Abstract
Invasive mold infections are a major cause of mortality in immunosuppressed and cancer patients. Diagnosis is challenging, requiring invasive procedures or reliance on fungal biomarkers with limited sensitivity and an inability to detect non-Aspergillus molds. Here, we perform whole-body 18F-fluorodeoxysorbitol (18F-FDS) positron emission tomography (PET) in nine prospectively enrolled patients with high-suspicion of invasive mold infections (eventually confirmed using culture or molecular assays, n = 4) or other pathologies (n = 5) with localization of 18F-FDS PET signal to infection sites as the primary outcome (NCT05611892). 18F-FDS PET (120 or 180 min after injection), rapidly detects and localizes invasive pulmonary and cerebral infections due to Aspergillus, non-Aspergillus (galactomannan-negative), or azole-resistant molds, and differentiates them from sterile inflammation or cancer. Moreover, 18F-FDS selectively and rapidly accumulates intracellularly in a range of clinically relevant molds, including azole-resistant molds, via a saturable process. In animals, 18F-FDS PET is able to detect and localize pulmonary and cerebral aspergillosis, as well as rhinosinusal infections due to Aspergillus, Rhizopus, and Mucor, confirming the clinical data. 18F-FDS can be easily synthesized from 18F-fluorodeoxyglucose (18F-FDG), which is widely available, and represents a promising, noninvasive diagnostic tool for detecting, localizing and monitoring of invasive mold infections throughout the body.
Similar content being viewed by others
Introduction
Invasive mold infections are an increasing global public health concern, affecting multiple organ systems (e.g., lung, brain), and associated with mortality rates as high as 85%1,2,3. The rising incidence of invasive mold infections is attributed to the expanding population of immunosuppressed patients3. The COVID-19 pandemic has also led to a dramatic increase in invasive mold infections, particularly pulmonary aspergillosis and mucormycosis4,5. Aspergillus spp. remains the major cause of invasive mold infections in hematologic cancer patients and transplant recipients. However, other opportunistic molds, which are even more challenging to diagnose and treat6, are on the rise due to several reasons, including wide-spread Aspergillus-targeted antifungal prophylaxis7,8. In 2022, the World Health Organization (WHO) categorized Aspergillus and Mucorales molds as Critical and High Priority Group pathogens, respectively9. Prompt identification of invasive mold infections and initiation of appropriate treatment is critical for improving patient outcomes.
When infections occur in relatively inaccessible sites, minimally invasive clinical samples (blood, urine, stool, or cerebrospinal fluid) can often yield non-diagnostic results or are otherwise insensitive for deep-seated infections. Although major strides have been made in the development of microbial cell-free DNA detection for infections10,11, clinically available biomarkers, namely galactomannan and 1,3-beta-D-glucan, have limited sensitivity and specificity for Aspergillus spp., and galactomannan is unable to reliably detect non-Aspergillus molds. Specifically, the overall sensitivity of serum / plasma galactomannan using the most current (2020) European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium (EORTC / MSGERC) guidelines is 65-71%, but lower in those with hematologic malignancies (58%), hematopoietic stem cell transplantation (65%), and non-neutropenic patients (24%)12,13. While galactomannan from the bronchoalveolar lavage (BAL) has a higher sensitivity (75–86%), BAL is an invasive process and cannot be performed in many cancer patients at risk for bleeding complications.
Importantly, all clinically available imaging tools such as radiography, ultrasonography, computed tomography (CT), and magnetic resonance imaging (MRI) rely on structural changes in anatomy or tissue morphology that are often delayed relative to the disease process, are non-specific, and reflect a combination of the infection and the host inflammatory response14,15,16. CT findings of pulmonary aspergillosis include the halo sign (nodule with surrounding ground-glass opacity), consolidations, and cavitary lesions, but these findings may also be present with other pathological processes17. Although 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) is highly sensitive for detecting infectious foci, including Aspergillosis18,19,20, it also reflects a combination of the infection and inflammation. Therefore, currently available imaging tools rely on host responses to infection that could be significantly altered in immunocompromised host and none can reliably differentiate infectious sites from sterile inflammatory or cancerous lesions14,15,16. Moreover, 18F-FDG PET may also have limited accuracy for detecting cerebral fungal infections19, presumably due to the high background signal in the brain. Additionally, biomarkers cannot provide spatial information on the location or extent of infection. Therefore, definitive diagnosis of invasive mold infections almost always requires invasive procedures such as BAL or deep tissue biopsy to obtain a relevant clinical sample, which is challenging, dangerous (e.g., brain biopsy), and delays the diagnosis. Because of these challenges, empiric antifungal treatment (prior to establishing a confirmatory diagnosis) is the norm in patients with suspected invasive mold infections. However, treatment can decrease the sensitivity of culture-based tests performed later. The development of rapid, whole-body, noninvasive, and specific diagnostics, able to detect invasive mold infections in patients who are often immunosuppressed and receiving empiric treatment, would be a crucial advancement in the field and could become the clinical standard of care. Although, there have been several efforts to develop pathogen-specific imaging approaches for Aspergillus using siderophores, labeled antibodies and sugars16,21,22,23,24,25,26,27,28,29,30, none have been prospectively studied in patients (other than a case report31), or applied to other mold infections. Moreover, there are some challenges in the use of labeled antibodies, namely slower clearance, potential for reduced penetration in necrotic tissues, as well as more complicated regulatory pathways (versus small molecules) for clinical translation. Therefore, there is an urgent need for sensitive, whole-body, noninvasive tools to diagnose invasive mold infections.
We have previously described 18F-fluorodeoxysorbitol (18F-FDS), a radioanalog of sorbitol (sugar alcohol), as a PET tracer to selectively image infections due to the Enterobacterales group of bacteria (e.g., Escherichia coli)32,33. However, Aspergillus can also utilize sorbitol as a carbon source34,35, and a recent study has shown that 18F-FDS PET can detect invasive pulmonary aspergillosis in animal models27. Our overall goals were to evaluate 18F-FDS PET to specifically detect and localize invasive mold infections by a wide range of clinically relevant mold-fungi in animal models and first-in-human studies.
Here, we perform whole-body 18F-FDS PET/CT in nine prospectively enrolled patients with either invasive mold infections (n = 4 patients) or other pathologies (n = 5 patients) (NCT05611892). We demonstrate that 18F-FDS PET can rapidly detect and localize invasive pulmonary and cerebral mold infections due to drug-susceptible or azole-resistant molds, and differentiate them from sterile inflammation and cancer. We also show that 18F-FDS selectively accumulates in a range of clinically relevant molds, but not by heat killed fungi or mammalian cells. Importantly, 18F-FDS is rapidly metabolized intracellularly by molds, via a saturable process. Tissue histology from eight unique patients with pulmonary, cerebral or rhinosinusal invasive mold infections, demonstrates the lesional biomass occupied by molds is ~50-fold higher than for bacterial infections. In animal studies, 18F-FDS PET/CT is able to detect and localize pulmonary and cerebral aspergillosis, as well as rhinosinusal infections due to several molds, confirming the clinical data. Importantly, 18F-FDS can be easily synthesized from commercially available 18F-FDG36, the most widely utilized PET tracer globally, by a simple one-step kit-based method, without the need for specialized radiosynthesis and purification facilities37, allowing on-demand synthesis and global availability of 18F-FDS.
