Abstract
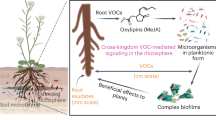
Plants influence each other chemically by releasing leaf volatiles and root exudates, but whether and how these two phenomena interact remains unknown. Here we demonstrate that volatiles that are released by herbivore-attacked leaves trigger plant–soil feedbacks, resulting in increased performance of different plant species. We show that this phenomenon is due to green leaf volatiles that induce jasmonate-dependent systemic defence signalling in receiver plants, which results in the accumulation of beneficial soil bacteria in the rhizosphere. These soil bacteria then increase plant growth and enhance plant defences. In maize, a cysteine-rich receptor-like protein kinase, ZmCRK25, is required for this effect. In four successive year-field experiments, we demonstrate that this phenomenon can suppress leaf herbivore abundance and enhance maize growth and yield. Thus, volatile-mediated plant–plant interactions trigger plant–soil feedbacks that shape plant performance across different plant species through broadly conserved defence signalling mechanisms and changes in soil microbiota. This phenomenon expands the repertoire of biologically relevant plant–plant interactions in space and time and holds promise for the sustainable intensification of agriculture.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The raw sequencing data on soil microbiota and maize transcriptomes are available in the Genome Sequence Archive of the National Genomics Data Center, China National Center for Bioinformation/Beijing Institute of Genomics, Chinese Academy of Sciences (CRA023167, CRA023173 and CRA023181) and are publicly accessible at https://ngdc.cncb.ac.cn/gsa. The illustrative video that describes the protocol for HAC exposure and the subsequent determination of PSF effects on succeeding plants is available via figshare at https://doi.org/10.6084/m9.figshare.28444481 (ref. 84). Source data are provided with this paper.
Code availability
The source code used for the soil microbiota analysis is available via GitHub at https://github.com/YongxinLiu/EasyAmplicon/releases/tag/v1.12.
References
Loreto, F. & D’Auria, S. How do plants sense volatiles sent by other plants? Trends Plant Sci. 27, 29–38 (2022).
Wang, N.-Q., Kong, C.-H., Wang, P. & Meiners, S. J. Root exudate signals in plant–plant interactions. Plant Cell Environ. 44, 1044–1058 (2021).
Kessler, A., Mueller, M. B., Kalske, A. & Chautá, A. Volatile-mediated plant–plant communication and higher-level ecological dynamics. Curr. Biol. 33, R519–R529 (2023).
Guerrieri, E. & Rasmann, S. Exposing belowground plant communication. Science 384, 272–273 (2024).
Maurya, A. K., Patel, R. C. & Frost, C. J. Acute toxicity of the plant volatile indole depends on herbivore specialization. J. Pest Sci. 93, 1107–1117 (2020).
Veyrat, N., Robert, C. A. M., Turlings, T. C. J. & Erb, M. Herbivore intoxication as a potential primary function of an inducible volatile plant signal. J. Ecol. 104, 591–600 (2016).
Hu, L. Integration of multiple volatile cues into plant defense responses. N. Phytol. 233, 618–623 (2022).
Scala, A., Allmann, S., Mirabella, R., Haring, M. A. & Schuurink, R. C. Green leaf volatiles: a plant’s multifunctional weapon against herbivores and pathogens. Int. J. Mol. Sci. 14, 17781–17811 (2013).
Schuman, M. C., Allmann, S. & Baldwin, I. T. Plant defense phenotypes determine the consequences of volatile emission for individuals and neighbors. eLife 4, e04490 (2015).
Frost, C. J. et al. Priming defense genes and metabolites in hybrid poplar by the green leaf volatile cis-3-hexenyl acetate. N. Phytol. 180, 722–733 (2008).
Karban, R., Yang, L. H. & Edwards, K. F. Volatile communication between plants that affects herbivory: a meta-analysis. Ecol. Lett. 17, 44–52 (2014).
