Abstract
Pre-mRNA secondary structures are hypothesized to regulate RNA processing pathways, but such structures have been difficult to visualize in vivo. Here, we characterize Saccharomyces cerevisiae pre-mRNA structures through transcriptome-wide dimethyl sulfate probing, enriching for low-abundance pre-mRNA through splicing inhibition. We cross-validate structures found from phylogenetic and mutational studies and identify structures within the majority of measured introns (79 of 88). We find widespread formation of ‘zipper stems’ between the 5′ splice site and branch point, ‘downstream stems’ between the branch point and the 3′ splice site, and previously uncharacterized long stems that distinguish pre-mRNA from spliced mRNA. Multi-dimensional chemical mapping reveals intron structures that independently form in vitro without the presence of binding partners, and structure ensemble prediction suggests that such structures appear in introns across the Saccharomyces genus. We further develop a high-throughput functional assay to characterize variants of RNA structure (VARS-seq), applying it to 135 sets of stems across 7 introns, identifying structured elements that alter retained intron levels at a distance from canonical splice sites. This transcriptome-wide inference of intron RNA structures introduces alternative paradigms and model systems for understanding how pre-mRNA folding influences gene expression.
Similar content being viewed by others
Main
Introns are widely prevalent features of eukaryotic genomes. Many genes contain long stretches of these non-coding RNA sequences, which are excised from mRNA precursors through RNA splicing. In the splicing reaction, the spliceosome precisely recognizes and positions three key intronic sequences termed the 5′ splice site, the branch point and the 3′ splice site, carrying out the two catalytic steps required for removing introns (Fig. 1a)1. Despite the prevalence of introns, the functional roles for many of them remain underexplored. In some cases, intron sequences beyond splice sites regulate gene expression by controlling splicing rates and promoting alternative splicing2,3. In addition, introns can contain functional non-coding RNAs, alter pre-mRNA decay rates and facilitate the evolution of new genes4,5,6,7,8,9,10.
a, Schematic of RNA splicing. b, Schematic of splicing inhibition, followed by the DMS-MaPseq experiment. c, Accumulation of pre-mRNA for RPL36B and MATa1, assessed using RT–PCR. This experiment was repeated with a biological replicate, with similar results. d, Read coverage across intron-containing pre-mRNA with (purple) and without (orange) pladB treatment. e, The RI fraction with and without pladB treatment. Points are plotted on a log scale, and the equal retained fraction from both conditions is indicated by the dashed line. RI, retained intron. f, Comparison of reactivity values for three introns with and without pladB treatment.
Intron RNA sequences are complex macromolecules that occupy an ensemble of secondary structures, including single- and double-stranded regions. Intron secondary structure can regulate splice-site selection and splicing efficiency, and structure in pre-mRNA can have a role in numerous other nuclear processes, such as RNA editing and RNA end processing11,12. S. cerevisiae provides a useful model system for studying the role of pre-mRNA secondary structures because the catalytic steps of splicing, spliceosomal machinery, RNA modification processes, and RNA-decay pathways are highly conserved across eukaryotes, from S. cerevisiae to humans1,13,14. Research on S. cerevisiae has revealed that intron stems called zipper stems can link the 5′ splice site to the branch point15,16,17,18, and hairpins can lower the effective distance between the branch point and 3′ splice site, facilitating efficient splicing19. In addition, pre-mRNA structures can bind to their protein products to regulate autogenous gene expression control at the level of splicing7,20,21,22.
These studies highlight various functions of pre-mRNA structures, but it remains unclear how generally these findings extend across the transcriptome. A comprehensive experimental survey of structures in introns across the transcriptome of any organism is lacking, and functional data that test the role of native intron structures are limited. Although scans for covariation have identified some potentially functional structures23, others uncovered in functional studies have been missed by these scans20,21,22,24. Moreover, while sequence variants have been used to test some intron structures3,25, the depth of mutagenesis has been limited26. At this stage, the structural landscape and functional roles for intron secondary structures across any transcriptome are only sparsely determined.
In vivo chemical probing provides an avenue for obtaining deep structural data on RNA across the transcriptome27. However, owing to their low abundance, introns typically escape structure detection and quantification by these methods. Here, we use splicing inhibition to enrich for unspliced pre-mRNA in transcriptome-wide structure probing experiments, identifying patterns in reactivity that distinguish structures in S. cerevisiae introns from coding regions in vivo. We combine this structural information with phylogenetic analysis across yeast genomes, and we develop a strategy for high-throughput functional analysis called VARS-seq to evaluate levels of unspliced and spliced RNA for variants of 135 sets of stems in 7 introns. Our combined structural, functional and evolutionary analysis provides an atlas of pre-mRNA structures, serving as a foundation for understanding the roles of introns in gene regulation.
Results
Transcriptome-wide structure probing with splicing inhibition in yeast
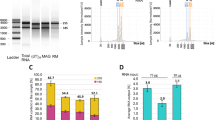
Structure probing experiments such as dimethyl sulfate (DMS) mutational profiling with sequencing (DMS-MaPseq)27 can be used to evaluate the formation of RNA secondary structures across the transcriptome. However, because splicing proceeds rapidly in yeast28, the coverage of pre-mRNA is limited in existing DMS-MaPseq datasets for S. cerevisiae, preventing the analysis of introns. Recently, S. cerevisiae strains with substitutions in the U2 snRNP component encoded by HSH155 (SF3B1 in human) have been developed that sensitize yeast to splicing inhibition by pladienolide B (pladB)29,30. Typically, splicing proceeds in two catalytic steps: the 5′ splice site and the branch point interact in the first catalytic step, and the 5′ splice site and the 3′ splice site participate in the second catalytic step (Fig. 1a)1. Using a pladB-sensitive yeast strain, we stalled the assembling pre-spliceosome (A complex) before the first catalytic step of splicing, chemically inhibiting splicing for 1 h before DMS treatment (Fig. 1b).
Treatment with pladB led to the accumulation of pre-mRNA, increasing the proportion of unspliced mRNA from RPL36B and MATa1 (Fig. 1c). DMS-MaPseq revealed increased intron retention in pladB-treated cells for many genes; intron read coverage increased from negligible levels in untreated cells to levels approaching those of coding regions (Fig. 1d). Most intron-containing genes showed increased intron retention following pladB treatment, yielding a median ratio of retained intron fraction (RI fraction) of 10.5 when comparing pladB and control conditions (Fig. 1e). Only 1 intron had an RI fraction less than 0.05, and 180 introns had an RI fraction above 0.5. Without pladB treatment, 143 of 288 annotated introns had an RI fraction less than 0.05, and only 28 introns had an RI fraction above 0.5. Longer introns (more than 200 nucleotides (nt)) and introns in ribosomal protein genes (RPGs) exhibited increased RI fractions upon pladB treatment compared with those of other introns (Supplementary Fig. 1a,b). We noted no coverage bias between the ends of introns (Supplementary Fig. 1c), suggesting that they are not partial degradation byproducts of nonsense-mediated decay (NMD), which predominantly involves XRN1-mediated 5′ to 3′ decay31,32. RI fractions for introns detected with and without pladB treatment are shown in Supplementary Table 1.
To assess the quality of our DMS-MaPseq reactivity data for introns, we examined control RNAs with known structure. DMS modifies A and C residues, leading to substitution frequencies of 2.7% and 2.3%, respectively, whereas unreactive G and U residues exhibited substitution frequency rates closer to the background level (0.3–0.4%) (Extended Data Fig. 1a). Data for rRNA aligned with prior experiments, showing that substitution frequencies for the 18S and 25S rRNA were highly correlated with values from an earlier DMS-MaPseq study (r2 = 0.83, Extended Data Fig. 1b)27. Moreover, DMS reactivity values, a proxy for RNA single-strandedness, permitted successful classification of accessible and inaccessible rRNA residues, with an area under the curve (AUC) of 0.91 (Extended Data Fig. 1b). Finally, the reactivity profiles for stem loops in HAC1 and ASH1 mRNA align with well-characterized secondary structures for these regions27, with base-paired residues being less reactive than residues in loops (Extended Data Fig. 1c).
Assessing structures from DMS-MaPseq after splicing inhibition
Using data generated from these DMS-MaPseq experiments, we predicted secondary structures with RNAstructure33,34. We also assigned confidence estimates for individual helices by performing non-parametric bootstrapping on reactivity values, using an approach that has empirically improved the quality of stem predictions35. To calibrate helix confidence estimate cut-offs, we made DMS-guided structure predictions for structured RNAs in our DMS-MaPseq dataset, including rRNAs, small nuclear RNAs (snRNAs), tRNAs and mRNA segments. The implementation of helix confidence estimate thresholds improved the positive predictive value (PPV) and F1 score for stem predictions (Extended Data Fig. 2). When using reactivity data for DMS-guided structure predictions with a helix confidence estimate threshold of 70%, stems with at least 5 base pairs (bp) had a PPV of 82.3% (Supplementary Table 2). This approach remained effective even for larger RNAs, such as the U1 snRNA (Extended Data Fig. 2d,e and Supplementary Table 2). We therefore designated stems with helix confidence estimates of >70% and at least 5 bp as high-confidence stems and focused subsequent analyses on them.
We next assessed the reproducibility of these high-confidence stems. Across all 259 introns, we identified 425 such stems, with 331 (77.6%) agreeing between replicate experiments. Introns with higher sequencing coverage had better replicability between experiments (Extended Data Fig. 3a). In the 88 introns with higher reactivity correlation between replicates (r2 > 0.6), 88.0% of high-confidence stems were consistent between replicates (Extended Data Fig. 3b). To confirm the presence of these stems, we carried out targeted probing for 30 introns to obtain higher-coverage data. DMS profiles for 24 of these introns yielded r2 > 0.9 between replicates of targeted probing (Extended Data Fig. 3c). In these introns, 90.5% of high-confidence stems identified by transcriptome-wide DMS-MaPseq matched structures from targeted probing (Extended Data Fig. 3d and Supplementary Fig. 2). To verify that DMS data informed predicted structures, we extracted RNA from cells and probed it with DMS after heat denaturation, revealing that only 10.4% of these stems were present in denatured RNA (Extended Data Fig. 3e and Supplementary Fig. 2). We focused all further analyses on the 88 introns with r2 > 0.6 from DMS-MaPseq, limiting analysis to high-confidence stems. We identified 301 stems across these 88 introns, with the majority (79 of 88) having at least 1 stem. With additional DMS-guided structure prediction approaches, we found that no introns included high-confidence pseudoknots, and all introns tested for multiple structures were best explained by a single conformation (see Supplementary Text). Supplementary Table 1 details introns’ replicate correlation, high-confidence stems and structures from transcriptome-wide and targeted probing.
To assess the impact of pladB treatment on intron folding, we examined the three introns that had sufficient DMS-MaPseq coverage, both with and without pladB treatment. The reactivity profiles of these introns were broadly similar, with many highly reactive positions shared between conditions (Fig. 1f). However, some intervals in the introns of RPL26B and RPS13 exhibited shifted reactivity profiles upon pladB treatment, surpassing the variation seen between replicates (Supplementary Fig. 3). When comparing conditions with and without pladB treatment, the reactivities had r2 values of 0.58, 0.86 and 0.71 for the introns in RPL26B, RPL28 and RPS13, respectively. To evaluate whether these differences affected structure prediction, we identified high-confidence stems predicted for these three introns with and without pladB treatment. We found that all high-confidence stems were shared across conditions (Supplementary Fig. 4), suggesting that structures obtained after pladB treatment could provide insights into the structures in untreated S. cerevisiae.
DMS support for previously reported yeast intron structures
Structures have been proposed for some introns in S. cerevisiae on the basis of functional experiments7,15,16,19,20,21,22,24, solved spliceosome structures including introns25,36, covariation scans that pinpoint functional base pairs23 and de novo prediction methods based on sequence conservation37. Using our DMS data, we evaluated the support for the presence of these proposed structures in vivo.
We first focused on seven introns that included regulatory structures identified in functional experiments: introns in RPS17B15,16, RPS23B19, RPL32 (ref. 20), RPS9A7, RPS14B21,22, RPL18A24 and RPS22B24. Most of these structures were supported by our DMS data (Fig. 2a–e), with only two exceptions (Supplementary Fig. 5a,b), validating structures found through experiments assessing mutants and compensatory mutants (Fig. 2g and Supplementary Text). By contrast, structures identified through computational predictions in Hooks et al.37 using CMfinder38, RNAz39 and Evofold40 showed low support from DMS data (Supplementary Fig. 6a–f), suggesting that these approaches are less reliable for structure identification (Fig. 2g and Supplementary Text). DMS-guided predictions include some false negatives (33.8% false negative rate, Extended Data Fig. 2a,b) and will miss structures that represent a minor portion of introns’ structural ensembles.
a–f, Helix confidence estimates and covariation for intron structures reported in RPL18A24 (a), RPS23B19 (b), RPS14B21 (c), the first intron in RPL7A (d), RPS9A (e) and RPS9B7 (f). Secondary structures are colored according to DMS reactivity, and helix confidence estimates are depicted as green percentages. The 5′ splice site, branch point and 3′ splice site sequences are circled in purple, blue and yellow, respectively. Covarying base pairs in RPS9A, RPS9B and RPL7A are marked with green boxes. g, Summary of the percentage of supported base pairs in structures proposed in prior functional studies, R-scape scans for covariation in multiple sequence alignments (MSAs), and other approaches using sequence alignments to pinpoint structures (Evofold, RNAz and cMfinder). A base pair is supported if it is included in a stem whose helix confidence estimate is >70%, and base-pair support statistics are computed on the basis of all base pairs in proposed structures (functional experiments, Evofold, RNAz, cMfinder) or significantly covarying base pairs (R-scape covariation).
We next evaluated structures detected using R-scape, which identifies pairs of residues with significant covariation compared with phylogenetic sequence backgrounds. In particular, we evaluated DMS support for the covarying residues identified in Gao et al.23 and those identified in our R-scape scan using less stringent cut-offs (Supplementary Fig. 7 and Methods). Seven of the eight S. cerevisiae introns that encode snoRNAs41 included significant covariation, suggesting that the remaining introns with covariation could also encode functional structures. Most intron structures with covarying residues were supported by DMS data (Fig. 2d–f and Supplementary Fig. 8), with one exception (Supplementary Fig. 5c and Supplementary Text), suggesting that covariation detected by R-scape42 can reliably identify structures (Fig. 2g). Thus, we found that, compared with structures predicted by CMfinder38, RNAz39 and Evofold40, those identified through functional experiments and R-scape42 covariation were more consistently supported by DMS data.
We also used our DMS data to identify high-confidence stems in tRNA introns in S. cerevisiae. Given that these tRNA introns are not processed by the standard spliceosome machinery, pladB treatment did not lead to their accumulation. Despite this, ten tRNA introns had sufficient coverage for structure analysis. Each of these introns included at least one high-confidence stem (Supplementary Fig. 9), aligning with prior pre-tRNA structure models, and again confirming structures from prior structural and functional experiments43,44.
Structural features found by probing S. cerevisiae pre-mRNA
Having established that our DMS-guided structure predictions are in agreement with previously identified intron structures, we next identified enriched structural features in our data. First, in vivo probing supports the widespread formation of zipper stems, that is, stems that reduce the distance between the 5′ splice site and branch point. Potential zipper stems have been noted in various introns17, and a zipper stem in the intron from RPS17B has been shown to be essential for efficient splicing15,16. To identify zipper stems across introns, we sought to precisely define the positional constraints on zipper stem formation, modeling intronic stems with varying linker lengths in the context of the A-complex spliceosome (Extended Data Fig. 4a,b and Methods). On the basis of this modeling, we defined zipper stems as the longest stem comprising 42 to 85 nt linking the stem, the 5′ splice site and branch point sequences. We found that high-confidence zipper stems (those with at least 6 bp and a minimum 70% helix confidence estimate) were predicted for 32 of 88 introns (Fig. 3a). In our targeted DMS probing of 30 introns, zipper stems were present in 25 of them (examples in Supplementary Fig. 2), whereas there were no zipper stems present when DMS data were collected after unfolding with heat denaturation, as expected (Extended Data Fig. 3d and Supplementary Fig. 2).
a, Reactivity support for zipper stems in RPL7A and RPS11A, and a pie chart representing the fraction of introns with zipper stems. 5′SS, 5′ splice site. b, Reactivity support for downstream stems connecting the branch point and 3′ splice site in RPL40B and RPS14A, and a pie chart representing the fraction of introns with downstream stems. 3′SS, 3′ splice site. c,The secondary structure of the intron in RPL28, predicted by RNAstructure guided by DMS reactivity. d, The top-scoring 3D model for the RPL28 intron in the context of the A complex spliceosome (PDB ID: 6G90)74, modeled using the secondary structure derived from DMS-MaPseq. e–h, Comparisons between introns and coding regions for the following secondary structure features: the Gini coefficient (e), normalized maximum extrusion from ends (f), longest stem length (g) and average helix confidence estimate (h). P values were computed using one-sided Wilcoxon ranked-sum tests to compare classes. In box plots, the median is the center white point, box limits are the 25th (Q1) and 75th (Q3) percentiles, and whiskers extend to the smallest and largest values that fall within 1.5 times the interquartile range below Q1 and above Q3. i, Proportion of nucleotides in high-confidence stems from sequence intervals surrounding the canonical 5′ splice site, branch point and 3′ splice site sequences. Intron sequence positions external to these intervals were marked as ‘distal from SS.’ j, Comparison of the protection by high-confidence stems between nucleotides in cryptic splice sites versus surrounding nucleotides. An example from RPL34A is shown with a stem occluding a cryptic 3′ splice site (red bracket). Secondary structures are colored by DMS reactivity and are annotated with helix confidence estimates. The 5′ splice site, branch point and 3′ splice site sequences are circled in purple, blue and yellow, respectively. In i–j, P values were computed with Chi-squared tests on 2 × 2 contingency tables.
