Abstract
Probiotic supplements have the potential to improve the gut flora and enhance nutritional efficacy and blood indices. In this study, sunflower seed meal based diet (SFM) supplemented with a commercial probiotic (protexin®) was examined to evaluate the effects on the survival, growth, digestibility, antioxidant status, carcass and hematology of Cirrhinus mrigala fingerlings. Fish were subjected to a 12-week feeding regimen to examine the efficacy of probiotics. Seven diets with different probiotic doses (0, 0.5, 1, 1.5, 2, 2.5, and 3 g kg−1) were prepared and randomly assigned to 21 tanks, each stocked with 15 fingerlings, resulting in a total of 315 fish utilized forthe experiment. Analysis showed that fish treated 2 g kg−1 probiotics level exhibited a remarkable 100% survival rate. The results indicated that probiotics (2 g kg−1) supplementation revealed notable differences in weight gain (19.76 g), weight gain% (283.15%), FCR (1.31) and SGR (1.49% d−1) than other probiotic concentrations and control. The dietary administration of 2 g kg−1 probiotics to C. mrigala resulted in significant improvements in digestibility, with values of 70.52% for protein, 70.30% for gross energy, and 73.71% for fat. Carcass composition also showed notable enhancements, with fat, ash, protein, and moisture content of 4.78%, 2.03%, 18.08%, and 75.11%, respectively. Overall findings indicated that adding 2 g kg⁻¹ probiotics showed improved hematological indices, such as increased red blood cell, hemoglobin and white blood cell count and enhanced antioxidant enzyme activity. Mineral analysis revealed optimal levels at 2 g kg−1 probiotics: Ca (72.57%), Na (73.08%), K (71.45%), P (74.70%), Fe (67.43%), Cu (63.62%), Mg (61.98%), and Al (62.18%). To sum up, the dose of 2 g kg−1 probiotics supplementation has a profoundly positive impact on growth, digestibility, health, and hematology in C. mrigala.
Similar content being viewed by others
Introduction
Probiotics are living bacteria that help animals maintain a healthy microbial balance inside their bodies, which is advantageous to their overall health. These bacteria include genus Bacillus, Clostridium, Staphylococcus, Lactobacillus and Aeromonas species, etc. Protexin, a commercially available probiotic, is a polymicrobial blend comprising a diverse array of microbes, including lactic acid bacteria (Lactobacillus plantarum, L. acidophilus), yeasts (Candida parapsilosis) and fungi (Aspergillus oryzae). Moreover, it is a multi-species probiotic which offers greater diversity in antimicrobial compounds and enhanced gut mucus adhesion, likely to have a more significant impact as feed supplements compared to mono-species probiotics1. The use of probiotics in aquaculture has been proven to have numerous benefits, including enhanced fish health, immunity, growth rate, gut integrity, and disease resistance2. Additionally, they boost digestive enzyme function, facilitating better nutrient absorption and energy allocation for fish development, providing a sustainable alternative to antibiotics in the industry3. Furthermore, multi-strain probiotics have been shown to enhance growth, hematology, intestinal health, digestibility, and immunity in various fish species, including Paralichthys olivaceus4, Cirrhinus cirrhosus5, and Acipenser baerii6.
Cirrhinus mrigala, commonly referred to as “mori”, is a bottom-feeding major carp that inhabits the fresh waters of Southeast Asia. Its commercial value in polyculture is well-documented, and the fish itself is prized for both its flavor and economic worth. Indian major carps (IMC) account for 8.7% of global carp production and Asian production accounts for 1 million metric tonnes annually7. Intensive farming, unregulated resource use, and lack of farmer expertise have increased disease outbreaks in C. mrigala (mrigal). However, effective health management strategies, including the application of probiotics as functional diet additives, have been developed to mitigate these impacts8. Farmed fish are an exceptional source of protein and micronutrients (vitamins and minerals)9,10,11. However, the high expense of aqua feed, which constitutes over 75% of aquaculture production costs, drives the market for mass production12. The industry relies on the production of affordable fish feed that adheres to nutritional standards. Although, fish meal (FM) and fish oil (FO) are unsuitable to fulfill this need because of their limited availability, unsustainable supply, high demand and expensive price9,13. Hence alternative, cheap plant by-products are needed to support and maintain aquaculture14.
One of the finest alternative plant by-products is sunflower seed meal (SFM), which boasts high contents of crude fiber, unsaturated fatty acids, and crude protein (about 50%)15. As a matter of fact, it is a leftover product from the oil extraction process16. This diet is characterized by high palatability, nutritional density, and accessibility, making it an optimal fish feed option17. After soybean (SBM), cottonseed (CSM), and canola meals (CM), it is the fourth-largest source of dietary protein worldwide18. This meal is well-liked because, in comparison to all other plant by-products, it has lower concentration of ANFs (anti-nutritional factors). Moreover, the high fibre content in SFM negatively impacts its nutrient availability19. These problems (ANFs and high fibre content) in the aquaculture sector have prompted researchers to look at additives like probiotics. Because probiotics mitigates these contents in plant-based ingredients by producing phytase, degrading oxalates, enhancing enzyme activity, altering gut microbiota, and binding ANFs, thereby improving nutrient bioavailability and digestive efficiency20,21.
This study examined the efficacy of supplementing a sunflower seed meal-based diet with a commercial probiotic (Protexin) on the health, growth, and efficiency of C. mrigala, providing a cost-effective and sustainable alternative to traditional fish feeds.
Materials and methods
This work was done at the GC University Faisalabad, Punjab, Pakistan. This region is located between the longitudes 73.0707° East and 31.4166° North.
Ethical statement
This research was conducted with utmost respect for ethical standards and in strict adherence to relevant laws and guidelines. The trial protocols received approval from the Animal Ethics Committee of GC University Faisalabad, ensuring compliance with ARRIVE (Animal Research Reporting of In Vivo Experiments) guidelines.
