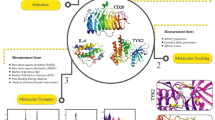
Abstract
Toad clothing (toad slough) detoxifies, reduces oedema and analgesia, is anti-inflammatory and anti-tumour, and regulates the immune system. Toad clothing’s mechanism is unclear. The molecular mechanism of toad clothing in rheumatoid arthritis (RA) therapy will be investigated in this research. We used Ultra-Performance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (UPLC-Q-TOF/MS), network pharmacology, molecular docking, and in vitro experiments to investigate the mechanism behind the anti-RA action of toad apparel. First, UPLC-Q-TOF/MS revealed 24 toad clothing ingredients. Network pharmacology predicted five key elements, six core targets, and the Phosphatidylinositol 3-Kinase/ Protein Kinase B (PI3K/AKT) signalling pathway. Then, molecular docking was used to validate the binding ability of core chemical constituents and core targets of toad clothing. The results showed that the primary constituent, resibufogenin, was bound tightly to six core targets and had a good affinity for key RA targets such as Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA), Epidermal Growth Factor Receptor (EGFR), and Janus Kinase 2 (JAK2). Cellular tests demonstrated that resibufogenin dramatically lowered Tumour Necrosis Factor-alpha (TNF-α), Interleukin-6 (IL-6), and Interleukin-1 beta (IL-1β) levels and regulated the PI3K/AKT signalling pathway. This research showed toad clothing’s anti-RA pharmacodynamic material basis and mechanism, offering a theoretical foundation for its creation, use, and clinical application.
Similar content being viewed by others
Introduction
Rheumatoid Arthritis (RA) is a chronic, systemic autoimmune disease that primarily affects joints, characterized by symmetrical polyarthritis and often accompanied by the involvement of extra-articular organs1,2. This disease is marked by persistent joint inflammation, cartilage destruction, and bone destruction, which can ultimately lead to joint deformity and loss of function3,4. Epidemiological surveys have shown that the global prevalence of RA is approximately 0.5–1.0%. RA can affect individuals of any age, with a peak incidence occurring between 30 and 50 years and a female-to-male incidence ratio of 2:1 to 3:15,6. RA treatment includes drug therapy, nondrug therapy, and surgery. In terms of drug therapy, Western medicine commonly employs nonsteroidal anti-inflammatory drugs, corticosteroids, and disease-modifying antirheumatic drugs. At the same time, these treatments are effective. Still, their long-term use can cause various adverse reactions7,8,9,10, such as gastrointestinal ulcers, osteoporosis, and hormone dependency, causing significant suffering for patients. Therefore, finding a drug with considerable efficacy and minimal adverse reactions in RA treatment is urgent.
Traditional Chinese and ethnic medicines, which are characterized by multitarget effects and minimal adverse reactions, offer unique advantages in the treatment of RA and play crucial roles in its therapy and management11,12. Toad clothing is a raw animal material commonly used in Chinese medicine clinics, and its medicinal use was first recorded in China’s first pharmacological monograph,"Shen Nong Ben Cao Jing”13 with the use of the toad clothing from Bufo gargarizans. During Bufo gargarizans’s growth and development, these toads naturally shed their keratinized clothing periodically, which is known as toad clothing. Toads undergo multiple molting cycles throughout their lifespan, each time replacing their old clothing with a new layer. The formation of toad clothing is closely related to the toad’s growth, development, and environmental factors. The frequency of molting is influenced by various factors, such as environmental humidity, temperature, and food availability. Under favorable conditions, toads can molt naturally every 1 to 3 months, and it is common for them to shed 1 to 3 layers of clothing per year. The molting process typically occurs at night, and the shed clothing is often consumed by the toads themselves, which is one of the reasons why toad clothing is rare. However, through artificial intervention and advanced collection techniques, the yield and quality of toad clothing can be significantly improved14. Toad clothing, also known as toad slough, is the horny skin shed naturally by toads during their growth and development, and it has various pharmacological effects, including detoxification, analgesia, anti-inflammatory, antitumor, immune regulation, and wound healing promotion15,16,17. Research has shown that toad clothing extracts have strong biological functions, especially anti-inflammatory and antitumor effects anti-rheumatoid arthritis18. However, the pharmacological basis and mechanism of action of toad clothing in treating RA remains unclear. Traditional Chinese medicine research often relies on empirical medicine and individualized treatment, lacking in-depth exploration of pharmacological substances and detailed elaboration of mechanisms of action. This limits the modernization of traditional Chinese medicine and prevents many potentially effective drugs from gaining wider application19.
Network pharmacology, which is based on the interaction network of"multiple genes, multiple targets, and multiple pathways,"provides predictive value for the targets and potential mechanisms of action of traditional Chinese medicines in treating various diseases. To better explore the potential molecular mechanisms of toad clothing components in treating RA20, this study systematically investigated its intervention mechanisms in RA from multiple perspectives, including chemical composition, target actions, and signaling pathways. Firstly, the chemical components of toad clothing were identified, and their potential active components, targets, and action pathways in RA treatment were analyzed. Subsequently, the binding capacity of core chemical components to core targets was predicted. Finally, the anti-rheumatoid arthritis effects and the mechanism of its key components were validated using an Lipopolysaccharide(LPS) induced mouse macrophage inflammation model. The workflow of this study was illustrated in Fig. 1.
This study used a schematic diagram of network pharmacology and molecular docking, as well as experimental verification.
Results
Identification of the chemical constituents of toad clothing
UPLC-Q-TOF/MS was employed to perform an entirely positive and negative ion scan of the methanol extract of toad clothing according to the mass spectrometry conditions under 4.2.3. The BPI chromatogram (Base Peak Ion Chromatogram) was a commonly used graph in mass spectrometry analysis. It displayed the information of the most intense ion at each time point in the Total Ion Current (TIC) mode, highlighting the strongest ion. BPI plots were obtained in the positive and negative ion modes (Fig. 2). Peak View software was needed for mass spectrometry data analysis, particularly for identifying and interpreting ion peaks. Raw data was generated by the Waters mass spectrometer and processing by Peak View 1.2 software, and the databases (including the Waters commercial database and the self-built database) were compared to further analyze and identify the chemical constituents of toad clothing based on excimer ion peak, retention time and primary and secondary mass spectrometry fragment information. Finally, 24 chemical constituents were determined (Table 1), including 17 steroids and 7 fatty acids. All compounds identified in this study were characterized via mass spectrometry-based analytical methods. Due to space constraints, only compounds 1, 6, and 7 were selected for detailed elucidation to exemplify the structural characterization workflow.
Representative BPI diagram of toad clothing extract in different ionization modes. (A) Negative ion mode. (B) Positive ion mode. BPI, base peak ion.
As shown in Fig. 3A, taking constituent one as an example, an component was identified at a retention time of 14.44 min with a protonated ion peak showing an m/z value of 403.2476. The most likely molecular formula, C24H34O5, was validated based on the nitrogen rule accurate mass calculator and theoretical mass (m/z 403.2479), with a mass error of 0.5 ppm. Three fragments were produced from the parent ion with an m/z value of 403.2476, namely m/z 385.2360, m/z 349.2153 and m/z 253.1957, due to the neutral losses of OH, 2H2O and C5H4O2, respectively. Among these fragments, m/z 403.2476 lost OH to form product ion at m/z 385.2360; similarly, m/z 385.2360 lost 2H2O, also yielding product ion at m/z 349.2153. Finally, the fragment at m/z 349.2153 was further broken down into ions at m/z 253.1957 by eliminating C5H4O2.
Mass Spectrum Fragmentation Pattern. (A) Mass Spectrum Fragmentation Pattern of constituent 1. (B) Mass Spectrum Fragmentation Pattern of constituent 6. (C) Mass Spectrum Fragmentation Pattern of constituent 7.
As shown in Fig. 3B, taking constituent six as an example, an impurity was identified at a retention time of 17.97 min with a protonated ion peak showing an m/z value of 400.2250. The most likely molecular formula, C24H34O5, was validated based on the nitrogen rule, Accurate Mass Calculator, and theoretical mass (m/z 401.2323), with a mass error of 0.4 ppm. Four fragments were produced from the parent ion with an m/z value of 400.2250, namely m/z 383.2143, m/z 365.2111, m/z 347.2009, and m/z 269.1897, due to the neutral losses of OH,2H2O, and C6H6, respectively. Among these fragments, m/z 400.2250 lost OH to form product ion at m/z 383.2143; similarly, m/z 383.2143 lost H2O, also yielding product ion at m/z 365.2111, m/z 365.2111 lost H2O also yielding product ion at m/z 347.2009. Finally, the fragment at m/z 347.2009 further broke down into ions at m/z 269.1897 by eliminating C6H6.