We next noted the presence of stems between the branch point and 3′ splice site across introns, which we term downstream stems. These stems are proposed to facilitate splicing by decreasing the effective distance between the branch point and 3′ splice site19. We found that downstream stems (those with at least 6 bp and a minimum 70% helix confidence estimate) are present in 21 of 88 introns (Fig. 3b). As with zipper stems, the reactivity profiles for downstream stems (for example, in introns in RPL40B, RPS14A and RPS23B) show higher reactivity in loop or junction residues than in base-paired positions, supporting their in vivo formation (Figs. 2b and 3b).
DMS reactivity patterns in some introns suggest the presence of elaborate extended secondary structures with long stems and multiway junctions. For example, the intron from RPL28 features a zipper stem and a downstream stem, and the pre-mRNA includes a three-way junction and long stems of more than 10 bp throughout the intron (Fig. 3c). When modeling the RPL28 intron in the context of the A-complex spliceosome, we observe that these structures enable internal intronic stems to extend beyond the core spliceosome, potentially allowing binding partners to avoid steric clashes with the splicing machinery (Fig. 3d). We note that the three-dimensional (3D) modeling approach here makes simplifying assumptions and samples only a few of many possible conformations (see Methods). However, sampled conformations suggest that intron structures can extend beyond the spliceosome, even for shorter introns such as that in RPL36B, facilitated by stems through the intron (Extended Data Fig. 4c).
Comparing intron structures with coding regions and unspliced decoys
Our DMS-guided structure predictions revealed that the properties of intron secondary structures are distinct from those of mRNA coding regions. As found previously in mammalian structure probing45, intron regions have higher Gini coefficients, a measure quantifying the extent to which reactivity values diverge from an even distribution (Fig. 3e). Therefore, introns include more non-random secondary structure elements than do coding regions. Additionally, intron structures predicted using DMS data extend further from their sequence endpoints than do coding-region structures, which we measure as the normalized maximum extrusion from ends (MEE) (Fig. 3f and Methods). Introns also include longer stems, with some high-confidence stems extending to more than 20 bp (Fig. 3g). Finally, intron stems have higher helix confidence estimates on average, suggesting that reactivity values better support intron secondary structures (Fig. 3h). These conclusions were reproduced when analyzing data from each DMS-MaPseq replicate separately (Supplementary Fig. 10).
The secondary structure patterns enriched in introns might distinguish spliced introns from other transcribed sequences that do not splice efficiently. To explore this possibility, we assembled a set of unspliced decoy introns in S. cerevisiae that included consensus 5′ splice site, branch point and 3′ splice site sequences in positions that matched canonical introns′ length distributions (Extended Data Fig. 5a). DMS-guided structure predictions for authentic intron sequences enriched for more stable zipper stems with lower folding free energy (ΔG), more stable downstream stems, longer stems and higher MEE (Extended Data Fig. 5b). In contrast to spliced introns, the DMS-guided secondary structures of unspliced decoys were not enriched for these features (Extended Data Fig. 5b). We conclude that, compared with coding regions and unspliced decoys, introns in S. cerevisiae adopt extended secondary structures with long stems, as supported by DMS data.
The enrichment of structural features in introns compared with coding regions and unspliced decoys suggests that these structures could have a functional role. To explore this possibility, we analyzed the placement of high-confidence stems in introns relative to the positions of canonical and cryptic splice sites. To evaluate structures surrounding canonical splice sites, we first identified sequence intervals in pre-mRNA surrounding the 5′ splice site, branch point and 3′ splice site, where structural conflicts with the spliceosome are anticipated (Methods). We noted that these intervals were significantly depleted of high-confidence stems when generating structures for introns in the context of the surrounding pre-mRNA sequence (Fig. 3i). However, the presence or absence of high-confidence stems involving these intervals near splice sites was not correlated with the fraction of spliced constructs, suggesting that other factors have a dominant role (Supplementary Fig. 11a). We next identified cryptic splice sites across introns, expecting that these sites could be occluded by structure. Indeed, in multiple introns, downstream stems between the branch point and 3′ splice site occluded cryptic 3′ splice sites (Supplementary Fig. 11b), aligning with prior work on an intron stem in RPS23B that blocks a cryptic 3′ splice site (Fig. 2b)19. Across introns, we noted that cryptic 3′ splice sites were enriched for high-confidence stems (Fig. 3j and Supplementary Fig. 11c), suggesting that these stems have a role in enforcing splicing fidelity by repressing the use of incorrect 3′ splice sites.
Evaluating S. cerevisiae intron RNA structures in vitro
To determine whether intron structures observed in vivo can form in vitro even when potential protein binding partners are missing, we probed isolated introns transcribed in vitro with DMS. For this, we used mutate-and-map readout through next-generation sequencing (M2-seq46), which can identify base-pairing residues in addition to providing average per-residue accessibility data. We applied in vitro M2-seq to five introns with zipper stems in DMS-MaPseq. In the case of the introns in QCR9 and RPL36B, Z-score plots from in vitro M2-seq included off-diagonal signals indicative of the presence of stems, and high base-pairing probabilities support the formation of these stems in vitro (Fig. 4a,b, Extended Data Fig. 6a,b and Supplementary Text). Secondary structures from M2-seq agree with the stems observed in vivo, with most high-confidence stems shared between these structures (Fig. 4c,d and Extended Data Fig. 6c,d). For the introns in RPS11A, RPL37A and RPS7B, although M2-seq Z-scores did not include off-diagonal signals, helix confidence estimates from M2-seq data supported stems observed in vivo (Extended Data Fig. 7). We additionally assayed intron structures in vitro by refolding RNA extracted from yeast and probing accessible residues with DMS (Methods). However, because only 3 introns reached a between-replicate reactivity correlation of r2 > 0.6 from this experiment (Supplementary Fig. 12), we focused our analyses on cases studied with in vitro M2-seq. Our in vitro M2-seq results suggest that intron sequences can form structures found in vivo, even outside the nucleus and without protein binding partners.
a, In vitro M2-seq Z scores for the intron in QCR9, with peaks representing helices annotated in red. b, In vitro chemical reactivity base-pairing probabilities for the QCR9 intron using one- and two-dimensional (1D and 2D) chemical reactivity from M2-seq, with peaks representing helices annotated in black. c,d, Secondary structure predictions guided by 1D and 2D DMS probing data for the intron in QCR9 from in vitro M2-seq (c) and in vivo DMS-MaPseq (d).
Structural landscape for S. cerevisiae introns
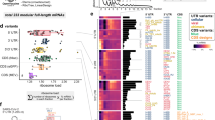
To understand the distribution of structural patterns in introns across the yeast transcriptome, we clustered introns on the basis of their structural features. For each intron, we assembled a full set of features using probing data (Supplementary Table 1): zipper stem and downstream stem free energy (Methods), the MEE, the longest stem length, the average helix confidence estimate, the maximum Gini coefficient window, and the accessibility across the 5′ splice site, branch point and 3′ splice site.
The clustered heatmap (Fig. 5) depicts the global distribution of secondary structure features across all introns with sufficient sequencing coverage. The first class of introns includes 24 introns with zipper stems; within this class, more stable zipper stems clustered together (class 1, yellow in Fig. 5). Class 2 includes nine introns that have both a zipper stem and downstream stem, with high average helix confidence estimates (class 2, brown in Fig. 5). The next class includes 12 introns with downstream stems (class 3, purple in Fig. 5). In S. cerevisiae, intron lengths are bimodal, with a set of shorter introns (shorter than 200 nt) and a set of longer introns (with most between 400 and 500 nt)47. Our structure probing data had sufficient coverage for 7 short introns and 81 long introns. Of the long introns, 55.6% include either a zipper stem or downstream stem in classes 1–3. Class 4 includes 13 structured long introns that include other structures with high helix confidence and long stems (red in Fig. 5). By contrast, the next class includes less structured long introns, which have neither high Gini coefficients nor long stems (Class 5, green in Fig. 5). Class 6 includes most short introns, which tend to be depleted of stems, without zipper stems, downstream stems or high helix confidence estimates (orange in Fig. 5). Finally, class 7 includes structured short introns with at least one high-confidence stem (blue in Fig. 5).
Heatmap and dendrogram summarizing intron structural classes, with hierarchical clustering based on secondary structure features. In addition to the features displayed on the heatmap, flags indicating whether zipper stems and downstream stems were present were included as features for hierarchical clustering. ΔG, folding free energy; len, length; SS, splice site; BP, branch point.
A high-throughput assay to test the function of intron stems
To assess the influence of intron structures on gene expression, we developed a high-throughput assay for evaluating variants of RNA structure (VARS-seq). Here, we used VARS-seq to measure spliced and unspliced mRNA levels for intron structure variants, anticipating that these structures might influence splicing or pre-mRNA decay rates. Structures that slow pre-mRNA decay or splicing could lead to an accumulation of unspliced mRNA, whereas structures that increase splicing rates could increase spliced mRNA levels (Fig. 6a). We chose seven structured introns to assay with VARS-seq, including introns with zipper stems (in RPL7A, QCR9, RPL36B and RPL28), covarying base pairs (in RPS9A, RPS9B and RPL7A), and long stems that distinguished pre-mRNA from coding RNA.
a, An overview of the structure-function experiment. ktx, transcription rate; kpre-mRNA, pre-mRNA decay rate; ksp, splicing rate; kmRNA decay, mRNA decay rate; klariat decay, intron lariat decay rate. b, Schematic for an example library design for one intron stem, with variants disrupting the stem and rescue sequences restoring base pairing. c–e, The effects of structure variants on the RI fraction for two regions of the RPL28 intron (c,d) and for intron stems in RPS9A (e). For a given stem set or loop, violin plots depict data for the wild-type sequence and all variant sequences; unique barcodes are shown as black points. Data for rescue sequences are shown when included in the intron library. P values are indicated for comparisons between the wild-type and variant sequence sets, and between the variant and rescue sequence sets. P values were computed using two-sided permutation tests for the difference in mean statistic. In box plots, the median is the center white point, box limits are the 25th (Q1) and 75th (Q3) percentiles and whiskers extend to the smallest and largest values that fall within 1.5 times the interquartile range below Q1 and above Q3. Secondary structures are colored according to reactivity data. Bars alongside the secondary structure indicate the stem and loop disruption sets, with each bar representing a set of variant sequences mutating nucleotides across the full extent of the bar. These bars are colored by the RI score for the corresponding stem or loop disruption set. The RI score is computed as the negative log (P value) when comparing RI values between wild-type and variant sequences; the sign is used to indicate the effect direction, with positive values (shown as blue) for a lower variant RI fraction compared with the wild type, and negative values (shown as red) for a higher variant RI fraction compared with the wild type. Green boxes in e indicate significantly covarying residues. f, RI fractions as measured by RT–qPCR for individual strains containing a set of wild-type, variant and rescue sequences for RPS9A stem 191–195 (top), with data shown for strains constructed with two different barcode sequences from three biological replicates (bottom). Data are presented as median values with 95% confidence intervals. P values are computed with two-way ANOVA tests with multiple comparisons. Exact P values for the low-RI-fraction barcode case are as follows: wild type versus 5′ mutant P < 1 × 10–4, wild type versus 3′ mutant P < 1 × 10–4, 5′ mutant versus rescue P < 1 × 10–4 and 3′ mutant versus rescue P = 0.0003. Exact P values for the high-RI-fraction barcode case are as follows: wild type versus 5′ mutant P = 0.018 and 5′ mutant versus rescue P = 0.021.
For each intron, we designed variants with systematically mutated secondary structures and rescued them with compensatory substitutions where possible (Supplementary Table 3). More specifically, for each target set of stems, we chose variants that were predicted to disrupt base-pairing in the stem set while maintaining base-pairing elsewhere in the structure (Fig. 6b and Extended Data Fig. 8a,b). When feasible within library length limits (Methods), we also included rescue sequences that were predicted to restore the native secondary structure (Fig. 6b and Extended Data Fig. 8a,b). We tested 4–8 distinct intron variants for each target stem set, with around 200 variant sequences for each intron (Supplementary Table 3). We integrated these intron variant libraries into the yeast genome48 in their native gene context (Fig. 6a and Extended Data Fig. 8c), installing unique barcodes to help match spliced RNAs to their original pre-mRNA variant (Extended Data Fig. 8c,d and Supplementary Fig. 13). With multiple barcodes and variant sequences assigned to each set of stems, we could observe subtle effects on gene expression due to changes in intron structure (Supplementary Text). Using our RNA-sequencing data, we computed two key readouts for each variant: the RI fraction (fraction of total RNA that is unspliced) and the normalized mRNA level (spliced mRNA levels normalized by representation in genomic DNA libraries).
Across the 7 tested introns, we detected statistically significant changes in RI and mRNA levels for 52 of 136 tested sets of stems and 4 of 15 tested loops (Extended Data Fig. 9 and Supplementary Fig. 14). Some stem disruptions resulted in significantly decreased RI levels (Fig. 6c), whereas others significantly increased them (Fig. 6d). For many introns, distinct variant sequences designed to disrupt overlapping sets of stems produced similar effects, providing a useful cross-check for our approach (Supplementary Fig. 14a–d). For instance, variants disrupting all 5 sets of stems and loops that included nucleotides 36–48 in the RPL28 intron significantly decreased the RI, suggesting that these nucleotides reduce splicing efficiency or slow pre-mRNA decay (Fig. 6c and Supplementary Fig. 14a). Similarly, all mutations to nucleotides 73–96 of the RPL28 intron led to increased RI fractions (Fig. 6d), suggesting that the wild-type nucleotides promote splicing or pre-mRNA decay. Unexpectedly, zipper stem variants in the RPL28 intron (Fig. 6c) lowered RI fraction, suggesting that not all zipper stems promote splicing by colocalizing splice sites16. In the context of secondary structures from DMS-MaPseq, these results demonstrate that intronic mutations can influence gene expression, even when they are structurally distant from splice sites (Fig. 6d and Supplementary Fig. 14).
In cases in which we could generate compensatory mutations, stem disruption and rescue variants pinpointed functional intronic structural elements. For instance, in the case of RPL36B, sequence variants in one region reduced normalized mRNA levels and rescue variants restored higher mRNA levels (Supplementary Fig. 15a), implicating the RNA structure, rather than its primary sequence, as the functional element. In the case of the intron in RPS9A, we identified a functional stem in which disruptions reduced the RI fraction, which was restored by stem rescue (stem 191–195, Fig. 6e). An analogous stem is present in the RPS9B intron, with similar effects (Stems 165–183, Supplementary Fig. 14b). These RPS9A and RPS9B stems likely influence gene expression by inhibiting splicing or pre-mRNA decay, aligning with covariation in these stems (Fig. 2e,f) and corroborating a prior study3 on RPS9A.
To validate our findings from VARS-seq, we constructed strains containing individual variant and rescue sequences for intron stems from RPL36B and RPS9A, and we used quantitative reverse transcription PCR (RT–qPCR) to measure mRNA levels. In the case of RPS9A, RT–qPCR data from individual strains recapitulated structure effects from VARS-seq along with the effects of barcode sequences (Fig. 6e,f). Indeed, when assessing a barcode that yielded low wild-type RI fractions (left, Fig. 6f), variant sequences lowered the RI, and rescue sequences restored it to wild-type levels, as determined by RT–qPCR. By contrast, individual strains designed to assess intron stems from RPL36B (Supplementary Fig. 15) did not show significantly different normalized mRNA levels as determined by RT–qPCR (Supplementary Fig. 15b,c). It is possible that effects found by aggregating data across variant and barcode sequences through VARS-seq could not be discerned when analyzing individual RPL36B variants in RT–qPCR. Owing to differences in dynamic range between VARS-seq and RT–qPCR, effects for higher RI barcodes on the intron in RPS9A are not visible with RT–qPCR (right, Fig. 6f), and significant zipper stem effects from RT–qPCR were not visible with VARS-seq for RPL36B (Supplementary Fig. 15b,c). To enable further analysis of intron stem variants, we summarize all variant and rescue comparisons from VARS-seq in heatmaps in Supplementary Figure 16, and we include per-barcode spliced and unspliced RNA counts in Supplementary Table 4.
