Abstract
Bone marrow adipocytes are known to have a critical role within the bone marrow niche. However, our understanding of bone marrow adipose tissue expansion with obesity and the role it plays in immune cell regulation and osteoclastogenesis is limited. Here, we showed the expansion of bone marrow adipocytes promoted osteoclast differentiation and subsequently led to obesity-related trabecular and cortical bone loss through a stimulatory effect of the PD-1/PD-L1 axis. Bone marrow adipocytes isolated from obese mice had increased Mcp-1 expression, a key regulator of osteoclastogenesis and myeloid cell accumulation. With the increase in bone marrow adipose tissue-derived Mcp-1, we found an increase in the number of PD-L1+ myeloid cells. While these cells inhibited activated T-cells, we found evidence of a stimulatory osteoclastogenic effect of PD-L1+ myeloid cells on PD-1-expressing osteoclast precursors. The inhibition of PD-1/PD-L1 signaling during early osteoclastogenesis prevented myeloid cell commitment and resulted in decreased cell fusion, supporting the role of PD-1/PD-L1 signaling in osteoclastogenesis. Using a bone marrow adipocyte depletion mouse model (BMAd-Pparg KO), we demonstrated that obese BMAd-Pparg KO mice had a reduced number of bone marrow PD-L1+ myeloid cells, accompanied by a decrease in PD-1+ osteoclast precursors. The reduction in these precursors resulted in fewer osteoclasts, subsequently leading to improved trabecular bone volume. Since osteoclasts are myeloid cell-derived, these results suggest that bone marrow adipocytes are critical for the commitment and differentiation of myeloid cells into osteoclasts. Targeting bone marrow adipogenesis could ameliorate enhanced osteoclastogenesis and provide a novel approach to treat obesity-related bone loss.

Obesity-induced expansion of BM adipocytes leads to PD-1/PD-L1-driven osteoclastogenesis and subsequent bone loss in obese, HFD-fed (OB-HFD) mice. After 12 weeks on a HFD, OB-HFD mice had a significant increase in BM adiposity and BMAT-derived Mcp-1 expression. The increase in BMAT-specific Mcp-1 expression was coupled with an increase in PD-1+ osteoclast (OC) precursors and PD-L1+ myeloid cells. In the context of obesity, the PD-1/PD-L1 axis has a stimulatory effect that enhances osteoclastogenesis and leads to trabecular and cortical bone loss. By depleting BM adipocytes with obesity, BMAT-derived Mcp-1 expression was decreased, as well as a decrease in PD-1+ OC precursors and PD-L1+ myeloid cells. This prevented obesity-related trabecular bone loss. Overall, this work demonstrated a strong correlation between BMAT expansion and PD-1/PD-L1-driven osteoclastogenesis as a mechanism for obesity-induced bone loss. (This image was created using BioRender).
Similar content being viewed by others
Introduction
Obesity is a major global health problem that affects one in eight adults 18 years and older.1 Obesity was initially thought to be advantageous for maintaining bone health due to greater bone mineral density observed in individuals who were overweight and obese.2,3 These observations were attributed to heavier individuals having greater weight-bearing or mechanical loading that stimulated bone-building cells.4,5 However, accumulating data suggest that obesity has a negative impact on bone health, resulting in increased obesity-related fractures due to compromised bone geometry and strength.3,6 In diet-induced obesity (DIO) murine models, the loss of trabecular bone has been attributed to increased osteoclastogenesis and osteoclast activity.2,7 However, the underlying biological mechanisms for these findings are not well understood.
DIO in rodents provides a useful model to interrogate the impact bone marrow (BM) adiposity has on trabecular and cortical bone volume.8,9 Bone marrow adipose tissue (BMAT), like white adipose tissue (WAT), expands with DIO. However, little is known about the role BMAT plays in the mechanisms of obesity-related bone loss. The notion that BMAT serves merely as an inert filler within the bone cavity has evolved over the last decade, and this tissue is now regarded as an essential secretory and metabolic organ.10,11,12 In addition to having local and systemic metabolic effects, the BM also functions as an immune regulatory organ.13 Immune cells that originate from the BM are found to infiltrate expanding WAT, where they play a crucial role in regulating metabolic functions and inflammation.14,15 WAT expansion with obesity leads to the recruitment and differentiation of monocytes to macrophages, the most prominent type of myeloid cell,16,17,18 emphasizing that the immune regulatory response within the BM is a critical element that may contribute to obesity-related peripheral inflammation and bone loss.
The innate and adaptive immune systems initiate immune activation to resolve inflammatory stimuli, but given the inflammatory nature of immune defenses, it is essential that immune activity is regulated.19,20 Failure to resolve acute immune responses has been implicated in perpetuating obesity-associated inflammation and peripheral tolerance (i.e., diminished peripheral immune response).21,22 Individuals with obesity have diminished immune cell activation as evidenced by higher rates of vaccine failure and greater complications from infection.21,23,24 In humans and rodent models, obesity is associated with an activated macrophage phenotype in multiple tissues, including the liver, adipose tissue, skeletal muscle, pancreatic islets, and the hypothalamus, which contributes to tissue inflammation and metabolic disease.25,26 Overactivation of innate immunity is caused by a dysregulation of the adaptive immune response. One way to regulate immune responses is through immune checkpoint proteins. Programmed death ligand-1 (PD-L1) is an immune checkpoint protein found on antigen-presenting cells, like macrophages and dendritic cells, which regulates the immune response by functioning as an immune response “brake”.27,28 PD-L1 expression regulates immune response when it interacts with the programmed death-1 (PD-1) receptor to promote the deactivation, functional exhaustion, and apoptosis of activated T-cells.29 However, elevated PD-L1 expression can lead to immunosuppression of the adaptive immune system. The increased macrophage phenotype may coincide with increased osteoclast activity, resulting in obesity-associated bone loss. Like macrophages, osteoclasts differentiate along the myeloid cell lineage.30 Taken together, the evidence suggests that obesity causes an increase in myelopoiesis and osteoclastogenesis, but the mechanism is unknown.
In this study, we aimed to investigate the role BMAT has on myelopoiesis and osteoclastogenesis. We hypothesized that the expansion of BMAT associated with DIO would have a negative impact on the BM microenvironment. Using a mouse model of high-fat diet (HFD)–induced obesity, we classified mice as obese based on pre-determined changes in body weight and fat mass. Obese mice showed a rapid increase in BM adiposity that altered the molecular phenotype of BMAT, which led to changes in immune cell function and skeletal homeostasis. Here, we report that in obese mice, the expansion in BMAT is directly associated with decreased trabecular and cortical bone volume through increased osteoclastogenesis by creating an immunosuppressive BM microenvironment through elevated PD-L1 signaling.
Results
Obesity parameters for HFD-fed male and female B6 mice
To characterize obesity in HFD-fed male and female mice, we examined age-related weight and fat mass changes in chow-diet-fed C57BL/6 J (B6) mice from 8 weeks to 20 weeks old (Fig. S1a–e). Based on the changes in body composition observed with aging, we considered male B6 mice after 4 weeks on a HFD (12 weeks old) to be obese if the change in body weight was >18% and the fold change in fat mass was >1.0 (where 1.0 equals 100%) while female mice had to have a change in weight >18% and a fold change in fat mass >0.85 (Fig. S1f). After 8 weeks on a HFD, male B6 mice (16 weeks old) were considered obese if the change in body weight was >38% and there was a fold change in fat mass >2.3, and female mice had to have a change in weight >28% and a fold change in fat mass >1.2 (Fig. S1f). To be considered obese after 12 weeks of HFD feeding (20 weeks old), male mice had to have a change in weight of >58% and a fold change in fat mass of >3.3. For female mice to be considered obese, there had to be a change in weight >38% and a fold change in fat mass >1.5 (Fig. S1f). It has been suggested that an underlying epigenetic mechanism established at birth causes varying changes in body weight and fat mass expansion in high-fat diet (HFD)-fed B6 mice.31,32 We anticipated that these pre-determined obesity parameters would remove possible epigenetic variation and allow us to investigate a mechanism for obesity-induced bone loss within a specific subset of HFD-fed mice.
Male B6 mice fed a HFD showed a significant increase in body weight compared to the LFD-fed mice after 2 weeks (Fig. S2a–c). By 4 weeks, obese, HFD-fed (OB-HFD) mice showed impaired glucose handling and mild insulin insensitivity (Fig. S2d, e). These metabolic changes were exacerbated after 8 weeks on the HFD, resulting in insulin resistance (Fig. S2f–j). After 12 weeks on a HFD, 34 out of 41 male B6 mice were considered obese based on our pre-determined obesity parameters, meaning 7 mice were considered non-obese, HFD-fed (NO-HFD) (Fig. 1a–c). We found there was a strong negative correlation between trabecular bone volume and changes in body weight (P = 3.6 × 10−6) as well as trabecular bone volume and the fold change in fat mass (P = 3.8 × 10−6) (Fig. 1d, e). Although NO-HFD males were considered non-obese based on our parameters, these mice weighed significantly more than the LFD-fed mice after 7 weeks on the HFD (Fig. S3a). Metabolically, OB-HFD and NO-HFD mice showed similar responses to glucose, and NO-HFD had a slight, but non-significant improvement in insulin handling (Fig. 1f, g). Circulating levels of adiponectin showed a mild decrease in OB-HFD (10.482 ±SD μg/mL) mice compared to the LFD and NO-HFD mice (12.937 and 11.568 ±SD μg/mL, respectively) (Fig. 1h). Serum leptin levels were lower in NO-HFD mice compared to the OB-HFD (Fig. 1i). OB-HFD and NO-HFD mice had a significant decrease in the serum levels of P1NP compared to the LFD mice, but no significant differences were observed between OB-HFD and NO-HFD mice (Fig. 1j). Both OB-HFD and NO-HFD mice had significantly greater serum levels of the osteoclast activity marker, TRAcP 5b, compared to LFD mice (Fig. 1k). Although it was not significant, NO-HFD mice had reduced TRAcP 5b serum levels compared to OB-HFD mice (59.174 vs 98.699 ±SD U/L) (Fig. 1k). Overall, these findings suggest that NO-HFD mice had improved systemic metabolism and potentially reduced osteoclast activity compared to OB-HFD mice.