As shown in Fig. 3C, taking constituent seven as an example, an impurity was identified at a retention time of 19.09 min with a protonated ion peak showing an m/z value of 403.2479. The most likely molecular formula, C24H34O5, was validated based on the nitrogen rule, Accurate Mass Calculator, and theoretical mass (m/z 403.2472), with a mass error of 1.7 ppm. Three fragments were produced from the parent ion with an m/z value of 403.2479, namely m/z 385.2346, m/z 367.2257, and m/z 349.2156, due to the neutral losses of OH and 2H2O, respectively. Among these fragments, m/z 403.2479 lost OH to form product ion at m/z 385.2346; similarly, m/z 385.2346 lost H2O, yielding product ion at m/z 367.2257. Finally, the fragment at m/z 367.2257 further broke down into ions at m/z 349.2156 by eliminating H2O.
Network pharmacology
Collection of potential targets
According to the results of mass spectrometry analysis, the active constituents of toad clothing were identified by literature reports. The information of 24 constituents was imported into the SwissTargetPrediction and PharmMapper database for target prediction, and 649 active constituent targets were obtained after redundancy elimination. Rheumatoid Arthritis targets were searched from the GeneCards database、DisGeNET database、DrugBank database Therapeutic Target Database (TTD)database, and OMIM database, and 2330 Rheumatoid Arthritis gene targets were screened according to filter conditions. The active constituent targets and disease targets were imported into Jvenn to make Venn diagrams and 288 intersecting targets were obtained as potential targets for the anti-rheumatoid arthritis action of toad clothing for further study (Fig. 4).
Venn diagram of the targets of toad clothing in Rheumatoid Arthritis.
PPI network analysis
To analyze the protein–protein interaction network, we interrogated the 288 target genes in the STRING database with the highest confidence set to 0.9 (Fig. 5A). The network included 288 protein nodes and 844 edges with an average node degree value of 5.86 and a clustering coefficient 0.47. Then, topology analysis was performed in Cytoscape 3.10.1 software using the plugin CytoNCA. Median values of betweenness centrality (BC), closeness centrality (CC), and degree centrality (DC) were used as limiting conditions for screening, and all genes with indicators more significant than the median were selected for subsequent analysis, and after two screenings, eight core targets were finally obtained, which were Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA), Signal Transducer and Activator of Transcription 3(STAT3), AKT Serine/Threonine Kinase 1 (AKT1),Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit beta (PIK3CB),Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit delta (PIK3CD), Epidermal Growth Factor Receptor (EGFR), Heat Shock Protein Family A Member 1 (HSP90AA1), and JAK2 (Fig. 5B, Table 2).
PPI network of potential target on Rheumatoid Arthritis. (A) Constructing a protein interaction network of Rheumatoid Arthritis target genes induced by toad clothing; (B) Screening of core targets of toad clothing in treating Rheumatoid Arthritis. PPI, Protein–Protein Interaction.
GO and KEGG pathway analysis
David database was applied to obtain the Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes(KEGG) enrichment of the above intersecting targets. The GO analysis for the anti-inflammatory action of toad clothing was obtained with screening criteria of P < 0.05. A total of 1023 entries were received, including 737 cellular Biological Processes (BP), 86 Cellular Components (CC) and 200 Molecular Functions (MF). The top 10 entries of biological processes, molecular functions and cellular components were visualized in Fig. 6A. GO analysis showed that the BP mainly involved signal transduction, chromatin remodelling, inflammatory response, and other processes. The CC mostly included plasma membrane, cytosol, cytoplasm, etc., and the MF mostly involved ATP binding, histone H2AXY142 kinase activity, histone H3Y41 kinase activity, etc. The enrichment results of the KEGG pathway were shown in Fig. 6B, and the number of targets and − lgP values of the enriched pathways were used to measure the degree of KEGG enrichment. Several pathways were implicated in inflammation occurrence and healing by the screened proteins. The three pathways with the lowest p-value were the PI3K-Akt signalling pathway, Ras-associated protein 1 signaling pathway (Rap1) signalling pathway and Ras Proto-Oncogene (Ras) signalling pathway. These three pathways were the primary mechanisms through which toad clothing exerted its anti-rheumatoid arthritis effects. Furthermore, we analyzed the PI3K-AKT signal pathway in the KEGG database. The font marked in red represented the key targets closely with Rheumatoid Arthritis in the treatment of toad clothing by the PI3K-AKT signal pathway (Fig. 6C).
GO and KEGG pathway analysis. (A) The top 10 significance of enriched GO terms analysis of therapy target genes of toad clothing on Rheumatoid Arthritis (P < 0.05). (B) Top 20 significance of enriched KEGG pathways and analysis of PI3K-AKT signal path in KEGG database (P < 0.05). (C) The font marked in red represents the target in the PI3K-AKT signal pathway closely related to treating Rheumatoid Arthritis with toad clothing. GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes; PI3K-AKT, Phosphatidylinositol 3-Kinase/Protein Kinase B.
Construction of constituent ‑target‑pathway -disease network diagram
A constituent-target-pathway-disease network diagram was constructed by combining the target of toad clothing in treating Rheumatoid Arthritis with the related pathways obtained by KEGG enrichment analysis with Cytoscape 3.10.1 software (Fig. 7). The network consisted of 333 nodes and 3489 edges. The highest EGFR degree value was 37. Therefore, EGFR was the key target of toad clothing against Rheumatoid Arthritis.
The constituent‑target‑pathway-disease network of toad clothing. Diamond shapes are used to represent signaling pathways, hexagons are used to represent bioactive constituents, blue circular nodes indicate target proteins, green triangle nodes indicate toad clothing drug, red ellipse nodes represent Rheumatoid Arthritis diseases, and the black edge stated that there was a connection between constituents, targets, drug, pathway and disease.
Molecular docking
The six core targets screened in the Protein–Protein Interaction (PPI) network were used as receptors, and the top 5 chemical constituents in the"drug-constituents-targets‑pathway-disease"network were used as ligands. AutoDock Vina was used for molecular docking. A smaller binding energy indicated a more substantial binding capacity; a binding energy < −5 kcal⋅mol − 1 indicated a better binding activity, and a binding energy < −7 kcal⋅mol − 1 indicated a vigorous binding activity. The results of molecular docking were shown in Fig. 8A. There were 14 groups with < −7 kcal⋅mol − 1 binding energies and three with < −9.5 kcal⋅mol − 1.These findings demonstrated that the majority of constituents exhibited favorable binding affinities with their respective targets, as evidenced by binding energies below the established threshold of −5 kcal⋅mol − 1. Compared with other target proteins, EGFR, JAK2, and PIK3CA had lower binding energies than other proteins, and resibufogenin and EGFR had the lowest binding energy (− 9.8 kcal⋅mol − 1), suggesting that these proteins might be potential anti-rheumatoid arthritis binding targets of toad clothing. Resibufogenin might be the key anti-rheumatoid arthritis constituent of toad clothing. PyMoLwas used to visualize the binding sites of constituents and targets with high free binding energy fraction, and it was observed that ligands and receptors could be hydrogen bonded (Fig. 8B). In particular, resibufogenin formed hydrogen bonds with amino acid residues MET-793, LEU-844, VAL-726 and TYR-869 in EGFR. It formed hydrogen bonds with the TRP-167, THR-163, TYR-159, and GLU-152 residues of PIK3CA and had a binding affinity.
Molecular docking heat map analysis and results of the lowest binding energy in each target with the selected bioactive constituent of toad clothing. (A) Heat map analysis of molecular docking of core constituents to core targets;(B) resibufogenin, EGFR, −9.8 kcal/mol; arenobufagin, EGFR, −9.8 kcal/mol; cinobufagin, JAK2, −9.7 kcal/mol; resibufogenin, PIK3CA, −8.3 kcal/mol. EGFR, epidermal growth factor receptor; JAK2, Janus Kinase 2; PIK3CA, hosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha.
