitively impaired patients.
Materials and methods
Study participants
This study was approved by the Ethics Committee of Hainan General Hospital (Hainan Hospital Affiliated to Hainan Medical University). Written informed consent has been secured from every participant or their respective legal guardians. AD was identified using the NINCDS-ADRDA diagnostic framework [17], and MCI was determined according to standard neuropsychological criteria [18]. Individuals classified as healthy controls (HC) were defined as individuals with no evidence of MCI or dementia, a Clinical Dementia Rating (CDR) of 0, and no previous affective or cognitive disorders. Cognitive normality was additionally verified using education-adjusted Mini-Mental State Examination (MMSE) cutoffs: scores of ≥17 for illiterate participants, ≥20 for primary education, and ≥24 for middle school education or above, considering both total and memory subscale scores [18, 19]. SCD was defined following current consensus criteria, requiring self- or informant-reported persistent cognitive concerns despite otherwise normal neuropsychological test performance [20]. A total of 380 participants were recruited, including healthy controls (HC), individuals with SCD, MCI, and AD. 26 participants were excluded due to incomplete MRI data, 20 unable to complete scales, 9 due to obvious brain lesions such as infarctions or tumors, and 12 due to excessive head movement, 2 was excluded due to hypothyroidism. Finally, 311 participants were included in this study (Supplementary Figure 1). To examine stage- specific effects, HC and SCD participants were combined into a cognitively unimpaired group (CU), while MCI and AD patients were grouped as cognitively impaired (CI), based on their objective cognitive performance.
Neuropsychological scale assessment
In this study, we used the Mini-Mental State Examination (MMSE) to assess participants’ overall cognitive function. MMSE is a 30-item screening tool used to evaluate cognitive abilities, including orientation, memory, attention, and language abilities. The total MMSE score represents the participant’s overall cognitive function. The Montreal Cognitive Assessment (MoCA) provides a quick evaluation of overall cognitive status, requiring roughly 10 min and scored on a 30-point scale, with lower values signifying greater impairment [21]. The assessment covers seven components: visuospatial/executive functions, naming, attention, orientation, language, abstraction, and delayed memory retrieval. The Shape Trail Test A (STT-A) assessed participants’ executive function. STT-A requires participants to connect numbers in ascending order by drawing lines. The time required to complete STT-A is considered an indicator of executive function. In the Rey Auditory Verbal Learning Test (RAVLT), participants listen to a list of 15 words repeated 5 times. After each presentation, participants are asked to recall the words (immediate recall and learning). After a 20-minute delay, participants are asked to recall these words again (delayed recall). We used long-term delayed free recall (AVLT-N5) and the total number of words recalled after 20 min (AVLT-N7) as indicators of memory function assessment. Participants’ language function was assessed using the semantic verbal fluency test (VFT) and Boston naming test (BNT). VFT requires participants to list as many animal names as possible within 60 s. We used the total number of animal names listed as an indicator of semantic fluency. BNT requires participants to name 30 objects/items printed in a book. To standardize across tests, all raw scores were Z-transformed. Composite scores for each cognitive domain were calculated by averaging Z-scores of tests belonging to the same domain: executive function (STT-A Z-score), memory (mean of AVLT-N5 and N7 Z-scores), and language (mean of VFT and BNT Z-scores).
MRI data acquisition
All participants underwent MRI examination using a 3.0 T MRI scanner (Prisma, Siemens) with a 64-channel head-neck receiver coil. The imaging protocol included three-dimensional magnetization prepared rapid acquisition gradient echo imaging (MPRAGE) sequence for anatomical imaging. We acquired routine images including T1-weighted imaging (T1WI), T2WI, and T2-FLAIR to exclude obvious brain lesions. Specific scanning parameters were as follows: (1) T1WI: TI = 900 ms; echo time (TE) = 17.0 ms; repetition time (TR) = 2000 ms; number of slices = 20; field of view (FOV) = 240 × 240 mm²; slice thickness = 6 mm, scan time 1 min 04 s. (2) T2-FLAIR: TI = 2500 ms; TE = 84.0 ms; TR = 9000 ms; number of slices = 20; FOV = 240 × 240 mm²; slice thickness = 6 mm; Bandwidth = 289 Hz/Px, scan time 1 min 48 s. (3) T2WI: flip angle = 150°; TE = 99.0 ms; TR = 6000 ms; number of slices = 20; FOV = 240 × 240 mm²; slice thickness = 6 mm; Bandwidth = 289 Hz/Px, scan time 1 min 20 s. (4) MPRAGE: TE = 2.26 ms; TR = 2300 ms; inversion time = 900 ms; flip angle = 8°; slice thickness = 1 mm; FOV = 256 × 256 mm²; voxel size = 1 × 1 × 1 mm³.
