Abstract
AgCu(Te, Se, S) alloys, as one of the rare p-type plastic inorganic thermoelectrics, are receiving striking attention for their application foreground in high-performing flexible thermoelectric generators. However, strategies to enhance their thermoelectric performance while maintaining exceptional plasticity remain largely unexplored. Here, we introduce a strategic vacancy-engineering approach to address this challenge. Using computational design as a guide, we carefully tune the cation vacancy concentration to optimize hole carrier concentration, achieving impressive ZTs of ~0.62 at 300 K and ~0.83 at 343 K in (AgCu)0.998Te0.8Se0.1S0.1, ranking among the highest in this class of material. Importantly, numerous diffuse Ag-S bonds combined with amorphous phase introdeuced by vacancy engineering ensure that (AgCu)0.998Te0.8Se0.1S0.1 retains high plasticity while having high performance. A novel flexible thermoelectric device, comprising ductile p-type (AgCu)0.998Te0.8Se0.1S0.1 and n-type commercial Bi2Te3, achieves an impressive power density of ~126 μW cm−2 under 25 K temperature difference, demonstrating significant application prospects for wearable electronics.
Similar content being viewed by others
Introduction
The depletion of natural fossil fuels, coupled with the resulting environmental challenges, has made the development of renewable energy technologies both necessary and urgent. Among the various green energy conversion technologies, thermoelectric systems have drawn widespread attention over the past few decades in the light of their unique ability to achieve direct and reversible energy conversion without generating pollution, noise, and requiring moving parts1,2,3,4,5. In traditional inorganic thermoelectric materials, the thermoelectric potential is typically parameterized by the dimensionless figure of merit, defined as6,7 \({ZT}=\frac{{S}^{2}\sigma }{\kappa }T=\frac{{S}^{2}\sigma }{{\kappa }_{l}+{\kappa }_{e}}T\), where σ, S, T, κ, κl, and κe represent the electrical conductivity, Seebeck coefficient, absolute temperature, total thermal conductivity, lattice thermal conductivity, and electronic thermal conductivity, respectively. The S2σ, known as the power factor, is a crucial parameter related to electrical transport. Meanwhile, the κ consists of two different parts: the electronic contribution (κe) and lattice vibration (κl)8. Since the σ, S, and κe are interdependent through carrier concentration (n), improving the ZT typically requires optimizing n via methods such as doping or alloying to achieve an ideal S2σ9. Simultaneously, to reduce κl and enhance the ZT, it is crucial to introduce lattice defects of varying dimensions that effectively scatter phonons across different wavelengths, without significantly impairing carrier mobility (μ)10.
With the swift advance of flexible electronics, the demand for flexible thermoelectric devices (F-TEDs) is growing significantly11. These devices must deliver both high thermoelectric performance and flexibility at ambient condition, enabling stable and efficient heat absorption from irregular heat sources for power generation or cooling on uneven surfaces12,13. Mainstream F-TEDs are currently fabricated using inorganic thin-film thermoelectric materials14,15,16, organic thermoelectric materials deposited on flexible substrates, and hybrid composites of both17,18. However, organic materials typically suffer from low S, resulting in ZT values that are several orders of magnitude lower than those of inorganic materials19. Even when organic/inorganic composites are engineered to boost the S, the improvement remains limited, and overall ZT performance is suboptimal20. Although inorganic thermoelectric materials pose a substantial in thermoelectric performance, undesirable brittleness restricts the further development of F-TEDs that heavily rely on these materials11,21.
To tackle this issue, recent discoveries have revealed that certain inorganic semiconductors, such as ZnS22, Ag (S, Se, Te) alloys23,24,25,26,27, Mg3(Bi, Sb)228,29, and van der Waals crystals like InSe30,31 and SnSe232, exhibit exceptional plastic deformability. Among these innovative plastic inorganic materials, Ag2(Te, Se, S)25,26 and Mg3(Bi, Sb)216,28 demonstrate moderate thermoelectric performance at ambient temperature, with ZT values of approximately 0.43 and 0.72, respectively. While these values significantly exceed those of organic materials, they still fall short compared to other conventional thermoelectric materials33,34,35. Furthermore, compared to the n-type Ag2(S, Se, Te) thermoelectrics, p-type pseudo-ternary AgCu(Se, S, Te) thermoelectric materials have recently been reported to exhibit more complex phase compositions, crystal structures, and enhanced physical properties25,26,27,36. AgCu(Se, S, Te) alloys achieved a maximum ZT of ~0.45 at 300 K, which is comparable to that of Ag2(S, Se, Te) pseudo-ternary alloys37,38,39,40. Additionally, F-TEDs fabricated using p-type AgCu(Se, S, Te) materials demonstrated a normalized power density of ~30 µW cm−2 K−2, surpassing that of organic F-TEDs25. However, pure AgCuTe is unstable at room temperature and typically contains trace amounts of α-Ag2Te as a secondary phase41. The presence of more compositional elements, alongside various compound defects caused by Cu vacancies or Ag interstitials, makes the physical properties of AgCu(Se, S, Te)-based alloys highly sensitive to phase composition. This sensitivity complicates efforts to effectively enhance the thermoelectric and mechanical properties of this material system, especially when attempting to improve ZT while maintaining high plasticity40. Consequently, discovering effective methods to significantly boost near-room-temperature thermoelectric performance compromising its plasticity has emerged as a critical research focus within the realm of AgCu(Se, S, Te) materials25,26,36.
Results and discussion
High thermoelectric performance and excellent plasticity in AgCu(Te, Se, S) alloys
To achieve a significant enhancement in near-room-temperature thermoelectric performance while maintaining high plasticity through simple compositional adjustments, this study employed a multi-step optimization process. First, element Se was introduced at the Te sites in AgCuTe to reduce the content of the secondary phase α-Ag2Te (Supplementary Fig. 1a). This modification resulted in a decrease in hole carrier density (nh) from 9.8 × 1018 cm−3 in undoped AgCuTe to 6.5 × 1018 cm−3 in AgCuTe0.9Se0.1, achieving a ZT value of 0.12 at 343 K (Supplementary Fig. 2f). Subsequently, S was introduced into the composition of AgCuTe0.9Se0.1 to further enhance the solubility of the secondary phase within the AgCuTe0.9Se0.1 matrix, adjusting the nh of AgCuTe0.8Se0.1S0.1 to 2.6 × 1018 cm−3 (Supplementary Fig. 3a). Additionally, the decrease in μ after introducing (Se, S) indicates enhanced alloy scattering of charge carriers in the AgCuTe0.9−ySe0.1Sy samples (Supplementary Fig. 3a). Next, vacancy engineering was applied to synergistically adjust the thermal and electrical transport properties of AgCuTe0.8Se0.1S0.1.
