Abstract
Staphylococcus aureus causes approximately 80% of skin and soft tissue infections (SSTIs). Collagen is the most abundant human extracellular matrix protein with critical roles in wound healing, and S. aureus encodes a collagen binding adhesin (Cna). The role of this protein during skin infections is unknown. Here we report that inability to bind collagen results in worsened pathology of intradermal Δcna S. aureus infection. WT/Cna+ S. aureus shows reduced infection severity, aggregate formation, and significantly improves clearance of bacteria. Cna binds to the collagen-like domain of serum C1q protein to reduce its opsonophagocytic functions. We demonstrate that infection of C1qKO mice with WT bacteria show results similar to the Δcna group. Conversely, inability to bind collagen results in an amplified inflammatory response caused in part by macrophage and neutrophil small molecule mediators released at the infection site (MMP-9, MMP-12, LTB4), leading to increased immune cell infiltration and death.
Similar content being viewed by others
Introduction
Collagen forms 30% of the protein dry weight in the human body1. With an essential role in wound healing, collagen is made up of 3 polypeptide alpha chains with characteristic Gly-X-X’ motifs that assemble to form a right-handed helical structure commonly identified as the ‘collagen like domain’2,3. Staphylococcus aureus is the most common cause of skin and soft tissue infections (SSTIs) such as abscesses, carbuncles and furuncles4,5. The treatment of these infections is often complicated by the acquisition of antibiotic resistance, the most common of which is the development of methicillin resistant S. aureus (MRSA)5,6. The success of S. aureus as a pathogen is in part because it is particularly adept at expressing numerous toxins that target host cells or evade immune responses7,8. One such evasion tactic is the expression of surface adhesins that bind host extracellular matrix components9. Among these proteins is the collagen binding adhesin (Cna). Multiple mouse models of intravenous injection demonstrate a role for Cna in the virulence of septic arthritis, keratitis and osteomyelitis caused by S. aureus, with the significance of Cna expression depending to some degree, on the model used10,11,12,13,14. There are currently no documented roles for Cna in the pathogenesis of S. aureus skin infection.
Cna is expressed by a subset of strains including the USA400 strain, MW2. USA400s are an epidemic MRSA with steadily reducing incidences in the United States, Europe and Canada over the past 15 years15,16,17,18. As a typical sortase anchored protein, Cna is attached to the cell wall via a C-terminal LPXTG motif19. The N-terminal A domain is characterized as being required for binding to collagen in the S. aureus strain Phillips20. The function of the B-repeat domain, that lies between the N-terminal A domain and the C-terminal anchor, is currently unknown21. USA300, a particularly problematic MRSA that has been implicated in a large percentage of SSTIs in the United States for more than a decade, does not express Cna6,22.
In addition to binding collagen, the A domain of Cna is also reported to bind the N-terminal collagen-like tail of serum protein C1q to prevent the classical pathway of complement mediated killing in S. aureus strain Phillips23. While the C-terminal globular head of C1q binds either directly to bacteria via pathogen associated molecular patterns (PAMPs), or to immunoglobulins on the bacterial surface, the N-terminal collagen like domain is recognized by innate immune cells24. Neutrophils and macrophages are key effectors in the phagocytic removal of opsonized bacteria. Following recognition of C1q bound to bacteria, a series of host proteolytic events result in the formation of C3b, via the C3 convertase (C4b2a). Deposition of C3b on the surface leads to bacterial uptake by neutrophils and macrophages which utilize toxic, antibacterial effectors to eliminate bacterial populations from the infection site25. Immune cells that are unable to phagocytose bacteria will likely use extracellular trap formation or frustrated phagocytosis as mechanisms to release antimicrobial compounds and kill bacteria26,27. Unlike phagocytosis, these processes can result in a higher degree of inflammation26,27,28,29. Among the proteins released by neutrophils, matrix metalloproteases (MMPs) are an important subclass of soluble mediators, with Zn2+ and Ca2+ dependent endopeptidase activities required for tissue remodeling. MMP-9 is released largely by neutrophils during degranulation but also by monocytes and macrophages. In addition to its collagenase activity, MMP-9 enhances TNF-α and IL-1β signaling and binds to CXCL8/KC, cleaving the cytokine to increase its chemotactic potency 10-fold30. Additionally, the collagenase activity of MMP-9 results in the formation of the inflammatory fragment, Pro-Gly-Pro which is also a potent neutrophil chemotactic factor, thereby augmenting the immune response to infection. Multiple reports demonstrate that MMP-9 is a significant contributing factor for pathogen control and removal31,32,33. The elastase MMP-12 is released primarily from macrophages and is required for their infiltration into infection sites. The hemopexin domain of this protein is reported to have bactericidal activity specifically against S. aureus34,35. Activation of MMPs also leads to the release of leukotriene B4 (LTB4) at the infection site. LTB4 binds to its cognate receptor BLT1, on leukocytes, to amplify immune influx. Overproduction of LTB4 can have detrimental effects, causing tissue damage to the host36. To control its activity, neutrophil and macrophage derived MMPs cause the activation of leukotriene hydrolases such as LTAH437. The contribution of these MMPs at the infection site is therefore significant to the inflammatory cascade induced during infection.
Our studies demonstrate that Cna-expressing S. aureus (Cna+) causes skin infections with significantly reduced bacterial loads accompanied by less inflammation, when compared with those caused by Cna-negative (Cna−) S. aureus. We show that binding to collagen and the collagen-like motif of C1q reduces bacterial spread and the inflammatory response to infection respectively. The two immune cell populations that we observe to be significantly affected by Cna-expressing bacteria are neutrophils and macrophages, both of which instigate a cycle of inflammation at the infection site, propagated by increased release of matrix metalloproteases and LTB4. Altogether this is the first report for the role of Cna in S. aureus skin infection and highlights the significance of collagen to infection outcomes.
Results
Expression of collagen binding adhesin (Cna) is sufficient to limit S. aureus abscess formation and bacterial burden
To evaluate the role of collagen binding adhesin in the outcome of S. aureus-associated skin infections, we utilized the clinical, MRSA USA400 MW2 strain of S. aureus that expresses Cna (hereafter WT), an isogenic mutant unable to synthesize Cna, (Δcna) and a corresponding Δcna strain where cna is ectopically expressed from a high copy expression vector Δcna:pCna (hereafter comp cna). In vitro adhesion assays demonstrate that expression of Cna by WT MW2 is sufficient for bacterial binding to collagen, a phenotype that is abrogated in the isogenic Δcna mutant (Fig. S1a). Collagen binding was restored in comp cna bacteria and bacterial growth of all three strains was found to be comparable (Fig. S1b). Furthermore, Cna was sufficient to induce collagen binding when heterologously expressed in the non-pathogenic Staphylococcus carnosus that does not encode a homolog of Cna (Fig. S1c).
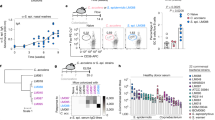
Since collagen is abundant in skin and is shown to be critical for the resolution of skin infection and wound healing, we utilized an established model of intradermal infection to determine the role of Cna in S. aureus virulence in vivo2,38. Following infection, we monitored abscess size and weight loss in female, age matched BALB/c mice over a 7-day period39,40. At the end of the experiment, lesions were excised and colony forming units per gram (CFU/g) of homogenized tissue were measured (Fig. 1a). WT and comp cna infected animals demonstrated weight loss comparable to the negative control group injected with saline, while Δcna infected mice lost ~10–20% of their initial weight (Fig. 1b). Mice infected with the Δcna mutant also formed discernably larger abscesses when compared with WT and comp cna infected groups (Fig. 1c, d). Larger lesions were accompanied by higher levels of recovered CFU/g (~1.5 log) of tissue in these mice, indicating exacerbated skin abscess infection in the absence of Cna (Fig. 1e).
a Method used for intradermal S. aureus infection and abscess formation. b Weight loss measured over the 7-day infection period in mice (n = 10 per bacterial strain) infected with WT MW2, isogenic Δcna or comp cna bacteria and calculated as a percentage of values at day 0. Asterisks indicate significant differences between WT and Δcna infected groups. c Images of abscess lesions taken at day 7, representative of mice infected with strains mentioned in (b). d Measurement of lesion sizes from mice infected with strains as above over a period of 7 days (n = 10 per bacterial strain). Measurements were made using ImageJ. Asterisks indicate significant differences between WT and Δcna infected groups. e Bacterial burdens enumerated per gram of homogenized tissue excised at day 7 post infection with strains as described above (n = 10 per bacterial strain). f Percent distribution of the cna gene as detected in S. aureus clones associated with human skin and soft tissue infections (n = 20) and colonizing healthy anterior nares (n = 30) g Abscess model of skin infection performed as described in (a), to compare pathology caused by USA300 (Cna−) and USA300:pCna (Cna+) bacteria (n = 5 mice per group). Weight loss was measured over 7 days and calculated as a percentage of values at day 0. h Representative images of lesions formed 7 days post infection with strains mentioned in (g). i Lesion sizes measured over 7 days post infection with strains as described in (g). Measurements were made using ImageJ. j Colony forming units (CFU) per gram of homogenized tissue enumerated from abscesses biopsied from mice infected with USA300 or USA300:pCna, at day 7 post inoculation (n = 5 mice). Results are representative of 3 (MW2) and 2 (USA300) independent analyses. Statistical analyses were performed with a one-way ANOVA with Tukey’s posttest (e) a two tailed Student’s t test (i) or a two-way ANOVA with Tukey’s posttest. Data are presented as median values ± SD. Groups with significant differences are denoted. *p < 0.05, **p < 0.01, ****p < 0.0001 (All adjusted p values are provided in Source Data file). a Created in BioRender. Urbina Martinez, M. (2026) https://BioRender.com/oa569ue.
To gauge the clinical relevance of findings with Cna, we assessed the prevalence of the cna gene in S. aureus isolates representing the major clonal types, from patients with primary SSTIs41. Of note, 60% of these isolates did not encode for cna (n = 20) (Fig. 1f and Table 1). Since nasal colonization is an important predisposing factor for skin infection, we also sequenced for the presence of the cna gene across the major clonal types in patients with positive nasal cultures for S. aureus42. We similarly found that ~60% of these isolates did not encode for cna (n = 30) (Fig. 1f and Table 2). The prevalence of Cna(−) strains was not specific to skin isolates and could similarly be found in patients with S. aureus pneumonia (n = 358) (Fig. S2a, Supplementary Data). Lastly, an analysis of available closed genomes of S. aureus showed that cna could only be identified in 38.4% of genomes (Fig. S2b). The presence of cna also varied across different clonal complexes (Fig. S2c). Among the cna(+) sequences, the predicted amino acid length of 2 identifiable pfam domains (pfam05738/cnaB-repeats and pfam17802) varied (Fig. S2d).
Considering the predominance for Cna(−) strains in patient samples, we performed a mouse intradermal infection using USA300, a dominant clinical strain of MRSA which does not encode the cna gene, or USA300 expressing cna from a high copy number vector, USA300:pCna (Fig. S1d)43. We infected mice as previously described and compared the progression of abscess formation with USA300 or USA300:pCna. As expected, we found that USA300 (Cna−) infected mice showed ~10% higher rates of weight loss when compared to the USA300:pCna group (Fig. 1g). This was comparable to observations made in mice infected with the Δcna isogenic mutant in the MW2 WT background. Similarly, we observed macroscopically larger abscesses, which was quantified by measuring lesion sizes over the course of infection (Fig. 1h, i). Lastly, these phenotypes were associated with ~2 log higher CFU/g of tissue in USA300 infected animals when compared to the USA300:pCna group (Fig. 1j).
