Abstract
The host-produced nodule specific cysteine-rich (NCR) peptides control the terminal differentiation of endosymbiotic rhizobia in the nodules of IRLC legumes. Although the Medicago truncatula genome encodes about 700 NCR peptides, only few of them have been proven to be crucial for nitrogen-fixing symbiosis. In this study, we applied the CRISPR/Cas9 gene editing technology to generate knockout mutants of NCR genes for which no genetic or functional data were previously available. We have developed a workflow to analyse the mutation and the symbiotic phenotype of individual nodules formed on Agrobacterium rhizogenes-mediated transgenic hairy roots. The selected NCR genes were successfully edited by the CRISPR/Cas9 system and nodules formed on knockout hairy roots showed wild type phenotype indicating that peptides NCR068, NCR089, NCR128 and NCR161 are not essential for symbiosis between M. truncatula Jemalong and Sinorhizobium medicae WSM419. We regenerated stable mutants edited for the NCR068 from hairy roots obtained by A. rhizogenes-mediated transformation. The analysis of the symbiotic phenotype of stable ncr068 mutants showed that peptide NCR068 is not required for symbiosis with S. meliloti strains 2011 and FSM-MA either. Our study reports that gene editing can help to elicit the role of certain NCRs in symbiotic nitrogen fixation.
Similar content being viewed by others
Introduction
Most legumes establish nitrogen-fixing symbiotic interaction with soil bacteria termed rhizobia. Compatible interaction between the host legumes and their rhizobia partner induces the formation of a specialised organ on the root, referred to as nodules. Rhizobia colonize the symbiotic cells of the nodules and differentiate into their symbiotic form, the bacteroids which convert the atmospheric nitrogen into ammonia1. The indeterminate nodules formed on the roots of temperate legumes, such as the model legume Medicago truncatula, possess a persistent apical meristem2. The continuous activity of the meristem (zone I) produces cells creating a developmental gradient in the nodules, whereupon these nodules are cylindrical in shape. The new cells mature, many of them become infected in zone II (infection zone) wherein bacteria are engulfed by a plant-derived membrane forming new organelles, the symbiosomes. Infected host cells and rhizobia differentiate in interzone (IZ) and the reduction of nitrogen takes place in zone III (nitrogen fixation zone). The completely developed and effectively nitrogen-fixing nodules produce leghemoglobin which gives wild type nodules their pink colour.
Rhizobia in the nodules of legumes belonging to the inverted repeat–lacking clade (IRLC), including M. truncatula, and some Dalbergoid species undergo irreversible differentiation mediated by nodule-specific cysteine-rich (NCR) peptides3,4,5. These host produced peptides contain four or six cysteine residues in conserved positions and possess signal peptides which target them to the symbiosomes through a nodule-specific protein secretory pathway6,7 where they govern the transition of rhizobia into bacteroids. The M. truncatula genome encodes a large family of NCR peptides with more than 700 members8,9. Although this high number of peptides show remarkable amino acid sequence variation, their function was initially presumed redundant. Nevertheless, recent studies have identified some peptides, NCR169, NCR211, NCR247, NCR343 and NCR-new35, crucial for the bacteroid differentiation and persistence in M. truncatula nodules but also found peptides (NCR178, NCR341, NCR344 and NCR345) which are not essential for nitrogen-fixing symbiosis10,11,12,13,14,15. The identification of most of these essential peptides was random during the forward genetic analyses of mutagenized M. truncatula populations. Therefore, the reverse genetic approach provides a comprehensive and targeted way to analyse the function of the NCR gene family members. The recently developed CRISPR/Cas9 (Clustered Regularly Interspaced Short Palindromic Repeats/CRISPR-associated protein 9) system is a simple and precise tool for targeted gene editing and analyse gene function in various plants16. Several reports described the application of CRISPR/Cas9 gene editing system in legume species (soybean17; peanut18; pea19; Lotus japonicus20 including M. truncatula21,22,23,24. Unfortunately, most legume species are largely recalcitrant to somatic embryogenesis and in vitro plant regeneration, therefore generating stable transformant legume plants using the Agrobacterium tumefaciens-mediated transformation system is a cumbersome and time-consuming process. In contrast, the A. rhizogenes-mediated hairy root transformation does not need complete plant regeneration, therefore it provides an efficient tool for a fast and large-scale functional analysis of root-related traits, such as symbiotic nodule development. Despite its robustness, the A. rhizogenes-mediated hairy root transformation has some disadvantages. For instance, the transformation efficiency varies, and gene transfer is not achieved in all root cells25, or the emerged hairy roots develop from multiple cells with different gene editing events which can result in mosaicism of transformed and untransformed cells or even multiple genotypes. This could cause trouble during gene editing of a wild-type genome to generate knockouts because a small amount of residual wild-type cells could affect the phenotype to be analysed. Therefore, the removal of untransformed or partially transformed roots and analysing the genotype of the whole hairy root is particularly important.
The CRISPR/Cas9 gene editing system, has been applied successfully to knockout and analyse the requirement of peptides NCR247, NFS1 and NFS212,26,27 using A. rhizogenes-mediated hairy root transformation. In these reports, the targeted NCR genes were selected based on preliminary genetic or functional studies. In this study, we optimized the CRISPR/Cas9 system using hairy root transformation to generate mutation in selected NCR genes and developed a workflow to conduct their functional analysis for symbiosis.
Results
Testing the efficiency of a modified vector for CRISPR/Cas9 gene editing system to target NCR169
To analyse the function of NCR genes, the CRISPR/Cas9 gene editing technology was used to generate composite Medicago truncatula plants mutated in hairy roots. In order to monitor the presence of CRISPR constructs, delivered into the genome of M. truncatula root cells using the Agrobacterium rhizogenes-mediated hairy-root system28, the gene coding for the DsRed fluorescent transformation marker was introduced into the CRISPR/Cas9 plant transformation vector pKSE401 resulting in the vector pKSE401-RR. The constitutively active red fluorescent marker allowed us to remove non-transgenic roots at the early stages to promote the growth of transgenic hairy roots. To test the efficiency of targeted mutagenesis of pKSE401-RR, we designed three different single guide RNA (sgRNA) constructs targeting the NCR169 gene (Fig. S1), previously proved to be essential for effective nitrogen-fixing symbiosis10. Each sgRNA (NCR169a, b and c) was designed directing to the first half of the coding sequence of NCR169 but the initial 5’ nucleotides for the protospacer sequences of NCR169b and NCR169c were C instead of the optimal G(/A) which is preferred by the AtU6 promoter (Fig. S1a;29). Nevertheless, we used these two suboptimal sgRNA constructs as well because the short, 189 bp long coding sequence of NCR169 constrained the available optimal target sequences. The three constructs carrying each sgRNAs were introduced into roots of wild-type M. truncatula 2HA genotype using A. rhizogenes-mediated hairy root transformation. At least ten transformed roots for each construct, detected by the DsRed fluorescent marker, were sampled and used for genomic DNA isolation. The region surrounding the targeted sequence in NCR169 was amplified by PCR. The sequence analysis of the amplicons detected only wild-type genotypes on all the NCR169b-transformed roots and roots transformed with the construct NCR169c were heterogeneous of wild-type and various mutant genotypes. However, the construct NCR169a generated mutations at higher efficiency and besides chimeric roots having both wild-type and distinct mutations, we could identify roots with homozygous or biallelic mutations for NCR169 (Fig. S1d). The results indicated that the presence of G at the 5’ end of the protospacer sequence (G(/A)N19NGG) is crucial and using C in this position greatly reduced the efficiency of genome editing which restricts the number of possible target sites in the case of small genes such as NCRs. In addition, the sequence analysis of amplicons containing the targeted region revealed that the sequences obtained by Sanger method was composed of multiplex sequence traces around the break site (Fig. S1b). An online tool, TIDE, enabling the decomposition of mixed sequence signals was applied30 that predicted the types of the mutation and the size of the deletion or insertion. For instance, the decomposition of one of the amplicons of NCR169c disclosed the mixture of wild-type and CRISPR/Cas9 edited sequences containing a 7 bp deletion and/or a one bp insertion (Fig. S1c).