Heil, M. & Karban, R. Explaining evolution of plant communication by airborne signals. Trends Ecol. Evol. 25, 137–144 (2010).
Brosset, A. & Blande, J. D. Volatile-mediated plant–plant interactions: volatile organic compounds as modulators of receiver plant defence, growth, and reproduction. J. Exp. Bot. 73, 511–528 (2021).
Pashalidou, F. G. et al. Plant volatiles induced by herbivore eggs prime defences and mediate shifts in the reproductive strategy of receiving plants. Ecol. Lett. 23, 1097–1106 (2020).
Freundlich, G. E., Shields, M. & Frost, C. J. Dispensing a synthetic green leaf volatile to two plant species in a common garden differentially alters physiological responses and herbivory. Agronomy 11, 958 (2021).
Maurya, A. K., Pazouki, L. & Frost, C. J. Priming seeds with indole and (Z)-3-hexenyl acetate enhances resistance against herbivores and stimulates growth. J. Chem. Ecol. 48, 441–454 (2022).
Erb, M. et al. Indole is an essential herbivore-induced volatile priming signal in maize. Nat. Commun. 6, 6273 (2015).
Hu, L., Ye, M. & Erb, M. Integration of two herbivore-induced plant volatiles results in synergistic effects on plant defence and resistance. Plant Cell Environ. 42, 959–971 (2019).
Riedlmeier, M. et al. Monoterpenes support systemic acquired resistance within and between plants. Plant Cell 29, 1440–1459 (2017).
Paudel Timilsena, B., Seidl-Adams, I. & Tumlinson, J. H. Herbivore-specific plant volatiles prime neighboring plants for nonspecific defense responses. Plant Cell Environ. 43, 787–800 (2020).
Howard, M. M., Bass, E., Chautá, A., Mutyambai, D. & Kessler, A. Integrating plant-to-plant communication and rhizosphere microbial dynamics: ecological and evolutionary implications and a call for experimental rigor. ISME J. 16, 5–9 (2021).
Hu, L. et al. Root exudate metabolites drive plant–soil feedbacks on growth and defense by shaping the rhizosphere microbiota. Nat. Commun. 9, 2738 (2018).
Gfeller, V. et al. Plant secondary metabolite-dependent plant–soil feedbacks can improve crop yield in the field. eLife 12, e84988 (2023).
Dudenhöffer, J.-H., Ebeling, A., Klein, A.-M. & Wagg, C. Beyond biomass: soil feedbacks are transient over plant life stages and alter fitness. J. Ecol. 106, 230–241 (2018).
Zhou, Y. et al. Crop rotation and native microbiome inoculation restore soil capacity to suppress a root disease. Nat. Commun. 14, 8126 (2023).
Zhang, H. et al. Cover crop rotation suppresses root-knot nematode infection by shaping soil microbiota. N. Phytol. 245, 363–377 (2025).
Kong, H. G., Song, G. C., Sim, H. J. & Ryu, C. M. Achieving similar root microbiota composition in neighbouring plants through airborne signalling. ISME J. 15, 397–408 (2021).
Kong, C. H. et al. Plant neighbor detection and allelochemical response are driven by root-secreted signaling chemicals. Nat. Commun. 9, 3867 (2018).
Christensen, S. A. et al. The maize lipoxygenase, ZmLOX10, mediates green leaf volatile, jasmonate and herbivore-induced plant volatile production for defense against insect attack. Plant J. 74, 59–73 (2013).
Hu, L., Zhang, K., Wu, Z., Xu, J. & Erb, M. Plant volatiles as regulators of plant defense and herbivore immunity: molecular mechanisms and unanswered questions. Curr. Opin. Insect Sci. 44, 82–88 (2021).
Wang, G. et al. Systemic root–shoot signaling drives jasmonate-based root defense against nematodes. Curr. Biol. 29, 3430–3438 (2019).