To verify that these phenotypes were not due to sex differences in our mouse experiments, we performed a similar infection study in male, age matched BALB/c mice. Consistent with the results from experiments with female mice of the same background, we found that male BALB/c mice infected with Δcna bacteria had visibly larger abscesses, with larger lesions sizes measured through the course of infection, in comparison to the WT infected group (Fig. S3a). While we did not observe significant weight loss (likely due to an overall higher starting weight), bacterial burdens were similarly ~1–1.5 log higher in Δcna infected mice, as compared with WT and comp cna infected controls (Fig. S3b, c). Since we observed similar phenotypes in male mice, considering that females are less affected by the stress of handling procedures, as well as the variability introduced in mixed sex studies due to differences in skin morphology and hormones, we utilized female mice for the remainder of the study44,45,46. Lastly, to confirm that the observed phenotypes were not due to an overwhelming of the immune system incurred by a heavy inoculation dose, we performed similar experiments comparing WT, Δcna and comp cna infected at a ten-fold lower dose of total bacteria. Differences between these strains were reproduced at this inoculum, demonstrating that the phenotypes we observed were not due to the effect of heavy dosage (Fig. S3d–f). Collectively these data confirm that the expression of Cna is sufficient to restrict the bacterial burden and gross pathology observed during S. aureus skin infection.
Hemolytic activity caused by S. aureus dermonecrotic proteins is a well-documented virulence mechanism in skin wounds47,48. To determine any differences in hemolytic activity of strains used in this study, bacteria were grown on 5% sheep blood agar. Strains from the same parental background showed similar zones of hemolysis (Fig. S4a). A transposon mutant of hla, the gene encoding the alpha hemolysin protein, in the MW2 background was used as a control. An isogenic mutant of cna in this hla::Tn background was used to confirm any additional activity dependent on Cna. We verified a lack of differences by performing a hemolysis activity assay with supernatants from these strains, using previously established methods49. Lysis of rabbit red blood cells was similar across all strains (Fig. S4b, c). These results indicate that there are no major differences in the hemolytic activity of strains. Alpha hemolysin is a pore forming toxin that has specifically been shown to contribute to skin inflammation following bacterial infection47,48. To confirm that the phenotypes we observed for Δcna in mice are independent from the effect of Hla, we performed an intradermal infection as described above, and compared a transposon mutant of hla, with an isogenic hla::Tn Δcna strain in the MW2 strain background. While we observed an overall reduction in lesion size, likely due to the activity of Hla, similar to previous results, the inability to express Cna caused a significant increase in lesion size, weight loss and CFU burden. These results indicate that our observed Δcna phenotypes were independent of alpha toxin (Fig. S4d–f).
Collagen binding dampens the inflammatory response to S. aureus in skin abscesses
S. aureus skin infections are often associated with an inflammatory immune response that causes worsened pathology50. Hematoxylin and Eosin (H&E) staining performed on longitudinal sections of abscess tissue excised at day 7 demonstrated marked accumulation of cells around the infection bolus (purple) in Δcna infected animals when compared to WT or comp cna S. aureus, which were comparable to control animals injected with saline51 (Fig. 2a). We therefore utilized a multiplex assay to measure the concentrations of inflammatory cytokines in these tissue samples. We observed a comprehensive increase in the inflammatory response measured from Δcna infected mice when compared to WT MW2 and comp cna controls (Fig. 2b). Specifically, this included significant increases in the concentrations of key cytokines previously demonstrated to be signatures of worsening S. aureus skin infection (USA300) including IL-6, TNF-α and IL-1β, as well as chemotactic and growth factors for hemopoietic cells, KC, MCP-1, G-CSF and GM-CSF52. Additionally, infection with Δcna bacteria caused a significant increase in the concentration of tissue inhibitor of matrix metalloprotease-1 (TIMP-1) (Fig. 2c). Collectively these results indicate that Δcna bacteria induce a more pronounced inflammatory response when compared with an isogenic Cna(+) counterpart. We performed a similar cytokine analysis using the dominant clinical strain USA300, which does not express Cna. These measurements were performed in comparison to a USA300:pCna, as described in Fig. 1. USA300, much like the MW2 Δcna infected mice, showed an overall increase in levels of inflammatory cytokines compared to the USA300:pCna infected group (Fig. 2d).
a Hematoxylin Eosin staining of tissue sections biopsied from mice infected with WT, isogenic Δcna or comp cna bacteria at day 7 post infection (n = 2 per group). Tissue section of mouse injected with saline is shown as a negative control. b Cytokine array performed on mouse tissue collected from animals infected as described for (a). Multiplexing analysis was performed to measure the concentration of cytokines using the Luminex™ 200 system by Eve Technologies Corp. Each column represents the log of the average of duplicate readings made from a single mouse (n = 5 per group). c Individual graphs to show differences in cytokine concentrations (picogram per mL) as made in (b), for 8 cytokines of interest generated from mice in response to WT, isogenic Δcna or comp cna bacteria. d Quantification of cytokine concentrations similar to (b), made from mice infected with USA300 or the isogenic USA300:pCna strain (n = 4 per group). e Summary of methods used to perform flow cytometry quantification of immune cells from abscesses infected with bacteria as described for (a). f Quantification of the total number of single immune cells using an antibody specific to CD45, from tissue samples collected as described in (f), for mice infected with bacterial strains described in (a) (n = 7 mice). g Differentiation of cells enumerated in (f), based on exclusion of Am Cyan viability dye(live) (g) from observably dead populations (j). h Sub populations of total CD45 cells classified as live (h) or dead (k) neutrophils or macrophages (i, l) based on staining with cell specific antibodies as described in Fig. S5a, b. Results are representative of three independent analyses. Statistical analyses were performed with a one-way ANOVA with a Tukey’s post test. Data are presented as median values ± SD. *p < 0.05, **p < 0.01, ****p < 0.0001 (All adjusted p values are provided in Source Data file). e created in BioRender. Urbina Martinez, M. (2026) https://BioRender.com/6auc6af.
Absence of Cna results in increased immune cell death
To quantify immune populations that contribute to increased inflammation in mice infected with Δcna bacteria, we performed flow cytometry on perfused abscess tissue collected at day 7, using previously published methods53 (Figs. 2e and S5a, b). While the total numbers of CD45+ immune cells (as a proportion of total single cells) were comparable between WT, Δcna and comp cna infected groups of mice (Fig. 2f), we observed a large disparity in the numbers of live, and dead/dying CD45+ populations between these groups, with Δcna infected tissue samples containing very few live CD45+ cells and a large population of observable dead CD45+ cells compared to WT infected samples. This phenotype was able to be complemented, as proportions of both live and dead CD45+ cells could be restored to WT levels in mice infected with the comp cna strain (Fig. 2g, j). These data indicate that the loss of Cna results in abscesses composed primarily of dead immune cells. Comparison of Δcna to WT and especially comp cna infected groups indicate that expression of Cna may promote the survival of neutrophils (Fig. 2h) and macrophages (Fig. 2i), since live populations of these cells were restored to significantly higher levels in comp cna infected tissue compared with Δcna groups. Further analysis demonstrated that the observed increase in dead CD45+ cells in the Δcna infected animals was driven largely by an increase in (intact but) dead/dying neutrophils (Fig. 2k). Interestingly, we did not observe this increase of dead/dying cells within the macrophage population (Fig. 2l). Although to a smaller extent, the decrease in live immune cells in Δcna infected tissue was also observed for additional CD45+ populations (Fig. S6a, b). Collectively these results demonstrate a decrease in total immune cell viability, particularly of neutrophils, during infection with S. aureus that does not express Cna.
In order to gain a deeper understanding of the progression of infection phenotypes associated with Cna, we assessed the immune profile of these infections at an earlier time point. Our results indicate that Δcna infected mice develop larger abscesses as early as day 3, when compared to WT and comp cna groups (Fig. 1d). H&E staining revealed pathology in Δcna infected mice at day 3 that was similar to that observed at day 7, although the levels of cellular influx were less pronounced (Fig. S6c–f). We observed that gross infection phenotypes were similar to day 7 including weight loss (Fig. S7a), lesion sizes (Fig. S7b), CFU burdens (Fig. S7c) and inflammatory cytokine concentrations (Fig. S7d).
We therefore used the previously described antibody panel to quantify immune cell populations present in day-3 abscess tissue, collected and performed as previously described (Fig. 2e). Similar to results from day 7, while the CD45+ cells (as a proportion of total single cells) were similar among S. aureus infected groups, once again we observed very few live immune cells in Δcna infected lesion tissue, compared to both WT and comp cna infected animals (Fig. S7e). Concurrently, we observed significantly higher numbers of dead/dying cells in Δcna infected abscesses compared to WT and comp cna infected groups, also largely driven by increases in dead/dying neutrophils (Fig. S7f). Significantly higher numbers of live macrophages could be observed in WT infected tissues, compared to Δcna samples (Fig. S7g). The decrease in total numbers of live cells was also evident in additional subpopulations of CD45+ cells (Fig. S7h).
To confirm our flow cytometry observations and provide spatial insights on the immune cells present around each abscess in vivo, we performed multispectral imaging on tissue sections collected 3 days post infection (Fig. 3a). WT/Cna+ S. aureus was observed as DAPI- stained aggregates surrounded by collagen (Fig. 3b). These were confirmed to be bacteria using a modified Gram stain (Fig. S9a). We found that of the CD45+ cells present, neutrophils and macrophages were observed surrounding but restricted from the infection in WT infected abscesses (Fig. 3b). In contrast, Δcna infections showed a higher number of neutrophils and decrease in observable macrophages, with both populations in close juxtaposition on the infection nidus, which were more diffused compared to both WT and comp cna infected tissue (Fig. 3c, d). Quantification of these immune cells in the tissue section corroborated our previous flow cytometry results to show comparable levels of CD45+ populations with higher numbers of neutrophils (~30%) in Δcna infected samples compared to both WT and comp cna sections, and numbers of macrophages remaining similar between groups (Fig. 3e). Together these results indicate that the absence of collagen leading to direct contact of bacteria with immune cells, causes a dysfunction in the innate immune response to infection. This results in bacterial persistence and an exaggerated inflammatory response. Since the most significant change in the innate immune response to Cna was observed with the neutrophil population, we decided to investigate this in greater detail.
a Multispectral quantitative pathology of skin abscess tissue excised from mice infected with WT, Δcna or comp cna bacteria 3 days post inoculation and stained with antibodies targeting 6 host proteins as described in the legend. Images depict staining of entire tissue section as used for quantitative analysis. b Images digitally zoomed in (X6.5) from sections shown in (a). Regions of interest are depicted in a as magenta boxes. Images demonstrate staining from tissues infected with WT (b), Δcna (c) or comp cna (d) and stained with antibodies targeting 6 host proteins as shown in legend. Images are separated according to channels that demonstrate spatial distribution of bacteria with collagen (DAPI, Collagen), CD45 cells, neutrophils (CD45, Ly6G) and macrophages (CD45, F480). e Quantification of staining demonstrated in (a) represented as total cells per millimeter of tissue sections infected with strains as described above. Staining of tissue was repeated for a total n = 2.