Enriched structural patterns across the Saccharomyces genus
Our transcriptome-wide structure mapping provides evidence for widespread structure in S. cerevisiae introns, and with VARS-seq, we find that intron structures can impact gene expression regulation. To extend our observations to other species in the Saccharomyces genus, we turned to computational structure prediction. We first evaluated whether de novo secondary structure prediction could recapitulate structural features observed through DMS-guided analysis, comparing introns with length-matched controls (Fig. 7a and Supplementary Fig. 17). For de novo structure prediction, as a first approach, we predicted minimum free-energy structures33; as a second approach, we generated secondary structure ensembles through stochastic sampling of structures49, which has previously enabled the study of structural patterns in introns16,19. With either of these approaches, structural features from de novo secondary structure prediction and DMS-guided structures were largely consistent (Fig. 7b, Supplementary Fig. 18 and Supplementary Text).
a, Workflow for comparisons between introns’ secondary structure ensembles and those of control sequences. b, Comparison of secondary structure feature enrichment between introns and control sequences for DMS-guided structure prediction (left; with folding engine RNAstructure, comparing introns to shifted genomic controls), de novo MFE structure prediction (middle; with folding engine RNAstructure, comparing introns with shifted genomic controls), and de novo ensemble structure prediction (right; with folding engine Vienna 2.0, comparing introns with shuffled sequence controls). *P < 0.01 by two-sided Wilcoxon ranked-sum test. Exact P values from left to right are as follows for the left panel: P < 1 × 10–4, P = 0.0003, P = 0.0007, P < 1 × 10–4, P < 1 × 10–4; for the middle panel: P < 1 × 10–4, P = 0.0072, P = 0.0008, P < 1 × 10–4, P = 0.0015; and for the right panel: P < 1 × 10–4, P = 0.0003, P = 0.0011, P < 1 × 10–4, P < 1 × 10–4. In the left and middle panels, 140 introns are compared; 288 introns are compared in the right panel. MFE, minimum free-energy; SS, splice site; BP, branch point; intron - control score, difference between intron and control score. c, Differences in zipper stem (top) and downstream stem (bottom) ΔG between introns and shuffled sequence controls for introns in the Saccharomyces genus, using Vienna 2.0 ensemble predictions. *P < 0.01 by two-sided Wilcoxon ranked-sum test. All P values for the zipper stem comparisons are <1 × 10–4. For each species, the number of introns compared for both stem types and the P value for the downstream stem comparison are as follows: smik (n = 279, P = 0.00150), skud (n = 279, P = 0.21), suva (n = 278, P = 0.07), cgla (n = 100, P < 1 × 10–4), kafr (n = 216, P = 0.83), knag (n = 175, P = 0.011), ncas (n = 250, P = 0.10), ndai (n = 218, P = 0.00056), tbla (n = 163, P = 0.0017), tpha (n = 143, P = 0.0005), kpol (n = 175, P < 1 × 10–4), zrou (n = 166, P = 0.28), tdel (n = 202, P = 0.58), klac (n = 151, P = 0.0026), agos (n = 185, P = 0.26), ecym (n = 19, P = 0.41), sklu (n = 229, P = 0.27), kthe (n = 215, P = 0.31) and kwal (n = 210, P = 0.011). d, Distribution of zipper stems across introns in the Saccharomyces genus. Green values on the heatmap indicate a predicted zipper stem; white indicates no predicted zipper stem; gray values indicate deleted introns. Ohnologous introns are combined into a single row, and a zipper stem is annotated if present in either homolog. The species represented in this figure are: Eremothecium gossypii (agos), Candida glabrata (cgla), Eremothecium cymbalariae (ecym), Kazachstania africana (kafr), Kluyveromyces lactis (klac), Kazachstania naganishii (knag), Vanderwaltozyma polyspora (kpol), Lachancea thermotolerans (kthe), Lachancea waltii (kwal), Naumovozyma castellii (ncas), Naumovozyma dairenensis (ndai), Saccharomyces kudiavzevii (skud), Saccharomyces mikatae (smik), Saccharomyces uvarum (suva), Tetrapisispora blattae (tbla), Torulaspora delbrueckii (tdel), Torulaspora phaffii (tpha) and Zygosaccharomyces rouxii (zrou). In box plots, the median is the center white point, box limits are the 25th (Q1) and 75th (Q3) percentiles, and whiskers extend to the smallest and largest values that fall within 1.5 times the interquartile range below Q1 and above Q3.
We next made de novo structure predictions across species in the Saccharomyces genus, focusing on 20 species with intron alignments from Hooks et al.50. Enrichment for numerous secondary structure patterns is conserved in introns across the Saccharomyces genus. In particular, introns across the genus include more stable zipper stems and downstream stems than do controls (Fig. 7c), along with higher MEE and shorter distances between the 5′ splice site and branch point (Supplementary Fig. 19a). Furthermore, many of these features remain enriched in comparison with phylogenetic controls, which include random sequences constructed to match the mutation and indel frequency between an intron and its homologous S. cerevisiae intron (Supplementary Fig. 19b).
Secondary structure patterns are maintained across Saccharomyces species despite extreme sequence-level divergence between introns, suggesting that these structures have conserved functions. Complete intron deletions between these species are common, with many introns having orthologs in only a subset of the Saccharomyces species (Supplementary Fig. 19c), and most intron sequences have low conservation (50 to 60%) between species (Supplementary Fig. 19d). Some key functional intervals in introns are more conserved across the Saccharomyces species, with higher sequence conservation (72.3% to 83.6%) across the seven small nucleolar RNA (snoRNAs) in these intron sequence alignments. By contrast, zipper stem regions in S. cerevisiae introns diverge substantially between Saccharomyces species; most zipper stems are only 20 to 30% conserved in the primary sequence. Strikingly, this structural motif appears in disparate introns across species, with many zipper stems present in only a small number of orthologous introns (Fig. 7d). It is possible that functional secondary structures in these regions have been challenging to find by covariation due to high intron sequence divergence in closely related fungal genomes.
Discussion
RNA structures have critical roles in regulating a wide array of nuclear processes, including transcription, RNA modification and editing and splicing. Intronic RNA structures are poised to participate in these processes, for instance by altering splicing kinetics, changing RNA decay rates, or interacting with other nuclear factors. Here, we sought to understand the structural landscape of introns in S. cerevisiae, a model system for eukaryotic splicing. First, we evaluated the presence of structural patterns in introns using transcriptome-wide DMS probing after splicing inhibition. This revealed extended secondary structures in introns that distinguished these regions from coding mRNA. These structural data enable the clustering of introns into seven classes, with most falling into classes that contain introns with zipper stems or downstream stems, long introns with intermediate structure, and unstructured short introns. In Extended Data Figure 10 and Supplementary Figure 20, we display secondary structures and reactivity profiles for all introns from DMS-MaPseq, grouped into these classes. With a high-throughput structure-function assay, VARS-seq, we used deep mutagenesis to identify intron structural elements that influence spliced and unspliced mRNA levels. Finally, through computational structure prediction, we identified signals for structure in introns across the Saccharomyces genus.
Structure probing experiments have enabled the assessment of RNA structure across the transcriptome, but these experiments often lack sufficient coverage to provide information for low-abundance transcripts, including many unspliced pre-mRNA molecules. Specific low-abundance introns can be probed by target-specific enrichment27,51 or nuclear RNA enrichment45,52, but here we enhance detection of pre-mRNA sequences generally by using global splicing inhibition. We expect that most pre-mRNAs in pladB-treated cells remain unspliced, as accumulating pre-mRNAs are expected to outnumber spliceosome components53. It is possible that splicing inhibition could alter pre-mRNA structure compared with that in untreated cells, as accumulating pre-mRNA could interact with the nuclear pore complex54, nuclear exosome55 or NMD machinery56. Nevertheless, for introns with sufficient coverage from untreated cells, we found similar high-confidence stems with and without splicing inhibition. In future work, it would be interesting to directly observe long-range base-pairing and higher-order RNA structures in introns using approaches such as PARIS57 or KARR-seq58 in pladB-treated cells.
Our data allowed us to discern structural patterns that distinguish introns from coding regions. For instance, with higher Gini coefficients and longer stems, stable structures are enriched in introns compared with coding regions. It is possible that introns’ intramolecular structures are necessary to avoid spurious interactions between intron RNA and other nucleic acids in the crowded nuclear environment, preventing dysregulation of gene expression. Additionally, although there is evidence across species for depletion of double-stranded RNA in coding RNA to avoid activating cytosolic antiviral cellular responses59, this selective force would not affect introns in the nucleus. Long stems could also be depleted from coding mRNA because cytosolic mRNAs with stable stems might accumulate stalled ribosomes, becoming subjected to decay pathways such as no-go decay31,60. Finally, without the evolutionary constraints of adhering to a coding sequence, introns might be free to form extended structures that can regulate a host of nuclear processes.
Given that structural motifs are enriched in S. cerevisiae introns, we hypothesized that these introns could harbor functional structures. Although bioinformatic scans for covariation have identified functional structures in a several S. cerevisiae introns (including RPS9A7, RPS9B, RPS13 (ref. 23), RPL7B23 and RPL7A), sequence-only analyses could miss functional structures owing to the limited power of existing intron alignments61, with high variation between aligned intron sequences. Indeed, tools such as R-scape can be limited by low sequence conservation in alignments42,61. By using the high-throughput VARS-seq analysis to evaluate intron variants and rescue sequences, we identified additional functional structures. Although prior intron variant libraries have been designed to assess the effects of sequence motifs62,63, libraries assessing the role for structured elements in introns have been limited. Using VARS-seq, we found that some introns included domains that could influence gene expression despite being distal from splice sites in the intron’s secondary structure. These examples add to the complexity of potential functional roles for introns9,10. Additional evaluation of individual strains using orthogonal assays could validate the effects for specific stems of interest, which we carried out for intron stem sets from RPS9A and RPL36B.
Together, our structural, computational and functional experiments point to structural patterns across introns that impact gene expression. In one common pattern, we found an enrichment for structures around cryptic splice sites and depletion of structures around canonical splice sites, potentially indicating a role for structures in encouraging the use of canonical sites. Another pattern was the formation of zipper stems that colocalize the 5′ splice site and the branch point. Introns included highly stable zipper stem structures in vivo, and these structures were further confirmed by multi-dimensional chemical mapping (M2-seq) in vitro. These zipper stems could enable efficient splicing by reducing the physical distance between the 5′ splice site and branch point64 or by enhancing specific interaction with the spliceosome, with intron helical density seen interacting directly with the E and pre-A spliceosome complexes25,36.
To investigate the mechanisms by which these and other structured elements regulate gene expression, customized experiments for individual introns are needed. Structures located close to splice sites could influence spliceosome recruitment, while structures farther away from splice sites might interact with other nuclear factors. Intron secondary structures can regulate splicing by sequestering alternative splice sites65,66, occluding exonic splicing enhancers67, physically bridging splice sites68, facilitating co-transcriptional splicing69 or mediating protein interactions that influence splicing patterns70. Additionally, these intronic structures could have regulatory functions in pathways orthogonal to splicing, much like the snoRNAs encoded in S. cerevisiae introns41. In fact, RNA secondary structures in introns are associated with RNA-binding proteins involved in transcription, tRNA and rRNA processing, ribosome biogenesis and assembly and metabolic processes71,72. Furthermore, secondary structures in introns could influence gene expression in auto-regulatory circuits, as seen previously in the cases of RPS14B21 or RPS9A7, with structural elements within a gene’s introns binding its protein products and thereby downregulating subsequent gene expression. Intron structures could also influence numerous pre-mRNA decay pathways, including nuclear retention followed by decay by the nuclear exosome, NMD and NMD-independent decay by cytoplasmic exonucleases31,32,54,55,73. Finally, structures can have a functional role in regulating adaptation to starvation by influencing the accumulation of introns under nutrient depletion5,6, and it will be interesting to explore these structures in these saturated-growth conditions.
Our work identifies a set of intron structures with properties distinct from those in coding regions, support for in vivo formation from DMS-MaPseq and signals in related yeast species. Furthermore, we identify structured intervals of introns that modulate gene expression. These functional experiments provide candidates for further mechanistic characterization and provide a glimpse into the broad regulatory potential for intron sequences beyond splice sites. The widespread presence of structured elements in S. cerevisiae introns raises the possibility that similar motifs and stable secondary structures play a role in introns in higher-order eukaryotes, perhaps forming regulatory elements in human pre-mRNA.
Methods
Strains, medium and growth conditions for DMS probing
The strain OHY001 was constructed from JRY8012 (ref. 75), which includes three deletions of ABC transporter genes (prd5::kanr, snq2::kanr, yor1::kanr) to reduce drug efflux. OHY001 was generated through CRISPR editing of JRY8012 to mutate portions of HSH155 HEAT repeat domains 15–16 to match the sequence found in human SF3B1 (see sequence in Supplementary Table 5).
Strains were grown at 30 °C on YPD plates and in YPD liquid medium. Single colonies of OHY001 were used to inoculate overnight cultures and grown to an optical density at 600 nm (OD600) of 0.5–0.6. Biological replicates were obtained from distinct single colonies.
Splicing inhibition and DMS treatment
We carried out splicing inhibition and DMS treatment for two biological replicates, only splicing inhibition for a no-modification control and only DMS treatment for another control. We cultured 15–60 ml to an OD600 of ~0.5 for each replicate of each condition (2 biological replicates of DMS treatment, 1 no-modification control, 1 no-pladB control and 2 biological replicates of in vitro DMS treatment). We treated cultures with 5 μM pladB (Cayman Chemicals), and we incubated cultures at 30 °C for 1 h with shaking. The condition without splicing inhibition was treated with an equal volume of DMSO.
For in vivo DMS modification, we treated cultures with 3% DMS or an equivalent volume of H2O for the no-modification control. Treated cells were incubated with occasional stirring in a water bath at 30 °C for 5 min, and the reaction was quenched by adding 20 ml stop solution (30% 2-mercaptoethanol, 50% isoamyl alcohol) for every 10 ml of culture. Cultures were mixed and then spun down for 3 min at 1,500g and 4 °C, and washed first with 5 ml wash solution (30% 2-mercaptoethanol) for every 10 ml of culture. A second wash was performed with 3 ml YPD per 10 ml culture. RNA was extracted using the YeaStar RNA Kit (Zymo Research), using 7.5 μl of Zymolase for every 2.5 ml of cell culture and shortening the Zymolase incubation to 15 min at 30 °C.
For in vitro DMS modification, we followed a protocol similar to the one used in Rouskin et al.76. We first obtained RNA from cultures after splicing inhibition with the YeaStar RNA Kit (Zymo Research). We then re-folded the RNA in vitro, first denaturing 200 μg of RNA at 95 °C for 2 min, cooling it on ice for 2 min, and then folding it at 30 °C for 30 min in 10 mM Tris HCl pH 8.0, 100 mM NaCl and 6 mM MgCl2. We treated RNA with 3% DMS at 30 °C for 5 min, and we quenched the reaction with 25% 2-mercaptoethanol. RNA was purified by ethanol precipitation and eluted in 20 μl RNase-free H2O.
RT–PCR for verifying splicing inhibition
As initial verification of splicing inhibition by pladB, we used RT–PCR to compare unmodified RNA extracted after 1 h of either 5 μM pladB or DMSO treatment. We first treated samples with TURBO DNase (Thermo Fisher). We then carried out reverse transcription with the iScript Reverse Transcription Supermix (Bio-Rad), using 10 μl of each RNA sample for a reaction that included the reverse transcriptase, and the remaining 10 μl for a control without the enzyme. PCR was then performed for 30 cycles at an annealing temperature of 56 °C using the NEBNext Ultra II Q5 Master Mix (New England Biolabs (NEB)) with primers RR063 and RR064 for RPL36B, and RR067 and RR070 for MATa1 (Supplementary Table 5).
DMS-MaPseq sequencing library preparation
To prepare DMS-treated RNA for sequencing, we first depleted the extracted RNA of rRNA using RNase H. We concentrated extracted RNA for each condition using RNA Clean and Concentrator-5 columns (Zymo Research), yielding between 46.1 and 200 μg RNA for each replicate of each condition. We pooled and concentrated 108 fifty-base oligonucleotides (RR-rRNAdep-1-108; Supplementary Table 5) that tiled the 5S, 5.8S, 18S and 25S rRNAs in S. cerevisiae. Up to 40 μg of total RNA was included in each rRNA depletion reaction, and multiple reactions were performed as needed for each sample. First, a 15-μl annealing reaction was prepared with total RNA, an equal mass of rRNA depletion oligonucleotides, and 3 μl of 5× hybridization buffer (500 mM Tris-HCl pH 7.5 and 1 M NaCl). For the annealing reaction, the reaction mix was heated to 95 °C for 2 min, and the temperature was ramped down to 45 °C by 0.1 °C s–1 with a thermocycler. Then, 7.5 μl of Hybridase Thermostable RNase H (Lucigen) with 2.5 μl of 10× digestion buffer (500 mM Tris-HCl pH 7.5, 1 M NaCl, 100 mM MgCl2) was preheated to 45 °C. We combed the annealing mix and the RNase H mix and incubated the reaction at 45 °C for 30 min. For each reaction, we used an RNA Clean and Concentrator-5 column with the size-selection protocol to exclude RNA and oligonucleotides below a size cutoff of 200 nt. We then treated each reaction with TURBO DNase. Reactions were purified and then eluted in 9 μl of RNase-free H2O.
