Abstract
Osteomyelitis, especially multidrug-resistant cases, remains a formidable clinical challenge due to recurrent infections and antibiotic limitations. Here, we present an injectable GaCuVan&HACHO-BSA hydrogel that self-assembles in situ within the bone marrow cavity—the epicenter of trained immunity—to simultaneously eradicate pathogens, induce innate immune memory, and regenerate bone. The hydrogel captured bacteria, virulence factors, and inflammatory mediators through multivalent interactions with enhanced injectability, biocompatibility, and sustained antigen release, making it ideal for minimally invasive treatment of osteomyelitis-related bone defects. Mechanistically, adsorbed pathogen signatures activated pattern recognition receptors, triggering metabolic reprogramming (elevated succinate/ATP/lactate), HIF-1α stabilization, amplified glycolysis and inflammation (COX2/iNOS/CD86), and pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) production—collectively inducing trained immunity with cross-protection against homologous/heterologous reinfection. Furthermore, glycyrrhizic acid promoted bone tissue repair and modulated immune responses. By converging antimicrobial defense, innate immune memory, and tissue repair into a single platform, this work redefines osteomyelitis management and advances immunomodulatory biomaterials for clinical translation.
Similar content being viewed by others
Introduction
Osteomyelitis, a chronic bone infection primarily caused by Staphylococcus aureus (S. aureus), is characterized by persistent infection, bone destruction, and abscess formation1. Traditional treatments, involving systemic antibiotics, often fail due to poor tissue penetration, bacterial biofilm formation, and increasing antibiotic resistance2,3,4. Even more problematic, problems with the healing of bone tissue and recurrent infections in patients with osteomyelitis continue to be major obstacles to clinical treatment, especially when the infection is caused by multidrug-resistant (MDR) bacteria, such as methicillin-resistant Staphylococcus aureus (MRSA)5,6,7,8. There is therefore an urgent need for new therapeutic strategies that can be effective against infection and bone repair simultaneously.
One promising approach to overcoming these challenges is the activation of trained immunity, a form of innate immune memory. Trained immunity is a form of innate immune memory where prior exposure to pathogens or immune modulators induces long-lasting functional reprogramming of immune cells like macrophages and neutrophils, enhancing their response to future infections9,10. Unlike adaptive immunity, it provides broad protection against various pathogens without requiring prior exposure to the same microorganism11,12,13. Although the concept of trained immunity has shown promising potential in treating bacterial diseases14, there have been no studies to date examining its use for osteomyelitis treatment.
Fortunately, hydrogels offer a promising direction for overcoming the limitations of traditional bacterial infection treatments and enhancing resistance to invading pathogens through the induction of trained immunity. Hydrogels can be engineered to provide a controlled, sustained release of immune-modulating agents at the site of infection, creating a localized environment that supports the activation of trained immunity15,16,17,18,19. Moreover, the use of a double-network hydrogel system enhances the material’s injectability, mechanical properties, and stability, making it ideal for minimally invasive treatment of complex bone defects16. This approach holds promise for activating trained immunity while overcoming the challenges associated with systemic treatments. Glycyrrhizic acid (Ga), a natural herbal small molecule extracted from liquorice, has a unique structure containing hydrophobic triterpenoid aglycones and hydrophilic Di-glucuronic acid units20. Studies have shown that the self-assembly process of Ga hydrogel is significantly accelerated in the presence of metal ions like Zn2+ and Cu2+21,22. In particular, Cu2+ plays an important role in bone tissue repair, which further promotes bone repair by regulating the osteogenic differentiation of bone marrow mesenchymal stem cells (BMSC) and promoting angiogenesis23. In addition, Ga can also regulate the Wnt/β-catenin signaling pathway, which is critical for promoting the osteogenic differentiation of BMSC, thereby effectively repairing bone defects24. It is worth noting that the dual-network structure of Ga-based hydrogels enhances mechanical strength, sustained release21,25,26, making them ideal candidates for both immune-stimulating carriers and immune-modulating therapeutic agents.
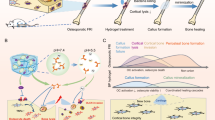
In this study, we developed an in situ self-assembled GaCuVan&HACHO-BSA hydrogel designed to specifically target the bone marrow cavity, where trained immunity is known to originate27, for the treatment and prevention of osteomyelitis (Fig. 1). Specifically, the hybrid hydrogel combines multiple physicochemical cross-linking networks, with the primary network formed by the complexation of vancomycin (Van)-grafted Ga and Cu2+ (GaCuVan) (Fig. 1A), and the secondary network (HACHO-BSA) formed by the cross-linking of the aldehyde groups of modified oxidized hyaluronic acid (HACHO) with multiple amino groups of bovine serum albumin (BSA) by Schiff base reaction (Fig. 1B). The introduction of hyaluronic acid (HA)-based networks is expected to confer macrophage targeting ability to hydrogel, as studies have shown that HA can bind to the CD44 receptor, which is highly expressed on the surface of macrophages during inflammation28,29. The dual-network structure improves the hydrogel’s mechanical properties and stability, allowing it to capture and adsorb various pathogen-associated molecules such as bacterial components, bacterial antigens, inflammatory cytokines, and serum proteins (Fig. 1C–F). These interactions, driven by hydrogen bonding, coordination, and Schiff base cross-linking, ensure continuous release of immunogenic components at the infection site. Mechanistically, these adsorbed pathogen signatures activate pattern recognition receptors (NOD-like receptor, TLR, and RIG-I-like receptor signaling pathways), driving metabolic reprogramming characterized by elevated succinate, ATP, and lactate levels. This metabolic shift stabilizes hypoxia-inducible factor (HIF-1α), amplifies glycolysis and inflammatory responses (via COX2, iNOS, and CD86 upregulation), and boosts production of pro-inflammatory cytokines (e.g., IL-1β, IL-6, TNF-α)—collectively inducing trained immunity (Fig. 1G). This mechanism not only enhances bacterial clearance (Fig. 1H) but also confers cross-protection against homologous and heterologous reinfections. Additionally, the hydrogel stimulates bone regeneration through the release of Ga and Cu²⁺, making it effective for treating osteomyelitis-related bone defects (Fig. 1I). More importantly, rats treated with the hydrogel exhibited strong immune protection, enabling them to resist secondary infections from both homologous and heterologous pathogens (Fig. 1J).
A Schematic illustration of GaCuVan self-assembly. B Schematic diagram illustrating the preparation of HACHO-BSA. C In vitro simulation of in situ gel formation of GaCuVan&HACHO-BSA hydrogel. D Mechanism of in situ gel formation. E In situ administration with GaCuVan&HACHO-BSA hydrogel. F (a) Recognition and capture of bacteria by the hydrogel via binding to specific terminal peptides on the bacterial cell wall. (b) Schematic illustrating the adsorption of bacterial antigens, serum albumin, and inflammatory factors by the hydrogel through Schiff base reactions, hydrogen bonding, and ionic coordination. G Mechanisms of induced trained immunity by the hydrogel. H Bacterial capture and bactericidal properties of the hydrogel. I Osteogenic effects of the hydrogel. J Induction of trained immunity and prevention of relapse by the hydrogel. Created in BioRender. Chen, H. (2025) https://BioRender.com/ompv5rx.
In summary, the GaCuVan&HACHO-BSA hydrogel offers a comprehensive solution for treating osteomyelitis by integrating infection control, bone repair, and trained immunity. Its unique dual-network structure, continuous antigen release, and ability to target immune cells at the infection site provide significant advantages over traditional treatments, making it a promising approach for managing osteomyelitis, particularly in patients with MDR infections.
Results
Design route, characterization, and mechanism of the GaCuVan&HACHO-BSA hydrogel
The two-step addition process for the construction of the double-network hydrogel was illustrated in the schematic diagram (Fig. 2A). In summary, the initial network was formed by the self-assembly of GaVan, which was induced by Cu2+. Subsequently, HACHO and BSA were mixed to form the second network, which was achieved through Schiff base formation, hydrogen bonding, and metal ion coordination. This process served to further enhance the stability of the hydrogel.
A Schematic illustration for the formation of GaCuVan&HACHO-BSA hydrogel, which forms the dual-network hydrogel (Created in BioRender. Chen, H. (2025) https://BioRender.com/40cd8b9). B Gelation time after addition of CuCl2 solution at different concentrations to GaVan solution at different W/W ratios. NG, GT > 2 and GT < 2 were assigned to samples with no self-assembly gelation, self-assembly gelation time greater than 2 min, and self-assembly gelation time within 2 min, respectively. C Gelation time after addition of BSA solution to HACHO solution at different W/W ratios. D Macroscopic observation of rapid gelation of GaCuVan mixed with HACHO-BSA with different dye colors. E Representative SEM images of GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels. n = 3 independent samples. Photographs of injectability (F), shape adaptability (G), and self-healing (H) of GaCuVan&HACHO-BSA hydrogel. I Frequency scan measurements in the rheological analysis of the different hydrogels. J Degradation profiles of different types of hydrogels in PBS at 37 °C. n = 4 independent samples. K In vivo degradation profiles of different hydrogels with time after subcutaneous implantation in Sprague-Dawley rats. n = 4 independent samples. L UV/Vis absorbance spectra of GaVan at concentrations ranging from 450 to 650 nm, measured by the vanillin-sulfuric acid method. Cumulative in vitro release behavior of Ga (M) and Van (N) from GaCuVan&HACHO-BSA hydrogel at 37 °C in PBS. n = 3 independent samples. O Comparison of the FT-IR spectra of GaVan, GaCuVan, HACHO, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels. P XPS spectrum of the prepared GaCuVan&HACHO-BSA hydrogel. Q Interaction energies between sub-docked molecules and the structure of different complexes. R Interaction analysis of three-dimensional docking results for multiple complexes. (a) Mode of interaction between Cu ions and GaVan molecules. Magnified image: Coordination formed between Cu ions and GaVan molecules. (b) Interaction mode between the HACHO molecule and the GaCuVan complex, the black frame represents that multiple hydrogen bonds between O and N atoms on the GaCuVan molecule and hydroxyl O atoms on the HACHO molecule. (c) Visualization and analysis of post-docking complex interactions between the GaCuVan&HACHO complex and BSA. The results showed that multiple sets of hydrogen bonds and coordination interactions were formed between several components to promote stable binding of GaCuVan&HACHO-BSA hydrogel. Data are presented as mean values ± SEM. A was generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
GaVan and HACHO were synthesized as described, and Fourier transform infrared spectroscopy (FT-IR) and 1H NMR spectra were used to verify the successful synthesis (Supplementary Fig. 1A, B) or the oxidation effectiveness (Supplementary Fig. 2). For structural stability of released Van, mass spectrometry analysis (Supplementary Fig. 1C) confirmed the intact molecular structure of Van in hydrogel release media, with the observed [M + H]+ peak at m/z 1450.43 matching the theoretical mass and ruling out hydrolysis or oxidation products. GaVan could self-assemble into hydrogels at a W/W of 2%, and its properties could be adjusted by adding particular metal ions21,22. Although several metal ions could promote the self-assembly process of GaVan according to the tube inversion test (Supplementary Fig. 3 and Supplementary Table 1), Cu2+ was chosen in subsequent studies to promote the hydrogelation of GaVan because of its important role as an essential trace metal in the human body, especially in the skeletal system23. Furtherly, different concentrations of Cu2+ solutions (0.1 mg/mL, 0.5 mg/mL, 1 mg/mL) and various mass ratios (0.5%, 1%, 2%, and 4%) of GaVan solutions were used to figure out the proper formula of GaCuVan hydrogel. Based on the gelation time (Fig. 2B and Supplementary Fig. 4), a mass ratio of 4% GaVan solution and 0.5 mg/mL Cu2+ solution, with gelation time of 1 min 36 s, was chosen to obtain the GaCuVan self-assembled hydrogel. The appropriate ratio of HACHO solution to BSA solution was also investigated in the gelation experiment (Fig. 2C and Supplementary Fig. 5). 10% W/W HACHO solution and 20% W/W BSA solution were used in subsequent studies, with a gelation time of 1 min 31 s.
In tissue engineering, biocompatibility is critical for in vivo bone defect repair. Studies have shown that low concentrations of copper ions (e.g., from 1 mmol/L copper nanoparticles) exhibited good biocompatibility with negligible cytotoxicity and no hemolysis30, while cytotoxicity in cell cultures emerged only when copper ion concentrations reached high levels (e.g., 152.7 ppm in Cu-MBG scaffolds after 7 days)31. However, high copper concentrations or burst release could reduce cell proliferation, creating an unfavorable microenvironment for cell adhesion and growth32.
While high ion concentrations could compromise cellular viability, we directly compared Cu²⁺, Mg²⁺, and Zn²⁺-containing hydrogels—synthesized using identical component concentrations to GaCuVan&HACHO-BSA—to clarify their safety and functional profiles. BMSC cultured with leachates from GaCuVan&HACHO-BSA or GaMgVan&HACHO-BSA (collected after 6 days of degradation) showed no significant cell death via live/dead staining (Supplementary Fig. 6), consistent with inductively coupled plasma optical emission spectrometry (ICP-OES) data. Cumulative Cu²⁺ release peaked at 75.80 ± 11.15 ppm—well below reported cytotoxic thresholds—and this concentration promoted osteogenic differentiation. For Mg²⁺, cumulative release reached 230.01 ± 15.11 ppm, which remained below its established cytotoxic threshold (320 ppm); however, this concentration failed to enhance BMSC osteogenesis, instead favoring chondrogenic differentiation33. In contrast, GaZnVan&HACHO-BSA leachates induced significant BMSC death within 48 h. ICP-OES confirmed cumulative Zn²⁺ release at 125.92 ± 12.08 ppm, exceeding the critical cytotoxic threshold (100 ppm), explaining this toxic effect34,35. These results further confirmed that Cu²⁺ was a suitable choice for subsequent experiments due to its favorable biocompatibility and ability to promote osteogenesis.
To understand the gelation of the double-network, methylene blue was added to the GaVan and Cu2+ mixed solution, while Nile red was used to mark the HACHO and BSA solution. As shown in Fig. 2D, the GaCuVan&HACHO-BSA cross-linked to form a hydrogel quickly within 1 min, possibly due to the formation of Schiff base bonds, hydrogen bonds, and coordination effects between different components. Scanning electron microscopy (SEM) images showed that all hydrogels had porous microstructures. However, the porous microstructure of the GaCuVan&HACHO-BSA hydrogel was more uniform and more interconnected, and the pore size decreased with the mixing of several components (Fig. 2E).
The injection test showed the injectability of GaCuVan&HACHO-BSA hydrogel without causing needle clogging (Fig. 2F), which was crucial for minimally invasive osteomyelitis treatment. After injection in containers with different shapes, the GaCuVan&HACHO-BSA hydrogel could completely fill the grooves (Fig. 2G). Upon removal from the groove, the GaCuVan&HACHO-BSA hydrogel formed a complete block of uniform thickness, indicating its shape adaptability and the ability to fill the complex, irregular environment in the bone marrow cavity. The cut-healing experiments showed that the two cut pieces of hydrogel could self-heal into a non-cracked bulk after 30 min (Fig. 2H), indicating the self-healing ability of the GaCuVan&HACHO-BSA hydrogel.
Rheological test (Fig. 2I) showed that the physico-chemical double cross-link network significantly improved the mechanical properties of the gel and gave it superior stability, since the GaCuVan&HACHO-BSA hydrogel had the highest and most stable storage modulus (G’) among all the groups of hydrogels. Hydrogels need to be biodegraded for their clinical therapeutic applications. To investigate in vitro degradation behavior, the GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels were co-incubated with phosphate-buffered saline (PBS) at 37 °C. The GaCuVan hydrogel was mostly degraded after 6 days, while GaCuVan&HACHO-BSA was degraded after 28 days, as shown in Fig. 2J. Time-course images of hydrogel degradation were added (Supplementary Fig. 7), which corroborated the degradation kinetics shown in Fig. 2J—confirming consistent structural breakdown over time: the GaCuVan&HACHO-BSA degradation was observed after 28 days. Additionally, lysozyme—a key enzyme abundant in infected microenvironments (e.g., 100–200 μg/mL in purulent exudates36,37)—was evaluated for its impact on hydrogel stability. As shown in Supplementary Fig. 8, GaCuVan&HACHO-BSA hydrogel incubated in PBS containing physiological lysozyme concentrations (100 μg/mL) at 37 °C exhibited no significant acceleration of degradation kinetics compared to lysozyme-free controls, demonstrating that the hydrogel maintained structural integrity in lysozyme-rich infected microenvironments to enable sustained drug release and immune modulation without premature breakdown. Subsequently, the GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels were implanted subcutaneously in the back of rats to investigate in vivo degradation behavior (Fig. 2K and Supplementary Fig. 9). The GaCuVan hydrogel degraded first after implantation into the body, while the GaCuVan&HACHO-BSA hydrogel was substantially degraded only by day 12 and had the longest lasting time in vivo. The degradation rules of several hydrogels were basically the same as in vitro, but the degradation rate was slightly faster than in vitro, which might be due to the abundance of biological enzymes in the body.
