Abstract
Accurate detection of nostril airflow is vital for real-time respiratory monitoring. However, the developed methods only rely on single stimulus sensing for nostril airflow, which is extremely susceptible to interference in the complex environment, and severely affects the accuracy of detection results. Here, a multimodal integrated eutectogel sensor is explored to simultaneously sense the pressure and temperature stimuli of nostril airflow, by independently outputting capacitance and resistance, respectively, without cross-coupling. The completely physical crosslinking and the synergistic interaction of hydroxyapatite and tannic acid within the network endow this eutectogel with extremely low modulus, remarkable self-healing efficiency, robust adhesion, good environmental stability, and bio-compatibility. A multimodal sensor is developed by integrating this synthetic eutectogel with circuit design, which exhibits superior pressure sensitivity compared to other reported gel-based sensors. As a proof of concept, this sensor is further explored to diagnose the traditional respiratory disease of obstructive sleep apnea syndrome by simultaneously detecting five kinds of stimuli in the sleeping process, greatly improving the accuracy and reliability of the detection results. This work provides a highly effective strategy for achieving ultrasensitive respiratory monitoring and forecasting respiratory diseases.
Similar content being viewed by others
Introduction
Respiration is an essential behavior for sustaining human life1,2. Respiratory behavior reflects the human health and physical or mental activity3. Real-time respiration monitoring can forecast, rule out, and diagnose some related diseases, such as obstructive sleep apnea syndrome (OSAS), cardiovascular disorders (CVD), asthma, or epilepsy4,5. Hence, highly efficient respiratory monitoring is vital for managing personal health and even saving human lives. However, the commercially available polysomnography (PSG) equipment can only be used in hospitals for patients owing to its cumbersome operation, high cost, and poor wearability6, which severely restricts the flexibility and extensive of respiratory monitoring for people with different needs.
Developing wearable population-wide electronics for continuous respiratory monitoring is urgently needed. Flexible electronic sensors have recently attracted great attention in personal healthcare, electrical skins, and artificial intelligence, Researchers benefit from their ability to transform different stimuli (strain, pressure, thermal, humidity, etc.) into directly readable electrical signals (current, voltage, resistance, capacitance, etc.)4,7,8,9,10,11,12,13. Great efforts have been devoted to studying respiratory-related behaviors using different modal sensors, such as monitoring the nostril airflow9,12,14,15,16, or recording the chest and abdomen movement in the inhaling/exhaling process8,11. However, current works on respiratory monitoring usually focus on transforming a single stimulus from a nostril airflow or chest/abdomen movement, which is extremely susceptible to interference from an uncontrollable external environment. This significantly hinders its potential application in continuous respiratory monitoring and in-home healthcare. Compared to sensing the single stimuli of pressure variation for the chest/abdomen, the simultaneously detectable signals from the nostril airflow during breathing are much more diverse, such as the temperature, humidity, pressure, or gas. Furthermore, some common breath-related diseases (Nasal Polyps, Nasal Septum, etc.) can be directly diagnosed by measuring individual airflow signals from the two nostrils in clinical settings17. Therefore, effective monitoring of the nostril airflow is extremely important for evaluating respiration status. If two or more types of signals from the nostril airflow are simultaneously detected but independently output as different electrical signals by one integrated sensor, the obtained results will be much more reliable and accurate. On the one hand, it can eliminate the interference of the external environment with temperature or humidity when it just senses one stimulus signal from nostril airflow. On the other hand, it can effectively avoid invalid diagnoses induced by motion artifacts when only chest/abdomen variation is monitored, which is susceptible to random body movements, especially in long-term sleeping respiratory monitoring.
Additionally, wearable comfortability, simple fabrication, and high sensitivity of the resultant sensor are the other key parameters that will help the practicability of respiratory monitoring equipment. Compared to the classical metal18, conductive fabric8,12, layer-by-layer composited nanogenerator1,9,11, or on-mask based sensors19, the conductive hydrogel-based resistive/capacitive sensor that is directly adhered to the skin possesses great advantages. This includes simple assembly and much higher comfortability and wearability due to the bio-compatibility, bio-tissue compliance, and comparable modulus to the natural skin10,20,21,22,23. Although some works tried to monitor the respiration by recording the humidity variation of nostril airflow or the chest/abdomen movement4,24, the single detection signal severely reduced the reliability and accuracy of results as mentioned above. The key cause is that the reported sensor cannot meet the requirement of high-pressure sensitivity for the weak nostril airflow. In addition, the appropriate modulus, high tissue adhesion, good environmental stability, and fast self-healing rate are beneficial to the high sensitivity and long service life for respiratory sensors, which also remains a great challenge for the currently developed gel sensors, especially for the hydrogel-based devices. Hence, developing a high-performance respiratory monitoring sensor with independent/simultaneous sensing of multiple signals from nostril airflow, high sensitivity, and good comfortability and usability is highly desired for meeting the increasing requirement of respiratory monitoring.
Herein, a multimodal ultrasensitive eutectogel-based respiratory sensor was developed, which independently and simultaneously detected pressure and temperature variations from nostril airflow, with negligible cross-coupling between these two detected stimuli. This exciting characteristic is ascribed to the remarkable performance of the synthesized eutectogel combined with the adjustment of the circuit connection for this sensor. As depicted in Fig. 1, a poly (N-Acryloyl 2-Glycine) composited eutectogel (labeled as ATH) was developed by physically crosslinking with tannic acid (TA) using hydroxyapatite (HAp) as the crosslinker in the deep eutectic solvents (DESs) of choline chloride (ChCl) and glycol. The optimized ATH6 gel exhibited comparable low modulus and superior self-healing efficiency due to the complete physical crosslinking interactions of the gel network. The strong adhesive strength to bio-tissue and other substrates was achieved, attributed to the multiple strong interactions between the gel and substrates. Besides, the utilization of DESs and bio-friendly raw materials endowed this gel with high ionic conductivity, good environmental stability, and high cell viability. The resulting sensor was simply fabricated by sandwiching a dielectric layer between two gel layers and attaching three wires, integrating a resistive sensor into a capacitive sensor. Notably, the much higher pressure sensitivity of this sensor compared to other reported gel-based pressure sensors ensured the good sensing ability for the weak airflow pressure of nasal breathing due to the low modulus and high ionic conductivity of this eutectogel. Combing its good durability and bio-compatibility, this integrated eutectogel sensor shows great advantages in continuous respiratory monitoring, and significantly improves the accuracy and reliability of the detected result for diagnosing respiration-related diseases by simultaneously displaying five monitoring channels, effectively avoiding environmental interference and motion artifacts when only a single stimulus is detected.
This eutectogel is prepared by completely physical crosslinking via the multiple interaction of PACG, TA, HAp and the solvent of ChCl/EG, which displays extremely low modulus, remarkable self-healing efficiency, robust adhesion, good environmental stability, and bio-compatibility. A multimodal sensor is developed by integrating this synthetic eutectogel with circuit design, which exhibits superior pressure sensitivity and can simultaneously sense the pressure and temperature stimuli of nostril airflow, by independently outputting capacitance and resistance, respectively, with non-interference.