The setting of feeding trial for fish
For the purpose of culturing Cirrhinus mrigala (average weight: 6.99 ± 0.7 g/fish; age: about 2 months), they were obtained from the local Fish Hatchery, Faisalabad. Soaking fish in a 0.5% saline solution (0.5 g NaCl/100 mL water) for 1–2 min was sufficient to eliminate ectoparasites and prevent fungal infections. This solution serves as a therapeutic agent, offering antibacterial, anesthetic, stress-reducing, and osmoregulatory benefits. Alternatively, other treatments like quick lime, potassium permanganate, and antibiotics can be employed22. Before the start of the experiment, acclimatization of fingerlings was done for 2 weeks in 70 L V-shaped specially assembled water tanks to allow fish to adjust to the experimental conditions and reduce stress. They were given basal feed (control diet or SFM-based diet) once per day throughout this duration. Throughout the study, continuous aeration via capillary method maintained optimal dissolved oxygen levels in all experimental tanks. Water quality parameters were rigorously controlled within precise ranges: temperature (25.8–29.6 °C), pH (7.5–8.5), and dissolved oxygen (DO: 5.8–7.4 mg/L). These parameters were monitored using calibrated instruments, specifically a thermometer, Jenway 3510 pH meter, and Jenway 970 dissolved oxygen meter, ensuring accurate and reliable data collection.
Feed ingredients and diet formation
Sunflower seeds and other ingredient components were sourced from the feed market and then used to formulate feed pellets. The sunflower seeds were defatted through a two-step process involving air drying followed by mechanical pressing. The defatted seeds were ground into a fine texture, passing through a 0.3 mm sieve, to produce the SFM. The standard chemical analyses (Table 1) were performed on the feed components before they were used to create the experimental diets. The test diets were supplemented with 1% chromic oxide (Cr2O3) as a non-digestible marker and 7.5% fish oil. Seven diets were created: one without probiotics (as a control/ 0 gkg−1) and six with graduated probiotic levels (0.5–3 g kg−1) (A multi-strain Probiotics: Protexin® CFUg−1 = 2 × 109, England) (Table 2). In this study, protexin referred to a multi-strain probiotic blend comprising Streptococcus faecium, Lactobacillus plantarum, L. bulgaricus, Bifidobacterium bifidum, Aspergillus oryzae, L. acidophilus, and Candida pintolopesii. The probiotics were mixed in 100 mL of distilled water (H2O) and then sprayed uniformly over the diets to achieve the desired concentrations23. A control diet, devoid of probiotics spraying, was prepared using the same methodology and ingredients as the experimental diets. To minimize probiotic leaching during immersion, wheat flour coating was applied to the feed pellets in our study, thereby enhancing retention and efficacy of probiotics on fish health24. The dietary components were mixed for 5 min using a mechanical mixer, then blended with water to develop a uniform dough. After that, the dough was extruded through an electric extruder, producing noodle-shaped pellets of consistent texture and composition. All diets were oven-dried (105 degrees for a day) and then preserved in a refrigerated environment at a temperature of 4 °C.
Bacterial colonization and viability in fish intestines were assessed using Kim and Austin’s25 modified method. Specifically, 0.1 g intestinal contents were homogenized in PBS, diluted (10−5), spread-plated, and incubated at room temperature for 48 h. Bacteria were identified based on colonial and morphological characteristics.
Sampling plan and feeding routine
The fingerlings received their formulated diets at a feeding rate of 4% of their live wet weight, administered in two equal portions daily, at 8 am and 4 pm. The weight of fingerlings was recorded bi-weekly, enabling adjustments to feeding rates in response to growth. After an hour of feeding, any food that wasn’t consumed, gathered and dried in an oven at 105 °C for 24 h and weighed using an electronic balance for further analysis of feed intake and feed conversion ratio (FCR). Feces were collected using siphoning methodology, employing fecal collection tubes and valves. Following each feeding session, residual feed was flushed out through valves, and tanks were replenished with fresh water to remove residual particles. Subsequently, fecal waste was extracted from the collection tubes. To maintain nutrient integrity, fecal material was carefully collected, dried at 65 °C in an oven for 24 h and the dried samples were then stored in airtight containers at −20 °C to maintain sample integrity. To minimize nutrient leaching, feces were collected shortly after defecation26.
Survival rate evaluation
The survival rate is used to measure fish’s ability and tolerance for sustaining its life. Fish in each tank were monitored daily to record their survival and death rates using the following formula.
Growth evaluation
Each tank (three tanks per treatment) of water had fifteen C. mrigala fingerlings. Fingerlings were weighed collectively, with careful handling to minimize stress. The growth performance was assessed by measuring initial and final weights, and calculating standard growth metrics such as SGR (specific growth rates), FCR (feed conversion ratio), and WG (weight gain). The standard formulae was used to calculate the productivity of fish population27.
Where FW, IW, WG, and FI are final weight, initial weight, weight gain and feed intake, respectively.
Carcass composition evaluation
This experiment was conducted for 12 weeks, and then feed, feces, and whole-body fish samples were pulverized and homogenized using a mortar and pestle before analysis and assessed using established procedures28. To avoid cross-contamination, personal protective equipment (gloves, lab coats), regular surface sanitation, laminar flow/biosafety cabinets, immediate sealing and storage and equipment (scoops, siphons, pipettes) were used29. A standardized oven-drying method, involving heating at 105 °C for 12 h, was used to evaluate the moisture content of the test diets and feces. The Kjeldahl technique and the petroleum ether extraction in a Soxhlet extractor were used to estimate the amount of crude protein and crude fat, respectively. Samples were burned in an electric furnace (Naberthern B170) at 650oC for 12 h to assess their ash composition.