To investigate the concentration of Ga and Van released from the hydrogel, the absorbance of Ga in the concentration range of 10–300 μg/mL and the absorbance of Van in the concentration range of 10–100 μg/mL were determined by ultraviolet and visible spectrophotometry (UV/Vis) (Fig. 2L and Supplementary Fig. 10). As shown in Fig. 2M, N, the concentration of Ga released from the hydrogel, as measured by UV/Vis, gradually increased over time, and the release rate of Ga slowly increased, approaching 80% release on the 12th day. Furthermore, the release curve of Van showed a similar trend to that of Ga, further demonstrating that the hydrogel could effectively release Ga and Van over a long period for sustained therapeutic effects in vivo.
FT-IR analysis (Fig. 2O) and X-ray photoelectron spectroscopy (XPS) analysis (Fig. 2P and Supplementary Fig. 11) were performed to verify the successful construction of the hydrogel. Initially, the FT-IR results showed the characteristic absorption peaks of Ga and Van, including C=O stretching vibration and C–O bond vibration. However, after Cu2+ complexation, the absorption peaks of these functional groups were slightly shifted, indicating that complexation had occurred. Furthermore, the characteristic absorption peaks of HACHO (such as C–H stretching vibration, etc.) were also verified, and at the same time, the characteristic peak of C=O stretching vibration showed a slight red shift, which further illustrated the enhancement of hydrogen bonding within the co-assembled hydrogel38. Taken together, these results supported the successful synthesis of the GaCuVan&HACHO-BSA hydrogel. Next, the XPS results showed that the characteristic peaks of Ga, Van, Cu2+, HACHO, and BSA (C 1s, O 1s, N 1s, Cu 2p, Cl 2p) were identified in the GaCuVan&HACHO-BSA hydrogel. And the changes in N 1s and C 1s spectra indicated the occurrence of Schiff base reaction, while the changes in O 1s and N 1s peaks revealed the further strengthening of hydrogen bonding. The above results fully demonstrated the successful synthesis of GaCuVan&HACHO-BSA hydrogel.
Binding energy of the GaCuVan&HACHO-BSA hydrogel components and molecular mechanism of gelation
To investigate the binding energy between the GaCuVan&HACHO-BSA hydrogel components, molecular docking was performed on Cu ions, GaVan molecules, and HACHO molecules sequentially. As shown in Fig. 2Q, the binding energy predicted by molecular docking between GaVan and Cu2+ was 0.998 kcal/mol; then, the binding energy predicted by molecular docking between HACHO and GaCuVan was 3.816 kcal/mol. The molecular docking binding energy between the GaCuVan&HACHO complexes and BSA was 8.854 kcal/mol.
Then, to study the mechanism of the GaCuVan&HACHO-BSA hydrogel gelling, the complex was visualized and analyzed after molecular docking (Fig. 2R). Initial visualization analysis showed that Cu2+ was bound to a cavity of GaVan, forming coordination with four surrounding O atoms to promote its stable association. In addition, five groups of hydrogen bonds were formed between the hydroxyl O atoms of HACHO and the O atoms of GaCuVan, as shown by the visualization analysis of the HACHO and GaCuVan complexes. Moreover, the hydroxyl O of HACHO also formed a hydrogen bond group with the N atom of GaCuVan, which further promoted the stable binding of the ternary complex. After adding BSA, a large number of hydrogen bonds were formed between the GaCuVan&HACHO complex and BSA, thereby promoting its binding to the cavity above the protein. Among them, Cu2+ in the ternary complex formed two coordination groups with the Asp135 amino acid side chain on BSA, in addition to forming four coordination groups with GaVan. At the same time, since GaVan and HACHO contained a large number of hydrogen bond acceptors and donors, a total of 26 hydrogen bond groups were formed between the ternary complex molecule and the protein, further promoting stable binding of the hydrogel.
In vitro simulation of the GaCuVan&HACHO-BSA hydrogel formation by adsorption of pathogen-related molecules, with favorable biomechanical properties
Hydrogels with rough, fibrous structures are efficient adsorbents for bacteria39. GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels, characterized by their porous structures and rough surface meshes (Fig. 2E), enable them with good bacteria adsorption ability. Van can bind to specific terminal peptides on the cell wall, effectively facilitating bacterial identification and capture. HACHO, a polyaldehyde-rich crosslinker, could promote gelation via Schiff base reactions and hydrogen bonding with a variety of pathogen-related molecules (such as bacterial antigens, inflammatory factors, serum proteins, etc.) in the infectious microenvironment. Therefore, GaCuVan&HACHO-BSA hydrogel not only recognized and trapped surrounding bacteria, but also adsorbed bacterial antigens, inflammatory factors, and serum proteins through Schiff base reactions, hydrogen bonding, and metal coordination (Fig. 3A). Additionally, groups treated with GelMA, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA for bacterial assays or mechanical tests were designated as Groups Ⅰ to Ⅳ (Ⅰ, GelMA; Ⅱ, GaCuVan; Ⅲ, HACHO-BSA; Ⅳ, GaCuVan&HACHO-BSA). The GelMA,GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels exhibited porosities of 42.7 ± 3.3%, 57.1 ± 3.5%, 55.6 ± 2.9%, and 49.4 ± 3.7%, with average pore diameters of 3.9 ± 1.7, 27.8 ± 3.2, 28.6 ± 2.6, and 19.8 ± 2.4 μm, respectively (Supplementary Fig. 12).
A Diagram showing the formation and the mechanism of GaCuVan&HACHO-BSA hydrogel, and subsequent experimental groups (Ⅰ, GelMA; Ⅱ, GaVan; Ⅲ, HACHO-BSA; Ⅳ, GaVan&HACHO-BSA) (Created in BioRender. Chen, H. (2025) https://BioRender.com/m80d37q). B, C After co-incubation of MRSA/S. aureus suspensions at a concentration of 106 CFU/mL with GelMA, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels in LB broth for 30 min, the bacterial colonies in the hydrogels and supernatants were analyzed by CFU counts, and the relative capture rates of different hydrogels for MRSA and S. aureus were qualified (C). ****p < 0.0001, ns no significance. D GelMA, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels were incubated with MRSA, and S. aureus for 30 min, followed by SYTOTM 9/PI staining, and representative CLSM fluorescence images were captured showing live bacteria (stained green with SYTOTM 9) and dead bacteria (stained red with PI) trapped on the hydrogel surfaces. E Quantitative analysis of MRSA and S. aureus SYTOTM 9 fluorescence intensity on the different hydrogel surfaces. F, G Each hydrogel group was incubated with MRSA and S. aureus for 12 h, followed by SYTOTM 9/PI staining, and representative CLSM fluorescence images of live and dead bacteria on the hydrogel surface were captured, followed by quantitative analysis of PI-positive cell ratio on the surface. H The concentration of LTA adsorbed on the different hydrogels was measured by UV-Vis after co-incubation in DMEM medium for 30 min or 12 h. The concentration of LPS (I) and TNF-α (J) adsorbed on different hydrogels after the same treatment was measured by ELISA. K Compression recovery properties of GaCuVan&HACHO-BSA hydrogel. L Representative compressive stress-strain curves for GaCuVan&HACHO-BSA hydrogel with 10%, 20% and 30% BSA concentrations. M Compressive strengths corresponding to GaCuVan&HACHO-BSA hydrogel with different BSA concentrations. N Representative compressive stress-strain curves for GaCuVan&HACHO-BSA hydrogel with and without MRSA. O Compressive strengths corresponding to GaCuVan&HACHO-BSA hydrogel with and without MRSA. The data in (B, C, E, G–J, M, O) were expressed as the mean ± s.d. (n = 5 biologically independent samples per group), with the results shown as mean ± SEM. Statistical significance and P values were determined by two-way ANOVA followed by Dunnett’s multiple comparison test (B) or one-way ANOVA followed by Tukey’s multiple comparison test (C, E, G–J, M, O). A was generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
In vitro experiments simulating the in vivo infection environment demonstrated that after 30 min of co-incubation with MRSA and S. aureus, GaCuVan, HACHO-BSA and GaCuVan&HACHO-BSA hydrogels all captured the bacteria vigorously, whereas GelMA hydrogel was ineffective as shown by a large number of bacteria in the supernatant, while few bacteria were associated with the gel (Fig. 3B, C and Supplementary Fig. 13). Notably, the GaCuVan&HACHO-BSA hydrogel achieved the highest capture rates of both MRSA (72 ± 4%) and S. aureus (76 ± 5%), leaving the lowest residual bacterial concentrations in the culture medium. To further investigate the full potential of hydrogel-based bacterial adsorption, we extended the incubation time to 12 h, allowing the bacteria to expand and fully disperse within these hydrogels. Strikingly, while prolonged incubation increased bacterial capture by HACHO-BSA hydrogel, no live bacteria were found in GaCuVan and GaCuVan&HACHO-BSA hydrogels (Supplementary Fig. 14). The increased Schiff-base and hydrogen bond interactions between the hydrogel and bacteria during extended incubation may contribute to the increased bacterial capture. Meanwhile, the sustained Van release over time resulted in the elimination of live bacteria in the GaCuVan and GaCuVan&HACHO-BSA hydrogels. Fluorescence staining revealed that the GaCuVan&HACHO-BSA hydrogel absorbed the thickest layer of bacteria (Fig. 3D, E), which was likely eradicated by Van after 12 h of incubation, without compromising the hydrogel’s bacterial capture efficiency (Fig. 3F, G and Supplementary Fig. 15). Additionally, the minimal bacterial adsorption by GelMA hydrogel in Fig. 3D, F arises from two sequential factors. First, its low porosity (42.7 ± 3.3%) and small pore diameter (3.9 ± 1.7 μm) inherently limit physical entrapment: as reported, hydrogels with <50% porosity show lower bacterial capture than those with 70–80% porosity, while pore diameters <3.67 μm enhance bacterial penetration resistance. This size-exclusion effect restricts MRSA and S. aureus (1 μm diameter) from infiltrating the dense GelMA network. Second, during the post-incubation PBS wash and 1 min agitation, GelMA’s reliance on physical entrapment alone allows superficially adsorbed bacteria to detach readily. In contrast, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogel exhibit additional chemical adhesion via hydrogen bonding and dynamic covalent interactions, stabilizing bacterial capture even after washing. This dual mechanism—physical entrapment plus chemical adhesion—explains the higher bacterial retention in composite hydrogels compared to GelMA.
As for inflammatory and virulence factors, we evaluated the absorption capacity of the four groups of hydrogels by incubating them in culture medium containing lipoteichoic acid (LTA), LPS and TNF-α for 30 min or 12 h (Fig. 3H–J and Supplementary Fig. 16). Within 30 min, the HACHO-BSA and GaCuVan&HACHO-BSA hydrogels adsorbed (16.856 ± 2.554) μg/mL and (22.275 ± 3.282) μg/mL of LTA, (23.971 ± 4.646) ng/mL and (34.757 ± 10.286) ng/mL of LPS, and (33.117 ± 2.75) ng/mL and (43.367 ± 9.083) ng/mL of TNF-α within 30 min, respectively. Notably, the adsorbed levels continued to increase by 12 h of incubation, surpassing the minimum concentrations required to induce the inflammatory responses in mouse-derived macrophages (Raw264.7) in vitro21. Apparently, the GaCuVan&HACHO-BSA hydrogel showed significantly higher adsorption of LTA, LPS, and TNF-α than the HACHO-BSA hydrogel.
Osteomyelitis is often associated with bone non-union and bone defects. Therefore, the developed hydrogels must have the ability not only to fill the defective bone area but also to provide structural support, requiring robust mechanical properties. The GaCuVan&HACHO-BSA hydrogel did not show any obvious cracks during the compression process and was able to recover to its original shape after the removal of the external force (Fig. 3K). As shown in Fig. 3L, M, increasing BSA content from 10% to 30%, the maximum compressive strain of the GaCuVan&HACHO-BSA hydrogel also increased accordingly, highlighting the importance of BSA concentration to the overall mechanical strength of the hydrogel. In addition, the addition of MRSA and S. aureus led to increased compressive strength (Fig. 3N, O and Supplementary Fig. 17), indicating that effective bacterial capture could improve the hydrogel’s mechanical strength by increasing the cross-linking. In short, the GaCuVan&HACHO-BSA hydrogel effectively captures bacteria, absorbs inflammatory factors and serum proteins, and achieves these functions through a combination of physical and chemical interactions. Its physical capture is driven by the hydrogel network structure and hydrogen bonding, while the chemical adsorption is mediated by Schiff base reactions and coordinated interactions.
In vitro biocompatibility and antimicrobial effects of GaCuVan&HACHO-BSA hydrogel
The GaCuVan&HACHO-BSA hydrogel, composed primarily of small plant-derived molecules and natural compounds, demonstrates favorable biocompatibility. The cellular viability effects of GaVan, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels on BMSC and human umbilical vein endothelial cells (HUVECs) were assessed over 1, 3, and 5 days of culture using Live/Dead staining and Cell Counting Kit-8 (CCK-8) assays. Live/Dead staining (Fig. 4A, B, Supplementary Figs. 18 and 19) revealed no significant morphological changes or increased cell death, with most cells showing good viability and a gradual increase in live cell numbers and density. Similarly, CCK-8 results confirmed that the hydrogels did not significantly affect BMSC or HUVECs proliferation (Fig. 4C, D). Further blood compatibility was assessed through hemolysis assays, which demonstrated no noticeable hemolysis when red blood cells (RBCs) were incubated with the hydrogels, confirming good blood compatibility of the hydrogels (Fig. 4E).
Representative Live/dead fluorescence images of BMSC (A) and HUVECs (B) stained by calcein-AM/PI after incubating with GaVan, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels for 24 h. The effects of GaCuVan&HACHO-BSA hydrogel components on cell viability of BMSC (C) and HUVECs (D) were further evaluated by CCK-8 assay. E Hemolysis assay and analysis of GaCuVan&HACHO-BSA hydrogel components. Representative standard plate coating images (F) and corresponding CFU count (J, K) of MRSA and S. aureus after treatments (T1, Control; T2, GaVan; T3, GaCuVan; T4, HACHO-BSA; T5, GaCuVan&HACHO-BSA; T6, Van). G, L, M) CLSM images of Live/dead MRSA and S. aureus after treatment with GaCuVan&HACHO-BSA hydrogel components and Van, with live cells stained positive for SYTOTM 9 (green) and dead cells visualized by PI staining (red), and the PI-positive cells were analyzed (L, M). Representative SEM (H) and TEM (I) images of MRSA and S. aureus before and after treatment with GaCuVan&HACHO-BSA hydrogel components and Van, showing cytoplasmic leakage, cell wall and membrane damage, and severe bacterial destruction, indicated by red arrows. MRSA (N)and S. aureus (O) 24-h growth curves after the above treatments. The data in (C–E, J–M) were expressed as the mean ± s.d. (n = 6 independent samples in (C, D, N, O), n = 5 independent samples in (E, J–M), with the results shown as mean ± SEM. Two-way ANOVA followed by Dunnett’s multiple comparison test was used for statistical analysis of (C, D). One-way ANOVA followed by Tukey’s multiple comparison test was used for statistical analysis of (E, J–M).
To evaluate in vivo biocompatibility—including long-term safety—we assessed blood samples and tissues (heart, liver, spleen, lung, kidneys) from rats injected with GaVan, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels in the tibia at both day 7 and day 28 post-administration, which covers the complete degradation cycle of the hydrogel (as the material is fully degraded within 28 days, longer observation periods are not necessary for the scope of this study). ELISA quantification of serum samples from these healthy rats revealed that all hydrogel-treated groups maintained low IL-6 and TNF-α levels at both time points, comparable to Sham controls (Supplementary Fig. 20), demonstrating an absence of cytokine storms and confirming the hydrogels’ in vivo safety profile. Hematological analyses (hemoglobin, platelet counts) and serum biochemistry assessments (liver function: alanine transaminase (ALT), aspartate transaminase (AST), γ-glutamyl transpeptidase (γ-GT); renal function: blood urea nitrogen (BUN), creatinine (CREA)) revealed no significant abnormalities across all groups at either time point, with GaCuVan&HACHO-BSA-treated rats showing profiles comparable to Sham (Supplementary Fig. 21). Histopathological examination via H&E staining confirmed no pathological changes, tissue damage, or inflammatory infiltrates in any organ at 7 or 28 days (Supplementary Fig. 22). Mechanistically, the hydrogel’s biocompatibility stems from its HA-based matrix—natural HA in HACHO-BSA is a low-immunogenic glycosaminoglycan, validated by the absence of fibrous capsule formation (Supplementary Fig. 23)—and renal/hepatic clearance of degradation byproducts, which induced no toxicity as evidenced by stable liver and kidney function markers across all time points. These consistent in vitro and in vivo safety profiles support broad clinical translation potential.
Next, we investigated the antimicrobial properties of the hydrogels, with experimental groups using GaVan, GaCuVan, HACHO-BSA, GaCuVan&HACHO-BSA, and Van designated as T1 (Control), T2 (GaVan), T3 (GaCuVan), T4 (HACHO-BSA), T5 (GaCuVan&HACHO-BSA), and T6 (Van). The standard plate coating method revealed that MRSA and S. aureus cultured in the presence of GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels failed to form colonies on LB agar, indicating significant bactericidal activity of these hydrogels, presumably owing to Van. In line with this, HACHO-BSA hydrogel did not inhibit bacterial growth, with colony counts around 109 CFU/mL similar to control (Fig. 4F, J, K).