Results
Fabrication and Mechanical performance of ATH eutectogel
ATH eutectogel was synthesized through photo-initiated free radical polymerization by mixing ACG (N-Acryloyl 2-Glycine) monomer, TA, and HAp in the ChCl/ glycol (1:2, M/M) solvent upon 365-nm irradiation, which was labeled as ATHx (x represents the weight ratio of HAp to ACG). The detailed information is presented in the Experimental section and Supporting information (Supplementary Table S1). In this ATH gel, the HAp played a key role in the crosslinking interaction via the strong coordination bonds with the polymer chains. At the same time, the TA also contributed to the hydrogen bonding of the gel network. As shown in Fig. 2a, the crosslinking network could be generated in the ATH6 system, and no gel formation was observed without HAp (ATH0). This result was also verified by testing the storage modulus (G′) and loss modulus (G″) for the ATH6 and ATH0 systems, respectively (Fig. 2b). The higher value of G′ compared to G″ clearly demonstrated the formation of the crosslinked gel in ATH6. Subsequently, the mechanical performance of the ATH gels, such as tensile and compressive properties, were systematically measured. The tensile stress (Fig. 2c) and fracture toughness (Supplementary Fig. 1a) increased as the weight ratio of HAp was increased from 2% to 20% for ATHx. Similar results were obtained in the compressive curves (Fig. 2e) and Young′s modulus (Fig. 2h), attributed to the much higher crosslinking density with increasing HAp. The tensile fracture strain of ATH2 to ATH20 was from 1062% to 544%, and the corresponding tensile fracture stress was from 9.7 kPa to 75.4 kPa (Fig. 2c). The elasticity of ATH was evaluated by taking ATH6 as the sample. Only a little hysteresis loop was observed in the successive stepwise tensile and compressive curves (Fig. 2d, f). The tensile and compressive stress increased stepwise as the strain increased from 100% to 700% for tensile (Fig. 2d) and from 20% to 80% for compression (Fig. 2f), respectively, meanwhile displaying the steady cyclic curves at a certain strain (Supplementary Fig. 1b, c). Moreover, ATH6 could recover to its original shape without any fracture when applying successive compression at 80% strain for at least 30 cycles (Fig. 2g), demonstrating the good fatigue resistance and elasticity of the gel. Notably, the ATH (ATH2 ∼ ATH20) gel exhibited extremely lower Young′s modulus (1.48 kPa to 12.87 kPa) compared to other reported gels for electrode or sensor application (Fig. 2h, i)25,26,27,28,29,30,31, which was similar to that of soft biological tissues and was a benefit to the skin compliance of the gel. To demonstrate the advantages of eutectogel over hydrogel, the corresponding ATH6 hydrogel (labeled as Hydrogel in this paper) was prepared just by replacing the ChCl/glycol deep eutectic solvent with the same amount of H2O. It showed much higher tensile stress, Young′s and storage modulus than that of ATH6 eutectogel (Supplementary Fig. 2a, b, c). As shown in Fig. 2j, the ATH6 and ATH20 eutectogels could transfer the fingerprint, and the softer the gel, the cleaner the fingerprint, while it was difficult for the corresponding hydrogel. The good elasticity and soft modulus of ATH eutectogel are vital to capture the brittle pressure variation of nostril airflow and also important for the tightness of the skin-sensor interface.
The gelation behavior (a) and the rheology test (b) of ATH6 and ATH0. Scale bars: 2 cm. The tensile (c) and the compressive (e) stress-strain curves for ATHx when varying the HAp content from 2% to 20%. The stepwise tensile (d) and compressive (f) curves of ATH6 eutectogel. g The successive compressive test at 80% strain for ATH6 eutectogel. h The Young’s modulus for ATHx eutectogel. Data are presented as the mean values ± SD, n = 5 independent samples. i Comparison of Young′s modulus for this work with other reported related works. j The fingerprint transformation by using ATH6 and ATH20 eutectogels and the corresponding hydrogel.
Adhesive performance and bio-compatibility of ATH eutectogel
Self-adhesive performance is extremely vital for fixing the sensor on the skin and avoiding interference signals from motion artifacts. The standard lap-shear test was utilized to quantitatively evaluate the adhesion property of ATH eutectogel. As depicted in Fig. 3a, b, the shear stress of ATH eutectogel to porcine skin was first increased and then decreased as the HAp content increased. This is because the adhesive performance was co-influenced by the bulk strength of the gel and the interfacial strength. The ATH6 displayed the highest shear strength (34.6 kPa) because of its higher bulk strength compared to that of ATH2, while the stiffer ATH12 and ATH20 decreased its interfacial strength. Considering the mechanical strength and adhesive performance results, the ATH6 was chosen as the optimized sample for subsequent tests. ATH6 exhibited strong adhesion to the smooth skin and the hairy skin, and reached ultra-conformal and seamless contact with the skin surface which was characterized using an optical microscope (Fig. 3e). Moreover, different kinds of substrates could also adhere strongly with ATH6 eutectogel, which exhibited much higher adhesive capability than hydrogel, especially for the nonpolar substrates (Supplementary Fig. 3a–d). Additionally, different external mechanical stimulus (such as the twisted treatment or continuously bending) generates negligible influence on the adhesion of ATH6 eutectogel to porcine skin (Supplementary Fig. 3e, f and Movie 1). Although the sweat on the skin has negative influence on the adhesion performance of ATH6 eutectogel, the adhesion strength on the sweaty skin (30 μL sweat deposited on the porcine skin, area: 1 × 1 cm2) still attained 23 kPa which was higher than that of many adhesive gels. The influences of temperature and storage time were investigated to demonstrate the adhesive stability in different environments. Considering the respiratory monitoring application, the varied temperature range was from 37 °C to −20 °C (Fig. 3c, d). The shear strength of ATH6 eutectogel and hydrogel nearly kept the same value at 25 °C and 37 °C. For ATH6 eutectogel, the shear strength increased sharply at lower temperatures and reached as high as 125.7 kPa at −20 °C. This is due to more intense hydrogen interactions between the polymer chains at lower temperatures and its good anti-freezing performance (Supplementary Fig. 4a–c). In contrast, the adhesion strength of hydrogel was seriously degraded, and it was even lost below zero because of water freezing, which could be detected by differential scanning calorimetry (DSC). Meanwhile, no obvious shear strength loss was observed for ATH6 eutectogel with a prolonged storage time of at least one week. However, the hydrogel sample lost the adhesion capability after 1 day due to the evaporation of water in the open-air environment (Fig. 3f). The result of the environmental stability test for ATH6 eutectogel and hydrogel could support the above explanation. The eutectogel kept the original state and weight at RT and 40% relative humidity (RH), while almost all the water within the hydrogel evaporated and a film sample was left after one week (Fig. 3g and Supplementary Fig. 5a). In addition, the ATH6 eutectogel was also put in the outdoor from 8:00 a.m. to 8:00 p.m. to mimic the practical application environment. It also kept the original weight, but the hydrogel severely lost the solvent after 3 h (Supplementary Fig. 5b). The good adhesion stability and the high adhesion strength of ATH6 eutectogel were ascribed to the synergistic effect of low modulus, the abundant adhesive groups (carboxyl, catechol, amide) and the utilization of DESs. They not only formed multiple interactions (mechanical anchoring, hydrogen bonding, electrostatic interaction, metal coordination interaction, and van der Waals interactions) with different substrates, but also maintained the original network structure in different environments.