Obesity parameters and metabolic phenotype in male B6 mice. a Body weight (g) measurements over 12 weeks for LFD and HFD-fed male B6 mice (n = 32-41). b Change in weight (%) (dotted line at 28% represents parameter for LFD and dotted line at 58% represents parameter for HFD). c Fold change in fat mass (g) (dotted line at 1.0 represents parameter for LFD and dotted line at 3.3 represents parameter for obese-HFD). d Pearson correlation analysis showing that the change in weight or fat mass fold change both negatively correlate to trabecular bone loss. e Correlation matrix that shows the change in weight or fat mass fold change both negatively correlate to trabecular bone loss (vertical line and horizontal line represent obesity cutoff). f Glucose tolerance test (GTT) for LFD (n = 26), obese HFD-fed (OB-HFD; n = 34) and non-obese HFD-fed (NO-HFD; n = 7). g Insulin tolerance test (ITT) for LFD, obese HFD-fed (OB-HFD), and non-obese HFD-fed (NO-HFD). h Serum adiponectin levels (n = 7). i Serum leptin levels. j Serum procollagen type I N-propeptide (P1NP) levels. k Serum tartrate-resistant acid phosphatase 5b (TRAcP 5b) levels. Analyses for a, f, and g were performed as 2-way ANOVA with Šídák’s multiple comparisons test. Significance for the post-hoc analysis for f, g was defined as: *P < 0.05 LFD vs. OB-HFD, #P < 0.05 LFD vs. NO-HFD-fed, and $P < 0.05 OB-HFD vs. NO-HFD-fed. Analyses for h, i and k were performed as a Kruskal-Wallis test with Dunn’s multiple comparison. Analysis for j was performed as a One-way ANOVA with Tukey’s multiple comparisons test
OB-HFD female B6 mice that met our pre-determined body parameters showed significant impairment to glucose handling after 4 and 8 weeks on a HFD, however, no evidence of insulin resistance was observed (Fig. S4a–j). During the 12 weeks on the HFD, females showed a significant increase in body weight after three weeks (Fig. S5a). Based on our pre-determined obesity parameters, 25 out of 36 female mice were considered obese, meaning 11 mice were considered non-obese (Fig. S5b, c). NO-HFD females did not weigh significantly more than the LFD-fed females at any point throughout the 12 weeks on a HFD, which was unlike males (Fig. S3b). Similar to males, female OB-HFD mice after 12 weeks had impaired glucose handling and responded poorly to insulin compared to the LFD-fed mice (Fig. S5d, e). Compared to OB-HFD, NO-HFD female mice showed improved glucose handling at early timepoints (15 and 30 minutes), but no improvements to insulin sensitivity were observed (Fig. S5d, e). Serum levels of adiponectin showed no significant difference in the female OB-HFD mice (17.413 ±SD μg/mL) compared to the LFD and NO-HFD mice (16.889 and 14.619 ±SD μg/mL, respectively) (Fig. S5f). Serum leptin levels were significantly increased in OB-HFD female mice compared to the LFD and OB-HFD (P = 0.000 2 and 0.000 1) (Fig. S5g). Unlike OB-HFD males, female OB-HFD mice had significantly lower P1NP levels compared to the LFD and NO-HFD mice (P = 0.016 6 and 0.006 6) (Fig. S5h). Of note, no significant difference in P1NP levels was observed between the LFD and NO-HFD mice (Fig. S5h). OB-HFD female mice also had significantly higher serum TRAcP 5b levels, compared to LFD mice. NO-HFD females showed no significant difference in TRAcP 5b levels compared to the OB-HFD mice, although the mean value was lower (22.65 vs 26.75 ±SD U/L) (Fig. S5i). The pre-determined obesity parameters for females showed a trend towards an inverse association between trabecular bone volume and change in body weight (−0.365 6, P = 0.051 1), but a significant negative correlation between trabecular bone volume and the fold change in fat mass (−0.378 7, P = 0.042 8) (Fig. S5j, k). In summary, OB-HFD female mice appeared to be metabolically similar to OB-HFD male mice. Also, NO-HFD females showed a slight decrease in osteoclast activity markers as well as evidence of increased osteoblast activity.
Trabecular and cortical bone loss in OB-HFD mice is the result of increased osteoclast resorption and defective osteoblast function
After 4 weeks, OB-HFD males showed no significant difference in trabecular or cortical bone parameters compared to LFD (Fig. S6a–g). Also, in OB-HFD males, no effect on osteoclast or osteoblast number and function was observed (Fig. S6h–o). But after 8 weeks, male OB-HFD mice had a significant decrease in trabecular bone volume of −26.30%, due to collective changes in trabecular parameters (trabecular spacing, trabecular number, and trabecular thickness) (Fig. S7a–e). However, no cortical bone loss was observed (Fig. S7f, g). The loss of trabecular bone was attributed to OB-HFD mice having a significant increase in mature osteoclast numbers (Fig. S7h, i). After 8 weeks, no difference in osteoblast number was observed, although there was a significant decrease in differentiated osteoblast function by histomorphometry (i.e., mineralizing surface, mineral apposition rate, and bone formation rate) (Fig. S7j–o). After 12 weeks on a HFD, OB-HFD male mice had a significant −34.14% decrease in tibial and −16.72% decrease in femoral trabecular bone volume compared to LFD, which was due to significant changes in trabecular parameters (Figs. 2a–d and S8a–d). In comparison, NO-HFD showed 42.60% greater trabecular bone volume compared to OB-HFD. Also, all trabecular parameters were improved in the NO-HFD mice, compared to the OB-HFD (Fig. 2b–d). In addition to trabecular bone loss, OB-HFD mice had a significant decrease in cortical thickness, but not overall cortical area, compared to LFD mice (Fig. 2e–g). The NO-HFD male mice showed a significant increase in cortical thickness compared to OB-HFD mice. The decrease in cortical bone resulted in OB-HFD mice having a lower cortical polar moment of inertia, which is an indirect measure of bone strength, compared to LFD (Fig. 2h). In contrast, NO-HFD mice had greater cortical strength (P = 0.003 0) compared to the OB-HFD mice (Fig. 2h). Static and dynamic histomorphometry revealed that the trabecular and cortical bone loss observed in the OB-HFD was the result of increased osteoclast number and surface, similar to the results seen after 8 weeks of a HFD (Figs. 2i, j and S8e). However, after 12 weeks, OB-HFD mice had no change in osteoblast number or osteoblast surface was observed, but there was a significant decrease in osteoblast function through diminished mineralizing surface and bone formation rate (Figs. 2k–n and S8f, g). Taken together, trabecular and cortical bone loss in OB-HFD males is the result of increased bone resorption as well as impairment in osteoblast function.
μCT and histomorphometric analysis showed OB-HFD mice have trabecular and cortical bone loss due to an increase in osteoclasts. a Tibial trabecular bone volume per total volume (Tb. BV/TV, %; n = 6-16). b Trabecular spacing (Tb.Sp, mm). c Trabecular number (Tb.N, mm-1). d Trabecular thickness (Tb.Th, mm). e Tibial cortical area per total area (Ct.Ar/Tt.Ar, %). f Representative coronal, mid-shaft μCT images of the proximal tibia (scale bar = 1 mm). g Tibial cortical thickness (Ct.Th, mm). h Cortical polar moment of inertia (Ct. pMOI, mm4). i Representative femoral images with immunohistochemistry for TRAP (pink) and F4/80 (brown) (scale bar = 20 μm). j Number of osteoclasts per bone perimeter (N.Oc/BPm, number per mm; n = 12-14). k Number of osteoblasts per bone perimeter (N.Ob/BPm, number per mm). l Mineralizing surface per bone surface (MS/BS, %). m Mineralizing apposition rate (MAR, μm/d). n Bone formation rate per bone surface (BFR/BS, %). Analyses for a and d–h were performed as a One-way ANOVA with Tukey’s multiple comparisons test. Analyses for b, c were performed as a Kruskal-Wallis test with Dunn’s multiple comparison
Female OB-HFD mice showed no significant differences in trabecular and cortical bone parameters after 4 or 8 weeks of a HFD (Fig. S9a–n). Although it was not significant, after 8 weeks, OB-HFD females had a −12.77% decrease in trabecular bone volume compared to LFD mice (Fig. S9h–k). By 12 weeks, OB-HFD females showed a significant decrease in trabecular bone volume compared to LFD (−18.77%, P = 0.037 4), which was mainly driven by an increase in trabecular spacing and a decrease in trabecular number (Fig. S10a–d). While no significant difference was observed between the OB-HFD and NO-HFD mice, the mean percentage for trabecular bone volume was higher in the NO-HFD mice by 10.78%. Unlike males, cortical thickness and cortical area were not significantly decreased in OB-HFD females; however, there was a significant decrease in cortical porosity compared to the LFD (Fig. S10e–h). Similar to males, OB-HFD females had a significant increase in osteoclast number (P = 0.038 2), as well as a significant decrease in osteoblast number (P = 0.007 9) that was coupled with a significant decrease in osteoblast function (i.e., mineralizing surface and bone formation rate) (Fig. S10i–o). In sum, OB-HFD females have trabecular, but not cortical, bone loss as a result of unbalanced bone remodeling through increased bone resorption and suppressed bone formation.
Bone marrow adiposity in OB-HFD mice increases osteoclastogenesis
Similar to WAT depots, BM adiposity (number and size) also increases with DIO. Male OB-HFD mice had a significant increase in BM adiposity after 4 weeks of a HFD (Fig. S11a–c). The expansion of BM adiposity continued after 8 and 12 weeks of a HFD (Figs. 3a–c and S11e–g). Despite being on a HFD, NO-HFD mice had minimal BMAT expansion (Fig. 3a–c). Due to NO-HFD mice having less BMAT expansion as well as improved bone volume, we speculated that increased BM adiposity might lead to osteoclast-driven bone loss. To test this, we co-cultured mature BM adipocytes with BM CD11b+ myeloid cells from LFD and OB-HFD mice during osteoclast differentiation. After 5 days, we saw a significant increase in the number and size of TRAP+ cells when co-cultured with OB-HFD BMAT (Fig. 3d–f). Since previous studies have shown myeloid cells from HFD-fed mice are programmed to more readily differentiate into osteoclasts, we also cultured BMAT from OB-HFD mice with CD11b+ myeloid cells from LFD mice. Interestingly, we saw a significant increase in the number of TRAP+ cells compared to osteoclast differentiation of LFD myeloid cells and LFD myeloid cells cultured with LFD BMAT. To better understand the effect BMAT has on osteoclastogenesis, we investigated the molecular phenotype of BM adipocytes isolated from OB-HFD mice. The gene expression of OB-HFD BMAT did not show an inflammatory profile (Fig. 3g). Instead, there was a significant increase in Foxp3 (unpaired t-test, P = 0.022), as well as monocyte-chemoattractant protein 1 (Mcp-1) and IL-10; two major factors that contribute to the recruitment and expansion of myeloid-derived suppressor cells. There was also a significant decrease in the inflammatory cytokine IL1β. In contrast, visceral perigonadal WAT (gWAT) from OB-HFD mice, which has some metabolic similarities to human omental WAT,33,34 showed an inflammatory phenotype with increased IL6, TNFα, and TLR4 (Mann-Whitney t-test, P = 0.001 5) expression (Fig. 3h). This is consistent with previous findings that have shown inflammatory WAT in DIO models in both rodents and humans.35,36,37,38 The only similarity between BMAT and gWAT was an increase in Mcp-1 expression. BMAT-derived Mcp-1 expression started as early as 8 weeks in OB-HFD mice, which coincides with trabecular bone loss in OB-HFD mice (Fig. S11h).