To visually assess antibacterial effects, SYTOTM 9/PI staining was performed (Fig. 4G, L, M and Supplementary Fig. 24). The control and HACHO-BSA groups showed clear green fluorescence, indicating live bacteria, while the GaVan, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA groups exhibited strong red fluorescence, indicative of bacterial death. Quantitative analysis of PI-positive cells confirmed these findings.
SEM and transmission electron microscopy (TEM) images revealed significant morphological damage to MRSA and S. aureus in GaVan, GaCuVan, and GaCuVan&HACHO-BSA groups, manifested by damage and rupture of the bacterial cell membrane (arrows) and leakage of the cytoplasm (stars) (Fig. 4H, I). Bacterial growth curves also confirmed that GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels effectively inhibited bacterial growth, with OD600 values remaining stable, while the control and HACHO-BSA groups showed typical growth curves (Fig. 4N, O). These results collectively demonstrated the potent bactericidal activity of GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels, with no cytotoxicity or adverse effects on blood or tissue biocompatibility, confirming their potential for clinical application.
In vitro immunomodulatory and oxidative stress regulation by GaCuVan&HACHO-BSA hydrogel
Macrophages are pivotal in the innate immune response, facilitating pathogen defense and wound healing following tissue injury40,41. Due to their high plasticity, macrophages can rapidly polarize into different phenotypes in response to environmental cues, playing a key role in immune regulation and tissue remodeling42. Previous studies have shown that Ga can modulate immune responses, promoting rapid wound healing in diabetic conditions21. We therefore hypothesized that the GaCuVan&HACHO-BSA hydrogel, formed by Ga self-assembly, might exert similar immunomodulatory effects.
To experimentally validate the role of HA in targeting macrophages via CD44 in vivo, we performed flow cytometric analysis of bone marrow tissues following injection of FITC-labeled HA (HA-FITC) into the bone marrow cavity. Six hours after injecting HA-FITC directly into the bone marrow cavity of rats, we harvested the bone marrow tissues and analyzed cellular fluorescence distribution using flow cytometry. Compared with the control group injected with free FITC, the HA-FITC group showed a significant increase in fluorescence intensity specifically within CD44⁺ cells (Supplementary Fig. 25). This result directly demonstrates that HA exhibits robust in vivo targeting capability to CD44⁺ macrophages within the bone marrow microenvironment, thereby validating the proposed CD44-mediated targeting mechanism.
To mimic the interaction of hydrogel with pathogen-related molecules in the bone marrow cavity, we incubated GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels with LTA (200 μg/mL) for 12 h in Dulbecco’s Modified Eagle Medium (DMEM). Following saturation with LTA, hydrogels were transferred to fresh media and co-cultured with Raw264.7 cells for 24 h; this preparation was specifically used for experiments investigating the regulation of immune cells and inflammatory cytokine secretion, as detailed in Fig. 5A, C–H.
A Schematic representation of the mechanism of activation of macrophage M1 polarization, APCs maturation and differentiation by GaCuVan&HACHO-BSA hydrogel after 12 h of co-incubation with LTA. B Schematic representation of GaCuVan&HACHO-BSA hydrogel after 6 days of co-incubation in DMEM medium, and the extract was harvested and co-cultured with Raw264.7 (Created in BioRender. Chen, H. (2025) https://BioRender.com/40jn2m1). C Cytoskeleton staining images of Raw264.7 cells cultured under standard conditions or co-incubated with GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels for 24 h, after pre-incubation of the hydrogels with DMEM containing LTA (200 μg/mL) for 12 h. qRT-PCR analysis and quantitation of miR-155 (D), IL-1β (E), and IL-6 (F) expression in Raw264.7 treated with GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels and LTA (10 μg/mL) for 24 h, after the different hydrogels were treated as before. Cytokine levels of TNF-α (G) and IL-10 (H) in culture supernatants of RAW264.7 co-incubated with differently treated hydrogels were detected by an ELISA kit. Representative micrographs (I, J) and corresponding quantitative analysis (M, N) of intracellular ROS and O2·− in LPS-stimulated Raw264.7 cells co-cultured under standard conditions or in GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogel extracts. Representative fluorescence images of CD86 (K) and CD206 (L) expressed by RAW264.7 were observed by CLSM after co-incubation with different extracts, and protein expression of CD86 (O) and CD206 (P) was quantified. The data in (D–H, M–P) were expressed as the mean ± s.d. (n = 3 biologically independent samples in (D–F), n = 5 independent samples in (G, H, M–P), with the results shown as mean ± SEM. Statistical analysis was performed using One-way ANOVA with Tukey’s multiple comparison test. A, B were generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
Morphological changes in macrophages were observed by cytoskeleton staining to assess the impact on maturation and differentiation. Free LTA-induced macrophage morphology served as a positive control (Fig. 5C and Supplementary Fig. 26). Macrophages exposed to HACHO-BSA and GaCuVan&HACHO-BSA hydrogels exhibited irregular dendritic or elongated morphology characteristic of M1 polarization, with multiple pseudopodia extending from the cell body. In contrast, macrophages incubated with GaCuVan hydrogel showed a more spherical shape, resembling the control group. The GaCuVan&HACHO-BSA hydrogel induced a more irregular morphology compared to HACHO-BSA alone, similar to the free LTA group. To investigate the effect of LTA-adsorbed hydrogels on antigen-presenting cell (APC) maturation and macrophage polarization, we performed quantitative real-time polymerase chain reaction (qRT-PCR) to measure the expression of miRNA-155 (Fig. 5D), a key gene in APC maturation, and pro-inflammatory cytokines IL-1β and IL-6 (Fig. 5E, F). Exposure to HACHO-BSA and GaCuVan&HACHO-BSA hydrogels upregulated miRNA-155 expression by 3.63-fold and 6.09-fold, respectively, compared to the control. In contrast, GaCuVan hydrogel did not significantly affect miRNA-155 expression, confirming that HACHO-BSA and GaCuVan&HACHO-BSA hydrogels promote APC maturation and differentiation, with GaCuVan&HACHO-BSA having the most significant effect across different groups. Further experiments showed that LTA-adsorbed HACHO-BSA and GaCuVan&HACHO-BSA hydrogels significantly increased IL-1β and IL-6 mRNA expression in macrophages, while GaCuVan hydrogel had no significant effect. ELISA analysis (Fig. 5G, H) of cytokine secretion revealed that TNF-α and IL-10 secretion was unaffected by GaCuVan hydrogel. However, LTA-adsorbed HACHO-BSA and GaCuVan&HACHO-BSA hydrogels significantly elevated TNF-α and suppressed IL-10 secretion, with GaCuVan&HACHO-BSA showing the most significant effect, comparable to free LTA. These results highlight that GaCuVan&HACHO-BSA hydrogel induced APCs maturation and macrophage M1 polarization more effectively than HACHO-BSA alone, likely due to higher LTA adsorption by the GaCuVan&HACHO-BSA hydrogel.
Chronic osteomyelitis is characterized by prolonged oxidative stress, which accelerates M1 polarization and hinders bone repair43. Antioxidants have been shown to reduce reactive oxygen species (ROS) levels and promote healing in osteomyelitis7,43,44. Notably, Ga also exhibits antioxidant properties in addition to its immunomodulatory effects. To explore the potential of GaCuVan&HACHO-BSA hydrogel in modulating oxidative stress, we established an in vitro chronic osteomyelitis model by stimulating RAW264.7 cells with LPS. GaVan, GaCuVan and GaCuVan&HACHO-BSA hydrogels were incubated in culture medium at 37 °C for 6 days (Fig. 5B). The hydrogel extracts were then applied to LPS-stimulated RAW264.7 cells to assess ROS scavenging activity. Using the fluorescent probe 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA), we observed that LPS significantly induced ROS production, as indicated by enhanced green fluorescence in RAW264.7 cells (Fig. 5I, M and Supplementary Fig. 27). In contrast, GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels significantly reduced ROS levels, suggesting strong antioxidant activity. These hydrogels also demonstrated effective scavenging of superoxide radicals (O2·−) with a scavenging rate of approximately 77.61% (Fig. 5J, N and Supplementary Fig. 28). To further evaluate the immunomodulatory effects of the hydrogels, we analyzed the expression of M1 macrophage markers (CD86) and M2 macrophage markers (CD206). RAW264.7 cells exhibited stronger CD86 fluorescence and weaker CD206 fluorescence, confirming LPS-induced M1 polarization (Fig. 5K, L, O, P). In comparison, cells treated with GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels showed weaker CD86 and stronger CD206 fluorescence, indicating that these hydrogels could inhibit M1 polarization and promote M2 polarization, which facilitates bone tissue healing after bacterial clearance. Therefore, in the absence of pathogen-associated molecules, GaCuVan&HACHO-BSA hydrogel effectively exerted antioxidant effects, inhibiting M1 polarization and promoting M2 polarization, which was pivotal for counteracting elevated ROS levels and immune dysregulation in chronic osteomyelitis.
In vitro osteogenic potential and mechanism of GaCuVan&HACHO-BSA hydrogel
Osteomyelitis often results in impaired osteoblast function, leading to abnormal bone repair and localized osteoporosis, which increases fracture risk43. Therefore, understanding the effects of the GaCuVan&HACHO-BSA hydrogel on bone repair, particularly its influence on osteoblast markers, genes, and proteins, is crucial for evaluating its therapeutic potential. To this end, alkaline phosphatase (ALP), an early marker of osteoblast differentiation, was first stained, and its activity was detected. Figure 6A, C shows the ALP staining (blue) in the blanked group, indicating successful differentiation of BMSC into osteoblasts when cultured in osteogenic induction medium. In comparison with the blanked group, GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels significantly enhanced ALP staining intensity (A) and activity (C), with the GaCuVan and GaCuVan&HACHO-BSA hydrogels showing the most significant effects among the four groups. In addition, as the co-culture time of hydrogels and BMSC increased from 7 days to 14 days, both the intensity and activity of ALP staining were further enhanced, indicating sustained promotion of osteogenic differentiation by the hydrogels. For further evaluation of markers of osteoblast differentiation and maturation, mineralized nodules were stained and quantitatively analyzed. These mineralized nodules are the classic morphological features of osteoblasts in the performance of osteogenic functions (Fig. 6B, D). Similarly, GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels displayed significantly more red-stained mineralized nodules than the blanked group, with the number and quantification of nodules increasing over time. Notably, the GaCuVan and GaCuVan&HACHO-BSA hydrogels still had the highest quantification of mineralized nodules. The above results indicated that Ga-containing hydrogels promoted early osteoblast differentiation and later mineralization. The presence of Cu2+ further enhanced the osteogenic effect.
Representative ALP staining micrographs (A) and corresponding ALP activity assay results (C) of BMSC after co-incubation with GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels on days 7 and 14. Osteogenic induction solutions were added to all groups. Alizarin Red S (ARS) staining of mineralized nodules (B) and corresponding quantitative analysis (D) after 14 and 21 days of co-incubation with BMSC using different hydrogels. qRT-PCR analysis and quantitation of Runx-2 (E), COL1A1 (F), ALP (G), OPG (H), and Osterix (I) expression in BMSC treated with osteogenic induction solution (β-Glycerophosphate: 10 mM, Ascorbic Acid: 300 nM, Dexamethasone: 10 nM) and co-cultured with GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels. J Representative immunofluorescence staining micrographs of BMSC co-incubation with different hydrogels. Green: Molecular probes labeling Runx-2, red: phalloidin labeling the cytoskeleton, and blue: DAPI labeling the nucleus. K Statistical analysis of the relative fluorescence density of Runx-2 protein. L The representative Western blot analysis images were used to assess the protein expression levels of genes related to osteogenic differentiation, including COL1A1, Runx-2, OPN, and BMP2 in BMSC (with GAPDH as the internal control). M–P Quantitative analysis of relative changes in protein expression ratios of COL1A1/GAPDH (M), Runx-2/GAPDH, OPN/GAPDH, and BMP2/GAPDH in BMSC. The data in (C–I, K, M–P) were expressed as the mean ± s.d. (n = 5 independent samples in C, D, K, n = 3 biologically independent samples in E–I, M–P), with the results shown as mean ± SEM. Statistical analysis was performed using One-way ANOVA with Tukey’s multiple comparison test.
Then, the expression of early genes (like ALP, Runx-2, Osterix) and late genes (like COL1A1, OPG, OPN, BMP2) during osteogenic differentiation of BMSC was detected by qRT-PCR, cell immunofluorescence, and Western blot (WB). Specifically, the mRNA levels of Runx-2, COL1A1, ALP, OPG, Osterix, OPN, and BMP2 were upregulated 1.64 ± 0.18, 1.58 ± 0.10, 1.44 ± 0.10, 1.76 ± 0.02, 1.32 ± 0.01, 2.40 ± 0.34, and 2.18 ± 0.07 times, respectively, in the GaVan group compared to the blanked group. Furthermore, BMSC co-cultured with GaCuVan and GaCuVan&HACHO-BSA hydrogels showed even higher upregulation than the GaVan group, with no significant statistical difference between the two hydrogel groups. It was worth noting that the GaCuVan and GaCuVan&HACHO-BSA hydrogels were the most effective in promoting the expression of osteogenesis-related mRNA and were superior to the GaVan hydrogel (Fig. 6E–I and Supplementary Fig. 29). To address component synergy, we evaluated Ga and Cu²⁺ alone in BMSC cultures. qRT-PCR showed that Ga and Cu²⁺ each enhanced RUNX-2 mRNA expression by 4.22 ± 0.99-fold and 4.49 ± 0.56-fold, respectively, compared to the blanked group, while HACHO-BSA alone had no effect (Supplementary Fig. 30). Similar trends were observed for COL1A1, ALP, OPG, and Osterix, demonstrating that both ions independently promoted osteogenesis—and exhibited the most significant effects when combined. Immunofluorescence analysis (Fig. 6J, K) revealed the highest expression of Runx-2 in both GaCuVan and GaCuVan&HACHO-BSA hydrogel groups, with significantly stronger green fluorescence compared to the blanked group. Although slightly weaker, expression in the GaVan group was still notably higher than in the blanked group. In addition, WB further confirmed the upregulation of COL1A1, Runx-2, OPN, and BMP2 proteins (Fig. 6L–P), consistent with the qRT-PCR and immunofluorescence results. Overall, GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels effectively promoted osteoblast differentiation and the expression of osteogenic genes and proteins at various stages of BMSC growth, with the GaCuVan and GaCuVan&HACHO-BSA hydrogels showing the most significant osteogenic effects.
In situ GaCuVan&HACHO-BSA hydrogel administration for osteomyelitis therapy
The GaCuVan&HACHO-BSA hydrogel effectively absorbs pathogen-related molecules, promoting the maturation and differentiation of APCs and inducing macrophage polarization to the M1 type. Moreover, the hydrogel could release Van and Ga to exhibit significant antibacterial activity and effectively promote osteogenic differentiation of BMSC. Inspired by these in vitro observations, we went on to validate its therapeutic effect on osteomyelitis in rats infected with drug-resistant bacteria. The experimental schedule for the treatment of osteomyelitis is shown in Fig. 7A and Supplementary Fig. 31. The experiment began with the establishment of the rat tibia infection model of MRSA and the injecting different hydrogels in situ on day 0, followed by sampling on the indicated days. Six groups of MRSA-infected rats were randomly assigned: G1, Sham group; G2, PBS group; G3, Van group; G4, GaVan group; G5, GaCuVan group; G6, GaCuVan&HACHO-BSA group.
A Experimental protocol for treating osteomyelitis by in situ hydrogel administration including constructing MRSA-infected osteomyelitis, in situ administration with different hydrogels, and then animal sampling and performing various experiments at different time points (G1, Sham; G2, PBS; G3, Van; G4, GaVan; G5, GaCuVan; G6, GaCuVan&HACHO-BSA) (Created in BioRender. Chen, H. (2025) https://BioRender.com/mo6lg6a). Representative MRSA bioluminescence images (B) and corresponding quantitative data (C) were analyzed using IVIS on days 1, 3, and 7 after injection of different hydrogels. Representative standard plate coating photographs (D) and CFU counts (E) of MRSA in bone marrow on days 7, 14, and 28 of infection, with PBS, GaVan, GaCuVan, GaCuVan&HACHO-BSA hydrogels, and Van pretreated in all rat groups. F Representative photographs of surrounding soft tissue (left) and tibia (right) from MRSA-infected rats after the above treatments, captured at days 7, 14, and 28 post-treatments. Changes in body temperature (G) and body weight (H) of rats were recorded throughout the treatment period. I Following treatment with the different hydrogels, routine blood tests were performed at days 7, 14, and 28. H&E staining (J) of bone marrow sections from osteomyelitis rats and the number of NEUT (K), indicated by red arrows, were analyzed after all rat groups were pretreated with PBS, GaVan, GaCuVan, GaCuVan&HACHO-BSA hydrogels, and Van. L Wright-Giemsa staining of bone marrow sections from osteomyelitis rats on days 14 and 28, with bacteria indicated by red arrows, was performed after all rat groups were pretreated with the above treatments. The data in (E, G, H, K) were expressed as the mean ± s.d. (n = 4 biologically independent samples per group), with the results shown as mean ± SEM. Statistical significance and P values were determined by two-way ANOVA followed by Dunnett’s multiple comparison test (G, H) or one-way ANOVA followed by Tukey’s multiple comparison test (E, K). A was generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
To track the development of MRSA-infected osteomyelitis, the small animal living imaging system (IVIS) was used to detect bioluminescent MRSA. Obvious bioluminescence was observed at the tibial injection site (red box) of rats in each group on day 1, and there was no significant difference in luminescence intensity, indicating that the tibial osteomyelitis model was successfully established (Fig. 7B, C). On days 3 and 7, MRSA bioluminescence was no longer detectable in the G6 group, while bioluminescence was overt in the other groups, indicating that in situ administration of the hydrogel could rapidly control bacterial infection, while other groups maintained a persistent infection state. This potent bactericidal effect could be attributed to the hydrogel’s ability to locally capture bacteria, releasing and maintaining effective Van concentrations in the bone marrow cavity. Additionally, it might involve the adsorption of pathogen-related molecules, which activate the immune response to combat microbial invasion.
