Abstract
While the clinical utility of conventional antibody therapies is undeniable, their therapeutic potential is often constrained high antigen loads and the recycling of antibody-antigen complexes via neonatal Fc receptor (FcRn). Here, we present a platform, based on a design similar to bispecific antibodies, FcγRIIb-Targeting Chimeras (FcRTAC). These constructs recognise antigens with one arm and bind FcγRIIb with the other arm to harness the unique endocytic properties of FcγRIIb to direct the recognized pathogenic antigens to lysosomes for irreversible degradation. The FcRTAC platform demonstrates broad therapeutic potential across multiple disease-relevant targets, including IgE, proprotein convertase subtilisin/kexin type 9 (PCSK9) and amyloid-β (Aβ). Notably, a single intravenous administration of blood brain barrier (BBB)-penetrating adeno-associated viral vector (AAV) encoding an Aβ-targeting FcRTAC construct achieves sustained therapeutic effects, establishing proof-of-concept for AAV-mediated delivery of an Aβ degrader as a strategy for Alzheimer’s disease treatment. Our comprehensive investigation of binding properties of FcRTACs reveals critical molecular determinants of function and enables development of optimized engineering approaches. In summary, our approach represents a versatile therapeutic platform for treating diverse diseases ranging from autoimmune disorders to neurodegenerative conditions, while simultaneously serving as a user-friendly, plug-and-play research tool for extracellular protein knockout in basic biological research.
Similar content being viewed by others
Introduction
The conventional antibodies, typically monoclonal IgG molecules, can neutralize biological functions by blocking molecular interactions. For example, adalimumab can specifically bind to soluble TNF-α to alleviate the progression of disease1,2. Omalizumab is a humanized monoclonal antibody that neutralizes free IgE and blocks its binding to FcεRI, thus preventing mast cell activation in moderate-to-severe allergic asthma3. Lecanemab, an anti- amyloid-β (Aβ) monoclonal antibody, can reduce the rate of disease progression and slow cognitive and functional decline in patients with early Alzheimer’s disease (AD). Lecanemab exhibits preferential binding to small Aβ oligomers and protofibrils, potentially inhibiting their aggregation into mature fibrils and amyloid plaques. Concurrently, it enhances microglia-mediated clearance of these soluble Aβ species through Fc receptor-dependent phagocytosis4,5. However, there are some limitations for the conventional antibodies6. The neutralizing capacity of conventional antibodies is fundamentally limited by their stoichiometric binding properties. This limitation arises from the bivalent nature of IgG antibodies, where each molecule contains only two antigen-binding sites. Therefore, high dose of conventional antibody is required to completely neutralize the function of the antigen when the antigen is present in abundance. For example, the recommended dose of eculizumab for the treatment of paroxysmal nocturnal hemoglobinuria is about 1000 mg every two weeks. Omalizumab requires 150–600 mg administered every 2–4 weeks according to clinical guidelines. When the serum level of pathogenic protein exceeds the maximum neutralizing capacity of therapeutic antibodies, the clinical efficacy will inevitably be compromised. In addition, binding of antibody to antigen can profoundly increase the half-life of antigen due to the recycling of the antibody-antigen complexes by the neonatal Fc receptor (FcRn) in the endosomes7.
To address these issues and reduce therapeutic dose requirements, diverse platforms of extracellular targeted protein degradation (eTPD) have been created to degrade extracellular antigens by trafficking them to the lysosome for degradation6. Igawa et al. pioneered the “sweeping antibody” technology to eliminate highly abundant soluble antigens from circulation. This technology combines a pH-dependent antigen-binding domain and an engineered Fc domain. The antibody binds to antigen at extracellular physiological pH, while dissociating in acidic endosomes following internalization. This mechanism directs antigens to lysosomal degradation, whereas the antibody is recycled via FcRn8,9. In addition, to enhance the cellular uptake of antigen-bound antibodies, the Fc regions can be engineered to increase their affinity for FcRn or FcγRIIb, or modified via surface charge adjustments. Notably, the sweeping antibody technology has already been clinically validated in therapeutics such as satralizumab (an anti-IL-6R antibody) for the treatment of neuromyelitis optica spectrum disorder (NMOSD). Igawa et al. have further identified additional targets for sweeping antibodies, including IgE and complement component C5a8. Bertozzi and co-workers pioneered the utilization of glycan-targeted recycling receptors such as the cation-independent mannose 6-phosphate receptor (CI-M6PR) or asialoglycoprotein receptor (ASGPR) to aid the lysosomal trafficking and degradation of antigen10,11. The group bioconjugated glycan ligands for the CI-M6PR or ASGPR to antigen-targeting antibody to produce constructs called lysosome-targeting chimeras (LYTAC). LYTAC molecules form a ternary complex with the target antigen and the receptor and induce internalization of the complex, leading to lysosome trafficking and degradation of target protein. Around the same period, several research groups developed innovative bifunctional molecules by strategically conjugating lysosome-targeting moieties with antigen-binding domains6,12. In addition to exploiting the internalizing receptor, researchers have developed bispecific antibodies antibody-based proteolysis-targeting chimeras (AbTACs) and proteolysis-targeting antibodies (PROTABs), two independently developed platforms sharing the same mechanistic paradigm that recruit membrane-bound E3 ligases for degradation of cell surface proteins13,14,15.
The extracellular target degraders can have significant advantages compared to the conventional neutralizing antibodies. At first, one eTPD class antibody can degrade antigens via lysosome or proteolysis mediated protein degradation so the eTPD class does not lead to the accumulation of the antigen-antibody immune complex. Secondly, some antigens such as disease-causing aggregates could be eliminated by eTPD. Nevertheless, there are many technical questions that remain to be addressed for each type of extracellular target degraders. The efficacy of sweeping antibodies may be attenuated by the excess of natural immunoglobulins in blood and tissues, and receptors are not efficiently internalized upon monomer antibody binding6. The proteolysis-targeting chimeras can degrade membrane proteins but not secreted proteins and extracellular aggregates. A common feature of all types of eTPDs is that the degraders dissociate from the receptor upon endocytosis, resulting in an extremely short half-life of eTPD. The bioconjugation chemistry creates CMC (chemistry, manufacturing, and controls) obstacles for scalable production of homogeneous bioconjugate LYTAC therapeutics.
Here we introduce FcγRIIb-Targeting Chimeras (FcRTAC), a bispecific antibody format that co-targets antigens and FcγRIIb. This design leverages the endocytic activity of FcγRIIb to direct extracellular soluble proteins and pathological aggregates toward lysosomal degradation (Fig. 1a). The platform effectively depletes diverse pathological targets in plasma, including IgE and PCSK9. Additionally, AAV-mediated delivery of an Aβ-targeting FcRTAC reduces hippocampal amyloid burden and rescues cognitive function in a mouse model. Furthermore, we elucidate the key molecular determinants governing FcRTAC functionality. This work establishes a versatile, genetically encodable strategy for degrading extracellular proteins, offering a promising therapeutic platform for a broad spectrum of diseases.
a Mechanism of action of FcRTAC. The FcRTAC binds the target antigen at one end and FcγRIIb at the other, triggering efficient FcγRIIb receptor-mediated endocytosis. Following internalization, the FcRTAC-antigen complex traffics to acidic endosomes where the antigen dissociates from FcRTAC due to pH dependent binding property and is subsequently targeted to lysosomes for proteolytic degradation, while the FcRTAC-FcγRIIb complex return to the cell surface for subsequent rounds of antigen capture. b Flow cytometry analysis of scFv-Fc (SKX-Fcs) binding to hFcγRIIb on Huvec-2b cells. Huvec-2b or control Huvec cells were incubated with various SKX-Fcs followed by an AF488-labeled secondary antibody. Fluorescence intensity was measured by flow cytometry to assess SKX-Fcs binding to FcγRIIb. The flow-cytometry gating strategy is shown in Supplementary Fig.14b. c Internalization assay of SKX-Fcs. SKX-Fcs were pre-mixed with a pH-sensitive dye-labeled anti-human Fc portion and incubated with Huvec-2b cells. Upon internalization, the pH-sensitive dye emitted red fluorescence in acidic endosomes. Fluorescence changes were monitored hourly over 24 h. d SPR assays for antibodies binding to hFcγRIIb at pH 7.4 and pH 6.0. The SK3-Fc or SK4-Fc was immobilized on a protein A biosensor chip, followed by the injection of hFcγRIIb over the flow cell. Solid curves represent raw data, while dashed curves indicate fitted curves. e Comparison of internalization efficiency for different FcRTAC formats. f,g SPR analysis of FcRTAC binding to FcγRIIb and IgE. FcRTACs were immobilized on a protein A biosensor chip, followed by sequential injection of hFcγRIIb and IgE. h Live-cell imaging of FcRTAC-mediated IgE endocytosis. Cy5-labeled IgE (red) was incubated with Huvec-2b cells in the presence or absence of FcRTACs for 1 h. Nuclei were counterstained with Hoechst (blue). Scale bar: 20 μm. Experiments were performed in triplicate and repeated three times with similar results. Panel (a) was created in BioRender. Zhao, J. (2025) https://BioRender.com/2nlicmd. Panel (e) was created in BioRender. Zhao, J. (2025) https://BioRender.com/abmfj7o.
Results
Screening of endocytosis inducing FcγRIIb-binding antibodies
In order to obtain anti-human FcγRIIb monoclonal antibodies, we employed a human single-chain variable Fragment (scFv) phage display library for screening binders to the extracellular region of human FcγRIIb (Supplementary Fig. 1a). This library has yielded OX40 binding antibody Nuvustotug which advanced into Phase I trials, CD40 agonistic antibodies and ASGPR binding antibodies16. After excluding redundant sequences or antibodies with poor expression yield, eight scFvs have been selected for conversion to Fc fusion for further characterization. The binding of these scFv-Fcs (SKX-Fcs) to FcγRIIb was detected by enzyme-linked immunosorbent assay (ELISA) (Supplementary Fig. 1b). Subsequently, we successfully generated a stable Huvec cell line expressing FcγRIIb (designated Huvec-2b) (Supplementary Fig. 1c, Supplementary Fig. 14a). Flow cytometry analysis showed that these SKX-Fcs bind with natural conformation of FcγRIIb on Huvec-2b (Fig. 1b, Supplementary Fig. 14b). We next assessed the internalization efficiency of the eight SKX-Fcs on the Huvec-2b cell line. As shown in Supplementary Fig. 1d, the SKX-Fcs mixed with a pH-sensitive dye labeled anti-human Fc Fab and then incubated with Huvec-2b cells. After internalization, the pH-sensitive dye showed red fluorescence in acidic endosomes for detection. The results showed that all eight SKX-Fcs can be internalized with the highest endocytosis of antibody observed for SK3-Fc and SK4-Fc (Fig. 1c). Therefore, SK3 and SK4 were selected for the subsequent research. The binding affinities of SK3-Fc and SK4-Fc to FcγRIIb were assessed by surface plasmon resonance (SPR) at both pH 6 and pH 7. SK3 bound FcγRIIb with an affinity approximately one order of magnitude greater than that of SK4 (Fig. 1d, Table 1).
Generation of FcγRIIb mediated FcRTAC to degrade IgE
Bispecific antibody based FcRTACs were designed with one arm binding FcγRIIb and the other arm binding target antigen to promote the clearance of target antigen. To determine the effect of spatial arrangement and inter-binder distance between the two FcγRIIb scFv domains, we engineered two distinct configurations of: in configuration FcRTAC-1, the FcγRIIb binder was fused to the carboxyl terminus (C-terminus) of the heavy chain of a pH-sensitive IgE targeting NK-2-1217; while in configuration FcRTAC-2, the binder was strategically positioned at the C-terminus of the light chain to maximize the inter-binder distance. We also engineered FcRTAC-3 containing a single FcγRIIb binder and two IgE binders using knob-in-hole (KIH) platform18 (Supplementary Fig. 1e). Through internalization assays using the Huvec-2b cell line, we observed that FcRTAC-3 failed to initiate endocytosis, suggesting that efficient receptor-mediated endocytosis requires molecular clustering facilitated by two FcγRIIb binders. FcRTAC-2 exhibited significantly lower cellular internalization efficiency compared to FcRTAC-1 (Fig. 1e). The reduced inter-binder distance in FcRTAC-1 compared to FcRTAC-2 likely facilitates more efficient receptor clustering, thereby enhancing its cellular internalization capacity.