Results
18F-FDS PET/CT selectively detects invasive mold infections
We prospectively enrolled nine newly identified patients with either invasive mold infections (n = 4) using the EORTC/MSGERC consensus definitions (Table S1) or uninfected controls with other pathologies (n = 5) (NCT05611892). Confirmatory diagnosis of mold infections was established in all four patients, using culture or molecular assays (Fig. 1, Table S2). Sterile inflammatory pathologies (n = 4) were determined clinically and/or by tissue biopsies and oncologic diagnosis (n = 1; breast cancer) was made via tissue biopsy (Fig. 1, Table S2)33. The median age was 59 years (range, 37 to 71 years), and 44% (4 of 9) were female. Importantly, the enrolled patients were representative of patients hospitalized in a tertiary care setting with several co-morbidities (Table S2). Whole-body 18F-FDS PET/CT performed in accordance with U.S. FDA guidelines was well tolerated in all patients. 18F-FDS kinetics were consistent with prior studies33, demonstrating rapid renal / hepatobiliary elimination and low background in unaffected brain and lung tissues.
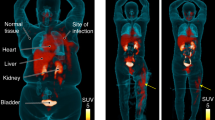
a Consort diagram for the study. Patients with confirmed invasive mold-related fungal infections and controls with confirmed inflammatory or oncologic disease (without infection) were prospectively enrolled. b Location of invasive mold infections and other pathologies. c 18F-FDS PET showing cranial (patient 1, left) and pulmonary (patient 4, right) mold infections with the lesions marked by red arrows. Panel (b) created using BioRender. Masias, Y. (2025) https://BioRender.com/w9dq20g.
To quantify the PET signal, spherical volumes of interest (VOIs) were drawn at the sites of pathology and unaffected sites of the same tissue to calculate the target-to-nontarget ratio (TNT). 18F-FDS PET signal was significantly higher at the sites of invasive mold infections with a TNT of 5.79 [interquartile range (IQR), 3.18 to 15.20] versus sterile inflammatory pathologies or cancer with a TNT of 1.40 (IQR, 1.01 to 1.93) (P = 0.008) (Fig. 2). Importantly, while the 18F-FDG PET clearly demonstrated the breast cancer lesion, no signal was noted on 18F-FDS PET (Fig. S1). Using a previously defined TNT cutoff of 3.033, 18F-FDS PET was able to detect and localize invasive pulmonary and cerebral infections in all patients with mold infections [Aspergillus, non-Aspergillus (galactomannan-negative) molds, or azole-resistant Aspergillus calidoustus)], despite having received up to seven days of antifungal treatments by the time of imaging. Moreover, lesions in all control patients had a TNT ratio of <3.
a To quantify the PET signal, spherical volumes of interest (VOIs) were drawn at the sites of pathology (red circle) and unaffected sites (yellow circle) of the same tissue to calculate the target-to-nontarget tissue ratio (TNT). b Median TNT for all enrolled patients are shown. 18F-FDS PET signal was significantly higher at the sites of invasive mold infections versus sterile inflammatory pathologies or cancer (P = 0.008). Data were derived from patients with invasive mold infections (n = 4) (patient 4 had pulmonary and cerebral lesions) and with inflammatory or oncologic diagnoses (n = 5). Data are shown on a log scale as median and interquartile range (IQR). Statistical analysis was performed using a two-tailed Mann-Whitney U test. L, liver; H, heart.
For pulmonary infections, 18F-FDS PET signal was significantly higher at infection sites with a TNT of 3.40 (IQR, 3.10 to 14.05) compared to sites with sterile inflammatory pathologies with a TNT of 1.79 (IQR, 1.57 to 2.17) (P < 0.001) (Fig. 3, S2–3, S6). Similarly, the 18F-FDS PET signal was significantly higher at the sites of invasive cerebral mold infections with a TNT of 6.39 (IQR, 5.19 to 16.92) (P = 0.007) (Fig. 4, S4–6). Importantly, 18F-FDS PET was able to identify a cerebral lesion in patient 4 with Cladophialophora bantiana pulmonary infection, which was previously missed on a brain MRI performed for clinical reasons (Fig. S4).
a Sagittal CT (left) with the corresponding transverse CT (middle) and 18F-FDS PET/CT (right) of a representative patient with confirmed invasive pulmonary mold infection (patient 3). A cavitary lesion is indicated by the yellow arrow. b Representative control patient with interstitial lung disease (control 1). Pulmonary lesions are marked with arrows. c TNT measurements derived from patients with invasive pulmonary mold infections (n = 3), and control patients (n = 4) with n = 3 VOIs per patient. 18F-FDS PET signal was significantly higher at infection sites compared to sites with sterile inflammatory pathologies (P < 0.001). Data are shown on a log scale as median ± IQR. Statistical analyses were performed using a two-tailed Mann-Whitney U test. A, aorta; H, heart; K, kidney; L, liver.
a Representative transverse (upper) and coronal (lower) CT (left) and 18F-FDS PET/CT (right) images from a patient with confirmed invasive cerebral mold infection (patient 1). b Representative control patients without cerebral disease (control 2). c Representative transverse MRI and 18F-FDS PET/MRI images from patient 1. The research PET was co-registered with the MRI performed for clinical reasons. d TNT measurements derived from patients with invasive cerebral mold infections (n = 2), and the control patients without cerebral pathology (n = 5) with n = 2 VOIs per patient (patient 4 with mold infection has 1 VOI only). 18F-FDS PET signal was significantly higher at the sites of invasive cerebral mold infections compared to controls (P = 0.007). Data are shown on a log scale as median ± IQR. Statistical analyses were performed using a two-tailed Mann-Whitney U test.
18F-FDS uptake by clinically relevant molds
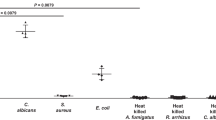
In vitro studies were performed to determine 18F-FDS uptake by a range of clinically relevant molds (Fig. 5a). Rapid 18F-FDS accumulation was noted in the live strains, whereas no uptake was noted in the heat killed controls (Fig. 5b–d) or in mammalian cells (THP-1 macrophages) (Fig. S7). Competitive inhibition tests conducted by co-incubation with increasing concentrations of unlabeled sorbitol decreased the 18F-FDS uptake (Fig. 5g, e), suggesting a saturable mechanism. Finally, all the 30 randomly selected clinical isolates, including Aspergillus spp., Rhizopus spp., Mucor spp., and other clinically relevant molds (Lichtheimia corymbifera, Syncephalastrum racemosum, Cunninghamella bertholletiae, Cladophialophora bantiana, and Fusarium solani), along with azole-resistant molds demonstrated robust 18F-FDS uptake. These findings confirm that sorbitol uptake is conserved across a range of clinically relevant molds (Fig. 5h).
a Methodology for 18F-FDS in vitro uptake. Briefly, molds were co-incubated with 18F-FDS for two hours, centrifuged to form a pellet and then washed. 18F-FDS uptake in the pellet was measured as becquerel (Bq) and corrected for protein (mg). Heat killed (HK) molds were generated by exposing them to 95 °C for 60 min. b–d 18F-FDS uptake by live and HK Aspergillus fumigatus (ATCC 1022) (red), Rhizopus arrhizus (green), and Mucor circinelloides (blue). The x-axis (time) in panel b is in min. e–g Competitive inhibition of 18F-FDS uptake with increasing concentrations of cold (unlabeled) sorbitol in Aspergillus (red), Rhizopus (green), and Mucor (blue). h 18F-FDS uptake (at 120 min) by the reference A. fumigatus (ATCC 1022) and 30 randomly selected clinical isolates representing a range of clinically relevant molds including Lichtheimia corymbifera, Syncephalastrum racemosum, Cunninghamella bertholletiae, Cladophialophora bantiana (clinical isolate from patient 4), and Fusarium solani. All assays were performed in triplicate. Data are shown as median ± IQR. Panel (a) created using BioRender. Masias, Y. (2025) https://BioRender.com/w9dq20g.