BMAT expansion in OB-HFD mice did not have an inflammatory phenotype and increased osteoclast differentiation. a Representative H&E images of the BMAT within the proximal tibia. b Number of BM adipocytes within the proximal tibia (BM.Ads/Ma.AR, number/mm2). c Average size of the BMAT within the proximal tibia (μm2). d Representative TRAP+ images after 5 days of osteoclast differentiation when co-cultured with BMAT. e Number of TRAP+ cells after 5 days of osteoclast differentiation when co-cultured with BMAT (n = 6; each dot represents an averaged technical replicate). f Average diameter of TRAP+ cells after 5 days of osteoclast differentiation when co-cultured with BMAT. g Immune and inflammatory gene expression of BM adipocytes relative to TBP (n = 7; represented as the mean with individual data points). h Immune and inflammatory gene expression of visceral, perigonadal WAT (gWAT) relative to TBP (n = 10; represented as the mean with individual data points). Analyses for b, c were performed as a One-way ANOVA with Tukey’s multiple comparisons test. Analyses for e, f were performed as a 2-way ANOVA with Tukey’s multiple comparisons test. Analyses for g, h were performed as a 2-way ANOVA with Šídák’s multiple comparisons test
Similar to males, OB-HFD females had a significant increase in BM adiposity after 4 weeks on HFD that continued after 8 and 12 weeks (Fig. S12a–i). In the NO-HFD females, we found greater heterogeneity in BMAT expansion than we previously observed with the NO-HFD males; however, NO-HFD females still had a significant decrease in BM adiposity compared to OB-HFD mice (Fig. S12g–i). Additionally, OB-HFD females had a significant increase in BMAT-derived Mcp-1 expression after 12 weeks of a HFD (Fig. S12j). Due to the increase in mature osteoclasts, the increase in BMAT-derived Mcp-1, and the lack of an inflammatory phenotype in BMAT in both male and female OB-HFD mice, we next sought to investigate the BM immune profile, specifically within the myeloid lineage, of OB-HFD mice.
Increased PD-L1+ myeloid cells in OB-HFD mice led to BM immunosuppression
After 12 weeks of a HFD, gWAT from OB-HFD male and female OB-HFD mice had an increase in the number of tissue F4/80+ macrophages, which directly contributes to the requirement of monocytes39,40 (Fig. S13a–h). To determine if obesity also induces the production of monocytes in the BM, we performed flow cytometric analysis of the BM myeloid cell compartment. After 12 weeks, OB-HFD males demonstrated an increase in the number of short-term hematopoietic stem cells (ST-HSCs), multipotent progenitors (MPPs), common myeloid progenitors (CMPs), and monocyte/macrophage pool, indicating activated myelopoiesis (Fig. S14a–f). Our data also demonstrated an increase in MHCIInegPD-L1+ myeloid cells (fully defined as CD11bhiCD11c+MHCIInegF4/80+PD-L1+) (Figs. 4a–f and S14i, j). The increase in MHCIInegPD-L1+ myeloid cells was accompanied by a decrease in CD4+ T-cells (P = 0.058 4) (Fig. S14k). It is important to note that we observed a mild, nonsignificant increase in B-cell precursors or mature B-cells, which may be in response to the decrease in CD4+ memory T-cells (Fig. S14g, h). PD-L1 is an inhibitory immune checkpoint protein that interacts with the PD-1 receptor found on T-cells, which controls the activation of T-cells. Of importance, CD4+ T-cells specifically recognize and interact with MHCII, meaning this population of myeloid cells also cannot activate CD4+ T-cells. The lack of MHCII expression and the increase in PD-L1 expression led us to believe that OB-HFD mice had an immunosuppressive BM microenvironment. In fact, an increase in MHCIInegPD-L1+ myeloid cells in OB-HFD mice was first observed after 8 weeks on a HFD in males and females (Figs. S14l–n and S15a–c). To test the inhibitory effect of these MHCIInegPD-L1+ cells, BM CD11b+ myeloid cells were isolated from OB-HFD mice and co-cultured with activated splenic T-cells from LFD mice. The BM myeloid cells from OB-HFD mice caused a significant decrease in moderately proliferative T-cells and a significant increase in poorly/non-proliferative T-cells (Fig. 4g, h). The increase in PD-L1 expression led us to investigate cell populations with increased PD-1 expression. Interestingly, we observed an increase in PD-1+ osteoclast precursors (defined as [B220/CD3/NK1.1]negc-kit+CD115+CD11blowPD-1+) (Fig. 4i–k). The increase in PD-1+ osteoclast precursors was observed in OB-HFD males after 8 weeks (P < 0.000 1) (Fig. S14o–q). Similar to males, OB-HFD females had a decrease in CD4+ T-cells after 8 weeks and 12 weeks that was coupled with an increase in immunosuppressive MHCIInegPD-L1+ myeloid cells (Fig. S15a–c, f–h). Similarly, OB-HFD females had a significant increase in PD-1+ osteoclast precursors after 8 and 12 weeks on a HFD (Fig. S15d, e, i, j). Collectively, these data show that obesity created an immunosuppressive BM microenvironment by promoting the generation of PD-L1-expressing myeloid cells. The increase in these immunosuppressive myeloid cells occurred after a significant increase in BMAT had been established. In addition to the increase in PD-L1+ myeloid, we also observed a significant increase in PD-1+ osteoclast precursors that coincided with our observations of trabecular bone loss. Due to these findings, we sought to further investigate the role PD-1/PD-L1 plays in osteoclastogenesis.
OB-HFD mice had BM immunosuppression through an increase in PD-L1+ myeloid cells. a Flow cytometric plots demonstrating subsets of CD11bhiCD11c+MHCIInegF4/80+ cells (outlined in red). b Flow cytometry plots showing the percentage of immunosuppressive PD-L1+F4/80+ cells (outlined in green). c Percentage of MHCIIneg and MHCII+ cells (n = 10). d Overall ratio of cells expressing MHCII compared to F4/80. e Number of PD-L1+ cells. f Percentage of PD-L1+ cells. g MLR showing T-cell proliferation rate when co-cultured with BM myeloid cells (n = 6). h Representative histogram of T-cell proliferation. i Number of PD-1+ osteoclast precursor cells (n = 10). j Percentage of PD-1+ osteoclast precursor cells. k Flow cytometric plots demonstrating subsets of [B220/CD3/NK1.1]negc-kit+CD115+CD11blowPD-1+cells (outlined in black). Analyses of c and g were performed as a 2-way ANOVA with Šídák’s multiple comparisons test
Stimulatory effect of the PD-1/PD-L1 axis on osteoclastogenesis
Osteoclasts differentiate from within the myeloid cell lineage. In OB-HFD mice, we observed a significant increase in PD-L1+ myeloid cells, PD-1+ osteoclast precursors, and mature osteoclasts. It is well-known that the PD-1/PD-L1 axis produces an inhibitory signal for T-cells.29,41 However, we speculated that the PD-1/PD-L1 axis had a stimulatory mechanism within the myeloid lineage that increases osteoclastogenesis. To test this, BM CD11b+ myeloid cells isolated from chow-fed B6 mice were treated with inhibitory anti-PD-1 or anti-PD-L1 during early (days 0-3) and late (days 3-6) osteoclast differentiation. We found the use of mCSF-1 in our osteoclast cultures increased PD-L1 expression (data not shown), thus creating an increase in PD-L1 expression without using myeloid cells from OB-HFD mice, which are primed to favor osteoclastogenesis.
Cultures treated with anti-PD-1 and anti-PD-L1 during early osteoclast differentiation (days 0-3) resulted in significantly fewer and smaller TRAP+ cells compared to the IgG control (Fig. 5a–d). Cultures treated with anti-PD-1 and anti-PD-L1 during late osteoclast differentiation (days 3-6) showed an increase in the number and size of TRAP+ cells compared to the cells treated during days 0-3 (Fig. 5a–d). We suspected that the interaction between PD-1 and PD-L1 on myeloid cells is necessary to initiate differentiation and commitment of myeloid cells to osteoclastogenesis (Fig. S16a). Cells treated from days 0-3 with anti-PD-1 or anti-PD-L1 had a decrease in expression of osteoclast commitment genes (Rank, Ostm1, c-Fos) as well as a decrease in fusion (Dc-stamp, Oc-stamp) and resorptive genes (Ctsk, Acp5) (Figs. 5e–j and S16b). We suspected early inhibition of PD-1/PD-L1 axis prevented myeloid cell commitment to osteoclast differentiation; thus, we anticipated the decrease in fusion and resorptive genes. In support of this, the blockade of the PD-1/PD-L1 axis during days 3-6 resulted in a significant increase in genes associated with osteoclast differentiation compared to days 0-3 (Figs. 5e–j and S16c). Specifically, we observed a significant increase in expression of commitment genes, Ostm1 and c-Fos, compared to days 0-3 (Fig. 5f, g). Late-stage inhibition of PD-1 and PD-L1 also increased resorptive genes, Ctsk and Acp5, compared to days 0-3 (Fig. 5i, j). These results were anticipated if the blockade of PD-1/PD-L1 signaling during late-stage differentiation did not affect myeloid cell commitment to osteoclast differentiation. Overall, there appeared to be an early stimulatory effect of PD-1/PD-L1 within the myeloid lineage during osteoclastogenesis.