Plasma biomarker analysis
We collected 2 mL of venous blood in tubes containing ethylenediaminetetraacetic acid (EDTA) and obtained plasma supernatant after centrifuging the samples at 2500 g/min for 15 min at 4 °C. All plasma samples were stored at −80 °C and thawed immediately on ice before analysis. Plasma Aβ, GFAP levels and NFL were measured using the single molecule array (Simoa™) platform (Quanterix Corporation, Billerica, Massachusetts, USA), following the manufacturer’s instructions strictly. Simoa detection used the following commercial kits: β-amyloid (1–42), β-amyloid (1–40), GFAP and NFL. All detections were performed in duplicate, reporting mean concentration values. For samples with concentrations below the lower limit of quantification, concentrations were set to half the lower limit of quantification.
White matter hyperintensity segmentation
White matter hyperintensity segmentation was completed using United Imaging software, based on a convolutional neural network called 2D VB-Net [22]. We input T1WI, T2WI, and T2-FLAIR images because clinically, these three scanning sequences are usually used together to determine the accurate extent of WMH and distinguish them from other brain lesions such as lacunar infarcts, periventricular spaces, and large infarcts. Kim et al. subdivided WMH into four categories based on their distance from ventricles, including juxWMH, pWMH, dWMH, and jcWMH [12]. juxWMH is defined as the region within 3 millimeters of the ventricles, pWMH ranges from 3 to 13 millimeters; jcWMH refers to WMH within 4 millimeters of the cortical-medullary junction, while dWMH is defined as the region between pWMH and jcWMH [12]. In our system, we adopted this WMH subclassification, allowing us to obtain their specific volumes as additional features. The specific analysis is as follows: we first used the 2D VB-Net structure to segment ventricles from MRI images, then constructed a distance map where voxels in ventricles have a distance of 0, and all other brain voxels are assigned values based on their nearest distance to any ventricular voxel. Based on this distance map, WMH throughout the brain were further subdivided into these four categories [22]. The 2D VB-Net segmentation algorithm we employed has been extensively validated in its original publication, where it demonstrated robust performance (Dice = 0.782) on a silver standard dataset labeled by five experienced neuroradiologists and showed excellent correlation with Fazekas visual rating scores, confirming its reliability for implementing Kim’s WMH classification system. To make WMH distribution more conform to normal distribution, we performed logarithmic (log₁₀) transformation on all WMH to obtain log-transformed juxtaventricular WMH (lgjuxWMH), log-transformed periventricular WMH (lgpWMH), log-transformed deep WMH (lgdWMH), and log-transformed juxtacortical WMH (lgjcWMH) for normalization.
Statistical analysis
Statistical analysis was performed using SPSS 19.0 (SPSS Inc., Chicago) and R software version 3.5.2 (R Core Team, www.R-project.org). For categorical data such as gender and APOE4 carrier status, chi-square tests were used for analysis. For continuous variables, the Kolmogorov-Smirnov test was first used to test whether continuous variables conformed to normal distribution. Data satisfying normal distribution were expressed as mean ± standard deviation, and categorical variables were expressed as numbers or percentages. Linear regression models were used to study relationships between Aβ42/Aβ40 and white matter hyperintensities and cognitive function, with gender, age, education level, and APOE4 carrier status as covariates for regression. Sensitivity analyses were performed by additionally adjusting for cardiovascular risk factors (hypertension, diabetes, hyperlipidemia) in all partial correlation models. In all models, plasma biomarkers (Aβ42/Aβ40) were entered as predictors, whereas WMH measures (lgjuxWMH, lgpWMH, lgdWMH, lgjcWMH) or cognitive outcomes (MMSE, MoCA, memory, executive, and language composite scores) were specified as dependent variables. Amos Graphics 24.0 software (SPSS Inc., Chicago) was used to construct structural equation models to evaluate relationships between plasma indicators, white matter hyperintensities in different regions, and memory, executive, and language functions. The PROCESS plugin in SPSS 19.0 (SPSS Inc., Chicago) was used for mediation analysis. Group comparisons were corrected using Bonferroni adjustment. Multiple correlations and mediation pathways were corrected using the Benjamini-Hochberg FDR procedure.
Results
Demographic characteristics
Table 1 shows characteristics of participants in each group. The four groups showed no differences in sex, APOE4 carrier status, hypertension and diabetes (all P > 0.05). Hyperlipidemia prevalence differed significantly across diagnostic groups (P < 0.001), with the highest prevalence in the SCD group. The ANOVA analysis revealed that the four groups showed significant differences in age. However, the post hocanalysis did not show any differences regarding age after Bonferroni correction. Education levels in MCI and AD groups were lower than HC and SCD groups (All P < 0.001, Bonferroni corrected). MMSE scores in MCI and AD groups were significantly lower than HC (P = 0.003; P < 0.001) and SCD (P < 0.001; P < 0.001; Bonferroni corrected) groups, and AD group had lower MMSE scores than MCI group (P < 0.001, Bonferroni corrected). MoCA scores in MCI and AD groups were significantly lower than HC (All P < 0.001) and SCD (P < 0.001; P < 0.001; Bonferroni corrected) groups, and AD group had lower MoCA scores than MCI group (P < 0.001, Bonferroni corrected).