In agreement with a previous report22, we also found that different genome editing events occurred in distinct hairy roots of a single plant which necessitates the analysis of individual nodules separately. These findings led us to the conclusion that the effectiveness of genome editing and the reliability of the identification of the nature of genome editing events needed to be improved in our system. Therefore (i) we replaced the Arabidopsis U6-26 promoter with the M. truncatula U6 (MtU6.6) promoter driving sgRNA synthesis, (ii) we developed a preparation procedure allowing phenotypic and genotypic analysis of individual nodules, and (iii) applied an NGS (Next Generation Sequencing) amplicon sequencing approach to assess allele distribution in individual nodules.
Inducing mutations in NCR169 and NCR-new35 using the optimized CRISPR/Cas9-mediated genome editing system
Previous studies found that the M. truncatula U6 promoter is optimal to express guide or hairpin RNAs efficiently in legume species21,31. Therefore, the Arabidopsis U6-26 promoter was replaced with MtU6.6 in the pKSE401-RR resulting in the vector pKSE466-RR. We inserted the sgRNAs NCR169a and another one, targeting the NCR-new35, of which requirement for effective symbiosis has been recently demonstrated13, into pKSE466-RR, respectively. To validate the efficiency of the pKSE466-RR vector construct, the empty vector and pKSE466-RR-NCR169a were introduced into wild-type M. truncatula 2HA plants using A. rhizogenes-mediated hairy root transformation and plants were inoculated with S. medicae WSM419. Nodules developed on DsRed fluorescent transformed roots were harvested 3–4 weeks post inoculation (wpi) with rhizobia, individually fixed and sections were stained with nucleic acid-binding dye SYTO13 (Fig. 1). Following image capturing, genomic DNA was purified from nodule sections and the regions surrounding the targeted sequences were amplified by PCR (Figs. 1, 2).
Plant gross and nodulation phenotype of Agrobacterium rhizogenes-mediated transgenic Medicago truncatula Jemalong 2HA hairy-roots mutagenized with CRISPR/Cas9 gene editing. No sign of nitrogen deficiency was observed on plants transformed with empty vector (a) or the construct targeting NCR068 (c) 4 weeks post inoculation with S. medicae WSM419. Transgenic roots were identified by fluorescence of DsRed protein. Elongated pink nodules, showing the characteristic features of indeterminate nodules, developed on transgenic roots. These nodules were fully colonized by rhizobia visualized by the fluorescent dye SYTO13 (d,f). (b) Targeted mutagenesis of NCR169 resulted in transgenic plants showing the symptoms of nitrogen starvation. Small, spherical and white ineffective nodules were formed on NCR169-targeted roots. (e) ncr169 mutant nodules did not contain infected cells in the region corresponding to the nitrogen fixation zone of wild-type plants. Inset images in (d–f) show the higher magnification of cells in nodule regions indicated by white rectangles. (g,h) The targeted regions were amplified and sequenced from the transgenic nodules. The sequence analysis revealed biallelic mutations causing frameshifts in edited NCR169 (g) and NCR068 (h) genes. ZII, infection zone; IZ, interzone; ZIII, nitrogen fixation zone; EV, empty vector.
The nodulation phenotype of hairy roots transformed with the construct pKSE466-RR-MtNCR169a was associated with the proportion of wild-type NCR169 allele or with the presence of mutant NCR169 lacking a single residue. White (Fix−) and pink (Fix+) nodules were randomly harvested 3–5 wpi with S. medicae WSM419 and the result of gene editing was identified by the sequencing of the targeted region using an Illumina platform. The ratios of reads assigned to mutant (brown ochre) and wild-type (blue) alleles are presented in pie charts. Fix- nodules (b,d, f) carried almost exclusively insertions or deletions resulting in frameshift in the coding sequence of NCR169 (a,c,e). Contrary, pink nodules (g,i,k) carried either unmodified alleles on a large scale (> 40%) (h,j) or a deletion of 3 base pairs that did not result in the loss of function of NCR169 (l).
The elongated nodules developed on empty vector-transformed roots were pink and showed the characteristic zonation of indeterminate nodules (Fig. 1a,d). The nodules on the roots of M. truncatula 2HA transformed with the pKSE466-RR-NCR169a were either pink and elongated, suggesting that these nodules were functional wild-type (Fig. 2g,i,k), or white undeveloped, implying that these were ineffective symbiotic nodules (Figs. 1b, 2b,d,f). To determine the nature of the mutations at the targeted region, the sequence analysis of the amplicons, generated from randomly selected elongated and undeveloped nodules, was carried out. The targeted amplicon sequences were determined using a next-generation sequencing (NGS) approach and the quantitative allele composition and the verification of genome editing mutations were analysed using the online tool CRISPResso232. The analysis revealed that all the white undeveloped nodules carried few base pair biallelic insertions or deletions at a high ratio and the very few reads of wild-type alleles identified in these nodules were probably derived from sporadic wild-type cells or the trace amount of wild-type contamination (Figs. 1b, 2a–f). The morphology and bacterial occupancy of these SYTO13-stained undeveloped nodules were very similar to the nodulation phenotype of the deletion mutant dnf7-2 showing no fluorescence in the nodule region proximal to the root (Fig. 1e). In addition, these plants, compared with empty-vector-transformed plants, showed the symptoms of nitrogen starvation (yellowish leaves and stunted growth habit) indicating the defect of symbiotic nitrogen fixation of these nodules (Fig. 1b). Contrary to these, the sequence analysis of the randomly selected elongated pink nodules showed that they either carried almost exclusively wild-type alleles (Fig. 2g,h) or they were heterozygous for example for a 4 bp deletion and the wild-type alleles (Fig. 2i,j). We also found a wild-type nodule that predominantly carried a homozygous deletion of 3 base pairs resulting in a deletion of the first single Glu residue of the mature peptide of NCR169 which mutation did not abolish the symbiotic function of NCR169 (Fig. 2k,l).