Finally, we performed studies to elucidate the metabolism of 18F-FDS by molds. We co-incubated actively growing Aspergillus fumigatus with 18F-FDS and analyzed the cytoplasmic fraction from the lysed fungal pellet by radio-thin layer chromatography (radio-TLC). 18F-FDS was rapidly metabolized intracellularly by Aspergillus in a manner similar to the intracellular metabolism of 18F-FDS in E. coli (Fig. S8).
Mold biomass occupies a significant proportion of the infected lesion
The in vitro uptake of 18F-FDS is lower in molds compared to E. coli (Fig. S7). However, in vivo visualization of infections by 18F-FDS PET is dependent on the total tracer uptake within the infected lesion. We therefore compared the biomass within infectious lesions occupied by molds and bacteria (Fig. 6). Infected tissue samples from eight unique patients with pulmonary, cerebral or rhinosinusal invasive Aspergillus infections from the Johns Hopkins Hospitals (patient characteristics are outlined in Table S3), or publicly available digitized slides of bacterial infections in patients38, were analyzed. Tissue from animals infected with Aspergillus, Rhizopus, Mucor or E. coli were also analyzed. High resolution images are shown in Fig. S9. The percentage of the infected lesion volume occupied by the mold or bacteria was calculated using automated segmentation (Fig. 6, S10). Data from human tissues demonstrated that the biomass occupied by Aspergillus was 11.71% (IQR 7.85 to 13.03%), which is ~50-fold higher than for bacterial infections at 0.24% (IQR 0.10 to 0.38%) (P < 0.001). Similar findings were observed in tissues obtained from animal studies (P = 0.016).
a Schematic showing the tissue volume occupied by mold and bacterial infections. b Methodology for quantifying the area and volume affected by mold infections in tissues. Automated segmentation of fungi is shown in red (middle panel). c Representative Grocott methenamine silver (GMS) stained histopathology slides from nine tissue samples from eight unique patients with pulmonary, cerebral, or rhinosinusal invasive Aspergillus infections; Scale bars = 50 µm. d Mold and bacterial biomass expressed as % of the infected lesion volume for infections in human and mouse tissues. Human data are derived from tissue samples from patients with pulmonary, cerebral or rhinosinusal invasive Aspergillus infections (n = 8; two tissue samples were analyzed for patient 4) from current studies, tissue slides from patients with bacterial infections (n = 4) (digital library supported by the National Library of Medicine), five slides from Aspergillus (n = 2 animals), Rhizopus (n = 2 animals) and Mucor (n = 2 animals) infected mice from current studies, and four slides from E. coli infected mice (n = 2 animals). Data from human and animal tissues demonstrated higher biomass occupied by Aspergillus in humans (P < 0.001) and by Aspergillus (P = 0.016), Rhizopus (P = 0.016) and Mucor (P = 0.016) in mice, compared to bacteria. Data are shown as median ± IQR. Statistical analyses were performed using a two-tailed Mann-Whitney U test.
18F-FDS PET localizes fungal infections in animal models
Next, we developed several clinically relevant mouse models of Aspergillus, Rhizopus, and Mucor infections and evaluated the ability of 18F-FDS PET to detect and localize the mold infections. Immunosuppressed mice were used for all studies. For pulmonary aspergillosis, mice were infected intratracheally with increasing doses of A. fumigatus conidia and the pulmonary 18F-FDS PET signal was measured, demonstrating a PET signal proportional to the infection burden (Fig. S11). Thereafter, we evaluated 18F-FDS PET in uninfected animals, animals with pulmonary aspergillosis and those with lipopolysaccharide (LPS)-induced sterile pulmonary inflammation (Fig. S12). Although substantial pulmonary disease was noted on CT and 18F-FDG PET in mice with sterile inflammation, no 18F-FDS PET signal was noted in these mice (Fig. S12b). However, mice with pulmonary aspergillosis demonstrated 18F-FDS PET signal, which colocalized with the lesions noted on CT (Fig. S12c). The pulmonary 18F-FDS PET signal was substantially higher in the mice with pulmonary aspergillosis versus those with sterile inflammation (Fig. S12g, P = 0.003). By contrast, 18F-FDG PET, which measures inflammation and is not specific for infection, could not distinguish mice with pulmonary aspergillosis versus those with sterile inflammation (Fig. S12h, P = 0.338). Postmortem pulmonary tissue histology demonstrated Aspergillus hyphae in the mice with pulmonary aspergillosis (Fig. S12i). We also performed studies with Pseudomonas, that can cause pneumonia in immunosuppressed patients. The pulmonary 18F-FDS PET signal was substantially higher in the mice with pulmonary aspergillosis versus those with Pseudomonas pneumonia (Fig. S13, P = 0.002). Importantly, the 18F-FDS PET signal from P. aeruginosa pneumonic lesions were no different than those noted with sterile pulmonary inflammation due to LPS. Consistent with our prior studies32, in vitro uptake of 18F-FDS by Pseudomonas was substantially lower than the uptake by molds (P = 0.003) (Fig. S13c).
We also developed mouse models of rhinosinusal mold infections due to A. fumigatus, Rhizopus arrhizus and Mucor circinelloides, and performed 18F-FDS PET, which was able to detect and localize the infection sites with significantly higher PET signals noted in mice with mold infections versus those with LPS-related systemic inflammation (Fig. S14, P < 0.002). Postmortem ex vivo biodistribution confirmed the imaging findings and tissue histology demonstrated fungal hyphae in the infected mice (Fig. S14e-f). Further, we performed 18F-FDS PET/CT in Aspergillus-infected mice with rhinosinusal infection to assess response to antifungal treatments. 18F-FDS PET signal in voriconazole treated (for 1 week) mice was significantly lower compared to untreated mice (P = 0.011) (Fig. S15).
Finally, we developed a mouse model of invasive cerebral aspergillosis, utilizing direct intracerebral infection with A. fumigatus. 18F-FDS PET was able to detect and localize the infection sites with significantly higher PET signal in mice with invasive cerebral aspergillosis compared to mice with LPS-related systemic inflammation (Fig. 7a–d, P < 0.001). Of note, the “ring enhancing” PET uptake pattern in mice (Fig. 7c) was similar to that noted in the patient with invasive cerebral aspergillosis (Fig. 4a), which is well described clinically39,40, and likely represents a lack of blood supply and dead tissues in the necrotic core of the lesion. Postmortem ex vivo biodistribution confirmed the imaging findings and tissue histology demonstrated fungal hyphae in the infected mice (Fig. 7e, f). Additionally, the 18F-FDS PET signal colocalized with the lesions noted on the MRI (Fig. 7h).