Plasma Aβ42/Aβ40 levels in AD patients were significantly lower than HC, SCD and MCI groups (P < 0.001; P < 0.001; P = 0.007; Bonferroni corrected). There were significant differences in Aβ42 levels among the four groups, with AD group significantly lower than SCD group (P = 0.002). GFAP in AD group was significantly higher than HC, SCD, and MCI groups (P < 0.001; P < 0.001; P = 0.001, Bonferroni corrected). NFL in AD group was significantly higher than HC, SCD, and MCI groups (All P < 0.001, Bonferroni corrected), and the MCI group also showed higher NFL levels than HC (P = 0.004, Bonferroni corrected) (Supplementary Figure 2). Compared to HC and SCD groups, MCI group showed significantly reduced memory function (all P < 0.001, Bonferroni corrected); compared to HC, SCD, and MCI groups, AD group showed significantly reduced memory function (all P < 0.001, Bonferroni corrected). Compared to HC and SCD group, MCI group performed worse in executive function (P = 0.001; P < 0.001; Bonferroni corrected). AD group performed worse in executive function than HC, SCD, and MCI groups (P < 0.001; P < 0.001; P < 0.001; Bonferroni corrected). Compared to HC and SCD groups, MCI group showed significantly reduced language function (all P < 0.001, Bonferroni corrected); compared to HC, SCD, and MCI groups, AD group showed significantly reduced language function (all P < 0.001, Bonferroni corrected). These overall and cognitive domain intergroup differences remained significant after removing the covariate of education level. eTable 1 shows the demographic and clinical characteristics of participants for the subgroup. CI participants were older and had lower education levels compared with CU individuals. The CI group also showed significantly lower Aβ42 and Aβ42/Aβ40 levels, as well as elevated GFAP and NFL concentrations. Consistent with these biomarker differences, CI participants exhibited markedly poorer global cognition and lower executive, memory, and language performance relative to the CU group.
Intergroup white matter hyperintensity differences
Figure 1 shows intergroup differences in white matter hyperintensities. Compared to HC, SCD, and MCI, AD group showed elevated lgjuxWMH (all P < 0.05, Bonferroni corrected), and MCI group showed elevated lgjuxWMH compared to HC and SCD group. Compared to HC, SCD and MCI group, AD group showed elevated lgpWMH (P < 0.001; P < 0.001; P = 0.001; Bonferroni corrected). There were no statistical differences in lgdWMH among the four groups. Compared to HC and SCD, AD group showed elevated lgjcWMH ((P < 0.001; P < 0.001; P = 0.004; Bonferroni corrected). All group differences in lgjuxWMH, lgpWMH, and lgjcWMH remained significant after additional adjustment for hypertension, diabetes, and hyperlipidemia (all Bonferroni-corrected P < 0.05), except for the previously observed increase in lgjuxWMH in the MCI group compared with the HC and SCD groups, which was no longer significant after covariate adjustment
Compared to HC, SCD, and MCI, AD group showed elevated juxWMH, and MCI group showed elevated lgjuxWMH compared to HC and SCD group (A). Compared to HC, SCD and MCI group, AD group showed elevated lgpWMH (B). There were no statistical differences in lgdWMH among the four groups (C). Compared to HC, SCD and MCI group, AD group showed elevated lgjcWMH (D). Abbreviations: AD, Alzheimer’s disease; dWMH, deep white matter hyperintensities; HC, healthy controls; MCI, mild cognitive impairment; jcWMH, juxtacortical white matter hyperintensities; juxWMH, juxtaventricular white matter hyperintensities; pWMH, periventricular white matter hyperintensities; SCD, subjective cognitive decline; lg represents log10 transformation of white matter hyperintensities.
Association between Aβ42/Aβ40 and cognitive function
After controlling for gender, age, education levels, lower Aβ42/Aβ40 ratios—reflecting greater amyloid pathology—were associated with poorer global cognition and domain-specific performance, including MMSE, MoCA, memory, executive, and language functions (Table 2). After including APOE4 as additional covariates, lower Aβ42/Aβ40 remained significantly related to worse executive and language function. For the cognitively unimpaired individuals, lower Aβ42/Aβ40 showed poorer MoCA and language functions. For the cognitively impaired patients, lower Aβ42/Aβ40 was demonstrated worse MMSE, MoCA and language functions.
Association between plasma biomarkers and white matter hyperintensities
After controlling for gender, age, education level, and APOE4 carrier status, higher amyloid pathology, indicated by lower Aβ42/Aβ40 levels were associated with greater WMH burden, including lgjuxWMH, lgpWMH, and lgjcWMH (Table 3). The subgroup results also showed similar trends. Partial correlation analysis showed that Aβ42/Aβ40 levels were negatively correlated with lgjuxWMH, lgpWMH, and lgjcWMH (r = −0.29, P < 0.001; r = −0.26, P < 0.001; r = −0.25, P < 0.001) after adjusting sex, age and educational levels. Aβ42 levels were negatively with lgjuxWMH, lgpWMH, and lgjcWMH (r = −0.15, P = 0.011; r = −0.13, P = 0.022; r = −0.16, P = 0.007), and Aβ40 levels were positively correlated with lgjuxWMH, and lgpWMH (r = 0.14, P = 0.019; r = 0.12, P = 0.040). Similarly, higher GFAP and NFL concentrations—reflecting greater astrocytic activation and axonal injury, were associated with higher lgjuxWMH and lgpWMH volumes (r = 0.22, P < 0.001; r = 0.19, P = 0.001; r = 0.22, P < 0.001) (Fig. 2). After FDR correction for multiple comparisons within each biomarker, all correlations remained significant except for Aβ40 associations.