Lopes, L. D., Wang, P., Futrell, S. L. & Schachtman, D. P. Sugars and jasmonic acid concentration in root exudates affect maize rhizosphere bacterial communities. Appl. Environ. Microbiol. 88, e0097122 (2022).
Xu, J. et al. Molecular dissection of rice phytohormone signaling involved in resistance to a piercing-sucking herbivore. N. Phytol. 230, 1639–1652 (2021).
Lebeis, S. L. et al. Salicylic acid modulates colonization of the root microbiome by specific bacterial taxa. Science 349, 860–864 (2015).
Hannula, S. E. et al. Persistence of plant-mediated microbial soil legacy effects in soil and inside roots. Nat. Commun. 12, 5686 (2021).
Raaijmakers, J. M. & Kiers, E. T. Rewilding plant microbiomes. Science 378, 599–600 (2022).
Sun, Y. et al. Plant receptor-like protein activation by a microbial glycoside hydrolase. Nature 610, 335–342 (2022).
Piovesana, M. et al. A point mutation in the kinase domain of CRK10 leads to xylem vessel collapse and activation of defence responses in Arabidopsis. J. Exp. Bot. 74, 3104–3121 (2023).
Yadeta, K. A. et al. A cysteine-rich protein kinase associates with a membrane immune complex and the cysteine residues are required for cell death. Plant Physiol. 173, 771–787 (2017).
Hoseinzadeh, A. H. et al. Comparative transcriptome provides molecular insight into defense-associated mechanisms against spider mite in resistant and susceptible common bean cultivars. PLoS ONE 15, e0228680 (2020).
Kalske, A. et al. Insect herbivory selects for volatile-mediated plant–plant communication. Curr. Biol. 29, 3128–3133 (2019).
Karban, R. Plant communication. Annu. Rev. Ecol. Evol. Syst. 52, 1–24 (2021).
Ameye, M. et al. Green leaf volatile production by plants: a meta‐analysis. N. Phytol. 222, 666–683 (2018).
Wang, J., Wu, D., Wang, Y. & Xie, D. Jasmonate action in plant defense against insects. J. Exp. Bot. 70, 3391–3400 (2019).
Frost, C. J. Information potential of an ubiquitous phytochemical cue. N. Phytol. 238, 1749–1751 (2023).
Zhou, H. et al. Efficacy of plant growth-promoting bacteria Bacillus cereus YN917 for biocontrol of rice blast. Front. Microbiol. 12, 684888 (2021).
Deng, C. et al. Molecular mechanisms of plant growth promotion for methylotrophic Bacillus aryabhattai LAD. Front. Microbiol. 13, 4291 (2022).
Passera, A. et al. Characterization of Lysinibacillus fusiformis strain S4C11: in vitro, in planta, and in silico analyses reveal a plant-beneficial microbe. Microbiol. Res. 244, 126665 (2021).
Planchamp, C., Glauser, G. & Mauch-Mani, B. Root inoculation with Pseudomonas putida KT2440 induces transcriptional and metabolic changes and systemic resistance in maize plants. Front. Plant Sci. 5, 719 (2015).
Nomura, K. et al. A bacterial virulence protein suppresses host innate immunity to cause plant disease. Science 313, 220–223 (2006).
Abramovitch, R. B. & Martin, G. B. Strategies used by bacterial pathogens to suppress plant defenses. Curr. Opin. Plant Biol. 7, 356–364 (2004).
Jiang, Q. et al. Two leucine-rich repeat receptor-like kinases initiate herbivory defense responses in tea plants. Hortic. Res. 12, uhae281 (2025).
Frisvold, G. B. How low can you go? Estimating impacts of reduced pesticide use. Pest Manage. Sci. 75, 1223–1233 (2019).