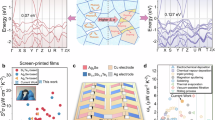
As shown in Fig. 1a, microstructural analysis confirms that (AgCu)0.998Te0.8Se0.1S0.1 contains point defects and multi-dimensional crystals, including nanophases and Cu-rich regions formed through vacancy engineering. Along with grain and phase boundaries, these features effectively scatter heat-carrying phonons of different wavelengths, reducing κl across operating temperatures. However, nanophases and lattice defects also act as charge carrier scattering centers, slightly decreasing μ and impacting electron transport properties. Moreover, incorporating S into the AgCuTe0.9Se0.1 matrix introduces numerous multi-center and diffuse Ag-S bonds, which are widely distributed and drive a brittleness-to-ductility transition23,25. These bonds increase cleavage energy, preventing fracture, while reducing slip energy barriers, allowing easier movement of atoms, defects, and dislocations, thereby enhancing mechanical properties23,26. Experimentally, the introduction of S significantly enhances the mechanical properties of AgCu(Te, Se, S) alloys. In the three-point bending test, the engineering strain increases from ~1% in AgCuTe0.9Se0.1 to ~10% in AgCuTe0.8Se0.1S0.1 (Fig. 1b). Additionally, all S-containing AgCu(Te, Se, S) alloys show excellent plastic deformation, achieving over 15% engineering compressive strain (Supplementary Fig. 4). As group VI chalcogens, Se and S substitution at Te sites in the AgCuTe lattice does not act as donor or acceptor dopants. Instead, incorporating Se and S into AgCuTe can: (i) suppress secondary phase formation, aiding n regulation; (ii) introduce point defects, enhancing phonon scattering; and (iii) form multi-center and diffuse Ag-S bonds distributed throughout the matrix, significantly improving mechanical properties. Figure 1c illustrates the significant deformability of this material, which can be manually bent to various extents, highlighting its potential in flexible electronics. Consequently, (AgCu)0.998Te0.8Se0.1S0.1 achieved a peak ZT value of ~0.62 at 300 K and ~0.83 at 343 K, the highest recorded for p-type ductile AgCu(Se, S, Te) alloys25,26,27 (Supplementary Fig. 5). In contrast, the cost-effective AgCu(Te, Se, S) alloys achieved a feasible combination of high ductility and high thermoelectric performance at room temperature, exceeding typical Bi2Te3-based materials42,43, ductile Ag2(Te, Se, S) alloys37,38,39,44,45,46,47, SnSe2-based materials48, and organic thermoelectric materials49,50,51,52, and matching up to the highest thermoelectric performance level of Mg3(Bi, Sb)2 and defective Bi2Te3 ductile inorganic materials28,29,53 (Fig. 1d).
a Schematic diagram of the microstructural characteristics of the as-prepared material (AgCu)0.998Te0.8Se0.1S0.1. b Engineering stress-strain curves for three-point bending tests for pristine AgCuTe and (AgCu)1−xTe0.9−ySe0.1Sy (x = 0, 0.002 and y = 0, 0.05, 0.08, 0.10, 0.12, 0.15, and 0.20) at room temperature. c Optical images of the (AgCu)0.998Te0.8Se0.1S0.1 materials with a thickness of 0.3 mm, showing good flexibility. d Figure of merit (ZT) and Seebeck coefficients (S) for this work compared with the reported works on ductile inorganic25,26,27,28,29,37,38,39,44,45,46,47,48,53, organic49,50,51,52, and typical Bi2Te3-based thermoelectrics42,43.
First-principles density functional theory (DFT) calculations were conducted on AgCu(Te, Se, S) alloys to elucidate the roles of (Se, S) and Ag/Cu vacancies in the thermoelectric performance of AgCuTe. Based on the hexagonal crystal structure, Fig. 2a–c display the calculated band structures for pristine AgCuTe (Ag24Cu24Te24), AgCuTe0.8Se0.1S0.1 (Ag24Cu24Te18Se3S3), and (AgCu)0.998Te0.8Se0.1S0.1 (Ag23Cu23Te18Se3S3). While the corresponding projected density of states (DOS) in Fig. 2d–f indicates that the maximum value of the valence band (VB) in all AgCu(Te, Se, S) materials is primarily derived from Te states, with Ag and Cu sites located deeper in the VB. This suggests that electrical transport is predominantly governed by the rigid anionic Te sublattice. The calculated band structures of AgCuTe0.9Se0.1 and AgCuTe0.9−ySe0.1Sy (y = 0.05, 0.10, 0.15, 0.20) are presented in Supplementary Fig. 6. First-principles calculations reveal that the electronic structure of hexagonal AgCuTe is metallic (Fig. 2a), resulting in a high μ of ~575 cm2 V−1 s−1 at 300 K, comparable to AgCuSe, which also exhibits a metallic electronic structure24,54,55. The incorporation of (Se, S) effectively modulates the band structure of AgCuTe, as evidenced by changes in the density-of-state (DOS) effective mass (m*) and deformation potential (Edef) of AgCuTe0.9−ySe0.1Sy (Supplementary Fig. 3b). Notably, AgCuTe0.8Se0.1S0.1 achieves maximum values for m* and minimum values for Edef, indicating weak scattering intensity between hole carriers and phonons. Consequently, with the increase of S and a decrease in σ, AgCuTe0.8Se0.1S0.1 obtained a ZT value of ~0.68 at 343 K (Supplementary Fig. 7c). Furthermore, Ag/Cu vacancies can gradually reduce the bandgap of AgCuTe0.8Se0.1S0.1, ultimately leading to (AgCu)0.996Te0.8Se0.1S0.1 (Ag21Cu21Te18Se3S3) exhibiting a vanishing bandgap (Supplementary Fig. 8b). This indicates that the introduced vacancies significantly increased the hole concentration, thereby enhancing σ.
a–f Calculated electronic band structures and projected density of states (DOS) of AgCuTe (Ag24Cu24Te24), AgCuTe0.8Se0.1S0.1 (Ag24Cu24Te18Se3S3) and (AgCu)0.998Te0.8Se0.1S0.1 (Ag23Cu23Te18Se3S3).