To evaluate bacterial load in the bone marrow cavity, tibia samples were taken from rats at 7, 14, and 28 days after different hydrogel treatments. After removing the epiphysis at both ends and washing out the infected marrow fluid with PBS, standard plate coating and CFU counting were performed. As shown in Fig. 7D, E, no bacterial colony growth was observed in the bone marrow fluid of the G1 group, while G2 had a CFU count of 1 × 106 CFU/mL. In comparison with the G2 group, the CFU counts of the G3-G5 groups did not change significantly, while the CFU counts of the G6 group decreased significantly. More significantly, by day 7, CFU had decreased by 5 log units in the G6 group, and by days 14 and 28, almost no bacterial survival was observed in the G6 group, while the rats in the G2–G5 groups still had obvious bacterial infections. The Van, GaVan, and GaCuVan groups showed comparable bacterial growth to the PBS group due to two key limitations. First, prior studies have shown that single-dose Van administration, whether via local injection or intravenous delivery, failed to resolve deep bone infections45. Second, GaCuVan hydrogel degraded within 2 days in vivo (Fig. 2K), leading to burst release of Van that could not maintain local bactericidal concentrations long enough to clear infections. This rapid degradation kinetics explained the lack of antibacterial activity in these groups. To address these limitations, we incorporated a chitosan-glycerol phosphate (CS-GP) thermosensitive hydrogel loaded with Van (Van/Gel) as a positive control46. At day 14, both Van/Gel and GaCuVan&HACHO-BSA groups achieved sterile bone marrow cultures, whereas PBS controls exhibited 5.70 ± 0.18 × 106 CFU/mL (Supplementary Fig. 32). This confirmed that sustained local antibiotic delivery—enabled by the hydrogel’s dual-network design—overcame the limitations of single-dose Van. These results aligned with prior reports highlighting the inadequacy of conventional Van regimens for deep osteomyelitis45, underscoring the necessity of our slow-release strategy. For subsequent experiments, to focus on investigating the mechanism by which our hydrogel induces trained immunity, we excluded the HACHO-BSA group (which showed no antibacterial activity) and the Van/Gel group (serving solely as a positive control for sustained drug delivery).
To further contextualize our hydrogel’s clinical relevance, we also compared it to a widely used clinical standard for osteomyelitis: Van-loaded poly(methylmethacrylate) (PMMA) bone cement (PMMA@Van)47. Using the same standard plate coating and bone marrow CFU counting assays, we found that PMMA@Van failed to effectively clear MRSA infections (Supplementary Fig. 32). This limitation stems from the inherent properties of non-degradable PMMA: antibiotic release from PMMA cement is slow and incomplete, leading to subtherapeutic local concentrations at infection foci48. This aligns with prior studies showing antibiotic-loaded PMMA struggles to treat chronic osteomyelitis caused by drug-resistant bacteria49, and these findings underscore that our hydrogel’s sustained-release strategy addresses an unmet clinical need by overcoming the key shortcomings of PMMA-based clinical delivery systems. For subsequent experiments, to maintain focus on dissecting the mechanism of trained immunity induction by our hydrogel, we further excluded the PMMA@Van group—this group served solely as a control to benchmark against clinical antibiotic-loaded bone cement therapy, with no relevance to mechanistic exploration of trained immunity.
To gain an intuitive understanding of the effect of MRSA infection on surrounding tissues, the changes in soft tissue and bone defects were also observed (Fig. 7F). In G2–G5 groups, the soft tissues around the infection were swollen and formed abscesses (large circles), particularly on day 7, while no abscesses appeared in the G1 and G6 groups during the entire observation period, indicating that the hydrogel could rapidly control the infection and inhibit the formation of abscesses. In addition, on the 7th day of MRSA infection, bone defects (small circles) were observed in all groups, but by day 28, the defects in each group had largely recovered, indicating that the bone defects had been repaired.
The basic vital signs of the rats, including body temperature (Fig. 7G), body weight (Fig. 7H), and blood routine (Fig. 7I), showed significant differences in the presence vs. absence of GaCuVan&HACHO-BSA hydrogel. Rats in the G6 group exhibited a rapid increase in body temperature within the first week post-hydrogel injection, which returned to normal starting in the second week, while other groups experienced prolonged fever. In addition, rats in the G6 group initially gained weight more slowly than normal controls during the first week but showed accelerated weight gain in the second week, approaching that of non-infected controls. In contrast, the other groups showed consistently slower weight gain. These findings suggest that the hydrogel effectively alleviated clinical symptoms caused by MRSA-infected osteomyelitis. Interestingly, blood analysis revealed that the number of white blood cells (WBCs) and neutrophils (NEUT) in group G6 were significantly higher than that in other groups on days 7 and 14 but returned to normal by day 28. This suggests that the early action of the hydrogel promoted the initiation and progression of the inflammatory response owing to in situ adsorption of various pathogen-related molecules, while later control of infection might be due to the presence of Ga, helping to restore immune balance.
To further assess the efficacy of the hydrogel in promoting inflammatory cell accumulation and eliminating bacterial load, rat bone marrow sections were stained with H&E, Wright-Giemsa staining. On day 14 of MRSA infection, a significant increase in NEUT was observed in the G2-G6 groups (arrows), with the highest NEUT accumulation in the G6 group (Fig. 7J, K and Supplementary Fig. 33). In addition, compared to day 24, NEUT levels were reduced in all groups on day 28, and the reduction was most significant in the G6 group, approaching the level of the G1 group. As shown in Fig. 7L and Supplementary Fig. 34, no bacteria were detected in the G1 and G6 groups on days 14 and 28 (arrows), whereas the G2–G5 groups maintained persistent bacterial infections. Additionally, we performed iNOS/CD68 immunofluorescence staining (M1 macrophage markers) on rat bone marrow sections. By day 14, the PBS, Van, GaVan, GaCuVan, and GaCuVan&HACHO-BSA groups all showed significantly increased iNOS/CD68 fluorescence, with the GaCuVan&HACHO-BSA group exhibiting the highest intensity (Supplementary Fig. 35). This indicated that the in situ GaCuVan&HACHO-BSA hydrogel injection promoted M1 macrophage polarization in bone marrow tissue to control bacterial infection.
In conclusion, in situ hydrogel effectively controlled bacterial infection by promoting inflammatory cell chemotaxis and modulating the immune microenvironment.
In vivo potential and mechanism of GaCuVan&HACHO-BSA hydrogel to promote osteomyelitis bone regeneration
Bone regeneration is equally crucial in osteomyelitis treatment, alongside bacterial eradication. To investigate the repair of bone defects in osteomyelitis, the bone defects in the tibia were monitored at 4 and 8 weeks. Micro-computed tomography (micro-CT) images of the damaged tibia showed that the bone defect (dashed box) in the Sham group was significantly smaller at 8 weeks than at 4 weeks, consistent with the normal healing rate in healthy rats (Fig. 8B, D). In contrast, the PBS and Van groups showed no effective repair, with the defect area unchanged, confirming that bacterial infection impedes bone regeneration. Notably, the GaVan, GaCuVan, and GaCuVan&HACHO-BSA groups demonstrated significantly better bone repair than the PBS group, especially at 8 weeks, with nearly complete healing. More importantly, the GaCuVan and GaCuVan&HACHO-BSA groups outperformed the GaVan group, suggesting that Ga and Cu2+ released from the hydrogel enhance osteomyelitis-induced bone regeneration. This conclusion was further supported by bone mineral density (BMD), bone volume fraction (BV/TV), trabecular thickness (Tb.Th), and trabecular separation (Tb.Sp) (Fig. 8C, E and Supplementary Fig. 36).
A In situ hydrogel administered in advance not only controlled infection in the bone marrow cavity, but also released Ga and Cu2+, which upregulated the expression of bone-forming factors (Runx-2, Osterix, OPG, COL1A1 and ALP), improved bone strength (increasing BMD, BV/TV and Tb.N and decreasing Tb.Sp) and supported bone regeneration and repair in osteomyelitis (Created in BioRender. Chen, H. (2025) https://BioRender.com/7zr6adl). Representative micro-CT images of rat tibias at 4 weeks (B) and 8 weeks (D), along with the corresponding quantitative analysis of bone mass and microarchitectural parameters, including BV/TV, Tb. Th, and Tb. Sp (C, E), were obtained after all rat groups were pretreated with PBS, GaVan, GaCuVan, and GaCuVan&HACHO-BSA hydrogels and Van. The red dotted circle represented the infected bone area. Representative Masson staining photographs of the tibia of rats with osteomyelitis (F), and corresponding quantification of the new bone area (G) at weeks 4 and 8 after in situ injection with different hydrogels. Representative COL1A1 immunohistochemistry images (H) of tibiae from rats treated with different hydrogels for 4 weeks and 8 weeks, and quantification of COL1A1 protein (I). Representative Runx-2 immunofluorescence images (J) of tibiae from rats treated with the same hydrogels as above and quantification of Runx-2 fluorescence intensity (K). The data in (C, E, G, I, K) were expressed as the mean ± s.d. (n = 4 biologically independent samples per group), with the results shown as mean ± SEM. Statistical analysis was performed using One-way ANOVA with Tukey’s multiple comparison test. A was generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
To further evaluate tibial bone repair, H&E staining and Masson staining were performed (Fig. 8F, G and Supplementary Fig. 37). After 4 and 8 weeks of treatment, the GaVan, GaCuVan and GaCuVan&HACHO-BSA groups had significantly larger areas of newly formed bone tissue, with a more organized and denser structure compared to the PBS and Van groups, among which the effect of the GaCuVan&HACHO-BSA group was most significant. In addition, bone repairs at 8 weeks were markedly improved compared to 4 weeks, further demonstrating the potential of the hydrogel in promoting bone regeneration.
To further elucidate the mechanism of bone repair in vivo, immunohistochemical staining, immunofluorescence staining, and quantitative analysis were performed for the early bone regeneration protein Runx-2 and the late bone regeneration protein COL1A1. As shown in Fig. 8H–K and Supplementary Fig. 38, the expression of Runx-2 and COL1A1 was significantly enhanced in the GaVan, GaCuVan, and GaCuVan&HACHO-BSA groups compared with the PBS and Van groups, among which the effect of the GaCuVan and GaCuVan&HACHO-BSA group was most significant. In conclusion, in situ inoculation of the hydrogel killed bacteria by activating the immune response. When the infection of the bone marrow cavity was well under control, bone regeneration and bone repair of osteomyelitis were facilitated as a result of upregulating the expression of Runx-2 through Ga and Cu2+, and the downstream bone formation-related factors such as COL1A1, resulting in increases in BMD, BV/TV, and Tb.N, while decreasing Tb.Sp (Fig. 8A).
GaCuVan&HACHO-BSA hydrogel functions as a vaccine, inducing trained immunity through metabolic reprogramming, preventing bacterial metastatic osteomyelitis
Local bacterial infections, if they remain unchecked, can lead to bacteremia or sepsis, causing systemic complications and organ damage8. To explore whether the GaCuVan&HACHO-BSA hydrogel could function as a vaccine capable of preventing recurrence of bacterial infections, the hydrogel treatment was administered on the first day after the establishment of the rat model of localized osteomyelitis, followed by induction of ectopic osteomyelitis in the contralateral hind limb 2 weeks later through MRSA infection (Fig. 9A). General condition of the rats, transcriptome sequencing (RNA-seq) analysis, histopathology, metabolic reprogramming and trained-immunity-related factors of bone marrow tissues were comprehensively evaluated at different time points post-infections. To further verify the potential mechanism of GaCuVan&HACHO-BSA hydrogel in stronger immune activation, rats with ectopic osteomyelitis after in situ inoculation of the hydrogel were randomly divided into six groups: G1, Sham group; G2, PBS group; G3, Van group; G4, GaVan group; G5, GaCuVan group; G6, GaCuVan&HACHO-BSA group.
A Ectopic osteomyelitis study protocols including pre-delivery of different hydrogels, establishment of ectopic MRSA-infected osteomyelitis, followed by treatments across groups (G1, Sham group; G2, PBS group; G3, Van group; G4, GaVan group; G5, GaCuVan group; G6, GaCuVan&HACHO-BSA group) and subsequent transcriptomic, immunological, histopathological, and bacteriological analyses (Created in BioRender. Chen, H. (2025) https://BioRender.com/n054ax4). B Heat map and functional classification of differentially expressed genes in bone marrow of rats with ectopic osteomyelitis pretreated with GaCuVan&HACHO-BSA hydrogel and Van (fold change ≥ 2 and q < 0.05). C GO enrichment analysis of the upregulated genes in GaCuVan&HACHO-BSA hydrogel group and Van group. D KEGG enrichment analysis of the upregulated DEGs in GaCuVan&HACHO-BSA hydrogel group and Van group. E–H GSEA analysis was performed on GO, KEGG, Reactome, and Wikipathway datasets from the rat, and the significantly enriched gene sets ranked in the top 5 by absolute NES value were selected by default for map display. Representative HIF-1α (I) and COX2 (J) immunofluorescence images of bone marrow sections on day 14 of MRSA infection with ectopic osteomyelitis and quantification of HIF-1α (K) and COX2 (L) protein. Cytokine levels of IL-17 (M), IL-6 (N), and TNF-α (O) in bone marrow from ectopic osteomyelitis rats on day 14 were detected by an ELISA kit. Cytokine levels of IL-17 (P), IL-6 (Q), and TNF-α (R) in serum from ectopic osteomyelitis rats on day 14 were measured using an ELISA kit. The data in (K–R) were expressed as the mean ± s.d. (n = 4 biologically independent samples per group), with the results shown as mean ± SEM. Statistical analysis was performed using One-way ANOVA with Tukey’s multiple comparison test. A was generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
To thoroughly analyze the pathways and mechanisms of GaCuVan&HACHO-BSA hydrogel in activating stronger immunity, RNA-seq analysis of bone marrow tissues of rats in each group was performed 14 days after administration. The results showed that a total of 184 differentially expressed genes (DEGs) related to hydrogel injection were identified in the bone marrow of rats in groups G1, G2 and G6 (fold change ≥ 2 and q < 0.05) (Fig. 9B and Supplementary Fig. 39). As shown in Fig. 9C, Gene Ontology (GO) database analysis revealed significant changes in differentially expressed genes in specific biological process, molecular function or cellular component (CC). Compared with G2 group, the expression of genes related to immune response, response to bacterium, immune system process, regulation of MyD88 independent toll-like receptor signaling pathway, response to external biotic stimulus, regulation of pattern recognition receptor signaling pathway, CXCR chemokine receptor binding and chemokine activity (e.g., Irf7, Dhx58, Ifit3, Cxcl2) were significantly increased in group G6 (red rectangle), further confirming the effect of GaCuVan&HACHO-BSA hydrogel in activating more robust immune response at the molecular level. In addition, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways enrichment analysis showed that the expression levels of genes involved in retinoic acid-inducible gene (RIG)-I-like receptor signaling pathway, IL-17 signaling pathway, nucleotide-binding oligomerization domain (NOD)-like receptor signaling pathway, Bacterial invasion of epithelial cells, Toll-like receptor (TLR) signaling pathway, NF-kappa B (NF-κB) signaling pathway and TNF signaling pathway activated after hydrogel treatment (red rectangle) (Fig. 9D). Reassuringly, gene set enrichment analysis (GSEA) also revealed that the hydrogel delivery can activate a number of changes in the immune response (Fig. 9E–H), including upregulation of genes in cytokines and inflammatory response pathway (like CXCL2, CSF3, CXCL3, IFNb1, IL2, IL6, IL11, CSF2, IL12b, TBX21, XCL1, CCL3, CCL4, IL12b, IL18), while downregulation of genes related to negative regulation of cytokine production involved in immune response, negative regulation of production of molecular mediator of immune response and IL-10 signaling pathways (like IFNb1, ARG1, RT1-M6-2, CD96, IL10, FOXP3, Zpbp2, CCL22). In general, activation of pattern recognition receptors (NOD-like receptor signaling pathway, TLR signaling pathway), IL-17 signaling pathway, and NF-κB signaling pathway is a core driver of trained immunity50,51. These pathways enhanced neutrophil recruitment, promoted macrophage activation, and induced epigenetic changes by upregulating pro-inflammatory cytokines (e.g., IL-1β, IL-6, TNF-α)52. In addition, activation of the RIG-I-like receptor signaling pathway and Th1 polarization (e.g., TBX21, IL12b) suggested that the hydrogel had potential broad-spectrum immune memory53.