Partial correlation analysis showed that Aβ42/Aβ40 levels were negatively correlated with lgjuxWMH, lgpWMH, and lgjcWMH (r = −0.29, P < 0.001; r = −0.26, P < 0.001; r = - 0.25, P < 0.001) (A–C)after adjusting sex, age and educational levels. Aβ42 levels were negatively with lgjuxWMH, lgpWMH, and lgjcWMH (r = −0.15, P = 0.011; r = −0.13, P = 0.022; r = −0.16, P = 0.007) (D–F), and Aβ40 levels were positively correlated with lgjuxWMH, and lgpWMH (r = 0.14, P = 0.019; r = 0.12, P = 0.040) (G–H). GFAP was positively correlated with lgjuxWMH volume (r = 0.22, P < 0.001) (I), and NFL levels were positively correlated with lgjuxWMH and lgpWMH volumes (r = 0.19, P = 0.001; r = 0.22, P < 0.001) (J–K). Abbreviations: AD, Alzheimer’s disease; dWMH, deep white matter hyperintensities; GFAP, glial fibrillary acidic protein; HC, healthy controls; jcWMH, subcortical white matter hyperintensities; juxWMH, juxtaventricular white matter hyperintensities; MCI, mild cognitive impairment; NFL, neurofilament light chain; pWMH, periventricular white matter hyperintensities; SCD, subjective cognitive decline. lg represents log10 transformation of white matter hyperintensities.
Association between white matter hyperintensities and global cognitive function
Figure 3 shows the relationship between white matter hyperintensities in different regions and overall cognitive function. After adjusting for gender, age, and education level, lgjuxWMH, lgpWMH, and lgjcWMH volumes showed significant negative correlations with MMSE (r = −0.21, P < 0.001; r = −0.18, P = 0.002; r = −0.21, P < 0.001, respectively) and MoCA scores (r = −0.32, P < 0.001; r = −0.25, P < 0.001; r = −0.19, P = 0.001, respectively). After FDR correction within each cognitive test, all correlations remained significant
After adjusting for gender, age, and education level, lgjuxWMH, lgpWMH, and lgjcWMH volumes showed significant negative correlations with MMSE (r = −0.21, P < 0.001; r = −0.18, P = 0.002; r = −0.21, P < 0.001, respectively) (A–C) and MoCA scores (r = −0.32, P < 0.001; r = −0.25, P < 0.001; r = −0.19, P = 0.001, respectively) (D–F). Abbreviations: AD, Alzheimer’s disease; dWMH, deep white matter hyperintensities; HC, healthy controls; jcWMH, subcortical white matter hyperintensities; juxWMH, juxtaventricular white matter hyperintensities; MCI, mild cognitive impairment; pWMH, periventricular white matter hyperintensities; SCD, subjective cognitive decline; lg represents log10 transformation of white matter hyperintensities.
Association between white matter hyperintensities and cognitive function
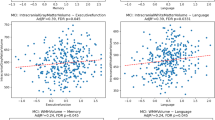
Figure 4 shows the relationship between white matter hyperintensities in different regions and cognitive function. After controlling for sex, age, and education, greater WMH burden was associated with worse cognitive performance across multiple domains. Higher lgjuxWMH volume was associated with worse memory and language functions (r = −0.16, P = 0.006; r = −0.34, P < 0.001) and longer executive function completion time (r = 0.18, P = 0.003). Higher lgpWMH volume was associated with worse memory, language functions (r = −0.13, P = 0.032; r = −0.24, P < 0.001) and executive function (r = 0.19, P = 0.002). Higher lgjcWMH volume was associated with worse with memory and language functions (r = −0.15, P = 0.011; r = −0.29, P < 0.001), while no significant association was found between lgjcWMH volume and executive function (r = 0.09, P = 0.123). All significant correlations survived FDR correction.
LgjuxWMH volume was associated with memory, executive, and language functions (r = −0.16, P = 0.006; r = 0.18, P = 0.003; r = −0.34, P < 0.001) (A–C). LgpWMH volume was negatively correlated with memory and language function (r = −0.13, P = 0.032; r = −0.24, P < 0.001) (D, F), but positively correlated with executive function (r = 0.19, P = 0.002) (E). LgjcWMH volume was negatively correlated with memory and language function (r = -0.15, P = 0.011; r = −0.29, P < 0.001) (G, I), but no significant association was found between lgjcWMH volume and executive function (r = 0.09, P = 0.123) (H). Abbreviations: AD, Alzheimer’s disease; dWMH, deep white matter hyperintensities; HC: healthy controls; jcWMH, subcortical white matter hyperintensities; juxWMH, juxtaventricular white matter hyperintensities; MCI, mild cognitive impairment; pWMH, periventricular white matter hyperintensities; SCD, subjective cognitive decline; lg represents log10 transformation of white matter hyperintensities.