Representative MIP (left) and coronal (right) 18F-FDS PET/CT from (a) uninfected, (b) LPS-induced systemic inflammation, and (c) Aspergillus-infected mice are shown. Lesions are marked with arrows. d 18F-FDS PET SUVr (normalized to tissues from uninfected animals) derived from uninfected mice (n = 8), mice with LPS-induced systemic inflammation (n = 7), and mice with Aspergillus infection (n = 7) demonstrate significantly higher PET signal in mice with invasive cerebral aspergillosis compared to mice with LPS-related systemic inflammation (P < 0.001). e Ex vivo quantification of 18F-FDS uptake (percent injected dose [%ID]/g) in postmortem brain tissues derived from uninfected mice (n = 3), mice with LPS-induced systemic inflammation (n = 4), and mice with Aspergillus infection (n = 3) show higher 18F-FDS activity in the brain of mice infected with Aspergillus compared to controls (P = 0.029). f Representative GMS-stained brain tissues from Aspergillus-infected mice show Aspergillus hyphae. Scale bars is 100 µm. g Representative coronal (upper left), transverse (middle left) and sagittal (lower left) MIP and coronal (upper right) and transverse (lower right) CT segmentation. h Representative coronal (upper) and transverse (lower) MRI T1 (left), T2 (middle), and 18F-FDS PET/MRI (right, T1 MRI) from Aspergillus-infected mice. Cerebral lesions are marked with arrows. Aspergillus fumigatus (ATCC 1022) was used for the studies. Data are shown on a log scale as median ± IQR. Statistical analyses were performed using a two-tailed (panel (d)) or one-tailed (panel (e)) Mann-Whitney U test. LPS, lipopolysaccharide. SUVr, standardized uptake value ratio.
Discussion
Here, we describe results from a prospective study evaluating 18F-FDS PET/CT to specifically detect and localize invasive mold infections (and differentiate them from other pathologies) in a patient population representative of a tertiary care hospital setting within seven days of initiation of antifungal treatments. Although larger studies are needed to better assess the diagnostic accuracy, by applying a previously defined33 TNT cutoff of 3, in this small study, 18F-FDS PET correctly classified all patients with and without invasive mold infections.
Differentiation of microbes by selective growth media, utilizing small molecules (mostly sugars and sugar alcohols), was historically a mainstay of clinical microbiology41. We (and others) have exploited this differential metabolism by mammalian cells versus pathogens42, to develop several pathogen-specific imaging approaches15,16. While we had originally developed 18F-FDS, a radioanalog of sorbitol (sugar alcohol), to selectively image infections due to the Enterobacterales group of Gram-negative bacteria32,33, a study by Kim et al. described the utility of 18F-FDS PET to detect and monitor invasive pulmonary aspergillosis in animal models27. However, the Kim et al. study could not explain why Aspergillus infections could be visualized well in vivo using 18F-FDS PET, even though the in vitro uptake of 18F-FDS is lower in molds compared to E. coli for the same mass. Although growth dynamics and the different morphotypes noted for fungi (but not for bacteria) could affect in vitro uptake, we hypothesized that differences in the biomass occupied by molds versus bacteria within infectious lesions may explain the excellent PET signal noted in vivo. Our study confirmed this, and we demonstrate that the biomass occupied by Aspergillus spp. in pulmonary, cerebral, or rhinosinusal infections is ~50-fold higher than for bacterial infections (Fig. 6). Similar findings were observed in tissues obtained from animal infections. These mechanistic findings have important implications for the development of novel pathogen-specific imaging tracers. Further, Lai et al. have hypothesized that the low uptake of 18F-FDS by Aspergillus is likely due to the need for induction of sorbitol dehydrogenase by sorbitol, which may not be achieved by PET tracers43. Our study disproves this hypothesis, demonstrating that Aspergillus rapidly accumulates 18F-FDS without the need for induction by sorbitol, via a saturable process (Fig. 5). Using radio-TLC studies, we further demonstrate that 18F-FDS is indeed rapidly metabolized intracellularly by A. fumigatus, which is identical to the intracellular metabolism of 18F-FDS noted in E. coli (Fig. S8).
Our data show that 18F-FDS-PET can distinguish invasive mold infections from normal tissue and sterile inflammation or cancerous lesions in humans. This is due to low tissue background signal from 18F-FDS in several tissues, including the lungs and the brain, which are the two primary sites for mold infections. Unlike molds and some other pathogens, mammalian cells cannot metabolize sorbitol, and substitution of the hydroxyl group by fluorine at the second position in 18F-FDS completely abrogates recognition by mammalian cells44. We have previously shown that cancer cell lines do not take up 18F-FDS32, and that 18F-FDS PET demonstrates low uptake in cancerous lesions in animal and human studies32,33. Some brain tumors or inflammatory pathologies have been reported to accumulate 18F-FDS45,46,47, but these reports describe pituitary lesions (outside the blood-brain barrier), and / or measure 18F-FDS uptake immediately after tracer injection (5 min in the case reports45,46 and 60 min in animal studies47), and thus have a higher background signal. Utilizing dynamic 18F-FDS PET over 120 min, we have demonstrated that while brain tumors can demonstrate initial PET uptake, this signal dissipates over time (after 60 min)32. Therefore, the findings previously noted in brain tumors or inflammatory pathologies are consistent with a non-specific, blood pool effect, consistent with capillary leak at the site of inflammation or tumor48. In our studies, imaging was performed 120 or 180 min after tracer injection with significant clearance of the background32,33,49, with the TNT measurements for invasive mold infections highest in the brain. For example, in patient 4, where imaging was performed 180 min after tracer injection, the 18F-FDS PET derived TNT in the right frontal cortex lesion was 17 (Fig. S4). The corresponding head MRI (performed for clinical reasons) reported postoperative changes from prior surgeries and white matter changes, but did not demonstrate evidence of pathologic enhancement.
18F-FDS PET also shows promise for the diagnosis and localization of non-Aspergillus molds, which may have a similar clinical presentation, are on the rise due to the use of Aspergillus-targeted antifungal prophylaxis7, and are not detected by current noninvasive biomarkers (i.e., galactomannan)6. In addition to Aspergillus spp., we demonstrate rapid 18F-FDS accumulation in a diverse group of clinical isolates, including azole-resistant molds, confirming that sorbitol uptake is a conserved process. Although, Aspergillus is known to metabolize sorbitol34,35, 18F-FDS accumulation by a range of clinically relevant mold fungi is a new finding from our studies. Our findings are further supported by clinically relevant animal models of invasive pulmonary, rhinosinusal and cerebral mold infections, and human studies. In one exemplary case, 18F-FDS PET was able to localize known pulmonary invasive mold infection due to C. bantiana in a patient with a negative galactomannan test. Here, 18F-FDS PET also identified a previously undiagnosed cerebral involvement with C. bantiana that was not detected on brain MRI performed for clinical reasons (Fig. S4). C. bantiana has a high propensity for dissemination to the brain and is associated with high mortality due to delays in diagnosis (median of 115 days)50. Early and accurate identification of cerebral infection has implications for treatment (the need for an antifungal agent with CNS penetration) and for prognostication. This example highlights many potential advantages of 18F-FDS PET, including its utility in diagnosing and localizing infections due to diverse clinically important molds not otherwise detected by currently available biomarkers, and a level of sensitivity superior to the best currently available imaging tools. Although larger clinical studies are needed to validate sensitivity, our data suggest that 18F-FDS PET is expected to have high sensitivity. Due to a lack of other noninvasive diagnostic tools for deep-seated invasive mold infections, the high sensitivity of 18F-FDS PET, could also be beneficial in ruling out mold infections, which may reduce the use of unnecessary empiric antifungal treatments. To our knowledge, in vivo detection of non-Aspergillus mold infections as well as the use of rhinosinusal and cerebral models of invasive mold disease, including non-Aspergillus molds, are novel and have not been reported previously.
