Abstract
This study investigates sperm telomere length (STL), gene polymorphisms in telomere-related pathway genes, and semen quality, were detected to evaluate their potential as a biomarkers of semen quality. Associationally, it explores between gene polymorphisms and STL, and semen quality. A total of 1,349 male volunteers from the Pu-yang Reproductive Medicine Center were selected as research subjects. Computer-aided sperm analysis analysis was employed to assess semen parameters. Real-time fluorescence quantitative PCR was utilized to measure STL, while polymerase chain reaction was used to detect gene polymorphisms. The correlation between STL, related gene polymorphisms, and semen parameters was subsequently analyzed. Among the 1,349 subjects, the proportions of semen volume, total sperm count, sperm concentration, progressive motility, and total sperm motility meeting the criteria exceeded 80%, whereas the proportion of normal morphology was only 32.46%. The results of the restricted cubic spline (RCS) analysis indicated that STL exhibtied a nonlinear relationship with sperm concentration (P < 0.001) and total sperm count (P = 0.007). After categorizing STL levels into quartiles, the risk of abnormalities in total sprem count (OR = 0.37[0.15,0.94], P = 0.037), sperm concentration (OR = 0.31[0.11,0.83], P = 0.020), progressive motility (OR = 0.27[0.12,0.61)], P = 0.002), and total vitality percentage (OR = 0.19[0.07,0.51], P = 0.001) was lower in the third quartile when compared to the first quartile. We explored the relationship between genetic variation in three key telomere pathway genes (POT1, TERF1, and TERT), STL, and semen parameters. STL levels were significantly higher in individuals carrying the POT1 rs1034794 mutant allele than in type individuals (β = 6.17, P = 0.005). We observed that the POT1 rs1034794 and rs10250202 variants were negatively correlated with total sperm count (β=-64.47), progressive motility (β1=-9.76; β2=-6.99), and total vitality percentage (β1=-11.80; β2=-7.04). The TERT rs2735940 variant was negatively correlated with Wobble (WOB) and Linearity (LIN). There is a non-linear relationship between STL and sperm concentration as well as total sperm count. STL may serve as potentially reliable biomarker of semen quality. Furthermore, single nucleotide polymorphisms (SNPs) in telomere-associated pathway genes (POT1, TERT) were significantly associated with sperm parameters and STL. This suggests a novel hypothesis thought polymorphisms in telomere pathway genes may indirectly regulate semen parameters by affecting STL.
Similar content being viewed by others
Introduction
Over the past few decades, an increasing number of couples have been suffering from infertility during their reproductive years. Infertility is defined as a couple’s failure to conceive after at least a year of unprotected regular sexual intercourse1, with male infertility accounting for more than half of these cases2. When assessing male fertility, clinical and departmental semen parameter tests are often used as surrogate and indirect evidence for male fertility assessment. Although semen quality analysis is currently the preferred test for diagnosing male infertility, some infertile patients still exhibit normal semen analysis results3,4. Moreover, the weak effects of cartain influencing factors on male reproductive health may be difficult to measure with conventional semen quality assessments alone. Therefore, further exploration for more sensitive biomarkers is needed5.
Another layer of complexity in the etiology of male infertility is the role of telomeres, which is currently receiving widespread attention. Telomeres are non-coding highly conserved tandem repeat DNA sequences (TTAGGG repeats) that play a crucial role in maintaining genomic integrity6,7. Several studies have investigated sperm telomere length(STL) and white blood cell telomere length (LTL) in the context of male infertility. One report indicated that infertile men exhibited significantly shorter mean values of LTL and STL compared to individuals of childbearing potential8. Similarly, the average telomere length of infertile men was significantly shorter than that of men of childbearing age. Additionally, sperm count and semen progressive motility were found to be related to telomere length9. However, some studies reported conflicting results, failing to establish an association between STL and sperm parameters such as sperm count, concentration, forward motility, and morphology. Most studies, however, identified a linear relationship between these factors, albeit in a small number of subjects10,11,12. This suggests that further research is necessary to elucidate the role of STL in assessing semen parameters using non-linear models. The restricted cubic spline (RCS) model can be applied to three types of analyses-inear, logistic, and Cox regression and offers the advantage of evaluating scenarios without a priori hypotheses regarding the shape of the dose-response association curve between semen parameters and STL.