The curves of lap shear test (a) and the calculated shear strength (b) of ATH to porcine skin at RT. Data are presented as the mean values ± SD, n = 5 independent samples. The curves of lap shear test (c) and the calculated shear strength (d) of ATH6 to porcine skin at different temperatures. Data are presented as the mean values ± SD, n = 5 independent samples. e The visual demonstration of adhesive performance for ATH6 eutectogel to smooth skin and hairy skin, and the seamless adhesion to porcine skin. Scale bars: 1 mm. f The shear strength of ATH6 eutectogel and hydrogel increases the storage time. Data are presented as the mean values ± SD, n = 5 independent samples. g The pictures of ATH6 eutectogel and hydrogel at different storage times. The cell viability (L929) demonstration of ATH6 eutectogel by varying the different concentrations (h) and co-culturing time (i). Data are presented as the mean values ± SD, n = 5 independent samples. j Photographs of the mouse attached with ATH6 eutectogel before and after 2 days (left, Scale bars: 5 mm). HE staining images (Scale bars: 200 μm) of skin from the mice attached without (control) and with ATH6 eutectogel for 2 days. Three animals per group.
Biocompatibility is also a key parameter for the material that directly adheres to the skin in practical applications. As shown in Fig. 3h, i, the ATH6 eutectogel displayed high cell viability (L929) of 94% even at 500 μg/mL and similar metabolic activity with the control sample on days 1, 3, and 5. After 24 h of firm adhesion with the skin of a volunteer, ATH6 eutectogel could be peeled off without any residue and irritative reaction (Supplementary Fig. 6). The skin irritation test was supplemented to further demonstrate the bio-compatibility of eutectogel by animal experiment (approval number 2024b177). As shown in Fig. 3j, ATH6 eutectogel has adhered to the depilated dorsal skin of mice. After 2 days, no irritative reaction was observed and the HE (hematoxylin-eosin) staining analysis shows this eutectogel did not induce any structural change in the skin. The good biocompatibility of ATH6 eutectogel was ascribed to the utilization of bio-friendly crude materials and solvent, including the monomer of N-Acryloyl 2-Glycine (ACG), HAp, and TA, and the DESs solvent of choline chloride and ethylene glycol, ensuring the bio-safety in the following nostril airflow monitoring.
Self-healing performance of ATH6 eutectogel
The fast and efficient self-healing capability is important for the long service life of the device. The tensile test was utilized to quantitatively measure the self-healing efficiency of ATH6 eutectogel. It reached 96.5% of self-healing efficiency after 3 h at RT (Fig. 4a, b), superior to other reported self-healing gels (eutectogels, hydrogel, and organogels) and elastomers which needed longer healing time or additional stimulus to reach the high healing efficiency (Fig. 4c)32,33,34,35,36,37,38,39,40,41,42,43,44,45,46. Additionally, two pieces of ATH6 eutectogel could be integrated into one just by attaching them together gently (Fig. 4f and Supplementary Fig. 7). This self-healed gel still demonstrated good stretchability, even resisting the sharp scissor for the healed junction and inflating the gel balloon to 16 times of original volume (Fig. 4f), and also recovering to its original conductivity (Supplementary Fig. 8). Furthermore, the cycling strain sweeping was conducted from 0.1% to 300% to evaluate the rheological recovery behavior of ATH6 eutectogel (Supplementary Fig. 9). When the larger strain of 300% was applied, the value of G′ became smaller than that of G″ because of the collapse of the gel network. While retaining the sample at 300% strain only for 100 s, the G′ could completely return to the initial value immediately when the strain was back to 0.1% due to the rapid restoration of the gel structure. These periodical and stable changes in modulus further verified the fast reconstruction for the ATH6 gel network. Furthermore, the self-healing efficiency of ATH6 under different temperatures was quantitatively detected as shown in Fig. 4d, and achieving high healing efficiency even at −20 °C (73.25%) (Fig. 4e). This good self-healing capability was ascribed to the completely physical crosslinking interactions of hydrogen bonds and coordination bonds within the ATH6 which were dynamically reversible, endowing it with superior self-healing performance (Fig. 4g).
a The tensile stress-strain curves of original ATH6 and self-healed eutectogels. b The calculated self-healing efficiency of ATH6 eutectogel with different self-healing time at RT. Data are presented as the mean values ± SD, n = 5 independent samples. c The comparison of the self-healing efficiency and time of this work with the previously reported works. d The tensile stress-strain curves of original and self-healed ATH6 eutectogel at different self-healing temperatures. e The calculated self-healing efficiency of ATH6 eutectogel at different temperatures. Data are presented as the mean values ± SD, n = 5 independent samples. f The visual demonstration of self-healed ATH6 eutectogel which can resist different kinds of extreme mechanical stimulus. Scale bars: 5 mm. g The self-healing mechanism of ATH6 eutectogel.
Multiple sensory capability of ATH6 eutectogel sensor
A sandwiched structure was designed by sandwiching a dielectric film (the commercial VHB 4905) between two ATH6 eutectogels and embedding three metal electrodes to construct the dual-modal eutectogel sensor (Fig. 5a). When the two electrodes were connected to the same piece of eutectogel, a temperature-sensitive resistive sensor was fabricated, because the ionic dissociation and transportation within the gel were sensitive to temperature. Moreover, the resistance change ratio was calculated using the equation of ΔR/R0 = (Rx–R0)/R0, where Rx and R0 represented the initial and real-time resistance, respectively. The parallel-plate capacitive sensor was formed by connecting two electrodes to the separated gels at the top and bottom. The applied pressure could cause the distance variation between two electrodes and generate the capacitance change. Furthermore, the capacitance change value was defined as ΔC/C0 = (Cx–C0)/C0, the Cx and C0 correspond to the initial and real-time capacitance, respectively. To detect the sensory relationship between the stimulus of pressure/thermal and the electrical signal of resistance and capacitance, and considering the practical application of nostril airflow monitoring, this integrated sensor was compressed under 0–250 Pa at different temperatures from 20 to 35 °C by utilizing the compression and heating mode of the Dynamic Mechanical Analyzer (DMA). In these defined ranges of pressure and temperature variation, the resistive change of this integrated sensor displayed a linear increase with the increasing temperature, and nearly no response of resistive change when the pressure was varied from 0 to 250 Pa (Fig. 5b). While the capacitive change was mainly influenced by the pressure stimulus with negligible influence of temperature (Fig. 5c). Hence, this result demonstrates the pressure and thermal stimuli can be simultaneously but independently sensed by this one integrated sensor by outputting separate capacitive and resistive signals, respectively, via utilizing the different sensitivity of capacitive and resistive sensor to pressure and temperature in this defined range. Compared to the previously reported multimodal sensors, this work not only decouples the pressure and temperature by this integrated capacitive and resistive sensor at a certain range21,24,47,48, but also has the advantages of utilizing just one kind of eutectogel which has robust tissue adhesion, superior self-healing, and good bio-compatibility, and this extremely facile fabrication of sensor could avoid the complex fabrication and multiple functional materials49, which is easy for achievement transformation. This remarkable performance of this sensor provides the great possibility for independent and simultaneous sensing of pressure and temperature from nostril airflow.