Hematology assessment
For blood sampling, three fish per tank (9 fish/triplicates) were taken. 1–2 mL blood sample was collected from each fish using sterilize syringes and stored in EDTA anticoagulant tubes. These samples were taken from the caudal vein after anesthesia was induced using a clove oil solution (50 mg/L). Hematological parameters assessed included packed cell volume (PCV) using micro-hematocrit, and white blood cell (WBC), platelet (PLT), and red blood cell (RBC) counts using a Neubauer counting chamber. The blood samples were centrifuged at 12,000 rpm for 5 min and hematocrit values were determined using a microhaematocrit reader. However, blood samples were diluted 1:100 in Hayem’s solution and then counted using microscopy at 400× magnification for RBC, PLT and WBC30. Hemoglobin (Hb) concentration was determined via cyanmethemoglobin spectrophotometric method as outlined by Wedemeyer and Yasutake31. In this protocol, Additionally, mean corpuscular volume (MCV), corpuscular hemoglobin concentration (MCHC) and mean corpuscular hemoglobin (MCH) were calculated as described by Khalid et al.26.
Nutrient digestibility
The nutrient digestibility of protein, fat and gross energy was evaluated by calculating the digestibility coefficients (ADC%) for feed and feces samples, determined using the standard formula as outlined by Khalid et al.26.
The concentration of chromic oxide, an inert digestibility marker, in feed and feces samples was quantified by spectrophotometric analysis at 350 nm after perchloric acid oxidation, based on the method outlined by Divakaran et al.32.
Mineral digestibility
Mineral concentrations were determined using Atomic Absorption Spectrophotometry (AAS; Hitachi UV-VIS 2001 spectrophotometer, US) after appropriate dilution, with calibrated standards prepared under standard conditions28,33. The samples were digested with a precisely formulated acid blend of nitric acid, sulfuric acid, and perchloric acid (10:0.5:2, v/v) prior to mineral analysis. AAS was employed to quantify the levels of key minerals, namely calcium, copper, phosphorus, magnesium, iron and aluminium. Sodium and potassium levels were measured using flame photometry such as firstly atomizing a sample containing elements like sodium or potassium into a fine mist, burning it in a flame to produce element-specific light, filtering the light, and measuring its intensity with a photoelectric cell sensor. The resulting electrical signal, displayed on a galvanometer, allows researchers to quantify the element after calibration34.
Phosphorus color reagent preparation involved dissolving ascorbic acid (10 g) and ammonium molybdate (1.25 g) in distilled water to create Solutions A and B, respectively. Combining 1 part Solution B with 5 parts Solution A yielded the reagent. Phosphorus analysis entailed adding 0.5 mL reagent to 1 mL sample, incubating at 40 °C for 1 h. The calibration curves were generated using phosphorus standard solutions. The phosphorus was then quantified calorimetrically at 720 nm, following the protocol by the Association of Official Analytical Chemists28,35.
Serum antioxidant enzyme
The blood samples (1–2 mL/fish) were drawn from caudal vein of fish using a 1 mL heparinized syringe. Fingerlings underwent gentle anesthesia using a 50 mg/L of clove oil solution, minimizing stress and promoting welfare. The precise species-specific dosage is essential to prevent adverse reactions36. The samples were promptly transferred to Eppendorf tubes and refrigerated at 4 °C for 24 h before further processing. After centrifugation (3000 × g, 10 min), the serum was isolated and preserved at −80 °C for further analysis. The activities of enzymes, such as superoxide dismutase (SOD), glutathione peroxidase (GPx) and catalase (CAT), were measured spectrophotometrically, adhering to established laboratory protocols37.
Statistical analysis
Finally, the data were subjected to a One-way Analysis of Variance (ANOVA)38 and Tukey’s Honesty Significant Difference Test (HSD) was used to analyze the differences between groups, and a p-value of less than 0.05 was considered statistically significant39. Graphical presentations were created using IBM’s statistical program, SPSS version 23.
Results
Growth parameters
Growth performance of C. mrigala improved substantially (P < 0.05) with probiotics supplemented SFM-based diets compared to the control group (Table 3). The highest survival rate (100%) was recorded at probiotic supplementation level of 2 g kg−1. In contrast, the control diet exhibited the lowest survival rate (91%). Maximum WG (19.76 g) of C. mrigala fingerlings was recorded when fed 2 g kg−1 probiotics supplement, closely followed by those receiving 1.5 g kg−1 probiotics. The minimum WG (16.24 g) was observed at control diet which was non-significant (P > 0.05) with 0.5 and 3 g kg−1 level-based test diets. Fingerlings fed the probiotic supplementation level of 2 g kg−1 level showed maximum weight gain% (283%) which was statistically similar (P > 0.05) to the values found in 1 (263.5%), 1.5 (272.6%) and 2.5 (264.3%) g kg−1 probiotics supplementation. Alternatively, the lowest weight gain% (232.56%) was observed in fingerlings fed the control. The lowest FCR (1.31) and highest SGR (1.49) values were achieved by fingerlings fed the diet having 2 g kg−1 probiotics supplement. Conversely, fish fed the control exhibited the highest FCR (1.78) and lowest SGR (1.34). According to quadratic regression analysis, the optimal levels of survival rate, weight gain, FCR and SGR for C. mrigala containing SFM diets were occurred at a probiotics dose of 2 g kg−1, with corresponding values of 1.79, 1.67, 1.93, and 1.67, respectively (Fig. 1).
Quadratic relationship between (A) weight gain (%), (B) feed intake (g), (C) FCR, (D) SGR of Cirrhinus mrigala (mori) fed with probiotics level-based diets.
Whole-body composition
In the Table 4, the findings of body composition revealed that C. mrigala fed on SFM based diet with supplementation of probiotics display a substantial difference (P < 0.05) in carcass composition than control diet without probiotics. The optimal nutrient composition in fish carcass was attained at a probiotic inclusion rate of 2 g kg−1, followed closely by the diet containing 1.5 g kg−1 probiotics supplementation. In contrast, minimum crude protein, ash and moisture contents were found when fed the control followed by 0.5 g kg−1 probiotic level-based diet. In the context of this feeding trial, there was a notable reduction (P < 0.05) in crude fat content of diet having 2 g kg−1 probiotics among all the probiotics supplemented experimental diets and control diet.