Protection against telomeric fusion is primarily regulated by the telomerase complex and the shelterin complex. Telomerase counteracts telomere fusion by adding telomere repeats to the ends of chromosomes13,14. Importantly, telomerase remains active in germ cells during spermatogenesis to ensure the transmission of full-length chromosomes to offspring. Human telomerase is composed of telomerase reverse transcriptase (TERT), telomerase RNA components (TERC), and a pseudouracil synthetase. TERT serves as the rate-limiting catalytic subunit of telomerase15. Previous studies utilizing TERT reporter mice have shown that high telomerase expression is characteristic of undifferentiated spermatogonia16. In mice deficient in telomerase RNA, subsequent generations display defects in spermatogenesis, increased apoptosis, and reduced testicular proliferation17. Telomere-associated shelterin protein complexes, which consist of six subunits (TRF1, TRF2, RAP1, TIN2, TPP1, and POT1), play a crucial role in telomere maintenance. A study demonstrated that TRF1, Speedy A, and Cdk2 protect telomeres from non-fusion within a densely populated microenvironment during spermatocyte meiosis18. Di(2-ethylhexyl) phthalate (DEHP) induces male reproductive damage by shortening telomeres and reducing levels of TRF1, TRF2, POT1, and TERT19. Genetic variations in telomere-related pathway genes may influence telomere length and chromosomal stability, thereby affecting disease susceptibility. Consequently, we hypothesized that single nucleotide polymorphisms (SNPs) in this pathway may be associated with male infertility risk, and analyzed eight common SNPs in telomere related genes.
To address the aforementioned gaps, a large cross-sectional study was conducted at the Reproductive Center of Pu-yang Maternal and Child Health Hospital in Henan Province, China. Computer-aided sperm analysis was employed to evaluate semen parameters. To gain a the comprehensive understanding of the effects of STL and their mechanisms on semen quality, we further examined STL and gene polymorphisms in telomere-related pathway genes.Additionally, the RCS model was applied to assess the non-linear relationship between STL and sperm parameters. The mediating effect of gene polymorphisms in telomere-related pathway genes was investigated to elucidate the mechanism by which STL influences semen parameters.
Materials and methods
Study population
Volunteers were recruited from the Reproductive Center of Puyang Maternal and Child Health Care Hospital in Henan Province, China, between December 2017 and August 2018. According to the criteria outlined in Fig. 1, a total of 1,349 individuals completed the study procedures, which included questionnaire surveys and routine physical examinations. The height and weight of the subjects were measured. ‘Never drinking alcohol’ is defined as consuming less than 15 g of alcohol per day.The samples collected from the population were not exposed to occupational or environmental metals. After semen analysis, the semen samples were storedt at −80℃ for research purposes only, not for conception. In summary, 1,349 male volunteers were recruited, of whom 841 provided semen samples, and 372 blood samples were collected during outpatient visits.
Flowchart of participant screening and enrollment.
Assessment of semen parameters
After a period of 2 to 7 days of abstinence, ejaculation was achieved through masturbation, and the semen was collected in a sterile, pre-weighed, wide-mouth plastic container. The semen sample was then placed in a 37 °C incubator and incubated until liquefaction occurred, or for a maximum of 1 h. The time taken to achieve complete liquefaction was recorded (< 1 h). Semen analysis was performed using computer-assisted sperm analysis techniques (CFT-9201, Jiangsu Ruiqi Life Science Co., Ltd., Jiangsu, China). The evaluation of semen parameters included basic semen parameters, sperm motility parameters and sperm morphological parameters. Sperm morphology was examined using a microscope. Following the analysis, the samples were stored at −80℃.
Genomic DNA extraction
Genomic DNA was extracted utilizing a commercial DNA extraction kit (Laifeng Biotechnology, Shanghai, China) in accordance with the manufacturer’s instructions. The extraction procedure comprised cell lysis, protein removal, DNA binding, washing, and elution. The extracted DNA was dissolved in sterile TE buffer (10 mM Tris-HCl, 1 mM EDTA, pH 8.0). The concentration and purity of the DNA were assessed using a microspectrophotometer (Thermo Fisher Scientific, USA). The A260/A280 ratios of all samples were confirmed to be between 1.8 and 2.0, indicating that the DNA quality was suitable for subsequent experiments. All DNA samples were stored at −80 °C until further analysis.
Measurement of the STL
Semen samples were incubated at 37 °C for subsequent analysis. Following the extraction of DNA from these samples, 536 were selected for STL detection. The relative telomere length of sperm DNA was measured using real-time PCR.Quantitative real-time PCR (qPCR) was conducted on a CFX96 Touch Real-Time PCR System (Bio-Rad, USA). The reaction mixture, totaling 20 µL, comprised of 10 µL of 2× SYBR Green Premix Pro Taq HS qPCR Kit (Accurate Biology, China), 0.4 µM of each forward and reverse primer, 20 ng of template DNA, and nuclease-free water to achieve the final volume. Each sample was analyzed in triplicate, and a no-template control (NTC) was included in each run to monitor for potential contamination.
The amplification protocol included an initial denaturation at 95 °C for 30 s, followed by 40 cycles of denaturation at 95 °C for 5 s and annealing/extension at 60 °C for 30 s, during which fluorescence signals were acquired. After amplification, a melting curve analysis was performed immediately conducted to verify the specificity of the amplicons by gradually heating the reaction from 65 °C to 95 °C with fluorescence measurements taken at every 0.5 °C increment.