Increased uptake of Δcna bacteria causes neutrophil lysis
Serum complement protein C1q plays an important role in the opsonophagocytosis of S. aureus54. C1q consists of a C-terminal globular head domain that binds to immunoglobulins or directly to the bacterial surface to activate the classical proteolytic pathway that leads to opsonization and phagocytosis of S. aureus55. The N- terminus of C1q is a collagenous tail domain that has previously been shown to bind to Cna derived from S. aureus strain Phillips in vitro and inhibit downstream activation of the classical pathway23 (Fig. S9b). To confirm similar in vitro binding of Cna to the C1q N-terminal tail in the MW2 strain background, we utilized a competitive enzyme linked immunosorbent assay with a C1q-coated surface to demonstrate a reciprocal relationship wherein the level of Cna bound to C1q progressively decreased when collagen was incubated in the presence of increasing concentrations of recombinant Cna (Fig. S9c). Since we observed increases in populations of dead neutrophils during in vivo infection (Fig. 2k), we sought to examine the effect of this interaction on downstream activation of the classical complement pathway. We opsonized WT, Δcna and comp cna and measured bound C3b or C4b. C4b is part of the C3 convertase and is cleaved to activate C3b, an opsonin with a central role in the complement pathway24,55. Using flow cytometry, we confirmed that Δcna bacteria exhibited significantly higher levels of C4b (Fig. 4a) and C3b (Fig. 4b) deposition, compared with both WT and comp cna strains, similar to results observed in strain Phillips. Consistent with the previous study, these results indicate that the presence of Cna reduces opsonization of bacteria by the classical complement pathway23. To assess the effect of this Cna mediated inhibition of C1q specifically on phagocytic activity, we measured bacterial uptake in the presence of primary human neutrophils using previously published methods56,57,58. We observed a significant increase in the uptake of Δcna bacteria (~20%) when compared to neutrophils exposed to WT or comp cna for 10 min (Fig. 4c). To attribute a direct role to C1q in this process, we opsonized bacteria with C1q-depleted serum (instead of pooled normal human serum) prior to performing the experiment. Under these conditions the differences observed in uptake between WT, Δcna and comp cna bacteria were abrogated (Fig. 4d). Collectively these results suggest that Cna binds to serum C1q, reducing complement activation and subsequent phagocytic uptake by neutrophils.
Flow cytometry of WT, Δcna or comp cna bacteria opsonized with 10% serum and stained with antibodies targeting serum complement proteins C4b (a) and C3b (b) (n = 3 biological replicates). c Uptake of WT, Δcna or comp cna bacteria by primary human neutrophils, following opsonization with 10% pooled human serum. CFU was enumerated 10 min post incubation, following which samples were treated with lysostaphin (5 min) to exclude extracellular populations. Experiment similar to (c) performed with bacteria opsonized with 10% C1q-depleted, pooled human serum (d). Experiments similar to (c), with intracellular (e) and total (f) bacterial survival calculated at 30 min post incubation with or without lysostaphin treatment, respectively. Confocal microscopy performed on WT, Δcna or comp cna (Green = Syto-9/live) opsonized as mentioned above in the presence of type 1 collagen and incubated with Cell Tracker Blue-labeled primary human neutrophils for 20 min, following which samples were stained with ethidium homodimer-1 (red) to visualize dead/dying cells. Arrows indicate likely location of collagen boundary (g, i) and lysed neutrophils (h). Statistical analyses were performed with a one-way ANOVA and Tukey’s post test. Data are presented as median values ± SD. *p < 0.05, **p < 0.01, ****p < 0.0001 (All adjusted p values are provided in Source Data file). All experiments were an n = 3 donors performed in duplicate.
We hypothesized that the increase in opsonophagocytic uptake observed in response to Δcna, would result in lytic neutrophil death. We therefore measured bacterial survival 30 min post incubation with neutrophils and observed significantly lower numbers of intracellular Δcna bacteria, compared to neutrophils that were exposed to WT or comp cna S. aureus (Fig. 4e). To understand if this was due to neutrophil lysis, bacterial release, and their inadvertent killing due to lysostaphin treatment, we measured bacterial survival in the absence of lysostaphin. We observed a significantly larger population of surviving Δcna bacteria compared with both the WT and comp cna under these conditions (Fig. 4f). These observations confirm that the Cna-C1q interaction decreases phagocytic uptake by neutrophils, inadvertently allowing for controlled, efficient killing of the pathogen by these cells. Conversely, in the absence of Cna, we find that neutrophils increasingly phagocytose bacteria which results in cell death and release of Cna(−) S. aureus.
Cna interacts with collagen to restrict neutrophil access to bacteria
C1q plays a central role in the opsonophagocytic response of neutrophils to bacteria55. We reasoned that both collagen and C1q would be present and capable of binding to WT Cna(+) bacteria in vivo. The binding of collagen, which has a higher affinity for Cna compared with C1q, would create a protective barrier or matrix to impede the entry of neutrophils and therefore phagocytosis3,59. Conversely, Δcna bacteria would present C1q appropriately, have no collagen matrix for protection and would be phagocytosed at higher rates (Fig. 6a, c). We therefore performed confocal microscopy on bacteria opsonized with pooled normal human serum in the presence of collagen and incubated with primary human neutrophils for 20 min, as described above. Binding to collagen restricted the direct access of neutrophils (blue) to WT (Fig. 4g) and comp cna bacteria (Fig. 4i). Loss of Syto-9 staining in bacteria closest to the periphery of the collagen-bound aggregate (compared with the center) indicated bacterial killing (Fig. 4g, i white arrows). In sharp contrast, Δcna bacteria largely caused lysis of neutrophils as evidenced by ethidium homodimer-1 staining (EthHD-1/ Fig. 4h, white arrows). In agreement with results from the opsonophagocytosis assay, we observed large numbers of Syto-9 labeled, extracellular Δcna bacteria, indicating bacterial survival (Fig. 4h). Together these results indicate that binding to collagen allows for controlled, efficient clearance of bacteria. Conversely, neutrophils have direct access to Δcna bacteria even in the presence of collagen, inadvertently causing them to lyse.
Since C1q and collagen both bind Cna, we performed an experiment similar to (Fig. 4g–i), with bacteria that were opsonized with C1q depleted serum with collagen, before incubation with human neutrophils. Under these conditions, we observed that neutrophils gained access to the collagen bound aggregates of WT and comp cna bacteria. This was associated with neutrophil lysis comparable to the response generated to Δcna bacteria in both C1q replete and depleted conditions. WT and comp cna were observed to elicit a response similar to Δcna bacteria in the absence of C1q including the presence of large numbers of bacteria (green) associated with EthHD-1-stained neutrophils (Fig. S9d, e).
Cna-C1q interaction dampens inflammation caused in response to S. aureus in vivo
Binding of C1q to Cna would occur via the N-terminal collagen-like domain of C1q and therefore make it unavailable for appropriate signaling23. We reasoned that this would result in decreased recognition and removal by immune cells in vivo, including neutrophils. Since Δcna bacteria present C1q appropriately, this would result in an inflammatory response, as previously observed with S. aureus60,61. To evaluate the contribution of the C1q-Cna interaction in vivo, we used the previously described abscess model of skin infection in C1q knockout mice (hereafter C1qKO) in the C57BL/6 WT background. Since our observations were made in the BALB/c mouse background, we performed a comparison of infection between C57BL/6 WT and C1qKO mice. As with previous results, we found that infection of C57BL/6 WT mice with Δcna bacteria resulted in a ~1.5 log increase in bacterial burdens (Fig. 5a) and abscess lesions (Fig. 5b). Weight loss was observed in WT and Δcna infected animals, unlike the comp cna group in C57BL/6 WT mice (Fig. S10a). We therefore measured the levels of inflammatory cytokines present in WT C57BL/6 abscesses and observed results similar to infections performed in female BALB/c mice (Fig. S10b), with Δcna infected abscesses containing significantly higher concentrations of key inflammatory markers when compared to both WT and comp cna groups (Fig. S10c). To understand the role of the C1q-Cna interaction in this model we similarly infected C1qKO mice with WT, Δcna or comp cna bacteria and observed an overall increase in the bacterial burden of WT and Δcna infected animals retrieved at day 7, when compared to previous observations (Fig. 5a). Additionally, we observed a reduction of the difference in bacterial CFU loads (~1 log) between WT and Δcna infected mice. WT and Δcna lesions sizes were more alike in the C1qKO mouse background and comp cna lesion sizes remained lower when compared to the other groups, likely due to the overexpression of Cna from the high copy number vector (Fig. 5a, c). Although overall the differences between WT and Δcna induced cytokines were smaller in the C1qKO mice, phenotypes from individual cytokines measured were similar to those recorded from WT mice. (Figs. 5d and S10e). Altogether our in vitro and in vivo results confirm that the absence of C1q assists bacterial survival in a Cna dependent manner and the Cna-C1q interaction reduces the severity of lesion formation by WT S. aureus but that additional factors likely contribute to the Cna-dependent differences in inflammatory response observed.
a CFU per gram of homogenized tissue enumerated from either WT C57/BL6 or C1qKO mice, 7 days post infection with WT, Δcna or comp cna bacteria. Representative images of lesions formed at day 7 post infection with lesion sizes measured over the course of the infection for each strain (n = 10 per group, bottom) of WT C57/BL6 (b) or C1qKO (c) mice. d Comprehensive view for the concentrations of inflammatory cytokines measured from abscess tissue 7 days after infection with WT, Δcna or comp cna, in the C1qKO mouse background. Concentrations are presented in logarithmic scale of picogram per mL homogenized tissue. Each column represents results from a single mouse performed in duplicate (n = 5 per group). e Graphical depiction of the known mechanisms by which matrix metalloproteases 9 and 12 can instigate a cascade of neutrophil and macrophage mediated inflammation of skin infection sites that is amplified by the interaction between leukotriene B4 and its receptor (BLT-1). Concentrations of MMP-2, pro-9 and 12 measured from abscess tissue excised from WT BL57 (f) or C1qKO (g) mice (n = 5) infected with WT, Δcna or comp cna bacteria. h Concentrations of leukotriene A-4 hydrolase (LTAH4) measured using an enzyme linked immunosorbent assay, from WT C57/BL6 mice infected with bacterial strains as described in (g) (n = 9 mice per bacterial strain). i Assay similar to (h) measuring the concentrations of leukotriene B4 from homogenized tissue samples (n = 5 mice per group). j Assay similar to (h), quantifying the concentrations of LTAH4 and performed with tissue samples from C1qKO mice infected with bacterial strains as described in (h) (n = 5 mice per group). Results are representative of two independent analyses. Statistical analyses were performed with a two-way ANOVA (b, c) or a one-way ANOVA (a, f–j) with Tukey’s posttest. Data are presented as median values ± SD. *p < 0.05, **p < 0.01 (All adjusted p values are provided in Source Data file). e created in BioRender. Urbina Martinez, M. (2026) https://BioRender.com/byzqrhz.