Nutrient digestibility
Analyzed nutrients% in feces and ND (%) coefficients (ADC%) of C. mrigala fed probiotics supplemented diets are significantly different (P < 0.05) as shown in Table 5; Figs. 2 and 3. It is obvious from the results that the lowest nutrient excretion in feces occurred in 2 g kg−1 of probiotics diet followed by 2.5 g kg−1 probiotic level based diet. The highest ADC% was observed at the 2 g kg⁻¹ level, indicating enhanced nutrient utilization compared to other levels. According to quadratic regression analysis, the optimum ADC% (GE, CP and EE) of C. mrigala for SFM based test diets were found at 1.79, 1.99 and 1.88 g kg−1 levels, respectively (Fig. 3). It was found that except for the 0.5 and 3 g kg−1 levels, probiotics supplemented diets did not result in a significant (P < 0.05) improvement in crude protein digestibility relative to the control diet.
Quadratic relationship of nutrient digestibility in feces of Cirrhinus mrigala (mori) fed probiotics level based diets (A) Protein (%), (B) Gross energy (g) and (C) Fat (%).
Quadratic relationship of nutrient digestibility ADC% of Cirrhinus mrigala (mori) fed probiotics level based diet (A) Protein (%), (B) Gross energy (g) and (C) Fat (%).
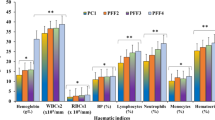
Hematological indices
Probiotics supplementation significantly (P < 0.05) enhanced the hematological indices of C. mrigala (Table 6). It was obvious that greatest values of RBCs, WBCs, PLT, Hb and Ht were found when C. mrigala fed with 2 g kg−1 probiotic level with SFM based diet. In contrast, these values differed significantly (P < 0.05) from other test diets supplemented with probiotics. The highest PCV (27%), MCH (63 pg) and MCV (197 fl.) values were found when fingerlings fed 2.5 g kg−1 probiotics. Conversely, the control achieved the lowest MCHC (101%), MCH (30 pg) and MCV (51 fl.) values. According to quadratic regression analysis, the highest values for RBCs and Hb in C. mrigala fed SFM diets were achieved at probiotic supplementation levels of 1.91 g kg−1 and 1.87 g kg−1, respectively, as illustrated in Figs. 4 and 5.
Quadratic relationship of hematological indices of Cirrhinus mrigala (mori) fed probiotics level based diet (A) RBCs, (B) Hb, (C) WBC and (D) PLT.
Quadratic relationship of hematological indices of Cirrhinus mrigala (mori) fed probiotics level based diet (A) MCH, (B) MCV, (C) PCV and (D) MCHC.
Mineral digestibility
The results of mineral analysis of feces and body are significant between each diet as shown in Tables 7 and 8; Figs. 6 and 7. The highest digestibility rates of essential minerals, including Ca (73%), Na (73%), K (71%), and P (75%), were shown when fish were fed a diet containing 2 g kg−1 probiotics. Whereas highest digestibility of Fe (69%), Cu (65%) and Al (65%) was noted at 2.5 g kg−1 probiotics dose followed by (Fe 67%, Cu 64% and Al 62%) the test diet V. Moreover, Mg (62%) exhibited a high digestibility and absorption rate when fish fed on 1.5 g kg−1 dose followed by the C. mrigala fingerlings fed on 2 g kg−1 probiotics dose. Minimum mineral digestibility was observed in C. mrigala fed the control diet.
Quadratic relationship of minerals (%) (A) Ca, (B) K, (C) Na, (D) Mg, (E) Fe, (F) P, (G) Cu and (H) Al in feces of Cirrhinus mrigala (mori) fed probiotics supplementation.
Quadratic relationship of mineral digestibility (%) (A) Ca, (B) K, (C) Na, (D) Mg, (E) Fe, (F) P, (G) Cu and (H) Al of Cirrhinus mrigala (mori) fed probiotics level based diet.
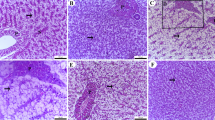
Antioxidant enzyme activity
Figure 8 shows that among all probiotic levels, the diet incorporated with 2 g kg−1 probiotics was the only treatment that significantly enhanced the activities of antioxidant enzymes, i.e., SOD, CAT, and GPx (P < 0.05) . However, all probiotic supplementation levels exhibited a significant increase (P < 0.05) in antioxidant enzyme activity, with varying degrees of improvement compared to the control diet.
Probiotic-induced benefits in antioxidant enzyme activities (%) in Cirrhinus mrigala (mori).
Discussion
Probiotics are beneficial microbes that improve fish welfare when administered in sufficient amounts, but require species-specific compatibility to colonize the intestine and exert positive effects. Additionally, it enhances the activity of digestive enzymes, optimizes nutrient absorption and energy utilization, and promotes healthy fish growth, offering a sustainable substitute to antibiotics in aquaculture3. In this research, it was shown that feeding commercial probiotics based on SFM to C. mrigala fingerlings improved their survival, growth parameters, digestibility, carcass composition, hematological indices, mineral utilization and antioxidant activity.
Growth performance
In the current investigation, no mortalities were recorded throughout the feeding experiment when probiotics administration level was 2 g kg−1. The live bacteria (probiotics) added to the SFM diet fed to the fingerlings targeted and eliminated undesirable gut bacteria, boosting digestive enzymes and enhancing digestion and feed intake. This replacement of harmful bacteria with beneficial ones resulted in increased immunity and improved survival rates. Notably, Paranita and Rahardjaet40 reported an 85% survival rate in catfish (Clarias sp.) fed 1 ml L−1 of commercial probiotics, whereas the present study achieved a 100% survival rate.