Inhibition of the PD-1/PD-L1 signaling during early osteoclast differentiation leads to a decrease in osteoclastogenesis. a Representative TRAP+ images after PD-1 and PD-L1 inhibition during days 0-3 of osteoclastogenesis (scale bar = 0.5 mm). b Representative TRAP+ images after PD-1 and PD-L1 inhibition during days 3-6 of osteoclastogenesis (scale bar = 0.5 mm). c Number of TRAP+ cells after PD-1 and PD-L1 inhibition during days 0-3 and days 3-6 (n = 8; each dot represents an averaged technical replicate). d Average diameter (μm) of TRAP+ cells after PD-1 and PD-L1 inhibition during days 0-3 and days 3-6. e Gene expression of Ostm1, an osteoclast lineage commitment gene, relative to β-Actin after PD-1 and PD-L1 inhibition during days 0-3 and days 3-6 (n = 6). f Gene expression of c-FOS, an osteoclast lineage commitment gene. g Gene expression of Dc-stamp, an osteoclast fusion gene. h Gene expression of Dc-stamp, an osteoclast fusion gene. i Gene expression of Ctsk, an osteoclast resorption gene. j Gene expression of Acp5, an osteoclast resorption gene. Analyses for c, d were performed as a 2-way ANOVA with Šídák’s multiple comparisons test. Analyses for e–i were performed as a 2-way ANOVA with Tukey’s multiple comparisons test
BMAT depletion in OB-HFD mice improves trabecular parameters by reducing BM immunosuppression
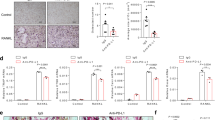
Our data indicated that the expansion of BMAT in OB-HFD mice leads to an increase in immunosuppressive myeloid cells and osteoclastogenesis. To test this, we used a BMAT-depleted mouse model (BMAd-Pparg KO) that prevents the maturation of lipid-laden adipocytes. In this model, obese mice were characterized by having a change in body weight >40% and a fold change in fat mass >2.3 (Fig. S17a–c). As previously reported,42,43 this mouse model only targets BMAT, while not affecting other fat depots. This was confirmed when we observed a significant increase in gWAT adiposity in the OB-HFD mice (Fig. S17d–f). After 12 weeks on a HFD, OB-HFD mice showed a similar metabolic response to glucose and insulin sensitivity as OB-HFD B6 mice (Fig. S17g–j). In this model, we verified that OB-HFD BMAd-Pparg KO mice had a −93.34% decrease in BM adipocytes as well as a decrease in adipocyte size compared to OB-HFD BMAd-Pparg WT (Fig. 6a–c). Isolated BMAT from OB-HFD BMAd-Pparg KO mice showed a significant decrease in Mcp-1 and IFNg expression (Fig. 6d, e). Interestingly, we noticed less accumulation of F4/80+ macrophages within the gWAT of OB-HFD BMAd-Pparg KO mice (Fig. 6f). The decrease in peripheral macrophages stemmed from a significant decrease in BM myelopoiesis (i.e., ST-HSCs, MPP, and F4/80+ macrophages) that also resulted in a decrease in PDL1+ myeloid cells that also resulted in a decrease in PDL1+ myeloid cells (Figs. 6g and S17k–m). We speculated that the reduction in PD-L1 expression was related to the decrease in BMAT-derived Mcp-1 and IFNg expression, two factors that regulate macrophage recruitment and PD-L1 expression. Consistent with our OB-HFD B6 data, OB-HFD BMAd-Pparg WT mice showed an increase in PD-1+ osteoclast precursors (Fig. 6h). However, OB-HFD BMAd-Pparg KO mice had a significant decrease in PD-1+ osteoclast precursors (Fig. 6h). Due to the decrease in BM PD-1 and PD-L1 expression, we anticipated that OB-HFD BMAd-Pparg KO mice would have less osteoclastic bone resorption. Regarding bone remodeling markers, there was no significant difference in serum P1NP levels, but OB-HFD BMAd-Pparg WT mice had a significant increase in CTX and TRAcP 5b, compared to LFD-fed BMAd-Pparg WT (Fig. 6i–k). Importantly, OB-HFD BMAd-Pparg KO mice had a significant decrease in CTX and TRAcP 5b levels (Fig. 6k). Altogether, these results indicated that BMAT depletion or the lack of BMAT accrual in DIO may protect bone volume by decreasing osteoclast activity.
BMAT depletion in obese mice improved BM immunosuppression through a decrease in PD-L1+ myeloid cells. a Number of BM adipocytes within the proximal tibia (BM.Ads/Ma.AR, number/mm2; n = 10-13). b Average size of the BMAT within the proximal tibia (μm2). c Representative H&E images of the BMAT within the proximal tibia (scale bar = 250 μm). d Immune and inflammatory gene expression of BM adipocytes from LFD mice relative to TBP (n = 8; represented as the mean with individual data points). e Immune and inflammatory gene expression of BM adipocytes from OB-HFD mice relative to TBP (n = 8; represented as the mean with individual data points). f F4/80 IHC of gWAT (red arrows indicate minimal macrophage infiltration in LFD-fed mice). g Number of PD-L1+ myeloid cells. h Number of PD-1+ osteoclast precursor. i Serum procollagen type I N-propeptide (P1NP) levels (n = 8). j Serum C-terminal telopeptide of type 1 collagen (CTX-1), a bone resorption marker. k Serum TRAcP 5b levels. Analyses for a, b and g–k were performed as 2-way ANOVA with uncorrected Fisher’s LSD. Analyses for d, e were performed as multiple unpaired t-tests and multiple Mann-Whitney tests, respectively
Based on our previous results in OB-HFD B6 mice, we speculated that the decrease in PD-1+ and PD-L1+ cells observed in OB-HFD BMAd-Pparg KO would decrease the number of mature osteoclasts, thus improving trabecular and cortical bone volume. Consistent with OB-HFD B6 mice, OB-HFD BMAd-Pparg WT had a significant decrease in trabecular bone volume (Fig. 7a–e). In comparison, OB-HFD BMAd-Pparg KO showed improvements to trabecular bone parameters, which led to an overall increase of 24.31% in trabecular bone volume. The trabecular bone in the OB-HFD BMAd-Pparg KO appeared to be more plate-like, providing additional support and stability, compared to the more rod-like phenotype seen in the OB-HFD BMAd-Pparg WT mice (Fig. 7f). We did not observe any cortical bone phenotype in this model (Fig. 7g, h). The trabecular bone loss seen in OB-HFD BMAd-Pparg WT was due to a significant increase in osteoclast number and activity, which is identical to the results seen in the OB-HFD B6 mice (Fig. 7i, j). However, unlike OB-HFD B6 mice, after 12 weeks on a HFD, OB-HFD BMAd-Pparg WT mice did not show any changes in osteoblast number or function (Fig. 7k–n). Most importantly, while no changes in osteoblasts were observed, OB-HFD BMAd-Pparg KO mice showed a significant decrease in osteoclast number and activity (Fig. 7i–n). In summary, the lack of BMAT accrual in OB-HFD mice resulted in a protective trabecular skeletal phenotype in part by reducing PD-L1 signaling to osteoclasts.
μCT and histomorphometric analysis showed that BMAT depletion in obese mice had improved trabecular bone due to a decrease in mature osteoclasts. a Representative μCT images of the proximal tibia (scale bar = 100 μm). b Tibial trabecular bone volume per total volume (Tb. BV/TV, %; n = 10-11). c Trabecular spacing (Tb.Sp, mm). d Trabecular number (Tb.N, 1/mm). e Trabecular thickness (Tb.Th, mm). f Trabecular structural model index (SMI). g Tibial cortical thickness (Ct.Th, mm). h Tibial cortical area per total area (Ct.Ar/Tt.Ar, %). i Number of osteoclasts per bone perimeter (N.Oc/BPm, number per mm; n = 8). j Eroded surface per bone surface (ES/BS; %). k Number of osteoblasts per bone perimeter (N.Ob/BPm, mm). l Mineralizing surface per bone surface (MS/BS, %). m Mineralizing apposition rate (MAR, μm/d). n Bone formation rate per bone surface (BFR/BS, %). Analyses for b–h were performed as a 2-way ANOVA with uncorrected Fisher’s LSD. Analyses for i–m were performed as a 2-way ANOVA with uncorrected Fisher’s LSD
Discussion
In this work, we show that BM adipocytes play a critical role in obesity-related BM immunosuppression and subsequent bone loss through PD-1/PD-L1 mediated osteoclastogenesis. Previous studies have reported inconsistent results regarding DIO and bone loss. At least one rodent study failed to show that DIO caused bone loss.9 Other studies showed that DIO leads to trabecular, but not cortical bone loss,44 or DIO leads to bone loss in males, but not females.45 Studies that report DIO-related bone loss in males,8,46 concluded that the observed bone loss is due to increased adipogenesis at the expense of osteoblastogenesis. However, these studies did not investigate possible pro-osteoclastic mechanisms of increased BM adiposity. To the best of our knowledge, this is the first study to provide a novel role of BMAT in the mediation of pro-osteoclastic PD-1/PD-L1 signaling in the context of obesity. This is also the first study to use pre-determined obesity parameters to investigate the effects of obesity on bone health. These parameters led to consistent trabecular bone loss in male and female B6 mice and cortical bone loss in males.
Our current results illustrate that the expansion of BMAT has a negative impact on the BM microenvironment in the context of DIO. In our OB-HFD mice, significant BMAT expansion happens as early as 4 weeks on a HFD, which is supported by previous findings.47 But the minimal BMAT accrual seen in NO-HFD mice, compared to the OB-HFD mice, was coupled with improved trabecular bone volume. NO-HFD mice had a decrease in circulating leptin levels compared to the OB-HFD, which may explain the lack of BMAT expansion. Despite being on a HFD, NO-HFD mice may still be responsive to leptin signaling, which depresses feeding and decreases adiposity.48,49
Phenotypically similar to DIO, calorie restriction models also show a dramatic and rapid increase in BM adiposity. However, previous work from our group has shown that the molecular phenotype of the BM adipocytes is remarkably different for each dietary intervention. In brief, we showed that BM adipocytes isolated from humans after a 10-day high-calorie diet showed an upregulation of FOXP3, the transcription factor that controls the development of regulatory T-cells (T-regs), which are critical in reducing inflammatory immune responses.50 After 10 days of fasting, the BM adipocytes had an upregulation of inflammatory genes (CP, CFH, IGFBP3).50 These findings illustrated the dynamic and differing effects that nutritional influences (i.e., acute overnutrition and acute calorie deprivation) have on BMAT metabolism and function within the BM microenvironment. Based on these findings, we know that not all BMAT expansion is the same and cannot be considered as such. Similar to our previous findings in humans, but contrary to other studies, BM adipocytes from OB-HFD mice did not have an inflammatory phenotype, which was opposite to the phenotype observed in visceral gWAT. Tencerova et al in 2018 showed a similar lack of BMAT inflammation after HFD,8 however, they did not find a significant increase in BMAT-derived Mcp-1 (or Ccl2) expression. We attributed this to their HFD-fed mice only having an increase in body weight of 29% and an increase in body fat mass of 212% after 12 weeks, whereas our mice had a change in body weight >58% and a fold change in fat mass >3.3 fold (or 330%). It has also been shown using genetic mouse models of obesity, such as leptin receptor-deficient (LepRdb/db) mice, that the expansion of BMAT correlates with elevated Mcp-1 levels.51 Given the role of Mcp-1 in promoting myeloid cell accumulation and osteoclast differentiation,51,52,53 these results indicate that the accrual of BMAT associated with obesity, but not necessarily a HFD, creates crosstalk between BM adipocytes and myeloid cells. In addition to its direct effects on myeloid cells and osteoclast differentiation, the secretion of Mcp-1 by BM adipocytes has been shown to fuel BMSC differentiation into new adipocytes, which disrupts the balance within the BM to favor adipogenesis and myelopoiesis.51,54 Studies have claimed an increase in BMAT-derived Mcp-1 signifies an inflammatory state within the BM microenvironment.51 However, our OB-HFD mice showed a significant increase in immunosuppressive PD-L1+ myeloid cells after 8 and 12 weeks of a HFD. We confirmed that the BM myeloid cells from OB-HFD mice were immunosuppressive and had a negative impact on T-cell proliferation. Interestingly, this immunosuppressive phenotype through elevated PD-L1 expression is similar to reported tumor microenvironments found with BM metastatic cancers,27,55,56,57,58,59 but has not been previously reported in non-tumor-bearing obese mice. We speculate the increase in PD-L1 expression seen in OB-HFD mice is related to the increase in Mcp-1 in part because previous cancer research has suggested the recruitment of myeloid cells via Mcp-1 creates an immunosuppressive tumor microenvironment by attracting tumor-associated macrophages, which are a major source of PD-L1.60
The inflammation commonly observed with obesity stems from macrophage accumulation in the WAT.18 Inflammatory responses triggered by myeloid cells after sensing tissue injury, similar to aberrant WAT expansion, can contribute to increased tissue inflammation unless regulated by the adaptive immune system.61,62 However, increased PD-L1 expression diminishes adaptive immune function, which is why there is continued inflammation in the WAT. It may seem paradoxical to identify obesity as a state of BM immunosuppression, since it is commonly referred to as a chronic inflammatory disease; however, we speculate that the increase in myelopoiesis within the BM also leads to an increase in circulating myeloid cells that accumulate within the peripheral WAT. In support of this, studies have suggested that the accumulation of macrophages in WAT with obesity is due to an influx of BM–derived precursors (i.e., monocytes) and their subsequent differentiation into mature F4/80-expressing macrophages.39 It has also been shown that individuals living with obesity have an increase in circulating monocyte populations.63,64,65 Overall, this suggests the changes in the BM are correlated with the systemic obesity phenotype. Thus, our OB-HFD mice demonstrate a simultaneous state of BM immunosuppression and WAT inflammation that is driven by myeloid cell differentiation and accumulation.