The Sperm DNA phase was calculated using the formula 2-△△Ct, where △Ct is defined as Ct of the target gene minus Ct of the internal control, and △△Ct is the difference between △Ct and the average △Ct of the control group. The primers for telomere fragments are detailed in Table S1.
Gene polymorphisms analysis
Sequence data for the target gene SNP loci were retrieved from the public NCBI dbSNP database, and allele-specific primers were designed. All primers were synthesized by Sangon Biotech Co., Ltd. (Shanghai, China). The PCR was conducted in a 25 µL reaction volume containing 12.5 µL of 2× Taq PCR MasterMix (with loading dye, Cowin Biotech, China), 0.4 µM of each forward and reverse primer, 100 ng of template DNA, and nuclease-free water to achieve the final volume. Amplification was performed on an Applied Biosystems Veriti Thermal Cycler according to the protocol detailed in Table S2.
To verify the specificity and size of the amplicons, 5 µL of each PCR product was analyzed using 2% agarose gel electrophoresis and visualized under a UV gel imaging system. For genotyping, the PCR products were digested with appropriate restriction endonucleases (Thermo Fisher Scientific, China) selected based on the nucleotide variations at each SNP locus. The 15 µL digestion reaction consisted of 8 µL of PCR product, 1.5 µL of 10× restriction enzyme buffer, 5–10 units of the enzyme, and nuclease-free water. The reaction mixture was incubated at 37 °C for a minimun of 4 h or overnight to ensure complete digestion.
The resulting fragments were separated by electrophoresis on either a 3% agarose gel or a 10% polyacrylamide gel, utilizing a DL2000 DNA Marker as a size standard. The digested fragment patterns were visualized and recorded using a gel imaging system. The genotype of each subject was determined by comparing the observed fragment sizes and numbers with the expected restriction digestion patterns. The primer sequences and corresponding restriction enzymes utilized for each SNP are listed in Tables S3 and S4, respectively.
Statistical analysis
All statistical analyses were conducted using SPSS version 22.0 (IBM, Inc.,New York, USA) and R version 4.3.1 (2023-06-16 ucrt). The differences in semen parameters and relative telomere length between the two or three groups were assessed using either t-tests or non-parametric tests.Baseline information, semen quality parameters, and sperm relative telomere length for the study population were described using pre-conversion data. Descriptive results are presented as mean ± SD, No.(%) or M (P25, P75).Subsequently, after adjusting for age, body mass index (BMI), smoking, alcohol consumption, and other factors, multiple linear regression employed to examine test the relationship between semen parameters, relative telomere length and telomere-related gene polymorphisms. To explore the potential dose-response relationship between sperm telomere length and semen quality, a restricted cubic spline model was utilized. This model analysis included sperm telomere length as acontinuous variable while adjusting for age, BMI, smoking, and alcohol consumption. Furthermore, semen quality performance is divided into dichotomous outcomes of ‘normal’ or ‘abnormal’ according to WHO guidelines, with reference levels as follows: sperm forward motility rate (sperm motility ≥ 32%), total motility (sperm motility ≥ 40%), total sperm count (≥ 39 million), volume (≥ 1.5 ml) and concentration (≥ 15 million/ml) (WHO, 2010). If a participant’s value was below the corresponding reference value, they were classified as abnormal. A multivariate logistic regression model was employed to evaluate the relationship between STL and binary classification outcomes. We adjusted for age (continuous), BMI(continuous), smoking (categorical), and drinking status (categorical). The regression results are presented as coefficients and 95% confidence intervals (CIs), unless otherwise stated.
Results
Participant characteristics
As illstrated in Table 1, the mean age of the survey participants was 29.49 years, with an average BMI was 24.92. Among the respondents, 47.04% of the males had attained a secondary school education, followed by 27.39% of with a junior college education and 21.23% holding a bachelor’s degree or higher. Notably, 46.70% of male participants reported never having smoked, while 58.32% indicated that they never consumed alcohol. In terms of semen analysis, among the five parameters assessed—semen volume, total sperm count, sperm concentration, progressive motility, and total sperm motility— of the 1349 subjects, the percentages meeting the standards set forth in the 5th WHO reference guide-lines were 93.39%, 88.88%, 88.88%, 80.22%, and 86.02%, respectively. However, only 32.46% of the subjects exhibited normal sperm morphology.
Distributions of genetic polymorphism
The distribution of genotypes and alleles along with the Hardy-Weinberg equilibrium test results for telomere-related gene polymorphisms, are summarized in Table 2. The genotypic distributions of TERF1 rs3863242, POT1 rs1034794, TERT rs2735940, TERT rs2736098, and TERT rs2736109 conform to Hardy-Weinberg equilibrium (P > 0.05).In contrast, POT1 rs10250202, TERT rs2736100, and TERC rs2293607 do not conform to Hardy-Weinberg equilibrium (P < 0.05).