HIF-1α regulates glycolytic genes in immune cells, which are critical for trained immunity54. To verify that GaCuVan&HACHO-BSA hydrogel induced trained immunity, the expression of HIF-1α and COX2 in bone marrow tissue sections was detected by immunofluorescence staining. As shown in Fig. 9I–L and Supplementary Fig. 41, the PBS group showed significantly increased HIF-1α and COX2 expressions compared to the Sham group. Following the hydrogel injection, HIF-1α and COX2 levels were significantly higher than in the PBS group, indicating enhanced glycolysis and an amplified inflammatory response in bone marrow tissue. Metabolic profiling of bone marrow tissues showed that hydrogel treatment increased lactate production (2.97 ± 0.11 mM vs. 0.97 ± 0.05 mM in Sham) and ATP levels (296.13 ± 6.25 nM vs. 202.80 ± 5.66 nM) (Supplementary Fig. 40A, B), indicative of enhanced glycolysis55. Concomitantly, succinate levels—key TCA cycle metabolites that stabilized HIF-1α56—were elevated (54.27 ± 1.94 μM vs. 23.25 ± 1.05 μM) (Supplementary Fig. 40C), directly linking metabolic rewiring to HIF-1α upregulation observed in bone marrow tissue. These data established a mechanistic chain where succinate-driven HIF-1α activation led to a glycolytic burst, ultimately promoting IL-1β/IL-17 production, thereby reinforcing the induction of trained immunity. Subsequent immunofluorescence and immunohistochemistry assays revealed significantly higher iNOS and CD86 expression in the GaCuVan&HACHO-BSA group, indicating a stronger immune response in the bone marrow tissue, potentially linked to trained immunity and macrophage activation (Supplementary Figs. 42 and 43). To evaluate the impact of the hydrogel on trained immunity-related inflammatory factors in osteomyelitis rats, we measured IL-17, IL-6, and TNF-α protein levels by ELISA (Fig. 9M–R). The results showed significantly higher levels of IL-17, IL-6, and TNF-α in both bone marrow and serum of GaCuVan&HACHO-BSA group, further supporting the hydrogel’s role in upregulating pro-inflammatory cytokines. Further analysis of blood routine, bone marrow H&E staining, and NEUT quantification (Supplementary Figs. 44 and 45) showed that WBC and NEUT counts were significantly higher in the G6 group on day 14 of infection. Additionally, the hydrogel reduced infection severity in rats with ectopic bacterial infections. Standard plate counts, CFU counts, and Wright-Giemsa staining of bone marrow tissue revealed almost no bacteria, further demonstrating the hydrogel’s enhanced bactericidal effect through trained immunity (Supplementary Figs. 46 and 47).
Previous studies have reported that 2-deoxyglucose (2-DG) can inhibit glycolysis by competing with glucose, thereby suppressing metabolism57,58. In our study, we aimed to further investigate the functional role of metabolic reprogramming in the context of trained immunity and its related outcomes in osteomyelitis. Initially, we treated osteomyelitis rats by in situ injection of the GaCuVan&HACHO-BSA hydrogel. After 3 weeks, we conducted a reinfection experiment where these rats were reinfected with MRSA at the same site. Subsequently, we divided them into two groups based on whether they were intraperitoneally injected with 2-DG or not. As shown by the standard plate coating and CFU counting in the bone marrow tissue in Supplementary Fig. 48A, B, no bacteria could be cultured in the GaCuVan&HACHO-BSA group, while a large number of bacteria appeared in the 2-DG-treated group. This indicates that the trained immunity induced by GaCuVan&HACHO-BSA, which is crucial for preventing the recurrence of osteomyelitis, was blocked after inhibiting glycolysis by 2-DG, highlighting the importance of metabolic reprogramming in this process. Moreover, to evaluate the impact of 2-DG on the trained immunity-related inflammatory factors induced by the hydrogel in osteomyelitis rats, we detected the protein levels of IL-6 and TNF-α by ELISA (Supplementary Fig. 48C). The results demonstrated that the levels of IL-6 and TNF-α in the bone marrow tissue of the GaCuVan&HACHO-BSA group were significantly increased. However, after treatment with 2-DG, the levels of IL-6 and TNF-α decreased significantly. This suggests that 2-DG, after inhibiting glycolysis, leads to a marked decline in the levels of trained immunity-related inflammatory factors induced by the hydrogel in rats, further strengthening the connection between metabolic reprogramming and the induction of trained immunity.
In summary, the hydrogel successfully induced trained immunity, eliminating ectopic infected bacteria by activating of pattern recognition receptors (NOD-like receptor signaling pathway, TLR signaling pathway, and RIG-I-like receptor signaling pathway), reshaping metabolism (HIF-1α), amplifying inflammation (COX2), activating stronger immune response (iNOS and CD86), and increasing pro-inflammatory factors (IL-17, IL-6, and TNF-α).
GaCuVan&HACHO-BSA hydrogel provided protective immunity in rats with recurrent osteomyelitis by inducing trained immunity
Bacterial recurrence is a significant challenge in osteomyelitis treatment, often involving recurrent infection with either the same or different bacteria, disease exacerbation, antibiotic resistance, and immune escape1. To address this, we developed the hydrogel to control MRSA spread and reduce relapse risk. Three weeks after injecting hydrogels in situ to cure MRSA-infected rats, we reinfected these rats at the same site with MRSA A0068, MRSA RJ0089, and S. aureus and left untreated thereafter (Fig. 10A). To simulate clinical osteomyelitis recurrence, we performed debridement and antibiotic drainage on rats not fully recovered before MRSA reinfection, effectively controlling the infection. Subsequently, the rats with recurrent osteomyelitis induced by different bacterial infections were randomly assigned into six groups: G1, Sham group; G2, PBS group; G3, Van group; G4, GaVan group; G5, GaCuVan group; G6, GaCuVan&HACHO-BSA group. As shown in Fig. 10B, C, rats in groups G2 to G5 exhibited significantly higher luminescence intensity than G1, confirming the successful establishment of the osteomyelitis recurrence model. Compared with the G2, rats pre-injected with the hydrogel showed significant luminescence inhibition on day 3 and near absence by day 7. Standard plate coating and CFU counting of bone marrow tissue revealed a significant reduction in MRSA A0068 counts in group G6, with no bacteria cultured from day 14 (Fig. 10D, E).
A A prophylactic regimen for recurrent osteomyelitis including pre-delivery of different hydrogels, antibiotic drainage after debridement surgery, and establishment of osteomyelitis with MRSA A0068, MRSA RJ0089, and S. aureus reinfection, followed by functional trained immunity validation and analyses of histopathological and bacteriological changes in rats across treatment groups (G1, Sham group; G2, PBS group; G3, Van group; G4, GaVan group; G5, GaCuVan group; G6, GaCuVan&HACHO-BSA group) (Created in BioRender. Chen, H. (2025) https://BioRender.com/2tx4t2f). Pre-injection with the GaCuVan&HACHO-BSA hydrogel was performed in situ in rats, and MRSA bioluminescence images (B) and corresponding quantitative analysis data (C) were obtained on days 1, 3, and 7 after osteomyelitis recurrence. Representative standard plate coating images (D) and CFU counts (E) of MRSA A0068 in bone marrow tissue on days 7, 14, and 21 of recurrent osteomyelitis in all rats. MRSA A0089 (F) and S. aureus (G) CFU counts in bone marrow were quantified at day 7, 14, and 21 in recurrent osteomyelitis. Wright-Giemsa staining results (H) and corresponding bacterial counts (J) (indicated by red arrows) of bone marrow sections from rats with osteomyelitis in S. aureus reinfection on days 14 and 21 were analyzed. Representative IL-1β/TNF-α immunofluorescence images (I) of tibiae from rats treated with different hydrogels for 14 and 21 days and quantification of IL-1β/TNF-α protein (K, L). The data in (E–G, J–L) were expressed as the mean ± s.d. (n = 4 biologically independent samples per group), with the results shown as mean ± SEM. Statistical analysis was performed using One-way ANOVA with Tukey’s multiple comparison test. A was generated using BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.
To verify the broad-spectrum immune memory induced by the hydrogel, bone marrow tissues from rats infected with MRSA RJ0089 and S. aureus were analyzed using standard plate counts and CFU assays. As shown in Fig. 10F, G and Supplementary Fig. 49, bacterial load in the G6 group significantly decreased by day 7, and no bacteria were cultured from day 14 onward, indicating broad-spectrum immune protection induced by the hydrogel. To further assess the immune response following re-challenge with S. aureus, NEUT counts in peripheral blood and bone marrow were analyzed (Supplementary Figs. 50 and 51). Notably, NEUT numbers in both peripheral blood and bone marrow of hydrogel-pretreated rats were significantly higher on days 7 and 14 after bacterial reinfection compared to other groups, returning to baseline levels by day 21. Giemsa staining of bone marrow cavities at day 14 post-reinfection showed no bacteria in the G6 group, whereas other groups still exhibited abundant bacterial presence (Fig. 10H, J). To confirm the enhanced secretion of pro-inflammatory cytokines, IL-1β and TNF-α expression was assessed by immunofluorescence (Fig. 10I, K, L and Supplementary Fig. 52). IL-1β and TNF-α levels were markedly higher in the G6 group, confirming the hydrogel’s ability to upregulate pro-inflammatory cytokine secretion.
In summary, these results emphasized that the GaCuVan&HACHO-BSA hydrogel induced trained immunity, provided strong, broad-spectrum immune memory, effectively preventing and protecting against recurrent osteomyelitis caused by different bacterial strains.
GaCuVan&HACHO-BSA hydrogel treatment to simulate complex clinical scenarios
To address the critical need for clinical translatability and navigate the challenges of real-world applications, we expanded our animal model validation by incorporating diabetic and aged rat osteomyelitis models. This allowed us to simulate complex clinical scenarios and systematically evaluate the hydrogel’s efficacy across diverse pathophysiological conditions—while also linking its performance in these models to its core mechanistic reliance on trained immunity.
To model diabetic osteomyelitis, 6-week-old rats were rendered diabetic via intraperitoneal streptozotocin (STZ, 50 mg/kg), with fasting blood glucose ≥16.7 mmol/L confirming model success. Following tibial MRSA inoculation, rats were treated with GaCuVan&HACHO-BSA hydrogel, which reduced bone marrow bacterial loads to below detectable levels at day 14. In contrast, the Van, GaVan, and GaCuVan groups showed no significant improvement relative to PBS (Supplementary Fig. 53A, B). Notably, in the heterotopic reinfection assay, diabetic rats treated with GaCuVan&HACHO-BSA hydrogel failed to control recurrent MRSA infection: bone marrow CFU counts reached 5.57 ± 0.42 × 105 CFU/mL at day 14, significantly higher than sham-treated controls (Supplementary Fig. 53C, D). This failure was likely driven by diabetic-induced immune dysregulation, which is known to impair trained immunity induction—hyperglycemia disrupts immune cell metabolism, weakens APC function, and compromises immune defense mechanisms that underpin trained immunity59,60. The hydrogel’s inability to prevent reinfection in diabetic hosts, despite clearing primary infection, underscores that its long-term protective effect (not just acute antibacterial action) depends on intact trained immunity machinery.
Additionally, we induced MRSA osteomyelitis in 18-month-old rats and assessed both primary infection clearance and heterotopic reinfection protection using bacterial plate counts and CFU assays. For primary tibial infections, rats were treated with GaCuVan&HACHO-BSA, Van, GaVan, or GaCuVan. At day 14, Van, GaVan, and GaCuVan groups showed robust bacterial growth, whereas GaCuVan&HACHO-BSA completely eradicated primary infections (Supplementary Fig. 54A, B). In heterotopic reinfection assays, where MRSA was inoculated into the contralateral limb after primary treatment, Van, GaVan, and GaCuVan groups again failed to control bacterial loads, while GaCuVan&HACHO-BSA prevented reinfection entirely (Supplementary Fig. 54C, D). This efficacy in aged rats aligned with reports that BCG-induced trained immunity could promote remyelination in senescent hosts61, underscoring the hydrogel’s ability to induce trained immunity in immunosenescent environments.
Collectively, these age- and diabetes-dependent outcomes further confirm that the hydrogel exerts its effects by inducing trained immunity rather than through other mechanisms: aged rats retained intact immune mechanisms necessary for trained immunity induction, so the hydrogel’s capacity to drive sterile inflammation and antigen presentation in the aged bone marrow microenvironment primed durable immune memory—evidenced by the absence of bacteria in both primary and recurrent infections. In contrast, diabetic hosts’ metabolic dysregulation undermined immune memory formation, preventing the hydrogel from exerting long-term protection. This stark contrast between the two models reinforces that the hydrogel’s unique value lies in its ability to induce trained immunity, rather than relying on non-specific antibacterial or immunomodulatory effects, and highlights its utility in immunosenescent rather than metabolically compromised settings.
Discussion
Osteomyelitis, particularly when caused by MDR bacteria62,63, remains a major clinical challenge due to factors such as antibiotic resistance, limited drug penetration, and recurrent infections4,64. While recent advancements in local drug delivery systems, such as hydrogels, have shown promise65,66, there has been no research to date focusing on using trained immunity for osteomyelitis treatment. In this study, we developed the GaCuVan&HACHO-BSA hydrogel as a targeted approach to osteomyelitis therapy, utilizing trained immunity activation to prevent infection recurrence, both from homologous and heterologous bacterial strains.
The hydrogel employs a unique double-network system that improves injectability, adaptability, and plasticity, making it ideal for minimally invasive treatments in complex bone defects. Unlike traditional therapies, the GaCuVan&HACHO-BSA hydrogel induces sustained, controlled immune activation over extended periods. This controlled release of immune-modulating factors ensures robust immune memory, enabling the body to respond more effectively to future infections. Additionally, the hydrogel’s ability to adsorb and capture pathogen-related factors (e.g., bacteria such as MRSA and S. aureus, and inflammatory mediators like TNF-α) at the infection site enhances local infection control. The efficacy of the hydrogel is derived from both physical adsorption (due to its network structure and hydrogen bonding) and chemical interactions (via Schiff base reactions and coordination).
Notably, incorporating HACHO into GaCuVan&HACHO-BSA hydrogel conferred three critical advantages: HACHO enhanced bacterial capture via Schiff base linkages with pathogens (Fig. 3), targeted macrophage CD44 receptors to initiate M1 polarization (Fig. 1G)28,29, and extended hydrogel degradation (Fig. 2K) to sustain Van release. These features integrated bacterial entrapment, immune modulation, and controlled drug delivery, enabling the hydrogel to resolve osteomyelitis through antibacterial action and immune microenvironment regulation.
An RNA-seq analysis of bone marrow tissues revealed that the hydrogel significantly upregulated genes associated with trained immunity, particularly pathways involving IL-17, TLR, and NF-κB signaling. These pathways are key drivers of trained immunity, enhancing neutrophil recruitment, macrophage activation, and epigenetic changes in immune cells. Additionally, the hydrogel activated pattern recognition receptors, including the NOD-like receptor signaling pathway, TLR signaling pathway, and RIG-I-like receptor signaling pathway, inducing further metabolic reprogramming. This metabolic shift, marked by enhanced glycolysis and amplified inflammatory response, is critical for trained immunity. Increased expression of iNOS and CD86 in bone marrow tissue further indicated a stronger immune response, potentially linked to trained immunity activation. The GaCuVan&HACHO-BSA hydrogel demonstrated significant efficacy in preventing recurrent osteomyelitis by reducing bacterial loads in bone marrow and enhancing the immune response during reinfection. The hydrogel also upregulated pro-inflammatory cytokines such as IL-1β, IL-6, IL-17, and TNF-α, which are essential for mounting a robust immune response. These findings support the broad-spectrum protection provided by the hydrogel against both homologous and heterologous bacterial infections, positioning it as a promising strategy for recurrent osteomyelitis treatment.
In addition to immune activation, the hydrogel’s ability to regulate bone repair is noteworthy. The release of Ga and Cu²⁺ ions enhances the immune response and stimulates osteogenic differentiation of BMSC through the Wnt/β-catenin signaling pathway. The upregulation of osteogenic genes, such as Runx-2 and Osterix, further supports bone regeneration, providing a dual benefit of infection control and bone repair.
Interestingly, compared to traditional hydrogels, the GaCuVan&HACHO-BSA hydrogel induces trained immunity through the following unique mechanisms: First, its direct injection into the bone marrow cavity ensures localized stimulation at the primary site for immune memory formation27. Second, the hydrogel uses HA to target pathogen-related molecules to macrophages, facilitating the activation of trained immunity. Third, continuous pathogen capture and the sustained release of immune-modulating factors provide prolonged immune stimulation, further promoting metabolic reprogramming and activation of key signaling pathways involved in trained immunity. These mechanisms differentiate the GaCuVan&HACHO-BSA hydrogel from other hydrogels, enabling it to effectively activate and maintain long-lasting immune memory.