We fragmented each reaction with 10X RNA Fragmentation Reagent (Ambion), incubating at 70 °C for 8 min. We added 1 μl of Stop Solution (Ambion), cleaned up reactions using RNA Clean and a Concentrator-5 column, and removed the 3′ phosphate groups left by fragmentation with 1.5 μl rSAP (NEB). We next ligated a universal cloning linker to the RNA to serve as a handle for reverse transcription. To prepare linker for this reaction, we first phosphorylated 1 nmol of the DNA universal cloning linker with a 3′ amino blocking group (oligo RR118; Supplementary Table 5) with T4 PNK (NEB), and we next adenylated the linker with Mth RNA Ligase (NEB). We then purified the reaction with an Oligo Clean and Concentrator column. We added adenylated linker in twofold molar excess to each RNA sample, with ligation using T4 RNA ligase 2 truncated KQ (NEB). The reaction was incubated at 25 °C for 2 h in a thermocycler. Ligated RNA was purified using an RNA Clean and Concentrator-5 column, followed by elution in 15 μl of RNase-free H2O. The excess DNA linker was then degraded with 1 μl of 5′ deadenylase (NEB) and 1 μl of RecJf (NEB) in a 20-μl reaction. The mixture was incubated at 30 °C for 1 h and purified with an RNA Clean and Concentrator-5 column.
We then proceeded to RT with mutational readthrough. For the RT primer, we used the oligonucleotide RR114 (Supplementary Table 5), which included a sequence complementary to the universal cloning linker, a 5′ phosphate modification that would allow for circularization after RT, a 10-nt randomized unique molecular identifier (UMI) sequence, and sequences complementary to Illumina sequencing primers to allow for PCR amplification of the final library. We added 2 μl of 5× TGIRT buffer (250 mM Tris-HCl pH 8.0, 375 mM KCl, 15 mM MgCl2), 0.5 μl of 2 μM RT primer, and 6 μl of the RNA sample. The reaction was denatured at 80 °C for 2 min and left at room temperature for 5 min. Then, 0.5 μl TGIRT enzyme (InGex), 0.5 μl SUPERase inhibitor, 0.5 μl 100 mM DTT, and 1 μl 10 mM dNTPs were added to the reaction. The reaction was incubated at 57 °C for 1.5 h in a thermocycler for RT. RNA was then degraded by adding 5 μl of 0.4 M NaOH for 3 min at 90 °C, and the reactions were neutralized by adding 5 μl of an acid quench mix (from a stock solution of 2 ml of 5 M NaCl, 2 ml of 2 M HCl and 3 ml of 3 M sodium acetate). The reaction was purified using an Oligo Clean and Concentrator column, with elution in 7.5 μl of RNase-free H2O. The cDNA was then purified with a denaturing PAGE (dPAGE) gel to remove excess RT primer, selecting the 200–400-nt size range. Purified cDNA was eluted using the ZR small-RNA PAGE Recovery Kit (Zymo Research).
The size-selected cDNA was circularized and then amplified for sequencing. For circularization, we used CircLigase ssDNA Ligase (Lucigen), with overnight incubation at 60 °C followed by 10 min at 80 °C. The circularized cDNA was purified with an Oligo Clean and Concentrator column and eluted in 7.5 μl of RNase-free H2O. Residual RT primer was removed by degrading linear DNA, first with treatment with RecJf, followed by ExoCIP A and ExoCIP B (NEB) treatment. The circularized cDNA was purified and eluted in 10 μl. We then carried out 9 cycles of indexing PCR to add i5 and i7 index sequences to the sequencing library, using the NEBNext Ultra II Q5 Master Mix (NEB) with 10 μl of cDNA and 2.5 μl of 10 μM primers including different index sequences for each sample (i5_4 and i7_4 for replicate 1, i5_3 and i7_3 for replicate 2, i5_5 and i7_5 for the no-modification control, and i5_2 and i7_2 for the -pladB control; Supplementary Table 5). Nine PCR cycles were carried out, with annealing at 70 °C for 30 s. The reaction mix was purified with the DNA Clean and Concentrator-5 kit (Zymo Research), with elution in 10 μl of H2O. To obtain sufficient material, we then carried out two more cycles of PCR for in vivo replicates 1 and 2 and the no-modification control, five more cycles for the no-pladB control, nine more cycles for in vitro replicate 1, and three more cycles for in vitro replicate 2. For the final PCR reaction, we used the same reaction conditions and cycling parameters as the first indexing PCR with primers P5 and P7 (Supplementary Table 5). We purified the final library with two rounds of bead purification with size selection to remove remaining excess RT primer. We used RNACleanXP beads (Beckman Coulter), mixing 42.5 μl beads with the 50 μl PCR reaction. For the first round of bead-based size selection, we performed elution in 50 μl of H2O, and for the second round, we performed elution in 20 μl of H2O. The sequencing libraries were quantified with a Qubit high sensitivity dsDNA Assay Kit (Invitrogen) and Bioanalyzer HS DNA assay. The dsDNA libraries for in vivo replicate 1, the no-modification control, and the no-pladB control were sequenced across three Illumina HiSeq lanes with paired-end reads of length 150. The dsDNA library for in vivo replicate 2 and the in vitro replicates were sequenced with NovaSeq S4 partial lanes; these also had a paired-end read length of 150.
DMS-MaPseq sequencing data analysis
We obtained 557 million reads for in vivo replicate 1, 1.30 billion reads for in vivo replicate 2, 375 million reads for the no-modification control, 169 million reads for the control without pladB, 335 million reads for in vitro replicate 1, and 174 million reads for in vitro replicate 2. We used UMI-tools77 to extract UMI tags from reads, and we used cutadapt to trim low-quality reads (Q-score cut-off of 20) and remove adapters. We then aligned sequencing reads to sequence sets of interest, including rRNA, introns, pre-mRNA ORFs, coding mRNA, decoy intron sequences (see below), and sequences for structured controls (the ASH1 and HAC1 mRNA sequences). S. cerevisiae intron annotations, including genomic coordinates and branch point positions, were obtained from Talkish et al.78. Coding ORF annotations were obtained from the Saccharomyces Genome Database79 for the S288C reference genome, and coding sequences corresponding to introns were identified for all cases except the two introns in snoRNAs (SNR17A and SNR17B). Paired-end alignment was performed with Bowtie2 (ref. 80) using the following alignment parameters in ShapeMapper 2 (ref. 81): –local –sensitive-local –maxins=800 –ignore-quals –no-unal –mp 3,1 –rdg 5,1 –rfg 5,1 –dpad 30. Alignments were merged, sorted, and indexed using samtools82. Reads with matching UMI tags were then deduplicated using the UMI-tools dedup function.
Mutational frequencies, coverage values and normalized reactivities were obtained by processing alignments using RNAframework83 executables. We first ran rf-count with the flag -m to compute mutation counts, using all other default parameters. We obtained coverage statistics for each sequence with rf-rctools stats, per-position mutation counts and coverage with rf-rctools view, and reactivity values with rf-norm (flags: -sm 2 -nm 2 -ow -rb AC -dw).
DMS-MaPseq data quality assessment
For each construct, per-base coverage was computed as the number of reads obtained from rf-rctools stats multiplied by the total read length and divided by the construct length. To compare the retained intron fraction across all introns before and after pladB treatment, these coverage values were obtained for all introns and all coding regions for genes containing introns in the conditions without and with pladB. The retained intron fraction was the ratio of these values. We removed introns in GCR1 from further consideration as the gene includes multiple distal alternative 5′ splice sites84. We combined data from two introns in SRC1 from nearby alternative 5′ splice sites differing by 4 nt. We additionally consolidated data from two-intron genes with multiple annotated isoforms (in RPL7B, VMA9, DYN2 and SUS1), in which annotated introns included both single individual introns and the longer intron representing the skipped isoform85. We used annotations for the ribosomal protein-coding genes from Hooks et al.50. We excluded eight snoRNA-containing introns from further analysis41, as the majority of these constructs’ reactivities represented the excised snoRNA structure rather than the complete intron structure.
We next found the Pearson correlation between intron reactivities for replicate 1 and replicate 2. Reproducibility between replicates is reported as the square of the Pearson correlation coefficient (r2) through the manuscript. A linear fit for log coverage versus replicate correlation for each intron indicated that the replicate correlation r was best approximated as 0.2 log (coverage) – 1.03. On the basis of this relationship, to reach r2 = 0.6, we used a coverage cut-off of 7,673, averaged between replicate 1 and replicate 2 in subsequent analyses.
To obtain the correlation between rRNA mutational frequencies in Zubradt et al.27 and our data, we obtained the paper’s DMS-MaPseq reads for S. cerevisiae with TGIRT reverse transcriptase (replicate 1 accession number SRX1959209, run number SRR3929621). We aligned these reads to the 18S and 25S yeast rRNA sequences with Tophat v2.1.0 (ref. 86) using the alignment parameters stated in Zubradt et al.27: -N 5 –read-gap-length 7 –read-edit-dist 7 –max-insertion-length 5 –max-deletion-length 5 -g 3. We obtained mutational frequencies with rf-count, as described above, and we compared them with our dataset.
We evaluated whether reactivity values could differentiate surface-accessible, unpaired residues from base-paired residues across the 18S and 25S rRNA. To identify surface-accessible residues in rRNA, we followed a similar protocol to that in Rouskin et al.76. With the S. cerevisiae ribosome structure (PDB ID 4V88 ref. 87), we computed solvent-accessible surface area (SASA) values for the rRNAs’ N1 atoms on A residues and N3 atoms on C residues in PyMOL, approximating DMS as a sphere with solvent_radius 3 and with dot_solvent and dot_density parameters set to 1. We determined the receiver operating characteristic curve for distinguishing unpaired and solvent-accessible rRNA residues from Watson–Crick base-paired positions (found with DSSR88), using a SASA cut-off of 2 to determine solvent accessibility.
DMS-guided structure prediction and validation
For introns with between-replicate r2 > 0.6, we performed secondary structure prediction by RNAstructure33 guided by DMS reactivity with 1,000 bootstrapping iterations, using the package Biers46 with default parameters for RNAstructure to obtain minimum free-energy structures and base-pair confidence matrices from bootstrapping. Structures were visualized with VARNA, Biers and RiboDraw (https://github.com/ribokit/RiboDraw).
To assess DMS-guided structure prediction, we performed structure prediction for a set of controls with known secondary structures. We obtained ground-truth secondary structures for the 5S, 5.8S, and 18S rRNA with DSSR88 from a eukaryotic ribosome structure (PDB ID: 4V88)87; for U5 snRNA from Nguyen et. al.89; for U1 snRNA from Li et. al.90; for tRNA from Rfam-derived secondary structures91; and for mRNA segments from Zubradt et al.27. We did not include control RNAs with known pseudoknots (for example, RNase P RNA92) because our structure predictions did not include pseudoknots. Additionally, we excluded control cases with multiple conformations, such as the U2 snRNA93. For DMS-guided structure prediction, 1,000 bootstrapping iterations were performed for all cases except the 18S rRNA, for which we performed 100 bootstrapping iterations. Positive stem predictions were cases in which predicted stems included at least 5 bp above the helix confidence estimate threshold. False positive predictions occurred when less than 50% of the predicted stem’s base pairs were included in the ground-truth structure. False negative predictions included stems of at least 5 bp in length in the native structure that had either low confidence or fewer than 50% of their base pairs correctly predicted.
We additionally explored other methods for structure prediction. First, using Arnie (https://github.com/DasLab/arnie) we ran ShapeKnots94 (allowing for pseudoknot prediction) on all 88 introns with sufficient coverage, guiding predictions with DMS data in 100 bootstrapping iterations. As a control, we predicted the RNase P RNA structure with ShapeKnots, comparing to the ground-truth secondary structure from PDB ID 6AGB ref. 92. We also generated predictions using DREEM95 for the following intron regions with high coverage from DMS-MaPseq (numbered from intron start): RPL28 75–300, RPL7A 120–340 (first intron), ECM33 35–270, RPL26B 300–400, RPS13 140–212, RPL25 148–255, RPS9B 147–266 and RPL30 25–165.
Targeted DMS probing of RNA in S. cerevisiae cells and heat-denatured RNA
To evaluate structures identified from transcriptome-wide DMS-MaPseq, we carried out targeted DMS probing for 30 introns that contained zipper stems (listed in Supplementary Table 6). For each of two biological replicates of targeted DMS probing, 5 ml of culture at an OD600 of ~0.5 were treated with 3% DMS, and RNA was extracted as described above. We additionally obtained DMS profiles for heat-denatured RNA. More specifically, for two biological replicates, we denatured 20 μg of RNA extracted from S. cerevisiae, denaturing RNA in a 500 μl volume with 1 mM EDTA by heating at 90 oC for 3 min. For these denatured RNA samples, we then incubated with 1.5% DMS for 1 min at room temperature, quenched the reactions with 500 μl 2-mercaptoethanol and purified RNA with RNA Clean and Concentrator-5 (Zymo Research). We depleted rRNA from all targeted probing samples with RNase H, as described above.
We then prepared sequencing libraries from the DMS-treated RNA samples, using targeted primers for RT and PCR to specifically assess the structure of our 30 target introns. We designed primers to amplify 220–270-nt intron segments, with overlapping intervals covering the full length of the intron (primers RR400–RR523 in Supplementary Table 5). Primers were designed to overlap exon and intron boundaries to ensure that unspliced pre-mRNA was specifically amplified. These primers were pooled for multiplexed PCR into four pools with Primer Pooler96 (setting the Mg2+ concentration to 0 mM, the Na concentration to 50 mM and the dNTP concentration to 0.8 mM), with the aim to minimize the formation primer dimers and other incorrect amplicons. Primers in Supplementary Table 5 are annotated with the resulting pool number.
DMS-treated RNA was reverse transcribed with each of the four RT primer pools. Each RT reaction was conducted with Induro reverse transcriptase (NEB) using 2 μl of 10 μM RT primer pool with 1 μg RNA in a 20-μl reaction. Reactions were incubated at 55 °C for 1 h for RT, and NaOH was used to degrade RNA, as described above. Purified cDNA was first amplified in a pooled format with a separate reaction for each primer pool, combining 20 μl of 10 μM PCR primer pool with 5 μl cDNA template and 25 μl Q5 master mix for an 11-cycle PCR reaction (annealing temperature 61 oC). DNA was purified with the DNA Clean and Concentrator-5 kit (Zymo Research). We next added adapters for Illumina sequencing, using primers RR524–RR647 (Supplementary Table 5) in individual PCR reactions with 15 cycles at an annealing temperature of 64 °C. Finally, i5 and i7 primers were added with 8 cycles of PCR with annealing temperature 70 °C (primers RR281, RR282 for targeted DMS probing replicate 1; RR283, RR284 for targeted DMS probing replicate 2; RR285, RR286 for denatured RNA probing replicate 1; and RR287, RR288 for denatured RNA replicate 2). To select amplicons with final library size between 350 and 400 bp, we used AMPure beads. Specifically, we used 28.5 μl of beads for a 50 μl sample volume to remove large fragments, and then added 13 μl of beads to the supernatant to capture the correct amplicon size range. The resulting library was quantified with Qubit and Bioanalyzer, and sequenced as a part of a NovaSeq X+ lane with a 2×150 cycle kit.
Data were processed using a similar workflow as described above for DMS-MaPseq. Targeted primers were removed using cutadapt, and primer-binding regions at the 5′ and 3′ end of each intron were masked during DMS-guided structure prediction. We obtained coverage of at least 100,000 for each of the 30 tested introns with targeted DMS probing.
Two-dimensional chemical mapping (M2-seq)
We carried out two-dimensional chemical probing to assess the formation of base pairs from DMS-MaPseq. DNA sequences for the introns from QCR9, RPL36B, RPS11A, RPL37A and RPS7B were obtained as gene fragments from Twist Biosciences (Supplementary Table 5). Each gene fragment included the full intron sequence, a T7 promoter, reference hairpins for normalizing structure probing data, and adapters for universal RT and PCR primers. To generate a pool of DNA variants through error-prone PCR, we first assembled a reaction mix with the following for each intron: 10 μl of 2 ng μl–1 template, 10 μl of 100 mM Tris pH 8.3, 2.5 μl of 2 M KCl, 3.5 μl of 200 mM MgCl2, 4 μl of 25 mM dTTP, 4 μl of 25 mM dCTP, 4 μl of 5 mM dATP, 4 μl of 5 mM dGTP, 2 μl of 25 mM MnCl2, 1 μl of Taq polymerase (Thermo Fisher), 2 μl of 100 mM primers (RR1 and RR107; Supplementary Table 5) and 51 μl of H2O. We carried out 24 cycles of PCR with a 64 °C annealing temperature, and samples were purified using RNACleanXP beads (Beckman Coulter) with an AMPure bead to sample volume ratio of 1.8. We then transcribed RNA in vitro with 5 μl of 1× T7 RNA polymerase (NEB), 2 μl of 1 M DTT, 6 μl of 25 mM NTPs, 5 μl of 40% PEG-8000, 5 μl of T7 transcription buffer (NEB) and template DNA in a final reaction volume of 50 μl. Reactions were incubated at 37 oC overnight. Samples were treated with TURBO DNase (Thermo Fisher). RNA was purified with RNACleanXP beads using a 70:30 ratio of beads to 40% PEG-8000 as the beads mixture.