Studies have shown a strong relationship between osteoclasts and immune cell activity,66,67 which is not surprising considering osteoclasts differentiate from within the myeloid cell lineage. The PD-1/PD-L1 axis is an immune cell checkpoint with a predominantly inhibitory effect on T-cells. However, PD-1 and PD-L1 can also be expressed in other cell types, including other non-immune cells. In tumor BM microenvironments, it has been shown that cancer-produced PD-L1 promoted osteoclastogenesis via PD-1 signaling on osteoclast precursors.57 It has also been shown that treatment with anti-PD-L1 antibodies suppresses bone metastasis of breast cancer and melanoma through inhibiting osteoclast differentiation and activity while enhancing immune responses in the BM microenvironment.57,58 By 8 weeks, our OB-HFD mice had an increase in PD-1+ osteoclast precursors and PD-L1+ myeloid cells that resulted in a loss of trabecular bone through increased osteoclast activity. After 12 weeks, the continued elevation of BM PD-L1+ myeloid cells, PD-1+ osteoclast precursors, and mature osteoclasts resulted in further trabecular bone loss as well as a decrease in cortical thickness and the indirect measurement of cortical strength. We speculate that the PD-1/PD-L1 axis in OB-HFD mice may have a stimulatory effect on osteoclastogenesis, similar to the effect observed in the tumor BM microenvironment. To investigate this potential role of the PD-1/PD-L1 signaling axis, we treated CD11b+ cells with anti-PD-1 and anti-PD-L1 antibodies during early (days 0-3) osteoclastogenesis. By preventing early PD-1/PD-L1 signaling, we observed a significant decrease in the number and size of mature osteoclasts. We also observed a decrease in osteoclast commitment genes, such as Ostm-1, c-Fos, and Rank, as well as osteoclast fusion (Calcrl, Nfatc1, Dc-stamp, Oc-stamp) and resorptive genes (Ctsk, Acp5, ATP6v0d2, Traf6, CD47). Due to these results, we speculated that PD-1 and PD-L1 play a role in myeloid cell commitment during early osteoclast differentiation. The implication that PD-1/PD-L1 signaling plays a vital role in osteoclastogenesis has been reported in previous cancer studies. After inoculation within the BM cavity with lung carcinoma cells, Pd1−/− mice showed a significant decrease in osteoclast number compared to tumor-bearing WT mice.57 It has also been reported that BM cells isolated from PD-L1−/− mice have impaired osteoclast differentiation and maturation.58 Expression levels of osteoclast-specific genes (Trap, Ctsk, Car2, Calcrl, Oscar, and Naftc1) were decreased in PD-L1−/− differentiation cultures.58 Taken together, these results strongly supported the role of the PD-1/PD-L1 signaling axis in the differentiation and lineage fate commitment of myeloid progenitors towards osteoclastogenesis.
Importantly, the increase in PD-L1+ myeloid cells in our OB-HFD mice was linked to the expansion of BMAT. When we depleted BMAT in HFD-fed mice, we saw a decrease in BMAT-derived Mcp-1 expression, PD-L1+ myeloid cells, and PD-1+ osteoclast precursors. By reducing the number of PD-L1-expressing cells within the BM, we observed significant improvements in trabecular bone parameters by reducing the number of mature osteoclasts. Osteoporosis and malignant states such as multiple myeloma and acute myeloid leukemia are also associated with increased BMAT expansion and greater bone resorption.68,69,70,71 Interestingly, current research is investigating methods to improve bone quality in these pathologies by administering treatments like intermittent PTH and Anti-SOST, which reduce BM adiposity and stimulate osteoblastic bone formation,72,73,74,75,76,77 further highlighting the link between BMAT accrual and detrimental bone effects.
There are several limitations to this study. First, we established obesity parameters based on changes in body weight and fat mass, which negatively correlated with bone volume in males and females. To avoid possible ascertainment bias in our analysis, weakening our conclusions about obesity-induced bone loss, we established these parameters a priori and with the support of previous findings from our laboratory. At the extreme ends of the distribution in HFD-fed mice, it has been shown that there can be a 4-fold difference in fat mass.31 In the absence of allelic variation, it was suggested that the phenotypic variation may be determined by either unstable stochastic mechanisms or by stable epigenetic modifications of genes.31,32 Serum lipid concentration, hormone imbalance, and transgenerational inheritance may be possible sources of epigenetic modifications associated with obesity.31,78,79 Also, epigenetic modifications that alter DNA methylation (i.e., hypomethylation or hypermethylation) in adipose tissue may be involved in obesity-related pathogenesis, as they affect the expression of genes associated with metabolism, fat storage, immune responses, and adipogenesis.80 The use of our obesity parameters reduced possible epigenetic variation, so that we could more conclusively establish a mechanism for obesity-induced bone loss. Of course, any conclusions about obesity-induced bone loss in humans would have to consider that heterogeneity. That being said, the sample sizes for NO-HFD mice were small (n = 6-9), and since the mice were group-housed, we were unable to determine if their food consumption was significantly less than the OB-HFD mice. Therefore, our findings regarding the metabolic differences between NO-HFD and OB-HFD mice could be considered speculative. As previously stated, the underlying epigenetic mechanism that causes varying obesity phenotypes is an area that requires further research. Secondly, we reported that the increase in PD-L1+ myeloid cells in OB-HFD was due to the rapid and significant expansion of BMAT. As such, one limitation is that we did not measure PD-L1 expression in NO-HFD mice. However, NO-HFD mice mimic our BMAT-depleted model since both were fed a HFD, but accrued minimal BMAT and did not have increased bone resorption. Additionally, although BM adipocytes from OB-HFD mice showed an increase in Mcp-1 and IL-10, it is possible that other adipokines, such as Cxcl1,81,82 Cxcl2 (or MIP-2),81,82 GM-CSF,83 Ccl3 (or MIP-1α),84,85 or Ccl5,86 might contribute to enhanced myeloid cell accumulation within the BM of OB-HFD mice. In addition to BM adipocytes, Mcp-1 can be secreted by BMSCs, osteoblasts, osteoclasts, and macrophages. Therefore, future research is needed to determine the impact of BMAT-derived Mcp-1 with DIO. Using the BMAd-Cre mice crossed with a Mcp-1flox/flox mouse, we would be able to determine the relationship between BMAT-derived MCP1, macrophage accumulation, and osteoclastogenesis. Additionally, our group is doing more research to further understand the stimulatory, pro-osteoclastic role of PD-1/PD-L1. We are using a mouse model with an inducible myeloid cell-specific (LysM-Cre) knockout of PD-L1 to better understand this mechanism, which will be published as a subsequent manuscript.
In conclusion, our results demonstrated a profound and novel role of BMAT in regulating immunosuppressive myeloid cell regulation and osteoclastogenesis. In OB-HFD mice, the rapid accrual of BMAT led to molecular changes that altered the myeloid cell population within the BM, resulting in elevated immunosuppressive PD-L1 expression. The elevated PD-L1 expression had a stimulatory effect within the myeloid lineage that pushed osteoclast precursors towards osteoclastogenesis and resulted in obesity-related bone loss. By genetically preventing the accrual of BMAT with DIO, BM immunosuppression and the detrimental effects on bone were significantly lessened. These findings suggest that therapies that decrease BMAT in individuals with obesity may reduce obesity-related complications by improving BM immunity. Also, our findings indicated that targeting PD-1/PD-L1 signaling may diminish osteoclastogenesis. Not only is this important in the context of obesity-related bone loss, but also in the treatment of metastatic breast cancer and multiple myeloma, both of which stimulate osteoclastogenesis and also have a higher prevalence of individuals who are obese. However, there have been studies that suggest immune checkpoint inhibitors function poorly in patients with bone metastases and do not result in the same survival benefits as patients with the same primary tumor.54 That being said, it has been shown that individuals who are obese (BMI > 30) have an increase in serum PD-1+ T-cells compared to healthy individuals (BMI < 30), and this T-cell dysfunction associated with obesity increased the efficacy of PD-1/PD-L1 blockade in tumor-bearing mice.87 These results were corroborated with clinical data showing improved outcomes in obese patients with cancer treatments using PD-1/PD-L1 inhibitors.87 Although these data do not show an increase of PD-1/PD-L1 within the BM, it does strengthen our findings by demonstrating that individuals who are obese have elevated PD-1/PD-L1 expression. However, given the limited clinical data, it remains unclear if immune checkpoint inhibitors would be beneficial for the treatment of bone metastatic tumors in individuals with obesity. But given the strong correlation between obesity and certain cancers, PD-L1 may become a biomarker for pathological obesity or used as a predictor for cancer incidences in individuals with obesity. Altogether, our work highlights the substantial impact BM adipocytes, previously regarded as inert “fillers” of the BM, have on immunity and the perpetuation of pathological conditions. Further research is needed to investigate the crosstalk between BM adipocytes and immune cells and how this relationship impacts immunopathology with obesity.
Materials and methods
Mice and dietary studies
All mouse procedures were performed under the approval of the Institutional Animal Care and Use Committee at the MaineHealth Institute for Research (under the protocol number 2019) and followed the US National Research Council’s Guide for the Care and Use of Laboratory Animals, the US Public Health Service’s Policy on Humane Care and Use of Laboratory Animals, and Guide for the Care and Use of Laboratory Animals. For each experiment, the numbers of mice used are provided within the corresponding figure legend.
C57BL/6J (B6) obesity studies
Male and female C57BL/6J (B6) mice (Jackson Laboratories, JAX:000664) were group-housed and randomly assigned to be fed a high-fat diet (HFD, 60% kcal; D12492, Research Diets Inc.) or a sucrose-matched low-fat diet (LFD, 10% kcal; D12450J, Research Diets Inc.) for 4, 8, or 12 weeks. Mice arrived at the animal facility at MaineHealth Institute for Research at 4 weeks old, which allowed for a 3-week habituation period before baseline body composition was measured. Starting at 8 weeks old, LFD-fed and HFD-fed mice were pair-fed with feed changes occurring three times a week (Monday, Wednesday, and Friday).