Czepak, M. P. et al. Mechanical damage in the tillering, development and productivity of wheat. Int. J. Plant Soil Sci. 26, 1–7 (2019).
ul Hassan, M. N., Zainal, Z. & Ismail, I. Green leaf volatiles: biosynthesis, biological functions and their applications in biotechnology. Plant Biotechnol. J. 13, 727–739 (2015).
von Merey, G. et al. Dispensing synthetic green leaf volatiles in maize fields increases the release of sesquiterpenes by the plants, but has little effect on the attraction of pest and beneficial insects. Phytochemistry 72, 1838–1847 (2011).
Liu, H. J. et al. High-throughput CRISPR/Cas9 mutagenesis streamlines trait gene identification in maize. Plant Cell 32, 1397–1413 (2020).
Maag, D. et al. 3-β-d-Glucopyranosyl-6-methoxy-2-benzoxazolinone (MBOA-N-Glc) is an insect detoxification product of maize 1,4-benzoxazin-3-ones. Phytochemistry 102, 97–105 (2014).
Ye, M. et al. A constitutive serine protease inhibitor suppresses herbivore performance in tea (Camellia sinensis). Hortic. Res. 10, uhad178 (2023).
Ye, M. et al. Indole primes defence signalling and increases herbivore resistance in tea plants. Plant Cell Environ. 44, 1165–1177 (2021).
Ye, M., Glauser, G., Lou, Y., Erb, M. & Hu, L. Molecular dissection of early defense signaling underlying volatile-mediated defense regulation and herbivore resistance in rice. Plant Cell 31, 687–698 (2019).
Erb, M. et al. Signal signature of aboveground-induced resistance upon belowground herbivory in maize. Plant J. 59, 292–302 (2009).
Chuang, W. P. et al. Herbivore cues from the fall armyworm (Spodoptera frugiperda) larvae trigger direct defenses in maize. Mol. Plant Microbe Interact. 27, 461–470 (2014).
Hu, L. et al. Soil chemistry determines whether defensive plant secondary metabolites promote or suppress herbivore growth. Proc. Natl Acad. Sci. USA 118, e2109602118 (2021).
Glauser, G., Vallat, A. & Balmer, D. Hormone profiling. Methods Mol. Biol. 1062, 597–608 (2014).
Sato, C., Seto, Y., Nabeta, K. & Matsuura, H. Kinetics of the accumulation of jasmonic acid and its derivatives in systemic leaves of tobacco (Nicotiana tabacum cv. Xanthi Nc) and translocation of deuterium-labeled jasmonic acid from the wounding site to the systemic site. Biosci. Biotechnol. Biochem. 73, 1962–1970 (2009).
Liu, Y. et al. Easyamplicon: an easy-to-use, open-source, reproducible, and community-based pipeline for amplicon data analysis in microbiome research. iMeta 2, e83 (2023).
Bolyen, E. et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 37, 852–857 (2019).
Rognes, T., Flouri, T., Nichols, B., Quince, C. & Mahe, F. VSEARCH: a versatile open source tool for metagenomics. PeerJ 4, e2584 (2016).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461 (2010).
Cole, J. R. et al. Ribosomal Database Project: data and tools for high throughput rRNA analysis. Nucleic Acids Res. 42, D633–D642 (2014).
Gephi: an open source software for exploring and manipulating networks. Proc. Int. AAAI Conf. Web Soc. Media 3, 361–362 (2009).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Pertea, M. et al. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 33, 290–295 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Benjamini, Y. & Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 29, 1165–1188 (2001).
Letunic, I., Khedkar, S. & Bork, P. SMART: recent updates, new developments and status in 2020. Nucleic Acids Res. 49, D458–D460 (2021).
Nelson, B. K., Cai, X. & Nebenfuhr, A. A multicolored set of in vivo organelle markers for co-localization studies in Arabidopsis and other plants. Plant J. 51, 1126–1136 (2007).
R Core Team. R: a language and environment for statistical computing. https://cran.r-project.org/doc/manuals/r-release/fullrefman.pdf (2025).