18F-FDS PET is a promising diagnostic tool that can be accessible to a range of institutions and patient populations. PET is now a routine clinical tool51, and is increasingly available both in high- and low-income and middle-income countries. Due to the short half-life of most PET radionuclides, e.g., 109 min for F-18, the tracer has to be synthesized on-site or at a nearby location, requiring dedicated infrastructure (cyclotron) and trained personnel, thus limiting on-demand supply. However, 18F-FDS can be synthesized rapidly from commercially available 18F-FDG by a simple, one-step kit-based method37, allowing on-demand synthesis (at room temperature) and global availability of 18F-FDS. Further, high specific activity (radioactivity per mass) is not required, as even low specific activity 18F-FDS (20–50 Ci/mM) produces optimal imaging characteristics32,42. We believe this is a major advantage that will allow global, on-demand 18F-FDS availability at any site with access to a PET scanner. Most patients suspected of invasive mold infections will undergo imaging studies such as CT or MRI as the current standard of care. 18F-FDS PET/CT could be incorporated into the current clinical workflow for these patients, especially in resource-rich settings. Given the rapid clearance of 18F-FDS, the radiation exposure from a whole-body 18F-FDS PET combined with a low-dose CT scan is estimated to be ~1 rem for an adult patient, which is less than the radiation exposures from a contrast-enhanced chest-abdomen-pelvis CT (~2 rem), routinely performed clinically52,53. PET imaging typically takes 15–60 min, performed 60–120 min after tracer administration, and provides results on the same day. In fact, the 18F-FDS PET/CT results in the current study were available much earlier than the conventional tests used to establish the confirmatory diagnosis for invasive mold infections. Newer technologies such as total-body PET scanners enable high-sensitivity imaging with dramatically improved signal-to-noise ratio and will further shorten the scan duration while increasing sensitivity54.
Our studies have limitations. Many PCR based tests (and fungal cultures) provide species or genus level identification and can also detect mixed mold infections (reported rarely in immunocompromised patients55). Although 18F-FDS PET cannot distinguish aspergillosis from other mold infections (e.g. mucormycosis) an approach combining 18F-FDS PET with galactomannan testing could aid in distinguishing between the two infections. Further, due to the need for invasive procedures, many mucormycosis cases remain undiagnosed or the diagnoses are significantly delayed. Therefore, 18F-FDS PET could not only help in the early diagnosis, but when positive, also justify the need for an earlier invasive procedure as well as highlight the best sites(s) to sample the infection, due to its high sensitivity and specificity. Moreover, our prior studies have shown that there is no correlation between the peripheral white blood cell or neutrophil counts with the 18F-FDS PET signal in the infected patients33, suggesting that 18F-FDS PET is not dependent on host inflammatory cells. Additionally, all animal experiments in the current studies utilized neutropenic mice (induced with cyclophosphamide), which did not preclude 18F-FDS PET to specifically detect, localize and monitor invasive fungal infections. We utilized stringent criteria in this clinical study (i.e., prospective enrollment, eventual confirmation of invasive mold infection from the infection site, and inclusion of patients who received ≤7 days of antifungal treatment prior to the 18F-FDS PET) strengthening the reliability and clinical relevance of our results, given that empiric antifungal treatments are often initiated before imaging studies or definitive testing for invasive mold infections are performed14. However, these criteria were also an impediment to patient recruitment, leading to a small sample size for our clinical study. A validated PET tracer in clinical use would not be subject to such study restrictions, so we believe that 18F-FDS PET will ultimately have applications in many more patients for whom early diagnosis and localization can significantly alter the diagnostic pathway, treatment, and prognosis. Moreover, due to the ease of tracer synthesis and on-demand availability with the one-step, solid-phase cartridge system37, this technology could be practically implemented in most settings. Notably, Enterobacterales bacteria (e.g., E. coli, Salmonella, etc.) also metabolize sorbitol and acute [high burden, 7 log10 colony forming units (CFU)/mL] infections would be visualized by 18F-FDS PET33. However, Enterobacterales bacteria are rarely in the differential for presentations consistent with invasive mold infections and patient characteristics, response to empiric antibiotics can easily distinguish Enterobacterales from mold infections. Similarly, while Pseudomonas pneumonia can present in this patient population, Pseudomonas do not accumulate 18F-FDS, and no 18F-FDS PET signal was noted in the mouse model of Pseudomonas pneumonia. Future studies will also evaluate whether 18F-FDS PET can be used to detect other invasive fungal infections, such as Candida and Cryptococcus.
In summary, we present data from a prospective clinical study evaluating whole-body 18F-FDS PET/CT in nine patients with high-suspicion of invasive mold infections or other pathologies. We demonstrate that 18F-FDS PET is feasible and can rapidly detect and localize invasive pulmonary and cerebral infections due to Aspergillus, non-Aspergillus, and azole-resistant molds, and differentiate them from sterile inflammation or cancer. Imaging data were confirmed by culture or molecular assays, and observations in humans were replicated using clinically relevant animal models. Animal studies also demonstrated that 18F-FDS PET can monitor the response to antifungal treatments. While larger clinical studies are needed to validate these findings, our data strongly support the role of 18F-FDS as an easily synthesizable PET tracer for noninvasive detection and localization of invasive mold infections throughout the body.
Methods
All protocols were approved by the Johns Hopkins University Biosafety, Radiation Safety, Animal Care and Use (MO22M354) and Institutional Review Board (IRB00097331, IRB00071002) Committees.
18F-FDS PET/CT in prospectively enrolled patients
18F-FDS was synthesized as a sterile solution with high specific activity and high radiochemical purity by the Johns Hopkins PET Center. This prospective observational study was performed in accordance with the U.S. FDA Radioactive Drug Research Committee guidelines56, with localization of 18F-FDS PET signal to the infection sites as the primary outcome measure. This study was registered on clinicaltrials.gov (NCT05611892)57. Nine patients (four with newly diagnosed invasive mold infections, and five control patients with other pathologies33) were prospectively recruited from the Johns Hopkins Hospitals between October 2016 to July 2024 using the following criteria (Table S1). Confirmatory diagnosis of mold infections was established in all four patients, using culture or molecular assays. Sterile inflammatory pathologies were determined clinically and/or by tissue biopsies, and oncologic diagnosis was made via tissue biopsy (Table S2). Written informed consent was obtained from each subject, physical examination performed by a trained physician and screening laboratory tests performed and reviewed by the study principal investigator to confirm eligibility. On the day of imaging, a low-dose CT, selected to minimize the radiation exposure, was performed. An intravenous injection of 18F-FDS (503 ± 191 MBq) was administered and PET was acquired at 120 min after tracer administration (except patient 4 where acquisition was at 180 min post-tracer injection) from the skull vertex to mid-thigh with 3 min per bed-position, using a Siemens Biograph PET/CT scanner. All patients were evaluated immediately after finishing the PET scan and 20-25 days to assess early and delayed adverse events.