According to this result, we constructed NK-2-12-SK3 and NK-2-12-SK4 by fusing SK3 or SK4 scFv to the C-terminus of IgE-targeting NK-2-12 heavy chain to deplete IgE. The representative purification results of NK-2-12-SK3 are shown in Supplementary Fig. 2a, b. The molecular weight of NK-2-12-SK3 was determined to be ~200 kDa by LC-MS (Supplementary Fig. 2c). Antibody thermal stability profiles were obtained using nanoDSF, revealing two characteristic melting temperatures: Tm1 (52.4 ± 0.5 °C) likely for scFv domain unfolding and Tm2 (82.8 ± 0.3 °C) for Fab domain denaturation (Supplementary Fig. 2d). The SPR results showed that both FcRTACs could simultaneously interact with IgE and FcγRIIb (Fig. 1f, g).
Both FcRTACs induced similar level of internalization by Huvec-2b cells. Moreover, the presence of IgE did not affect the internalization efficiency of FcRTACs, thereby indicating that the binding of FcRTACs to IgE does not affect antibody internalization (Supplementary Fig. 3a). NK-2-12-SK3 and NK-2-12-SK4 can considerably increase the uptake of IgE by Huvec-2b cells (Fig. 1h). Meanwhile, the IgE levels in the culture supernatants of the NK-2-12-SK3 or NK-2-12-SK4 treatment groups were lower than those in the vehicle control group (Supplementary Fig. 3b).
FcRTAC rapidly eliminated plasma targets and the clearance efficiency is enhanced by pH-sensitive binding to the target
To evaluate the efficacy of FcRTAC in enhancing antigen clearance and to investigate whether its performance can be optimized through pH-sensitive antigen binding, we conducted an in vivo experiment using humanized FcγRIIb mice. The experimental groups included: (1) pH-sensitive IgE-targeting FcRTACs (NK-2-12-SK3 and NK-2-12-SK4) (Supplementary Fig. 4), (2) pH-insensitive IgE FcRTACs (Omalizumab-SK3 and Omalizumab-SK4), (3) pH-sensitive NK-2-12, and (4) pH-insensitive IgE-targeting antibody Omalizumab. Except for the commercially available Omalizumab, NK-2-12 and all FcRTACs harbor the N297A mutation, which serves to abrogate Fc domain binding to Fc receptors. These agents were administered via intraperitoneal (i.p.) injection followed by introduction of IgE. Subsequently, total IgE and free IgE levels were quantified using ELISA (Fig. 2a). Omalizumab treatment rapidly decreased free IgE while significantly increased total plasma IgE level compared to the IgE only group (Fig. 2b-e), which recapitulated the clinical phenomenon that the total IgE are accumulated to levels 5-10 times the basal level of IgE after omalizumab administration. In contrast to Omalizumab, Omalizumab-SK3 treatment reduced the total IgE level (Fig. 2b) and Omalizumab-SK4 didn’t increase the total IgE level compared with IgE only group (Fig. 2d). Both NK-2-12-SK3 and NK-2-12-SK4 significantly accelerated the clearance of total IgE compared to Omalizumab-SK3 or Omalizumab-SK4 treatment and reduced both total IgE and free IgE levels to below the detection level (Fig. 2b–e). In conclusion, our findings demonstrate that IgE-targeting FcRTACs, particularly those engineered with pH-dependent binding properties, exhibit potent IgE clearance capacity.
a Schematic overview of the IgE elimination experiment. Humanized FcγRIIb mice (b–e; n = 5 mice per group) or FcγRIIb-/- mice (f, g; n = 4 mice per group) were injected with 5 mg/kg FcRTACs,followed by injection of 2.5 mg/kg IgE 30 min later. Blood samples were collected at the indicated time points (red arrows) to measure the serum levels of total IgE (b, d, f, g) and free IgE (c, e). Data are presented as means ± SEM. Statistical significance was determined using two-way ANOVA (*P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, the exact P value were provided in Supplementary Table 5). The curves for “Only IgE”, “Omalizumab”, and “NK-2-12” are shared between panels b and d, as well as c and e. Panel (a) was created in BioRender. Zhao, J. (2025) https://BioRender.com/53ug670.
To examine whether antigen elimination by the FcRTACs is dependent on the FcγRIIb receptor, we analyzed the antigen clearance activity of FcRTAC in FcγRIIb-/- mice. The IgE clearance mediated by different FcRTACs was largely attenuated in FcγRIIb-/- mice (Fig. 2f, g), confirming that FcγRIIb receptor mediated the antigen clearance capacity of FcRTAC. To assess the toxicity of FcRTACs, FcγRIIb humanized mice were intravenously injected with NK-2-12-SK3, NK-2-12-SK4, or Omalizumab at a high dose of 10 mg/kg. Blood samples were collected and different organs were dissected for analysis following drug administration. The levels of AST, ALT and LDH in the blood samples were not significantly elevated (Supplementary Fig. 5a–c) and H&E staining showed that FcRTAC did not lead to inflammation and injury in different organs including spleen and liver (Supplementary Fig. 5d).
We performed pharmacokinetic analysis of NK-2-12-SK3 in humanized FcRn mice. NK-2-12-SK3 demonstrated significantly shorter serum half-lives than Omalizumab (Supplementary Fig. 6a). The observed acceleration in serum clearance is probably mediated by the drug’s preferential redistribution from circulation to cellular surfaces expressing FcγRIIb. The half-life extending Fc variants like YTE (M252Y/S254T/T256E) are commonly employed to prolong serum persistence19. Surprisingly, the YTE-modified NK2-12-SK3 (YTE) showed only marginal improvement on serum half-life over NK-2-12-SK3 (Supplementary Fig. 6a). This phenomenon may be explained by the higher affinity of SK3 for FcγRIIb (KD ~ 10-9 M) compared to Fc-YTE binding to FcRn (KD ~ 0.1 uM at pH 6.0). As a result, NK-2-12-SK3 shows preferential retention on FcγRIIb rather than undergoing FcRn-mediated release into circulation. Tissue distribution analysis revealed that NK-2-12-SK3 preferentially accumulated in FcγRIIb-expressing organs, particularly the liver, within 2 h post-injection (Supplementary Fig. 6b, c).
To evaluate whether the FcRTAC can be used to deplete diverse pathological antigens beyond IgE, two distinct PCSK9-targeting antibody variants: 300 N (pH-insensitive) and L30H (pH-sensitive)20, along with their corresponding FcRTACs (300N-SK3 and L30H-SK3) were generated (Supplementary Fig. 7a and Supplementary Table 1). In PCSK9 humanized mouse models, neither 300 N nor L30H alone showed significant reduction in circulating PCSK9 levels, whereas the derived FcRTACs demonstrated PCSK9 clearance capacity. Among these, L30H-SK3 achieved the most pronounced reduction in plasma PCSK9 concentrations (Supplementary Fig. 7b).
Aβ-targeting FcRTAC promotes the microglia mediated clearance of pathological Aβ
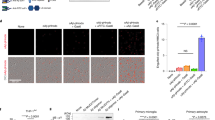
We next investigated whether FcRTAC platform could be applied to reduce the Aβ burden in a mouse model with amyloid pathology. We analyzed the single-cell sequencing data of the mouse brain (GEO GSE132042). The analysis revealed that microglia (Iba1) and macrophages (Lyz2) within the mouse brain exhibit high expression levels of FcγRIIb (Fig. 3a). Therefore Aβ-targeting FcRTAC Lecanemab-SK3 (Lec-SK3) were designed to enhance microglia mediated clearance of Aβ. The Fc portion of Lec-SK3 contained an N297A mutation, which abrogates Fc domain binding to Fc receptors (Fig. 3b, c). Lec-SK3 could efficiently promote the microglial cell uptake of fluorescence labeled Aβ (Fig. 3d).
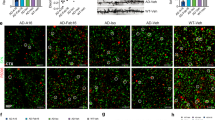
a Single-cell analysis reveals expression of FcγRIIb by various cell types in mouse brain. b Schematic diagram illustrating the structure of Lecanemab SK3 (Lec-SK3) and Lecanemab-N297A (Lec). c Schematic diagram illustrating how Lec-SK3 enhances FcγRIIb-mediated internalization of Aβ oligomers. d Representative confocal images showing Aβ oligomers (green) internalization in microglia treated with unlabeled Lec-SK3 (upper) or Cy5-labeled Lec-SK3 (lower). Cell nuclei were stained with DAPI (blue). Scale bar: 2 μm. Experiments were performed in triplicate and repeated three times with similar results. eExperimental timeline of 2.5-month-old male WT or 5xFAD mice with bilateral i.c.v. injection of saline, 2 mg/kg Lec-SK3 or Lec. Spontaneous alternation rate (f), exploration frequency and dwelling duration in the novel arm (g) of the Y-maze were assessed post-injection of saline, Lec-SK3 or Lec (n = 7 mice per group). h Discrimination index (%) and recognition index (%) of the NOR test were determined following the injection of saline, Lec-SK3, or Lec (n = 7 mice per group). i Representative confocal z-stack images (left) and 3D reconstructed images (right) of microglia (IBA1, green) and Aβ plaque (6E10, red) in CA1 regions of hippocampus from mice. Scale bar, 5 μm. Experiments were performed in triplicate and repeated three times with similar results. j Representative confocal images and quantifications of microglia (IBA1, green) and Aβ plaque (6E10, red) in CA1 regions of hippocampus from mice (n = 9 independent fields from 3 mice per group). Cell nuclei were labeled with DAPI (blue). Scale bar, 100 μm. k Western blotting and comparisons of MAP2 and PSD95 in hippocampus of 5xFAD mice with different treatments (n = 4 mice per group). Data are presented as means ± SEM. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons test (*P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001, the exact P value were provided in Supplementary Table 6). Panel (b) was created in BioRender. Zhao, J. (2025) https://BioRender.com/kr4x49j. Panel (c) was created in BioRender. Zhao, J. (2025) https://BioRender.com/6beshm0. Panel (k) was Created in BioRender. Zhao, J. (2025) https://BioRender.com/l2up9v3.
Lec-SK3 or Lec (N297A Fc-mutated Lecanemab) was administered to 5×FAD mice and their wild-type (WT) littermate controls on a C57BL/6 J background via i.c.v. injection. One-week post-treatment, behavior outcomes were assessed (Fig. 3e). Lec-SK3 and Lec treatment both significantly improved spontaneous alternation and Lec-SK3 induced a significantly greater improvement than Lec (Fig. 3f), demonstrating its superior efficacy in restoring hippocampal-dependent working memory21. Furthermore, Lec-SK3 significantly increased both novel-arm entry percentage and dwelling duration (Fig. 3g), confirming its benefits for hippocampal memory encoding and entorhinal-hippocampal attentional processing22. In the NOR test, Lec-SK3-treated mice showed significantly higher discrimination and recognition indices compared to Lec-treated mice (Fig. 3h), indicating improved non-spatial recognition memory and entorhinal cortical function23. Collectively, these results demonstrate that Lec-SK3 alleviates both spatial and non-spatial memory deficits more effectively than Lec.
To investigate the effects of Lec-SK3 on Aβ plaques in vivo, brains were collected one week after treatment. Both treatments significantly reduced Aβ1–42 levels in the mouse hippocampus, with Lec-SK3 exerting the most pronounced effect (Supplementary Fig. 8). Quantitative immunofluorescence analysis was subsequently performed to assess Aβ plaque burden and microglial density in the hippocampal CA1 subregion. The levels of Aβ plaques and microglial gliosis were significantly reduced in Lec-SK3-treated mice, indicating that Lec-SK3 decreases Aβ burden and alleviates microglial overactivation. Lec treatment also reduced Aβ plaque levels compared to saline-treated mice; however, microglial levels in Lec-treated mice were comparable to those in the saline-treated group (Fig. 3i, j). To evaluate the neuroprotective effect of Lec-SK3, we assessed the levels of microtubule-associated protein 2 (MAP2) and postsynaptic density protein 95 (PSD95) in the hippocampus. This choice was based on their well-established roles in synaptic dysfunction during prodromal Aβ pathology (Braak stages II-III), as well as the known causal relationship between Aβ oligomer-induced synaptic toxicity and the depletion of these key scaffolding proteins24,25. As shown in Fig. 3k, Lec-SK3 restored the expression levels of MAP2 and PSD95, thereby reversing Aβ-induced neuronal complexity loss in this AD model.