BMAd-Pparg KO and obesity studies
BMAT-depleted mice were generously donated for use by Dr. Ziru Li at MaineHealth Institute for Research. These mice were generated and previously characterized by Drs. Ziru Li and Ormond MacDougald.42,43 Starting at 8-10 weeks old, male BMAd-Pparg KO and littermate controls, BMAd-Pparg WT, were randomly assigned to be fed a high-fat diet (HFD, 60% kcal; D12492, Research Diets Inc.) or a sucrose-matched low-fat diet (LFD, 10% kcal; D12450J, Research Diets Inc.) for 12 weeks. LFD-fed and HFD-fed mice were pair-fed, and feed was changed three times a week (Monday, Wednesday, and Friday). Baseline body composition measurements were obtained one week prior to the start of dietary intervention.
Body composition measured with nuclear magnetic resonance (NMR)
Body composition (body fat, lean mass, and free fluid) was analyzed by Minispec NMR (Bruker Corp; MaineHealth Institute for Research), which allowed for non-invasive, serial body composition measurements without the use of anesthesia. The Minispec uses the contrasting hydrogen density and/or hydrogen spin properties from adipose tissue and muscle for estimating body composition. A quality control check of NMR parameters using a standard provided by the manufacturer was performed before testing. NMR measurements were taken for baseline body composition and 2 days prior to euthanasia.
Insulin and glucose tolerance testing
For insulin (ITT) and glucose tolerance tests (GTT), mice were fasted for 6 h, then a baseline fasted blood glucose was measured (Timepoint 0). For ITT, insulin diluted in sterile PBS was injected intraperitoneally (IP) at a dose of 1 U/kg. For GTT, pharmaceutical-grade 10% dextrose USP (ICU Medical Inc.) was injected IP at a dose of 1 000 mg/kg. Blood glucose was measured 15-, 30-, 45-, 60-, and 120-minutes post injection using the AlphaTrak 2 (Zoetis, Parsippany-Troy Hills, NJ). ITT and GTT were completed 2 weeks and 1 week prior to euthanasia, respectively.
Serum collection and ELISA analyses
Prior to blood collection, mice were fasted for 6 h. Blood was collected using a terminal blood collection protocol. Samples were allowed to clot at RT for 1-2 h before being centrifuged at 5 500 g for 15 minutes. After centrifugation, the upper phase containing serum was transferred to a new, sterile microcentrifuge tube and stored at −80°C. Serum protein analysis of adiponectin (Mouse Adiponectin/Acrp30 Quantikine ELISA, R&D Systems MRP300), leptin (Mouse/Rat Leptin Quantikine ELISA, R&D Systems MOB00B), TRAP (Mousetrap TRAcP 5b ELISA, Immunodiagnostic Systems SB-TR103), CTX-1 (Mouse CTX ELISA Kit, Immunodiagnostic Systems AC-06F1), and P1NP (Rat/Mouse P1NP EIA, Immunodiagnostic Systems AC-33F1) were measured by ELISA according to manufacturer’s instructions. Each sample was run in duplicate and then averaged. Serum concentrations were determined using a standard curve as recommended by the manufacturer.
Micro-computed tomography
Tibial micro-computed tomography (μCT) analyses were performed at MaineHealth Institute for Research. Bones were fixed in 10% formalin and placed on a rocker for 24 h before being rinsed, placed in 70% ethanol, and stored at 4°C. All bones were scanned using the same instrument under the same conditions, in accordance with the American Society for Bone and Mineral Research guidelines.88 A high-resolution desktop micro-tomographic imaging system (μCT40, Scanco Medical AG, Brüttisellen, Switzerland) was used to assess trabecular bone microarchitecture and cortical bone morphology in the proximal metaphysis and mid-diaphysis of the tibia. This machine was also used to assess trabecular bone microarchitecture and cortical bone morphology in the distal metaphysis and mid-diaphysis of the tibia, respectively.
Dynamic and static histomorphometry
Prior to euthanasia, mice were IP injected with calcein (20 mg/kg) at intervals dependent on age and in accordance with double labeling protocols.89 Bones were fixed in 10% formalin and placed on a rocker for 24 h before being rinsed, placed in 70% ethanol, and stored at 4°C in amber vials (12 mL vials, Agilent Technologies 5183-4322). Following μCT analysis, femurs were embedded in plastic and sectioned for static and dynamic histomorphometry. All static and dynamic histomorphometric analyses were performed on femurs and reported according to the criteria previously established by the American Society for Bone Mineral Research and in accordance with the Skeletal Research Histomorphometry Core at Massachusetts General Hospital.90 Static parameters were measured in the distal femoral metaphysis 0.3 mm below the epiphyseal growth plate, using an OsteoMeasure image analyzer (Osteometrics, Atlanta, GA, USA). Measurements of mineral apposition rate, mineralized surface per bone surface (MS/BS), and bone formation rate (BFR/BS) were obtained for trabecular and cortical bone sections based on calcein labels using the OsteoMeasure (version 1.0.4.4, OsteoMetrics, Decatur, GA).
Histology and immunohistochemistry (IHC)
Tibial and white adipose tissue H&E
Bones and visceral, gonadal white adipose tissue (gWAT) were fixed in 10% formalin for 24 h and 72 h, respectively. Tissues were then rinsed and stored in 70% ethanol at 4°C. Bones were decalcified in 14% EDTA after 8-10 days by changing the solution every other day and rocking the bones at 4°C. Bones and WAT were paraffin-embedded and sectioned at 5 and 10 μm thickness, respectively. Bones were sectioned in the coronal orientation. Slides were then stained with hematoxylin and eosin (H&E) and mounted with resin-based medium. H&E slides were used to quantify adipocyte ghosts residing in the bone marrow and adipose tissues.
Tartrate-resistant acid phosphatase (TRAP) and F4/80 IHC in femurs and white adipose tissue
Bones and gWAT were fixed in 10% formalin for 24 and 72 h, respectively, then rinsed and stored in 70% ethanol at 4 ℃. Bones were decalcified in 14% EDTA after 8-10 days by changing the solution every other day and rocking the bones at 4°C. Tissues were paraffin-embedded and sectioned at 5 μm thickness. Femurs were double-labeled using F4/80 immunostaining and TRAP enzyme staining, and F4/80 immunostaining was performed on gWAT. For F4/80 IHC, slides were deparaffinized and then rehydrated through xylene and graded ethanol. 3% hydrogen peroxide in PBS was applied for 10 minutes to quench peroxidases, and enzymatic epitope retrieval was performed using trypsin (Abcam ab970) for 2 minutes at RT, and non-specific binding was blocked using 5% normal goat serum (Abcam ab7481) in 0.1% PBS-Triton X100 for 30 minutes at RT. The F4/80 antibody (BioRad MCA497GA) was diluted 1:2 000 in blocking buffer, then slides were incubated with the antibody overnight at 4°C. Slides were washed in PBS-Tx, and anti-rat HRP polymer (Abcam ab214882) was applied for 30 minutes. After washing, DAB substrate was applied for 5 minutes before rinsing and proceeding to TRAP staining. TRAP staining buffer was prepared following the manufacturer’s instructions (Sigma 387 A). Slides were placed in pre-warmed TRAP staining solution, incubated at 37°C for 30 minutes, then rinsed in distilled water. Slides were counterstained with hematoxylin and mounted in aqueous media.
Cell quantification
Image acquisition
Histologic slides were photographed using a Keyence Fluorescence Microscope BZ-X800 (10x plan fluor PhL, Brightfield/Phase contrast) using an automated image capture software, BZ-X Analyzer, to create high-resolution images. The images were exported as .tiff files prior to quantification/analysis. These images were also “image-stitched” with the BZ-X Analyzer software to produce a composite image of the entire slide.
Quantification of adipocytes within the bone marrow and white adipose tissue
Adipocytes were quantified as described.72 In brief, a blinded analysis of BM adipocytes and gWAT was performed using Fiji/ImageJ software (version 2.16.0/1.54p). A global scale was set based on the microscope imaging objective and settings. Color images were converted to 32-bit, and then a threshold using the Huang filter was applied. Adipocytes were analyzed based on size and circularity. Yellow overlay masks identifying adipocytes were used for visibility purposes (Fig. S18a, b). For each sample, 2-3 sequential sections were quantified and then averaged.
Quantification of F4/80+ Cells
F4/80+ macrophages were manually counted using the Cell Counter plugin in ImageJ/Fiji. For each image, twelve 3.3 × 3.3 mm2 regions of interest were used (indicated by the blue squares) (Fig. S18c). The number of F4/80+ macrophages within each region of interest was counted (indicated by the red arrow heads), then averaged per sample (Fig. S18d). The results were displayed as number per mm2.
Quantification of TRAP+ Cells
TRAP+ cells from in vitro experiments were quantified with Fiji/ImageJ. A global scale was set based on the microscope imaging objective and settings. A threshold was applied to the color image using Adjust Color threshold. The thresholding method was set to Huang, the threshold color was red, the color space was HSB, and dark background was checked. The parameters were adjusted until TRAP+ cells were highlighted in red. Images were converted to a binary image, then analyzed based on size and circularity. Yellow overlay masks identifying cells were used for visibility purposes (Fig. S18e). When determining TRAP+ cell number and size, two wells were quantified and then averaged for each sample/condition.
In vitro experiments
For BM cultures, red blood cells were lysed using 1 mL per sample of RT ACK lysing buffer (Thermo Scientific A1049201). After several minutes, cells were poured over a 40 μm filter into 20 mL of DPBS, then centrifuged at 500 g for 5 minutes. Base media consisted of filtered α-MEM supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin. Osteoclast differentiation was stimulated by supplementing our base media with m-CSF (25 ng/mL; PeproTech 315-02) and RANKL (50 ng/mL; PeproTech 310-01). For all osteoclast differentiation studies, BM CD11b+ myeloid cells were magnetic-activated cell sorted (MACS) from the BM using a negative sorting method with Anti-PE microbeads (Miltenyi Biotec 130-048-801) and MACS cell separation LS columns (Miltenyi Biotec 130-042-401) in accordance with the manufacturer’s instructions prior to plating.
BM adipocyte and osteoclast co-culture
CD11b+ myeloid cells sorted from the BM of LFD and OB-HFD B6 mice were plated in a 24-well plate with osteoclast differentiation media (Day 0) with 24-well transwell inserts. BM adipocytes were separated from the BM cells of LFD and HFD-fed B6 mice using centrifugation, as described,91 then added inside the transwell inserts. This method of isolating BM adipocytes results in a heterogeneous population, and due to the fragility of BM adipocytes, there is no method, currently, to purify these samples. However, we have optimized this isolation method to maximize the collection of mature adipocytes and minimize contamination of other bone marrow cells.91 Myeloid cells and BM adipocytes were co-cultured for 3 days, at which time a media change occurred (Day 3). On day 6, osteoclast cultures were fixed with 2.5% glutaraldehyde in PBS, then stained for TRAP (Sigma 387 A).