Fish digestibility (ADC%) and feed efficiency are negatively impacted by the presence of ANFs in plant-based fish meals. A crucial aspect of this study lies in probiotics’ ability to mitigate ANFs and fibre content inherent in plant-based ingredients. ANFs, such as phytates, oxalates, and saponins, and fibre content impair mineral bioavailability and digestive efficiency. Probiotics supplementation can effectively mitigate these content present in plant-based ingredients, enhancing nutrient bioavailability and digestive efficiency. Probiotics produce phytase enzymes, breaking down phytates and releasing bound minerals. Moreover, they degrade oxalates, reducing mineral precipitation and produce enzymes (e.g., amylase, protease) to counteract ANF-induced enzyme inhibition. Additionally, they alter gut microbiota, enhancing nutrient absorption and reducing ANF effects and can bind or adsorb ANFs, reducing their bioavailability20,21. To address this issue, probiotics are added to feed to enhance the efficiency of digestive enzymes and nutrient absorption, leading to increased growth. Our findings indicate that a probiotic inclusion level of 2 g kg−1 yielded the best growth outcomes in C. mrigala, including maximum WG, highest WG%, lowest FCR, and greatest SGR. Likewise, Labeo rohita fingerlings fed with plant-based diets showed improved the growth performance when supplemented with probiotics41. Moreover, Eissa et al.23 found that incorporating probiotic (P. acidilactici) improved survival and growth performance (WG% 262, FCR 1.3, and SGR 2.1) in sea bass at 3 g kg−1. This enhancement was attributed to reduced pH in the gastrointestinal tract (GIT), creating an optimal environment for digestive enzymes. Another recent research found that feeding 2 g kg−1 probiotics Cyprinus carpio fingerlings in addition to a plant-based corn-gluten diet resulted in the best growth outcomes with maximum WG of 19.43 g and SGR of 1.4742.
Carcass composition
Probiotics in fish feed enhance the gut microbiota, leading to improved nutrient absorption and alterations in the proportions of the various components of fish carcass. In this study, C. mrigala fingerlings fed SFM with commercial probiotics levels exhibited a non-linear relationship. The overall results showed that 2 g kg−1 probiotics supplementation in SFM-fed C. mrigala fingerlings resulted in optimal carcass composition values, including crude protein (18.5%), ash content (1.93%), crude fat (4.55%) and moisture content (75.02%). Similarly, Eissa et al.23 found that 3 g kg−1 of commercial probiotics improved sea bass (Dicentrarchus labrax) carcass composition. A related research on Catla catla by Shahzad et al.43 reported that probiotic supplementation in a rapeseed meal showed similar carcass results to our study, with crude protein (18.84%) and crude fat (9%) values. Consequently, it can be concluded that various species, feeding habits, and environmental factors contribute to variations in findings.
Digestibility
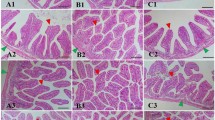
Our findings revealed that the supplementation of probiotics at an optimal level of 2 g kg−1 yielded the maximum ADC% in C. mrigala fish. The enhanced nutrient absorption is likely a result of the positive effects of probiotics in the intestinal tract. By supplementing diets with probiotics, the stimulation of innate digestive enzymes, such as protease, lipase and amylase, is stimulated, leading to improved nutrient breakdown and absorption (amino acids and lipids)44. According to Lee et al.45 diets supplemented with the probiotic Bacillus resulted in significantly higher ADC values in shrimp (Litopenaeus vannamei) compared to the control diet without probiotics. Multi-species probiotics have an advantage over single-strain probiotics, as they significantly improve intestinal morphology, including villi length, width, and area, as well as goblet cell density, leading to enhanced nutrient absorption. Likewise, a research by Shekarabi et al.6 revealed that a multi-strain probiotic blend exhibited synergistic effects, amplifying the activity of multiple enzymes, which led to more efficient nutrient digestion and a significant boost to the wellbeing of Siberian sturgeon. The findings by Hossain et al.5 demonstrated that mixed-species probiotics can have a profound impact on enhancing intestinal microbiota and morphology. Since C. mrigala is an agastric fish species, its feed remains in the small intestine for a longer period, thereby increasing nutrient absorption through the gut46.
Hematology
Hematological parameters serve as a valuable indicator for monitoring the health status and well-being of fish, as they reflect changes in physiological and pathological conditions. Elevated Hb and RBC counts indicate high oxygen-carrying capacity in the blood47. The findings of this study suggest that a 2 g kg−1 probiotic incorporation is the optimal dosage for enhancing blood parameters. Research conducted by various authors5,48,49 has investigated the impact of probiotics on hematological parameters in L. rohita, Barbonymus gonionotus and C. cirrhosus, respectively. These findings are consistent with the current study, indicating that a probiotic blend, including Bacillus spp. and Lactobacillus spp., can substantially enhance the count of RBCs and WBCs. Similarly, Tanveer et al.50 observed that the addition of a 2 g kg−1 mixed-strain probiotic blend (E. faecalis, B. subtilis and B. licheniformis) resulted in enhanced hematological parameters in Clarius batrachus. In contrast, Shekarabi et al.6 demonstrated that with the exception of marked improvements in WBCs and neutrophils, the blood indices of Siberian sturgeon fed a mixture of multi-enzymes and probiotics in diet did not exhibit significant changes.
Antioxidant activity
SOD, GPx, and CAT serve as indicators of the innate immune response45. The results of current research indicate that incorporating 2 g kg−1 of mixed-strain probiotics significantly improved the activity of serum antioxidant enzymes in C. mrigala. Tanveer et al.50 revealed that adding a 2 g kg−1 mixed-strain probiotic blend (B. subtilis, B. licheniformis and E. faecalis), significantly ameliorated antioxidant enzyme activity in C. batrachus. Although reactive oxygen species (ROS) generated during oxidative injury can inflict cell damage and tissue disruption, probiotics have emerged as a natural defense, possessing antioxidant properties that protect hosts from oxidative damage, as supported by a growing evidence51,52. Gobi et al.53 found that supplementing tilapia feed with B. licheniformis Dahb1 probiotics at a level of 107 CFU g−1 enhanced antioxidant enzymes activities in the fish.