Tissue matrix metalloproteases contribute to innate immune cell dysfunction and increased immune cell influx
We observed a significantly higher level of the tissue inhibitor of matrix metalloprotease-1 (TIMP-1) measured from Δcna infected mouse tissue, when compared to WT and comp cna groups at both day 3 (Fig. S7) and 7 (Fig. 2b, c) post infection. This suggested that the host immune system may be attempting to control the levels of inflammatory MMPs in the tissue bed during Δcna infection. MMP-2 and 9 are gelatinases that also act as collagenases, while MMP-12 is a macrophage elastase33,62. MMP-9 is specifically released by neutrophils in response to the small molecule mediator, leukotriene B4 (LTB4), from macrophages63. LTB4 interacts with the leukotriene B4 1 receptor (BLT-1) expressed on leukocytes, to activate the release of MMP-9 from neutrophils which degrades collagen to form the proinflammatory, neutrophil chemotactic peptide, Pro-Gly-Pro. Binding of Pro-Gly-Pro to CXCR1/2, results in recruitment of additional neutrophils to the site of infection (Fig. 5e). This would result in increased levels of the chemokines KC and MCP-1 similar to our observations at day 3 (Fig. S7) and 7 (Fig. 2b, c) and the large influx of neutrophils as observed in vivo (Figs. 2 and 3).
Concentrations of MMP-2, 9 and 12 measured from abscess tissues were significantly higher in the Δcna infected group when compared to WT and comp cna (Fig. 5f). Since both the C-terminal globular domain and N- terminal collagen tail of C1q are recognized by macrophages via the gC1qR and cC1qR/calreticulin receptors respectively, we asked whether this inflammatory cascade was disrupted in the absence of C1q. While C1qKO mice showed higher levels of all MMPs measured, when compared to WT C57BL/6 mice, levels of MMPs in WT and comp cna infected groups were similar to those measured from Δcna infected mice (Fig. 5g). To terminate this inflammatory cycle, proinflammatory, MMP-12- secreting macrophages release leukotriene A4 hydrolase (LTAH4) which directly binds and inactivates Pro-Gly-Pro37. Δcna infected mice contained significantly higher levels of LTAH4 compared with WT and comp cna groups (Fig. 5h), and these differences were lost in C1qKO mice (Fig. 5i). Since LTAH4 is also required for the formation of LTB4, we reasoned that LTAH4 binding to increased levels of Pro-Gly-Pro would reciprocally limit the concentrations of released LTB4. Indeed, we observed a significant reduction in concentrations of this small molecule in Δcna infected animals, compared with WT and comp cna infected mice (Fig. 5j).
Since LTB4 binds to its receptor, BLT1, in order to elicit an influx of immune cells and consequent inflammation, we treated mice with the BLT-1 inhibitor, U-75302 (or ethanol as vehicle control), following infection with either WT, Δcna or comp cna bacteria using previously published methods64,65,66. Inhibitor was delivered intradermally on alternate days for a week, at which point the experiment was terminated and results recorded (Fig. S11a). Previous studies with USA300 (Cna-) S. aureus have shown that infection leads to production of LTB4 and that inhibiting BLT1 leads to an increase in bacterial burdens67. We therefore expected to see an increase in bacterial burdens in all infected groups. Since our observations indicate that the immune system is attempting to control LTB4- associated inflammation specifically in response to Δcna, we expected to see larger differences in Δcna mice treated with the BLT1 inhibitor. We found that Δcna infections led to larger abscesses compared with WT and comp cna infected mice in the vehicle control group. Although overall the lesion sizes were smaller, treatment with the inhibitor significantly increased lesion sizes of Δcna infected mice, compared to all other groups. The inhibitor increased lesion sizes for mice treated with WT bacteria as well, in comparison to the vehicle control (Fig. S11b, c). While there were no significant changes in weight loss, treatment with inhibitor caused a ~2 log increase of bacterial burdens in both WT and Δcna burdens, likely accounting for increased lesion sizes in these groups (Figs. S11d and S12a). Of note, comp cna lesion sizes and bacterial burdens did not change when BLT1 was inhibited, indicating that overexpression of Cna can abrogate the effects of BLT1 inhibition. These results recapitulate previous observations for the role of BLT1 signaling in controlling S. aureus infection (USA300) and additionally demonstrate that BLT1 contributes in a Cna-dependent manner67.
Since BLT1 signaling affects the influx of immune cells, including neutrophils, we confirmed BLT1 inhibition by measuring the levels of the neutrophil chemokine KC in mice infected with respective strains with or without inhibitor. We found that the levels of KC decrease in all groups of mice treated with the inhibitor (Fig. S11e). A similar trend was also recorded for IL-1β, MIP-2α and MCP-1, as previously observed (Fig. S12b–d)68. To confirm that BLT1 inhibition was playing a role in reducing the MMP mediated inflammatory cycle, we measured the levels of MMPs present in these tissues. We saw a significant decrease in proMMP-9 (Fig. S11f), MMP-2 (Fig. S11g) and MMP-12 (Fig. S11h) specifically in Δcna mice treated with the inhibitor when compared to vehicle controls. Of note, in all outputs of BLT1 function measured, significant differences were observed when comparing Δcna infected groups with and without inhibitor. Together these results confirm that the MMP induced cycle of inflammation and immune influx plays a significant role in controlling bacterial infection in skin abscesses, and that this occurs in a Cna-dependent manner.
Discussion
Studies characterizing the cna gene of S. aureus were performed over a decade ago11,12,14. Our knowledge of its importance to S. aureus pathogenesis, however, is extremely limited. Published reports while informative, were performed in three infection environments, namely eye, heart and bone, utilizing the less characterized strains of S. aureus: Phillips and CYL31613,14,69. These previous studies do not examine Cna in the context of skin colonization or SSTIs despite the essential role for collagen in the resolution of skin infection and the overwhelming presence of S. aureus in a majority of purulent SSTIs41,70. Here we provide a comprehensive analysis for the role of Cna, specifically during S. aureus skin infections. We demonstrate that the inability to directly bind collagen is associated with worsened outcomes (Fig. 1a–e), and that Cna(−) S. aureus is prevalent among major nose, skin and pneumonia clones as well as available reference genomes of S. aureus (Figs. 1f and S2a, b). Furthermore, we establish that Cna is necessary and sufficient to reduce the severity of skin infection caused by the predominant, clinically relevant Cna(−) strain, USA300, in a mouse model of infection (Fig. 1g–j). These results demonstrate the importance of considering Cna expression of S. aureus strains especially when studying skin infection phenotypes.
Multiple comparative analyses that investigate the role of S. aureus adhesins to inflammation do not identify Cna as an important virulence determinant71. In one study that included skin samples from patients with atopic dermatitis, psoriasis and normal skin colonized with S. aureus, absence of Cna expressed from S. aureus resulted in no observable decrease in bacterial binding to the stratum corneum72. An analysis of cna sequences identified from available closed genomes indicates that the length and sequence of Cna protein varies between strains and clonal complexes (Fig. S2c, d). Further studies to specifically characterize the prevalence and sequence of isolates lacking the cna gene, as well as the role of the B-repeats, are therefore imperative to our knowledge of S. aureus infections73. Of note, Panton Valentine leukotoxin (PVL) is epidemiologically linked to primary, purulent SSTIs in humans but does not contribute to infections in mice74,75,76. Although we see no correlation between the presence of pvl and cna in skin associated isolates (Tables 1 and 2) and MW2 and USA300, the strains used in our study, both express PVL, further studies in human tissue and primary cell models will determine if PVL can contribute to immune cell death.
Biochemical characterization of the Cna protein from strain Phillips, identifies the N-terminal A domain as the ligand binding region of the protein and demonstrates that this domain additionally binds to complement protein C1q to prevent the opsonization of S. aureus23. Authors in these studies used RBC lysis as a functional read out of complement activation. Here, we additionally demonstrate opsonophagocytosis as a more relevant endpoint (Fig. 4a–c)77,78. By using primary human neutrophils, we build on previous findings to show that when C1q is not sequestered by binding to Cna, this results in a likely uncontrolled mechanism of cell death with increased neutrophil lysis accompanied by bacterial survival (Fig. 6a, c). It is plausible that while Cna-C1q binding affects opsonization and uptake of bacteria as shown here, toxins released by S. aureus are responsible for bacterial escape and neutrophil lysis. Leukotoxins are widely characterized as causing immune cell lysis, with LukAB being particularly significant at allowing intracellular bacteria to lyse neutrophils from within79,80,81. This activity occurs in concert with alpha hemolysin (Hla), a secreted, pore-forming toxin82,83. Further studies that expand on the effect of Cna and its interactions with host ligands, with emphasis on the neutrophil response to S. aureus, are required in order to further evaluate potential roles for these toxins. Here we demonstrate that competition between collagen and C1q for binding to Cna correlates with the downstream neutrophil response, and that the ability to fine-tune ligand binding (collagen vs. C1q) is necessary to protect S. aureus from immune clearance mechanisms23. Indeed, when Cna is ectopically produced from a high copy expression vector, bacterial survival in C1qKO mice is comparable to WT and comp cna S. aureus, in WT C57BL/6 mice, indicating that increased binding to collagen may compensate for the absence of binding events that occur with the C1q N-terminal domain (Fig. 5a). Of note, C57BL/6 showed behavioral differences that indicated stress, compared with BALB/c mice, likely contributing to the variability observed in weight loss and lesion size. Further studies to construct C1qKOs in BALB/c mice and alternatively understand if there are inherent differences in the skin response to S. aureus between species of mice will provide additional insight into these observations.
a The N-terminal collagen-like domain of C1q binds to Cna and reduces complement deposition and opsonophagocytosis. Expression of Cna allows S. aureus to bind to collagen and restrict bacterial contact with immune cells such as neutrophils. Bacteria that are taken up are eliminated by neutrophils. b The neutrophil response to Cna(+) S. aureus activates the release of the neutrophil chemokine KC, causing an influx of neutrophils. Macrophages at the site of infection release MMP-12 and leukotriene B4 which interacts with its receptor, BLT1, on neutrophils, macrophages and other cells. This causes the activation of MMP-9 which cleaves collagen, forming the highly inflammatory molecule, Pro-Gly-Pro. MMP-2 (collagenase) is released by epithelial cells and fibroblasts contributing to inflammation and production of Pro-Gly-Pro. Neutrophil receptors CXCR1/2 interact with KC and Pro-Gly-Pro which causes the influx of additional neutrophils, thereby controlling infection. c Bacteria lacking the ability to express Cna cannot directly bind to collagen. C1q is not sequestered in the absence of Cna, causing increased bacterial uptake by neutrophils. This allows direct contact between neutrophils and bacteria. This leads to neutrophil lysis and inflammation. d The neutrophil response to Cna(−) bacteria results in neutrophil lysis, release of higher levels of KC and influx of a larger number of neutrophils (compared to Cna(+) conditions). This amplifies the cycle of inflammation shown in c and leads to the production of significantly higher levels of leukotriene hydrolase 4 (LTAH4) from macrophages (as compared to Cna(+)), an enzyme that breaks down LTB4. Due to the influx and death of neutrophils this inflammation cycle perpetuates. Created in BioRender. Urbina Martinez, M. (2026) https://BioRender.com/rk7djos.