In conclusion, the GaCuVan&HACHO-BSA hydrogel offered a comprehensive solution for treating osteomyelitis by integrating pathogen clearance, trained immunity activation, and bone regeneration, mechanisms that collectively addressed the multifaceted challenges of low local antibiotic efficacy, impaired bone repair, insufficient immune activation, and recurrent infections. Its unique double-network architecture enabled synergistic functions: HACHO-BSA-mediated bacterial capture and macrophage targeting, Ga/Cu²⁺-driven osteogenic differentiation, and sustained Van release, directly tackling the core limitations of conventional therapies. This study established the first hydrogel-based platform to harness trained immunity for osteomyelitis, validated by RNA-seq-defined pathway activation and in vivo protection against homologous/heterologous reinfections. By merging antibacterial chemotherapy with metabolic reprogramming to initiate trained immunity, this approach transcended traditional hydrogel therapies, presenting a paradigm shift for managing drug-resistant bone infections with broad clinical translation potential.
Notably, three key considerations guide future work: First, the hydrogel’s efficacy in metabolically compromised hosts (e.g., diabetic models) remains constrained by impaired trained immunity formation, highlighting the need for combinatorial strategies to mitigate metabolic dysregulation. Second, validating long-term persistence (6 months or longer) in large animal models will be critical for chronic osteomyelitis, where sustained infection control and bone regeneration are paramount. Third, mechanical optimization for large load-bearing applications will expand its utility, as enhancing compressive strength and stability under dynamic stress is key. Additionally, two limitations warrant attention: untested performance in autoimmune-mediated bone pathologies (e.g., rheumatoid arthritis-associated osteomyelitis), where dysregulated immunity may interact unpredictably with its mechanisms; and incomplete mechanistic exploration of trained immunity induction. Specifically, further clarification of the epigenetic reprogramming underlying trained immunity would benefit from the integration of RNA-seq and ATAC-seq, though this line of investigation is more appropriately reserved for future work. Future work will evaluate safety in autoimmune models and integrate RNA-seq, ATAC-seq, single-cell sequencing, and histone modification assays to clarify immune crosstalk, epigenetic memory, and the precise epigenetic reprogramming process. Addressing these will strengthen clinical applicability across diverse patient populations.
Methods
Ethical statement
Animal experiments were ethically approved by the Institutional Animal Care and Use Committee of Shanghai Jiaotong University (protocol number: 2021022401), in accordance with the National Guidelines for the Care and Use of Laboratory Animals in Research Institutions. All male Sprague-Dawley rats were maintained under standard laboratory conditions, including a constant temperature of 25 ± 2 °C, humidity of 50 ± 5%, and housed under specific pathogen-free conditions with a 12-h light/dark cycle. Rats were allowed free access to food and water throughout the study.
Materials for experiments
All raw materials were purchased from commercial suppliers. Glycyrrhizic acid (Ga), vancomycin (Van), N-hydroxysuccinimide (NHS), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), BSA, Copper chloride (CuCl2·2H2O), GelMA, LTA, lipopolysaccharide (LPS), TNF-α, Methylene blue, Nile red, Vanillin, PI, Calcein acetoxymethyl ester (Calcein-AM), DCFH-DA, Dihydroethidium, β-Glycerophosphate, Ascorbic Acid and Dexamethasone from Sigma-Aldrich Co., Ltd. (USA). hyaluronic acid (HA, Mw: 70W-140W) and Sodium Periodate (NaIO4) from Adamas Co., Ltd. (China). CCK-8, Live/Dead Staining Kit, BCIP/NBT Alkaline Phosphatase Color Development Kit, and Alizarin Red Solution from Beyotime Biotechnology Co., Ltd. (China). Fetal bovine serum (FBS), DMEM, PBS, penicillin, and streptomycin from Thermo Fisher Scientific Co., LTD. (USA). ELISA Kits for LPS, TNF-α, IL-6, IL-10, and IL-17 were purchased from Dakewe Bio-engineering Co., LTD. (China). S. aureus (ATCC43300) was obtained from the American Type Culture Collection. Clinical isolates of MRSA A0068 and MRSA RJ0089 were sourced from Ruijin Hospital and confirmed to be MDR (Supplementary Table 2). The primer sequences and antibodies used in this study are listed in Supplementary Tables 3 and 4.
Cells
HUVECs and Raw264.7 cell lines, sourced from the Cell Bank of the Chinese Academy of Sciences in Shanghai, China, were cultured separately in DMEM medium (Gibco) at 37 °C in a humidified incubator with 5% CO2.
BMSCs were isolated from the bone marrow of 4-week-old male Sprague-Dawley rats according to the following protocol. First, the rats were euthanized by cervical dislocation, and their tibias and femurs were carefully excised under sterile conditions. The bones were then flushed with sterile PBS using a 1 mL syringe to collect the bone marrow cells. The marrow was passed through a 70 μm cell strainer to remove any debris, and the cell suspension was centrifuged at 300 × g for 5 min to pellet the cells. The supernatant was discarded, and the cell pellet was resuspended in complete culture medium (DMEM supplemented with 10% FBS and 1% penicillin-streptomycin). The cells were then seeded into a culture dish and incubated at 37 °C with 5% CO2. Non-adherent cells were removed by changing the medium after 24 h. The adherent cells were cultured and passaged when they reached 80–90% confluence.
Synthesis of GaVan, HACHO, and GelMA hydrogel
To prepare GaVan, 20 mg of Ga was dissolved in 20 mL of deionized water, followed by the addition of 8.81 mg of Van dissolved in deionized water at a 4:1 molar ratio. NHS and EDC were then added to the reaction mixture at 1.2 times the molar amount of the reactants. NHS and EDC were dissolved in PBS buffer and slowly added to the Ga and Van solution, followed by stirring at room temperature for 4 h while adjusting the pH to 7.4. Upon completion of the reaction, the product was transferred into a 3500 Da dialysis bag and dialyzed for 24 h to remove unreacted NHS, EDC, and other small molecular impurities. The product was then freeze-dried to obtain the Ga-Van complex.
For the synthesis of HACHO, a 100 mg/mL NaIO₄ solution was slowly added to a 12.5 mg/mL HA solution at a stirring speed of 600 rpm. The reaction was conducted in the dark at room temperature for 24 h. After the reaction, 1.5 mL of ethylene glycol was added to terminate the reaction. The product was then transferred to a 3500 Da dialysis bag and dialyzed in deionized water for 7 days, with water changed twice daily to remove unreacted small molecules. Finally, the product was freeze-dried for 3 days to obtain HACHO, which was stored at −20 °C. Hydroxylamine hydrochloride titration assays determined that the oxidation degree of HACHO stood at 93.2%.
GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels synthesis
Ten milliliters of deionized water was preheated to 60 °C, 400 mg of GaVan and 5 mg of CuCl2 were weighed and dissolved therein, and the incubation was continued at 60 °C to promote rapid dissolution of the components to form the GaCuVan hydrogel. Next, for the preparation of the HACHO-BSA hydrogel, 1 g of HACHO was dissolved in 10 mL of a solution containing 20% BSA. This mixture was thoroughly mixed at 60 °C to obtain the HACHO-BSA hydrogel. To prepare the GaCuVan&HACHO-BSA hydrogel, the precursor A solution (10 mL, 800 mg of GaVan, and 40% BSA) and precursor B solution (10 mL, 2 g of HACHO, and 1 mg/mL of CuCl2) were rapidly mixed in a 1:1 volume ratio.
Characterization of GaCuVan&HACHO-BSA hydrogel
The gelation time of different concentrations of GaVan and Cu²⁺ was measured, while the gelation time of different concentrations of HACHO and BSA was evaluated to determine the optimal formulation for hydrogel formation. The gelation time of GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels was investigated using methylene blue and Nile red staining, and images were taken to capture the rapid gelation process. The microstructure and morphology of GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels were characterized through SEM (JSM-7800F, JEOL). In order to determine the standard concentration profile of GaVan, the absorbance curves of different concentrations of GaVan were plotted using UV-visible spectrophotometry, after the solution was pre-reacted with vanillin-sulfuric acid solution. The release profiles of Ga and Van from GaCuVan&HACHO-BSA hydrogel were monitored by sampling the release medium at specific time points and analyzing it using UV-Vis spectrophotometry. Chemical interactions between different hydrogels were analyzed via FT-IR (Nicolet 6700, USA) spectroscopy. XPS was used to characterize the chemical composition of GaCuVan&HACHO-BSA hydrogel. 1H NMR spectroscopy was performed to analyze the chemical structures of Ga, Van, GaVan, HA, and HACHO. For mass spectrometry analysis, the degradation supernatant of the hydrogel was collected, and the chemical structure of Van in the supernatant was identified using a mass spectrometer. To investigate the targeting effect of HA on CD44⁺ macrophages, FITC and HA-FITC were injected into the bone marrow cavity of rats. Six hours post-injection, flow cytometry was performed to detect FITC fluorescence in CD44⁺ cells. The flow cytometry gating strategy was as follows: first, cell debris and dead cells were excluded on FSC/SSC dot plots to gate intact cells; single cells were isolated using FSC-A/FSC-H gating to eliminate cell aggregates; FITC-negative gates were set based on the group (without HA uptake); CD44⁺ populations were defined with the blank group as the negative control.
Cytocompatibility and ion release profiles of metal ion-containing hydrogels
To evaluate the cytocompatibility and ion release behavior of metal ion-containing hydrogels, GaCuVan&HACHO-BSA, GaMgVan&HACHO-BSA, and GaZnVan&HACHO-BSA hydrogels were prepared using identical component concentrations.
For ion release measurement, hydrogels (1 mL) were incubated in 10 mL of PBS at 37 °C with gentle agitation (50 rpm). After 6 days of degradation, the supernatant (leachate) was collected and centrifuged (3000 × g, 10 min) to remove particulates. The concentrations of Cu²⁺, Mg²⁺, and Zn²⁺ in the leachates were quantified using ICP-OES (Thermo Scientific iCAP 7400) with calibration curves generated from standard solutions (0.1–100 ppm).
For cytocompatibility assessment, BMSC (5 × 104 cells/well in 24-well plates) were cultured with 6-day hydrogel leachates (1:1 dilution with medium) for 24 h. Live/dead staining was performed using calcein-AM and PI, with fluorescence images captured to evaluate cell viability (n = 4 independent experiments).
Plasticity, injectability, and self-healing of GaCuVan&HACHO-BSA hydrogel
To evaluate the plasticity of the hydrogel, the hydrogel was poured into the pentagram and circle models and removed with tweezers after gel formation to observe its shape. To evaluate the injectability, the hydrogels were injected with a 1 mL syringe, and different letters were injected as needed. The self-healing properties of the hydrogels were tested by pre-staining the hydrogels with two types of stains, methylene blue and Nile red. Then, both hydrogels were cut and allowed to heal for a period of time at room temperature after bonding. The healing efficiency of the repaired hydrogel was evaluated based on the restoration of mechanical strength and visual appearance of the repaired hydrogel.
Mechanical testing of GaCuVan&HACHO-BSA hydrogel
Rheological evaluation was performed on cylindrical hydrogel samples (diameter: 10 mm) using a stress-controlled Discovery DHR-2 rheometer (TA Instruments, USA) with a frequency sweep at room temperature, covering an angular frequency range of 0.1–100 rad/s, and the hydrogels were divided into GaCuVan, HACHO-BSA, GaCuVan&HACHO-BSA groups. Cylindrical GaCuVan&HACHO-BSA hydrogel samples (8 mm in diameter and 10 mm in height) were placed on the sample stage of a universal testing machine (Instron 4202, USA) with a 200 N load cell, and compression tests were performed at an ultimate strain fixed at 80%.
Degradation of hydrogels
For in vitro degradation assessment, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogels (1 mL each) were prepared in glass vials and cross-linked for 4 h at room temperature to ensure structural stability. Degradation was initiated immediately after this cross-linking period by adding 10 mL of PBS as the degradation medium. At predetermined intervals, the medium was fully aspirated, and hydrogels were gently blotted dry to measure residual weight (n = 4 per group per time point). The mass remaining ratio was calculated as:
Degradation was set to begin immediately following the 4-h cross-linking period. Time-course imaging was performed exclusively for GaCuVan&HACHO-BSA hydrogels, which were photographed at each time point using a digital camera under standardized lighting conditions to visually document structural changes. To evaluate the impact of lysozyme—an enzyme abundant in infected microenvironments—on hydrogel stability, GaCuVan&HACHO-BSA hydrogels (1 mL) were subjected to the same protocol but using PBS containing 100 μg/mL lysozyme (physiological concentration) as the degradation medium, with measurements taken at identical intervals (n = 4 per time point). All in vitro degradation experiments were conducted at 37 °C with gentle agitation (50 rpm) to simulate physiological conditions.
For in vivo degradation assessment, pre-formed hydrogel discs (6 mm diameter, 2 mm thickness) were prepared by casting each hydrogel (GaCuVan, HACHO-BSA, GaCuVan&HACHO-BSA) into cylindrical molds and allowing them to cross-link for 4 h. Male Sprague-Dawley rats (250–300 g, n = 4 per group) were anesthetized, and the dorsal skin was sterilized. Subcutaneous pockets (1 × 1 cm) were created bilaterally, and one pre-formed hydrogel disc was implanted into each pocket. At 2, 4, 8, 12, 16, and 20 days post-implantation, rats were euthanized (n = 4 per time point per group), and residual hydrogel mass was retrieved, gently washed with PBS to remove surrounding tissues, blotted dry, and weighed. The degradation ratio was calculated using the same formula as in vitro.
Molecular recognition studies between multiple molecules in the hydrogel
The amino acid sequences of the two protein molecules were downloaded from the NCBI database, and the structures were modeled from scratch using I-TASSER software. Meanwhile, GaVan and HACHO were used as the initial structures and processed with AutoDock Tools 1.5.6 to preserve the original charges of the proteins. In this study, a total of three docking steps were required: first, Cu2+ was docked as a ligand with GaVan, and HACHO molecules were used as ligands for the second docking step. Subsequently, the GaCuVan&HACHO complex molecules obtained from the previous two docking steps were used as ligands in the third step using BSA docking. All ligand molecules in the process were used to optimize the molecular structure and calculate the PM3 atomic charge for subsequent molecular docking using the MOPAC program, and the ligand structure was processed using AutoDock Tools 1.5.6 to generate a pdbqt file for docking. Global molecular docking was performed in this study using both molecular docking methods. Molecular docking was implemented using the AutoDock 4.2.6 software package, with the center coordinates of the docking box set to encompass the entire receptor structure in its entirety, the number of lattice points in each direction of XYZ set to 100 × 100 × 100, the number of docking times set to 50, and the rest of the parameters used as default values. The result of molecular docking may have inappropriate atomic contacts in the spatial structure, and energy optimization can be used to release these forces into a more stable structure.
Preparation and characterization of GelMA hydrogel
Prepared by dissolving 3 g GelMA (15% w/v) in 20 mL PBS at 50 °C, followed by 0.1 g lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP, 0.5% w/v) under dark stirring, the solution (1 mL) was UV-cross-linked (365 nm, 30 W/cm2, 10 min) in a cubic mold (1 cm3) and washed with PBS to remove unreacted initiators. The microstructure and morphology of GelMA hydrogel were characterized through SEM (JSM-7800F, JEOL).
Adsorption of pathogen-associated molecules in GaCuVan&HACHO-BSA hydrogel
MRSA/S. aureus (1 × 106 CFU/mL) and different hydrogels (GelMA, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA groups) were co-cultured in LB broth and placed in a bacteria shaker at 37 °C for 30 min or 12 h. The supernatant and hydrogels were separated, and the separated hydrogels were washed with PBS and agitated for 1 min. Subsequently, the bacteria on the hydrogels were separated by ultrasonic shaking (1 W/cm2) and resuspended in the same volume of PBS. Standard plate plating, CFU enumeration, and SYTOTM 9/PI staining were performed. DMEM medium containing LTA (200 μg/mL), LPS (300 ng/mL), and TNF-α (300 ng/mL), and different hydrogels were co-cultured and incubated in a water bath at 37 °C for 30 min or 12 h. Then, UV-Vis and ELISA were performed to detect changes in LTA, LPS, and TNF-α concentrations in the supernatants. Hydrogel samples were formed by adding 10%, 20%, and 30% BSA or adding MRSA/S. aureus to GaCuVan&HACHO-BSA hydrogel, and their compression properties were tested using a universal testing machine.