Mineral analysis
The results of our investigation revealed that a probiotics inclusion rate of 2 g kg−1 yielded the best mineral digestibility outcomes in C. mrigala fingerlings. Notably, the highest digestibility rates were observed at this probiotic level, with values of 73%, 73%, 71%, and 75% for calcium, sodium, potassium, and phosphorus, respectively, when fish were fed a diet based on SFM. Similarly, according to Hussain et al.54, Oreochromis niloticus when received 3 g kg−1 of probiotic-enriched feed, exhibited the lowest fecal mineral excretion and highest mineral absorption rate in the body. The reduced mineral absorption in plant-based diets may be due to the presence of ANFs, which inhibit mineral uptake55. However, the addition of probiotics to plant-based diets can enhance mineral digestion by boosting the synthesis of digestive enzymes (amylase, lipase and protease), detoxifying harmful compounds, and reducing gut pH, thereby improving nutrient absorption. Although probiotics have been widely explored for their health benefits, the scientific understanding of their effects on mineral digestibility remains limited. As a result, there is a pressing need for further investigation into the relationships between probiotics, mineral digestibility, and overall nutrient bioavailability.
Moreover, our study also faces limitations, such as a controlled environment that may not accurately reflect commercial-scale animal production settings. Additionally, temperature, humidity, and other environmental factors may impact outcomes. A more in-depth examination is required to explore the interactions between probiotics and dietary supplements, and to elucidate their molecular mechanisms improving health and nutritional parameters.
Conclusion
Probiotics have been shown to be a valuable additive in fish feed, enhancing survival rates, growth performance, carcass composition, digestibility, hematology, antioxidant status, and mineral status. This study reveals that a probiotics dose of 2 g kg−1 is optimal for optimizing these parameters. Researchers are currently exploring methods to boost the stability of probiotics in fish diet, with the goal of maximizing their positive and environmentally friendly impacts.
Data availability
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request.
References
Firouzbakhsh, F., Noori, F., Khalesi, M. K. & Jani-Khalili, K. Effects of a probiotic, protexin, on the growth performance and hematological parameters in the Oscar (Astronotus ocellatus) fingerlings. Fish. Physiol. Biochem. 37, 833–842. https://doi.org/10.1007/s10695-011-9481-4 (2011).
El-Saadony, M. T. et al. The functionality of probiotics in aquaculture: an overview. Fish. Shellfish Immunol. 117, 36–52. https://doi.org/10.1016/j.fsi.2021.07.007 (2021).
Mohammadian, T., Monjezi, N., Peyghan, R. & Mohammadian, B. Effects of dietary probiotic supplements on growth, digestive enzymes activity, intestinal histomorphology and innate immunity of common carp (Cyprinus carpio): a field study. Aquaculture 549, 737787. https://doi.org/10.1016/j.aquaculture.2021.737787 (2022).
Niu, K. M. et al. Effects of dietary multi-strain probiotics supplementation in a low fishmeal diet on growth performance, nutrient utilization, proximate composition, immune parameters, and gut microbiota of juvenile olive flounder (Paralichthys olivaceus). Fish. Shellfish Immunol. 93, 258–268. https://doi.org/10.1016/j.fsi.2019.07.056 (2019).
Hossain, M. K. et al. Multi-species probiotics improve growth, intestinal microbiota and morphology of Indian major carp mrigal Cirrhinus cirrhosus. Saudi J. Biol. Sci. 29 (9), 103399. https://doi.org/10.1016/j.sjbs.2022.103399 (2022).
Shekarabi, S. P. H., Ghodrati, M., Dawood, M. A., Masouleh, A. S. & Roudbaraki, A. F. The multi-enzymes and probiotics mixture improves the growth performance, digestibility, intestinal health, and immune response of siberian sturgeon (Acipenser baerii). Ann. Anim. Sci. 22 (3), 1063–1072. https://doi.org/10.2478/aoas-2022-0006 (2022).
FAO. Food and agriculture organization of the United Nations (2014). https://www.fao.org/3/i4040e/i4040e.pdf
FAO. Cirrhinus mrigala. In Cultured aquatic species fact sheets. Text by Ayyappan, S. Edited and compiled by Valerio Crespi and Michael New. CD-ROM (multilingual) (2009).
9. FAO. The State of World Fisheries and Aquaculture 2018: Meeting the Sustainable Development Goals. (Food and Agriculture Organization (FAO) of the United Nations. https://www.fao.org/documents/card/en/c/I9540EN/ (2018).
Nölle, N. et al. Fish as a source of (micro) nutrients to combat hidden hunger in Zambia. Food Secur. 12 (6), 1385–1406. https://doi.org/10.1007/s12571-020-01060-9 (2020).
Ahmad, B. et al. Efficacy of acidified phytase supplemented cottonseed meal based diets on growth performance and proximate composition of Labeo rohita fingerlings. Braz. J. Biol 83, 2023. https://doi.org/10.1590/1519-6984.247791 (2021).
12. FAO. The State of World Fisheries and Aquaculture 2020. Sustainability in action. https://doi.org/10.4060/ca9229en (2020).
Cottrell, R. S., Blanchard, J. L., Halpern, B. S., Metian, M. & Froehlich, H. E. Global adoption of novel aquaculture feeds could substantially reduce forage fish demand by 2030. Nat. Food. 1 (5), 301–308. https://doi.org/10.1038/s43016-020-0078-x (2020).
Tabassum, S. et al. Partial replacement of fish meal with Moringa oleifera leaf meal in practical diets of Cirrhinus mrigala fingerlings. Braz. J. Biol. 83, 2023. https://doi.org/10.1590/1519-6984.246333. (2021).