Anti-PD-1 and anti-PD-L1 inhibitory osteoclast cultures
CD11b+ myeloid cells were sorted from the BM of chow-fed, 18–20-week-old B6 mice (established colony within the Rosen Laboratory at MaineHealth Institute for Research) and plated in a 24-well plate with osteoclast differentiation media. The myeloid cells were then treated with IgG2a and IgG2b (Bio X Cell BE0089 and BE0090), Anti-PD1 (Clone 29 F.1A12, Bio X Cell BE0273), or Anti-PDL1 (Clone 10 F.9G2, Bio X Cell BE0101) at concentrations of 10 ng/mL from Days 0-3 (early-stage osteoclast differentiation) or Days 3-6 (late-stage osteoclast differentiation). Media changes occurred on days 3 and 6. On day 7, TRIzol was added for subsequent RNA extraction, or osteoclast cultures were fixed with 2.5% glutaraldehyde, then stained for TRAP.
Mixed lymphocyte reaction (MLR)
Using a crude isolation method, naïve splenic T-cells were isolated from LFD-fed mice, then stained with CellTrace CFSE (Thermo Scientific C34554). Intracellular enzymes convert CellTrace CFSE into a fluorescent form with an emission wavelength of 488 nm. As cells divide, the dye is equally distributed between the daughter cells, causing the total fluorescence intensity to be cut in half with each new generation. After confirming with flow cytometry that the cells were stained, but not proliferating due to the lack of additional mean fluorescent intensity peaks, we activated the T-cells with CD3/CD28 Dynabeads (Thermo Scientific 11452D). Activated T-cells were plated in a 96-well round-bottom plate. Then, T-cells were cultured with BM CD11b+ myeloid cells isolated from LFD-fed or OB-HFD B6 mice at a 2:1 ratio (T-cells:Myeloid cells). T-cell proliferation was measured using flow cytometry after 5 days.
Quantitative RT-PCR (qRT-PCR)
Total RNA was extracted from cultured cells, isolated BM adipocytes,91 and visceral gonadal WAT (gWAT) using TRIzol (Thermo Fischer Scientific). A total of 500 ng of RNA was converted to cDNA (High-Capacity cDNA Kit; Invitrogen 4368813), then diluted 1:10. Quantification of mRNA expression was carried out using AzuraQuant Green Fast qPCR Mix (Azura Genomics AZ-2120), and thermocycling reactions were completed using a CFX-384 (Bio-Rad Laboratories). Data were analyzed using Bio-Rad CFX Manager 3.1 and Excel using the delta-delta CT method. Technical duplicates were run, then averaged for each biological data point. TATA-binding protein (TBP) and β-Actin were used as housekeeping genes.
Flow cytometric analysis
BM cells from LFD-fed and OB-HFD mice were used for flow cytometric analysis as described.92 In brief, red blood cells were lysed using 1 mL per sample of RT ACK lysing buffer. After several minutes, cells were poured over a 40 μm filter into 20 mL of DPBS, then centrifuged at 500 g for 5 minutes. Cells were resuspended in cold FACS buffer (PBS/0.5% BSA/2 mmol/L EDTA) and counted using Trypan. A cell concentration of 106 cells/mL was stained for cell-surface antigens and used for flow cytometric analysis. Data acquisition was performed on a MacsQuant Analyzer 10 (Miltenyi Biotec., Inc.), and the data were analyzed using WinList or FlowJo 10.10.0 software. Viable and non-viable cells were distinguished using DAPI (to detect dead nucleated cells) (Miltenyi Biotec 130-111-570).
Statistical analyses
All data were graphed and analyzed using GraphPad Prism 10.4.1 software. Data are visualized as individual data points with an error bar that represents the mean or as averaged data bars with error bars that represent the standard deviation (SD), unless otherwise noted. To determine the appropriate statistical analysis, Shapiro-Wilk normality testing was performed. Based on normality testing, the appropriate parametric or nonparametric statistical tests were performed. Two variable analyses (i.e., effect between LFD and HFD-fed mice) were performed as an unpaired t-test or Mann-Whitney test. When comparing the three variables (i.e., effect between LFD, OB-HFD, and NO-HFD mice), a one-way ANOVA with Tukey’s multiple comparison test or a Kruskal-Wallis with Dunn’s multiple comparison test was performed, unless otherwise noted. When comparing multiple variables (e.g., effect of genotype and diet), a 2-way ANOVA with Tukey’s multiple comparison, Sidak’s multiple comparison, or uncorrected Fisher’s least significant difference (LSD) test was performed. The appropriate post-hoc analyses used for one-way and 2-way ANOVAs are indicated within the figure legends. Significance was defined as having a P-value < 0.05, and the numerical P-values are shown.
References
Islam, A. N. M. S. et al. The global burden of overweight-obesity and its association with economic status, benefiting from STEPs survey of WHO member states: a meta-analysis. Prev. Med. Rep. 46, 102882 (2024).
Cao, J. J. Effects of obesity on bone metabolism. J. Orthop. Surg. Res. 6, 1–7 (2011).
Gower, B. A. & Casazza, K. Divergent effects of obesity on bone health. J. Clin. Densitom. 16, 450 (2013).
Rinonapoli, G. et al. Obesity and bone: a complex relationship. Int. J. Mol. Sci. 22, 13662 (2021).
Iwaniec, U. T. & Turner, R. T. Influence of body weight on bone mass, architecture and turnover. J. Endocrinol. 230, R115–R130 (2016).
Jensen, V. F. H., Mølck, A. M., Dalgaard, M., McGuigan, F. E. & Akesson, K. E. Changes in bone mass associated with obesity and weight loss in humans: applicability of animal models. Bone 145, 115781 (2021).
Halade, G. V., El Jamali, A., Williams, P. J., Fajardo, R. J. & Fernandes, G. Obesity-mediated inflammatory microenvironment stimulates osteoclastogenesis and bone loss in mice. Exp. Gerontol. 46, 43–52 (2011).
Tencerova, M. et al. High-fat diet–induced obesity promotes expansion of bone marrow adipose tissue and impairs skeletal stem cell functions in mice. J. Bone Miner. Res. 33, 1154–1165 (2018).
Doucette, C. R. et al. A high fat diet increases bone marrow adipose tissue (MAT) but does not alter trabecular or cortical bone mass in C57BL/6J mice. J. Cell Physiol. 230, 2032–2037 (2015).
De Paula, F. J. A. & Rosen, C. J. Marrow adipocytes: origin, structure, and function. Annu. Rev. Physiol. 82, 461–484 (2020).
Suchacki, K. J., Cawthorn, W. P. & Rosen, C. J. Bone marrow adipose tissue: formation, function and regulation. Curr. Opin. Pharmacol. 28, 50–56 (2016).
Devlin, M. J. & Rosen, C. J. The bone-fat interface: basic and clinical implications of marrow adiposity. Lancet Diabetes Endocrinol. 3, 141–147 (2015).
Zhao, E. et al. Bone marrow and the control of immunity. Cell. Mol. Immunol. 9, 11–19 (2012).
Kawai, T., Autieri, M. V. & Scalia, R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol. Cell Physiol. 320, C375–C391 (2021).
Man, K., Kallies, A. & Vasanthakumar, A. Resident and migratory adipose immune cells control systemic metabolism and thermogenesis. Cell. Mol. Immunol. 19, 421–431 (2021).
Nagareddy, P. R. et al. Adipose tissue macrophages promote myelopoiesis and monocytosis in obesity. Cell Metab. 19, 821 (2014).
Li, X. et al. Adipose tissue macrophages as potential targets for obesity and metabolic diseases. Front. Immunol. 14, 1153915 (2023).
Surmi, B. K. & Hasty, A. H. Macrophage infiltration into adipose tissue: initiation, propagation and remodeling. Future Lipidol. 3, 545 (2008).
Cronkite, D. A. & Strutt, T. M. The regulation of inflammation by innate and adaptive lymphocytes. J. Immunol. Res. 2018, 1467538 (2018).
Wang, R. et al. The interaction of innate immune and adaptive immune system. MedComm 5, e714 (2024).
Andersen, C. J., Murphy, K. E. & Fernandez, M. L. Impact of obesity and metabolic syndrome on immunity. Adv. Nutr. 7, 66–75 (2016).
Rius, B. et al. Resolution of inflammation in obesity-induced liver disease. Front. Immunol. 3, 257 (2012).
Painter, S. D., Ovsyannikova, I. G. & Poland, G. A. The weight of obesity on the human immune response to vaccination. Vaccine 33, 4422 (2015).
Shaikh, S. R., MacIver, N. J. & Beck, M. A. Obesity dysregulates the immune response to influenza infection and vaccination through metabolic and inflammatory mechanisms. Annu. Rev. Nutr. 42, 67 (2022).
Rohm, T. V., Meier, D. T., Olefsky, J. M. & Donath, M. Y. Inflammation in obesity, diabetes, and related disorders. Immunity 55, 31–55 (2022).
Singer, K. et al. Diet-induced obesity promotes myelopoiesis in hematopoietic stem cells. Mol. Metab. 3, 664–675 (2014).
Han, Y., Liu, D. & Li, L. PD-1/PD-L1 pathway: current researches in cancer. Am. J. Cancer Res. 10, 727 (2020).
Li, W. et al. Correlation between PD-1/PD-L1 expression and polarization in tumor-associated macrophages: a key player in tumor immunotherapy. Cytokine Growth Factor Rev. 67, 49–57 (2022).
Lin, X. et al. Regulatory mechanisms of PD-1/PD-L1 in cancers. Mol. Cancer 23, 1–50 (2024).
Sun, Y. et al. Macrophage-osteoclast associations: origin, polarization, and subgroups. Front. Immunol. 12, 778078 (2021).
Koza, R. A. et al. Changes in gene expression foreshadow diet-induced obesity in genetically identical mice. PLoS Genet. 2, e81 (2006).
Yazbek, S. N., Spiezio, S. H., Nadeau, J. H. & Buchner, D. A. Ancestral paternal genotype controls body weight and food intake for multiple generations. Hum. Mol. Genet. 19, 4134 (2010).
Chusyd, D. E., Wang, D., Huffman, D. M. & Nagy, T. R. Relationships between rodent white adipose fat pads and human white adipose fat depots. Front. Nutr. 3, 186521 (2016).
Börgeson, E., Boucher, J. & Hagberg, C. E. Of mice and men: pinpointing species differences in adipose tissue biology. Front. Cell Dev. Biol. 10, 1003118 (2022).
Ellacott, K. L. J., Murphy, J. G., Marks, D. L. & Cone, R. D. Obesity-induced inflammation in white adipose tissue is attenuated by loss of melanocortin-3 receptor signaling. Endocrinology 148, 6186–6194 (2007).
Chait, A. & den Hartigh, L. J. Adipose tissue distribution, inflammation and its metabolic consequences, including diabetes and cardiovascular disease. Front. Cardiovasc. Med. 7, 22 (2020).
Visser, M., Bouter, L. M., McQuillan, G. M., Wener, M. H. & Harris, T. B. Elevated C-reactive protein levels in overweight and obese adults. JAMA 282, 2131–2135 (1999).
Yang, R. Z. et al. Acute-phase serum amyloid A: an inflammatory adipokine and potential link between obesity and its metabolic complications. PLoS Med. 3, 0884–0894 (2006).
Weisberg, S. P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Amano, S. U. et al. Local proliferation of macrophages contributes to obesity-associated adipose tissue inflammation. Cell Metab. 19, 162–171 (2014).