We proceeded with structure probing, RT, and library preparation for these RNA pools. We prepared 3 μl of 12.5 pmol RNA for each intron for DMS treatment and a no-modification control. We denatured the RNA by unfolding at 95 °C for 2 min, and left it on ice for 1 min. To fold the RNA, we added 5 μl of 5× folding buffer (1.5 M sodium cacodylate pH 7.0. 50 mM MgCl2) and 14.5 μl of RNase-free H2O, and incubated the mixture for 30 min at 37 °C. We modified the RNA by adding 2.5 μl of 15% DMS (DMS condition) or 100% ethanol (no-modification control), and heating at 37 °C for 6 min. The reaction was quenched by adding 25 μl of 2-mercaptoethanol and purified by AMPure bead purification, followed by elution in 7 μl of RNase-free H2O. We reverse-transcribed the modified RNA with mutational read-through using TGIRT, as described above, in a 10 μl reaction volume. For reverse transcription, we used FAM-labeled primers that included a distinct index sequence for each construct and condition (RTB primers in Supplementary Table 5). The RT reaction was incubated at 57 °C for 3 hours, RNA was degraded with NaOH, and the solution was neutralized with an acid quench (see above). cDNA was purified using RNACleanXP beads and then amplified with PCR to add Illumina adapters for sequencing, using Phusion high-fidelity DNA polymerase (NEB) for 20 cycles at annealing temperature 65 °C with MaP forward and reverse primers (Supplementary Table 5). The resulting libraries were sequenced in two partial sequencing runs using Illumina MiSeq v3 600-cycle reagent kits, providing 300-nt paired-end reads.
M2-seq sequencing reads were processed using the M2seq package46. First, barcodes for the different introns and conditions were demultiplexed, obtaining at least 500,000 reads for each intron. The ShapeMapper 1.2 (ref. 81) software was used to align reads using Bowtie2 and compute mutation rates. The output from ShapeMapper was processed by the simple_to_rdat.py script to obtain RDAT files for both DMS and no-modification conditions, and processed with the Biers package46 to generate Z-score plots, base-pairing probability matrices, and predicted secondary structures. These secondary structure predictions were made using RNAstructure’s Fold executable33 with 500 bootstrapping iterations, guiding predictions with both one-dimensional and two-dimensional DMS reactivity data. Structures were visualized using VARNA and Biers.
Assessing proposed functional structures with DMS data
Using our DMS data, we assessed previously proposed intron structures. Structures from Hooks et al.37 were obtained by request. For structures with covarying residues, DMS-guided structure predictions were displayed when agreeing with proposed covarying base pairs (in RPS9A, RPS9B, and RPL7A), and structures from CaCoFold97 were displayed otherwise (RPS13).
To expand the set of covarying residues identified across introns, we used the protocol and software developed by Gao et al.23 with thresholds as follows. We obtained all complete and chromosome-level assemblies of genomes in the Ascomycota phylum from NCBI GenBank98 (107 sequences, accessed on 17 August 2021). For each S. cerevisiae intron, we generated multiple-sequence alignments in flanked mode with a non-coding threshold of 50. We then applied R-scape42 with an E-value threshold of 0.05, and we generated structures with CaCoFold97. We identified all introns with at least one significantly covarying pair between two non-consecutive residues in a stem with at least three base pairs. We evaluated the presence of these covarying residues in minimum free-energy secondary structures from ViennaRNA 2.0 (ref. 49). The set of snoRNA-containing introns was obtained from the S. cerevisiae snoRNA database41.
Zipper stem identification and stability calculation
We identified the positional constraints for forming a stem between the 5′ splice site and branch point (‘zipper stems’) by modeling 3D structures for introns in the context of the spliceosome using the Rosetta protocol FARFAR2 (ref. 99). For test constructs, we used variants of the RPS17B intron, which includes a previously characterized zipper stem16. To identify the minimum linker length for which a zipper stem is sterically compatible with the spliceosome, we built models of the RPS17B intron with a range of linker lengths between the 5′ splice site, zipper stem, and branch point. We based models on an A-complex spliceosome structure (PDB ID: 6G90)74. The tested variants included 36, 40, 45, 50, and 56 total nucleotides between the 5′ splice site, zipper stem strands, and the branch point. All models were built using Rosetta (release v3.10) for RNA homology modeling100.
More specifically, for each modeled RPS17B variant, we used Rosetta’s rna_thread to replace the pre-mRNA nucleotides resolved in the A-complex structure with the RPS17B sequence. We docked the RPS17B zipper stem into the binding site for an intron stem in the U1 snRNP by aligning the E-complex structure including a zipper stem (PDB ID: 6N7R ref. 25) with the A-complex structure in PyMOL. For each variant, 1,250 structures were sampled using Rosetta’s rna_denovo. The A-complex spliceosome, docked zipper stem, and rethreaded RPS17B residues were treated as rigid bodies, and remaining linker residues in RPS17B were sampled freely. To accelerate structure sampling, we generated models using three scoring terms, only including rna_vdw at weight 10.0 (the RNA van der Waals score term), rnp_vdw at weight 50.0 (the RNA–protein van der Waals score term), and linear_chainbreak at weight 10.0 (a score term penalizing chain breaks). We collected linear chain break scores for each of the top ten sampled structures, monitoring whether linker lengths were too short to extend between docked nucleotides.
The 3D modeling approaches used here make numerous approximations that could lead to inaccuracies. First, components such as the docked zipper stem are included as a rigid body, which precludes capturing local dynamics in these components. Second, we used a simplified score function and did not model explicit waters and ions in our system. Without additional score terms (for example electrostatics), we cannot model specific interactions accurately. Finally, these intron structures are highly flexible, but we analyzed only a small set of structures to obtain statistics. These structural models can be used to estimate pre-mRNA length constraints, rather than to analyze detailed structural features and interactions. Therefore, despite modeling limitations, this structural modeling can provide guidelines for zipper stem formation.
On the basis of the linear chain break scores, to designate a zipper stem, we required at least 42 nt connecting the zipper stem to the 5′ splice site and branch point. Additionally, these stems were required to begin at least 10 nt from the 5′ splice site and end at least 20 nt from the branch point, as closer positions could not form base pairs in the context of the A complex. Finally, we required that the linker between zipper stems and the 5′ splice site and branch point be at most 85 nt total, as zipper stems must colocalize the 5′ splice site and branch point. For a given secondary structure, we first identified the longest zipper stem satisfying these constraints with at most 5 bulge nucleotides, and we computed the stem’s ΔG stability with RNAcofold101. Vienna 2.0 carries out free-energy calculations using a nearest-neighbor energetic model, which computes the total free energy by summing energies for stacking neighboring base pairs (including enthalpic and entropic contributions). As a caveat, these calculations will not capture non-local interactions that might be modeled by sampling 3D structures. Free energies were computed at the default temperature (37 °C) and salt concentration (1.021 M).
Analyzing intron secondary structures surrounding canonical splice sites
We designated intervals surrounding the 5′ splice site, branch point, and 3′ splice site where pre-mRNA secondary structures would clash with the spliceosome. We identified the number of pre-mRNA nucleotides that threaded through structures of the E, A, pre-B, B, Bact, C and C* state spliceosome25,74,102,103,104,105,106. We noted that the following nucleotides threaded through the spliceosome in at least one state: 16 nt upstream and 10 nt downstream of the 5′ splice site; 33 nt upstream and 21 nt downstream of the branch point; and 8 nt upstream and 20 nt downstream of the 3′ splice site. We expected that these sequence intervals would be depleted of structure. We generated DMS-guided secondary structure predictions for each intron along with 50 upstream and downstream nucleotides, and we analyzed the proportion of nucleotides occluded by high-confidence stems.
Analyzing intron secondary structures surrounding cryptic splice sites
We identified cryptic splice sites by searching for alternative 5′ splice sites, branch points, and 3′ splice sites in relevant intron regions. More specifically, we searched for any splice site sequences that had appeared in either canonical introns or previously annotated proto introns78. We noted that at least 42 nt were required between the 5′ splice site and branch point for pre-mRNA to be compatible with spliceosome structures (see above). Thus, we located cryptic 5′ splice site sequences by searching for alternative 5′ splice sites up to 42 nt upstream of the canonical branch point. Similarly, we located cryptic branch point sequences between 42 nt downstream of the 5′ splice site and the canonical 3′ splice site. Finally, we identified cryptic 3′ splice sites within the intron that were at least 10 nt downstream of the branch point sequence. Using intron structures derived from DMS probing data, including 50 nt of surrounding context, we evaluated the protection of these cryptic sites by high-confidence stems, comparing with the background protection of nucleotides in each region.
We additionally tested whether cryptic 3′ splice sites used only upon Prp18p inactivation (GAG, UG, CG and GG splice sites) were protected107, finding a significant enrichment of secondary structure at these cryptic 3′ splice sites relative to surrounding background (291/1,559 = 18.6% protection at these sites relative to 526/4,280 = 12.3% protection at background sites; P < 0.001). However, cryptic 3′ splice sites matching the standard consensus sequence (UAG, CAG or AAG) were more protected (53/183 = 29.0% protection).
DMS structure analysis for introns and coding regions
We made reactivity-guided secondary structure predictions to identify structural features of S. cerevisiae introns and coding sequences for intron-containing genes. To count the number of introns that included zipper stems, we used the zipper stem criteria identified through spliceosome modeling as described above. We additionally identified ‘downstream stems’ between the branch point and 3′ splice site with at least 6 base pairs and bootstrapping support at least 70%.
To calculate secondary structure properties, we used both fine-and coarse-grained graphs. Fine-grained graphs included a node for every base pair and single-stranded position, with edges between consecutive positions or base pairs. Coarse-grained graphs included a single node for each stem, junction, loop, single-stranded 5′ end, and single-stranded 3′ end. Fine-grained graphs were used to compute the maximum extrusion from ends, using the arnie package108 to find the shortest path length from each position to either end of the sequence. The maximum value for this shortest path across all nodes yielded the maximum extrusion from ends, providing the effective distance between the sequence ends and the position farthest from these ends. The values for maximum extrusion from the ends were normalized by the length of each construct. Coarse-grained graphs were used to identify the longest stem with at most 10 total loop nucleotides and at least 90% bootstrapping probability. Additionally, coarse-grained graphs were used to compute average helix confidence estimates for all stems with a length of at least 6. Finally, Gini coefficients were calculated on the basis of mutational frequency values for intron and coding regions, using windows of 20 nt and scanning in intervals of 10 nt. To calculate the Gini index, we required that every position had per-position coverage at least 15,346 as computed from rf-rctools view, corresponding to 0.6 Pearson correlation of reactivity profiles between replicates.
Modeling of intron stems in spliceosome structure
We modeled the RPL28 and RPL36B introns in the context of the A-complex spliceosome (PDB ID: 6G90)74, using a protocol similar to that for RPS17B zipper stem modeling, described above. Intron nucleotides resolved in the PDB structure were replaced by the corresponding sequences from RPL28 and in RPL36B with the rna_thread application109. The secondary structure used for modeling was obtained from RNAstructure prediction guided by DMS data, and helices were supplied as rigid bodies. 4564 models were sampled using the Rosetta application rna_denovo99 with the simplified score function described above.
Comparison of introns with decoys
To determine whether secondary structure features can distinguish authentic introns from unspliced genomic sequences that match splicing motifs, we assembled a set of decoy introns. To find these decoy introns, we first computed the position weight matrix (PWM) for the 5′ splice site (6 nt with the consensus sequence GUAUGU), branch point (8 nt with the consensus sequence UACUAACN) and 3′ splice site (3 nt with the consensus sequence YAG) for all annotated S. cerevisae introns. We then obtained three length distributions from annotated introns: the complete intron length, the distance between the 5′ splice site and branch point, and the distance between the branch point and 3′ splice site. To find decoy introns, we scanned all genes for intervals containing 5′-splice-site, branch-point, and 3′-splice-site sequences matching the PWMs and three length distributions for introns, using cut-offs that captured at least 95% of canonical introns. We filtered these decoy sequences to exclude annotated introns. For each authentic intron and decoy sequence, we assembled length-matched control sequences that were shifted in the genome by 500 nt. DMS-guided structure predictions and structural features were computed for each intron, decoy and matched control sequence with sufficient coverage, as described above. Additionally, we computed the distance between the 5′ splice site and branch point as the length of the shortest path in the secondary structure graph between these sites. We then determined whether authentic introns and decoy intron sequences enriched for these structural features more than shifted genomic controls.
Visualizing the structure landscape for S. cerevisiae introns
We used secondary structure features from DMS-MaPseq to classify S. cerevisiae introns. For each intron, we included the following metrics: the sequence length, the presence of a zipper stem, zipper stem free energy, the presence of a downstream stem, downstream stem free energy, maximum extrusion from ends, the longest stem length, the average helix confidence estimate, the maximum Gini coefficient, and average DMS accessibility across splice sites and branch points. Hierarchical clustering was performed using scipy’s hierarchy module110 with Ward linkage and optimal leaf ordering, and Seaborn’s clustermap was used to generate a dendrogram and heatmap.
Designing intron structure variants for structure-function assay
We chose seven structured introns to mutate systematically by identifying secondary structures with zipper stems, covarying residues, and long stems. Intron library sequences were constrained to 300 nt, including fixed primer binding sites, leaving around 200–250 nt downstream from the 5′ splice site as the variable region. For each intron, we designated stem and loop sets, with each stem set containing one or more continuous stems, and each loop set containing a single junction or loop.
For each stem set and loop set, we generated candidate variants and scored these candidates on the basis of the desired secondary structure properties. For each stem set, we first generated 10,000 randomized and 10,000 shuffled sequences mutating the 5′ strands of the stems. Additionally, if the stem’s 3′ end fell within the library’s length constraints, we generated rescue sequences for each variant, installing compensatory mutations in the 3′ strand. For each loop set, we generated 20,000 random and shuffled variants. For each variant and rescue sequence, we calculated the minimum free energy structure with Contrafold111 within the full intron context. We then assessed whether the variant disrupted any targeted stems without altering the remainder of the secondary structure. More specifically, for each variant, we computed two penalty scores: a variable region penalty and a constant region penalty. The variable region penalty was the sum of the number of paired residues in the variable region along with the number of base pairs present in this region. The constant region penalty focused on nucleotides outside the variable region, computing the number of native base pairs disrupted by the variant, and adding half of the number of base pairs gained in the variant. If applicable, we calculated the rescue penalty as the number of base pairs altered between the native secondary structure and the rescue sequence. We chose the top 4–8 unique variants per region on the basis of the sum of the variable region, constant region and rescue penalties. Our variants did not affect the 5′ splice site, branch point or 3′ splice site. We additionally included the wild-type sequence for each set. The final chosen variant sequences and their penalty scores are in Supplementary Table 3.
Designing gene context for intron variants
Introns were cloned into their native gene context to better mimic the effects of structures in native S. cerevisiae on gene expression. Each construct included the promoter TDH3 (which has been shown to induce high expression levels48), the 5′ untranslated region (UTR) beginning at the transcription start site, the full gene including the intron sequence and the ADH1 terminator (shown to yield high mRNA half life112) (Extended Data Fig. 8c). For each gene, we used the YeasTSS database to identify the most prominent transcription start site to start the 5′ UTR113. When cloning intron libraries into this gene context, we included a 8-nt fixed sequence in the 5′ UTR to distinguish the inserted gene from the endogenous copy (AGCGGACG for QCR9, RPL28, RPS9A and RPL36B; AGAAGACG for RPS14B; AGAAGAGC for RPL7A and AACTGCCC for RPS9B). Additionally, we included a random 12-nt (12N) barcode in the 5′ UTR to identify variants even after introns are spliced out. We ran simulations sampling 12N barcodes to confirm that, for our expected number of clones (10,000–50,000), most barcodes would have an edit distance of at least two from all others (Extended Data Fig. 8d). We therefore expected that these barcodes would enable unambiguously assigning spliced reads to intron variants.
Growth media for structure variant library assay
We used the following growth media and plates for our structure–function assay:
-
YPD medium: YPD powder (Difco; yeast extract 10 g l–1, peptone 20 g l–1, dextrose 20 g l–1)
-
YPD plates: Difco YPD powder 50 g l–1, Difco agar powder 20 g l–1
-
SD-LEU plates: premix from Takara Bio
-
SD-HIS plates: premix from Takara Bio
Constructing yeast background strain for integrating structural variant libraries
We integrated our variant library into the yeast genome using homologous recombination and CRISPR–Cas9-induced double-stranded breaks. We chose the ARS416 locus as our integration site, because it is a highly efficient site114. To ensure that our library was integrated in the expected locus, we first constructed the background strain PLH001, which inserted a partial LEU2 selection marker at this locus, beginning at the second codon of the coding sequence. We then designed our structure variant library constructs to include the LEU2 promoter and start codon, selecting for integration at the correct locus with SD-LEU.