Herve, M. RVAideMemoire: testing and plotting procedures for biostatistics version 0.9-83-7. https://cran.r-project.org/web/packages/RVAideMemoire/ (2023).
Lenth, R. V. Least-squares means: the R package lsmeans. J. Stat. Softw. 69, 1–33 (2016).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 39–140 (2010).
McMurdie, P. J. & Holmes, S. phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8, e61217 (2013).
Hu, L. Video shows the protocol for HAC exposure and subsequent plant soil feedback experiments. figshare https://doi.org/10.6084/m9.figshare.28444481 (2025).
Acknowledgements
We thank R. Li at Zhejiang University for sharing the rice aoc mutant and its WT Xiushui11. This research was supported by the National Key Research and Development Project of China (grant no. 2021YFD1900200 to L.H.); the National Natural Science Foundation of China (grant nos 42377285 to L.H. and 32372775 to M.Y.); Zhejiang Provincial Natural Science Foundation of China (grant nos LR25C160002 to M.Y. and LR25D010001 to L.H.); Hainan Province Science and Technology Special Fund (grant no. ZDYF2024XDNY161 to L.H.); the Elite Youth Program of Chinese Academy of Agricultural Sciences; the 111 Project (grant no. B17039 to J.M.); the Swiss National Science Foundation (grant no. 200355 to M.E.); the Swiss State Secretariat for Education, Research, and Innovation (Project CANWAS to M.E.); and the University of Bern.
Author information
Authors and Affiliations
Contributions
M.Y., J.X., M.E. and L.H. designed the study. J.X., M.E., C.A.M.R., M.Y., L.H., J.M.W. and J.M.R. devised the experimental design and supervised the project. L.H., K.Z., Y.X., X.Z., J.M.W., X.O., Z.W., J.M.R., Y.H., B.M., M.Y. and Z.S. collected and analysed the data. L.H., M.Y., M.E. and J.X. wrote the initial draft of the manuscript. All authors read and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Haiyan Chu, Jurgen Engelberth and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Dispensers emit physiologically relevant levels of GLVs.
a, Experimental setup of the simulated herbivory treatment. b, Volatile emissions from control (Con) and herbivory-induced wild-type (WT) maize plants. The herbivory-induced maize plants were treated by simulated herbivory for 1.5 h. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, ***P < 0.001). c, Diagram of genomic structure of ZmIGL gene regions edited by CRISPR-Cas9. Bars indicate exons and lines represent introns. Scale bar represents 100 bp. d, Volatile emissions from WT plants and igl mutants that were induced by simulated herbivory for 1.5 h. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, ***P < 0.001). e, Diagram of genomic structure of ZmLOX10 gene regions with mutated position indicated. Bars indicate exons and lines represent introns. Scale bar represents 100 bp. f, Volatile emissions from WT plants and lox10 mutants that were induced by simulated herbivory for 1.5 h. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, *P < 0.05; ***P < 0.001). g–i, Release rate of (Z)-3-hexenal (HAL, g), (Z)-3-hexen-1-ol (HOL, h), and (Z)-3-hexenyl acetate (HAC, i) from herbivory-induced WT maize plants and capillary dispensers. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Raw data and exact P values for all comparisons in this figure are provided in the Source Data. j–k, GC/MS selected ion chromatograms of HAL (j) and HOL (k) emitted from herbivory-induced maize plants and capillary dispensers. DMNT, 4,8-dimethyl-1,3(E),7-nonatriene. L.O.D., below the limit of detection.