All associations remained largely unchanged after adjusting for hypertension, diabetes, and hyperlipidemia (see Supplementary Material: Supplementary text).
Path Analysis
juxWMH volume mediated the effect of plasma Aβ42/Aβ40 on cognitive function (Fig. 5; Table 4). As the direct pathway from plasma Aβ42/Aβ40 to cognitive function was not statistically significant, juxWMH may serve as a mediator linking Aβ42/Aβ40 to cognitive function. Table 4 presents the following analysis results: path coefficients, model fit indices, and indirect effect values.
This figure describes the path analysis diagram of Aβ42/Aβ40. In the model, Aβ42/Aβ40 serves as the predictor variable, juxWMH, pWMH, and jcWMH serve as mediating variables. Memory, language, and executive functions are considered outcome variables. Numbers on paths represent statistically significant standardized coefficients. Abbreviations: dWMH, deep white matter hyperintensities; jcWMH, juxtacortical white matter hyperintensities; juxWMH, juxtaventricular white matter hyperintensities; pWMH, periventricular white matter hyperintensities.
Subgroup mediation analysis
Mediation analyses were conducted using bootstrapping (5,000 iterations) with age, sex, and education as covariates. After FDR correction, seven pathways remained significant, all showing partial mediation (Fig. 6). In cognitively unimpaired individuals, juxtaventricular WMH mediated Aβ42/Aβ40 associations with MoCA (indirect: β = 0.06, P = 0.047, 22.5%) and language (indirect: β = 0.07, P = 0.025, 20.4%) (Fig. 6A, B). In cognitively impaired individuals, five pathways showed mediation: Aβ42/Aβ40 through juxtaventricular WMH to MoCA (indirect: β = 0.06, P = 0.013, 22.2%) and language (indirect: β = 0.06, P = 0.032, 25.8%); GFAP through juxWMH to MoCA (indirect: β = −0.05, P = 0.013, 15.8%) and language (indirect: β = −0.05, P = 0.041, 17.6%); Aβ42/Aβ40 through periventricular WMH to MoCA (indirect: β = 0.04, P = 0.048, 15.0%) (Fig. 6C–G).Direct effects remained significant in all pathways (P < 0.05), indicating partial mediation with WMH accounting for 15–26% of biomarker-cognition associations.
In cognitively unimpaired individuals, juxtaventricular WMH partially mediated associations between plasma Aβ42/Aβ40 and MoCA and language function (A–B). (C–G) In cognitively impaired individuals, juxtaventricular WMH partially mediated associations between Aβ42/Aβ40 and MoCA (C) and language (D), and between GFAP and MoCA (E) and language (F). Periventricular WMH partially mediated the association between Aβ42/Aβ40 and MoCA (G). ***P < 0.001, **P < 0.01, and *P < 0.05 with FDR correction Bootstrapping with 5,000 iterations. GFAP, glial fibrillary acidic protein; lgjuxWMH, log-transformed juxtaventricular white matter hyperintensities; lgpWMH, log-transformed periventricular white matter hyperintensities. MoCA, Montreal Cognitive Assessment.
Discussion
This study aimed to explore the associations between plasma biomarkers, different regional WMH volumes at different anatomical locations, and cognitive function. The results showed that lower plasma Aβ42/Aβ40 ratios were associated with higher volumes of juxWMH, pWMH, and jcWMH. Additionally, white matter hyperintensities in various regions showed significant correlations with cognitive function. Further mediation analysis revealed differential patterns across cognitive stages: in CU individuals, juxWMH and pWMH partially mediated associations between plasma Aβ42/Aβ40 and cognitive function, while in CI patients, mediation patterns expanded to include neuroinflammatory and neurodegeneration pathways across multiple anatomically-defined WMH categories.
Previous studies reported that overall WMH burden in AD patients is usually higher than in healthy populations [4, 5], but systematic studies on regional-specific characteristics of WMH are still lacking. Some research found that AD patients are more prone to posterior WMH accumulation [6, 8], which may have potential predictive value for sporadic AD [23]. In this study, we classified WMH into three anatomical locations: juxWMH, pWMH, and jcWMH for the first time, and quantitatively analyzed their volumes using automated methods. Results showed that these three regional WMH gradually increased in volume during progression from HC to AD, showing a clear gradient change trend. Several hypotheses have been proposed for the mechanisms of increased white matter hyperintensity volume in AD patients and its relationship with cognitive impairment. The “additive effect” or “interactive effect” theory suggests that WMH, as a type of cerebral small vessel disease, may produce synergistic or cumulative effects with AD neuropathology, accelerating the appearance and development of clinical symptoms. Alternatively, WMH in AD may reflect specific non-vascular mechanisms, such as neurodegenerative changes induced by AD-related pathology [24, 25].