Structural characterizations of AgCu(Te, Se, S) alloys
To investigate the crystal structure and phase composition of AgCuTe0.9−ySe0.1Sy (y = 0, 0.05, 0.08, 0.10, 0.12, 0.15, and 0.20) and (AgCu)1−xTe0.8Se0.1S0.1 (x = 0, 0.002, 0.003, 0.004), X-ray diffraction (XRD) was performed on all samples at room temperature, with results displayed in Fig. 3 and Supplementary Fig. 9. As shown in Fig. 3a, the primary diffraction peaks of pristine AgCuTe are classified as belonging to the hexagonal phase, while additional diffraction peaks attributed to the α-Ag2Te phase (P121/C1) are observed as a common secondary phase within the matrix41. Notably, the intensity of these secondary phase peaks diminishes as the introduction of 10% Se, indicating that Se doping effectively suppresses the formation of the secondary phase in the AgCuTe lattice. Furthermore, the complete disappearance of the second-phase peaks after adding S to the AgCuTe0.9Se0.1 matrix demonstrates that S significantly increases the solubility of α-Ag2Te in the AgCuTe0.9Se0.1 lattice. The diffraction patterns for both AgCuTe0.9−ySe0.1Sy and (AgCu)1−xTe0.8Se0.1S0.1 samples are primarily attributed to the main hexagonal phase. For (AgCu)1−xTe0.8Se0.1S0.1 (x = 0, 0.002, 0.003, 0.004), the peaks gradually shift to the right with increasing Ag/Cu vacancy concentration (Fig. 3b), indicative of lattice contraction. Since the atomic radius of S (1.04 Å) is smaller than that of Te (1.37 Å), increasing S content in AgCu(Te, Se, S) alloys contracts the lattice. As a result, diffraction peaks shift from lower to higher angles (Supplementary Fig. 9b), and lattice parameters gradually decrease (Supplementary Fig. 9c) as S increases to 10% and Te decreases to 80%. Further S addition does not change the parameters, indicating a solubility limit of ~10% for S in AgCuTe0.9Se0.1. Figure 3c–f presents the backscattered electron (BSE) images from scanning electron microscopy (SEM) of the polished surfaces of AgCuTe0.8Se0.1S0.1 and (AgCu)0.998Te0.8Se0.1S0.1 (the insets show secondary electron (SE) images), along with the corresponding energy-dispersive X-ray spectroscopy (EDS) images. As noted, the addition of S and Se almost entirely suppresses the formation of the secondary phase in the AgCuTe0.8Se0.1S0.1 matrix, leading to a homogeneous distribution of elements within the AgCuTe0.8Se0.1S0.1 matrix (Fig. 3c). EDS mapping confirms that Se is homogeneously distributed, indicating that 10% Se fully substitutes Te and dissolves into the AgCuTe matrix, effectively suppressing secondary phase formation (Supplementary Fig. 1b). In contrast, S shows an inhomogeneous distribution in AgCuTe0.75Se0.1S0.15 and AgCuTe0.7Se0.1S0.2 (Supplementary Figs. 1c and 1d), suggesting a solubility limit of ~10% for S in AgCuTe0.9Se0.1. Interestingly, the introduction of Ag/Cu vacancies results in the emergence of a small amount of Cu-rich phase (Fig. 3d) (the relevant EDS point scan results for AgCuTe, AgCuTe0.8Se0.1S0.1, and (AgCu)0.998Te0.8Se0.1S0.1 are shown in Supplementary Fig. 10), which may contribute to the increased nh and the relatively low κl. Figure 3g–i displays the SEM-SE images of (AgCu)0.998Te0.8Se0.1S0.1 along different fracture directions. The grain distribution exhibits no significant orientation, indicating that the material displays an isotropic polycrystalline structure.
a X-ray diffraction (XRD) and b magnified XRD patterns of AgCuTe, AgCuTe0.9Se0.1, and AgCuTe0.8Se0.1S0.1 and (AgCu)1−xTe0.8Se0.1S0.1 (x = 0, 0.002, 0.003, and 0.004). Backscattering electron (BSE) images for c AgCuTe0.8Se0.1S0.1 and d (AgCu)0.998Te0.8Se0.1S0.1 and e–f corresponding energy dispersive X-ray spectroscopy (EDS) elemental mapping results. g–i The SEM-SE (secondary electron) images of (AgCu)0.998Te0.8Se0.1S0.1 with different fracture directions.
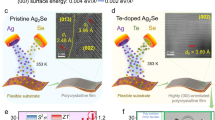
We also examined transmission electron microscopy (TEM) characterization on the sample (AgCu)0.998Te0.8Se0.1S0.1 to determine the microstructure and nanostructure of the material with introduced vacancies. Figure 4a, b displays low-magnification TEM images of (AgCu)0.998Te0.8Se0.1S0.1 and the corresponding selected area electron diffraction (SAED) pattern, indicating that the image was taken along the [0\(\bar{1}\)1] zone axis of hexagonal AgCuTe. Notably, Fig. 4c reveals regions with distinct contrast, suggesting a multiphase structure at this magnification, consistent with the SEM images of (AgCu)0.998Te0.8Se0.1S0.1. The corresponding SAED pattern for Fig. 4c is presented in Fig. 4d, exhibiting clear evidence of a clear nanocrystalline or multiphase structure. Figure 4e exhibits a high-resolution TEM (HRTEM) image taken from a regular area of Fig. 4a, revealing a clear lattice structure that indicates excellent crystallinity of the phase. However, despite the overall ordered arrangement of the lattice, some lattice distortions are evident in the image. The corresponding strain maps, shown in Fig. 4f and derived from Fig. 4e, indicate strong strains in different directions, which lead to lattice distortions. These distortions may be attributed to point defects, such as cation vacancies or the substitution of S and Se for Te. Figure 4g displays a magnified HRTEM image of grain boundary between two grains of (AgCu)0.998Te0.8Se0.1S0.1 along with the corresponding fast Fourier transform (FFT) patterns of the two grains. The lattice mismatch at the grain boundary arises from orientation differences between adjacent nanocrystalline grains. Additionally, high magnification scanning transmission electron microscopy (STEM) image and the corresponding EDS images (Fig. 4i) confirm that the dark field regions in Fig. 4h are predominantly composed of Cu, Se, and S, indicating the formation of a nanoscale secondary phase. This nanoscale secondary phase may result from local nanoscale segregation due to the introduction of cation vacancies. Thus, the presence of various types of lattice defects—including multiple point defects, grain boundaries, phase boundaries, and nanoscale precipitates—collectively contributes to phonon scattering sources. This scattering can affect phonons of different wavelengths, resulting in a lower κl and a slight decrease in μ.
a, b Low- and c, d high-magnification transmission electron microscopy (TEM) images and corresponding select area electron diffraction (SAED) patterns of the samples prepared using focused ion beam (FIB) technology. e High-resolution TEM (HRTEM) image taken from a normal area. f Corresponding strain maps. g HRTEM image taken from the grain boundary. h–i Scanning transmission electron microscopy (STEM) image and corresponding EDS maps of Ag, Cu, Te, Se, S.