We also applied the developed CRISPR/Cas9 toolkit to target NCR-new35, a recently identified NCR gene that is crucial for the persistence of bacteroids in M. truncatula cv. Jemalong nodules13. The empty vector and the pKSE466-RR-NCRnew35 constructs were introduced into the roots of M. truncatula 2HA and M. truncatula ecotype R108 plants using A. rhizogenes-mediated hairy root transformation. Nodules on empty vector-transformed roots displayed the characteristic zonation of indeterminate nodules showing cells occupied by rhizobia (Fig. S2e,m). In contrast, undeveloped nodules were observed on roots of M. truncatula 2HA and R108 plants transformed with the editing construct of NCR-new35 (Figs. S2, S3g–r, 4e–h). The sequence analysis showed that undeveloped nodules, randomly collected from transformed roots of M. truncatula 2HA and R108 plants, carried biallelic or monoallelic homozygous mutations in the amplicons containing the target sequence in MtNCR-new35 (Fig. 4q,r). SYTO13-staining revealed that these nodules did not show the typical zonation and only their apical part fluoresced indicating that the region corresponding to the prospective nitrogen fixation zone was devoid of rhizobia (Fig. S2f–h, n–p). The morphology and bacterial occupancy of genome edited nodules were in agreement with the nodulation phenotype of Mtsym19 and Mtsym20 nodules defective in NCR-new3513. These results also revealed that NCR-new35 is crucial for the symbiotic interactions not only between M. truncatula 2HA and S. medicae WSM419 but between M. truncatula R108 and S. meliloti FSM-MA as well.
Selecting of NCR genes to be targeted by CRISPR/Cas9 editing
The M. truncatula genome contains a large gene family coding for more than 700 NCR peptides but only a few of them, NCR169, NCR211, NCR247, NCR343 and NCR-new35, have been proven to be crucial for symbiosis10,11,12,13,14,15. Apart from the fact that these peptides contained four cysteine residues in conserved positions, no other common features could be identified10,11,13. Therefore, it was not straightforward to ascertain which additional NCR peptides are essential and should be targeted. Despite all that, we have set up a pipeline to select NCR genes to be targeted (Fig. 3). We have analysed several features of more than 700 predicted NCR genes identified in the M. truncatula genome9 and selected NCR peptides based on whether they had predicted correct signal peptides and contained four cysteine residues in conserved positions (Fig. 3a). We have further analysed the expression profile of the diminished list of 248 NCR genes in the database obtained by laser-capture microdissection of M. truncatula nodules33. We identified 11 NCRs with no reads, which are certainly pseudogenes and found 65 NCRs which are expressed at low level (< 10,000 reads) in nodules compared with other NCRs. The recently identified NCR-new35 shows the lowest expression level among the crucial NCRs13 and thus, we have selected genes from the 172 NCRs showing similar or higher expression level as NCR-new35 (Fig. 3c,d). These NCR genes were analysed to predict whether they contained sequences which were amenable to CRISPR-based editing and the M. truncatula genome was analysed for off-target sites using the online prediction tool CRISPOR34. The isoelectric points (pI) of crucial NCR peptides vary between 4.78 and 10.15 implying that anionic, neutral and cationic NCR peptides could be essential for nitrogen-fixing symbiosis in M. truncatula. For this reason, the gene of NCR peptides with wide range of charges were selected to be edited (Fig. 3b). (v) Finally, we have selected four NCR genes, NCR068, NCR089, NCR128 and NCR161, to be targeted by CRISPR/Cas9 editing. The pI of the encoded mature peptides varied between 5.75 and 8.34 (Fig. 3b). NCR128 and NCR161 are induced in the late infection zone and showed expression in the interzone but NCR068 and NCR089 are preferentially expressed in the interzone and at lower level in the nitrogen fixation zone based on the LCM transcription data (Fig. 3c,33). The transcription activity of the four selected NCRs was lower than NCR169 and NCR211 but similar or higher than NCR-new35 (Fig. 3d).
Selection of NCR genes to be targeted by CRISPR/Cas9 mutagenesis. (a) 679 M. truncatula NCR genes were grouped based on the presence of a signal peptide in their encoded peptide sequence and containing four cysteine residues in conserved positions. (b) the 248 NCR peptides complying with all the three features (a) were classified based on the charge of the encoded mature peptides. NCR peptides crucial for symbiosis are highlighted in red, peptides targeted in this study by gene editing are in black and peptides found to be non-essential in the former study13 are in purple. (c) Relative spatial expression of four NCR genes essential for symbiosis and four NCRs mutagenized using CRISPR/Cas9 method in this study. Expression was determined by RNAseq of nodule zones obtained with laser-capture microdissection described in the study33. (d) The total read number of the NCR transcripts identified by RNAseq and analysed in panel (c). All the four NCRs selected for gene editing show higher transcriptional activity than NCR-new35 which shows the lowest expression activity among the crucial NCR genes. (e) The cleavage sites of Cas9 on the sequences of NCR genes targeted by CRISPR/Cas9 gene editing in this study. The target sites of each guideRNA were designed upstream of the nucleotides coding for the fourth conserved cysteines of each NCR peptides. ZIII, nitrogen fixation zone; IZ, interzone; IID, distal part of infection zone; IIP, proximal part of infection zone; pI, isoelectric point.
Inducing mutations in selected NCR genes using the CRISPR/Cas9 system
To induce mutations in genes NCR068, NCR089, NCR128 and NCR161 by CRISPR/Cas9-mediated cleavage, sgRNA constructs targeting a single site of each gene were designed and introduced along with the control empty vector and the constructs targeting NCR-new35 and NCR169 into M. truncatula 2HA roots using A. rhizogenes-mediated hairy root transformation. Transformed plants were inoculated with S. medicae WSM419 at 2–3 weeks after removal of non-transformed roots and the symbiotic phenotype of transformed plants were scored at 4–7 wpi (Fig. 4). The aerial part of plants transformed with empty vector or the constructs targeting NCR068 did not show the symptoms of nitrogen deficiency (Fig. 1a,c) and plants transformed with constructs targeting genes NCR089, NCR128 and NCR161 displayed similar growth habit (data not shown) indicating the effective symbiotic nitrogen fixation capacity of these plants. Nodules formed on roots transformed with sgRNA constructs of the four selected NCRs were elongated and pink suggesting that they were functional nodules (Fig. 4j,l,n,p). The staining of nodule sections with SYTO13 revealed that the bacterial colonization of nodules targeted for mutagenesis of genes NCR068, NCR089, NCR128 and NCR161 was identical to the wild-type indeterminate nodules developed on empty-vector transformed roots (Fig. 4i–p). Contrary, 2HA plants carrying mutations in NCR169 or NCR-new35 in each hairy root displayed retarded growth, showed the symptoms of nitrogen starvation characteristically (Fig. 1b) and slightly elongated white nodules were found on their roots (Figs. 4f, S2 and S3). In addition, the occupancy of nodules mutagenized for NCR169 and NCRnew-35 showed the phenotype characteristic for the previously described deletion mutants10,13 (Figs. 1b, 4e–h, S2, S3).