Our results provide further evidence for the vital role that neutrophils and macrophages play during resolution of skin infections (Fig. 6)50,84. In this work, the absence of viable macrophages was accompanied by increased numbers of dead neutrophils and prolonged inflammation in mice infected with Δcna bacteria, providing further validation of the communication between neutrophils and macrophages that is essential for resolution of skin infection. Hla has documented roles in macrophage lysis, leading to reduced neutrophil infiltration and bacterial clearance during skin infection85. Increased bacterial survival in the absence of Cna may result in higher local concentrations of Hla and therefore more lysed macrophages. Whether Hla plays an additional role in the lack of live macrophages observed in our studies, remains to be assessed and would provide a deeper understanding of the triggers that cause macrophage activation, small molecule release and therefore neutrophil infiltration. Similarly, the increased concentrations of matrix metalloproteases may be a direct effect of the presence of a larger number of lysed neutrophils. One report indicates that matrix metalloproteases 1, 2, 3 and 9 can cleave the collagen-like domain of C1q and result in neutrophil reactive oxygen burst. This may contribute to the killing of bacteria that are exposed to neutrophils (Fig. 4g–i)86. Lastly, while we see a rise in observable, dead neutrophils when mice are infected with Δcna bacteria, we do not observe these differences in macrophage populations at the timepoints examined (Fig. 2j). While this work focuses on the neutrophil response, C1q interacts with macrophages to control inflammation in vivo. It is plausible that sequestration of C1q by Cna causes additional reductions in inflammation due to altered macrophage signaling87,88. Additionally, macrophages can suffer numerous fates following resolution of inflammation including conversion to endothelial cells or fibroblasts89. Recently, fibroblasts have been demonstrated to be an important source of collagen that is utilized as nutrition by S. aureus during pulmonary infections. Of note, these studies were performed in strains that do not express Cna90. The fate and function of macrophages and monocytes following exposure to S. aureus in the context of Cna, remains to be resolved91,92.
Results presented here translate in vitro experiments for the first time to demonstrate a direct association between the Cna-C1q interaction and control of inflammation in vivo (Fig. 5a–d). It is important to note, however, that C1q is one of many host proteins that contain a collagen-like motif, any of which could be present in the abscess microenvironment and contribute to sequestration of available ligand binding domains on Cna93,94. Additionally, host proteins that bind the N-terminus of C1q, such as MBL-associated serine proteases, could compete with Cna for binding95. Similarly, most studies focus on the ability of Cna to bind type 1 collagen. The ratio of type 1/3 collagen is crucial for wound healing and most matrix metalloproteases bind multiple types of collagens. This may influence the outcome of infection in the context of Cna, and remains to be studied62,96.
We demonstrate that the immune response generated by S. aureus- collagen interactions in a mouse model of infection is, in part, due to the degree of activation of an inflammatory cycle involving matrix metalloproteases and LTB4. When collagen is not sequestered around the infection, this causes a significant amplification of this cycle, leading to increased levels of MMP-2, pro-MMP9, MMP-12 and LTAH4 (with subsequent decrease in LTB4) (Fig. 5e–h). These results signify that the host immune system may attempt to control inflammation during Δcna infection. Blocking LTB4 activity by intradermally inhibiting its immune receptor BLT-1, resulted in decreased levels of neutrophil chemokines accompanied by increased bacterial burdens and lesion sizes particularly in the Δcna infected group (Figs. 6e and S11b–d). This was similar to previous studies demonstrating the contribution of LTB4 in controlling the spread of USA300 skin infection in mice. LTB4 signaling is complex and can influence inflammation in multiple ways64,67. Further studies are required to determine how LTAH4 is activated as well as the role of collagen degradation products to the response observed in vivo.
USA300 (Cna−) and MW2 (Cna+) are both successful, clinically isolated strains of S. aureus that differ in their ability to express Cna17,22,97. This, together with our findings, indicates that rather than determining bacterial survival, the expression of Cna may dictate the nature and length of infection caused by S. aureus, as well as the degree of inflammation achieved as a consequence. Our current work reveals that rather than assisting in infection exacerbation and dissemination of S. aureus such as would be expected of a canonical virulence factor, Cna is involved in concealing bacteria from the immune system in a mouse model of infection. This allows bacteria to quiescently establish an infection bolus, likely until favorable conditions for growth become scarce and dissemination is required for survival. These findings also indicate that the loss of cna may have provided an evolutionary advantage to enhance S. aureus dissemination98,99. Conversely, expression of Cna by a subset of strains may allow them to persist for long periods in the community, either as colonizers or as chronic, biofilm-associated infections13,14,69.
S. aureus cells expressing Cna accumulate collagen, which may serve as a ’self’ signal that allows immune evasion. Bacteria afford the time required to proliferate and form a collagen shield by binding to collagen-like domains of the major innate, bacterial recognition protein, C1q. This function potentially extends to additional pathogen recognition molecules, many of which express similar domains93,94. Additionally, bacteria that are recognized and engulfed by immune cells presumably utilize one or more bacterial toxins to kill these cells, allowing S. aureus to survive58,100,101. We demonstrate that the loss of cna promotes the expansion of S. aureus infection in mice. It is well established that when S. aureus is present in sufficient numbers, it is not easily eliminated by the immune system58,100,102. Our results with murine skin infections confirm this and show that this leads to massive immune cell death, likely caused due to bacterial virulence properties that are similar to Cna expressing cells. The zone of necrotic cells, largely neutrophils, presumably shields bacteria from the entry and function of additional immune cells into the infection bolus. Altogether, utilizing a murine model of infection, this work establishes a significant role for collagen interactions in S. aureus skin infections and demonstrates its substantial immune evasion properties, while setting a precedence for deeper investigation into the collagen binding properties of additional pathogens (Fig. 6).
Methods
Bacterial strains and growth conditions
Bacterial strains used in this study are listed in Table 3. Unless otherwise indicated, all strains of Staphylococcus aureus were grown in tryptic soy broth (TSB) at 37 °C with shaking. Overnight bacterial suspensions were sub-cultured and grown to the exponential phase for all in vitro assays. This corresponded to an optical density of 0.42 (O.D. 600). Antibiotics were added during growth for strains expressing plasmids. Escherichia coli ER2566 (used for purification of recombinant Cna, Fig. S9c) was grown in Luria Bertani Broth using methods described below for protein purification.
Generation of bacterial mutants and complementation
Strain MW2 Δcna was created by transducing the Δcna::tetM cassette from a previously published strain AH4770 into the wild-type MW2 using a phage 11103,104. The resulting mutant was validated through PCR for the cna gene. Strains MW2 hla::Tn and MW2 hla::Tn Δcna were created by transducing the hla::Tn transposon from the Nebraska Transposon Mutant Library into the MW2 wild-type and MW2 Δcna strains using the phage 11103,105. Mutants were validated by PCR for the hla gene. The cna complementing plasmid was generated by amplifying cna with its promoter region using the Q5 polymerase from WT MW2 genomic DNA using the primers:
F-ctcggtaccttaggaggatgattatttatgaacaagaacgtgttgaa
R- acagctatgacatgattacgaattcttatgagttaaatctttttcttaaaattaaatac
KpnI and EcoRI were used to digest this fragment, which was subsequently ligated into an E.coli-S.aureus shuttle expression vector pCM28 digested with the same enzymes106. The resulting plasmid was propagated in E.coli NEB 5α and was subsequently electroporated into the intermediate host S. aureus RN4220, from where it was moved by electroporation into the final staphylococcal strain of interest. The resulting plasmid was confirmed through Sanger sequencing. To ensure plasmid stability inside staphylococci, it was maintained with 10 µg/mL chloramphenicol.
Murine skin abscess model of infection
Six-week-old mice were obtained from the Jackson Laboratory and allowed to acclimate for 1 week prior to experiments. Unless otherwise indicated, experiments were performed in female BALB/cJ mice (Strain #:000651, RRID:IMSR_JAX:000651). Experiments in Fig. 5 were performed with WT C57/BL6J (Strain #:000664 RRID:IMSR_JAX:000664) or C1qKO background (B6(Cg)-C1qatm1d(EUCOMM)Wtsi/TennJ, Strain #:031675 RRID:IMSR_JAX:031675). Controlled conditions (12-h light/dark cycles, 20–22 °C temperature, 30–70% humidity) were provided with ad libitum access to food and water. All infections were performed on 7-week-old mice by inoculating 108 total bacteria in 50 uL intradermally as previously described39,40. Briefly, overnight bacterial cultures were sub-cultured and grown to an appropriate OD and resuspended in saline. Mouse stomachs were shaved and treated with Nair, hair removal cream, 1 day prior to inoculation of bacteria. Abscess formation was monitored with imaging over the span of 7 days. Mouse weight loss was measured daily. On day 7, mice were sacrificed, and abscesses excised. Abscess tissue was resuspended in 500 uL phosphate-buffered saline and homogenized with physical disruption using (0.1 mm beads, Biospec Mini- Beadbeater). Colony-forming units were calculated from homogenate and plotted per gram of tissue. Lesion sizes were measured using ImageJ 1.54p software. For BLT-1 inhibitor studies, 10ug U-75302 was diluted in saline (Cayman Chem, 70705) and intradermally injected at or near to the location of infection, 1 day post bacterial inoculation. Ethanol was used for the vehicle control as previously described64. These injections were performed on days 1, 3 and 5.
Detection of cna from clinical isolates
We assessed the presence of cna by analyzing the genome sequences of isolates representing the major clonal types of methicillin-susceptible and -resistant S. aureus isolates from two previously published studies of primary purulent SSTI and nasal colonization41,42. Briefly, libraries were prepared and sequenced at the NYU Langone Genome Technology Center using an Illumina NovaSeq to produce paired-end 150 bp reads. Reads were filtered and trimmed with fastp v0.20.1 using default settings107. Confindr v0.7.4 identified within-species contamination and isolates with >10% contamination were excluded108. Filtered reads were then assembled with Unicycler v0.4.8 in conservative mode109. Taxonomic classification of assemblies was performed using GTDBTK v1.5.1; non–S. aureus isolates were excluded110. S. aureus sequence types and clonal complexes were determined with MLST (https://github.com/tseemann/mlst). We used BLAST v2.12.0+ to search for the cnaB gene with the KEGG sequence (ID: MW2612), considering a genome cnaB positive if a BLAST hit showed an E-value < 10−20 111,112.
Mouse histology
Abscess tissue was fixed in 10% formalin and submitted to the Gates Histology Services Core Lab (University of Colorado Anschutz Medical Campus) for microtomy and hematoxylin-eosin staining. Standard regressive H&E was performed on 4um sections of tissue113. Images were analyzed using QuPath-0.5.0-x64.
Cytokine measurements
Cytokine concentrations were measured from mouse tissue homogenate that was treated with protease inhibitor (Sigma) and stored in PBS at −80 °C before being analyzed by Eve Technologies. Briefly, the multiplexing analysis was performed using the Luminex™ 200 system (Luminex, Austin, TX, USA) by Eve Technologies Corp. (Calgary, Alberta). Forty-five markers were simultaneously measured in the samples using Eve Technologies’ Mouse Cytokine 45-Plex Discovery Assay®. Assay sensitivities of these markers range from 0.3–30.6 pg/mL for the 45-plex. Cytokines measured at levels out of the range of the assay/given extrapolated values for all groups of mice, were eliminated from further analysis. Individual analyte sensitivity values are available in the MilliporeSigma MILLIPLEX® MAP protocol.