Effect of STL on semen parameters
The results of RCS analysis indicated that STL exhibited a nonlinear relationship with sperm concentration(P < 0.001) and total sperm count (P = 0.007). The RCS curve illustrated this nonlinearity, revealing two inflection points at 0.77 and 3.64. Specifically, STL was negatively correlated with sperm concentration when STL was less than 0.77 and greater than 3.64, while a positive correlaion was observed when STL fell within the range of 0.77 to 3.64.Similarly, the RCS curve demonstrated a nonlinearity between STL and total sperm count, with inflection points at 0.68 and 3.55. STL was negatively correlated with total sperm count when STL was below 0.68, whereas a positive correlation was noted within the range of 0.68–3.55 (Fig. 2, Figure S1).
Upon categorizing the population by STL level into quartiles, we found that the risk of abnormalities in total sperm count (OR = 0.37(0.15, 0.94), P = 0.037), sperm concentration (OR = 0.31 (0.11, 0.83), P = 0.020), progressive motility (OR = 0.27 (0.12, 0.61), P = 0.002), and total vitality percentage (OR = 0.19 (0.07, 0.51), P = 0.001) was significantly lower in the third quartile compared to the lowest quartile (Table S5, Fig. 3B).
Furthermore, when categorizing the population into two groups based on WHO semen parameter criteria (normal or abnormal), we observed that the STL in the normal group was significantly greater than that in the abnormal group across all parameters, including sperm concentration, total sperm count, progressive motility, and total vitality percentage. (P < 0.05)(Tables S6,Fig. 3A). We employed a linear regression model to analyze the relationship between relative telomere length and semen parameters.After adjusting for age, BMI, smoking and drinking habits, STL was found to be positively correlated with total sperm count (β = 0.159 (0.009, 0.309), P = 0.037) and sperm concentration (β = 0.177 (0.043, 0.312), P = 0.010). No significant correlation was identified with other semen parameters (P > 0.05) (Tables S7).
Illustrates the relationship between Ssperm Ttelomere Llength and the semen quality in male adult males, analyzed using restricted cubic splines.
Illustrates the relationship between sperm telomere length and semen parameters. (A) The Mann-Whitney U test was utilized to assess the differences in relative telomere length between groups with normal and abnormal seminal quality. STL denotes sperm telomere length.Statistical significance is indicated as follows: ns (P > 0.05), * (P < 0.05), and ** (P < 0.01) in comparedison to the normal group. (B) Adjusted odds ratios [95% confidence interval (CI)] for semen quality were calculated according to quartiles of relative telomere length levels, as included in the logistic regression analysis model. Relative telomere length was incorporated into the conditional logistic regression model, and the adjusted odds ratio (OR) was derived after controlling for confounding variables such as age, BMI, and smoking/drinking habits. STL: again refers to sperm telomere length.
Effect of gene polymorphisms on semen parameters
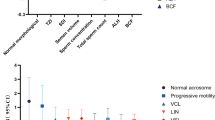
The non-parametric analysis of semen parameters across different genotypes revealed statistically significant differences in total sperm count among the three genotypes of POT1 rs10250202(AA, AC and CC). However no significant differences were observed in semen parameters for other polymorphic loci (Table S8). After adjusting for age, BMI, and smoking/drinking habits, we employed a linear regression model to analyze the relationship between gene polymorphisms and semen parameters. Our findings indicate that the mutant homozygous AA genotype of POT1 rs1034794 is negatively correlated with sperm forward motility (β=−9.76(−18.88,−0.64,)P = 0.036) and total motility (β=−11.80(−21.49,−2.10༉P = 0.017) when compared to the wild homozygous TT genotype. For POT1rs10250202, the AA genotype group, exhibited lower total sperm count, progressive motility and total vitality percentage compared to the CC genotype group (P < 0.05 for all). Additionally, the mutant homozygous CC and heterozygous TC genotypes of TERT rs2735940 were negatively correlated with STR, WOB and LIN when compared to the wild homozygous TT genotype (P < 0.05 for all). Conversely, the AA genotype group of TERT rs2736100 demonstrated significantly higher STR, WOB, and LIN compared to the GG group (P < 0.01 for all).Furthermore, the wobble (WOB) in the heterozygous AG genotype of TERT rs2736109 was higher than that in the GG group (β = 7.88(0.85,14.92) P = 0.028). No significant associations were found between TERF 1rs3843242, TERC rs2293607 and TERTrs 2736098 with semen quality parameter (Table S9-1, S9-2, S9-3, and Fig. 4).
After categorizing the population into two groups based on the WHO semen parameter criteria (normal or abnormal), a univariate logistic regression analysis was conducted to examine the relationship between genotype and semen parameters. As presented in Table S10, POT1 rs1034794 genotype AA (OR = 3.844 (1.353, 10.922) P = 0.012) was identified as a non-protective factor for progressive motility when compared to the genotype TT. Furthermore, in comparison to the genotype CC of POT1 rs10250202, genotype AA and AC were found to be non-protective factor for forward motility (ORAA=5.108(1.431, 18.235) PAA=0.012; ORAC=4.419(1.308, 14.930) PAC=0.017) and total viability (ORAA=4.932(1.065, 22.847) PAA=0.041; ORAC=4.419(1.086, 20.489) PAC=0.038).