Thermoelectric properties of (AgCu)1−xTe0.8Se0.1S0.1 alloys
To unravel the impact of rationally introducing Ag/Cu vacancies on the thermoelectric transport properties of AgCuTe0.8Se0.1S0.1, we assessed the thermoelectric performance of (AgCu)1−xTe0.8Se0.1S0.1 (x = 0, 0.002, 0.003, and 0.004) over a temperature range of 300 to 343 K. All samples were prepared using a melting-annealing-quenching-sintering method, indicating that they had not yet reached their final thermodynamic equilibrium state. Consequently, when the temperature reached the phase transition temperature and subsequently decreased to room temperature, the phase composition of these materials would change. Therefore, within the 300–343 K temperature range, the thermoelectric performance of these samples exhibited high reproducibility and stability, as the highest measured temperature remained below the phase transition temperature. Supplementary Figs. 11 and 12 illustrate the thermoelectric performance of AgCuTe0.9−ySe0.1Sy (y = 0, 0.05, 0.08, 0.10, 0.12, 0.15, and 0.20). Among these samples containing S, the one with a nominal composition of AgCuTe0.8Se0.1S0.1 achieved a superior ZT value of approximately 0.68 at 343 K. Compared to the predicted ZT values, the thermoelectric performance based on AgCuTe0.8Se0.1S0.1 can be further improved (Supplementary Fig. 7c). In semiconductors, vacancies are stable point defects formed when atoms or ions leave their lattice positions. They distort the crystal structure and affect electrical and thermal transport properties. Understanding vacancy formation and evolution is key to precisely controlling vacancy concentration in thermoelectric materials. As an essential optimization strategy, vacancy engineering helps balance electrical and thermal transport. Based on charge counting, each Ag or Cu vacancy generates one hole in AgCu(Te, Se, S) alloys. Introducing a small number of Ag/Cu vacancies in AgCuTe0.8Se0.1S0.1 optimizes n, enhancing electrical performance. At the same time, these vacancies act as phonon scattering centers, reducing high-frequency phonon transport and lowering κl. Figure 5a shows the variation of the S with temperature. All samples exhibit positive S, confirming their p-type semiconductor behavior. As the concentration of Ag/Cu vacancies increases, the S decreases, although all samples show an increasing trend in S at the operating temperature. The peak S of AgCuTe0.8Se0.1S0.1 is 256.63 µV K−1 at 343 K. Figure 5b depicts the variation of σ with temperature. With the increasing fraction of Ag/Cu vacancies, the σ significantly increased from 116.146 cm−1 to 214.08 S cm−1 at 300 K. Although S decrease slightly, the product S2σ improved from 7.10 µW cm−1 K−2 at x = 0 to 8.15 µW cm−1 K−2 at x = 0.002 at room temperature (Fig. 5c).
Temperature-dependent a Seebeck coefficients (S), 2% error bar is employed. b Electrical conductivity (σ), 5% error bar is employed. c Power factor (S2σ), 7% error bar is employed. d Measured room-temperature carrier concentration (nh) and carrier mobility (µ), 5% error bars are employed. e Calculated room-temperature density-of-state effective mass (m*) and deformation potential (Edef) via single parabolic band (SPB) model. Temperature-dependent f total thermal conductivity (κ) and g lattice thermal conductivity (κl), 3% error bars are employed. h Figure of merit (ZT), 10% error bar is employed. i Comparison of predicted ZT by the single parabolic band (SPB) model and measured ZT as a function of nh.
To comprehend the experimentally acquired electrical transport performance, we measured the nh and µ at ambient temperature for all (AgCu)1−xTe0.8Se0.1S0.1 (x = 0, 0.002, 0.003, and 0.004) samples, as shown in Fig. 5d, to verify the impact of Ag/Cu vacancies on the electronic transport properties. It is evident that the introduced Ag/Cu vacancies create additional acceptor defects within the matrix, resulting in an increase in nh that leads to higher σ. Consequently, as x increases from 0 to 0.004, the nh rises significantly from 2.6 to 5.5 × 1018 cm−3. However, the µ slightly decreases from 278.81 cm2 V−1 s−1 for AgCuTe0.8Se0.1S0.1 to 242.96 cm2 V−1 s−1 for (AgCu)0.996Te0.8Se0.1S0.1. As shown in Fig. 1a, The reduction in µ may be ascribed to strengthened carrier scattering due to phase segregation and point defects, as well as a slight increase in carrier-carrier scattering. Thus, the enhancement of nh is answerable for the increase in σ. Based on the single parabolic band (SPB) model, Fig. 5e illustrates the relationship between m* and Edef. As the proportion of Ag/Cu vacancies increases, the m* initially rises before showing a downward trend, while the Edef exhibits a completely opposite trend. Additionally, both the m* and Edef reach their maximum and minimum values when the Ag/Cu vacancy concentration is 0.002, indicating a weaker scattering intensity between carriers and phonons. This allows the sample to achieve high S2σ while maintaining a relatively low κ.
Regarding the thermal transport properties, Fig. 5f shows the temperature-dependent κ values for (AgCu)1−xTe0.8Se0.1S0.1 (x = 0, 0.002, 0.003, and 0.004) in the operating temperature range of 300 to 343 K. It can be observed that the sample without vacancies exhibits the lowest κ. The introduction of Ag/Cu vacancies slightly increases the κ, which is attributed to the enhanced contribution from σ. To better understand the thermal transport characteristics, the κe was calculated using the formula κe = LσT. Here, L denotes the Lorenz number obtained through the SPB model. Figure 5g illustrates the κl calculated by subtracting κe from κ. All AgCu(Te, Se, S) alloys in this work show relatively low κl. As liquid-like materials, the local vibrations of Ag/Cu atoms in the hexagonal AgCuTe phase generate low-frequency optical phonons, contributing to the experimental heat capacity (Cp) at low temperatures41. These optical phonons strongly couple with acoustic phonons, softening the lattice and further reducing κl. As shown in Supplementary Fig. 13, since Se and S have lower atomic masses than Te, Cp gradually increases with the addition of Se and S across the studied temperature range. It can be observed that samples with an Ag/Cu vacancy ratio greater than 0.002 have a higher κl than other samples. Both AgCuTe0.8Se0.1S0.1 and (AgCu)0.998Te0.8Se0.1S0.1 exhibit the lowest κl of approximately 0.26 W m−1 K−1 at 343 K. The suppression of κl is primarily due to strong phonon scattering arising from phase segregation and enhanced point defects. Figure 5h shows the variation of the ZT with temperature from 300 to 343 K. Under favourable conditions with S2σ of 8.37 µW cm−1 K−2 and relatively low κl of 0.26 W m−1 K−1 at 343 K, (AgCu)0.998Te0.8Se0.1S0.1 demonstrates a ZT value of ~0.83 at 343 K, which is consistent with the predicted value (Fig. 5i), indicating that nh has been optimized to an optimal level in our case. Additionally, the ratio of weighted mobility (μw) to κl and the maximum engineering strain from three-point bending tests at room temperature serve as indicators for evaluating the synergy between thermoelectric and mechanical properties (Supplementary Fig. 14). Both AgCuTe0.8Se0.1S0.1 and (AgCu)0.998Te0.8Se0.1S0.1 exhibit significantly higher μw/κl and three-point bending strain compared to other AgCu(Te, Se, S) alloys in this study, confirming their superior thermoelectric and mechanical performance. The S2σ and κ of (AgCu)0.998Te0.8Se0.1S0.1 showed almost identical results across three measurements (Supplementary Fig. 15), confirming good thermal stability of the samples in the temperature range of 300 K to 343 K. In addition, the high reproducibility of the thermoelectric performance of (AgCu)0.998Te0.8Se0.1S0.1 was also investigated using three different batches of samples, as shown in Supplementary Fig. 16.