We further assessed the therapeutic efficacy of Lec-SK3 by bilateral i.c.v. co-administration with Aβ oligomers. For comparative evaluation, Lec and the mouse IgG2a isotype control (Lec-mIgG2a) were included in the experiment. Mirroring the results observed in the 5×FAD mouse model, Lec-SK3 administration demonstrated significant improvement in working memory performance. Notably, at equivalent doses, Lec-SK3 exhibited superior efficacy compared to both Lec and Lec-mIgG2a (Supplementary Fig. 9a, b). To validate the mechanistic dependence of Lec-SK3’s therapeutic efficacy on FcγRIIb engagement, we performed bilateral i.c.v. co-injections of Lec-SK3 and Aβ oligomers in WT, Fcer1g-KO (Fcer1g-chain deficient), and FcγRIIb-/- mice. While Lec-SK3 restored working memory in both WT and Fcer1g-KO mice, its therapeutic effect was completely abolished in FcγRIIb-/- mice (Supplementary Fig. 9c, d). These results unequivocally demonstrate that FcγRIIb is indispensable for Lec-SK3 mediated cognitive improvement.
Aβ targeting FcRTAC delivery via a AAV vector improves learning behavior and pathology in Aβ pathology mouse model
Brain accumulation of therapeutic antibodies is severely limited by the BBB. AAV variants with engineered capsids such as AAV-PHP. eB have been developed to efficiently penetrate the BBB and transduce neurons26, allowing for sustained and local production of transgene in the brain. We designed AAV-PHP. eB to express the Lecanemab N297A Fc variant (AAV-Lec) or the Lec-SK3 N297A Fc variant (AAV-Lec-SK3) (Fig. 4a).
a Schematic diagram of AAV virus sequence. b Binding to FcγRIIb (left) and antibody concentrations (right) in mouse brain homogenates were measured via ELISA at 20 days after i.c.v. injection of AAV (n = 4 mice per group). Data are presented as means ± SD. Statistical significance was determined by Unpaired t test. c Experimental timeline for 2-month-old male WT or 5×FAD mice receiving bilateral i.c.v. injections of saline, AAV-Lec-SK3, AAV-Lec, or AAV-GFP. Spontaneous alternation rate (d), exploration frequency (e), and dwelling duration (f) in the novel arm of the Y-maze were assessed at 20 and 60 days post-injection of saline, AAV-Lec-SK3, AAV-Lec, or AAV-GFP (n = 6 mice per group). g Schematic representation of NOR test. h Discrimination index (%) of the NOR test was determined following the injection of saline, AAV-Lec-SK3, AAV-Lec, or AAV-GFP (n = 6 mice per group). Data are presented as means ± SEM. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons test (*P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001, the exact P value were provided in Supplementary Table 7). Panel (a) was created in BioRender. Zhao, J. (2025) https://BioRender.com/zteancb. Panel (c) was created in BioRender. Zhao, J. (2025) https://BioRender.com/53ug670.
WT mice received i.c.v. injections of either AAV-Lec, AAV-Lec-SK3 or AAV-GFP. Faint GFP fluorescence was initially detected near the cerebral ventricles at 10 days post-injection of AAV-GFP, with significantly enhanced expression observed by 20 days post-injection (Supplementary Fig. 10). Surprisingly, Lec-SK3 expression was not detected in brain homogenates of WT mice 20 days after injection (Fig. 4b, c). We hypothesized that Lec-SK3 is endocytosed by binding to FcγRIIb receptors on microglia or macrophages in brain. To test this hypothesis, AAV-Lec-SK3 was administered to FcγRIIb-/- mice and the expression of the Lec-SK3 could be detected 20 days after i.c.v administration of AAV-Lec-SK3 (Fig. 4b), supporting the hypothesis that the expressed Lec-SK3 is cleared in an FcγRIIb-dependent manner.
To evaluate the therapeutic efficacy of AAV-Lec-SK3 in the 5×FAD mouse model, we performed bilateral i.c.v. injections of either AAV-Lec-SK3 or AAV-Lec. Behavioral assessments including Y-maze and NOR task were performed at 20- and 60-days post-injection (Fig. 4c). In the Y-maze behavioral assessment, mice treated with AAV-Lec-SK3 demonstrated significant cognitive enhancement, as evidenced by increased spontaneous alternation rates and novel arm exploration at both 20- and 60-day time points (Fig. 4d–f). In contrast, AAV-Lec-treated animals are less effective to improve either short-term working memory or long-term spatial memory performance throughout the evaluation period (Fig. 4d–f). The AAV-Lec-SK3-treated mice showed a significantly higher discrimination index in the NOR test, suggesting that AAV-Lec-SK3 treatment could improve non-spatial recognition memory as well (Fig. 4g, h).
To further evaluate the clinical feasibility of AAV-Lec-SK3, we also examined its therapeutic effects after i.v. administration for 60 days (Fig. 5a). Although AAV-Lec-treated animals exhibited a modest improvement trend in Y-maze performance compared to controls, this difference did not achieve statistical significance. In contrast, our results demonstrate the superior efficacy of AAV-Lec-SK3 in enhancing cognitive function, underscoring its therapeutic potential for AD (Fig. 5b, c).
a Experimental timeline for 2-month-old male WT or 5×FAD mice receiving bilateral i.v. injections of saline, AAV-Lec-SK3, AAV-Lec, or AAV-GFP. b,c, Spontaneous alternation rate (b) and discrimination index of the NOR test (c) were assessed 60 days post-injection of saline, AAV-Lec-SK3, AAV-Lec, or AAV-GFP (n = 6 mice per group). d Representative confocal images (left panel) and 3D-reconstructed images (right panel) of microglia (green) and Aβ plaques (red) in the CA1 region of the hippocampus at 60 days post-treatment. Scale bar: 5 μm. e Representative confocal images and quantification of microglia (green) and Aβ plaques (red) in the CA1 region of the hippocampus from mice 60 days post-treatment. Cell nuclei were stained with DAPI (blue). Scale bar: 50 μm. Quantification of Aβ and IBA1 levels using 9 independent fields from 3 mice per group. f Representative Western blot analysis of MAP2 and PSD95 in mice hippocampus 60 days post-treatment. g Quantification of MAP2 and PSD95 in panel (f) (n = 5 mice per group). Data are presented as means ± SEM. Statistical significance was determined by one-way ANOVA with Dunnett’s multiple comparisons test (*P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001, the exact P value were provided in Supplementary Table 8). Panel (a) was created in BioRender. Zhao, J. (2025) https://BioRender.com/53ug670, https://BioRender.com/s49qd4y. Panel (f) was Created in BioRender. Zhao, J. (2025) https://BioRender.com/l2up9v3.
To evaluate the effect of AAV-Lec-SK3 on the neuropathology of AD, we measured Aβ1-42 levels in the hippocampus of mice using ELISA. As expected, Aβ1-42 levels were significantly reduced in the AAV-Lec-SK3-treated group. In contrast, AAV-Lec treatment did not significantly reduce Aβ1-42 levels compared to the vehicle-treated group (Supplementary Fig. 11). Immunofluorescence staining revealed a reduction in both Aβ burden and microglial abundance in the AAV-Lec-SK3-treated group (Fig. 5d, e). While AAV-Lec treatment demonstrated a modest reduction in Aβ burden relative to controls, this effect was significantly less pronounced than that achieved with AAV-Lec-SK3 treatment. Furthermore, AAV-Lec administration did not produce a significant decrease in IBA1+ microglia levels (Fig. 5d, e). Next, we assessed the expression levels of MAP2 and PSD95 in the hippocampus across different treatment groups. Both MAP2 and PSD95 levels were significantly elevated following i.v. administration of AAV-Lec-SK3, whereas AAV-Lec treatment failed to induce a significant change in PSD95 levels compared with the control group (Fig. 5f, g). These findings support the therapeutic efficacy of Lec-SK3 in reversing the synaptic damage characteristic of AD pathology.
Mechanistic insight into the FcRTAC
Next we employed the IgE-targeting antibody as a model system to elucidate the molecular mechanisms underlying FcRTAC-mediated antigen clearance. We systematically evaluated the contribution of distinct endocytic pathways to receptor internalization using specific pharmacological inhibitors. Both NK-2-12-SK3 and NK-2-12-SK4 exhibited significant sensitivity to chlorpromazine, a clathrin-dependent endocytosis inhibitor, suggesting the predominant role of clathrin-mediated endocytosis in FcRTAC internalization (Fig. 6a, b). In contrast, nystatin-mediated inhibition of caveolae-dependent endocytosis showed no significant effect on internalization efficiency (Fig. 6a, b). NK-2-12-SK3-mediated internalization was independent of both macropinocytosis and phagocytosis, NK-2-12-SK4 showed partial dependence on these pathways, as evidenced by its sensitivity to cytochalasin D and amiloride treatment (Fig. 6a, b).
a, b Effect of inhibitors targeting different endocytic pathways on FcRTAC internalization. Huvec-2b cells were pre-treated with endocytosis inhibitors for 1 h, followed by the addition of pH-sensitive reagent-prelabeled NK-2-12-SK3 (a) or NK-2-12-SK4 (b). Fluorescence changes were monitored hourly over 12 h. Chlorpromazine: clathrin-mediated endocytosis inhibitor; cytochalasin D: macropinocytosis and phagocytosis inhibitor; amiloride (EIPA): macropinocytosis inhibitor; nystatin: caveolae pathway inhibitor. c–e, Live-cell imaging of FcRTAC-mediated IgE endocytosis. Cy3-labeled IgE (red) was incubated with Huvec-2b cells in the presence or absence of Cy5-labeled FcRTACs (white) for 2 h. Cells were stained with LysoTracker (green) and Hoechst (blue) for confocal imaging. Images were acquired using a 40× objective on a confocal microscope (c, scale bar: 20 μm) or a super-resolution microscope (d and e; scale bar: 10 μm). f Subcellular distribution of FcγRIIb and FcRTACs in Huvec-2b cells. Huvec-2b cells were incubated with Cy5-labeled FcRTACs (red) for 1 h. After fixation and permeabilization, cells were stained for Lamp-1 (green) and FcγRIIb (white) using primary antibodies and their corresponding fluorescent secondary antibodies. Nuclei were stained by DAPI (blue). Images were acquired using a 40× objective on a confocal microscope. Scale bar: 10 μm. c–f Experiments were performed in triplicate and repeated three times with similar results.
The subcellular localization of endocytosed IgE and different FcRTACs was detected with immunofluorescence microscopy. The Cy3 labeled IgE and Cy5 labeled FcRTACs were added to Huvec-2b cell. The results showed that intracellular IgE was only detected in cells treated with NK-2-12-SK3 or NK-2-12-SK4, but not in the untreated cells (Fig. 6c). We also observed that the endocytosed IgE colocalized with lysosome, indicating that the endocytosed antigen trafficked to lysosomes for degradation (Fig. 6d, e). Interestingly, the subcellular location of NK-2-12-SK3 and NK-2-12-SK4 exhibited different patterns (Fig. 6d, e). A significant fraction of NK-2-12-SK3 was localized to the cell plasma membrane, in addition to its cytosolic localization, suggesting that NK-2-12-SK3 may be more efficiently recycled back to the plasma membrane in relative to NK-2-12-SK4 (Fig. 6d). In contrast, the strong colocalization of NK-2-12-SK4 with the lysosome marker protein lysosomal-associated membrane protein 2 (LAMP2) staining indicated that NK-2-12-SK4 is more likely to enter the lysosome for degradation (Fig. 6e).
We subsequently examined the potential colocalization between different FcRTACs and FcγRIIb. The majority of FcγRIIb receptors were found on the plasma membrane, with a small fraction detected intracellularly. Notably, our observations revealed the absence of receptor localization within the lysosomal compartment, indicating that the FcγRIIb receptor undergoes recycling (Fig. 6f). Immunofluorescence analysis revealed distinct subcellular distribution patterns between the two constructs: NK-2-12-SK3 colocalized with FcγRIIb receptors in plasma membrane and endosomal structures, with minimal localization observed in lysosomes. In contrast, NK-2-12-SK4 exhibited significantly reduced cytoplasmic colocalization with FcγRIIb receptors but demonstrated enhanced accumulation within lysosomal compartments (Fig. 6f).