Murine skin abscess flow cytometry
A murine model of skin abscess infection with S. aureus was performed as described above. Skin abscesses were harvested using a 6 mm biopsy punch following perfusion of the animal with PBS and heparin. Skin punches were minced and incubated for 2 h at 37 °C with shaking in RPMI 1640 w/o phenol red (Fischer Scientific, Catalog No: 11-835-055) with Miltenyi Multi Tissue Dissociation Kit 1 enzymes (volumes according to the manufacturer). Following enzymatic digestion, samples were mechanically digested using the Miltenyi Gentle Macs dissociator (program “Multi H”). Single cell suspensions were prepared from digested abscess samples by filtering samples through a 70 mm cell strainer and pelleting cells by centrifugation (300 × g, 5 min). Remaining red blood cells were removed by resuspending the cell pellet in red blood cell lysis buffer (150 mM NH4Cl, 10 mM KHCO3, 0.1 mM Na2EDTA; pH 7.2) for 2 min at room temperature and washing with RPMI 1640 (Gibco). Cells were then pelleted by centrifugation and resuspended in MACS buffer (Phosphate-buffered saline with 0.5% BSA and 2 mm EDTA, pH 7.2). Single cell suspensions were first stained with eBioscience Fixable Viability Dye eFluor 506 (Catalog # 65-0866-18) in PBS for 30 min at room temperature. Cells were stained with the anti-mouse surface antibodies in MACS buffer for 30 min at room temperature (see Fig. S2b for details on antibodies). After surface antibody staining, the cells were fixed (30 min at room temperature) using the FoxP3 fixation/permeabilization kit (Thermo Fisher Scientific, Catalog # 00-5523-00). Stained cells were analyzed on a BD LSRFortessa (BD Biosciences) using the BD FacsDiva software (v9) or Cytoflex. Data were analyzed with BD FlowJo software v 10.10.0. Gating strategy was adapted from a previous study and can be found in Fig. S2a53. Since dead cells often exhibit auto fluorescence, we performed fluorescence minus one (FMO) controls on Δcna-infected abscess tissue (which contains largely dead immune cells) for the viability dye to assess background fluorescence of eFluor 506 within CD45+, Ly6G+ and CD64+ populations (Fig. S5a). Similarly, FMO controls for each of three markers, CD45+, Ly6G+ and CD64+ in Δcna-infected abscess tissue, confirmed little to no autofluorescence by these fluorophores, despite the large proportion of dead cells within these tissues (Fig. S5b). S5b). All FMO controls in Fig. S8 represent staining by the entire flow cytometric panel outlined in Fig. S5b, except for one fluorophore of interest (e.g., Viability-eFluor 506 in panel a, and CD45-BUV395, Ly6G-APC, or CD64-BV650, respectively, in panel b).
C4b and C3b binding assays
To measure the differences in C3 binding between various strains, bacteria were grown overnight in TSB and brought up to an O.D of 0.42 were washed 3 times with PBS and re-suspended in 10% pooled human serum for 30 min. After repeating the washing step, each sample was stained with rabbit monoclonal Alexa Fluor 647-labeled anti-C3 antibody (Abcam ab196639), or isotype IgGκ control (BD 565571), FITC labeled anti-C4b antibody (Thermo Fisher, PA1-28407) or isotype IgG control (Invitrogen #11-4614-80) for 30 or 20 min respectively, at room temperature. Samples were then washed 3 times with PBS and resuspended in PBS for analysis by flow cytometry, using the BD LSR Fortessa (BD Biosciences) and BD FacsDiva software (v9).
Neutrophil isolation
Isolation of primary human neutrophils was performed using whole blood collected from healthy, consenting human volunteers. Briefly, whole blood components were separated using a Ficoll Hypaqe based density gradient. Following removal of peripheral blood monocytes and lymphocytes, red blood cells were lysed with water and remaining neutrophils were resuspended in 0.9% sodium chloride. Following re-suspension in phosphate buffered saline neutrophils were enumerated using a hemocytometer. Neutrophils were used at a concentration of 4 × 106cells/mL as previously published57.
Neutrophil opsonophagocytosis assays
S. aureus was incubated with primary, blood-derived human neutrophils isolated as previously described57. To do this, overnight cultures of bacteria grown in tryptic soy broth were diluted 1:100 into fresh medium and grown to an O.D of 0.42, corresponding to ~1 × 108 CFU/mL. Cultures were then washed 3 times with phosphate buffered saline (PBS) and re-suspended in 10% pooled normal or C1q depleted human serum (Complement Tech, Catalog No: NHS, A300 respectively) for 30 min, in order to opsonize bacteria. Cultures were washed 3 times with PBS and resuspended in 4 × 106 neutrophils/mL, to bring the multiplicity of infection to 1 (neutrophil): 25 (Bacteria). Samples were then incubated at 37 degrees for 10 (uptake) or 30 (survival) min and treated with 10 ug/mL lysostaphin for 5 min to eliminate extracellular bacteria, as previously described56. Intracellular bacterial survival was assessed by plating serial dilutions on tryptic soy agar. Percent survival was calculated by comparing survival for each strain at 10 and 30 min, to its bacterial inoculum present at time 0.
Neutrophil confocal microscopy
Bacterial strains were opsonized as described above either with 20 μg/ml type I rat tail collagen (Corning®, Cat No: 354236), or without collagen as a control. Staining was performed using previously published methods58. Briefly, neutrophils were isolated as described above (4 × 106/mL) and stained with Cell Tracker Blue CMAC (Thermo Fisher, Cat No: C2110). Opsonized bacteria labeled with Syto-9 (Invitrogen, Cat No: S34854) were washed and incubated with neutrophils (1:25) for 20 min in the presence of and Ethidium Homodimer-1 (Invitrogen, Cat No: E1169), similarly to the method described above. Samples were washed (X3 PBS) and centrifuged (800 × g) for 15 min, pellets re-suspended in minimal volume and mounted onto glass slides with Pro Long Gold Antifade (Invitrogen, Cat No: P36930) with coverslips. Images were collected using the Olympus FV1000 confocal laser scanning microscope (Advanced Light Microscopy Core, University of Colorado Anschutz Medical Campus) and images acquired with the FluoView system (Version 4.2.4.5). Images were acquired with the XY scan mode, LSM observation mode, one-way scan direction and Line Kalman integration type. A total of 405 nm (PMT 431 V) laser transmissivity was 4.8%, 488 nm (PMT 311 V) laser transmissivity was 3.0% and 543 nm (316 V) laser transmissivity was 12.0% Numerical apertures was 1.4 and scan speed was set at 12.5 [us/Pixel]. Images were not processed.
Matrix metalloproteinase measurements
MMP concentrations were measured from mouse tissue homogenate that was treated with protease inhibitor and stored in PBS at −80 °C before being analyzed by Eve Technologies. Briefly, the multiplexing analysis was performed using the Luminex™ 200 system (Luminex, Austin, TX, USA) by Eve Technologies Corp. (Calgary, Alberta). Five markers were simultaneously measured in the samples using Eve Technologies’ Mouse MMP 5-Plex Discovery Assay® (MilliporeSigma, Burlington, Massachusetts, USA) according to the manufacturer’s protocol. The 5-plex consisted of MMP-2, MMP-3, MMP-8, proMMP-9 and MMP-12. Assay sensitivities of these markers range from 1.6–8.4 pg/mL for the 5-plex. Individual analyte sensitivity values are available in the Millipore Sigma MILLIPLEX® MAP protocol.
Measurement of LTAH4 and LTB4 from abscess tissue
Enzyme-linked immunosorbent assays were performed to measure the concentration of LTAH4 (Biomatik, Cat No: EKN46742-96T) and LTB4 (Avantar, Cat No: 76576-968) according to the manufacturer’s protocol. Quantification was done using abscess tissue that was biopsied on the same day (day 7 post infection), homogenized as described above and re-suspended in PBS to be used immediately.
Immunofluorescence staining
Performed on paraffin-embedded tissue sections excised from 7-week-old, female BALB/c mice which were euthanized 3 days post inoculation with WT, Δcna or comp cna bacteria, or saline as a negative control. Analysis done by the Human Immune Monitoring Shared Resource, University of Colorado Cancer Center, who can be contacted directly for further technical information on the Vectra Polaris system used for these studies114. Imaging shown includes antibodies against CD11b (1:1000 Novus, Cat No: NB110-89474), Ly6G (1:100 Cell Signaling Tech, Cat No: 87048S), COL1A1 (1:100 Cell Signaling Tech, Cat No: 72026S), CD45 (1:100 Cell Signaling Tech, Cat No: 70257S), F480 (1:100 Cell Signaling Tech, Cat No: 30325S)114,115.
Statistical analysis
All statistical analysis was performed using GraphPad Prism Version 10.4.0 (527). All experiments were performed in triplicate with a minimum n = 3 and plotted with standard deviation. Two-tailed, unpaired Student's t test with an F-test to compare variance was performed where appropriate. Significance was assessed as p < 0.05. One-way or two-way analysis of variance was performed with Tukey’s least significant difference post-hoc test where applicable. Bartlett’s test was used to assess the extent of variability between groups and variance was found to be similar. Sample sizes were chosen using a pilot experiment in triplicate that achieved approximately a specified half-width with 95% confidence interval (α = 0.05).
Ethics statement
Experiments with animals were reviewed and approved by the Institutional Animal Care and Use Committee at the University of Colorado Anschutz Medical Campus (IACUC #00486). Primary human neutrophils were isolated from healthy human donors after obtaining informed, written consent from each donor. This was done according to the protocol approved by the University of Colorado Anschutz Medical Campus institutional review board (IRB #17-1926).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings made here are provided in the article, Source data file, and Supplementary Information (Figshare: https://doi.org/10.6084/m9.figshare.30473018). Further inquiries and information on reagents and resources should be directed to (and will be fulfilled by) the lead contact, A.R.H. (alexander.horswill@cuanschutz.edu). Source data are provided with this paper.
References
Ricard-Blum, S. The collagen family. Cold Spring Harb. Perspect. Biol. 3, 1–19 (2011).
Mathew-Steiner, S. S., Roy, S. & Sen, C. K. Collagen in Wound Healing. Bioengineering (Basel). 8, 63 (2021).
Valotteau, C. et al. Single-cell and single-molecule analysis unravels the multifunctionality of the Staphylococcus aureus collagen-binding protein Cna. ACS Nano 11, 2160–2170 (2017).
Del Giudice, P. Skin infections caused by Staphylococcus aureus. Acta Derm. Venereol. 100, 208–215 (2020).
Esposito, S., Noviello, S. & Leone, S. Epidemiology and microbiology of skin and soft tissue infections. Curr. Opin. Infect. Dis. 29, 109–115 (2016).
Kaye, K. S., Petty, L. A., Shorr, A. F. & Zilberberg, M. D. Current epidemiology, etiology, and burden of acute skin infections in the United States. Clin. Infect. Dis. 68, S193–S199 (2019).
Otto, M. Staphylococcus aureus toxins. Curr. Opin. Microbiol. 17, 32–37 (2014).