Prototype flexible thermoelectric modules based on (AgCu)1−xTe0.8Se0.1S0.1 alloys
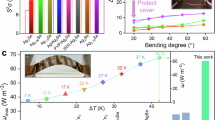
We also evaluated the mechanical properties of the ZT-optimized samples to demonstrate that this strategy does not significantly compromise the plasticity of the materials. The secondary phase Ag2Te in AgCuTe0.8Se0.1S0.1 is almost completely dissolved into the matrix. As shown in Fig. 3c, all elements are uniformly distributed, promoting the formation of multi-center and diffuse Ag-S bonds, which lower slip barrier energy and increase cleavage energy, enhancing mechanical stability during deformation. As seen in Fig. 1a, the large atomic mass difference between guest atoms (Se/S) and the host atom (Te) creates mass field fluctuations that effectively hinder phonon transport. While strain field fluctuations from atomic radius differences further enhance phonon scattering, they also disrupt the periodic potential field, reducing μ (Supplementary Fig. 3a). As a result, AgCuTe0.8Se0.1S0.1 achieves a ZT of 0.68 at 343 K (Supplementary Fig. 11f) due to an improved S2σ and reduced κl. Additionally, the widespread Ag-S bonds allow the material to sustain nearly 10% strain in three-point bending tests (Fig. 6a). Introducing Ag/Cu vacancies slightly reduces ductility due to point defect dispersion strengthening56, but with a low vacancy concentration (0.2%), (AgCu)0.998Te0.8Se0.1S0.1 achieves a higher ZT of 0.83 at 343 K and can endure over 8% strain before fracture. Moreover, the thermoelectric performance of (AgCu)0.998Te0.8Se0.1S0.1 remains stable and reproducible under repeated bending. As shown in Supplementary Fig. 17, a ~0.05 mm thick sample exhibits less than a 5% variation in S and σ after 100 bending cycles at a fixed angle. This result suggests that ductile AgCu(Te, Se, S) alloys can bend without fracturing, and the strain has minimal effect on electrical properties. Figure 6b, c shows the fracture morphologies of (AgCu)0.998Te0.8Se0.1S0.1 after bending and compression, revealing stepped and wavy features, which confirm its flexibility and deformation capability. Although AgCu(Te, Se, S) contains high-cost elements, it exhibits excellent thermoelectric and mechanical properties comparable to commercial Bi2Te3, making it a strong candidate for applications. Moreover, its cost-effective melting process makes it suitable for large-scale production.
a Engineering stress-strain curves for three-point bending tests for pristine AgCuTe, Se-doped sample, (Se, S)-doped sample and further vacancy-introduced sample. Fracture surface morphologies of (AgCu)0.998Te0.8Se0.1S0.1 after b bending test and c compression test. (d) Fabricated in-plane F-TEDs based on (AgCu)0.998Te0.8Se0.1S0.1 with its schematics at the top. e, f Measured voltages (V) and output powers (P) of the device as a function of loading current (Iload) at different temperature differences (ΔTs). g The measurement output power density (ω) varies with ΔT. h Optical image of the in-plane F-TED adhered to a human arm. i Variation of measured open-circuit voltage (Voc) over time for the in-plane F-TED adhered to the human arm under static state, moving and static state with alcohol sprayed at the bottom of the device.
The results indicate that the carefully optimized pseudo-ternary AgCu(Te, Se, S) solid solution exhibits good ductility and high thermoelectric performance, making it well-suited for F-TED applications. To verify the application potential of the as-designed AgCu(Te, Se, S) alloys, we used commercial Bi2Te3 as the n-type leg (its thermoelectric properties are shown in Supplementary Fig. 18) and (AgCu)0.998Te0.8Se0.1S0.1 as the p-type leg to fabricate a flexible in-plane F-TED with 6 pairs of thermoelectric legs. The schematic diagram of the manufactured F-TED is displayed in Fig. 6d. Additionally, based on the high-performance (AgCu)0.998Te0.8Se0.1S0.1, the prototype flexible in-plane and out-of-plane thermoelectric modules were fabricated (Supplementary Fig. 19). First, the n-type leg (commercial Bi2Te3) and the p-type leg (AgCu)0.998Te0.8Se0.1S0.1) were cut into thermoelectric legs measuring 2 × 0.5 × 10 mm3. Next, these thermoelectric legs were connected in the series to copper foil using high-conductivity silver paste. Finally, the legs were stuck to a flexible polyimide (PI) film substrate to obtain final flexible in-plane thermoelectric device. The optical images of the fabricated devices are shown in Fig. 6d and Supplementary Fig. 19. As illustrated in Fig. 6e-f, the open-circuit voltage (Voc) and maximum power output (Pmax) of the device increased with the temperature difference (ΔT), reaching 53.5 mV and 15.12 µW, respectively, at ΔT = 25 K.