The strain PLH001 was constructed from background strain BY4741 (Mata, his3Δ1, leu2Δ0, met15Δ0, ura3Δ0). A partial LEU2 selection marker and a complete HIS3 selection marker were integrated into the ARS416 gene locus114. LEU2 and HIS3 were obtained from the pHLUM plasmid115, adding homology arms by PCR for genomic integration into ARS416. These constructs were cloned into the pUC19 backbone116 using NEBuilder Hifi DNA Assembly (NEB) (Extended Data Fig. 8c and insert sequence RRLH in Supplementary Table 5). We obtained a linear insert for transformation with PCR, which was integrated into the ARS416 locus using SD-HIS selection117.
Constructing plasmid backbones for structure variant library
We first cloned a plasmid backbone for each intron variant containing the intron’s gene context (pRR1–pRR7), and we then cloned our intron libraries into this background. For each intron, the background gene consisted of the TDH3 promoter, the 5′ UTR, the gene and intron excluding the variant library, the ADH1 terminator and homology arms for insertion into the ARS416 locus (Extended Data Fig. 8c). All PCR reactions in the following sections were carried out with the NEBNext Ultra II Q5 Master Mix (NEB). We used the following gene fragments from Integrated DNA Technologies to build gene background constructs: (1) gene blocks with the homology arm and TDH3 promoter; (2) a gene block with the ADH1 terminator, LEU2 promoter, LEU2 start codon and LEU2 homology arm; and (3) gene blocks with the target gene and constant regions in the intron (gblockRR4-12; Supplementary Table 5). Gene blocks were amplified with PCR using primers RR222–RR233 (Supplementary Table 5) and cloned into the background plasmid using a NEBuilder Hifi DNA Assembly (NEB) reaction.
Cloning structure variant library with high-efficiency transformation
Intron libraries were obtained as one oPool per intron from Integrated DNA Technologies, and the seven sub-libraries were amplified by PCR. For each intron, we carried out eight parallel PCR reactions with limited PCR cycling to reduce bias. We used the following primer pairs: RR273 and RR249 for RPS14B; RR274 and RR251 for RPS9B; RR275 and RR253 for RPS9A; RR276 and RR255 for QCR9; RR277 and RR257 for RPL36B; RR279 and RR261 for RPL28; and RR280 and RR263 for RPL7A (Supplementary Table 5). Primers included overhangs for plasmid insertion, and forward primers included 12N random barcodes. PCR was carried out with 30 ng template and 14 amplification cycles. The eight PCR reactions were concentrated together with the QIAQuick PCR Purification Kit (Qiagen) and gel purified with a MinElute PCR Purification Kit (Qiagen). Plasmid backbones pRR1–pRR7 were linearized using PCR primers RR223 and RR239–RR245 using a 3.5-min extension time, gel purified, and treated with DpnI (NEB). We transformed NEB Stable Competent E. coli cells (NEB) with NEBuilder HiFi DNA Asssembly (NEB) reactions for each of our 7 sub-libraries, using 2.5 μl of ligation product for 32 μl of competent cells. We obtained 6,000–46,000 clones per sub-library (30–230× library coverage) and validated 8–16 clones per sub-library with Sanger sequencing. Plasmid DNA was extracted using the ZymoPURE II Plasmid Midiprep Kit (Zymo Research).
Yeast genomic integration for structure variant library assay
The plasmid library was linearized by restriction digest. For the initial digestion, we combined 3 μl of XcmI (NEB) with 10 μl of 10X R2.1 buffer (NEB) and added 5 μg of each plasmid sub-library in a total volume of 100 μl. Reactions were left at 37 °C for 60 min and at 65 °C for 20 min. Then, for the second digestion, we added 2.0 μl of 2.5 M NaCl, 4.16 μl of Tris HCl pH 7.9, and 1.5 μl of the BglII enzyme, followed by an incubation at 37 °C for 60 min. To increase the efficiency of integration, the linearized plasmid library was transformed with Cas9. We obtained 50 μg of plasmid p426 (ref. 114) with the ZymoPUREII Plasmid Maxiprep Kit (Zymo Research) and linearized with AhdI digest.
We used heat-shock transformation in PLH001 with the linearized Cas9 plasmid and library insert for genomic integration with the lithium acetate (LiAc), single-stranded DNA (ssDNA) and polyethylene glycol (PEG) transformation protocol117. For each sub-library transformation, we used 5 μg linearized plasmid library and 5 μg linearized p426 plasmid to transform 25 ml of cells at an OD600 of 0.5. We plated transformed cells on SD-LEU selection plates. As negative controls, we transformed DNA from one sub-library into BY4741, and we transformed p426 alone without a library insert into PLH001. We saw no colony growth for either of these negative control conditions on SD-LEU selection plates. We obtained 9,000–35,000 colonies for each sub-library, yielding 45–175× library coverage for each intron. We verified correct integration for four to eight colonies per sub-library with Sanger sequencing.
Genomic DNA sequencing for structural variant library assays
We sequenced genomic DNA to assign barcodes to each designed variant sequence and to obtain transformation frequencies. We extracted DNA using the YeaStar Genomic DNA Kit (Zymo Research). We then carried out an indexing PCR with limited cycles to amplify target intron library regions and add i5 and i7 indices for Illumina sequencing with the primers RR281–RR294 (Supplementary Table 5). These primers amplified the target sub-libraries including barcodes, with forward primers binding to the fixed 8-nt sequences that distinguished the inserted intron library from the endogenous gene copy. For each indexing PCR, we used 50 ng genomic DNA template and 7 amplification cycles. We then amplified our indexing PCR sample with P5 and P7 primers, now with 11–14 PCR cycles and annealing at 66 °C. Samples were purified using RNACleanXP beads (Beckman Coulter) with a AMPure bead to sample volume ratio of 0.8 to remove primer dimers from the sample. The sequencing libraries were quantified and the libraries were sequenced using an Illumina MiSeq v3 600-cycle kit.
Targeted RNA sequencing for structure variant library assay
We aimed to sequence RNA to obtain levels of spliced and unspliced RNA. We started overnight cultures from 100 μl of yeast sub-library glycerol stocks, and we used 5 ml culture per sub-library in two columns from the YeaStar RNA Kit (Zymo Research), with 2.5 μl of Zymolase for every 2.5 ml starting cell culture. We pooled RNA extracted from each sub-library, combining 50 μg total RNA from each sub-library to yield 350 μg total RNA. For RNA sequencing, as described previously, we first depleted rRNA from our total RNA with RNase H, using 50-mer oligos to tile rRNA sequences. We then carried out size selection with RNA Clean and Concentrator-5 kit (Zymo Research), followed by treatment with TURBO DNase, and purified reactions with the RNA Clean and Concentrator-5 kit (Zymo Research).
To assess the removal of contaminating genomic DNA from our total RNA, we carried out a reverse transcription reaction with and without reverse transcriptase and checked for amplification of control RNA regions with PCR. For reverse transcription, we used the iScript Reverse Transcription Supermix (Bio-Rad). We purified samples with Oligo Clean and Concentrator columns (Zymo Research), with elution in 15 μl H2O. We then performed PCR with the MATa1 and RPL36B primers as described above.
We next generated cDNA using the DMS-MaPseq library-preparation strategy described above. In brief, first, we fragmented our RNA and purified with the RNA Clean and Concentrator-5 kit (Zymo Research). We then removed 3′ phosphate groups from fragmentation with rSAP (NEB) treatment followed by rSAP inactivation. We prepared adenylated linker from RR118 (Supplementary Table 5), and we then ligated this linker to our RNA sample with a T4 RNA ligase 2 truncated KQ (NEB) reaction, adding adenylated linker in 2× molar excess of the RNA sample. Excess linker was degraded with 5′ deadenylase (NEB) and RecJf (NEB) exonuclease. For reverse transcription, we used the TGIRT enzyme (InGex), using the reverse transcription primer RR301 which includes a 10-nt UMI and Illumina adapters (Supplementary Table 5). We degraded RNA and then purified cDNA with an Oligo Clean and Concentrator column (Zymo Research).
Next, we amplified cDNA with targeted PCR primers for each sub-library to generate final sequencing samples. We first carried out a short indexing PCR for each sample. For these PCR reactions, we used the following primer pairs: RR281 and RR302; RR283 and RR303; RR285 and RR304; RR287 and RR305; RR289 and RR306; RR291 and RR307; and RR293 and RR308 (Supplementary Table 4). Our forward PCR primers included the 8-nt fixed sequence that distinguished the inserted library from the endogenous gene. These primers allowed for the amplification of cDNA, including barcode sequences and the splice site junction. Additionally, these primers included unique i5 and i7 index sequences. For indexing PCR, we used Q5 with seven amplification cycles. We then carried out 24–28 additional PCR cycles with P5 and P7 primers (Supplementary Table 5). Finally, to obtain double-stranded DNA for sequencing, we carried out two rounds of size-selection and purification with RNACleanXP beads (Beckman Coulter), using an AMPure bead to sample volume ratio of 0.7, followed by elution in 15 μl H2O. The sequencing libraries were sequenced using a partial Illumina NovaSeq S4 lane, providing around 700 million paired-end reads of length 150.
Genomic DNA sequencing data analysis for structure variant library
Genomic DNA sequencing provided links from barcodes to intron variants along with quantification for the number of transformants for each variant. To prepare genomic DNA sequencing reads for analysis, we first used UMI-tools77 to extract barcodes and then error-corrected paired-end reads using bbmerge118, removing Illumina adapter sequences and ensuring consistency between paired-end reads.
We used the following pipeline to assemble consensus intron variant sequences for each barcode using the fgbio toolkit (fulcrumgenomics.github.io/fgbio/). First, paired-end reads were aligned to introns using BWA119, alignments were sorted using fgbio’s SortBam, and mate-pairs were annotated with fgbio’s SetMateInformation. We then grouped reads with the same barcode using fgbio’s GroupReadsByUmi with the adjacency method. Consensus reads were called using fgbio’s CallMolecularConsensusReads with the flags -m 20 -M 5 to require that bases have a Q score above 20, and that at least 5 reads covered each consensus sequence. We then filtered these sequences using fgbio’s FilterConsensusReads with the flags -M 5 -q 20 -N 20 -E 0.01, additionally requiring that consensus bases have a quality of at least 20, and reads have average error rates lower than 0.01. We merged paired-end reads using bbmerge118, obtaining consensus length distributions. Finally, to ensure that barcodes are mapped primarily to single consensus reads, we kept the primary consensus sequence for each barcode and required that at most 5% of reads map to secondary consensus sequences, removing 0.4–4.7% of barcodes.
From our consensus sequences, we obtained barcode mappings, along with transformation frequencies, for designed variants. We collated barcodes that mapped to designed variant sequences, and found at least ten unique barcodes for many of the designed variants. We additionally found that some colonies had consensus sequence variants that were mutations of wild-type sequences or designed variants. From these sequences, we retrieved mutated QCR9 branch point sequences as a control sample. To obtain transformation frequencies, we found read coverage using the output from fgbio’s FilterConsensusReads.
RNA-sequencing data analysis for structure variant library
To process RNA-sequencing data, we first extracted barcodes (one for each transformant) and UMIs (one for each cDNA molecule) from our sequencing reads with UMI-tools77. We then removed Illumina adapters from the reads using cutadapt, additionally trimming bases with a Q-score <20. We then aligned reads to spliced isoform reference sequences. For our reference sequence annotations, we collated all possible isoforms capturing spliced and unspliced transcripts (four isoforms for two-intron RPL7A and two for all other genes), and we generated gff annotation files with gmap120. We used TopHat2 (ref. 86) to align sequencing reads, using the following flags:–no-novel-juncs -T –b2-mp 3,1 –b2-rdg 5,1 –b2-rfg 5,1 –segment-length 20 –segment-mismatches 3 –read-gap-length 7 –read-edit-dist 50 -m 1 –max-insertion-length 19 –max-deletion-length 19. We generated fasta files from the resulting alignment files with samtools82.
We classified each aligned sequence into one of three categories: unspliced, spliced at the expected junction, or other, requiring 14 nt of agreement at the unspliced or spliced junction. Sequences that did not include canonical spliced or unspliced junctions were classified as alternative splicing events if they appeared in at least ten UMIs per barcode and at least three barcodes per variant. For canonical spliced or unspliced junctions, we counted reads after deduplicating UMIs. We then computed two metrics for each barcode: the retained intron fraction and the normalized mRNA level. The retained intron fraction was the ratio between the unspliced read counts and total read counts. The normalized mRNA level was the ratio between the spliced read count and the transformation frequency from genomic DNA sequencing. For each intron sub-library, we used the barcode-variant correspondence from genomic DNA sequencing to collect retained intron fractions and normalized mRNA levels.
For all the stem and loop sets assessed in each intron sub-library, we compared RI fractions and normalized mRNA levels between the wild-type sequence versus stem disruption variants, and the stem disruption variants versus the rescue sequences. Comparisons were made using two-sided permutation tests (SciPy110) for the difference in mean statistic, with 100,000 resamples of data labels per comparison.
RT–qPCR from individual variant strains to validate VARS-seq
We first cloned intron variants of RPS9A and RPL36B individually into the corresponding plasmid backbones to generate complete gene constructs with individual variant introns. For RPS9A, we cloned in eight intron variants, including the wild type, 5′ mutant, 3′ mutant, and rescue sequences for two different barcodes (from gblock_RR15-18 with two barcodes subsequently added by PCR). For RPL36B, we cloned eight intron variants including the wild type, 5′ mutant, 3′ mutant, and rescue sequences for two different stem sets labeled ‘short’ and ‘long’ (cloning from gblock_RR19-26). For RPS9A, gblocks were amplified by PCR with primers RR253 and RR337 to include the barcode expected to have high retained intron levels, and with primers RR253 and RR338 to include the barcode expected to have low levels of retained introns. For RPL36B, gblocks were amplified with primers RR343 and RR345 for the short stem set and with primers RR345 and RR344 for the long stem set. Intron library inserts were assembled into the corresponding linearized backbones (pRR5 and pRR7) using NEBuilder HiFi DNA Assembly (NEB) as described above, with cloning in NEB Stable Competent E. coli cells (NEB).
We next integrated these intron variant constructs into S. cerevisiae to analyze the effects of these variants on splicing. Plasmids extracted from correct clones were linearized by restriction digest, with a first restriction digest by XcmI (NEB) and a second digest with BglII (NEB), as described above. These linearized plasmids included complete gene constructs for RPS9A or RPL36B with intron variants, homology arms to the genomic integration site, and a partial LEU2 selection cassette that would be completed by genomic integration into PLH001. Linearized plasmids were transformed into PLH001 and plated on SD-LEU selection plates. We verified clones with Sanger sequencing of extracted genomic DNA.
We next extracted RNA from each strain and evaluated levels of spliced and unspliced transcripts with qPCR. To extract RNA, we used the YeaStar RNA Kit (Zymo Research) with 5 ml culture with an OD600 of 0.5–0.6, as described above. We treated the samples with TURBO DNase (Thermo Fisher) and cleaned up samples with Zymo5 RNA columns. We then carried out reverse transcription with the iScript Reverse Transcription Supermix (Bio-Rad) along with a no-reverse-transcriptase control, and we purified samples with Oligo Clean and Concentrator columns (Zymo Research).
With this reverse transcription product, we then carried out qPCR with the iTaq Universal SYBR Green Supermix (Bio-Rad), using 1 μl of forward and reverse primer in a 20-μl reaction. Forward primers were designed to avoid the endogenous gene locus by including the fixed 8-nt sequence inserted into the constructs. Reverse primers were designed to amplify only the spliced construct (binding to the exon junction) or only the unspliced construct (binding within the intron). For RPS9A we used primers RR313 and RR314 to amplify the spliced product, and RR317 and RR319 to amplify the unspliced product. For RPL36B we used primers RR346 and RR347 to amplify the spliced product, and RR346 and RR348 to amplify the unspliced product. A region of ACT1 was used as a control interval, with primers RR341 and RR342. All primers were verified by checking amplicons on agarose gels.
De novo computational structure prediction and structure metrics
We predicted secondary structure ensembles for S. cerevisiae introns and control sequence sets with de novo structure prediction. For direct comparison between DMS-guided RNAstructure prediction and de novo RNAstructure prediction, we computed features in both cases only for the 161 introns with a coverage of 1,971 (corresponding to r2 > 0.25) from DMS-MaPseq; for other cases, we made predictions for the complete set of introns filtered to exclude sequences smaller than 50 nt or larger than 600 nt. Three control sequence sets were assembled. First, for the ‘shifted control,’ the genome coordinates for each intron were shifted 500 nt downstream of the 5′ splice site. For the ‘sequence-matched control,’ the shifted control’s sequences were replaced at the positions of splice sites to match the corresponding intron. The final set, the ‘shuffled control,’ was obtained by shuffling each intron’s sequence randomly.