Wijayanti, I., AZ, Y. S., Khotijah, L. & April The evaluation of sunflower seed meal as protein source in lamb ration. IOP Conf. Ser. Earth Environ. Sci. 478 (1), 012028. https://doi.org/10.1088/1755-1315/478/1/012028 (2020).
Saleh, A. A. et al. Effects of sunflower meal supplementation as a complementary protein source in the laying hen’s diet on productive performance, egg quality, and nutrient digestibility. Sustainability 13 (6), 3557. https://doi.org/10.3390/su13063557 (2021).
Adeleke, B. S. & Babalola, O. O. Oilseed crop sunflower (Helianthus annuus) as a source of food: Nutritional and health benefits. Food Sci. Nutr. 8 (9), 4666–4684. https://doi.org/10.1002/fsn3.1783 (2020).
Anjum, M. A. et al. Assessment of poultry feed ingredients used in commercial compound feed. Pak J. Life Soc. Sci. 12 (2), 69–73 (2014). https://www.academia.edu/download/35363250/Assesment_of_feed.pdf
Kaur, R. & Ghoshal, G. Sunflower protein isolates-composition, extraction and functional properties. Adv. Colloid Interface Sci. 306, 102725. https://doi.org/10.1016/j.cis.2022.102725 (2022).
Samtiya, M., Aluko, R. E. & Dhewa, T. Plant food anti-nutritional factors and their reduction strategies: an overview. Food Prod. Process. Nutr. 2, 1–14. https://doi.org/10.1186/s43014-020-0020-5 (2020).
Joudaki, H. et al. Microbial phytases: properties and applications in the food industry. Curr. Microbiol. 80 (12), 374. https://doi.org/10.1007/s00284-023-03471-1 (2023).
Rowland, S. J. Diseases of Australian native freshwater fishes with particular emphasis on the ectoparasitic and fungal diseases of Murray Cod (Maccullochellapeeli), golden perch (Macquariaambigua) and silver perch (Bidyanusbidyanus), 21–23 (1991). https://agris.fao.org/agris-search/search.do?recordID=US201300710251
Eissa, E. S. H. et al. Assessing the influence of dietary Pediococcus acidilactici probiotic supplementation in the feed of European sea bass (Dicentrarchus labrax L.) (Linnaeus, 1758) on farm water quality, growth, feed utilization, survival rate, body composition, blood biochemical parameters, and intestinal histology. Aquacult. Nutr. 2022, 1–11. https://doi.org/10.1155/2022/5841220 (2022).
Chomová, N. et al. Development and evaluation of a fish feed mixture containing the probiotic lactiplantibacillus plantarum prepared using an innovative pellet coating method. Front. Vet. Sci. 10, 1196884. https://doi.org/10.3389/fvets.2023.1196884 (2023).
Kim, D. H. & Austin, B. Innate immune responses in rainbow trout (Oncorhynchus mykiss, Walbaum) induced by probiotics. Fish. Shellfish Immunol. 21 (5), 513–524. https://doi.org/10.1016/j.fsi.2006.02.007 (2006).
Khalid, M. A., Hussain, S. M., Sultana, S., Asrar, M. & Sharif, A. Impact of different types of biochar supplemented Moringa oleifera seed meal-based diets on nutrient utilization, hematology and growth performance of Cirrhinus mrigala fingerlings. Pak. J. Agric. Sci. 59 (5). https://doi.org/10.21162/PAKJAS/22.90 (2022).
NRC (National Research Council). Nutrient Requirements of fish (National Academy Press, 2011).
28. AOAC (Association of Official Analytical Chemists). Official Methods of Analysis. 15th Edn, 1094 (Association of Official Analytical Chemists, 1995).
Jagtap, G. A., Badge, A., Kohale, M. G. & Wankhade, R. S. The role of the Biosafety Cabinet in preventing infection in the Clinical Laboratory. Cureus 15 (12). https://doi.org/10.7759/cureus.51309 (2023).
Kefas, M., Abubakar, K. A. & Ja’afaru, A. Haematological indices of tilapia (Oreochromis niloticus) from Lake Geriyo, Yola, Adamawa State, Nigeria. Int. J. Fish. Aquac Stud. 3 (1), 9–14 (2015).
Wedemeyer, G. A. & Yasutake, W. T. Clinical methods for the assessment of the effects of environmental stress on fish health. Department of the Interior, Fish, and Wildlife Service, 89. https://doi.org/10.12691/ajfst-7-5-1 (1977).
Divakaran, S., Obaldo, L. G. & Forster, I. P. Note on the methods for determination of chromic oxide in shrimp feeds. J. Agric. Food Chem. 50, 464–467. https://doi.org/10.1021/jf011112s (2002).
Ahmad, N. et al. Dietary exposure of Cr nanoparticles to Catla catla fingerlings: effects on mineral digestibility and carcass composition. Aquac Rep. 30, 101598. https://doi.org/10.1016/j.aqrep.2023.101598 (2023).
Hald, P. M. The flame photometer for the measurement of sodium and potassium in biological materials, pp. 499–510 (1947).
McKie, V. A. & MccleAry, B. V. A novel and rapid colorimetric method for measuring total phosphorus and phytic acid in foods and animal feeds. J. AOAC Int. 99 (3), 738–743. https://doi.org/10.5740/jaoacint.16-0029 (2016).
Witeska, M., Kondera, E., Ługowska, K. & Bojarski, B. Hematological methods in fish–not only for beginners. Aquaculture 547, 737498. https://doi.org/10.1016/j.aquaculture.2021.737498 (2022).
McCord, J. M. & Fridovich, I. Superoxide dismutase: an enzymic function for erythrocuprein (hemocuprein). J. Biol. Chem. 244 (22), 6049–6055. https://doi.org/10.1016/S0021-9258(18)63504-5 (1969).