The FcRTAC incorporating an affinity-matured FcγRIIb binder, demonstrated more sustained antigen clearance efficiency
Through sequence alignment analysis, we identified a high degree of similarity between SK3 and SK4 (Fig. 7a). Competitive SPR assays revealed that the binding of one antibody to FcγRIIb prevented the binding of the other antibody to the receptor (Fig. 7b), indicating that SK3 and SK4 recognize overlapping epitopes on FcγRIIb. Consistent with the affinity measurements of SK3 and SK4 monoclonal antibodies (Fig. 1), NK-2-12-SK3 exhibited significantly higher binding affinity for FcγRIIb compared to NK-2-12-SK4 in the same SPR experiment (Table 2). To elucidate the structural basis for SK3’s higher FcγRIIb binding affinity relative to SK4, we determined their complex structures by X-ray crystallography. SK4 Fab was successfully co-crystallized with the extracellular region of FcγRIIb and the structure was further determined at 1.66 Å. Detailed crystallographic statistics were summarized in Supplementary Table 2. The structure reveals that the epitope recognized by the SK4 Fab is primary located in the second N-terminal domain of FcγRIIb (Fig. 7c). The total buried surface area of the FcγRIIb epitope is approximately 800 Ų.
a Alignment of SK3 and SK4 Sequences. HCDRs and LCDRs are indicated with red. b SPR-based competition assay for SK3 and SK4. NK-2-12-SK3 was immobilized on a protein A biosensor chip, followed by injection of hFcγRIIb over the flow cell. Subsequently, SK4 Fab and hFcγRIIb were injected. c Structure of the SK4 Fab in complex with FcγRIIb (PDB ID: 9M5B). The heavy and light chains of the SK4 Fab are shown in orange and light blue, respectively. The ribbon diagram of FcγRIIb is colored green, with its surface illustrated in white and the contact area with SK4 Fab highlighted in brown. d Comparison of the calculated SK3 scFv-FcγRIIb complex structure versus the SK4 Fab-FcγRIIb complex structure. e SPR-based affinity measurements of SK3 and SK3hi binding to hFcγRIIb at pH 7.4 and pH 6.0. SK3 or SK3hi was immobilized on a protein A biosensor chip, followed by injection of hFcγRIIb over the flow cell. Solid curves represent raw data, while dashed curves indicate fitted curves. f Live-cell imaging of NK-2-12-SK3hi-mediated IgE endocytosis. Cy3-labeled IgE (red) was incubated with Huvec-2b cells in the presence or absence of Cy5-labeled NK-2-12-SK3hi (white) for 2 h. Cells were stained with LysoTracker (green) and Hoechst (blue) for confocal imaging. Images were acquired using a super-resolution confocal microscope. Scale bar: 5 μm. Experiments were performed in triplicate and repeated three times with similar results. g, h The rechallenge experiment. FcγRIIb humanized mice (n = 4 mice per group) were i.p. injected with 2.5 mg/kg IgE following by injection of 2.5 mg/kg NK-2-12-SK3 or NK-2-12-SK3hi. One day later, mice were rechallenged with 2.5 mg/kg IgE without FcRTACs. Blood samples were collected to measure serum IgE levels. Data are presented as means ± SEM. Statistical comparisons were performed using Mixed-effects analysis (*P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, the exact P value were provided in Supplementary Table 9).
The heavy chains of SK4 contributed to the majority of interactions with FcγRIIb. Specifically, Tyr160 of FcγRIIb is a key residue recognized by SK4, engaging with 9 amino acids of SK4, including heavy chain residues Gly33, Gly50, Ile51, Ser52, Trp53, Arg57, Arg58, Glu99, and light chain residue Arg96. Notably, the main-chain of Tyr160 form two hydrogen bonds with the main-chain nitrogen atom of Trp53 and the side-chain carboxyl group of Glu99 in the heavy chain, respectively. Additionally, Thr161 of FcγRIIb forms two hydrogen bonds with the main chain atoms of SK4 heavy chain residues Gly33 and Glu99. Tyr163 of FcγRIIb also establishes a hydrogen bond with the backbone atom of SK4 heavy chain residues Tyr101. Furthermore, Asn157, Gly159, and Leu162 of FcγRIIb each form a hydrogen bond with Trp53, Arg57, and His31 of the SK4 heavy chain, respectively. Additionally, Lys116 and Lys120 of FcγRIIb form salt bridges with Asp102 and Asp56 of the SK4 heavy chain (Supplementary Table 3).
Unable to obtain the experimental structure of SK3 in complex with FcγRIIb, we built a model of the SK3-FcγRIIb complex using the SWISS-MODEL homology modeling pipeline with the structure of the SK4-FcγRIIb complex as the template due to the high sequence similarity and overlapping epitopes between SK3 and SK4. Comparison of the calculated SK3 scFv-FcγRIIb complex structure with the SK4 Fab-FcγRIIb complex structure showed substantial overlap between SK3 scFv and the variable region of SK4 (Fig. 7d). The model revealed that all three CDRs of the heavy chain contribute significantly to the interaction with FcγRIIb, while the light chain exhibits minimal involvement in receptor binding. The HCDR3 of SK3 is longer than that of SK4, resulting in a larger FcγRIIb interface relative to SK4 (Fig. 7d).
We hypothesized that FcγRIIb binders with increased affinity under acidic endosomal conditions were preferentially recycled to the plasma membrane, leading to multiple cycles of antigen clearance. Based on the structural information, we engineered SK3 variants with optimized FcγRIIb binding to further improve its therapeutic efficacy. A focused mutagenesis library was generated by introducing mutations into all three CDRs of the SK3 heavy chain. Following iterative biopanning against FcγRIIb under acidic conditions, a variant designated SK3hi, exhibited one order of magnitude higher affinity compared to the parental SK3 molecule under both physiological (pH 7.4) and endosomal (pH 6.0) conditions (Fig. 7e and Table 2, Supplementary Table 4). Confocal microscopy analysis demonstrated that NK2-12-SK3hi exhibited preferential plasma membrane localization with no detectable accumulation in lysosomal compartments (Fig. 7f). In a re-challenge experiment, mice receiving SK3hi-based FcRTAC maintained significantly better IgE clearance capacity than those treated with SK3-based FcRTAC post IgE re-injection on the second day, in the absence of additional FcRTAC administration (Fig. 7g, h).
Subsequently we built a phage display library by incorporating histidine substitutions at FcγRIIb-interacting residues in the SK3 to select for pH-dependent binders. We identified SK3ph, a variant containing three histidine substitutions, as the most strongly enriched clone after panning (Table 2). SPR analysis revealed that SK3ph binds FcγRIIb with a KD of 2.65 × 10-9 M at pH 7.4, which weakened to 9.37 × 10-9 M at pH 6.0 (Supplementary Fig. 12a, Supplementary Table 4). The antigen clearance of FcRTAC-SK3ph is comparable to NK-2-12-SK3 (Supplementary Fig. 12b). This pH-sensitive FcγRIIb dissociation translates to improved serum persistence of FcRTAC-SK3ph in vivo (Supplementary Fig. 12c). The data suggest that pH-dependent FcγRIIb dissociation facilitates FcRTAC recycling through the FcRn pathway, ultimately leading to its release back into circulation and the extended half-life in vivo (Supplementary Fig. 12d).
Benchmarking FcRTAC against sweeping technology and ASGPR-targeting degraders
To evaluate FcRTAC’s performance relative to the sweeping antibody technology27, we engineered NK-2-12-V12, a pH-dependent hIgG1 variant with selectively enhanced human FcγRIIb binding affinity (Fig. 8a)28. The cellular internalization assays revealed that NK-2-12-V12 exhibited significantly reduced uptake efficiency compared to NK-2-12-SK3 (Fig. 8b). To delineate the mechanisms underlying NK-2-12-V12 internalization, we systematically evaluated the contribution of distinct endocytic pathways using pathway-specific pharmacological inhibitors. Treatment with chlorpromazine (clathrin inhibition), cytochalasin D (actin polymerization blockade), and amiloride (macropinocytosis suppression) significantly impaired NK-2-12-V12 uptake (Fig. 8c), demonstrating concurrent involvement of clathrin-mediated endocytosis, phagocytosis, and macropinocytosis. In contrast, nystatin-mediated disruption of caveolae-dependent endocytosis had no significant effect on internalization efficiency. The subcellular localization of endocytosed IgE and NK-2-12-V12 was assessed using immunofluorescence microscopy after incubating Huvec-2b cells with labeled IgE and NK-2-12-V12. The majority of NK-2-12-V12 was localized to the cell membrane, with only a minor fraction detected in the cytosol. In contrast, a substantial proportion of NK-2-12-SK3 was internalized into the cytoplasm in addition to its presence on the cell surface (Fig. 8d). High-resolution microscopy demonstrated distinct IgE trafficking patterns: while IgE primarily localized to the cell surface with minimal lysosomal colocalization following NK-2-12-V12 treatment, NK-2-12-SK3 treatment induced significant lysosomal recruitment of IgE (Fig. 8e). While cell biology studies revealed distinct mechanisms of action between FcRTAC and sweeping antibodies, NK-2-12-V12 demonstrated comparable IgE depletion efficacy to FcRTAC at 5 mg/kg (Fig. 8f). Notably, the sweeping antibody mechanism conferred prolonged serum persistence (Fig. 8g).
a Schematic diagram of the structure of NK-2-12-v12. b Internalization assay of NK-2-12-V12. Antibodies were pre-mixed with a pH-sensitive dye-labeled anti-human Fc and incubated with Huvec-2b cells. Fluorescence changes were monitored hourly over 24 h. c, Internalization assay of NK-2-12-V12 in presence of endocytosis inhibitors. d, e Live-cell imaging of bispecific antibodies-mediated IgE endocytosis. Cy3-labeled IgE (red) was incubated with Huvec-2b cells in the presence of Cy5-labeled designated antibodies (white) for 1 h. Cells were stained with LysoTracker (green) and Hoechst (blue) for confocal imaging. Images were acquired using a 40× objective (d, scale bar: 40 μm) or a 60× objective confocal microscope (e left panel, scale bar: 20 μm) and a super-resolution microscope (e middle and right panel; scale bar: 10 μm). Experiments were performed in triplicate and repeated three times with similar results. f FcγRIIb humanized mice were injected with 5 mg/kg NK-2-12-SK3 or NK-2-12-V12, followed by administration of 2.5 mg/kg IgE after 30 min. Blood samples were collected to measure the serum IgE levels (n = 4 mice per group). Data are presented as means ± SEM. Statistical significance was determined using two-way ANOVA (*P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, the exact P value were provided in Supplementary Table 10). The curves for “Only IgE” is shared between Supplementary Fig. 4. g FcγRIIb humanized mice were injected with 5 mg/kg of NK-2-12-SK3 and NK-2-12-V12. Blood was collected from the tail vein of mice at indicated time points and serum antibody levels were quantitatively detected by ELISA (n = 5 mice per group). Panel (a) was created in BioRender. Zhao, J. (2025) https://BioRender.com/g5wqarw.
Various eTPD strategies exploit ASGPR-mediated clearance11,12,29, we also evaluated FcRTAC in comparison with ASGPR-targeting eTPD approaches. Phage display screening yielded multiple ASGPR-specific scFvs (Supplementary Fig. 13a), with AT5-Fc and AT10-Fc emerging as optimal candidates due to their high internalization efficiency in HepG2 (Supplementary Fig. 13b, c). These lead binders were subsequently engineered into bispecific antibodies featuring an IgE-targeting arm and an ASGPR-binding arm (Supplementary Fig. 13d). The internalization efficiency of both NK-2-12-AT5 and NK-2-12-AT10 in HepG2 cells is reduced compared to NK-2-12-SK3 in HUVEC-2b cells (Fig. 8d, Supplementary Fig. 13e, f). While NK-2-12-AT5 effectively reduced IgE levels in mice, its efficacy was modestly lower than that of FcRTAC NK-2-12-SK3 (Supplementary Fig. 13g). Collectively, these data establish that either receptor can serve as a platform for developing eTPDs capable of mediating robust antigen clearance from circulation.