a The schematic design of dual-modal eutectogel-based sensor, and the sensing mechanism of resistance and capacitance for this sensor upon thermal or pressure stimulus. The response curves of resistance (b) and capacitance (c) for pressure vibration from 0 to 250 Pa and temperature change from 20 to 35 °C.
Before simultaneously detecting the two signals from nostril airflow using this dual-modal eutectogel-based sensor, the separate sensing capability of pressure and temperature through capacitance and resistance was investigated, respectively. High-pressure sensitivity is vital for monitoring the weak pressure signal from nostril airflow. Hence, the pressure sensitivity S was first calculated by drawing the curve of the capacitance change versus pressure, and the slop of this curve was defined as S (0.42 kPa−1, R2 = 0.99, Fig. 6a). Additionally, the fast response time of 15 ms and recovery time of 17.5 ms were displayed when touching the sensor under a successively instantaneous pressing behavior, and the stable periodical signal of capacitive change was present (Fig. 6b). A radar map (Fig. 6c)13,24,37,48,50,51,52 and a detailed table (Supplementary Table 2) were made to compare the comprehensive performance of this work with other related gel or elastomer -based flexible sensors. This ATH6 based sensor could decouple the compressive pressure and temperature under a certain range, simultaneously possessing good self-adhesion performance and comparable pressure and thermal sensitivity. When a certain pressure from 40 Pa to 190 Pa was applied to the sensor, the capacitive change displayed stepwise increase with increasing the pressure and steady cyclic signals at a certain pressure, respectively (Fig. 6d). Even the subtle difference in the moving rate for the eyeball was clearly recognized from the capacitance signal when the sensor was adhered to the left eyelid, strongly demonstrating the high sensitivity of this sensor (Fig. 6e). Furthermore, the durability was tested by compressing this sensor at a simulating nostril airflow pressure of 70 Pa under the successive loading-unloading mode (Fig. 6f). The almost constant of capacitance change was observed at least 10,000th cycle, showing the superior durability and environmental stability of this sensor, due to the strong interactions between the gel network and the stable DESs. The above good pressure sensitivity and durability of this sensor are critical for the long-time service life and use-cost in its practical application.
a The sensing sensitivity (S) of capacitance for this sensor upon pressure stimulus from 0 to 200 Pa. b The response curve of capacitive variation when applying successively instantaneous pressing behavior to the sensor. c A radar map for comparing this work and the previously reported gel or elastomer-based sensors. d The response curves of capacitive variation for this sensor upon applying different pressures. e Detecting different rates of eye movement by adhering this sensor on the upper eyelid. f Demonstration of the sensing stability for this sensor upon applying pressure of 70 Pa for 10,000 cycles.
Respiratory behavior involves the nostril airflow and the chest/abdomen movement. The respiratory rate and intensity are the key parameters for evaluating the status of a person and are extremely important for early diagnosis of related diseases. In this work, four different respiratory states were stimulated, including apnea, fast, deep, and normal breathing behavior. The sensor was self-adhered on the skin below the nostril, as well as the chest and abdomen positions. Firstly, the pressure variation from nostril airflow and chest and abdomen movement in the respiratory behavior was continuously monitored in the capacitive mode. The relative capacitive change was constant when holding breath to simulate the apnea. In contrast with the electrical signal at normal state (ΔC/C0 = 0.30%, 24 bpm), the bigger capacitive variation (ΔC/C0 = 0.38%, 17 bpm) of deep respiratory and the shorter time interval (ΔC/C0 = 0.20%, 30 bpm) of fast respiratory were displayed, clearly indicating the stronger intensity and faster frequency corresponded to the deep and fast respiration, respectively (Fig. 7a). A similar change pattern of capacitive variation was displayed in real-time monitoring of chest and abdomen movement for respiratory behavior (Fig. 7b, c, Supplementary Movie 2 and 3). In contrast, the relative capacitive change of chest/abdomen breath was much higher than that of nose breath. This demonstrates that the weak pressure stimulus from nostril airflow could indeed be precisely recorded by this sensor. Specifically, the change in the absolute value for capacitance was the opposite for inhaling and exhaling behavior in respiratory monitoring via nostril airflow and chest/abdomen breath (the insets of Fig. 7a, b, and c). For nose breathing, the exhalation induced an increase in capacitance because of the generated pressure from the nostril airflow. Conversely, the act of exhalation via chest/abdomen breathing resulted in a decrease of capacitive signal, ascribing to the comparable volume shrinkage of the chest and abdomen when exhaling. Interestingly, the sensor even detected the difference between the left and right nostrils (Fig. 7d). One of the nostrils, called the main breathing nostril, exhibited much higher respiratory intensity than that of another accessory breathing nostril. Meanwhile, the different physiological activities of a person could also be differenced by monitoring the pulse, which was calculated from the stable and clear capacitive signal. Figure 7e showed the pulse could be gradually recovered to the normal state by increasing the rest time after running. The above results strongly confirm that this sensor possesses extraordinary sensitivity for weak pressure detection, providing the potential application of this sensor in disease diagnosis via monitoring the pressure variation.
The curves of capacitive variation for monitoring the respiratory behavior from nostril airflow (a), chest (b), and abdomen (c) movement by mimicking different respiratory behaviors. d The capacitive variation for detecting left and right nostril airflow by adhering the sensor on the corresponding subnostril. e Monitoring pulse variation after resting different time on capacitive mode. f Detecting nostril or mouth airflow by mimicking different respiratory behavior on resistive mode, via monitoring the thermal variation of airflow. Scale bars: 1 cm. g Demonstration of the sensing performance for the self-healed sensors by monitoring the temperature variation of nostril airflow on resistive mode.