Experimental verification in vitro
Cell viability assay
Molecular docking results showed that resibufogenin (Fig. 9A) was the main constituent in the anti-RA effect of toad clothing. RAW264.7 cells, a murine macrophage cell line, exhibit strong phagocytic activity and stable genetic background, making them an ideal cellular model for studying rheumatoid arthritis21.To further verify the anti-RA action of resibufogenin, the impact of different concentrations of resibufogenin on cell toxicity of RAW264.7 was evaluated using the CCK-8 method. The LPS-induced RAW264.7 cells were treated with varying doses of resibufogenin, and the expression of NO, TNF-α, IL- 6 and IL-1β inflammatory mediators and cytokines in the cell supernatant was also determined to assess the anti-inflammatory effect of resibufogenin. An in vitro cytotoxicity assay was performed using the CCK-8 kit to determine the direct impact of resibufogenin on cell viability. As shown in Fig. 9B, various concentrations of the sample solution (2.5,5,10,20,40, 80,160 μM) were used to treat RAW264.7 cells. Cells in the blank group were not treated with sample solutions. As shown in Fig. 9C, after co-incubation with LPS (1 μg/ml) for 24 h, RAW264.7 cells were treated with various concentrations of the sample solution (2.5, 5, 10, 20, 40, 80, 160 μM). The cells in the blank group and the LPS group were not treated with the sample solution. The results showed no cytotoxic effect in the range of 2.5–80 μM.
Toxicity of Resibufogenin (RBG) on RAW264.7 cells. The chemical structure of RBG is drawn using ChemDraw software (A). RAW264.7 cells were treated with RBG (0.25–160 μM) in the absence (B)or presence (C) of LPS (1 μg/ml) for 24 h, then the cell viability was detected by CCK-8 assay. RAW264.7 cells were treated with RBG (10,20,40,80 μM) in the absence of LPS (1 μg/ml) for 24 h; then the supernatant was collected for the detection of NO with Griess reagent22(D). Data are mean ± SD of a minimum of three independent experiments. #P < 0.05, ##P < 0.01 versus Control; *P < 0.05 and **P < 0.01 versus LPS. RBG, resibufogenin; LPS, Lipopolysaccharide; CCK, Cell Counting Kit.
Anti-RA activity of resibufogenin in LPS-stimulated RAW264.7 cells
Figure 9B demonstrated that 10 μM resibufogenin significantly enhanced cell viability (130 ± 8%, p < 0.01). However, this pro-proliferative effect might confound anti-inflammatory assessments, as excessive cell proliferation could dilute inflammatory signals and obscure the drug’s true anti-inflammatory activity. Different doses of resibufogenin (80,40,20 μM) and LPS (1 μg/ml) were co-incubated with RAW264.7 cells. The concentration of NO in the cell supernatant was determined to observe the effect of resibufogenin on NO and to evaluate the anti-inflammatory activity of resibufogenin. According to Fig. 9D, RAW264.7 cells stimulated with LPS showed significantly higher NO levels in the supernatant than in the control group. Compared with the LPS group, NO concentration was significantly lower in the resibufogenin group at 80, 40 and 20 μM concentrations in a dose-dependent manner. All concentrations were statistically different from the LPS group. TNF-α, IL-6, and IL-1β are important pro-inflammatory cytokines in the inflammatory response. Studies have shown that reducing the expression of pro-inflammatory cytokines suppresses the inflammatory response and inhibits oxidative stress23. To further validate the anti-inflammatory activity of resibufogenin, we detected the protein expression level of the pro-inflammatory cytokines TNF-α, IL-6, and IL-1β by ELISA. According to Fig. 10A–C, compared to the normal group, TNF-α, IL-6, and IL-1β secretion levels in the LPS group significantly increased by 13.35, 18.02, and 2.03-fold, respectively. Relative to the LPS group, 20 μM drug treatment reduced TNF-α, IL-6, and IL-1β levels by 0.92, 0.56, and 0.67-fold; 40 μM treatment reduced them by 0.86, 0.28, and 0.54-fold; and 80 μM treatment reduced them by 0.26, 0.10, and 0.54-fold. These results demonstrated that resibufogenin significantly inhibited pro-inflammatory mediators and cytokine release in RAW 264.7 cells induced by LPS.
Effect of RBG on RAW264.7 cells. (A-C) Effects of RBG on TNF-α, IL-6 and IL-1β secretion in RAW264.7 cells stimulated by LPS. #P < 0.05, ##P < 0.01 versus Control; *P < 0.05 and **P < 0.01 versus LPS. RBG, resibufogenin; TNF-α, Tumour Necrosis Factor-alpha; IL-6, Interleukin-6; IL-1β, Interleukin-1 beta; LPS, Lipopolysaccharide.
Resibufogenin exerts anti-rheumatoid arthritis effects by inhibiting the expression of PIK3CA, EGFR, and JAK2 genes
Molecular docking results suggested that PIK3CA, EGFR, JAK2 were the primary targets of resibufogenin against RA. As shown in Fig. 11A-C, qRT-PCR results indicated that the mRNA levels of PIK3CA, EGFR, and JAK2 in the LPS group were increased by 14.11, 1.37, and 1.61-fold compared to the normal group. Compared to the LPS group, at a concentration of 20 μM, the mRNA levels of PIK3CA, EGFR, and JAK2 were reduced by 0.50, 0.32, and 0.70-fold, respectively; at a concentration of 40 μM, they were reduced by 0.45, 0.27, and 0.52-fold; and at a concentration of 80 μM, they were reduced by 0.37, 0.23, and 0.39-fold, respectively. These findings suggested that suppressing these gene expressions by resibufogenin might reduce inflammatory mediators, thereby helping control RA progression. Therefore, PIK3CA, EGFR, and JAK2 might emerge as potential vital targets for the treatment of RA.
Effects of RBG on LPS-treated RAW264.7 cells. (A-C) the mRNA expression levels of PIK3CA, EGFR, and JAK2 in RAW264.7 cells stimulated by LPS. #P < 0.05, ##P < 0.01 versus Control; *P < 0.05 and **P < 0.01 versus LPS. RBG, resibufogenin; LPS, Lipopolysaccharide; PIK3CA, hosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha; EGFR, epidermal growth factor receptor; JAK2, Janus Kinase 2.
Resibufogenin regulates the PI3K/AKT signalling pathway to mitigate inflammation
Network pharmacology analysis (KEGG 2025) identified the PI3K/AKT signaling pathway as a key mediator of resibufogenin’s therapeutic effects against RA. Therefore, we quantitatively analyzed the expression levels of key PI3K/AKT pathway proteins (PI3K, p-PI3K AKT, p-AKT) by Western blot analysis. As shown in Fig. 12A-C, compared to the normal group, the protein expression of p-PI3K/PI3K and p-AKT/AKT in the LPS group was increased by 2.35 and 1.63-fold, respectively. Compared to the LPS group, at a concentration of 20 μM, the protein expression of p-PI3K/PI3K and p-AKT/AKT was reduced by 1.03 and 0.77-fold, respectively; at a concentration of 40 μM, it was reduced by 1.00 and 0.52-fold; and at a concentration of 80 μM, it was reduced by 0.42 and 0.19-fold, respectively. These findings suggested that resibufogenin might modulate inflammatory responses through the PI3K/AKT pathway.
The protein expression levels of p-PI3K and p-AKT in RAW264.7 cells treated by RBG. (A) The protein expression levels of p-PI3K and p-AKT were measured by western blot assay. (B, C) Quantitative analysis of protein expression levels of p-PI3K and p-AKT. #P < 0.05, ##P < 0.01 versus Control; *P < 0.05 and **P < 0.01 versus LPS. All data were analyzed as mean ± SD (n = 3)—one-way ANOVA followed by the SNK method. p-PI3, Phosphorylated Phosphoinositide 3-kinase; p-AKT, phosphorylated Protein Kinase B; RBG, resibufogenin; LPS, Lipopolysaccharide.
Discussion
Inflammation is a core pathological feature of RA, causing significant harm to patients. It leads to synovial hyperplasia, vascular membrane formation, and cartilage and bone tissue erosion, resulting in joint deformities and loss of function24. Patients often suffer from persistent pain, restricted movement, and a significantly reduced quality of life. In addition, inflammatory factors can affect the entire body, increasing the risk of cardiovascular diseases and damaging multiple organs, including the lungs, eyes, and nervous system, potentially leading to complications such as conjunctivitis, scleritis, and neuritis, thus severely threatening patient health25. However, the bioactive molecules and mechanisms responsible for the anti-RA effects of toad clothing remain unclear. The toxicity, single-target mechanisms, and drug resistance issues of Tripterygium wilfordii and sinomenine underscore the urgent need for novel traditional Chinese medicines (TCMs). Toad clothing-derived compounds, exhibit multi-target effects and low toxicity, positioning them as promising candidates for rheumatoid arthritis (RA) therapy.Therefore, this study utilized ultra-performance liquid chromatography-mass spectrometry (UPLC-Q-TOF/MS) to identify 24 chemical constituents in toad clothing. It aimed to explore the potential pharmacologically active substances and mechanisms for its anti-RA effects.