Effect of gene polymorphisms in telomere-related pathway genes on semen parameters.
We analyzed the relationship between gene polymorphisms and semen parameters utilizing a linear regression model. All models adjusted for age, BMI, duration of abstinence,, smoking status and alcohol consumption.
Effect of gene polymorphisms on STL
Non-parametric tests revealed significant differences in the relative telomere length among the TERC rs2293607 genotypes (AA, GA, and GG).However, no significant differences were observed in relative telomere length across other polymorphic loci (see Table S11). The linear regression analysis of gene polymorphisms and relative telomere length is presented in Table 2. POT1 rs1034794 genotype AA exhibited a positive correlation with relative telomere length(β = 6.17 (1.89, 10.44) P = 0.005).Similarly, the TERC rs2293607 genotype AA also showed a was positive correlation with relative telomere length(β = 3.74 (0.11,7.38)) P = 0.044).
Discussion
Our study evaluated five semen parameters in 1,349 subjects: volume, total sperm count, sperm concentration, progressive motility rate, and total motility rate. According to the fifth edition of the World Health Organization’s reference guidelines, the proportion of subjects meeting the standards for these parameters was significantly higher, while the proportion of subjects with normal morphology was relatively lower. This indicates a significant and widespread decline in semen quality among the male population in the Shandong region. From 2008 to 2014, the semen volume, sperm concentration, progressive motility rate, and total sperm count in this group decreased by approximately 2.73%, 6.89%, 1.38%, and 9.84% respectively each year20. Additionally, the concentration and proportion of morphologically normal sperm in donors at a human sperm bank in Hunan Province, China, decreased from 68 × 10^6 per ml to 47 × 10^6 and from 31.8% to 10.8% respectively21.
In recent years, the influence of sperm telomere length (STL) on male fertility has garnered increasing attention. This study, for the first time, reveals a non-linear association between STL and semen parameters through a large sample analysis, while also exploring the role of telomere-related gene polymorphisms. Multiple studies have consistently demonstrated that men experiencing infertility, but without known structural abnormalities in the urinary system, exhibit shorter sperm telomere lengths compared to healthy controls3,8. Our research findings further indicate that across all parameters—including sperm concentration, total sperm count, forward motility rate, and total motility percentage—the STL in the normal group is significantly longer than that in the abnormal group. These results suggest that telomere length may serve as a diagnostic biomarker for male infertility. A meta-analysis has indicated a linear positive correlation between telomere length and semen parameters22. Additionally, a study by Kariati et al.23, which involved 73 male volunteers aged 31 to 52 years, observed a positive correlation between telomere length and sperm count. Furthermore, the crucial role of sperm telomere length in reproduction has been confirmed by animal studies, which noted a positive correlation between sperm telomere length and sperm motility, as well as membrane integrity in bulls, alongside a negative correlation with sperm malformations24.Through a linear regression model, our study corroborated previous research findings, demonstrating a positive correlation between telomere length and total sperm count as well as sperm concentration. Notably, while the association between STL and total sperm count was statistically significant in our model, the confidence interval of the effect estimate approached the null value (OR = 0.37 [0.15, 0.94], P = 0.037; β = 0.159 [0.009, 0.309], P = 0.037). This suggests that although the likelihood of a positive correlation and protective relationship is substantial, the strength of this effect in a clinical context may be limited. These findings are near the significance threshold and warrant careful interpretation. From a clinical perspective, these results indicate that STL is not a definitive biomarker for abnormal sperm count but may serve as a component within a broader multi-factorial framework. Its clinical utility may reside in risk stratification, enabling the identification of individuals with shorter telomeres (especially those in the lowest quartile) for more aggressive lifestyle interventions or earlier fertility assessments. However, the moderate effect size implies that STL alone is insufficient for clear diagnosis or prognosis, and its measurement should be considered alongside other established clinical parameters. Furthermore, we employed a restricted cubic spline (RCS) model to reveal a more complex non-linear relationship. When STL falls within the moderate length range, for instance, between 0.77 and 3.64, it is positively correlated with sperm concentration, and within the range of 0.68 to 3.55, it is positively correlated with total sperm count. This finding was further substantiated by the logistic regression results, which indicated that STL in the third quartile exhibited a clear protective effect on semen parameters. This suggests the existence of an optimal ‘healthy window’ for telomere length, rather than a simple linear relationship where longer telomeres are always better.