(a–p) Nodulation phenotype of M. truncatula 2HA (e,f,i–p) and R108 (g,h) plants mutagenized using the CRISPR/Cas9 method with A. rhizogenes-mediated hairy root transformation. Transformation with gene editing construct designed to target NCR-new35 (e–h) resulted in ineffective white and slightly elongated nodules while the homozygous or biallelic mutant alleles of NCR068 (i,j), NC089 (k,l), NCR128 (m,n) and NCR161 (o,p) were similar to the elongated and pink nodules formed on empty vector-transformed roots of M. truncatula 2HA (a,b) and R108 (c,d) plants. Images of nodule sections following SYTO13 staining and whole nodules were taken 4–5 weeks post inoculation with S. medicae WSM419 used for M. truncatula 2HA and S. meliloti FSM-MA for R108 plants. (q–v) Allele-specific results of CRISPR/Cas9 editing of the targeted NCR genes identified by next generation sequencing on Illumina platform. Black and brown rectangles show deleted and inserted nucleotides, respectively.
To verify the result of CRISPR/Cas9 genome editing, the targeted regions were amplified from nodule sections following microscopic analysis and sequenced using a MiSeq platform. The sequence analysis of the amplicons was carried out with the online tool CRISPResso2. The Cas9-induced mutations were predominantly indels, and the frequencies of nodules containing only indel alleles varied between 47 and 100% (Table 1). The genotyping also revealed that individual nodules were often chimeric containing more than two alleles or the ratio of the two alleles deviated from the expected 1:1 ratio indicating that the tissues of these nodules on hairy roots originated from multiple founder cells with different genetic backgrounds. The identified indels generated frameshift mutations in the targeted NCR genes presumably producing malfunctioning peptides. Nevertheless, all the nodules carrying biallelic mutations in the targeted NCRs were elongated and pink, indicating the presence of leghemoglobin, and were fully colonized by rhizobia indicating that peptides NCR068, NCR089, NCR128 and NCR161 are not required for the effective symbiotic nitrogen fixation between M. truncatula 2HA and S. medicae WSM419 (Figs. 4i–p and S4).
ncr068 plants, regenerated from gene edited hairy roots, form effective symbiosis with the most commonly used laboratory rhizobium strains
To analyse the symbiotic phenotype of genome editing mutants in more detail, we regenerated transgenic plants from hairy roots mutagenized for NCR genes. As a proof of concept, we cut root segments that had been transformed with the editing construct of NCR068 and stable transformants were regenerated using a slightly modified formerly published protocol35. DNA was purified from the leaves of hairy root-derived regenerated plants and the sequence analysis revealed that all the plants were homozygous containing a single adenine insertion in NCR068 (Fig. 5g). The one bp insertion generates a frame shift in the coding sequence of NCR068 and the mutant sequence encodes a chimera peptide containing 27 NCR068-specific and 23 non-NCR068-specific residues before termination. The fourth cysteine residue in the conserved position is lacking in the chimeric NCR068 peptide and based on the necessity of cysteines for the function of NCRs in planta10,13, we reckoned that the mutant NCR068 peptide does not function correctly.
(a) Stable ncr068 mutants, regenerated from segments of transgenic hairy roots with induced mutations in NCR068 using the CRISPR method, retained wild type symbiotic phenotype similar to wild-type (WT) M. truncatula 2HA plants 3 wpi with S. medicae WSM419. (b,c) The gene edited nodules developed on A. rhizogenes-transformed hairy-roots were elongated and pink with colonized cells in the nitrogen fixation zone. (d,e) Nodules developed on the roots of regenerated stable ncr068 mutants were similar to the nodules found on hairy-roots mediated by A. rhizogenes. (f) The selected hairy root (NCR068-5_S138) was chimeric and composed of cells with homozygous insertions of A or G in the targeted region of NCR068. (g) The regenerated stable ncr068 mutants (Ac2211) carried a homozygous A insertion causing a frameshift in the coding sequence of NCR068. (h) The alignment of WT and mutant NCR068 peptide sequences encoded by the mutated gene in the stable ncr068 mutant. Identical residues are highlighted by black background, conserved Cys residues are in red. SP, signal peptide; MP, mature peptide.
To confirm the symbiotic phenotype of regenerated ncr068 mutant, T1 seeds were synchronously germinated and planted into substrate with no nitrogen content. Three wpi with S. medicae WSM419, ncr068 stable transformants did not show symptoms of nitrogen starvation and developed like wild type M. truncatula 2HA plants (Fig. 5). Nodules, formed on the roots of stable transformant ncr068 mutant plants were elongated similarly what we found on the hairy roots of composite plants induced by the transformation construct targeting NCR068 (Fig. 5c,e). The staining of ncr068 nodule sections with SYTO13 revealed infected cells in interzone and nitrogen fixation zone fully occupied with elongated bacteria, similarly as it was found in wild-type nodules and NCR068-targeted hairy roots (Figs. 1d,f, 5b,d). The nodulation phenotype of the regenerated mutant ncr068 confirmed that the peptide NCR068 is not essential for the nitrogen-fixing symbiosis between M. truncatula and S. medicae WSM419.
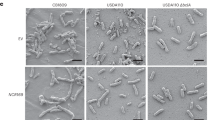
Different symbiotic efficiency has been reported for Sinorhizobium sp.36,37, therefore we assayed the strain-dependent nodulation phenotype of regenerated ncr068 mutant in response to S. meliloti strains 2011 and FSM-MA compared with S. medicae WSM419. The growth habit of wild-type M. truncatula 2HA and ncr068 mutant plants was similar following inoculation with any of the tested strains (Fig. 6). We observed a similar shoot dry weight of wild-type and ncr068 plants inoculated with S. meliloti FSM-MA or S. medicae WSM419 (Fig. S5). In agreement with published results38, the performance of wild-type and, in our study ncr068 plants as well, inoculated with S. medicae WSM419 superseded the plants inoculated with S. meliloti 2011 (Fig. S5). The dnf7-2 mutant showed the previously described ineffective symbiotic phenotype with all three tested rhizobia strains (Fig. 6). In line with the growth habit, the nodules developed on wt and ncr068 roots inoculated with the highly compatible strains S. medicae WSM419 and S. meliloti FSM-MA were cylindrical, pink and fully colonized by rhizobia but nodules elicited by the less effective strain S. meliloti 2011 were only slightly elongated (Fig. 6). In brief, no difference in plant development and nodule structure was noticed between wt and ncr068 plants with the tested rhizobia which suggests that the symbiotic phenotype of plants lacking NCR068 is not strain-dependent.
Stable ncr068 T1 mutant plants inoculated with three rhizobia strains show wild-type symbiotic phenotype. Wild-type M. truncatula 2HA (WT), the deletion mutant deficient in NCR169 (dnf7-2) and ncr068 mutant plants were inoculated with S. medicae WSM419, S. meliloti FSM-MA or S. meliloti 2011 and scored for symbiotic phenotype at 5 wpi. WT and ncr068 plants showed similar growth habit but presented less effective symbiotic interaction with S. meliloti 2011 compared with the two other strains. Mutant dnf7-2 showed nitrogen deficiency with all three tested rhizobia strains. SYTO13-staining revealed a similar colonization of WT and ncr068 nodules while only bacteria in infection threads and sporadic saprophytic bacteria fluoresced in the basal part of dnf7-2 nodules.