Based on the precise detection of pressure stimulus for respiratory behavior in the capacitive mode, the thermal variation of nostril airflow was further investigated in the resistive mode. The temperature coefficient of resistance (TCR), which represents the thermal sensitivity, was evaluated according to the equation (TCR = [(RT–R0)/R0]/δT). The calculated TCR was −1.67% °C−1 (R2 = 0.99, Supplementary Fig. 12). When applying the increased temperature stimulus to the sensor, not only activating the ionic transport within the gel network, but also promoting the dissociation of Ca2+ within the polymer network and increasing the concentration of charge carriers, both of them contributed to the decreased resistance at higher temperature, which is beneficial to detect the small thermal variation of nostril airflow. When monitoring the thermal variation of airflow from the nose and mouth by changing four kinds of respiratory patterns, a similar law was displayed (Fig. 7f). No electrical variation was observed when simulating apnea. The larger signal and faster frequency appeared when simulating the deep and fast respiration, respectively, compared to the normal state. Specifically, at deep and normal respiration states, the resistive variation of airflow from mouth breathing was obviously bigger than that of nose breathing. This is attributed to the much more exhaled hot airflow from the mouth, which induces a larger temperature difference than that of nostril airflow. This can be used to distinguish breathing behavior from nose or mouth and is important for evaluating personal sleep quality. In order to investigate the effect of eutectogel to the capacitive sensing. Two control capacitive sensors were fabricated which utilizing the polyethylene (PE) film and metal Cu sheet as the control dielectric layer and control electrode, respectively, ascribing to their incompressibility and super high modulus and stiffness. And the corresponding schematic illustration for sensor structure was shown in Supplementary Fig. 11a, which were labeled as sensor I, II, and III, respectively. When applied pressure of 15, 30 or 150 Pa to the sensor, the relative value of capacitive variation of sensor I and II is periodical for one certain pressure and stepwise increased as increasing the pressure (Supplementary Fig. 11b). Notably, in contrast to sensor I, the sensor II displayed the similar value of ΔC/C0 at comparable small pressure of 15 and 30 Pa, but showed smaller value of ΔC/C0 at 150 Pa (Supplementary Fig. 11b). Meanwhile, the sensor III cannot detect the capacitive variation at 15 Pa, and also exhibited much smaller value of ΔC/C0 at 30 and 150 Pa compared to that of sensor I and II (Supplementary Fig. 11b). This experiment clearly demonstrated the capacitive variation was mainly ascribed to the expanding area of eutectogel at comparable small pressure (C = εS/4πkd, S is the effective area of the conducting layer, d is the thickness of the dielectric layer, ε is the dielectric constant of the dielectric layer, and k is the electrostatic constant). While for large pressure, the capacitive variation was ascribed to the synergistic effect of expanding area of eutectogel and the reduced thickness variation of VHB. And the result of nostril airflow monitoring by using these three sensors also verified this effect (Supplementary Fig. 11c). As mentioned above, this eutectogel possessed superior self-healing efficiency. To investigate the reusability of this fabricated gel sensor, the sensor was deliberately cut into two halves, and the resistance value of the original and self-healed sensor for monitoring the successive nose breathing behavior was compared. As shown in Fig. 7g, the outputted electrical signal could recover to the original value when the gel was self-healed, and no electrical signal degraded even after the 5th healing. And the healed sensor could also reproduce the extremely same resistive variation when mimicking the different respiratory patterns (Supplementary Fig. 13). Even after healing at an extreme environment of −20 °C, the sensor could display good sensing capability (Supplementary Fig. 14), ascribing to the excellent self-healing efficiency at −20 °C (Fig. 4e) and the remarkable ionic conductivity at −20 °C (Supplementary Fig. 10b). This excellent self-healing performance could also be reflected in the mode of the capacitive pressure sensor (Supplementary Fig. 15). The above results demonstrate not only the weak pressure stimuli but also the small thermal variation could be precisely detected by outputting different kinds of electrical signals. This provides a solid foundation for monitoring the nostril airflow via the dual mode.
Monitoring nostril airflow via the dual mode of pressure and temperature
Based on the above results, the nostril airflow was monitored real-time by adhering to the bimodal sensor below the left nose (Fig. 8a). The airflow field and the pressure distribution from exhaled airflow were simulated (ANSYS WORKBENCH CFX), respectively, and the maximum pressure to the sensor was less than 10 Pa. Simultaneously, a higher temperature stimulus from exhaled airflow was applied to the sensor, and the highest temperature value was about 35 °C. As shown in Fig. 5b, c, the pressure variation from 0 to 10 Pa and the thermal stimulus from 20 to 35 °C to the corresponding electrical signal of capacitance and resistance displayed a linear relationship without cross-coupling. Hence, the capacitive signal reflected the pressure variation of nostril airflow, while the resistive signal represented the thermal stimuli of nostril airflow. As shown in Fig. 8a, the pressure and temperature of nostril airflow were indeed simultaneously detected by outputting independent electrical signals (Supplementary Movie 4). Notably, the two monitored signals matched perfectly, and the peak trend of capacitive and resistive variation was the opposite, ascribing to the exhaled airflow generated pressure stimuli, which increased the capacitance value while the higher temperature made the resistance decrease, which was consistent with the above-mentioned result. Moreover, the different respiratory behaviors were mimicked and monitored. In the apnea state, a straight line was observed because of no variation in capacitance and resistance. When switching into the deep, normal, and fast respiratory mode, both the capacitive and resistive variation curves changed accordingly. The corresponding respiratory frequencies for the deep, normal, and fast states were 14, 19, and 32 bpm, respectively. To further investigate the superior sensitivity for simultaneous and independent detection of pressure and temperature, the subtle water droplets with different weights and temperatures were applied to this dual-modal sensor. As shown in Fig. 8b, when fixing the temperature of water droplets at 27 °C and just increasing the weight of water droplets from 5, 15 to 30 mg, the capacitive variation value increased stepwise. The resistive variation was nearly kept constant, and the little difference in resistance was ascribed to the different contact areas when varying the weight of the droplet. When fixing the weight of water droplets at 30 mg and just increasing their temperature from 27, 28 to 29 °C, the capacitive variation was nearly the same while the resistive variation increased stepwise. To demonstrate the advantage of this bimodal sensor, the pressure and temperature variation of nostril airflow were simultaneously monitored by this bimodal sensor at different environmental temperatures (18, 25, 40 °C). As shown in Supplementary Fig. 16, the outside temperature indeed generates severe influence on the thermal detection of nostril airflow via resistive mode, while the capacitive signal sensing the pressure variation of nostril airflow keeps a stable value at these different outside temperatures. These results strongly demonstrate the pressure and temperature stimulus from one subject could be simultaneously but independently detected by this fabricated bimodal eutectogel-based sensor, via outputting different electrical signals without cross-coupling, thereby effectively eliminating interference between the different stimuli when relying on one kind of electrical signal output.
a Simultaneous detection of the pressure and temperature variation of nostril airflow by outputting different kinds of electrical signals of capacitance and resistance, respectively, the airflow field and pressure distribution of exhaled nostril airflow was simulated, and the thermal distribution of nostril airflow was visualized by an infrared camera. b The sensing test of water droplets to the dual-modal sensor by varying the weight or temperature of the water droplet.