Dal Peraro, M. & van der Goot, F. G. Pore-forming toxins: ancient, but never really out of fashion. Nat. Rev. Microbiol. 14, 77–92 (2016).
Bhattacharya, M. & Horswill, A. R. The role of human extracellular matrix proteins in defining Staphylococcus aureus biofilm infections. FEMS Microbiol. Rev. 48, 2 (2024).
Thomas, M. G., Peacock, S., Daenke, S. & Berendt, A. R. Adhesion of Staphylococcus aureus to collagen is not a major virulence determinant for septic arthritis, osteomyelitis, or endocarditis. J. Infect. Dis. 179, 291–293 (1999).
Nilsson, I. M., Patti, J. M., Bremell, T., Höök, M. & Tarkowski, A. Vaccination with a recombinant fragment of collagen adhesin provides protection against Staphylococcus aureus-mediated septic death. J. Clin. Investig. 101, 2640–2649 (1998).
Patti, J. M. et al. Molecular characterization and expression of a gene encoding a Staphylococcus aureus collagen adhesin. J. Biol. Chem. 267, 4766–4772 (1992).
Elasri, M. O. et al. Staphylococcus aureus collagen adhesin contributes to the pathogenesis of osteomyelitis. Bone 30, 275–280 (2002).
Rhem, M. N. et al. The collagen-binding adhesin is a virulence factor in Staphylococcus aureus keratitis. Infect. Immun. 68, 3776 (2000).
Four pediatric deaths from community-acquired methicillin-resistant Staphylococcus aureus—Minnesota and North Dakota, 1997–1999. MMWR Morb Mortal Wkly Rep Vol. 48 707–10 (1999).
Groom, A. V. et al. Community-acquired methicillin-resistant Staphylococcus aureus in a rural American Indian community. JAMA 286, 1201–1205 (2001).
Vignaroli, C., Varaldo, P. E. & Camporese, A. Methicillin-resistant Staphylococcus aureus USA400 Clone, Italy. Emerg. Infect. Dis. 15, 995 (2009).
Golding, G. R. et al. High rates of Staphylococcus aureus USA400 infection, Northern Canada. Emerg. Infect. Dis. 17, 722 (2011).
Mazmanian, S. K., Liu, G., Ton-That, H. & Schneewind, O. Staphylococcus aureus sortase, an enzyme that anchors surface proteins to the cell wall. Science 285, 760–763 (1999).
Zong, Y. et al. A ‘Collagen Hug’ model for Staphylococcus aureus CNA binding to collagen. EMBO J. 24, 4224–4236 (2005).
Snodgrass, J. L. et al. Functional analysis of the Staphylococcus aureus collagen adhesin B domain. Infect. Immun. 67, 3952–3959 (1999).
Carrel, M., Perencevich, E. N. & David, M. Z. USA300 methicillin-resistant Staphylococcus aureus, United States, 2000-2013. Emerg. Infect. Dis. 21, 1973–1980 (2015).
Kang, M. et al. Collagen-binding microbial surface components recognizing adhesive matrix molecule (MSCRAMM) of gram-positive bacteria inhibit complement activation via the classical pathway. J. Biol. Chem. 288, 20520 (2013).
Giang, J. et al. Complement activation in inflammatory skin diseases. Front. Immunol. 9, 639 (2018).
Pouw, R. B. & Ricklin, D. Tipping the balance: intricate roles of the complement system in disease and therapy. Semin. Immunopathol. 43, 757–771 (2021).
Fuchs, T. A. et al. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 176, 231–241 (2007).
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Padmore, T., Stark, C., Turkevich, L. A. & Champion, J. A. Quantitative analysis of the role of fiber length on phagocytosis and inflammatory response by alveolar macrophages. Biochim. Biophys Acta Gen. Subj. 1861, 58–67 (2017).
Castanheira, F. V. S. & Kubes, P. Neutrophils and NETs in modulating acute and chronic inflammation. Blood 133, 2178–2185 (2019).
Kandhwal, M. et al. Role of matrix metalloproteinase in wound healing. Am. J. Transl. Res. 14, 4391 (2022).
Taylor, J. L. et al. Role for matrix metalloproteinase 9 in granuloma formation during pulmonary Mycobacterium tuberculosis infection. Infect. Immun. 74, 6135–6144 (2006).
Malik, M. et al. Matrix metalloproteinase 9 activity enhances host susceptibility to pulmonary infection with type A and B strains of Francisella tularensis. J. Immunol. 178, 1013–1020 (2007).
Elkington, P. T. G., O’Kane, C. M. & Friedland, J. S. The paradox of matrix metalloproteinases in infectious disease. Clin. Exp. Immunol. 142, 12 (2005).
Pidwill, G. R., Gibson, J. F., Cole, J., Renshaw, S. A. & Foster, S. J. The role of macrophages in Staphylococcus aureus infection. Front. Immunol. 11, 620339 (2021).
Houghton, A. M. G., Hartzell, W. O., Robbins, C. S., Gomis-Rüth, F. X. & Shapiro, S. D. Macrophage elastase kills bacteria within murine macrophages. Nature 460, 637 (2009).
Brandt, S. L. et al. Excessive localized leukotriene B4 levels dictate poor skin host defense in diabetic mice. JCI Insight 3, e120220 (2018).
Stsiapanava, A. et al. Binding of Pro-Gly-Pro at the active site of leukotriene A4 hydrolase/aminopeptidase and development of an epoxide hydrolase selective inhibitor. Proc. Natl. Acad. Sci. USA 111, 4227–4232 (2014).
Roy, S. et al. Staphylococcus aureus biofilm infection compromises wound healing by causing deficiencies in granulation tissue collagen. Ann. Surg. 271, 1174–1185 (2020).
Brown, M. M. et al. Novel peptide from commensal Staphylococcus simulans blocks methicillin-resistant Staphylococcus aureus quorum sensing and protects host skin from damage. Antimicrob. Agents Chemother. 64, e00172-20 (2020).
Kwiecinski Id, J. M. et al. Staphylococcus aureus adhesion in endovascular infections is controlled by the ArlRS-MgrA signaling cascade. https://doi.org/10.1371/journal.ppat.1007800 (2019).
Pelzek, A. J. et al. Human memory B cells targeting Staphylococcus aureus exotoxins are prevalent with skin and soft tissue infection. mBio 9, e02125-17 (2018).
Phillips, M. et al. Preventing surgical site infections: a randomized, open-label trial of nasal mupirocin ointment and nasal povidone-iodine solution. Infect. Control Hosp. Epidemiol. 35, 826–832 (2014).
Foster, C. E. et al. Adhesin genes and biofilm formation among pediatric Staphylococcus aureus isolates from implant-associated infections. PLoS ONE 15, e0235115 (2020).
Chi, L. et al. Sexual dimorphism in skin immunity is mediated by androgen-ILC2-dendritic cell axis. Science 384, eadk6200 (2024).
Sensini, F. et al. The impact of handling technique and handling frequency on laboratory mouse welfare is sex-specific. Sci. Rep. 10, 1–9 (2020).
Azzi, L., El-Alfy, M., Martel, C. & Labrie, F. Gender differences in mouse skin morphology and specific effects of sex steroids and dehydroepiandrosterone. J. Invest. Dermatol. 124, 22–27 (2005).
Kennedy, A. D. et al. Targeting of alpha-hemolysin by active or passive immunization decreases severity of USA300 skin infections in a mouse model. J. Infect. Dis. 202, 1050 (2010).
Yang, C., Robledo-Avila, F. H., Partida-Sanchez, S. & Montgomery, C. P. α-Hemolysin-mediated endothelial injury contributes to the development of Staphylococcus aureus-induced dermonecrosis. Infect. Immun. 92, e0013324 (2024).
Bernheimer, A. W. [30] Assay of hemolytic toxins. Methods Enzymol. 165, 213–217 (1988).
Brandt, S. L., Putnam, N. E., Cassat, J. E. & Serezani, C. H. Innate Immunity to Staphylococcus aureus: evolving paradigms in soft tissue and invasive infections. J. Immunol. 200, 3871–3880 (2018).
Fischer, A. H., Jacobson, K. A., Rose, J. & Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. 2008, pdb.prot4986 (2008).
Chan, L. C. et al. Protective immunity in recurrent Staphylococcus aureus infection reflects localized immune signatures and macrophage-conferred memory. Proc. Natl. Acad. Sci. USA 115, E11111–E11119 (2018).
Yu, Y. R. A. et al. A protocol for the comprehensive flow cytometric analysis of immune cells in normal and inflamed murine non-lymphoid tissues. PLoS ONE 11, e0150606 (2016).
Chen, X., Schneewind, O. & Missiakas, D. Engineered human antibodies for the opsonization and killing of Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 119, e2114478119 (2022).
Ricklin, D., Hajishengallis, G., Yang, K. & Lambris, J. D. Complement: a key system for immune surveillance and homeostasis. Nat. Immunol. 11, 785–797 (2010).
Thammavongsa, V., Missiakas, D. M. & Schneewind, O. Staphylococcus aureus degrades neutrophil extracellular traps to promote immune cell death. Science 342, 863–866 (2013).
Nauseef, W. M. Isolation of human neutrophils from venous blood. Methods Mol. Biol. 412, 15–20 (2007).
Bhattacharya, M. et al. Leukocidins and the nuclease Nuc prevent neutrophil mediated killing of Staphylococcus aureus biofilms. Infect. Immun. 88, e00372-20 (2020).
Herman-Bausier, P. et al. Mechanical strength and inhibition of the Staphylococcus aureus collagen-binding protein Cna. mBio 7, e01529–16 (2016).
Kitur, K. et al. Necroptosis promotes Staphylococcus aureus clearance by inhibiting excessive inflammatory signaling. Cell Rep. 16, 2219 (2016).
Liu, Q., Mazhar, M. & Miller, L. S. Immune and inflammatory reponses to Staphylococcus aureus skin infections. Curr. Dermatol. Rep. 7, 338 (2018).
Krishnaswamy, V. R., Mintz, D. & Sagi, I. Matrix metalloproteinases: the sculptors of chronic cutaneous wounds. Biochim. Biophys Acta Mol. Cell Res. 1864, 2220–2227 (2017).
Chou, R. C. et al. Lipid-cytokine-chemokine cascade drives neutrophil recruitment in a murine model of inflammatory arthritis. Immunity 33, 266 (2010).
Shao, W.-H., Del Prete, A., Bock, C. B. & Haribabu, B. Targeted disruption of leukotriene B4 receptors BLT1 and BLT2: a critical role for BLT1 in collagen-induced arthritis in mice. J. Immunol. 176, 6254–6261 (2006).
Prado, M. K. B. et al. Leukotriene B4 is essential for lung host defence and alpha-defensin-1 production during Achromobacter xylosoxidans infection. Sci. Rep. 7, 1–13 (2017).
Kuraishi, Y., Nagasawa, T., Hayashi, K. & Satoh, M. Scratching behavior induced by pruritogenic but not algesiogenic agents in mice. Eur. J. Pharm. 275, 229–233 (1995).
Brandt, S. L. et al. Macrophage-derived LTB4 promotes abscess formation and clearance of Staphylococcus aureus skin infection in mice. PLoS Pathog. 14, e1007244 (2018).