Secondary structure ensemble predictions for each set of sequences were obtained either through minimum free energy predictions or by sampling suboptimal secondary structures. For suboptimal structure sampling, 1,000 structures were sampled using the Arnie package (https://github.com/DasLab/arnie) to call structure prediction executables from Vienna 2.0 (ref. 49) and RNAstructure33. We additionally made structure predictions with 50 nt of surrounding context upstream and downstream of the intron. For each intron and matched control, we computed average values across these structure ensembles for the zipper stem free energy, downstream stem free energy, length of the longest stem, maximum extrusion from ends, and distance between the 5′ splice site and branch point. As described previously, we compared the intron and control values using the non-parametric Wilcoxon ranked-sum test.
Structure prediction for Saccharomyces genus
We obtained multiple sequence alignments for introns in 20 species in the Saccharomyces genus from Hooks et al.50. To count the number of orthologs for each intron, we found all the species with non-empty sequences aligned to the S. cerevisiae intron. We additionally counted the number of zipper stem orthologs by finding all the non-empty regions aligned to the longest zipper stem in each S. cerevisiae intron. Finally, we computed the average percentage sequence conservation across each complete intron and across the intron’s zipper stem, if one was present.
We next evaluated secondary structure feature enrichment for the introns across the Saccharomyces genus. We tested each intron in these 20 species against matched control sets, with each intron having a matching shuffled sequence control. We additionally compared the secondary structure ensembles of all the introns in each of these species’ genomes to phylogenetic controls. To construct phylogenetic control sequence sets, we measured mutation and indel rates between each intron and its homologous S. cerevisiae intron, and we created an intron’s matched control sequence by inserting mutations and indels randomly into the homolog S. cerevisiae intron at this measured average frequency. As described earlier for the analysis of S. cerevisiae introns, for each of these comparisons, we predicted secondary structure ensembles for the intron set and control set, computed secondary structure features, and compared between sequence sets with a Wilcoxon ranked-sum test.
Reagent and resource sharing
Yeast strains are available upon request from the authors. Requests for resources and reagents should be directed to the corresponding author, Rhiju Das (rhiju@stanford.edu).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All sequencing data are available at the Gene Expression Omnibus (GEO) accession number GSE209857. DMS-derived secondary structures are included in Supplementary Table 1. Processed DMS reactivity data, control RNA secondary structures and base-pairing probabilities from structure prediction with bootstrapping can be found at https://github.com/ramyarangan/DMS_intron_analysis. VARS-seq intron variant sequences along with spliced and unspliced read counts are included in Supplementary Tables 3 and 4. Source data for Figs. 1–3 and 5–7, along with Extended Data Figs. 1–5, 8 and 9 are provided with this paper. Source data for the Supplementary figures have been provided as a Supplementary Data File. Source data for Supplementary Fig. 14 is included in Supplementary Table 4.
Code availability
Code for assessing DMS-MaPseq reactivity values, evaluating control RNA structures, and analyzing DMS-derived secondary structures is available at https://github.com/ramyarangan/DMS_intron_analysis. Code for computational secondary structure prediction, secondary structure feature calculation, statistical comparisons, and evolutionary analyses is available at https://github.com/ramyarangan/pre-mRNA_Secstruct. Code for designing VARS-seq sequences and analyzing gDNA and RNA sequencing data from VARS-seq is available at https://github.com/ramyarangan/VARSseq.
References
Wilkinson, M. E., Charenton, C. & Nagai, K. RNA splicing by the spliceosome. Annu. Rev. Biochem. 89, 359–388 (2020).
Wang, Z. & Burge, C. B. Splicing regulation: from a parts list of regulatory elements to an integrated splicing code. RNA 14, 802–813 (2008).
Petibon, C., Parenteau, J., Catala, M. & Elela, S. A. Introns regulate the production of ribosomal proteins by modulating splicing of duplicated ribosomal protein genes. Nucleic Acids Res. 44, 3878–3891 (2016).
Lukacisin, M., Espinosa-Cantu, A. & Bollenbach, T. Intron-mediated induction of phenotypic heterogeneity. Nature 605, 113–118 (2022).
Morgan, J. T., Fink, G. R. & Bartel, D. P. Excised linear introns regulate growth in yeast. Nature 565, 606–611 (2019).
Parenteau, J. et al. Introns are mediators of cell response to starvation. Nature 565, 612–617 (2019).
Plocik, A. M. & Guthrie, C. Diverse forms of RPS9 splicing are part of an evolving autoregulatory circuit. PLoS Genet. 8, e1002620 (2012).
Vincenti, S., De Chiara, V., Bozzoni, I. & Presutti, C. The position of yeast snoRNA-coding regions within host introns is essential for their biosynthesis and for efficient splicing of the host pre-mRNA. RNA 13, 138–150 (2007).
Jo, B. S. & Choi, S. S. Introns: the functional benefits of introns in genomes. Genomics Inf. 13, 112–118 (2015).
Chorev, M. & Carmel, L. The function of introns. Front Genet 3, 55 (2012).
Warf, M. B. & Berglund, J. A. Role of RNA structure in regulating pre-mRNA splicing. Trends Biochem. Sci. 35, 169–178 (2010).
Scharfen, L. & Neugebauer, K. M. Transcription regulation through nascent RNA folding. J. Mol. Biol. 433, 166975 (2021).
Andjus, S., Morillon, A. & Wery, M. From yeast to mammals, the nonsense-mediated mRNA decay as a master regulator of long non-coding rnas functional trajectory. Noncoding RNA 7, 44 (2021).
Roundtree, I. A., Evans, M. E., Pan, T. & He, C. Dynamic RNA modifications in gene expression regulation. Cell 169, 1187–1200 (2017).
Charpentier, B. & Rosbash, M. Intramolecular structure in yeast introns aids early steps of in vitro spliceosome assembly. RNA 2, 509–522 (1996).
Rogic, S. et al. Correlation between the secondary structure of pre-mRNA introns and the efficiency of splicing in Saccharomyces cerevisiae. BMC Genomics 9, 355 (2008).
Parker, R. & Patterson, B. Architecture of Fungal Introns: Implications for Spliceosome Assembly (Academic Press, 1987).
Howe, K. J. & Ares, M. Jr. Intron self-complementarity enforces exon inclusion in a yeast pre-mRNA. Proc. Natl Acad. Sci. USA 94, 12467–12472 (1997).
Meyer, M., Plass, M., Perez-Valle, J., Eyras, E. & Vilardell, J. Deciphering 3′SS selection in the yeast genome reveals an RNA thermosensor that mediates alternative splicing. Mol. Cell 43, 1033–1039 (2011).
Vilardell, J. & Warner, J. R. Regulation of splicing at an intermediate step in the formation of the spliceosome. Genes Dev. 8, 211–220 (1994).
Li, Z., Paulovich, A. G. & Woolford, J. L. Jr. Feedback inhibition of the yeast ribosomal protein gene CRY2 is mediated by the nucleotide sequence and secondary structure of CRY2 pre-mRNA. Mol. Cell. Biol. 15, 6454–6464 (1995).
Fewell, S. W. & Woolford, J. L. Jr. Ribosomal protein S14 of Saccharomyces cerevisiae regulates its expression by binding to RPS14B pre-mRNA and to 18S rRNA. Mol. Cell. Biol. 19, 826–834 (1999).
Gao, W., Jones, T. A. & Rivas, E. Discovery of 17 conserved structural RNAs in fungi. Nucleic Acids Res. 49, 6128–6143 (2021).
Danin-Kreiselman, M., Lee, C. Y. & Chanfreau, G. RNAse III-mediated degradation of unspliced pre-mRNAs and lariat introns. Mol. Cell 11, 1279–1289 (2003).
Li, X. et al. A unified mechanism for intron and exon definition and back-splicing. Nature 573, 375–380 (2019).
Libri, D., Stutz, F., McCarthy, T. & Rosbash, M. RNA structural patterns and splicing: molecular basis for an RNA-based enhancer. RNA 1, 425–436 (1995).
Zubradt, M. et al. DMS-MaPseq for genome-wide or targeted RNA structure probing in vivo. Nat. Methods 14, 75–82 (2017).
Wallace, E. W. J. & Beggs, J. D. Extremely fast and incredibly close: cotranscriptional splicing in budding yeast. RNA 23, 601–610 (2017).
Hansen, S. R., Nikolai, B. J., Spreacker, P. J., Carrocci, T. J. & Hoskins, A. A. Chemical inhibition of pre-mRNA splicing in living Saccharomyces cerevisiae. Cell Chem. Biol. 26, 443–448 (2019).
Hunter, O. et al. Broad variation in response of individual introns to splicing inhibitors in a humanized yeast strain. RNA 30, 149–170 (2024).
Parker, R. RNA degradation in Saccharomyces cerevisae. Genetics 191, 671–702 (2012).
Losson, R. & Lacroute, F. Interference of nonsense mutations with eukaryotic messenger RNA stability. Proc. Natl Acad. Sci. USA 76, 5134–5137 (1979).
Reuter, J. S. & Mathews, D. H. RNAstructure: software for RNA secondary structure prediction and analysis. BMC Bioinf. 11, 129 (2010).
Cordero, P., Kladwang, W., VanLang, C. C. & Das, R. Quantitative dimethyl sulfate mapping for automated RNA secondary structure inference. Biochemistry 51, 7037–7039 (2012).
Kladwang, W., VanLang, C. C., Cordero, P. & Das, R. Understanding the errors of SHAPE-directed RNA structure modeling. Biochemistry 50, 8049–8056 (2011).
Zhang, Z. et al. Structural insights into how Prp5 proofreads the pre-mRNA branch site. Nature 596, 296–300 (2021).
Hooks, K. B., Naseeb, S., Parker, S., Griffiths-Jones, S. & Delneri, D. Novel intronic RNA structures contribute to maintenance of phenotype in Saccharomyces cerevisiae. Genetics 203, 1469–1481 (2016).
Yao, Z., Weinberg, Z. & Ruzzo, W. L. CMfinder—a covariance model based RNA motif finding algorithm. Bioinformatics 22, 445–452 (2006).
Gruber, A. R., Neubock, R., Hofacker, I. L. & Washietl, S. The RNAz web server: prediction of thermodynamically stable and evolutionarily conserved RNA structures. Nucleic Acids Res. 35, W335–W338 (2007).
Pedersen, J. S. et al. Identification and classification of conserved RNA secondary structures in the human genome. PLoS Comput. Biol. 2, e33 (2006).
Piekna-Przybylska, D., Decatur, W. A. & Fournier, M. J. New bioinformatic tools for analysis of nucleotide modifications in eukaryotic rRNA. RNA 13, 305–312 (2007).
Rivas, E., Clements, J. & Eddy, S. R. A statistical test for conserved RNA structure shows lack of evidence for structure in lncRNAs. Nat. Methods 14, 45–48 (2017).
Ogden, R. C., Lee, M. C. & Knapp, G. Transfer RNA splicing in Saccharomyces cerevisiae: defining the substrates. Nucleic Acids Res. 12, 9367–9382 (1984).
Trotta, C. R. et al. The yeast tRNA splicing endonuclease: a tetrameric enzyme with two active site subunits homologous to the archaeal tRNA endonucleases. Cell 89, 849–858 (1997).
Sun, L. et al. RNA structure maps across mammalian cellular compartments. Nat. Struct. Mol. Biol. 26, 322–330 (2019).
Cheng, C. Y., Kladwang, W., Yesselman, J. D. & Das, R. RNA structure inference through chemical mapping after accidental or intentional mutations. Proc. Natl Acad. Sci. USA 114, 9876–9881 (2017).
Neuveglise, C., Marck, C. & Gaillardin, C. The intronome of budding yeasts. C. R. Biol. 334, 662–670 (2011).
Lee, M. E., DeLoache, W. C., Cervantes, B. & Dueber, J. E. A highly characterized yeast toolkit for modular, multipart assembly. ACS Synth. Biol. 4, 975–986 (2015).
Lorenz, R. et al. ViennaRNA package 2.0. Algorithms Mol. Biol. 6, 26 (2011).
Hooks, K. B., Delneri, D. & Griffiths-Jones, S. Intron evolution in Saccharomycetaceae. Genome Biol. Evolution 6, 2543–2556 (2014).
Bubenik, J. L. et al. RNA structure probing to characterize RNA-protein interactions on a low abundance pre-mRNA in living cells. RNA 27, 343–358 (2020).
Liu, Z. et al. In vivo nuclear RNA structurome reveals RNA-structure regulation of mRNA processing in plants. Genome Biol. 22, 11 (2021).
Ares, M. Jr., Grate, L. & Pauling, M. H. A handful of intron-containing genes produces the lion’s share of yeast mRNA. RNA 5, 1138–1139 (1999).
Galy, V. et al. Nuclear retention of unspliced mRNAs in yeast is mediated by perinuclear Mlp1. Cell 116, 63–73 (2004).
Bousquet-Antonelli, C., Presutti, C. & Tollervey, D. Identification of a regulated pathway for nuclear pre-mRNA turnover. Cell 102, 765–775 (2000).
Sayani, S., Janis, M., Lee, C. Y., Toesca, I. & Chanfreau, G. F. Widespread impact of nonsense-mediated mRNA decay on the yeast intronome. Mol. Cell 31, 360–370 (2008).
Lu, Z. et al. RNA duplex map in living cells reveals higher-order transcriptome structure. Cell 165, 1267–1279 (2016).
Wu, T. et al. KARR-seq reveals cellular higher-order RNA structures and RNA-RNA interactions. Nat. Biotechnol. 42, 1909–1920 (2024).
Barak, M. et al. Purifying selection of long dsRNA is the first line of defense against false activation of innate immunity. Genome Biol. 21, 26 (2020).
Simms, C. L., Yan, L. L. & Zaher, H. S. Ribosome collision is critical for quality control during no-go decay. Mol. Cell 68, 361–373 (2017).
Rivas, E., Clements, J. & Eddy, S. R. Estimating the power of sequence covariation for detecting conserved RNA structure. Bioinformatics 36, 3072–3076 (2020).
Schirman, D., Yakhini, Z., Pilpel, Y. & Dahan, O. A broad analysis of splicing regulation in yeast using a large library of synthetic introns. PLoS Genet. 17, e1009805 (2021).
Rosenberg, A. B., Patwardhan, R. P., Shendure, J. & Seelig, G. Learning the sequence determinants of alternative splicing from millions of random sequences. Cell 163, 698–711 (2015).
Abelson, J. et al. Conformational dynamics of single pre-mRNA molecules during in vitro splicing. Nat. Struct. Mol. Biol. 17, 504–512 (2010).
Tan, J. et al. A disease-causing intronic point mutation C19G alters tau exon 10 splicing via RNA secondary structure rearrangement. Biochemistry 58, 1565–1578 (2019).
Shilo, A., Tosto, F. A., Rausch, J. W., Le Grice, S. F. J. & Misteli, T. Interplay of primary sequence, position and secondary RNA structure determines alternative splicing of LMNA in a pre-mature aging syndrome. Nucleic Acids Res. 47, 5922–5935 (2019).
Saha, K. et al. Structural disruption of exonic stem-loops immediately upstream of the intron regulates mammalian splicing. Nucleic Acids Res. 48, 6294–6309 (2020).
Graveley, B. R. Mutually exclusive splicing of the insect Dscam pre-mRNA directed by competing intronic RNA secondary structures. Cell 123, 65–73 (2005).
Saldi, T., Riemondy, K., Erickson, B. & Bentley, D. L. Alternative RNA structures formed during transcription depend on elongation rate and modify RNA processing. Mol. Cell 81, 1789–1801 (2021).
Fish, L. et al. A prometastatic splicing program regulated by SNRPA1 interactions with structured RNA elements. Science 372, eabc7531 (2021).
Casas-Vila, N., Sayols, S., Perez-Martinez, L., Scheibe, M. & Butter, F. The RNA fold interactome of evolutionary conserved RNA structures in S. cerevisiae. Nat. Commun. 11, 2789 (2020).
Gosai, S. J. et al. Global analysis of the RNA-protein interaction and RNA secondary structure landscapes of the Arabidopsis nucleus. Mol. Cell 57, 376–388 (2015).
Hilleren, P. J. & Parker, R. Cytoplasmic degradation of splice-defective pre-mRNAs and intermediates. Mol. Cell 12, 1453–1465 (2003).
Plaschka, C., Lin, P. C., Charenton, C. & Nagai, K. Prespliceosome structure provides insights into spliceosome assembly and regulation. Nature 559, 419–422 (2018).
Jeong, H., Herskowitz, I., DKroetz, D. L. & Rine, J. Function-altering SNPs in the human multidrug transporter gene ABCB1 identified using a Saccharomyces-based assay. PLoS Genet. 3, e39 (2007).
Rouskin, S., Zubradt, M., Washietl, S., Kellis, M. & Weissman, J. S. Genome-wide probing of RNA structure reveals active unfolding of mRNA structures in vivo. Nature 505, 701–705 (2014).