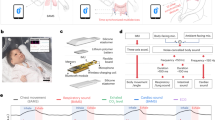
Obstructive sleep apnea syndrome (OSAS) is one typical respiratory disease in clinical practice, that is likely to induce some severe illness. Effectively monitoring respiratory behavior in sleep is extremely crucial for assessing individual health and even saving human lives. Figure 9 shows the evident characteristic of OSAS is the intermittent cessation of breathing during sleep because of the pharyngeal narrowing. In clinical practice, sleep apnea is defined as a complete airflow cessation period of at least 10 s. Herein, the bimodal sensor was further developed to monitor OSAS by adhering four sensors on the corresponding body positions, below the nose, chest, abdomen, and wrist, to independently but simultaneously detect the nostril airflow, chest/abdomen movement, and pulse. As proof of concept, the breathing was first conducted at a normal state for 36 s. Then, it was deliberately held for 24 s to simulate the apnea state and recovered to normal breathing again, and this process was repeated two times. Figure 9 shows the consistent, stable, and periodical electrical signal at the initial normal state for the five kinds of stimulus from four different body positions. This included the temperature response of nostril airflow and the pressure detection from nostril airflow, chest, abdomen, and pulse. The calculated respiratory rate and pulse rate were about 25 rpm and 72 bpm, respectively. For the second stage of apnea, nearly flat curves of capacitive variation for the pressure stimulus of nostril airflow, chest, and abdomen were observed. This was because no pressure variation was observed for these body positions at this state. In contrast, the value of resistive variation increased until a platform was achieved, ascribed to the gradually increased resistance when stopping the thermal stimulus from nostril airflow. Meanwhile, the pulse rate evidently decreased to 62 bpm because of the airflow obstruction, further strongly demonstrating the occurrence of an apnea event. And the corresponding resistive and capacitive variation gradually returned to the normal state when recovering normal breathing for these five outputted electrical signals. Furthermore, a similar curve appeared when applying the repeated behaviors. The severity of OSAS was evaluated by recording the number and duration time of apnea events per hour in the personal sleep. The above results clearly verified that this kind of eutectogel-based bimodal sensor could achieve superior sensing capability for simultaneously monitoring five different stimuli by outputting independent electrical signals. This significantly improved the accuracy and reliability of OSAS diagnosis and effectively avoided the motion artifact and interference of the environment, which usually occurred when solely a single stimulus is monitored or only the same electrical signal is output for different stimuli.
The schematic illustration of the symptoms of OSAS disease, and monitoring the apnea state and the respiratory recovery process when mimicking OSAS via simultaneously displaying five kinds of channels.
Discussion
In this work, a ATH eutectogel was synthesized by completely physical crosslinking of PACG, TA, and HAp in DESs. The optimized ATH6 gel exhibited low modulus and superior self-healing efficiency compared to other reported gel or elastomers. While the strong adhesive strength to the porcine skin or other substrates was achieved, simultaneously possessing good environmental stability and bio-compatibility. The fabricated dual-mode sensor displayed pressure sensitivity superior to previous reports, which could precisely detect the weak pressure stimulus of nostril airflow. Hence, this kind of ATH6 gel-based sensor could simultaneously detect the pressure and thermal stimuli from the nostril airflow, while outputting independent electrical signals of capacitance and resistance, respectively, without cross-coupling. This remarkable benefit significantly improved the accuracy and reliability of the detection result for nostril airflow, which effectively excluded the interference of the external environment when solely a single stimulus is monitored or only the same electrical signal is output for different stimuli. In contrast with the clinical instrument of polysomnography (PSG) which needs two kinds of sensors to be simultaneously fixed to monitor the pressure and temperature of nostril airflow, this kind of bimodal sensor integrated into one single sensor, greatly improving the comfortability, wearability, and portability. In proof of concept, this constructed sensor was applied to real-time monitor and evaluate the severity of OSAS, via simultaneously monitoring five kinds of stimuli in sleeping behavior, demonstrating superior sensing capability and highly improving the accuracy and reliability of detection results. This work provides a potentially effective approach to real-time monitoring of sleeping respiration and is a strong tool for forecasting respiratory or sleep-related diseases.
Methods
Materials
Nano-sized HAp was purchased from Macklin Biochemical Technology Co., Ltd (Shanghai, China). TA and 2-Hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone (I2959) were purchased from Sigma Aldrich Life Science & Technology Co., Ltd (Shanghai, China). Choline chloride (CHCl), ethylene glycol (EG), and acryloyl chloride were purchased from Adamas-beta Co., Ltd N-Acryloyl 2-Glycine (ACG) was synthesized according to previous work (glycine, acryloyl chloride and NaOH were as the crude materials, which reacted at RT for 1 h)53. All other chemicals and solvents were analytical reagents. All animal experiments were conducted with the permission of the Laboratory Animal Ethics Committee of South China Agricultural University (approval number 2024b177). Six female SD rats with an average body weight of 160 g were used. All animals were purchased and raised by the animal management of South China Agricultural University. The rats were housed in the controlled animal facilities with relative humidity at 55–60% and temperature conditions at 22–25 °C under 12/12-h light/dark cycle, with access to food and water ad libitum. Each animal was used for only one experiment. And the rats were euthanized at second day via intraperitoneal injection of an excessive amount of anesthetic. And the treated skins were photographed and processed for HE staining.
Preparation of deep eutectic solvents (DESs)
Simply, choline chloride and ethylene glycol were mixed at a 1:2 M/M ratio. The mixture was continuously stirred at 80 °C until a transparent liquid was obtained.
Preparation of ATH eutectogels
Appropriate amounts of ACG, HAp, and TA were first dissolved in DESs, according to Table S1. Then, 1 wt% I2959 (relative to the total mass of ACG) was added and stirred thoroughly until complete dissolution. The mixture was added to a polytetrafluoroethylene (PTFE) mold, covered with a glass sheet, and then irradiated with UV light (λ = 365 nm) for 20 min. A series of ATH eutectogels were obtained and labeled as ATHx, in which x represents the weight ratio of HAp to ACG.
Characterization of the mechanical strength of eutectogels
The tensile and compression tests were carried out using a universal testing machine (model 2 kN, CMT1203). For the tensile tests, dumbbell-shaped strip specimens of 3 mm thick, 3 mm wide, and 65 mm length were prepared. The tensile rate was 100 mm/min. For the compression test, the shape of the eutectogel samples was cylindrical, with a diameter of 12 mm and a height of 10 mm. The compression rate was fixed at 10 mm/min. Cyclic tests were carried out as follow-up tests immediately after the initial loading. For the high- and low- temperature tensile tests, the eutectogels were allowed to balance at the tested temperatures for 6 h. Then, the tensile tests were performed at a speed of 100 mm/min. Data are expressed as mean ± s. d (n = 5).
Measurement of adhesive shear strength
The adhesive shear strength of the gel for different substrates was determined by the lap shear test on a universal testing machine equipped with a 2 kN load cell at a constant speed of 50 mm*min−1 according to ASTM F2255-05 standards. The gels were cut into a size of 10 mm × 10 mm × 1 mm, sandwiched between two substrates, and pressed with a 500 g weight for 1 min. The substrates included fresh porcine skin, steel, copper, wood, rubber, and plastic. Moreover, the fresh porcine skin was purchased from a local grocery shop. For the high-low temperature lap-shear test, the gels were equilibrated at the corresponding temperature for 2 h and then tested at 50 mm/min.