This study employed UPLC-Q-TOF/MS technology to identify 24 bioactive constituents in toad clothing. As shown in Fig. 2, these chemical constituents were determined, including 17 steroids and 7 fatty acids. The analysis revealed the main constituents of toad clothing that might participate in the treatment of RA, including resibufogenin, arenobufagin, cinobufagin, cinobufotalin, and hydroxystearic acid. Research indicates that resibufogenin has significant pharmacological activity and promising prospects in areas such as anti-ageing26, anti-breast cancer27, anti-pancreatic cancer28, anti-colorectal cancer29, and immune regulation30. Arenobufagin exhibits pharmacological activity against hepatocellular carcinoma31, colorectal cancer32, and gastric cancer33. Cinobufagin has shown effects against gastric cancer34 and hepatocellular carcinoma35. However, there are few reports on the use of these constituents for treating RA. The active constituents identified through network pharmacology are likely the key substances contributing to the anti-RA effects of toad clothing.
Furthermore, to further elucidate the anti-RA mechanisms of toad clothing, 288 overlapping targets of RA and toad clothing were identified by analyzing several databases, and a PPI network was constructed from these targets (Fig. 4). We constructed a protein–protein interaction (PPI) network, which predicted six core targets, including STAT3, EGFR, PIK3CA, AKT1, JAK2, and HSP90AA1(Fig. 5). Our selection of EGFR, STAT3, AKT1, PIK3CA, HSP90AA1, and JAK2 as core targets was predicated on the following rationale: First, PIK3CA has been conclusively demonstrated to play a pivotal role in RA pathogenesis, with its mutational status showing direct correlation with disease progression and exhibiting well-characterized pharmacological properties supported by extensive inhibitor development studies. Second, although PIK3CB and PIK3CD belong to the same protein family, accumulated evidence indicates their contributions to RA pathogenesis are more ancillary, exhibiting less prominent involvement in critical signaling cascades compared to the central regulatory position of PIK3CA36. Future investigations will systematically elucidate the mechanistic roles of PIK3CB and PIK3CD, particularly their potential synergistic interactions with other signaling molecules in RA pathophysiology. Previous studies have shown that these targets are closely related to inflammatory diseases. Among these critical targets, STAT3 plays a pivotal role in the pathogenesis of RA by regulating the expression of various inflammatory cytokines, such as IL-6, IL-21, and IL-2337. Moreover, STAT3 also plays a crucial role in the chronic inflammation and bone destruction seen in RA. Activating inflammatory cytokines such as IL-1, TNF, and IL-6 further induce the expression of IL-6 family cytokines and Receptor Activator of Nuclear Factor-Kappa B Ligand(RANKL), thus promoting inflammation and bone destruction38. In RA patients, the expression of EGFR in synovial tissue is significantly increased.
EGFR expression was also elevated in the collagen-induced arthritis (CIA) mouse model, suggesting its importance in the pathogenesis of RA39. Applying EGFR inhibitors effectively reduces EGFR expression in the synovium, suppressing inflammation and joint destruction40. The PIK3CA gene encodes the catalytic subunit α of phosphoinositide 3-kinase (PI3K), and its mutation leads to the activation of the PI3K/AKT/Mammalian Target of Rapamycin(mTOR) signalling pathway, which causes vascular abnormalities and hyperplasia in various tissues. This pathway’s activation may play an essential role in the pathogenesis of RA. Although current research mainly focuses on the relationship between the PIK3CA gene and tumours, such as the antitumor activity of copanlisib in PIK3CA-mutated tumours, these studies also provide indirect evidence for the potential role of PIK3CA in RA41. Studies on AKT1 in RA mainly focus on the abnormal activation of signalling pathways and their effects on disease progression42. According to current research, the PI3K/AKT pathway is widely present and abnormally activated in RA synovial cells and is closely related to the abnormal apoptosis of synovial fibroblasts (FLSs) in RA patients43. Specific inhibitors such as LY294002 (which affects the PI3K/AKT pathway) have been shown to have significant therapeutic effects in the CIA mouse model44. JAK2 plays a vital role in RA research. JAK2, a member of the JAK family, participates in the signalling pathways of inflammatory cytokines such as interferon-γ, IL-6, IL-12, and IL-23, which are crucial in autoimmune inflammatory diseases. These cytokines activate the JAK2/STAT signalling pathway, promoting the activation of T helper cell subgroups, thus playing an essential role in the pathological process of RA45,46. Studies have shown that HSP90AA1 exacerbates bone destruction in RA by activating the TNF Receptor-Associated Factor 6/Nuclear Factor of Activated T Cells, Cytoplasmic 1 (TRAF6/NFATc1) signalling pathway47. The core targets predicted by network pharmacology for toad clothing’s intervention in RA are mainly related to inflammation and angiogenesis, providing a reference for further experiments. In conclusion, these studies indicate that the core targets we have identified play essential roles in inflammatory responses, and inhibiting these targets may be an effective way for toad clothing to combat RA.
Gene Ontology (GO) functional enrichment analysis showed that entries closely related to inflammation, protein phosphorylation, intracellular signal transduction, serine phosphorylation, and positive phosphoinositide 3-kinase (PI3K) regulation were dominant. Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis further revealed that the signalling pathways through which toad clothing affects RA mainly involve the PI3K-AKT, Mitogen-Activated Protein Kinase (MAPK) and Ras Proto-Oncogene (Ras) pathways. Among them, the PI3K-AKT signalling pathway plays a key role in the onset of RA. It plays a crucial role in various cellular processes, including cell proliferation, apoptosis, growth, differentiation, transcription, and migration. It is also a key factor in inhibiting inflammation. As an intracellular phosphoinositide kinase, PI3K produces specific phosphorylation reactions; AKT1, a protein kinase family member, is a core regulator in the pathway, influencing tumour cell proliferation and metabolism48. This prediction suggested that toad clothing may intervene in RA by regulating the PI3K-AKT signalling pathway. Molecular docking, a computerized target prediction tool, is mainly used to predict ligand-target interactions and aids in identifying lead constituents with significant activity. To further validate the accuracy of our network pharmacology results, five core constituents were successfully connected to six core targets with good binding ability. Among them, resibufogenin exhibited the best binding ability to EGFR (−9.8 kcal/mol), suggesting that resibufogenin may be the key constituent in exerting anti-RA effects. Resibufogenin was identified as the principal anti-rheumatoid arthritis (RA) constituent based on three key pharmacological advantages: (1) superior polypharmacological properties, demonstrating enhanced mean binding affinity (−7.9 kcal⋅mol − 1) compared to Arenobufagin across multiple targets; (2) greater network centrality (degree value 176 vs 163), indicating its dominant position in RA-associated signaling pathways; and (3) well-established mechanistic evidence for NF-κB pathway inhibition49. Although Arenobufagin exhibited strong binding to EGFR (−9.8 kcal⋅mol − 1), its limited network contribution and insufficient mechanistic validation in RA pathogenesis supported the selection of Resibufogenin as the most promising therapeutic candidate.
Macrophages are immune cells closely related to the onset of inflammation, and NO, TNF-α, IL-6, and IL-1β are important pro-inflammatory factors. The secretion of these inflammatory factors can induce an inflammatory response in the body, making the LPS-induced RAW264.7 cell50 inflammation model widely used in modern pharmacology to evaluate the RA response of various drugs. Based on the network analysis and molecular docking results, we finally selected resibufogenin, which showed the lowest binding energy to each core target, for in vitro anti-inflammatory validation. The LPS-induced RAW264.7 mouse macrophage inflammation model was used to detect the expression of pro-inflammatory factors (NO, TNF-α, IL-6, IL-1β). As shown in Fig. 9D and Fig. 10, the results showed that resibufogenin significantly reduced the levels of inflammatory factors and pro-inflammatory mediators in LPS-induced RAW264.7 cells, indicating that resibufogenin could alleviate the inflammatory response in LPS-induced RAW264.7 cells.
The PI3K-Akt pathway is a crucial signaling pathway within cells, involved in regulating processes such as cell proliferation, survival, metabolism, and migration. In rheumatoid arthritis (RA), the aberrant activation of the PI3K-Akt pathway is closely associated with the excessive secretion of inflammatory factors.Through bioinformatics technology, it was identified that PIK3CA, EGFR, and JAK2 genes are core targets of the PI3K/AKT pathway. As shown in Fig. 11, qPCR analysis of these core targets revealed that resibufogenin significantly reduced the mRNA levels of PIK3CA, EGFR, and JAK2 in LPS-induced RAW264.7 cells. Additionally, As shown in Fig. 12, western blotting demonstrated that resibufogenin inhibited the phosphorylation of PI3K and AKT. Toad clothing may exert its anti-inflammatory effects by suppressing the excessive activation of the PI3K-Akt pathway, thereby reducing the release of inflammatory factors such as TNF-α, IL-6, IL-1β.