Discussion
NCR peptides, present solely in IRLC clade and Dalbergoid legumes, modulate the differentiation of nitrogen-fixing endosymbiont bacteria4,5,8. NCRs compose a large gene family with more than 700 members9 in M. truncatula and because of this large number of NCRs, their function was thought to be redundant. The identification of few individual NCRs crucial for nitrogen-fixing symbiosis disproved this concept10,11,12,13,14,15. These essential NCRs except for NCR247, which was analysed by reverse genetic approach, were identified fortuitously using deletion mutant collections of M. truncatula. In addition, the large deletions in mutants dnf4-1 and NF-FN9363 unveiled NCR peptides, NCR178, NCR341, NCR344 and NCR345, which are not crucial for the effective nitrogen-fixing symbiosis with the studied rhizobia strains11,13. Moreover, no specific chemical properties, charge or amino acid composition have been observed for the NCRs which would discriminate between crucial and non-crucial NCR peptides. Therefore, the targeted gene editing is the most promising tool to ascertain the need of a particular NCR peptide for nitrogen-fixing symbiosis. In this study, we used the CRISPR/Cas9 technology in the model legume M. truncatula to knock out four selected NCR genes encoding for peptides with four conserved cysteine residues representing the anionic, neutral and cationic members of the gene family.
The A. rhizogenes-mediated hairy root transformation produces chimeric root system composed of different ratios of transformed and non-transformed roots depending on the efficiency of transformation. To identify transformed roots in a convenient and non-destructive way and improve gene editing efficiency, a modified vector system carrying the DsRed transformation visible marker and single-site sgRNAs under the control of the M. truncatula U6 promoter was applied to generate mutations in the selected NCR genes in M. truncatula 2HA and R108 plants. Despite of its robustness, the A. rhizogenes-mediated hairy root transformation system has the disadvantage of occasionally emerging chimeric roots of a mixture of transformed and -non-transformed cells. In addition, the CRISPR/Cas9 system is not necessarily competent to edit the target sites in every cell or independent gene editing events occur in several transformed cells. Our results provided evidence of both cases; we found chimeric roots containing the mixture of wild-type and mutant alleles with different ratios, these roots mostly retained wild-type nodulation phenotype, and we detected roots having multi-allelic mutations at target sites. To eliminate these interfering effects and reveal the association between CRISPR/Cas9-induced mutations and the symbiotic phenotype without a doubt, we have sequenced the amplified fragment population of the targeted sites of SYTO13-stained and examined sections of individual nodules. This enabled the precise analysis of a large set of nodules and gene editing events could be coupled with the phenotype of each nodule.
We used two essential NCRs, NCR169 and NCR-new35, to prove the efficiency of the CRISPR/Cas9 gene editing tool in our A. rhizogenes-mediated hairy root transformation system and selected four NCRs (NCR068, NCR089, NCR128 and NCR161) to assay their requirement for symbiotic nitrogen fixation in M. truncatula. We generated mutant versions of the four previously non-investigated NCRs and the analysis of the colonization of the corresponding nodules indicated that these four NCRs are not essential for the symbiotic interaction between M. truncatula 2HA and S. medicae WSM419. The removal of variable number of non-transgenic roots from each plant and the potential presence of root cells with non-edited genome did not enable the reliable analysis of the nitrogen starvation phenotype (stunted growth, yellow leaves) of the shoots of transformed plants. The nodules of all the previously identified mutants defective in essential NCRs were white and slightly elongated and showed defects in colonization of the region corresponding to the nitrogen fixation zone10,11,13,14,15. Therefore, we assessed the symbiotic phenotype of genome edited nodules mostly based on their size, colour, and the colonization of the nitrogen fixation zone. The coordinated sequence and phenotype analyses which we carried out on every single nodule, clearly demonstrated that CRISPR/Cas9-induced mutations generating corrupted open reading frames in the coding sequence of NCR068, NCR089, NCR128 and NCR161 did not affect the nodulation phenotype of gene edited roots, thus these NCR peptides are not essential for the nitrogen-fixing symbiosis between M. truncatula 2HA and S. medicae WSM419. Although the four target NCR genes were selected based on the common features of the encoded essential peptides (four cysteine residues in conserved positions, predicted signal peptide, nodule-specific expression, unique sequence, various pI;10,11,13), these criteria were not adequate to identify essentials NCR peptides. This conclusion was supported by the genetic studies that identified non-essential peptides, NCR341, NCR344 and NCR345, possessing these features. The four NCR peptides targeted in this study extend the group of NCRs proved to be non-required for nitrogen fixation between M. truncatula and S. medicae WSM419 under the symbiotic conditions used in this study. Our findings show that peptides NCR068, NCR089, NCR128 and NCR161 are either not crucial or have minor or redundant roles in symbiosis. Alternatively, these peptides may be required under specific symbiotic or environmental conditions or essential for strain-specific symbiotic interaction with M. truncatula.
We have demonstrated that the A. rhizogenes-mediated hairy root transformation was successfully adapted to screen the requirement of NCR peptides for nitrogen fixation in M. truncatula. We have regenerated plants of gene edited knockout of NCR068 gene directly from transformed hairy root segments to carry out a more detailed analysis of CRISPR/Cas9-induced mutations in stable transformants. A previous proteomic study identified peptide NCR068 in bacteroids39 and we anticipated that NCR068 may have a crucial role in symbiosis. The mutant plants carrying a frame-shift mutation, induced by the insertion of a single base pair, in NCR068 in their germline showed similar nodulation phenotype to nodules found on A. rhizogenes-mediated mutant hairy roots proving the efficiency and usability of CRISPR/Cas9-mediated mutagenesis using hairy root transformation. The stable ncr068 mutant enabled us to assay the symbiotic phenotype with S. medicae strain WSM419 and two S. meliloti strains, 2011, a laboratory reference strain and FSM-MA, which is highly effective symbiotic partner of M. truncatula A17 and R10837. All the three tested rhizobia strains induced the formation of wild-type nodules on the root of ncr068, indicating that NCR068 is not crucial for the interaction with these strains. However, we cannot exclude that NCR068 is required for the symbiosis with other rhizobia strains or essential under specific environmental conditions.
Our study demonstrates that an optimized gene editing CRISPR/Cas9 vector construct and the developed workflow (Fig. S6) to analyse nodules developed on hairy roots mediated by A. rhizogenes- transformation is an effective system to analyse the symbiotic function of NCR genes. Although the selected NCR peptides were found to be non-essential for nitrogen-fixing symbiosis between M. truncatula and the studied rhizobia strains, the application of the technology can identify additional indispensable and crucial NCR peptides for nitrogen-fixing symbiosis.
Materials and methods
Plant material, growth conditions, bacterial strains and nodulation assay
Medicago truncatula cv. Jemalong 2HA line40 (2HA) and the M. truncatula R108 accession (R108) used for transformation experiments and M. truncatula dnf7-2 used as a symbiotic control mutant were described and used previously10,41. All these M. truncatula lines are available for and widely used by the M. truncatula community and we have a permission to collect seeds. All the methods were performed in accordance with the relevant institutional, national, and international guidelines and legislation.