Test for the self-healing performance
A dumbbell-shaped eutectogel (3 mm thick, 3 mm wide, and 65 mm long) was cut into two parts with a razor blade to evaluate the self-healing ability of the ATH gel. The two separated parts were placed in contact with each other in a PTFE mold for a certain time, and tested the tensile curve at the same temperature by using the high-low temperature chamber of the universal testing machine. To evaluate the self-healing efficiency, the tensile fracture strain of the original and self-healed eutectogels were measured under the same conditions. The self-healing efficiency (HE) was defined as Eq. (1):
where SH and SO are the tensile fracture strain of the self-healed eutectogel and the original eutectogel, respectively.
Determination for the long-term stability of ATH
The long-term stability of the gel was assessed by recording the weight change of the gel in different environments. The relative humidity (RH) of the indoor environment (RT) was about 40%, and the temperature was 25 °C, while the outdoor RH was 55%–70% and the temperature was 25 °C–30 °C. Weight retention was calculated by Wt/W0 ×100%.
Wt and W0 represent the post-storage weight and the initial weight of the gel, respectively. Five parallel specimens were tested.
Differential scanning calorimetry (DSC)
The anti-freezing properties of ATH6 eutectogel and the corresponding hydrogel were determined using differential scanning calorimetry (DSC, Netzsch 214 polyma, Germany) under an N2 atmosphere. The temperature range was from 20 to −80 °C at a cooling rate of −10 °C min−1.
Rheological measurements
Rheological tests were carried out on a rotational rheometer (TA Discovery HR-2) with parallel plates (diameter of 20 mm). In the linear viscoelastic region, a constant strain amplitude of 0.5% and a constant frequency of 1 Hz were tested for time sweep. To evaluate the self-healing properties of the gels, large strain of 300% and small strain of 0.1% were respectively maintained for 100 s, and alternately repeated four times. All gel samples were prepared as circles with a diameter of 20 mm and a thickness of 1.5 mm.
In vitro cell compatibility tests
The CCK-8 method was used to evaluate the cell viability of this eutectogel. The gel samples were soaked in Duchenne Modified Eagle’s Medium (DMEM) and left at 37 °C for 24 h to obtain the leach liquor, and the pure DMEM as the control sample. Fibroblasts were inoculated into 96-well plates at 5 × 103 cells per well. After the cells adhered to the bottom of the well plates, the different concentrations of leach liquor (50 µg/mL, 100 µg/mL, 200 µg/mL, 500 µg/mL) were co-incubated in a CO2 incubator (37 °C, 5% CO2) for 1, 3 and 5 days. At each predetermined time point, the optical density values at 450 nm were measured using the microplate reader.
Construction and characterization of multimodal eutectogel-based sensor
A dielectric film (VHB 4905, 3 M) was sandwiched between two eutectogels, and three wires were inserted into the eutectogels. The capacitance and resistance signals were detected by a digital multimeter (LinkZill TruEbox 01RC, China). To detect the sensory relationship between the stimulus of pressure/thermal and the electrical signal of resistance and capacitance, DMA (DMA850, TA) was utilized by varying the pressure from 0 to 250 Pa at different temperatures from 20 to 35 °C. The infrared images of the sensors were recorded by an infrared thermal imager (FLUKE Tis75).
Statistics and reproducibility
All experiments were repeated independently with similar results at least five times.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings of this study are available within this article, its supplementary information, and source data file. Source data are provided with this paper.
References
Su, Y. et al. Self-powered respiration monitoring enabled by a triboelectric nanogenerator. Adv. Mater. 33, 2101262 (2021).
Shen, S. et al. Advances in wearable respiration sensors. Mater. Today 72, 140–162 (2024).
Li, Y. et al. Integrated wearable smart sensor system for real-time multi-parameter respiration health monitoring. Cell Rep. Phys. Sci. 4, 101191 (2023).
Ni, Y. et al. Environmental stability stretchable organic hydrogel humidity sensor for respiratory monitoring with ultrahigh sensitivity. Adv. Funct. Mater. 34, 2402853 (2024).
Hussain, T., Ullah, S., Fernández-García, R. & Gil, I. Wearable sensors for respiration monitoring: a review. Sensors 23, 7518 (2023).
Zavanelli, N. et al. At-home wireless monitoring of acute hemodynamic disturbances to detect sleep apnea and sleep stages via a soft sternal patch. Sci. Adv. 7, eabl4146 (2021).
Yeon, H. et al. Long-term reliable physical health monitoring by sweat pore–inspired perforated electronic skins. Sci. Adv. 7, eabg8459 (2021).
Ning, C. et al. Helical fiber strain sensors based on triboelectric nanogenerators for self-powered human respiratory monitoring. ACS Nano 16, 2811 (2022).
Wang, M. et al. Air-flow-driven triboelectric nanogenerators for self-powered real-time respiratory monitoring. ACS Nano 12, 6156–6162 (2018).
Liu, J. et al. Multimodal hydrogel-based respiratory monitoring system for diagnosing obstructive sleep apnea syndrome. Adv. Funct. Mater. 32, 2204686 (2022).
Peng, X. et al. All-nanofiber self-powered skin-interfaced real-time respiratory monitoring system for obstructive sleep apnea-hypopnea syndrome diagnosing. Adv. Funct. Mater. 31, 2103559 (2021).
Fang, Y. et al. A deep‐learning‐assisted on‐mask sensor network for adaptive respiratory monitoring. Adv. Mater. 34, 2200252 (2022).
Lee, S. et al. Nanomesh pressure sensor for monitoring finger manipulation without sensory interference. Science 370, 966–970 (2020).
Jagadev, P. & Giri, L. I. Non-contact monitoring of human respiration using infrared thermography and machine learning. Infrared Phys. Techn. 104, 103117 (2020).
Dai, J. et al. Ultrafast response polyelectrolyte humidity sensor for respiration monitoring. ACS Appl. Mater. Interfaces 11, 6483–6490 (2019).
Huang, Y. et al. Liquid metal-based epidermal flexible sensor for wireless breath monitoring and diagnosis enabled by highly sensitive SnS2 nanosheets. Research 2021, 9847285 (2021).
Ottaviano, G. & Fokkens, W. J. Measurements of nasal airflow and patency: a critical review with emphasis on the use of peak nasal inspiratory flow in daily practice. Allergy 71, 162–174 (2016).
Feng, B. et al. Nacre-inspired, liquid metal-based ultrasensitive electronic skin by spatially regulated cracking strategy. Adv. Funct. Mater. 31, 2102359 (2021).
Ye, L. et al. Face mask integrated with flexible and wearable manganite oxide respiration sensor. Nano Energy 112, 108460 (2023).
Wang, H. et al. An integrally formed Janus hydrogel for robust wet-tissue adhesive and anti-postoperative adhesion. Adv. Mater. 35, 2300394 (2023).