Extended Data Fig. 2 HAC promotes maize performance via PSFs.
a–c, Chlorophyll content (a), root (b) and total biomass (c) of wild-type maize plants which were growing in soils of control (Con)- or HAC-exposed receiver plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, *P < 0.05; **P < 0.01). d–f, Independent repetition experiment of HAC-induced PSFs in Switzerland. Shoot biomass (d), larval weight gain (e) and leaf damage (f) of WT maize plants which were growing in soils of Con- or HAC-exposed receiver plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, *P < 0.05; **P < 0.01). g–i, HAC triggers PSFs via a receiver plant rather than soil directly. Shoot biomass (g), larval weight gain (h) and leaf damage (i) of wild-type maize plants growing in soils which were directly exposed by Con or HAC volatiles. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. No significant difference was observed between soil types. Raw data and exact P values for all comparisons in this figure are provided in the Source Data.
Extended Data Fig. 3 The influences of exposure frequency, removing and legacy time on PSFs.
a–c, Shoot biomass (a), larval weight gain (b) and leaf damage (c) of wild-type maize plants growing in soils of Con- or HAC-exposed receiver plants. The HAC exposure frequency over different consecutive days was indicated. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (ANOVA followed by pairwise comparisons of FDR-corrected LSMeans, *P < 0.05; ***P < 0.001). d–f, Shoot biomass (d), larval weight gain (e) and leaf damage (f) of wild-type maize plants growing in soils of Con- or HAC-exposed receiver plants. The soils were left in greenhouse with different days after removing the receiver plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (ANOVA followed by pairwise comparisons of FDR-corrected LSMeans, *P < 0.05; **P < 0.01; ***P < 0.001). g–i, Shoot biomass (g), larval weight gain (h) and leaf damage (i) of wild-type maize plants growing in soils of Con- or HAC-exposed receiver plants which were removed at different times after exposure. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (ANOVA followed by pairwise comparisons of FDR-corrected LSMeans, *P < 0.05; **P < 0.01; ***P < 0.001). Raw data and exact P values for all comparisons in this figure are provided in the Source Data.
Extended Data Fig. 4 Herbivory-induced plant volatiles (HIPVs) from rice or tea plants promote the performance and resistance of succeeding maize plants.
a–c, Growth phenotypes (a), shoot biomass (b), caterpillar weight gain (c) of wild-type (WT) maize plants growing in soils of Con- or HIPV-exposed rice WT plants or aoc mutants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (ANOVA followed by pairwise comparisons of FDR-corrected LSMeans, *P < 0.05; **P < 0.01; ***P < 0.001). d–e, Shoot biomass (d) and caterpillar weight gain (e) of WT maize plants growing in soils of Con- or HIPV-exposed tea receiver plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, *P < 0.05; ***P < 0.001). Raw data and exact P values for all comparisons in this figure are provided in the Source Data.
Extended Data Fig. 5 Chemical and physical properties of field soils.
The soil pH (a), content of sand (b), clay (c), silt (d), dissolved organic carbon (DOC, e), available nitrogen (N, f), potassium (K, g), phosphorous (P, h), copper (Cu, i), zinc (Zn, j), magnesium (Mg, k), manganese (Mn, l), iron (Fe, m), silicon (Si, n), molybdenum (Mo, o) and nickel (Ni, p) in field soils. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Raw data for this figure are provided in the Source Data. DW, dry weight.
Extended Data Fig. 6 Soil fungi in the rhizosphere of HAC-exposed maize receiver plants.