Smith, T., Heger, A. & Sudbery, I. UMI-tools: modeling sequencing errors in unique molecular identifiers to improve quantification accuracy. Genome Res. 27, 491–499 (2017).
Talkish, J. et al. Rapidly evolving protointrons in Saccharomyces genomes revealed by a hungry spliceosome. PLoS Genet. 15, e1008249 (2019).
Cherry, J. M. et al. Saccharomyces Genome Database: the genomics resource of budding yeast. Nucleic Acids Res. 40, D700–D705 (2012).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Busan, S. & Weeks, K. M. Accurate detection of chemical modifications in RNA by mutational profiling (MaP) with ShapeMapper 2. RNA 24, 143–148 (2018).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Incarnato, D., Morandi, E., Simon, L. M. & Oliviero, S. RNA Framework: an all-in-one toolkit for the analysis of RNA structures and post-transcriptional modifications. Nucleic Acids Res. 46, e97 (2018).
Kawashima, T., Douglass, S., Gabunilas, J., Pellegrini, M. & Chanfreau, G. F. Widespread use of non-productive alternative splice sites in Saccharomyces cerevisiae. PLoS Genet. 10, e1004249 (2014).
Hossain, M. A., Rodriguez, C. M. & Johnson, T. L. Key features of the two-intron Saccharomyces cerevisiae gene SUS1 contribute to its alternative splicing. Nucleic Acids Res. 39, 8612–8627 (2011).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Ben-Shem, A. et al. The structure of the eukaryotic ribosome at 3.0 A resolution. Science 334, 1524–1529 (2011).
Lu, X. J. & Olson, W. K. 3DNA: a software package for the analysis, rebuilding and visualization of three-dimensional nucleic acid structures. Nucleic Acids Res. 31, 5108–5121 (2003).
Nguyen, T. H. D. et al. Cryo-EM structure of the yeast U4/U6.U5 tri-snRNP at 3.7 A resolution. Nature 530, 298–302 (2016).
Li, X. et al. CryoEM structure of Saccharomyces cerevisiae U1 snRNP offers insight into alternative splicing. Nat. Commun. 8, 1035 (2017).
Kalvari, I. et al. Rfam 14: expanded coverage of metagenomic, viral and microRNA families. Nucleic Acids Res. 49, D192–D200 (2021).
Lan, P. et al. Structural insight into precursor tRNA processing by yeast ribonuclease P. Science 362, eaat6678 (2018).
Rodgers, M. L. et al. Conformational dynamics of stem II of the U2 snRNA. RNA 22, 225–236 (2016).
Hajdin, C. E. et al. Accurate SHAPE-directed RNA secondary structure modeling, including pseudoknots. Proc. Natl Acad. Sci. USA 110, 5498–5503 (2013).
Tomezsko, P. J. et al. Determination of RNA structural diversity and its role in HIV-1 RNA splicing. Nature 582, 438–442 (2020).
Brown, S. S. et al. PrimerPooler: automated primer pooling to prepare library for targeted sequencing. Biol. Methods Protoc. 2, bpx006 (2017).
Rivas, E. RNA structure prediction using positive and negative evolutionary information. PLoS Comput. Biol. 16, e1008387 (2020).
Benson, D. A. et al. GenBank. Nucleic Acids Res. 41, D36–D42 (2013).
Watkins, A. M., Rangan, R. & Das, R. FARFAR2: improved de novo rosetta prediction of complex global RNA folds. Structure 28, 963–976 (2020).
Watkins, A. M., Rangan, R. & Das, R. Using Rosetta for RNA homology modeling. Methods Enzymol. 623, 177–207 (2019).
Bernhart, S. H. et al. Partition function and base pairing probabilities of RNA heterodimers. Algorithms Mol. Biol. 1, 3 (2006).
Bai, R., Wan, R., Yan, C., Lei, J. & Shi, Y. Structures of the fully assembled Saccharomyces cerevisiae spliceosome before activation. Science 360, 1423–1429 (2018).
Plaschka, C., Lin, P. C. & Nagai, K. Structure of a pre-catalytic spliceosome. Nature 546, 617–621 (2017).
Yan, C., Wan, R., Bai, R., Huang, G. & Shi, Y. Structure of a yeast activated spliceosome at 3.5 A resolution. Science 353, 904–911 (2016).
Galej, W. P. et al. Cryo-EM structure of the spliceosome immediately after branching. Nature 537, 197–201 (2016).
Yan, C., Wan, R., Bai, R., Huang, G. & Shi, Y. Structure of a yeast step II catalytically activated spliceosome. Science 355, 149–155 (2017).
Roy, K. R. et al. Spliceosomal mutations decouple 3’ splice site fidelity from cellular fitness. Preprint at bioRxiv https://doi.org/10.1101/2023.01.12.523824 (2023).
Hagberg, A., Schult, D. & Swart, P. Exploring network structure, dynamics, and function using NetworkX. In Proc. 7th Python in Science conference (SciPy 2008) (eds. Varoquaux, G., Vaught, T. & Millman, J.) 11–15 (2008).
Cheng, C. Y., Chou, F. C. & Das, R. Modeling complex RNA tertiary folds with Rosetta. Methods Enzymol. 553, 35–64 (2015).
Virtanen, P. et al. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat. Methods 17, 261–272 (2020).
Do, C. B., Woods, D. A. & Batzoglou, S. CONTRAfold: RNA secondary structure prediction without physics-based models. Bioinformatics 22, e90–e98 (2006).
Curran, K. A., Karim, A. S., Gupta, A. & Alper, H. S. Use of expression-enhancing terminators in Saccharomyces cerevisiae to increase mRNA half-life and improve gene expression control for metabolic engineering applications. Metab. Eng. 19, 88–97 (2013).
McMillan, J., Lu, Z., Rodriguez, J. S., Ahn, T. H. & Lin, Z. YeasTSS: an integrative web database of yeast transcription start sites. Database (Oxford) 2019, baz048 (2019).
Reider Apel, A. et al. A Cas9-based toolkit to program gene expression in Saccharomyces cerevisiae. Nucleic Acids Res. 45, 496–508 (2017).
Mulleder, M. et al. A prototrophic deletion mutant collection for yeast metabolomics and systems biology. Nat. Biotechnol. 30, 1176–1178 (2012).
Norrander, J., Kempe, T. & Messing, J. Construction of improved M13 vectors using oligodeoxynucleotide-directed mutagenesis. Gene 26, 101–106 (1983).
Gietz, R. D. & Woods, R. A. Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Methods Enzymol. 350, 87–96 (2002).
Bushnell, B., Rood, J. & Singer, E. BBMerge—accurate paired shotgun read merging via overlap. PLoS ONE 12, e0185056 (2017).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Wu, T. D. & Watanabe, C. K. GMAP: a genomic mapping and alignment program for mRNA and EST sequences. Bioinformatics 21, 1859–1875 (2005).
Acknowledgements
We thank K. B. Hooks (Faculty of Life Sciences, University of Manchester, M13 9PT, United Kingdom) and D. Delneri (Faculty of Life Sciences, University of Manchester, M13 9PT, United Kingdom) for providing secondary structure data upon request. We thank A. Hoskins, J. Puglisi, T. Lan and S. Rouskin for illuminating discussions on this project, and we thank V. V. Topkar and I. Zheludev for advice on experimental protocols. We thank Stanford University for providing computational resources and support for the Sherlock 2.0 cluster that contributed to these results. This work was supported by the National Science Foundation Graduate Research Fellowship Program grant no. 1650114 (R.R.); the Gerald J. Lieberman Fellowship (R.R.); the National Institutes of Health grant R35GM145266 (M.A.), a Stanford Bio-X Interdisciplinary Initiative Award (R.D.); the National Institutes of Health grant R35GM122579 (R.D.) and the Howard Hughes Medical Institute (HHMI) Investigator Program (R.D.). This article is subject to HHMI’s Open Access to Publications policy. HHMI lab heads have previously granted a nonexclusive CC BY 4.0 license to the public and a sublicensable license to HHMI in their research articles. Pursuant to those licenses, the author-accepted manuscript of this article can be made freely available under a CC BY 4.0 license immediately upon publication. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
R.R. and R.D. conceived the project. R.R. designed and performed experiments and carried out computational analyses. O.H. and M.A. developed the S. cerevisiae strain OHY001 used in the project. R.H. conducted in vitro and targeted DMS probing experiments. P.P. tested alternate structure prediction approaches. R.R., M.A. and R.D. interpreted the results. R.R. wrote the initial draft, and R.R., M.A. and R.D. wrote the final manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Structural & Molecular Biology thanks Aaron Hoskins and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor Dimitris Typas was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 DMS-MaPseq data quality.
A) Per-base mutational frequencies. B) Accuracy of mutational frequency values for rRNA residues. C) HAC1 and ASH1 positive control structures with overlaid reactivity profiles.
Extended Data Fig. 2 Support from DMS reactivity for in vivo formation of control structures.
A) PPV, B) sensitivity, and C) F1 score for structure prediction of a set of control RNA structures (rRNAs, tRNAs, snRNAs, and mRNAs; Supplementary Table 2), using RNAstructure guided by DMS with varying helix confidence estimate cutoffs for calling stems. The black dotted line represents the helix confidence estimate 0.7 chosen in this paper. The red dotted line represents the PPV, sensitivity, and F1 score for Vienna RNA structure prediction without using DMS data. D)-E) DMS-MaPseq structure prediction for the U1 snRNA compared to the native secondary structure. D) DMS-guided secondary structure prediction for the U1 snRNA, with reactivity values overlaid and helix confidence estimates indicated in green percentages. E) Native secondary structure for the U1 snRNA, with DMS reactivity overlaid along with helix confidence estimates. For the native structure, helix confidence estimates were computed as the percent of bootstrapping iterations where the helix was recovered when sampling DMS reactivity values and making structure predictions.
Extended Data Fig. 3 DMS-MaPseq and targeted DMS probing reproducibility between replicates.
A) r2 between replicates for each intron in S. cerevisiae versus the average sequencing coverage between replicates. B) Bar graph comparing the number of high confidence stems that are shared vs discordant between replicates for introns in different ranges of replicate correlation. C) r2 between replicates of targeted DMS probing for 30 introns without (left) or with (right) heat denaturation. D) The number of high confidence stems found in 24 introns with r2 > 0.9 from targeted DMS probing and r2 > 0.6 from DMS-MaPseq. E) The number of high confidence stems found in 22 introns with r2 > 0.9 from targeted DMS probing of denatured RNA and r2 > 0.6 from DMS-MaPseq.
Extended Data Fig. 4 Structural modeling with the A state spliceosome.
A) Sample Rosetta models of introns with varying linker lengths to identify linker lengths for zipper stems compatible with the A state spliceosome structure. B) Penalty for chain breaks as modeled linker length increases. Data are presented as mean values +/- standard deviation. C) Top 3 models for RPL36B intron in the context of the A state spliceosome, with the RPL36B intron secondary structure specified from DMS-MaPseq.
Extended Data Fig. 5 Secondary structure features for introns and non-splicing decoy sequences.
A) Intron and decoy sequence schematic. Nucleotide lengths depicted are representative lengths for introns and decoy sequences, with decoy sequences chosen to have 5’ splice site, branch point, and 3’ splice site placements matching length distributions from canonical introns (see Methods). B) Secondary structure features are enriched in standard canonical spliced introns in yeast, but not in decoy sequences (genomic intervals which match splice site sequences and yet do not splice). *p-value < 0.01 by two-sided Wilcoxon ranked-sum test. Left: N = 140 canonical introns were compared to controls, yielding p-values (left to right): <1E-4, 0.00027, 0.00072, <1E-4, <1E-4. Right: N = 167 decoy introns were compared to controls, yielding p-values (left to right): 0.74, 0.23, 0.68, 0.37, 0.10. Embedded box plots mark the median as the center white point and include a box from the 25th (Q1) to 75th (Q3) percentile, extending whiskers to the smallest and largest value that fall within 1.5 times the interquartile range below Q1 and above Q3.
Extended Data Fig. 6 Multidimensional chemical mapping for RPL36B.
A) In vitro M2-seq Z scores for the intron in RPL36B, with peaks representing helices annotated in red. B) In vitro chemical reactivity base-pairing probabilities for RPL36B using 1D and 2D chemical reactivity from M2-seq. Secondary structure predictions guided by 1D and 2D DMS probing data for the intron in RPL36B C) from in vitro M2-seq, and D) from in vivo DMS-MaPseq.
Extended Data Fig. 7 Base-pairing probabilities and secondary structure predictions from in vitro M2-seq and in vivo DMS-MaPseq.
Base-pairing probabilities and secondary structure predictions are shown for introns in RPS11A (A, B), RPL37A (C, D), RPS7B (E, F). (B, D, F) Secondary structures are guided by 1D and 2D DMS probing data from M2-seq (left) or by 1D DMS probing data from in vivo DMS-MaPseq (right).
Extended Data Fig. 8 Structure variant library design to measure spliced and unspliced RNA levels for intron variants.
A) Computational pipeline for designing variant and rescue sequences to assess stem and loop sets in an intron. B) Sample base-pair probability matrix comparison between a set of wildtype (left), variant (middle), and rescue (right) sequences. The stem set targeted by this variant and rescue sequence are bracketed. C) Constructs used for constructing the landing pad strain LPL001 (top) and for transforming the intron library via genomic integration into LPL001 (bottom). D) Based on simulations sampling 10,000 or 50,000 random barcodes, the number of barcodes within 2, 3, or 4 edit distance of another barcode. The vertical line indicates the chosen barcode length (12).
Extended Data Fig. 9 Effects of structure variants on retained intron levels for RPS9A variants.
For a given stem set or loop, violin plots depict data for the wildtype sequence and all variant sequences, with points for each unique barcode. Data for rescue sequences are shown when included in the intron library. p-values (computed by two-sided permutation tests) are indicated for comparisons between wildtype and variant sequences, and between variant and rescue sequences. There are 67 wildtype sequences compared to variant sequences in each plot. The numbers of variant sequences are as follows: 12 for stem 51-58, 75 for stem 73-79, 11 for stems 73-84, 39 for stems 73-96, 10 for stems 102-114, 81 for stem 191-195, 44 for stems 184-195, 60 for stems 182-195, 74 for stems 171-195, and 51 for loop 196-205. The numbers of rescue sequences are as follows: 18 for stem 191-195, 46 for stems 184-195, 25 for stems 182-195, and 24 for stems 171-195. Embedded box plots mark the median as the center white point and include a box from the 25th (Q1) to 75th (Q3) percentile, extending whiskers to the smallest and largest value that fall within 1.5 times the interquartile range below Q1 and above Q3. Secondary structures are colored by reactivity data, and bars alongside the secondary structure indicate stem and loop disruption sets, with each bar representing variant sequences mutating nucleotides across the full extent of the bar. These bars are colored by the retained intron (RI) score for the interval. The RI score is the negative log(p-value) comparing RI values between wildtype and variant sequences, and the sign indicates the effect direction, with positive values (shown as blue) for lower variant RI compared to wildtype, and negative values (shown as red) for higher variant RI.
Extended Data Fig. 10 One-page overview of secondary structures and overlaid DMS reactivity profiles for all introns with sufficient coverage from DMS-MaPseq.
Introns are grouped into classes from hierarchical clustering (Fig. 5). Secondary structures are depicted schematically using RiboGraphViz (https://github.com/DasLab/RiboGraphViz).
Supplementary information
Supplementary Information (download PDF )
Supplementary Text, Supplementary Figs. 1–20, Supplementary Table 2 and Source data for Supplementary Fig. 13.
Supplementary Tables (download XLSX )
Supplementary Tables 1 and 3–6.
Supplementary Data 1 (download ZIP )
Source data for supplementary figures.
Source data
Source Data Fig. 1 (download XLSX )
Source data for plots.
Source Data Fig. 1 (download PDF )
Unprocessed gels for Fig. 1.
Source Data Fig. 2 (download XLSX )
Source data for plots.
Source Data Fig. 3 (download TXT )
Source data for plots.
Source Data Fig. 5 (download XLSX )
Source data for plots.
Source Data Fig. 6 (download XLSX )
Source data for plots.
Source Data Fig. 7 (download XLSX )
Source data for plots.
Source Data Extended Data Fig. 1 (download XLSX )
Source data for plots.
Source Data Extended Data Fig. 2 (download XLSX )
Source data for plots.
Source Data Extended Data Fig. 3 (download XLSX )
Source data for plots.
Source Data Extended Data Fig. 4 (download TXT )
Source data for plots.
Source Data Extended Data Fig. 5 (download XLSX )
Source data for plots.
Source Data Extended Data Fig. 8 (download XLSX )
Source data for plots.
Source Data Extended Data Fig. 9 (download XLSX )
Source data for plots.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rangan, R., Huang, R., Hunter, O. et al. Comprehensive analysis of Saccharomyces cerevisiae intron structures in vivo. Nat Struct Mol Biol 32, 1488–1502 (2025). https://doi.org/10.1038/s41594-025-01565-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41594-025-01565-x
This article is cited by
-
Mechanism of alternative splicing of yeast HEH1 through competing 5’ splice sites
Nature Communications (2025)