Beyond regional patterns of WMH accumulation, our study reveals location- specific relationships between WMH subtypes and different cognitive domains. Numerous studies have confirmed that white matter hyperintensity burden is closely related to memory decline and executive dysfunction in elderly individuals [26]. However, research on whether WMH in different anatomical locations have distinct effects on various cognitive functions remains scarce. Our study further found that juxWMH, pWMH, and jcWMH were all significantly associated with memory, executive function, and language abilities, whereas dWMH showed no significant association, suggesting that different location-specific WMH may represent heterogeneous pathological mechanisms. While previous studies mostly focused on overall WMH [26], few studies have deeply explored the effects of each location-specific WMH on distinct cognitive domains. For example, WMH located in frontal and prefrontal regions are more likely to affect executive function [27], while WMH located in deep frontal or occipital regions are more likely to affect memory abilities [28]. Our study additionally demonstrates, for the first time, an association between WMH and language function, suggesting that WMH may affect broader cognitive domains beyond memory and executive function.
We also found that lower plasma Aβ42/Aβ40 levels were associated with higher juxWMH, pWMH, and jcWMH volumes, even after adjusting for gender, age, education years, and APOE4 gene carrier status, suggesting strong independence. Previous studies have shown that both CSF Aβ content [29] and amyloid deposition on PET [30] are associated with WMH, particularly in the splenium of the corpus callosum. Cerebral amyloid angiopathy may partially explain increased WMH in AD [30, 31], although some evidence suggests that this association is not fully mediated by cerebral amyloid angiopathy [32]. Plasma tau protein levels also correlate positively with WMH volume, especially in AD populations [33], suggesting that WMH may also participate in neurodegenerative processes by promoting tau protein deposition beyond amyloid pathology. Interestingly, while plasma Aβ42/Aβ40-WMH associations remained significant after APOE4 adjustment, the Aβ42/Aβ40-memory relationship was attenuated while executive and language associations persisted. This differential pattern suggests APOE4 may specifically influence amyloid-memory pathways. In summary, our results support the existence of certain biological connections between plasma Aβ levels and multiple regional WMH volumes. Future research should further explore the spatial heterogeneity relationships between amyloid protein and tau deposition in different brain regions and cognitive impairment.
A central finding of our study is the mediating role of lgjuxWMH between plasma biomarkers and cognitive function. Notably, we observed that juxWMH plays a mediating role between plasma Aβ42/Aβ40 and cognitive function. juxWMH specifically refers to white matter hyperintensity regions within 3 millimeters of the ventricular wall, usually considered related to cerebrospinal fluid leakage, reflecting a type of non-ischemic white matter lesion [12]. This finding suggests that in the cognitive impairment process of AD, non-ischemic mechanisms may play key roles, and juxWMH may constitute a type of AD-specific pathological marker. Previous studies indicated that destruction of ependymal cell structure may cause cerebrospinal fluid leakage, thereby affecting the structural stability of adjacent brain tissue and penetrating arteries, and may cause blood-brain barrier dysfunction. Increased blood-brain barrier permeability is considered one of the important mechanisms of AD occurrence [34], and juxWMH may be an early manifestation of this mechanism.
Importantly, this study reveals differential mediating patterns of WMH across cognitive stages, with important pathophysiological implications. In cognitively unimpaired individuals, only juxtaventricular and periventricular WMH partially mediated associations between plasma Aβ42/Aβ40 and cognitive functions, suggesting early-stage amyloid pathology primarily affects cognition through specific white matter locations. In CI patients, mediation involved multiple location-specific WMH and incorporated neuroinflammatory markers, indicating a transition from pure amyloid- mediated effects to synergistic multi-pathological mechanisms. This transition from pure amyloid-mediated effects to synergistic multi-pathological mechanisms likely reflects the pathological cascade of AD, where early amyloid deposition triggers secondary neuroinflammation and axonal damage, forming a vicious cycle that accelerates cognitive decline. These findings provide crucial insights for understanding AD pathological progression and developing stage-specific therapeutic interventions. These findings have important clinical implications: combining plasma biomarkers with location-specific WMH assessment may improve early detection and risk stratification in AD, while stage-dependent mediation patterns suggest that interventions may need to target amyloid and vascular pathways in preclinical stages and expand to anti-neuroinflammatory approaches in symptomatic patients.
This study has some limitations. First, as a cross-sectional observational study, we cannot dynamically track the evolution process of WMH in AD progression, limiting causal inference. Second, although we adjusted for major cardiovascular risk factors and our findings remained robust, other contributors (e.g.,detailed lipid measures) were not assessed, and cardiovascular risk factors were coded only as binary variables. Third, this study sample consists entirely of Asian populations, and limitations in racial and genetic background may affect the broad applicability of research results. Fourth, limited by sample size, this study has limited statistical power, and future studies should expand sample sizes to improve research robustness and generalizability. Finally, this study did not include PET imaging or cerebrospinal fluid biomarker detection, and therefore could not further validate the direct relationship between WMH and brain Aβ and tau burden. Future longitudinal studies incorporating PET and CSF measures benefit from advanced machine-learning frameworks such as large language models-assisted planning or graph-based deviation modeling [35, 36] to refine WMH subtyping and capture subtle, stage-dependent spatial patterns.