Biocompatibility assessment
For the Live/Dead staining assay, BMSC/HUVECs were treated with GaVan, GaCuVan, HACHO-BSA, GaCuVan&HACHO-BSA hydrogel groups, and incubated for 1, 3, and 5 days. After the incubation period, the cells were washed with PBS to remove excess media. Then, the cells were stained with the Live/Dead Staining Kit, following the manufacturer’s protocol. The staining solution consisted of calcein-AM (green fluorescence for live cells) and PI (red fluorescence for dead cells), which were applied to the cells and incubated for 30 min at 37 °C. After incubation, the cells were washed again with PBS, and fluorescence images were captured using CLSM (LSM800, Zeiss). The proportion of live and dead cells was quantified by analyzing the fluorescence intensity using ImageJ software. For cell viability and proliferation, BMSC/HUVECs were cultured under the same conditions. The CCK-8 assay was performed to evaluate cell viability. At each time point, 10 μL CCK-8 solution was added to each well and incubated at 37 °C for 1 h. Absorbance at 450 nm was measured using a microplate reader (Infinite F50, Tecan, Switzerland). To isolate pure RBCs, 1 mL of rat blood was collected and centrifuged at 2000 rpm for 15 min. The resulting pellet was washed five times with saline to remove any residual components. Next, a 4% RBCs solution was prepared and incubated with different types of hydrogels, water, or PBS at 37 °C for 4 h. After incubation, the samples underwent a second centrifugation, and the supernatant was collected. The absorbance of the supernatant at 542 nm was measured using a microplate reader. The hemolysis rate was then calculated using the following formula:
Antibacterial test and bacterial morphology
The antibacterial and inhibitory effects of Van and different hydrogels (GaVan, GaCuVan, HACHO-BSA, GaCuVan&HACHO-BSA) against MRSA and S. aureus were evaluated using the standard plate count method, Live/dead staining, and growth curve analysis. For the standard plate count, each hydrogel type was mixed with a bacterial suspension (1 × 106 CFU/mL) containing LB broth and incubated at 37 °C for 12 h. After incubation, the suspension was serially diluted and plated on LB agar plates, followed by incubation at 37 °C for another 12 h, and CFU counts were evaluated. For Live/dead staining, the bacterial suspension (1 × 106 CFU/mL) was incubated with each hydrogel for 12 h at 37 °C. Bacteria were then stained with SYTO™ 9/PI for 15 min and observed under a fluorescence microscope. The percentage of dead bacteria was calculated using ImageJ software. In addition, bacteria and hydrogels were co-incubated for 12 h, fixed with glutaraldehyde for 2 h, and dehydrated through a gradient of ethanol solutions. The samples were then air-dried, and bacterial morphology was observed by SEM. After co-incubation of bacteria with the different hydrogels for 12 h at 37 °C, the samples were fixed with 2.5% glutaraldehyde for 2 h. After fixation, the samples were washed, dehydrated through an ethanol gradient, and embedded in resin. Ultrathin sections were prepared and observed under TEM to examine bacterial morphology. For growth curve analysis, bacteria (5 × 105 CFU/mL) were co-incubated with each hydrogel type at 37 °C for 24 h. Absorbance at 600 nm was measured using a microplate reader at appropriate time intervals.
ELISA assay
After co-incubation of different hydrogels (GelMA, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA) with DMEM medium containing LPS (300 ng/mL) or TNF-α (300 ng/mL) for 30 min or 12 h at 37 °C, the concentration of remaining LPS or TNF-α in the supernatant was measured under the guidance of the manufacturer’s instructions by corresponding ELISA Kits. The concentrations of LPS or TNF-α adsorbed by each group of hydrogels were calculated with the following formula: LPS/TNF-α (ng/mL) = (A0−A1) ng/mL. A0 is the initial concentration of LPS or TNF-α in the medium. A1 is the concentration of LPS orTNF-α remaining in the supernatant after co-incubation with different hydrogels. Cytokine levels of TNF-α and IL-10 in the culture supernatant of RAW264.7 co-cultured with different hydrogels were detected using the corresponding ELISA kits.
Antioxidant efficiency of the GaCuVan&HACHO-BSA hydrogel
To evaluate the antioxidant capacity of the different hydrogels, Raw264.7 cells were co-cultured with each hydrogel type for 12 h. First, Raw264.7 were seeded in 24-well plates at a density of 1 × 105 cells per well and allowed to adhere overnight under standard culture conditions (37 °C, 5% CO₂). The different hydrogels were then introduced to the cells and incubated for 24 h to facilitate interaction. For assessment with the DCFH-DA probe, cells were first treated with 10 μM DCFH-DA for 30 min at 37 °C. DCFH-DA diffuses into cells and is deacetylated by cellular esterases to non-fluorescent DCFH, which is subsequently oxidized by ROS to the highly fluorescent dichlorofluorescein (DCF). After incubation, the fluorescence intensity was measured using a fluorescence microplate reader, providing an indication of intracellular ROS levels. In parallel, the Dihydroethidium assay was used to specifically detect superoxide anion radicals. After the 24-h co-culture period, cells were incubated with 5 μM dihydroethidium for 30 min at 37 °C. Dihydroethidium permeates cell membranes and reacts with superoxide to form the fluorescent product ethidium, which intercalates with DNA and emits red fluorescence upon excitation. Fluorescence was quantified using a fluorescence microscope and analyzed using ImageJ software to determine relative levels of superoxide production.
ALP staining and Alizarin Red S (ARS) staining
To evaluate the osteogenic differentiation of BMSC cultured with different hydrogels, ALP staining and ARS staining were performed at different time points. For ALP staining, BMSC at a density of 2 × 105 cells per well in 6-well plates were incubated with the different hydrogels in osteogenic differentiation medium (DMEM containing 10% FBS, 10 nM dexamethasone, 10 nM β-glycerophosphate, and 300 μM ascorbic acid) for 7 and 14 days. After the incubation period, the cells were washed twice with PBS and fixed with 4% paraformaldehyde for 30 min at room temperature. ALP activity was then detected using the BCIP/NBT Alkaline Phosphatase Color Development Kit according to the manufacturer’s instructions. Cells were incubated with the ALP staining solution for 30 min at 37 °C, and positive ALP activity was visualized as blue/purple staining under a light microscope. For ARS staining, BMSC co-cultured with hydrogels were incubated in osteogenic medium for 14 and 21 days. After incubation, the cells were washed twice with PBS and fixed in 4% paraformaldehyde for 30 min at room temperature. To detect mineralization, the fixed cells were stained with 1% Alizarin Red S solution (pH 4.2) for 30 min at room temperature. The staining solution was then removed, and the cells were extensively washed with distilled water to remove excess dye. Mineralized nodules, indicated by red staining, were observed under a light microscope. For quantification, the Alizarin Red S stain was eluted with 10% cetylpyridinium chloride (CPC) for 30 min, and the absorbance at 562 nm was measured using a microplate reader to assess the degree of mineralization.
Quantification of mRNA and the quantitative qRT-PCR
Total RNA was extracted from cells using TRIzol reagent (Invitrogen, Carlsbad, CA) following the manufacturer’s instructions. One microgram of the isolated RNA was used for complementary DNA synthesis with the RevertAid First-Strand cDNA Synthesis Kit (TaKaRa, Dalian, China). The cDNA was then amplified using qRT-PCR, which was performed on the ABI 7500 Sequencing Detection System (Applied Biosystems, USA), utilizing the SYBR Premix Ex Tag Kit (TaKaRa, Dalian, China). Primer sequences employed in this study are provided in Supplementary Table 3.
Immunofluorescence staining
The Raw264.7 and BMSC were initially fixed with 4% paraformaldehyde for 15 min, then treated with 0.1% Triton X-100 for 10 min. After fixation, cells were incubated with 3% BSA in PBS for 30 min to block non-specific binding. Following this, the cells were incubated overnight at 4 °C with mouse primary antibodies: anti-CD86 (1:200; #24595, Cell Signaling Technology), anti-CD206 (1:200; #91882, Cell Signaling Technology), and anti-Runx-2 (1:200; 20700-1-AP, Proteintech). Goat Anti-Rabbit IgG H&L (Alexa Fluor® 488) and Goat Anti-Rabbit IgG H&L (Alexa Fluor® 647) were applied at room temperature for 1 h. To counterstain, 4′,6-diamidino-2-phenylindole (DAPI) was used for the nucleus and Alexa Fluor 594 phalloidin (Life Technologies, USA) for the cytoskeleton. The immunofluorescence images were captured using an LSCM (LSM800, Zeiss, Germany) and analyzed with ImageJ to quantify the relative fluorescence intensity of CD86, CD206, and Runx-2.
Western blotting analysis
Proteins were extracted from BMSC cell lysates and separated using 7.5 to 12.5% SDS-polyacrylamide gel electrophoresis, followed by transfer to a polyvinylidene difluoride membrane (0.45 μm; Millipore, Bedford, MA, USA). To block non-specific binding, the membrane was incubated with 5% nonfat dry milk in tris-buffered saline containing Tween-20 for 30 min. The membrane was then incubated at 4 °C overnight with primary antibodies targeting various proteins, including anti-COL1A1 (1:1000; 66761-1-Ig, Proteintech), anti-Runx-2 (1:1000; 20700-1-AP, Proteintech), anti-OPN (1:1000; 22952-1-AP, Proteintech), anti-BMP2 (1:1000; 66383-1-Ig, Proteintech), and anti-GAPDH (1:1000; ab8245, Abcam). Afterward, the membrane was incubated with the appropriate horseradish peroxidase-conjugated secondary antibody (1:2000; ab6721 or ab6728, Abcam) for 1 h. Antibody detection was performed using the Enhanced Chemiluminescence Western Blot System (Thermo Fisher Scientific, Pierce, Rockford, IL, USA).
Biocompatibility assessment methods
For in vivo biocompatibility evaluation, 40 male Sprague-Dawley rats (6 weeks old, 200–250 g) were randomly divided into 5 groups (n = 8 per group): Sham (tibia injected with PBS), GaVan, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogel groups. Hydrogels (200 μL) were injected into the tibial medullary cavity of anesthetized rats, with the Sham group receiving an equal volume of PBS.
At 7 and 28 days post-injection, blood samples (1 mL) were collected from the retro-orbital sinus of 4 rats per group under anesthesia. Hematological analyses (hemoglobin concentration and platelet count) were performed using an automated hematology analyzer (Sysmex XN-1000). Serum was separated by centrifugation (3000 × g, 10 min, 4 °C) for further assessments: liver function indices (ALT, AST, γ-GT) and renal function markers (BUN, CREA) were quantified via commercial kits (Nanjing Jiancheng Bioengineering Institute) following the manufacturer’s protocols; additionally, serum levels of IL-6 and TNF-α were measured using ELISA kits according to the manufacturer’s instructions to evaluate systemic inflammatory responses.
At the same time points, rats were euthanized, and major organs (heart, liver, spleen, lung, and kidney) were harvested, fixed in 4% paraformaldehyde for 24 h, embedded in paraffin, and sectioned (5 μm). Sections were stained with H&E and examined under a light microscope to evaluate histopathological changes (e.g., tissue damage, inflammatory infiltration, or structural abnormalities).
Establishment of rat osteomyelitis model and in situ GaCuVan&HACHO-BSA hydrogel delivery
The rat osteomyelitis model was established by inoculating 6-week-old male Sprague-Dawley rats (200–250 g) with 1 × 106 CFU of MRSA into the tibial bone marrow cavity. Prior to inoculation, the rats were anesthetized using 3% isoflurane, and the tibial region was sterilized with iodine to prevent contamination. A 1-cm incision was made on the anteromedial aspect of the right tibia to expose the bone surface, followed by drilling a 2-mm diameter defect using a medical drill. Through this defect, 200 µL of MRSA suspension (1 × 106 CFU mL−1) was injected into the marrow cavity, except for the sham group, which received saline. The defect was sealed with bone wax, and the surgical site was sutured. Rats were housed under standard conditions, monitored for infection signs (redness, swelling, mobility limitation), and body weight/temperature were recorded daily.
For in situ hydrogel delivery, 200 µL of GaVan, GaCuVan, HACHO-BSA, and GaCuVan&HACHO-BSA hydrogel, or CS-GP thermosensitive hydrogel loaded with Van (Van/Gel, positive control46) were injected into the tibial marrow cavity 1 day post-MRSA inoculation. The Van group received intramedullary injection of Van solution (10 mg/kg, 200 µL), while the positive control and sham groups received saline. PMMA@Van pellets were filled into the bone marrow cavity according to a previously reported method47. Post-injection, rats were monitored for adverse effects, and blood counts were analyzed at days 7 and 28.
Antibacterial effects of GaCuVan&HACHO-BSA hydrogel in vivo
The IVIS system (PerkinElmer) was used to record MRSA bioluminescence in the tibia of rats with osteomyelitis on days 1, 3, and 7 and to assess the severity of infection. Rats on days 7, 14, and 28 of MRSA infection were also harvested for tibial bone marrow. 0.1 mL of the bone marrow dilution was applied to LB culture plates. Plates were incubated at 37 °C for 24 h for colony documentation and CFU count.
Histological examination
On days 14 and 28, the rats were euthanized, and their tibiae were harvested. Immediately following collection, the tibiae were fixed in 4% paraformaldehyde and decalcified in EDTA for 4 weeks. After decalcification, the tissues underwent dehydration through a graded ethanol series. Subsequently, the dehydrated tissues were embedded in paraffin, sectioned, and stained. H&E and Wright-Giemsa staining were carried out to assess inflammation and bacterial infiltration associated with osteomyelitis. All semi-quantitative analyses were conducted using ImageJ software.
Micro-CT imaging and histopathologic detection of bone defects in osteomyelitis
Rats were executed at 4 and 8 weeks after different hydrogels (GaVan, GaCuVan, and GaCuVan&HACHO-BSA groups) administration, and tibiae were collected. Bone specimens were fixed in 4% paraformaldehyde for 24 h and then scanned using the micro-CT system (Skyscan 1172, Aartselaar, Belgium) at 15-µm voxel size. The acquired 3D images were reconstructed and analyzed to assess BMD, BV/TV, Tb.N, and Tb.Sp. These parameters were quantitatively analyzed using micro-CT software (CTAn, Bruker) to assess the extent of bone repair and osseointegration. For histopathologic examination, tibiae were decalcified in EDTA solution for 4 weeks and then embedded in paraffin. Tissue sections (5 μm thick) were stained with H&E to visualize the general morphology of the tissues and evaluated for collagen deposition and bone formation using Masson staining. In addition, immunohistochemical staining was performed to assess the expression of COL1A1. Runx-2 immunofluorescence staining was used to assess osteogenic differentiation. Histologic and immunohistochemical analyses were performed in a blinded fashion to ensure fair and accurate interpretation of the bone repair process.
Modeling of ectopic osteomyelitis in rats
Prior to infection, male Sprague-Dawley rats (6 weeks old, 200–250 g) were randomized into six groups: G1, Sham; G2, PBS; G3, Van; G4, GaVan; G5, GaCuVan; G6, GaCuVan&HACHO-BSA. The in situ osteomyelitis rat model was established as described above and pre-delivered with different hydrogels. To establish the ectopic osteomyelitis model, MRSA suspension (1 × 106 CFU/mL MRSA) was injected into the bone marrow cavity of the contralateral tibia to induce osteomyelitis. Infection was confirmed by monitoring clinical signs such as redness and swelling at the injection site, and then samples were collected at the indicated time points (days 7 and 14).
In vivo investigation of the induction of trained immunity
Bone marrow tissues from rats on day 14 of infection were subjected to RNA-seq analysis to investigate changes in gene expression associated with osteogenesis and immune responses. Total RNA was extracted from tibial bone marrow tissues of rats with osteomyelitis using TRIzol® reagent (Invitrogen, USA). The A260/A280 absorbance ratio of RNA samples was measured using Nanodrop ND-2000 (Thermo Scientific, USA), and the RIN values were determined using Agilent Bioanalyzer 4150 (Agilent Technologies, CA, USA) to determine the RIN value of RNA. PE libraries were prepared according to the instructions of the ABclonal mRNA-seq Lib Prep Kit (ABclonal, China). Finally, sequencing was performed on the Illumina Novaseq 6000/MGISEQ-T7 sequencing platform. GO functional enrichment of differential genes and KEGG pathway enrichment analysis were performed using the clusterProfiler R software package. Conventional enrichment analysis based on the hypergeometric distribution relies on significantly up- or down-regulated genes and tends to miss some genes that are not significantly differentially expressed but are biologically important. GSEA does not require the specification of an explicit differential gene threshold; all genes are ranked according to the degree of differential expression in the two sample sets, and then a statistical method is used to test whether a predetermined set of genes is enriched at the top or bottom of the ranked list.
For histopathological analysis, tibiae were decalcified in EDTA solution for 4 weeks and embedded in paraffin. Tissue sections (5 μm thick) were subjected to H&E staining to visualize NEUT infiltration in the contralateral bone marrow cavity and Wright-Giemsa staining to assess bacterial load in the bone marrow. Bacterial growth in the exudate was further evaluated by bacterial plate culture and CFU count. Immunofluorescence staining was performed to assess the expression of HIF-1α and COX2, markers of metabolic reprogramming and inflammation, respectively, as well as iNOS, a marker of M1 macrophage polarization. Immunohistochemical staining of CD86 was conducted to evaluate M1 macrophage polarization in the bone marrow. Additionally, ELISA was used to measure the production of lactate, ATP, and succinate, and the levels of IL-17, IL-6, and TNF-α in both bone marrow tissue and serum. All histological and immunohistochemical analyses were carried out in a blinded manner to ensure unbiased evaluation of trained immunity activation.
To investigate the effect of glycolysis inhibitor on GaCuVan&HACHO-BSA hydrogel-induced trained immunity, osteomyelitis rats were treated with in situ injection of GaCuVan&HACHO-BSA hydrogel following our established protocol. Three weeks post-treatment, the rats were reinfected with MRSA at the same site and randomized into two groups based on intraperitoneal injection of 2-DG (2 mg/g): the GaCuVan&HACHO-BSA group and the GaCuVan&HACHO-BSA + 2-DG group. On day 14 after MRSA reinfection, rats were euthanized, and their tibias were harvested. Bacterial burden was evaluated using standard plate counting and CFU assays. Additionally, the levels of IL-6 and TNF-α in bone marrow tissue were measured by ELISA.