In conclusion, this study successfully used UPLC-Q-TOF–MS technology to comprehensively analyze the chemical constituents of toad clothing and preliminarily reveal its mechanism of action in treating RA. This research lays the foundation for the pharmacological activity of the substance and quality control. Additionally, through network pharmacology, we successfully identified the material basis and mechanism of action for toad clothing’s treatment of RA, providing scientific evidence for its clinical application. Compounds with low toxicity were defined as those exhibiting minimal or negligible harmful effects on organisms (including humans) at specified doses. According to international standards (e.g., OECD 423), compounds with acute oral LD50 > 2000 mg/kg (rat) were classified as low-toxicity, while those > 5000 mg/kg were considered practically non-toxic. Existing studies demonstrated significant toxicity differences among toad-derived materials: toad venom (LD50 = 31.62 mg/kg) showed the highest toxicity, followed by toad skin, while toad clothing (shed skin) exhibited relatively lower toxicity due to reduced content of active constituents51. However, the absence of reported LD50 values for toad clothing limited comprehensive safety evaluation. To address this gap, systematic LD50 determination experiments were planned following OECD guidelines to establish complete toxicological profiles and provide reliable safety data for clinical applications. This investigation was designed to fill current research gaps and support the development of toad clothing as a low-toxicity natural medicine. Our study has several limitations. Firstly, we didn’t compare the active component of toad clothing, Resibufogenin, with other anti-RA drugs or assess its combination therapy potential, which limits our understanding of its clinical potentials. Secondly, the complex anti-RA mechanism of Resibufogenin involves multiple pharmacological pathways Please list some of uninvolved pathways, and list the pathways talked in this study. While we’ve identified some key targets and pathways deleted this, list the involved pathways, further research is needed to fully elucidate these mechanisms. In terms of study design, we used the RAW264.7 cell line to demonstrate Resibufogenin’s effects in-vitro, while the understanding of Resibufogenin’s biological effects in-vivo were still unknown, as well as its impact on other key cell types, such as synovial fibroblasts and T/B cells. Moreover, the lack of in-vivo experiments is a significant limitation. Future animal studies and clinical trials are necessary to confirm these findings and to strengthen the reliability and generalizability of our conclusions.
Materials and methods
Materials and reagents
Toad clothing was purchased from Anhui Chanxin Biotechnology Co., Ltd. (Huangshan, China) in May 2023 and authenticated by Long Qingde, a professor at Guizhou Medical University. The amount of toad clothing purchased was 260 g, and the powdered yield after grinding was 90.9%. Before the experiment, the sample materials were dried, pulverized, sieved and then stored in a desiccator at room temperature. Resibufogenin was acquired from Med Chem Express (Monmouth Junction, NJ, USA). Lipopolysaccharides Purchased from Beijing Solarbio Science & Technology Co., Ltd. (Beijing, China).
UPLC-Q-TOF/MS analysis of toad clothing chemical constituents
Laboratory instrument
Instruments for this experiment included: H-Class Ultra High-Performance Liquid chromatograph (Waters, Milford, MA, USA), Sciex Triple Time-of-Flight 4600 Liquid Chromatography/Mass Spectrometry (Waters, Milford, MA, USA), Agilent Eclipse XDB-C18(100 mm × 2.1 mm, 1.8 μm, (Waters, Milford, MA, USA),1/10000ME104、1/1000000XPR2 Electronic Balance(Mettler-Toledo, Shanghai, China).
Sample preparation
0.2 G of toad clothing51 powder was placed in a round-bottom flask, 25 ml of methanol was added, and the extract was heated to reflux for 1 h, filtered, Concentrate the filtrate, and diluted the concentrated solution to a final volume of 5 ml with methanol. UPLC-Q-TOF/MS is a highly sensitive and highly resolved analytical technique, suitable for the rapid separation and precise identification of chemical components in complex samples. Its high separation efficiency and sensitivity have made it an ideal choice for the analysis of chemical constituents of traditional Chinese medicine52. The quantitative analysis of Resibufogenin in toad clothing was limited by insufficient sample quantities; therefore, this study employed the literature-reported concentration range (31.3–80 μg/g) as reference values53. Subsequent investigations were planned to perform systematic quantification using UPLC-Q-TOF/MS.
UPLC-Q-TOF/MS qualitative analysis
The conditions for chromatographic analysis were as follows: chromatographic separation was performed on an Agilent Eclipse XDB-C18(100 mm × 2.1 mm, 1.8 μm), The mobile phase was composed of 0.1% formic acid solution (solvent A) and formic acid acetonitrile solution (solvent B). The gradient elution setup was as follows: 0–3 min, 5% B;3–10 min, 5%–30% B, 10–15 min, 30%–39% B; 15–25 min,39% B; 25–30 min, 39%–95% B; 30–35 min, 95% B;35–35.1 min, 95%–5% B; 35.1–40 min, 5% B. The flow rate was set at 0.3 ml min − 1, the column temperature was 30℃, and the injection volume was 5μL.
The conditions for mass spectrometry analysis were as follows: the capillary voltages were set at 5 kV (positive ion mode) and 4.5 kV (negative ion mode). Nitrogen was used as the drying gas, and the source temperature was set to 500 ℃. The sample cone voltage was 100 V, the collision energy was 40e V, and the mass scan range was m/z 120 to 1500 Da54.
Network pharmacology
Collection of active constituents and targets of drugs
The Chemical Abstracts Service (CAS) numbers of the toad clothing chemical components identified by UPLC-Q-TOF/MS were entered into the PubChem (https://pubchem.ncbi.nlm.nih.gov/)platform for searching to screen the chemical constituents with Simplified Molecular Input Line Entry System (SMILES) and 2D Structure-Data File (2D-SDF) file, and the literature was reviewed to complement the chemical constituents that did not have SMILES and 2D-SDF file, but with significant biological activity. SMILES and 2D-SDF files, which are standard representations of chemical structures, played a key role in the storage and exchange of molecular information. Afterwards, SwissTargetPrediction (https://www.swisstargetprediction.ch/) and PharmMapper database (https://lilab-ecust.cn/pharmmapper/index.html) were used to import the structures of the chemical ingredients55. Then, the targets with a probability greater than zero and Norm Fit greater than 0.2 were selected and imported into the Uniport online website(https://www.uniprot.org/) to be converted to the appropriate gene symbols. Lastly, the targets of the candidate ingredients were determined by combining and deleting duplicates from the obtained targets.
Collection of rheumatoid arthritis-related targets and potential therapeutic targets
The GeneCards, DisGeNET, DrugBank, TTD, and OMIM databases provided valuable information about genes, diseases, drugs, and therapeutic targets, and have been essential resources in biomedical research. GeneCards (https://previous.genecards.org/), DisGeNET (https://disgenet.com/), DrugBank (https://go.drugbank.com), TTD (https://db.idrblab.net/ttd/) and OMIM (https://www.omim.org/) databases were searched for targets related to Rheumatoid Arthritis with the keyword Rheumatoid Arthritis. GeneCards search results were analyzed using the median screening method 2 times to remove many targets with relevance scores < 3.11. The targets with relevance scores ≥ 3.11 in GeneCards search results were selected and merged with OMIM, DisGeNET, DrugBank, and TTD database search results to obtain Rheumatoid Arthritis-related targets after removing duplicates. Then, the mutual targets of chemical constituents and diseases were obtained through the Jvenn(https://jvenn.toulouse.inra.fr/) platform.
Construction of protein–protein interaction networks and screening of core targets
To establish the PPI network, the obtained"candidate constituent-disease” common targets were added to the STRING (https://cn.string-db.org/) database. The species was limited to“Homo sapiens”; the confidence level was set to 0.9 as a threshold. Subsequently, Cytoscape3.10.1 software was used to analyze the downloaded Tab-Separated Values (TSV) format result files visually. Network Analyzer tools were used to analyze the topological properties and relationships between nodes in networks, which helped to understand the structures and functions of biological networks. The Network Analyzer plugin was employed to compute the betweenness centrality (BC), closeness centrality (CC) and degree value (Degree). Network topology parameters were calculated to quantitatively characterize the structural organization and functional attributes of the component–target interactome. Key metrics included degree centrality (which measures node connectivity to identify pivotal hubs), closeness centrality (which assesses the average shortest path length to reflect information transmission efficiency), betweenness centrality (which quantifies a node’s role as a bridge along shortest paths to pinpoint regulatory bottlenecks), and clustering coefficient (which evaluates the interconnectivity among a node’s neighbors to reveal functional modules). These parameters were standardized using the Network Analyzer tool in Cytoscape, enabling systematic identification of core targets, functional modules, and regulatory pathways. This approach provided critical insights into the mechanisms underlying component–target interactions, aligned with established network pharmacology frameworks, and ensured robust topological characterization of complex biological systems. Lastly, common targets above the average of the three were selected as the core targets.