Steel, R. G. D., Torrie, J. H. & Dickey, D. A. Principles and Procedures of Statistics, 3rd Edn, 336–352 (McGraw Hill international Book Co. Inc., 1996).
Snedecor, G. W. & Cochran, W. G. Statistical Methods, 8th Edn, 503 (Iowa State University Press, 1991).
Paranita, A. & Rahardja, B. S. Effect of probiotics addition on total organic matter and survival rate of catfish (Clarias sp.) maintenance using recirculating aquaculture system (RAS). IOP Conf. Ser. Earth Environ. Sci. 1036 (1), 012089. https://doi.org/10.1088/1755-1315/1036/1/012089 (2022).
Aman, S., Tabssum, F., Hussain, A., Jabeen, S. & Qazi, J. I. Growth performance of Labeo rohita fingerlings fed with probiotic added plant by-products-based feeds. Pak. J. Zool. 1, 1–6. https://doi.org/10.17582/journal.pjz/20220224060200 (2022).
Hussain, S. M. et al. Efficacy of probiotics supplementation on growth performance, carcass composition and hematological parameters of Cyprinus carpio fingerlings fed corn gluten meal-based diet. Braz. Arch. Biol. Technol. 64. https://doi.org/10.1590/1678-4324-2021200187 (2021).
Shahzad, M. M. et al. N. improvement in body composition and blood parameters of Catla catla fingerlings by supplementing rapeseed meal based diet with probiotics. Pak. J. Zool. 1–9. https://doi.org/10.17582/journal.pjz/20211011061010 (2022).
Xie, J. J. et al. Effects of dietary mixed probiotics on growth, non-specific immunity, intestinal morphology and microbiota of juvenile pacific white shrimp, Litopenaeus vannamei. Fish. Shellfish Immunol. 90, 456–465. https://doi.org/10.1016/j.fsi.2019.04.301 (2019).
Lee, C. et al. Dietary supplementations of Bacillus probiotic improve digestibility, growth performance, innate immunity, and water ammonia level for Pacific white shrimp, Litopenaeus vannamei. Aquac. Int. 29 (6), 2463–2475. https://doi.org/10.1007/s10499-021-00760-z (2021).
Soni, N. & Ujjania, N. C. Gut contents analysis and preponderance index based study on feeding habit of Cirrhinus mrigala from Ukai Dam. J. Fish. Life Sci. 3 (1), 19–21 (2018).
Yaqub, A., Nasir, M., Kamran, M., Majeed, I. & Arif, A. Immunomodulation, fish health and resistance to Staphylococcus aureus of Nile tilapia (Oreochromis niloticus) fed diet supplemented with zinc oxide nanoparticles and zinc acetate. Biol. Trace Elem. Res. 201, 4912–4925. https://doi.org/10.1007/s12011-023-03571-w (2023).
Jahan, N., Islam, S. M., Rohani, M. F., Hossain, M. T. & Shahjahan, M. Probiotic yeast enhances growth performance of rohu (Labeo rohita) through upgrading hematology, and intestinal microbiota and morphology. Aquaculture 545, 737243. https://doi.org/10.1016/j.aquaculture.2021.737243 (2021).
Salam, M. A. et al. Gut probiotic bacteria of Barbonymus gonionotus improve growth, hematological parameters and reproductive performances of the host. Sci. Rep. 11 (1), 10692. https://doi.org/10.1038/s41598-021-90158-x (2021).
Tanveer, A. et al. Effect of multi-strain probiotics on the growth, hematological profile, blood biochemistry, antioxidant capacity, and physiological responses of Clarias batrachus fingerlings. Aquac. Int. 32 (2), 1817–1833. https://doi.org/10.1007/s10499-023-01245-x (2024).
Rajput, I. R. et al. Supplementary effects of Saccharomyces boulardii and Bacillus subtilis B10 on digestive enzyme activities, antioxidation capacity and blood homeostasis in broiler (2013).
Krishnamurthy, P. & Wadhwani, A. Antioxidant enzymes and human health. Antioxid. Enzyme. 1, 3–18. https://doi.org/10.5772/48109 (2012).
Gobi, N. et al. Dietary supplementation of probiotic Bacillus licheniformis Dahb1 improves growth performance, mucus and serum immune parameters, antioxidant enzyme activity as well as resistance against Aeromonas hydrophila in tilapia Oreochromis mossambicus. Fish. Shellfish Immunol. 74, 501–508. https://doi.org/10.1016/j.fsi.2017.12.066 (2018).
Hussain, S. M. et al. Efficacy of probiotics supplementation on mineral digestibility, haematological parameters and carcass composition of Oreochromis niloticus fingerlings fed canola meal based diets. Pak. J. Zool. 50 (5). https://doi.org/10.17582/journal.pjz/2018.50.5.1825.1834 (2018).
Hussain, S. M. et al. Substitution of fishmeal: highlights of potential plant protein sources for aquaculture sustainability. Heliyon https://doi.org/10.1016/j.heliyon.2024.e26573 (2024).
Acknowledgements
The authors are grateful for the financial support received from the HEC of Pakistan, under Project No. 20-4892/NRPU/R&D/HEC/14/1145. We also express our sincere gratitude to the Researchers Supporting Project (RSP2025R48) at King Saud University, Riyadh, Saudi Arabia, for their contribution to this research.
Author information
Authors and Affiliations
Contributions
Writing-original draft: Danish Riaz. Software, Supervision, Methodology, Data curation: Syed Makhdoom Hussain. Investigation, Formal analysis, Data curation, Writing—review and editing, conceptualization: Shafaqat Ali and Joanna Nowosad. Writing—review and editing, Data curation, Formal analysis: Konrad Turkowski and Khalid A. Al-Ghanim.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Riaz, D., Hussain, S.M., Ali, S. et al. Evaluation of protexin probiotics on the growth, and health of Cirrhinus mrigala (Mrigal). Sci Rep 15, 6172 (2025). https://doi.org/10.1038/s41598-025-89495-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89495-y