Conclusion
This study explored associations between plasma biomarkers, reginal WMH volumes, and cognitive function across the AD spectrum. We found differential mediation patterns across cognitive stages: in cognitively unimpaired individuals, juxWMH and pWMH primarily mediated amyloid-related cognitive effects, while in cognitively impaired patients, mediation expanded to include neuroinflammatory and neurodegeneration pathways across multiple reginal WMH volumes. This suggests a pathological cascade from early amyloid-driven changes to multi-factorial processes. JuxWMH serves as an important “bridging” marker linking plasma biomarkers to cognitive decline in a stage-dependent manner, supporting the potential of combined plasma and imaging biomarkers for stratified intervention in AD.
Data availability
The data that support the findings of this study are available on request from the corresponding author when the appropriate data sharing agreements are consented.
References
Cox SR, Lyall DM, Ritchie SJ, Bastin ME, Harris MA, Buchanan CR, et al. Associations between vascular risk factors and brain MRI indices in UK Biobank. Eur Heart J. 2019;40:2290–300.
Kloppenborg RP, Nederkoorn PJ, Geerlings MI, van den Berg E. Presence and progression of white matter hyperintensities and cognition: a meta-analysis. Neurology. 2014;82:2127–38.
Bolandzadeh N, Davis JC, Tam R, Handy TC, Liu-Ambrose T. The association between cognitive function and white matter lesion location in older adults: a systematic review. BMC Neurol. 2012;12:126.
Wang YL, Chen W, Cai WJ, Hu H, Xu W, Wang ZT, et al. Associations of white matter hyperintensities with cognitive decline: A longitudinal study. J Alzheimers Dis. 2020;73:759–68.
Damulina A, Pirpamer L, Seiler S, Benke T, Dal-Bianco P, Ransmayr G, et al. White matter hyperintensities in Alzheimer’s disease: A lesion probability mapping study. J Alzheimers Dis. 2019;68:789–96.
Yoshita M, Fletcher E, Harvey D, Ortega M, Martinez O, Mungas DM, et al. Extent and distribution of white matter hyperintensities in normal aging, MCI, and AD. Neurology. 2006;67:2192–8.
Lorenzini L, Ansems LT, Lopes Alves I, Ingala S, Vállez García D, Tomassen J, et al. Regional associations of white matter hyperintensities and early cortical amyloid pathology. Brain Commun. 2022;4:fcac150.
Pålhaugen L, Sudre CH, Tecelao S, Nakling A, Almdahl IS, Kalheim LF, et al. Brain amyloid and vascular risk are related to distinct white matter hyperintensity patterns. J Cereb Blood Flow Metab. 2021;41:1162–74.
Soriano-Raya JJ, Miralbell J, López-Cancio E, Bargalló N, Arenillas JF, Barrios M, et al. Deep versus periventricular white matter lesions and cognitive function in a community sample of middle-aged participants. J Int Neuropsychol Soc. 2012;18:874–85.
Silbert LC, Nelson C, Howieson DB, Moore MM, Kaye JA. Impact of white matter hyperintensity volume progression on rate of cognitive and motor decline. Neurology. 2008;71:108–13.
Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol. 1987;149:351–6.
Kim KW, MacFall JR, Payne ME. Classification of white matter lesions on magnetic resonance imaging in elderly persons. Biol Psychiatry. 2008;64:273–80.
Li RR, He YS, Liu M, Nie ZY, Huang LH, Lu Z, et al. Analysis of correlation between cerebral perfusion and KIM score of white matter lesions in patients with Alzheimer’s disease. Neuropsychiatr Dis Treat. 2019;15:2705–14.
Marnane M, Al-Jawadi OO, Mortazavi S, Pogorzelec KJ, Wang BW, Feldman HH, et al. Periventricular hyperintensities are associated with elevated cerebral amyloid. Neurology. 2016;86:535–43.
Osborn KE, Liu D, Samuels LR, Moore EE, Cambronero FE, Acosta LMY, et al. Cerebrospinal fluid β-amyloid(42) and neurofilament light relate to white matter hyperintensities. Neurobiol Aging. 2018;68:18–25.
Teunissen CE, Verberk IMW, Thijssen EH, Vermunt L, Hansson O, Zetterberg H, et al. Blood-based biomarkers for Alzheimer’s disease: Towards clinical implementation. Lancet Neurol. 2022;21:66–77.
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr., Kawas CH, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7:263–9.
Bondi MW, Edmonds EC, Jak AJ, Clark LR, Delano-Wood L, McDonald CR, et al. Neuropsychological criteria for mild cognitive impairment improves diagnostic precision, biomarker associations, and progression rates. J Alzheimers Dis. 2014;42:275–89.
Albert MS, DeKosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7:270–9.
Jessen F, Amariglio RE, Buckley RF, van der Flier WM, Han Y, Molinuevo JL, et al. The characterisation of subjective cognitive decline. Lancet Neurol. 2020;19:271–8.
Roalf DR, Moore TM, Mechanic-Hamilton D, Wolk DA, Arnold SE, Weintraub DA, et al. Bridging cognitive screening tests in neurologic disorders: A crosswalk between the short Montreal Cognitive Assessment and Mini-Mental State Examination. Alzheimers Dement. 2017;13:947–52.