Discussion
In this study, we have developed a class of bispecific antibody-based FcRTAC that simultaneously engages FcγRIIb for lysosomal trafficking and disease-specific targets for degradation. The FcRTAC platform demonstrates remarkable versatility, enabling the conversion of virtually any conventional neutralizing antibody into a potent antigen-degrading therapeutic, as exemplified by our successful implementation across multiple target classes, including IgE, PCSK9, and Aβ-specific antibodies. Specifically, FcRTACs successfully achieved complete IgE clearance, addressing the antigen accumulation problem of the conventional anti-IgE therapies. On a mouse model for AD-related amyloidosis, Aβ-targeted FcRTACs demonstrated significantly enhanced clearance efficiency compared to standard antibody approaches, with corresponding improvements in cognitive function. These findings establish FcRTAC as a transformative strategy for diseases requiring antigen depletion, particularly in scenarios where conventional antibodies hit efficacy ceilings due to high target burden.
The modularity of this bispecific antibody-based platform enables the comprehensive engineering of FcRTAC. For example, the antigen targeting arm can be engineered to be pH sensitive so it can bind with target antigen at physiological pH while dissociate from antigen at acidic pH. Consequently, the antigen dissociates from the FcRTAC complex within the endosomal compartment and trafficks to lysosomes for degradation. Both arms of FcRTAC can be obtained by experiment based methods such as hybridoma technology, phage or yeast display30 or the emerging artificial intelligence driven protein design as exemplified by the design of endocytosis-triggering binding proteins (EndoTags)31. Moreover, other lysosomal targeting receptors (LTRs) could be used to mediate antigen degradation by lysosomes12,32,33,34,35,36.
The selection of appropriate receptors for eTPD development requires careful consideration including the receptor’s endocytic efficiency, recycling kinetics, tissue distribution profile and physiological compatibility. For example, Banik, S. M. et al. established CI-M6PR as the foundational platform for LYTAC development10. Subsequently Anh et al. used CRISPR knockout screening to identify critical regulators of CI-M6PR targeting LYTAC and the retromer complex has been shown to limit LYTAC activity through CI-M6PR recycling. Consistent with these findings, we demonstrate that pH-sensitive antigen-binding domains represent a receptor-agnostic eTPD design strategy.
A critical pharmacokinetic limitation of FcRTAC therapeutics is their accelerated clearance via target-mediated drug disposition (TMDD), which necessitates frequent dosing regimens to maintain therapeutic efficacy. The underlying mechanism involves the formation of a tertiary complex between circulating FcRTAC, soluble antigen, and cell surface FcγRIIb, which undergoes FcγRIIb-mediated endocytosis, simultaneously removing both the therapeutic agent and its target from circulation. Following internalization, FcRTAC molecules face two distinct fates: (1) lysosomal trafficking and degradation following receptor dissociation, or (2) recycling to the plasma membrane in complex with FcγRIIb or FcRn for subsequent rounds of antigen clearance. Our findings demonstrate that antibodies with enhanced FcγRIIb binding affinity under acidic endosomal conditions (e.g. SK3) are preferentially maintained in complex with FcγRIIb. This stable interaction promotes receptor recycling to the plasma membrane, thereby augmenting antigen clearance efficiency. Therefore, enhancing FcRTAC’s FcγRIIb binding in endosomal compartments represents a promising strategy to extend its therapeutic efficacy. For instance, SK3hi—engineered for optimal FcγRIIb affinity under acidic endosomal conditions—demonstrates significantly prolonged activity persistence. However, the SK3hi-mediated enhancement of antigen-SK3hi complex recycling could potentially compromise antigen clearance efficiency when using pH-independent antigen-binding domains. To maximize target elimination, high-affinity FcγRIIb binders should be combined with pH-sensitive antigen-binding domains. Complementary approaches could include chemical biology strategies to develop covalent receptor-binding moieties, improving recycling efficiency37,38. Alternatively, modulating FcγRIIb binding affinity to favor FcRn-mediated recycling, as exemplified by the pH-sensitive variant SK3ph, exhibits extended serum half-life. Furthermore, the extended serum persistence observed with sweeping antibodies supports that modulating cellular internalization kinetics represents a viable strategy to prolong therapeutic serum half-life. The modularity of FcRTAC platform enable antibody engineering to optimize both pharmacokinetic profiles and antigen clearance efficiency for specific clinical indications, ranging from acute conditions requiring rapid target depletion to chronic diseases needing sustained therapeutic activity. Given FcRTAC’s bispecific linker function, increasing FcRTAC concentration did not proportionally enhance functional immune complex formation. Supraoptimal dosing not only reduced therapeutic efficacy but also risked antagonistic effects-a manifestation of the classic high-dose hook effect, underscoring the importance of dose optimization to achieve maximal therapeutic benefit.
Monoclonal antibodies generally demonstrate limited BBB penetration owing to their substantial molecular size. This challenge can be addressed through simultaneous targeting of transferrin receptor (TfR), CD98 heavy chain or insulin receptor to mediate transcytosis39,40,41,42,43. The ‘brainshuttle’ antibody trontinemab, a bispecific molecule with monovalent transferrin receptor 1 binding and bivalent Aβ targeting, shows rapid amyloid plaque clearance in AD patients42. Antibodies can be engineered to incorporate TfR-binding moieties, potentially enabling efficient CNS delivery for neurological disorders. For example, Michelle E. Pizzo et al. developed an anti-Aβ antibody engineered with a TfR-based domain for brain-targeted antibody delivery and bearing asymmetric Fc effector-reducing mutations, which mitigates ARIA-like lesions in a mouse model of amyloid pathology44. Brain-penetrant AAV offer an alternative strategy for delivering therapeutic proteins across the BBB, enabling sustained intracranial production of anti-Aβ FcRTACs. We observed that i.v. administration of AAV-Lec demonstrated significantly enhanced therapeutic efficacy compared to i.c.v. delivery. We attribute this differential efficacy to distinct biodistribution patterns: i.v. administration enabled whole brain delivery and widespread transgene expression45,46,47,48, whereas i.c.v. administration resulted in more restricted distribution and localized expression. This study establishes the first proof-of-concept for AAV-mediated delivery of an Aβ degrader as a therapeutic strategy for AD, representing a significant advancement in the field of targeted protein degradation for neurodegenerative disorders.
Furthermore, FcRTAC’s constitutive engagement of FcγRIIb in peripheral tissues could potentially restrict its therapeutic utility for CNS disorders. To address this limitation, we propose implementing a proteolytically activated antibody design49. FcRTAC can be engineered with a masking peptide covalently attached to its FcγRIIb-binding domain through a CNS-specific protease-cleavable linker. This innovative design would maintain FcRTAC in an inactive state during systemic circulation while allowing selective activation within the CNS microenvironment through protease-mediated unmasking.
The Lec-SK3 FcγRTAC system demonstrates high-efficiency Aβ clearance without inducing sustained microglial activation, as indicated by the absence of reactive gliosis. This finding is consistent with the TAM receptor (Tyro3, Axl, and Mer)-dependent microglial phagocytosis reported by Jung et al., where Aβ clearance occurs independently of gliosis induction50. Although the precise mechanisms underlying reduced gliosis of Lec-SK3 remain to be elucidated, our findings further support Jung et al.’s findings, demonstrating that microglial phagocytic activity can be functionally uncoupled from classical activation states. Importantly, this reveals a distinct mechanism from the conventional Aβ depletion immunotherapy, which typically depend on FcγR activation to drive Aβ phagocytosis51.
It has been reported that Aβ oligomers bind to neuronal receptors (e.g., PrPC/mGluR5), triggering calpain-1-mediated MAP2 proteolysis and Mdm2-dependent PSD95 ubiquitination. This process leads to the reduction of these proteins and subsequent synaptic loss52. The well-established causal relationship between Aβ deposition and the depletion of MAP2 and PSD95 provides mechanistic evidence that the therapeutic efficacy of Lec-SK3 stems from its ability to effectively clear pathogenic Aβ species, thereby reversing the synaptic damage characteristic of AD brain pathology.
In summary, we have developed bispecific antibody-based FcRTAC as a targeted protein degradation platform that directs extracellular soluble antigens and pathological protein aggregates to lysosomes for destruction, addressing unmet needs in two major therapeutic areas: (1) diseases characterized by high levels of pathogenic circulating proteins and (2) amyloid disorders involving misfolded protein aggregation. Through systematic mechanistic studies, we have elucidated the molecular principles governing FcRTAC activity and established a mechanistic framework for developing antigen-depleting therapeutics. This innovative approach represents a versatile therapeutic platform for treating diverse diseases ranging from autoimmune disorders to neurodegenerative conditions, and offers a powerful research tool for extracellular protein knockout in basic biological studies.
Methods
Mice
C57BL/6 J mice were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. Humanized FcγRIIb mice (NM-HU-2000010), FcγRIIb-/- mice (NM-KO-00130), Fcer1g-KO mice (NM-KO-190187), humanized FcRn mice (NM-HU-190070) and humanized PCSK9 mice (NM-HU-00075) were purchased from Shanghai Model Organisms Center, Inc. Humanized FcγRIIb mice were generated by CRISPR/Cas9-mediated homologous recombination to exclusively replace the mouse Fcgr2b extracellular domain (exons 3-4) with human Fcgr2b sequences while retaining native transmembrane and intracellular regions. 5×FAD mice (C001541) were purchased from Cyagen. All mice were bred and maintained in our pathogen-free facilities. During the feeding process, these mice were allowed to freely feed and drink. The temperature of the feeding environment was 22–24 °C, the humidity was 40–60%, the lighting and dark time were 12 h each, and the lighting time was 8:00–20:00. At the end of the experiments, animals were euthanized by cervical dislocation under deep anesthesia. Briefly, animals were first placed in the Rodent Anesthesia System (PerkinElmer, RAS-4) and deeply anesthetized with 5% isoflurane (repu) until the loss of consciousness and pedal reflexes. Cervical dislocation was then swiftly performed to ensure immediate death. All animal experiments were conducted in accordance with the protocols approved by the Institutional Animal Care and Use Committee of ShanghaiTech University (20241210001 for IgE elimination mouse model) and Nankai University (2024-SYDWLL-000076 for the IgE or PCSK9 elimination model and 2024-SYDWLL-000412 for the AD models).
Phage display screening of human FcγRIIb binders
A naive human single chain variable fragment (scFv) library was used for panning against human FcγRIIb as previously described16. This naive antibody library was constructed from peripheral blood mononuclear cells (PBMCs) isolated from 30 healthy donors. Variable heavy (VH) and light (VL) chain genes were amplified via PCR using degenerate primers targeting conserved framework regions. The VH and VL domains were connected by a flexible (Gly4Ser)3 linker using overlap extension PCR. The assembled scFv fragments were subsequently cloned into a phagemid display vector pCGMT3 using restriction enzyme cloning. The resulting library achieved a diversity of ~1010 individual clones, as determined by colony counting. To validate the library quality, 100 randomly selected clones were picked for Sanger sequencing analysis.
The phage library was incubated with biotinylated human FcγRIIb ectodomain (ACROBiosystems, CDB-H82E0) for 2 h at room temperature, followed by the capture of phage-antigen complexes using Dynabeads M280 (Invitrogen, 11206D). Bound phages were then eluted with Glycine-HCl (pH 2.2) for 10 min at room temperature and neutralized with Tris-HCl (pH 8) to a final pH of 7.5. The eluted phages were amplified for the next round of bio-panning. After a total of three rounds of panning, FcγRIIb-binding phages were enriched, and individual phage clones were amplified and subjected to phage ELISA. The phagemid of the positive clones in ELISA was isolated with plasmid miniprep kit (TIANGEN, DP118-02) for Sanger DNA sequencing.
For the affinity maturation of SK3, the SK3 mutagenesis phage display libraries were incubated with biotinylated human FcγRIIb ectodomain (ACROBiosystems, CDB-H82E0) in a pH 6 buffer for 2 h at room temperature. Subsequently, Dynabeads M280 (Invitrogen, 11206D) were used to capture the phage-antigen complexes. Then, unbound phages were washed away with 0.05% PBST at pH 6, the beads were resuspended in a pH 7 buffer and incubated at room temperature for 1 h to elute antibody variants with low affinity at pH 7. The eluted phages were amplified for the next round of bio-panning. After a total of four rounds of panning, individual phage clones were then amplified, and the entire process of phage ELISA was performed in pH 6 buffer. Plasmid miniprep kits (TIANGEN, DP118-02) were used to isolate phagemid from positive clones for sequencing.