Oxidative stress can compromise the integrity of telomeres and disrupt telomerase activity, which is primarily responsible for maintaining telomere length25,26. Shortened telomeres may subsequently impede chromosome meiosis, leading to apoptosis during the first meiotic division, thereby adversely affecting the development of reproductive cells. Consequently, excessively short telomeres may result in a reduction in sperm quantity and semen concentration27. Our research supports this pathway, revealing that the STL of all abnormal semen parameter groups was significantly shorter than that of the normal group. However, this study also addresses the potential risks associated with excessively long telomeres. We hypothesize that overly long telomeres may lead to increased telomere fragility and the formation of abnormal structures28,29,30, which could counteract their normal functions and even have detrimental effects on sperm production. This offers a plausible explanation for the absence of a clear protective effect in the second and fourth quartiles.
Currently, the assessment of sperm quality using the WHO guidelines does not consider molecular genetic pathways, such as sperm telomere length, sperm DNA integrity, and sperm lipid peroxidation levels. Cells with shorter telomeres exhibit increased DNA fragmentation, which may contribute to reduced cell motility and vitality. Our research has also revealed a strong positive correlation between sperm telomere length and both sperm motility and the percentage of forward-moving sperm. This suggests that these factors may be interconnected in relation to diminished sperm quality and could also be implicated in poor reproductive outcomes. Therefore, alongside conventional sperm parameters, the quality of sperm and its fertilization capability may also hinge on molecular genetic analysis results. Meta-analysis data indicate that sperm telomere length (STL) has a positive linear correlation with semen parameters. The optimal critical value for diagnosing male infertility using STL is 1.0, with a sensitivity and specificity of 80%. This suggests that telomere length may serve as a potential biomarker for assessing sperm quality and could be utilized as an additional sperm parameter, offering new insights into the diagnosis of male infertility and serving as a foundation for clinical research and practice.
At the genetic level, we are the first to measure the polymorphisms of telomerase-related genes to assess their relationship with STL and semen quality. We first conducted a Hardy-Weinberg equilibrium (HWE) test for the genotype distribution. The genotype distributions of TERF1 rs3863242, POT1 rs1034794, TERT rs2735940, TERT rs2736098, and TERT rs2736109 were in accordance with HWE (P > 0.05), indicating that the data quality of these loci was reliable and the population was genetically stable at these loci, not affected by significant confounding factors such as typing errors, selection, or population stratification. In contrast, significant deviations from HWE were observed for POT1 rs10250202, TERT rs2736100, and TERC rs2293607 (P < 0.05). This requires caution when interpreting the association results of these specific variants. We speculate that the deviations observed in these specific telomere maintenance-related genes may suggest their association with potential disease susceptibility or biological processes31,32. Additionally, we found that the purity of the mutant AA was positively correlated with STL compared to the wild-type POT1 rs1034794 purity. Furthermore, we discovered that the mutant AA purity of POT1 rs1034794 was negatively correlated with the forward motility and total motility of sperm compared to the wild-type purity TT. Similarly, the AA genotype of POT1 rs10250202 was associated with lower total sperm count and motility. These results collectively indicate that certain variations in the POT1 gene may disrupt telomere length homeostasis, leading to telomere dysfunctionally elongated, ultimately damaging semen quality28,33,34. This is highly consistent with the non-linear effect of STL that we observed, where specific gene mutations drive telomere elongation beyond the optimal range, which is also detrimental to male fertility. Moreover, our analysis of TERT gene polymorphisms further reveals the complexity of the telomere maintenance system. For example, the AA genotype of rs2736100 was associated with higher STR, WOB, and LIN values, while the TC/CC genotype of rs2735940 was associated with lower these parameters. These findings suggest that different TERT variations may affect sperm functional dynamics through regulating telomerase activity in different ways35.
This study examines the relationship between single nucleotide polymorphisms and semen quality as well as relative telomere length. It provides the first comprehensive results of polymorphic studies and reveals and a nonlinear relationship between sperm telomere length and semen quality. These findings may serve as a valuable tool for assessing the fertilization potential of sperm, enhancing diagnostic accuracy, preventing male infertility, and offering insights into the success rates of assisted reproduction. However, our study has several limitations. This study was designed as a single-center investigation with a limited sample size, which may affect the generalizability of the results. More importantly, the cross-sectional study design inherently limits the ability to infer causal relationships, as it cannot clearly establish the temporal sequence between variables or the potential causal pathways. Furthermore, potential confounding factors, such as environmental exposures and epigenetic influences, were not fully considered. Additionally, we did not completely exclude other genetic abnormalities that could contribute to semen anomalies, and the single-center design may introduce selection bias. Future research should focus on conducting large-scale, multi-center longitudinal studies or prospective cohorts to further validate the current findings and explore the underlying mechanisms in greater depth.
Conclusions
Our findings reveal a non-linear relationship between sperm telomere length (STL) and both sperm concentration and total sperm count, highlighting STL as a potential biomarker for semen quality. Furthermore, polymorphisms in telomere-related genes, such as POT1 and TERT, were found to be associated with sperm parameters and STL, suggesting that genetic variations in the telomere pathway may influence semen quality through their effects on telomere length. Future studies should investigate the molecular mechanisms by which telomere length and associated genes regulate sperm development and evaluate the clinical utility of STL in male fertility diagnostics.