a, The phytohormone concentrations in the rhizosphere soil of receiver plants after HAC exposure. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (two-sided Student’s t test, *P < 0.05; **P < 0.01). Raw data and exact P values for all comparisons in this panel are provided in the Source Data. b, The information of lox8 maize mutant. Diagram of genomic structure of ZmLOX8 gene with transposon insertion indicated. Bars indicate exons and lines represent introns. Scale bar represents 100 bp. c–e, Concentrations of 12-oxophytodienoic acid (OPDA, c), jasmonic acid (JA, d), and JA-isoleucine (JA-Ile, e) in wild-type (WT) and lox8 mutant plants after HAC exposure. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (ANOVA followed by pairwise comparisons of FDR-corrected LSMeans, *P < 0.05; **P < 0.01; ***P < 0.001). Raw data and exact P values for all comparisons in this panel are provided in the Source Data. FW, fresh weight. f, Shannon index of fungal communities in the rhizosphere of control (Con)- or HAC-exposed maize receiver plants There are eight biological replicates for each treatment. Data points represent individual replicate samples. g, Unconstrained PCoA with Bray-Curtis distance showing that the rhizosphere fungal communities of Con-exposed maize receiver plants separate from those of HAC-exposed receiver plants in the first axis (P < 0.01, permutational multivariate analysis of variance [PERMANOVA] by Adonis). There are eight biological replicates for each treatment. Data points represent individual replicate samples. h–i, Phylum- (g) and genus (h)-level distribution of fungus communities in the rhizosphere of Con- and HAC-exposed WT receiver plants. There are eight biological replicates for each treatment. j, Manhattan plot showing fungal OTUs enriched in the rhizosphere of Con- or HAC-exposed receiver plants. Each dot or triangle represents a single OTU. OTUs enriched in Con- or HAC-exposed soil are represented by filled or empty triangles, respectively. Differential OTU abundance was analyzed using two-sided Wilcoxon rank-sum tests, with P values corrected by the FDR method (P < 0.05). OTUs are arranged in taxonomic order and colored according to the phylum. CPM, counts per million. k, Rhizofungal co-occurrence networks of Con- and HAC-exposed receiver plants. The networks were constructed based on Spearman correlation analysis of taxonomic profiles (P < 0.05). The nodes in the network represent genus and links indicate potential microbial interactions. Node size is proportional to degree. l, Soil microbiota topological features of co-occurrence networks in the rhizosphere of Con- or HAC-exposed receiver plants. NaN, not a Number.
Extended Data Fig. 7 The influence of soil bacteria on plant growth and resistance.
a–l, Shoot biomass (a–d), larval weight gain (e–h), and leaf damage (i–l) of wild-type (WT) maize plants inoculated with 18 bacterial strains which correspond to the OTUs that are enriched in the rhizosphere of HAC-exposed plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between bacteria inoculation and buffer treatments (two-sided Student’s t test, *P < 0.05; **P < 0.01; ***P < 0.001). m–n, Bacteria complementation restores HAC-triggered PSF effects. Shoot biomass (m) and caterpillar weight gain (n) of WT maize plants growing in soils of control (Con)-exposed receiver plants. The soils were individually complemented with different bacteria strains. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Different letters denote significant differences between treatments (ANOVA followed by multiple comparisons of FDR-corrected LSMeans, P < 0.05). Raw data and exact P values for all comparisons in this figure are provided in the Source Data.
Extended Data Fig. 8 Soil bacteria in the rhizosphere of wild-type plants and lox8 mutants after HAC exposure.
a–b, Shoot biomass (a) and caterpillar weight gain (b) of wild-type (WT) plants growing in soils of control (Con)- or JA-complemented lox8 receiver plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between treatments (ANOVA followed by pairwise comparisons of FDR-corrected LSMeans, *P < 0.05). Raw data and exact P values for all comparisons in this panel are provided in the Source Data. Soils were either left untreated or X-ray sterilized. c, Shannon index of bacterial communities in the rhizosphere of Con- or HAC-exposed WT and lox8 plants There are eight biological replicates for each treatment. Data points represent individual replicate samples. d, Unconstrained PCoA with Bray-Curtis distance of the rhizosphere bacterial communities of WT and lox8 plants after Con or HAC exposure (P < 0.001, permutational multivariate analysis of variance [PERMANOVA] by Adonis). There are eight biological replicates for each treatment. Data points represent individual replicate samples. e, Rhizobacterial co-occurrence networks of Con- and HAC-exposed receiver plants. The networks were constructed based on Spearman correlation analysis of taxonomic profiles (P < 0.05). The nodes in the network represent genus and links indicate potential microbial interactions. Node size is proportional to degree.