To isolate SK3 variants with pH-dependent FcγRIIb binding, the histidine scanning scFv library of SK3 was generated, in which each CDR residue of heavy chain can either be retained as its original residue or mutated to a histidine residue. Three rounds of phage display selection were performed against biotinylated FcγRIIb (100 nM, pH 7.4) with magnetic bead capture, stringent washing (PBST, pH 7.4), and acidic elution (PBS, pH 6.0). Enriched clones were sequenced and analyzed for pH-sensitive binding profiles.
Protein expression, purification and analysis
ScFv-Fc (SKX-Fc) proteins were generated by fusing FcγRIIb-binding scFvs to the N-terminus of the Fc fragment of human IgG1 via a glycine-serine (G4S)3 linker. FcRTACs were created by fusing an FcγRIIb-binding scFv to the C-terminus of the heavy chain of a full-length IgG (including Omalizumab, NK-2-12, 300 N, L30H and lecanemab). The N297A mutation was introduced into the Fc region of all the FcRTACs. The SKX-Fcs and FcRTAC constructs were cloned into the mammalian expression vector pTT5. The plasmids were transfected into HEK293F cells to express FcRTACs over a 5-day period in Freestyle medium (Thermo, cat 12338018). The cells were centrifuged at 200 × g for 10 min at 4 °C, and the supernatants were collected and filtered through a 0.45 μm filter (Cobetter, OBT-1051). Protein purification was carried out using a HiTrap Protein A column (GE, 17040301) on an ÄKTA purifier chromatography system. The antibodies were further purified by gel filtration chromatography using a Superose 6 Increase 10/300 column (Cytiva, 29091596) with PBS buffer. The proteins were concentrated using Ultra Centrifugal Filters (Merck/Millipore, UFC903024). Antibody NK-2-12-SK3 was analyzed by LC-MS using an ACQUITY UPLC Peptide BEH C4 column (300 Å,1.7 μm,2.1 × 100 mm) with mobile phases A (0.1% FA/H2O) and B (0.1% FA/ACN), separated at a flow rate of 0.2 mL/min, and detected on a ZenoTOF 7600 mass spectrometer (Sciex). Data were deconvoluted to obtain intact molecular weights. The thermal stability of antibodies was analyzed using nanoDSF technology on a Prometheus NT.48 (NanoTemper). Samples (2 mg/mL) were added into 1.5 mL EP tubes (30–40 μL per tube), automatically aspirated by a single capillary, and subjected to thermal denaturation from 20 °C to 95 °C at 1.0 °C/min.
The knob-in-hole (KIH) strategy was used to construct heteroFcRTACs containing one copy of FcγRIIb-binding scFv and two copies of IgE binder. The Fc of one antibody heavy chain contained the Y407T mutation and had an FcγRIIb-binding scFv fused to its C-terminus followed by a His tag. The Fc of the other antibody heavy chain contained the T366Y mutation and had a flag tag fused to its C-terminus. The heterobispecific antibody was expressed in HEK293F as described above and purified by Flag/His tandem affinity purification using anti-Flag G1 afffinity resin (GenScript, L00432) and high affinity Ni-Charged resin (GenScript, L00666).
Purification of FcγRIIb for structure determination: The extracellular domain of FcγRIIb and the SK4 Fab were constructed with a 6×His tag at the C-terminal end (at the heavy chain of the SK4 Fab). For FcγRIIb extracellular domain expression, the mannosidase I inhibitor, kifunensine (MedChemExpress, HY-19332), was supplemented to the HEK293F cell culture to a final concentration of 5 µM. The 6×His-tagged proteins were purified using a nickel column (HisTrap HP, GE Healthcare, 17524801). The SK4 Fab was further purified by gel filtration chromatography using a Superdex 75 Increase 10/300 column (Cytiva, 29148721). The affinity-purified FcγRIIb was treated with 10 μL Endo Hf (NEB, P0703S) per mg of FcγRIIb for deglycosylation. Subsequently, it was further purified using a Superdex 200 Increase 10/300 column (Cytiva, 28990944).
Flow cytometry
Huvec or Huvec-2b cells were incubated with 2 μg/mL anti-FcγRIIb SKX-Fcs protein at 4 °C for 30 min. The cells were washed with PBS supplemented with 2% FBS and then stained with AF488 goat anti-human IgG (H + L) (Invitrogen, A-11013) at 1:500 dilution for 30 min. After two washes, fluorescence was detected by flow cytometry, and the data were analyzed with Flowjo X.
SPR assays
For dissociation constant determination, all assays were performed using a Protein A biosensor chip (Cytiva, 29127556) in a Biacore T200 instrument (Cytiva). The antibodies were captured onto the chip by injecting the antibody solution (80 µg/mL) at a rate of 10 µL/min until a signal of approximately 150 RU (response units) was reached. Subsequently, the analyte (recombinant human FcγRIIb), was injected onto the chip at a rate of 30 µL/min for 120 s and then allowed to dissociate for 240 s. Before the next cycle, the chip was regenerated using Glycine-HCl (pH 1.5) at a flow rate of 30 µL/min for 30 s. All binding reactions were fitted to a 1:1 binding kinetic model using the Biacore T200 evaluation software to calculate the affinity and kinetic parameters.
To determine the ability of a FcRTACs to simultaneously bind to two different antigens with each of its arms, a SPR-based assay was performed on a BIAcore T200 instrument. The assay used a sequential capture method in which the FcRTACs was immobilized on a Protein A biosensor chip by injection at a flow rate of 30 µL/min for 180 s, resulting in a surface density of approximately 20 RU. Subsequently, human FcγRIIb protein in PBS-T buffer (0.05% Tween 20) was injected onto the chip at a flow rate of 30 µL/min for 90 s to determine if it binds to the immobilized FcRTACs. Finally, IgE protein was injected onto the chip at a flow rate of 30 µL/min to assess whether the FcRTACs can also bind to IgE. All binding signals were analyzed using the Biacore T200 evaluation software.
The competition assay was conducted for FcRTACs NK-2-12-SK3 and NK-2-12-SK4 using a Protein A biosensor chip in a Biacore T200 instrument (Cytiva). The first FcRTACs was diluted in PBS-T buffer (0.05% Tween 20) and injected at a flow rate of 10 µL/min for 60 s. Human FcγRIIb was injected onto the chip at a flow rate of 30 µL/min for a duration of 90 s. Subsequently, the second FcRTACs was introduced at the same flow rate in the buffer containing the human FcγRIIb. All binding signals were analyzed using the Biacore T200 evaluation software.
Endocytosis tracking assay
The SKX-Fcs proteins, FcRTACs, or control antibodies were labeled for 15 min with the Incucyte® FabFluor-pH Antibody Labeling Dye (Sartorius, 4722) at a molar ratio of 1:3. The dye is an anti-Fc Fab fragment conjugated with a pH-sensitive fluorophore, which is non-fluorescent at pH 7.4 and becomes increasingly brighter as the pH drops when the labeled antibodies travel through endosome/lysosomal compartments (pH 4.5–5.5). Huvec-2b cells (10,000 cells/well) in 96-well plates were incubated overnight with each of the pre-labeled proteins while being protected from light. The plates were then imaged in the Live Cell Analysis System (Sartorius, IncuCyte Zoom HD/2CLR) at 60 min intervals for 24 h. The percentage of lysosomal routing was calculated as the ratio of the Fluorescent Area (fluorescent-positive cells) to the Phase Area (total cell confluency) using the Live Cell Analysis System (Sartorius, IncuCyte Zoom HD/2CLR). The analysis was performed using integrated software (GraphPad Prism 10). To investigate the cellular internalization capacity of NK-2-12-V12, antibodies were pre-mixed with a pH-sensitive dye-labeled anti-human Fc and incubated with Huvec-2b cells. The plates were imaged using the Live Cell Analysis System (Sartorius, IncuCyte S3 HD/2CLR), which exhibits higher sensitivity for detecting low levels of internalization compared to the IncuCyte Zoom HD/2CLR.
To investigate the mechanism of entry into cells for SKX-Fcs, Huvec-2b cells were pre-incubated with one of the endocytosis inhibitors for one hour prior to the addition of the pre-labeled proteins. The inhibitors included Chlorpromazine (Sigma, 31679), Cytochalasin D (MCE, HY-N6682), Amiloride (MCE, HY-101840), and Nystatin (Solarbio, N8040).
Immunofluorescence microscopy and live-cell imaging
For live-cell imaging experiments, IgE and FcRTACs were labeled with Cy3-NHS (Lumiprobe, 11320) and Cy5-NHS (Lumiprobe, 13320), respectively. The labeled IgE and FcRTACs were added to Huvec-2b cells (1 × 104 cells/well) in 96-well plates and incubated for 5 h at 37 °C. The lysosomal dye LysoTracker (Solarbio, L7400, 1:500 dilution) and the nuclear dye Hoechst (Yeasen, 40731ES10, 1:1000) were added at the end of the incubation period and incubated for another 1 h. Images were acquired using a spinning disk confocal microscope (Olympus, SpinSR10), with a 40×objective (UPlanXAPO, 40× /0.95 air objective), using 405 nm, 488 nm, 561 nm, and 640 nm lasers for Hoechst, LysoTracker, Cy3, and Cy5 signals, respectively.
For ultrahigh-resolution immunofluorescence microscopy experiments, images were acquired using a spinning disk confocal super-resolution microscope (Olympus, SpinSR10+OSR) with a 60× objective (UPlanApo, 60×/1.5 oil objective). The fluorescence intensity of the region of interest (ROI) was corrected by the unbleached control region and then normalized to the pre-bleached intensity of the ROI. The corrected and normalized data were fitted to a single exponential model using OSR (the OlyVIA software).
For the colocalization analysis of FcγRIIb with FcRTACs, Huvec-2b cells were incubated with Cy5-NHS (Lumiprobe, 13320) labeled antibodies for 1 h. After incubation, the cells were fixed with 4% histiocyte fixative solution (Solarbio, P1110) for 10 min, followed by three PBS washes. Subsequently, the cells were permeabilized with PBS containing 0.2% Triton X-100 for 5 min, followed by another three PBS washes. The anti-FcγRIIb antibody (Thermo, MA550351, 1:500 dilution) and anti-Lamp-1 antibody (Santa, sc-20011, 1:500 dilution) were applied to the cells for 1 h, followed by three PBS washes. Secondary antibodies CP555 anti-rabbit (proteintech, RGAR003, 1:1000 dilution) and AF488 anti-mouse (Invitrogen, A11017, 1:2000 diluted) were then added and incubated for another hour, followed by three PBS washes. Following the final PBS wash, the cells were mounted with mounting medium containing DAPI (Abcam, ab104139, 1:1000 dilution) for imaging under an LSM900 inverted microscope. Image analysis was performed using the ZEN2 (blue version) v.2.0.0 software.
In vivo IgE clearance
8-weeks female humanized FcγRIIb mice and FcγRIIb-/- mice were injected with 5 mg/kg of FcRTACs, followed by an injection of 2.5 mg/kg IgE 30 min later. Blood samples were collected at 1, 2, 3, 8, and 22 h after IgE injection. No other human IgG was co-administered in the experiment. Serum levels of total IgE and free IgE were measured via ELISA.
In vivo imaging
For the in vivo fluorescence imaging study, NK-2-12-SK3 was conjugated to Cy5.5-NHS (Lumiprobe, 17020) following the manufacturer’s protocol. Thirty minutes before administering 2.5 mg/kg of IgE via the tail vein, mice received an intravenous injection of 5 mg/kg of Cy5.5-labeled NK-2-12-SK3. Imaging was carried out two hours after IgE administration. Mice were anesthetized with Isoflurane, placed on a black background plate, and imaged using the IVIS Lumina II system. Following euthanasia by cervical dislocation, internal organs were excised for further imaging. Image acquisition and analysis were performed using Living Imaging software version 4.4.