Data availability
Contact the corresponding author for obtaining.
References
Datta, J. et al. Prevalence of infertility and help seeking among 15 000 women and men. Hum. Reprod. 31 (9), 2108–2118. https://doi.org/10.1093/humrep/dew123 (2016).
Concepción-Zavaleta, M. et al. Assessment of hormonal status in male infertility. An update. Diabetes Metab. Syndr. 16 (3), 102447. https://doi.org/10.1016/j.dsx.2022.102447 (2022).
Randell, Z. et al. Sperm telomere length in male-factor infertility and reproduction. Fertil. Steril. 121 (1), 12–25. https://doi.org/10.1016/j.fertnstert.2023.11.001 (2024).
Agarwal, A. et al. Male infertility. Lancet 397 (10271), 319–333. https://doi.org/10.1016/S0140-6736(20)32667-2 (2021).
Esteves, S. C. Evolution of the world health organization semen analysis manual: where are we? Nat. Rev. Urol. 19 (7), 439–446. https://doi.org/10.1038/s41585-022-00593-2 (2022).
Schmidt, J. C. & Cech, T. R. Human telomerase: biogenesis, trafficking, recruitment, and activation. Genes Dev. 29 (11), 1095–1105. https://doi.org/10.1101/gad.263863.115 (2015).
Engin, A. B. & Engin, A. The connection between cell fate and telomere. Adv. Exp. Med. Biol. 1275, 71–100. https://doi.org/10.1007/978-3-030-49844-3_3 (2021).
Darmishonnejad, Z. et al. Relationship between sperm telomere length and sperm quality in infertile men. Andrologia 52 (5), e13546. https://doi.org/10.1111/and.13546 (2020).
Liu, S. Y. et al. Association study of telomere length with idiopathic male infertility. Yi Chuan. 37 (11), 1137–1142. https://doi.org/10.16288/j.yczz.15-267 (2015).
Thilagavathi, J. et al. Analysis of sperm telomere length in men with idiopathic infertility. Arch. Gynecol. Obstet. 287 (4), 803–807. https://doi.org/10.1007/s00404-012-2632-8 (2013).
Berneau, S. C. et al. Associations of sperm telomere length with semen parameters, clinical outcomes and lifestyle factors in human normozoospermic samples. Andrology 8 (3), 583–593. https://doi.org/10.1111/andr.12734 (2020).
Gentiluomo, M. et al. Telomere length and male fertility. Int. J. Mol. Sci. 22 (8), 3959. https://doi.org/10.3390/ijms22083959 (2021). Published 2021 Apr 12.
Artandi, S. E. & DePinho, R. A. Telomeres and telomerase in cancer. Carcinogenesis 31 (1), 9–18. https://doi.org/10.1093/carcin/bgp268 (2010).
Dahse, R., Fiedler, W. & Ernst, G. Telomeres and telomerase: biological and clinical importance. Clin. Chem. 43 (5), 708–714 (1997).
Yan, L. et al. Genetic variants in telomerase reverse transcriptase (TERT) and telomerase-associated protein 1 (TEP1) and the risk of male infertility. Gene 534 (2), 139–143. https://doi.org/10.1016/j.gene.2013.11.008 (2014).
Pech, M. F. et al. High telomerase is a hallmark of undifferentiated spermatogonia and is required for maintenance of male germline stem cells. Genes Dev. 29 (23), 2420–2434. https://doi.org/10.1101/gad.271783.115 (2015).
Lee, H. W. et al. Essential role of mouse telomerase in highly proliferative organs. Nature 392 (6676), 569–574. https://doi.org/10.1038/33345 (1998).
Wang, L. et al. Dual roles of TRF1 in tethering telomeres to the nuclear envelope and protecting them from fusion during meiosis. Cell. Death Differ. 25 (6), 1174–1188. https://doi.org/10.1038/s41418-017-0037-8 (2018).
Zhu, X., Fu, H., Sun, J., Di, Q. & Xu, Q. N6-methyladenosine modification on Hmbox1 is related to telomere dysfunction in DEHP-induced male reproductive injury. Life Sci. 309, 121005. https://doi.org/10.1016/j.lfs.2022.121005 (2022).
Wang, L. et al. Decline of semen quality among Chinese sperm bank donors within 7 years (2008–2014). Asian J. Androl. 19 (5), 521–525. https://doi.org/10.4103/1008-682X.179533 (2017).
Huang, C. et al. Decline in semen quality among 30,636 young Chinese men from 2001 to 2015. Fertil. Steril. 107 (1), 83–88e2. https://doi.org/10.1016/j.fertnstert.2016.09.035 (2017).
Yuan, Y. et al. Sperm telomere length as a novel biomarker of male infertility and embryonic development: A systematic review and meta-analysis. Front. Endocrinol. (Lausanne). 13, 1079966. https://doi.org/10.3389/fendo.2022.1079966 (2023). Published 2023 Jan 11.