Liu, J. et al. Anti-bacterial silk-based hydrogels for multifunctional electrical skin with mechanical-thermal dual sensitive integration. Chem. Eng. J. 426, 130722 (2021).
Liu, Z. et al. Highly compressible and superior low temperature tolerant supercapacitors based on dual chemically crosslinked PVA hydrogel electrolytes. J. Mater. Chem. A 8, 6219–6228 (2020).
Liu, Z. et al. Highly compressible hydrogel sensors with synergistic long-lasting moisture, extreme temperature tolerance and strain-sensitivity property. Mater. Chem. Front. 4, 3319 (2020).
Liu, J. et al. Multimodal hydrogel-based respiratory monitoring system for diagnosing obstructive sleep apnea syndrome. Adv. Func. Mater. 32, 2204686 (2022).
Zhang, L. et al. Fully organic compliant dry electrodes self-adhesive to skin for long-term motion-robust epidermal biopotential monitoring. Nat. Commun. 11, 4683 (2020).
Ohm, Y. et al. An electrically conductive silver–polyacrylamide–alginate hydrogel composite for soft electronics. Nat. Electron. 4, 185–192 (2021).
Yang, Q. et al. Photocurable bioresorbable adhesives as functional interfaces between flexible bioelectronic devices and soft biological tissues. Nat. Mater. 20, 1559–1570 (2021).
Yu, Z. & Wu, P. Underwater communication and optical camouflage Ionogels. Adv. Mater. 33, 2008479 (2021).
Wang, C. et al. On-skin paintable biogel for long-term high-fidelity electroencephalogram recording. Sci. Adv 8, eabo1396 (2022).
Bao, B. et al. Rapid fabrication of physically robust hydrogels. Nat. Mater. 22, 1253–1260 (2023).
Cai, L. et al. Soft composite gels with high toughness and low thermal resistance through lengthening polymer strands and controlling filler. Adv. Funct. Mater. 33, 2207143 (2023).
Liu, Y. et al. Super stable, highly ion-conductive and transparent eutecto-/hydro-gel promotes wearable electronic and visual strain sensing. Chem. Eng. J. 461, 141965 (2023).
Sun, X. et al. Tough and ultrastretchable liquid-free ion conductor strengthened by deep eutectic solvent hydrolyzed cellulose microfibers. Adv. Funct. Mater. 32, 2202533 (2022).
Yao, P. et al. Environmentally stable, robust, adhesive, and conductive supramolecular deep eutectic gels as ultrasensitive flexible temperature sensor. Adv. Mater. 35, 2300114 (2023).
Li, B., Xu, F., Guan, T., Li, Y. & Sun, J. Self-adhesive self-healing thermochromic ionogels for smart windows with excellent environmental and mechanical stability, solar modulation, and antifogging capabilities. Adv. Mater. 35, 2211456 (2023).
Hu, R. et al. An ultrahighly stretchable and recyclable starch-based gel with multiple functions. Adv. Mater. 35, 2303632 (2023).
Zhang, X. et al. Tough liquid-free ionic conductive elastomers with robust adhesion and self-healing properties for ionotronic devices. Adv. Funct. Mater. 34, 2307400 (2024).
Xu, L. et al. A transparent, highly stretchable, solvent-resistant, recyclable multifunctional ionogel with underwater self-healing and adhesion for reliable strain sensors. Adv. Mater. 33, 2105306 (2021).
Ye, H., Wu, B., Sun, S. & Wu, P. Self-compliant ionic skin by leveraging hierarchical hydrogen bond association. Nat. Commun. 15, 885 (2024).
Guan, L. et al. Balloon inspired conductive hydrogel strain sensor for reducing radiation damage in peritumoral organs during brachytherapy. Adv. Funct. Mater. 32, 2112281 (2022).
Hou, K.-X. et al. A puncture-resistant and self-healing conductive gel for multifunctional electronic skin. Adv. Funct. Mater. 31, 2107006 (2021).
Wei, Y. et al. MXene-based conductive organohydrogels with long-term environmental stability and multifunctionality. Adv. Funct. Mater. 30, 2005135 (2020).
Yiming, B. et al. A mechanically robust and versatile liquid-free ionic conductive elastomer. Adv. Mater. 33, 2006111 (2021).
Zhang, P. et al. Dynamically crosslinked dry ion-conducting elastomers for soft iontronics. Adv. Mater. 33, 2101396 (2021).
Ma, J. et al. Mechanochromic, shape-programmable and self-healable cholesteric liquid crystal elastomers enabled by dynamic covalent boronic ester bonds. Angew. Chem. Int. Ed. 61, e202116219 (2022).
Gu, C. et al. A full-device autonomous self-healing stretchable soft battery from self-bonded eutectogels. Adv. Mater. 35, 2208392 (2023).
Wu, H. et al. Strong adhesive, high conductive and environmentally stable eutectogel based on “Water in deep eutectic Solvent”: ultrasensitive flexible temperature and strain sensors. Chem. Eng. J. 497, 154883 (2024).
Quan, Q. et al. Tough and stretchable phenolic-reinforced double network deep eutectic solvent gels for multifunctional sensors with environmental adaptability. Adv. Funct. Mater. 33, 2303381 (2023).
Yang, R. et al. Multimodal sensors with decoupled sensing mechanisms. Adv. Sci. 9, 2202470 (2022).
Shen, Z., Zhu, X., Majidi, C. & Gu, G. Cutaneous ionogel mechanoreceptors for soft machines, physiological sensing, and amputee prostheses. Adv. Mater 33, 2102069 (2021).
Liu, Y. et al. Versatile ion-gel fibrous membrane for energy-harvesting iontronic skin. Adv. Funct. Mater. 33, 2303723 (2023).
Zhang, Y. et al. Highly stable flexible pressure sensors with a quasi-homogeneous composition and interlinked interfaces. Nat. Commun. 13, 1317 (2022).
Yu, J. et al. Robust hydrogel adhesives for emergency rescue and gastric perforation repair. Bioact. Mater. 19, 703–716 (2023).
Acknowledgements
This work was financially supported by the Guangdong Basic and Applied Basic Research Foundation (2025B1515020031, 2023A1515012167), National Key Research and Development Program of China (2024YFA1107600). No formal approval for the experiments involving human volunteers was required, which was waived by the Medical Ethics Committee of Guangzhou Medical University. The volunteers took part following informed consent.
Author information
Authors and Affiliations
Contributions
T.L. and Q.W. contributed equally to this work, and they carried out most experiments. Z.L. conceived this idea, supervised the work, and wrote the manuscript. H.L., X.Z., X.Y., J.L., and Z.N. assisted in characterization of some materials. B.Z. and Q.W. assisted in supervising the work. All authors discussed the results and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, T., Wu, Q., Liu, H. et al. A crosslinked eutectogel for ultrasensitive pressure and temperature monitoring from nostril airflow. Nat Commun 16, 3334 (2025). https://doi.org/10.1038/s41467-025-58631-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-58631-7