Figure 6g shows the dependence of ω (power density: Pmax/A, where A represents the cross-sectional area of the thermoelectric legs) on the ΔT. The planar F-TED, composed of 6 pairs of AgCu(Te, Se, S) and Bi2Te3 legs, exhibited a maximum ω of approximately 126 μW cm−2 under a measured ΔT of 25 K. As shown in Fig. 6h, this planar F-TED can be easily attached to an arm using tape. As a continuous heat source, the human body naturally maintains a ΔT with its surroundings. Physical activity further increases heat generation, resulting in an even larger ΔT between the body and the environment. Using a portable infrared detection device, we can accurately and in real time quantify the ΔT under various motion states while wearing the device (Supplementary Fig. 20). Under ambient conditions, the Voc and Pmax of the device were approximately 8.5 mV and 0.39 μW, respectively (Supplementary Fig. 19a). By continuously operating the device to enhance convective heat transfer, a greater ΔT across the device can be achieved, resulting in increased Voc and Pmax values of approximately 18.5 mV and 1.86 μW, respectively (Supplementary Fig. 19b). Spraying alcohol on the underside of the device induces an even greater ΔT, yielding higher Voc and Pmax values of about 48.5 mV and 13.7 μW, respectively (Supplementary Fig. 19c). Considering the length of the thermoelectric legs (Ld), the corresponding normalized power density ωn = 0.0126 W m−1. PmaxLd/A for typical in-plane F-TED is summarized in Supplementary Fig. 21. The ωn value for the in-plane F-TED based on 6 pairs of AgCu(Te, Se, S)/Bi2Te3 is significantly higher than that of organic in-plane F-TEDs57,58,59,60 and inorganic in-plane F-TEDs16. Figure 6i presents the time-voltage characteristics of the device, showing that the Voc remains relatively stable under different motion states, confirming the operational stability of the as-designed device. Although (AgCu)0.998Te0.8Se0.1S0.1 demonstrates high thermoelectric performance near room temperature, the interface between the material and the electrode remains a key limitation for in-plane and out-of-plane thermoelectric modules. Due to significant contact resistance between AgCu(Te, Se, S) alloys and electrodes, the internal resistance is approximately 7.5 Ω, 25 Ω, and 50 Ω in flexible in-plane and out-of-plane thermoelectric modules. This leads to lower output performance compared to the theoretical output voltage and power (Supplementary Fig. 22). Given the high room-temperature ZT of the flexible p-type (AgCu)0.998Te0.8Se0.1S0.1, minimizing the interface resistance of the F-TED is anticipated to further enhance performance and facilitate its future applications in flexible electronics.
In summary, we have theoretically and experimentally investigated the effects of targeted vacancy engineering on the thermal and electrical transport properties of AgCuTe0.8Se0.1S0.1. The finely tuned Ag/Cu vacancies optimized the hole carrier density, significantly enhancing the σ. Additionally, the formation of multi-scale secondary phases and the introduction of point defects, driven by vacancy engineering, contribute to a low κl of ~0.26 W m−1 K−1 at 343 K. As a result, the synergistic effects of electronic and thermal transport properties in this multiphase material enable the p-type (AgCu)0.998Te0.8Se0.1S0.1 to achieve a peak ZT value of approximately 0.62 at 300 K and around 0.83 at 343 K, the highest values for ductile AgCu(Se, S, Te) alloys. The incorporation of S into AgCuTe0.9Se0.1 alloys induces a vast majority of multi-center and diffuse Ag-S bonds, significantly altering the mechanical properties and enhancing the ductility of AgCuTe0.9−ySe0.1Sy. This improvement is attributed to a decrease in slip barriers and an increase in cleavage energy, resulting in a near 10% bending strain in AgCuTe0.8Se0.1S0.1. Furthermore, the F-TEDs fabricated from these AgCu(Te, Se, S) materials exhibit considerable an ω of ~126 μW cm−2 under a ΔT of 25 K, highlighting their potential for wearable applications.
Methods
Materials synthesis
Pristine AgCuTe and (AgCu)1−xTe0.9−ySe0.1Sy (x = 0, 0.002, 0.003, 0.004 and y = 0, 0.05, 0.08, 0.10, 0.12, 0.15, 0.20) samples were synthesized through a melting-annealing-quenching method combined with spark plasma sintering (SPS). The detailed preparation process is illustrated in Supplementary Fig. 23. High-purity raw materials, including Ag (99.99%, Sigma-Aldrich, Australia), Cu (99.99%, Alfa Aesar, United States), Se (99.999%, Sigma-Aldrich, Australia), Te (99.999%, Sigma-Aldrich, Australia), and S (99.999%, Sigma-Aldrich, Australia), were precisely weighed based on predetermined stoichiometric ratios. To prevent reactions between the elements and the quartz tube, each element was loaded into quartz tubes lined with a thin layer of pyrolytic carbon and vacuum sealed. The tubes were gradually heated to 773 K over 12 h to minimize sulfur evaporation, then heated to 1273 K over 5 h and held at this temperature for 48 h. Subsequently, the tubes were cooled to 873 K for over 4 h, annealed at this temperature for 3 days, and then quenched into ice water. The materials were ground into powder using an agate mortar after cooling in liquid nitrogen. The powders were then packed into a 12.7 mm graphite die and sintered under vacuum using an SPS system (SUGA SPSS2200) at 773 K for 5 min under a pressure of ~4.0 kN. The obtained pellets were cut and polished into a cuboid of ~2.5 mm × 2.5 mm × 10 mm for electrical measurements and a disc with diameter of ~10 mm and thickness of ~1 mm for thermal and Hall measurements.
Structural characterizations
Grazing incidence XRD analysis was conducted using a Rigaku Smart Lab instrument with Cu Kα radiation over an angular range of 10° to 80° in 0.02° increments to determine the crystal orientation of pristine AgCuTe and (AgCu)1−xTe0.9−ySe0.1Sy (x = 0, 0.002, 0.003, 0.004 and y = 0, 0.05, 0.08, 0.10, 0.12, 0.15, 0.20) samples. Morphological analysis, mapping, and composition assessment were conducted using a Hitachi SU7000 SEM, while EDS was performed with an Oxford Ultim Max 100 EDS detector. The microstructures of (AgCu)0.998Te0.8Se0.1S0.1 sample were studied by a JEOL 2100 TEM.
Thermoelectric performance evaluation
The σ and S were simultaneously measured using a commercial ZEM-3 system (ULVAC) with measurement uncertainties of σ and S about ±5% and ±2%, respectively. The κ was determined by κ = DρCp, where D is the thermal diffusivity measured with a laser flash thermal analyzer (LFA 467, NETZSCH) (within ±3% uncertainty) under an argon atmosphere, Cp is the specific heat estimated via the Dulong-Petit approximation, and ρ is the volumetric density. The calculated uncertainty of ZT is within ±10%. The Hall coefficient (RH) was obtained using the van der Pauw method, with n calculated as n = 1/(eRH) and μ as μ = σRH, where e is the elementary charge. The bending test was conducted with a dynamic mechanical analyzer (DMA 850, TA Instrument) at room temperature with a loading rate of 0.1 mm/min. The specimens’ dimensions for the bending test were 1 × 0.5 × 10 mm3.