For sterilization and scarification, seeds were soaked in concentrated sulfuric acid for 8 min, then rinsed with distilled water. Sterilized seeds were vernalized for 3 days on inverted 0.8% (w/v) water agar plates at 4 °C and germinated overnight at room temperature before transformation. Seedlings were grown in zeolite substrate (Geoproduct Kft., Mád, Hungary) for five days before inoculation with rhizobia. Plants were grown in SANYO HM350 environmental test chambers at 24 °C at 16/8 h light/dark periods.
M. truncatula 2HA plants were inoculated with Sinorhizobium (Ensiferi) medicae strain WSM419 (SmWSM419) carrying pXLGD4 plasmid42, constitutively expressing the lacZ reporter gene under the control of the hemA promoter. To test strain specific symbiotic phenotype of ncr068 mutants, T1 plants were inoculated with S. meliloti 2011, S. meliloti FSM-MA and SmWSM419 (pXLGD4) strains. M. truncatula R108 plants were inoculated with S. meliloti FSM-MA. The rhizobium strains were grown overnight in liquid TA medium. The bacteria were pelleted, resuspended and diluted to OD600 0.1 in nitrogen free liquid Fahraeus medium43 and used for inoculation.
Construction of the CRISPR/Cas9 binary vector used for gene editing
For CRISPR/Cas9-based gene editing experiments, a modified version of the pKSE401 vector44 was generated. DsRed fluorescent protein under the control of the 830 bp promoter of UBQ10 gene from Arabidopsis thaliana was inserted into the EcoRI digested vector to allow the identification of transformed roots based on their fluorescence. This generated construct (pKSE401_RR) was further modified by replacing the AtU6 promoter with MtU6.6 promoter31,45 resulting in the construct pKSE466_RR. The MtU6.6 fragment was amplified from the genomic DNA of M. truncatula A17 using the primers MtU6.6_prom_InF_F1 and MtU6.6_prom_InF_R along with the guide cassette fragment amplified from pKSE401_RR using the primers pKSE401_empty_InF_F2 and pKSE401_InF_R2 (Table S1) and the amplified fragments were assembled into the HindIII digested pKSE401_RR vector backbone using In-Fusion cloning (Takara Bio, Kusatsu, Japan).
Selection of targeted genes and generating CRISPR/Cas9 constructs
To select the M. truncatula NCR genes to be targeted, we have analysed the previously collected and described NCR peptide sequences9. During the selection, first the signal peptide prediction was repeated with the SignalP 5.0 tool46. If the prediction did not detect signal peptide, the whole peptide sequence was considered as mature peptide. Further on, NCR peptides possessing four cysteine residues in conserved position (Xn–C–X5–C–Xn–C–X4–C–Xn)8 were selected. The transcriptional activity of this set of NCR genes was analysed in the RNAseq data of M. truncatula nodules33 and genes with more than 10,000 reads were retained. The final set of 172 NCR genes was classified based on their isoelectric point listed in the collection of NCR peptides9. Our preference was to select NCR genes to be gene-edited encoding for cationic, neutral and anionic peptides. After that, we have selected NCRs preferentially expressed in the infection and interzones. According to these preferences, we heuristically selected target NCR genes that met the following criteria that an optimal sgRNA could be designed either in the upstream region of the gene close to the sequence coding for the first residues of the mature peptide, or at least upstream from the last conserved cysteine residue using the online tool available at http://crispor.tefor.net/34 using the M. truncatula A17 (MtrunA17r5.0) genome as reference47. This allowed us to disrupt the conserved cysteine residues which are essential for the function of NCR peptides in planta 10,13. Targets with GN19 NGG motif were preferred for optimal expression controlled by the MtU6.6 promoter. To minimize the risk of off-target mutations all selected sgRNA had at least two mismatches compared with potential off-target sites (Table S2), which were identified by searching the M. truncatula genome using an online prediction tool CRISPOR34. Among the potential targets that met all our criteria, we have particularly favoured NCR068, because it was previously detected in bacteroids isolated from M. truncatula nodules39. Plasmid constructs were assembled following the protocol described by Xing et al.44.
Agrobacterium rhizogenes-mediated hairy root transformation
CRISPR constructs were introduced into the ARqua1 strain of Agrobacterium rhizogenes and used for hairy root transformation of M. truncatula 2HA and R108 seedlings as described by Boisson-Dernier et al.28. Each CRISPR construct was transformed into the roots of at least 35 plants and experiments were repeated at least twice. Following transformation, the seedlings were grown for one week in square petri dishes containing antibiotic-free Fahraeus medium and then seedlings were transferred to selective Fahraeus medium supplemented with 25 mg/l kanamycin. To prevent root growth into the agar, the roots were carefully positioned between two layers of Whatman filter paper. Following the plant growth for additional two weeks, non-transgenic roots, identified based on the absence of red fluorescence, were excised. After that, plants were transferred into zeolite supplemented with Fahraeus medium containing 2 mM NH4NO3. Plants were further grown in plastic boxes to preserve rhizobium-free conditions and inoculated with rhizobia after 2–3 weeks. Prior to inoculation with rhizobia, newly emerged non-transgenic roots were excised again.
Embryonic induction and plant regeneration from hairy-root cultures
Plantlets were regenerated from transformed hairy roots grown on plate under sterile conditions using a slightly modified protocol described previously35. Three-week-old transgenic hairy roots were identified by DsRed fluorescence using a Leica MZ10-F fluorescence stereo microscope (Leica Microsystems GmbH, Wetzlar, Germany) and cut into approximately 0.5–1 cm segments and placed on modified MTR-2 medium (Murashige and Skoog medium including vitamins supplemented with 5.0 mg/l 2,4-Dichlorophenoxyacetic acid, 0.5 mg/l 6-Benzylaminopurine (BAP), 3% (w/v) sucrose, 50 mg/l kanamycin, 250 mg/l cefotaxime solidified with 0.8% (w/v) agar) to induce primary calli. After 2–3 weeks of incubation in dark at room temperature, calli were transferred to the modified MTR-3 medium supplemented with BAP (Murashige and Skoog medium including vitamins supplemented with 0.5 mg/l BAP, 2% (w/v) sucrose, 50 mg/l kanamycin, 250 mg/l cefotaxime solidified with 0.8% (w/v) agar) to induce embryonic calli and plates were kept in plant growing chambers with a photopheriod of 16 h of light and 8 h darkness at 24 °C. The calli were transferred to fresh medium in 1–2-week intervals. Shoots developed on MTR-3 + BAP medium were put on MTR-3 medium (without selective agent) in glass tubes and transferred to fresh medium on every 2–5 weeks to promote development and induce roots. Well-grown shoots were propagated by cutting the apical 2–3 cm of the shoots and they were transferred into separate tubes. After rooting, the T0 regenerated plants were transferred into jars containing zeolite, and watered with Fahraeus medium supplemented with 2 mM NH4NO3. The jars were kept in growth chambers under the same condition as described above and humidity in the jars was gradually decreased. Once new healthy leaves emerged, the plantlets were inoculated and tested for nodulation phenotype or alternatively, they were transferred to nutrient-rich soil in the greenhouse for seed collection.