Construction of drug-constituents-targets-pathway-disease network
The chemical constituents, common target genes and Rheumatoid Arthritis diseases of toad clothing were imported into Cytoscape3.10.1, and a “drug-constituents-targets-pathway-disease"network diagram of the anti-Rheumatoid Arthritis activity of toad clothing was constructed. The software Network Analyzer tool was utilized to calculate network topology parameters. The top five compounds (Resibufogenin, Arenobufagin, Cinobufagin, Cinobufotalin, and Hydroxystearic acid) were selected based on their degree centrality rankings within the constructed"drug-component-target-pathway-disease"network. This prioritization criterion ensured that the chosen compounds exhibited the most significant interactions with core disease targets, thereby confirming their biological relevance and therapeutic potential for subsequent experimental validation.
GO and KEGG enrichment analysis
The cross-targets were uploaded to the David database (https://david.ncifcrf.gov/) for gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG)56,57,58 pathway enrichment Analysis. The restricted species was Homo sapiens; the screening criterion was P < 0.05. The visualization and mapping were performed using online tools such as the bioinformatics platform (https://www.bioinformatics.com.cn/).
Molecular docking
The 2D structures of the core ligand constituents were obtained from the PubChem database in SDF format and saved in mol2 format after energy minimization using Chem 3D 19.0. The obtained core targets were downloaded from the PDB database (https://www.rcsb.org/) in Protein Data Bank (PDB) format files. TSV, mol2, and PDBQT format files served as standard data storage formats, ensuring data compatibility and readability. PyMOL software was a powerful tool in molecular visualization, widely used for viewing and analyzing molecular structures. The PyMoL software removed water molecules and ligands from protein structures. AutoDockTools software was an important tool in molecular docking research, which was used to prepare and analyze molecular docking studies.Then, the proteins and constituents were hydrogenated with AutoDockTools-1.5.7 software and saved as pdbqt format files. Finally, the AutoDock Vina software was utilized for molecular docking. The heat map was drawn using online tools, such as the bioinformatics platform (https://www.chiplot.online/), to display the free binding energy, and the visual analysis was carried out using PyMoL software59. Here was a brief explanation and standard for using AutoDock Vina software for molecular docking: first, downloaded the protein structure from the PDB database, removed water molecules, added hydrogen, and calculated charges, saving it as.pdbqt format; at the same time, downloaded small molecule structures from databases such as PubChem, added hydrogen, and calculated charges, saving them as.pdbqt format. Next, determined the binding site, set the center coordinates and size of the docking box, and created a configuration file (conf.txt) containing the names of the receptor and ligand and docking parameters. Then, ran AutoDock Vina using the command vina –config conf.txt for docking. After the docking was complete, a result file containing the binding energies of different conformations was generated. Finally, used AutoDock Tools or PyMol to view the results and analyze the conformation with the lowest binding energy. During the docking process, the default scoring function of AutoDock Vina was used to evaluate the binding energy, and the binding affinity between molecules was judged by the size of the binding energy59.
In vitro experiments
Cell culture
RAW264.7 cells were originated from Wuhan Pricella Biotechnology Co., Ltd and cultured in high-sugar Dulbecco’s Modified Eagle Medium (DMEM)(Thermo Fisher Scientific, Waltham, MA, USA) media supplemented with 10% fetal bovine (Gibco, Los Gatos, CA, USA), Chinaserum and 1% penicillin–streptomycin in the fully humidified incubator at 37 ℃ with 5% CO2.
Cytotoxicity assay
RAW264.7 cells were inoculated in 96-well plates (1 × 104 cells/well) and cultured for 24 h. Then, 100μL of medium containing different concentrations of sample solutions (2.5 μM-160 μM) was added. After each well’s 24 h incubation period, the medium was taken out. After adding 10% Cell Counting Kit (CCK) (Invigentech, Irvine, CA, USA) solution, the mixture was incubated at 37 ℃ for one h60. The absorbance at 450 nm was read using a spectrophotometer (Bio Tek, Winooski, VT, USA). The Optical Density (OD) values were used to calculate cell viability.
Determination of nitric oxide and inflammatory cytokines
RAW264.7 cells were inoculated into a 96-well plate (1 × 104 cells/well) and cultured for 24 h. The 1 μg/mL LPS concentration was selected as the optimal dose for inducing M1 macrophage polarization in RAW264.7 cells, as validated by published studies. This concentration robustly enhanced pro-inflammatory cytokine expression (e.g., TNF-α, IL-6) while maintaining cellular viability and model stability61. Regarding the selection of resibufogenin (RBG) dosage, we conducted cytotoxicity assays on RAW264.7 cells using a gradient of RBG concentrations (0, 2.5, 5, 10, 20, 40, 80, and 160 μmol/L). Results indicated that ≤ 80 μmol/L RBG exhibited minimal cytotoxicity. Further anti-inflammatory efficacy analyses demonstrated that RBG at 20–80 μmol/L significantly and dose-dependently reduced LPS-induced pro-inflammatory cytokine (e.g., TNF-α, IL-6) expression in RAW264.7 cells, confirming its therapeutic efficacy within this range. Afterwards, Resibufogenin and different concentrations of constituents mixed with LPS (the final concentration of LPS was one µg/mL, and the samples were 20,40 and 80 µM) were incubated in the complete medium for 24 h.The supernatant was collected to determine the content of NO, TNF-α, IL-6, IL-1β62. The Griess assay protocol was incorporated into the revised manuscript as follows: Nitrite \({\rm (NO_2^1)}\), the stable oxidation product of nitric oxide (NO), was quantified spectrophotometrically. Cell culture supernatants were clarified by centrifugation (12,000 × g, 5 min). Then, 50 μL of the supernatant was mixed with 50 μL of Griess Reagent I (1% sulfanilamide in 5% phosphoric acid) and 50 μL of Griess Reagent II (0.1% N-(1-naphthyl) ethylenediamine dihydrochloride). After incubation at room temperature for 10 min, absorbance was measured at 540 nm using a microplate reader (Thermo Scientific Multiskan FC).Nitrite concentrations were calculated using a sodium nitrite standard curve (0–100 μM)62. The contents of Nitric oxide (NO) were determined by Gris’s reagents (Biyuntian Biotechnology Co., Ltd., Shanghai, China), TNF-α, IL-6, IL-1β and concentrations were determined by ELISA kit (Jianglai Biotechnology Co., Ltd., Shanghai, China). ELISA Plate Reader: Model Synergy H1 (BioTek Inc., USA) for ELISA detection. In the ELISA experiment, we followed the following standard steps: Sample preparation: collected more than 3 groups of cell supernatant samples, usually diluted 1:50 to 1:100 to reduce background interference caused by non-specific antibody binding.After homogenization in lysis buffer with lysis buffer, the cell lysate was centrifuged at 10,000 × g for 5 min at 4°C, and the supernatant was taken for immediate detection or aliquoted and stored at −80 °C. Coating and blocking: The ELISA plate was coated with the capture antibody and incubated overnight at 4 °C. Unbound sites were blocked with bovine serum albumin (BSA), incubated at room temperature for 1–2 h. Antibody incubation: Primary antibodies were diluted 1:1000–1:5000, and secondary antibodies were diluted 1:2000–1:10,000. Enzyme-labeled secondary antibodies were used, which could be horseradish peroxidase (HRP) or alkaline phosphatase (AP).Detection: After adding the substrate, a color change was produced depending on the enzyme, and the absorbance was measured using a microplate reader to draw a standard curve for quantitative analysis of the target antigen. These steps ensured the accuracy and sensitivity of the detection62.
Quantitative real-time polymerase chain reaction (qRT-PCR) analysis for gene expression
Total RNA from RAW264.7 cells was extracted utilizing the TaKaRa (Kusatsu, Shiga, Japan) Kit, 1 μg of which was applied for cDNA synthesis. qRT-PCR was followed using SYBR Green Master Mix with Roche LightCycler 480 real-time PCR system63. The relative mRNA expression of tested genes was calculated using the 2-ΔΔCt method using Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH) as the housekeeper gene. Primers involved in our work are listed in Table 3.