Module fabrication and test
High-performance p-type (AgCu)0.998Te0.8Se0.1S0.1 thermoelectric legs and n-type commercial Bi2Te3 with dimensions of 2 × 0.5 × 10 mm3 and 2 × 2 × 0.5 mm3 were used for fabricating in-plane and out-of-plane thermoelectric modules, respectively. The thickness of (AgCu)0.998Te0.8Se0.1S0.1 legs is first cut to 1 mm by a diamond-wire-saw machine and then by manually polishing to 0.5 mm. flexible PI films, Cu sheets and Cu wire were used as support bases and conducting wires. Then, high conductive Ag paste was used to connect thermoelectric legs and Cu sheets. A home-build instrument with a source meter (K2400, Keithley) was used to measure the output voltage and source current of the thermoelectric modules. The temperature gradient is applied along the length direction in the in-plane module and thickness direction in the out-of-plane module by one-side heating and the other-side cooling.
Data availability
The data generated in this study is provided in the Source Data file. Source data are provided with this paper.
References
Wang, D. et al. Multi-heterojunctioned plastics with high thermoelectric figure of merit. Nature 632, 528–535 (2024).
Xie, L. et al. Screening strategy for developing thermoelectric interface materials. Science 382, 921–928 (2023).
He, J. & Tritt, T. M. Advances in thermoelectric materials research: looking back and moving forward. Science 357, eaak9997 (2017).
Mao, J. et al. High thermoelectric cooling performance of n-type Mg3Bi2-based materials. Science 365, 495–498 (2019).
Qin, Y. et al. Grid-plainification enables medium-temperature PbSe thermoelectrics to cool better than Bi2Te3. Science 383, 1204–1209 (2024).
Li, N. et al. IV-VI/I-V-VI2 thermoelectrics: recent progress and perspectives. Adv. Funct. Mater. 34, 2405158 (2024).
Shi, X.-L. et al. Advancing flexible thermoelectrics for integrated electronics. Chem. Soc. Rev. 53, 9254–9305 (2024).
Snyder, G. J. & Toberer, E. S. Complex thermoelectric materials. Nat. Mater. 7, 105–114 (2008).
Massetti, M. et al. Unconventional thermoelectric materials for energy harvesting and sensing applications. Chem. Rev. 121, 12465–12547 (2021).
Shi, X.-L., Chen, W.-Y., Tao, X., Zou, J. & Chen, Z.-G. Rational structure design and manipulation advance SnSe thermoelectrics. Mater. Horiz. 7, 3065–3096 (2020).
Chen, W. et al. Nanobinders advance screen-printed flexible thermoelectrics. Science 386, 1265–1271 (2024).
Shi, X.-L. et al. Advances in flexible inorganic thermoelectrics. EcoEnergy 1, 296–343 (2023).
Shi, X.-L. et al. Weavable thermoelectrics: advances, controversies, and future developments. Mater. Futures 3, 012103 (2024).
Jin, Q. et al. Flexible layer-structured Bi2Te3 thermoelectric on a carbon nanotube scaffold. Nat. Mater. 18, 62–68 (2019).
Tang, X., Li, Z., Liu, W., Zhang, Q. & Uher, C. A comprehensive review on Bi2Te3-based thin films: Thermoelectrics and beyond. Interdiscipl Mater. 1, 88–115 (2022).
Hu, B. et al. High-performing flexible Mg3Bi2 thin-film thermoelectrics. Adv. Sci. 11, 2409788 (2024).
Kim, N. et al. Elastic conducting polymer composites in thermoelectric modules. Nat. Commun. 11, 1424 (2020).
Blackburn, J. L., Ferguson, A. J., Cho, C. & Grunlan, J. C. Carbon-nanotube-based thermoelectric materials and devices. Adv. Mater. 30, 1704386 (2018).
Xu, S. et al. Conducting polymer-based flexible thermoelectric materials and devices: from mechanisms to applications. Prog. Mater. Sci. 121, 100840 (2021).
Cao, T., Shi, X.-L. & Chen, Z.-G. Advances in the design and assembly of flexible thermoelectric device. Prog. Mater. Sci. 131, 101003 (2023).
Paul, B., Björk, E. M., Kumar, A., Lu, J. & Eklund, P. Nanoporous Ca3Co4O9 thin films for transferable thermoelectrics. ACS Appl Energy Mater. 1, 2261–2268 (2018).
Oshima, Y., Nakamura, A. & Matsunaga, K. Extraordinary plasticity of an inorganic semiconductor in darkness. Science 360, 772–774 (2018).
Shi, X. et al. Room-temperature ductile inorganic semiconductor. Nat. Mater. 17, 421–426 (2018).
Yang, Q. et al. Incommensurately modulated structure in AgCuSe-based thermoelectric materials for intriguing electrical, thermal, and mechanical properties. Small 19, 2300699 (2023).
Yang, Q. et al. Flexible thermoelectrics based on ductile semiconductors. Science 377, 854–858 (2022).
Shen, K. et al. Ductile P-type AgCu(Se,S,Te) thermoelectric materials. Adv. Mater. 36, 2407424 (2024).
Yin, T. et al. Effects of Ag off-stoichiometry on mechanical and thermoelectric properties of ductile AgCuSe0.6S0.4. Mater. Today Phys. 43, 101402 (2024).
Zhao, P. et al. Plasticity in single-crystalline Mg3Bi2 thermoelectric material. Nature 631, 777–782 (2024).
Li, A. et al. High performance magnesium-based plastic semiconductors for flexible thermoelectrics. Nat. Commun. 15, 5108 (2024).
Wong, L. W. et al. Deciphering the ultra-high plasticity in metal monochalcogenides. Nat. Mater. 23, 196–204 (2024).
Wei, T.-R. et al. Exceptional plasticity in the bulk single-crystalline van der Waals Semiconductor InSe. Science 369, 542 (2020).
Deng, T. et al. Plastic/ductile bulk 2D van der Waals single-crystalline SnSe2 for flexible thermoelectrics. Adv. Sci. 9, 2203436 (2022).
Hong, M., Li, M., Wang, Y., Shi, X.-L. & Chen, Z.-G. Advances in versatile GeTe thermoelectrics from materials to devices. Adv. Mater. 35, 2208272 (2023).
Zhang, D. et al. High thermoelectric performance in earth-abundant Cu3SbS4 by promoting doping efficiency via rational vacancy design. Adv. Funct. Mater. 33, 2214163 (2023).