ELISA
The method for measuring serum total and free IgE levels was described previously17,53. In brief, to evaluate serum total IgE levels,96-well ELISA plates were coated with anti-human IgE monoclonal antibody (SouthernBiotech, 9240-01, HP6061, 2 μg/mL). Anti-human IgE antibody (SouthernBiotech, 9250-05, HP6029-HRP, 1: 2000 dilution) was used as detection antibody to probe the captured IgE. For measuring free IgE levels, FcεRI (SinoBiological, 13193-H08H, 1 μg/mL) was coated onto 96-well ELISA plates and anti-human IgE antibody (HP6061, 2 μg/mL) and HRP-conjugated goat anti-mouse IgM (Abcam, Ab97230, 1:2000 dilution) were sequentially applied to probe the captured free IgE. 3,3’,5,5’-Tetramethylbenzidine (TMB, Solarbio, PR1200) was used as the substrate for color development, and the absorbance at 450 nm was measured with a plate reader (TECAN, 16039400). The concentration of total or free serum IgE was calculated from the standard curve generated using the purified IgE protein.
To determine the serum half-life of the FcRTACs, the concentration of the FcRTACs was measured by ELISA. AffiniPure F(ab’)2 fragment goat anti-Human IgG (Jackson ImmunoResearch, 109-006-098, 1 μg/mL) was used for coating the plates to capture IgG, and peroxidase-labeled AffiniPure goat anti-human IgG (H + L) (Jackson ImmunoResearch, 109-005-003, 1:10000 dilution) was used to detect the captured IgG.
All procedures were performed according to the manufacturer’s instructions. Following sacrifice and dissection, hippocampus of mice was isolated and rinsed on ice with PBS supplemented with a fresh protease inhibitor cocktail, then homogenized. The homogenate was transferred to a centrifuge tube and centrifuged at 5,000 × g for 10 min at 4 °C. The levels of Aβ1-42 in supernatant was quantified using an ELISA kit (Elabscience, Cat. No.: E-EL-M3010).
Crystallization, data collection and structure determination
The protein complex consisting of the extracellular domain of FcγRIIb and the SK4 Fab was prepared by mixing them at a molar ratio of 1.2:1 and incubated at room temperature for 2 h. The complex was purified using a Superdex 200 Increase 10/300 column with a buffer consisting of 50 mM HEPES, 150 mM KCl, and pH 7.5. The protein sample was concentrated to 10 mg/mL. Crystals were obtained using the vapor diffusion method by mixing the protein sample with a crystallization buffer containing 0.2 M NaCl, 0.1 M BIS-TRIS at pH 6.5, and 25% w/v polyethylene glycol (PEG) 3350 at a 1:1 (v/v) ratio. Prior to flash-freezing in liquid nitrogen, the crystals were cryoprotected by soaking them in the crystallization buffer supplemented with 20% (v/v) glycerol. X-ray data were collected on the BL19U1 beamline at the Shanghai Synchrotron Radiation Facility (SSRF), China. Data integration and scaling were performed using XDS. The structure was determined by molecular replacement using PHASER. Models were iteratively adjusted manually with Coot and refined with Phenix REFINE. Phasing and refinement statistics are summarized in Supplementary Table 2.
AAV injections
The Lec and Lec-SK3 heavy and light chains were connected via T2A peptides and cloned into AAV backbone plasmids. The transgene expression was driven by the EF1α promoter. All AAV-PHP. eB viruses were manufactured by Guangzhou Paizhen Biotechnology Co. The virus was produced by triple plasmid transfection of HEK293 cells. After several days of transfection, the supernatant was collected and concentrated using the PEG method. Subsequently, the sample underwent ultracentrifugation, ultrafiltration, and chromatographic purification, followed by sterilization through 0.22 µm filtration. The final genome titer of the AAV-PHP. eB vector was 1 × 1013 GC/mL. For i.c.v. injection of AAV, For i.v. injection of AAV, 2-month-old male 5×FAD mice and C57BL/6 J (WT) mice were used. 5×FAD mice were divided into 4 groups (n = 6) and injected with 3.5 × 1010 genome copies (GC) of AAV-GFP, AAV-Lec, AAV-Lec-SK3 or saline into bilateral lateral ventricles. 5×FAD mice were randomly assigned to four groups (n = 6). Each group received a retrobulbar injection of 3.5 × 1010 GC of AAV-GFP, AAV-Lec, AAV-Lec-SK3 or saline. WT mice injected with saline as a control. AAV vectors were delivered intravenously via retro-orbital sinus injection to adult mice. All injections were performed under isoflurane inhalation anesthesia (induction = 2% isoflurane 1 L/min O2, maintenance = 1% isoflurane 1 L/min O2).
Behavioral tests
Spontaneous alternation test in Y-maze. The Y-maze is a three-arm horizontal maze (40 cm long and 10 cm wide with 12 cm- high walls) in which the arms are symmetrically disposed at 120° angles from each other. The task was carried out on day 1 of behavioral tests. Mice were placed at the end of one arm and allowed to move freely through the maze during an 8 min session. The number of total arm choices and sequence of arm choices were recorded. The percentage of spontaneous alternation is calculated by dividing the number of successful alternations by the total number of arm entries minus two. Before each trial, the interior of the maze was sprayed with a 75% ethanol solution to erase any scent cues.
Spatial novelty recognition test in Y-maze. Two experiments were included, with an interval of 2 h. The first experiment was the acquisition period, one arm was closed, and the animals were allowed to explore freely in the other two arms for 3 min. The second experiment (recall phase) was performed 2 h later. All arms were opened, and the animals were free to move in three arms for 3 min. The time and distance of exploration in each arm were recorded. The time and distance of exploration in the new arm of the memory impaired were shortened. The final parameters to be given include the number of arm entries in each arm, the time and distance explored in each arm.
Mice were acclimated to the experimental room and apparatus for three consecutive days prior to the NOR test. The NOR test was conducted in a custom-made, square acrylic arena (30 × 30 x 28 cm3, width × depth × height), following established protocols50. The task consisted of three sequential phases: habituation, training (sample phase), and testing. During the habituation phase, mice were allowed to freely explore the empty arena for 10 min. 24 h post-habituation, the training phase commenced; mice were placed into the arena containing two identical sample objects placed on the two ends 10 cm away from a side wall for 10 min. Following a 24 h interval, the testing phase was performed: mice were reintroduced to the arena where one familiar object had been replaced with a novel object for a final 10 min session. The arena and objects were thoroughly cleaned with 75% ethanol between trials. All behavioral assessments were performed by researchers blinded to group assignment. The discrimination index (DI) was calculated using the formula: DI = (Time exploring novel objects - Time exploring familiar objects) / (Time exploring novel objects + Time exploring familiar objects) × 100%. The recognition index (RI) was calculated using the formula: RI = Time exploring novel objects / (Time exploring novel objects + Time exploring familiar objects) × 100%.
Stereotaxic co-injection of Aβ oligomers and antibodies into mouse lateral ventricles
Stereotaxic surgery was conducted under anesthesia. A 1.25 μL volume of soluble Aβ oligomers (AβO, 250 μM) was bilaterally injected into the lateral ventricles of WT mice, while control mice received an equivalent volume of saline.
To evaluate the effects of Lec-SK3, Lec-mIgG2a (MedChemExpress, Cat. No.: HY-P990110), or Lec-N297A, a 1.25 μL volume of each antibody (50 μg) was co-administered with 1.25 μL of soluble AβO (250 μM) into the lateral ventricles of WT mice. Parallel control animals received saline injections instead.
To determine whether Lec-SK3’s therapeutic efficacy depends on FcγRIIb engagement, Lec-SK3 (50 μg in 1.25 μL) and 1.25 μL of soluble AβO (250 μM) were co-injected into the lateral ventricles of WT, FcγRIIb -/-, or Fcer1g-KO mice.
All experiments were performed using 2.5-month-old male mice.
Immunohistochemistry
The immunohistochemistry of mice brain was performed as previously described54. Mice were anaesthetized with avertin (20 μL/g) by i.p. injection and perfused with saline followed by 4% PFA. Brains were post-fixed in 4% PFA at 4 °C overnight and gradiently transferred to 20%, 30% and 40% sucrose in saline for 24 h. Brains were embedded in OCT compound (Sakura, #4583) and sectioned into 20 μm coronal slices on cryo-stat microtomes (Leica). Sections were permeabilized by blocking buffer (4% BSA, 0.3% Triton X-100 in 1 × TBS) for 1 h at room temperature followed by incubation with appropriate primary antibodies for 24 h at 4 °C. The primary antibodies used were as follows: rabbit anti-Iba1 (Wako, 019-19741, 1:200 dilution), rabbit anti-PSD95 (Abcam, ab18258, 1:200 dilution), mouse anti-Aβ (Biolegend, 803001, 1:200 dilution). The sections were washed with TBST (0.1% Tween 20 in 1 × TBS) and stained with Fluorescein (FITC) AffiniPure™ Goat Anti-Mouse IgG (H + L) (Jackson, 115-095-003, 1:200 dilution) or Rhodamine (TRITC) AffiniPure™ Goat Anti-Rabbit IgG (H + L) (Jackson, 111-025-003, 1:200 dilution) in TBST for 1 h at room temperature. The sections were washed and mounted on slide glasses. The sections were washed with TBST (0.1% Tween 20 in 1 × TBS), and Vectashield with DAPI (Vector Lab, SP-8500-15) was used as mounting medium. Samples were stored at −20 °C before imaging on a confocal laser-scanning microscope. All confocal images from the brain sections were acquired using Leica TCS SP8 (×40 oil immersion optical lens) or Zeiss LSM800 (×20 lens) and quantified with Zeiss LSM800. For the internalization of Aβ in microglia, images were also reconstructed by Imaris (Bitplane) followed the manufacturer instructions. For quantification of abundance of Aβ and microglia, the fluorescent intensity of Aβ and microglia was measured by the Measure function in ImageJ.
Immunoblotting
Total protein lysate from hippocampus of mice was extracted with RIPA lysis buffer (recipe) (Epizyme, PC101), sonicated on ice, and centrifuged at 13400 g at 4 °C. Anti-MAP2 (Abcam, ab32454, 1:1000 dilution), Anti-PSD95 (Abcam, ab18258, 1:1000 dilution), or Anti-β-Actin (Easybio, BE0021, 1:5000 dilution) and Goat Anti-Mouse IgG (H&L)-HRP Conjugated (Easybio, BE0102, 1:5000 dilution) or Goat Anti-Rabbit IgG (H&L)-HRP Conjugated (Easybio, BE0101, 1:5000 dilution) were used as primary antibody and secondary antibody of WB (Figs. 3k, 5f). Data quantification was performed by ImageJ software (version 1.53).
Assessment of toxicity
8-weeks female humanized FcγRIIb mice received a single i.v. injection of BsAbs at a dosage of 20 mg/kg. The mice were euthanized 24 h post treatment.
Blood routine test: About 0.15 mL blood were collected via posterior orbital venous plexus with EDTA-K2 anticoagulant tube. Blood samples were tested using a fully automated blood cell analyzer (Mindray, BC-6100Plus).
Blood biochemical test: Blood samples were collected via posterior orbital venous plexus using EDTA-K2 anticoagulant tube 24 h after administration. The blood samples were centrifuged at 2000 rpm for 8 min and diluted 7.5 times (about 1.5 mL) at room temperature and sent to Shanghai Jinyu Medical Laboratory Center for testing.
Histopathological examination:Organs including heart, liver, spleen,kidney and lung were harvested, fixed with 10% formalin overnight. The samples were immersed in paraformaldehyde for 24–72 h to be dehydrated in Leica ASP300S instrument. Then the required position of the samples was adjusted, and the samples were embedded with HistoCore Arcadia H to make paraffin blocks. HistoCore BIOCUT microtome was used to cut into 4 μm sections for Hematoxylin and eosin (H&E) staining. (hematoxylin: Zhuhai Beso Biotechnology Co.LTD, BA4041, eosin: Zhuhai Beso Biotechnology Co.LTD, BA4099), and pathological image scanners were used to observe the tissue morphology and structure.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All experimental data are provided in the manuscript. The structures determined in this study have been deposited in the Protein Data Bank (PDB) under accession codes: 9M5B (SK4 Fab in complex with FcγRIIb). Materials used in this study will be made available under an appropriate Materials Transfer Agreement. Source data are provided with this paper.