Western blotting
Protein samples (20 μg per lane) were resolved on 10% SDS-PAGE gels using the Bio-Rad Mini-PROTEAN Tetra System. Electrophoresis was performed at 80 V for 30 min through the stacking gel, followed by 120 V for 90 min through the resolving gel. Proteins were then transferred to PVDF membranes (activated in methanol for 1–2 min and equilibrated in transfer buffer for 5 min) using the Bio-Rad Trans-Blot Turbo System at a constant current of 180 mA for 120 min in an ice bath. Membranes were blocked with 5% non-fat milk (Thermo Fisher Scientific) for 1 h at room temperature, followed by overnight incubation at 4 °C with primary antibodies (PI3K, p-PI3K, AKT, p-AKT; Cell Signaling Technology; 1:1000 diluted in 5% BSA). After thorough washing, membranes were incubated with HRP-conjugated secondary antibodies (goat anti-rabbit IgG; Bioworld; 1:5000 diluted in 5% milk) for 1 h at room temperature. Protein bands were visualized using the Bio-Rad ChemiDoc MP Imaging System with enhanced chemiluminescence (ECL) substrate. RAW247.6 cells were inoculated in 6-well plates and incubated in a medium containing various resibufogenin concentrations (0, 20, 40 and 80 μM/L) for the indicated time. Cell lysates were prepared with Radioimmunoprecipitation Assay (RIPA) buffer supplemented with phosphatase and protease inhibitors—the Bicinchoninic Acid Assay (BCA)protein assay kit quantified protein concentrations. Protein samples were heated at 95 °C for 10 min following diluted in 5 × loading buffer. Then, each sample was fractionated on 10% Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis (SDS-PAGE) (Solarbio, Beijing, China) gels and transferred to the Polyvinylidene Fluoride (PVDF)membrane.SDS-PAGE Electrophoresis System: Bio-Rad Mini-PROTEAN Tetra Cell (Bio-Rad Laboratories, USA) for protein separation. The PVDF membranes were blocked with blocking solution for one h and incubated with different primary antibodies (Cell Signaling Technology, Danvers, MA, USA) (1:1000) overnight at 4 °C. Transfer System: Bio-Rad Trans-Blot Turbo Transfer System (Bio-Rad Laboratories, USA) for Western blot transfer.Then, a Horseradish Peroxidase (HRP)-conjugated secondary antibody (Bioworld, Bloomington, MN, USA) (1:1000) was added at room temperature, and the membrane was washed with 1 × Tris Buffered Saline with Tween (TBST)64. Chemiluminescence Imager: Bio-Rad ChemiDoc MP Imaging System (Bio-Rad Laboratories, USA) for protein band detection.Proteins were visualized with enhanced chemiluminescence (ECL) detection reagents. Semi-quantitative analysis was performed by using Image J software. Target protein levels were normalized by the level of GAPDH (Bioworld, Bloomington, MN, USA).
Statistical analysis
All the experiments were performed at least three times. Data were analyzed by GraphPad Prism 9 (GraphPad Software, San Diego, CA, USA) and showed as mean ± SD. GraphPad Prism is a commonly used software in the biosciences for data analysis and chart creation, providing a wealth of statistical analysis features. Statistical analysis used one-way ANOVA followed by the Tukey test. All statistical tests with P < 0.05 were taken as statistically significant differences.
Data availability
All data are contained in the article. The datasets used and/or analyzed during the current study are available from the first author(T.W.)or the corresponding author (J.X.) (363912577@qq.com) upon reasonable request.
References
Di Matteo, A., Bathon, J. M. & Emery, P. Rheumatoid arthritis. Lancet 402, 2019–2033 (2023).
Gravallese, E. M. et al. What Is Rheumatoid Arthritis?. N. Engl. J. Med. 390, e32 (2024).
Gan, P.-R., Wu, H., Zhu, Y.-L., Shu, Y. & Wei, Y. Glycolysis, a driving force of rheumatoid arthritis. Int. Immunopharmacol. 132, 111913 (2024).
Yang, M. et al. Rheumatoid arthritis increases the risk of malignant neoplasm of bone and articular cartilage: A two-sample bidirectional mendelian randomization study. Arthritis Res. Ther. 25, 219 (2023).
Zhang, M., Li, M., Hu, H., Li, X. & Tian, M. Global, regional, and national burdens of rheumatoid arthritis in young adults from 1990 to 2019. Arch. Med. Sci. 20, 1153–1162 (2024).
Luo, P. et al. Metabolic Syndrome Is Associated With an Increased Risk of Rheumatoid Arthritis: A Prospective Cohort Study Including 369,065 Participants. J. Rheumatol. 51, 360–367 (2024).
Burmester, G. R. & Pope, J. E. Novel treatment strategies in rheumatoid arthritis. Lancet 389, 2338–2348 (2017).
Lewis, M. J. Predicting best treatment in rheumatoid arthritis. Semin. Arthritis Rheum. 64S, 152329 (2024).
Prasad, P. et al. Rheumatoid arthritis: Advances in treatment strategies. Mol. Cell Biochem. 478, 69–88 (2023).
Brown, P., Pratt, A. G. & Hyrich, K. L. Therapeutic advances in rheumatoid arthritis. BMJ 384, e070856 (2024).
Liang, Y., Liu, M., Cheng, Y., Wang, X. & Wang, W. Prevention and treatment of rheumatoid arthritis through traditional Chinese medicine: role of the gut microbiota. Front. Immunol. 14, 1233994 (2023).
Wang, Y. et al. Traditional herbal medicine: Therapeutic potential in rheumatoid arthritis. J. Ethnopharmacol. 279, 114368 (2021).
D, Z. et al. Advances on Delivery System of Active Ingredients of Dried Toad Skin and Toad Venom. International journal of nanomedicine 19, (2024).
H, T., F, Z., Bs, Y. & Bt, Z. Combinational Antitumor Strategies Based on the Active Ingredients of Toad Skin and Toad Venom. Drug design, development and therapy 18, (2024).
Zhao, X. et al. Anti-rheumatoid arthritis effects of iridoid glucosides from Lamiophlomis rotata (Benth.) kudo on adjuvant-induced arthritis in rats by OPG/RANKL/NF-κB signaling pathways. J. Ethnopharmacol. 266, 113402 (2021).
Sun, M., Han, X., Liu, X. & Xu, Y. Cinobufacini suppresses malignant behaviors of endometrial cancer by regulating NF-κB pathway. Biotechnol. Genet. Eng. Rev. 40(3), 2221–2233 (2023).
Uttra, A. M. et al. Ribes orientale: A novel therapeutic approach targeting rheumatoid arthritis with reference to pro-inflammatory cytokines, inflammatory enzymes and anti-inflammatory cytokines. J. Ethnopharmacol. 237, 92–107 (2019).
Zheng, Y. et al. Screening of Bufadienolides from Toad Venom Identifies Gammabufotalin as a Potential Anti-inflammatory Agent. Planta Med. 88, 43–52 (2022).
Li, W. et al. Prevention and treatment of inflammatory arthritis with traditional Chinese medicine: Underlying mechanisms based on cell and molecular targets. Ageing Res. Rev. 89, 101981 (2023).
Xie, S. et al. Cinobufagin Modulates Human Innate Immune Responses and Triggers Antibacterial Activity. PLoS ONE 11, e0160734 (2016).
Yang, X. et al. Lonicerin alleviates the progression of experimental rheumatoid arthritis by downregulating M1 macrophages through the NF-κB signaling pathway. Phytother. Res. 37, 3939–3950 (2023).
Lee, H. S. et al. Anti-inflammatory effects of Allium cepa L. peel extracts via inhibition of JAK-STAT pathway in LPS-stimulated RAW264.7 cells. J. Ethnopharmacol. 317, 116851 (2023).
Sahoo, D. K. et al. Oxidative stress, hormones, and effects of natural antioxidants on intestinal inflammation in inflammatory bowel disease. Front. Endocrinol. 14, 1217165 (2023).
Guagnano, M. T. et al. Improvement of Inflammation and Pain after Three Months’ Exclusion Diet in Rheumatoid Arthritis Patients. Nutrients 13, 3535 (2021).
Komatsu, N. & Takayanagi, H. Mechanisms of joint destruction in rheumatoid arthritis - immune cell-fibroblast-bone interactions. Nat. Rev.. Rheumatol. 18, 415–429 (2022).
Takaya, K., Asou, T. & Kishi, K. Identification of resibufogenin, a component of toad venom, as a novel senolytic compound in vitro and for potential skin rejuvenation in male mice. Biogerontology 24, 889–900 (2023).
Yang, T. et al. Resibufogenin Suppresses Triple-Negative Breast Cancer Angiogenesis by Blocking VEGFR2-Mediated Signaling Pathway. Front. Pharmacol. 12, 682735 (2021).
Han, Q. et al. Resibufogenin suppresses colorectal cancer growth and metastasis through RIP3-mediated necroptosis. J. Transl. Med.. 16, 201 (2018).