Zhu W, Huang H, Zhou Y, Shi F, Shen H, Chen R, et al. Automatic segmentation of white matter hyperintensities in routine clinical brain MRI by 2D VB-Net: A large-scale study. Front Aging Neurosci. 2022;14:915009.
Brickman AM, Zahodne LB, Guzman VA, Narkhede A, Meier IB, Griffith EY, et al. Reconsidering harbingers of dementia: progression of parietal lobe white matter hyperintensities predicts Alzheimer’s disease incidence. Neurobiol Aging. 2015;36:27–32.
Garnier-Crussard A, Cotton F, Krolak-Salmon P, Chételat G. White matter hyperintensities in Alzheimer’s disease: Beyond vascular contribution. Alzheimers Dement. 2023;19:3738–48.
Garnier-Crussard A, Bougacha S, Wirth M, Dautricourt S, Sherif S, Landeau B, et al. White matter hyperintensity topography in Alzheimer’s disease and links to cognition. Alzheimers Dement. 2022;18:422–33.
Puzo C, Labriola C, Sugarman MA, Tripodis Y, Martin B, Palmisano JN, et al. Independent effects of white matter hyperintensities on cognitive, neuropsychiatric, and functional decline: a longitudinal investigation using the National Alzheimer’s Coordinating Center Uniform Data Set. Alzheimers Res Ther. 2019;11:64.
Tullberg M, Fletcher E, DeCarli C, Mungas D, Reed BR, Harvey DJ, et al. White matter lesions impair frontal lobe function regardless of their location. Neurology. 2004;63:246–53.
Brugulat-Serrat A, Salvadó G, Sudre CH, Grau-Rivera O, Suárez-Calvet M, Falcon C, et al. Patterns of white matter hyperintensities associated with cognition in middle-aged cognitively healthy individuals. Brain Imaging Behav. 2020;14:2012–23.
Walsh P, Sudre CH, Fiford CM, Ryan NS, Lashley T, Frost C, et al. CSF amyloid is a consistent predictor of white matter hyperintensities across the disease course from aging to Alzheimer’s disease. Neurobiol Aging. 2020;91:5–14.
Graff-Radford J, Arenaza-Urquijo EM, Knopman DS, Schwarz CG, Brown RD, Rabinstein AA, et al. White matter hyperintensities: relationship to amyloid and tau burden. Brain. 2019;142:2483–91.
Greenberg SM, Charidimou A. Diagnosis of cerebral amyloid angiopathy: Evolution of the boston criteria. Stroke. 2018;49:491–7.
Lee S, Zimmerman ME, Narkhede A, Nasrabady SE, Tosto G, Meier IB, et al. White matter hyperintensities and the mediating role of cerebral amyloid angiopathy in dominantly-inherited Alzheimer’s disease. PLoS One. 2018;13:e0195838.
Laing KK, Simoes S, Baena-Caldas GP, Lao PJ, Kothiya M, Igwe KC, et al. Cerebrovascular disease promotes tau pathology in Alzheimer’s disease. Brain Commun. 2020;2:fcaa132.
Pantoni L, Garcia JH. Pathogenesis of leukoaraiosis: a review. Stroke. 1997;28:652–9.
Zhang Y, Wang C, Qi J, Peng Y. Leave it to large language models! correction and planning with memory integration. Cyborg Bionic Syst. 2024;5:0087.
Mencattini A, Curci G, Riccardi A, Casti P, D’Orazio M, Filippi J, et al. MEA-Based graph deviation network for early autism syndrome signatures in human forebrain organoids. Cyborg Bionic Syst. 2025;6:0441.
Funding
This work was supported in part by the Key Science and Technology Project of Hainan Province (ZDYF2024SHFZ058, ZDYF2023SHFZ096), the National Natural Science Foundation of China (82572211, 82271977, 82160327), the Hainan Academician Innovation Platform Scientific Research Project (YSPTZX202514), the Hainan Province ‘Nanhai Rising Star’Healthcare Talent Platform Project (NHXXWJW-2023003), the Hainan Provincial Natural Science Foundation of China (825RC870), the Hainan Province Clinical Medical Center, and the Innovation Platform for Academicians of Hainan Province.
Author information
Authors and Affiliations
Contributions
HC, FC: conceptualization. HC, YG, WH, FC: study design. HC, YG: imaging and data processing. HC, YG, WH, KZ, TL, FC: analysis and statistics. HC, FC: original draft. HC, YG, WH, KZ, TL, FC review and editing. All authors contributed to the article and approved of the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors have no conflict of interest to declare.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Hainan General Hospital in accordance with the Declaration of Helsinki. All participants and/or their respective Legally Authorized Representative (when applicable) provided their written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, H.J., Guo, Y., Huang, W. et al. Stage-Dependent mediation of white matter hyperintensities between plasma biomarkers and cognitive function in Alzheimer’s disease. Transl Psychiatry 16, 140 (2026). https://doi.org/10.1038/s41398-026-03927-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-026-03927-5