References
Lerner, R. A. Manufacturing immunity to disease in a test tube: the magic bullet realized. Angew. Chem. Int Ed. Engl. 45, 8106–8125 (2006).
Carter, P. J. & Lazar, G. A. Next generation antibody drugs: pursuit of the ‘high-hanging fruit. Nat. Rev. Drug Discov. 17, 197–223 (2018).
Saini, S. S. et al. Efficacy and safety of omalizumab in patients with chronic idiopathic/spontaneous urticaria who remain symptomatic on H1 antihistamines: a randomized, placebo-controlled study. J. Invest Dermatol 135, 67–75 (2015).
Fertan, E. et al. Lecanemab preferentially binds to smaller aggregates present at early Alzheimer’s disease. Alzheimers Dement 21, e70086 (2025).
Lord, A. et al. An amyloid-beta protofibril-selective antibody prevents amyloid formation in a mouse model of Alzheimer’s disease. Neurobiol. Dis. 36, 425–434 (2009).
Wells, J. A. & Kumru, K. Extracellular targeted protein degradation: an emerging modality for drug discovery. Nat. Rev. Drug Discov. 23, 126–140 (2024).
Wang, W., Wang, E. Q. & Balthasar, J. P. Monoclonal antibody pharmacokinetics and pharmacodynamics. Clin. Pharm. Ther. 84, 548–558 (2008).
Igawa, T., Haraya, K. & Hattori, K. Sweeping antibody as a novel therapeutic antibody modality capable of eliminating soluble antigens from circulation. Immunol. Rev. 270, 132–151 (2016).
Igawa, T. et al. Antibody recycling by engineered pH-dependent antigen binding improves the duration of antigen neutralization. Nat. Biotechnol. 28, 1203–1207 (2010).
Banik, S. M. et al. Lysosome-targeting chimaeras for degradation of extracellular proteins. Nature 584, 291–297 (2020).
Ahn, G. et al. LYTACs that engage the asialoglycoprotein receptor for targeted protein degradation. Nat. Chem. Biol. 17, 937–946 (2021).
Caianiello, D. F. et al. Bifunctional small molecules that mediate the degradation of extracellular proteins. Nat. Chem. Biol. 17, 947–953 (2021).
Cotton, A. D., Nguyen, D. P., Gramespacher, J. A., Seiple, I. B. & Wells, J. A. Development of Antibody-Based PROTACs for the Degradation of the Cell-Surface Immune Checkpoint Protein PD-L1. J. Am. Chem. Soc. 143, 593–598 (2021).
Marei, H. et al. Antibody targeting of E3 ubiquitin ligases for receptor degradation. Nature 610, 182–189 (2022).
Siepe, D. H., Picton, L. K. & Garcia, K. C. Receptor Elimination by E3 Ubiquitin Ligase Recruitment (REULR): A Targeted Protein Degradation Toolbox. ACS Synth. Biol. 12, 1081–1093 (2023).
Wang, Y. et al. High-throughput functional screening for next-generation cancer immunotherapy using droplet-based microfluidics. Sci. Adv. 7, eabe3839 (2021).
Li, N. et al. Reduction of circulating IgE and allergens by a pH-sensitive antibody with enhanced FcγRIIb binding. Mol. Ther. 32, 3729–3742 (2024).
Ridgway, J. B., Presta, L. G. & Carter, P. Knobs-into-holes’ engineering of antibody CH3 domains for heavy chain heterodimerization. Protein Eng. 9, 617–621 (1996).
Dall’Acqua, W. F., Kiener, P. A. & Wu, H. Properties of human IgG1s engineered for enhanced binding to the neonatal Fc receptor (FcRn). J. Biol. Chem. 281, 23514–23524 (2006).
G. D. Yangkopoulos, N. J. P., A. J. Murphy & N. Star. Anti-PCSK9 antibodies with pH-dependent binding characteristics. US patent CN104540852A (2018).
Kim, H. Y. et al. EPPS rescues hippocampus-dependent cognitive deficits in APP/PS1 mice by disaggregation of amyloid-β oligomers and plaques. Nat. Commun. 6, 8997 (2015).
Wu, C. et al. Disrupted presynaptic nectin1-based neuronal adhesion in the entorhinal-hippocampal circuit contributes to early-life stress-induced memory deficits. Transl. Psychiatry 12, 141 (2022).
Monteiro, S. et al. Absence of IFNγ promotes hippocampal plasticity and enhances cognitive performance. Transl. Psychiatry 6, e707 (2016).
Koleske, A. J. Molecular mechanisms of dendrite stability. Nat. Rev. Neurosci. 14, 536–550 (2013).
Kallergi, E. et al. Dendritic autophagy degrades postsynaptic proteins and is required for long-term synaptic depression in mice. Nat. Commun. 13, 680 (2022).
Goertsen, D. et al. AAV capsid variants with brain-wide transgene expression and decreased liver targeting after intravenous delivery in mouse and marmoset. Nat. Neurosci. 25, 106–115 (2022).
Iwayanagi, Y. et al. Inhibitory FcγRIIb-Mediated Soluble Antigen Clearance from Plasma by a pH-Dependent Antigen-Binding Antibody and Its Enhancement by Fc Engineering. J. Immunol. 195, 3198–3205 (2015).
Mimoto, F. et al. Engineered antibody Fc variant with selectively enhanced FcγRIIb binding over both FcγRIIa(R131) and FcγRIIa(H131). Protein Eng. Des. Sel. 26, 589–598 (2013).
Wu, Y. et al. Aptamer-LYTACs for Targeted Degradation of Extracellular and Membrane Proteins. Angew. Chem. Int. Ed. Engl. 62, e202218106 (2023).
Chao, G. et al. Isolating and engineering human antibodies using yeast surface display. Nat. Protoc. 1, 755–768 (2006).
Huang, B. et al. Designed endocytosis-inducing proteins degrade targets and amplify signals. Nature 638, 796–804 (2025).
Liu, Y. et al. Targeted protein degradation via cellular trafficking of nanoparticles. Nat. Nanotechnol. 20, 296–302 (2025).
Zhang, B. et al. Insulin-like Growth Factor 2 (IGF2)-Fused Lysosomal Targeting Chimeras for Degradation of Extracellular and Membrane Proteins. J. Am. Chem. Soc. 145, 24272–24283 (2023).
Pance, K. et al. Modular cytokine receptor-targeting chimeras for targeted degradation of cell surface and extracellular proteins. Nat. Biotechnol. 41, 273–281 (2023).
Zhou, Y., Teng, P., Montgomery, N. T., Li, X. & Tang, W. Development of Triantennary N-Acetylgalactosamine Conjugates as Degraders for Extracellular Proteins. ACS Cent. Sci. 7, 499–506 (2021).
Zhang, D. et al. Transferrin receptor targeting chimeras for membrane protein degradation. Nature 638, 787–795 (2025).
Xiao, Y. et al. A covalent peptide-based lysosome-targeting protein degradation platform for cancer immunotherapy. Nat. Commun. 16, 1388 (2025).
Zhang, H. et al. Covalently Engineered Nanobody Chimeras for Targeted Membrane Protein Degradation. J. Am. Chem. Soc. 143, 16377–16382 (2021).
Khoury, N. et al. Fc-engineered large molecules targeting blood-brain barrier transferrin receptor and CD98hc have distinct central nervous system and peripheral biodistribution. Nat. Commun. 16, 1822 (2025).
Banks, W. A., Rhea, E. M., Reed, M. J. & Erickson, M. A. The penetration of therapeutics across the blood-brain barrier: Classic case studies and clinical implications. Cell Rep. Med 5, 101760 (2024).
Abbott, A. Brain drugs can now cross the once impenetrable blood-brain barrier. Nature 641, 1086–1088 (2025).
Grimm, H. P. et al. Delivery of the Brainshuttle™ amyloid-beta antibody fusion trontinemab to non-human primate brain and projected efficacious dose regimens in humans. MAbs 15, 2261509 (2023).
Wells, R. C. et al. Dual targeting of transferrin receptor and CD98hc enhances brain exposure of large molecules. Cell Rep. 44, 116038 (2025).
Pizzo, M. E. et al. Transferrin receptor-targeted anti-amyloid antibody enhances brain delivery and mitigates ARIA. Science 389, eads3204 (2025).
Chan, K. Y. et al. Engineered AAVs for efficient noninvasive gene delivery to the central and peripheral nervous systems. Nat. Neurosci. 20, 1172–1179 (2017).
Hu, S., Yang, T. & Wang, Y. Widespread labeling and genomic editing of the fetal central nervous system by in utero CRISPR AAV9-PHP.eB administration. Development 148, dev195586 (2021).
Luoni, M. et al. Whole brain delivery of an instability-prone Mecp2 transgene improves behavioral and molecular pathological defects in mouse models of Rett syndrome. Elife 9, e52629 (2020).
Konno, A. & Hirai, H. Efficient whole brain transduction by systemic infusion of minimally purified AAV-PHP.eB. J. Neurosci. Methods 346, 108914 (2020).
Polu, K. R. & Lowman, H. B. Probody therapeutics for targeting antibodies to diseased tissue. Expert Opin. Biol. Ther. 14, 1049–1053 (2014).
Jung, H. et al. Anti-inflammatory clearance of amyloid-β by a chimeric Gas6 fusion protein. Nat. Med 28, 1802–1812 (2022).
Wilcock, D. M. et al. Microglial activation facilitates Abeta plaque removal following intracranial anti-Abeta antibody administration. Neurobiol. Dis. 15, 11–20 (2004).
Amin, L. & Harris, D. A. Aβ receptors specifically recognize molecular features displayed by fibril ends and neurotoxic oligomers. Nat. Commun. 12, 3451 (2021).
Hamilton, R. G., Marcotte, G. V. & Saini, S. S. Immunological methods for quantifying free and total serum IgE levels in allergy patients receiving Omalizumab (Xolair) therapy. J. Immunol. Methods 303, 81–91 (2005).
Wang, H. et al. Self-Propelled Nanomotors with an Alloyed Engine for Emergency Rescue of Traumatic Brain Injury. Adv. Mater. 34, e2206779 (2022).
Acknowledgements
This work was funded by grants from by the National Natural Science Foundation of China (grant no. 82261138553 and 82373898 to H.Z., grant no. 82241058 to X.X.), Natural Science Foundation of Tianjin (grant no. 25JCJQJC00260 to X.X.), the Scientific Instrument and Technology R&D Project of Nankai. University (grant no. 24NKSYJS04) and ShanghaiTech University Shanghai Institute for Advanced Immunochemical Studies (SIAIS) research funding. We gratefully acknowledge the technical support provided by the following core facilities: the Discovery Technology Platform at SIAIS, ShanghaiTech University for IncuCyte operation; the Biomedical Big Data Platform at SIAIS, ShanghaiTech University for data analysis services; the State Key Laboratory of Medicinal Chemical Biology for SPR instrumentation; and the Flow Cytometry Platform at College of Life Science, Nankai University for flow cytometry analysis. We would also like to thank the staff from the BL19U1 beamline (https://cstr.cn/31129.02.NFPS.BL19U1) of National Facility for Protein Science in Shanghai (NFPS) at Shanghai Synchrotron Radiation Facility (SSRF) for assistance during data collection. We sincerely thank Jiliang Zhao, a member of Hongkai Zhang’s laboratory, for preparing the figure schematics using BioRender.
Author information
Authors and Affiliations
Contributions
H.Z., X.X. and H.Y. conceived the study. H.Z., M.D., X.X. and H.Y. supervised the project. H.Z., M.D., X.X., H.Y., X.L., N.L. and T.L. wrote the paper. H.Z., M.D., T.L. and N.L. designed all the experiments. N.L., T.L., Z. Zhao, H.L., N.G., B.D., Wenli Wang, Y.J., W.M., X. Zhang, Y.W., Z. Zhang, Y.B., X.W., C.W., J.L., X.G., F.Z., Wei Wang, L.L., X. Zhu., Y.L., Y.D., G.H., P.W., S.G., S.W., A.C., Y.H. performed the experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Du, M., Li, N., Li, T. et al. Chimeras co-targeting antigens and FcγRIIb trigger degradation of extracellular soluble proteins and pathological aggregates. Nat Commun 17, 514 (2026). https://doi.org/10.1038/s41467-025-67207-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67207-4