Study on the non-specific protective immunity provided by GaCuVan&HACHO-BSA hydrogel in rats with recurrent osteomyelitis
Male Sprague-Dawley rats (6 weeks old, 200–250 g) were used to model recurrent osteomyelitis and to evaluate the effect of pre-delivering with hydrogel. The rats were randomized into six groups: G1, Sham; G2, PBS; G3, Van; G4, GaVan; G5, GaCuVan; G6, GaCuVan&HACHO-BSA. Administration was performed 3 weeks prior to the induction of osteomyelitis to allow sufficient time for activation of the immune response, and bone marrow cleaning and antibiotic drainage were performed prior to secondary infection with MRSA. To induce recurrent osteomyelitis, 1 × 106 CFU/mL MRSA suspension was used for infection at the same site of in situ osteomyelitis. At the indicated time points (days 7, 14, and 21 after recurrent infection), the rats were euthanized, and their tibiae were harvested for further analysis using the IVIS system and bacteriologic tests (standard plate counts and CFU assays) to assess the recurrence of osteomyelitis.
To investigate the cross-protective effect of the hydrogel through trained immunity, bacterial loads of MRSA RJ0089 and S. aureus were assessed using standard plate counts and CFU assays. For rats infected with S. aureus to model recurrent osteomyelitis, bone marrow tissue sections were subjected to H&E staining to visualize NEUT infiltration in the marrow cavity and Wright-Giemsa staining to assess bacterial load. Additionally, immunofluorescence staining was performed to assess the expression of IL-1β and TNF-α, markers indicative of trained immunity induction, in the bone marrow tissues of these rats.
Evaluation of GaCuVan&HACHO-BSA hydrogel in clinically relevant osteomyelitis models
To assess clinical translatability, two preclinical models were established: diabetic and aged rat MRSA osteomyelitis, with experimental groups including Sham, PBS, Van, GaVan, GaCuVan, and GaCuVan&HACHO-BSA.
For the diabetic model, 6-week-old Sprague-Dawley rats (n = 48, 200–250 g)were rendered diabetic via intraperitoneal injection of streptozotocin (STZ, 50 mg/kg). Diabetes was confirmed by fasting blood glucose ≥16.7 mmol/L 72 h post-injection. MRSA osteomyelitis was induced by tibial inoculation of 1 × 106 CFU/mL MRSA suspension, followed by immediate administration of treatments. At 14 days post-infection, primary infection clearance was evaluated by harvesting tibial bone marrow for bacterial quantification. For heterotopic reinfection assays, rats with cleared primary infections underwent contralateral tibial inoculation with the same MRSA suspension at day 14, with bacterial burden assessed 14 days later via standard plate counting and CFU assays.
In the aged model, 18-month-old Sprague-Dawley rats (n = 48, 540–600 g) were subjected to tibial MRSA inoculation (1 × 106 CFU/mL) to induce primary osteomyelitis, followed by treatment administration. Primary infection clearance was evaluated at 14 days via bone marrow CFU counting. For heterotopic reinfection, MRSA was inoculated into the contralateral tibia 14 days after primary treatment, with bacterial loads quantified 14 days post-reinfection using plate counting and CFU assays.
Statistical analysis
The statistical significance of all comparative studies in this research was analyzed using GraphPad Prism (version 9.50). For parametric tests, normality of data distribution was assumed without formal testing. One-way ANOVA was employed for group comparisons, followed by Tukey’s multiple comparison test. A P value of <0.05 was considered statistically significant for all analyses.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The primary data supporting the findings of this study can be found within the paper and its SI file. The data for Figs. 1–10, Supplementary Figs. 1–54, and Supplementary Tables 1–4 are included in the Source data file accompanying this paper. Any further requests for information should be directed to the corresponding authors, who will provide the necessary details. The raw sequence data have been submitted to the NCBI Short Read Archive (SRA) with bioproject accession number PRJNA1380635. Source data are provided with this paper.
References
Masters, E. A. et al. Skeletal infections: microbial pathogenesis, immunity and clinical management. Nat. Rev. Microbiol. 20, 385–400 (2022).
Flemming, H. C. et al. Biofilms: an emergent form of bacterial life. Nat. Rev. Microbiol. 14, 563–575 (2016).
Kavanagh, N. et al. Staphylococcal osteomyelitis: disease progression, treatment challenges, and future directions. Clin. Microbiol. Rev. 31, https://doi.org/10.1128/cmr.00084-00017 (2018).
Moriarty, T. F. et al. Fracture-related infection. Nat. Rev. Dis. Prim. 8, 67 (2022).
Cheng, Y. et al. Guanidinium-decorated nanostructure for precision sonodynamic-catalytic therapy of MRSA-infected osteomyelitis. Adv. Mater. 34, e2206646 (2022).
Turner, N. A. et al. Methicillin-resistant Staphylococcus aureus: an overview of basic and clinical research. Nat. Rev. Microbiol. 17, 203–218 (2019).
Ma, L. et al. A Janus-ROS healing system promoting infectious bone regeneration via sono-epigenetic modulation. Adv. Mater. 36, e2307846 (2024).
Qiao, Y. et al. Treatment of MRSA-infected osteomyelitis using bacterial capturing, magnetically targeted composites with microwave-assisted bacterial killing. Nat. Commun. 11, 4446 (2020).
Netea, M. G. et al. Defining trained immunity and its role in health and disease. Nat. Rev. Immunol. 20, 375–388 (2020).
Ochando, J., Mulder, W. J. M., Madsen, J. C., Netea, M. G. & Duivenvoorden, R. Trained immunity—basic concepts and contributions to immunopathology. Nat. Rev. Nephrol. 19, 23–37 (2023).
Netea, M. G. et al. Trained immunity: a program of innate immune memory in health and disease. Science 352, aaf1098 (2016).
Domínguez-Andrés, J. et al. Trained immunity: adaptation within innate immune mechanisms. Physiol. Rev. 103, 313–346 (2023).
Ding, C. et al. Inducing trained immunity in pro-metastatic macrophages to control tumor metastasis. Nat. Immunol. 24, 239–254 (2023).
Chaumond, E. et al. Development of innate immune memory by non-immune cells during Staphylococcus aureus infection depends on reactive oxygen species. Front. Immunol. 14, 1138539 (2023).
Wang, F. et al. In situ vaccination with an injectable nucleic acid hydrogel for synergistic cancer immunotherapy. Angew. Chem. Int. Ed. 63, e202315282 (2024).
Liang, X. et al. A spontaneous multifunctional hydrogel vaccine amplifies the innate immune response to launch a powerful antitumor adaptive immune response. Theranostics 11, 6936–6949 (2021).
Fu, W. et al. Injectable hydrogel mucosal vaccine elicits protective immunity against respiratory viruses. ACS Nano 18, 11200–11216 (2024).
Zheng, B. et al. Sendai virus-based immunoadjuvant in hydrogel vaccine intensity-modulated dendritic cells activation for suppressing tumorigenesis. Bioact. Mater. 6, 3879–3891 (2021).
Ji, P. et al. Modular hydrogel vaccine for programmable and coordinate elicitation of cancer immunotherapy. Adv. Sci. 10, e2301789 (2023).
Kim, S. W. et al. Glycyrrhizic acid affords robust neuroprotection in the postischemic brain via anti-inflammatory effect by inhibiting HMGB1 phosphorylation and secretion. Neurobiol. Dis. 46, 147–156 (2012).
Qian, Y. et al. Immunoregulation in diabetic wound repair with a photoenhanced glycyrrhizic acid hydrogel scaffold. Adv. Mater. 34, e2200521 (2022).
Pi, W. et al. A metal ions-mediated natural small molecules carrier-free injectable hydrogel achieving laser-mediated photo-Fenton-like anticancer therapy by synergy apoptosis/cuproptosis/anti-inflammation. Bioact. Mater. 29, 98–115 (2023).
Li, S. et al. Ultrasound-activated probiotics vesicles coating for titanium implant infections through bacterial cuproptosis-like death and immunoregulation. Adv. Mater. 36, e2405953 (2024).
Xu, H. et al. Glycyrrhizic acid alters the hyperoxidative stress-induced differentiation commitment of MSCs by activating the Wnt/β-catenin pathway to prevent SONFH. Food Funct. 14, 946–960 (2023).
Xue, W. et al. A dual-network nerve adhesive with enhanced adhesion strength promotes transected peripheral nerve repair. Adv. Funct. Mater. 33, 2209971 (2023).
Kim, Y. S. et al. Chondrogenesis of cocultures of mesenchymal stem cells and articular chondrocytes in poly(l-lysine)-loaded hydrogels. J. Control. Release 328, 710–721 (2020).
Mitroulis, I. et al. Modulation of myelopoiesis progenitors is an integral component of trained immunity. Cell 172, 147–161.e112 (2018).
Fu, Y. J. et al. A gas therapy strategy for intestinal flora regulation and colitis treatment by nanogel-based multistage NO delivery microcapsules. Adv. Mater. 36, e2309972 (2024).
Shinn, J. et al. Antioxidative hyaluronic acid–bilirubin nanomedicine targeting activated hepatic stellate cells for anti-hepatic-fibrosis therapy. ACS Nano 18, 4704–4716 (2024).
Kargozar, S., Montazerian, M., Hamzehlou, S., Kim, H. W. & Baino, F. Mesoporous bioactive glasses: promising platforms for antibacterial strategies. Acta Biomater. 81, 1–19 (2018).
Wu, C. et al. Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomaterials 34, 422–433 (2013).
Wang, Y., Zhang, W. & Yao, Q. Copper-based biomaterials for bone and cartilage tissue engineering. J. Orthop. Transl. 29, 60–71 (2021).
Yu, H. et al. Hydrogel microspheres for stem cell recruitment and induction of directed differentiation in osteoarthritis therapy. Chem. Eng. J. 497, 154798 (2024).
Tang, H. et al. Zinc-cobalt bimetallic organic frameworks with antioxidative and osteogenic activities for periodontitis treatment. Small 21, e2412065 (2025).
Bai, L. et al. Multifunctional scaffold comprising metal-organic framework, hydrogel, and demineralized bone matrix for the treatment of steroid-induced femoral head necrosis. Small 21, e2407758 (2025).
Ferraboschi, P., Ciceri, S. & Grisenti, P. Applications of lysozyme, an innate immune defense factor, as an alternative antibiotic. Antibiotics 10, 1534 (2021).
Markart, P., Korfhagen, T. R., Weaver, T. E. & Akinbi, H. T. Mouse lysozyme M is important in pulmonary host defense against Klebsiella pneumoniae infection. Am. J. Respir. Crit. Care Med. 169, 454–458 (2004).
Zhao, C. et al. Molecular co-assembled strategy tuning protein conformation for cartilage regeneration. Nat. Commun. 15, 1488 (2024).
Dsouza, A. et al. Multifunctional composite hydrogels for bacterial capture, growth/elimination, and sensing applications. ACS Appl. Mater. Interfaces 14, 47323–47344 (2022).
Eming, S. A., Wynn, T. A. & Martin, P. Inflammation and metabolism in tissue repair and regeneration. Science 356, 1026–1030 (2017).
Chen, H. et al. Urchin-like ceria nanoparticles for enhanced gene therapy of osteoarthritis. Sci. Adv. 9, eadf0988 (2023).
Shapouri-Moghaddam, A. et al. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 233, 6425–6440 (2018).
Yang, N. et al. Magnesium implants with alternating magnetic field-enhanced hydrogen release and proton depletion for anti-infection treatment and tissue repair. Bioact. Mater. 38, 374–383 (2024).
Chen, H. et al. MicroRNA-224-5p nanoparticles balance homeostasis via inhibiting cartilage degeneration and synovial inflammation for synergistic alleviation of osteoarthritis. Acta Biomater. 167, 401–415 (2023).
Zhang, X. et al. A sonosensitive diphenylalanine-based broad-spectrum antimicrobial peptide. Nat. Biomed. Eng. 9, 1349–1365 (2025).
Tao, J. et al. Injectable chitosan-based thermosensitive hydrogel/nanoparticle-loaded system for local delivery of vancomycin in the treatment of osteomyelitis. Int. J. Nanomed. 15, 5855–5871 (2020).
Wu, Y. et al. Peptide polymer-doped cement acting as an effective treatment of MRSA-infected chronic osteomyelitis. Adv. Funct. Mater. 32, 2107942 (2022).
Webb, J. C. & Spencer, R. F. The role of polymethylmethacrylate bone cement in modern orthopaedic surgery. J. Bone Jt. Surg. 89, 851–857 (2007).
Hope, P. G., Kristinsson, K. G., Norman, P. & Elson, R. A. Deep infection of cemented total hip arthroplasties caused by coagulase-negative staphylococci. J. Bone Jt. Surg. 71, 851–855 (1989).
Moerings, B. G. J. et al. Dectin-1b activation by arabinoxylans induces trained immunity in human monocyte-derived macrophages. Int. J. Biol. Macromol. 209, 942–950 (2022).
Rosati, D. et al. Candida albicans N-linked mannans potentiate the induction of trained immunity via dectin-2. J. Infect. Dis. 230, 768–777 (2024).
Liu, G. et al. Bacteria-derived nanovesicles enhance tumour vaccination by trained immunity. Nat. Nanotechnol. 19, 387–398 (2024).
Zhang, C. et al. EZH2 identifies the precursors of human natural killer cells with trained immunity. Cancer Biol. Med. 18, 1021–1039 (2021).
Cheng, S. C. et al. mTOR- and HIF-1α-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 345, 1250684 (2014).
Cai, H. et al. Lactate activates trained immunity by fueling the tricarboxylic acid cycle and regulating histone lactylation. Nat Commun. 16, 3230 (2025).
Tannahill, G. M. et al. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 496, 238–242 (2013).
Chen, Y. et al. Macrophage-targeted efferocytosis therapy promotes diabetic wound healing via cellular level debridement. Adv. Funct. Mater. 35, 2503035 (2025).
Liu, L. et al. Proinflammatory signal suppresses proliferation and shifts macrophage metabolism from Myc-dependent to HIF1α-dependent. Proc. Natl. Acad. Sci. USA 113, 1564–1569 (2016).
Nobs, S. P. et al. Lung dendritic-cell metabolism underlies susceptibility to viral infection in diabetes. Nature 624, 645–652 (2023).
Gray, V. et al. Hyperglycemia-triggered lipid peroxidation destabilizes STAT4 and impairs anti-viral Th1 responses in type 2 diabetes. Cell Metab. 36, 2511–2527.e2517 (2024).
Cantuti-Castelvetri, L. et al. Defective cholesterol clearance limits remyelination in the aged central nervous system. Science 359, 684–688 (2018).
Wang, S. et al. Hematoma-like dynamic hydrogelation through natural glycopeptide molecular recognition for infected bone fracture repair. Bioact. Mater. 30, 73–84 (2023).
Lu, R. et al. A multifunctional tissue-engineering hydrogel aimed to regulate bacterial ferroptosis-like death and overcoming infection toward bone remodeling. Adv. Sci. 11, e2309820 (2024).
Feng, W. et al. Cascade-targeting poly(amino acid) nanoparticles eliminate intracellular bacteria via on-site antibiotic delivery. Adv. Mater. 34, e2109789 (2022).
Zhang, Y. et al. Enhanced chemoradiotherapy for MRSA-infected osteomyelitis using immunomodulatory polymer-reinforced nanotherapeutics. Adv. Mater. 36, e2304991 (2024).
Li, D. et al. A trained immunity inducer-adjuvanted nanovaccine reverses the growth of established tumors in mice. J. Nanobiotechnology 21, 74 (2023).
Acknowledgements
This research was funded by the Natural Science Foundation of Shanghai (23ZR1456800 to L.M.), the National Natural Science Foundation of China (82002188 to L.M.), the Shanghai Municipal Health Commission General Project (202240185 to L.M.), and the Shanghai Jiao Tong University “Medical and Research” initiative (YG2021ZD07 to L.M.). The authors are grateful to Dr. Xin Li from the Instrumental Analysis Center at Shanghai Jiao Tong University for their support with the relevant tests in this paper.
Author information
Authors and Affiliations
Contributions
H.C., L.W., Q.Y., M.W., and M.L. (Min Lu) were responsible for the overall study design. H.C., L.D., and M.L. (Min Lu) came up with the research idea and were involved in developing the materials used. H.C., C.W., H.F., M.L. (Ming Li), and R.D. conducted the in vitro experiments and performed data analysis. H.C., Y.M., J.J. (Jiahao Jiang), and L.W. carried out the in vivo osteomyelitis studies, including experimental design and analysis. H.C., J.J. (Jiahao Jiang), and Q.Y. performed histological analysis, immunofluorescence, and sequencing analysis. H.C. was in charge of histological evaluation and scoring. H.C., L.D., Q.Y., and L.W. were involved in the preparation of the figures. The manuscript was written by H.C., M.W., and M.L. (Ming Li), with contributions from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Zhiwen Zeng, Maziar Divangahi, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, H., Wei, L., Yu, Q. et al. Injectable hydrogels for osteomyelitis treatment induce metabolic reprogramming for protection against reinfection. Nat Commun 17, 1613 (2026). https://doi.org/10.1038/s41467-026-68318-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68318-2