Human histology
Infected tissue samples from eight unique patients with confirmed pulmonary, cerebral or rhinosinusal invasive Aspergillus spp. infections from Johns Hopkins Hospitals were obtained (patient characteristics outlined in Table S3). Multiple random slides were obtained from each tissue sample, stained with GMS, digitally scanned and analyzed at 40x magnification. Additionally, publicly available digitized slides of bacterial infections in patients (case IDs: 963, 749, 1692, 1688) from Infectious Diseases Images eMicrobes Digital Library were analyzed38. The percentage of area occupied by the mold or bacteria was calculated using automated segmentation (ImageJ) after standardizing the area analyzed in each sample (Fig. 6, S10).
In vitro assays
A. fumigatus (ATCC 1022), or clinical isolates from the Johns Hopkins Hospitals clinical microbiology laboratory including Rhizopus arrhizus and Mucor circinelloides were grown on potato dextrose agar (PDA) plates at 37 °C and conidia were collected in sterile phosphate buffered saline (PBS) with Tween 80 (0.01%). A manual count of the conidia density (conidia/mL) was performed using a hemocytometer. Fungal cultures were heat killed by exposure to 95 °C heat for 60 min. 18F-FDS (5 μCi/mL) was co-incubated with the fungi grown at 37 °C with shaking and samples were collected at different time points followed by pelleting and washing thrice with PBS. Activity for each sample pellet was measured immediately using an automated gamma counter (PerkinElmer). Counts were corrected for background and decay and normalized to pellet weight (mg). Bacterial and mammalian cells (THP-1) studies were performed as described before32. All studies were performed in triplicate samples.
Metabolite studies
A. fumigatus (ATCC 1022) or E. coli (ATCC 25922) cultures were incubated with 18F-FDS. Aliquots of 1 mL were pelleted by centrifugation and washed with PBS. The cells were lysed and the cytoplasmic fraction was separated by ultracentrifugation and filtered (0.22 µm syringe). The solute was analyzed on a radio-TLC (Eckert & Ziegler) using 18F-FDS and 18F-FDG as reference.
Animal studies
Female BALB/c mice (6-8 weeks old, Charles River) were housed in pre-sterilized, filter-topped cages, with unrestricted access to both food and water [medicated to prevent bacterial superinfection prophylaxis with tetracycline (Sigma)]. All animals received anesthesia with 2% isoflurane and O2 mixture for infection inoculation, LPS-induced inflammation and imaging.
For the mold infection studies, mice were immunosuppressed using cortisone acetate (Sigma, 200 mg/Kg) administered subcutaneously at day five and one prior to mold infection and cyclophosphamide (Sigma, 150 mg/Kg) administered intraperitoneally at day four and one day before infection27,58. For invasive pulmonary aspergillosis, anesthetized mice were hung by their upper incisors and secured to a support. A superficial 1 cm incision was made in the midline of the neck to visualize and successfully cannulate the trachea59. The intubation was confirmed upon connection to a ventilator, and then 40 µL suspension of fungal conidia [7 × 106 A. fumigatus (ATCC 1022) conidia/mouse] was used for the infection followed by two days of incubation prior to imaging. Rhinosinual infection was carried out by a slow intranasal instillation of 15 µL of the fungal conidia suspension [1 × 108 A. fumigatus (ATCC 1022), Rhizopus arrhizus or Mucor circinelloides conidia per mouse) under anesthesia with two days of incubation prior to imaging. M. circinelloides is one of the most common mucoralean fungi isolated from clinical samples60, and causes human mucormycosis including fungal rhinosinusitis61. In a subset of mice with rhinosinusal Aspergillus-infection, voriconazole (MedChem; 40 mg/kg/day) was administered daily (5-days a week) via oral gavage for one week, starting two days after infection. For invasive cerebral aspergillosis, anesthetized mice were infected with 3 µL suspension of fungal conidia [1 × 106 A. fumigatus (ATCC 1022) conidia/mouse] via a burr hole using a Hamilton syringe (Hamilton, 88,000) and stereotaxic instrument (David KOPF instrument, model 900) with the following coordinates: 2 mm dorsal to bregma, 2 mm lateral to middle line and 2 mm deep. After the inoculation, the hole was sealed with bone wax and the scalp was closed using a surgical staple and the infection incubated for one day prior to imaging. The model of sterile pneumonia was developed using an intratracheal instillation of 30 µL of LPS solution (5 µg/g). For the Pseudomonas pneumonia model, mice were immunosuppressed with cyclophosphamide (Sigma, 150 mg/kg, administered intraperitoneally on day 4 and one day before infection). On the day of infection 40 µL of a Pseudomonas aeruginosa (ATCC 10145) suspension (2 × 105 CFU/mouse) was administered via intratracheal instillation, and imaging performed 3 days later. The pulmonary bacterial burden was assessed immediately after imaging. For systemic inflammation mice were intraperitoneally administered 50 µL of LPS solution four days prior (2 µg/g) and the day of imaging (6 µg/g)62,63.
Imaging: Animals were injected via tail vain with a bolus of 8.89 MBq ± 1.43 of 18F-FDG or 8.54 ± 1.56 MBq of 18F-FDS. A 15 min PET was acquired 45 min after injection of 18F-FDG, and 120 min after injection of 18F-FDS, using a nanoScan PET/CT (Mediso). A CT scan was obtained for anatomical reference. MRI was performed using a 7 T preclinical MRI (MR Solutions) and mouse 1H body coils with 1 mm slices and fat saturation. The T1W and T2W sequences utilized a repetition time of 1000 ms and an echo time of 11 and 45 ms, respectively.
Ex vivo Biodistribution: These studies were performed immediately after the completion of the imaging studies. Animals were sacrificed, and the tissues were perfused by infusing 6 mL of PBS per animal through the left ventricle. The organs were extracted thereafter and the activity in each organ was measured immediately using an automated gamma counter (PerkinElmer) and presented as the percentage injected dose per gram of tissue (%ID/g).
Histopathology: Slides were prepared from tissues fixed in 4% paraformaldehyde from our current mold infection animal models. Multiple random slides were obtained from each tissue sample, stained with GMS, digitally scanned and analyzed as described above. Fixed tissue samples from mice with E. coli myositis with a target implantation of 7 log10 CFU units were obtained from our prior studies32. Multiple random slides were obtained from each tissue sample, Gram stained, digitally scanned and analyzed as described above.
Image analysis
Human images were analyzed using Mirada XD™ 3.6.8 (Mirada Medical) and PMOD version 3.402 (PMOD Technologies LLC), while the animal images were analyzed using VivoQuant 2020 (Invicro)32,33,64. Three-dimensional VOIs were drawn manually using the CT as a reference and the PET activity was measured. When multiple VOIs were drawn in the same patients, they were from independent lesions or different parts of large lesions (with no overlap of any VOI). The human PET data are presented as TNT which is the ratio of the mean PET signal at the sites of pathology to the unaffected sites of the same tissue. The animal PET data are presented as SUVr which is the ratio of the mean PET signal at the sites of pathology to the same tissue from uninfected animals. All data were visualized using AMIRA 5.2.1 (Visage Imaging, Inc.) or AMIDE 1.0.6 (Andreas Loening).
Statistical analysis
Data were analyzed using Prism 10.0 (GraphPad Software Inc). All data are represented as median ± IQR or mean ± SD and comparisons were made using one-tailed or two-tailed Mann-Whitney U test. P values ≤ 0.05 were considered statistically significant. No sex-based analyses were performed because the study design was not explicitly set up to examine sex differences.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data are available in the main text or the supplementary materials. Source data file has been provided with this paper. Source data are provided with this paper.