Liu, L. et al. Resibufogenin suppresses transforming growth factor-β-activated kinase 1-mediated nuclear factor-κB activity through protein kinase C-dependent inhibition of glycogen synthase kinase 3. Cancer Sci. 109, 3611–3622 (2018).
Wang, S. et al. Metabolic reprogramming by traditional Chinese medicine and its role in effective cancer therapy. Pharmacol. Res. 170, 105728 (2021).
Yang, Y. et al. Arenobufagin regulates the p62-Keap1-Nrf2 pathway to induce autophagy-dependent ferroptosis in HepG2 cells. Naunyn Schmiedebergs Arch. Pharmacol. 397, 4895–4909 (2024).
Wang, M. et al. Arenobufagin inhibits lung metastasis of colorectal cancer by targeting c-MYC/Nrf2 axis. Phytomedicine 127, 155391 (2024).
Deng, Y.-Q. et al. Compound-composed Chinese medicine of Huachansu triggers apoptosis of gastric cancer cells through increase of reactive oxygen species levels and suppression of proteasome activities. Phytomedicine 123, 155169 (2024).
Yang, A. et al. Cinobufagin restrains the growth and triggers DNA damage of human hepatocellular carcinoma cells via proteasome-dependent degradation of thymidylate synthase. Chem. Biol. Interact 360, 109938 (2022).
Xiong, X. et al. Inhibition of autophagy enhances cinobufagin-induced apoptosis in gastric cancer. Oncol. Rep. 41, 492–500 (2019).
Damian, L., Lebovici, A., Pamfil, C., Belizna, C. & Vulturar, R. Rheumatoid Arthritis and CLOVES Syndrome: A Tricky Diagnosis. Diagnostics 10, 467 (2020).
Samuels, J. S. et al. Prostaglandin E2 and IL-23 interconnects STAT3 and RoRγ pathways to initiate Th17 CD4+ T-cell development during rheumatoid arthritis. Inflamm Res. 67, 589–596 (2018).
Hu, L., Liu, R. & Zhang, L. Advance in bone destruction participated by JAK/STAT in rheumatoid arthritis and therapeutic effect of JAK/STAT inhibitors. Int. Immunopharmacol. 111, 109095 (2022).
Shu, H. et al. Transcriptomics-based analysis of the mechanism by which Wang-Bi capsule alleviates joint destruction in rats with collagen-induced arthritis. Chin Med. 16, 31 (2021).
Ge, Y. et al. Identification and validation of hub genes of synovial tissue for patients with osteoarthritis and rheumatoid arthritis. Hereditas 158, 37 (2021).
Choi, J. et al. Integrated mutational landscape analysis of uterine leiomyosarcomas. Proc. Natl. Acad. Sci. U. S. A. 118, e2025182118 (2021).
Ye, H. et al. CD4 T-cell transcriptome analysis reveals aberrant regulation of STAT3 and Wnt signaling pathways in rheumatoid arthritis: Evidence from a case-control study. Arthritis Res. Ther. 17, 76 (2015).
Glaviano, A. et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol. Cancer 22, 138 (2023).
He, J. et al. Arecoline hydrobromide suppresses PI3K/AKT pathway in rheumatoid arthritis synovial fibroblasts and relieves collagen-induced arthritis in mice. Int. Immunopharmacol. 124, 110925 (2023).
Yan, Y. et al. Jolkinolide B ameliorates rheumatoid arthritis by regulating the JAK2/STAT3 signaling pathway. Phytomedicine 124, 155311 (2024).
Zhong, X. et al. Periplogenin inhibits pathologic synovial proliferation and infiltration in rheumatoid arthritis by regulating the JAK2/3-STAT3 pathway. Int. Immunopharmacol. 128, 111487 (2024).
Zeng, P., Huang, H. & Li, D. Combining bioinformatics, network pharmacology, and artificial intelligence to predict the mechanism of resveratrol in the treatment of rheumatoid arthritis. Heliyon 10, e37371 (2024).
Ba, X. et al. WTD Attenuating Rheumatoid Arthritis via Suppressing Angiogenesis and Modulating the PI3K/AKT/mTOR/HIF-1α Pathway. Front. Pharmacol. 12, 696802 (2021).
Gao, Y. et al. Resibufogenin, one of bufadienolides in toad venom, suppresses LPS-induced inflammation via inhibiting NF-κB and AP-1 pathways. Int. Immunopharmacol. 113, 109312 (2022).
Linghu, K. et al. Small molecule α-methylene- γ-butyrolactone, an evolutionarily conserved moiety in sesquiterpene lactones, ameliorates arthritic phenotype via interference DNA binding activity of NF- κ B. Acta Pharm. Sin. B 14, 3561–3575 (2024).
Yang, Q. et al. Angel of human health: current research updates in toad medicine. Am. J. Trans. Res. 7, 1 (2015).
Chen, J. et al. UPLC-Q-TOF-MS based investigation into the bioactive compounds and molecular mechanisms of Lamiophlomis Herba against hepatic fibrosis. Phytomedicine 121, 155085 (2023).
Gao, H. et al. Chemical constituents from Bufonis periostracum and their antitumor activity in vitro. Ch. J. Ch. Mater. Med. 36, 2207–2210 (2011).
Basit, A. et al. UPLC-Q-TOF-MS profiling of Viola stocksii Boiss and evaluation of aphrodisiac potential and risk factors associated with erectile dysfunction. J. Ethnopharm. 321, 117477 (2024).
Nogales, C. et al. Network pharmacology: curing causal mechanisms instead of treating symptoms. Trends Pharmacol. Sci. 43, 136–150 (2022).
Kanehisa, M. & Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 28, 27–30 (2000).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28, 1947–1951 (2019).
Kanehisa, M., Furumichi, M., Sato, Y., Matsuura, Y. & Ishiguro-Watanabe, M. KEGG: biological systems database as a model of the real world. Nucleic Acids Res. 53, D672–D677 (2025).
Eberhardt, J., Santos-Martins, D., Tillack, A. F. & Forli, S. AutoDock Vina 1.2.0: New Docking Methods, Expanded Force Field, and Python Bindings. J. Chem. Inf. Model. 61, 3891–3898 (2021).
Zhao, J. et al. iPSC-derived exosomes as amphotericin B carriers: A promising approach to combat cryptococcal meningitis. Front. Microbial. 16, 1531425 (2025).
Li, X., Qi, H., Zhang, X. & Liang, H. Zeng N Jing-Fang n-butanol extract and its isolated JFNE-C inhibit ferroptosis and inflammation in LPS induced RAW264.7 macrophages via STAT3/p53/SLC7A11 signaling pathway. J. Ethnopharmacol. 316, 116689 (2023).
Tu, C. et al. Promoting the healing of infected diabetic wound by an anti-bacterial and nano-enzyme-containing hydrogel with inflammation-suppressing, ROS-scavenging, oxygen and nitric oxide-generating properties. Biomaterials 286, 121597 (2022).
Li, L., Guo, X. & Wang, S. Selection and evaluation of reference genes for qRT-PCR in Inonotus obliquus. Front. Microbial. 16, 1500043 (2025).
Owen, C. & Fader, K. A. Hassanein M Western blotting: evolution of an old analytical method to a new quantitative tool for biomarker measurements. Bioanalysis 16, 319–328 (2024).
Acknowledgements
Special Project for Scientific and Technological Research on Traditional Chinese Medicine and Ethnic Medicine of Guizhou Province Administration of Traditional Chinese Medicine, QZYY-2024-012. National Natural Science Foundation of China, 82401632. Guizhou Province Science and Technology Fund, QKHJC‐ZK [2023] ‐302.
Funding
Guizhou Province Science and Technology Fund,QKHJC‐ZK [2023] ‐302,Special Project for Scientific and Technological Research on Traditional Chinese Medicine and Ethnic Medicine of Guizhou Province Administration of Traditional Chinese Medicine,QZYY-2024-012,National Natural Science Foundation of China,82401632
Author information
Authors and Affiliations
Contributions
Conceptualization, writing the original draft, data curation, T.W. and J.X.; participation in experiments, T.W. and H.X.; providing advice about the experimental procedure, J.Z., M.Y. and Z.Z.; funding acquisition, J.X. and W.Z.; writing-review and editing, supervision and project administration, J.L. and J.X. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, T., Xia, H., Zhu, J. et al. UPLC-Q-TOF/MS, network pharmacology, molecular docking, and experimental validation to explore the mechanisms of toad clothing on rheumatoid arthritis. Sci Rep 15, 30005 (2025). https://doi.org/10.1038/s41598-025-16053-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16053-x