Patsoukis, N., Wang, Q., Strauss, L. & Boussiotis, V. A. Revisiting the PD-1 pathway. Sci. Adv. 6, eabd2712 (2020).
Li, Z. et al. Lipolysis of bone marrow adipocytes is required to fuel bone and the marrow niche during energy deficits. Elife 11, e78496 (2022).
Li, Z. et al. Constitutive bone marrow adipocytes suppress local bone formation. JCI Insight 7, e160915 (2022).
Cao, J. J., Gregoire, B. R. & Gao, H. High-fat diet decreases cancellous bone mass but has no effect on cortical bone mass in the tibia in mice. Bone 44, 1097–1104 (2009).
Gautam, J. et al. Micro-architectural changes in cancellous bone differ in female and male C57BL/6 mice with high-fat diet-induced low bone mineral density. Br. J. Nutr. 111, 1811–1821 (2014).
Scheller, E. L. et al. Changes in skeletal integrity and marrow adiposity during high-fat diet and after weight loss. Front. Endocrinol. 7, 102 (2016).
Suresh, S., Alvarez, J. C., Dey, S. & Noguchi, C. T. Erythropoietin-induced changes in bone and bone marrow in mouse models of diet-induced obesity. Int. J. Mol. Sci. 21, 1657 (2020).
Song, W. et al. Obesity, but not high-fat diet, is associated with bone loss that is reversed via CD4+CD25+Foxp3+ Tregs-mediated gut microbiome of non-obese mice. npj Sci. Food 7, 1–14 (2023).
Myers, M. G., Leibel, R. L., Seeley, R. J. & Schwartz, M. W. Obesity and leptin resistance: distinguishing cause from effect. Trends Endocrinol. Metab. 21, 643 (2010).
Costa, S. N. et al. Altered metabolomics and inflammatory transcriptomics in human bone marrow adipocytes after acute high calorie diet and acute fasting. Front. Endocrinol. 16, 1591280 (2025).
Wan, S., Xie, J., Liang, Y. & Yu, X. Pathological roles of bone marrow adipocyte-derived monocyte chemotactic protein-1 in type 2 diabetic mice. Cell Death Discov. 9, 412 (2023).
Ferland-McCollough, D. et al. MCP-1 feedback loop between adipocytes and mesenchymal stromal cells causes fat accumulation and contributes to hematopoietic stem cell rarefaction in the bone marrow of patients with diabetes. Diabetes 67, 1380–1394 (2018).
Miyamoto, K. et al. MCP-1 expressed by osteoclasts stimulates osteoclastogenesis in an autocrine/paracrine manner. Biochem. Biophys. Res. Commun. 383, 373–377 (2009).
Joseph, G. J., Johnson, D. B. & Johnson, R. W. Immune checkpoint inhibitors in bone metastasis: clinical challenges, toxicities, and mechanisms. J. Bone Oncol. 43, 100505 (2023).
Sun, C., Mezzadra, R. & Schumacher, T. N. Regulation and function of the PD-L1 checkpoint. Immunity 48, 434–452 (2018).
Bader, J. E. et al. Obesity induces PD-1 on macrophages to suppress anti-tumour immunity. Nature 630, 968–975 (2024).
Wang, K. et al. PD-1 blockade inhibits osteoclast formation and murine bone cancer pain. J. Clin. Invest. 130, 3603 (2020).
Zuo, H. & Wan, Y. Inhibition of myeloid PD-L1 suppresses osteoclastogenesis and cancer bone metastasis. Cancer Gene Ther. 29, 1342–1354 (2022).
Ren, Y., Bäcker, H., Müller, M. & Kienzle, A. The role of myeloid derived suppressor cells in musculoskeletal disorders. Front. Immunol. 14, 1139683 (2023).
Wang, L., Lan, J., Tang, J. & Luo, N. MCP-1 targeting: shutting off an engine for tumor development. Oncol. Lett. 23, 26 (2021).
del Fresno, C. & Sancho, D. Myeloid cells in sensing of tissue damage. Curr. Opin. Immunol. 68, 34–40 (2021).
Reyes, M. et al. White adipose tissue-infiltrated CD11b+ myeloid cells are a source of S100A4, a new potential marker of hepatic damage. Eur. J. Endocrinol. 184, 533–541 (2021).
Devêvre, E. F. et al. Profiling of the three circulating monocyte subpopulations in human obesity. J. Immunol. 194, 3917–3923 (2015).
Friedrich, K. et al. Perturbation of the monocyte compartment in human obesity. Front. Immunol. 10, 458434 (2019).
Krinninger, P. et al. Peripheral monocytes of obese women display increased chemokine receptor expression and migration capacity. J. Clin. Endocrinol. Metab. 99, 2500–2509 (2014).
Caetano-Lopes, J., Canhão, H. & Fonseca, J. E. Osteoimmunology—the hidden immune regulation of bone. Autoimmun. Rev. 8, 250–255 (2009).
del Fattore, A. & Teti, A. The tight relationship between osteoclasts and the immune system. Inflamm. Allergy Drug Targets 11, 181–187 (2012).
Veldhuis-Vlug, A. G. & Rosen, C. J. Clinical implications of bone marrow adiposity. J. Intern. Med. 283, 121–139 (2018).
Austin, M. J., Kalampalika, F., Cawthorn, W. P. & Patel, B. Turning the spotlight on bone marrow adipocytes in haematological malignancy and non-malignant conditions. Br. J. Haematol. 201, 605 (2023).
Caers, J. et al. Neighboring adipocytes participate in the bone marrow microenvironment of multiple myeloma cells. Leukemia 21, 1580–1584 (2007).
Shafat, M. S. et al. Leukemic blasts program bone marrow adipocytes to generate a protumoral microenvironment. Blood 129, 1320–1332 (2017).
Costa, S., Fairfield, H. & Reagan, M. R. M. R. Inverse correlation between trabecular bone volume and bone marrow adipose tissue in rats treated with osteoanabolic agents. Bone 123, 211–223 (2019).
Costa, S. et al. Sclerostin antibody increases trabecular bone and bone mechanical properties by increasing osteoblast activity damaged by whole-body irradiation in mice. Bone 147, 115918 (2021).
Nardone, V., D’Asta, F. & Brandi, M. L. Pharmacological management of osteogenesis. Clinics 69, 438 (2014).
Ominsky, M. S., Boyce, R. W., Li, X. & Ke, H. Z. Effects of sclerostin antibodies in animal models of osteoporosis. Bone 96, 63–75 (2017).
Li, X. et al. Sclerostin antibody treatment increases bone formation, bone mass, and bone strength in a rat model of postmenopausal osteoporosis. J. Bone Miner. Res. 24, 578–588 (2009).
Jilka, R. L. Molecular and cellular mechanisms of the anabolic effect of intermittent PTH. Bone 40, 1434–1446 (2007).
Sandovici, I., Morais, T., Constância, M. & Monteiro, M. P. Epigenetic changes associated with obesity-related metabolic comorbidities. J. Endocr. Soc. 9, bvaf129 (2025).
Dekkers, K. F. et al. Blood lipids influence DNA methylation in circulating cells. Genome Biol. 17, 138 (2016).
Kasinska, M. A., Drzewoski, J. & Sliwinska, A. Epigenetic modifications in adipose tissue—relation to obesity and diabetes. Arch. Med. Sci. 12, 1293–1301 (2016).
Zhou, C., Gao, Y., Ding, P., Wu, T. & Ji, G. The role of CXCL family members in different diseases. Cell Death Discov. 9, 1–12 (2023).
Hardaway, A. L., Herroon, M. K., Rajagurubandara, E. & Podgorski, I. Marrow adipocyte-derived CXCL1 and CXCL2 contribute to osteolysis in metastatic prostate cancer. Clin. Exp. Metastasis 32, 353–368 (2015).
Lotfi, N. et al. Roles of GM-CSF in the pathogenesis of autoimmune diseases: an update. Front. Immunol. 10, 1265 (2019).
Yu, D. et al. CCL3 in the bone marrow microenvironment causes bone loss and bone marrow adiposity in aged mice. JCI Insight 8, e159107 (2023).
Kennedy, A. et al. Impact of macrophage inflammatory protein-1α deficiency on atherosclerotic lesion formation, hepatic steatosis, and adipose tissue expansion. PLoS One 7, e31508 (2012).
Chan, P. C., Lu, C. H., Chien, H. C., Tian, Y. F. & Hsieh, P. S. Adipose tissue-derived CCL5 enhances local pro-inflammatory monocytic MDSCs accumulation and inflammation via CCR5 receptor in high-fat diet-fed mice. Int. J. Mol. Sci. 23, 14226 (2022).
Wang, Z. et al. Paradoxical effects of obesity on T cell function during tumor progression and PD-1 checkpoint blockade. Nat. Med. 25, 141–151 (2019).
Bouxsein, M. L. et al. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Miner. Res. 25, 1468–1486 (2010).
Vignery, A. & Baron, R. Dynamic histomorphometry of alveolar bone remodeling in the adult rat. Anat. Rec. 196, 191–200 (1980).
Dempster, D. W. et al. Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 28, 2–17 (2013).
Costa, S. N., Chlebek, C. & Rosen, C. J. Isolation and characterization of primary bone marrow adipocytes in rodent models. Methods Mol. Biol. 2885, 87–98 (2025).
Yin, H. et al. Protective role of ErbB3 signaling in myeloid cells during adaptation to cardiac pressure overload. J. Mol. Cell Cardiol. 152, 1 (2020).
Acknowledgements
This study was supported by the NIAMS grant RO1 AR073774 (C.J.R.) and the NHLBI grant R01 HL136560 (S.R.). The NMR body composition data reported here were supported by the services of the Physiology Core of the COBRE in Mesenchymal and Neural Regulation of Metabolic Networks (2P20GM121301). μCT analyses and images were performed and generated by the Small Animal Imaging Core at MaineHealth Institute for Research, which is supported by the Northern New England Clinical and Translational Research Network (U54 GM115516 from the National Institutes of Health). The histological processing was performed by the COBRE Histology and Imaging Core at the University of New England, Biddeford, Maine (NIGMS P30GM103643). Static and dynamic histomorphometry analyses were performed at Massachusetts General Hospital, Center for Skeletal Research, which is supported by an NIH-funded program (NIAMS P30 AR075042).
Author information
Authors and Affiliations
Contributions
S.N.C. is responsible for the conceptualization, methodology, validation, formal analysis, investigation, drafting the original manuscript, visualization, and project administration. C.C. is responsible for investigation and visualization. L.G. is responsible for the investigation. P.C. is responsible for the visualization of histological images. S.R. is responsible for the conceptualization, validation, formal analysis, investigation, visualization, and funding acquisition. C.J.R. is responsible for conceptualization, validation, supervision, and funding acquisition. All authors revised and approved this manuscript for publication. S.R. and C.J.R. are the corresponding authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Costa, S.N., Chlebek, C., Gray, L. et al. Expansion of bone marrow adipocytes in obese mice leads to PD-L1-driven bone marrow immunosuppression and osteoclastogenesis. Bone Res 14, 32 (2026). https://doi.org/10.1038/s41413-026-00509-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41413-026-00509-5