Jiang, B. et al. High figure-of-merit and power generation in high-entropy GeTe-based thermoelectrics. Science 377, 208–213 (2022).
Li, N.-H., Zhang, Q., Shi, X.-L., Jiang, J. & Chen, Z.-G. Silver copper chalcogenide thermoelectrics: advance, controversy, and perspective. Adv. Mater. 36, 2313146 (2024).
Yang, S. et al. Ductile Ag20S7Te3 with excellent shape-conformability and high thermoelectric performance. Adv. Mater. 33, 2007681 (2021).
Chen, H. et al. High-entropy cubic pseudo-ternary Ag2(S, Se, Te) materials with excellent ductility and thermoelectric performance. Adv. Energy Mater. 14, 2303473 (2024).
Wu, H. et al. Optimized thermoelectric performance and plasticity of ductile semiconductor Ag2S0.5Se0.5 via dual-phase engineering. Adv. Energy Mater. 13, 2302551 (2023).
Wu, H. et al. Sn-doping-induced biphasic structure advances ductile Ag2S-based thermoelectrics. Adv. Sci. 11, 2408374 (2024).
Roychowdhury, S. et al. Soft phonon modes leading to ultralow thermal conductivity and high thermoelectric performance in AgCuTe. Angew. Chem. 130, 4107–4111 (2018).
Hu, L. et al. Leveraging deep levels in narrow bandgap Bi0.5Sb1.5Te3 for record-High zTave near room temperature. Adv. Funct. Mater. 30, 2005202 (2020).
Jabar, B. et al. Enhanced power factor and thermoelectric performance for n-type Bi2Te2.7Se0.3 based composites incorporated with 3D topological insulator nanoinclusions. Nano Energy 80, 105512 (2021).
Liang, J. et al. Flexible thermoelectrics: from silver chalcogenides to full-inorganic devices. Energy Environ. Sci. 12, 2983–2990 (2019).
Liang, J. et al. Crystalline structure-dependent mechanical and thermoelectric performance in Ag2Se1‐xSx system. Research 2020, 6591981 (2020).
Liang, X. & Chen, C. Ductile inorganic amorphous/crystalline composite Ag4TeS with phonon-glass electron-crystal transport behavior and excellent stability of high thermoelectric performance on plastic deformation. Acta Mater. 218, 117231 (2021).
Liang, J., Zhang, X. & Wan, C. From brittle to ductile: a scalable and tailorable all-inorganic semiconductor foil through a rolling process toward flexible thermoelectric modules. ACS Appl Mater. Interfaces 14, 52017–52024 (2022).
Deng, T. et al. High thermoelectric power factors in plastic/ductile bulk SnSe2-based crystals. Adv. Mater. 36, 2304219 (2024).
See, K. C. et al. Water-processable polymer−nanocrystal hybrids for thermoelectrics. Nano Lett. 10, 4664–4667 (2010).
Xuan, Y. et al. Thermoelectric properties of conducting polymers: The case of poly(3-hexylthiophene). Phys. Rev. B 82, 115454 (2010).
Ju, H. & Kim, J. Chemically exfoliated SnSe Nanosheets and Their SnSe/Poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) composite films for polymer based thermoelectric applications. ACS Nano 10, 5730–5739 (2016).
Zhang, K., Qiu, J. & Wang, S. Thermoelectric properties of PEDOT nanowire/PEDOT hybrids. Nanoscale 8, 8033–8041 (2016).
Deng, T. et al. Room-temperature exceptional plasticity in defective Bi2Te3-based bulk thermoelectric crystals. Science 386, 1112–1117 (2024).
Han, C. et al. Ambient scalable synthesis of surfactant-free thermoelectric CuAgSe nanoparticles with reversible metallic-n-p conductivity transition. J. Am. Chem. Soc. 136, 17626–17633 (2014).
Ishiwata, S. et al. Extremely high electron mobility in a phonon-glass semimetal. Nat. Mater. 12, 512–517 (2013).
Shi, X.-L. et al. Optimization of sodium hydroxide for securing high thermoelectric performance in polycrystalline Sn1-xSe via anisotropy and vacancy synergy. InfoMat 2, 1201–1215 (2020).
Li, C. et al. A simple thermoelectric device based on inorganic/organic composite thin film for energy harvesting. Chem. Eng. J. 320, 201–210 (2017).
Tang, J., Chen, R., Chen, L., Bazan, G. C. & Liang, Z. Semiconducting polymer contributes favorably to the Seebeck coefficient in multi-component, high-performance n-type thermoelectric nanocomposites. J. Mater. Chem. A 8, 9797–9805 (2020).
Chen, Y., He, M., Tang, J., Bazan, G. C. & Liang, Z. Flexible thermoelectric generators with ultrahigh output power enabled by magnetic field–aligned metallic nanowires. Adv. Electron Mater. 4, 1800200 (2018).
Zhou, W. et al. High-performance and compact-designed flexible thermoelectric modules enabled by a reticulate carbon nanotube architecture. Nat. Commun. 8, 14886 (2017).
Acknowledgements
This work was financially supported by the Australian Research Council and the QUT Capacity Building Professor Program. The authors thank National Computational Infrastructure for providing computation allocation (Project wk98), and the Australian Microscopy & Microanalysis Research Facility for providing characterization facilities. This work was enabled by the use of the Central Analytical Research Facility hosted by the Institute for Future Environments at QUT. We thank Wenyi Chen, Boxuan Hu, Yongqi Chen, Liangcao Yin, and Qingfeng Liu for their valuable discussions.
Author information
Authors and Affiliations
Contributions
Z.-G.C. and X.-L.S. supervised the whole project. N.-H.L. prepared materials and measured the thermoelectric properties, mechanical properties, carried out the scanning electron microscope, designed device structures, fabricated devices and measured the performance of thermoelectric modules. S.-Q.L. carried out the scanning transmission electron microscopy study. W.-Y. L. provided the sample of n-type Bi2Te3. N.-H.L., M.Z., T.-Y.C., W.-D.L., D.-C.Q., X.-L.S. and Z.-G.C. discussed the results. M.L. conducted the DFT calculations. N.-H.L., X.-L.S. and Z.-G.C. analyzed the data and wrote the manuscript. All the authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Nagendra S. Chauhan, Yoshiki Takagiwa, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, NH., Shi, XL., Liu, SQ. et al. Strategic vacancy engineering advances record-high ductile AgCu(Te, Se, S) thermoelectrics. Nat Commun 16, 2812 (2025). https://doi.org/10.1038/s41467-025-58104-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-58104-x