Scott, M. J. et al. Leukotriene B4 receptor (BLT-1) modulates neutrophil influx into the peritoneum but not the lung and liver during surgically induced bacterial peritonitis in mice. Clin. Diagn. Lab Immunol. 11, 936 (2004).
Hienz, S. A., Schennings, T., Heimdahl, A. & Flock, J. I. Collagen binding of Staphylococcus aureus is a virulence factor in experimental endocarditis. J. Infect. Dis. 174, 83–88 (1996).
Ray, G. T., Suaya, J. A. & Baxter, R. Incidence, microbiology, and patient characteristics of skin and soft-tissue infections in a U.S. population: a retrospective population-based study. BMC Infect. Dis. 13, 252 (2013).
Cho, S. H. et al. Preferential binding of Staphylococcus aureus to skin sites of Th2-mediated inflammation in a murine model. J. Invest. Dermatol. 116, 658–663 (2001).
Cho, S. H., Strickland, I., Boguniewicz, M. & Leung, D. Y. M. Fibronectin and fibrinogen contribute to the enhanced binding of Staphylococcus aureus to atopic skin. J. Allergy Clin. Immunol. 108, 269–274 (2001).
Symersky, J. et al. Structure of the collagen-binding domain from a Staphylococcus aureus adhesin. Nat. Struct. Biol. 4, 833–838 (1997).
Del Giudice, P. et al. Panton-valentine leukocidin-positive Staphylococcus aureus strains are associated with follicular skin infections. Dermatology 222, 167–170 (2011).
Friesen, J. et al. Panton-Valentine leukocidin–positive Staphylococcus aureus in skin and soft tissue infections from primary care patients. Clin. Microbiol. Infect. 26, 1416.e1–1416.e4 (2020).
Jin, Y., Zhou, W., Ge, Q., Shen, P. & Xiao, Y. Epidemiology and clinical features of skin and soft tissue infections caused by PVL-positive and PVL-negative methicillin-resistant Staphylococcus aureus isolates in inpatients in China: a single-center retrospective 7-year study. Emerg. Microbes Infect. 13, 2316809 (2024).
Vandendriessche, S., Cambier, S., Proost, P. & Marques, P. E. Complement receptors and their role in leukocyte recruitment and phagocytosis. Front. Cell Dev. Biol. 9, 624025 (2021).
Boero, E. et al. Purified complement C3b triggers phagocytosis and activation of human neutrophils via complement receptor 1. Sci. Rep. 13, 1–17 (2023).
Spaan, A. N. et al. The staphylococcal toxin panton-valentine leukocidin targets human C5a receptors. Cell Host Microbe 13, 584–594 (2013).
Spaan, A. N., van Strijp, J. A. G. & Torres, V. J. Leukocidins: staphylococcal bi-component pore-forming toxins find their receptors. Nat. Rev. Microbiol. 15, 435–447 (2017).
Yoong, P. & Torres, V. J. The effects of Staphylococcus aureus leukotoxins on the host: cell lysis and beyond. Curr. Opin. Microbiol. 16, 63–69 (2013).
DuMont, A. L. et al. Staphylococcus aureus elaborates leukocidin AB to mediate escape from within human neutrophils. Infect. Immun. 81, 1830–1841 (2013).
DuMont, A. L. et al. Staphylococcus aureus LukAB cytotoxin kills human neutrophils by targeting the CD11b subunit of the integrin Mac-1. Proc. Natl. Acad. Sci. USA 110, 10794–10799 (2013).
Forde, A. J. et al. Metabolic rewiring tunes dermal macrophages in staphylococcal skin infection. Sci. Immunol. 8, eadg3517 (2023).
Abtin, A. et al. Perivascular macrophages mediate neutrophil recruitment during bacterial skin infection. Nat. Immunol. 15, 45–53 (2013).
Ruiz, S. S. et al. Digestion of C1q collagen-like domain with MMPs-1,−2,−3, and-9 further defines the sequence involved in the stimulation of neutrophil superoxide production. Artic. J. Leukoc. Biol. 66, 416–422 (1999).
Benoit, M. E., Clarke, E. V., Morgado, P., Fraser, D. A. & Tenner, A. J. Complement protein C1q directs macrophage polarization and limits inflammasome activity during the uptake of apoptotic cells. J. Immunol. 188, 5682 (2012).
Galvan, M. D., Foreman, D. B., Zeng, E., Tan, J. C. & Bohlson, S. S. Complement component C1q regulates macrophage expression of Mer tyrosine kinase to promote clearance of apoptotic cells. J. Immunol. 188, 3716 (2012).
Sinha, M. et al. Direct conversion of injury-site myeloid cells to fibroblast-like cells of granulation tissue. Nat. Commun. 9, 936 (2018).
Urso, A. et al. Staphylococcus aureus adapts to exploit collagen-derived proline during chronic infection. Nat. Microbiol. 1–16 https://doi.org/10.1038/s41564-024-01769-9 (2024).
Gordon, S., Plüddemann, A. & Martinez Estrada, F. Macrophage heterogeneity in tissues: phenotypic diversity and functions. Immunol. Rev. 262, 36–55 (2014).
Jopling, C., Boue, S. & Belmonte, J. C. I. Dedifferentiation, transdifferentiation and reprogramming: three routes to regeneration. Nat. Rev. Mol. Cell Biol. 12, 79–89 (2011).
Bidula, S., Sexton, D. W. & Schelenz, S. Ficolins and the recognition of pathogenic microorganisms: an overview of the innate immune response and contribution of single nucleotide polymorphisms. J. Immunol. Res. 2019, 3205072 (2019).
Lacroix, M. et al. Interaction of complement defence collagens C1q and mannose-binding lectin with BMP-1/tolloid-like proteinases. Sci. Rep. 7, 1–13 (2017).
Rosbjerg, A. et al. C1q/MASP complexes—hybrid complexes of classical and lectin pathway proteins are found in the circulation. J. Immunol. 213, 998–1007 (2024).
Singh, D., Rai, V. & Agrawal, D. K. Regulation of collagen I and collagen III in tissue injury and regeneration. Cardiol. Cardiovasc. Med. 7, 5 (2023).
Tong, S. Y. C., Davis, J. S., Eichenberger, E., Holland, T. L. & Fowler, V. G. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 28, 603–661 (2015).
Thomer, L., Schneewind, O. & Missiakas, D. Pathogenesis of Staphylococcus aureus bloodstream infections. Annu. Rev. Pathol. 11, 343–364 (2016).
Kwiecinski, J. et al. Staphylokinase promotes the establishment of Staphylococcus aureus skin infections while decreasing disease severity. J. Infect. Dis. 208, 990–999 (2013).
Bhattacharya, M. et al. Staphylococcus aureus biofilms release leukocidins to elicit extracellular trap formation and evade neutrophil-mediated killing. Proc. Natl. Acad. Sci. USA 115, 7416–7421 (2018).
Vandenesch, F., Lina, G. & Henry, T. Staphylococcus aureus hemolysins, bi-component leukocidins, and cytolytic peptides: a redundant arsenal of membrane-damaging virulence factors? Front. Cell Infect. Microbiol. 2, 12 (2012).
Thammavongsa, V., Kim, H. K., Missiakas, D. & Schneewind, O. Staphylococcal manipulation of host immune responses. Nat. Rev. Microbiol. 13, 529–43 (2015).
Novick, R. P. [27] Genetic systems in Staphylococci. Methods Enzymol. 204, 587–636 (1991).
Kwiecinski, J. M. et al. Staphylococcus aureus adhesion in endovascular infections is controlled by the ArlRS-MgrA signaling cascade. PLoS Pathog. 15, e1007800 (2019).
Fey, P. D. et al. A genetic resource for rapid and comprehensive phenotype screening of nonessential Staphylococcus aureus genes. mBio https://doi.org/10.1128/mBio.00537-12.Editor (2013).
Pang, Y. Y. et al. agr-Dependent interactions of Staphylococcus aureus. USA300 with human polymorphonuclear neutrophils. J. Innate Immun. 2, 546–559 (2010).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Low, A. J., Koziol, A. G., Manninger, P. A., Blais, B. & Carrillo, C. D. ConFindr: rapid detection of intraspecies and cross-species contamination in bacterial whole-genome sequence data. PeerJ 7, e6995 (2019).
Wick, R. R., Judd, L. M., Gorrie, C. L. & Holt, K. E. Unicycler: resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput. Biol. 13, e1005595 (2017).
Chaumeil, P. A., Mussig, A. J., Hugenholtz, P. & Parks, D. H. GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database. Bioinformatics 36, 1925–1927 (2020).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Kanehisa, M. & Goto, S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Luna, L. G., et al. Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology. 3rd ed, Blakiston Division (McGraw-Hill, 1968).
Steinhart, B. et al. The spatial context of tumor-infiltrating immune cells associates with improved ovarian cancer survival. Mol. Cancer Res. 19, 1973–1979 (2021).
Hoyt, C. C. Multiplex immunofluorescence and multispectral imaging: forming the basis of a clinical test platform for immuno-oncology. Front. Mol. Biosci. 8, 674747 (2021).
Kwiecinski, J. M. et al. Staphylococcus aureus uses the ArlRS and MgrA cascade to regulate immune evasion during skin infection. Cell Rep. 36, 109462 (2021).
Wormann, M. E., Reichmann, N. T., Malone, C. L., Horswill, A. R. & Grundling, A. Proteolytic cleavage inactivates the Staphylococcus aureus lipoteichoic acid synthase. J. Bacteriol. 193, 5279–5291 (2011).
Kreiswirth, B. N. et al. The toxic shock syndrome exotoxin structural gene is not detectably transmitted by a prophage. Nature 305, 709–712 (1983).
Acknowledgements
The authors would like to thank members of the Horswill and Doran groups for their critical evaluation of the data in this manuscript. We also acknowledge the Human Immune Monitoring Shared Resource (RRID:SCR_021985) within the University of Colorado Human Immunology and Immunotherapy Initiative and the University of Colorado Cancer Center (P30CA046934) for their expert assistance in the analysis of multispectral quantitative pathology. We thank Laura Hoaglin HT(ASCP) with the Gates Histology Services Core Lab (University of Colorado Anschutz Medical Campus) for microtomy and hematoxylin-eosin staining. B.S. is funded by the National Institutes of Health (NIH) awards R01AI137336, R01AI193629, and R01AI140754. K.S.D. is funded by NIH awards R01NS116716 and R01AI153332. A.R.H. is funded by NIH award AI083211 and the Department of Veteran’s Affairs award BX002711.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.B., J.M.K., and A.R.H.; Methodology: M.B., B.L.S., J.M.K., M.P., G.P., and A.R.H.; Investigation: M.B., B.L.S., and J.M.K.; Writing—original draft: M.B.; Writing—review and editing: M.B., B.L.S., J.M.K., B.S., K.S.D., and A.R.H.; Funding acquisition: B.S., K.S.D., and A.R.H.; Supervision: A.P., B.S., K.S.D., and A.R.H.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bhattacharya, M., Spencer, B.L., Kwiecinski, J.M. et al. Collagen binding adhesin restricts Staphylococcus aureus skin infection. Nat Commun 17, 1980 (2026). https://doi.org/10.1038/s41467-026-68788-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68788-4