Phenotypic characterization of transformed plants and microscopy analysis
To observe intact roots and nodules and detect fluorescence of transformed roots, a Leica MZ10F fluorescent dissecting microscope equipped with ET DsRED, and GFP LP filters was used (Leica Microsystems). Transgenic nodules were harvested, promptly immersed into ice-cold 1xPhosphate Buffered Saline (PBS, pH 7.4) and fixed with 4% paraformaldehyde. Fixed nodules were embedded in 5% (w/v) agarose gel prepared in 1xPBS and 100 µm tick longitudinal sections were prepared with Leica VT 1200S vibratome. Nodule sections were stained in 1xPBS solution containing 5 µM SYTO13 dye (Thermo Fisher Scientific, Waltham, MA, USA) for 30 min at room temperature. Images of stained nodule sections were captured using a Leica SP5 AOBS confocal laser scanning microscope (Leica Microsystems).
DNA isolation from sections of transgenic nodules
To purify DNA from fixed nodule sections, 2–8 sections per nodule sample were collected after SYTO13 staining and microscopic analysis, and transferred into nodule lysis buffer [100 mM Tris–HCl (pH 8), 0,5 M EDTA (pH 8), 0.2% SDS, 5 mM NaCl, Proteinase K 0.02 µg/µl]. The samples were homogenized using glass pestles and incubated at 56 °C overnight. Proteinase K was inactivated by incubating the samples at 95 °C for 15 min. Samples were spun down by microcentrifuge and cooled to room temperature. Subsequently 2 µL RNase A (from 10 mg/ml stock) was added and incubated at room temperature for 2 min. After that, 200 µL binding buffer [2.55 M Guanidium-HCl, 10% Triton X-100, 37% ethanol] was added and briefly vortexed, and 200 µL of 96% ethanol was added to the mix and vortexed immediately. The mixture was centrifuged at 8000 rpm for 30 s to pellet the tissue debris. The supernatants were then transferred to QIAprep Spin Miniprep columns (Qiagen, Hilden, Germany). Following centrifugation at 8000 rpm for 1 min, the flow-through was discarded and columns were washed two times with 500 µL washing solution (10 mM Tris–HCl pH 7.5, 80% ethanol). After the second washing step flow-through was discarded and columns were centrifuged at 13,000 rpm for 3 min. DNA elution was performed by incubating the columns with 35–50 µL of de-ionized water at 50 °C for 3 min and eluted DNA was collected by centrifugation at 13,000 rpm for 1 min.
Sequence analysis of targeted genes in gene-edited nodules
The analysis of the result of target site mutagenesis of transgenic roots targeted by the sgRNAs NCR169a, NCR169b and NCR169c was performed by Sanger sequencing of PCR fragments amplified from the target region using the ncr169_F1 and ncr169_R17 primer pair. To analyse the edits of targeted regions of genes NCR169, NCR-new35, NCR068, NCR128, NCR089 and NCR161 in nodule samples, the PCR amplified fragments were analysed by next-generation sequencing (NGS). The genomic regions of appr. 200 bp, including the target sites, were amplified using GoTaq™ G2 polymerase (Promega corp. Madison WI, USA) according to the manufacturer's instructions and gene specific primer pairs containing 5’ adaptor sequences (Table S1). The samples were sequenced on Illumina platform, generating 10,000 reads per sample. NGS sequence analysis was carried out by using CRISPresso2 program32 using default parameters.
Data availability
All data supporting the findings of this study are available within the paper and the corresponding supplementary materials published online.
References
Jones, K. M., Kobayashi, H., Davies, B. W., Taga, M. E. & Walker, G. C. How rhizobial symbionts invade plants: The Sinorhizobium–Medicago model. Nat. Rev. Microbiol. 5, 619–633 (2007).
Vasse, J., De Billy, F., Camut, S. & Truchet, G. Correlation between ultrastructural differentiation of bacteriods and nitrogen fixation in alfalfa nodules. J. Bacteriol. 172, 4295–4306 (1990).
Mergaert, P. et al. Eukaryotic control on bacterial cell cycle and differentiation in the Rhizobium–legume symbiosis. Proc. Natl. Acad. Sci. 103, 5230–5235 (2006).
Van de Velde, W. et al. Plant peptides govern terminal differentiation of bacteria in symbiosis. Science 327, 1122–1126 (2010).
Czernic, P. et al. Convergent evolution of endosymbiont differentiation in dalbergioid and inverted repeat-lacking clade legumes mediated by nodule-specific cysteine-rich peptides. Plant Physiol. 169, 1254–1265 (2015).
Wang, D. et al. A nodule-specific protein secretory pathway required for nitrogen-fixing symbiosis. Science 327, 1126–1129 (2010).
Yang, J. et al. A signal peptide peptidase is required for ER-symbiosome proximal association and protein secretion. Nat. Commun. 14, 4355 (2023).
Mergaert, P. et al. A novel family in Medicago truncatula consisting of more than 300 nodule-specific genes coding for small, secreted polypeptides with conserved cysteine motifs. Plant Physiol. 132, 161–173 (2003).
Montiel, J. et al. Morphotype of bacteroids in different legumes correlates with the number and type of symbiotic NCR peptides. Proc. Natl. Acad. Sci. U. S. A. 114, 5041–5046 (2017).
Horvath, B. et al. Loss of the nodule-specific cysteine rich peptide, NCR169, abolishes symbiotic nitrogen fixation in the Medicago truncatula dnf7 mutant. Proc. Natl. Acad. Sci. U. S. A. 112, 15232–15237 (2015).
Kim, M. et al. An antimicrobial peptide essential for bacterial survival in the nitrogen-fixing symbiosis. Proc. Natl. Acad. Sci. U. S. A. 112, 15238–15243 (2015).
Sankari, S. et al. A haem-sequestering plant peptide promotes iron uptake in symbiotic bacteria. Nat. Microbiol. 7, 1453–1465 (2022).
Horváth, B. et al. The Medicago truncatula nodule-specific cysteine-rich peptides, NCR343 and NCR-new35 are required for the maintenance of rhizobia in nitrogen-fixing nodules. New Phytol. 239, 1974–1988 (2023).
Zhang, R. et al. Nodule-specific cysteine-rich peptide 343 is required for symbiotic nitrogen fixation in Medicago truncatula. Plant Physiol. 193(3), 1897–1912. https://doi.org/10.1093/plphys/kiad454 (2023).
Gao, F. et al. NCR343 is required to maintain the viability of differentiated bacteroids in nodule cells in Medicago truncatula. New Phytol. 240(2), 815–829. https://doi.org/10.1111/nph.19180 (2023).
Zhang, Y., Malzahn, A. A., Sretenovic, S. & Qi, Y. The emerging and uncultivated potential of CRISPR technology in plant science. Nat. Plants 5, 778–794 (2019).
Le, H. et al. CRISPR/Cas9-mediated knockout of Galactinol synthase-encoding genes reduces Raffinose family oligosaccharide levels in soybean seeds. Front. Plant Sci. 11, 2033 (2020).