References
McCarthy, M., Rosengart, A., Schuetz, A. N., Kontoyiannis, D. P. & Walsh, T. J. Mold infections of the central nervous system. N. Engl. J. Med 371, 150–160 (2014).
Lee, S. O. Diagnosis and treatment of invasive mold diseases. Infect. Chemother. 55, 10–21 (2023).
Denning, D.W. Global incidence and mortality of severe fungal disease. Lancet Infect. Dis. 24, e428–e438 (2024).
Hoenigl, M. et al. COVID-19-associated fungal infections. Nat. Microbiol 7, 1127–1140 (2022).
Pasquier, G. COVID-19-associated mucormycosis in India: why such an outbreak?. J. Mycol. Med 33, 101393 (2023).
Lamoth, F. & Kontoyiannis, D. P. Therapeutic challenges of non-aspergillus invasive mold infections in immunosuppressed patients. Antimicrob. Agents Chemother. 63, e01244–e01319 (2019).
Wattier, R. L. et al. A prospective, International Cohort Study of invasive mold infections in children. J. Pediatr. Infect. Dis. Soc. 4, 313–322 (2015).
Hoenigl, M. et al. Global guideline for the diagnosis and management of rare mould infections: an initiative of the European Confederation of Medical Mycology in cooperation with the International Society for Human and Animal Mycology and the American Society for Microbiology. Lancet Infect. Dis. 21, e246–e257 (2021).
World Health Organization. WHO fungal priority pathogens list to guide research, development and public health action. https://www.who.int/publications/i/item/9789240060241 (2022).
Bergin, S. P. et al. Plasma microbial cell-free DNA sequencing in immunocompromised patients with pneumonia: a prospective observational study. Clin. Infect. Dis. 78, 775–784 (2024).
Blauwkamp, T. A. et al. Analytical and clinical validation of a microbial cell-free DNA sequencing test for infectious disease. Nat. Microbiol 4, 663–674 (2019).
Mercier, T. et al. Defining galactomannan positivity in the updated EORTC/MSGERC consensus definitions of invasive fungal diseases. Clin. Infect. Dis. 72, S89–S94 (2021).
Zhou, W. et al. Diagnostic value of galactomannan antigen test in serum and bronchoalveolar lavage fluid samples from patients with nonneutropenic invasive pulmonary aspergillosis. J. Clin. Microbiol 55, 2153–2161 (2017).
Ordonez, A.A., et al. Molecular imaging of bacterial infections: Overcoming the barriers to clinical translation. Sci. Transl. Med. 11, eaax8251 (2019).
Chen, X. et al. Visualizing bacterial infections with novel targeted molecular imaging approaches. J. Infect. Dis. 228, S249–S258 (2023).
Lai, J., Wang, B., Petrik, M., Beziere, N. & Hammoud, D. A. Radiotracer development for fungal-specific imaging: past, present, and future. J. Infect. Dis. 228, S259–S269 (2023).
Kuhlman, J. E. et al. CT of invasive pulmonary aspergillosis. AJR Am. J. Roentgenol. 150, 1015–1020 (1988).
Leroy-Freschini, B. et al. 18F-FDG PET/CT for invasive fungal infection in immunocompromised patients. QJM 111, 613–622 (2018).
Ankrah, A.O., et al. The Added Value of [(18)F]FDG PET/CT in the Management of Invasive Fungal Infections. Diagnostics 11, 137 (2021).
Sharma, P., Mukherjee, A., Karunanithi, S., Bal, C. & Kumar, R. Potential role of 18F-FDG PET/CT in patients with fungal infections. AJR Am. J. Roentgenol. 203, 180–189 (2014).
Petrik, M. et al. 68Ga-siderophores for PET imaging of invasive pulmonary aspergillosis: proof of principle. J. Nucl. Med. 51, 639–645 (2010).
Petrik, M. et al. 68Ga-triacetylfusarinine C and 68Ga-ferrioxamine E for Aspergillus infection imaging: uptake specificity in various microorganisms. Mol. Imaging Biol. 16, 102–108 (2014).
Rolle, A. M. et al. ImmunoPET/MR imaging allows specific detection of Aspergillus fumigatus lung infection in vivo. Proc. Natl Acad. Sci. USA 113, E1026–1033 (2016).
Davies, G. et al. Towards translational immunoPET/MR imaging of invasive pulmonary aspergillosis: the humanised monoclonal antibody JF5 detects aspergillus lung infections in vivo. Theranostics 7, 3398–3414 (2017).
Thornton, C. R. Molecular imaging of invasive pulmonary aspergillosis using immunoPET/MRI: the future looks bright. Front. Microbiol. 9, 691 (2018).
Henneberg, S., et al. Antibody-guided in vivo imaging of Aspergillus fumigatus lung infections during antifungal azole treatment. Nat. Commun. 12, 1707 (2021).
Kim, D. Y., et al. In vivo imaging of invasive aspergillosis with (18)F-fluorodeoxysorbitol positron emission tomography. Nat. Commun. 13, 1926 (2022).
Lai, J. et al. PET imaging of Aspergillus infection using Zirconium-89 labeled anti-beta-glucan antibody fragments. Eur. J. Nucl. Med Mol. Imaging 51, 3223–3234 (2024).
Shah, S. et al. Development and preclinical validation of 2-deoxy 2-[(18)F]fluorocellobiose as an Aspergillus-specific PET tracer. Sci. Transl. Med. 16, eadl5934 (2024).
Basuli, F. et al. Fully automated cassette-based synthesis of 2-Deoxy-2-[(18)F]fluorocellobiose using trasis allinone module. J. Label. Comp. Radiopharm. 67, 308–313 (2024).
Schwenck, J., et al. Antibody-guided molecular imaging of aspergillus lung infections in leukemia patients. J. Nucl. Med. https://doi.org/10.2967/jnumed.121.263251 (2022).
Weinstein, E. A. et al. Imaging Enterobacteriaceae infection in vivo with 18F-fluorodeoxysorbitol positron emission tomography. Sci. Transl. Med 6, 259ra146 (2014).
Ordonez, A.A., et al. Imaging Enterobacterales infections in patients using pathogen-specific positron emission tomography. Sci. Transl. Med. 13, eabe9805 (2021).
Elshafei, A. M. & Abdel-Fatah, O. M. Evidence for a non-phosphorylated route of galactose breakdown in cell-free extracts of Aspergillus niger. Enzym. Micro. Technol. 29, 76–83 (2001).
Koivistoinen, O. M., Richard, P., Penttila, M., Ruohonen, L. & Mojzita, D. Sorbitol dehydrogenase of Aspergillus niger, SdhA, is part of the oxido-reductive D-galactose pathway and essential for D-sorbitol catabolism. FEBS Lett. 586, 378–383 (2012).
Li, Z. B. et al. The synthesis of 18F-FDS and its potential application in molecular imaging. Mol. Imaging Biol. 10, 92–98 (2008).
Mota, F., De Jesus, P. & Jain, S. K. Kit-based synthesis of 2-deoxy-2-[(18)F]-fluoro-D-sorbitol for bacterial imaging. Nat. Protoc. 16, 5274–5286 (2021).
Partners’ Infectious Disease Images. Infectious Diseases Images eMicrobes Digital Library. https://www.idimages.org/atlas (2024).
DeLone, D. R. et al. Disseminated aspergillosis involving the brain: distribution and imaging characteristics. AJNR Am. J. Neuroradiol. 20, 1597–1604 (1999).