Cariati, F. et al. Investigation of sperm telomere length as a potential marker of paternal genome integrity and semen quality. Reprod. Biomed. Online. 33 (3), 404–411. https://doi.org/10.1016/j.rbmo.2016.06.006 (2016).
Iannuzzi, A. et al. Evaluation of bovine sperm telomere length and association with semen quality. Theriogenology 158, 227–232. https://doi.org/10.1016/j.theriogenology.2020.09.019 (2020).
Aeby, E. et al. Peroxiredoxin 1 protects telomeres from oxidative damage and preserves telomeric DNA for extension by telomerase. Cell. Rep. 17 (12), 3107–3114. https://doi.org/10.1016/j.celrep.2016.11.071 (2016).
He, Y. et al. Structures of telomerase at several steps of telomere repeat synthesis. Nature 593 (7859), 454–459. https://doi.org/10.1038/s41586-021-03529-9 (2021).
Hemann, M. T. et al. Telomere dysfunction triggers developmentally regulated germ cell apoptosis. Mol. Biol. Cell. 12 (7), 2023–2030. https://doi.org/10.1091/mbc.12.7.2023 (2001).
DeBoy, E. A. et al. Familial clonal hematopoiesis in a long telomere syndrome. N Engl. J. Med. 388 (26), 2422–2433. https://doi.org/10.1056/NEJMoa2300503 (2023).
Wang, C. et al. ILF3 safeguards telomeres from aberrant homologous recombination as a telomeric R-loop reader. Protein Cell. 15 (7), 493–511. https://doi.org/10.1093/procel/pwad054 (2024).
Rocca, M. S. et al. Sperm telomere length as a parameter of sperm quality in normozoospermic men. Hum. Reprod. 31 (6), 1158–1163. https://doi.org/10.1093/humrep/dew06 (2016).
Wittke-Thompson, J. K., Pluzhnikov, A. & Cox, N. J. Rational inferences about departures from Hardy-Weinberg equilibrium. Am. J. Hum. Genet. 76 (6), 967–986. https://doi.org/10.1086/430507 (2005).
Xu, J. et al. Positive results in association studies are associated with departure from Hardy-Weinberg equilibrium: hint for genotyping error? Hum. Genet. 111 (6), 573–574. https://doi.org/10.1007/s00439-002-0819-y (2002).
Baumann, P. & Price, C. Pot1 and telomere maintenance. FEBS Lett. 584 (17), 3779–3784. https://doi.org/10.1016/j.febslet.2010.05.024 (2010).
Takasugi, T. et al. Pot1b -/- tumors activate G-quadruplex-induced DNA damage to promote telomere hyper-elongation. Nucleic Acids Res. 51 (17), 9227–9247. https://doi.org/10.1093/nar/gkad648 (2023).
Sheng, X. et al. TERT polymorphisms modify the risk of acute lymphoblastic leukemia in Chinese children. Carcinogenesis 34 (1), 228–235. https://doi.org/10.1093/carcin/bgs325 (2013).
Acknowledgements
We would like to express our sincere gratitude to the participants, CDC professionals, doctors, and nurses, and all those involved in this study.
Funding
This work was supported by Shenzhen Park Project (Contract No. HZQSWS-KCCYB-2024031) of Hetao Shenzhen-Hong Kong Science and Technology Innovation Cooperation Zone.The National Natural Science Foundation of China (82273663). Key Research Projects of Higher Education Institutions in Henan Province (26B330005). Guangzhou Basic and Applied Basic Research Foundation (2023A03J0240), Natural Science Foundation of Guangdong Province (2022A1515012443).Shenzhen portion of the Hetao Shenzhen-Hong Kong Science and Technology Innovation Cooperation Zone(program grant # HZQSWS-KCCYB-2024031.)The funders were not involved in the study design, data collection, analysis and interpretation, and manuscript writing.
Author information
Authors and Affiliations
Contributions
Conceptualization, X.J. and J.C.; methodology, G.Z. and J.C.; software, S.Z.; validation, K.F., J.C. and J.L.; formal analysis, H.J.; investigation, X.Q. and J.C.; resources, S.Z.; data curation, J.C.; writing—original draft preparation, X.J. and S.Z.; writing—review and editing, J.C. and S.Z.; visualization, L.T and Y.Y.; supervision, A.U.; project administration, Z.G.; funding acquisition, Z.G.and J.L.and A.W. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Institutional review board statement
Study procedures were performed in accordance with the Declaration of Helsinki ethical principles for medical research involving human subjects. The study was approved by the Ethics Committee of Xinxiang Medical University, and written informed consent was obtained from all participants (Reference Number:XXLL-20170311).
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jia, X., Cao, J., Zhang, S. et al. Effect of telomere length and related gene polymorphism in signaling pathway on semen quality. Sci Rep 16, 6575 (2026). https://doi.org/10.1038/s41598-025-34054-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-34054-8