Shu, H., Luo, Z., Peng, Z. & Wang, J. The application of CRISPR/Cas9 in hairy roots to explore the functions of AhNFR1 and AhNFR5 genes during peanut nodulation. BMC Plant Biol. 20, 417 (2020).
Li, G. et al. Development of an Agrobacterium-mediated CRISPR/Cas9 system in pea (Pisum sativum L.). Crop J. 11, 132–139 (2023).
Wang, L. et al. CRISPR/Cas9 knockout of leghemoglobin genes in Lotus japonicus uncovers their synergistic roles in symbiotic nitrogen fixation. New Phytol. 224, 818–832 (2019).
Meng, Y. et al. Targeted mutagenesis by CRISPR/Cas9 system in the model legume Medicago truncatula. Plant Cell Rep. 36, 371–374 (2017).
Zhang, H. et al. Efficient generation of CRISPR/Cas9-mediated homozygous/biallelic Medicago truncatula mutants using a hairy root system. Front. Plant Sci. 11, 1–11 (2020).
Confalonieri, M. et al. CRISPR/Cas9-mediated targeted mutagenesis of CYP93E2 modulates the triterpene saponin biosynthesis in Medicago truncatula. Front. Plant Sci. 12, 1–16 (2021).
Jain, D., Jones, L. & Roy, S. Gene editing to improve legume-rhizobia symbiosis in a changing climate. Curr. Opin. Plant Biol. 71, 102324 (2023).
Limpens, E. et al. RNA interference in Agrobacterium rhizogenes-transformed roots of Arabidopsis and Medicago truncatula. J. Exp. Bot. 55, 983–992 (2004).
Wang, Q. et al. Host-secreted antimicrobial peptide enforces symbiotic selectivity in Medicago truncatula. Proc. Natl. Acad. Sci. U. S. A. 114, 6854–6859 (2017).
Yang, S. et al. Microsymbiont discrimination mediated by a host-secreted peptide in Medicago truncatula. Proc. Natl. Acad. Sci. U. S. A. 114, 6848–6853 (2017).
Boisson-Dernier, A. et al. Agrobacterium rhizogenes-transformed roots of Medicago truncatula for the study of nitrogen-fixing and endomycorrhizal symbiotic associations. Mol. Plant-Microbe Interact. 14, 695–700 (2001).
Wang, D. et al. Optimized CRISPR guide RNA design for two high-fidelity Cas9 variants by deep learning. Nat. Commun. 10, 1–14 (2019).
Brinkman, E. K., Chen, T., Amendola, M. & Van Steensel, B. Easy quantitative assessment of genome editing by sequence trace decomposition. Nucleic Acids Res. 42, 1–8 (2014).
Jacobs, T. B., LaFayette, P. R., Schmitz, R. J. & Parrott, W. A. Targeted genome modifications in soybean with CRISPR/Cas9. BMC Biotechnol. 15, 1–10 (2015).
Clement, K. et al. CRISPResso2 provides accurate and rapid genome editing sequence analysis. Nat. Biotechnol. 37, 224–226 (2019).
Roux, B. et al. An integrated analysis of plant and bacterial gene expression in symbiotic root nodules using laser-capture microdissection coupled to RNA sequencing. Plant J. 77, 817–837 (2014).
Concordet, J. P. & Haeussler, M. CRISPOR: Intuitive guide selection for CRISPR/Cas9 genome editing experiments and screens. Nucleic Acids Res. 46, W242–W245 (2018).
Crane, C., Wright, E., Dixon, R. A. & Wang, Z. Y. Transgenic Medicago truncatula plants obtained from Agrobacterium tumefaciens-transformed roots and Agrobacterium rhizogenes-transformed hairy roots. Planta 223, 1344–1354 (2006).
Terpolilli, J. J., O’Hara, G. W., Tiwari, R. P., Dilworth, M. J. & Howieson, J. G. The model legume Medicago truncatula A17 is poorly matched for N2 fixation with the sequenced microsymbiont Sinorhizobium meliloti 1021. New Phytol. 179, 62–66 (2008).
Kazmierczak, T. et al. Specific host-responsive associations between Medicago truncatula accessions and Sinorhizobium strains. Mol. Plant-Microbe Interact. 30, 399–409 (2017).
Larrainzar, E., Gil-Quintana, E., Seminario, A., Arrese-Igor, C. & González, E. M. Nodule carbohydrate catabolism is enhanced in the Medicago truncatula A17-Sinorhizobium medicae WSM419 symbiosis. Front. Microbiol. 5, 447 (2014).
Durgo, H. et al. Identification of nodule-specific cysteine-rich plant peptides in endosymbiotic bacteria. Proteomics 15, 2291–2295 (2015).
Rose, R. J., Nolan, K. E. & Bicego, L. The development of the highly regenerable seed line Jemalong 2HA for transformation of Medicago truncatula—Implications for regenerability via somatic embryogenesis. J. Plant Physiol. 155, 788–791 (1999).
Domonkos, Á. et al. NAD1 controls defense-like responses in Medicago truncatula symbiotic nitrogen fixing nodules following rhizobial colonization in a BacA-independent manner. Genes 8, 387 (2017).
Leong, S. A., Williams, P. H. & Ditta, G. S. Analysis of the 5’ regulatory region of the gene for δ-aminolevulink add synthetase of Rhizobium meliloti. Nucleic Acids Res. 13, 5965–5976 (1985).
Fahraeus, G. The infection of clover root hairs by nodule bacteria studied by a simple glass slide technique. J. Gen. Microbiol. 16, 374–381 (1957).
Xing, H. L. et al. A CRISPR/Cas9 toolkit for multiplex genome editing in plants. BMC Plant Biol. 14, 1–12 (2014).
Kim, G. B. & Nam, Y. W. Isolation and characterization of Medicago truncatula U6 promoters for the construction of small hairpin RNA-mediated gene silencing vectors. Plant Mol. Biol. Report. 31, 581–593 (2013).
Almagro Armenteros, J. J. et al. SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat. Biotechnol. 37, 420–423 (2019).
Pecrix, Y. et al. Whole-genome landscape of Medicago truncatula symbiotic genes. Nat. Plants 4, 1017–1025 (2018).
Acknowledgements
This work was supported by the Hungarian National Research Fund/National Research, Development and Innovation Office grants OTKA 119652, 129547, PD-132495 and 132646 as well as by the Collaborative Research Programme ICGEB Research Grant HUN17-03. We thank H. Cs. Tolnainé for her skillful technical assistance and Z. Tóth for support in confocal microscopy imaging.
Author information
Authors and Affiliations
Contributions
J.B.B. and P.K conceived the project. B.G., J.B.B., D.Á. and B.H. performed the experiments and analysed the data J.B.B. and P.K. wrote the manuscript that all authors reviewed.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Güngör, B., Biró, J.B., Domonkos, Á. et al. Targeted mutagenesis of Medicago truncatula Nodule-specific Cysteine-Rich (NCR) genes using the Agrobacterium rhizogenes-mediated CRISPR/Cas9 system. Sci Rep 13, 20676 (2023). https://doi.org/10.1038/s41598-023-47608-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-023-47608-5