Tempkin, A. D., Sobonya, R. E., Seeger, J. F. & Oh, E. S. Cerebral aspergillosis: radiologic and pathologic findings. Radiographics 26, 1239–1242 (2006).
Mac Faddin, J.F. Biochemical tests for identification of medical bacteria, (Williams & Wilkins, Baltimore, 1976).
Ordonez, A. A. et al. A systematic approach for developing bacteria-specific imaging tracers. J. Nucl. Med. 58, 144–150 (2017).
Lai, J. et al. Evaluation of 2-[(18)F]-fluorodeoxysorbitol PET imaging in preclinical models of aspergillus infection. J. Fungi 8, 25 (2021).
Scott, M. E. & Viola, R. E. The use of fluoro- and deoxy-substrate analogs to examine binding specificity and catalysis in the enzymes of the sorbitol pathway. Carbohydr. Res. 313, 247–253 (1998).
Kong, Z., Wang, Y. & Ma, W. & Cheng, X. Role of (18)F-fluorodeoxyglucose (FDG) and (18)F-2-fluorodeoxy sorbitol (FDS) in autoimmune hypophysitis: a case report. BMC Endocr. Disord. 20, 84 (2020).
Cheng, X., Zhu, W. & Cui, R. Increased 18F-2-fluorodeoxysorbitol (18F-FDS) activity in a pituitary spindle cell carcinoma. Clin. Nucl. Med 41, 953–955 (2016).
Hugon, G., et al. [(18)F]2-fluoro-2-deoxy-sorbitol PET imaging for quantitative monitoring of enhanced blood-brain barrier permeability induced by focused ultrasound. Pharmaceutics 13, 1752 (2021).
Javadi, M. S., Ordonez, A. A. & Jain, S. K. Re: Increased 18F-2-fluorodeoxysorbitol (18F-FDS) activity in a pituitary spindle cell carcinoma. Clin. Nucl. Med 42, 649 (2017).
Zhu, W., et al. Biodistribution and Radiation Dosimetry of the Enterobacteriaceae-Specific Imaging Probe [F]Fluorodeoxysorbitol Determined by PET/CT in Healthy Human Volunteers. Mol. Imaging Biol. 18, 782–787 (2016).
Chakrabarti, A. et al. Brain abscess due to Cladophialophora bantiana: a review of 124 cases. Med Mycol. 54, 111–119 (2016).
Hricak, H. et al. Medical imaging and nuclear medicine: a Lancet Oncology Commission. Lancet Oncol. 22, e136–e172 (2021).
Wright, W. F. & Auwaerter, P. G. Fever and fever of unknown origin: review, recent advances, and lingering dogma. Open Forum Infect. Dis. 7, ofaa132 (2020).
Bharucha, T. et al. Diagnostic yield of FDG-PET/CT in fever of unknown origin: a systematic review, meta-analysis, and Delphi exercise. Clin. Radio. 72, 764–771 (2017).
Henrich, T. J., Jones, T., Beckford-Vera, D., Price, P. M. & VanBrocklin, H. F. Total-body PET imaging in infectious diseases. PET Clin. 16, 89–97 (2021).
McLintock, L. A., Gibson, B. E. & Jones, B. L. Mixed pulmonary fungal infection with Aspergillus fumigatus and Absidia corymbifera in a patient with relapsed acute myeloid leukaemia. Br. J. Haematol. 128, 737 (2005).
FDA. Guidance for industry and researchers: the radioactive drug research committee: human research without an investigational new drug application. (2010).
University, J.H. Biodistribution and pathophysiology study of 18F-fluorodeoxysorbitol (18F-FDS) in diseased patients. (ClinicalTrials.gov, 2024).
Wang, Y. et al. Detection of Aspergillus fumigatus pulmonary fungal infections in mice with (99m)Tc-labeled MORF oligomers targeting ribosomal RNA. Nucl. Med Biol. 40, 89–96 (2013).
Villabona-Rueda, A., Wang, D. & D’Alessio, F.R. Experimental model to evaluate resolution of pneumonia. J. Vis. Exp. https://doi.org/10.3791/63925 (2023).
Badali, H. et al. Epidemiology and antifungal susceptibilities of mucoralean fungi in clinical samples from the United States. J. Clin. Microbiol 59, e0123021 (2021).
Erami, M., et al. Microbial and clinical epidemiology of invasive fungal rhinosinusitis in hospitalized COVID-19 patients, the divergent causative agents. Med. Mycol. 61, myad020 (2023).
Liverani, E. et al. LPS-induced systemic inflammation is more severe in P2Y12 null mice. J. Leukoc. Biol. 95, 313–323 (2014).
Xingi, E., et al. LPS-induced systemic inflammation affects the dynamic interactions of astrocytes and microglia with the vasculature of the mouse brain cortex. Cells 12, 1418 (2023).
Chen, X., et al. Dynamic PET reveals compartmentalized brain and lung tissue antibiotic exposures of tuberculosis drugs. Nat. Commun. 15, 6657 (2024).
Acknowledgments
We thank all the subjects who participated in the study. We would also like to thank Jeff Leal (Johns Hopkins Hospitals) for curating the human imaging data, Kelly Flavahan (Johns Hopkins Hospitals) for assistance with PET/CT imaging, and Warda Memon (Johns Hopkins Hospitals) for assistance with curating the clinical isolates. This work was funded by the U.S. National Institutes of Health R01-AI145435-A1 (S.K.J.), R01-AI153349 (S.K.J.), R01-HL131829 (S.K.J.), S10-OD030381-A1 (S.K.J.), S10-OD034232 (M.A.L.), K08-AI139371 (E.W.T.) and funds from the Hankins Young Investigator Award (S.K.J.).
Author information
Authors and Affiliations
Contributions
C.E.R-G. and S.K.J. conceptualized and designed the studies. M.S., A.K., and ML.S. performed the in vitro studies. M.S. and L.S.C. performed bacterial metabolite studies. C.E.R-G. developed the animal models and performed the animal studies with help from Y.M-L. and X.C. O.J.N-M., E.W.T., M.A.L., and S.K.J. wrote the protocol for the 18F-FDS PET clinical studies with support from S.S. and S.X.Z. O.J.N-M. and X.C. coordinated the clinical studies and E.W.T., K.R.C., O.S.K., and S.K.J. recruited and consented the patients. S.X.Z. provided clinical isolates and human histology slides. C.E.R-G. analyzed the data with support from O.J.N-M., M.S., and Y.M-L. S.K.J. provided funding and supervised the project. C.E.R-G. and S.K.J. wrote the manuscript with substantial input from all co-authors.
Corresponding authors
Ethics declarations
Competing interests
S.K.J. is a co-inventor on pending patent USPA #63/071,755 on the solid-phase cartridge to formulate ready-to-use 18F-FDS and pending patent USPA #63/699,572 on Noninvasive Detection of Invasive Mold Infections by 18F-FDS PET, filed by Johns Hopkins University. All other authors declare that they have no competing interests.
Peer review
Peer review information
Nature Communications thanks Jung-Joon Min, and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ruiz-Gonzalez, C.E., Nino-Meza, O.J., Singh, M. et al. 18F-Fluorodeoxysorbitol PET for noninvasive detection of invasive mold infections: preclinical and first-in-human studies. Nat Commun 16, 6395 (2025). https://doi.org/10.1038/s41467-025-61700-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-61700-6