Extended Data Fig. 9 Soil bacteria change the expression of receptor-like kinases.
a, Expression levels of nine receptor-like kinase, Zm00001eb291400, Zm00001eb304650, Zm00001eb323660, Zm00001eb323640, Zm00001eb334650, Zm00001eb325290, Zm00001eb239210, Zm00001eb442380 and Zm00001eb325300, in maize roots after inoculation with 12 bacterial strains which correspond to the OTUs that are enriched in the rhizosphere of HAC-exposed plants. Data are presented as mean + SEM. The exact number of biological replicates is indicated on each bar. Data points represent individual replicate samples. Asterisks denote significant differences between bacteria inoculation and buffer treatments (two-sided Student’s t test, *P < 0.05; **P < 0.01; ***P < 0.001). Raw data and exact P values for all comparisons in this figure are provided in the Source Data. b, Correlations between bacteria-triggered plant growth, herbivore resistance and the expression of nine receptor-like kinases. Relative shoot biomass, larval weight, and damage area (bacteria/control) is correlated with relative expression levels of nine receptor-like kinase genes (bacteria/control) after inoculation with 12 bacterial strains which correspond to the OTUs that are enriched in the rhizosphere of HAC-exposed plants. Exact P values and Pearson’s r of correlations are shown.
Extended Data Fig. 10 Protein alignment of ZmCRK25 with homologous proteins in Arabidopsis.
a, Schematic representation of ZmCRK25 domain composition and organization based on conserved domain analysis. The numbers indicate amino acids positions of the ZmCRK25 protein domains. The positions of the signal peptide (red color), two salt stress response/antifungal domains (stress-antifung), transmembrane (blue color), protein kinase (Pkinase), and low complexity region (purple color) are shown. b, The amino acid sequence of ZmCRK25 was aligned by ClustalW with homologous sequences of CRKs in Arabidopsis: AtCRK25 (AT4G05200.2), AtCRK10 (AT4G23180.1), AtCRK29 (AT4G21410.3). c, Knockout of ZmCRK25. Diagram of genomic structure of ZmCRK25 gene regions edited by CRISPR-Cas9. Bars indicate exons and lines represent introns. Scale bar represents 100 bp.
Supplementary information
Supplementary Data 1 (download XLSX )
Microbiome statistics. This file contains lists for the taxonomies, sequences and statistical details (fold change, abundance and edgeR’s likelihood ratio statistic with the corresponding FDR-corrected P values) of the differentially abundant bacterial and fungal OTUs in the rhizosphere of control (Con)- and HAC-exposed plants.
Supplementary Data 2 (download TXT )
Sequences of the 18 selected HAC rhizosphere-enriched bacteria for the inoculation experiments.
Supplementary Data 3 (download XLSX )
Differentially expressed genes of maize plants growing in soils of control- or HAC-exposed receiver plants by RNA-sequencing.
Supplementary Table 1 (download XLSX )
QRT-PCR primers of target genes.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, L., Zhang, K., Xu, Y. et al. Herbivory-induced green leaf volatiles increase plant performance through jasmonate-dependent plant–soil feedbacks. Nat. Plants 11, 1001–1017 (2025). https://doi.org/10.1038/s41477-025-01987-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41477-025-01987-x
This article is cited by
-
Crystal ball time
Nature Plants (2026)
-
Root-knot nematode Meloidogyne incognita uses secondary-metabolite-mediated soil microbiome shifts to locate host plants
Nature Plants (2026)
-
Caterpillar-induced plant-soil feedback affects resistance in wild and cultivated cabbage
Plant and Soil (2026)
-
Airborne cues shape soil legacies
Nature Plants (2025)
-
Rhizosphere Streptomyces confers dual-mode resistance to root-knot nematodes through nematicidal metabolites and JA-mediated immunity in maize
Soil Ecology Letters (2025)